Abstract
The study was conducted to evaluate the microbiological and chemical profiles of elephant grass inoculated with and without different wild strains of lactic acid bacteria. Silage was prepared of four treatments and one control with three replicates as control (EKC, adding 2 ml/kg sterilizing water), Lactobacillus plantarum (USA commercial bacteria) (EKP), Lactobacillus plantarum (EKA), Pediococcus acidilactici (EKB), and Pediococcus acidilactici (SKD) isolated from King grass. Silage were prepared using polyethylene terephthalate bottles, and incubated at room temperature for different ensiling days. The pH and acetic acid (AA) were significantly (P < 0.05) reduced and lactic acid (LA), butyric acid (BA), and ethanol were significantly increased (P < 0.05) at 3, 5, 7, and 14 days in treatment groups as compared to control. Water-soluble carbohydrate (WSC) and NH3–N concentration was not affected at days 3, 5, and 7, but significantly (P < 0.05) reduced at 14 days in treatment groups as compared to control. The LA, BA, and ethanol were significantly (P < 0.05) increased and AA, WSC NH3–N, and yeast were significantly (P < 0.05) decreased at 30 days of ensiling in treatment groups as compared to control. It is recommended that the inoculation of LAB could improve the fermentation quality of elephant grass silage and further effort is needed to evaluate these effects on silage produced on farm scale and on animal production performance.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Elephant grass can be attributed to the role that it plays as the major livestock feed in smallholder dairy production systems in China and other parts of the world in tropical and subtropical areas (Desta et al. 2016; Gulfam et al. 2016). Ensiling is a traditional preservation method of green plant material, which aimed the availability of nutritious and palatable feed for animals throughout the year. Moreover, during the fermentation process, the epiphytic lactic acid bacteria (LAB) on forage utilizes water-soluble carbohydrates (WSC) to produce lactic acid (LA); as a result, pH is dropped in silage and reduced the risk of a clostridial fermentation by the native bacterial population(Ni et al. 2014). According to the review and literature, 75% studies have reported that inoculated silages reduced pH, NH3, and enhanced lactic acid production. Furthermore, it was reported that mostly, tropical and subtropical grasses are very difficult to be ensiled due to their low WSC, low lactic acid bacteria, and high buffer capacity (Desta et al. 2016). The epiphytic microorganisms existed naturally in forage crops are responsible for silage fermentation and also influence the effectiveness of silage bacterial inoculation. Consequently, the previous studies have focused on how to get better fermentation quality by isolating LAB, ideally competent of dominating lactic fermentation from epiphytic LAB of forage or silage (Liu et al. 2012). Different chemicals and biological additives have been used to improve silage fermentation. Many researchers used different silage additives such as molasses, formic acid, cellulose, fibrolytic enzyme, and LAB to obtain good quality silage (Guo et al. 2014; Ni et al. 2014; Desta et al. 2016).
The applications of the above additives during the ensiling improve the fermentation quality, rapid pH, and NH3 decline and enhanced lactic acid. Therefore, some species of Lactobacillus such as Lactobacillus plantarum, Pediococcus species, and Enterococcus species are commonly used as additives. The LAB can improve the level of acidification and fermentation quality by decreasing pH, NH3, dry matter, and protein degradation of different grass silages (Driehuis et al. 2001; Wrobel et al. 2008; Shah et al. 2017).
The objective of the present study was to find the effect of microbiological and chemical profiles of elephant grass inoculated with and without different wild strains of lactic acid bacteria.
Materials and methods
Experimental site
A field experiment was conducted at the Institute of Ensiling and Processing of Grass, Nanjing Agricultural University, Weigang No. 1, Nanjing 210095, P. R China. Elephant grass was planted on 08 June 2016 in an experimental field (Humid subtropical climate, Latitude 32°01′59.81′N, Longitude 118°50′13.63″E, and Altitude of 17 m above mean sea level). Elephant grass was harvested at the mature stage on 01 December 2016.
