Abstract
Withdrawal from opioids involves a negative affective state that promotes maintenance of drug-seeking behavior and relapse. As such, understanding the neurobiological mechanisms underlying withdrawal from opioid drugs is critical as scientists and clinicians seek to develop new treatments and therapies. In this review, we focus on the neural systems known to mediate the affective and somatic signs and symptoms of opioid withdrawal, including the mesolimbic dopaminergic system, basolateral amygdala, extended amygdala, and brain and hormonal stress systems. Evidence from preclinical studies suggests that these systems are altered following opioid exposure and that these changes mediate behavioral signs of negative affect such as aversion and anxiety during withdrawal. Adaptations in these systems also parallel the behavioral and psychological features of opioid use disorder (OUD), highlighting the important role of withdrawal in the development of addictive behavior. Implications for relapse and treatment are discussed as well as promising avenues for future research, with the hope of promoting continued progress toward characterizing neural contributors to opioid withdrawal and compulsive opioid use.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Opioid use disorder (OUD) is a chronic relapsing disorder characterized by various symptoms, including unsuccessful efforts to decrease use, craving, continued use despite negative consequences, and the development of withdrawal symptoms that may be relieved by taking more of the drug (American Psychiatric Association, 2013). More than 26 million people worldwide are affected by OUD, including more than 80,000 fatal overdoses in the USA in 2021 (Vos et al. 2017; Centers for Disease Control and Prevention, 2022). While gold-standard treatments such as methadone maintenance therapy are effective when individuals are in treatment programs, relapse remains a serious concern after the completion of these programs (Bell and Strang 2020; Magura and Rosenblum 2001), underscoring the need for better treatments that prevent relapse long-term.
Critically, the characteristics of OUD exist in a repetitive cycle in which individuals suffering from addiction experience negative emotion (e.g., aversion, anxiety) followed by intense craving and temptation to use the drug again, which has the potential to induce relapse (Koob and Volkow 2010). Relief of negative affect is also thought to contribute to the addictive cycle via negative reinforcement mechanisms (Koob 2013). Understanding the neurobiological mechanisms that produce negative emotional signs and symptoms of opioid withdrawal is therefore an important foundation for properly treating patients with OUD.
The aversive symptoms of withdrawal from drugs of abuse oppose the initial rewarding, euphoric effects of drug use (Solomon and Corbit 1974; Koob et al. 1989). For opioids, this may include somatic signs such as body aches and chills, gastrointestinal upset, hyperalgesia, and affective signs such as anxiety, irritability, and reduced motivation for natural rewards (Koob et al. 1989; Pergolizzi et al. 2020). Importantly, this subsequent aversive process relies on some of the same neural circuits that mediate the euphoric and reinforcing effects of opioids (Stinus et al. 1990; Vargas-Perez et al. 2009; Radke et al. 2011). Affective signs of withdrawal from opioids are observed after one or a few exposures in humans (Longnecker et al. 1973; Jones 1980) and animals (Schulteis et al. 1997; Parker and Joshi 1998; Harris and Gewirtz 2004; Rothwell et al. 2012) and intensify in severity with additional drug exposure (Kenny et al. 2006; Edwards et al. 2012; Williams et al. 2012). Thus, as the cycle of addiction continues, relief of withdrawal is an important factor driving continuation of drug use (Weiss et al. 2001; Hutcheson et al. 2001; Koob and Volkow 2010; Piper 2015; Kosten and Baxter 2019).
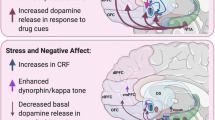
A critical distinction in the motivation to continue using drugs despite the aversive symptoms of withdrawal is between somatic vs. affective withdrawal signs, and contemporary literature argues that it is primarily the affective symptoms of withdrawal that contribute to addiction rather than somatic signs (Koob and Volkow 2010). However, symptoms traditionally considered to be entirely physical also induce negative affect and as such may serve as powerful motivators to continue opioid use, particularly in the immediate term when withdrawal begins. Because of their critical contribution to the cycle of addiction, this review will focus on the neural systems known to mediate the signs and symptoms of both affective and somatic signs of opioid withdrawal from a circuit perspective. A robust literature suggests that the functions of mesolimbic dopaminergic system, basolateral amygdala, extended amygdala (i.e., central nucleus of the amygdala, bed nucleus of the stria terminalis, and nucleus accumbens shell), and brain and hormonal stress systems are altered following opioid exposure (Table 1). These changes mediate behavioral signs of negative affect such as aversion and anxiety during withdrawal and parallel the development of drug-seeking behavior. Additionally, peripheral opioid receptors, the locus coeruleus, and the periaqueductal gray are important mediators of somatic signs of opioid withdrawal.
Mesolimbic dopamine system
The mesolimbic dopamine system consists primarily of dopamine (DA) neurons in the ventral tegmental area (VTA) that project to various limbic structures, including the amygdala and nucleus accumbens (NAc) (Kiyatkin 1995). Early studies demonstrated that animals robustly respond for intracranial stimulation of the VTA (Olds and Milner 1954; Crow 1972) and NAc (Phillips and Fibiger 1980; Cole et al. 2018) and since then an abundance of research has demonstrated the significance of the mesolimbic DA system in motivated behavior. The functions ascribed to the mesolimbic DA system are vast and varied, including roles in learning and plasticity, salience, value, effort, and choice (Wise 2004; Keiflin and Janak 2015; Berke 2018; Piantadosi et al. 2021). Reward prediction error theory, which is based on the observation that DA neuron firing rates are increased following unexpected presentation of a reward or reward-predictive cue (Schultz et al. 1993), has been particularly influential in the addiction field. Because all major drugs of abuse acutely increase extracellular DA levels in the NAc (Di Chiara and Imperato 1988), some have argued that inflated reward prediction errors in the presence of drugs enhance learning about drugs and drug-paired cues (Ahmed 2004; Redish 2004; Keiflin and Janak 2015). Sensitization of neural responses to drug-paired cues in turn elicits drug-seeking behavior, often at the expense of other available rewards and long after drug use has ceased (Robinson and Berridge 2001).
Because the VTA and NAc support the positive reinforcing effects of opioids (Koob and Volkow 2010), it is not surprising that affective signs of withdrawal arise from compensatory mechanisms in these same structures. Observations of increased cFos expression in the NAc shell during opiate withdrawal support this idea (Walters et al. 2000; Gracy et al. 2001). Further, infusion of an opioid receptor antagonist into the NAc of dependent animals induces conditioned place aversion (Stinus et al. 1990; Carlezon and Thomas 2009). A state of anxiety-like behavior can also be observed during spontaneous withdrawal from an intra-VTA infusion of morphine (Radke et al. 2011). In general, withdrawal from opioids is associated with decreases in mesolimbic DA neuron firing and DA release (Pothos et al. 1991; Diana et al. 1995; Fox et al. 2017; George et al. 2022). Reductions in DA signaling disrupt baseline reward sensitivity, as evidenced by increases in intracranial self-stimulation (ICSS) thresholds that occur during withdrawal (Kenny et al. 2006), and produce behavioral signs of withdrawal such as place aversion and increases in the startle reflex that can be reversed with infusions of DA receptor agonists into the NAc (Chartoff et al. 2009; Radke and Gewirtz 2012).
Intracellularly, the mu-opioid receptor (MOR) exerts an inhibitory influence on the membrane potential via coupling to the Gi intracellular signaling pathway, which inhibits adenylyl cyclase activity (Chakrabarti et al. 1995). As a compensatory mechanism, activity of adenylyl cyclase and other components of the cAMP signaling pathway are upregulated (Sharma et al. 1975; Chan and Lutfy 2016). This causes increased “rebound” firing in MOR-expressing VTA GABAergic neurons during the opioid-withdrawn state (Bonci and Williams 1997). Greater inhibition from these MOR-expressing GABAergic cells in the VTA therefore results in decreases in DA neuron activity during withdrawal (Diana et al. 1995; 1999). Projections from MOR-expressing neurons outside of the VTA, for example, in the medial habenula (Boulos et al. 2020) and rostromedial tegmental nucleus (Kaufling and Aston-Jones 2015; Bobzean et al. 2019), also contribute to modulations of DA neuron firing during withdrawal. Another potential regulator of DA release during withdrawal are the cholinergic interneurons of the striatum, which modulate DA release by inducing action potentials in the axons of DA neurons (Liu et al., 2022). Cholinergic interneurons express MORs (Ponterio et al. 2013; Mamaligas et al. 2016; Collins et al. 2019), and acetylcholine release is increased during precipitated withdrawal (Rada et al. 1991, 1996).
Decreases in baseline sustained (i.e., tonic) DA neuron activity that occur during opioid withdrawal (Kaufling and Aston-Jones 2015; George et al. 2022) may serve to make opioid-induced changes in phasic DA signaling more salient during withdrawal. In other words, withdrawal may be critical to the development of addiction because it enhances the “signal-to-noise” ratio of the DA response (Wanat et al. 2009; Zhang et al. 2012). This enhanced phasic DA signal that occurs after reexposure to drugs or drug-paired cues under withdrawal conditions may serve as a critical mechanism in the motivation to continue drug use.
In addition to reductions in DA levels, opioid withdrawal is characterized by plasticity in glutamatergic signaling in the NAc, originating from cortical, thalamic, or amygdalar inputs (Zhu et al. 2016; Hearing et al. 2018). In morphine withdrawal-exposed striatal cells, evidence shows increased phosphorylation of the AMPA receptor subunit GluR1 and increased AMPA receptor-mediated currents (Chartoff et al. 2003). Additionally, experimentally elevating GluR1 levels in the NAc enhances drug-induced CPA and AMPA receptor antagonist infusion in the NAc shell of morphine-dependent rats prevents naloxone-induced withdrawal CPA (Carlezon and Thomas 2009). Together, these findings suggest that heightened striatal glutamate is another important mediator of withdrawal-induced aversion (Russell et al. 2016).
Basolateral amygdala
The basolateral amygdala (BLA) signals the value of environmental stimuli, and as such has an important role in associative processes necessary for emotional learning (Wassum and Izquierdo 2015). Through its connections with areas such as the thalamus and sensory cortices, the BLA receives information about the external environment and guides behavior through its connections with the central amygdala (CeA), NAc, dorsal medial striatum, and bed nucleus of the stria terminalis (BNST) (Weller and Smith 1982; van Vulpen and Verwer 1989; McDonald 1998; Ambroggi et al. 2008; Vertes et al. 2015; Kim et al. 2017). The role of BLA in learning about aversive and anxiogenic stimuli is well documented (Maren 2003; O’Neill et al. 2018), so it is not surprising that conditioned opioid withdrawal memories are stored here (Frenois et al. 2002; Hellemans et al. 2006; Lucas et al. 2008; Lyons et al. 2013; Franco-García et al. 2022). It is important to note that this function of associating the negative emotional effects of withdrawal with previously neutral cues is likely the primary role of the BLA in opioid withdrawal, rather than mediating aversion itself. As evidence of this, BLA lesions prevent the acquisition of conditioned withdrawal behavior (i.e., suppression of a food-seeking response) in morphine-dependent rats but do not eliminate the acute expression of withdrawal signs (Schulteis et al. 2000). Precipitated withdrawal alone (without conditioning) increases cFos expression in a number of limbic brain regions, but not in the BLA (Frenois et al. 2002). Further, lesions of BLA reduced but did not eliminate acquisition of morphine withdrawal-induced CPA, suggesting that this manipulation does not prevent withdrawal-induced aversion (Watanabe et al. 2002b).
The BLA-dependent mechanisms underlying conditioned opioid withdrawal include a number of neurochemical systems and connections with cortical and hippocampal circuits. Reduced DA transmission in the BLA is correlated with the magnitude of withdrawal-induced place aversion (García-Pérez and Milanés 2020). BLA projections to the prelimbic cortex express cFos in response to withdrawal-induced CPA and optogenetically inhibiting these neurons eliminates expression of CPA (Song et al. 2019). Similarly, contextual fear conditioning is stronger when performed in a context previously paired with opioid withdrawal, and this effect involves projections from prelimbic cortex to BLA (Seno et al. 2022). Recruitment of BLA during opioid withdrawal also involves the corticotropin releasing hormone (CRH) system, as a CRH receptor 1 antagonist prevents acquisition of morphine CPA and withdrawal-induced increases in BDNF expression in this region (Martínez-Laorden et al. 2020).
