Abstract
Stress exposure during early stages of life elevates the risk of developing psychopathologies and psychiatric illness in later life. The brain and immune system are not completely developed by birth and therefore continue develop after birth; this post birth development is influenced by several psychosocial factors; hence, early life stress (ELS) exposure can alter brain structural development and function. A growing number of experimental animal and observational human studies have investigated the link between ELS exposure and increased risk of psychopathology through alternations in the immune system, by evaluating inflammation biomarkers. Recent studies, including brain imaging, have also shed light on the mechanisms by which both the innate and adaptive immune systems interact with neural circuits and neurotransmitters, which affect psychopathology. Herein, we discuss the link between the experience of stress in early life and lifelong alterations in the immune system, which subsequently lead to the development of various psychiatric illnesses.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Early life stress
- Psychopathology
- Psychiatric illness
- Inflammation
- Cytokines
- Neuroinflammation
- Immune system
1 Introduction
Both positive and negative experiences in early life can have profound effects on mammalian brain development. In particular, early life stress (ELS) is associated with increased risk of both mental and physical health deterioration throughout life [1,2,3]. Approximately 12% of youth, from infancy to age 18 years, experiences ELS to such an extent that they will have mental and physical health discrepancies throughout life [4, 5]. These individuals who account for almost half of all mental disorders [6] experience approximately 44% increase in strokes and heart attacks [7] and elevated risk of death by age 50 years [8, 9].
Generally, two main criteria, namely, the developmental age range of early life and the characteristics of stressful events, should be considered when defining ELS. Previous studies have considered an upper age limit between 12 and 18 years as early life. Regarding the stress criterion, various models have proposed that stress is experienced when an individual faces a threatening situation for which adequate coping skills are not available. Disruption to physiological homeostasis also induces stress responses. A certain event occurring within a defined developmental term may be classified as ELS. Thus, ELS can be defined as an injury, potential of injury, or threat of injury generally caused by a child’s caregiver [10]. This definition includes different stressful experiences, such as the death of a caregiver, neglect, bullying, emotional maltreatment, and physical and sexual abuse. However, emotional and physical abuse and neglect are the most common patterns of harm reported [11].
The most major forms of ELS in individuals are neglect (physical or emotional), abuse (physical, emotional, or sexual), and parental loss (death, or separation). The number of abused children reached approximately 520,000 in the United Kingdom in 2011, which has since increased [12]. According to a large-scale epidemiological study on adverse childhood events, approximately 65% of individuals in the United States have been exposed to at least one event, while 12.5% have been exposed to as many as four [13]. Adults reporting ≥4 ELS are 4.6 times more likely to experience depression and 12.2 times more likely to attempt suicide compared to those with no ELS exposure. Gilbert et al. found that children self-reporting physical and emotional abuse is estimated at up to 29% and 33%, respectively, in Eastern Europe [14].
Approximately 8% of males and 20% of females experience childhood sexual abuse, with the highest prevalence in Africa (34.4%), followed by Asia, America (10.1%), and Europe (9.2%) [15]. Physical abuse, the intentional use of physical force that harms the child’s development, survival, or dignity, is estimated at 17.6% [16]. Meanwhile, psychological abuse, the failure to give children enough supportive environment, may also be more than physical and sexual abuse but is more difficult to estimate [17]. Neglect, the failure of a caregiver to provide for a child’s basic needs, is the most major ELS affecting 78.5% of children in the general population. Other forms of ELS include natural disasters, physical diseases, surgeries, accidents, and events such as terrorism or war. Less salient experiences with significant distress on children include poverty, unstable families, poor parental care, and dysfunctional relationships between parent and children. ELS can be often complex, with different forms simultaneously coexisting and can happen as chronic or ongoing stress.
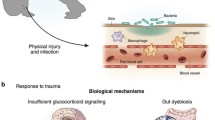
ELSs are associated with an elevated risk of noncommunicable diseases in adulthood [18,19,20,21] and premature mortality [22], possibly mediated by a dysfunctional immune system, particularly chronic low-grade inflammation [23,24,25]. The dysregulated immune responses could be prone to developmental programming attributed to ELS exposure that only trigger an excessive stress response at onset, but also influencing long-term stress responses, leading to chronic low-grade inflammation [26, 27]. The immune system responds to foreign invaders [28], and both human and animal researches have shown that ELS can cause persistent inflammation, which could develop psychiatric problems through effects on brain development and response to stressors [29].
The brain and immune system are not fully developed at birth, yet have minimal functions in newborns that enable adjustment to limited and expected stimuli. The ongoing maturity of the immune system throughout infant and childhood indicates that environmental effects and stimulation during childhood can seriously affect the immune system. Therefore, the brain and immune system experience during early postnatal development progressively increases their repertoire to maximize adaptation to stimuli specific to the individual’s own environment [1, 30, 31]. Notably, ELS gives rise to various aberrations in brain circuitry, cognitive function, and general health [32,33,34] and the immune system may also play a unifying role in the pathophysiology of these multifactorial diseases related to ELS. Herein, we provide an overview of the current evidence connecting ELS to elevated inflammation and subsequent risk of psychiatric disorders.
2 Early Life Stress and Inflammation
2.1 Experimental Animal Studies
The first report for the effect of ELS on the immune system came to light from experimental animal models more than half a century ago. Mouse handled before the process of weaning exhibited a decreased rate of development in transplanted tumor [35] and elevated serum antibody titer in response to the bacterial protein flagellin [36]. These results attracted interest in the area of developmental psychoneuroimmunology [37,38,39], which facilitated subsequent studies on the association between ELS and immune functioning in later life in rats and nonhuman primates [40, 41].
Experimental animal models have expanded our understanding of the relationship between ELS and immune system abnormalities and allowed for invasive procedures to investigate immune function since components in the immune system can be targeted with drugs during and after ELS to determine the adverse health outcomes. ELS in rats has been manipulated through various experimental models with heterogeneous effects on parental caregiving behavior, such as nursery rearing, maternal separation (MS), maternal deprivation, neonatal handling, and dexamethasone exposure. Measures of immune function range from pro-inflammatory cytokines and chemokines in the plasma and antigen-specific immune response to pro-inflammatory gene expression in the brain and gut microbiota.
MS has been commonly used in animal models of ELS. In nonhuman primates, MS increased macrophage activity [42] and upregulated long-term pro-inflammatory gene transcription in monocytes [43]. In rats, MS elevated core temperature [40] and pro-inflammatory cytokines in the plasma [44, 45]. These findings demonstrated the association between MS and inflammatory processes in later life.
Using a mouse model, a previous study has reported that MS results in a loss of prefrontal cortex (PFC) interneurons [46], underlying a supposed mechanism of schizophrenia associated with inflammatory and excitotoxic damage [47]. In an animal model undergoing repeated MS (RMS), elevated hippocampal interleukin-1β (IL-1β) mRNA levels approximately 20 times that of the control have been reported [48]. One study also showed elevated NF-α expression in the PFC of animals sacrificed on the day of their final MS episode [49], while another reported that MS animals sacrificed immediately after their final episode had higher hypothalamic tumor necrosis factor-α (TNF-α) than those with a single episode of MS sacrificed simultaneously [48]. In animals sacrificed on the final day of MS, elevated interleukin-10 (IL-10) expression was identified in the PFC and small intestine but not the hippocampus or serum [49, 50].
In pubs, MS during brain development is associated with reduced lipopolysaccharide-binding protein expression in the hippocampus [51] and decreased microglial cell numbers in the midbrain [52]. In contrast, early MS in adult animals increases synaptic levels of pro-inflammatory cytokine interleukin-1 (IL-1) receptor [53], elevates the number and motility of cortical microglial processes [54], and exacerbates microglial activation [55]. Moreover, mice experiencing MS have a higher elevation in body core temperature after a second MS, increased cytokine expression followed by viral infection [56], and greater cortical microglial activation following exposure to chronic food-restriction stress [55]. Although the peripheral response to ELS may not be mostly activated or suppressed, ELS-linked early immune programming seems to sensitize later pro-inflammatory processes and result in higher risk to depression and anxiety in adulthood [57]. Increased heart rate and inflammatory responses to a physiological stressor [58], as well as elevated TNF-α and interferon-γ (IFN-γ) and corticosterone levels, and anxiety-like behavior [59] in maternally deprived rodents have been shown.
ELS studies have recently reported overall increases in activation and number of microglial cells in various brain regions. Microglial cells sensitized in early life could show a dysregulated response and morphological changes in later life [60]. ELS may therefore convert a neuroprotective state to a pro-inflammatory state in microglia [48]. Moreover, microglial activation and increases could facilitate brain maturation [61]. Ex vivo studies on the early MS-induced damage on microglia have reported an overall elevation in the proportion of cells with an activated morphology in the hippocampus [48, 62, 63] and medulla [64]. Furthermore, using captured microglial cells in vivo, one study showed that somatosensory stimulation in adulthood caused a significantly higher increase in microglial motility in MS mice compared to controls which could also affect microglia–synapse interactions and neuronal function [54].
Psychosocial stressors other than immune stimuli can also provoke a microglial response that may induce different responses to threats [65]. Gong et al. reported that 1 day of brief social isolation at postnatal day (P)14 elevated microglial density in the hippocampus, presumably by facilitating increases of these cells [66]. Four days following isolation (P14–17), the number of cells was restored to normal levels. In contrast, a week of brief social isolation (P14–21) in adulthood decreased microglial cell number in the dentate gyrus of the hippocampus. Concordantly, social defeat in adolescent mice triggers early increase of ionized calcium-binding adapter molecule (IBA-1) in the hippocampus and a following decrease in microglial cells and IBA-1 expression in adulthood [67, 68]. Furthermore, a milder social defeat paradigm adopted during the adolescent stage elevated microglia number, IBA-1 expression, and the size of soma in the ventral tegmental area of pups [69]. Adult mice experiencing repeated social defeat show significant elevation in neutrophils and CD11b+LyC6high monocytes in the spleen and circulation [70, 71]. Splenic dendritic cells from mice experiencing repeated social defeat have shown greater surface expression of major histocompatibility complex class I, CD80, and CD44, suggesting an activated state [72]. Exposure to repeated social defeat in mice and low socioeconomic status in humans can also lead to a relative expansion of a transcriptional protein associated with immature pro-inflammatory monocytes in peripheral blood mononuclear cells [73]. Therefore, various types of social stressors in early life independently impact the development of the immune system, although the dysfunctional relationship between mother and infant may negatively affect health outcome.
