Abstract
The dietary uptake of Cd and Pb may contribute to the decline of migratory red-crowned cranes (Grus japonensis) on the Asian mainland. To uncover the relevance of this hypothesis, we determined the concentrations of Pb and Cd as well as further macro and trace elements (Ca, Mg, Cu, Zn, Hg and As) in the gastric contents, gastric wall, intestinal wall, liver, kidney, muscle, and feathers of two individuals found dead in Zhalong Wetland in Northeastern China. Indeed, the Pb concentrations in the liver and kidney tissues was with 31.4 and 60.3 mg kg−1 dry weight (dw), respectively, above concentrations considered as potentially toxic level in common birds (i.e. 30 mg kg−1). These Pb concentration may have possibly been associated with lethal toxicosis in this endangered species suggesting Pb as major threat for G. japonensis populations. Thus, the inputs of Pb into Zhalong Wetland should be reduced to maintain and reestablish environmental conditions supporting the population development of these migratory red-crowned cranes in the Zhalong Wetland, a critical crane habitat for the long-term sustainability of this species.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Excessive exposure to trace elements has been extensively investigated because of their toxic effects on biotic systems (Burger and Gochfeld 1993; Eisler 2000). Thus, the assessment of trace element concentrations in birds may provide early warning of changes in their population health (Friend 1985; Dauwea et al. 2005; Scheuhammer 1996). The liver and kidneys of wild animals are commonly used for such evaluations (Wiemeyer and Withers 2004; Orłowski et al. 2012; Kim and Oh 2014), because most toxic trace elements mainly accumulate in these organs (Agusa et al. 2005). However, most baseline tissue levels from environmental exposure and lethal thresholds are described in common birds or mammals. Whether these established thresholds are applicable to rare water birds, such as cranes, remains unknown. For example, Pb toxicosis is a commonly reported cause of toxicity among birds, but lethal levels are variable among tissues and species (Locke and Thomas 1996). For instance, a Pb concentration of 72 mg kg−1 dry weight (dw) in the liver of an endangered whooping crane (Grus americana) is associated with poisoning (Snyder et al. 1992). Pb concentrations ranging from 15.5 to 17.7 mg kg−1 dw have been detected in the kidney of dead nestling rooks (Corvus grugilegus) (Orłowski et al. 2012).
Red-crowned crane (Grus japonensis) is an endangered species in East Asia with a small global population of 2750 mature individuals (BirdLife International 2012). The resident population in Japan remains stable (Teraoka et al. 2007), while the migratory population of the Asian mainland has declined due to the loss and degradation of their habitat (i.e. wetlands) for agricultural and industrial development (Harris 2008). Thus, red-crowned cranes increasingly rely on agricultural lands in northeastern China, placing the cranes at greater risk for poisoning (Su and Zhou 2012; Luo et al. 2014a, b). A previous study found a common inorganic pesticide and relatively high Pb and Cd concentrations in the sediment and prey of red-crowned cranes in this region (Luo et al. 2013, Luo et al. 2014a, Luo et al. 2014b). These conditions probably enhance the exposure of these cranes to potentially toxic doses of pesticides and trace elements (Luo et al. 2015a). However, published toxicological analyses among red-crowned cranes have been rarely performed.
Our study aimed to determine the distribution and concentration of trace elements in tissues of two dead red-crowned cranes. The findings from this case study may be used to optimize environmental management.
Materials and Methods
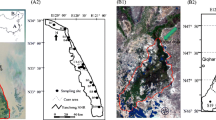
Two moribund red-crowned cranes were collected in the field in July 2014 in Zhalong Wetland (Table 1). Before the cranes’ death, they exhibited loss of appetite, lethargy, weakness, emaciation, tremors, and drooped wings. The cranes had neither wounds nor external parasites, suggesting the birds likely consumed toxic substances or suffer from disease. The carcasses of the birds were immediately transported under refrigeration to the laboratory for dissection. An infectious disease was ruled out on the basis of gross pathology beyond the emaciated state of the birds.