Although studies using place conditioning have found that BLA’s role is primarily an associative one, other studies have implicated this structure in opioid withdrawal-induced defensive behaviors. Pharmacological inactivation of the BLA prevents elevations of the startle reflex, a behavioral index of anxiety-like behavior, during withdrawal from a single morphine exposure (Harris et al. 2006). Chemogenetic inhibition of BLA inputs and projections to the ventral hippocampus have also recently been shown to attenuate anxiogenic behaviors as measured on the elevated plus maze and open field test 2 weeks after cessation of chronic opioid exposure (Deji et al. 2022).
In addition to being required for forming memories of withdrawal, the BLA may mediate enhancements in the incentive value of rewards experienced during opioid withdrawal. Receipt of reward during opioid withdrawal enhances reward-seeking in rats (Hutcheson et al. 2001) and an elegant demonstration by the Wassum lab established that this effect is blocked by the inactivation of MORs in the BLA (Wassum et al. 2016). These BLA MORs appear to be necessary for recall of outcome-specific reward memories, and their recruitment by exogenous opioids likely plays an important role in promoting drug taking behaviors (Lichtenberg and Wassum 2017). Another intriguing study in mice exposed to chronic morphine and withdrawal suggests that this treatment enhances learning about fearful stimuli by enhancing GluA1-dependent plasticity in the BLA (Pennington et al. 2020). Thus, the role of withdrawal in shaping associated learning processes in the BLA appears to apply to both aversive and appetitive stimuli.
Central amygdala
The central amygdala (CeA) mediates the expression of behavioral output in a wide range of circumstances, both aversive and appetitive, and plays an important role in action selection and incentive motivation (Fadok et al. 2018; Warlow and Berridge 2021). This latter function of the CeA is particularly relevant to the maladaptive seeking responses that characterizes OUD. CeA plasticity induced by repeated cycle of opioid exposure and withdrawal may narrow and intensify behaviors directed toward drugs and drug-paired cues. In support of this idea, CeA has been shown to be necessary for the expression of conditioned and unconditioned negative emotional signs of opioid withdrawal.
Lesioning the CeA significantly reduces morphine withdrawal-induced CPA in rats (Watanabe et al. 2002b), suggesting that this nucleus is critical for the production of avoidance responses during withdrawal. The central importance of the CeA in withdrawal-induced aversion is also supported by observations of cFos expression and increases in CRE-mediated transcription in this region after morphine withdrawal (Stornetta et al. 1993; Gracy et al. 2001; Shaw-Lutchman et al. 2002; Frenois et al. 2002; Veinante et al. 2003; Baidoo et al. 2021) and the finding that CPA and suppression of operant responding for food can be precipitated following systemic morphine injection via intra-CeA infusion of a MOR antagonist (Heinrichs et al. 1995; Criner et al. 2007). Anxiety-like behavior, measured as startle potentiation, during acute opioid withdrawal is also prevented when the CeA is pharmacologically inhibited (Harris et al. 2006; Cabral et al. 2009).
A number of neurochemical systems in the CeA have been implicated in opioid withdrawal. Withdrawal-induced CPA is attenuated by intra-CeA infusion of glutamate receptor antagonists (Watanabe et al. 2002a; Ishida et al. 2008) and targeted deletion of NMDA receptors (Glass et al. 2008). The GluA1 subunit of the AMPA receptor is upregulated in the CeA during morphine withdrawal and suppression of GluA1 prevented elevated morphine-seeking behavior in withdrawn rats (Hou et al. 2015) (although this same treatment enhanced morphine withdrawal CPA, see Cai et al. 2020). Increased basal GABAergic transmission has also been demonstrated during opioid withdrawal, an effect that is mediated by activations in cAMP signaling (Bajo et al. 2014; Guo et al. 2019). Specifically, firing of CeA parvalbumin inhibitory interneurons, which regulate the activity of principal CeA neurons, is increased during withdrawal from morphine. Further, optogenetic inhibition of these neurons reduced expression of withdrawal-induced CPA, avoidance of the open arms on the elevated plus maze, and anhedonia in a saccharin preference test, whereas stimulation increased these affective withdrawal signs (Wang et al. 2016).
An important player in the CeA’s response to opioid withdrawal is the neuropeptide CRH. CRH receptor blockade in the CeA reduces CPA and lesion of CeA CRH-containing neurons attenuates expression of conditioned operant suppression of food seeking (Heinrichs et al. 1995; McNally and Akil 2002), suggesting an important role for CRH projections to and from this nucleus. In addition to attenuating negative affective signs of withdrawal, inhibition of the parvalbumin interneurons described above reduces upregulated levels of CRH mRNA in the CeA (Wang et al. 2016). There is also evidence to suggest that reciprocal connections between the VTA and CeA CRH neurons are required for opioid withdrawal-induced CPA. 6-Hydroxydopamine lesions of the CeA-projecting VTA DA neurons impair the formation of a place aversion during withdrawal (Xu et al. 2012). Further, CRH-containing neurons that project to the VTA are activated during withdrawal and participate in the formation of withdrawal-induced CPA (Jiang et al. 2021). Considering that stimulation of CRH neurons in the CeA has been shown to increase incentive motivation (Baumgartner et al. 2021), activation of this system during opioid withdrawal may serve as a neural substrate for the intense, maladaptive seeking behavior that characterizes addiction.
Bed nucleus of the stria terminalis
The role of the BNST, which is also part of the extended amygdala, in opioid withdrawal fits with its known role in mediating sustained defensive responses and stress-related behaviors (Lebow and Chen 2016). The BNST makes connections with brain regions already discussed, such as the amygdala, NAc, and VTA and projects to and influences neurons in the paraventricular nucleus of the hypothalamus that regulate the hypothalamic–pituitary–adrenal (HPA) axis (Song et al. 2020). Afferent projections from the locus coeruleus and other noradrenergic nuclei in the brainstem are also present here and play an important role in BNST activity during opioid withdrawal.
As evidence of the BNST’s involvement in opioid withdrawal, cFos is enhanced in this region during withdrawal and is correlated with the level of withdrawal-induced place aversion (Gracy et al. 2001; Frenois et al. 2002; Veinante et al. 2003; Nakagawa et al. 2005). Infusion of a MOR antagonist into the BNST also precipitates withdrawal, as measured by suppression of operant responding for food (Criner et al. 2007). Inactivation of BNST reduces the magnitude of withdrawal-potentiated startle during acute opioid withdrawal (Harris et al. 2006) and BNST lesions reduce withdrawal following chronic morphine exposure (Nakagawa et al. 2005). BNST neurons are more excitable following a history of opioid dependence (Francesconi et al. 2017), and this may result from changes in local inhibitory signaling. For example, systemic treatment with the GABA-B receptor agonist baclofen reduces morphine withdrawal-induced anxiety-like behavior and prevents decreases in BNST brain derived neurotrophic factor (BDNF) (Pedrón et al. 2016). Changes in BNST GABAergic signaling have also been observed during morphine withdrawal in mice, although the direction of the effects diverged in males and females (Luster et al. 2020). Activation of BNST during withdrawal appears to be mediated through projections from the CeA, as CeA lesions prevent withdrawal-induced increases in cFos immunoreactivity in the BNST (Nakagawa et al. 2005).
The BNST has the most significant density of noradrenergic inputs over any other brain region, and BNST norepinephrine (NE) signaling is important in mediating negative affect during opioid withdrawal. Extracellular levels of NE are increased in the ventral BNST after chronic morphine treatment (Fuentealba et al. 2000). Additionally, enhanced NE increases withdrawal-induced place aversion, whereas inhibiting NE activity blocks this behavior (Brownstein and Palkovits 1984; Aston-Jones et al. 1999; Delfs et al. 2000). The effects of NE are mediated by ꞵ-adrenergic receptors, as suggested by findings that withdrawal-induced cFos expression in the BNST is reduced by pretreatment with a ꞵ-adrenergic receptor antagonist and that intra-BNST ꞵ receptor antagonism reduces withdrawal-induced CPA (Aston-Jones et al. 1999). A major source of BNST NE during withdrawal are the brainstem nuclei that project via the ventral noradrenergic bundle, i.e., the A1 and A2 cell groups (Aston-Jones et al. 1999; Delfs et al. 2000).
Stress systems
Opioid withdrawal is known to induce a robust peripheral stress response through activation of the HPA axis and the sympathetic nervous system. Release of cortisol and NE from the adrenal glands circulates systemically and impacts circuits throughout the central nervous system. Thus, activation of stress systems has the potential to impact all of the circuits discussed above and more.
The production and secretion of glucocorticoids (e.g., cortisol in humans and corticosterone in rodents) is controlled by the HPA axis. Neurons in the paraventricular nucleus of the hypothalamus (PVN) release CRH into the anterior pituitary, which releases adrenocorticotropic hormone (ACTH) into the bloodstream to trigger glucocorticoid secretion from the adrenal cortex (Fulford and Harbuz 2005; Watson and Mackin 2006; Glynn et al. 2013). During withdrawal from chronic morphine treatment, corticosterone and ACTH levels are increased in rodents (Fuertes et al. 2000; Houshyar et al. 2003; Almela et al. 2012; Navarro-Zaragoza et al. 2021). Additionally, formation of a withdrawal-induced place aversion and induction of delta FosB expression in NAc, BNST, CeA, and PVN is impaired following adrenalectomy (García-Pérez et al. 2012, 2016; García-Pérez and Milanés 2020), although somatic withdrawal signs remain intact. Clinical findings also suggest increased HPA activity in humans during withdrawal. Increased cortisol has been observed during spontaneous and naloxone-precipitated withdrawal in individuals who are dependent on opioids (Tennant et al. 1991; Culpepper-Morgan and Kreek 1997; Nava et al. 2006; Zhang et al. 2008).
HPA axis activation results from activity in the PVN during withdrawal, as evidenced by cFos expression in CRH-containing neurons and increases in CRH mRNA in the PVN (McNally and Akil 2002; Houshyar et al. 2003; Hamlin et al. 2004), although decreases in CRH mRNA have also been observed (Milanés et al. 2002). The important role that CRH plays in withdrawal has also been demonstrated in mice with genetic deletion of CRH-1 or CRH-2 receptors (Contarino and Papaleo 2005; Ingallinesi et al. 2012; García-Carmona et al. 2015a) and through systemic pharmacological blockade of CRH receptors (Stinus et al. 2005; Skelton et al. 2007). It is important to note here that systemic manipulations such as these cannot differentiate between the actions of CRH in the HPA axis and in other extra-hypothalamic brain regions such as the CeA. Noradrenergic projections from the A1 and A2 cell groups are one mechanism through which PVN CRH neurons are recruited during withdrawal. Direct noradrenergic projections to PVN are activated during withdrawal from chronic morphine exposure (Fuertes et al. 2000; Benavides et al. 2003). The BNST is also critical in regulating PVN CRH activity (Forray and Gysling 2004), meaning that NE release here likely also contributes to HPA axis activation during opioid withdrawal, although this has yet to be demonstrated.
Despite robust evidence for glucocorticoid release during opioid withdrawal and the impacts of adrenalectomy on place aversion mentioned above (García-Pérez et al. 2016; García-Pérez and Milanés 2020), blockade of glucocorticoid receptors (GR) during withdrawal has been found ineffective in modifying affective signs of withdrawal (Solecki et al. 2019). The adrenal gland is also an important source of systemic epinephrine/norepinephrine that is activated by sympathetic projections from the spinal cord, which receive inputs from central nervous system nuclei, including the PVN (Hosoya et al. 1991). In human patients, naloxone-precipitated withdrawal increases plasma concentrations of epinephrine and NE (Kienbaum et al. 1998), which may underlie some of the effects of adrenalectomy on withdrawal-induced aversion. Systemic treatment with β adrenergic receptor antagonists attenuates affective signs of opioid withdrawal (Solecki et al. 2019), but much like systemic studies of the CRH system, it is difficult to ascribe these effects to sympathoadrenal vs. central mechanisms. Thus, the exact mechanisms through which the HPA axis contributes to the affective component of withdrawal have yet to be fully clarified.
