Abstract
Introduction and hypothesis
Levator ani muscle (LAM) avulsion is associated with pelvic organ prolapse (POP). There is limited information on the prevalence of LAM avulsion in Chinese women with POP. This study evaluated the prevalence of LAM avulsion in women presenting with POP and the effects on their quality of life (QoL).
Methods
This prospective study was conducted in a tertiary urogynecology center. Chinese women presenting with POP were recruited and completed standardized questionnaires exploring their symptoms and QoL and underwent gynecological examination to assess the stage of POP and involved compartments. Four-dimensional translabial ultrasound (US) was performed on all women, and evaluation of LAM avulsion was done offline.
Results
Three hundred and ninety-eight women completed the study. The prevalence of LAM avulsion was 38.9% [95% confidence interval (CI) 34.1–43.7%]; 31 (7.8%), 26 (6.5%), and 98 (24.6%) had left, right, and bilateral LAM avulsion, respectively. LAM avulsion was associated with a more advanced stage of prolapse (P < 0.005) and prolapse of stage ≥II of all three compartments. Bilateral LAM avulsion was associated with a more severe stage of prolapse. More women with LAM avulsion reported bothersome symptoms of prolapse and had higher Pelvic Organ Prolapse Distress Inventory (POPDI) and Urinary Incontinence Questionnaire (UIQ) scores, implying more impairment in QoL. However, after multivariate linear regression analysis, POP stage remained a significant predictor of POPDI and UDI scores (P ≤ 0.006); LAM avulsion was not a predictor (P = 0.2 and 0.27, respectively).
Conclusions
LAM avulsion was detected in 39% of Chinese women with POP. It was associated with a more advanced POP stage, and these women had more impairment of QoL. However, LAM avulsion was not an independent factor influencing the QoL of these women.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The levator ani muscle (LAM) is important for pelvic floor support [1]. LAM avulsion was found in 15–21% of women after vaginal deliveries, with the first delivery associated with the highest risk [2,3,4,5]; risk was greater after forceps delivery [2, 6]. Although LAM avulsion may result in a reduction in pelvic floor muscle contractility [4, 7], it was not associated with symptoms of pelvic floor disorders and impaired quality of life (QoL) in the months following delivery [6, 8].
Mean latency between first vaginal delivery and presentation of pelvic organ prolapse (POP) has been reported to be 33.5 years [9], and women with POP had impaired QoL [10]. LAM avulsion has been found in 32–80% of women with POP [9, 11, 12]. This condition is important because it increases the risk of prolapse recurrence, even after surgical treatment [12]. However, previous studies on the prevalence of LAM avulsion in women with POP mainly involved Caucasian women. It is known that there are differences in pelvic floor muscle biometry and pelvic organ mobility between nulliparous East Asian and Caucasian women [13]. There are also possible differences labor management during delivery, such as rates of epidural analgesia and episiotomy in different populations, which may affect the incidence of pelvic floor trauma [2, 14].
The aim of this study was to define the prevalence of LAM avulsion in Chinese women presenting with POP and to study the impact of LAM avulsion on their QoL.
Materials and methods
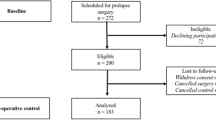
This prospective observational study was conducted in a tertiary urogynecology center. Chinese women, who were referred with a chief complaint of POP and were not on active treatment for it, were invited to participate in the study. Women currently managed with a vaginal ring pessary or who were not able to fill in the standard questionnaires were excluded. Written consent was obtained, and the study was approved by the institutional ethics committee (CREC 2015.125).