Recent studies on the gut microbiota found that MS in rodent and nonhuman primate models also has transient and long-term effects on gut microbiota [59, 74]. ELS-induced changes of the microbiome in murine models continue during adulthood [75,76,77] and are linked to anxiety-like behaviors and activation of systems involved in stress [78]. Rats exposed to stress show inflammation, altered gastrointestinal function and leaky gut, and disturbances of immune activity [78]. Moreover, gut microbiota and dysregulated inflammation in rats or mice exposed to stress can regulate the metabolism of tryptophan to kynurenine or 5-hydroxytryptamine (5-HT) [79,80,81]. Inflammatory cytokines such as IFN-γ and interleukin-6 (IL-6) enhance indoleamine-2,3-dioxygenase (IDO) production, which subsequently metabolizes tryptophan to kynurenine, increasing kynurenine production and decreasing 5-HT levels [80]. Elevated kynurenine/tryptophan ratios have been recognized in rats with depression-like behavior, together with elevated pro-inflammatory cytokines and altered gut microbiota [81]. Furthermore, a study on Flexibacter and Prevotella, in connection with colitis, revealed that they were more abundant in the gut of MS rats [82], and concordantly, Wong et al. showed that caspase-1 inhibition, an inflammasome factor, restored stress-induced gut microbiota alterations [83].
3 Early Life Stress and Inflammation
3.1 Observational Human Studies
ELS can affect the immune system at the time of exposure [57, 84] and alter its normal developmental trajectory [85]. The exaggerated effects of ELS on the immune system are long term, resulting in chronic low-grade inflammation throughout life [86]. A large population-based study of almost 12,000 participants observed an association between increased white blood cell counts and ELS [87].
A meta-analysis demonstrated that adults with ELS have higher levels of C-reactive protein (CRP) and the major pro-inflammatory cytokines IL-6 and TNF-α as compared to adults without ELS [88]. Another meta-analysis reported a significant correlation between ELS and inflammatory markers, with effect sizes being greatest for TNF-α, followed by IL-6 and CRP. A recent meta-analysis showed a relationship between ELS and IL-1β, IL-6, TNF-α, and CRP, but not interleukin-2 (IL-2), interleukin-4 (IL-4), interleukin-8 (IL-8), IL-10, or fibrinogen [89]. Moreover, Baumeister et al. reported that various types of ELS exposure differentially resulted in alterations of inflammatory markers [88]. Notably, physical and sexual abuse were associated with significantly elevated TNF-α and IL-6, but not CRP, which is mainly associated with parental absence during the early developmental period. Although the underlying pathophysiology remains nebulous, factors such as situation and duration of stress exposure may interact with individual stress types to regulate immune response.
Furthermore, a meta-analysis of 27 studies conducted by Kuhlman et al. evaluated the relation between ELS and inflammation in those under 18 years of age [90] and found small associations between ELS and inflammation that were statistically significant only for CRP. Meanwhile, other longitudinal studies have reported an association between ELS and elevated inflammatory markers in early adulthood [91, 92], thereby providing evidence for the relationships between ELS and increased peripheral CRP levels, particularly profound in those who develop subsequent depression in adult life [93, 94]. Danese et al. reported elevated inflammation levels in abused children who had depression at the age of 12 years compared to normal controls [95]. Increased CRP levels were also identified in 10-year-old children with recent onset of maltreatment and a genotype predisposing to elevated CRP levels [93]. Exposure to stressful events before the age of 8 years is associated with increased CRP at 10 and 15 years old [25]. Peripheral low-grade inflammation may describe the relationships between early-life stress and various physical or mental health outcomes [96,97,98]. To the best of our knowledge, there has been one study investigating the relationship between ELS and inflammation in preschool-aged children (3–5 years) which showed increased IL-1β levels in connection with ELS [99]. In healthy community samples, ELS has been related to elevated IL-6, in response to the Trier Social Stress Test [100] and IL-1β, interleukin-12 (IL-12), and TNF-α levels [101]. Furthermore, recent findings suggest that repeated exposure to ELS is connected with significant increases in soluble urokinase plasminogen activator receptor (suPAR) in young adulthood [102] and mid-adulthood [92]. During activated immune and pro-inflammatory states, suPAR is discharged into the systemic bloodstream by cleavage of the membrane-bound urokinase receptor (uPAR) [103, 104] expressed on endothelial cells and immune cells [105]. While CRP and IL-6 are influenced by acute fluctuations in inflammation, such as during infections [106, 107], suPAR reflects a person’s overall immune activity level and is predicted to be involved in low-grade chronic inflammation, tissue and organ damage [108, 109], development and progression of disease, adverse clinical outcomes, and mortality [110, 111] and thus is an effective and additional measure of persistent inflammatory response. Concordantly, suPAR is associated with ELS [92, 102] and adult stressful life events [112], whereas IL-6 and CRP are not persistently related to these kinds of stressors.
Granulocyte function, evaluated by ex vivo killing of Staphylococcus aureus, is decreased by 20% in children with divorced parents [113], which is in line with the findings of elevated vulnerability to Staphylococcus aureus in children exposed to acute or chronic family stress [114]. Moreover, reduced natural killer (NK) cell activity was related to stressful events in adolescents with depression or conduct disorder [115] and adult females with breast cancer [116]. However, increased NK cell activity has been identified in children whose parents showed more chronic stress, which is also associated with greater rates of febrile diseases in childhood, while this was not associated with NK cell activity [117]. Furthermore, Wyman et al. studied a younger population undergoing immune assessment [117] and observed an elevation in NK cell activity [118]. Meanwhile, Ayaydin et al., in a relatively small number of participants, also showed no significant difference in NK cell activity between control and sexually abused adolescents [119]. Sexual or physical abuse is associated with lower salivary IgA levels in young females, even though adult sexual victimization appeared to mediate this relationship [120].
Evidence of the relationship between ELS and increased reactive oxygen species (ROS) production, oxidative stress, and mitochondrial activity, which are associated with pro-inflammatory cytokine from different immune cells, has been reported [121]. Dysfunctional cellular immunity caused by repeated viral infections and reactivation of viruses elevates inflammatory markers including IL-6 and CRP [122]. Also, individuals exposed to ELS showed increased immune activation with higher CD25 expression, major histocompatibility complex, class II, DR (HLA-DR), or implicating CD8 T cells [123]. Moreover, adolescents exposed to ELS have decreased NK and NK T cells and increased circulating and senescent T cells with the activation markers CD3+/CD69+ and CD2+/CD4+/CD25+ [124, 125].
Positron emission tomography (PET) imaging of the mitochondrial translocator protein (TSPO) can provide insights on the microglial activation in the human brain. One study found that after peripheral lipopolysaccharide (LPS) injection, TSPO expression uniformly increased across the brain [126]. These findings allow for deeper assessment of neuroinflammatory markers to investigate microglial activation during brain injury and neurodegeneration. Interestingly, only one study has investigated microglial activation using TSPO-PET in individuals exposed to ELS [71, 127]; therefore, further investigations on the relationship between ELS and microglial activation are warranted.
Also, in line with findings from animal studies, results from human studies suggest that females may have higher sensitivity to stressful events in provoking a pro-inflammatory response than males [128, 129]. Moreover, evidence from an environmental risk study indicates that the levels of inflammation were already increased in children exposed to ELS who developed depression at the age of 12 years as compared to controls [95], while sex differences may influence the susceptibility to cause a pro-inflammatory state post ELS exposure [130]. CRP concentrations in 18-year-old females were significantly correlated with childhood victimization, yet no such correlation was observed in their male counterparts. According to the study by Entringer et al., the relationship between CRP levels and maltreatment was significantly mediated by child sex and were higher in the maltreated girls compared to the control group which was stable over the 2-year follow-up period, whereas no relationship between maltreatment and CRP levels was shown, suggesting that following ELS exposure at a very young age in girls, the effect of maltreatment may immediately emerge in an inflammation process and exacerbate over time.
In summary, peripheral inflammation caused by ELS can influence the brain and change neural activity through various routes, such as humorally via active transport of cytokines stimulated by the release of second messenger or cellular routes involving macrophage-like cells residing in circumventricular organs. Microglia can be activated by peripheral inflammation that enter the brain across the blood-brain barrier (BBB) with different routes [131]. Subsequently, microglia cells affect cell proliferation and survival in the brain based on their inflammatory state [126]. Microglial cells can undergo several alterations [132] such as pro-inflammatory cytokine production and expression of cell surface antigens related to oxidative stress in the brain. Peripheral LPS injection, used for immune challenge in primates, can increase TSPO expression uniformly across the brain. Recently, alterations in the gut microbiome have been reported in adults with ELS-induced PTSD [133]. Children exposed to ELS have also been reported in a study to exhibit gut microbiome alterations, with gut bacteria levels associated with PFC activation in an emotional face experiment [134]. Generally, the association between ELS-linked gut dysbiosis and inflammation is likely bidirectional [135, 136].
