Abstract
Thermal modification can change the color and improve the dimensional stability and decay resistance of wood without the use of chemical modifiers. However, the impact of extracts from thermally modified wood (TMW) on the performance of TMW remains obscured. To achieve high-value utilization of TMW extracts and to regulate the properties of the TMW through extracts, the impacts of TMW acetone extracts on the color, dimensional stability, and decay resistance of TMW were investigated. The 180, 200 and 220 °C thermally modified spruce (Picea asperata Mast.) and poplar (Populus tomentosa Carr.) with or without acetone extract were used for CIELab color system, anti-swelling efficiency (ASE), and decay resistance assessments. Fourier infrared spectroscopy (FTIR) and X-ray photoelectron spectroscopy (XPS) were used to characterize extracted and unextracted TMW. The results indicated that the extract showed positive effects on the color, dimensional stability (220 °C), and decay resistance of TMW, and this effect weakened with increasing temperature. The removal of the extract resulted in a reduction in brightness difference (∆L*) and total color difference (∆E*) of the TMW, with the most pronounced impact observed at 180 °C. The removal of the extract enhanced the dimensional stability of the TMW at 180 and 200 °C but decreased it at 220 °C. The decay resistance of wood thermally modified at 180, 200, and 220 C decreased after extraction, and was most significant at 180 °C. The antifungal activity of the extract was related to the production of vanillin, isovanillic acid, syringic acid, and syringaldehyde. Extract from TMW played an important role in the properties of TMW, serving as a potential means for adjusting its properties or as a natural wood modifier.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The dimensional instability and biodegradability of wood limit its application (Spear et al. 2021). The dimensional stability of wood can be significantly improved by thermal modification, which is widely used in Europe and other regions (Dubey et al. 2011). In addition, the decay resistance (Chaouch et al. 2010; Tripathi et al. 2014) and surface color (Li et al. 2018; Nguyen et al. 2018) of wood can also be improved by heating with simple equipment, especially the sapwood of some species with poor decay resistance and color. Regrettably, the mechanical strength of wood decreased due to thermal modification and became significant with increasing temperature (Korkut et al. 2008). The modulus of rupture (MOR) and modulus of elasticity (MOE) of wood treated at 200 °C for 24 h decreased by 44-50% and 4–9% respectively (Bekhta and Niemz, 2003). Some studies indicated that extracts from thermally modified wood (TMW) showed a positive effect on its decay resistance and color (Esteves et al. 2008; Sandoval-Torres et al. 2010). In previous studies, it has been confirmed that the extract of TMW can be used to improve decay resistance and change color of wood without reducing its mechanical strength (Bi et al. 2022).
The degradation of cellulose and hemicellulose during thermal modification has generally been considered a prime key for improving decay resistance and reducing hygroscopicity (Candelier et al. 2016). The degradation of cellulose, hemicellulose, and lignin which are the nutritional sources of wood decay fungi, results in a reduction of nutritional and hydrophilic hydroxyl groups (Costa et al. 2019; Hill et al. 2021). The reduction of hygroscopicity impeded expansion induced by increased moisture, and is not conducive to the growth of fungi and the transportation of small molecular substances that degrade the cell wall, thus improving the dimensional stability and decay resistance of wood (Sivrikaya et al. 2015). However, some researchers disputed the significant correlation between wood hygroscopicity and decay resistance (Hakkou et al. 2006), and the decay resistance of TMW is also affected by pseudo-lignin and small molecular anti-fungal active compounds (Jiang et al. 2022).
Furthermore, the new extracts with antifungal activity produced by thermal modification play an important role in improving decay resistance (Chu et al. 2019). Kamdem et al. (2002) subjected maritime pine (Pinus pinaster Aiton), spruce, and poplar wood to thermal modification, followed by water or chloroform extraction. That study indicated that the decay resistance of this wood improved by heating, while its decay resistance reduced after water or chloroform extraction. In addition, Candelier et al. (2020) found that acetone extract of thermally modified ash (Fraxinus excelsior L.) wood showed antifungal activity against wood decay fungi. Isovanillic acid, vanillin, and other compounds in these extracts inhibited the growth of wood decay fungi (Candelier et al. 2020; Bi et al. 2022). Vanillin has been proven to inhibit enzyme activity, and damage cell membranes, ATP, and other physiological and biochemical indicators of wood decay fungi (Bi et al. 2021a). This indicated the utilization of the extract from thermally modified wood (TMW) can improve the decay resistance of wood without mechanical loss.
Color is one of the main physical properties of wood. The chromophores and (or) auxochromes of hemicellulose, lignin, and extract changed after heating, resulting in the color change of TMW (Qu et al. 2021). The wood extract rich in compounds with unsaturated conjugated structures, such as tannin, phenol, stilbene, and quinone, exhibited susceptibility to change upon heating (Mayer et al. 2006; Zhu et al. 2021). The new extracts produced by the degradation of the original wood extract, hemicellulose, and lignin during the thermal modification process led to the change of chromogenic groups and auxochrome groups, thereby changing the color of the wood (Chen et al. 2012; Gao et al. 2015). Furthermore, the study indicated that the extractives can hinder the degradation of thermally modified Scots pine (Pinus sylvestris L.) during weathering (Shen et al. 2016).
However, the effect of extracts on color, dimensional stability, and decay resistance of TMW has not garnered sufficient attention, warranting further research. The objective of this study was to investigate the influence of thermally modified wood extracts (air atmosphere) on their color, dimensional, and decay resistance. This study can provide a theoretical basis for utilizing extracts to modulate the color, dimensional stability, and decay resistance of TMW. Additionally, the processing residues of TMW can be reused, providing novel materials for plant-derived wood modifiers.
2 Materials and methods
2.1 Materials
Wood preparation: Poplar (Populus tomentosa Carr.) and spruce (Picea asperata Mast.) trees were selected from plantations at Northwest A&F University in the Yangling District, Shaanxi Province, China. Clean, defect-free blocks measuring 20 mm × 20 mm × 10 mm (R × T × L) and 20 mm × 20 mm × 20 mm (R × T × L) were cut from 45 cm diameter sapwood (strong permeability and poor durability) of poplar and spruce wood. The lumber was air-dried and then small shavings were cut from the lumber and ground to pass a 40-mesh screen. The wood blocks and ground wood were oven-dried at 60 °C to 8% moisture content and weighed (nearest 0.01 g).
