Abstract
The trend towards functional foods and healthy diets encourages researchers to produce new probiotic food products with improved probiotic delivery methods. Recently, edible films have been applied as carriers for probiotics, improving their survival in the foods and the gastrointestinal tract. This article is structured as follows: a description of edible films as probiotic carriers is followed by an introduction of the materials used as edible films in three main chapters (1) hydrocolloids (polysaccharides and proteins), (2) lipids and (3) composites. Then, probiotic viability in all mentioned edible films is discussed in detail and the impact of prebiotic incorporation is mentioned. Microbial, physicochemical, and sensory properties of edible film containing probiotics in food matrices are reviewed. Finally, the application of probiotic edible films in different food products is described.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Probiotics can play a beneficial role in the human intestinal tract ecosystem (Abd El-Salam and El-Shibiny 2015). Today, increasing knowledge about the influence of diet on health leads to a continuous increase in the consumers’ demand for functional food products, which exhibit minimum changes in organoleptic properties (Lusk 2019). This fact leads to an increasing attention of researchers to investigate certain characteristics of functional food products.
During industrial food processing and products’ shelf life, probiotics are exposed to many challenges such as temperature, oxidative, acute toxic factors (e.g. hydrogen peroxide), water vapor, osmotic and mechanical stresses, and acid–base changes (Iaconelli et al. 2015). Also, probiotics are adversely affected by low pH of the stomach, enzymatic action of pepsin, protease-rich conditions of the intestine, and contact with bile salts during their passage through the gastrointestinal tract (GIT) (Tripathi and Giri 2014). To provide health benefits, probiotics must be able to survive until their consumption and then successfully proliferate in the gut (Farnworth and Champagne 2010). The cell survival in probiotic food products is still challenging (Haffner et al. 2016; Maciel et al. 2014). Anhydrobiotics technology i.e. the encapsulation of viable cells via extrusion, spray-drying, emulsion, coacervation, and electrospraying, is the most popular method to maintain maximum viability of probiotics (Foroutan et al. 2017; Prasanna and Charalampopoulos 2018). One of the newest techniques to improve the survivability of probiotics is to use edible films, that are plasticized thin layered biopolymer structures and that protect them, besides having the potential to favor consumer health (Gialamas et al. 2010; Kanmani and Lim 2013; López de Lacey et al. 2012, 2014; Romano et al. 2014; Soukoulis et al. 2016).
It is possible to improve the functionalities of food products with edible films, such as incorporating nutrients, antioxidants, and antimicrobial agents. On the other hand, most of the edible films include high amounts of fiber, which are recommended in a healthy diet to weight control. In addition, probiotics can be incorporated into edible polymer matrices (via spray, freeze or fluidized bed drying) to improve food stability, functionality, and safety. Probiotic edible films are easier to prepare than encapsulated probiotics and are produced at lower cost because of lower equipment requirements (Altamirano-Fortoul et al. 2012). The application of edible films in the food industry can affect shelf life by reducing harmful reactions (Falguera et al. 2011). Furthermore, probiotic edible films can avoid gas exchange, moisture loss, photodegradation, oxidation, and growth of pathogenic microorganisms (Altamirano-Fortoul et al. 2012). Moreover, probiotic edible films could improve probiotic survival rates during storage and consumption and control probiotic dosage (Soukoulis et al. 2016).
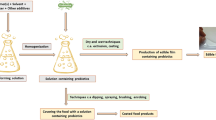
The terms edible films and edible coatings represent different concepts. Edible films are thin solid layers that can be applied as a wrapping for foodstuff, while the edible coatings are formed directly on the surface of the food products (such as fruits), usually by immersing the products in a solution of structural matrix-forming materials (Silva-Weiss et al. 2013). Edible films can be applied to all kinds of food products like a cover, but usually, they are used for foods that cannot be immersed in the film-forming solution such as bakery products. However, probiotic edible films could not be a replacement for common packaging materials because these films are eaten with foods; therefore the products need an external packaging (such as paper packaging) to prevent external contamination. The difference between the probiotic edible coating and the probiotic edible film is demonstrated in Fig. 1. Since edible films are eaten with the products, nutritional and organoleptic characteristics of edible films can be improved by inserting several components such as organic acids, essential oils, and chitosan (Rojas-Graü et al. 2009).
In recent years, probiotic immobilization in edible films has become considerably valuable (Guimaraes et al. 2018; Kanmani and Lim 2013; López de Lacey et al. 2014; Romano et al. 2014; Soukoulis et al. 2016). There are a few review articles about probiotic edible films (Espitia et al. 2016; Guimaraes et al. 2018; Pandhi et al. 2019; Pavli et al. 2018), but this study is a comprehensive review about probiotic edible films in foodstuff. It highlights the scientific investigations about probiotic incorporation in edible films and summarizes the materials used for edible films and probiotic applications in edible food packaging. In addition, the physicochemical and organoleptic properties of probiotic-containing edible films and probiotic viability in edible films are discussed. For this purpose, all recent and important published studies are reviewed comprehensively.
2 General aspects of probiotics: importance and application
Probiotics are defined as “viable microorganisms that, while ingested in sufficient amounts, exert health benefits on the host” (Huq et al. 2013). The main properties of probiotic strains are resistance to gastrointestinal conditions, adhesion to human epithelial cells or mucus, antimicrobial effects against pathogenic bacteria via the production of antimicrobial agents or competition for growth factors or nutrients and binding sites (FAO/WHO 2002). Some of the reported health effects include improvement of GIT health (Thushara et al. 2016), reduction of serum cholesterol levels (Choi and Chang 2015), and biosorption of toxins (Zoghi et al. 2014, 2017) and heavy metals (Hadiani et al. 2018a, b) from foodstuff. To get the beneficial effects of probiotics, a daily intake of 107–109 Colony Forming Units (CFU)/g of probiotic viable cells per person for humans is recommended (Haffner et al. 2016).
3 Edible films as probiotic carriers
Although edible films are not a replacement for an external packaging, they enhance food protection by reducing the moisture loss and gas transfer between food and environment (Soukoulis et al. 2014a, b). Nowadays, edible films are applied as a carrier as well as a controlled release system for some drugs, antioxidants, antimicrobial agents. Since edible films act as water and oxygen barriers, enhancement of quality and shelf life of food products is the main result. Edible films are a “green” replacement for petroleum-based films and can be consumed with the food products (Soukoulis et al. 2014a, b). Immobilization of viable cells in biopolymer networks is a well-known technique to enhance microbial stability. Edible films have appropriate chemical and physical properties, and due to their sustainable nature they are proposed as bioactive compounds carriers (Falguera et al. 2011).