4 Inflammation and Psychiatric Illness
4.1 Experimental Animal Studies
Animal models are beneficial to investigate the pathophysiology due to their flexibility in randomly assigning animals to different rearing environments and allow for directly investigating the brain and immune system using techniques thought to be too invasive in humans. Inflammation can increase animals’ responses to rewarding stimuli with reinforcers such as food or electrical stimulation [137]. Initial findings linking the immune function and psychiatric etiology, particularly mood disorders, originated from studies involving humans and animals with acute infection showing stereotypical behaviors as featured by anhedonia, anorexia, and reduced grooming [138]. In line with the hypothesis of this “sickness behavior” with evolutionarily inflammatory origins, gene knockout models in rodents have been strongly beneficial in emphasizing the causal relationship of inflammatory cytokines (including IL-1β and TNF-α) in developing social withdrawal, sickness behaviors, and anhedonia [137]. Also, the development of sickness behavior led by a pro-inflammatory state is attenuated by treatment with IL-10 and aggravated in mice that are IL-10-deficient [137]. An elevation in cytokine serum levels may correspondingly elevate oxidative stress and reduce availability of serotonin and other neurotransmitters, along with activities of the hypothalamic-pituitary-adrenal (HPA) axis in the brain [137, 139]. Acute induction of pro-inflammatory agents, such as LPS or typhoid vaccination, can trigger transient and similar symptoms [140]. Rodents exposed to MS show dysfunction in PFC-mediated behaviors including social interactions [141], learned helplessness [142], and cognitive function [46] in adolescence and elevated peripheral inflammatory cytokines IL-β and IL-6 [45]. Findings in rodents have suggested that this immune-to-brain traffic can control the cortico-amygdala circuitry involved in threat processing and is connected with enhanced anxiety-like behaviors [143,144,145]. Pigs with MS show sickness-like behavior that is buffered with anti-inflammatory treatment [146, 147], indicating that pro-inflammatory processes can influence early responses to ELS. Social withdrawal, lethargy, and anhedonia related to exposure to pro-inflammatory agents may be part of the organism’s evolutionary effort to use all its resources for fighting foreign invaders and overcoming diseases [148]. Giovanoli et al. have investigated if an anti-inflammatory medication with minocycline in early life during peripubertal adversity exposure could prevent the following occurrence of adult behavioral problems [149]. Notably, rats deficient in the inflammasome NLRP3 showed improvement in both pro-inflammatory state and cognitive function and reduced both systemic inflammation and functional decline during aging [150].
5 Inflammation and Psychiatric Illness
5.1 Observational an Experimental Human Studies
Experimental findings suggest that inflammation can decrease neural activity to reward circuit, as shown by studies that induced inflammation with low-dose bacterial stimuli [151] or investigating the effects of immune-activating treatments on neural reward circuit [152]. Induction of pro-inflammatory states in humans produces a clinical response similar to major depression [153]. Patients with some types of cancer or hepatitis C treated with interferon-α (IFN-α) also develop depressive symptoms within weeks [140, 154]. Additional experimental evidence related to the inflammation as the pathophysiology of mood disorders comes from the antidepressant effects of anti-inflammatory medications. These experimental human studies proposed that inflammation can modulate neural circuit activity linked to rewards independently in different processes that may lead inflammation in those exposed to ELS.
Recently, a meta-analysis showed that cytokine inhibitors and nonsteroidal anti-inflammatory drugs can have small to moderate antidepressant effects [155]. Moreover, patients administered minocycline exhibited a greater decrease in negative symptoms in two clinical trials comparing minocycline versus placebo [156, 157]. Pro-inflammatory cytokines may also reduce executive control-related processes associated with PFC in the brain where it is linked to decision making, executive control, and regulation of reward and threat-related predisposition [158, 159]. Cytokine increase may alternate microglia in the cortex, thereby causing structural and functional changes, which increases the risk of mental illness [160]. Concordantly, alterations in microglial activation have been observed in several psychiatric disorders including schizophrenia [161], depression [162], and anxiety [163].
Microglia plays a major role in the adaptive immune response in the central nervous system (CNS) that can modulate neuronal function not only during inflammation but also in synaptic pruning [164] and plasticity during development [165, 166]. A recent TSPO-PET study showed elevated microglial activity in patients with schizophrenia and persons who are even at ultrahigh risk of psychosis. Moreover, increased microglial activity was positively associated with greater symptom severity in the high-risk population [167], suggesting a relationship between neuroinflammation and psychotic symptoms.
In line with these findings, human observational studies over the past 30 years have emphasized the role of the immune function in the pathophysiology of several psychiatric disorders, including schizophrenia, depression, bipolar affective disorder [168, 169], obsessive–compulsive disorder [170, 171], and posttraumatic stress disorder (PTSD), along with an increase of suicidal attempt [172]. A meta-analysis controlling the effect of antipsychotics in schizophrenia showed persistently increased levels of several immune proteins released from macrophages, such as IL-12, TNF-α, and IFN-α [97]. Interestingly, cell cultures from patients with schizophrenia also produce greater levels of circulating IL-1 and IL-8, thereby confirming the role of immunity-related pathophysiology in schizophrenia. Studies on obsessive–compulsive disorder reported polymorphisms in the TNF-α gene [173] and elevation in plasma TNF-α cytokine levels [174,175,176]; based on the individual, cytokine gene polymorphisms may manifest differently [173]. Other prospective studies also showed that elevated IL-6 and CRP were significantly associated with depressive symptoms later in life. Longitudinal studies have found that increase inflammatory levels in patients with depression likely result from a bidirectional relationship between inflammation and depression over time [177]. A meta-analysis of clinical studies found that patients with depression show a slight elevation in several inflammatory biomarkers [178]. Concordantly, longitudinal associations between inflammation and subsequent psychopathology were shown in participants with psychosis [179], depression [171], and PTSD [180, 181].
Associations between inflammation and psychopathology have been best investigated in depression [182]. Patients with depression show immune cell profiles featured by systemic low-grade inflammation [183]. A cross-sectional meta-analysis investigated alterations in inflammation in depressed adults and characterized depression by a small increase of serum inflammatory markers [178]. Anti-inflammatory medications showed antidepressant effects in a subset of depressed patients with elevated baseline levels of inflammation [184]. Group differences between inflammation in patients with depression and controls likely attributed to the bidirectional relationship between depression and inflammation [29, 177]. Increased levels of pro-inflammatory cytokines, such as IL-6 and TNF-α, are associated with depressed mood [185,186,187], and decreased levels of the anti-inflammatory cytokine IL-10 have been shown in depression [188]. A meta-analysis demonstrated increased TNF-α and IL-6 in patients with depression [189]. Moreover, a study by Miller and Cole showed that the transition to depression was associated with relative increases in CRP and IL-6 levels in individuals exposed to ELS, indicating that ELS can potentially enhance a phenotype wherein depression and inflammation occurred simultaneously [190].
Concordantly, patients with bipolar disorder also have increased levels of inflammation [191], TLR-mediated intracellular signaling [45], and toll-like receptors (TLRs) in peripheral monocytes and lymphocytes. Moreover, elevated NLRP3 levels were found in the frontal cortex of patients with bipolar disorder, which is associated with elevated levels of IL-6, IL-1, TNF-α, and IL-10 [192]. Meta-analyses of clinical studies found that patients with bipolar disorder have small to moderate elevation of both pro-inflammatory cytokines [193] and CRP [194] levels compared to controls. An elevated inflammation state can predict poor treatment prognosis in bipolar disorder [195]. These relationships can reflect the negative outcomes in individuals exposed to ELS [29]. Systemic inflammation in patients with bipolar disorder can be identified not only during active episodes, but also in euthymic phases [194], indicating that inflammation may be a trait marker rather than a state marker for bipolar disorder.
Although there have been limited findings from cross-sectional human studies, increased IL-6 and CRP levels is associated with psychosis [3, 196], as supported by longitudinal studies involving the general population, including the Avon Longitudinal Study of Parents and Children birth cohort. Furthermore, greater levels of pro-inflammatory cytokines in childhood are associated with an elevated risk for psychosis in adolescence and young adulthood [179, 197]. A meta-analysis controlling antipsychotics persistently showed increased TNF-α, interleukin-12 (IL-12), and IFN-γ in patients with schizophrenia [97]. Furthermore, schizophrenic patients show a moderate to large increase in pro-inflammatory cytokines [97] and CRP [198]. Initial evidence also suggests that elevated baseline inflammatory levels can be predictive of poor treatment response in first-episode psychosis [126]. Indeed, a study on both chronic psychotic disorders and first-episode psychosis showed that several inflammatory markers appear to be trait markers and showed no reduction following antipsychotic medication [97, 198].
Patients with PTSD also exhibited increased inflammation levels. A meta-analysis suggests that patients with PTSD have moderate to large elevation in several pro-inflammatory cytokines [199] after controlling the effect of comorbid depression [199]. Genetic [181] and longitudinal [180] studies have suggested that inflammation can be a preexisting susceptibility factor for patients with PTSD exposed to ELS rather than a simple correlation of disease severity, subjective distress, or dysfunctional coping strategies after PTSD development.
6 Early Life Stress and Psychiatric Illness
6.1 Experimental Animal Studies
Animal studies of ELS on psychiatric disorders found that associations between early contexts of stress and later emotional and behavioral abnormalities are likely causal in nature [200,201,202,203,204,205,206]. Studies using mouse [202, 205] and nonhuman primate models [203, 204, 206] have found that ELS from MS can negatively influence the emotional and behavioral development and impair cognitive functioning, in line with the seminal studies of clinical observations by Spitz [207] and Bowlby [208] on the effects of MS on psychiatric disorders. Indeed, animals exposed to ELS show behavioral despair and learned helplessness [200], dysfunctional fear conditioning [200], and avoidant behaviors. Surprisingly, sensitization in guinea pigs was first identified when two, 3-h separations at a 24-h interval increased the number of 1-min intervals that guinea pig pups spent showing a passive, depressive-like response on the second day of separation [40]. Although the effects of induced ELS can be different based on the protocol used and the animals’ gender and age, the findings of these experimental studies strongly indicate a causative role of ELS in psychopathology in the late stage.
