Abstract
Purpose
Our study was designed to investigate premaxillary–maxillary suture growth in fetuses from the first trimester of pregnancy using the B‑ultrasound technique in order to determine the suture fusion time.
Methods
We selected 169 healthy Han singleton pregnancies as subjects. All subjects received routine pregnancy tests and were divided into three groups based on the gestational age of the fetus: group 1, the 11th gestational week; group 2, the 12th gestational week; and group 3, the 13th gestational week. Fetal biometric measurements were recorded during consecutive prenatal ultrasonographic examinations. These measurements included nuchal translucency thickness, crown–rump length, and premaxillary–maxillary length. Intergroup comparisons were performed using analysis of variance (ANOVA).
Results
The premaxillary–maxillary suture grows gradually and its measured length at the 11th, 12th and 13th week was 0.54 cm, 0.65 cm, and 0.74 cm, respectively. We observed a significant linear correlation between the premaxillary–maxillary length and the week of gestation in the first trimester. The growth rate of the maxilla at the 11th, the 12th and the 13th week are significantly different with a descending order of growth rates being week 12, week 11 and week 13, with the 12th week rate being the most rapid.
Conclusion
The premaxillary and maxillary growth at 11 and 12 gestational weeks in the first trimester steadily accelerated, peaking at the 12th week. The rate of growth slows down after week 12 which may be associated with the fusion of the premaxillary–maxillary suture.
Zusammenfassung
Ziel
Die vorliegende Studie wurde durchgeführt, um die Entwicklung der Sutur zwischen Prämaxilla und Maxilla bei Föten im ersten Schwangerschaftsdrittel mittels B‑Ultraschalltechnik zu untersuchen und den Zeitpunkt der Suturverknöcherung zu bestimmen.
Methode
An der Studie nahmen 169 gesunde Schwangere (Mehrlingsgebärende ausgeschlossen) teil. Alle Teilnehmerinnen wurden regelmäßig routinemäßig untersucht und wurden in drei Gruppen eingeteilt, basierend auf dem Gestationsalter des Fötus: Gruppe 1: 11. Schwangerschaftswoche (SSW); Gruppe 2: 12. SSW, und Gruppe 3: 13. SSW. Die biometrische Vermessung der Föten wurde während der konsekutiven pränatalen Ultraschalluntersuchungen durchgeführt. Diese Messungen enthielten Nackentransparenzmessung, Schädel-Steiß-Länge und Länge der Prämaxilla-Maxilla. Vergleiche zwischen den Gruppen wurden mittels Varianzanalyse (ANOVA) durchgeführt.
Ergebnisse
Die Sutur Prämaxilla-Maxilla entwickelte sich kontinuierlich, wobei ihre Länge in der 11., 12. und 13. SSW 0,54 cm, 0,65 cm und 0.74 cm beträgt. Wir konnten eine signifikante lineare Korrelation zwischen Prämaxilla-Maxilla-Länge und der SSW im ersten Schwangerschaftsdrittel beobachten. Das maxilläre Wachstum in der 11., 12. und 13. SSW unterscheidet sich signifikant, wobei die Wachstumsrate in der 12. SSW am höchsten ist, gefolgt von der 11. und 13. SSW.
Schlussfolgerung
Das Wachstum der Prämaxilla und Maxilla nahm in der 11. und 12. SSW stetig zu, und wies einen Wachstumsgipfel in der 12. SSW auf. Die Wachstumsrate verlangsamte sich nach der 12. SSW, was sich möglicherweise auf die Verknöcherung der Sutur Prämaxilla-Maxilla zurückführen lässt.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Angle class III malocclusion is one of the most common malocclusions for children in China. This malformation can be etiologically summarized as maxillary deficiency, mandibular excess, or a combination of both. Maxillary deficiency accounts for a large percentage of these malocclusions; however, the pathogenesis of maxillary deficiency is still unknown [20,21,22, 26]. Our animal studies showed that early ossification of the premaxillary–maxillary suture may induce extensive craniomaxillofacial morphological abnormality in rats (including the midface), which was similar to the facial characteristics in human class III malocclusion. Thus, we can infer that the midfacial hypoplasia is related to the development and growth of the premaxillary–maxillary suture [6, 18]. Due to the different prevalence rates of class III malocclusion between Mongoloid and Caucasian races, we propose that class III malocclusion is related to the different time points for the ossification of the premaxillary–maxillary suture (PMS).
