Abstract
Controlled Zn-mediated grafting of a thin layer of a diazonium salt was used to functionalise a carbon electrode with ruthenium(II)-tris-bipyridine (Ru)-labelled DNA for use as a capture probe in an electrochemiluminescent genosensor. A secondary reporter probe was labelled with a ferrocene (Fc) molecule, and in the presence of the single-stranded DNA target a genocomplex formed, where the Fc-label effectively quenched the electrochemiluminescence of the signal emitted from the Ru-label. The spacing of the labels for maximum sensitivity and minimum detection limit was optimised, and the signal reproducibility and stability of the method was established.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Electrochemiluminescence (ECL) is a detection technique that combines electrochemical and photochemical processes. It has long attracted much attention because it incorporates the advantages of both methods for biomolecule detection, resulting in high sensitivity and versatility [1, 2]. In ECL, high-energy electron-transfer reactions are performed on electrodes via the application of a specific potential to generate excited states that emit light [3, 4]. Exploiting potential provides a more selective source of excitation than is afforded optically [5], and also simplifies the instrumentation for the construction of portable devices [6, 7]. The use of ECL transduction has been reported for the detection of DNA [8] and proteins [9] immobilised on different substrates including carbon [10], gold [11], and indium tin oxide [12], using diverse ECL-active species as labels [13, 14]. The most commonly used ECL detection method is based on the co-reactant mechanism [15, 16], and the coordination compounds formed by ruthenium(II) and pyridinic derivatives as chelates, either as a single molecule or in combination with nanostructures [2, 17], have been reported as preferable luminophores, using tripropylamine (TPA) as co-reactant [18, 19]. In this system the same applied potential is able to oxidise both the Ru(bpy) 2 +3 (luminophore) and the TPA (co-reactant), producing strong oxidative species. From the reaction of these two species the excited state Ru(bpy) 2 + *3 is formed, which emits light [20]. ECL “on/off” strategies have recently been reported for clinical analytes [21] using immuno [22] and DNA [23, 24] detection, where ferrocene derivatives are used to quench the ruthenium complex signal (“off”) because they have good quenching efficiency and stability in different chemical media [1, 25–27]. Although ECL is a very sensitive method [28], one of its limitations is the high potentials required to excite the active centre, and this is particularly critical for surface-immobilised ECL strategies. Therefore the robustness of the surface chemistry determines the reproducibility of the ECL signal, which has two main components: the stability of the ECL response after applying consecutive pulses once the surface has been modified (with acceptable RSD values <5 %) [1, 14, 15, 29], and the inter-sensor reproducibility associated with sensor fabrication, which varies from 2 [1, 2, 15] to 10 % [16] depending on the substrate used.
Consequently, in ECL measurements a strong and stable covalent bond, which is able to resist the high applied potentials, is crucial. The methods based on diazonium derivatives immobilised on gold or carbon surfaces have been revealed to be stable at the potential used in ECL detection [30, 31].
In this work an ECL sensor for the detection of a Francisella tularensis DNA target was developed. The Francisella subspecies tularensis is one of the causative agents of the disease tularemia [32], and current methods for detecting this bacterium involve time-consuming culturing of suspect pathogens [33] or, alternatively, the detection of antibodies, which cannot be achieved until at least two weeks after infection [34]. These disadvantages render rapid DNA detection an attractive strategy. One method of detecting the subspecies tularensis is the use of the specific sequences of tul4 [35], which was used as a model target for the present study.
In this work, an “on/off” approach was developed based on a sandwich-type detection of DNA by using two DNA sequences complementary to almost consecutive regions of the DNA target: a capture DNA probe labelled with ruthenium(II) tris-bipyridine (Rubpy), and a secondary DNA reporter probe labelled with ferrocene. In the presence of single-stranded target DNA the probes hybridise to their complementary sequences on the target, bringing them into close proximity, permitting energy transfer, and thus facilitating the quenching effect of the ferrocene on the Rubpy signal. This on/off sandwich approach also increases the specificity, because two regions of the target hybridise with two different probes. The system was optimised in the solution phase and then, using the optimised experimental conditions, a solid-phase system using stable diazonium grafting was investigated [31].