Polyethylene gloves were used throughout the dissection procedure to prevent external contamination. The proventriculus and ventriculus (stomach) of the red-crowned cranes were dissected from between the esophagus and small intestine. Each stomach was incised in half with a razor blade and the contents were flushed into a Petri dish for inspection. Likely lead-containing particles, such as shotgun pellets, were not detected in the stomach. Food items were examined and sorted using a zoom stereomicroscope and divided into three categories: animal, plant, and mineral according to Orłowski et al. (2009). Additionally, mineral particle size was determined using the wet sieve method (Gee and Bauder 1982). The extracted fraction was dried to weigh for contents of 0.2–2, 2–5, and 5–10 mm in size, and classified as coarse sand, fine gravel, and media gravel, respectively, based on the U.S. Department of Agricultural soil texture criteria (Gent 1983).
Eight red-crowned crane samples were sampled during 2010–2014 with the aid of fire inspectors at the Zhalong Wetland (Table 1; Fig. 1). The direct causes of death of these cranes were identified as starvation resulting from food shortage under freezing conditions and collision with power lines, as determined by pathological inspection (Luo et al. 2015a). For the purposes of this report, the eight cranes served as negative comparisons to the 2 cases of suspected poisoning.
Approximately 1–2 g of liver, kidney, gastric wall, intestine, pectoral skeletal muscle, and stomach content samples (wet weight) were collected for acid digestion. Several flight feathers were also collected. Each sample was acid-digested in a microwave in accordance with USEPA (1996) methods. The techniques used in our study were described by Luo et al. (2015a). Ca, Mg, Cu, Zn, Pb, Cd, and As concentrations in the six sample types were determined through inductively coupled plasma-mass spectrometry (Agilent 7500ce, Agilent Technologies, Inc., Santa Clara, CA, USA). We estimated the precision and accuracy of the analyses on the basis of two certified reference materials (Beijing Shiji Ouke Bio-tech Co., Ltd): Pseudoscianea crocea (GBW08573) for Cu (1.36 ± 0.13 mg kg−1), Zn (28.8 ± 1.4 mg kg−1), Pb (8.8 ± 1.10 mg kg−1), Cd (0.014 ± 0.001 mg kg−1), and As (5.08 ± 0.39 mg kg−1). The results agreed with the certified values for all metals, with average recovery rates of 102 %, 94 %, 103 %, 105 %, and 102 % for Cu, Zn, Pb, Cd, and As, respectively. We determined the T-Hg concentration in internal tissues and feathers by using a mercury analyzer (Tekran 2600 CVAFS, Tekran Instrument Corporation, Knoxville, USA) in accordance with the methods described in our previous study (Luo et al. 2014b). The detection limit for all elements was 5 ng kg−1. Reusable materials, such as Petri dishes for examining stomach contents, were acid-washed. The samples were analyzed in triplicate at a relative standard deviation lower below 1.5 %. The concentrations were expressed as mg kg−1 dw.
Results and Discussion
The stomach contents of the two cranes were dominated by maize compared to available controls, and lacked animal residues (Table 2). The mean tissue element concentrations determined from the two poisoned cranes exhibited the following order: Ca > Mg > Zn > Pb > Cu > Cd > Hg > As (Table 3). The concentrations of macro elements Ca and Mg were predominant in the cranes, with concentrations ranging from 0.14 to 1.38 g kg−1 and from 0.28 to 1.54 g kg−1, respectively; these concentrations were similar to control cranes (Luo et al. 2015a). The mean tissue concentrations of the trace elements Cu and Zn were high in the liver (24.2–47.3 mg kg−1 dw for Cu, and 263–469 mg kg−1 dw for Zn), kidney (38.8–42.5 mg kg−1 dw for Cu, and 210–370 mg kg−1 dw for Zn), and muscle (23.9–41.5 mg kg−1 for Cu, and 190–269 mg kg−1 dw for Zn). By contrast, the concentrations were relatively low in the feathers (5.98–21.7 mg kg−1 for Cu, and 88.5–174 mg kg−1 dw for Zn). The concentrations of Cu, Zn, Cd, Hg, and As in the two poisoned cranes were generally similar to those in the eight control birds. The stomach contents and the tissues from the two poisoned cranes contained elevated Pb levels compared with the control cranes. The average Pb concentrations in the kidney and liver tissues of the two examined cranes reached 31.4 and 60.3 mg kg−1 dw, respectively. By comparison, the Pb concentrations were 1.83 and 1.85 mg kg−1 dw in the same tissues of the controls. The average Pb concentrations in the tissues of the two poisoned cranes displayed a decreasing pattern: kidney > gastric wall > liver > gut wall > feather > muscle.