Neural circuits mediating somatic withdrawal signs
Some of the most prominent aspects of the opioid withdrawal syndrome include the somatic signs and symptoms. In humans, the somatic withdrawal syndrome begins within 24 h of discontinuing opioids and reaches its peak within the next few days, though this time course can vary due to individual differences and whether an individual is discontinuing short or long-acting opioids (Dunn et al. 2019). Typical somatic signs of withdrawal include gastrointestinal issues such as diarrhea and nausea and vomiting, changes in pain sensitivity including muscle and joint aches and hyperalgesia, hot and cold flashes, piloerection, pupil dilation, lacrimation, rhinorrhea, and yawning (Bradley et al. 1987; Dunn et al. 2019). Many of these signs can be measured in rodent models of opioid withdrawal, and species-specific signs can be observed as well. Scales of somatic withdrawal in rodents frequently include signs such as jumping, rearing, wet dog shakes, abdominal constrictions, diarrhea, salivation, hunched posture, and teeth chattering (Gellert and Holtzman 1978; Maldonado et al. 1992a, b; Maldonado and Koob 1993).
In general, somatic signs and symptoms of opioid withdrawal can be attributed to compensatory rebound activation and neuronal hyperexcitability following opioid-induced inhibition. Peripheral MORs are found in the terminals of afferent sensory neurons and in enteric neurons (Stein 2013; Galligan and Akbarali 2014), and these receptors are responsible for the gastrointestinal effects and other peripheral signs such as lacrimation, rhinorrhea, and salivation (Bianchetti et al. 1986; Maldonado, Negus, Koob, 1992a). For example, withdrawal-induced diarrhea was shown to be dependent on peripheral opioid receptors by administration of the peripherally selective opioid antagonist SR 58,002 C, which induces diarrhea, but not jumping, in morphine-dependent mice (Bianchetti et al. 1986). Similarly, the widely available over-the-counter anti-diarrheal medication, loperamide, primarily reduces diarrhea by activating MORs in the gut (Kang et al. 2016).
Within the central nervous system, MOR antagonism in a wide array of brain regions precipitates withdrawal signs in rodents, with especially strong effects observed in two contiguous regions of the brainstem, the periaqueductal gray (PAG) and locus coeruleus (LC) (Maldonado et al. 1992a, b; Koob et al. 1992). Infusion of the opioid receptor antagonist methylnaloxonium into the PAG produced signs such as teeth chatter, jumping, rearing, piloerection, and loss of body weight in rats (Maldonado et al. 1992a, b) as well as severe agitation and development of a conditioned place aversion (Stinus et al. 1990). Manipulation of protein kinases in this region also attenuates withdrawal behaviors (Maldonado et al. 1995; Punch et al. 1997). Further, PAG neurons express cFos following precipitated withdrawal (Chieng et al. 1995) and have been shown to develop electrophysiological tolerance and withdrawal following opioid exposure (Chieng and Christie 1996). This sensitivity of the PAG to withdrawal is not surprising considering the large number of opioid receptors expressed in this region and a well-characterized role in opioid-induced analgesia via descending projections to the dorsal horn of the spinal cord. Because of its important role in modulation of pain, the PAG is also the primary mediator of the enhanced pain sensitivity characteristic of withdrawal. Withdrawal-induced hyperexcitability in GABAergic interneurons of the PAG reduces the ability of PAG output neurons to suppress ascending pain signals (Hack et al. 2003; Bagley et al. 2005, 2011).
Another brain region that has received a great deal of attention for its role in somatic signs of opioid withdrawal is the LC, the primary source of NE in the brain. Rasmussen and colleagues demonstrated that the time course of multiple somatic withdrawal signs paralleled increased activity in the LC (Rasmussen et al. 1990). Increased cFos activity has also been observed in the LC during precipitated withdrawal (Hayward et al. 1990; Alvarez-Bagnarol et al. 2022). Increases in LC activity are mediated by excitatory inputs from the nucleus paragigantoceullularis and orexinergic inputs from the lateral hypothalamus in addition to MOR antagonism (Rasmussen and Aghajanian, 1989; Akaoka and Aston-Jones, 1991; Aghajanian et al. 1994; Hooshmand et al. 2019). Maldonado and Koob showed that lesioning the LC in dependent rats reduces a wide breadth of somatic withdrawal signs, including jumping, hyperactivity, and wet dog shakes (Maldonado and Koob 1993). Additionally, methylnaloxonium injection directly into the LC enhances somatic withdrawal signs in dependent rats (Maldonado et al. 1992a, b) while intra-LC AMPA receptor antagonism (Rasmussen et al. 1996; Taylor et al. 1998), GABA-B receptor agonism (Riahi et al. 2009), and orexin-1 receptor antagonism (Azizi et al. 2010) reduces their expression. A role for noradrenergic involvement in withdrawal symptoms is further demonstrated in clinical data with α2 adrenergic agonists demonstrating promise for reduction of withdrawal signs and improvement of treatment outcomes in addicted individuals (Gowing et al. 2016). Despite this robust evidence suggesting a role for the LC, studies using neurochemical lesion techniques to target LC noradrenergic neurons have failed to find a role for these neurons in the somatic component of withdrawal (Chieng and Christie, 1995; Christie et al. 1997; Caillé et al., 1999). The precise role of the LC in somatic signs of withdrawal therefore remains an unresolved issue (Williams et al. 2001).
Discussion
Taken together, the evidence reviewed suggests that opioid withdrawal results in severely dysregulated reward and stress systems. Because preclinical studies in rodents do not provide insight into an animal’s affective consciousness it is impossible to conclude that brain regions such as the amygdala or NAc mediate the subjective emotional experience of withdrawal. Still, the reward and stress systems reviewed above can be said to work together to produce behaviors that represent negative emotional states, such as aversion, anhedonia, and anxiety. Further, neural changes in these circuits parallel the psychological phenomena of OUD, such as an unusual focus on drug reward at the expense of natural rewards and self-medication or relapse in response to anxiety or stress.
Role in addiction
A number of theoretical frameworks have been proposed to explain the psychology of addictive behavior, and there is ongoing debate over the relative contribution of negative affect, habit, decision-making processes, and incentive motivation to maladaptive drug seeking (Field and Kersbergen, 2020; Hogarth 2020; Epstein 2020). Relief of withdrawal has long been seen as a key driver of continued drug use although thinking about the nature of this contribution has shifted over time (Wikler 1948; Dole et al. 1966; Koob and Volkow 2010). One important observation that strongly supports the supposition that withdrawal is critical to initiating and maintaining the cycle of addiction is the observation that opioids are most likely to be abused when administered intermittently (vs. continuously). For example, rapidly delivered and short-acting heroin has a greater abuse liability than long-acting methadone (Stimmel and Kreek 2000). Preclinical markers of addiction such as psychomotor sensitization and opioid self-administration are similarly observed following intermittent opioid exposure only (Vanderschuren et al. 1997; Rothwell et al. 2010; Yu et al. 2014; Lefevre et al. 2020; Fragale et al. 2021). As such, withdrawal, or perhaps more specifically reexposure to opioids during withdrawal, drives the development of addictive behaviors by creating an altered neural state that contributes to maladaptive drug-induced plasticity. These withdrawal-induced changes in the function of the mesolimbic dopamine system, BLA, CeA, and BNST consequently set the stage for opioids and opioid-paired cues to become more salient, valued, and motivating than other stimuli.
An important line of evidence suggesting a role for opioid withdrawal in promoting addiction comes from the experiences of patients with OUD. For example, a study of patients seeking treatment for prescription opioid use found withdrawal avoidance to be the number one factor motivating current use (Weiss et al. 2014) and multiple other studies have found at least a partial role for withdrawal when examining factors that motivate opioid use (Heiwe et al. 2011; Harocopos et al., 2016; Stumbo et al. 2017; Frank et al. 2016; Cicero and Ellis 2017; AbdelWahab et al. 2018). Fear of withdrawal also contributes to risky decision making and continued use despite negative consequences. Individuals with OUD report making risky medical decisions (Summers et al. 2018) and avoiding treatment for their drug use (Mitchell et al. 2009) over concerns related to opioid withdrawal.
The preclinical literature also supports a role for withdrawal in motivating opioid consumption, although the literature is mixed. Injection of naloxone in morphine-dependent rats increases responding for heroin at low doses (e.g., 0.01 mg/kg), but higher doses of naloxone (e.g., 0.03 mg/kg) do the opposite (Carrera et al. 1999). Stimuli conditioned to acutely precipitate heroin withdrawal can also stimulate heroin self-administration behavior in rats and blunt the sensitivity of the brain reward system, as measured with ICSS (Kenny et al. 2006). Experience with heroin during withdrawal has also been shown to be necessary for withdrawal to motivate future drug seeking in rats (Hutcheson et al. 2001). Work in rhesus monkeys has further demonstrated that heroin (vs. food) choice increases following cessation of 21-h heroin access (Negus, 2006; Negus and Rice 2009). A number of pharmacological manipulations that reduce signs of opioid withdrawal reduce self-administration behaviors, including reinstatement, in rats, and these manipulations can include a wide range of pharmacological targets including α1 adrenergic receptors (Greenwell et al. 2009), CRF (Park et al. 2015), PPAR-γ, (de Guglielmo et al. 2017), dopamine D3 receptors (de Guglielmo et al. 2019), and 5α-Reductase (Bosse et al. 2021). However, drugs targeting withdrawal signs did not reduce heroin choice in monkeys (Negus and Rice 2009) and have not yet successfully translated to the clinic. In contradiction to findings supporting a role for withdrawal in opioid consumption, another line of evidence suggests that opioid withdrawal-induced anhedonia (i.e., increases in ICSS thresholds) is associated with reduced morphine self-administration behaviors (Holtz et al. 2015; Swain et al. 2020). Examinations of the influence of withdrawal on heroin seeking also fail to find an effect of naloxone injection on reinstatement of lever pressing (Shaham and Stewart 1995; Shaham et al. 1996).
While early theories postulated a role for relief of physical signs of withdrawal in addiction (Wikler 1948; Dole et al. 1966), views have shifted to consider the affective signs and symptoms of withdrawal to be of primary motivational significance (Koob and Volkow 2010). One reason for this distinction is that while the somatic component of opioid withdrawal resolves in a few days to weeks, affective signs can persist much longer. Additionally, the low abuse liability of many pharmacologically active substances that nonetheless produce tolerance and a state of withdrawal upon abstinence (e.g., caffeine) argues against the idea that physical dependence alone is sufficient for addiction (Heinz et al., 2020). Indeed, opioids themselves can produce opioid withdrawal syndrome and opioid dependence separately from OUD (Kosten and Baxter 2019; Ballantyne et al. 2019). Preclinical work also suggests a dissociation in the neural substrates mediating the affective and somatic components of withdrawal (see mechanisms reviewed above; Frenois et al. 2002) and that somatic signs are not predictive of affective withdrawal or opioid self-administration behaviors (Mucha 1987; Swain et al. 2020). That said, while affective withdrawal signs may be more reflective of neural plasticity in circuits responsible for addictive behavior, somatic signs should not be completely dismissed when considering the role of withdrawal in addiction.
One reason to consider a role for somatic withdrawal in OUD is that physical and emotional signs are not entirely separable. Although not typically life-threatening, acute somatic withdrawal signs such as aches and pains, nausea and diarrhea, and hot/cold flashes are severely unpleasant (Dunn et al. 2019). As such, the symptoms of opioid withdrawal are thought to resemble a severe flu-like illness (Farrell 1994) that individuals are highly motivated to avoid (Summers et al. 2018). These symptoms therefore construct a highly salient event composed of multiple aversive physical symptoms, and there is evidence to suggest that at least some addicted individuals continue drug use to avoid these symptoms. For instance, opioid-dependent patients cite a fear or concern about increased pain sensitivity as one of the primary reasons why they wish not to experience withdrawal (Stumbo et al. 2017; Frank et al. 2016). Because many of the symptoms of opioid withdrawal are uncomfortable and painful, it is not possible to categorize them as purely “physical.” There may also be overlap between the physical and emotional aspects of withdrawal as anticipation of somatic withdrawal signs produces distress and anxiety (Bruneau et al. 2021). It should further be noted that there is overlap in the neural structures demonstrated to produce affective and somatic signs of withdrawal in animal models. For example, the dopaminergic system contributes to somatic withdrawal signs in rats (Harris and Aston-Jones 1994; Chartoff et al. 2006) and methylnaloxonium infused into the PAG of morphine-dependent rats produces a place aversion (Stinus et al., 1990). Therefore, it is important to recognize that physical dependence also contributes to negative affect and that avoidance of these symptoms is in fact a powerful influence in maintaining opioid use.