Cranial sutures are membranous gaps that remain between the boney plates of the skull as the bones grow and develop. These intervening fibrous sutures act as flexible joints between the developing bones allowing the skull to change shape and grow during development. Under normal conditions, complete fusion of the cranial bones usually does not occur until adulthood. There are many sutures located in the cranium and maxillofacial area. In the cranium, there are three sutures, i. e., the coronal suture, the sagittal suture, and the lambdoid suture. For the maxillofacial area, there are many more sutures, including the midpalatal suture, the zygomaticomaxillary suture, and the premaxillary–maxillary suture. In addition, the premaxilla, the main structure of the middle facial area, connects with the maxilla, basal bone, vomer, and frontal bone by the corresponding sutures. In humans, all sutures in the maxillofacial area remain patent except for the PMS, which begins to fuse around the time of birth. In all other mammalian species, the PMS remains patent. This leads to the characteristic facial profile of human beings, such as the anterior nasal spine [3, 15, 16]. As the size of the anterior nasal spine correlates with facial prognathism and the timing of premaxillary–maxillary sutural fusion, Mooney et al. suggested that the timing of PMS fusion may have implications for models of midfacial growth [16]. By examining dry skulls, they demonstrated that PMS fusion time correlates significantly with midfacial morphology and may explain the distinctive racial variations between black and white populations in midfacial profiles established early in fetal development and maintained postnatally [15]. In addition, studies by Holton et al. showed that restriction of sutural growth via rigid plate fixation can make a highly significant reduction in facial projection and overall size, which is the characteristic configuration of the modern human [6]. Furthermore, our own studies show that PMS premature fusion can induce extensive abnormal craniofacial form, which is associated with the characteristics of class III malocclusion [18]. Therefore, some scholars proposed that if the PMS remains open, we may be able to stimulate the formulation of the maxilla and the midface with the use of anteroposterior expansion of the maxilla for the treatment of class III malocclusion and in cases of nasal obstruction [24]. However, due to the rapid fetal development time and the lack of effective examination techniques, PMS fusion time is still unresolved and controversial. Some scholars considered that human premaxilla first appears at gestational week 7 and begins to fuse with the maxillary bone at gestational week 9 until around birth [2]. Other scholars, however, have demonstrated suture fusion at gestational week 14 [27, 28]. Some scholars revealed that the premaxillary–maxillary suture remains open during childhood or partly fused throughout life, and they postulate that the premaxilla could be used as the basis for therapeutics for the middle third of growth [24].
With constant improvement in ultrasonic technology, B‑ultrasound has become an important means of examination in the first trimester today. Not only normal fetal development, but also major fetal malformations can now be found with B‑ultrasound in the first trimester. Due to a strong correlation between fetal nuchal translucency (NT) and fetal health, a NT scan by ultrasonography has become an essential means for fetal health screening in the first trimester in recent years [10, 11, 23]. Some scholars used B‑ultrasound to measure the maxillary lengths of the fetus from 11 +0 to 13 +6 gestational weeks and proposed a linear correlation between maxillary lengths and gestation time in weeks [19]. However, the possibility that the PMS may be fusing during this time suggests that the rate of increase of the premaxillary and maxillary length could be slowed. In this study, we hypothesize that the PMS is fusing at some time during these 3 weeks and as a result, this fusion leads to a non-linear growth rate.
Subjects and methods
Subjects
For this study, we selected 169 healthy Han nationality women with normal singleton pregnancies and administered routine pregnancy tests in the Hangzhou Fuyang Woman and Children Hospital. The pregnant women were ages 29.19 ± 4.72 years and at 11 +0 to 13 +6 gestational weeks (12.52 ± 0.68 weeks). All subjects were divided into three groups according to their week of gestation (Table 1). The ages of the pregnant women in each group showed no significant differences (F = 0.47, P = 0.63). This study has been reviewed and approved by the medical ethics review board in the Children’s Hospital Zhejiang University School of Medicine and has obtained the informed consent of the patients.