Materials and methods
Chemicals
All reagents were commercially available and used as received. Ruthenium(II)-tris-bipyridine-N-hydroxysuccinimide, N-ethyl-N′-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDC), N-hydroxysuccinimide (NHS), streptavidin, NaH2PO4.H2O, Na2HPO4, 3-hydroxypicolinic acid, strontium nitrate, potassium ferricyanide(III), potassium ferrocyanide(II), and tetramethylbenzidine (TMB) liquid substrate system were purchased from Sigma–Aldrich (Spain). H2SO4 was purchased from Scharlau (Spain), and dimethyl sulfoxide from Abcam plc. The Sephadex G-25 size-exclusion column was from Pierce, the magnetic beads from SIMAG, and the 3,5-bis(4-aminophenoxy) benzoic acid from TCI. All solutions were prepared with Milli-Q water (18 MΩ cm; Millipore). All HPLC-grade oligonucleotides were provided by Biomers.net (Ulm, Germany). The DNA targets and ferrocene-labelled DNA (Fc–DNA) were used as received and the Rubpy–DNA conjugates were prepared as described below. The DNA sequences used were:
For studying the effect of the separation between the luminescent ruthenium(II) tris-bipyridine (Rubpy) group attached to the DNA and the quencher ferrocene (Fc) linked to the secondary probe:
-
5′-TGGGCGTTAAACGTGACA-3′-NH2 was used for preparing the Rubpy–DNA:
-
Rubpy–DNA: TGGGCGTTAAACGTGACA-3′-NHCO-Rubpy; m/z = 6370.69
-
Fc–DNA: Fc-5′-ACCGAGACGAATAGGTAT-3′
-
Targets containing spacer T (n = 0, 1, 2, 3, 4, 5, and 6):
-
5′-ATACCTATTCGTCTCGGT (T)n TGTCACGTTTAACGCCCA-3′
-
For detection of Francisella tularensis DNA by ECL:
-
Biotin-TEG-5′-CTTAGTAATTGGGAAGCTTGTATCATGGCACTTAGAA-3′-NH2 was used for preparing the Rubpy–DNA capture probe:
-
Biotin-TEG-5′-CTTAGTAATTGGGAAGCTTGTATCATGGCACTTAGAA-3′-NHCO-Rubpy;
-
m/z = 12815.74
-
Fc–DNA secondary probe: Fc-5′-TTCTGGAGCCTGCCATTGTAAT-3′
-
Target: 5′ATTACAATGGCAGGCTCCAGAAGGTTCTAAGTGCCATGATACAAGCTTCCCAATTACTAAGTATGCTGAGAAGAACGATAAAACTTGGGCA-3′
Instrumentation
Electrochemical (EC) measurements were performed on a PC-controlled PGSTAT12 Autolab potentiostat (EcoChemie, The Netherlands). The electrochemiluminescence (ECL) experiments were performed using a device assembled at Institut für Mikrotechnik Mainz GmbH (IMM), Germany. The ECL equipment contained a photomultiplier (Hamamatsu H10682-01) connected to a potentiostat manufactured by PALM INSTRUMENTS BV to supply the voltage to the working electrode. The screen-printed-carbon-electrodes configuration (DRP-110) of working electrode: carbon disk (φ = 4 mm); reference electrode: silver; counter electrode: carbon, was used in both EC and ECL experiments. The carbon electrodes were activated by cycling three times from 0 to −1.2 V in 0.5 mol L−1 KOH at 50 mV s−1 scan rate. The quality of the cleaning step was checked using cyclic voltammetry in 1 mmol L−1 K4[Fe(CN)6]/K3[Fe(CN)6] with 50 mmol L−1 Sn(NO3)2 as supporting electrolyte. The CV was recorded from −0.2 to 0.4 V at 100 mV s−1.