Implications for relapse and treatment
In the early stages of abstinence from opioids, negative emotional signs and symptoms of withdrawal contribute to the motivation to continue drug use as individuals take drugs to avoid the associated pain and dysphoria. As discussed above, exposure to drugs during this state promotes the neural plasticity that underlies compulsive use and drug craving (Koob 2020). Although this acute phase of withdrawal is a temporary state, persistent emotional dysregulation and sensitization of pain and stress systems far beyond the detoxification stage leave individuals vulnerable to opioid craving months or even years after abstinence is achieved (Sinha 2009; Koob 2020). Cues conditioned to withdrawal can also trigger craving and promote renewed drug taking (Pantazis et al. 2021). Thus, the state of protracted withdrawal is an important contributor to relapse in individuals recovering from OUD.
In animals, relapse is modeled by examining reinstatement of drug seeking following exposure to drugs, drug-paired cues, stress, or withdrawal-paired cues (Shaham and Stewart 1995; Kenny et al. 2006; Mantsch et al. 2016; Bossert et al. 2019). It is notable that many of the neural circuits and neurotransmitters implicated in affective withdrawal from opioids have also been identified as participants in reinstatement, particularly when it is induced by stress (for a thorough review of the stress-induced reinstatement model see Mantsch et al. 2016). These overlapping neural circuits include the CeA, BNST, VTA, and NAc and within these roles for NE, CRH, DA, and glutamate have been found. Precipitators of relapse such as stress consequently engage the same neural circuits that are sensitized during repeated cycles of drug exposure and withdrawal. Thus, withdrawal is critical in setting the stage for relapse to be initiated by pain, stress, and cues in the long term.
The complexity and interconnectivity of the circuits recruited during opioid withdrawal may explain, at least in part, the difficulty of treating OUD and preventing relapse. Some of the most successful treatments available today help patients achieve abstinence by avoiding the withdrawn state. For instance, pharmacological maintenance therapies such as methadone and buprenorphine are long-acting opioid receptor agonists (Joseph et al. 2000). The chronic, stable levels of opioid receptor agonism provided by these pharmacotherapies prevent the highs and lows associated with opioid use, including the acute and protracted withdrawal states. Of course, despite the success and critical importance of pharmacological maintenance therapies to current OUD treatment strategies, a limitation of these treatments is that they do not correct the long-lasting brain circuit alterations that come along with addiction and influence complex phenomena such as salience, value, or motivation. For example, methadone maintenance significantly reduces heroin reinstatement after a priming injection of heroin; however, that study saw no effect of methadone maintenance on stress-induced reinstatement (Leri et al. 2004). Furthermore, although methadone maintenance therapy is effective at reducing relapse while individuals are currently undergoing treatment (Bell and Strang 2020), relapse to opioids still remains a significant concern after completion of methadone therapy programs (Magura and Rosenblum 2001). Thus, although methadone maintenance therapy is effective in the short-term, it does not adequately address the issue of long-term relapse susceptibility. In this sense, it may allow providers and patients to feel comfortable for as long as the individual is in treatment, but any holistic treatment plan that does not adequately prevent relapse years later ultimately fails to achieve the goal of long-term success. Thus, the challenge for the future is to develop treatment strategies that normalize some of the long-term neural circuit alterations resulting from opioid use and withdrawal.
Future directions
Given the importance of withdrawal in maintaining the cycle of addiction and setting the stage for relapse in abstinent patients, continued research into the neural mechanisms underlying withdrawal-induced negative emotional states is necessary. Contemporary research has begun to assess the influence of additional neurotransmitter systems and brain circuits, and this work has outlined some promising new avenues for OUD treatments. In terms of opioid receptors, most of the studies reviewed above have focused on the role of MORs. A few recent studies suggest that kappa opioid receptor (KOR) stimulation, which is known to inhibit striatal DA release and increase aversion (for review, see Bruijnzeel 2009), may also contribute to aversion during withdrawal. For instance, systemic KOR antagonism reduces morphine withdrawal-induced CPA (Kelsey et al. 2015). This effect may be mediated by KORs in the BLA and CeA, which are thought to be involved in anxiety-like behavior (Knoll et al. 2011). Advanced genetic and neural circuit approaches (e.g., selective gene knockout, optogenetics) will help determine the exact mechanisms by which KORs may be involved in the described circuit. Another popular treatment currently used in OUD patients is α2 adrenergic agonists. As has been discussed, the adrenergic system plays a crucial role in the physical symptoms of withdrawal and α2 agonists are quite effective at reducing these symptoms (Gowing et al. 2016). Furthermore, the use of α2 agonists decreases stress-induced drug seeking in rats as well as craving in humans (Sinha et al. 2011).
Another promising target is the cannabinoid type 1 receptor (CB1R). This system is increasingly being implicated in anxiety and negative affect (Witkin et al. 2005) and modulation of CB1R signaling in the BNST, BLA, CeA, and insular cortex all attenuate morphine withdrawal-induced place aversion (Wills et al. 2016, 2017). Other neurotransmitter systems such as adenosine (Jafarova Demirkapu et al. 2020) and orexin (Laorden et al. 2012) have also been implicated in withdrawal and are worthy of further investigation.
In addition to new neurochemical targets, future studies into the role that cortical circuits play in withdrawal are also needed. The insular cortex, known for its role in interoception and self-awareness, has been identified as a potential therapeutic target for addictive disorders (Naqvi et al. 2007; Dinur-Klein et al. 2014; Droutman et al. 2015). This region is highly interconnected with the brain circuits reviewed here, including the BLA, CeA, and BNST, and it receives DAergic inputs from the VTA (Gogolla 2017). Inactivation of the insula in rats has been shown to disrupt the formation of morphine withdrawal-induced CPA (Li et al. 2013) as do manipulations of CB1R signaling in this area (Wills et al. 2016). The anterior cingulate cortex (ACC) is another cortical region implicated in emotional processes and highly interconnected with subcortical circuits known to participate in withdrawal. Neuroimaging suggests that the ACC is activated during opioid withdrawal in men (Chu et al. 2015) and inhibition of this region has recently been demonstrated to prevent somatic signs of opioid withdrawal in male mice (McDevitt et al. 2021).
Finally, female subjects are currently vastly underrepresented in the literature on the neural mechanisms of opioid withdrawal. The majority of studies reviewed here were conducted exclusively in male subjects. While many areas of addiction neuroscience research have made progress in regard to the study of sex differences (Radke et al. 2021), sex differences in opioid withdrawal remain almost entirely unexplored. Male rodents are known to exhibit greater somatic signs of morphine withdrawal than females (Craft et al. 1999; Kest et al. 2001; Radke et al. 2013; Bobzean et al. 2019), which we speculate may depend on the PAG given demonstrated sex differences in opioid-induced analgesia mediated by this region (Loyd and Murphy 2009). In regard to affective signs of withdrawal, conditioning to morphine withdrawal may be equivalent in male and female mice (García-Carmona et al. 2015b), although at least one report suggests that the acquisition of place aversion is dependent on estradiol in female rats (Martinez-Casiano et al. 2015). Similar increases in the acoustic startle response during withdrawal from acute morphine in male and female rats (Radke et al. 2013, 2015) have also been observed. Many studies additionally find sex differences in self-administration of opioids (Lynch and Carroll 1999; Cicero et al. 2003; Mavrikaki et al. 2017; Fulenwider et al. 2019; Smethells et al. 2020; Monroe and Radke 2021; Radke et al. 2021); however, several studies also demonstrate a lack of sex differences under certain self-administration parameters (Venniro et al. 2019; Venniro et al. 2017; Fredriksson et al. 2020; Bossert et al. 2022). Further research is needed to clarify the influence of biological sex on opioid-seeking behaviors and the extent to which withdrawal contributes to these behaviors in males vs. females.
Summary
In sum, negative affect during opioid withdrawal is a crucial motivational phenomenon in the cycle of addiction. It involves brain systems underlying reward and motivated behavior, learning and memory, and stress. These systems interact with one another under conditions of dysregulated reward systems, hyperactive stress, and enhanced sensitivity to environmental cues, contributing to the complicated pathophysiology underlying opioid addiction. Continued progress in this area over the next several decades has the potential to lead to new and better treatments for OUD and other addictive disorders.