Methods
An ultrasound 730 machine equipped with a 4 MHz abdominal probe (GE Healthcare, Chicago, IL, USA) was used for this study. During image acquisition, the long axis of the probe was brought parallel to the median line of the sagittal view of the fetal body and an image of the sagittal view was acquired. The direction of the probe was adjusted to maximize the manifestation of nuchal translucency thickness (NT), crown–rump length (CRL), and premaxillary–maxillary length (MaxL). A method of real-time measurement was adopted to measure NT, CRL, and MaxL by an experienced specialist in ultrasonic imaging (H.W.Q.).
NT measurement
According to the measurement method in the literature [17], the two hyperechogenic regions were measured at the bottom of the nuchal dorsum displayed in the sagittal view, where NT (in cm) was defined as the maximum distance. In this study, we only include fetuses whose NT was less than 0.3 cm, which we considered to be normal based on the literature [8].
MaxL measurement
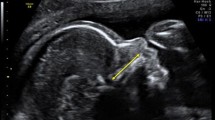
The maximum distance was measured around the hyperechogenic tissue displayed on the middle face of a fetus when its face was presented in the midsagittal position (Fig. 1). This distance (in cm) included the premaxilla, the maxillary palatal process and the palatal horizontal process. These structures appear rod-shaped in the ultrasonic picture and we measured these rod-shaped bones from the inner to the outer margins along the mid-line.
Ultrasound midsagittal plane view, MaxL is defined by a line segment between the inner and the outer margins along the mid-line, including premaxilla, maxillary palatal process, and palatal horizontal process. MaxL premaxillary–maxillary length
Midsagittalebene im Ultraschall. MaxL ist definiert durch die Linie zwischen den äußeren und inneren Rändern entlang der Mittellinie, die Prämaxilla, maxillären Gaumenfortsatz und horizontalen Gaumenfortsatz enthält. MaxL „premaxillary–maxillary length“
CRL measurement
CRL is the maximum distance (in cm) measured from the top of the head to the bottom of the buttocks of a fetus when its face is presented in the mid sagittal position (Fig. 2).
Results
MaxL at different gestational weeks
MaxL is 0.54 cm at 11 weeks, 0.65 cm at 12 weeks, and 0.74 cm at 13 weeks (Table 2). This suggests that MaxL continuously increased from the 11th week to the 13th week, while the MaxL growth increment (0.11 cm) from the 11th week to the 12th week was greater than the growth increment (0.09 cm) from the 12th week to the 13th week (P < 0.001). These results suggest that the rate of MaxL growth before 12 weeks is greater than the rate of growth after 12 weeks.
Correlation between MaxL and the weeks of gestation and between MaxL and CRL
There was a moderate but significant correlation between MaxL and gestational age (Table 3), with a Pearson correlation coefficient of 0.713 (P < 0.001). Similarly, a statistical correlation was found between MaxL and CRL with a correlation coefficient of 0.697 (P < 0.001).
MaxL growth rate difference
Based on the correlation between MaxL and the gestational weeks, linear regression relationships were analyzed for these different stages (Fig. 3). The linear regression equation for MaxL (y) and the gestational weeks (x) was found to be as follows: y = 0.18x − 1.60, R2 = 0.31 at the 11th week, y = 0.22x − 2.04, R2 = 0.37 at the 12th week, and y = 0.16x − 1.45, R2 = 0.24 at the 13th week, respectively. For these results, the gestational age of 12 weeks had the greatest slope, relative to 11 weeks and 13 weeks (The slope increase for the MaxL and the gestational week data was 12th week > 11th week > 13th week). From this analysis, we conclude that MaxL growth rates for these 3 weeks were different, reached a peak level in week 12, then a slowing of the growth rate by the 13th week.