In general, the clinical signs of the poisoned cranes before their death and the results of postmortem tissue analyses indicated that excessive intake of Pb might have been associated with the death of the red-crowned cranes. In support of this assumption, a summary of toxic effect Pb level in kidney and liver of birds is provided in Table 4.
In the past 30 years, habitat loss has forced the cranes to rely on crops and farmlands in Zhalong Wetland (Su and Zhou 2012; Luo et al. 2014a). High corn contents left in the stomach of the red-crowned cranes in this case study supported the point. High anthropogenic inputs from the residential, agricultural, and industrial zones surrounding Zhalong Wetland in the past decades also increased the concentrations of trace elements in the sediments and prey of cranes (Luo et al. 2013, 2014b). These changes may ultimately pose a risk to cranes and other birds that rely on food and water from this habitat (Luo et al. 2013, 2015a). However, the two dead cranes only exhibited high concentrations of Pb among the several trace elements analyzed. The Zn concentration of <210 mg kg−1 in the liver was significantly below the toxic level of 2100 mg kg−1 (United States Department of the Interior 1998). By contrast, whooping cranes with a Zn concentration of 420 mg kg−1 dw in the liver are diagnosed with Zn toxicosis (Spalding et al. 1997). The Zn concentration of 150 mg kg−1 wet weight (approximately 430 mg kg−1 dw) in the kidney and the Zn concentration of 150 mg kg−1 wet weight (approximately 450 mg kg−1 dw) in the liver of free-flying trumpeter swans (Cygnus buccinator) are considered elevated (Carpenter et al. 2004) compared with the normal range of 30 mg kg−1 wet weight to 100 mg kg−1 wet weight (approximately 100 to 300 mg kg−1 dw) (Puls 1994). In the present study, the detected Zn concentrations in the liver and kidney of crane specimens were within the normal range set by Puls (1994) and below the toxic level previously reported. Similar to Zn, the detected Cu, Hg, and Cd concentrations in the liver did not exceed the toxicity thresholds: 540 mg kg−1 for Cu and 20 to 24 mg kg−1 dw for Hg (United States Department of the Interior 1998), and 40 mg kg−1 dw for Cd (Eisler 2000).
The degree of Pb toxicity varies according to age, sex, and species (Eisler 1988; Burger and Gochfeld 1993; Orłowski et al. 2012). The Pb concentrations in the liver and kidney of the two dead cranes were similar to the lethal threshold of >24 and 20–30 mg kg−1 dw suggested by Clark and Scheuhammer (2003) and Friend (1985). Teraoka et al. (2007) reported similar liver Pb level, ranging from 30 to 60 mg kg−1, in four dead red-crowned cranes in Japan; however, these concentrations were significantly higher than those reported by Takazawa et al. (2004), Friend (1985), and Clark and Scheuhammer (2003). In addition, Snyder et al. (1992) reported a case of lead toxicosis in a whooping crane from Colorado in the United States. Pb was detected in the liver at 20 mg kg−1 wet weight (approximately 50–70 mg kg−1 dw) and in the kidney at 10 mg kg−1 wet weight (approximately 25–30 mg kg−1 dw), similar to those detected in the red-crowned cranes in the present study. The observed symptoms, such as loss of appetite, lethargy, weakness, emaciation, tremors, and drooped wings, of the moribund red-crowned cranes are typical signs of Pb poisoning (Eisler 1988). These symptoms supported our initial assumption of Pb poisoning in this case. Furthermore, Pb concentration of 30 mg kg−1 dw in the liver or kidney appears to be associated with lethal toxicosis in the red-crowned crane species.
In birds, the background concentrations of Ca and Mg in the liver range from 0.2 to 0.4 g kg−1 dw and from to 0.8 g kg−1 dw, respectively (Wiemeyer and Withers 2004; Custer et al. 2003). According to Gonzalez (1988) and Scheuhammer (1996), high loads of toxic trace elements, such as Pb and Cd, in the avian body likely reduces Ca and Mg contents; as a consequence, Ca and Mg deficiencies occur. We found that Ca and Mg levels in the liver of the dead samples were approximately two times higher than the background concentrations reported by Wiemeyer and Withers (2004) and Custer et al. (2003) in the present study. Therefore, the increased concentrations of Pb did not induce marked Ca or Mg deficiencies. This conflicting result may be obtained because the high content of minerals in the stomach (Table 2) acted as the source of Ca and Mg for the physiological need of cranes and compensated the possible loss induced by the increased Pb concentration in this case; this observation was supported by the finding of Orłowski et al. (2009).