7 Early Life Stress and Psychiatric Illness
7.1 Observational Human Studies
Individuals exposed to ELS are 1.3–3.1 times more likely to result in lifetime major depressive disorder or dysthymia, based on the frequency, type and severity, and stressful events [209,210,211]. Although exposure to ELS can increase the risk of many psychiatric disorders, the relationship between ELS and various types of psychiatric etiology have not been clarified [6, 212,213,214,215]. One study showed that ELS predicts several psychiatric disorders, including schizophrenia, depression, bipolar disorder, and PTSD [216,217,218]. ELS from childhood neglect has also been related to later changes in reward function in individuals [219]. Activation of the nucleus accumbens [220] and other basal ganglia regions [221] associated with the reward system decreases in teenagers exposed to ELS. Therefore, individuals exposed to ELS have an increased lifelong risk of major depression including an early-onset and elevated comorbidity [213, 222]. Individuals with present depression and a history of ELS are also more likely to show high levels of high-sensitivity CRP compared to controls. Notably, this association is not likely to be suggested by retrospectively biased reports of individuals in depression at the time of ELS assessment as the evidence is persistent with those from official records and prospective evaluations of maltreatment investigated in childhood [223]. Moreover, this is also unlikely to be described by the effects confounded genetically because a higher risk of depression in individuals exposed to ELS has been identified within twin studies [209].
Also, a history of ELS is highly associated with patients with bipolar disorder and can predict an unfavorable illness course and clinical symptoms such as higher severity of manic, psychotic, and depressive symptoms, a higher suicidal attempt, higher risk of comorbid substance use disorders, anxiety disorders, elevated risk of rapid cycling, and increased occurrence of depressive and manic episodes [224]. Moreover, ELS predicts an increased number of psychotic disorders such as schizophrenia or schizoaffective disorder later in life [217] Furthermore, ELS is related to an elevated risk of PTSD [218] and is associated with more complex symptoms including dysfunctional interrelationship, dysregulated emotion, and poor self-concept [225].
8 Discussion
In this chapter, we provided an overview of the literature on early-life stress, inflammation, and psychiatric illness. This section reviews how ELS affects the psychopathology of psychiatric illness via inflammation. In the past, the brain is thought to be immune-privileged with highly controlled innate and adaptive immunity, especially inflammation in the blood-brain barrier. It has increasingly become evident that the immune-privileged property of the brain is complicated and not absolute [226]. The brain immune system is not only associated with the peripheral immune system [137] but also actively contributes to normal brain development and functioning [227]. The immune system in the brain has different cells, such as T cells and microglia, and proteins such as chemokines or cytokines that play essential roles to maintain homeostasis in the CNS resting state. Microglia monitor the surrounding extracellular space during the resting state for infection and eliminate cellular debris as well as maintain neurogenesis and inactive or dysfunctional synaptic structures. Conversely, during a pro-inflammatory state, microglia produce inflammatory cytokines and other molecules and clean up triggering foreign invaders through phagocytosis. T cells originating from the lymphoid hematopoietic cell scan and detect signals cascaded from brain into the CSF during the resting state. Meanwhile, during the pro-inflammatory state, T cells release cytokines (e.g., IL-4) that stimulate astrocytes to lead the production of brain-derived neurotrophic factor (BDNF) and control inflammatory activity in parenchymal and meningeal myeloid cells such as microglia and induce a protective immune response that may be associated with aggravated results for brain function. Moreover, cytokines accumulating in the microglia and T cells at the resting states play a critical role in hippocampus-linked learning and memory processes, putatively via supporting long-term potentiation, whereas cytokines during the pro-inflammatory state enhance neuroinflammation and decrease monoaminergic transmission and trigger glutamate transmission and the HPA axis–mediated neuroendocrine stress response. Furthermore, a higher level of cytokines inhibits BDNF and cholinergic transmission [137, 227]. Notably, there are various routes through which inflammatory cytokines can increase synaptic monoamine availability; these routes can play a fundamental role in the mechanism underlying the pathophysiology of psychiatric illness [228]. An increased level of IDO is also associated with cytokine-induced monoamine neurotransmitter changes by converting the metabolism of tryptophan more into the kynurenine pathway but less into the 5-hydroxyindoleacetic acid, thereby reducing serotonin production. Subsequently, the neurotoxic metabolite quinolinic acid from microglia, monocytes, and macrophages in the CNS originates from kynurenine [229, 230]. Quinolinic acid stimulates N-methyl-D-aspartate receptors for glutamate and glutamate release by astrocyte and blocks glutamate reuptake by astrocytes [231], which directly affect glutamate metabolism to ultimately increase excitotoxicity and decrease efficient neurogenesis, finally resulting in increased glutamate both inside and outside the synapse. Therefore, elevated glutamate also increases excitotoxicity and decreases the production of BDNF [232].
Concordantly, high levels of nitric oxide (NO) [233] released from microglia in the inflammatory state can promote more neuronal cytotoxicity and apoptosis [234, 235] and contribute to neuronal loss in schizophrenia and Alzheimer’s diseases [236,237,238]. Thus, ELS sensitize microglial activation resulting in a lower threshold for a reactive state and subsequently increasing inflammatory cytokine levels and dysregulated neurotransmission, which can explain psychopathologies of psychiatric illness caused by ELS.
As mentioned above, previous studies have linked the peripheral immune system and the brain immune system; researchers have recognized that the immune-privileged property of the brain is complicated and not absolute. The humoral pathway refers to the cytokine passage through regions such as the circumventricular organs with increased permeability in the BBB and elevated binding of cytokines to saturable transport molecules on the BBB. The neural pathway [137] refers to the binding of peripheral cytokines to peripheral afferent nerve fibers, such as the vagus nerve, which subsequently triggers ascending catecholaminergic fibers in the CNS and/or brings back cytokine signals in the central part [139]. The signal transduction pathway refers to the triggering by peripheral cytokines from cell surface receptors on endothelial cells and astrocytes in the brain that maintain the BBB, subsequently stimulating more cytokine production by these cells. The transmembrane pathway refers to the active transport of cytokines (TNF-α, IL-6, IL-1) through saturable carrier proteins to enter the BBB. Finally, the cellular pathway refers to the trafficking of activated immune cells, typically monocytes, to the brain vasculature and parenchyma. Through these pathways, peripheral inflammation can trigger neuroinflammation in the brain [137, 239]. For example, peripheral induction of LPS in rodents increases the production of pro-inflammatory cytokines [240] and microglia activation and inhibited adult neurogenesis in the brain [241].
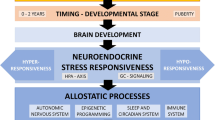
Chronic stress in early life causes repeated and prolonged HPA overactivation, which can subsequently cause less compensation in reduced signaling through epigenetic alterations in the glucocorticoid receptor [242] and promote resistance to the function of cortisol to control the inflammatory state. Experimental human studies have found that traumatic experiences during childhood are associated with allele-specific DNA demethylation related to glucocorticoid response elements (FKBP5 gene), which is related to the subsequently reduced sensitivity of peripheral blood immune cells to the inhibitory function of glucocorticoids on LPS-induced production of IL-6 in vitro [243]. A longitudinal study also showed that adolescents exposed to harsh familial treatment showed decreased sensitivity of glucocorticoid over time and elevated ex vivo cytokine responses to LPS administration [244].
Furthermore, the alteration of colonization and composition of the gut microbiota might be influenced by ELS, which could affect immune development as well as brain development via inflammatory signal transmission through metabolic alterations or the vagus nerve [135]. Experimental animals findings also showed that MS during the first year of life causes a significant reduction in fecal lactobacilli [74] with long-term alterations on the composition of the microbiota in the gut being apparent in later life [59].
Interestingly, recent meta-analytical findings in animal models showed that ELS is linked to a small increase in the risk of obesity [19], as individuals with ELS may be less sensitive to reward and hence may be involved in more dysfunctional appetitive behaviors, such as eating fast foods or more high-calorie food items. Also, given that ELS can potentiate HPA axis activation and related unpleasant feelings, individuals with ELS eat more to decrease HPA axis activation. Elevated pro-inflammatory cytokines by adipocytes can trigger a systemic inflammatory state in individuals with obesity [245]. Moreover, individuals with ELS may have dysfunction in hormonal pathways regulating thermogenesis and lipolysis including the leptin pathway or the HPA axis [19].
Previous studies have also reported that individuals exposed to ELS are at increased risk of sleep disturbances [246, 247], which showed stronger relationship for participants with more severe maltreatment exposures [246] regardless of concurrent PTSD or depressive disorders [248] Furthermore, MS in rodents can disrupt sleep architecture and decrease total sleep; meanwhile, in humans, MS can induce sleep deprivation and loss, which elevates the expression and levels of pro-inflammatory cytokines [249, 250].
In line with the biological and evolutional aspects of the bidirectional associations between the brain and immune system, the critical targets primary related to inflammation in the brain include those brain regions associated with both motivation and motor activity such as arousal, anxiety, and alarm. In other words, the main neurocircuits affected by inflammation involve the reward and anxiety circuits. Dopamine as a core neurotransmitter plays a critical role in the reward circuit and inflammatory cytokines have been demonstrated to reduce the production of dopamine in the basal ganglia, which is involved in decreased motivation and activation of the reward circuit in the ventral striatum [151, 152, 251]. Accumulated imaging studies such as PET, functional magnetic resonance imaging (fMRI), and magnetic resonance spectroscopy (MRS) have shown decreases in reward activation in the striatum, showing strong reproducibility and validity of the cytokine-induced alterations of the brain in nondepressed individuals peripherally administered LPS, typhoid vaccination, or IFN-α [151, 152, 252,253,254]. Notably, fMRI studies also found that inflammation-mediated reduction of positive reward activation is related to elevated sensitivity to negative stimuli and decreased activations in the substantia nigra in the basal ganglia [254, 255]. According to studies on positive valence systems, peripheral administration of typhoid vaccine and LPS decreases responses to reward in the ventral striatum [151, 152]. Inflammatory cytokines in dopaminergic pathways also induce a state of reduced motivation. Moreover, elevated inflammation is related to elevated responses to anxiety and threat neurocircuitry, involving the amygdala, dACC, and insula [155, 256, 257]. Notably, the dACC and amygdala are regions with elevated responses in patients with depression, anxiety, and neuroticism [258]. Thus, elevated oral IL-6 expression is strongly associated with increased response of the amygdala to social evaluation stressor, with subjects showing the highest IL-6 responses to stress, indicating greatest functional connectivity within threat circuitry, which involves the dorsomedial PFC and amygdala [259]. Similarly, elevated concentrations of oral IL-6 and soluble TNF receptor 2 in response to an induced social anxiety condition, such as a public speaking, are strongly associated with the activation of the dACC to a social rejection task [257]. Indeed, these findings are related to the trafficking of monocytes to the amygdala led by social defeat stress in rodents [145]. Considering negative valence systems, typhoid vaccine decreases the relationship between the sACC and the amygdala and elevates the activation in the sACC while processing emotional faces [158]; peripheral administration of LPS also enhances activation in the amygdala in conditions of socially threatening stimuli [260]. Subsequently, alterations in reward and threat processing are critical potential mediators led by the effect of systemic inflammation on behavioral responses.