Women were asked to complete the validated Chinese Pelvic Floor Distress Inventory (PFDI) and Pelvic Floor Impact Questionnaire (PFIQ) during their first consultation to explore their symptoms and health-related QoL [15]. They were also asked to grade the impact of pelvic floor disorders using a 1- to 10-cm visual analog scale, with a higher score referring to greater impairment. Demographic data and body mass index (BMI) were obtained. Women were then assessed by gynecologists who were blinded to the above information to determine prolapse stage according to the Pelvic Organ Prolapse Quantification (POP-Q) system and the involved compartments [16]. Women who were not able to give consent and/or to fill in the PFDI and PFIQ were excluded from the study.
A standard translabial ultrasound (US) scan was performed to assess pelvic floor anatomy. A GE Voluson 730 3D US system (GE Medical systems, Zipf, Austria) with a 4- to 8-MHz 3D/4D autosweep transducer was used for image acquisition. The transducer was placed on the perineum in the midsagittal plane immediately after voiding, with women in the supine position. US volumes with a sweep angle of 85 ° were obtained at rest, during maximal pelvic floor muscle contraction, and during maximal Valsalva [17]. The volume data sets were saved and analyzed in a standard manner. Offline analysis of US data was done remote from the actual examination. At most, three contractions and three Valsalva were performed.
The LAM was assessed using tomographic US imaging on volumes obtained at maximal pelvic floor contraction at 2.5-mm slice intervals from 5-mm below to 12.5-mm above the plane of minimal hiatal dimensions, which is the horizontal line between the pubic symphysis and anorectal junction in the midsagittal plane [18]. LAM avulsion was defined according to the minimal criteria for complete avulsion of the LAM in at least three central slices obtained at the level of minimal hiatal dimension and 2.5 and 5 mm above [18]. In doubtful cases, measurements of the levator–urethral gap were made by placing calipers in the center of the hypoechogenic structure that indicates the urethra and on the most medial aspect of the muscle insertion [18]. A cutoff of 23.65 mm for the levator–urethral gap has 92% sensitivity and 95% specificity for diagnosing LAM avulsion in Chinese women with POP [19]. In cases where the woman could not perform pelvic floor contraction, the volume at rest was used for analysis.
The terminology in this manuscript followed the IUGA-ICS joint report on terminology for female pelvic floor dysfunction [20].
Sample size
LAM avulsion was found in 15–21% of women after vaginal deliveries and in 32–80% of women with POP [2,3,4, 9, 11, 12]. We assume its prevalence in Chinese women with POP to be 40%, with an accepted error of 5%, thereby making the sample size needed 369.
Statistical analysis
Descriptive analysis was used to study the prevalence of LAM avulsion, POP stage, and demographic data. Chi-squared test and Mann–Whitney U test were used for statistical comparisons. Multivariate linear regression analysis was used to assess POP stage and LAM avulsion to predict women’s QoL. P < 0.05 was considered statistically significant.
Results
A total of 458 women were referred to our unit with the chief complaint of POP during the study period. However, 14 (3.1%) were already being managed with a vaginal ring pessary, and another 46 (10%) could not complete the questionnaire due to time constraints, illiteracy, or mental illness, leaving a total of 398 (86.9%) women with complete data sets. Mean age was 62.4 ± 10.1 years (range 33–88), mean parity 3.0 ± 1.6, and mean BMI 25.2 ± 3.7 kg/m2. Twenty-three women had a history of hysterectomy, among whom, two had a history of vaginal hysterectomy and pelvic floor repair. Although they experienced POP recurrence, they did not receive treatment before attending the clinic. Overall, 9.0%, 65.9%, 22.1%, and 3.0% had stage I, II, III, and IV POP, respectively; 83.3%, 58.3%, and 26.9% had stage ≥II anterior, middle, and posterior compartment prolapse, respectively.
LAM avulsion was found in 155 women [38.9%, 95% confidence interval (CI) 34.1–43.7%]. In all, 31 (7.8%) had left, 26 (6.5%) right, and 98 (24.6%) bilateral LAM avulsion. There was no significant difference in age, BMI, parity, and number of vaginal births for women with or without LAM avulsion. Women who had LAM avulsion were associated with a more advanced stage of prolapse (P < 0.005) (Table 1). LAM avulsion was also associated with prolapse of stage ≥II in all three compartments. Bilateral LAM avulsion was associated with more severe stage of prolapse (Table 2).