The linear regression equation between MaxL and the gestational weeks (GWs) showing that the gestational age of 12 weeks had the greatest slope, compared to the 11 weeks and 13 weeks. The conclusion can be drawn that MaxL growth rates during these 3 weeks are different, reaching the peak in week 12 and then slowing down. MaxL premaxillary–maxillary length
Die lineare Regression zwischen MaxL und den Gestationswochen (GWs) zeigt, dass das Gestationsalter von 12 Wochen im Vergleich zu 11 und 13 Wochen die größte Steigung hatte. Daraus lässt sich schließen, dass die MaxL-Wachstumsraten während dieser 3 Wochen unterschiedlich sind, den Höhepunkt in Woche 12 erreichen und danach eine Verlangsamung eintritt. MaxL „premaxillary–maxillary length“
Discussion
NT length and normal fetus examination in the first trimester
Fetal nuchal translucency (NT) length is, in fact, the lymph fluid collecting within the nape of the fetal neck during the first trimester. It increases during the 11th week and disappears at the end of the 13th week. In 1992, Nicolaides et al. [17] first found that the NT length of the early fetus (11 +0 to 13 +6 week) was related to some chromosome disorders and fetal malformations. In their analysis, they proposed that the fetuses whose NT thickness was less than 0.3 cm could be considered normal. Since then, additional studies have documented this phenomenon [8, 10, 11, 23]. Today, NT thickness measurement has become a popular sonographic technique for screening the health of children in the first trimester.
Researchers have shown that NT in most normal fetuses is less than 0.25 cm and if NT length is greater than 0.30 cm, chromosome abnormalities, congenital heart malformations, and facial deformities should be taken into consideration [1, 5, 8, 9, 13]. Our study shows that the mean value of NT was 0.13 ± 0.06 cm with a minimum value of 0.04 cm and a maximum value of 0.26 cm. For all 169 subjects in our study the values for NT were in the normal range. The percentage of low NT fetuses, whose NT is under 0.1 cm, is 30.8%, while 0.1–0.2 cm accounts for 63.3%. Together, these account for 94.1%. We found that values of 0.2–0.25 cm account for 5.3% and values over 0.25 cm account for 0.6%, together accounting for 5.9%. Therefore, in our study, NT measurements conformed to the definition of normal fetus NT values suggested by Nicolaides et al. [17].
Ossification time of the premaxillary–maxillary suture
The PMS exists in the early fetus, whereas its ossification time has long been a matter of controversy. Some scholars consider that the PMS starts to ossify after 9 weeks of pregnancy and completes the ossification process at birth [2]. Others are convinced that the fusion of the PMS ends after 14 weeks of pregnancy [27, 28]. Zhang et al. investigated the ossification centers and time points for the maxillofacial region in aborted fetuses, discovering that the maxillary ossification centers appeared at the 8th week of pregnancy, while the zygomatic process, palatal process, frontal process, and fossa canina emerged from their ossification centers after the 9th week. In addition, they also found that the premaxillary ossification center appeared at the 12th week, and quickly grew into processes, and then the premaxillary palatal process fused with the maxillary palatal process, which completely ossified at the 14th gestational week [27, 28]. The newest research shows that the premaxillary–maxillary suture remains open even after birth and the premaxilla exists non-fused or partly fused throughout life [24]. Abnormal growth of the premaxilla in relation to the maxilla, for example by precocious fusion, may be correlated with maxillofacial malformations such as prognathism [24] and class III malocclusion [6, 18].
The findings in this study show that the rate of MaxL increase for the 11th, 12th, and 13th week were different as the slope of the regression lines in these respective weeks was different. The growth differences suggest that during the earlier weeks of gestation, the PMS increased at a faster rate. This may be related to the state of suture patency or may be related to incomplete ossification of the PMS in which the connective tissue of sutures have a significant effect on ossification and increased the bone length. The linear regression equations for MaxL and the weeks of gestation for the time points of this study revealed that the slope of week 12 was the greatest, while the slope of week 13 was the lowest. These results suggest that the fusing state of PMS was toward the end of gestational week 12. The more fused the area of the PMS, the less increase found for MaxL. The lower slope at week 13 showed that a more fused area of the PMS existed at this time point than at week 12. Thus, after the 12th week, MaxL growth rate begins to slow and thus, its increment decreased. This observation is in accord with the growth patterns described in suture biology [7, 25].