The white-rot fungus, Trametes versicolor (L. ex Fr.) Quél. (Isolate # CFCC5336) and brown-rot fungus, Gloeophyllum trabeum (Pers.: Fr.) Murr. (Isolate # CFCC86617) were grown on 3.7% potato dextrose agar (PDA) at 28 ± 2 °C and 80 ± 5% relative humidity (RH) for 5–7 days prior to use.
2.2 Preparation of thermally modified wood and extraction
In our previous study, acetone extract with the strongest antifungal activity had a significant effect on the color of thermally modified poplar wood, and it was chosen as the extraction solvent (Bi et al. 2021b, 2022). The poplar wood blocks, spruce wood blocks, or ground wood with constant weight were respectively put into the oven with the side and top vents open, and thermally treated at 180 °C, 200 °C, or 220 °C for 6 h. Subsequently, the samples were cooled to 60 °C and the resulting thermally modified wood (TMW) was stored in a desiccator. Twelve pieces of thermally modified poplar (TMP) and spruce (TMS) wood were placed in the extraction tube of the Soxhlet extractor, and 1000 mL of acetone was poured into a round bottom flask of the Soxhlet extractor. The assembled Soxhlet extractor was immersed in a water bath and heated to produce four reflux changes per hour. The extracted TMW samples were obtained after 72 h of maintenance.
2.3 Color measurement
The color of wood was deepened by heating, being an eco-friendly and effective way to change the color of fast-growing wood species with poor color. The thermal degradation of the cellulose, hemicellulose, lignin, and original wood extracts produced new compounds, and the impact on the color of TMW was explored in this study.
The color coordinates (L*, a*, b*) of the 20 mm × 20 mm × 20 mm (R × T × L) TMP and TMS, with or without acetone extraction, were measured by the CIELab system with a CHNspec CS-802 Spectrophotometer (Hangzhou CHNspec Technology Co., Ltd., Hangzhou, Zhejiang, China) equipped with D65 light source (Bi et al. 2021b; Vuthiganond et al. 2020). The samples were measured at a 10-degree observation angle with a 15 mm aperture at the point of measurement. Each treatment was repeated three times. The color coordinates were measured at three locations on each sample, and the mean value was calculated. The total color difference (∆E*) was calculated according to Eq. 1.
where ∆L*, ∆a*, and ∆b* are the differences of L*, a*, and b* between non-extracted and extracted samples, respectively. The positive values of ∆L*, ∆a*, and ∆b* indicate that the color of the sample after extraction tended to be brighter, redder, or yellower than before treatment. The negative values indicate darker, greener, or bluer.
2.4 Dimensional stability test of extracted and unextracted TMW
2.4.1 Anti-swelling efficiency of extracted and unextracted TMW
Twelve pieces of 20 mm × 20 mm × 20 mm extracted or unextracted TMP and TMS were placed in a temperature-humidity chamber at 22 °C and allowed to stand for 4 days at 65%, 75%, 85%, 75%, and 65% relative humidity. Sample size and mass were measured after each humidity balance. The hygroscopicity of wood samples under various humidity conditions was calculated according to Eq. 2, and the hygroscopicity analysis curve was drawn. The moisture absorption (MA) of the moisture absorption-desorption cycle and anti-swelling efficiency (ASE) of absorption were calculated according to Eqs. 3–4, and the results were averaged.
where MA is moisture absorption of wood, %; Ma is the mass of the sample before each moisture balance, g; Mb is the mass of the sample after each moisture balance, g. ∆V is the change rate of wood volume,%; Va is the volume of sample before each moisture balance, mm3; Vb is the volume of sample after each moisture balance, mm3; ASE is anti-swelling efficiency of samples, %; ∆V0 is the change rate of original wood volume,%; ∆V1 is the change rate of treated wood volume, %.
2.4.2 Water contact angle of extracted and unextracted TMW
The SDC-100 contact angle meter was used to measure the water contact angle (°) of extracted or unextracted TMW. The samples were placed at 22 °C and 65% relative humidity for one week to attain a constant weight before testing. A 5 µL drop of distilled water was deposited on the cross-section of the wood sample using the sessile drop method and the water contact angle was recorded at 60 s. The test was repeated three times for each treatment. Each treatment was repeated three times.
2.5 Decay resistance test of extracted and unextracted TMW
The decay resistance test referenced the XP CEN/TS 15083-1 (2006) standard with slight modifications. Decay tests were performed in Petri dishes (Bi et al. 2022). The potato dextrose agar (PDA) medium, sterilized in a 121 °C autoclave for 30 min, was poured into a 90 mm glass Petri dish, and cooled to a solid state. The 4.4 mm disk was cut from the active edge of the activated white-rot fungi (T. versicolor) and brown-rot fungi (G. trabeum) colonies and placed at the center of the PDA medium. These Petri dishes were then incubated in darkness at 28 °C and 80% relative humidity for 7 days. The 20 mm × 20 mm × 10 mm (R × T × L) extracted or unextracted TMW was wrapped in sterile gauze and sterilized in an autoclave at 105 °C for 30 min. Sterilized samples were placed on a medium full of mycelium and incubated in the dark at 80% RH and 28 °C for 15 weeks. The mycelium was scraped off the blocks and oven dried (60 °C) and weighed. Each treatment was replicated on eight wood samples per fungus. The mass loss rate (ML) was calculated according to Eq. (5).
where m1 is the mass of the wood block before fungal exposure, m2 is the mass of the wood block after fungal exposure.
2.6 Antifungal activity test of TMW extract
The inhibitory effect of acetone extracts from wood thermally modified at 180 °C, 200 °C, and 220 °C on the growth of wood decay fungi was used to investigate the effect of TMW extracts on the decay resistance of TMW. The acetone extracts of TMP and TMS were prepared by ultrasound-assisted solvent extraction according to the previously described method (Bi et al. 2022). TMP or TMS extracts were dissolved in anhydrous ethanol to prepare a 300 mg/mL stock solution. 107 µL, 213 µL or 427 µL of the stock solution was mixed with 7920 µL, 7814 µL or 7624 µL of sterilized PDA medium (80 °C) to prepare extract-containing media at concentrations of 4 mg/mL, 8 mg/mL, and 16 mg/mL respectively. The 4.4 mm disks were placed at the center of the PDA medium and incubated in darkness at 28 °C and 80% RH for 14 days. Each group was assessed on three plates per fungus. Colony diameter (mm) was measured and photographed on the 2nd, 4th, 6th, 8th, 14th, and 21st days.