As immune stimulation also profoundly affects the perinatal brain development processes involved in cognitive function, some experimental animal studies showed that infection and systemic inflammation during prenatal or neonatal periods impair learning, memory, and attention [261,262,263,264]. Meanwhile, observational human studies found a relationship between prenatal exposure to infection and elevated risk of schizophrenia [265, 266]. Elevated levels of the systemic inflammatory marker IL-6 during childhood are significantly related to an elevated risk of causing psychosis and depression in young adult [179].
Accumulated experimental and observational studies in animals and humans have suggested bidirectional relationships between psychiatric illness and inflammation in peripheral and neuroinflammation over time [177], indicating that susceptibilities associated with emotional and behavioral symptoms and dysfunctional perception of distress could elevate inflammatory responses and sensitization over time or vice versa. Thus, the severities and frequencies of stressful events could be affected by an individual’s susceptibilities, such as personality traits or attachment style, and their environmental risk factors, both of which are critical risk factors for ELS [267].
References
Danese A, McEwen BS. Adverse childhood experiences, allostasis, allostatic load, and age-related disease. Physiol Behav. 2012;106(1):29–39.
Ehlert U. Enduring psychobiological effects of childhood adversity. Psychoneuroendocrinology. 2013;38(9):1850–7.
Miller GE, Chen E, Parker KJ. Psychological stress in childhood and susceptibility to the chronic diseases of aging: moving toward a model of behavioral and biological mechanisms. Psychol Bull. 2011;137(6):959–97.
Felitti VJ, Anda RF, Nordenberg D, Williamson DF, Spitz AM, Edwards V, et al. Relationship of childhood abuse and household dysfunction to many of the leading causes of death in adults. The adverse childhood experiences (ACE) study. Am J Prev Med. 1998;14(4):245–58.
Kalmakis KA, Chandler GE. Health consequences of adverse childhood experiences: a systematic review. J Am Assoc Nurse Pract. 2015;27(8):457–65.
Kessler RC, McLaughlin KA, Green JG, Gruber MJ, Sampson NA, Zaslavsky AM, et al. Childhood adversities and adult psychopathology in the WHO world mental health surveys. Br J Psychiatry. 2010;197(5):378–85.
Korkeila J, Vahtera J, Nabi H, Kivimaki M, Korkeila K, Sumanen M, et al. Childhood adversities, adulthood life events and depression. J Affect Disord. 2010;127(1–3):130–8.
Chen E, Turiano NA, Mroczek DK, Miller GE. Association of reports of childhood abuse and all-cause mortality rates in women. JAMA Psychiat. 2016;73(9):920–7.
Kelly-Irving M, Lepage B, Dedieu D, Bartley M, Blane D, Grosclaude P, et al. Adverse childhood experiences and premature all-cause mortality. Eur J Epidemiol. 2013;28(9):721–34.
Sideli L, Mule A, La Barbera D, Murray RM. Do child abuse and maltreatment increase risk of schizophrenia? Psychiatry Investig. 2012;9(2):87–99.
Scherrer JF, Waterman BM, Heath AC, Bucholz KK, True WR, Jacob T. Are substance use, abuse and dependence associated with study participation? Predictors of offspring nonparticipation in a twin-family study. J Stud Alcohol. 2004;65(1):140–4.
Radford L, Corral S, Bradley C, Fisher H, Bassett C, Howat N, Collishaw S. Child abuse and neglect in the UK today. London: NSPCC; 2011.
Middlebrooks J, Auedage N. The effects of childhood stress on health across the lifespan. Atlanta, GA: CDC; 2008.
Gilbert R, Widom CS, Browne K, Fergusson D, Webb E, Janson S. Burden and consequences of child maltreatment in high-income countries. Lancet. 2009;373(9657):68–81.
Verdolini N, Attademo L, Agius M, Ferranti L, Moretti P, Quartesan R. Traumatic events in childhood and their association with psychiatric illness in the adult. Psychiatr Danub. 2015;27(Suppl 1):S60–70.
Dubowitz H, Pitts SC, Black MM. Measurement of three major subtypes of child neglect. Child Maltreat. 2004;9(4):344–56.
Holmes WC, Slap GB. Sexual abuse of boys: definition, prevalence, correlates, sequelae, and management. JAMA. 1998;280(21):1855–62.
Bennouna-Greene M, Bennouna-Greene V, Berna F, Defranoux L. History of abuse and neglect in patients with schizophrenia who have a history of violence. Child Abuse Negl. 2011;35(5):329–32.
Danese A, Tan M. Childhood maltreatment and obesity: systematic review and meta-analysis. Mol Psychiatry. 2014;19(5):544–54.
Hughes K, Bellis MA, Hardcastle KA, Sethi D, Butchart A, Mikton C, et al. The effect of multiple adverse childhood experiences on health: a systematic review and meta-analysis. Lancet Public Health. 2017;2(8):e356–e66.
Tsehay M, Necho M, Mekonnen W. The role of adverse childhood experience on depression symptom, prevalence, and severity among school going adolescents. Depress Res Treat. 2020;2020:5951792.
Bellis MA, Hughes K, Leckenby N, Hardcastle KA, Perkins C, Lowey H. Measuring mortality and the burden of adult disease associated with adverse childhood experiences in England: a national survey. J Public Health (Oxf). 2015;37(3):445–54.
Flouri E, Francesconi M, Midouhas E, Lewis G. Prenatal and childhood adverse life events, inflammation and depressive symptoms across adolescence. J Affect Disord. 2020;260:577–82.
O'Connor TG, Willoughby MT, Moynihan JA, Messing S, Vallejo Sefair A, Carnahan J, et al. Early childhood risk exposures and inflammation in early adolescence. Brain Behav Immun. 2020;86:22–9.
Slopen N, Kubzansky LD, McLaughlin KA, Koenen KC. Childhood adversity and inflammatory processes in youth: a prospective study. Psychoneuroendocrinology. 2013;38(2):188–200.
Bucci M, Marques SS, Oh D, Harris NB. Toxic stress in children and adolescents. Adv Pediatr Infect Dis. 2016;63(1):403–28.
Franke HA. Toxic stress: effects, prevention and treatment. Children (Basel). 2014;1(3):390–402.
Johnson SB, Riley AW, Granger DA, Riis J. The science of early life toxic stress for pediatric practice and advocacy. Pediatrics. 2013;131(2):319–27.
Danese A, van Harmelen AL. The hidden wounds of childhood trauma. Eur J Psychotraumatol. 2017;8(sup5):137584.
Bateson P, Barker D, Clutton-Brock T, Deb D, D'Udine B, Foley RA, et al. Developmental plasticity and human health. Nature. 2004;430(6998):419–21.
Greenough WT, Black JE, Wallace CS. Experience and brain development. Child Dev. 1987;58(3):539–59.
Anda RF, Brown DW, Dube SR, Bremner JD, Felitti VJ, Giles WH. Adverse childhood experiences and chronic obstructive pulmonary disease in adults. Am J Prev Med. 2008;34(5):396–403.
Brown DW, Anda RF, Felitti VJ, Edwards VJ, Malarcher AM, Croft JB, et al. Adverse childhood experiences are associated with the risk of lung cancer: a prospective cohort study. BMC Public Health. 2010;10:20.
Dube SR, Fairweather D, Pearson WS, Felitti VJ, Anda RF, Croft JB. Cumulative childhood stress and autoimmune diseases in adults. Psychosom Med. 2009;71(2):243–50.
Ader R, Friedman SB. Social factors affecting emotionality and resistance to disease in animals. V. Early separation from the mother and response to a transplanted tumor in the rat. Psychosom Med. 1965;27:119–22.
Solomon GF, Levine S, Kraft JK. Early experience and immunity. Nature. 1968;220(5169):821–2.
Ader R. Developmental psychoneuroimmunology. Dev Psychobiol. 1983;16(4):251–67.
Coe CL. Developmental psychoneuroimmunology revisited. Brain Behav Immun. 1996;10(3):185–7.
Danese A. Developmental psychoneuroimmunology: from bench to bedside. Brain Behav Immun. 2014;36:27–8.
Hennessy MB, Deak T, Schiml-Webb PA, Carlisle CW, O'Brien E. Maternal separation produces, and a second separation enhances, core temperature and passive behavioral responses in Guinea pig pups. Physiol Behav. 2010;100(4):305–10.
Shanks N, Lightman SL. The maternal-neonatal neuro-immune interface: are there long-term implications for inflammatory or stress-related disease? J Clin Invest. 2001;108(11):1567–73.
Coe CL, Rosenberg LT, Levine S. Prolonged effect of psychological disturbance on macrophage chemiluminescence in the squirrel monkey. Brain Behav Immun. 1988;2(2):151–60.
Cole SW, Conti G, Arevalo JM, Ruggiero AM, Heckman JJ, Suomi SJ. Transcriptional modulation of the developing immune system by early life social adversity. Proc Natl Acad Sci U S A. 2012;109(50):20578–83.
Reus GZ, Dos Santos MA, Abelaira HM, Ribeiro KF, Petronilho F, Vuolo F, et al. Imipramine reverses alterations in cytokines and BDNF levels induced by maternal deprivation in adult rats. Behav Brain Res. 2013;242:40–6.
Wieck A, Andersen SL, Brenhouse HC. Evidence for a neuroinflammatory mechanism in delayed effects of early life adversity in rats: relationship to cortical NMDA receptor expression. Brain Behav Immun. 2013;28:218–26.