Edible films have the potential to create bespoke structures to enhance mechanical properties, increase shelf-life, and maintain structural integrity. Furthermore, they could be versatile and feasible carriers for the delivery of probiotics (Soukoulis et al. 2014b).
4 Incorporation of probiotics in edible films
Two major processes provide edible films:
-
1.
In the wet process, biopolymers (such as methylcellulose) and other additives (such as plasticizers) are dissolved in distilled water, the film-forming solution is provided after homogenization, and then the solvent is evaporated.
-
2.
The dry process is based on the thermoplastic behavior that some proteins and polysaccharides show at low moisture levels in pressing molding and extrusion (Guimaraes et al. 2018).
Tapia et al. (2007) first investigated the entrapment of probiotics into edible films. Since then, several studies have evaluated the probiotic incorporation in edible films via the direct method, where the probiotic cells are added into the film-forming solution and the probiotic edible film is obtained by a casting method in a forced-air oven, and allowed to dry at room temperature (Kanmani and Lim 2013). Table 1 summarizes previous studies about edible films that have been used for probiotic incorporation in food products. Different parameters such as the presence of oxygen scavenging agents, the biopolymer and plasticizer type and amount, and adding prebiotics have recently been evaluated (Gialamas et al. 2010; López de Lacey et al. 2014; Romano et al. 2014; Soukoulis et al. 2013; Soukoulis et al. 2014b; Soukoulis et al. 2016).
5 Materials used for probiotic edible films
The materials used for the preparation of edible films can be categorized into three classes: hydrocolloids, lipids, and composites (Rojas-Graü et al. 2009). Some other materials such as plasticizers are also added to the film-forming solutions to enhance their stability or to improve the mechanical properties (Valencia-Chamorro et al. 2011).
5.1 Hydrocolloids
The hydrocolloids consist of proteins and polysaccharides. Proteins that are used for probiotic edible films include gelatin, wheat gluten, corn zein, soy protein, casein, and collagen. Protein-based probiotic edible films are prepared from protein solutions as the solvent (ethanol, water or their mixture) evaporates. It is reported that using protein could improve probiotic survival in edible films via scavenging free radicals and conveying nutrients (Burgain et al. 2013). In addition, applying proteins could lead to the formation of less porous and more compact structures (Soukoulis et al. 2016). Polysaccharides for probiotic edible films include cellulose derivatives, inulin, dextrans, alginate, starch derivatives, carrageenan, pectin derivatives, seaweed extracts, chitosan, and galactomannans. Polysaccharide-based probiotic edible films have good mechanical properties and form good odor, oxygen, and oil barriers, but their major disadvantage is their moisture permeability, due to their hydrophilic properties (Ramos et al. 2012). Protein edible films have better mechanical and barrier properties than polysaccharide edible films, but they present poor water resistance (Suput et al. 2015).
Alginates have good film-forming features and can provide crystalline and water-soluble edible films as probiotic carriers. Alginate is a common term for the alginic acid salts. Since alginate can eliminate lipid oxidation and delay dehydration, it has been applied mainly for meat products (Nayik et al. 2015).
Several investigations have studied pectin as a material for probiotic edible films (Espitia et al. 2016; Soukoulis et al. 2017). Pectins are structural constituents of plant cell walls and they are a common type of gelling agent.
Starch made edible films are usually tasteless, odorless, colorless, and transparent with low oxygen permeability. Starch edible films have good barrier properties to CO2. Physicochemical and functional properties of starch-based probiotic edible films depend on the amylose/amylopectin ratio (Basiak et al. 2017). Additionally, cross-linked, oxidized, substituted, and acid-hydrolyzed starches are provided as a result of chemical modifications that have an impact on the edible film properties as probiotic carrier. Acid hydrolysis of starches decreases swelling power and increases solubility compared to native starches (Shah et al. 2016).
Gelatin-based probiotic edible films have poor water vapor barrier properties and are applied to meats, due to their ability in reducing oxygen, oil and moisture transportation (López de Lacey et al. 2012; Soukoulis et al. 2016). Gelatin has antimicrobial and antioxidant activities.
5.2 Lipids
Lipids used for probiotic edible films include vegetable oils, natural waxes, acetoglycerides, resins, and fatty acids. These compounds present some disadvantages such as chemical and mechanical instabilities and organoleptic quality reduction (Pavli et al. 2018). Therefore, lipids are commonly combined with other materials such as proteins or polysaccharides to improve specific characteristics of lipid-based edible films (Suput et al. 2015). Lipid-based edible films are good barriers to moisture transfer, but they have weak gas permeability and mechanical properties compared to polysaccharides and protein-based edible films (Guimaraes et al. 2018).
5.3 Composites
Composites are defined as probiotic edible films containing a blend of polysaccharides, proteins, and lipids. The purpose of providing composite edible films is to modify the properties of the edible film for specific applications such as carrying probiotics (Soukoulis et al. 2014a). A combination of materials can give more efficient properties to probiotic edible films because each material has its unique and limited functions (Rojas-Graü et al. 2009).
5.4 Plasticizers
Plasticizers are low molecular weight components, usually hydrophilic compounds, that decrease the glass transition temperature, increase toughness, flexibility and the tear resistance of the edible films. The plasticizers increase the intermolecular spacing, the mobility of polar polymer chains, and decrease congested intermolecular forces and polymer chain gumminess. The most frequently used plasticizers include glycerol, sucrose, sorbitol, and polyethylene glycol (Ramos et al. 2012). Using plasticizers is essential for the formation of edible films, especially when polysaccharides or proteins are used as materials. However, it may increase the lethality of the embedded probiotic cells, due to osmolysis and increasing exposure to oxygen. To control this, some approaches including the incorporation of free radical scavenger compounds, promoting probiotic cell adhesion properties and suppression of matrix glass transition temperature are used (Burgain et al. 2013). Selecting a plasticizer for an edible film is based on the adaptability and persistence of the plasticizer, and the required physical characteristics of the films. Using sorbitol in the films leads to thermal stability, high thickness, and density. Applying glycerol increases moisture content. Moreover, edible films including glycerol show higher gas permeability in comparison to edible films including sorbitol (Pérez and Dufour 2017).
Rompothi et al. (2017) postulated that glycerol supplied better plasticizer efficiency than sorbitol. They also have concluded that enhancing plasticizer concentration led to increasing solubility, elongation, water vapor permeability, and seal strength, but decrease oxygen permeability, tensile strength, and elastic modulus. However, Krogars et al. (2003) have found that applying an equal amount of glycerol and sorbitol as a plasticizer was more effective than applying sorbitol or glycerol alone.