2.7 Chemical characterization
2.7.1 FTIR analysis
The extracted or unextracted thermally modified wood meals were placed in an oven at 105 °C for 6 h to remove moisture. Those ground wood samples were subjected to FTIR analysis using the KBr plate method on a Bruker Vertex 70 FTIR Spectrometer (Bruker Optics Ltd, Coventry, UK) The KBr plates were scanned 32 times at a resolution of 4 cm− 1 in the spectral region 4,000-400 cm− 1.
2.7.2 XPS analysis
Slices measuring 10 mm × 1 mm × 10 mm (R × T × L) were cut from the radial faces of extracted or unextracted TMW. XPS analyses were carried out with a Thermo Escalab 250XI spectrometer (Thermo Fisher Scientific, Massachusetts, USA). 284.6 eV was used as an internal reference to correct the binding energies (Yan et al. 2021). The carbon spectrum (C1s) was de-convoluted into C1, C2, C3, and C4 peaks corresponding to C-C/C-C/C-H, C-O/C-O-C, -C-O/O-C-O, and O-C-O/C(= O)OH with binding energies (BE) of 284.6 eV, 286.4 eV, 287.8 eV, and 289.2 eV, respectively (Acheampong et al. 2018; Inari et al. 2011).
2.8 GC-MS analysis of extracts
The acetone extract of 180 °C, 200 °C, and 220 °C TMW (TMP and TMS) was dissolved in methanol (GC) to prepare sample solutions of 1 mg/mL. These sample solutions were then filtered through a 0.22 μm organic nylon filter at least three times to remove any particulates. The resulting filtrate was analyzed on a GCMS-QP2010 GC-MS spectrometer (Shimadzu Corporation, Kyoto, Japan) with a TG-5MS fused silica column (30 m × 0.25 mm id., film thickness 0.25 μm). The technical parameters were referenced to previous reports and their mass spectrum were identified using the National Institute of Standards and Technology library database (NIST08s. LIB). The top ten compounds in content were listed, and the results were only for reference.
2.9 Statistical analysis
The results were analyzed using a one-way ANOVA (P = 0.05) using the SPSS Statistics 22.0 software program. Results were expressed as mean ± SD, significant difference P < 0.05 (*), and highly significant difference P < 0.01 (**).
3 Results and discussion
3.1 Effects of extract on the color of thermally modified wood
The color of both poplar and spruce wood underwent significant changes following thermal modification, and the total color difference (∆E*) increased with rising treatment temperatures, reaching 53.7 and 49.6 at 220 °C (Figs. 1 and 2). The color of poplar and spruce wood tended to be darker, redder, and yellower after thermal modification at 180 °C, while the color tended to be darker, redder, and bluer after thermal modification at 200 and 220 °C. However, the color of spruce was different from poplar and tended to be greener after thermal modification at 220 °C. The effect of thermal modification on wood color has been extensively studied, and its color change is highly dependent on the wood properties, and also on the intensity of treatment (Zhan et al. 2022). However, the study on the effect of TMW extract on its color was insufficient.
Lightness difference ΔL* (a), red-green axis chromaticity index difference Δa* (b), yellow-blue axis chromaticity index difference Δb* (c), and the total color difference ΔE* (d) of extracted/unextracted poplar wood thermally modified at 180 °C, 200 and 220 °C. Note: ns means no significant difference, * means significant difference (P < 0.05), and ** means extremely significant difference (P < 0.01)
The effect of TMP or TMS acetone extracts on the color of these two thermally modified woods exhibited similarity, with a more pronounced effect observed in spruce wood (Fig. 2) compared to poplar wood (Fig. 1). The effect of extracts on ∆a* and ∆b* of TMW was not significant and lacked a consistent pattern. The ∆L* and ∆E* of TMP and TMS wood were reduced after the removal of the extract, and this effect was significant for wood thermally modified at 180 °C. Additionally, this effect diminished with increasing treatment temperatures. This indicated that the extract positively influenced the deepening of TMW color, particularly significant at 180 °C temperatures. Hemicellulose and lignin of wood underwent slight degradation among the three major components of wood at 180 °C, exerting a slight impact on wood color (Mehrotra et al. 2010; Ling et al. 2016). At this time, changes in the extract significantly influenced the color of TMW. It is reported that 180 °C was a critical temperature for the color changes of TMW, the color change was related to the degradation of hemicellulose and the increase in relative lignin content at that temperature (Zhan et al. 2022; González-Peña and Hale 2009). Severe degradation of hemicellulose at temperatures above 200 °C led to an increase in the content of lignin containing many chromogenic and auxochrome groups, a primary cause for wood discoloration (Huang et al. 2010). At this point, the impact of the extract was not significant.
3.2 Effects of extract on the dimensional stability of thermally modified wood
3.2.1 Analysis of water contact angle and moisture absorption of extracted or unextracted TMW
The water contact angle (°) of the wood cross-section significantly increased after thermal modification at 180 °C, 200 °C, and 220 °C (Fig. 3). The water contact angles of TMP and TMS increased from 73.9° or 50.2° to over 100° at 60 s. The extracts had no significant effect on the hydrophobic angle of TMP, and slight differences at different temperatures might be due to experimental errors.
The moisture absorption of poplar and spruce wood decreased after thermal modification under 180 °C, 200 and 220 °C, particularly noticeable at 200 and 220 °C (Fig. 4). The average moisture absorption of TMP and TMS samples at 220 °C during the moisture absorption analysis cycle decreased from 10.8% to 10.9% (original wood) to 6.9% and 7.4%, respectively. The moisture absorption of TMP and TMS gradually reduced with the increase of thermal modification temperature, indicating that thermal modification can reduce the moisture absorption of wood, regardless of extract removal. This was consistent with previous reports (Metsa-Kortelainen et al. 2006).
