Abstract
The naturally fragmented habitat on the tallest African mountains provides a good model system to study vicariance, dispersal, and hybridization. Many mountains are separated by lowland that likely was unsuitable for high-alpine plants even during cold climatic periods. We explore the relative importance of these processes using two endemic sister species: the widespread Ethiopian/eastern East African Carex monostachya and the mainly western East African C. runssoroensis. These bog-forming sedges co-occur in some mountains and are hypothesized to hybridize. The two species were distinctly differentiated for genome-wide amplified fragment length polymorphisms (AFLPs), also in one mountain where they co-occur. However, the plants from another mountain showed strong signals of admixture. The results suggest initial divergence into one western and one northern/eastern lineage, followed by long-distance dispersal resulting in secondary contact zones. In addition within species genetic diversity was clearly structured with distinct genetic groups on some, but not all mountains. Differentiation levels varied considerably and did not always correspond to the extent of lowland habitat between mountains. The narrow Rift Valley in the otherwise nearly contiguous highlands in Ethiopia appears to present a much stronger barrier to dispersal than the extensive lowlands separating Ethiopia from East Africa. This may be a general pattern since it has been documented also for other afro-alpine species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
All species evolve in a particular geographic context. The geographic template interacts with species traits such as dispersal ability and habitat requirements to shape biodiversity. Naturally fragmented habitats such as altitudinal zones in alpine areas are good model systems to study the relative importance of vicariance, dispersal, secondary contact and hybridization in the evolution of plant taxa. Past climate fluctuations have caused altitudinal shifts of vegetation zones resulting in species’ range expansions or contractions and in some cases connectivity between distributed fragments which presently are separated by barriers of unsuitable habitat (Ehrich et al. 2007; Kebede et al. 2007). The eastern African high mountains provide an extreme example of such a fragmented system and have attracted the interest of generations of biogeographers. The tropical afro-alpine flora offers famous examples of peculiar life forms, local endemism, and striking biogeographic connections to remote parts of the world (Hedberg 1957, 1965; Knox and Palmer 1995, 1998; Koch et al. 2006; Assefa et al. 2007; Ehrich et al. 2007; Popp et al. 2008; Gehrke and Linder 2009). Repeated long-distance colonizations and subsequent radiations seem to have played a central role in the history of this flora, which appears to be the youngest of all African floras (Plio-Pleistocene origin; Gehrke and Linder 2014; Linder 2014).
Little is, however, known about the phylogeographic processes operating within the afro-alpine system. Many of the mountains are separated by vast areas of lower altitude habitat unsuitable for plants adapted to high-alpine conditions, creating a landscape of “sky-islands” (Hedberg 1964, 1969). In such a geographic context, it is likely that vicariant evolution after initial colonization of single mountains or mountain groups results in distinct genetic differentiation. Population divergence may be predicted to increase with increasing distance across unsuitable habitat between mountains, resulting in a pattern of isolation by distance. Stochasticity in the colonization process can, however, lead to different patterns. Moreover, intermountain dispersal leading to secondary contact or even hybridization between species and between different intraspecific lineages may also be important and depend on the nature and the relative strength of dispersal barriers. Tropical high-alpine plant species are most interesting in this respect since their populations probably were isolated from each other also during cold periods of the Pleistocene, when the afro-alpine habitat extended to lower altitudes than today (Flenley 1979; Bonnefille et al. 1990).
Some recent studies based on AFLP and/or plastid DNA sequence variation have explored phylogeographic patterns in species that are typical of the uppermost ericaceous and alpine vegetation zones. They revealed cases of distinct genetic differentiation into two or more lineages with no evidence of secondary contact, suggesting a long history of isolation after initial colonization of different mountains or mountain groups (Erica trimera, Gizaw et al. 2013; Deschampsia cespitosa, Masao et al. 2013). Other species, on the contrary, show no geographic structuring of their genetic diversity at all, suggesting recent colonization of several mountains from a single source and/or extensive recent dispersal (Erica arborea, Gizaw et al. 2013; Koeleria capensis, Masao et al. 2013) and indicating that intermountain gene flow may be more common than previously thought. Others still not only show distinct genetic structuring, but also clear-cut episodes of intermountain dispersal followed by hybridization between divergent lineages (Wondimu et al. 2014). Finally, the strength of barriers seems to differ among species, resulting in different geographic structuring of their genetic diversity. Most surprising is that the narrow Rift Valley that cuts through the otherwise nearly contiguous highland in Ethiopia may correspond to stronger genetic differentiation than the extensive area of unsuitable lowland habitat that separates the Ethiopian mountains from the East African ones (Wondimu et al. 2014). The most striking result overall is thus that the inferred species histories are very different from each other. Therefore, more case studies of species with different habitat requirements and dispersal ability are needed to be able to develop a general phylogeographic model for the afro-alpine flora.
