Abstract
Purpose
The psychosocial health of mother is crucial for healthy prenatal period and early childhood. We investigated the prevalence and risk factors of maternal depression during pregnancy and postpartum among women who participated in a home visitation program in Seoul, South Korea (Seoul Healthy First Step Project, SHFSP).
Methods
We analyzed 80,116 women who participated in the SHFSP, which was launched by Seoul metropolitan government in 2013, and defined peripartum depression as a score ≥ 10 on the Korean version of the Edinburgh Postnatal Depression Scale (EPDS). Sociodemographic factors and psychosocial health status were evaluated through a standardized questionnaire completed by participants upon program registration. We calculated the prevalence of women at risk for peripartum depression and evaluated associated factors by multivariable logistic regression analysis.
Results
Prevalence of women at risk for peripartum depression was 17.7% (prepartum depression: 14.2%, postpartum depression: 24.3%). Younger maternal age, low income (OR 2.40, 95% CI 2.03–2.84), disability (2.61, 1.96–3.47), single parenthood (3.27, 2.69–3.99), and smoking (2.02, 1.44–2.83) increased the peripartum depression risk. Furthermore, experience of stress, change, or loss over the past 12 months (3.36, 3.22–3.50), history of treatment for emotional issues (2.47, 2.27–2.70), experience of child abuse (1.91, 1.74–2.11), and domestic violence (2.25, 1.81–2.80) increased the risk for peripartum depression, whereas having helpers for the baby (0.62, 0.58–0.67), having someone to talk with (0.31, 0.27–0.35), and considering oneself confident (0.30, 0.29–0.31) decreased the risk.
Conclusions
Policies to reduce and manage peripartum depression should be strengthened, with a focus on high-risk pregnant and puerperal women.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The prenatal period and early childhood are critical for lifelong health and wellbeing [1, 2]. Investments in the early years of life contribute to better health in adulthood, thereby reducing the socioeconomic burden of healthcare [1, 2]. Furthermore, health disparities develop during these periods [3]. Therefore, it is pivotal to create an environment where all children can have a healthy prenatal period and early childhood.
The psychosocial health of mothers is crucial for a healthy prenatal period and early childhood because the mother is usually the most important caregiver during these periods. Women at risk for peripartum depression may experience impaired mother–child interaction, low cognitive function, and developmental delays in children [4,5,6], and depression during pregnancy causes suboptimal fetal outcomes, including low birth weight and preterm birth [7]. Therefore, it is important to identify mothers with depressive symptoms and provide appropriate interventions. Home visitation programs in several countries have improved maternal and child health outcomes including maternal depressive symptoms [8,9,10].
Several theoretical models explain the mechanisms of peripartum depression. Biological theories include changes in the hypothalamic–pituitary–adrenal axis and the levels of gonadal hormones during and after pregnancy [11]. Psychosocial theories, including psychodynamic theory, behavioral theory, and social and interpersonal theory, suggest that depression is triggered by interpersonal events and psychosocial stressors [11]. Specifically, sociodemographic factors, psychosocial factors, and behavioral factors may contribute to the development of peripartum depression, whereas social support can mitigate these effects (Fig. 1).
The prevalence of peripartum depression exceeded 10% worldwide in a recent meta-analysis [12] and was approximately 40.5–61.4% in one Korean study; however, that study included a small number of participants and the presence of depression was evaluated by a self-report measure [13]. The overall prevalence estimates evaluated by symptom scales was 1.6 times higher than those evaluated by diagnostic instruments [12]. Another Korean study, which evaluated postpartum depression by the depression diagnosis codes in the national health insurance database, reported the prevalence of postpartum depression to be 1.4% [14]. In these studies, women with peripartum depression reported symptoms, such as not being able to laugh, blaming oneself, being anxious for no good reason, difficulty sleeping, feeling sad, feeling so unhappy that it causes oneself to cry, and the thought of harming oneself. Several factors, including low socioeconomic status, immigration status, lack of social support, and violence and abuse within the family have been reported to increase the risk for peripartum depression [15,16,17], indicating the need for societal interventions in the management of women with peripartum depression. In contrast, a healthy diet, skin-to-skin care, high social support, and adequate partner support have been reported to protect against peripartum depression [16, 18, 19].