The hygroscopicity of poplar or spruce wood thermally modified at 180 and 200 °C decreased with the extract removal, while the hygroscopicity of 220 °C TMP or TMS was increased by the removal of extractives. The effect of extracts on the moisture absorption of TMS (P < 0.05) was more significant than that of TMP (P˃0.05), and the hygroscopicity of the 180 and 200 °C TMS after removal of the extract decreased from 10.1 to 8.9% and 8.6–7.4%, respectively (Fig. 4d and e). This might be due to the different reactions in the thermal modification process caused by the different hemicellulose, lignin, and extracts of spruce and poplar wood. For example, the mass loss after thermal modification and acetone extract yield of 180 °C TMS (1.8% and 3.1%) were higher than those of TMP (1.2% and 2.8%), while acetone is a highly polar solvent and its extract is hydrophilic (Smeds et al. 2021; Korenman et al. 2010). The lower moisture absorption of TWS might be due to the removal of more acetone extracts with hydrophilic. Hill et al. (2021) proposed that the extract produced by thermal modification under oxygen conditions containing water adsorption sites can lead to an increase in wood hygroscopicity. The decrease in hygroscopicity of 180 or 200 °C TMW after acetone extraction might be attributed to the removal of hydrophilic acetone extract, leading to a decrease in water adsorption sites. The analysis results of FTIR and XPS in Sect. 3.3 can support this conclusion to some extent.
The results indicated that the removal of 180 or 200 °C TMS extracts would reduce the hygroscopicity of the TMS. The spruce thermally modified at lower temperatures (180 and 200 °C) can achieve the same hygroscopicity as the spruce thermally modified at higher temperatures by removing the extract.
3.2.2 Analysis of anti-swelling efficiency of extracted or unextracted TMW
The anti-swelling efficiency (ASE) served as an indicator of the wood’s capacity to resist deformation in high humidity environments, and a larger value indicated better dimensional stability. The average ASE of poplar wood can be improved through thermal modification at 180 °C, 200 °C, and 220 °C (Fig. 5a), while the ASE of spruce wood can be improved by thermal modification at 200 and 220 °C (Fig. 5b). The ASE of wood thermally modified at 180 and 200 °C was improved after removing its extract, while the ASE of wood thermally modified at 220 °C was reduced after its extract removal (P < 0.05). This result was consistent with the conclusion of hygroscopicity and might be related to the different reactions of the main components of wood at different temperatures. As mentioned in the hygroscopicity section, the removal of 180 and 200 °C TMW hygroscopic extracts led to reduced hygroscopicity and additional space, which may be the possible reason for the improvement of ASE. The decrease in ASE of 220 °C TMW after extraction might be attributed to the exposure of more free hydroxyl groups. Källbom et al. (2018) thermally modified Scots pine wood according to the ThermoWood® D process (with a peak temperature of 212 °C) and extracted it with water, acetone, and ethanol/toluene. The results indicated a decreased swelling rate of TMW after extraction, suggesting that cell wall disruption at this temperature, coupled with extract removal, created additional space related to new adsorption sites within the cell wall. The relative content of free hydroxyl groups in TMW and its acetone extracts at different temperatures was investigated using FTIR and XPS to elucidate this result.
3.3 FTIR and XPS analysis of the extracted or unextracted TMW
3.3.1 FTIR analysis of the extracted or unextracted TMW
FTIR and XPS were used to analyze the relative content of free hydroxyl in TMW with or without extraction at different temperatures. The characteristic peak intensities of 180 °C, 200 and 220 °C TMW decreased after extraction, especially for 220 °C (Fig. 6). This indicated the degradation of lignin and hemicellulose and the extraction of degradation products. The absorption peak at 3670 − 3230 cm− 1 was assigned to the stretching vibration of hydroxyl groups, and the narrowing of the peak or shift towards a lower wavenumber indicated an increase in free hydroxyl groups (Gadhave et al. 2019). The hydroxyl stretching vibration peaks of 180 °C TMP (3401 cm− 1) and TMS (3435 cm− 1) after extract removal, shifted towards lower wavenumbers (3381 cm− 1 and 3389 cm− 1). This indicated that the removal of the extract at this temperature reduced the free hydroxyl content of the TMW, resulting in decreased hygroscopicity and increased ASE. It was speculated that the acetone extract might have stronger hydrophilicity, and the removal of the extract led to a reduction in the free hydroxyl content of TMW. There was no significant change in the hydroxyl stretching vibration peaks of 200 °C TMW, and it was consistent with the conclusion that the moisture absorption and ASE changes of TMW at 200 °C were minimal after extract removal.
The hydroxyl stretching vibration peaks of 220 °C TMP (3367 cm-1) and TMS (3348 cm-1) shifted towards high wavenumbers (3397 cm-1 and 3356 cm-1) with the removal of the extract, indicating that the free hydroxyl content of TMW increased due to the removal of the extract at this temperature. This was one of the reasons for the increase in moisture absorption and decrease of ASE of 220 °C TMP after removing the extract. This might be due to the intense degradation of hemicellulose and the slight degradation of lignin at 220 °C, and more free hydroxyl groups were exposed after the removal of the extract.
3.3.2 XPS analysis of the extracted or unextracted TMS
The removal of extracts significantly impacted the 180 and 220 °C TMS, and XPS was used to analyze the chemical composition of its surface (Fig. S1 and Table 1). The carbon spectrum (C1s) was de-convoluted into C1, C2, C3, and C4 peaks with ∑X2 less than 40 (Fig. S1). The C1, C2, C3, and C4 peaks corresponded to C-C/C = C/C-H, C-O/C-O-C, -C = O/O-C-O, and O-C = O/C(= O)-OH with binding energies (BE) of 284.6 eV, 286.4 eV, 287.8 eV, and 289.2 eV, respectively (Acheampong et al. 2018; Huang et al. 2019). C1 is mainly attributed to lignin and extracts of wood, such as fatty acids, fats, and waxes. C2 and C3 are mainly attributed to the hydroxyl of hemicellulose and cellulose and the carbonyl of hemicellulose and lignin, respectively. C4 is mainly attributed to the ester and carboxyl groups of hemicellulose and extract (Lyu et al. 2019; Sernek et al. 2004; Ozen et al. 2014). The relative percentages of C1 in 180 °C TMS decreased compared to the original spruce, attributed to the loss of extracts of spruce such as fatty acids, resin acids, and glycerol esters (Inari et al. 2011; Popescu et al. 2009). The relative percentages of C1 in 220 °C TMS were higher than those of 180 °C TMS, which was attributed to the degradation of hemicellulose leading to a decrease in the relative percentages of C2. The relative percentages of C4 in spruce treated at 180 and 220 °C increased, indicating the production of extracts containing ester and carboxyl groups after thermal modification. These extracts might have antifungal activity, making a partial contribution to improving the decay resistance of TMW.