Brenhouse HC, Andersen SL. Nonsteroidal anti-inflammatory treatment prevents delayed effects of early life stress in rats. Biol Psychiatry. 2011;70(5):434–40.
Behrens MM, Sejnowski TJ. Does schizophrenia arise from oxidative dysregulation of parvalbumin-interneurons in the developing cortex? Neuropharmacology. 2009;57(3):193–200.
Roque A, Ochoa-Zarzosa A, Torner L. Maternal separation activates microglial cells and induces an inflammatory response in the hippocampus of male rat pups, independently of hypothalamic and peripheral cytokine levels. Brain Behav Immun. 2016;55:39–48.
Giridharan VV, Reus GZ, Selvaraj S, Scaini G, Barichello T, Quevedo J. Maternal deprivation increases microglial activation and neuroinflammatory markers in the prefrontal cortex and hippocampus of infant rats. J Psychiatr Res. 2019;115:13–20.
Moya-Perez A, Perez-Villalba A, Benitez-Paez A, Campillo I, Sanz Y. Bifidobacterium CECT 7765 modulates early stress-induced immune, neuroendocrine and behavioral alterations in mice. Brain Behav Immun. 2017;65:43–56.
Wei L, Simen A, Mane S, Kaffman A. Early life stress inhibits expression of a novel innate immune pathway in the developing hippocampus. Neuropsychopharmacology. 2012;37(2):567–80.
Chocyk A, Dudys D, Przyborowska A, Majcher I, Mackowiak M, Wedzony K. Maternal separation affects the number, proliferation and apoptosis of glia cells in the substantia nigra and ventral tegmental area of juvenile rats. Neuroscience. 2011;173:1–18.
Viviani B, Boraso M, Valero M, Gardoni F, Marco EM, Llorente R, et al. Early maternal deprivation immunologically primes hippocampal synapses by redistributing interleukin-1 receptor type I in a sex dependent manner. Brain Behav Immun. 2014;35:135–43.
Takatsuru Y, Nabekura J, Ishikawa T, Kohsaka S, Koibuchi N. Early-life stress increases the motility of microglia in adulthood. J Physiol Sci. 2015;65(2):187–94.
Brenhouse HC, Thompson V. Maternal separation increases IBA-1 expression: a microglia activation marker in the prefrontal cortex of adolescent males following a second hit of stress (abstract). Soc Biol Psychiatry. 2015;77:52s.
Avitsur R, Hunzeker J, Sheridan JF. Role of early stress in the individual differences in host response to viral infection. Brain Behav Immun. 2006;20(4):339–48.
Hennessy MB, Deak T, Schiml-Webb PA. Early attachment-figure separation and increased risk for later depression: potential mediation by proinflammatory processes. Neurosci Biobehav Rev. 2010;34(6):782–90.
Loria AS, Pollock DM, Pollock JS. Early life stress sensitizes rats to angiotensin II-induced hypertension and vascular inflammation in adult life. Hypertension. 2010;55(2):494–9.
O'Mahony SM, Marchesi JR, Scully P, Codling C, Ceolho AM, Quigley EM, et al. Early life stress alters behavior, immunity, and microbiota in rats: implications for irritable bowel syndrome and psychiatric illnesses. Biol Psychiatry. 2009;65(3):263–7.
Tay TL, Sagar DJ, Grun D, Prinz M. Unique microglia recovery population revealed by single-cell RNAseq following neurodegeneration. Acta Neuropathol Commun. 2018;6(1):87.
Cowan CS, Callaghan BL, Kan JM, Richardson R. The lasting impact of early-life adversity on individuals and their descendants: potential mechanisms and hope for intervention. Genes Brain Behav. 2016;15(1):155–68.
Delpech JC, Wei L, Hao J, Yu X, Madore C, Butovsky O, et al. Early life stress perturbs the maturation of microglia in the developing hippocampus. Brain Behav Immun. 2016;57:79–93.
Saavedra LM, Fenton Navarro B, Torner L. Early life stress activates glial cells in the hippocampus but attenuates cytokine secretion in response to an immune challenge in rat pups. Neuroimmunomodulation. 2017;24(4–5):242–55.
Baldy C, Fournier S, Boisjoly-Villeneuve S, Tremblay ME, Kinkead R. The influence of sex and neonatal stress on medullary microglia in rat pups. Exp Physiol. 2018;103(9):1192–9.
Frank MG, Fonken LK, Watkins LR, Maier SF. Microglia: neuroimmune-sensors of stress. Semin Cell Dev Biol. 2019;94:176–85.
Gong Y, Tong L, Yang R, Hu W, Xu X, Wang W, et al. Dynamic changes in hippocampal microglia contribute to depressive-like behavior induced by early social isolation. Neuropharmacology. 2018;135:223–33.
Zhang Y, Xu H, Wang J, Ren F, Shao F, Ellenbroek B, et al. Transient upregulation of immune activity induced by adolescent social stress is involved in cognitive deficit in adult male mice and early intervention with minocycline. Behav Brain Res. 2019;374:112136.
Zhang Y, Xu H, Zhang F, Shao F, Ellenbroek B, Wang J, et al. Deficiencies of microglia and TNFalpha in the mPFC-mediated cognitive inflexibility induced by social stress during adolescence. Brain Behav Immun. 2019;79:256–66.
Lo Iacono L, Catale C, Martini A, Valzania A, Viscomi MT, Chiurchiu V, et al. From traumatic childhood to cocaine abuse: the critical function of the immune system. Biol Psychiatry. 2018;84(12):905–16.
Gomez-Gonzalez B, Escobar A. Prenatal stress alters microglial development and distribution in postnatal rat brain. Acta Neuropathol. 2010;119(3):303–15.
Rupprecht R, Papadopoulos V, Rammes G, Baghai TC, Fan J, Akula N, et al. Translocator protein (18 kDa) (TSPO) as a therapeutic target for neurological and psychiatric disorders. Nat Rev Drug Discov. 2010;9(12):971–88.
Zhao Q, Peng C, Wu X, Chen Y, Wang C, You Z. Maternal sleep deprivation inhibits hippocampal neurogenesis associated with inflammatory response in young offspring rats. Neurobiol Dis. 2014;68:57–65.
Zhao Q, Xie X, Fan Y, Zhang J, Jiang W, Wu X, et al. Phenotypic dysregulation of microglial activation in young offspring rats with maternal sleep deprivation-induced cognitive impairment. Sci Rep. 2015;5:9513.
Bailey MT, Coe CL. Maternal separation disrupts the integrity of the intestinal microflora in infant rhesus monkeys. Dev Psychobiol. 1999;35(2):146–55.
Garcia-Rodenas CL, Bergonzelli GE, Nutten S, Schumann A, Cherbut C, Turini M, et al. Nutritional approach to restore impaired intestinal barrier function and growth after neonatal stress in rats. J Pediatr Gastroenterol Nutr. 2006;43(1):16–24.
Jasarevic E, Howard CD, Misic AM, Beiting DP, Bale TL. Stress during pregnancy alters temporal and spatial dynamics of the maternal and offspring microbiome in a sex-specific manner. Sci Rep. 2017;7:44182.
Jasarevic E, Rodgers AB, Bale TL. A novel role for maternal stress and microbial transmission in early life programming and neurodevelopment. Neurobiol Stress. 2015;1:81–8.
Gareau MG, Silva MA, Perdue MH. Pathophysiological mechanisms of stress-induced intestinal damage. Curr Mol Med. 2008;8(4):274–81.
Carlessi AS, Borba LA, Zugno AI, Quevedo J, Reus GZ. Gut microbiota-brain axis in depression: the role of neuroinflammation. Eur J Neurosci. 2021;53(1):222–35.
Christmas DM, Potokar J, Davies SJ. A biological pathway linking inflammation and depression: activation of indoleamine 2,3-dioxygenase. Neuropsychiatr Dis Treat. 2011;7:431–9.
Kelly JR, Borre Y, Brien CO, Patterson E, El Aidy S, Deane J, et al. Transferring the blues: depression-associated gut microbiota induces neurobehavioural changes in the rat. J Psychiatr Res. 2016;82:109–18.
Pusceddu MM, El Aidy S, Crispie F, O'Sullivan O, Cotter P, Stanton C, et al. N-3 polyunsaturated fatty acids (PUFAs) reverse the impact of early-life stress on the Gut microbiota. PLoS One. 2015;10(10):e0139721.
Wong ML, Inserra A, Lewis MD, Mastronardi CA, Leong L, Choo J, et al. Inflammasome signaling affects anxiety-and depressive-like behavior and gut microbiome composition. Mol Psychiatry. 2016;21(6):797–805.
Hennessy MB, Paik KD, Caraway JD, Schiml PA, Deak T. Proinflammatory activity and the sensitization of depressive-like behavior during maternal separation. Behav Neurosci. 2011;125(3):426–33.
Coe CL, Lubach G, Ershler WB. Immunological consequences of maternal separation in infant primates. New Dir Child Dev. 1989;45:65–91.
Hertzman C, Boyce T. How experience gets under the skin to create gradients in developmental health. Annu Rev Public Health. 2010;31:329–47. 3p following 47
Surtees P, Wainwright N, Day N, Luben R, Brayne C, Khaw KT. Association of depression with peripheral leukocyte counts in EPIC-Norfolk—role of sex and cigarette smoking. J Psychosom Res. 2003;54(4):303–6.
Baumeister D, Akhtar R, Ciufolini S, Pariante CM, Mondelli V. Childhood trauma and adulthood inflammation: a meta-analysis of peripheral C-reactive protein, interleukin-6 and tumour necrosis factor-alpha. Mol Psychiatry. 2016;21(5):642–9.
Tursich M, Neufeld RW, Frewen PA, Harricharan S, Kibler JL, Rhind SG, et al. Association of trauma exposure with proinflammatory activity: a transdiagnostic meta-analysis. Transl Psychiatry. 2014;4:e413.
Kuhlman KR, Horn SR, Chiang JJ, Bower JE. Early life adversity exposure and circulating markers of inflammation in children and adolescents: a systematic review and meta-analysis. Brain Behav Immun. 2020;86:30–42.