UV–visible spectra were recorded using a temperature-controlled Cary 100 Bio spectrophotometer (Varian) in 1 cm quartz cells. Plate optical densities were recorded using a Wallac Victor2 1420 Multilabel counter from Perkin Elmer. The fluorescence experiments were performed using a Cary Eclipse spectrofluorimeter. The excitation wavelength was set at 460 nm. The fluorescence spectra were recorded in the wavelength interval 540–700 nm, with excitation and emission slits of 10 nm and a scan rate of 240 nm min−1. All experiments were performed in triplicate and the average value was used.
The analyses of the conjugates were performed using an Applied Biosystems Voyager STR MALDI-TOF spectrometer. Samples were prepared using 3-hydroxypicolinic acid in 50:50 (v/v) acetonitrile–water buffered with 0.05 mol L−1 diammonium acetate as matrix. Linear positive mode was used for detection.
Experimental
For synthesis of the conjugates Rubpy–DNA, ruthenium(II) tris-bipyridine N-hydroxysuccinimide was first dissolved in DMSO, and then water was slowly dropped to avoid precipitation (important: the percentage (v/v) of DMSO with respect to the final volume of water was less than 10 %). The resulting Rubpy solution was then mixed with a 25 μmol L−1 solution of biotin–TEG–DNA–NH2 probe in PBS (pH = 7.8) in a 1:8 DNA–Rubpy molar ratio. The reaction was performed by shaking at room temperature. Because the Rubpy is very sensitive to light, the conjugation was performed in light-shielded containers. After 1 h, the product obtained was purified using the NAP G-25 size-exclusion column (DNA quality) to separate the uncoupled Rubpy. This step was achieved following step 7 of the procedure MSD® TAG-NHS-Ester, MSD Labeling method. The column was pre-equilibrated with PBS and the absorbance of the fractions obtained was measured. Finally the non-labelled DNA was isolated from the conjugate using carboxyl magnetic beads. The particles (500 μL) were washed with 1 mL MES two times and the carboxyl groups were activated by adding EDC and NHS at 80 and 40 mg mL−1, respectively, over 20 min. The activated particles were separated using a magnet and washed with PBS. Then the DNA conjugates were mixed with the magnetic beads and shaken for 2 h. The beads containing the non-labelled DNA were separated by magnet and the supernatant was characterised by mass spectroscopy and UV–visible spectroscopy.
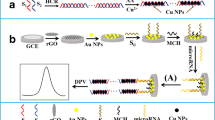
For ECL measurements, carbon surfaces were grafted with 3,5-bis(4-diazophenoxy) benzoic acid tetrafluoroborate [31]. The details of the synthesis of the diazonium salt and its immobilisation on carbon electrodes via Zn-mediated grafting were as reported elsewhere [31]. After activation of the carboxylate groups of the diazonium-salt-coated surface using 20 μL EDC–NHS (2:1 molar ratio), 1 μmol L−1 streptavidin was linked via amide-bond formation. After washing, the biotin–Rubpy–DNA capture probe, previously dissolved in PBS buffer at pH 7.4 to a concentration of 1 μmol L−1, was immobilised on the modified carbon surfaces over 1 h at 25 °C. After rinsing the electrode, the ECL signal was recorded at a potential pulse of +1 V for 5 s (using 100 mmol L−1 TPA as coreactant, PBS pH 7.8). The measurements were recorded 10 times to check the reproducibility of the signal.
After the modification of the electrode with Rubpy–DNA, a differential pulse voltammetry (DPV) signal was recorded in 0.1 mol L−1 PB pH 7.4, over a potential range of 0.1–1.1 V and with a scan rate of 50 mV s−1. This revealed that the target and secondary Fc–DNA were hybridised, and the DPV signal was then recorded again, but in the potential range 0.1–0.4 V to detect the ferrocene on the surface. In the absence of the target, the signal from ferrocene was absent as expected.
After the immobilisation of the Rubpy–DNA capture probe to the surface following the procedure described above, the target sequence at different concentrations (0, 0.1, 0.5, 0.7, 0.8, 1, 2, 3, 4, 5 nmol L−1) was dissolved in PB buffer (pH 7.4, with 0.8 mol L−1 NaCl) and hybridised to the capture element over 1 h. Finally the secondary Fc–DNA probe was hybridised to the target in a molar ratio 1:1 with respect to the Rubpy–DNA, and the detection was performed using the same conditions described above, before and after interaction with Fc–DNA.