The relative percentages of C2 in 180 °C TMS decreased after extract removal, indicating the presence of free hydroxyl groups in the extract of wood thermally modified at this temperature. The hydroxyl percentages of TMW at 180 °C decreased with extract removal, resulting in decreased hygroscopicity. The relative percentages of C2 in 220 °C TMS were lower than those in the original spruce wood, while it decreased after removing the extract. This indicated that the hydroxyl content of the extract was lower at high temperatures, but it exposed more hydroxyl groups of TMW after the removal of the extract, leading to an increase in hygroscopicity. This was consistent with the conclusion of Sect. 3.3.1. Additionally, the relative percentages of C4 in 180 and 220 °C TMS significantly decreased after extract removal, indicating the extraction of newly produced extracts with antifungal activity, potentially contributing to a decrease in wood decay resistance.
3.4 Effects of extract on the decay resistance of thermally modified wood
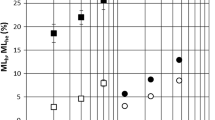
The decay resistance of poplar and spruce wood against brown-rot fungi (G. trabeum) or white-rot fungi (T. versicolor) can be significantly improved by thermally modified at 200 °C or 200 °C and 220 °C, respectively (Fig. 7). The decay resistance of the 180 °C, 200 °C, and 220 °C TMW decreased after extraction, and the degree of decrease weakened with the temperature increasing.
The mass loss of 220 °C TMP and TMS exposed to T. versicolor after 15 weeks of incubation was 14.0% and 3.5% (Fig. 7a and c), significantly lower than the mass loss of original wood (39.0% and 24.0%). Similarly, their mass loss exposure to G. trabeum after 15 weeks of incubation was 0.9% and 0.4% (Fig. 7b and d), significantly lower than the mass loss of original wood (34.8% and 12.5%). The extract had a non-significant effect on the decay resistance of TMW at this temperature. Some studies have indicated that the improvement in decay resistance of wood thermally modified at 220 °C might be due to the combined effect of the degradation of three major nutrients, leading to nutrient source loss, decreased hygroscopicity, formation of pseudo-lignin and antifungal active compounds (Jiang et al. 2022; Mohammed et al. 2006). For example, the study has indicated that the moisture content and holocellulose of 230 °C TMW (Pinus spp., Fraxinus mandshurica, and Quercus mongolica) significantly decreased compared to 180 °C TMW, which is only half of that of untreated wood (Li et al. 2009).
Mass loss of extracted/unextracted poplar (a, b) and spruce (c, d) wood thermally modified at 180 °C, 200 °C, and 220 °C exposed to T. versicolor and G. trabeum. Note: ns means no significant difference, * means significant difference (P < 0.05), and ** means extremely significant difference (P < 0.01)
The decay resistance of poplar and spruce wood against G. trabeum fungi can be significantly improved by thermal modification at 200 °C, but their decay resistance to T. versicolor cannot be improved. The positive effect of pseudo-lignin on the decay resistance of TMW to brown-rot fungi, with little effect on white-rot fungi, might be a contributing factor (Jiang et al. 2022). Additionally, hemicellulose and lignin were partially degraded at 200 °C, and brown-rot fungi might lose more nutrients than white-rot fungi. The removal of extract had no significant effect on the decay resistance of 200 °C TMW (P ˃ 0.05), except for the decay resistance of TMP to G. trabeum. The mass loss of 200 °C TMP after extraction exposed to T. versicolor and G. trabeum increased from 41.4% and 2.3–44.8% and 10.2%, respectively. The extract with antifungal activity was only one of the contributing factors to the improved decay resistance of 200 and 220 °C TMW, and the role of extract needs to be further studied.
Thermal modification of poplar and spruce wood at 180 °C cannot effectively improve their decay resistance. The removal of the extract significantly impacted the decay resistance of 180 °C TMP against T. versicolor and TMS against G. trabeum (P < 0.05). The mass loss of TMP after extraction exposed to T. versicolor and G. trabeum increased from 32.5% and 16.9–47.4% and 22.2%, respectively. Additionally, the mass loss of TMS after extraction exposed to G. trabeum increased from 14.8 to 27.1%, respectively. Jiang et al. (2022) indicated that the pseudo-lignin produced by hydrothermal treatment of wood at 180 °C was minimal and insufficient to affect the decay resistance of TMW. The degradation of the three major nutrients was weaker at this temperature and there was no significant improvement in hygroscopicity (Fig. 4). The results indicated that the extracts of 180 °C TMP and TMS can significantly improve the decay resistance of 180 °C TMW, and the extract played an important role in the decay resistance of TMW at this temperature.
3.5 Effect of thermally modified wood extracts on fungi growth
In order to further analyze the effect of TMW extracts on the decay resistance of TMW, the antifungal activity of 180 °C, 200 °C, and 220 °C TMW extracts was analyzed. The brown-rot fungi (G. trabeum) or white-rot fungi (T. versicolor) grew well on the non-amended control PDA, completely covering the agar over the 8-day incubation period. The average colony diameter (mm) of both fungi grown on PDA amended with 16, 8, and 4 mg/mL extracts are shown in Fig. 8, Table S1, and Table 2. Both fungi grown on PDA amended with ethanol completely covered the agar over the 21-day incubation period. The average colony diameter of both fungi grown on PDA amended with 16 mg/mL extracts of 180 °C, 200 and 220 °C TMW (TMP and TMS) were not increased (Fig. 8), indicating that all extracts at this concentration can completely inhibit the growth of wood decay fungi.
8 mg/mL extract of 220 °C TMS cannot completely inhibit the growth of wood decay fungi, but their growth was slower than the control (Table S1). The growth of both tested fungi can be completely inhibited by other TMP and TMS extracts at different temperatures. Both tested fungi cannot be inhibited by 4 mg/mL extract of 180 °C, 200 °C, and 220 °C TMP and TMS (Table 2). However, their growth was slower than control and solvent control. The results indicated that the acetone extracts of TMW treated at 180 °C, 200 °C, and 220 °C showed satisfactory antifungal activity against wood decay fungi, and their growth was completely inhibited at 16 mg/mL. There was no significant difference in the antifungal activity of extracts from TMW treated at different temperatures. Ethanol extract of Forsythia suspensa, the essential oils of Betula pendula and Syzygium aromaticum, and water extract of teak (Tectona grandis L. f.) were reported to inhibit the growth of wood decay fungi at a concentration of 30 mg/mL, 10%, and 20 mg/mL, respectively (Zhao et al. 2021; Pánek et al. 2014; Brocco et al. 2017). This result indicated that TMW extracts might have more potential as wood preservatives.