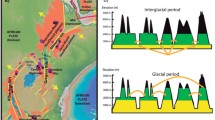
Here we selected two endemic and ecologically important sister species of sedges. Carex monostachya A. Rich. is widespread in Ethiopia and eastern East Africa, whereas C. runssoroensis K. Schum. has a mainly western range in East Africa (Fig. 1). They evolved after one of at least 13 independent colonizations of the afro-alpine region by Carex (Gehrke and Linder 2009) and are most closely related to the circumarctic/circumboreal C. capitata L. (Starr and Ford 2009; Gehrke 2011). Carex monostachya is characterized by a distinctly triangular culm (at least just below the inflorescence), long (up to 30 cm) leaves, and a utricle with scabrid hairs, whereas C. runssoroensis has a terete (round) culm, undeveloped or up to 5 cm long leaves, and a glabrous or sparsely scabrid utricle (Verdcourt 2010; Gehrke 2011). The two species grow in similar, moist habitats and often dominate the vegetation in bogs and swamps. Both typically form large, dense tussocks up to 1.5 m high, but can also grow as creeping, rhizomatous perennials (Hedberg 1957; Haines and Lye 1983; Verdcourt 2010; Gehrke 2011). In East Africa, they mainly occur in the uppermost afro-alpine zone and sometimes in the ericaceous belt, spanning (2400–) 2750–4300 (–4500) m altitude (Hedberg 1957, 1964; Gehrke 2011). In Ethiopia, C. monostachya is only reported from the alpine zone proper (3750–4200 m; Lye 1997; occasionally down to 3400 m according to material in the National Herbarium of Ethiopia). Carex runssoroensis is most common in mountains along the western branch of the Rift Valley (Ruwenzori and Virunga Mts), but it has also been reported to extend eastwards to some mountains along the eastern branch of the Rift (Elgon/Cherangani Hills, Aberdare and Kenya; Fig. 1; Verdcourt 2010; Gehrke 2011). The plants in Mt Aberdare and Mt Kenya have been described as a distinct variety, var. aberdarensis, because of their thinner culms, hyaline (as opposed to brown/blackish) margin of the pistillate scales, and creeping (as opposed to densely tufted) habit (Verdcourt 2010; Gehrke 2011). Carex monostachya is reported both from Ethiopia (where it has its main occurrences in the Simen and Bale Mts) and from the eastern Rift mountains in East Africa (Kilimanjaro, Aberdare, Kenya, and Elgon; Hedberg 1957; Lye 1997; Verdcourt 2010; Fig. 1).
a Total geographic distribution of the afro-alpine endemics C. monostachya (yellow) and C. runssoroensis (green) based on Hedberg (1957), Haines and Lye (1983), Lye (1997), Verdcourt (2010), Gehrke (2011), and herbarium records from the National Herbarium of Ethiopia, Addis Ababa University (ETH), Ethiopia. The two species have been reported to co-occur in three mountain areas (Mt Elgon including the adjacent Cherangani Hills, Mt Kenya and Mt Aberdare; shown in both yellow and green). b Sampling sites for this study and geographic distribution of the three main genetic groups inferred from Bayesian clustering of the AFLP data (light gray the Simen group in C. monostachya; black the Bale/EA group in C. monostachya; white C. runssoroensis). In Ethiopia, we sampled the two areas that harbor the main occurrences of C. monostachya (Simen Mts and Bale Mts). Note that we did not succeed in locating C. runssoroensis in Mt Kenya, where it has been reported as rare (Gehrke 2011)
Hybridization between the two species has been suggested based on morphology in the three mountains where they are reported to co-occur (Elgon, Aberdare, and Kenya). Haines and Lye (1983) described and illustrated putative hybrids. Because of continuous variation in morphological characters they suggested that all plants occurring on Mt Elgon may belong to a hybrid swarm between the two species. Verdcourt (2010) tentatively accepted their hybrid hypothesis for Mt Elgon and also accepted C. runssoroensis var. aberdarensis as a distinct taxon endemic to Mt Aberdare and Mt Kenya. He stated that further studies are needed ‘to consider these hybrids in conjunction with true nature of var. aberdarensis’, implicitly suggesting that the plants on Mt Aberdare and Mt Kenya also may represent hybrids (see also Gehrke 2011).
For this study, we sampled C. monostachya and C. runssoroensis across most of their ranges (Fig. 1) and used genome-wide amplified fragment length polymorphisms (AFLPs) to explore to what degree genetic diversity is structured according to individual mountains, to assess whether the strength of barriers between mountains increases with increasing extent of unsuitable habitat separating them, and to identify potential episodes of intermountain dispersal, secondary contact, and admixture. In particular, we test whether there is a genetic signature of hybridization in the Mt Elgon plants as suggested by Haines and Lye (1983), and whether C. runssoroensis var. aberdarensis in the eastern mountains also represents an interspecific hybrid (cf. Verdcourt 2010). We assume that the reported co-occurrence of the two species in three eastern mountains resulted from dispersal of C. runssoroensis from its main western range towards the east, and thus represent secondary contact zones. If range expansion was recent, we predict that the eastern populations of C. runssoroensis have low levels of genetic distinctiveness and harbor little diversity because of colonization after long-distance dispersal. Moreover, as both of these sister species are wind-pollinated, lack obvious adaptations to long-distance dispersal, and have similar habitat requirements, we expect to find a similar degree of genetic structuring in the two.
Materials and methods
Sampling
Carex monostachya and C. runssoroensis were collected from seven high mountains/mountain systems in Ethiopia, Kenya, Tanzania and Uganda between 2007 and 2009 (Fig. 1, “Appendix 1”). Whenever possible, four spatially separated 100 m × 100 m plots were selected on each mountain and fresh leaf tissue from five individual plants, referred to as a population sample, was collected from each plot and dried in silica gel. Three of the five plants collected from each population were pressed and deposited in the following herbaria: one in the National Herbarium of Ethiopia, Addis Ababa University (ETH), Ethiopia; one in the Natural History Museum, University of Oslo (O), Norway; and the third voucher was deposited according to the country of collection, i.e., in the East African Herbarium (EA), Kenya; Makerere University Herbarium (MHU), Uganda; or in the National Herbarium of Tanzania (NHT), Tanzania. In Ethiopia, we selected the two areas that harbor the main occurrences of C. monostachya (Simen Mts and Bale Mts). For logistical reasons, we were not able to collect in some smaller mountains in Ethiopia or in the western Virunga Mts in East Africa (Fig. 1). In Mt Elgon, we collected two closely situated populations, of which one (KN0396) was morphologically intermediate between the species and one (KN0391) was morphologically most similar to C. runssoroensis. Since both of them showed a genetic signature of admixture (see Results) we designate them as putative hybrids in the following. The remaining material consisted of 19 populations identified as C. monostachya and seven populations identified as C. runssoroensis based on morphology.
