Abstract
Purpose
To report second-look arthroscopic assessment after all-arthroscopic autologous chondrocyte implantation (ACI) for articular cartilage defects at the patella.
Methods
A second-look arthroscopy after all-arthroscopic ACI using chondrospheres® (ACT3D) was performed in 30 patients with 30 full-thickness retropatellar cartilage defects. The mean time from ACI to second-look arthroscopy was 14.9 ± 16.3 (6–71) months. The quality of cartilage regeneration was evaluated by the International Cartilage-Repair Score (ICRS)—Cartilage Repair Assessment (CRA).
Results
Eleven lesions (36.7%) were classified as CRA grade I (normal) and 19 lesions (63.3%) as grade II (nearly normal). Concerning the degree of defect repair, 25 lesions (83.3%) were repaired up to the height of the surrounding articular retropatellar cartilage. Five lesions (16.7%) showed 75% repair of defect depth. The border zone was completely integrated into the surrounding articular cartilage shoulder in 28 lesions (93.3%) and demarcated within 1 mm in 2 lesions (6.7%). Macroscopically and by probing, 12 lesions (40%) had intact smooth surface, 17 lesions (56.7%) had fibrillated surface and 1 lesion (3.3%) had small, scattered fissures. A negative correlation was found between the overall repair assessment score and the defect size (r2 = − 0.430, p = 0.046) and between integration into border zone and defect size (r2 = − 0.340, p = 0.045). A positive correlation was found between macroscopic appearance and age (r2 = + 0.384, p = 0.036).
Conclusions
All-arthroscopic ACI using chondrospheres® (ACT3D) for full-thickness retropatellar articular cartilage defects proved to be reproducible and reliable. The advantage of the procedure is that it is minimal invasive. Arthroscopic second-look demonstrated a high grade of normal or nearly normal cartilage regeneration. Although statistically significant differences were not observed, larger defect size and younger age may compromise the result of overall repair.
Level of evidence
III.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Retropatellar chondral defects may cause anterior knee pain and the prevalence of patellofemoral joint damage assessed by magnetic resonance images is reported to be between 15 and 20% [33]. The cartilage defects are often diagnosed arthroscopically and the prevalence is secondary to cartilage lesions at the medial femoral condyle [3, 32]. Trauma, patella dislocation and abnormal joint loading can contribute to the cartilage damage, which may gradually deteriorate due to alterations from normal weight bearing, leading to osteoarthritis [17].
Several surgical treatment options have been proposed for retropatellar chondral defects. Arthroscopic chondral debridement [9], microfracture [19, 22, 26], autologous matrix-induced chondrogenesis [11, 22, 28] and osteochondral transplantation [4, 15] are discussed among treatment options. Recently autologous chondrocyte implantation (ACI) has been reported to be one of the surgical options with good outcomes [11, 18, 27]. Several authors report good clinical results after ACI at the patella [5, 6, 12,13,14, 16, 18, 20, 21, 25, 26, 34,35,36]. However, second-look arthroscopy was rarely done. Even if it was performed, the number of patients was few and limited [12, 13, 36]. Therefore, the visual aspect after ACI for retropatellar cartilage defects is still unknown.
Full size articular cartilage defects at the patella are usually treated by an arthrotomy and open ACI [25, 26].
The purpose of this study is to report second-look results of cartilage regeneration after arthroscopic ACI for the treatment of full size retropatellar cartilage defects. The advantage of the method is that it is minimal invasive allowing for easier rehabilitation. The hypothesis is that retropatellar arthroscopic ACI may be reliable leading to good clinical results.