Few studies have investigated the prevalence of peripartum depression in Korea, and a large difference in the prevalence estimates is observed among the studies [13, 14]. Furthermore, studies investigating the factors associated with peripartum depression among a large number of Korean women at the population level are lacking. Therefore, in this study, we aimed to investigate the prevalence of women at risk for peripartum depression and associated factors among Korean women using data from the Seoul Healthy First Step Project (SHFSP), which targeted all pregnant women in Seoul. We hypothesized that women with low socioeconomic status, social disadvantages, and psychological problems would be at greater risks for peripartum depression.
Materials and methods
Study participants
We used the database from the SHFSP, which was launched by the Seoul metropolitan government in 2013 to support women with young children and to improve the health and development of babies [20]. In this program, registered nurses provide universal home visitation to all pregnant women during pregnancy or within 4 weeks after their delivery. For families who need continuing assistance, 25 or more sustained home visits are provided until the child reaches 2 years of age. At the time of registration, the data regarding the sociodemographic characteristics, psychosocial health status, and the parenting environment of pregnant women were collected using a standardized questionnaire. Therefore, the psychosocial health status was evaluated during the prepartum period for those who had been registered during pregnancy, while it was evaluated during the postpartum period for those who had been registered after delivery. The SHFSP initially took place in 3 districts of Seoul in 2013 and gradually expanded to all 25 districts in Seoul in 2020. In 2019, the database included 31.8% of all births in Korea [21]. More detailed information about the SHFSP has been reported previously [20]. The Institutional Review Board of Kangwon University approved the study, which analyzed the secondary data, excluding personal identifiers (KWNUIRB-2020-06-003). Among the 86,561 participants in the SHFSP database, we excluded 6,445 participants with missing responses for any of the survey items; thus, 80,116 participants were included in the final analysis (Fig. 2). The differences in the characteristics between the included and excluded participants are shown in Supplementary Table 2.
Measures
Definition of peripartum depression
Peripartum depression was evaluated using the Korean version of the Edinburgh Postnatal Depression Scale (EPDS) [22]. The EPDS consists of 10 items, including 8 items evaluating depressive symptoms and 2 items evaluating anxiety symptoms. The score for each item ranges from 0 to 3; thus, the total score ranges from 0 to 30, with a higher score indicating severe depressive symptoms. Initially, a threshold score of ≥ 13 was found to have a sensitivity of 86% and specificity of 78% in detecting depressed women [22]. However, a cutoff score of ≥ 10 has been proposed to reduce the failure to detect cases with depression to < 10% because it is important not to miss actual cases of peripartum depression considering its adverse impact on mothers and children [22]. In a Korean validation study, the cutoff points of 9/10 had sensitivity and specificity values of 76.7% and 87.1%, respectively, while the cutoff points of 12/13 had sensitivity and specificity values of 43.3% and 95.2%, respectively [23]. Therefore, we determined the participants to have peripartum depression when their EPDS score was ≥ 10.
Psychosocial health status
The psychosocial health status was evaluated by the following questions, as shown in Supplementary Table 1: “Do you have someone who can help with the baby?”, “Do you have someone to talk about your feelings and worries?”, “Have you ever experienced serious stress, change, or loss over the past 12 months?”, “Do you consider yourself confident?”, “Have you ever been treated for emotional issues?”, “Have you ever been physically, emotionally, or sexually abused in your childhood?”, and “Do you need any help for domestic violence?”. The participants provided responses of either “yes” or “no”.
Sociodemographic and lifestyle factors
The age of the participants was categorized into ≤ 19, 20–29, 30–39, and ≥ 40 years old. The economic status of the participants was categorized as either “low income” or “others”. The participants were asked whether they were basic livelihood security program recipients or from a secondary lower-income family, which refers to those whose recognized amount of income was below the 50/100 of the standard median income in Korea. Those who corresponded to these categories were classified as low income. Furthermore, the participants were asked whether they had any physical or mental disability. We also evaluated the presence of a husband/partner and the participants’ country of origin. Lifestyle factors, including alcohol consumption and smoking status were evaluated. Alcohol consumption was evaluated by the following question, “do you drink alcohol?”, and the smoking status was evaluated by the following question, “do you smoke currently?” For alcohol consumption and smoking behaviors, the participants provided responses of either “yes” or “no”.