AFLP analysis
Total genomic DNA was extracted from silica gel-dried leaf tissue using an automated GeneMole® robot and the MoleStrip™ Plant DNA Mini Kit following the manufacturer’s instructions (QIAGEN Nordic, Oslo, Norway). The quality of extracted DNA was assessed on 1 % TBE-agarose gels. AFLP profiles were generated following Gaudeul et al. (2000), except that the PCR reaction volumes were reduced by 50 % and pre-selective PCR products were diluted ten times. An initial screening of 24 combinations of selective primers was performed on eight individuals of each species from different geographical regions. Three primer combinations resulting in AFLP profiles with many polymorphic and well-separated fragments were selected for full analysis of the total data set: 6FAM-EcoRI-ATG/MseI-CG, VIC-EcoRI-AAG/MseI-CC, NED-EcoRI-AAC/MseI-CG.
For each sample, 2 μL 6-FAM, 2 μL VIC and 3 μL NED labeled selective PCR products were diluted in 14 μL distilled water. A total volume of 3.5 μL of the diluted reaction was mixed with 11.7 μL formamide and 0.3 μL GeneScan™ 500 ROX™ internal-lane size standard and denatured at 95 °C for 5 min, cooled on ice before run on ABI 3100 sequencer (Applied Biosystems). The raw data were analyzed using the ABI prism GeneScan analysis software version 3.7 (Applied Biosystems) and imported for scoring into GeneMapper version 4.0 (Applied Biosystems). AFLP fragments in the size range 50–500 base pairs (bp) were automatically scored as present (1) or absent (0). To assess reproducibility, nine samples were extracted twice and the AFLP procedure was repeated. Reproducibility (1-error rate) was estimated as the average proportion of correctly replicated bands (Bonin et al. 2004). Markers with low reproducibility were discarded.
Data analyses
Genetically homogenous groups in the total dataset were inferred using the Bayesian clustering algorithm implemented in STRUCTURE v 2.3.3 (Pritchard et al. 2000). We used the recessive allele model to take into account the dominant nature of AFLP data (Falush et al. 2007) together with an admixture model with correlated allele frequencies as well as the no admixture model with uncorrelated allele frequencies. Based on the results of preliminary analyses we selected the admixture model with correlated allele frequencies for the final analyses. The analyses were carried out at the Lifeportal, University of Oslo (http://www.lifeportal.uio.no) for K = 1–10 and 10 replicate runs for each K, using a burn-in period of 2 × 105 and 106 iterations. We used the R-script STRUCTURE-SUM (Ehrich 2007) to summarize the results and determine the most likely value of K (i.e., number of genetic groups) based on the estimated posterior log likelihood, L(K), and the similarity among different runs for the same K (Nordborg et al. 2005). As additional selection criterion we calculated the rate of change in the likelihood between successive runs, ∆K, as a function of K (Evanno et al. 2005). The average estimate of individual admixture across the replicated runs for the selected K was calculated using the program CLUMPP (Jakobsson and Rosenberg 2007) and the result was graphically visualized using the program DISTRUCT (Rosenberg 2004). Each individual was assigned to a certain genetic group if the proportion of ancestry in this group (q) was higher than 90 %, and to a hybrid between genetic groups if 10 % < q < 90 %. We considered symmetric assignments around q = 50 % to be consistent with first generation (F1) hybrids; otherwise with later generation hybrids. The widths of 90 % posterior probability intervals (PI) for q were also taken into account. Genetic variation within and between populations and groups of populations was assessed using Analyses of Molecular Variance (AMOVA) in ARLEQUIN version 3 (Excoffier and Schneider 2005). Significance of differentiation was evaluated with 1000 permutations.
Pairwise genetic similarity among AFLP multilocus genotypes was calculated using Dice’s coefficient in NTSYSpc version 2.11a (Rohlf 2000) and visualized using Principal Coordinate Analysis (PCoA). In addition, a Neighbor-Net diagram (Bryant and Moulton 2004) based on uncorrected p-distance was constructed using SplitsTree4 version 4.12.6 (Huson and Bryant 2006) and support for branches was estimated from 1000 bootstrap replicates using TreeCon version 1.3b (Van de Peer and De Wachter 1994). Gene diversity estimates were obtained for each species in total, for each population, for each mountain, and for genetic groups identified by STRUCTURE. We used the R-script AFLPdat (Ehrich 2006) to calculate the proportion of polymorphic markers (P) and Nei’s gene diversity (D; estimated as the average proportion of pairwise differences among genotypes; Kosman 2003). Genetic distinctiveness or rarity was quantified as frequency-down-weighted marker values (DW) according to Schönswetter and Tribsch (2005) using AFLPdat (Ehrich 2006). DW values were obtained for each population and mountain both with reference to the respective species and with reference to the total dataset comprising the two species and their putative hybrid. Pairwise differentiation among the identified genetic groups and the two putative hybrid populations was quantified by F ST (equivalent to ΦST; ARLEQUIN version 3.0; Excoffier and Schneider 2005) and levels of significance were estimated from 1000 permutations. For each species (excluding hybrids), we tested for increased differentiation with geographic distance performing a Mantel tests (Mantel 1967). We used Slatkin’s linearized F ST (estimated as F ST/(1 − F ST), Rousset 1997) and the natural logarithm of geographic distance, and carried out 1000 permutations in ARLEQUIN version 3.0 (Excoffier and Schneider 2005).