3.6 Chemical composition of extracts from thermally modified poplar and spruce wood
The primary components (top ten compounds) of original wood (Table S2) and 180 °C, 200 °C, and 220 °C TMW in the resulting GC-MS chromatograms were listed based on peak area (Tables 3 and 4). The compound with the highest relative content (reference only) of acetone extract from poplar sapwood was 4 H-1-Benzopyran-4-one, 2,3-dihydro-5-hydroxy-2-(4-hydroxyphenyl)-7-methoxy-, (S)-, followed by Naringenin, and the other compounds were various, including esters, organic acids, and alcohols. The extract of spruce sapwood contained 25% of 4’-methoxy-neneneea-2-hydroxystilbene, 8.9% of 2-(4-methylphenyl) benzoic acid, along with other compounds such as esters and phenols. The antifungal activity of these compounds against wood decay fungi has not been reported.
Aromatic aldehyde and aromatic acid compounds such as vanillin, syringaldehyde (benzaldehyde, 4-hydroxy-3,5-dimethoxy-), syringic acid (benzoic acid, 4-hydroxy-3,5-dimethoxy-), 4-ethoxybenzoic acid (benzoic acid, 4-hydroxy-), and isovanillic acid (3-hydroxy-4-methoxybenzoic acid) appeared in the acetone extract of poplar wood after thermal modification (Table 3). Vanillin, syringaldehyde, syringic acid, and isovanillic acid have been proven to show antifungal activity against wood decay fungi (Bi et al. 2021a, 2022; Esteves et al. 2008). These compounds contributed to the antifungal activity of TMW extracts. The relative content of aromatic aldehydes and aromatic acids increased and then decreased with the increase of thermal modification temperature, and the content of aromatic acids was gradually higher than that of aromatic aldehydes.
The compounds with antifungal activity such as isovanillic acid, vanillin, syringic acid, and syringaldehyde appeared in the acetone extract of spruce wood after thermal modification, which was the same as that of poplar (Table 4). The main compounds of TMS acetone extract were isovanillic acid and vanillin, while the TMP extract was mainly syringic acid. This was related to the lignin types of softwood (mainly guaiacyl phenylpropane units) and hardwood (mainly guaiacyl and syringyl phenylpropane units). The relative content of aromatic aldehyde and aromatic acid compounds exhibited an initial increase followed by a decrease with the rise in thermal modification temperature. The results indicated that the antifungal activity compounds from lignin degraded, such as isovanillic acid, vanillin, syringic acid, and syringaldehyde, might be the source of the antifungal activity of TMW extracts.
4 Conclusion
The effect of acetone extracts of thermally modified poplar and spruce wood on the color, dimensional stability, and decay resistance of thermally modified wood was studied. The results suggest the extract positively affected the decay resistance, color, and dimensional stability (220 °C) of thermally modified wood. The brightness difference ∆L* and total color difference ∆E* of TMW decreased with the removal of the extract, and this effect weakened with the increase of treatment temperature, significantly at 180 °C. The dimensional stability of 180 and 200 °C TMW was improved due to the removal of the extract. The moisture absorption and anti-swelling efficiency of extracted 180 °C TMP and TMS were increased from 20.6% and − 6.9–34.2% and 14.1%, respectively. The removal of extracts led to a reduction in the decay resistance of TMW. The mass loss of 180 °C TMP exposed to white-rot fungi and brown-rot fungi increased from 32.5% and 16.9–47.4% and 22.2% after extract removal, respectively. The extract showed satisfactory antifungal activity against wood decay fungi, and the compounds with antifungal activity such as vanillin, isovanillic acid, syringaldehyde, and syringic acid in the extract played an important role.
References
Acheampong A, Okyem S, Akoto CO, Baah KA (2018) Antioxidant, antimicrobial and FTIR analysis of methanol root extract of Cnestis ferruginea and ethanol root extract of Citrus limon. J Pharmacogn Phytochem 7(4):2938–2946. https://www.phytojournal.com/archives?year=2018&vol=7&issue=4&ArticleId=5393
Bekhta P, Niemz P (2003) Effect of high temperature on the change in color, dimensional stability, and mechanical properties of spruce wood. Holzforschung 57(5):539–546. https://doi.org/10.1515/HF.2003.080
Bi ZJ, Zhao Y, Morrell JJ, Lei YF, Yan L (2021a) The antifungal mechanism of Konjac flying powder extract and its active compounds against wood decay fungi. Ind Crop Prod 164(1):113406. https://doi.org/10.1016/j.indcrop.2021.113406
Bi ZJ, Yuan JY, Morrell JJ, Yan L (2021b) Effects of extracts on the colour of thermally modified Populus tomentosa Carr. Wood Sci Technol 55:1075–1090. https://doi.org/10.1007/s00226-021-01304-7
Bi ZJ, Morrell JJ, Lei YF, Yan L, Ji MT (2022) Eco-friendly and mildly modification of wood cell walls with heat treated wood extracts to improve wood decay resistance. Ind Crop Prod 184:115079. https://doi.org/10.1016/j.indcrop.2021.113406
Brocco VF, Paes JB, Gonçalves da Costa L, Brazolin S, Arantes MDC (2017) Potential of teak heartwood extracts as a natural wood preservative. J Clean Prod 142:2093–2099. https://doi.org/10.1016/j.jclepro.2016.11.074
Candelier K, Thevenon MF, Petrissans A, Dumarcay S, Gerardin P, Petrissans M (2016) Control of wood thermal treatment and its effects on decay resistance: a review. Ann for Sci 73:571–583. https://doi.org/10.1007/s13595-016-0541-x