Statistical analysis
We evaluated the cumulative prevalence of women at risk for depression according to the EPDS scores in all peripartum women as well as in prepartum and postpartum women, as well as the median and interquartile range (IQR) of the EPDS scores in peripartum, prepartum, and postpartum women. We compared the socioeconomic characteristics, psychosocial health status, and parenting environment of the study participants with and without depression using the chi-square test. A multivariable analysis was performed using a multiple logistic regression model to identify the factors associated with peripartum depression. In model 1, a crude analysis was performed to evaluate the association between each factor and peripartum depression. In model 2, a multivariable analysis was performed after adjusting for the timing of the EPDS measurement, maternal age, economic status, disability, single parent status, multicultural family status, questions evaluating the psychosocial health status, and lifestyle factors. A sensitivity analysis was performed with a definition of peripartum depression as an EPDS score ≥ 13. Furthermore, a stratified analysis was performed according to the timing of the EPDS measurement. All analyses were conducted with the IBM SPSS Statistics for Windows version 23.0 (IBM Corp., Armonk, NY, USA).
Results
Prevalence of women at risk for peripartum depression
Table 1 and Fig. 3 show the distribution of the EPDS scores in the 80,116 women. The prevalence of women at risk for peripartum depression was 17.7% in all women (prepartum depression: 14.2%, postpartum depression: 24.3%) when using a cutoff of ≥ 10 (Table 1). The median EPDS score in all peripartum women was 5 (IQR 3–8) (Fig. 1). The prevalence of women at risk for peripartum depression was 7.5% (prepartum depression: 5.6%, postpartum depression: 11.0%) with a cutoff of ≥ 13. The prevalence of women who have been treated for emotional issues in the study population was 3.9% (Table 2). The median EPDS score in women who have been treated for emotional issues was 9 (IQR 6–13), whereas the score in women without a history of treatment for emotional issues was 5 (IQR 3–8).
Comparison of the socioeconomic characteristics, psychosocial health status, and parenting environment between women with and without peripartum depression
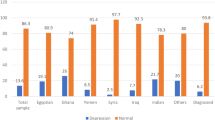
Table 2 shows that the prevalence of women at risk for peripartum depression was the highest among teenagers and the lowest among those aged 30–39 years and was higher in the low income group, those with a disability, single parents, and those from foreign countries than their counterparts. As for the psychosocial health status, the prevalence of women at risk for peripartum depression was higher in participants who had experienced stress, change, or loss over the past 12 months, had been treated for emotional issues, had any experience of being abused during childhood, and reported needing any help for domestic violence. In contrast, the prevalence of women at risk for peripartum depression was lower when participants had someone who could help with the baby, had someone to talk with, and considered themselves confident. The prevalence of women at risk for peripartum depression was higher among current drinkers and smokers.
Factors associated with peripartum depression with multivariable analysis
Table 3 presents the univariable and multivariable analyses of factors associated with maternal depression. In the univariable analysis, all the variables considered in this study were significantly associated with peripartum depression. At least fourfold differences in the odds of maternal depression were detected for most of the risk factors: 7.34 for smoking; 6.58 for needing help for domestic violence; 6.17 for being a single parent; 4.61 for having been treated for emotional issues; 4.37 for experiencing stress, change, or loss; 4.34 for experiencing child abuse; 4.31 for low income; and 4.18 for alcohol consumption. When an EPDS score of ≥ 13 was employed for defining peripartum depression, the magnitude of association measured by odds ratios (ORs) and 95% confidence intervals (CIs) was even greater (Supplementary Table 3). The univariable analysis results also showed that women who had someone who could help with the baby, had someone to talk with, and considered themselves confident were less likely to have low odds of having peripartum depression. For example, the OR for having someone to talk with was 0.12 (0.11–0.13).