Materials and methods
From 2012 to 2018, 30 patients with 30 full-thickness retropatellar cartilage defects underwent second-look arthroscopy after all-arthroscopic ACI at our institution. The patients’ demographic data are shown in Table 1. The mean size of the cartilage defects was 4.4 ± 3.7 cm2 (0.9–14) and the mean time from ACI to second-look arthroscopy was 14.9 ± 16.3 months (6–71). All patients were clinically and radiologically assessed before the ACI. In case of patella malalignment, additional procedures such as a medial tibia tubercle transfer (Elmslie–Trillat) or/and a medial patellofemoral ligament reconstruction (MPFL-R) were combined with the ACI. Other combined surgeries were partial meniscus resection, anterior cruciate ligament reconstruction and autologous matrix-induced chondrogenesis (AMIC) for the medial femoral condyle (MFC), which were performed simultaneously to the ACI. In total, 21 patients (70%) had concomitant surgeries. All surgeries were performed by the senior surgeon. This study was approved by the local IRB of the ATOS Clinic Heidelberg (ID 02/2020).
All-arthroscopic ACI
The procedure of ACI needs two stages: cartilage-harvest and implantation of the cultured chondrocytes. After arthroscopic inspection of the joint, the retropatellar chondral defect was assessed. In case of indication for ACI, a cartilage biopsy was taken from the intercondylar notch and the chondrocytes were cultivated for 8 weeks [7, 10] (co.don AG, Berlin, Germany). At the time of arthroscopic ACI, the retropatellar cartilage defect was debrided with a curette and a shaver. The arthroscopic fluid was retrieved from the joint to create ideally dry conditions for the implantation. Dry-arthroscopically, the three-dimensional autologous chondrocyte spheroids (chondrospheres®) were placed into the defect using a special applicator and were spread evenly with a probe. The adhesion of spheroids to the defect was obtained within several minutes. Detailed data of the cultured spheroids in this study are shown in Table 2.
Postoperative rehabilitation
Free range of motion supported by a CPM device twice a day for 30 min and physiotherapy were encouraged from the second postoperative day. 10 kg weight bearing was allowed in week 1–6, 20 kg in week 7–8, followed by full weight bearing thereafter. Swimming and cycling were encouraged from week 8 and jogging earliest 1 year postoperatively. High-intensity sports were permitted 1–1.5 years postoperatively depending on the type of sports.
Second-look arthroscopy
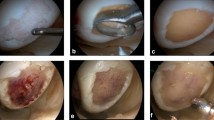
The quality of the recovered cartilage was assessed visually and also by probing according to the International Cartilage-Repair Score (ICRS)—Cartilage Repair Assessment (CRA) [2]. The assessment was performed by the senior surgeon. An example is shown in Fig. 1. The reasons for the second-look arthroscopy are shown in Table 3.
Statistical analysis
Data were analysed using the Statistical Package for Statcel 4 (OMS, Saitama, Japan). All measured values were expressed as means ± standard deviation (SD) and range. Person’s correlation coefficient was applied to analyse correlations between predictors and ICRS-CRA. P < 0.05 was considered for statistical significance.
Results
According to the ICRS-CRA, 11 lesions (36.7%) were classified as grade I (normal) and 19 lesions (63.3%) as grade II (nearly normal). None of the lesions was abnormal or severely abnormal. Concerning the degree of defect repair, 25 lesions (83.3%) were repaired up to the height of surrounding cartilage and 5 lesions (16.7%) showed 75% repair of defect depth. The border zone was completely integrated with surrounding cartilage in 28 lesions (93.3%) and demarcated within 1 mm in 2 lesions (6.7%). Macroscopically, 12 lesions (40%) had intact smooth surface, 17 lesions (56.7%) had fibrillated surface and 1 lesion (3.3%) had small, scattered fissures. The detailed result of ICRS-CRA is shown in Table 4. The mean time from ACI to second-look arthroscopy was 14.9 months; however, three patients demonstrated relatively longer intervals: 53, 62 and 71 months, respectively. The ICRS-CRA for these three patients was 11, 11 and 12 (maximum score is 12), respectively. There was no drop out from the study. All patients treated with a retropatellar ACI at our institution and who had a second look have been included in this study.
A negative correlation was found between the ICRS-CRA and the defect size (r2 = − 0.430, p = 0.046), and integration into border zone and defect size (r2 = − 0.340, p = 0.045). A positive correlation was found between the macroscopic appearance of the ACI and age (r2 = + 0.384, p = 0.036). The other predictors (sex, BMI, operated side, defect size, number of spheroids, size of spheroids, number of spheroids for size of defect, concomitant surgeries and time from ACI) did not have correlations with ICRS-CRA (Table 5). In this analysis, the detected correlation coefficients are in a weak to moderate category.