Our study shows that mean MaxL for fetuses during the 11th and the 13th week was 0.65 cm and there exists a significantly positive correlation between MaxL and gestational weeks (R2 = 0.51). MaxL at the 12th week in our study was longer than in the study by Cicero et al., while it was shorter at the 13th week in our study than in their study [4]. Based on these results, we speculate that the ossification time of the PMS between Chinese Mongoloid and western Caucasian populations is different which could explain the different facial morphology or the differences in malocclusion. Taken together with the findings from the Chinese scientists Zhang et al. [27, 28], we conclude that the ossification time of PMS in China is earlier than that in Western countries. We suggest that the earlier ossification restrains the maxillary growth in the sagittal direction, which may lead to a shorter maxillary length and higher morbidity of Class III malocclusion in eastern Asia [12]. Further, the ossification time of PMS may occur at different times in different races. For example, the PMS ossification time for Caucasians of European origin may be significantly earlier than in people of African ancestry. This would explain why Caucasians of European origin have a more obvious anterior spina nasalis than people of African ancestry. A further speculation suggests that perhaps when compared with modern man, Neanderthal individuals had a more prominent midface due to the longer patent time of their PMS [14].
Although PMS fusion investigated during the various gestational stages cannot be confirmed by the ultrasonographic technique applied alone, the ultrasonographic scan is a good way to study the development, growth, and fusion of the PMS in real time at each gestational stage. However, due to the very complicated structure of the maxillofacial suture and the sophisticated mechanism of craniofacial development in the first trimester, more in depth investigations are needed in the future to confirm the role of PMS suture fusion in the observed morphological variations.
References
AlSarheed M (2015) A comparative study of oral health amongst trisomy 21 children living in Riyadh, Saudi Arabia: Part 1 caries, malocclusion, trauma. Saudi Dent J 27:220–223. https://doi.org/10.1016/j.sdentj.2015.03.003
Barteczko K, Jacob M (2004) A re-evaluation of the premaxillary bone in humans. Anat Embryol (berl) 207:417–437. https://doi.org/10.1007/s00429-003-0366-x
Behrents RG, Harris EF (1991) The premaxillary-maxillary suture and orthodontic mechanotherapy. Am J Orthod Dentofacial Orthop 99:1–6. https://doi.org/10.1016/s0889-5406(05)81673-7
Cicero S, Curcio P, Rembouskos G et al (2004) Maxillary length at 11–14 weeks of gestation in fetuses with trisomy 21. Ultrasound Obstet Gynecol 24:19–22. https://doi.org/10.1002/uog.1077
Gu LL (2012) Study on new technique of prenatal ultrasound screening and diagnosis of maxillofacial structural malformations in first-trimester. Southern medical university, Guangdong (Master’s thesis)
Holton NE, Franciscus RG, Nieves MA et al (2010) Sutural growth restriction and modern human facial evolution: an experimental study in a pig model. J Anat 216:48–61. https://doi.org/10.1111/j.1469-7580.2009.01162.x
Ishii M, Sun J, Ting MC et al (2015) The development of the calvarial bones and sutures and the pathophysiology of craniosynostosis. Curr Top Dev Biol 115:131–156. https://doi.org/10.1016/bs.ctdb.2015.07.004
Karki S, Joshi KS, Tamrakar SR et al (2013) Nuchal translucency in normal fetus and its variation with increasing crown rump length (CRL) and gestational age. Kathmandu Univ Med J 11:282–286. https://doi.org/10.3126/kumj.v11i4.12522
Lauridsen H, Hansen BF, Reintoft I et al (2005) Short hard palate in prenatal trisomy 21. Orthod Craniofac Res 8:91–95. https://doi.org/10.1111/j.1601-6343.2005.00318.x