Results
The final dataset consisted of 120 plants successfully analyzed for AFLPs, including 84 plants (19 populations) of C. monostachya, 29 plants (7 populations) of C. runssoroensis, and 7 plants (2 populations) of the putative hybrid from Mt Elgon. The three primer combinations resulted in a total of 262 markers, of which 95.2 % were polymorphic. Monomorphic markers were removed before further analyses. Reproducibility as estimated from the nine duplicated individuals was 95.5 %.
The STRUCTURE analyses did not converge on one solution for K = 2. However, the result for K = 2 with highest L(K) clearly separated the two species and indicated mixed ancestry for both populations from Mt. Elgon. Both mean L(K) and ∆K indicated K = 3 as the optimal number of groups (Figs. 1, 2a). This was corroborated by the high correlation coefficient between replicated runs for K = 3. One group consisted of all C. runssoroensis samples (Mt Aberdare and Mt Ruwenzori). The samples of C. monostachya were divided into two genetic groups, one confined to the Simen Mts in northwestern Ethiopia (the Simen group) and one which was widespread, occurring in the Bale Mts in southeastern Ethiopia as well as in the East African mountains Mt Kenya, Mt Aberdare, and Mt Kilimanjaro (the Bale/EA group). As was the case for K = 2, all plants collected from Mt Elgon showed mixed ancestry and were assigned partially to the C. runssoroensis genetic group and partially to the Bale/EA group of C. monostachya (Figs. 1, 2a).
a Genetic structuring inferred from Bayesian clustering of the AFLP data in the total dataset comprising 84 plants (19 populations) of C. monostachya, 29 plants (7 populations) of C. runssoroensis, and 7 plants from two putative hybrid populations. The graphs to the left show log likelihood, L(K), as a function of K ranging from 1 to 10 (top), the rate of change in the probability between successive runs, ∆K, as a function of K (calculated according to Evanno et al. 2005; middle), and average similarity coefficients for the pairwise comparisons among 10 runs for a given K (calculated according to Rosenberg et al. 2002; bottom). A graphical representation of the assignment pattern for K = 3 is shown to the right, with shading identifying the genetic groups (see Fig. 1) and symbols identifying the mountains. b Principal Coordinates Analysis (PCoA) based on Dice’s coefficient of similarity between the AFLP phenotypes observed in the total dataset. Shading and symbols as in a. c Neighbor-Net diagram based on uncorrected p-distance computed among the AFLP phenotypes observed in the total dataset. Branch support was estimated using 1000 bootstrap replicates. Shading and symbols as in a
The assignment patterns differed between the two putative hybrid populations from Mt Elgon. The four plants from the KN0396 population, which were recognized as intermediate between the two species based on morphology, showed a symmetric pattern of genetic admixture between the Bale/EA group and the C. runssoroensis group, with the 90 % PI for q including 0.5 for all individuals. The three plants from the KN0391 population, which were morphologically most similar to C. runssoroensis, showed an asymmetric pattern of admixture with ~70 % of the genome assigned to the C. runssoroensis group and ~30 % (90 % PI excluded 0.5 for all three individuals) assigned to the Bale/EA group of C. monostachya. Apart from these Mt Elgon populations, we observed very little admixture in the dataset (a few plants from Simen Mts, Bale Mts, and Mt Kilimanjaro; Fig. 2a).
Both the PCoA and the Neighbor-Net analyses clearly distinguished the two species and the two distinct phylogeographic groups in C. monostachya, and supported the inference of mixed ancestry of the Mt Elgon populations (Fig. 2b, c). The first two axes of the PCoA separated the three genetic groups identified by STRUCTURE, with a major split along the first axis (explaining 17.6 % of variation) separating C. runssoroensis from C. monostachya. The second axis (13.0 %) separated the Simen group from the Bale/EA group of C. monostachya. The third axis (10.7 %) revealed a subdivision of the C. runssoroensis group into two distinct subgroups corresponding to Mts Ruwenzori and Aberdare. In accordance with the STRUCTURE analysis, the putative hybrids from Mt Elgon were placed between C. runssoroensis and C. monostachya along the first PCoA axis, with the KN0391 population closest to C. runssoroensis (Fig. 2b). In the Neighbor-Net analyses, the C. monostachya samples formed a distinct cluster with bootstrap support of 94 % (Fig. 2c). Within this cluster, the Bale/EA group and the Simen group were separated as two clusters with bootstrap support of 70 and 96 %, respectively. The C. runssoroensis samples were divided into two distinct clusters with high bootstrap support (99–100 %), corresponding to Mts Aberdare and Ruwenzori. The putative hybrids from Mt Elgon were placed between the two species, but clustered with C. runssoroensis (bootstrap support 94 %).