Danese A, Pariante CM, Caspi A, Taylor A, Poulton R. Childhood maltreatment predicts adult inflammation in a life-course study. Proc Natl Acad Sci U S A. 2007;104(4):1319–24.
Rasmussen LJH, Moffitt TE, Eugen-Olsen J, Belsky DW, Danese A, Harrington H, et al. Cumulative childhood risk is associated with a new measure of chronic inflammation in adulthood. J Child Psychol Psychiatry. 2019;60(2):199–208.
Cicchetti D, Handley ED, Rogosch FA. Child maltreatment, inflammation, and internalizing symptoms: investigating the roles of C-reactive protein, gene variation, and neuroendocrine regulation. Dev Psychopathol. 2015;27(2):553–66.
Danese A, Moffitt TE, Pariante CM, Ambler A, Poulton R, Caspi A. Elevated inflammation levels in depressed adults with a history of childhood maltreatment. Arch Gen Psychiatry. 2008;65(4):409–15.
Danese A, Caspi A, Williams B, Ambler A, Sugden K, Mika J, et al. Biological embedding of stress through inflammation processes in childhood. Mol Psychiatry. 2011;16(3):244–6.
Fagundes CP, Glaser R, Kiecolt-Glaser JK. Stressful early life experiences and immune dysregulation across the lifespan. Brain Behav Immun. 2013;27(1):8–12.
Miller BJ, Buckley P, Seabolt W, Mellor A, Kirkpatrick B. Meta-analysis of cytokine alterations in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry. 2011;70(7):663–71.
Slavich GM, Irwin MR. From stress to inflammation and major depressive disorder: a social signal transduction theory of depression. Psychol Bull. 2014;140(3):774–815.
Tyrka AR, Parade SH, Valentine TR, Eslinger NM, Seifer R. Adversity in preschool-aged children: effects on salivary interleukin-1beta. Dev Psychopathol. 2015;27(2):567–76.
Carpenter LL, Gawuga CE, Tyrka AR, Lee JK, Anderson GM, Price LH. Association between plasma IL-6 response to acute stress and early-life adversity in healthy adults. Neuropsychopharmacology. 2010;35(13):2617–23.
Li L, Chassan RA, Bruer EH, Gower BA, Shelton RC. Childhood maltreatment increases the risk for visceral obesity. Obesity (Silver Spring). 2015;23(8):1625–32.
Rasmussen LJH, Moffitt TE, Arseneault L, Danese A, Eugen-Olsen J, Fisher HL, et al. Association of adverse experiences and exposure to violence in childhood and adolescence with inflammatory burden in young people. JAMA Pediatr. 2020;174(1):38–47.
Dekkers PE, ten Hove T, te Velde AA, van Deventer SJ, van Der Poll T. Upregulation of monocyte urokinase plasminogen activator receptor during human endotoxemia. Infect Immun. 2000;68(4):2156–60.
Ostrowski SR, Piironen T, Hoyer-Hansen G, Gerstoft J, Pedersen BK, Ullum H. Reduced release of intact and cleaved urokinase receptor in stimulated whole-blood cultures from human immunodeficiency virus-1-infected patients. Scand J Immunol. 2005;61(4):347–56.
Blasi F, Carmeliet P. uPAR: a versatile signalling orchestrator. Nat Rev Mol Cell Biol. 2002;3(12):932.
Hunter CA, Jones SA. IL-6 as a keystone cytokine in health and disease. Nat Immunol. 2015;16(5):448–57.
Rhodes B, Furnrohr BG, Vyse TJ. C-reactive protein in rheumatology: biology and genetics. Nat Rev Rheumatol. 2011;7(5):282–9.
Desmedt S, Desmedt V, Delanghe JR, Speeckaert R, Speeckaert MM. The intriguing role of soluble urokinase receptor in inflammatory diseases. Crit Rev Clin Lab Sci. 2017;54(2):117–33.
Lyngbaek S, Sehestedt T, Marott JL, Hansen TW, Olsen MH, Andersen O, et al. CRP and suPAR are differently related to anthropometry and subclinical organ damage. Int J Cardiol. 2013;167(3):781–5.
Eugen-Olsen J, Andersen O, Linneberg A, Ladelund S, Hansen TW, Langkilde A, et al. Circulating soluble urokinase plasminogen activator receptor predicts cancer, cardiovascular disease, diabetes and mortality in the general population. J Intern Med. 2010;268(3):296–308.
Rasmussen LJ, Ladelund S, Haupt TH, Ellekilde G, Poulsen JH, Iversen K, et al. Soluble urokinase plasminogen activator receptor (suPAR) in acute care: a strong marker of disease presence and severity, readmission and mortality. A retrospective cohort study. Emerg Med J. 2016;33(11):769–75.
Bourassa KJ, Rasmussen LJH, Danese A, Eugen-Olsen J, Harrington H, Houts R, et al. Linking stressful life events and chronic inflammation using suPAR (soluble urokinase plasminogen activator receptor). Brain Behav Immun. 2021;97:79–88.
Bartlett JA, Demetrikopoulos MK, Schleifer SJ, Keller SE. Phagocytosis and killing of Staphylococcus aureus: effects of stress and depression in children. Clin Diagn Lab Immunol. 1997;4(3):362–6.
Meyer RJ, Haggerty RJ. Streptococcal infections in families. Factors altering individual susceptibility. Pediatrics. 1962;29:539–49.
Birmaher B, Rabin BS, Garcia MR, Jain U, Whiteside TL, Williamson DE, et al. Cellular immunity in depressed, conduct disorder, and normal adolescents: role of adverse life events. J Am Acad Child Adolesc Psychiatry. 1994;33(5):671–8.
Witek Janusek L, Tell D, Albuquerque K, Mathews HL. Childhood adversity increases vulnerability for behavioral symptoms and immune dysregulation in women with breast cancer. Brain Behav Immun. 2013;30(Suppl):S149–62.
Wyman PA, Moynihan J, Eberly S, Cox C, Cross W, Jin X, et al. Association of family stress with natural killer cell activity and the frequency of illnesses in children. Arch Pediatr Adolesc Med. 2007;161(3):228–34.
Naliboff BD, Benton D, Solomon GF, Morley JE, Fahey JL, Bloom ET, et al. Immunological changes in young and old adults during brief laboratory stress. Psychosom Med. 1991;53(2):121–32.
Ayaydin H, Abali O, Akdeniz NO, Kok BE, Gunes A, Yildirim A, et al. Immune system changes after sexual abuse in adolescents. Pediatr Int. 2016;58(2):105–12.
Waldron JC, Scarpa A, Kim-Spoon J, Coe CL. Adult sexual experiences as a mediator between child abuse and current secretory immunoglobulin a levels. J Interpers Violence. 2016;31(5):942–60.
Boeck C, Koenig AM, Schury K, Geiger ML, Karabatsiakis A, Wilker S, et al. Inflammation in adult women with a history of child maltreatment: the involvement of mitochondrial alterations and oxidative stress. Mitochondrion. 2016;30:197–207.
Bennett JM, Glaser R, Malarkey WB, Beversdorf DQ, Peng J, Kiecolt-Glaser JK. Inflammation and reactivation of latent herpesviruses in older adults. Brain Behav Immun. 2012;26(5):739–46.
Elwenspoek MMC, Sias K, Hengesch X, Schaan VK, Leenen FAD, Adams P, et al. T cell Immunosenescence after early life adversity: association with cytomegalovirus infection. Front Immunol. 2017;8:1263.
Elwenspoek MMC, Hengesch X, Leenen FAD, Schritz A, Sias K, Schaan VK, et al. Proinflammatory T cell status associated with early life adversity. J Immunol. 2017;199(12):4046–55.
Elwenspoek MMC, Kuehn A, Muller CP, Turner JD. The effects of early life adversity on the immune system. Psychoneuroendocrinology. 2017;82:140–54.
Mondelli V, Vernon AC, Turkheimer F, Dazzan P, Pariante CM. Brain microglia in psychiatric disorders. Lancet Psychiatry. 2017;4(7):563–72.
Chen MK, Guilarte TR. Translocator protein 18 kDa (TSPO): molecular sensor of brain injury and repair. Pharmacol Ther. 2008;118(1):1–17.
Rampp C, Eichelkraut A, Best J, Czamara D, Rex-Haffner M, Uhr M, et al. Sex-related differential response to dexamethasone in endocrine and immune measures in depressed in-patients and healthy controls. J Psychiatr Res. 2018;98:107–15.
Rohleder N, Schommer NC, Hellhammer DH, Engel R, Kirschbaum C. Sex differences in glucocorticoid sensitivity of proinflammatory cytokine production after psychosocial stress. Psychosom Med. 2001;63(6):966–72.
Baldwin JR, Arseneault L, Caspi A, Fisher HL, Moffitt TE, Odgers CL, et al. Childhood victimization and inflammation in young adulthood: a genetically sensitive cohort study. Brain Behav Immun. 2018;67:211–7.
Cattaneo A, Macchi F, Plazzotta G, Veronica B, Bocchio-Chiavetto L, Riva MA, et al. Inflammation and neuronal plasticity: a link between childhood trauma and depression pathogenesis. Front Cell Neurosci. 2015;9:40.
Walker FR, Beynon SB, Jones KA, Zhao Z, Kongsui R, Cairns M, et al. Dynamic structural remodelling of microglia in health and disease: a review of the models, the signals and the mechanisms. Brain Behav Immun. 2014;37:1–14.
Hemmings SMJ, Malan-Muller S, van den Heuvel LL, Demmitt BA, Stanislawski MA, Smith DG, et al. The microbiome in posttraumatic stress disorder and trauma-exposed controls: an exploratory study. Psychosom Med. 2017;79(8):936–46.
Callaghan BL, Fields A, Gee DG, Gabard-Durnam L, Caldera C, Humphreys KL, et al. Mind and gut: associations between mood and gastrointestinal distress in children exposed to adversity. Dev Psychopathol. 2020;32(1):309–28.
Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. 2012;13(10):701–12.