Preparation of the experimental silages
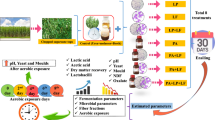
Fresh elephant grass was chopped into 1–2 cm long pieces using a knife and chopped in a stationary machine (Sh-2000, Shanghai Donxe Industrial Co., Ltd., China) and ensiled in anaerobic polyethylene terephthalate bottles of 1 L capacity. Each silo contained 600 g of fresh elephant grass treated with the following five treatments before ensiling: Control (EKC), lactobacillus plantarum (MTD/1CB, Ecosyl Products Inc. USA commercial bacteria) (EKP), Lactobacillus plantarum (EKA), Pediococcus acidilactici (EKB), and Pediococcus acidilactici (EKD) isolated from King grass. The number of bacteria of each strain was adjusted at 1 × 105 cfu/g. After treating and mixing, each treatment (three samples per treatment) was packed into polyethylene terephthalate bottles (silos), followed by sealing with a screw top and kept at the ambient temperature. The screw top was fitted with a synthetic fermentation trap filled with water to prevent the entry of the air. Each triplicate silo for each treatment was opened on days 3, 5, 7, 14, and 30.
Collection of samples for fermentative quality and chemical composition
Silages samples were taken at 3, 5, 7, 14, and 30 days. 35 g silage subsamples were macerated with 70 ml of distilled water and stored in a refrigerator at 4 °C for 24 h. After 24 h, the silage extract was filtered through double-layered cheesecloth and a filter paper (Xinhua Co., China) and pH of silage extract was recorded by pH meter (HANNA pH 211, Hanna Instruments Italia Sel, Italy). The filtered solution was stored at − 20 °C for further analysis of NH3–N and organic acid. 80 g silage subsamples were taken at the same days after silo opening of the plastic pouches and taken in the oven at 65 °C for 48 h. After 48 h, the silage subsamples were weighted for dry mater (DM). Dry mater was recorded and the silage samples were grinded by high-speed universal grinder (Hainai ship Hi-100C, HainaiYinjiang Litong-trade company Lit., Zhejiang, China). The silage powder was used for the WSC, neutral detergent fiber (NDF), and acid detergent fiber (ADF). Dry matter content of fresh and treated silage subsample was determined by the method (International 2005), while NDF and ADF were determined by the method described by (Van Soest et al. 1991). Water-soluble carbohydrates (WSC) were analyzed by a reaction with anthrone reagent (Arthur Thomas 1977). VFAs and LA were determined with gas chromatography (Shimadzu GC-17A, Japan, with 12 m capillary column, condition: column temperature 130 °C, and injection temperature 220 °C) and contents of NH3–N were analyzed using the methodology of (Barker and Summerson 1941; Chaney and Marbach 1962).
Microbial population examination
20 g silage subsample with 180 ml sterilized water (NaCl8.5%) using a medium-speed shaker incubator (Crystal, 1S-RDV1) for 2 h and, after 2 h, made a serial dilution in sterilized water. Lactic acid bacteria were examined in Man Rogosa and Sharp (MRS) agar media, Yeasts were examined on Potato dextrose agar (PDA), and aerobic bacteria were examined on nutrient agar (NA) plates and were taken in an incubator at 37 °C for 3 days. After the microbial data examined, the data were transformed to log10 and obtained on a wet weight basis.
Statistical analysis
The experiment was conducted in a 5 × 5 factorial design (5 inoculants × 5 fermentation periods) using a randomized design with three replicates. The model includes the effects of inoculant (I), fermentation period (P), and the interaction I × P. The data were analyzed using GLM procedure of (SPSS, version 12.0). Means of the significantly affected traits were separated by Duncan Multiple Range Test (Duncan 1955). P value less than 0.05 was considered to be statistically significant. The linear regression relationships between different inoculants and different fermentation periods were quantified using Microsoft Excel 2010 (Microsoft Corporation, Redmond, WA, USA).