Results and discussion
First, a model system based on short sequences, specifically designed not to have self-complementarity leading to secondary structures, was used to study the effect of the separation between the luminescent Rubpy and the quencher ferrocene (Fig. 1). Rubpy was linked to DNA via amide-bond formation between the activated carboxylic group of a Rubpy derivative and aminated DNA, and the bioconjugate was purified using column chromatography. It was then evaluated using UV–visible spectroscopy at 260 nm to detect DNA, at 460 nm to measure the characteristic bands of the Rubpy metal-to-ligand charge transfer, and at 277 nm to measure the π → π* ligand charge-transfer transition, which appears 10 nm shifted to blue with respect to the original Rubpy. The expected m/z = 12815.74 of the bioconjugate was observed using MALDI-TOF.
Rubpy–DNA and Fc–DNA were added in a 1:1 molar ratio to target-DNA sequences with spacers ranging from 0 to 6 bases, the genocomplex was incubated protected from light. A control was performed in the absence of target DNA, and no quenching was observed. The same procedure was followed to study the system using fluorescence detection and the results were compared, with the control response regarded as a 100 % signal (Fig. 1).
Comparing both methods, two bases in the spacer of the target was found to be the optimum compromise between proximity (needed for energy transfer) and steric hindrance (which could affect the hybridisation of strands) because it obtained the highest quenching effect, giving a decrease in signal of 85 % and 95 % for ECL and fluorescence, respectively (Fig. 1). It should be noted that the concentration of the probes used for ECL analysis was four orders of magnitude lower than that for fluorescence analysis, revealing the increased sensitivity achievable using ECL detection.
Once the spacing of Fc to Rubpy labels had been optimised, the target system of Francisella tularensis was studied. Any potential cross-reactivity between the two probes to be used was evaluated, and no interaction was observed (Electronic Supplementary Material (ESM) Fig. S1a). The pH for maximising ECL (basic) [36] whilst not inhibiting hybridisation was evaluated by immobilising the capture probe on a streptavidin plate and using the secondary probe labelled with HRP for detection by colorimetric enzyme-linked oligonucleotide assay, and pH 7.8 was chosen as an optimum compromise between efficient DNA hybridisation and TPA deprotonation (ESM Fig. S1b) [12].
Finally, using the optimised conditions of two-base spacing between labels and a pH of 7.8, a 22-mer-Fc–DNA probe and a 37-mer-Rubpy probe were hybridised with 91-mer-Francisella tularensis target in solution phase as a proof-of-concept with a real system. The evaluation was performed using two concentrations of target DNA, 0.15 and 0.5 μmol L−1. The controls of Rubpy–DNA and Fc–DNA in the absence of DNA target were observed, as expected, to be maximum and minimum, respectively. When DNA target was introduced in a quantity of 0.5:0.15:0.5 μmol L−1 notable quenching of approximately 60 % was observed, and when the concentration of DNA target was increased the ECL signal was further reduced, indicating that the quenching effect is proportional to the target concentration (ESM Fig. S2).
Having obtained a proof-of-concept in the homogenous solution phase, the possibility of transferring the system to an immobilised, heterogeneous phase was investigated. This is highly attractive in terms of simplicity and potentially increased sensitivity and multiplexing capability. However, it is known that thiolated DNA desorbs from gold electrodes at potentials of > ca. 1 V vs. Ag, and thus grafting of a bipodal diazonium salt on carbon electrodes was used. This diazonium salt, 3,5-bis(4-diazophenoxy)benzoic acid tetrafluoroborate, has recently been revealed to be stable at high positive potential because of the formation of covalent bonds with carbon. Furthermore, the immobilisation strategy involves a simple 5 min Zn-mediated grafting without the necessity of using a potentiostat, resulting in an almost monolayer formation brought about by a lack of radicals [31].