In the multivariable model simultaneously adjusted for the timing of the EPDS measurement, maternal age, economic status, disability, single parent status, multicultural family status, questions evaluating the psychosocial health status, and lifestyle factors, the ORs for all variables except for the period of EPDS measurement and an age group of 20–29 years moved toward 1 (Table 3). The OR for the timing of the EPDS measurement was greater in multivariable analyses (OR 2.11, 95% CI 2.03–2.20) than the OR in the univariable analysis (1.95, 1.88–2.02). In the multivariable analysis, the OR was greater for the low income group (1.35, 1.11–1.64), those with a disability (1.43, 1.01–2.02), and for single parents (1.31, 1.04–1.65) than their counterparts. Compared with those aged ≥ 40 years, the odds for peripartum depression were higher among those aged 20–29 years (1.17, 1.05–1.31). Furthermore, the odds for peripartum depression increased when the participants had experienced stress, change, or loss over the past 12 months (3.36, 3.22–3.50), had been treated for emotional issues (2.47, 2.24–2.70), had experienced abuse during childhood (1.91, 1.74–2.11), and reported needing any help for domestic violence (2.25, 1.81–2.80). The odds decreased when participants had someone who could help with the baby (0.62, 0.58–0.67), had someone to talk with (0.31, 0.27–0.35), and considered themselves confident (0.30, 0.29–0.31). Furthermore, current smokers (2.02, 1.44–2.83) had greater odds of peripartum depression. The sensitivity analysis with an EPDS score ≥ 13 for defining peripartum depression produced similar results; however, the magnitude of association was relatively greater than that when an EPDS score ≥ 10 was used as the cutoff for defining depression (Supplementary Table 3).
The stratified analysis according to the timing of the EPDS measurement showed that the associated factors for prepartum depression remained consistent with the overall results (Supplementary Table 4). The associated factors for postpartum depression were similar in the univariable analysis; however, in the multivariable model, the associations of maternal age, economic status, disability, single parent status, multicultural family status, and lifestyle factors with postpartum depression were weakened (Supplementary Table 5). Overall, the association between psychosocial health status and postpartum depression was significant and consistent.
Discussion
The prevalence of women at risk for peripartum depression in Korean women was 17.7% and was higher during the postpartum period (24.3%) than the prepartum period (14.2%) when using an EPDS cutoff of ≥ 10. With a cutoff ≥ 13, the overall prevalence substantially reduced to 7.5% (prepartum depression: 5.6%, postpartum depression: 11.0%). Younger maternal age, low income, disability, being a single parent, being from a foreign country, and smoking increased the risk for peripartum depression. Furthermore, various aspects of psychosocial status and family dynamics were associated with peripartum depression. Specifically, psychological stress, a history of being treated for emotional issues, and experiences of child abuse and domestic violence increased the risk, whereas having someone who can provide help, having someone to talk with, and self-confidence decreased the risk. The magnitude of the association measured by ORs and 95% CIs was greater when a more conservative definition of maternal depression (EPDS ≥ 13) was used than when an EPDS cutoff of ≥ 10 was employed for defining depression.
Globally, the prevalence of prenatal depression has been reported to range from 15 to 65%, with generally higher rates reported in low- and middle-income countries than in high-income countries [24]. The pooled prevalence of postpartum depression among healthy mothers from a recent meta-analysis was 17%; significant differences were found with respect to geographical regions, with the Middle East having the highest prevalence and Europe having the lowest [25]. These reported prevalence rates may not be directly compared as studies have used different tools for diagnosing depression. One meta-analysis reported that the prevalence estimates evaluated by symptom scales was 1.6 times higher than those evaluated by diagnostic instruments [12], whereas another study reported that the prevalence of depression was similar regardless of the type of diagnostic tool used [25]. In our analysis, we evaluated the prevalence of women at risk for peripartum depression using the EPDS with a cutoff of ≥ 10 as recommended by a Korean validation study [23]. A sensitivity analysis with a cutoff of ≥ 13 led to a substantially lower prevalence of 7.5%. A cutoff ≥ 13 had a sensitivity of 43.3% in a Korean study, suggesting a large number of missing true cases of peripartum depression [23].