Results
Chemical composition of elephant grass before ensiling and after ensiling is presented in Table 1 and Figs. 1, 2, and 3, respectively. The pH and acetic acid (AA) were significantly (P < 0.05) reduced and lactic acid, butyric acid (BA), and ethanol were significant (P < 0.05) at 3, 5, 7, and 14 days in treatment groups as compared to control. The DM was not significantly affected among the treatment and control groups at 3, 5, 7, and 14 days of ensiling period, but the value was numerically increased and decreased. Water-soluble carbohydrate (WSC) and NH3–N concentration were not affected at days 3, 5, and 7, but significantly reduced at 14 days in treatment groups as compared to control. Microbial composition of elephant grass treated with lactic acid bacteria strains during ensiling is presented in Fig. 4. The microbial (LAB) counting of the elephant grass silage during different ensiling days (3, 5, 7, and 14) was significantly (P < 0.05) increased in all treatments groups as compared to control. Aerobic bacteria and yeast were significantly deceased at 3, 5, 7, and 14 days in all treatments groups as compared to control. The regression analysis of pH, AA, PA, BA, DM, and WSC results was obtained from experiment, there were highly positive relationships between fermentation period and different inoculant effects, as shown in Figs. 5, 7, 8, 9, 10 and 13, and the relationships were expressed by a lozenge function during the fermentation periods. The R 2 values ranged from 0.953 to 0.449, 0.999 to 0.975, 0.547 to 0.728, 0.716 to 0.0499, 0.699 to 0.002, and 0.741 to 0.204 during the fermentation periods, respectively. The NH3–N, AB, and yeast results were obtained from experiment, there were poorly positive relationships between fermentation period and different inoculant effects, as shown in Figs. 12, 15, and 16, and the relationships were expressed by a lozenge function during the fermentation periods. The R 2 values ranged from 0.1638 to 0.416, 0.353 to 0.863, and 0.244 to 0.693 during the fermentation periods, respectively. The LA, ethanol, and LAB results were obtained from experiment, there were not found relationships between fermentation periods and different inoculant effects, as shown in Figs. 6, 11, and 14, and the relationships were expressed by a lozenge function during the fermentation periods. The R 2 values ranged from 0.978 to 0.895, 0.584 to 0.641, and 0.825 to 0.914 during the fermentation periods, respectively. Chemical composition and fermentation characteristics of different LAB additives on 30 days of elephant grass silage are described in Table 2. The DM, LA, BA, and ethanol were significantly (P < 0.05) increased and AA, WSC, NH3–N, and yeast were significantly (P < 0.05) decreased in all treatments groups as compared to control. Propionic acid (PA) neutral detergent fiber (NDF), acid detergent fiber (ADF), and aerobic bacteria (AB) were not affected among the treatment and control groups, but the values were numerically increased and decreased.
Regression analysis between different inoculants (I) and different fermentation periods (P) on lactic acid (LA). (EKC, adding 2 ml/kg sterilizing water), Lactobacillus plantarum (commercial bacteria) (EKP), Lactobacillus plantarum (EKA), Pediococcus acidilactici (EKB), and Pediococcus acidilactici (EKD)
Regression analysis between different inoculants (I) and different fermentation periods (P) on acetic acid (AA). (EKC, adding 2 ml/kg sterilizing water), Lactobacillus plantarum (commercial bacteria) (EKP), Lactobacillus plantarum (EKA), Pediococcus acidilactici (EKB), and Pediococcus acidilactici (EKD)
Regression analysis between different inoculants (I) and different fermentation periods (P) on propionic acid (PA). (EKC, adding 2 ml/kg sterilizing water), Lactobacillus plantarum (commercial bacteria) (EKP), Lactobacillus plantarum (EKA), Pediococcus acidilactici (EKB), and Pediococcus acidilactici (EKD)
Regression analysis between different inoculants (I) and different fermentation periods (P) on butyric acid (BA). (EKC, adding 2 ml/kg sterilizing water), Lactobacillus plantarum (commercial bacteria) (EKP), Lactobacillus plantarum (EKA), Pediococcus acidilactici (EKB), and Pediococcus acidilactici (EKD)