In our approach, this organic phase was linked to streptavidin and used to anchor the labelled biotin–Rubpy–DNA capture probe. Stability of immobilisation was tested by applying a potential of +1 V vs. Ag. The stability and reproducibility of the signal was recorded, and remained unaffected during at least ten cycles of application of potential (RSD 3 %) (Fig. 2a).
(a) The ECL signal from carbon-modified Rubpy–DNA for ten consecutive pulses of potential application. (b) Initial ECL signal from immobilised Rubpy–DNA, (c) ECL signal after hybridisation with target DNA and Fc–DNA, and (d) ECL signal after denaturation of genocomplex with 0.1 mol L−1 NaOH. Potential pulse of +1 V vs. Ag for 5 s (100 mmol L−1 TPA in PB pH 7.8)
Figure 2 shows the switch on/off process used for detecting the DNA target. First, the baseline ECL signal from the immobilised Rubpy–DNA was recorded (Fig. 2b), and then the target DNA and the reporter Fc–DNA probe were added and the signal was recorded again (Fig. 2c). To establish that the quenching of the Rubpy signal is caused by ferrocene and is not the result of damage to the phase, alkaline conditions were used to denature the genocomplex and revert to the Rubpy–DNA probe. The ECL signal was successfully recovered (RSD 4.5 %) (Fig. 2d), clearly revealing the quenching effect of ferrocene and the possibility of re-using the phase for repeated measurements of target DNA.
The presence of the Rubpy and ferrocene-labelled probes was further established by recording differential pulse voltammograms (Fig. 3a, b). The significant difference in current intensity between Rubpy and ferrocene can be attributed to the fact that the amount of ferrocene present depends on the target-DNA concentration, whereas a monolayer of the immobilised labelled DNA probe would result in a higher amount of Rubpy being present. The surface confinement of the ferrocene group was also confirmed using cyclic voltammetry, which revealed linear behaviour of cathodic and anodic peak currents vs. scan rate (Fig. 3c).
This method was tested for quantitative detection of the Francisella tularensis target DNA. A calibration curve (Fig. 4) was constructed with different target concentrations, using the optimised experimental conditions. Each measurement was performed in triplicate using three different electrodes. The RSD of the measurements (10 % in the linear range) indicates good reproducibility of screen-printed electrodes. The quenching of the signal was proportional to the concentration of the DNA target in the range 0–1 nmol L−1, with a limit of detection of 0.1 nmol L−1.
Conclusions
A method based on Zn-mediated immobilised 3,5-bis(4-diazophenoxy)benzoic acid tetrafluoroborate was revealed to be efficient for the surface confinement of a captured Rubpy-labelled DNA probe and its subsequent use for DNA-target detection by ECL quenching by a Fc-labelled reporter probe. As a result of the robustness of the thin layer of covalent linker, a stable and reproducible ECL signal from Rubpy–DNA was recorded during ten cycles after applying +1 V vs. Ag. Experimental conditions were optimised and detection of a subnanomolar concentration of DNA target was achieved.