Potential biases should be considered when interpreting our results because other toxic substances from pesticides were not examined in this study. However, the increased Pb concentrations detected in the tissues of the two red-crowned cranes suggested that the inputs of Pb into Zhalong Wetland should be reduced. Thus, the healthy state of this critical crane habitat can be maintained for the long-term sustainability of the crane population.
References
Agusa T, Matsumoto T, Ikemoto T, Anan Y, Kubota R, Yasunaga G, Kunito T, Tanabe S, Ogi H, Shibata Y (2005) Body distribution of trace elements in black-tailed gulls from Rishiri Island, Japan: age-dependent accumulation and transfer to feathers and eggs. Environ Toxicol Chem 24:2107–2120
BirdLife International (2012) Grus japonensis: IUCN 2012. IUCN red list of threatened species, Version 2012. 4. http://www.birdlife.org/datazone/speciesfactsheet. Accessed 5 May 2013
Burger J, Gochfeld M (1993) Lead and cadmium accumulation in eggs and fledgling seabirds in the New York Bight. Environ Toxicol Chem 12:261–267
Carpenter JW, Andrews GA, Beyer WN (2004) Zinc toxicosis in a free-flying trumpeter swan (Cygnus buccinator). J Wildl Dis 40:769–774
Clark AJ, Scheuhammer AM (2003) Lead poisoning in up-land foraging birds of prey in Canada. Ecotoxicology 12:23–30
Custer CM, Custer TW, Anteau MJ, Afton AD, Wooten DE (2003) Trace elements in lesser scaup (Aythya affinis) from the Mississippi flyway. Ecotoxicology 12:47–54
Dauwea T, Janssens E, Bervoetsb L, Blustb R, Eens M (2005) Heavy-metal concentrations in female laying great tits (Parus major) and their clutches. Arch Environ Contam Toxicol 49:249–256
Degernes LA (2008) Waterfowl toxicology: a review. Veterinary Clinics of North America; exotic animal. Practice 11:283–300
Eisler R (1988) Lead hazards to fish, wildlife, and invertebrates: a synoptic review. United States Fish and Wildlife Service Biology Report 85(1.14)
Eisler R (2000) Handbook of chemical risk assessment: health hazards to humans, plants, and animals, vol 1. Metals. Lewis Publishers, Boca Raton
Friend M (1985) Interpretation of criteria commonly used to determine lead poisoning problem areas. Leafleft 2. United States Fish and Wildlife Service. Fish and wildlife, Washington DC
Gee GW, Bauder JW (1982) Particle-size analysis. In: Miller RH, Keeney DR (eds) Methods of soil analysis: part 2. Agronomy monographs: no. 9, 2nd edn. ASA, Madison, pp 383–411
Gent JA Jr (1983) A computer program for determining particle size distribution and soil textural class. Commun Soil Sci Plant 14:347–351
Gonzalez LM (1988) Organochlorine and heavy metal contamination in the eggs of the Spanish imperial eagle (Aquila heliaca adalberti) and accompanying changes in eggshell morphology and chemistry. Environ Pollut 51:241–258
Harris J (2008) Cranes respond to climate change. ICF Bugle 34(1–3):14–15
Kim J, Oh J-M (2014) Assessment of lead exposure in waterfowl species, Korea. Arch Environ Contam Toxicol 67:529–534
Locke LN, Thomas NJ (1996) Lead poisoning of waterfowl and raptors. In: Fairbrother A, Locke LN, Hoff GL (eds) Noninfectious diseases of wildlife, 2nd edn. Iowa State University Press, Ames, pp 108–117
Luo J, Yin X, Ye Y, Wang Y, Zang S, Zhou X (2013) Pb and Cd bioaccumulations in the habitat and preys of red-crowned cranes (Grus japonensis) in Zhalong Wetland, Northeastern China. Biol Trace Elem Res 156:134–143
Luo J, Ye Y, Gao Z, Wang Y, Wang W (2014a) Characterization of heavy metal contamination in the habitat of red-crowned crane (Grus japonensis) in Zhalong Wetland, Northeastern China. Bull Environ Contam Toxicol 93:327–333