Discussion
The most important finding in this study is that full-thickness retropatellar articular cartilage lesions can regenerate to a normal or nearly normal cartilage appearance at second-look arthroscopy in the majority of cases when treated by an arthroscopic ACI. A larger defect size had a negative effect on the overall repair assessment score and the integration of the ACI into the border zone, whereas age at time of ACI had a positive correlation with the macroscopic cartilage appearance.
Previously, several studies reported a hypertrophy of the ACI when combined with a periosteal flap: Adachi et al. reported results of second-look arthroscopy after ACI in 15 patients with retropatellar full-thickness cartilage defects [1]. The ACI was performed in an open fashion using a periosteal membrane. Two years postoperatively nine patients were classified as grade II (nearly normal), five were as grade III (abnormal) and one was as grade IV (severely abnormal) according to the ICRS-CRA. The authors reported that hypertrophy was the most commonly seen complication due to the use of periosteal membrane. Similarly, Henderson et al. used an ACI with periosteal flap to repair full size articular cartilage defects at the patella [16]. A total of 23 out of 44 patients were re-operated due to hypertrophy or extrusion related to the periosteal patch. In addition, Gillogly et al. reported a high reoperation rate after ACI combined with periosteal flap for retropatellar cartilage lesions [12]. Of 25 knees, 10 knees (40%) underwent subsequent surgery for arthroscopic debridement for graft and for removal of a loose body. Zarkadis et al. reported a series of 51 knees who underwent patellofemoral ACI with a periosteal membrane (11 knees, 15%) or a type I/III collagen membrane (40 knees, 85%) [36]. Twenty-five percent of patients had a second-look arthroscopy showing a complete incorporation of the regenerate cartilage. Four patients (5%) required a surgical debridement for hypertrophy of the periosteal flap. A lower number of revisions were observed when no periosteal flap was used. Vanlauwe et al. treated 38 patellofemoral cartilage defects with ACI using a bilayered collagen membrane. Nine patients (25%) underwent a re-arthroscopy, four (11.1%) for hypertrophic tissue and two (5.6%) for partial loosening of less than 20% of the repaired tissue [34].
Arthroscopic results were reported by Gobbi et al. [13] performing ACI using HYAFF11 (Hyalograft C) in 25 retropatellar lesions. Six knees (24%) underwent a second-look arthroscopy at a mean time of 15.7 months which revealed a CRA grade II (nearly normal) in all repaired lesions.
In contrast, in the present study, the CRA was normal in 40% and nearly normal in 60% of patients which is a remarkable number of good cartilage repair and only 2 reoperations due to hypertrophy.
In the present study, larger size lesions tend to have a lower overall assessment score and a reduced integration into border zone. Previously, Niemeyer demonstrated that no correlation was found between the size of the defect and morphological aspects on MRI [24]. However, MRI results might be difficult to be interpreted and may be less precise compared to a visual inspection by arthroscopy. Gobbi et al. reported of six patients who underwent a second-look arthroscopy in lesions with an average size of 4.5 cm2. Most tissue repairs were well integrated with the surrounding cartilage, however, the detailed scores were not mentioned in their study [13]. Similar results were reported by Zarkadis et al. who performed a second-look arthroscopy in six patients. All showed a complete incorporation of the regenerate cartilage after ACI in defects with an average size of 4.3 cm2 [36]. In the present study, the cartilage defects were an average of 4.43 cm2 large, which was almost equivalent to the previous reports. A strength of the study is that the number of second-look arthroscopies was much higher (n = 30) than previously reported, which could give more detailed insight on the relationship between lesion size and arthroscopic findings.