Total genetic diversity when pooling all genotyped individuals was 0.150 in C. monostachya, 0.137 in C. runssoroensis, and 0.117 in the putative hybrids (“Appendix 1”). Average within-population gene diversity was similar in C. monostachya (0.089 ± 0.021) and the putative hybrid (0.088 ± 0.026), but lower in C. runssoroensis (0.049 ± 0.017; “Appendix 1”). When pooled by mountain, C. monostachya had highest gene diversity in Mt Kilimanjaro and Mt Aberdare (0.118–0.123) and lowest in the Bale Mts (0.082; Table 1). Carex runssoroensis had higher diversity in Mt Ruwenzori (0.070) than in Mt Aberdare (0.039). Average genetic rarity calculated for the total dataset (DWtotal; Table 1) was highest in the putative hybrid and in the Aberdare populations of both species, and lowest in the Bale and Kenya populations of C. monostachya. In the separate estimation for each species (DWseparate), genetic rarity in C. monostachya increased with increasing gene diversity, and in C. runssoroensis, the least diverse Aberdare populations showed higher genetic rarity than the Ruwenzori populations. A total of 94 AFLP markers were private to C. monostachya, 22 to C. runssoroensis and 10 to the putative hybrid (Fig. 3; Table 1). Of the ten markers private to the putative hybrid, two were fixed or shared between populations and four were private to a single population. The putative hybrid uniquely shared 19 markers with C. monostachya and six with C. runssoroensis. The Simen populations of C. monostachya harbored many more private markers (29) than the populations in other mountains (4–13; Table 1).
Pair-wise comparisons of private and shared AFLP markers observed in C. monostachya, C. runssoroensis and their putative hybrids (see Table 1 for details)
The non-hierarchical AMOVAs assigned 41.86 and 67.58 % of the total variation to among-population variation in C. monostachya and C. runssoroensis, respectively (Table 2). In hierarchical AMOVAs, a larger proportion of genetic variation was found among the three genetic groups (37.81 %) than between the two described species (32.96 %). In C. monostachya, 33.71 % of the variation was assigned to between-group variation (Bale/EA vs Simen). The proportion of variation assigned to among-mountain variation was much lower in the Bale/EA group of C. monostachya (22.27 %) than in C. runssoroensis (71.86 %). Less variation was assigned to between-group variation when comparing the putative hybrids with C. runssoroensis (23.10 %) than with the Bale/EA group of C. monostachya (30.31 %; Table 2). The pairwise F ST values indicated stronger differentiation between the two species than between the two genetic groups in C. monostachya (Table 3). The two hybrid populations were less differentiated from the Bale/EA group than from the Simen group of C. monostachya, and they were strongly differentiated from each other. In both species, the Mantle tests revealed a significant pattern of isolation by distance (C. monostachya: regression coefficient b = 0.132, P < 0.001; C. runssoroensis: b = 0.614, P = 0.004).
Discussion
Our results provide new insights into the history of the plants growing in the highly fragmented and fascinating, but understudied ecosystem restricted to the highest mountains in eastern Africa. In an ecologically important group of sedges, we found that vicariant evolution after initial colonization has resulted in divergent lineages in some, but not all mountains. We also found that the two sister species are clearly differentiated genetically and form secondary contact zones, of which at least one has led to interspecific hybridization. This has most likely happened after long-distance dispersal across a vast area of unsuitable habitat (the Ugandan lowland gap, ~550 km of less than 1500 m altitude).
In the most widespread species, C. monostachya, we found no clear relationship between the degree of genetic differentiation among mountains and the extent of unsuitable lowland habitat separating them, although the overall genetic structure, consisting of geographically co-localized genetic groups, resulted in an overall pattern of isolation by distance. We identified only two distinct genetic groups in this species, one confined to a single mountain range (the Simen Mts in Ethiopia) and one shared among four geographically well-separated mountain systems (the Bale Mountains in Ethiopia and three eastern East African mountains; Fig. 1). This suggests that the narrow Rift Valley cutting through the Ethiopian Highlands represents a strong barrier although it only spans a short geographic distance (<75 km) between two areas that both probably provided more or less continuous, extensive alpine habitat during cold climatic periods (~80 % of the highlands >3000 m; Yalden 1983; Gottelli et al. 2004). The Rift Valley bottom is situated at 750 m of altitude in the northeast, progressively raises southwestwards to 1700 m, and then drops to 1100 m in the south (Chorowicz 2005). Thus, even if the high-alpine habitat may have extended as much as 1000 m downwards during cold periods (Moreau 1963; Coetzee 1964; Flenley 1979; Jacob et al. 2015), the Rift Valley in Ethiopia may have acted as an important biogeographic barrier by preventing gradual migration of high-alpine organisms (Arctander et al. 1999; Gottelli et al. 2004).
In contrast, the East African mountains are separated from the Ethiopian Highlands by a vast lower lying gap (~830 km between the Bale Mts and Mt Kenya), but this gap seems to represent a much weaker barrier to dispersal for Carex. A large part of the gap, which also is believed to have served as an efficient barrier against gradual migration of afro-alpine plants also during the Pleistocene cold periods (Hedberg 1969, 1986), is situated at an altitude of ~1000 m (~250 km; Brühl 1997). In addition the eastern East African mountains are separated by large lower lying areas at altitudes of 1000–2000 m, but these gaps seem neither to constitute strong barriers against gene flow in C. monostachya (Figs. 1, 2).
In eastern Africa, studies of sediment cores have demonstrated cycles of varying temperature and humidity, and there is evidence for extensive earlier glaciations on some mountains (e.g., Bale Mts, Mt Kilimanjaro, Mt Kenya; Van Zinderen Bakker 1962; Mohammed and Bonnefille 1998; Trauth et al. 2005). During the Pleistocene, the northern ice ages corresponded more or less to cold and dry periods in the African tropics (Bonnefille et al. 1990) and the present vegetation belts of the afro-alpine region may have been pushed down by 1000 m (Moreau 1963; Flenley 1979). Increased aridity may, however, have limited expansion of plants growing in moist habitats. Gradual migration of the two moisture-demanding sedges between mountains during cold and dry periods seems therefore unlikely. The fact that these sedges do occur in many isolated mountains can therefore be taken as evidence for high long-distance dispersal ability, in spite of their lack of obvious morphological adaptations to dispersal. The finding of secondary contact zones between them provides additional evidence for high dispersal ability. We therefore conclude that distinct genetic differentiation among the populations of C. monostachya occurring in four widely separated mountain systems likely has been prevented by high levels of gene flow mediated by long-distance dispersal, possibly by strong winds or birds as suggested by Hedberg (1969). It is also possible that these mountains were colonized only very recently from a single source population, but this explanation also necessitates long-distance dispersal.