6 Probiotic viability in edible films
The edible films can maintain probiotic viability in the GIT (Soukoulis et al. 2014b). The survival of many probiotic strains have been widely evaluated under various conditions, whilst only a few investigations are accessible dealing with the viability of probiotics in edible films to evaluate their suitability as probiotic carriers (Guimaraes et al. 2018). Since the film-forming procedure and the chemistry of the edible films affect the probiotic viability (both post-processing and post-ingestion), they are critical factors (Soukoulis et al. 2017). While a complete mechanistic understanding of probiotic stability in edible film matrices during storage is not available, it is reported that steric hindrance of solutes and the interaction via hydrogen bonding with the polar head groups of membrane phospholipids, the presence of free radical scavenging agents and nutrients can be possible factors (Kanmani and Lim 2013; Soukoulis et al. 2013). Furthermore, the molecular mobility of solutes driven by the structural state of the immobilizing matrix can also affect the viability of probiotics. Therefore, obtaining low water matrices with low permeability to gases consisting of free radical scavenging agents is an effective procedure for improving probiotics’ survival in food products (Soukoulis et al. 2013).
The type of material used for probiotic-containing edible films might also cause considerable injuries because of osmotic stress (Bustos and Bórquez 2013). For instance, polysaccharides influence the viability of probiotics during the drying process and the storage period (Yonekura et al. 2013). In addition, the presence of high amounts of solutes, as well as the rubbery physical state (solutes’ increased molecular mobility) in the edible films, simplifies the occurrence of chemical and enzymatic reactions that injure cellular structures such as phospholipid membrane bilayers (Fu and Chen 2011).
The viability of Lactobacillus (L.) casei in sodium caseinate films with sorbitol as a plasticizer was studied by Gialamas et al. (2010). Direct entrapment of probiotics into the film-forming solution led to increased viability at both selected temperatures (4 °C and 25 °C). In addition, they stated that adding sorbitol increased viability due to its action as a protective agent for probiotics during drying or low water activity storage. The mechanism by which sorbitol and related polyols exert this protective impact could be due to the interaction between phospholipid membranes and sugars, helping to retain their fluidity (López de Lacey et al. 2012). Kanmani and Lim (2013) investigated probiotic viability in pullulan and starch-based edible films. L. rhamnosus GG, L. reuteri and L. acidophilus were entrapped into films of starches, pullulan, and their combination and were stored at 4 °C and 25 °C. Maximum probiotic viability was observed in pullulan films at 25 °C up to 30 days. Inserting starch to the pullulan films has a negative impact on cell viability. After 60 days of storage at 25 °C, no viable probiotic cells were observed, probably because of increased bacterial metabolism. In contrast, the pullulan and the pullulan-starch films maintained probiotic viability > 80% at 4 °C after 30 days, maybe because of decreased bacterial metabolism.
Concha-Meyer et al. (2011) incorporated lactic acid bacteria (LAB) into alginate films and used it to pack salmon. They reported that the LAB strain growth and viability in the edible film was improved after getting in touch with the salmon, maybe due to nutrient diffusion. They also claimed that the water activity of the edible film after drying (aw = 0.91) affected probiotic viability because a low enhancement of this value (aw = 0.92) was observed at the contact time with the salmon. Consequently, a decrease in water activity (or increase in osmotic stress) can impact the probiotic viability (Prasad et al. 2003). As stated previously, whey protein can partially decrease osmotic stress and increase adhesion that can lead to an improved survival rate (Soukoulis et al. 2017). Edible films without probiotics were applied as control samples in all studies.
Pavli et al. (2017) studied the viability of L. plantarum and L. pentosus in sodium alginate edible films. The storage temperature had no impact on the survival of the probiotic cells. The viability was reduced after contact of edible films with ham slices at all temperatures, maybe because of the drying process and the subsequent stress. The viability of Bifidobacterium (B.) animalis Bb-12 and L. casei-01 in edible films were investigated by Pereira et al. (2016). They stated that whey protein had a positive impact on the viability of the probiotic strain during storage, due to its nutritional value and by increasing the buffering capacity.
In general, adding plasticizers into edible films enhances the molecular mobility of water and accelerates fatal enzymatic and chemical reactions. Low molecular mobility is obtained at low storage temperature and low water content (Tymczyszyn et al. 2012). Moreover, the permeability of the film to gases such as water vapor and oxygen can also affect adversely the viability of probiotic cells. Edible film composition (protein and polysaccharides type, type and amount of plasticizers, presence of prebiotics) and storage temperature play a key role in probiotics stability (Kanmani and Lim 2013; Soukoulis et al. 2014b).
6.1 Effect of incorporation of prebiotics
Prebiotics are mostly consisting of nonstarch polysaccharides and oligosaccharides that beneficially influence the host by selectively stimulating the activity or growth of one or a limited number of probiotic bacterial species in the colon, and therefore enhance their survivability for host health (Gallego and Salminen 2016). Although inserting prebiotics is a favorable strategy for probiotic protection, only a few researchers have investigated the use of prebiotics in probiotic-containing edible films to maintain stability and functionality of the entrapped probiotics.
The interaction of phosphate groups of lipid membranes with fructooligosaccharides reduces the phase transition temperatures. In addition, the dynamic mechanical analysis showed that the addition of fructooligosaccharides into edible films changes the hydrogen bonds in polymer molecules, decreases the glass transition temperature, thus indicating the plasticizer effect of fructooligosaccharides.
Therefore, the incorporation of fructooligosaccharides into the probiotic-containing edible films contributes to protecting probiotics during dehydration and storage (Romano et al. 2014). Moreover, inserting prebiotics into gelatin edible films led to a uniform and more compact structure, with no detectable interspaces, thus prebiotics could act as fillers in gelatin-based edible films (Soukoulis et al. 2014a).
7 Inhibitory effects of probiotic edible films against other microbial species
In addition to probiotic delivery to the consumers, probiotic edible films are important for increasing food stability and safety as the probiotics control the growth of spoilage microorganisms via competition or produce antimicrobial substances. It is well established that LAB show antimicrobial activity by the production of antifungal peptides, acids, and bacteriocins. Therefore, the incorporation of LAB into edible films also exhibits antimicrobial features (Cizeikiene et al. 2013) and reinsures the microbiological safety of the food products (García-Argueta et al. 2013).