Regression analysis between different inoculants (I) and different fermentation periods (P) on dry mater (DM). (EKC, adding 2 ml/kg sterilizing water), Lactobacillus plantarum (commercial bacteria) (EKP), Lactobacillus plantarum (EKA), Pediococcus acidilactici (EKB), and Pediococcus acidilactici (EKD)
Regression analysis between different inoculants (I) and different fermentation periods (P) on water-soluble carbohydrates (WSC). (EKC, adding 2 ml/kg sterilizing water), Lactobacillus plantarum (commercial bacteria) (EKP), Lactobacillus plantarum (EKA), Pediococcus acidilactici (EKB), and Pediococcus acidilactici (EKD)
Regression analysis between different inoculants (I) and different fermentation periods (P) on lactic acid bacteria (LAB). (EKC, adding 2 ml/kg sterilizing water), Lactobacillus plantarum (commercial bacteria) (EKP), Lactobacillus plantarum (EKA), Pediococcus acidilactici (EKB), and Pediococcus acidilactici (EKD)
Regression analysis between different inoculants (I) and different fermentation periods (P) on aerobic bacteria (AB). (EKC, adding 2 ml/kg sterilizing water), Lactobacillus plantarum (commercial bacteria) (EKP), Lactobacillus plantarum (EKA), Pediococcus acidilactici (EKB), and Pediococcus acidilactici (EKD)
Discussion
The most important purpose of using LAB inoculants is to get a lactic acid kind fermentation that results in well preserved silage at the time of silage making. It is commonly believed that microbial inoculation of silage has helpful effects on fermentation by lowering pH and BA and enhancing the level of lactic acid (Nkosi et al. 2010). The pH of the silages after 90 days of ensiling was decreased to 4.6 which are measured for suitable silages with a DM content of 350 g/kg (Charmley et al. 2006). In the present study, the representative of pH is also 4.4 and LAB inoculants improved the formation of LA and decreased pH as well as concentrations of BA and NH3–N compared to control. (Aksu et al. 2004) reported that the LAB improved LA concentrations and declined pH in maize silage. Low pH, low NH3–N concentration, low numbers of yeast, and mould indicated that silages with or without additives were preserved well. These results showed that anaerobic environment prevailed surrounded by the bales and confirmation for unwanted microbial movement was quite small. According to Keles et al. (2009), who reported that when baled silages are wrapped with a sufficient level of plastic stretch film, the anaerobic environment produced is sufficient to reduce undesirable microbial action; on that condition, the physical honesty of the plastic seal is maintained.
Water-soluble carbohydrates are regarded as important substrates for the development of LAB for suitable fermentation (Ni et al. 2014). According to Desta et al. (2016), the concentration of WSC more than 30 g/kg DM in herbage is significant for successful fermentation. The WSC concentration in potato mix up before ensiling was 78 g/kg DM, analytical of enough WSC for capable fermentation. The remaining WSC was lower in the inoculated silages compared to the control after 90 days of ensiling, and represented that WSC was better utilized by LAB in the inoculated silages (Desta et al. 2016; Gulfam et al. 2016). Ammonia N in silage reflects the level of protein degradation and generally proteolysis harmfully affects the use of N by ruminants (Wilkinson 2005; Wrobel et al. 2008). A good quality silages should be contain less than 100 g/kg NH3–N/TN (Aksu et al. 2004; Gulfam et al. 2016), and the elephant grass silages had NH3–N/TN concentrations of less than 100 g/kg NH3–N/TN (Table 2). However, inoculating the elephant grass at ensiling with both Lactobacillus plantarum and Pediococcus acidilactici reduced the NH3–N/TN concentration compared to control, behind conclusion of many researchers (Nkosi et al. 2010; Liu et al. 2012; Guo et al. 2014; Pholsen et al. 2016). These LAB inoculants had a helpful effect on pH lowering and resulting in a decreased NH3–N/TN formation in silage. Desta et al. (2016) and Shah et al. (2017) reported that a rapid decreasing pH is desirable to decrease the quantity of protein degradation in the silo. The present study also shows decreasing pH, AA, WSC and NH3–N/TN concentrations in all treatment groups as compared to the control.