It is therefore difficult to explain why gene flow has not prevented the strongly vicariant pattern observed between the C. monostachya populations from the opposite sides of the narrow Rift Valley in Ethiopia. Notably, this pattern has also been reported in two other afro-alpine species, of which one is clearly adapted to long-distance dispersal by wind (Wondimu et al. 2014), and it may thus represent a common pattern. Another common pattern seems to be that the western and eastern mountains in East Africa tend to harbor populations that are genetically highly divergent, but that occasional dispersal followed by admixture does occur across the vast Ugandan lowland gap separating them (this study; Mugizi et al. submitted; Gizaw et al. unpublished data).
Vicariant speciation (in the sense that species originate as a result of isolation of populations in different mountains) is generally considered to be a prominent process shaping afro-alpine plant diversity (Hedberg 1957, 1969; Knox and Palmer 1995, 1998). The two sister species of sedges studied here probably also originated in different mountains; origin by divergence within a single mountain is unlikely because they have similar habitat requirements and often dominate in similar, moist vegetation types. Since C. runssoroensis is most abundant and harbors most genetic diversity and private markers in the west (Mt Ruwenzori), a reasonable hypothesis is that it originated there and later spread across the vast Ugandan gap into the range of the eastern C. monostachya. This resulted in secondary contact zones in three eastern mountains, of which at least one resulted in hybridization (Figs. 1, 2). We found, however, no evidence for recent range expansion of C. runssoroensis. Its eastern populations in Mt Aberdare were distinctly differentiated from the western ones and contained considerable genetic rarity and private markers (Fig. 3; Table 1), suggesting that the expansion took place a long time ago. This is consistent with the finding of considerable genetic rarity and private markers in the hybrids from Mt Elgon, although this alternatively can be explained by under-sampling of the variation in the parental species.
Our data provided strong evidence for hybridization between the two species. The plants from Mt Elgon showed clear signals of genetic admixture as predicted from the hypothesis of Haines and Lye (1983). All individuals in our sampling from Mt Elgon were admixed and seem to represent either F1 hybrids (KN0396) or backcrosses to C. runssoroensis (KN0391). This suggests that hybrids between the two species are at least partly fertile, as frequently reported for other species pairs in Carex (e.g., Reznicek 1990; Choler et al. 2003; Volkova et al. 2008; Korpelainen et al. 2010). We found, however, no evidence of hybrid origin of the plants from Mt Aberdare assigned to C. runssoroensis var. aberdarensis (Fig. 2; cf. Verdcourt 2010; Gehrke 2011). It is possible that more extensive sampling on this mountain would reveal hybridization between the two species occurring there. A more thorough study is also needed to assess whether ‘pure’ populations of the two species occur on Mt Elgon in addition to the hybrid populations, and if so whether the two species sometimes grow in mixed populations.
Our study supports recognition of two distinct and allopatric infraspecific taxa in C. runssoroensis, as originally suggested by Kükenthal (1909) and later accepted by Hedberg (1957) and Verdcourt (2010) at the variety level (var. runssoroensis and var. aberdarensis). Given the distinct genetic divergence we observed between them it seems reasonable to recognize them at the subspecies level, but more work is necessary to test whether they can be consistently separated based on morphological characters. There are different opinions on whether they can be separated based on habit (tussock-forming vs mat-forming with creeping rhizomes; compare Hedberg 1957; Verdcourt 2010), but this may turn out to vary within both of them. In Carex nigra, for example, it was recently shown that populations forming conspicuous tussocks and often referred to as a distinct species, rather represent an ecotype that has originated repeatedly from different populations with creeping rhizomes (Jiménez-Mejías et al. 2012). A morphological analysis is also necessary to assess whether the two distinct genetic groups we observed within C. monostachya should be recognized taxonomically. This species was originally described based on material from the Simen Mountains in Ethiopia, whereas material from Mt Kilimanjaro in East Africa has been described as a distinct species, C. triquetrifolia Boeck. (Verdcourt 2010).
In conclusion, this study adds to a growing body of evidence demonstrating a high diversity of phylogeographic patterns in the high-alpine flora of Africa (Gizaw et al. 2013; Masao et al. 2013; Wondimu et al. 2014). Such diversity may arise because of characteristics of dispersal vectors or differences between species in, e.g., dispersal ability, habitat requirement, or time since first arrival to eastern Africa and to different mountains. The diversity may also to some degree have arisen by chance as a result of long-distance colonization, which is a highly stochastic process (Nathan 2006). A well-structured comparative study of many extensively sampled species is clearly required to identify the relative importance of the various processes shaping biodiversity in this extremely fragmented sky-island system.