The prevalence of women at risk for depression was higher during the postpartum period than the prepartum period in this study. A recent multicenter study in Italy reported the prevalence of prepartum and postpartum depression to be 6.4% and 19.9%, respectively [26]. On the contrary, the prevalence of depression was higher during the prepartum period than the postpartum period in Australia and New Zealand [27, 28]. The symptoms of prepartum depression include diminishing capacity for self-care, such as inadequate nutrition, alcohol abuse, and poor antenatal clinic attendance as well as depressive mood and anxiety [29]. Therefore, prepartum depression may restrict the optimal fetal monitoring and normal growth of the fetus. Symptoms of postpartum depression include inappropriate parenting behaviors in addition to neglecting one’s well-being and physical health [30]. After delivery, women often experience high levels of stress from the delivery process and due to the new role of parents, which they are not used to. Furthermore, they experience significant environmental changes both in the work place and at home.
Younger maternal age, low income, disability, being a single parent, and smoking increased the risk for peripartum depression in this study. Prior studies have reported similar findings [31]. In general, a younger maternal age was associated with peripartum depression [32,33,34], and indicators of low socioeconomic status, including low education, low income, and unemployment, all independently increased the risk for peripartum depression [32, 35, 36]. Women with disabilities had a 1.6 times higher risk of experiencing symptoms of postpartum depression than healthy women in one study [37]. Furthermore, compared with married women, cohabitating women or single parents had a higher risk of postpartum depression [38]; however, one study reported that single women were not more likely to be depressed than married women after controlling for relationship quality [39]. Smoking was reported to increase the risk for peripartum depression among Korean women in previous studies [40, 41]. Diverse changes caused by cigarette smoking, such as elevated levels of pro-inflammatory cytokines, oxidative stress, and cortisol contribute to the development of depression [40, 42].
In this study, stress, change, or loss over the past 12 months, a history of treatment for emotional issues, and experiences of child abuse and domestic violence increased the risk for peripartum depression. Similarly, stressful life events, antenatal depression and anxiety, abuse (sexual, physical, or psychological), and domestic violence have been reported to be the predisposing factors of peripartum depression [43,44,45,46].
On the contrary, the risk for peripartum depression was reduced when women had helpers for their babies, had someone to talk to, and considered themselves confident. Low social support has long been known to increase the risk for maternal depression [16]. The recent studies found that increased social support could reduce the risk for maternal depression [47, 48]. In particular, receiving support for infant care was associated with lower depressive symptoms; additionally, support from family, friends, and special people were associated with lower odds for maternal depression [40]. Therefore, official government programs that send a person to each woman to help with her baby and simply to talk could eventually reduce peripartum depression. Furthermore, significant relationships of maternal confidence and maternal parental self-efficacy with postpartum depression were found [49, 50]. Thus, interventions promoting maternal self-confidence are needed to prevent peripartum depression.
In our analysis stratified by the timing of the EPDS measurement, younger maternal age, low income, disability, being a single parent, multicultural family status, alcohol consumption, and smoking increased the risk for prepartum depression; additionally, the psychosocial health status was associated with prepartum depression. On the other hand, the associations of sociodemographic and behavioral factors with postpartum depression were weakened, and only the psychosocial health status remained to be associated with postpartum depression. These results correspond to the psychosocial model of antenatal depression, postnatal depression, and parenting stress suggested by Leigh et al. [29]. In the prepartum period, predisposing factors such as young age, low income, and low education may directly influence the depressive symptoms. In the postpartum period, parenting stress interacts with depressive symptoms in addition to the existing antenatal predisposing factors [29]. Given that prepartum depression is a strong risk factor for postpartum depression, intervention at prepartum periods may substantially reduce the prevalence of both prepartum and postpartum depression. The treatment of depression as well as social support and appropriate education during pregnancy significantly lowered the levels of depressive symptoms and major depressive episodes in the postpartum period [51]. Therefore, early intervention during pregnancy is needed especially among women with social disadvantages, psychological problems, and experiences of child abuse and domestic violence.
Some countries are already providing home visiting programs during the prenatal and early childhood periods to support women with babies [52,53,54]. The SHFSP, which was initially launched in 3 districts of Seoul in 2013 and fully expanded to all 25 districts in 2020, may help reduce maternal depression by making mothers more self-confident and increasing their self-efficacy [9, 10]. Home visiting nurses in the SHFSP have referred women to a local suicide prevention center or to a mental health welfare center depending on the severity of depressive symptoms. Women with an EPDS score ≥ 13 or women with a self-harm score ≥ 1 and having the potency to harm themselves or their baby are referred to a suicide prevention center. In addition, women who do not correspond to the above categories but manifest depressive symptoms are referred to a mental health welfare center and are provided with emotional support and education.