Candelier K, Thévenon MF, Collet R, Gérardin P, Dumaray S (2020) Anti-fungal and anti-termite activity of extractives compounds from thermally modified ash woods. Maderas-Cienc Tecnol 22(2):223–240. https://doi.org/10.4067/S0718-221X2020005000209
Chaouch M, Petrissans M, Petrissans A, Gerardin P (2010) Use of wood elemental composition to predict heat treatment intensity and decay resistance of different softwood and hardwood species. Polym Degrad Stabil 95(12):2255–2259. https://doi.org/10.1016/j.polymdegradstab.2010.09.010
Chen Y, Fan YM, Gao JM, Li HK (2012) Coloring characteristics of in situ lignin during heat treatment. Wood Sci Technol 46(1–3):33–40. https://doi.org/10.1007/s00226-010-0388-5
Chu DM, Zhang XY, Mu J, Avramidis S, Xue L, Li YS (2019) A greener approach to byproducts from the production of heat-treated poplar wood: analysis of volatile organic compound emissions and antimicrobial activities of its condensate. J Clean Prod 213:521–527. https://doi.org/10.1016/j.jclepro.2018.12.163
Costa JC, Branco JM, Cames AF (2019) Thermal modification of most representative Portuguese wood species. A preliminary study. Rev Constr 18(3):488–500. https://doi.org/10.7764/RDLC.18.3.488
Dubey MK, Pang SS, Walker J (2011) Effect of oil heating age on colour and dimensional stability of heat-treated pinus radiata. Eur J Wood Prod 69(2):255–262. https://doi.org/10.1007/s00107-010-0431-0
Esteves B, Graça J, Pereira H (2008) Extractive composition and summative chemical analysis of thermally treated eucalypt wood. Holzforschung 62(3):344–351. https://doi.org/10.1515/HF.2008.057
Gadhave RV, Sanjivkasbe P, Mahanwar PA, Gadekar PT (2019) Synthesis and characterization of lignin-polyurethane based wood adhesive. Int J Adhes Adhes 95:102427. https://doi.org/10.1016/j.ijadhadh.2019.102427
Gao S, Chen Y, Gao JM (2015) Effect of extractives on Color changes of Eucalyptus urophylla × Eucalyptus grandis and its Spectrum Analysis. J Northeast for Univ 43(4):73–76. http://doi.org/CNKI:SUN:DBLY 0.2015-04-016
González-Peña MM, Hale MDC (2009) Colour in thermally modified wood of beech, Norway spruce and scots pine. Part 1: Colour evolution and colour changes. Holzforschung 63(4):385–393. https://doi.org/10.1515/HF.2009.078
Hakkou M, Pétrissans M, Gérardin P, Zoulalian A (2006) Investigations of the reasons for fungal durability of heat-treated beech wood. Polym Degrad Stabil 91:393–397. https://doi.org/10.1016/j.polymdegradstab.2005.04.042
Hill C, Altgen M, Rautkari L (2021) Thermal modification of wood-a review: chemical changes and hygroscopicity. J Mater Sci 56(11):6581–6614. https://doi.org/10.1007/s10853-020-05722-z
Huang RF, Lv JX, Cao YJ, Zhao X, Zhao YK, Zhou YD (2010) Impact of heat treatment on chemical composition of Chinese white poplar wood. J Beijing for Univ 32(3):155–160. https://doi.org/10.3724/SP.J.1011.2010.01351
Huang YG, Li GY, Chu FX (2019) Modification of wood cell wall with water-soluble vinyl monomer to improve dimensional stability and its mechanism. Wood Sci Technol 53(3):1051–1060. https://doi.org/10.1007/s00226-019-01112-0
Inari GN, Petrissans M, Dumarcay S, Lambert J, Ehrhardt JJ, Sernek M, Gerardin P (2011) Limitation of xps for analysis of wood species containing high amounts of lipophilic extractives. Wood Sci Technol 45(2):369–382. https://doi.org/10.1007/s00226-010-0324-8
Jiang JY, Peng Y, Ran YY, Cao JZ (2022) Pseudo lignin formed from hygrothermally treated holocellulose and its effect on fungal degradation. Ind Crop Prod 184:115004. https://doi.org/10.1016/j.indcrop.2022.115004
Källbom S, Moghaddam MS, Walinder MEP (2018) Liquid sorption, swelling and surface energy properties of unmodified and thermally modified scots pine heartwood after extraction. Holzforschung 72(3):251–258. https://doi.org/10.1515/hf-2017-0064
Kamdem DP, Pizzi A, Jermannaud A (2002) Durability of heat-treated wood. Holz Roh- Werkst 60(1):1–6. https://doi.org/10.1007/s00107-001-0261-1
Korenman YI, Mokshina NY, Zykov AV (2010) Distribution coefficients of vitamin B-2 in hydrophilic organic solvent-aqueous salt solution systems. Russ J Phys Chem A 84(3):415–418. https://doi.org/10.1134/S0036024410030131
Korkut S, Akgul M, Dundar T (2008) The effects of heat treatment on some technological properties of scots pine (Pinus sylvestris L.) wood. Bioresource Technol 99(6):1861–1868. https://doi.org/10.1016/j.biortech.2007.03.038
Li HM, Chen RW, Yan T, Chen KH (2009) Chemical composition, decay resistance and mold inhibition of heat-treated lumber. China Wood Ind 23(3):46–48. https://doi.org/10.19455/j.mcgy.2009.03.015
Li RR, Xu W, Wang XD, Wang CG (2018) Modeling and predicting of the color changes of wood surface during CO2 laser modifcation. J Clean Prod 183:818–823. https://doi.org/10.1016/j.jclepro.2018.02.194
Ling Z, Ji Z, Ding DY, Cao JZ, Xu F (2016) Microstructural and topochemical characterization of thermally modified poplar (Populus cathayaha) cell wall Bioresources 11(1):786–799. https://bioresources.cnr.ncsu.edu/resources/microstructural-and-topochemical-characterization-of-thermally-modified-poplar-populus-cathayaha-cell-wall/
Lyu WH, Shi Y, Zheng YX, Liu X (2019) XPS and FTIR studies of fungus-stained Daemonorops margaritae. J for Res 30(2):739–743. https://doi.org/10.1007/s11676-018-0598-5
Mayer I, Koch G, Puls J (2006) Topochemical investigations on wood extractives and their influence on color changes in American black cherry (Prunus serotina Borkh). Holzforschung 60:589–594. https://doi.org/10.1515/HF.2006.100
Mehrotra R, Singh P, Kandpal H (2010) Near infrared spectroscopic investigation of the thermal degradation of wood. Thermochim Acta 507–508:60–65. https://doi.org/10.1016/j.tca.2010.05.001