References
Arctander P, Johansen C, Coutellec-Vreto MA (1999) Phylogeography of three closely related African bovids (tribe Alcelaphini). Mol Biol Evol 16:1724–1739
Assefa A, Ehrich D, Taberlet P, Nemomissa S, Brochmann C (2007) Pleistocene colonization of afro-alpine ‘sky islands’ by the arctic-alpine Arabis alpina. Heredity 99:133–142
Bonin A, Bellemain E, Eidesen PB, Pompanon F, Brochmann C, Taberlet P (2004) How to track and assess genotyping errors in population genetics studies. Mol Ecol 13:3261–3273
Bonnefille R, Roeland JC, Guiot J (1990) Temperature and rainfall estimates of the past 40000 years in equatorial Africa. Nature 346:347–349
Brühl C (1997) Flightless insects: a test case for historical relationships of African mountains. J Biogeogr 24:233–250
Bryant D, Moulton V (2004) NeighborNet: an agglomerative algorithm for the construction of phylogenetic networks. Mol Biol Evol 21:255–265
Choler P, Erschbamer B, Tribsch A, Gielly L, Taberlet P (2003) Genetic introgression as a potential to widen a species’ niche: insights from the alpine Carex curvula. Proc Natl Acad Sci USA 101:171–176
Chorowicz J (2005) The East African rift system. J Afr Earth Sci 43:379–410
Coetzee JA (1964) Evidence for a considerable depression of the vegetation belts during the upper Pleistocene on the East African mountains. Nature 204:564–566
Ehrich D (2006) AFLPdat: a collection of R functions for convenient handling of AFLP data. Mol Ecol Notes 6:603–604
Ehrich D (2007) Structure-sum v. 2007: a series of R functions for summarizing the outputs of the program structure ver. 2.2. (Unpublished, available from the author upon request)
Ehrich D, Gaudeul M, Assefa A, Koch MA, Mummenhoff K, Nemomissa S, Brochmann C (2007) Genetic consequences of Pleistocene range shifts: contrast between the Arctic, the Alps and the East African mountains. Mol Ecol 16:2542–2559
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620
Excoffier LLG, Schneider S (2005) ARLEQUIN version 3.0: an integrated software package for population genetics data analysis. Evol Bioinform Online 1:47–50
Falush D, Stephens M, Pritchard J (2007) Inference of population structure using multilocus genotype data: dominant markers and null allele. Mol Ecol Notes 7:574–578
Flenley JR (1979) The Equatorial rain forest: a geological history. Butterworths, London
Gaudeul M, Taberlet P, Till-Bottraud I (2000) Genetic diversity in an endangered alpine plant, Eryngium alpinum L. (Apiaceae), inferred from amplified fragment length polymorphism markers. Mol Ecol 9:1625–1637
Gehrke B (2011) Synopsis of Carex (Cyperaceae) from sub-Saharan Africa and Madagascar. Bot J Linn Soc 166:51–59
Gehrke B, Linder HP (2009) The scramble for Africa: pan-temperate elements on the African high mountains. Proc R Soc Lond, Ser B: Biol Sci 276:2657–2665
Gehrke B, Linder HP (2014) Species richness, endemism and species composition in the tropical Afroalpine flora. Alp Bot 124:165–177
Gizaw A, Kebede M, Nemomissa S, Ehrich D, Bekele B, Mirré V, Popp M, Brochmann C (2013) Phylogeography of the heathers Erica arborea and E. trimera in the afro-alpine ‘sky islands’ inferred from AFLPs and plastid DNA sequences. Flora 208:453–463
Gottelli D, Marino J, Sillero-Zubiri C, Funk SM (2004) The effect of the last glacial age on speciation and population genetic structure of the endangered Ethiopian wolf (Canis simensis). Mol Ecol 13:2275–2286
Haines RW, Lye KA (1983) The sedges and rushes of East Africa. East African Natural History Society, Nairobi
Hedberg O (1957) Afroalpine vascular plants. A taxonomic revision. Symb Bot Ups 15:1–411
Hedberg O (1964) Features of Afroalpine plant ecology. Almqvist and Wiksells, Uppsala
Hedberg O (1965) Afroalpine flora elements. Webbia 19:519–529
Hedberg O (1969) Evolution and speciation in a tropical high mountain flora. Bot J Linn Soc 1:135–148
Hedberg O (1986) Origin of the afroalpine flora. In: Vuilleumier F, Monastero M (eds) High altitude tropical biogeography. Oxford University Press, New York, pp 443–468
Huson DH, Bryant D (2006) Application of phylogenetic networks in evolutionary studies. Mol Biol Evol 23:254–267
Jacob M, Annys S, Frankl A, De Ridder M, Beeckman H, Guyassa E, Nyssen J (2015) Tree line dynamics in the tropical African highlands—identifying drivers and dynamics. J Veg Sci 26:9–20
Jakobsson M, Rosenberg NA (2007) CLUMPP: a cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics 23:1801–1806
Jiménez-Mejías P, Luceño M, Lye KA, Brochmann C, Gussarova G (2012) Genetically diverse but with surprisingly little geographical structure: the complex history of the widespread herb Carex nigra (Cyperaceae). J Biogeogr 39:2279–2291
Kebede M, Ehrich D, Taberlet P, Nemomissa S, Brochmann C (2007) Phylogeography and conservation genetics of a giant lobelia (Lobelia giberroa) in Ethiopian and Tropical East African mountains. Mol Ecol 16:1233–1243
Knox EB, Palmer JD (1995) Chloroplast DNA variation and the recent radiation of the giant senecios (Asteraceae) on the tall mountains of eastern Africa. Proc Natl Acad Sci USA 92:10349–10353
Knox EB, Palmer JD (1998) Chloroplast DNA evidence on the origin and radiation of the giant Lobelias in Eastern Africa. Syst Bot 23:109–149