There are several limitations in this study. First, it is not possible to derive cause-and-effect relationships between various factors and peripartum depression due to the cross-sectional design of the study. Second, the study participants are women who registered for the program voluntarily and do not represent all pregnant women of Seoul. Third, recall bias might have influenced the classifications of each variable, as most variables in this study were derived from survey responses. Fourth, we excluded 6445 participants from the original 86,561 participants in the SHFSP database. As differences in the socioeconomic and psychosocial characteristics between the included and excluded participants exist (Supplementary Table 2), it may have influenced the results. Despite these limitations, this study included a sufficiently large number of participants from the community to analyze the factors associated with peripartum depression.
In conclusion, peripartum depression is quite prevalent among Korean women, especially those with social disadvantages, psychological problems, and previous experiences of child abuse and domestic violence. Since we found a lower risk of depression among women, who had someone to get help from or talk with, and among those who had confidence, a home visitation program, like the SFHSP could be a good method to help prevent or alleviate maternal depression.
Availability of data and material
The data are available upon request from the corresponding author.
References
Hoffman DJ, Reynolds RM, Hardy DB (2017) Developmental origins of health and disease: current knowledge and potential mechanisms. Nutr Rev 75(12):951–970
Campbell F, Conti G, Heckman JJ et al (2014) Early childhood investments substantially boost adult health. Science 343(6178):1478–1485
Shonkoff JP, Boyce WT, McEwen BS (2009) Neuroscience, molecular biology, and the childhood roots of health disparities: building a new framework for health promotion and disease prevention. JAMA 301(21):2252–2259
McManus BM, Poehlmann J (2012) Maternal depression and perceived social support as predictors of cognitive function trajectories during the first 3 years of life for preterm infants in Wisconsin. Child Care Health Dev 38(3):425–434
McManus BM, Poehlmann J (2012) Parent–child interaction, maternal depressive symptoms and preterm infant cognitive function. Infant Behav Dev 35(3):489–498
Deave T, Heron J, Evans J, Emond A (2008) The impact of maternal depression in pregnancy on early child development. BJOG 115(8):1043–1051
Jarde A, Morais M, Kingston D et al (2016) Neonatal outcomes in women with untreated antenatal depression compared with women without depression: a systematic review and meta-analysis. JAMA Psychiat 73(8):826–837
Hans SL, Edwards RC, Zhang Y (2018) Randomized controlled trial of Doula-home-visiting services: impact on maternal and infant health. Matern Child Health J 22(Suppl 1):105–113
Sandner M, Cornelissen T, Jungmann T, Herrmann P (2018) Evaluating the effects of a targeted home visiting program on maternal and child health outcomes. J Health Econ 58:269–283
Tandon D, Mackrain M, Beeber L, Topping-Tailby N, Raska M, Arbour M (2020) Addressing maternal depression in home visiting: Findings from the home visiting collaborative improvement and innovation network. PLOS ONE 15(4):e0230211
Abdollahi F, Lye MS, Zarghami M (2016) Perspective of postpartum depression theories: a narrative literature review. N Am J Med Sci 8(6):232–236
Woody CA, Ferrari AJ, Siskind DJ, Whiteford HA, Harris MG (2017) A systematic review and meta-regression of the prevalence and incidence of perinatal depression. J Affect Disord 219:86–92
Park JH, Karmaus W, Zhang H (2015) Prevalence of and risk factors for depressive symptoms in Korean women throughout pregnancy and in postpartum period. Asian Nurs Res 9(3):219–225
Youn H, Lee S, Han SW et al (2017) Obstetric risk factors for depression during the postpartum period in South Korea: a nationwide study. J Psychosom Res 102:15–20