Metsa-Kortelainen S, Antikainen T, Viitaniemi P (2006) The water absorption of sapwood and heartwood of scots pine and Norway spruce heat-treated at 170°C, 190°C, 210°C and 230°C. Holz Roh Werkst 64(3):192–197. https://doi.org/10.1007/s00107-005-0063-y
Mohammed H, Petrissans M, Gerardin P, Zoulalian A (2006) Investigations of the reasons for fungal durability of heat-treated beech wood. Polym Degrad Stabil 91(2):393–397. https://doi.org/10.1016/j.polymdegradstab.2005.04.042
Nguyen THV, Nguyen TT, Ji XD, Guo MH (2018) Predicting color change in wood during heat treatment using an artificial neural network model. BioResources 13(3):6250–6264. https://bioresources.cnr.ncsu.edu/wp-content/uploads/2018/06/BioRes_13_3_6250_Nguyen_NJG_Predicting_Color-Change_Wood_Heat_ANN-_Model_13888.pdf
Ozen E, Yeniocak M, Goktas O, Alma HM, Yilmaz F (2014) Antimicrobial and antifungal properties of madder root (Rubia tinctorum) colorant used as an environmentally-friendly wood preservative. BioResources 9(2):1998–2009. https://doi.org/10.15376/biores.9.2.1998-2009
Pánek M, Reinprecht L, Hulla M (2014) Ten essential oils for beech wood protection-efficacy against wood-destroying fungi and moulds, and effect on wood discoloration. BioResources 9:5588–5603. https://bioresources.cnr.ncsu.edu/BioRes_09/BioRes_09_3_5588_Panek_RH_Essential_Oils_Wood_Protection_5577.pdf
Popescu CM, Tibirna CM, Vasile C (2009) Xps characterization of naturally aged wood. Appl Surf Sci 256(5):1355–1360. https://doi.org/10.1016/j.apsusc.2009.08.087
Qu L, Qian J, Gao J, Wang Z, Yi S (2021) Effect of aluminum sulfate impregnation heat joint treatment on color changes of Chinese fir. Wood Sci Tech 55:1–23. https://doi.org/10.1007/s00226-020-01249-3
Sandoval-Torres S, Jomaa W, Marc F, Puiggali JR (2010) Causes of color changes in wood during drying. For Stud China 12(4):167–175. https://doi.org/10.1007/s11632-010-0404-8
Sernek M, Kamke FA, Glasser WG (2004) Comparative analysis of inactivated wood surfaces. Holzforschung 58(1):22–31. https://doi.org/10.1515/HF.2004.004
Shen H, Cao J, Sun W, Peng Y (2016) Influence of post-extraction on photostability of thermally modified scots pine wood during artificial weathering. BioRresource 11(2):4512–4525. https://bioresources.cnr.ncsu.edu/resources/influence-of-post-extraction-on-photostability-of-thermally-modified-scots-pine-wood-during-artificial-weathering/
Sivrikaya H, Can A, Conde M (2015) Comparative biological resistance of differently thermally modified wood species against decay fungi, reticulitermes grassei and hylotrupes bajulus. Maderas-Cienc y Tecnol 17(3):559–570. https://doi.org/10.4067/S0718-221X2015005000050
Smeds AI, René Herrera Rahkila J, Willfor SM (2021) Extractives in betula celtiberica stemwood and isolation and identification of diarylheptanoids in the hydrophilic extract. Holzforschung 75(11):1001–1011. https://doi.org/10.1515/hf-2021-0022
Spear MJ, Curling SF, Dimitriou A, Ormondroyd GA (2021) Review of functional treatments for modified wood. Coatings 11(3):327. https://doi.org/10.3390/coatings11030327
Tripathi S, Pant H, Kashyap AK (2014) Decay resistance against basidiomycetes fungi of heat-treated pinus roxburghii and mangifera indica wood. J Trop for Sci 26(2):203–207. https://doi.org/10.1093/forestry/cpt033
Vuthiganond N, Nakpathom M, Mongkholrattanasit R (2020) Azoic deep dyeing of silk and UV protection using plant polyphenols and diazonium coupling. Fiber Polym 21(5):1052–1060. https://doi.org/10.1007/s12221-020-9057-y
Yan L, Zeng FY, Chen ZJ, Chen S, Lei YF (2021) Improvement of wood decay resistance by salicylic acid / silica microcapsule: effects on the salicylic leaching, microscopic structure and decay resistance. Int Biodeter Biodegr 156:105134. https://doi.org/10.1016/j.ibiod.2020.105134
Zhan TY, Zhu JY, Liu ZT, Li T, Peng H, Jiang JL, Lyu J (2022) Meta-analysis of chromatic properties of heat-treated wood. Eur J Wood Prod 80(4):851–858. https://doi.org/10.1007/s00107-022-01831-5
Zhao XQ, Wang L, Ren SM, Hu Z, Wang YM (2021) One-pot synthesis of Forsythia@carbon quantum dots with natural anti-wood rot fungus activity. Mater Des 206:109800. https://doi.org/10.1016/j.matdes.2021.109800
Zhu T, Sheng JL, Chen JT, Ren K, Wu ZZ, Wu H, Li J, Lin JG (2021) Staining of wood veneers with anti-uv property using the natural dye extracted from dalbergia cohinchinensis. J Clean Prod 284(4):124770. https://doi.org/10.1016/j.jclepro.2020.124770
Acknowledgements
This study was supported by the National Natural Science Foundation of China (31971590) and the 111 project (D21027).
Author information
Authors and Affiliations
Contributions
ZJ Bi: Conceptualization, Methodology, Investigation, Writing - Origianl Draft, and Visualization. XJ Zhou: Writing - review & editing and Methodology. J Chen: Investigation (Sect. 2.3 and 2.7) and Data Curation. YF Lei: Supervision and Validation. L Yan: Resources, Supervision, Funding acquisition, Project administration. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest exists in the submission of this manuscript, and the manuscript is approved by all authors for publication.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bi, Z., Zhou, X., Chen, J. et al. Effects of extracts on color, dimensional stability, and decay resistance of thermally modified wood. Eur. J. Wood Prod. 82, 387–401 (2024). https://doi.org/10.1007/s00107-023-02024-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00107-023-02024-4