Gialamas et al. (2010) stated that edible films containing L. sakei caused a significant reduction of Listeria monocytogenes in beef in comparison to the control. The inhibitory effect of L. sakei against Listeria monocytogenes has been related to direct competition for nutrients or to lactic acid production. The application of edible films containing LAB for chilled fish inhibited the growth of Photobacterium phosphoreum (López de Lacey et al. 2012). Sánchez-González et al. (2013) evaluated the inhibitory effect of L. plantarum incorporated in different edible films against Listeria innocua. They stated that polysaccharide-based edible films had significant activity against Listeria innocua, while protein-based edible films did not show this activity, may be due to the delay in bacteriocin production. They concluded that the material of the edible film and the time of bacteriocin production are the major parameters. Sánchez-González et al. (2014) confirmed the inhibitory impact of L. reuteri and L. acidophilus incorporated in sodium caseinate and methylcellulose against Listeria innocua for a week. Both film formulations have led to a decrease of approximately 1.5 log cycles of Listeria innocua growth at the end of storage. Thus, it can be concluded that the inhibitory activity is attributed to bacteriocin production, not to competitive growth, and the nature of the edible film matrix influences the bacteriocin production. In another study, L. paracasei and B. lactis embedded in the agar-based edible film into hake fillets caused a reduction of H2S-producing microorganism counts during the storage period (López de Lacey et al. 2014). Edible films without incorporating probiotics were applied as control samples in all studies. Thus, probiotic edible films could be used as an alternative method of food preservation.
8 Probiotic-containing edible film characterization
8.1 Physicochemical properties
The physical and chemical properties of edible films should be analyzed simultaneously for the design of appropriate edible films with suitable plasticization properties providing a suitable environment that guarantees the viability of the probiotic.
8.2 Mechanical properties
Edible films, as probiotic carriers, should have appropriate mechanical strength and extensibility to resist food processing, packaging, and storage stress (Garavand et al. 2017). The presence of structural deficiencies, the structural adaptation of the major components of edible films, the distribution and density of the intra- and intermolecular interactions between the polymer chains, and the type and amount of plasticizers have been reported to affect the mechanical profile of edible films as probiotic carriers (Falguera et al. 2011).
The plasticizer glycerol is an effective parameter that determines the mechanical properties of edible films as probiotic carriers. It reduces the intermolecular forces between polymers, reducing the tensile strength and increasing the elongation at break (Rouhi et al. 2017). Moreover, the presence of cavities and holes could decrease the edible film flexibility and tensile strength. Therefore, edible films must have good mechanical properties to protect probiotics, resist food processing, handling, and storage stresses. Gialamas et al. (2010) demonstrated that no significant changes were observed in tensile strength, elongation at break, and modulus of elasticity of sodium caseinate edible films containing probiotics, because of the relatively low mass of the added probiotics with little impact on the mechanical properties of protein-based edible films. Consequently, the cellulose-based edible films, with greater mechanical resistance, are a little more sensitive to the incorporation of the probiotic cells.
8.3 Thickness
Thickness is a crucial parameter that affects the water vapor permeability, transparency, and mechanical features of the edible films to improve the film's ability to carry probiotics and to increase the mechanical integrity of foods (Ghanbarzadeh and Almasi 2011). The thickness of edible films depends on the preparation technique and drying conditions (Galus and Lenart 2012). Soukoulis et al. (2014a) stated that no significant impact on thickness was observed by the addition of L. rhamnosus GG cells into the probiotic-containing edible film. Conversely, Soukoulis et al. (2014b) in another study demonstrated that the inclusion of probiotics in film-forming solutions changed the film thickness.
8.4 Moisture content
Since edible films consist of hygroscopic materials, and a high water content is harmful for probiotic preservation, the control of the moisture content during food processing and storage is important (Soukoulis et al. 2014b). The moisture content after drying influences the rate of probiotic viability during long storage periods and simplifies the melting of edible films in the mouth (Kanmani and Lim 2013). Glycerol can retain the water content in the edible films and thus prevent water evaporation (Thakhiew et al. 2010).
8.5 Water vapor permeability
Water vapor permeability is one of the most critical properties of edible films as probiotic carriers, that can be influenced by parameters such as solubility coefficient, diffusion rate, hydrophobic ratio, the integrity of the film, interactions between the functional groups of the polymers, crystalline ratio, thickness, and amorphous ratio (Kanmani and Lim 2013).Water vapor permeability of the edible films is influenced by the mobility of polymer chains (Su et al. 2010). The probiotic cells, as discontinuous particles, might inhibit the chain mobility of the polymers in the film matrix (Guimaraes et al. 2018). Moreover, increased water vapor permeability may improve edible film solubility, which is one of the main advantages to releasing probiotics (Kanmani and Lim 2013).
8.6 Sensory properties
Acceptable sensory properties of probiotic products are required for achieving commercial success in the market. Therefore, sensory assessment is essential before starting a new production. It is well established that inserting free probiotics into food products can significantly alter their sensory properties. Entrapment of probiotics into edible films can control undesirable modifications in sensory properties (Corona-Hernandez et al. 2013). Although there are some studies regarding the sensory properties of edible coating containing probiotics, there is a gap of knowledge dealing with edible films containing probiotics.
The color and optical properties of edible films are important because they directly affect the appearance of food products and consumers’ preferences. Transparency is one of the usual optical properties of edible films. Inserting probiotics into the edible films may influence the light that passes through the edible film, perhaps because of the increasing dispersion of light (Kanmani and Lim 2013). According to Martins et al. (2012), the moisture content of probiotic-containing edible films can affect the light reflections on the film surface. Moreover, Ghanbarzadeh and Almasi (2011) observed an improvement in optical properties and yellowness reduction by inserting carboxymethyl cellulose to starch-based films. The shine of the edible films depends on the surface morphology achieved during film drying. Generally, the smoother the surface of the edible films, the higher the brightness (Ward and Nussinovitch 1996).
Due to the difference between the density of cells and the polymer solution during the drying stage, probiotics are present on the surface of the edible films and form a continuous layer. The number of probiotic cells accumulated on the edible film surface depends on the viscosity of the film-forming dispersion (Ly et al. 2008). The surface charge of the probiotics is essential because of the determination of electrostatic interactions with charged polymers. Generally, parameters such as the crystallites’ mean size and crystallinity, the amount of plasticizer and its type as well as structural conformation, the refractive index, and compatibility of the film compounds affect the opacity of edible films (Fakhouri et al. 2013).