Biaggi A, Conroy S, Pawlby S, Pariante CM (2016) Identifying the women at risk of antenatal anxiety and depression: a systematic review. J Affect Disord 191:62–77
Zhao XH, Zhang ZH (2020) Risk factors for postpartum depression: An evidence-based systematic review of systematic reviews and meta-analyses. Asian J Psychiatr 53:102353
Gelaye B, Rondon MB, Araya R, Williams MA (2016) Epidemiology of maternal depression, risk factors, and child outcomes in low-income and middle-income countries. Lancet Psychiatry 3(10):973–982
Milgrom J, Hirshler Y, Reece J, Holt C, Gemmill AW (2019) Social support-a protective factor for depressed perinatal women? Int J Environ Res Public Health 16(8):1426
Pilkington PD, Milne LC, Cairns KE, Lewis J, Whelan TA (2015) Modifiable partner factors associated with perinatal depression and anxiety: a systematic review and meta-analysis. J Affect Disord 178:165–180
Khang YH, Cho SH, June KJ, Lee JY, Kim YM, Cho HJ (2018) The Seoul Healthy First Step Project: introduction and expansion, program content and performance, and future challenges. J Korean Soc Matern Child Health 22(2):63–76
Lee JY, Khang YH, June KJ, Cho SH, Cho HJ, Kim YM (2020) Report for the Seoul healthy fist step project. Seoul Health Foundation, Seoul
Cox JL, Holden JM, Sagovsky R (1987) Detection of postnatal depression. Development of the 10-item Edinburgh postnatal depression scale. Br J Psychiatry 150:782–786
Kim YK, Hur JW, Kim KH, Oh KS, Shin YC (2008) Clinical application of Korean version of Edinburgh postnatal depression scale. J Korean Neuropsychiatr Assoc 47(1):36–44
Dadi AF, Miller ER, Bisetegn TA, Mwanri L (2020) Global burden of antenatal depression and its association with adverse birth outcomes: an umbrella review. BMC Public Health 20(1):173
Shorey S, Chee CYI, Ng ED, Chan YH, Tam WWS, Chong YS (2018) Prevalence and incidence of postpartum depression among healthy mothers: a systematic review and meta-analysis. J Psychiatr Res 104:235–248
Cena L, Mirabella F, Palumbo G, Gigantesco A, Trainini A, Stefana A (2021) Prevalence of maternal antenatal and postnatal depression and their association with sociodemographic and socioeconomic factors: a multicentre study in Italy. J Affect Disord 279:217–221
Ogbo FA, Eastwood J, Hendry A et al (2018) Determinants of antenatal depression and postnatal depression in Australia. BMC Psychiatry 18(1):49
Underwood L, Waldie KE, D’Souza S, Peterson ER, Morton SMB (2017) A longitudinal study of pre-pregnancy and pregnancy risk factors associated with antenatal and postnatal symptoms of depression: evidence from growing up in New Zealand. Matern Child Health J 21(4):915–931
Leigh B, Milgrom J (2008) Risk factors for antenatal depression, postnatal depression and parenting stress. BMC Psychiatry 8:24
Stewart DE, Vigod SN (2019) Postpartum depression: pathophysiology, treatment, and emerging therapeutics. Annu Rev Med 70:183–196
Norhayati MN, Hazlina NHN, Asrenee AR, Emilin WMAW (2015) Magnitude and risk factors for postpartum symptoms: a literature review. J Affect Disord 175:34–52
Kozinszky Z, Dudas RB, Csatordai S et al (2011) Social dynamics of postpartum depression: a population-based screening in South-Eastern Hungary. Soc Psychiatry Psychiatr Epidemiol 46(5):413–423
Quelopana AM, Champion JD, Reyes-Rubilar T (2011) Factors associated with postpartum depression in Chilean women. Health Care Women Int 32(10):939–949
Sword W, Landy CK, Thabane L et al (2011) Is mode of delivery associated with postpartum depression at 6 weeks: a prospective cohort study. BJOC 118(8):966–977
Chien LY, Tai CJ, Ko YL, Huang CH, Sheu SJ (2006) Adherence to “Doing-the-month” practices is associated with fewer physical and depressive symptoms among postpartum women in Taiwan. Res Nurs Health 29(5):374–383
Rubertsson C, Wickberg B, Gustavsson P, Rådestad I (2005) Depressive symptoms in early pregnancy, two months and one year postpartum-prevalence and psychosocial risk factors in a national Swedish sample. Arch Womens Ment Health 8(2):97–104
Mitra M, Lezzoni LI, Zhang J, Long-Bellil LM, Smeltzer SC, Barton BA (2015) Prevalence and risk factors for postpartum depression symptoms among women with disabilities. Matern Child Health J 19(2):362–372