References
AbdelWahab MA, Abou El Magd SF, Grella CE et al (2018) An examination of motives for tramadol and heroin use in an Egyptian sample. J Addict Dis 37:123–134
Aghajanian GK, Kogan JH, Moghaddam B (1994) Opiate withdrawal increases glutamate and aspartate efflux in the locus coeruleus: an in vivo microdialysis study. Brain Res 636(1):126–130
Ahmed SH (2004) Addiction as compulsive reward prediction. Science 306:1901–1902
Akaoka H, Aston-Jones G (1991) Opiate withdrawal-induced hyperactivity of locus coeruleus neurons is substantially mediated by augmented excitatory amino acid input. J Neurosci 11.12 (1991):3830–3839
Almela P, Navarro-Zaragoza J, García-Carmona J-A et al (2012) Role of corticotropin-releasing factor (CRF) receptor-1 on the catecholaminergic response to morphine withdrawal in the nucleus accumbens (NAc). PLoS ONE 7:e47089
Alvarez-Bagnarol Y, Marchette RC, Francis C, Morales M, Vendruscolo LF (2022) Neuronal correlates of hyperalgesia and somatic signs of heroin withdrawal in male and female mice. Eneuro 9(4)
Ambroggi F, Ishikawa A, Fields HL, Nicola SM (2008) Basolateral amygdala neurons facilitate reward-seeking behavior by exciting nucleus accumbens neurons. Neuron 59:648–661
American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (5th ed.). https://doi.org/10.1176/appi.books.9780890425596
Aston-Jones G, Delfs JM, Druhan J, Zhu Y (1999) The bed nucleus of the stria terminalis. A target site for noradrenergic actions in opiate withdrawal. Ann N Y Acad Sci 877:486–498
Azizi H, Mirnajafi-Zadeh J, Rohampour K, Semnanian S (2010) Antagonism of orexin type 1 receptors in the locus coeruleus attenuates signs of naloxone-precipitated morphine withdrawal in rats. Neurosci Lett 482:255–259
Bagley EE, Chieng BCH, Christie MJ, Connor M (2005) Opioid tolerance in periaqueductal gray neurons isolated from mice chronically treated with morphine. Br J Pharmacol 146:68–76
Bagley EE, Hacker J, Chefer VI et al (2011) Drug-induced GABA transporter currents enhance GABA release to induce opioid withdrawal behaviors. Nat Neurosci 14:1548–1554
Baidoo N, Wolter M, Holahan MR et al (2021) The effects of morphine withdrawal and conditioned withdrawal on memory consolidation and c-Fos expression in the central amygdala. Addict Biol 26:e12909
Bajo M, Madamba SG, Roberto M, Siggins GR (2014) Acute morphine alters GABAergic transmission in the central amygdala during naloxone-precipitated morphine withdrawal: role of cyclic AMP. Front Integr Neurosci 8:45
Ballantyne JC, Sullivan MD, Koob GF (2019) Refractory dependence on opioid analgesics. Pain 160:2655–2660
Baumgartner HM, Schulkin J, Berridge KC (2021) Activating corticotropin-releasing factor systems in the nucleus accumbens, amygdala, and bed nucleus of stria terminalis: incentive motivation or aversive motivation? Biol Psychiatry 89:1162–1175
Bell J, Strang J (2020) Medication treatment of opioid use disorder. Biol Psychiat 87(1):82–88
Benavides M, Laorden ML, García-Borrón JC, Milanés MV (2003) Regulation of tyrosine hydroxylase levels and activity and Fos expression during opioid withdrawal in the hypothalamic PVN and medulla oblongata catecholaminergic cell groups innervating the PVN. Eur J Neurosci 17:103–112
Berke JD (2018) What does dopamine mean? Nat Neurosci 21:787–793
Bianchetti A, Guidice A, Nava F, Manara L (1986) Dissociation of morphine withdrawal diarrhea and jumping in mice by the peripherally selective opioid antagonist SR 58002 C. Life Sci 39:2297–2303
Bobzean SAM, Kokane SS, Butler BD, Perrotti LI (2019) Sex differences in the expression of morphine withdrawal symptoms and associated activity in the tail of the ventral tegmental area. Neurosci Lett 705:124–130
Bonci A, Williams JT (1997) Increased probability of GABA release during withdrawal from morphine. J Neurosci 17:796–803
Bosse GD, Cadeddu R, Floris G, Farero RD, Vigato E, Lee SJ, ... & Peterson RT (2021) The 5α-reductase inhibitor finasteride reduces opioid self-administration in animal models of opioid use disorder. J Clin Invest 131(10)
Bossert JM, Hoots JK, Fredriksson I et al (2019) Role of mu, but not delta or kappa, opioid receptors in context-induced reinstatement of oxycodone seeking. Eur J Neurosci 50:2075–2085
Bossert JM, Townsend EA, Altidor LK-P, Fredriksson I, Shekara A, Husbands S, et al (2022) Sex differences in the effect of chronic delivery of the buprenorphine analogue BU08028 on heroin relapse and choice in a rat model of opioid maintenance. Br J Pharmacol 179:227–241
Boulos LJ, Ben Hamida S, Bailly J et al (2020) Mu opioid receptors in the medial habenula contribute to naloxone aversion. Neuropsychopharmacology 45:247–255
Bradley BP, Gossop M, Phillips GT, Legarda JJ (1987) The development of an opiate withdrawal scale (OWS). Br J Addict 82:1139–1142
Brownstein MJ, Palkovits M (1984) Classical transmitters in the CNS, Pt 1. Handbook of Chem Neuroanatomy 2:23–54
Bruijnzeel AW (2009) kappa-Opioid receptor signaling and brain reward function. Brain Res Rev 62:127–146
Bruneau A, Frimerman L, Verner M et al (2021) Day-to-day opioid withdrawal symptoms, psychological distress, and opioid craving in patients with chronic pain prescribed opioid therapy. Drug Alcohol Depend 225:108787
Cabral A, Ruggiero RN, Nobre MJ et al (2009) GABA and opioid mechanisms of the central amygdala underlie the withdrawal-potentiated startle from acute morphine. Prog Neuropsychopharmacol Biol Psychiatry 33:334–344
Cai Y-Q, Hou Y-Y, Pan ZZ (2020) GluA1 in central amygdala increases pain but inhibits opioid withdrawal-induced aversion. Mol Pain 16:1744806920911543
Caillé S, Espejo EF, Reneric J-P et al (1999) Total neurochemical lesion of noradrenergic neurons of the locus ceruleus does not alter either naloxone-precipitated or spontaneous opiate withdrawal nor does it influence ability of clonidine to reverse opiate withdrawal. J Pharmacol Exp Ther 290:881–892
Carlezon WA Jr, Thomas MJ (2009) Biological substrates of reward and aversion: a nucleus accumbens activity hypothesis. Neuropharmacology 56(Suppl 1):122–132
Carrera MR, Schulteis G, Koob GF (1999) Heroin self-administration in dependent Wistar rats: increased sensitivity to naloxone. Psychopharmacology 144:111–120
Centers for Disease Control and Prevention (2022) U.S. overdose deaths in 2021 increased half as much as in 2020 — but are still up 15%. Centers for Disease Control and Prevention. https://www.cdc.gov/nchs/pressroom/nchs_press_releases/2022/202205.htm
Chakrabarti S, Prather PL, Yu L et al (1995) Expression of the μ-opioid receptor in CHO cells: Ability of μ-opioid ligands to promote α-azidoanilido[32P]GTP labeling of multiple G protein α subunits. J Neurochem 64:2534–2543
Chan P, Lutfy K (2016) Molecular changes in opioid addiction: the role of adenylyl cyclase and cAMP/PKA system. Prog Mol Biol Transl Sci 137:203–227
Chartoff EH, Papadopoulou M, Konradi C, Carlezon WA (2003) Effects of naloxone-precipitated morphine withdrawal on glutamate-mediated signaling in striatal neurons in vitro. Ann N Y Acad Sci 1003:368–371
Chartoff EH, Mague SD, Barhight MF et al (2006) Behavioral and molecular effects of dopamine D1 receptor stimulation during naloxone-precipitated morphine withdrawal. J Neurosci 26:6450–6457
Chartoff EH, Barhight MF, Mague SD et al (2009) Anatomically dissociable effects of dopamine D1 receptor agonists on reward and relief of withdrawal in morphine-dependent rats. Psychopharmacology 204:227–239
Chieng B, Christie MJ (1996) Local opioid withdrawal in rat single periaqueductal gray neurons in vitro. J Neurosci 16:7128–7136
Chieng B, Keay KA, Christie MJ (1995) Increased fos-like immunoreactivity in the periaqueductal gray of anaesthetised rats during opiate withdrawal. Neurosci Lett 183:79–82
Christie MJ, Williams JT, Osborne PB, Bellchambers CE (1997) Where is the locus in opioid withdrawal? Trends Pharmacol Sci 18:134–140
Chu LF, Lin JC, Clemenson A et al (2015) Acute opioid withdrawal is associated with increased neural activity in reward-processing centers in healthy men: a functional magnetic resonance imaging study. Drug Alcohol Depend 153:314–322
Cicero TJ, Ellis MS (2017) Understanding the demand side of the prescription opioid epidemic: does the initial source of opioids matter? Drug Alcohol Depend 173(Suppl 1):S4–S10
Cicero TJ, Aylward SC, Meyer ER (2003) Gender differences in the intravenous self-administration of mu opiate agonists. Pharmacol Biochem Behav 74:541–549
Cole SL, Robinson MJF, Berridge KC (2018) Optogenetic self-stimulation in the nucleus accumbens: D1 reward versus D2 ambivalence. PLoS ONE 13:e0207694
Collins AL, Aitken TJ, Huang I-W et al (2019) Nucleus accumbens cholinergic interneurons oppose cue-motivated behavior. Biol Psychiatry 86:388–396
Contarino A, Papaleo F (2005) The corticotropin-releasing factor receptor-1 pathway mediates the negative affective states of opiate withdrawal. Proc Natl Acad Sci U S A 102:18649–18654
Craft RM, Stratmann JA, Bartok RE et al (1999) Sex differences in development of morphine tolerance and dependence in the rat. Psychopharmacology 143:1–7
Criner SH, Liu J, Schulteis G (2007) Rapid neuroadaptation in the nucleus accumbens and bed nucleus of the stria terminalis mediates suppression of operant responding during withdrawal from acute opioid dependence. Neuroscience 144:1436–1446
Crow TJ (1972) A map of the rat mesencephalon for electrical self-stimulation. Brain Res 36:265–273
Culpepper-Morgan JA, Kreek MJ (1997) Hypothalamic-pituitary-adrenal axis hypersensitivity to naloxone in opioid dependence: a case of naloxone-induced withdrawal. Metabolism 46:130–134
de Guglielmo G, Kallupi M, Scuppa G et al (2017) Pioglitazone attenuates the opioid withdrawal and vulnerability to relapse to heroin seeking in rodents. Psychopharmacology 234:223–234
de Guglielmo G, Kallupi M, Sedighim S et al (2019) Dopamine D3 receptor antagonism reverses the escalation of oxycodone self-administration and decreases withdrawal-induced hyperalgesia and irritability-like behavior in oxycodone-dependent heterogeneous stock rats. Front Behav Neurosci 13:292
Deji C, Yan P, Ji Y et al (2022) The basolateral amygdala to ventral hippocampus circuit controls anxiety-like behaviors induced by morphine withdrawal. Front Cell Neurosci 16:894886
Delfs JM, Zhu Y, Druhan JP, Aston-Jones G (2000) Noradrenaline in the ventral forebrain is critical for opiate withdrawal-induced aversion. Nature 403:430–434
Di Chiara G, Imperato A (1988) Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proc Natl Acad Sci U S A 85:5274–5278
Diana M, Pistis M, Muntoni A, Gessa G (1995) Profound decrease of mesolimbic dopaminergic neuronal activity in morphine withdrawn rats. J Pharmacol Exp Ther 272:781–785
Diana M, Muntoni AL, Pistis M et al (1999) Lasting reduction in mesolimbic dopamine neuronal activity after morphine withdrawal. Eur J Neurosci 11:1037–1041
Dinur-Klein L, Dannon P, Hadar A et al (2014) Smoking cessation induced by deep repetitive transcranial magnetic stimulation of the prefrontal and insular cortices: a prospective, randomized controlled trial. Biol Psychiatry 76:742–749
Dole VP, Nyswander ME, Kreek MJ (1966) Narcotic blockade. Arch Intern Med 118:304–309
Droutman V, Read SJ, Bechara A (2015) Revisiting the role of the insula in addiction. Trends Cogn Sci 19:414–420
Dunn KE, Huhn AS, Bergeria CL et al (2019) Non-opioid neurotransmitter systems that contribute to the opioid withdrawal syndrome: a review of preclinical and human evidence. J Pharmacol Exp Ther 371:422–452
Edwards S, Vendruscolo LF, Schlosburg JE et al (2012) Development of mechanical hypersensitivity in rats during heroin and ethanol dependence: alleviation by CRF1 receptor antagonism. Neuropharmacology 62:1142–1151
Epstein DH (2020) Let’s agree to agree: a comment on Hogarth (2020), with a plea for not-so-competing theories of addiction. Neuropsychopharmacology 45:715–716
Fadok JP, Markovic M, Tovote P, Lüthi A (2018) New perspectives on central amygdala function. Curr Opin Neurobiol 49:141–147
Farrell M (1994) Opiate withdrawal. Addiction 89(11):1471–1475
Field M, Kersbergen I (2020) Are animal models of addiction useful? Addiction 115:6–12
Forray MI, Gysling K (2004) Role of noradrenergic projections to the bed nucleus of the stria terminalis in the regulation of the hypothalamic–pituitary–adrenal axis. Brain Res Rev 47:145–160
Fox ME, Rodeberg NT, Wightman RM (2017) Reciprocal catecholamine changes during opiate exposure and withdrawal. Neuropsychopharmacology 42:671–681
Fragale JE, James MH, Aston-Jones G (2021) Intermittent self-administration of fentanyl induces a multifaceted addiction state associated with persistent changes in the orexin system. Addict Biol 26:e12946
Francesconi W, Szücs A, Berton F et al (2017) Opiate dependence induces cell type-specific plasticity of intrinsic membrane properties in the rat juxtacapsular bed nucleus of stria terminalis (jcBNST). Psychopharmacology 234:3485–3498
Franco-García A, Fernández-Gómez FJ, Gómez-Murcia V, Hidalgo JM, Milanés MV, Núñez C (2022) Molecular Mechanisms Underlying the Retrieval and Extinction of Morphine Withdrawal-Associated Memories in the Basolateral Amygdala and Dentate Gyrus. Biomedicines 10(3):588
Frank JW, Levy C, Matlock DD, Calcaterra SL, Mueller SR, Koester S, Binswanger IA (2016) Patients’ perspectives on tapering of chronic opioid therapy: a qualitative study. Pain Med 17(10):1838–1847
Fredriksson I, Applebey SV, Minier-Toribio A, Shekara A, Bossert JM, Shaham Y (2020) Effect of the dopamine stabilizer (-)-OSU6162 on potentiated incubation of opioid craving after electric barrier-induced voluntary abstinence. Neuropsychopharmacology 45:770–779
Frenois F, Cador M, Caillé S et al (2002) Neural correlates of the motivational and somatic components of naloxone-precipitated morphine withdrawal: brain areas underlying morphine withdrawal. Eur J Neurosci 16:1377–1389
Fuentealba JA, Forray MI, Gysling K (2000) Chronic morphine treatment and withdrawal increase extracellular levels of norepinephrine in the rat bed nucleus of the stria terminalis. J Neurochem 75:741–748
Fuertes G, Laorden ML, Milanés MV (2000) Noradrenergic and dopaminergic activity in the hypothalamic paraventricular nucleus after naloxone-induced morphine withdrawal. Neuroendocrinology 71:60–67
Fulenwider HD, Nennig SE, Hafeez H, Price ME, Baruffaldi F, Pravetoni M, Cheng K, Rice KC, Manvich DF, Schank JR (2020) Sex differences in oral oxycodone self‐administration and stress‐primed reinstatement in rats. Addict Biol 25(6)
Fulford AJ, Harbuz MS (2005) An introduction to the HPA axis. In Techniques in the behavioral and neural sciences (Vol. 15, pp. 43–65). Elsevier
Galligan JJ, Akbarali HI (2014) Molecular physiology of enteric opioid receptors. Am J Gastroenterol Suppl 2:17–21
García-Carmona J-A, Baroja-Mazo A, Milanés M-V, Laorden ML (2015a) Sex differences between CRF1 receptor deficient mice following naloxone-precipitated morphine withdrawal in a conditioned place aversion paradigm: implication of HPA axis. PLoS ONE 10:e0121125
García-Pérez D, Milanés MV (2020) Role of glucocorticoids on noradrenergic and dopaminergic neurotransmission within the basolateral amygdala and dentate gyrus during morphine withdrawal place aversion. Addict Biol 25:e12728
García-Pérez D, Laorden ML, Milanés MV, Núñez C (2012) Glucocorticoids regulation of FosB/ΔFosB expression induced by chronic opiate exposure in the brain stress system. PLoS ONE 7:e50264
García-Pérez D, Ferenczi S, Kovács KJ et al (2016) Different contribution of glucocorticoids in the basolateral amygdala to the formation and expression of opiate withdrawal-associated memories. Psychoneuroendocrinology 74:350–362
Gellert VF, Holtzman SG (1978) Development and maintenance of morphine tolerance and dependence in the rat by scheduled access to morphine drinking solutions. J Pharmacol Exp Ther 205:536–546
George, B. E., Dawes, M. H., Peck, E. G., & Jones, S. R. (2022). Altered Accumbal Dopamine Terminal Dynamics Following Chronic Heroin Self-Administration. International Journal of Molecular Sciences, 23(15), 8106.