Luo J, Ye Y, Wang Y (2014b) Dietary exposure of the red-crowned crane (Grus japonensis) to total and methyl mercury in Zhalong Wetland, Northeastern China. Biol Trace Elem Res 159:210–218
Luo J, Ye Y, Gao Z, Wang W (2015a) Essential and nonessential elements in red-crowned cranes (Grus japonensis) of Zhalong Wetland, Northeastern China. Toxicol Environ Chem 96:1096–1105
Luo J, Ye Y, Gao Z, Wang W (2015b) Stomach content and faecal analysis of red-crowned cranes (Grus japonensis) in Zhalong Wetland, Northeastern China. Biologia 70:542–546
Orłowski G, Kasprzykowski Z, Zawada Z, Kopij G (2009) Stomach content and grit ingestion by Rook Corvus frugilegus nestlings. Ornis Fenn 86:117–122
Orłowski G, Kamiński P, Kasprzykowski Z, Zawada Z, Koim-Puchowska B, Szady-Grad M, Klawe JJ (2012) Essential and nonessential elements in nesting rooks Corvus frugilegus from eastern Poland with a special emphasis on their high cadmium contamination. Arch Environ Contam Toxicol 63:601–611
Pain DJ, Meharg AA, Ferrer M, Taggart M, Penteriani P (2005) Lead concentrations in bones and feathers of the globally threatened Spanish imperial eagle. Biol Conserv 121: 603–610
Puls R (1994) Mineral levels in animal health: diagnostic data, 2nd edn. Sherpa International, Clearbrook, British Columbia, p 349
Scheuhammer AM (1987) The chronic toxicity of aluminum, cadmium, mercury, and lead in bird: a review. Environ Pollut 46:263–295
Scheuhammer AM (1996) Influence of reduced dietary calcium on the accumulation and effects of lead, cadmium, and aluminum in birds. Environ Pollut 94:337–343
Snyder SB, Richard MJ, Thilsted JP, Drewien RC (1992) Lead poisoning in a whooping crane. Technical Report 12. In: Proceedings, 1988 North American Crane Workshop, Nongame Wildlife Program, Florida Fish and Wildlife Conservation Commission, Tallahassee, FL, USA, pp 207–210
Spalding MG, Nesbitt SA, Folk MJ, McDowell LR, Sepulveda MS (1997) Metal consumption by whooping cranes and possible zinc toxicosis. N Am Crane Workshop Proc 7:237–242
Su L, Zhou F (2012) Status, threats and conservation needs for the continental population of the red–crowned crane. Chin Birds 3:147–164
Takazawa Y, Kitamura K, Yoshikane Y, Morita M (2004) Discovery of fenthion poisoning in two Japanese cranes (Grus japonensis) found dead in Hokkaida, Japan. Bull Environ Contam Toxicol 73:947–954
Teraoka H, Kumagai Y, Iwai H, Haraguchi K, Ohba T, Nakai K, Satoh H, Sakamoto M, Momose K, Masatomi H, Hiraga T (2007) Heavy metal contamination status of Japanese cranes (Grus japonensis) in east Hokkaido, Japan–extensive mercury pollution. Environ Toxicol Chem 26:307–312
United States Department of the Interior. 1998. Guidelines for interpretation of the biological effects of selected constituents in biota, water, and sediment. National Irrigation Water Quality Program Information Report No. 3
USEPA (1996) Method 3050B-Acid digestion of sediments, sludges, and soils. United States Environmental Protection Agency, Washington DC
Wiemeyer SN, Withers D (2004) Metals and trace elements in livers of American white pelicans at Anaho Island. Nevada 2004. United States Fish and Wildlife Service, Nevada Fish and Wildlife Office, Reno
Acknowledgments
This research was supported by the Heilongjiang provincial nature fund under Grant No. C2016058.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Luo, J., Ye, Y., Gao, Z. et al. Lead in the Red-Crowned Cranes (Grus japonensis) in Zhalong Wetland, Northeastern China: A Report. Bull Environ Contam Toxicol 97, 177–183 (2016). https://doi.org/10.1007/s00128-016-1853-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-016-1853-0