Li ZH, Hong Y, Qin LL et al (2016) The value of nuchal translucency thickness in the fetal chromosome abnormality screening. J Pract Med 32:402–405
Liao D (2015) Correlation of fetal nuchal translucency sonography and abnormal fetal development. Chin J Fam Plann 23:239–241
Mak KL (1969) An analysis of treated orthodontic patients in Hong Kong. Dent Mag Oral Top 86:258–259
Markov D, Jacquemyn Y, Leroy Y (2003) Bilateral cleft lip and palate associated with increased nuchal translucency and maternal cocaine abuse at 14 weeks of gestation. Clin Exp Obstet Gynecol 30:109–110
Maureille B, Bar D (1999) The premaxilla in Neandertal and early modern children: ontogeny and morphology. J Hum Evol 37:137–152. https://doi.org/10.1006/jhev.1999.0312
Mooney MP, Siegel MI (1986) Developmental relationship between premaxillary-maxillary suture patency and anterior nasal spine morphology. Cleft Palate J 23:101–107
Mooney MP, Siegel MI (1991) Premaxillary-maxillary suture fusion and anterior nasal tubercle morphology in the chimpanzee. Am J Phys Anthropol 85:451–456. https://doi.org/10.1002/ajpa.1330850408
Nicolaides KH, Azar G, Byrne D et al (1992) Fetal nuchal translucency: ultrasound screening for chromosomal defects in first trimester of Pregnaney. BMJ 304:867–869. https://doi.org/10.1136/bmj.304.6831.867
Ruan WH, Winger JN, Yu JC et al (2008) Induced premaxillary suture fusion: class III malocclusion model. J Dent Res 87:856–860. https://doi.org/10.1177/154405910808700901
Shyu IL, Yang MJ, Wang HI et al (2014) Fetal maxillary and mandibular length in normal pregnancies from 11 weeks’ to 13+6 weeks’ gestation: a Taiwanese study. Taiwan J Obstet Gynecol 53:53–56. https://doi.org/10.1016/j.tjog.2012.05.003
Singh GD (1999) Morphologic determinants in the etiology of class III malocclusions: a review. Clin Anat 12:382–405. https://doi.org/10.1002/(SICI)1098-2353(1999)12:5<382::AID-CA9>3.0.CO;2-0
Stojanović Z, Nikolić P, Nikodijević A (2013) Cephalometric assessment of maxillary length in Serbian children with skeletal class III. Vojnosanit Pregl 70:645–652. https://doi.org/10.2298/vsp110224042s
Szuhanek C, Gâdea PE, Sişu AM et al (2011) Cephalometric investigation of Class III dentoalveolar malocclusion. Rom J Morphol Embryol 52:1343–1346
Tan CY, Qi H, Zhang AQ et al (2012) The evaluation of nuchal translucency abnormality in 115 cases. Chin J Prac Gynecol Obstet 28:56–58
Trevizan M, Consolaro A (2017) Premaxilla: an independent bone that can base therapeutics for middle third growth! Dental Press J Orthod 22:21–26. https://doi.org/10.1590/2177-6709.22.2.021-026.oin
Wu BH, Kou XX, Zhang C et al (2017) Stretch force guides finger-like pattern of bone formation in suture. PLoS ONE 12:e177159. https://doi.org/10.1371/journal.pone.0177159
Xue F, Wong RW, Rabie AB (2010) Genes, genetics, and Class III malocclusion. Orthod Craniofac Res 13:69–74. https://doi.org/10.1111/j.1601-6343.2010.01485.x
Zhang WJ, Fang JX (2000) Embryonic development for the jaws. J Linyi Med Coll 22:161–162
Zhang WJ, Ma YZ, Meng QL et al (1999) Observation of primary ossification center of fetal cranium viscerale. J Linyi Med Coll 21:5–6
Acknowledgements
This study was funded by the key project of the medical science and technology plan of Zhejiang province (2012ZDA028).
The authors are very grateful to James L. Borke, Professor and Assistant Dean of the College of Dental Medicine, Western University of Health Sciences, Pomona, CA, USA and Raymond Wong, Professor of Pediatric Dentistry from California, San Francisco, CA, USA, for reviewing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
W.-H. Ruan, W.-Q. Han, M.-l. Huang, K.-l. Huang and L.-l. Jin declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Ruan, Wh., Han, Wq., Huang, Ml. et al. Premaxillary–maxillary suture development in the first trimester. J Orofac Orthop 80, 25–31 (2019). https://doi.org/10.1007/s00056-018-0163-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00056-018-0163-z