Glass MJ, Hegarty DM, Oselkin M et al (2008) Conditional deletion of the NMDA-NR1 receptor subunit gene in the central nucleus of the amygdala inhibits naloxone-induced conditioned place aversion in morphine-dependent mice. Exp Neurol 213:57–70
Glynn LM, Davis EP, Sandman CA (2013) New insights into the role of perinatal HPA-axis dysregulation in postpartum depression. Neuropeptides 47:363–370
Gogolla N (2017) The insular cortex. Curr Biol 27:R580–R586
Gowing L, Farrell M, Ali R, White JM. Alpha2‐adrenergic agonists for the management of opioid withdrawal. Cochrane Database of Systematic Reviews 2016, Issue 5. Art. No.: CD002024. DOI: 10.1002/14651858.CD002024.pub5.
Gracy KN, Dankiewicz LA, Koob GF (2001) Opiate withdrawal-induced fos immunoreactivity in the rat extended amygdala parallels the development of conditioned place aversion. Neuropsychopharmacology 24:152–160
Greenwell TN, Walker BM, Cottone P et al (2009) The α1 adrenergic receptor antagonist prazosin reduces heroin self-administration in rats with extended access to heroin administration. Pharmacol Biochem Behav 91:295–302
Guo L-B, Yu C, Ling Q-L et al (2019) Proteomic analysis of male rat nucleus accumbens, dorsal hippocampus and amygdala on conditioned place aversion induced by morphine withdrawal. Behav Brain Res 372:112008
Hack SP, Vaughan CW, Christie MJ (2003) Modulation of GABA release during morphine withdrawal in midbrain neurons in vitro. Neuropharmacol 45:575–584
Hamlin AS, Buller KM, Day TA, Osborne PB (2004) Effect of naloxone-precipitated morphine withdrawal on c-fos expression in rat corticotropin-releasing hormone neurons in the paraventricular hypothalamus and extended amygdala. Neurosci Lett 362:39–43
Harocopos A, Allen B, Paone D (2016) Circumstances and contexts of heroin initiation following non-medical opioid analgesic use in New York City. Int J Drug Policy 28:106–112
Harris GC, Aston-Jones G (1994) Involvement of D2 dopamine receptors in the nucleus accumbens in the opiate withdrawal syndrome. Nature 371:155–157
Harris AC, Gewirtz JC (2004) Elevated startle during withdrawal from acute morphine: a model of opiate withdrawal and anxiety. Psychopharmacology 171:140–147
Harris AC, Atkinson DM, Aase DM, Gewirtz JC (2006) Double dissociation in the neural substrates of acute opiate dependence as measured by withdrawal-potentiated startle. Neuroscience 139:1201–1210
Hayward MD, Duman RS, Nestler EJ (1990) Induction of the c-fos proto-oncogene during opiate withdrawal in the locus coeruleus and other regions of rat brain. Brain Res 525:256–266
Hearing M, Graziane N, Dong Y, Thomas MJ (2018) Opioid and psychostimulant plasticity: targeting overlap in nucleus accumbens glutamate signaling. Trends Pharmacol Sci 39:276–294
Heinrichs SC, Menzaghi F, Schulteis G et al (1995) Suppression of corticotropin-releasing factor in the amygdala attenuates aversive consequences of morphine withdrawal. Behav Pharmacol 6:74–80
Heinz A, Daedelow LS, Wackerhagen C, Di Chiara G (2020) Addiction theory matters—why there is no dependence on caffeine or antidepressant medication. Addict Biol 25:e12735
Heiwe S, Lönnquist I, Källmén H (2011) Potential risk factors associated with risk for drop-out and relapse during and following withdrawal of opioid prescription medication. Eur J Pain 15:966–970
Hellemans KGC, Everitt BJ, Lee JLC (2006) Disrupting reconsolidation of conditioned withdrawal memories in the basolateral amygdala reduces suppression of heroin seeking in rats. J Neurosci 26:12694–12699
Hogarth L (2020) Addiction is driven by excessive goal-directed drug choice under negative affect: translational critique of habit and compulsion theory. Neuropsychopharmacol. https://doi.org/10.1038/s41386-020-0600-8
Holtz NA, Radke AK, Zlebnik NE et al (2015) Intracranial self-stimulation reward thresholds during morphine withdrawal in rats bred for high (HiS) and low (LoS) saccharin intake. Brain Res 1602:119–126
Hooshmand B, Azizi H, Ahmadi-Soleimani SM, Semnanian S (2019) Synergistic effect of orexin-glutamate co-administration on spontaneous discharge rate of locus coeruleus neurons in morphine-dependent rats. Neurosci Lett 706:12–17
Hosoya Y, Sugiura Y, Okado N et al (1991) Descending input from the hypothalamic paraventricular nucleus to sympathetic preganglionic neurons in the rat. Exp Brain Res 85:10–20
Hou Y-Y, Cai Y-Q, Pan ZZ (2015) Persistent pain maintains morphine-seeking behavior after morphine withdrawal through reduced MeCP2 repression of GluA1 in rat central amygdala. J Neurosci 35:3689–3700
Houshyar H, Gomez F, Manalo S et al (2003) Intermittent morphine administration induces dependence and is a chronic stressor in rats. Neuropsychopharmacol 28:1960–1972
Hutcheson DM, Everitt BJ, Robbins TW, Dickinson A (2001) The role of withdrawal in heroin addiction: enhances reward or promotes avoidance? Nat Neurosci 4:943–947
Ingallinesi M, Rouibi K, Le Moine C et al (2012) CRF2 receptor-deficiency eliminates opiate withdrawal distress without impairing stress coping. Mol Psychiatry 17:1283–1294
Ishida S, Shimosaka R, Kawasaki Y et al (2008) Involvement of the amygdala on place aversion induced by naloxone in single-dose morphine-treated rats. Yakugaku Zasshi 128:395–403
Jafarova Demirkapu M, Yananlı HR, Kaleli M et al (2020) The role of adenosine A1 receptors in the nucleus accumbens during morphine withdrawal. Clin Exp Pharmacol Physiol 47:553–560
Jiang C, Yang X, He G, Wang F, Wang Z, Xu W, Mao Y, Ma L, Wang F (2021) CRHCEA→VTA inputs inhibit the positive ensembles to induce negative effect of opiate withdrawal. Mol Psychiatry 26(11):6170–6186
Jones RT (1980) Dependence in non-addict humans after a single dose of morphine. In: Way EL (ed) Endogenous and exogenous opiate agonists and antagonists. Pergamon, pp 557–560
Joseph H, Stancliff S, Langrod J (2000) Methadone maintenance treatment (MMT): a review of historical and clinical issues. Mt Sinai J Med 67:347–364
Kang J, Compton DR, Vaz RJ, Rampe D (2016) Proarrhythmic mechanisms of the common anti-diarrheal medication loperamide: revelations from the opioid abuse epidemic. Naunyn Schmiedebergs Arch Pharmacol 389:1133–1137
Kaufling J, Aston-Jones G (2015) Persistent adaptations in afferents to ventral tegmental dopamine neurons after opiate withdrawal. J Neurosci 35:10290–10303
Keiflin R, Janak PH (2015) Dopamine prediction errors in reward learning and addiction: from theory to neural circuitry. Neuron 88:247–263
Kelsey JE, Verhaak AMS, Schierberl KC (2015) The kappa-opioid receptor antagonist, nor-binaltorphimine (nor-BNI), decreases morphine withdrawal and the consequent conditioned place aversion in rats. Behav Brain Res 283:16–21
Kenny PJ, Chen SA, Kitamura O et al (2006) Conditioned withdrawal drives heroin consumption and decreases reward sensitivity. J Neurosci 26:5894–5900
Kest B, Palmese CA, Hopkins E et al (2001) Assessment of acute and chronic morphine dependence in male and female mice. Pharmacol Biochem Behav 70:149–156
Kienbaum P, Thürauf N, Michel MC et al (1998) Profound increase in epinephrine concentration in plasma and cardiovascular stimulation after mu-opioid receptor blockade in opioid-addicted patients during barbiturate-induced anesthesia for acute detoxification. Anesthesiology 88:1154–1161
Kim J, Zhang X, Muralidhar S et al (2017) Basolateral to central amygdala neural circuits for appetitive behaviors. Neuron 93:1464-1479.e5
Kiyatkin EA (1995) Functional significance of mesolimbic dopamine. Neurosci Biobehav Rev 19:573–598
Knoll AT, Muschamp JW, Sillivan SE et al (2011) Kappa opioid receptor signaling in the basolateral amygdala regulates conditioned fear and anxiety in rats. Biol Psychiatry 70:425–433
Koob GF (2013) Negative reinforcement in drug addiction: the darkness within. Curr Opin Neurobiol 23:559–563
Koob GF (2020) Neurobiology of opioid addiction: opponent process, hyperkatifeia, and negative reinforcement. Biol Psychiatry 87:44–53
Koob GF, Volkow ND (2010) Neurocircuitry of addiction. Neuropsychopharmacology 35:217–238
Koob GF, Stinus L, Le Moal M, Bloom FE (1989) Opponent process theory of motivation: neurobiological evidence from studies of opiate dependence. Neurosci Biobehav Rev 13:135–140
Koob GF, Maldonado R, Stinus L (1992) Neural substrates of opiate withdrawal. Trends Neurosci 15:186–191
Kosten TR, Baxter LE (2019) Review article: effective management of opioid withdrawal symptoms: a gateway to opioid dependence treatment. Am J Addict 28:55–62
Laorden ML, Ferenczi S, Pintér-Kübler B et al (2012) Hypothalamic orexin—a neurons are involved in the response of the brain stress system to morphine withdrawal. PLoS ONE 7:e36871
Lebow MA, Chen A (2016) Overshadowed by the amygdala: the bed nucleus of the stria terminalis emerges as key to psychiatric disorders. Mol Psychiatry 21:450–463
Lefevre EM, Pisansky MT, Toddes C et al (2020) Interruption of continuous opioid exposure exacerbates drug-evoked adaptations in the mesolimbic dopamine system. Neuropsychopharmacology 45:1781–1792
Leri F, Tremblay A, Sorge RE, Stewart J (2004) Methadone maintenance reduces heroin- and cocaine-induced relapse without affecting stress-induced relapse in a rodent model of poly-drug use. Neuropsychopharmacol 29:1312–1320
Li C-L, Zhu N, Meng X-L et al (2013) Effects of inactivating the agranular or granular insular cortex on the acquisition of the morphine-induced conditioned place preference and naloxone-precipitated conditioned place aversion in rats. J Psychopharmacol 27:837–844
Lichtenberg NT, Wassum KM (2017) Amygdala mu-opioid receptors mediate the motivating influence of cue-triggered reward expectations. Eur J Neurosci 45:381–387
Yu G, Zhang F-Q, Tang S-E et al (2014) Continuous infusion versus intermittent bolus dosing of morphine: a comparison of analgesia, tolerance, and subsequent voluntary morphine intake. J Psychiatr Res 59:161–166
Liu C, Cai X, Ritzau-Jost A, Kramer PF, Li Y, Khaliq ZM, ... & Kaeser PS (2022) An action potential initiation mechanism in distal axons for the control of dopamine release. Science 375(6587):1378–1385
Longnecker DE, Grazis PA, Eggers GW Jr (1973) Naloxone for antagonism of morphine-induced respiratory depression. Anesth Analg 52:447–453
Loyd DR, Murphy AZ (2009) The role of the periaqueductal gray in the modulation of pain in males and females: are the anatomy and physiology really that different? Neural Plast 2009:462879