Regarding PFDI question 4: “Do you usually have sensation of bulging or protrusion from vaginal area?” and question 5: “Do you usually have a bulge or something falling out that you can see or feel in the vaginal area?” More women in the LAM avulsion group reported that symptoms bothered them (avulsion vs no avulsion group: question 4, 80% vs 70.4%; question 5, 81% vs 72%, both P = 0.04). PFDI subscale scores were higher for the avulsion group than for the no avulsion group, but only the POPDI reached statistical significance (P = 0.035) (Table 3). PFIQ Urinary Impact Questionnaire (UIQ) score was significantly higher for the group with LAM avulsion [20.8 (3.3–72.1) vs 12.6 (0–44.7); P = 0.049] but not for the Pelvic Organ Prolapse Impact Questionnaire (POPIQ) or the Colorectal Anal Impact Questionnaire (CRAIQ) (Table 3). The UIQ travel and social subscale scores were significantly higher in women with LAM avulsion (both P = 0.03). There was also a tendency for higher self-reported VAS scores in the group with LAM avulsion: 5.2 [standard deviation (SD) 2.7] vs 4.7 (2.9), P = 0.07, although this did not reach statistical significance.
Women who had stage III or IV POP had higher PFDI subscale scores than women who had stage I or II POP (Table 3). After conducting multivariate linear regression analysis to assess POP stage and LAM avulsion to predict QoL, only POP stage remained a significant predictor of POPDI (P < 0.005) and UDI (P = 0.006) scores; LAM avulsion was not a predictor (P = 0.2 and 0.27, respectively).
Discussion
Translabial US is very useful in the investigation of pelvic floor anatomy, with detachment of the puborectalis muscle clearly visualized. Furthermore, US is usually more widely available than magnetic resonance imaging (MRI). It is encouraging that the same method for diagnosing LAM avulsion using translabial US has been adopted in many previous publications, allowing comparison between different studies [18]. A more complicated scoring system is not needed, as it has not been shown to be superior to the discrete diagnostic method [18, 21].
The prevalence of LAM avulsion in women with pelvic floor disorders who presented to urogynecology units were reported to be 15–37% [7, 21, 22]. Among them, 41% had bilateral LAM avulsion [7]. In women who would receive POP surgery, the prevalence was 53.6% [11]. In this study, we found that 38.9% of women with POP had LAM avulsion. Our prevalence is expected to be higher if the previous studies included women presenting not only for POP but also for urinary incontinence, fecal incontinence, or defecatory problems [7, 21, 22]. It was lower, though, in women receiving surgical treatment [11]. This could be because women with more severe prolapse had a higher tendency to choose surgical treatment and that LAM avulsion is a risk factor for vaginal pessary expulsion [10, 23].
Previous research has confirmed the association of LAM avulsion with anterior- and middle-compartment prolapse and significant prolapse defined as stage ≥ II[12]. However, there is limited information between LAM avulsion and prolapse stage. Here, we report that the rate of LAM avulsion increases with prolapse stage, with > 50% of women with LAM avulsion if they had stage III or IV prolapse. Looking for LAM avulsion, especially in women with advanced prolapse stages, may help with counseling on management options and treatment efficacy. LAM avulsion increases the risk of failure in retaining a vaginal pessary and in having a higher rate of recurrence after surgical repair [11, 23].