References
Meng L, Hongmei Y, Chao M, Yan Z, Shenguang G, Jinghua Y, Mei Y (2014) A sensitive signal-off aptasensor for adenosine triphosphate based on the quenching of Ru(bpy)3 2+-doped silica nanoparticles electrochemiluminescence by ferrocene. Sensors Actuators B Chem 191:377–383
Wang H, Xu G, Dong S (2003) Electrochemiluminescence sensor using tris(2,2′-bipyridyl)ruthenium(II) immobilized in Eastman-AQ55D-silica composite thin-films. Anal Chim Acta 480:285–290
Richter MM (2004) Electrochemiluminescence (ECL). Chem Rev 104:3003–3036
Bard AJ (2004) Electrogenerated chemiluminescence. CRC Press, New York
Hu L, Xu G (2010) Applications and trends in electrochemiluminescence. Chem Soc Rev 39:3275–3304
Hao N, Xiong MJ, Zhang D, Xu JJ, Chen HY (2013) Portable thermo-powered high-throughput visual electrochemiluminescence sensor. Anal Chem 85:11715–11719
Cao WJ, Liu X, Yang E, Wang E (2002) New technique for capillary electrophoresis directly coupled with end-column electrochemiluminescence detection. Electrophoresis 23:3683–3691
Wu MS, He LJ, Xu JJ, Chen HY (2014) Sensitive electrochemiluminescence detection of c-Myc mRNA in breast cancer cells on a wireless bipolar electrode. Anal Chem 86:4559–4565
He Y, Chai Y, Yuan R, Wang H, Bai L, Liao N (2014) A supersandwich electrochemiluminescence immunosensor based on mimic-intramolecular interaction for sensitive detection of proteins. Analyst 139:5209–5214
Wang J, Shan Y, Zhao WW, Xu JJ, Chen HY (2011) Gold nanoparticle enhanced electrochemiluminescence of CdS thin films for ultrasensitive thrombin detection. Anal Chem 83:4004–4011
Zhao HF, Liang RP, Wang JW, Qiu JD (2014) One-pot synthesis of GO/AgNPs/luminol composites with electrochemiluminescence activity for sensitive detection of DNA methyltransferase activity. Biosens Bioelectron 63:458–464
Chen Y, Qiu B, Jiang Y, Lin Z, Sun J, Zhang L, Chen G (2009) Detection of hypoxanthine based on the electrochemiluminescent of 6-(4-methoxyphenyl)-2-methylimidazo[1,2-a]pyrazin-3(7H)-one on the electrically heated indium–tin-oxide electrode. Electrochem Commun 11:2093–2096
Yuanyuan C, Yifeng T (2014) The enhanced electrochemiluminescence of luminol by resonance energy transfer with solid-phase CdTe quantum dots. Electrochim Acta 135:187–191
Liu L, Ma Q, Yang L, Liu YZ, Su X (2014) A novel signal-off electrochemiluminescence biosensor for the determination of glucose based on double nanoparticles. Biosens Bioelectron 63:519–524
He Y, Chai Y, Yuan R, Wang H, Bai L, Cao Y, Yuan Y (2013) An ultrasensitive electrochemiluminescence immunoassay based on super sandwich DNA structure amplification with histidine as a co-reactant. Biosens Bioelectron 50:294–299
Gu W, Xu Y, Lou B, Lyu Z, Wang E (2014) One-step process for fabricating paper-based solid-state electrochemiluminescence sensor based on functionalized grapheme. Electrochem Commun 38:57–60
Sun Q, Zou G, Zhang X (2011) Electrochemiluminescence DNA sensor based on hairpin structure DNA as recognition element and Ru(bpy)3 2+-doped silica nanoparticles as signal-producing compound. Electroanalysis 23:2693–2698
Yin XB, Dong S, Wang E (2004) Analytical applications of the electrochemiluminescence of tris (2,2′-bipyridyl) ruthenium and its derivatives. TrAC Trends Anal Chem 23:432–441
Zhou X, Zhu D, Liao Y, Liu WG, Liu H, Ma Z, Xing D (2014) Synthesis, labeling and bioanalytical applications of a tris(2,2′-bipyridyl)ruthenium(II)-based electrochemiluminescence probe. Nat Protoc 9:1146–1159
Su M, Liu S (2010) Solid-state electrochemiluminescence analysis with coreactant of the immobilized tris(2,2(′)-bipyridyl) ruthenium. Anal Biochem 402:1–12