The cell wall apparatus (NDF and ADF) of silages was not changed by the inculcation of LAB. This indicates that LAB both homofermentative and heterofermentative have no direct effects on these nutrients. Homofermentative LAB and enzyme combination stabilizer did not also change the level of cell wall apparatus and WSC concentration of silages. This suggests that enzymes did not change the fiber degradation of high DM-baled triticale silage. Similar results (Meeske et al. 2002) and Desta et al. (2016) reported that homofermentative LAB and enzyme combination had no effect on cell wall apparatus of baled barley and Napier grass silage with a DM of 298–328 g/kg.
Zhang et al. (2011) reported that high levels of ethanol accretion may well increase the fermentation quality of the Napier grass silage, because these results show that ethanol played an important role in inhibition of aerobic bacteria and yeast growth. The present study also improved the ethanol level at 30 days of ensiling.(Filya et al. 2007; Shah. 2017) found that increase microbial number increases LA, ethanol and decreased pH, AA, WSC, NH3–N, yeast, and aerobic bacteria in inoculated silage compared with the control. (Amanullah et al. 2014; Kim et al. 2015) reported that at some stage of ensiling, there was no significant (P < 0.05) variation in DM, CP, NDF, and ADF between treatment and control group. This result of our study granted with previously reported study, where King grass (Pennisetum purpureophoides) was treated with Epiphytic Lactic Acid Bacteria and Tannin of Acacia (Santoso et al. 2011). Throughout ensiling period, protein is degraded to peptides and free amino acid by plant proteases (Owens et al. 2002). In result, degradation of amino acids to ammonia and non-protein nitrogenous fraction is mostly due to proteolytic clostridia. The formation of AA is the sign of wasteful fermentation or of secondary fermentation of LA–BA and degradation of amino acid to NH3–N/TN by method of formation of AA from the carbon structure of the amino acid (Driehuis et al. 2001; Filya et al. 2007; Pholsen et al. 2016).
Conclusions
It is concluded that the inoculation of LAB improved the fermentative characteristics of elephant grass. Further studies should be conducted to evaluate the effects on farm scale and animal production performance.
References
Aksu T, Baytok E, Bolat D (2004) Effects of a bacterial silage inoculant on corn silage fermentation and nutrient digestibility. Small Rumin Rese 55:249–252
Amanullah S, Kim D, Lee H, Joo Y, Kim S, Kim S (2014) Effects of microbial additives on chemical composition and fermentation characteristics of barley silage. Asian Australas J Animal Sci 27:511
Arthur Thomas T (1977) An automated procedure for the determination of soluble carbohydrates in herbage. J Sci Food Agric 28:639–642
Barker SB, Summerson WH (1941) The colorimetric determination of lactic acid in biological material. J Biol Chem 138:535–554
Chaney AL, Marbach EP (1962) Modified reagents for determination of urea and ammonia. Clin Chem 8:130–132
Charmley E, Nelson D, Zvomuya F (2006) Nutrient cycling in the vegetable processing industry: utilization of potato by-products. Can J Soil Sci 86:621–629
Desta ST, Yuan X, Li J, Shao T (2016) Ensiling characteristics, structural and nonstructural carbohydrate composition and enzymatic digestibility of Napier grass ensiled with additives. Bioresour Technol 221:447–454
Driehuis F, Oude Elferink S, Van Wikselaar P (2001) Fermentation characteristics and aerobic stability of grass silage inoculated with Lactobacillus buchneri, with or without homofermentative lactic acid bacteria. Grass Forage Sci 56:330–343
Duncan DB (1955) Multiple range and multiple F tests. Biometrics 11:1–42
Filya I, Muck R, Contreras-Govea F (2007) Inoculant effects on alfalfa silage: fermentation products and nutritive value. J Dairy Sci 90:5108–5114