In the current study, the macroscopic appearance of the regenerated cartilage at time of second-look had a positive correlation with the patient’s age at time of ACI. In the literature, there are reports that younger patients are more suitable candidates for an ACI due to their biological healing potential [23]. However, Zarkadis et al. concluded that age < 30 years was a risk factor for failure of a retropatellar ACI. The authors discuss that failures in the general population may be due to an age-related decline in cartilage quality and viability. In contrast, a younger military population tends to fail for activity-related reasons and due to the inability to self-modify professional activity [36]. In an evaluation of MACI procedures, Enea et al. pointed out, that CRA values at second-look were significantly higher in patients older than 40 years [8]. The histological examination of biopsies also showed that the outcome was not significantly related either to the macroscopic appearance of the lesion nor to the patient’s functional status.
In the past, it was impossible to treat a retropatellar full size articular cartilage lesion with an arthroscopic ACI. So far, the surgical procedure required an arthrotomy [5, 6, 12,13,14, 16, 18, 20, 21, 34,35,36]. In contrast, the procedure used in this study allowed an all-arthroscopic ACI as previously described [29,30,31]. The technique could be of benefit for the patients because it is less invasive, reduce pain and encourage a faster rehabilitation.
In our study, there was no correlation between the result of second-look arthroscopy and the number of spheroids per unit area. This is similar to the findings by Niemeyer et al. [24]. They demonstrated that a higher number of spheroids per unit area had a trend towards better defect filling, while the outcomes were similar regardless of the number of spheroids after 12 months.
There are some limitations to this study. First, there was no assessment of patient’s clinical symptoms. Many patients underwent concomitant patella-related surgical procedures which makes isolated assessment of patients’ cartilage-related clinical symptoms difficult. Second, no biopsies were performed for not to harm the repair tissue. Thus, the relationship between morphologic and histological results cannot be discussed. The mean time from ACI to second-look arthroscopy was 14.9 months. Although three patients with relatively long intervals after ACI demonstrated a good overall repair assessment score, the results must be considered early-stage results after ACI. Unfortunately, we do not have any histologies of the regenerated cartilage. However, this was done in the interest of the patients for not to damage the ACI and compromise the clinical result.
The study is of clinical relevance. It is the first to show that arthroscopic retropatellar ACI is reliable and does lead to reproducible good results of cartilage regeneration controlled by second-look arthroscopy. The findings support the use of spheroids for arthroscopic retropatellar ACI and do show that open surgery for retropatellar ACI may be abandoned in the future. Further studies are necessary to support the current findings.
Conclusion
All-arthroscopic ACI using chondrospheres® (ACT3D) for full-thickness retropatellar articular cartilage defects proved to be reproducible and reliable. The advantage of the procedure is that it is minimal invasive. Arthroscopic second-look demonstrated a high grade of normal or nearly normal cartilage regeneration. Although statistically significant differences were not observed, larger defect size and younger age may compromise the result of overall repair.
References
Adachi N, Ochi M, Deie M, Nakamae A, Kamei G, Uchio Y et al (2014) Implantation of tissue-engineered cartilage-like tissue for the treatment for full-thickness cartilage defects of the knee. Knee Surg Sports Traumatol Arthrosc 22(6):1241–1248
Brittberg M, Winalski CS (2003) Evaluation of cartilage injuries and repair. J Bone JtSurg Am 85-A(Suppl 2):58–69