Tavera-Quiroz et al. (2015), applied edible films of methylcellulose containing L. plantarum to green apple baked snacks. The analyzed sensory attributes included taste, color, texture, appearance, and overall acceptability. The snacks' taste score rating with the probiotic-containing edible film was considerably lower than that of the control without the probiotic edible film. In addition, the color-related findings were nearly identical, but the texture values of control samples were slightly higher than that of the snacks with probiotic edible film, perhaps because of their higher moisture content. Similarly, Alvarez (2012) stated that edible film with a specified composition (milk whey, inulin, and glycerol) can maintain the textural properties of the broccoli during 15 days of storage. Sodium alginate edible films containing probiotics significantly influenced the aroma and taste attributes of ham slices and the total organoleptic scores. The researchers demonstrated that these disadvantages were partially controlled when a high-pressure processing treatment was applied before using the probiotic-containing edible films, probably due to a lower level of probiotic cells number in the ham slices (Pavli et al. 2017).
9 Application of probiotic edible films in food products
Compared to conventional dehydrated microcarriers, probiotic edible films could provide significant benefits, e g. increasing the shelf life period, for intermediate moisture foods, because of their ability to maintain their physical state and biological activity during food storage. Therefore, several applications for probiotic edible films such as fruits and vegetables, bakery and confectionery products, olives, dairy products, fishery products, cereal bars, and meat products have been studied (Altamirano-Fortoul et al. 2012; De Prisco and Mauriello 2016; Espitia et al. 2016; López de Lacey et al. 2012; Soukoulis et al. 2017; Tavera-Quiroz et al. 2015).
The first application of probiotic edible films was evaluated for fresh fruits (Tapia et al. 2007). After that, probiotic edible films were applied to carrots (Shigematsu et al. 2019), cherry tomatoes, and Thompson grapes (Dianin et al. 2019). Probiotic edible films for fresh fruits and vegetables could control ripeness by decreasing oxygen diffusion and inhibiting the growth of spoilage microorganisms, so that metabolic activity and softening changes will be reduced. Therefore, the freshness of the fruits, the quality characteristics such as color, acid, sugar and flavor and the nutritional factors can be maintained for a longer time (Pandhi et al. 2019).
Although there are some investigations (Concha-Meyer et al. 2011; Gialamas et al. 2010; López de Lacey et al. 2012, 2014) about using probiotic edible films in meat and fishery products, further studies are needed. The efficiency of sodium-alginate edible films as the probiotic carrier, was evaluated for application in sliced ham with or without high-pressure processing pretreatment. The results of the study demonstrated that probiotic strains were successfully carried by edible films in the meat products irrespective of the high-pressure processing treatment (Pavli et al. 2017). In another study, L. acidophilus and B. bifidum were inoculated into edible gelatin films for preserving hake fish (Merluccius merluccius) (López de Lacey et al. 2012). Adding green tea extract into probiotic edible films (containing L. paracasei and B. lactis) resulted in better chemical and microbial stabilities and prolonged the shelf-life of hake fillets for at least one week (López de Lacey et al. 2014).
Some researchers focused on the effect of the application of probiotic edible films on the shelf-life and quality factors in bakery and confectionery products (Altamirano-Fortoul et al. 2012; Soukoulis et al. 2014b, 2017; Tavera-Quiroz et al. 2015). The outward appearance of baked products is the main quality factor that influences the visual sense of the consumers. Soukoulis et al. (2017) found that no significant difference was observed in sensory and thermo-physical characteristics of bread samples in bread conventional packages and bread with the probiotic edible film.
10 Conclusions
In this article, the probiotic incorporation into edible films to improve probiotic viability during the storage period and processing of the food product has been reviewed. Application of edible films as a carrier of probiotics has several benefits. Indeed, many of these films contain high amounts of fiber, which are recommended in a healthy diet. Fibers can also stimulate satiation signals in the colon which may be useful for weight control.
A wide variety of edible film materials with different properties can be applied which may influence the properties of the final products. Although the physicochemical properties of edible films can be influenced by the incorporation of probiotics, using edible films containing probiotics does not alter the sensory characteristics of the food products significantly. In general, the application of the edible film is a promising strategy to enhance probiotics' survival during storage time.
Although the incorporation of probiotic bacteria into many different edible materials is reported and survival and properties of films have been studied, there is a lack of in vivo studies and investigation of survival and colonization properties of such entrapped probiotics in the human body. In addition, there is no report on the inhibitory effects of probiotic films against pathogens in vivo. What is still mystifying scientists in the field of probiotic-containing edible films, is that how an entrapped microorganism can be released from the film, activated and localized during the short time of passage (on average a few hours) in the GIT.
Moreover, many technological and economic aspects of manufacturing processes need to be streamlined. Further studies must be carried out to apply different probiotics and prebiotics in new edible films and apply them to foods familiar for the consumer. Each geographic region should provide edible films with their localized and accessible probiotics, prebiotics and film materials to apply in their localized foods to increase the efficacy and health benefits of functional foods.