Gulfam A, Guo G, Tajebe S, Chen L, Liu Q, Yuan X, Bai Y, Saho T (2016) Characteristics of lactic acid bacteria isolates and their effect on the fermentation quality of Napier grass silage at three high temperatures. J Sci Food Agric 97:1931–1938
Guo G, Yuan X, Li L, Wen A, Shao T (2014) Effects of fibrolytic enzymes, molasses and lactic acid bacteria on fermentation quality of mixed silage of corn and hulless–barely straw in the Tibetan Plateau. Grassland Sci 60:240–246
International A (2005) Official methods of analysis of AOAC International. AOAC International 5:1269–1278
Keles G, O’Kiely P, Lenehan JJ, Forristal PD (2009) Conservation characteristics of baled grass silages differing in duration of wilting, bale density and number of layers of plastic stretch-film. Irish J Agric Food Res 1:21–34
Kim DH, Amanullah SM, Lee HJ, Joo YH, Kim SC (2015) Effect of microbial and chemical combo additives on nutritive value and fermentation characteristic of whole crop barley silage. Asian Australas J Animal Sci 28:1274
Liu Q, Chen M, Zhang J, Shi S, Cai Y (2012) Characteristics of isolated lactic acid bacteria and their effectiveness to improve stylo (Stylosanthes guianensis Sw.) silage quality at various temperatures. Anim Sci J 83:128–135
Meeske R, Van der Merwe G, Greyling J, Cruywagen C (2002) The effect of adding an enzyme containing lactic acid bacterial inoculant to big round bale oat silage on intake, milk production and milk composition of Jersey cows. Anim Feed Sci Technol 97:159–167
Ni K, Wang Y, Pang H, Cai Y (2014) Effect of cellulase and lactic acid bacteria on fermentation quality and chemical composition of wheat straw silage. Am J Plant Sci 5:1877
Nkosi B, Meeske R, Van der Merwe H, Groenewald I (2010) Effects of homofermentative and heterofermentative bacterial silage inoculants on potato hash silage fermentation and digestibility in rams. Anim Feed Sci Technol 157:195–200
Owens V, Albrecht K, Muck R (2002) Protein degradation and fermentation characteristics of unwilted red clover and alfalfa silage harvested at various times during the day. Grass Forage Sci 57:329–341
Pholsen S, Khota W, Pang H, Higgs D, Cai Y (2016) Characterization and application of lactic acid bacteria for tropical silage preparation. Anim Sci J 87:1202–1211
Santoso B, Hariadia BT, Manik H, Abubakar H (2011) Silage quality of king grass (Pennisetum purpureophoides) treated with epiphytic lactic acid bacteria and tannin of acacia. Media Peternak 34:140
Shah. YX, Zhihao D, Siran W, Tao S (2017) Effects of lactic acid bacteria on ensiling characteristics, chemical composition and aerobic stability of king grass. J Animal Plant Sciences 3:747–755
Van Soest PJ, Robertson JB, Lewis BA (1991) Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J Dairy Sci 74:3583–3597
Wilkinson J (2005) Silage. Chapter 19: Analysis and clinical assessment of silage. Chalcombe Publications, Southampton
Wrobel B, Zielinska A, Suterska A (2008) Evaluation of quality and aerobic stability of grass silage treated with bacterial inoculants containing Lactobacillus buchneri. In: Proceedings, 13th international conference on forage conservation, Nitra, Slovak Republic, pp 122–123
Zhang L, Yu C, Shimojo M, Shao T (2011) Effect of different rates of ethanol additive on fermentation quality of Napiergrass (Pennisetum purpureum). Asian Australas J Animal Sci 24:636–642
Acknowledgements
This work were supported by 31672488, 2016YFC0502005 and 2016ZDKJZC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors confirm that there is no conflict of interests.
Additional information
Communicated by Erko Stackebrandt.
Rights and permissions
About this article
Cite this article
Shah, A.A., Xianjun, Y., Zhihao, D. et al. Microbiological and chemical profiles of elephant grass inoculated with and without Lactobacillus plantarum and Pediococcus acidilactici . Arch Microbiol 200, 311–328 (2018). https://doi.org/10.1007/s00203-017-1447-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-017-1447-1