Shortly after delivery, women with avulsion noticed reduced pelvic floor strength and had a lower POP-Q Aa and Ba point and a higher POPDI general score [8, 24]. However, there was no association of LAM avulsion and impaired QoL when women were followed up 1 year after delivery [8]. It may be doubtful that LAM avulsion impaired QoL in women with POP. Until only recently, Abdool et al. reported that significant associations between awareness and visualization of a vaginal lump by women with POP and LAM avulsion [25]. Our findings from PFDI questions 4 and 5 were consistent with their findings. However, interference with everyday life was not associated with avulsion in their study [25]. We found that the PFDI-POPDI and PFIQ-UIQ scores were higher for the LAM avulsion than the no avulsion group. There was also a tendency of higher self-reported VAS score for women with LAM avulsion. However, the difference on QoL scores between women with or without LAM avulsion was small. On multivariate regression analysis, only POP stage was a predicator of POPDI and UIQ scores; LAM avulsion was not a predictor. This implies that POP stage exerts more influence on QoL of women, and women with a more advanced stage had qreater QoL impairment compared with women with a milder POP stage. Presence of LAM avulsion was not an independent factor affecting QoL. This was consistent with Endress et al.’s findings [26]. Apart from LAM avulsion, other pelvic floor trauma may contribute to POP and impaired QoL of women, e.g., mobility of pelvic organs or irreversible hiatal distension.
The strength of our study included a reasonably large sample size and a homogenous group of women of the same ethnicity, for which there is limited information in the literature. We also used standardized questionnaires to study pelvic floor symptoms and QoL. However, since only Chinese women were studied, results may not be generalizable to other populations.
In conclusion, 39% of women with POP had LAM avulsion. LAM avulsion was associated with a more advanced POP stage. Women with more advanced POP stage had greater QoL impairment. However, LAM avulsion was not an independent factor influencing QoL.
References
DeLancey JO. The anatomy of the pelvic floor. Curr Opin Obstet Gynecol. 1994;6:313–6.
Chan SC, Cheung YK, Yiu KW, Lee LL, Pang WL, Choy KW, et al. Prevalence of levator ani muscle injury in Chinese women after first delivery. Ultrasound Obstet Gynaecol. 2012;39:704–9.
Van Delft K, Sultan AH, Thakar R, Schwertner-Tiepelmann N, Kluivers K. The relationship between postpartum levator ani muscle avulsion and signs and symptoms of pelvic floor dysfunction. BJOG. 2014;121:1164–71. discussion 1172
Guzman Rojas R, Wong V, Shek KL, Dietz HP. Impact of levator trauma on pelvic floor muscle function. Int Urogynecol J. 2014;25:375–80.
Chan SS, Cheung RY, Lee LL, Choy KW, Chung TKH. A longitudinal follow-up of levator ani muscle avulsion: Does a second delivery affect it? Ultrasound Obstet Gynecol. 2016; https://doi.org/10.1002/uog.16009. Chan SS, Cheung RY, Lee LL, Choy KW, Chung TKH. A longitudinal follow-up of levator ani muscle avulsion: Does a second delivery affect it? Ultrasound Obstet Gynecol. 2017;50:110–5.
Chung MY, Wan OY, Cheung RY, Chung TK, Chan SS. Prevalence of levator ani muscle injury and health-related quality of life in primiarous Chinese women after instrumental delivery. Ultrasound Obstet Gynecol. 2015;45:728–33.
Steensma AB, Konstantinovic ML, Burger CW, de Ridder D, Timmerman D, Deprest J. Prevalence of major levator abnormalities in symptomatic patients with an underactive pelvic floor contraction. Int Urogynecol J. 2010;21:861–7.
Chan SS, Cheung RY, Yiu KW, Lee LL, Chung TK. Effect of levator ani muscle injury on primiparous women during the first year after childbirth. Int Urogynecol J. 2014;25:1381–8.
Thomas V, Shek KL, Guzman Rojas R, Dietz HP. Temporal latency between pelvic floor trauma and presentation for prolapse surgery: A retrospective observational study. Int Urogynecol J. 2015;26:1185–9.
Chan SS, Cheung RY, Yiu KW, Lee LL, Pang AW, Chung TK. Symptoms, quality of life, and factors affecting women’s treatment decisions regarding pelvic organ prolapse. Int Urogynecol J. 2012;23:1027–33.