References
Abd El-Salam MH, El-Shibiny S (2015) Preparation and properties of milk proteins-based encapsulated probiotics: a review. Dairy Sci Technol 95(4):393–412
Abdollahzadeh E, Ojagh SM, Imani Fooladi AA et al (2018) Effects of probiotic cells on the mechanical and antibacterial properties of sodium-caseinate films. Appl Food Biotechnol 5(3):155–162. https://doi.org/10.22037/afb.v5i3.20360
Altamirano-Fortoul R, Moreno-Terrazas R, Quezada-Gallo A, Rosell CM (2012) Viability of some probiotic coatings in bread and its effect on the crust mechanical properties. Food Hydrocoll 29(1):166–174. https://doi.org/10.1016/j.foodhyd.2012.02.015
Alvarez VT (2012) Effect of edible coating with added probiotics (Lactobacillus casei Shirota) on the textural, microbiological and physicochemical characteristics on minimally processed broccoli during its shelf life. [Undergraduate Thesis] Toluca, Edo Mex: Universidad Autónoma del Estado de México
Basiak E, Lenart A, Debeaufort F (2017) Effect of starch type on the physico-chemical properties of edible films. Int J Biol Macromol 98:348–356. https://doi.org/10.1016/j.ijbiomac.2017.01.122
Behboudi-Jobbehdar S, Soukoulis C, Yonekura L, Fisk I (2013) Optimization of spray-drying process conditions for the production of maximally viable microencapsulated L. acidophilus NCIMB 701748. Dry Technol 31(11):1274–1283. https://doi.org/10.1080/07373937.2013.788509
Burgain J, Gaiani C, Cailliez-Grimal C et al (2013) Encapsulation of Lactobacillus rhamnosus GG in microparticles: Influence of casein to whey protein ratio on bacterial survival during digestion. Innov Food Sci Emerg Technol 19:233–242. https://doi.org/10.1016/j.ifset.2013.04.012
Bustos P, Bórquez R (2013) Influence of osmotic stress and encapsulating materials on the stability of autochthonous Lactobacillus plantarum after spray drying. Dry Technol 31(1):57–66
Choi EA, Chang HC (2015) Cholesterol-lowering effects of a putative probiotic strain Lactobacillus plantarum EM isolated from kimchi. LWT Food Sci Technol 62:210–217. https://doi.org/10.1016/j.lwt.2015.01.019
Cizeikiene D, Juodeikiene G, Paskevicius A, Bartkiene E (2013) Antimicrobial activity of lactic acid bacteria against pathogenic and spoilage microorganism isolated from food and their control in wheat bread. Food Control 31(2):539–545. https://doi.org/10.1016/j.foodcont.2012.12.004
Concha-Meyer A, Schobitz R, Brito C, Fuentes R (2011) Lactic acid bacteria in an alginate film inhibit Listeria monocytogenes growth on smoked salmon. Food Control 22:485–489. https://doi.org/10.1016/j.foodcont.2010.09.032
Corona-Hernandez RI, Alvarez-Parilla E, Lizardi-Mendoza J et al (2013) Structural stability and viability of microencapsulated probiotic bacteria: a review. Compr Rev Food Sci Food Saf 12:614–628. https://doi.org/10.1111/1541-4337.12030
Dafe A, Etemadi H, Dilmaghani A, Mahdavinia GR (2017) Investigation of pectin/starch hydrogel as a carrier for oral delivery of probiotic bacteria. Int J Biol Macromol 97:536–543. https://doi.org/10.1016/j.ijbiomac.2017.01.060
De Prisco A, Mauriello G (2016) Probiotication of foods: a focus on microencapsulation tool. Trends Food Sci Technol 48:27–39. https://doi.org/10.1016/j.tifs.2015.11.009
Dianin I, Oliveira Junior A, Pimentel T et al (2019) Edible biofilms formulated with whey proteins isolate and L. casei probiotic culture: characterization and application in tomatoes or grapes. Chem Eng Trans 75:469–474. https://doi.org/10.3303/CET1975079
Ebrahimi B, Mohammadi R, Rouhi M et al (2018) Survival of probiotic bacteria in carboxymethyl cellulose-based edible film and assessment of quality parameters. LWT Food Sci Technol 87:54–60. https://doi.org/10.1016/j.lwt.2017.08.066
Espitia PJ, Batista RA, Azeredo HM, Otoni CG (2016) Probiotics and their potential applications in active edible films and coatings. Food Res Int 90:42–52. https://doi.org/10.1016/j.foodres.2016.10.026
Fakhouri FM, Costa D, Yamashita F et al (2013) Comparative study of processing methods for starch/gelatin films. Carbohyd Polym 95(2):681–689. https://doi.org/10.1016/j.carbpol.2013.03.027
Falguera V, Quintero JP, Jimenez A et al (2011) Edible films and coatings: structures, active functions and trends in their use. Trends Food Sci Technol 22(6):292–303. https://doi.org/10.1016/j.tifs.2011.02.004
FAO/WHO (2002) Report of a Joint FAO/WHO Working Group on drafting guidelines for the evaluation of probiotics in food, London, Ontario, Canada
Farnworth ER, Champagne C (2010) Production of probiotic cultures and their incorporation into foods. In: Watson ER, Preedy VR (eds) Bioactive foods in promoting health. Academic Press, London, pp 3–17
Foroutan NS, Tabandeh F, Khodabandeh M et al (2017) Isolation and identification of an indigenous probiotic Lactobacillus strain: its encapsulation with natural branched polysaccharids to improve bacterial viability. Appl Food Biotechnol 4(3):133–142
Fu N, Chen XD (2011) Towards a maximal cell survival in convective thermal drying processes. Food Res Int 44:1127–1149. https://doi.org/10.1016/j.foodres.2011.03.053
Gallego CG, Salminen S (2016) Novel probiotics and prebiotics: how can they help in human gut microbiota dysbiosis? Appl Food Biotechnol 3(2):72–81
Galus S, Lenart A (2012) Development and characterization of composite edible films based on sodium alginate and pectin. J Food Eng. https://doi.org/10.1016/j.jfoodeng.2012.03.006
Garavand F, Rouhi M, Razavi SH et al (2017) Improving the integrity of natural biopolymer films used in food packaging by crosslinking approach: a review. Int J Biol Macromol 104:687–707. https://doi.org/10.1016/j.ijbiomac.2017.06.093
García-Argueta I, Dublán-García O, Quintero-Salazar B et al (2013) Effect of lactic acid bacteria on the textural properties of an edible film based on whey, inulin and gelatin. Afr J Biotechnol 12(19):2659–2669. https://doi.org/10.5897/AJB2013.12134
García-Argueta I, Quintero-Salazar B, Dominguez-Lopez A et al (2016) Effect of edible coating based on whey, inulin and gelatine with Lactobacillus casei on the textural and sensorial properties of a cracker cookie. J Probiotics Health 4(3):153–159. https://doi.org/10.4172/2329-8901.1000153
