Abstract
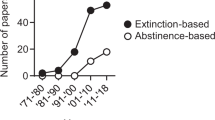
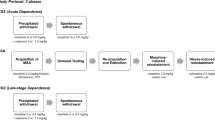
Opioid abuse in humans is characterized by discontinuous periods of drug use and abstinence. With time, the probability of falling into renewed drug consumption becomes particularly high and constitutes a considerable problem in the management of heroin addicts. The major problem in the treatment of opioid dependence still remains the occurrence of relapse, to which stressful life events, renewed use of heroin, and exposure to drug-associated environmental cues are all positively correlated. To study the neurobiology of relapse, many research groups currently use the reinstatement animal model, which greatly contributed to disentangle the mechanisms underlying relapse to drug-seeking in laboratory animals. The use of this model is becoming increasingly popular worldwide, and new versions have been recently developed to better appreciate the differential contribution of each opioid receptor subtype to the relapse phenomenon. In this chapter we review the state of the art of our knowledge on the specific role of the opioid receptors as unrevealed by the reinstatement animal model of opioid-seeking behavior.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
United Nation Office on Drugs and Crime (UNODC) (2013) World drug report. p. 18
Epstein DH, Preston KL, Stewart J et al (2006) Toward a model of drug relapse: an assessment of the validity of the reinstatement procedure. Psychopharmacology 189:1–16
Tzschentke TM (2007) Measuring reward with the conditioned place preference (CPP) paradigm: update of the last decade. Addict Biol 12:227–462
Arnold JM, Roberts DC (1997) A critique of fixed and progressive ratio schedules used to examine the neural substrates of drug reinforcement. Pharmacol Biochem Behav 57:441–447
Shalev U, Grimm JW, Shaham Y (2002) Neurobiology of relapse to heroin and cocaine seeking: a review. Pharmacol Rev 54:1–42
Mello NK, Mendelson JH, Bree MP (1981) Naltrexone effects on morphine and food self-administration in morphine-dependent rhesus monkeys. J Pharmacol Exp Ther 218:550–557
Mello NK, Bree MP, Mendelson JH (1983) Comparison of buprenorphine and methadone effects on opiate self-administration in primates. J Pharmacol Exp Ther 225:378–386
Matthes HW, Maldonado R, Simonin F et al (1996) Loss of morphine-induced analgesia, reward effect and withdrawal symptoms in mice lacking the mu-opioid-receptor gene. Nature 383:819–823
Liang J, Li Y, Ping X et al (2006) The possible involvement of endogenous ligands for mu-, delta- and kappa-opioid receptors in modulating morphine-induced CPP expression in rats. Peptides 27:3307–3314
Le Merrer J, Becker JA, Befort K et al (2009) Reward processing by the opioid system in the brain. Physiol Rev 89:1379–1412
Lutz PE, Kieffer BL (2013) The multiple facets of opioid receptor function: implications for addiction. Curr Opin Neurobiol 23:473–479
De Wit H, Stewart J (1983) Drug reinstatement of heroin-reinforced responding in the rat. Psychopharmacology 79:29–31
Stewart J (1983) Conditioned and unconditioned drug effects in relapse to opiate and stimulant drug self-administration. Prog Neuropsychopharmacol Biol Psychiatry 7:591–597
Fattore L, Spano MS, Cossu G et al (2003) Cannabinoid mechanism in reinstatement of heroin-seeking after a long period of abstinence in rats. Eur J Neurosci 17:1723–1726
Fattore L, Spano S, Cossu G et al (2005) Cannabinoid CB(1) antagonist SR 141716A attenuates reinstatement of heroin self-administration in heroin-abstinent rats. Neuropharmacology 48:1097–1104
Spano MS, Fattore L, Fratta W et al (2007) The GABAB receptor agonist baclofen prevents heroin-induced reinstatement of heroin-seeking behavior in rats. Neuropharmacology 52:1555–1562
Parker LA, Mcdonald RV (2000) Reinstatement of both a conditioned place preference and a conditioned place aversion with drug primes. Pharmacol Biochem Behav 66:559–561
Ribeiro Do Couto B, Aguilar MA et al (2003) Reinstatement of morphine-induced conditioned place preference in mice by priming injections. Neural Plast 10:279–290
Mueller D, Perdikaris D, Stewart J (2002) Persistence and drug-induced reinstatement of a morphine-induced conditioned place preference. Behav Brain Res 136:389–397
Ribeiro Do Couto B, Aguilar MA et al (2005) Long-lasting rewarding effects of morphine induced by drug primings. Brain Res 1050:53–63
Stewart J, Wise RA (1992) Reinstatement of heroin self-administration habits: morphine prompts and naltrexone discourages renewed responding after extinction. Psychopharmacology 108:79–84
Stewart J (1984) Reinstatement of heroin and cocaine self-administration behavior in the rat by intracerebral application of morphine in the ventral tegmental area. Pharmacol Biochem Behav 20:917–923
Shaham Y, Stewart J (1996) Effects of opioid and dopamine receptor antagonists on relapse induced by stress and re-exposure to heroin in rats. Psychopharmacology 125:385–391
Shaham Y, Rajabi H, Stewart J (1996) Relapse to heroin-seeking in rats under opioid maintenance: the effects of stress, heroin priming, and withdrawal. J Neurosci 16:1957–1963