Curl WW, Krome J, Gordon ES, Rushing J, Smith BP, Poehling GG (1997) Cartilage injuries: a review of 31,516 knee arthroscopies. Arthroscopy 13(4):456–460
Donoso R, Figueroa D, Espinoza J, Yañez C, Saavedra J (2019) Osteochondral autologous transplantation for treating patellar high-grade chondral defects: a systematic review. Orthop J Sports Med 7(10):2325967119876618
Ebert JR, Fallon M, Smith A, Janes GC, Wood DJ (2015) Prospective clinical and radiologic evaluation of patellofemoral matrix-induced autologous chondrocyte implantation. Am J Sports Med 43(6):1362–1372
Ebert JR, Schneider A, Fallon M, Wood DJ, Janes GC (2017) A comparison of 2-year outcomes in patients undergoing tibiofemoral or patellofemoral matrix-induced autologous chondrocyte implantation. Am J Sports Med 45(14):3243–3253
EMA (2017) Spheroids of human autologous matrix-associated chondrocytes. https://www.ema.europa.eu/en/medicines/human/EPAR/spherox#product-information-section. Accessed 23 Oct 2020
Enea D, Cecconi S, Busilacchi A, Manzotti S, Gesuita R, Gigante A (2012) Matrix-induced autologous chondrocyte implantation (MACI) in the knee. Knee Surg Sports Traumatol Arthrosc 20(5):862–869
Federico DJ, Reider B (1997) Results of isolated patellar debridement for patellofemoral pain in patients with normal patellar alignment. Am J Sports Med 25(5):663–669
Fickert S, Gerwien P, Helmert B, Schattenberg T, Weckbach S, Kaszkin-Bettag M et al (2012) One-year clinical and radiological results of a prospective, investigator-initiated trial examining a novel, purely autologous 3-dimensional autologous chondrocyte transplantation product in the knee. Cartilage 3(1):27–42
Fossum V, Hansen AK, Wilsgaard T, Knutsen G (2019) Collagen-covered autologous chondrocyte implantation versus autologous matrix-induced chondrogenesis: a randomized trial comparing 2 methods for repair of cartilage defects of the knee. Orthop J Sports Med 7(9):2325967119868212
Gillogly SD, Arnold RM (2014) Autologous chondrocyte implantation and anteromedialization for isolated patellar articular cartilage lesions: 5- to 11-year follow-up. Am J Sports Med 42(4):912–920
Gobbi A, Kon E, Berruto M, Filardo G, Delcogliano M, Boldrini L et al (2009) Patellofemoral full-thickness chondral defects treated with second-generation autologous chondrocyte implantation: results at 5 years’ follow-up. Am J Sports Med 37(6):1083–1092
Gomoll AH, Gillogly SD, Cole BJ, Farr J, Arnold R, Hussey K et al (2014) Autologous chondrocyte implantation in the patella: a multicenter experience. Am J Sports Med 42(5):1074–1081
Hangody L, Dobos J, Baló E, Pánics G, Hangody LR, Berkes I (2010) Clinical experiences with autologous osteochondral mosaicplasty in an athletic population: a 17-year prospective multicenter study. Am J Sports Med 38(6):1125–1133
Henderson IJ, Lavigne P (2006) Periosteal autologous chondrocyte implantation for patellar chondral defect in patients with normal and abnormal patellar tracking. Knee 13(4):274–279
Insall J, Falvo KA, Wise DW (1976) Chondromalacia patellae. A prospective study. J Bone JtSurg Am 58(1):1–8
Kon E, Filardo G, Gobbi A, Berruto M, Andriolo L, Ferrua P et al (2016) Long-term results after hyaluronan-based MACT for the treatment of cartilage lesions of the patellofemoral joint. Am J Sports Med 44(3):602–608
Kreuz PC, Steinwachs MR, Erggelet C, Krause SJ, Konrad G, Uhl M et al (2006) Results after microfracture of full-thickness chondral defects in different compartments in the knee. OsteoarthrCartil 14(11):1119–1125
Macmull S, Jaiswal PK, Bentley G, Skinner JA, Carrington RW, Briggs TW (2012) The role of autologous chondrocyte implantation in the treatment of symptomatic chondromalacia patellae. Int Orthop 36(7):1371–1377
Meyerkort D, Ebert JR, Ackland TR, Robertson WB, Fallon M, Zheng MH et al (2014) Matrix-induced autologous chondrocyte implantation (MACI) for chondral defects in the patellofemoral joint. Knee Surg Sports Traumatol Arthrosc 22(10):2522–2530