Lucas M, Frenois F, Vouillac C et al (2008) Reactivity and plasticity in the amygdala nuclei during opiate withdrawal conditioning: differential expression of c-fos and arc immediate early genes. Neuroscience 154:1021–1033
Luster BR, Cogan ES, Schmidt KT et al (2020) Inhibitory transmission in the bed nucleus of the stria terminalis in male and female mice following morphine withdrawal. Addict Biol 25:e12748
Lynch WJ, Carroll ME (1999) Sex differences in the acquisition of intravenously self-administered cocaine and heroin in rats. Psychopharmacology 144:77–82
Lyons D, de Jaeger X, Rosen LG et al (2013) Opiate exposure and withdrawal induces a molecular memory switch in the basolateral amygdala between ERK1/2 and CaMKIIα-dependent signaling substrates. J Neurosci 33:14693–14704
Magura S, Rosenblum A (2001) Leaving methadone treatment: lessons learned, lessons forgotten, lessons ignored. The Mount Sinai J Med, New York 68(1):62–74
Maldonado R, Koob GF (1993) Destruction of the locus coeruleus decreases physical signs of opiate withdrawal. Brain Res 605(1):128–138
Maldonado R, Negus S, Koob GF (1992a) Precipitation of morphine withdrawal syndrome in rats by administration of mu-, delta- and kappa-selective opioid antagonists. Neuropharmacology 31:1231–1241
Maldonado R, Stinus L, Gold LH, Koob GF (1992b) Role of different brain structures in the expression of the physical morphine withdrawal syndrome. J Pharmacol Exp Ther 261:669–677
Maldonado R, Valverde O, Garbay C, Roques BP (1995) Protein kinases in the locus coeruleus and periaqueductal gray matter are involved in the expression of opiate withdrawal. Naunyn Schmiedebergs Arch Pharmacol 352:565–575
Mamaligas AA, Cai Y, Ford CP (2016) Nicotinic and opioid receptor regulation of striatal dopamine D2-receptor mediated transmission. Sci Rep 6:37834
Mantsch JR, Baker DA, Funk D et al (2016) Stress-induced reinstatement of drug seeking: 20 years of progress. Neuropsychopharmacology 41:335–356
Maren S (2003) The amygdala, synaptic plasticity, and fear memory. Ann N Y Acad Sci 985:106–113
Martinez‐Casiano K, Ramos‐Ortolaza D, Chamorro J, Torres‐Reveron A (2015) Development of conditioned place aversion to spontaneous morphine withdrawal during estradiol replacement. FASEB J 29:LB639
Martínez-Laorden E, Navarro-Zaragoza J, Milanés M-V et al (2020) Conditioned aversive memory associated with morphine withdrawal increases brain-derived neurotrophic factor in dentate gyrus and basolateral amygdala. Addict Biol 25:e12792
Mavrikaki M, Pravetoni M, Page S et al (2017) Oxycodone self-administration in male and female rats. Psychopharmacology 234:977–987
McDevitt DS, McKendrick G, Graziane NM (2021) Anterior cingulate cortex is necessary for spontaneous opioid withdrawal and withdrawal-induced hyperalgesia in male mice. Neuropsychopharmacology 46:1990–1999
McDonald AJ (1998) Cortical pathways to the mammalian amygdala. Prog Neurobiol 55:257–332
McNally GP, Akil H (2002) Role of corticotropin-releasing hormone in the amygdala and bed nucleus of the stria terminalis in the behavioral, pain modulatory, and endocrine consequences of opiate withdrawal. Neuroscience 112:605–617
Milanés MV, Laorden ML, Angel E et al (2002) Effect of naloxone-precipitated morphine withdrawal on CRH and vasopressin mRNA expression in the rat hypothalamic paraventricular nucleus. Neurosci Lett 334:58–62
Mitchell SG, Kelly SM, Brown BS et al (2009) Incarceration and opioid withdrawal: the experiences of methadone patients and out-of-treatment heroin users. J Psychoactive Drugs 41:145–152
Monroe SC, Radke AK (2021) Aversion-resistant fentanyl self-administration in mice. Psychopharmacology 238:699–710
Mucha RF (1987) Is the motivational effect of opiate withdrawal reflected by common somatic indices of precipitated withdrawal? A place conditioning study in the rat. Brain Res 418:214–220
Nakagawa T, Yamamoto R, Fujio M et al (2005) Involvement of the bed nucleus of the stria terminalis activated by the central nucleus of the amygdala in the negative affective component of morphine withdrawal in rats. Neuroscience 134:9–19
Naqvi NH, Rudrauf D, Damasio H, Bechara A (2007) Damage to the insula disrupts addiction to cigarette smoking. Science 315:531–534
Nava F, Caldiroli E, Premi S, Lucchini A (2006) Relationship between plasma cortisol levels, withdrawal symptoms and craving in abstinent and treated heroin addicts. J Addict Dis 25:9–16
Navarro-Zaragoza J, Martínez-Laorden E, Teruel-Fernández FJ et al (2021) Naloxone-induced conditioned place aversion score and extinction period are higher in C57BL/6J morphine-dependent mice than in Swiss: Role of HPA axis. Pharmacol Biochem Behav 201:173106
Negus SS (2006) Choice between heroin and food in nondependent and heroin-dependent rhesus monkeys: effects of naloxone, buprenorphine, and methadone. J Pharmacol Exp Ther 317(2):711–723
Negus SS, Rice KC (2009) Mechanisms of withdrawal-associated increases in heroin self-administration: pharmacologic modulation of heroin vs food choice in heroin-dependent rhesus monkeys. Neuropsychopharmacology 34:899–911
O’Neill P-K, Gore F, Salzman CD (2018) Basolateral amygdala circuitry in positive and negative valence. Curr Opin Neurobiol 49:175–183
Olds J, Milner P (1954) Positive reinforcement produced by electrical stimulation of septal area and other regions of rat brain. J Comp Physiol Psychol 47(6):419
Pantazis CB, Gonzalez LA, Tunstall BJ, Carmack SA, Koob GF, Vendruscolo LF (2021) Cues conditioned to withdrawal and negative reinforcement: Neglected but key motivational elements driving opioid addiction. Sci Adv 7(15):eabf0364
Park PE, Schlosburg JE, Vendruscolo LF et al (2015) Chronic CRF1 receptor blockade reduces heroin intake escalation and dependence-induced hyperalgesia. Addict Biol 20:275–284
Parker LA, Joshi A (1998) Naloxone-precipitated morphine withdrawal induced place aversions: effect of naloxone at 24 hours postmorphine. Pharmacol Biochem Behav 61:331–333
Pedrón VT, Varani AP, Balerio GN (2016) Baclofen prevents the elevated plus maze behavior and BDNF expression during naloxone precipitated morphine withdrawal in male and female mice. Synapse 70:187–197
Pennington ZT, Trott JM, Rajbhandari AK et al (2020) Chronic opioid pretreatment potentiates the sensitization of fear learning by trauma. Neuropsychopharmacology 45:482–490
Pergolizzi JV Jr, Raffa RB, Rosenblatt MH (2020) Opioid withdrawal symptoms, a consequence of chronic opioid use and opioid use disorder: current understanding and approaches to management. J Clin Pharm Ther 45:892–903
Phillips AG, Fibiger HC (1980) The role of dopamine in maintaining intracranial self-stimulation in the ventral tegmentum, nucleus accumbens, and medial prefrontal cortex. Canadian J Psychology/revue Canadienne De Psychol 32:58
Piantadosi PT, Halladay LR, Radke AK, Holmes A (2021) Advances in understanding meso-cortico-limbic-striatal systems mediating risky reward seeking. J Neurochem 157:1547–1571
Piper ME (2015) Withdrawal: expanding a key addiction construct. Nicotine Tob Res 17:1405–1415
Ponterio G, Tassone A, Sciamanna G et al (2013) Powerful inhibitory action of mu opioid receptors (MOR) on cholinergic interneuron excitability in the dorsal striatum. Neuropharmacology 75:78–85
Pothos E, Rada P, Mark GP, Hoebel BG (1991) Dopamine microdialysis in the nucleus accumbens during acute and chronic morphine, naloxone-precipitated withdrawal and clonidine treatment. Brain Res 566:348–350
Punch LJ, Self DW, Nestler EJ, Taylor JR (1997) Opposite modulation of opiate withdrawal behaviors on microinfusion of a protein kinase A inhibitor versus activator into the locus coeruleus or periaqueductal gray. J Neurosci 17:8520–8527
Rada P, Mark GP, Pothos E, Hoebel BG (1991) Systemic morphine simultaneously decreases extracellular acetylcholine and increases dopamine in the nucleus accumbens of freely moving rats. Neuropharmacology 30:1133–1136
Rada PV, Mark GP, Taylor KM, Hoebel BG (1996) Morphine and naloxone, IP or locally, affect extracellular acetylcholine in the accumbens and prefrontal cortex. Pharmacol Biochem Behav 53:809–816
Radke AK, Gewirtz JC (2012) Increased dopamine receptor activity in the nucleus accumbens shell ameliorates anxiety during drug withdrawal. Neuropsychopharmacology 37:2405–2415
Radke AK, Rothwell PE, Gewirtz JC (2011) An anatomical basis for opponent process mechanisms of opiate withdrawal. J Neurosci 31:7533–7539
Radke AK, Holtz NA, Gewirtz JC, Carroll ME (2013) Reduced emotional signs of opiate withdrawal in rats selectively bred for low (LoS) versus high (HiS) saccharin intake. Psychopharmacology 227:117–126
Radke AK, Gewirtz JC, Carroll ME (2015) Effects of age, but not sex, on elevated startle during withdrawal from acute morphine in adolescent and adult rats. Behav Pharmacol 26:485–488
Radke AK, Sneddon EA, Monroe SC (2021) Studying sex differences in rodent models of addictive behavior. Curr Protoc 1:e119
Rasmussen K, Aghajanian GK (1989) Withdrawal-induced activation of locus coeruleus neurons in opiate-dependent rats: attenuation by lesions of the nucleus paragigantocellularis. Brain Res 505:346–350
Rasmussen K, Beitner-Johnson DB, Krystal JH, Aghajanian GK, Nestler EJ (1990) Opiate withdrawal and the rat locus coeruleus: behavioral, electrophysiological, and biochemical correlates. J Neurosci 10(7):2308–2317
Rasmussen K, Kendrick WT, Kogan JH, Aghajanian GK (1996) A selective AMPA antagonist, LY293558, suppresses morphine withdrawal-induced activation of locus coeruleus neurons and behavioral signs of morphine withdrawal. Neuropsychopharmacology 15:497–505
Redish AD (2004) Addiction as a computational process gone awry. Science 306:1944–1947
Riahi E, Mirzaii-Dizgah I, Karimian SM et al (2009) Attenuation of morphine withdrawal signs by a GABAB receptor agonist in the locus coeruleus of rats. Behav Brain Res 196:11–14
Robinson TE, Berridge KC (2001) Incentive-sensitization and addiction. Addiction 96:103–114
Rothwell PE, Gewirtz JC, Thomas MJ (2010) Episodic withdrawal promotes psychomotor sensitization to morphine. Neuropsychopharmacology 35:2579–2589
Rothwell PE, Thomas MJ, Gewirtz JC (2012) Protracted manifestations of acute dependence after a single morphine exposure. Psychopharmacology 219:991–998
Russell SE, Puttick DJ, Sawyer AM et al (2016) Nucleus accumbens AMPA receptors are necessary for morphine-withdrawal-induced negative-affective states in rats. J Neurosci 36:5748–5762