Abdul Jalil SS, Guzman Rojas R, Dietz HP. Does it matter whether levator avulsion is diagnosed pre- or postoperatively? Ultrasound Obstet Gynecol. 2016;10(48):516–9.
Dietz HP, Simpson JM. Levator trauma is associated with pelvic organ prolapse. BJOG. 2008;115:979–84.
Cheung RY, Shek KL, Chan SS, Chung TK, Dietz HP. Pelvic floor muscle biometry and pelvic organ mobility in east Asian and Caucasian nulliparae. Ultrasound Obstet Gynecol. 2015;45:599–604.
Shek KL, Dietz HP. Intrapartum risk factors for levator trauma. BJOG. 2010;117:1485–92.
Chan SS, Cheung RY, Yiu AK, Li JC, Lai BP, Choy KW, et al. Chinese validation of pelvic floor distress inventory and pelvic floor impact questionnaire. Int Urogynecol J. 2011;22:1305–12.
Bump RC, Mattiasson A, Bo K, Brubaker LP, DeLancey JO, Karskov P, et al. The standardization of terminology of female pelvic organ prolapse and pelvic floor dysfunction. Am J Obstet Gynecol. 1996;175:10–7.
Dietz HP. Pelvic floor ultrasound: A review. Am J Obstet Gynecol. 2010;202:321–34.
Dietz HP, Bernado MJ, Kirby A, Shek KL. Minimal criteria for the diagnosis of avulsion of the puborectalis muscle by tomographic ultrasound. Int Urogynecol J. 2011;22:699–704.
Zhuang RR, Song YF, Chen ZQ, Ma M, Huang HJ, Chen JH, et al. Levator avulsion using a tomographic ultrasound and magnetic resonance-based model. Am J Obstet Gynecol. 2011;205:232.e1–8.
Haylen BT, de Ridder D, Freeman RM, Swift SE, Berghmans B, Lee J, et al. An international Urogynecologic association (IUGA)/international continence society (ICS) joint report on the terminology for female pelvic floor dysfunction. Int Urogynecol J. 2010;21:5–26.
Trutnovsky G, Kamisan Atan I, Ulrich D, Martin A, Dietz HP. Levator ani trauma and pelvic organ prolapse – A comparison of three translabial ultrasound scoring system. Acta Obstet Gynecol Scand. 2016;95:1411–7.
Dietz HP, Steensma AB. Prevalence of major abnormalities of the levator ani in urogynaecological patients. BJOG. 2006;113:225–30.
Cheung RY, Lee JH, Lee LL, Chung TK, Chan SS. Levator ani muscle avulsion is a risk factor for vaginal pessary expulsion within 1 year for pelvic organ prolapse. Ultrasound Obstet Gynecol. 2017; https://doi.org/10.1002/uog.17407.
Dietz HP, Shek KL, Chantarasorn V, Langer SE. Do women notice the effect of childbirth-related pelvic floor trauma? Aust N Z J Obstet Gynecol. 2012;52:277–81.
Abdool Z, Dietz HP, Lindeque BG. Prolapse symptoms are associated with abnormal functional anatomy of the pelvic floor. Int Urogynecol J. 2017; https://doi.org/10.1007/s00192-017-3280-0.
Endress E, Shek KL, Dietz HP. Levator avulsion is not associated with symptom bother of female pelvic organ prolapse. Arch Gynecol Obstet. 2015;292:629–33.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Financial disclaimers
None.
Rights and permissions
About this article
Cite this article
Yu, C.H., Chan, S.S.C., Cheung, R.Y.K. et al. Prevalence of levator ani muscle avulsion and effect on quality of life in women with pelvic organ prolapse. Int Urogynecol J 29, 729–733 (2018). https://doi.org/10.1007/s00192-017-3454-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-017-3454-9