Migliorini F, Eschweiler J, Maffulli N, Driessen A, Rath B, Tingart M et al (2021) Management of patellar chondral defects with autologous matrix induced chondrogenesis (AMIC) compared to microfractures: a four years follow-up clinical trial. Life (Basel) 11(2):141
Nawaz SZ, Bentley G, Briggs TW, Carrington RW, Skinner JA, Gallagher KR et al (2014) Autologous chondrocyte implantation in the knee: mid-term to long-term results. J Bone JtSurg Am 96(10):824–830
Niemeyer P, Laute V, John T, Becher C, Diehl P, Kolombe T et al (2016) The effect of cell dose on the early magnetic resonance morphological outcomes of autologous cell implantation for articular cartilage defects in the knee: a randomized clinical trial. Am J Sports Med 44(8):2005–2014
Ogura T, Bryant T, Merkely G, Minas T (2019) Autologous chondrocyte implantation for bipolar chondral lesions in the patellofemoral compartment: clinical outcomes at a mean 9 years’ follow-up. Am J Sports Med 47(4):837–846
Olivos Meza A, Cortés González S, Ferniza Garza JJ, Pérez Jiménez FJ, Enrique VC, Ibarra C (2019) Arthroscopic treatment of patellar and trochlear cartilage lesions with matrix encapsulated chondrocyte implantation versus microfracture: quantitative assessment with MRI T2-mapping and MOCART at 4-year follow-up. Cartilage. https://doi.org/10.1177/19476035198359091947603519835909
Peterson L, Vasiliadis HS, Brittberg M, Lindahl A (2010) Autologous chondrocyte implantation: a long-term follow-up. Am J Sports Med 38(6):1117–1124
Schiavone Panni A, Del Regno C, Mazzitelli G, D’Apolito R, Corona K, Vasso M (2018) Good clinical results with autologous matrix-induced chondrogenesis (Amic) technique in large knee chondral defects. Knee Surg Sports Traumatol Arthrosc 26(4):1130–1136
Siebold R, Karidakis G, Feil S, Fernandez F (2016) Second-look assessment after all-arthroscopic autologous chondrocyte implantation with spheroides at the knee joint. Knee Surg Sports Traumatol Arthrosc 24(5):1678–1685
Siebold R, Karidakis G, Fernandez F (2014) Clinical outcome after medial patellofemoral ligament reconstruction and autologous chondrocyte implantation following recurrent patella dislocation. Knee Surg Sports Traumatol Arthrosc 22(10):2477–2483
Siebold R, Suezer F, Schmitt B, Trattnig S, Essig M (2018) Good clinical and MRI outcome after arthroscopic autologous chondrocyte implantation for cartilage repair in the knee. Knee Surg Sports Traumatol Arthrosc 26(3):831–839
Solheim E, Krokeide AM, Melteig P, Larsen A, Strand T, Brittberg M (2016) Symptoms and function in patients with articular cartilage lesions in 1,000 knee arthroscopies. Knee Surg Sports Traumatol Arthrosc 24(5):1610–1616
Stefanik JJ, Niu J, Gross KD, Roemer FW, Guermazi A, Felson DT (2013) Using magnetic resonance imaging to determine the compartmental prevalence of knee joint structural damage. OsteoarthrCartil 21(5):695–699
Vanlauwe JJ, Claes T, Van Assche D, Bellemans J, Luyten FP (2012) Characterized chondrocyte implantation in the patellofemoral joint: an up to 4-year follow-up of a prospective cohort of 38 patients. Am J Sports Med 40(8):1799–1807
von Keudell A, Han R, Bryant T, Minas T (2017) Autologous chondrocyte implantation to isolated patella cartilage defects. Cartilage 8(2):146–154
Zarkadis NJ, Belmont PJ Jr, Zachilli MA, Holland CA, Kinsler AR, Todd MS et al (2018) Autologous chondrocyte implantation and tibial tubercle osteotomy for patellofemoral chondral defects: improved pain relief and occupational outcomes among US army service members. Am J Sports Med 46(13):3198–3208
Funding
No funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors have no potential conflict of interest.
Ethical approval
The study was approved by the local ethics committee of the ATOS hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sumida, Y., Nakamura, K., Feil, S. et al. Good healing potential of patellar chondral defects after all-arthroscopic autologous chondrocyte implantation with spheroids: a second-look arthroscopic assessment. Knee Surg Sports Traumatol Arthrosc 30, 1535–1542 (2022). https://doi.org/10.1007/s00167-021-06584-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-021-06584-x