Koch MA, Kiefer C, Ehrich D, Vogel J, Brochmann C, Mummenhoff K (2006) Three times out of Asia Minor: the phylogeography of Arabis alpina L. (Brassicaceae). Mol Ecol 15:825–839
Korpelainen H, Virtanen V, H KK Väre (2010) Hybridization and introgression in Carex aquatilis and C. paleacea. Plant Syst Evol 287:141–151
Kosman E (2003) Nei’s gene diversity and the index of average differences are identical measures of diversity within populations. Plant Pathol 52:533–535
Kükenthal G (1909) Cyperaceae-Caricoideae. In: Engler A (ed) Das Pflanzenreich, vol. 4 (21). Wilhelm Engelmann, Leipzig, pp 1–824
Linder HP (2014) The evolution of African plant diversity. Front Ecol Evol 2:1–14
Lye KA (1997) 212. Cyperaceae: 19. Carex. In: Edwards S, Demissew S, Hedberg I (eds) Flora of Ethiopia and Eritrea. Addis Ababa University, Addis Ababa, pp 391–511
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Masao CA, Gizaw A, Piñeiro R, Tusiime FM, Wondimu T, Abdi AA, Popp M, Gussarova G, Lye KA, Munishi P, Nemomissa S, Brochmann C (2013) Phylogeographic history and taxonomy of some afro-alpine grasses assessed based on AFLPs and morphometry: deschampsia cespitosa, D. angusta and Koeleria capensis. Alp Bot 123:107–122
Mohammed MU, Bonnefille R (1998) A late Holocene pollen record from a highland peak at Tamsaa, Bale Mountains, South Ethiopia. Glob Planet Chang 16–17:121–129
Moreau RE (1963) Vicissitides of the African biomes in the late Pleistocene. Proc Zool Soc Lond 141:395–421
Nathan R (2006) Long-distance dispersal of plants. Science 313:786–788
Nordborg M, Hu TT, Ishino Y, Jhaveri J, Toomajian C et al (2005) The pattern of polymorphism in Arabidopsis thaliana. PLoS Biol 3(7):e196
Popp M, Gizaw A, Nemomissa S, Suda J, Brochmann C (2008) Colonization and diversification in the African ‘sky islands’ by Eurasian Lychnis L. (Caryophyllaceae). J Biogeogr 35:1016–1029
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Reznicek AA (1990) Evolution in sedges (Carex, Cyperaceae). Can J Bot 68:1409–1432
Rohlf F (2000) NTSYSpc: Numerical taxonomy and multivariate analysis system. Version 2.11a. Exeter Software, Setauket, New York
Rosenberg NA (2004) DISTRUCT: a program for the graphical display of population structure. Mol Ecol Notes 4:137–138
Rosenberg NA, Pritchard JK, Weber JL, Cann HM, Kidd KK, Zhivotovsky LA, Feldman MV (2002) Genetic structure of human populations. Science 298:2381–2385
Rousset F (1997) Genetic differentiation and estimation of gene flow from F-statistics under isolation by distance. Genetics 145:1219–1228
Schönswetter P, Tribsch A (2005) Vicariance and dispersal in the alpine perennial Bupleurum stellatum L. (Apiaceae). Taxon 54:725–732
Starr JR, Ford BA (2009) Phylogeny and evolution in Cariceae (Cyperaceae): current knowledge and future directions. Bot Rev 75:110–137
Trauth MH, Maslin MA, Deino A, Strecker MR (2005) Late cenozoic moisture history of East Africa. Science 309:2051–2053
Van de Peer Y, De Wachter R (1994) TREECON for Windows: a software package for the construction and drawing of evolutionary trees for the Microsoft Windows environment. Comput Appl Biosci 10:569–570
Van Zinderen Barker EM (1962) A late-glacial and post-glacial climatic correlation between East Africa and Europe. Nature 194:201–203
Verdcourt B (2010) Carex. In: Beentje HJ, Ghazanfar SA (eds) Flora of Tropical East Africa. Richmond Kew Publishing, Kew
Volkova PA, Shipunov AB, Elven R, Brochmann C (2008) The seashore sedges of the Russian Kola Peninsula: how many species? Flora 203:523–533
Wondimu T, Gizaw A, Tusiime FM, Masao CA, Abdi AA, Gussarova G, Popp M, Nemomissa S, Brochmann C (2014) Crossing barriers in an extremely fragmented system: two case studies in the afro-alpine sky island flora. Plant Syst Evol 300:415–430
Yalden DW (1983) The extent of high ground in Ethiopia compared to the rest of Africa. SINET 6:35–38
Acknowledgments
This study was funded by The Norwegian Programme for Development, Research and Higher Education (NUFU) Project No 2007/1058 (AFROALP-II-Afroalpine ‘sky islands’: genetic versus taxonomic biodiversity, climate change, and conservation) to S. Nemomissa and C. Brochmann. We thank Kåre Lye for verifying the identifications of the herbarium material and Berit Gehrke for helpful comments and discussions. We are grateful to the other members of AFROALP-II team, in particular Geoffrey Mwachala (Kenya), Pantaleo Munishi (Tanzania), and Gerald Eilu (Uganda) for facilitating collection permits and for helping to organize the field work in their respective countries. We also thank two anonymous reviewers for helpful comments and corrections.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
See Table 4.
Rights and permissions
About this article
Cite this article
Gizaw, A., Wondimu, T., Mugizi, T.F. et al. Vicariance, dispersal, and hybridization in a naturally fragmented system: the afro-alpine endemics Carex monostachya and C. runssoroensis (Cyperaceae). Alp Botany 126, 59–71 (2016). https://doi.org/10.1007/s00035-015-0162-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00035-015-0162-2