Ghanbarzadeh B, Almasi H (2011) Physical properties of edible emulsified films based on carboxymethyl cellulose and oleic acid. Int J Biol Macromol 48(1):44–49. https://doi.org/10.1016/j.ijbiomac.2010.09.014
Gialamas H, Zinoviadou KG, Biliaderis CG, Koutsoumanis KP (2010) Development of a novel bioactive packaging based on the incorporation of Lactobacillus sakei into sodium-caseinate films for controlling Listeria monocytogenes in foods. Food Res Int 43:2402–2408. https://doi.org/10.1016/j.foodres.2010.09.020
González-Cuello RE, Ortega-Toro R, Zapateiro LG (2018) Effect of Lactobacillus acidophilus addition on mechanical and barrier properties of binary films during storage. Contemp Eng Sci 11(6):269–282. https://doi.org/10.12988/ces.2018.8117
Guimaraes A, Abrunhosa L, Pastrana LM, Cerqueira MA (2018) Edible films and coatings as carriers of living microorganisms:a new strategy towards biopreservation and healthier foods. Compr Rev Food Sci Food Saf 17:594–614. https://doi.org/10.1111/1541-4337.12345
Hadiani MR, Khosravi-Darani K, Rahimifard N, Younesi H (2018a) Assessment of mercury biosorption by Saccharomyces cerevisiae: response surface methodology for optimization of low Hg(II) concentrations. J Environ Chem Eng 6(4):4980–4987. https://doi.org/10.1016/j.jece.2018.07.034
Hadiani MR, Khosravi-Darani K, Rahimifard N, Younesi H (2018b) Biosorption of low concentration levels of Lead(II) and Cadmium(II) from aqueous solution by Saccharomyces cerevisiae: response surface methodology. Biocatal Agric Biotechnol 15:25–34. https://doi.org/10.1016/j.bcab.2018.05.001
Haffner F, Diab R, Pasc A (2016) Encapsulation of probiotics: insights into academic and industrial approaches. AIMS Mater Sci 3(1):114–136. https://doi.org/10.3934/matersci.2016.1.114
Huq T, Khan A, Khan R et al (2013) Encapsulation of probiotic bacteria in biopolymeric system. Crit Rev Food Sci Nutr 53(9):909–916. https://doi.org/10.1080/10408398.2011.573152
Iaconelli C, Lemetais G, Kechaou N et al (2015) Drying process strongly affects probiotics viability and functionalities. J Biotechnol 214:17–26. https://doi.org/10.1016/j.jbiotec.2015.08.022
Kanmani P, Lim ST (2013) Development and characterization of novel probiotic-residing pullulan/starch edible films. Food Chem 141:1041–1049. https://doi.org/10.1016/j.foodchem.2013.03.103
Krogars K, Heinämäki J, Karjalainen M et al (2003) Enhanced stability of rubbery amylose-rich maize starch films plasticized with a combination of sorbitol and glycerol. Int J Pharmacol 251(1–2):205–208
López de Lacey AM, López-Caballero ME, Gómez-Estaca J et al (2012) Functionality of Lactobacillus acidophilus and Bifidobacterium bifidum incorporated to edible coatings and films. Innov Food Sci Emerg Technol 16:277–282. https://doi.org/10.1016/j.ifset.2012.07.001
López de Lacey AM, López-Caballero ME, Montero P (2014) Agar films containing green tea extract and probiotic bacteria for extending fish shelf-life. LWT Food Sci Technol 55(2):559–564. https://doi.org/10.1016/j.lwt.2013.09.028
Lusk JL (2019) Consumer beliefs about healthy foods and diets. PLoS One 14(10):e0223098. https://doi.org/10.1371/journal.pone.0223098
Ly MH, Aguedo M, Goudot S et al (2008) Interactions between bacterial surfaces and milk proteins, impact on food emulsions stability. Food Hydrocoll 22:742–751. https://doi.org/10.1016/j.foodhyd.2007.03.001
Maciel GM, Chaves KS, Grosso CRF, Gigante ML (2014) Microencapsulation of Lactobacillus acidophilus La-5 by spray-drying using sweet whey and skim milk as encapsulating materials. J Dairy Sci 97:1–8. https://doi.org/10.3168/jds.2013-7463
Martins JT, Cerqueira MA, Bourbon AI et al (2012) Synergistic effects between k-carrageenan and locust bean gum on physicochemical properties of edible films made thereof. Food Hydrocoll 29(2):280–289. https://doi.org/10.1016/j.foodhyd.2012.03.004
Nayik GA, Majid I, Kumar V (2015) Developments, in edible films and coatings for the extension of shelf life of fresh fruits. Am J Nutr Food Sci 2(1):16–20
Pandhi S, Kumar A, Alam T (2019) Probiotic edible films and coatings:concerns, applications and future prospects. J Pack Technol Res 3(3):261–268. https://doi.org/10.1007/s41783-019-00069-6
Pavli F, Kovaiou I, Apostolakopoulou G et al (2017) Alginate-based edible films delivering probiotic bacteria to sliced ham pretreated with high pressure processing. Int J Mol Sci 18:1867. https://doi.org/10.3390/ijms18091867
Pavli F, Tassou C, Nychas GJE, Chorianopoulos N (2018) Probiotic incorporation in edible films and coatings:bioactive solution for functional foods. Int J Mol Sci 19(1):150. https://doi.org/10.3390/ijms19010150
Pereira O, Soares J, Sousa S et al (2016) Edible films as carriers for probiotic bacteria. LWT Food Sci Technol 73:543–550. https://doi.org/10.1111/1541-4337.12345
Pereira O, Soares J, Monteiro M et al (2019) Cereal bars functionalized through Bifidobacterium animalis subsp. lactis BB-12 and inulin incorporated in edible coatings of whey protein isolate or alginate. Food Funct 10:6892–6902. https://doi.org/10.1039/C9FO00370C
Pérez EE, Dufour D (2017) Native and modified starches as matrix for edible films and covers. Nutr Food Sci Int J 3(3):555–615. https://doi.org/10.19080/NFSIJ.2017.03.555615
Phovisay S, Siriwoharn T, Surawang S (2018) Effect of drying process and storage temperature on probiotic Lactobacillus casei in edible films containing prebiotics. Food Appl Biosci J 6:105–116
Prasad J, McJarrow P, Gopal P (2003) Heat and osmotic stress responses of probiotic Lactobacillus rhamnosus HN001 (DR20) in relation to viability after drying. Appl Environ Microbiol 69(2):917–925. https://doi.org/10.1128/AEM.69.2.917-925.2003
Prasanna PHP, Charalampopoulos D (2018) Encapsulation of Bifidobacterium longum in alginate-dairy matrices and survival in simulated gastrointestinal conditions, refrigeration, cow milk and goat milk. Food Biosci 21:72–79. https://doi.org/10.1016/j.fbio.2017.12.002
Ramos OL, Fernandes JC, Silva SI et al (2012) Edible films and coatings from whey proteins: a review on formulation, and on mechanical and bioactive properties. Crit Rev Food Sci Nutr 52:533–552. https://doi.org/10.1080/10408398.2010.500528