Schulteis G, Heyser CJ, Koob GF (1997) Opiate withdrawal signs precipitated by naloxone following a single exposure to morphine: potentiation with a second morphine exposure. Psychopharmacology 129:56–65
Schulteis G, Ahmed SH, Morse AC et al (2000) Conditioning and opiate withdrawal. Nature 405:1013–1014
Zhang G-F, Ren Y-P, Sheng L-X et al (2008) Dysfunction of the hypothalamic–pituitary–adrenal axis in opioid dependent subjects: effects of acute and protracted abstinence. Am J Drug Alcohol Abuse 34:760–768
Schultz W, Apicella P, Ljungberg T (1993) Responses of monkey dopamine neurons to reward and conditioned stimuli during successive steps of learning a delayed response task. J Neurosci 13:900–913
Seno FZ, Sgobbi RF, Nobre MJ (2022) Contributions of the GABAergic system of the prelimbic cortex and basolateral amygdala to morphine withdrawal-induced contextual fear. Physiol Behav 254
Shaham Y, Stewart J (1995) Stress reinstates heroin-seeking in drug-free animals: an effect mimicking heroin, not withdrawal. Psychopharmacology 119:334–341
Shaham Y, Rajabi H, Stewart J (1996) Relapse to heroin-seeking in rats under opioid maintenance: the effects of stress, heroin priming, and withdrawal. J Neurosci 16:1957–1963
Sharma SK, Klee WA, Nirenberg M (1975) Dual regulation of adenylate cyclase accounts for narcotic dependence and tolerance. Proc Natl Acad Sci U S A 72:3092–3096
Shaw-Lutchman TZ, Barrot M, Wallace T et al (2002) Regional and cellular mapping of cAMP response element-mediated transcription during naltrexone-precipitated morphine withdrawal. J Neurosci 22:3663–3672
Sinha R (2009) Modeling stress and drug craving in the laboratory: implications for addiction treatment development. Addict Biol 14:84–98
Sinha R, Shaham Y, Heilig M (2011) Translational and reverse translational research on the role of stress in drug craving and relapse. Psychopharmacology 218:69–82
Skelton KH, Oren D, Gutman DA et al (2007) The CRF1 receptor antagonist, R121919, attenuates the severity of precipitated morphine withdrawal. Eur J Pharmacol 571:17–24
Smethells JR, Greer A, Dougen B, Carroll ME (2020) Effects of voluntary exercise and sex on multiply-triggered heroin reinstatement in male and female rats. Psychopharmacology 237:453–463
Solecki WB, Kus N, Gralec K et al (2019) Noradrenergic and corticosteroid receptors regulate somatic and motivational symptoms of morphine withdrawal. Behav Brain Res 360:146–157
Solomon RL, Corbit JD (1974) An opponent-process theory of motivation. I. Temporal Dynamics of Affect Psychol Rev 81:119–145
Song Y, Meng Q-X, Wu K et al (2020) Disinhibition of PVN-projecting GABAergic neurons in AV region in BNST participates in visceral hypersensitivity in rats. Psychoneuroendocrinology 117:104690
Song J, Shao D, Guo X, Zhao Y, Cui D, Ma Q, ... & Zheng P (2019) Crucial role of feedback signals from prelimbic cortex to basolateral amygdala in the retrieval of morphine withdrawal memory. Sci Adv 5(2):eaat3210
Stein C (2013) Opioid receptors on peripheral sensory neurons. Madame Curie Bioscience Database Available from: https://www.ncbi.nlm.nih.gov/books/NBK6242/.
Stimmel B, Kreek MJ (2000) Neurobiology of addictive behaviors and its relationship to methadone maintenance. Mt Sinai J Med 67:375–380
Stinus L, Le Moal M, Koob GF (1990) Nucleus accumbens and amygdala are possible substrates for the aversive stimulus effects of opiate withdrawal. Neuroscience 37:767–773
Stinus L, Cador M, Zorrilla EP, Koob GF (2005) Buprenorphine and a CRF1 antagonist block the acquisition of opiate withdrawal-induced conditioned place aversion in rats. Neuropsychopharmacology 30:90–98
Stornetta RL, Norton FE, Guyenet PG (1993) Autonomic areas of rat brain exhibit increased Fos-like immunoreactivity during opiate withdrawal in rats. Brain Res 624:19–28
Stumbo SP, Yarborough BJH, McCarty D, Weisner C, Green CA (2017) Patient-reported pathways to opioid use disorders and pain-related barriers to treatment engagement. J Subst Abuse Treat 73:47–54
Summers PJ, Hellman JL, MacLean MR et al (2018) Negative experiences of pain and withdrawal create barriers to abscess care for people who inject heroin. A mixed methods analysis. Drug Alcohol Depend 190:200–208
Swain Y, Muelken P, Skansberg A et al (2020) Higher anhedonia during withdrawal from initial opioid exposure is protective against subsequent opioid self-administration in rats. Psychopharmacology 237:2279–2291
Taylor JR, Punch LJ, Elsworth JD (1998) A comparison of the effects of clonidine and CNQX infusion into the locus coeruleus and the amygdala on naloxone-precipitated opiate withdrawal in the rat. Psychopharmacology 138:133–142
Tennant F, Shannon JA, Nork JG et al (1991) Abnormal adrenal gland metabolism in opioid addicts: implications for clinical treatment. J Psychoactive Drugs 23:135–149
van Vulpen EH, Verwer RW (1989) Organization of projections from the mediodorsal nucleus of the thalamus to the basolateral complex of the amygdala in the rat. Brain Res 500:389–394
Vanderschuren LJ, Tjon GH, Nestby P et al (1997) Morphine-induced long-term sensitization to the locomotor effects of morphine and amphetamine depends on the temporal pattern of the pretreatment regimen. Psychopharmacology 131:115–122
Zhang L, Dong Y, Doyon WM, Dani JA (2012) Withdrawal from chronic nicotine exposure alters dopamine signaling dynamics in the nucleus accumbens. Biol Psychiatry 71:184–191
Zhu Y, Wienecke CFR, Nachtrab G, Chen X (2016) A thalamic input to the nucleus accumbens mediates opiate dependence. Nature 530:219–222
Vargas-Perez H, Ting-A-Kee R, van der Kooy D (2009) Different neural systems mediate morphine reward and its spontaneous withdrawal aversion. Eur J Neurosci 29:2029–2034
Veinante P, Stoeckel M-E, Lasbennes F, Freund-Mercier M-J (2003) c-Fos and peptide immunoreactivities in the central extended amygdala of morphine-dependent rats after naloxone-precipitated withdrawal. Eur J Neurosci 18:1295–1305
Venniro M, Zhang M, Shaham Y, Caprioli D (2017) Incubation of methamphetamine but not heroin craving after voluntary abstinence in male and female rats. Neuropsychopharmacology 42(5):1126–1135
Venniro M, Russell TI, Zhang M, Shaham Y (2019) Operant social reward decreases incubation of heroin craving in male and female rats. Biol Psychiatry 86(11):848–856
Vertes RP, Linley SB, Hoover WB (2015) Limbic circuitry of the midline thalamus. Neurosci Biobehav Rev 54:89–107
Vos T et al (2017) Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet 390(10100):1211–1259
Walters CL, Aston-Jones G, Druhan JP (2000) Expression of fos-related antigens in the nucleus accumbens during opiate withdrawal and their attenuation by a D2 dopamine receptor agonist. Neuropsychopharmacology 23:307–315
Wanat M, Willuhn I, Clark J, Phillips P (2009) Phasic dopamine release in appetitive behaviors and drug addiction. Current Drug Abuse Reviewse 2:195–213
Wang L, Shen M, Jiang C, Ma L, Wang F (2016) Parvalbumin interneurons of central amygdala regulate the negative affective states and the expression of corticotrophin-releasing hormone during morphine withdrawal. Int J Neuropsychopharmacol 19(11)
Warlow SM, Berridge KC (2021) Incentive motivation:‘wanting’roles of central amygdala circuitry. Behav Brain Res 411:113376
Wassum KM, Izquierdo A (2015) The basolateral amygdala in reward learning and addiction. Neurosci Biobehav Rev 57:271–283
Wassum KM, Greenfield VY, Linker KE et al (2016) Inflated reward value in early opiate withdrawal. Addict Biol 21:221–233
Watanabe T, Nakagawa T, Yamamoto R et al (2002a) Involvement of glutamate receptors within the central nucleus of the amygdala in naloxone-precipitated morphine withdrawal-induced conditioned place aversion in rats. Jpn J Pharmacol 88:399–406
Watanabe T, Yamamoto R, Maeda A et al (2002b) Effects of excitotoxic lesions of the central or basolateral nucleus of the amygdala on naloxone-precipitated withdrawal-induced conditioned place aversion in morphine-dependent rats. Brain Res 958:423–428
Watson S, Mackin P (2006) HPA axis function in mood disorders. Psychiatry 5:166–170
Weiss RD, Potter JS, Griffin ML et al (2014) Reasons for opioid use among patients with dependence on prescription opioids: the role of chronic pain. J Subst Abuse Treat 47:140–145
Weiss F, Ciccocioppo R, Parsons LH, Katner S, Liu X, Zorrilla EP, Valdez GR, Ben-Shahar O, Angeletti S, Richter RR. (2001) Compulsive drug‐seeking behavior and relapse. Neuroadaptation, stress, and conditioning factors. Ann N Y Acad Sci 937(1):1–26
Weller KL, Smith DA (1982) Afferent connections to the bed nucleus of the stria terminalis. Brain Res 232:255–270
Wikler A (1948) Recent progress in research on the neurophysiologic basis of morphine addiction. Am J Psychiatry 105:329–338
Williams JT, Christie MJ, Manzoni O (2001) Cellular and synaptic adaptations mediating opioid dependence. Physiol Rev 81:299–343
Williams AM, Reis DJ, Powell AS et al (2012) The effect of intermittent alcohol vapor or pulsatile heroin on somatic and negative affective indices during spontaneous withdrawal in Wistar rats. Psychopharmacology 223:75–88
Wills KL, Petrie GN, Millett G et al (2016) Double dissociation of monoacylglycerol lipase inhibition and CB1 antagonism in the central amygdala, basolateral amygdala, and the interoceptive insular cortex on the affective properties of acute naloxone-precipitated morphine withdrawal in rats. Neuropsychopharmacology 41:1865–1873
Wills KL, DeVuono MV, Limebeer CL et al (2017) CB1 receptor antagonism in the bed nucleus of the stria terminalis interferes with affective opioid withdrawal in rats. Behav Neurosci 131:304–311
Wise RA (2004) Dopamine, learning and motivation. Nat Rev Neurosci 5:483–494
Witkin JM, Tzavara ET, Nomikos GG (2005) A role for cannabinoid CB1 receptors in mood and anxiety disorders. Behav Pharmacol 16:315–331
Xu W, Li YH, Tan BP et al (2012) Inhibition of the acquisition of conditioned place aversion by dopaminergic lesions of the central nucleus of the amygdala in morphine-treated rats. Physiol Res 61:437–442
Funding
This work was supported by the NIH grants R15 AA027915 (AKR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Monroe, S.C., Radke, A.K. Opioid withdrawal: role in addiction and neural mechanisms. Psychopharmacology 240, 1417–1433 (2023). https://doi.org/10.1007/s00213-023-06370-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-023-06370-2