Leri F, Rizos Z (2005) Reconditioning of drug-related cues: a potential contributor to relapse after drug reexposure. Pharmacol Biochem Behav 80:621–630
Liu H, Lai M, Zhou X et al (2012) Galantamine attenuates the heroin seeking behaviors induced by cues after prolonged withdrawal in rats. Neuropharmacology 62:2515–2521
Zhang F, Zhou W, Liu H et al (2005) Increased c-Fos expression in the medial part of the lateral habenula during cue-evoked heroin-seeking in rats. Neurosci Lett 386:133–137
Berger AC, Whistler JL (2011) Morphine-induced mu opioid receptor trafficking enhances reward yet prevents compulsive drug use. EMBO Mol Med 3:385–397
Finn AK, Whistler JL (2001) Endocytosis of the mu opioid receptor reduces tolerance and a cellular hallmark of opiate withdrawal. Neuron 32:829–839
Nathan PJ, Bush MA, Tao WX et al (2012) Multiple-dose safety, pharmacokinetics, and pharmacodynamics of the μ-opioid receptor inverse agonist GSK1521498. J Clin Pharmacol 52:1456–1467
Cambridge VC, Ziauddeen H, Nathan PJ et al (2013) Neural and behavioral effects of a novel mu opioid receptor antagonist in binge-eating obese people. Biol Psychiatry 73:887–894
Giuliano C, Robbins TW, Wille DR et al (2013) Attenuation of cocaine and heroin seeking by μ-opioid receptor antagonism. Psychopharmacology 227:137–147
Cummins E, Boughner E, Leri F (2014) Cue-induced renewal of heroin place preference: involvement of the basolateral amygdala. Neuroreport 25:297–302
Lu L, Chen H, Su W et al (2005) Role of withdrawal in reinstatement of morphine-conditioned place preference. Psychopharmaocology 181:90–100
Pfeiffer A, Brantl V, Herz A et al (1986) Psychotomimesis mediated by kappa opiate receptors. Science 233:774–776
Carey AN, Borozny K, Aldrich JV et al (2007) Reinstatement of cocaine place-conditioning prevented by the peptide kappa-opioid receptor antagonist arodyn. Eur J Pharmacol 569:84–89
Redila VA, Chavkin C (2008) Stress-induced reinstatement of cocaine seeking is mediated by the kappa opioid system. Psychopharmacology 200:59–70
Land BB, Bruchas MR, Schattauer S et al (2009) Activation of the kappa opioid receptor in the dorsal raphe nucleus mediates the aversive effects of stress and reinstates drug seeking. Proc Natl Acad Sci U S A 106:19168–19173
Zhou Y, Leri F, Grella SL et al (2013) Involvement of dynorphin and kappa opioid receptor in yohimbine-induced reinstatement of heroin seeking in rats. Synapse 67:358–361
Bruchas MR, Land BB, Chavkin C (2010) The dynorphin/kappa opioid system as a modulator of stress-induced and pro-addictive behaviors. Brain Res 1314:44–55
Cordery SF, Taverner A, Ridzwan IE et al (2012) A non-rewarding, non-aversive buprenorphine/naltrexone combination attenuates drug-primed reinstatement to cocaine and morphine in rats in a conditioned place preference paradigm. Addict Biol. doi:10.1111/adb.12020
Abdelhamid EE, Sultana M, Portoghese PS et al (1991) Selective blockage of delta opioid receptors prevents the development of morphine tolerance and dependence in mice. J Pharmacol Exp Ther 258:299–303
Zhu Y, King MA, Schuller AG et al (1999) Retention of supraspinal delta-like analgesia and loss of morphine tolerance in delta opioid receptor knockout mice. Neuron 24:243–252
Ciccocioppo R, Martin-Fardon R, Weiss F (2002) Effect of selective blockade of mu(1) or delta opioid receptors on reinstatement of alcohol-seeking behavior by drug-associated stimuli in rats. Neuropsychopharmacology 27:391–399
Marinelli PW, Funk D, Harding S et al (2009) Roles of opioid receptor subtypes in mediating alcohol-seeking induced by discrete cues and context. Eur J Neurosci 30:671–678
Kotlinska JH, Gibula-Bruzda E, Pachuta A et al (2010) Influence of new deltorphin analogues on reinstatement of cocaine-induced conditioned place preference in rats. Behav Pharmacol 21:638–648
Le Merrer J, Faget L, Matifas A et al (2012) Cues predicting drug or food reward restore morphine-induced place conditioning in mice lacking delta opioid receptors. Psycho-pharmacology 223:99–106
Lenard NR, Daniels DJ, Portoghese PS et al (2007) Absence of conditioned place preference or reinstatement with bivalent ligands containing mu-opioid receptor agonist and delta-opioid receptor antagonist pharmacophores. Eur J Pharmacol 566:75–82
Chefer VI, Shippenberg TS (2009) Augmentation of morphine-induced sensitization but reduction in morphine tolerance and reward in delta-opioid receptor knockout mice. Neuropsychopharmacology 34:887–898
Le Merrer J, Plaza-Zabala A, Del Boca C et al (2011) Deletion of the delta opioid receptor gene impairs place conditioning but preserves morphine reinforcement. Biol Psychiatry 69:700–703
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this protocol
Cite this protocol
Fattore, L., Fadda, P., Antinori, S., Fratta, W. (2015). Role of Opioid Receptors in the Reinstatement of Opioid-Seeking Behavior: An Overview. In: Spampinato, S. (eds) Opioid Receptors. Methods in Molecular Biology, vol 1230. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-1708-2_24
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1708-2_24
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-1707-5
Online ISBN: 978-1-4939-1708-2
eBook Packages: Springer Protocols