Kiernan K, Pickett KE (2006) Marital status disparities in maternal smoking during pregnancy, breastfeeding and maternal depression. Soc Sci Med 63(2):335–346
Akincigil AS, Munch S, Niemczyk KC (2010) Predictors of maternal depression in the first year postpartum: marital status and mediating role of relationship quality. Soc Work Health Care 49(3):227–244
Cui M, Kimura T, Ikehara S et al (2020) Prenatal tobacco smoking is associated with postpartum depression in Japanese pregnant women: the Japan environment and children’s study. J Affect Disord 264:76–81
Pooler J, Perry DF, Ghandour RM (2013) Prevalence and risk factors for postpartum depressive symptoms among women enrolled in WIC. Matern Child Health J 17(10):1969–1980
Yanbaeva DG, Dentener MA, Creutzberg EC, Wesseling G, Wouters EFM (2007) Systemic effects of smoking. Chest 131(5):1557–1566
Gaillard A, Strat YL, Mandelbrot L, Keïta H, Dubertret C (2014) Predictors of postpartum depression: prospective study of 264 women followed during pregnancy and postpartum. Psychiatry Res 215(2):341–346
Escribà-Agüir V, Artazcoz L (2011) Gender differences in postpartum depression: a longitudinal cohort study. J Epidemiol Community Health 65(4):320–326
Silverman ME, Loudon H (2010) Antenatal reports of pre-pregnancy abuse is associated with symptoms of depression in the postpartum period. Arch Womens Ment Health 13(5):411–415
Dennis CL, Vigod S (2013) The relationship between postpartum depression, domestic violence, childhood violence, and substance use: epidemiologic study of a large community sample. Violence Against Women 19(4):503–517
Li Y, Long Z, Cao D, Cao F (2017) Social support and depression across the perinatal period: a longitudinal study. J Clin Nurs 26(17–18):2776–2783
Tambag H, Turan Z, Tolun S, Can R (2018) Perceived social support and depression levels of women in the postpartum period in Hatay. Turkey Niger J Clin Pract 21(11):1525–1530
Arante FO, Tabb KM, Wang Y, Faisal-Cury A (2020) The Relationship between postpartum depression and lower maternal confidence in mothers with a history of depression during pregnancy. Psychiatr Q 91(1):21–30
Leahy-Warren P, McCarthy G, Corcoran P (2012) First-time mothers: social support, maternal parental self-efficacy and postnatal depression. J Clin Nurs 21(3–4):388–397
Sockol LE, Epperson CN, Barber JP (2013) Preventing postpartum depression: a meta-analytic review. Clin Psychol Rev 33(8):1205–1217
Health Resources & Services Administration (2021) The maternal, infant, and early childhood home visiting program. https://mchb.hrsa.gov/maternal-child-health-initiatives/home-visiting-overview. Accessed 15 May 2021
National Health Service (2021) Family nurse partnership. https://fnp.nhs.uk/. Accessed 15 May 2021
Australian Government Department of Health (2020) Pregnancy, birth and baby. https://about.healthdirect.gov.au/pregnancy-birth-and-baby. Accessed 15 May 2021
Funding
This work was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant Number: HI19C0481, HC19C0048).
Author information
Authors and Affiliations
Contributions
H-JC: conceptualization, methodology, validation, writing—review and editing, supervision, SYK: methodology, formal analysis, writing—original draft, writing—review and editing, visualization, Y-HK: methodology, validation, writing—reviewing and editing, funding acquisition, KJJ: methodology, validation, writing—reviewing and editing, S-HC: methodology, validation, writing—reviewing and editing, JYL: methodology, validation, writing—reviewing and editing, Y-MK: methodology, validation, writing—reviewing and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kang, S.Y., Khang, YH., June, K.J. et al. Prevalence and risk factors of maternal depression among women who participated in a home visitation program in South Korea. Soc Psychiatry Psychiatr Epidemiol 57, 1167–1178 (2022). https://doi.org/10.1007/s00127-022-02226-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00127-022-02226-w