Gao W, Zhang A, Chen Y, Chen Z, Chen Y, Lu F, Chen Z (2013) A novel probe density controllable electrochemiluminescence biosensor for ultra-sensitive detection of Hg2+ based on DNA hybridization optimization with gold nanoparticles array patterned self-assembly platform. Biosens Bioelectron 49:139–145
Zhang M, Ge S, Li W, Yan M, Song X, Yu J, Xua W, Huang J (2012) Ultrasensitive electrochemiluminescence immunoassay for tumor marker detection using functionalized Ru-silica@nanoporous gold composite as labels. Analyst 137:680–685
Yao W, Wang L, Wang H, Li L, Zhang N, Pan L, Xing N (2013) An electrochemiluminescent DNA sensor based on nano-gold enhancement and ferrocene quenching. Biosens Bioelectron 40:356–361
Stobiecka M, Molinero AA, Chałupa A, Hepel M (2012) Mercury/homocysteine ligation-induced ON/OFF-switching of a T–T mismatch-based oligonucleotide molecular beacon. Anal Chem 84:4970–4978
Cao W, Ferrance JP, Demas J, Landers JP (2006) Quenching of the electrochemiluminescence of ruthenium(II) by ferrocene and its potential application to quantitative DNA detection. J Am Chem Soc 128:7572–7578
Chen L, Cai Q, Luo F, Chen X, Zhu X, Qiu B, Lin Z, Chen G (2010) A sensitive aptasensor for adenosine based on the quenching of Ru(bpy)3 2+-doped silica nanoparticle ECL by ferrocene. Chem Commun 46:7751–7753
Liao Y, Yuan R, Chai Y, Mao L, Zhuo Y, Yuan Y, Bai L, Yuan S (2001) Electrochemiluminescence quenching via capture of ferrocene-labeled ligand-bound aptamer molecular beacon for ultrasensitive detection of thrombin. Sensors Actuators B Chem 158:393–399
Wei H, Wang E (2011) Electrochemiluminescence of tris(2,2′-bipyridyl) ruthenium and its applications in bioanalysis: a review. Luminescence 26:77–85
Lv X, Li Y, Yan T, Pang X, Hu L, Du B, Wei Q (2015) An electrochemiluminescent immunosensor based on CdS–Fe3O4 nanocomposite electrodes for the detection of Ochratoxin A. New J Chem. doi:10.1039/c5nj00320b
Piper DJE, Barbante GJ, Brack N, Pigram PJ, Hogan CF (2011) Highly stable ECL active films formed by the electrografting of a diazotized ruthenium complex generated in situ from the amine. Langmuir 27:474–480
Torréns M, Ortiz M, Turner APF, Beni V, O’Sullivan CK (2015) Controlled Zn-mediated grafting of thin layers of bipodal diazonium salt on gold and carbon substrates. Chem Eur J 21:671–681
Jones RM, Nicas M, Hubbard A, Sylvester MD, Reingold A (2005) The infectious dose of Francisella tularensis (Tularemia). Appl Biosaf 10:227–239
Haristoy X, Lozniewski A, Tram C, Simeon D, Bevanger L, Lion C (2003) Francisella tularensis Bacteremia. J Clin Microbiol 41:2774–2776
Carlsson HE, Lindberg AA, Lindberg G, Hederstedt B, Karlsson KA, Agell BO (1979) Enzyme-linked immunosorbent assay for immunological diagnosis of human tularemia. J Clin Microbiol 10:615–621
Celebi B, Kilic S, Yesilyurt M, Acar B (2014) Evaluation of a newly-developed ready-to-use commercial PCR kit for the molecular diagnosis of Francisella tularensis. Mikrobiyol Bul 48:135–142
Gao W, Xia XH, Xu JJ, Chen HY (2007) Three-dimensionally ordered macroporous gold structure as an efficient matrix for solid-state electrochemiluminescence of Ru(bpy)3 2+/TPA system with high sensitivity. J Phys Chem C 33:12213–12219
Acknowledgments
This work was performed with partial financial support from the Commission of the European Communities specific RTD programme (FP7-2010-SEC-261810) and from National Plan “Cardioscreening” (BIO2011-25369). MT thanks the Generalitat de Catalunya for an FI pre-doctoral scholarship.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 120 kb)
Rights and permissions
About this article
Cite this article
Torréns, M., Ortiz, M., Bejarano-Nosas, D. et al. Electrochemiluminescent DNA sensor based on controlled Zn-mediated grafting of diazonium precursors. Anal Bioanal Chem 407, 5579–5586 (2015). https://doi.org/10.1007/s00216-015-8765-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-015-8765-4