Rojas-Graü MA, Soliva-Fortuny R, Martin-Belloso O (2009) Edible coatings to incorporate active ingredients to fresh-cut fruits: a review. Trends Food Sci Technol 20:438–447. https://doi.org/10.1016/j.tifs.2009.05.002
Romano N, Tavera-Quiroz MJ, Bertola N et al (2014) Edible methylcellulose-based films containing fructo-oligosaccharides as vehicles for lactic acid bacteria. Food Res Int 64:560–566. https://doi.org/10.1016/j.foodres.2014.07.018
Rompothi O, Pradipasena P, Tananuwong K et al (2017) Development of non-water soluble, ductile mung bean starch based edible film with oxygen barrier and heat sealability. Carbohydr Polym 157(10):748–756. https://doi.org/10.1016/j.carbpol.2016.09.007
Rouhi M, Razavi SH, Mousavi SM (2017) Optimization of crosslinked poly (vinyl alcohol) nanocomposite films for mechanical properties. Mater Sci Eng C 71:1052–1063. https://doi.org/10.1016/j.msec.2016.11.135
Sánchez-González L, Quintero Saavedra JI, Chiralt A (2013) Physical properties and antilisterial activity of bioactive edible films containing Lactobacillus plantarum. Food Hydrocoll 33(1):92–98. https://doi.org/10.1016/j.foodhyd.2013.02.011
Sánchez-González L, Quintero Saavedra JI, Chiralt A (2014) Antilisterial and physical properties of biopolymer films containing lactic acid bacteria. Food Control 35:200–206. https://doi.org/10.1016/j.foodcont.2013.07.001
Shah U, Naqash F, Gani A, Masoodi FA (2016) Art and science behind modified starch edible films and coatings: a review. Compr Rev Food Sci Food Saf 15:568–580. https://doi.org/10.1111/1541-4337.12197
Shigematsu E, Dorta C, Santos DN et al (2019) Edible coating with coconut water to preserve probiotic strains and sensory characteristics of minimally processed carrots. Int Food Res J 26(4):1285–1292
Silva-Weiss A, Ihl M, Sobral PJA et al (2013) Natural additives in bioactive edible films and coatings: functionality and applications in foods. Food Eng Rev 5:200–216. https://doi.org/10.1007/s12393-013-9072-5
Singh P, Magalhães S, Alves L et al (2018) Cellulose-based edible films for probiotic entrapment. Food Hydrocoll. https://doi.org/10.1016/j.foodhyd.2018.08.057
Soukoulis C, Behboudi-Jobbehdar S, Yonekura L et al (2013) Impact of milk protein type on the viability and storage stability of microencapsulated Lactobacillus acidophilus NCIMB 701748 using spray drying. Food Bioprocess Technol 1:1–14
Soukoulis C, Behboudi-Jobbehdar S, Yonekura L et al (2014a) Stability of Lactobacillus rhamnosus GG in prebiotic edible films. Food Chem 159(100):302–308. https://doi.org/10.1016/j.foodchem.2014.03.008
Soukoulis C, Yonekura L, Gan HH et al (2014b) Probiotic edible films as a new strategy for developing functional bakery products: the case of pan bread. Food Hydrocoll 39:231–242. https://doi.org/10.1016/j.foodhyd.2014.01.023
Soukoulis C, Singh P, Macnaughtan W, Parmenter C, Fisk ID (2016) Compositional and physicochemical factors governing the viability of Lactobacillus rhamnosus GG embedded in starch-protein based edible films. Food Hydrocoll 52:876–887. https://doi.org/10.1016/j.foodhyd.2015.08.025
Soukoulis C, Behboudi-Jobbehdar S, Macnaughtan W et al (2017) Stability of Lactobacillus rhamnosus GG incorporated in edible films: impact of anionic biopolymers and whey protein concentrate. Food Hydrocoll 70:345–355. https://doi.org/10.1016/j.foodhyd.2017.04.014
Su JF, Huang Z, Zhao YH et al (2010) Structure and properties of carboxymethyl cellulose/soy protein isolate blend edible films cross linked by Maillard reactions. Carbohydr Polym 79:145–153
Suput DZ, Lazic VL, Popovic SZ, Hromis NM (2015) Edible films and coatings-sources, properties and application. Food Feed Res 42:11–22. https://doi.org/10.5937/FFR1501011S
Tapia MS, Rojas-Graü MA, Rodríguez EJ et al (2007) Alginate- and gellan-based edible films for probiotic coatings on fresh-cut fruits. J Food Sci 72:E190–E196. https://doi.org/10.1111/j.1750-3841.2007.00318.x
Tavera-Quiroz MJ, Romano N, Mobili P et al (2015) Green apple baked snacks functionalized with edible coatings of methylcellulose containing Lactobacillus plantarum. J Funct Foods 16:164–173. https://doi.org/10.1016/j.jff.2015.04.024
Thakhiew W, Devahastin S, Soponronnarit S (2010) Effects of drying methods and plasticizer concentration on some physical and mechanical properties of edible chitosan films. J Food Eng 99(2):216–224. https://doi.org/10.1016/j.jfoodeng.2010.02.025
Thushara RM, Gangadaran S, Solati Z, Moghadasian MH (2016) Cardiovascular benefits of probiotics: a review of experimental and clinical studies. Food Funct 7(2):632–642. https://doi.org/10.1039/c5fo01190f
Tripathi MK, Giri SK (2014) Probiotic functional foods: survival of probiotics during processing and storage. J Funct Foods 9:225–241. https://doi.org/10.1016/j.jff.2014.04.030
Tymczyszyn EE, Sosa N, Gerbino E et al (2012) Effect of physical properties on the stability of Lactobacillus bulgaricus in a freeze dried galacto-oligosaccharides matrix. Int J Food Microbiol 155:217–221. https://doi.org/10.1016/j.ijfoodmicro.2012.02.008
Valencia-Chamorro SA, Palou L, del Río MA, Pérez-Gago MB (2011) Antimicrobial edible films and coatings for fresh and minimally processed fruits and vegetables: a review. Crit Rev Food Sci Nutr 51:872–900. https://doi.org/10.1080/10408398.2010.485705
Ward G, Nussinovitch A (1996) Gloss properties and surface morphology relationships of fruits. J Food Sci 61(5):973–977
Yonekura L, Sun H, Soukoulis C, Fisk I (2013) Microencapsulation of Lactobacillus acidophilus NCIMB 701748 by spray drying: impact of soluble fibres on storage stability and survival after in vitro digestion. J Funct Foods 6:205–214. https://doi.org/10.1016/j.jff.2013.10.008
Zoghi A, Khosravi-Darani K, Sohrabvandi S (2014) Surface binding of toxins and heavy metals by probiotics. Mini Rev Med Chem 14:84–98
Zoghi A, Khosravi-Darani K, Sohrabvandi S et al (2017) Effect of probiotics on patulin removal from synbiotic apple juice. J Sci Food Agric 97(8):2601–2609. https://doi.org/10.1002/jsfa.8082
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zoghi, A., Khosravi-Darani, K. & Mohammadi, R. Application of edible films containing probiotics in food products. J Consum Prot Food Saf 15, 307–320 (2020). https://doi.org/10.1007/s00003-020-01286-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00003-020-01286-x