Hooper LV, Littman DR, Macpherson AJ. Interactions between the microbiota and the immune system. Science. 2012;336(6086):1268–73.
Dantzer R, O'Connor JC, Freund GG, Johnson RW, Kelley KW. From inflammation to sickness and depression: when the immune system subjugates the brain. Nat Rev Neurosci. 2008;9(1):46–56.
Hart BL. Biological basis of the behavior of sick animals. Neurosci Biobehav Rev. 1988;12(2):123–37.
Miller AH, Maletic V, Raison CL. Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol Psychiatry. 2009;65(9):732–41.
Reichenberg A, Yirmiya R, Schuld A, Kraus T, Haack M, Morag A, et al. Cytokine-associated emotional and cognitive disturbances in humans. Arch Gen Psychiatry. 2001;58(5):445–52.
Holland FH, Ganguly P, Potter DN, Chartoff EH, Brenhouse HC. Early life stress disrupts social behavior and prefrontal cortex parvalbumin interneurons at an earlier time-point in females than in males. Neurosci Lett. 2014;566:131–6.
Leussis MP, Freund N, Brenhouse HC, Thompson BS, Andersen SL. Depressive-like behavior in adolescents after maternal separation: sex differences, controllability, and GABA. Dev Neurosci. 2012;34(2–3):210–7.
Frank MG, Watkins LR, Maier SF. Stress- and glucocorticoid-induced priming of neuroinflammatory responses: potential mechanisms of stress-induced vulnerability to drugs of abuse. Brain Behav Immun. 2011;25(Suppl 1):S21–8.
Wohleb ES, Hanke ML, Corona AW, Powell ND, Stiner LM, Bailey MT, et al. ta-adrenergic receptor antagonism prevents anxiety-like behavior and microglial reactivity induced by repeated social defeat. J Neurosci. 2011;31(17):6277–88.
Wohleb ES, Powell ND, Godbout JP, Sheridan JF. Stress-induced recruitment of bone marrow-derived monocytes to the brain promotes anxiety-like behavior. J Neurosci. 2013;33(34):13820–33.
Hennessy MB, Schiml-Webb PA, Miller EE, Maken DS, Bullinger KL, Deak T. Anti-inflammatory agents attenuate the passive responses of Guinea pig pups: evidence for stress-induced sickness behavior during maternal separation. Psychoneuroendocrinology. 2007;32(5):508–15.
Perkeybile AM, Schiml-Webb PA, O'Brien E, Deak T, Hennessy MB. Anti-inflammatory influences on behavioral, but not cortisol, responses during maternal separation. Psychoneuroendocrinology. 2009;34(7):1101–8.
Hartung HP, Heininger K, Schafer B, Fierz W, Toyka KV. Immune mechanisms in inflammatory polyneuropathy. Ann N Y Acad Sci. 1988;540:122–61.
Giovanoli S, Engler H, Engler A, Richetto J, Feldon J, Riva MA, et al. Preventive effects of minocycline in a neurodevelopmental two-hit model with relevance to schizophrenia. Transl Psychiatry. 2016;6:e772.
Youm YH, Grant RW, McCabe LR, Albarado DC, Nguyen KY, Ravussin A, et al. Canonical Nlrp3 inflammasome links systemic low-grade inflammation to functional decline in aging. Cell Metab. 2013;18(4):519–32.
Eisenberger NI, Berkman ET, Inagaki TK, Rameson LT, Mashal NM, Irwin MR. Inflammation-induced anhedonia: endotoxin reduces ventral striatum responses to reward. Biol Psychiatry. 2010;68(8):748–54.
Capuron L, Pagnoni G, Drake DF, Woolwine BJ, Spivey JR, Crowe RJ, et al. Dopaminergic mechanisms of reduced basal ganglia responses to hedonic reward during interferon alfa administration. Arch Gen Psychiatry. 2012;69(10):1044–53.
Smith RS. The macrophage theory of depression. Med Hypotheses. 1991;35(4):298–306.
Musselman DL, Lawson DH, Gumnick JF, Manatunga AK, Penna S, Goodkin RS, et al. Paroxetine for the prevention of depression induced by high-dose interferon alfa. N Engl J Med. 2001;344(13):961–6.
Kohler O, Benros ME, Nordentoft M, Farkouh ME, Iyengar RL, Mors O, et al. Effect of anti-inflammatory treatment on depression, depressive symptoms, and adverse effects: a systematic review and meta-analysis of randomized clinical trials. JAMA Psychiat. 2014;71(12):1381–91.
Chaudhry IB, Hallak J, Husain N, Minhas F, Stirling J, Richardson P, et al. Minocycline benefits negative symptoms in early schizophrenia: a randomised double-blind placebo-controlled clinical trial in patients on standard treatment. J Psychopharmacol. 2012;26(9):1185–93.
Levkovitz Y, Mendlovich S, Riwkes S, Braw Y, Levkovitch-Verbin H, Gal G, et al. A double-blind, randomized study of minocycline for the treatment of negative and cognitive symptoms in early-phase schizophrenia. J Clin Psychiatry. 2010;71(2):138–49.
Harrison NA, Brydon L, Walker C, Gray MA, Steptoe A, Critchley HD. Inflammation causes mood changes through alterations in subgenual cingulate activity and mesolimbic connectivity. Biol Psychiatry. 2009;66(5):407–14.
Juengling FD, Ebert D, Gut O, Engelbrecht MA, Rasenack J, Nitzsche EU, et al. Prefrontal cortical hypometabolism during low-dose interferon alpha treatment. Psychopharmacology. 2000;152(4):383–9.
Reus GZ, Fries GR, Stertz L, Badawy M, Passos IC, Barichello T, et al. The role of inflammation and microglial activation in the pathophysiology of psychiatric disorders. Neuroscience. 2015;300:141–54.
van Berckel BN, Bossong MG, Boellaard R, Kloet R, Schuitemaker A, Caspers E, et al. Microglia activation in recent-onset schizophrenia: a quantitative (R)-[11C]PK11195 positron emission tomography study. Biol Psychiatry. 2008;64(9):820–2.
Torres-Platas SG, Cruceanu C, Chen GG, Turecki G, Mechawar N. Evidence for increased microglial priming and macrophage recruitment in the dorsal anterior cingulate white matter of depressed suicides. Brain Behav Immun. 2014;42:50–9.
Frick LR, Williams K, Pittenger C. Microglial dysregulation in psychiatric disease. Clin Dev Immunol. 2013;2013:608654.
Paolicelli RC, Bolasco G, Pagani F, Maggi L, Scianni M, Panzanelli P, et al. Synaptic pruning by microglia is necessary for normal brain development. Science. 2011;333(6048):1456–8.
Parkhurst CN, Yang G, Ninan I, Savas JN, Yates JR 3rd, Lafaille JJ, et al. Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell. 2013;155(7):1596–609.
Tremblay ME, Lowery RL, Majewska AK. Microglial interactions with synapses are modulated by visual experience. PLoS Biol. 2010;8(11):e1000527.
Bloomfield PS, Selvaraj S, Veronese M, Rizzo G, Bertoldo A, Owen DR, et al. Microglial activity in people at ultra high risk of psychosis and in schizophrenia: an [(11)C]PBR28 PET brain imaging study. Am J Psychiatry. 2016;173(1):44–52.
Ligthart S, Vaez A, Vosa U, Stathopoulou MG, de Vries PS, Prins BP, et al. Genome analyses of >200,000 individuals identify 58 loci for chronic inflammation and highlight pathways that link inflammation and complex disorders. Am J Hum Genet. 2018;103(5):691–706.
Speer K, Upton D, Semple S, McKune A. Systemic low-grade inflammation in post-traumatic stress disorder: a systematic review. J Inflamm Res. 2018;11:111–21.
Jones KA, Thomsen C. The role of the innate immune system in psychiatric disorders. Mol Cell Neurosci. 2013;53:52–62.
Valkanova V, Ebmeier KP, Allan CL. CRP, IL-6 and depression: a systematic review and meta-analysis of longitudinal studies. J Affect Disord. 2013;150(3):736–44.
Courtet P, Jaussent I, Genty C, Dupuy AM, Guillaume S, Ducasse D, et al. Increased CRP levels may be a trait marker of suicidal attempt. Eur Neuropsychopharmacol. 2015;25(10):1824–31.
Cappi C, Muniz RK, Sampaio AS, Cordeiro Q, Brentani H, Palacios SA, et al. Association study between functional polymorphisms in the TNF-alpha gene and obsessive-compulsive disorder. Arq Neuropsiquiatr. 2012;70(2):87–90.
Denys D, Fluitman S, Kavelaars A, Heijnen C, Westenberg H. Decreased TNF-alpha and NK activity in obsessive-compulsive disorder. Psychoneuroendocrinology. 2004;29(7):945–52.
Konuk N, Tekin IO, Ozturk U, Atik L, Atasoy N, Bektas S, et al. Plasma levels of tumor necrosis factor-alpha and interleukin-6 in obsessive compulsive disorder. Mediat Inflamm. 2007;2007:65704.
Monteleone P, Catapano F, Fabrazzo M, Tortorella A, Maj M. Decreased blood levels of tumor necrosis factor-alpha in patients with obsessive-compulsive disorder. Neuropsychobiology. 1998;37(4):182–5.
Matthews KA, Schott LL, Bromberger JT, Cyranowski JM, Everson-Rose SA, Sowers M. Are there bi-directional associations between depressive symptoms and C-reactive protein in mid-life women? Brain Behav Immun. 2010;24(1):96–101.
Howren MB, Lamkin DM, Suls J. Associations of depression with C-reactive protein, IL-1, and IL-6: a meta-analysis. Psychosom Med. 2009;71(2):171–86.
Khandaker GM, Pearson RM, Zammit S, Lewis G, Jones PB. Association of serum interleukin 6 and C-reactive protein in childhood with depression and psychosis in young adult life: a population-based longitudinal study. JAMA Psychiat. 2014;71(10):1121–8.
Eraly SA, Nievergelt CM, Maihofer AX, Barkauskas DA, Biswas N, Agorastos A, et al. Assessment of plasma C-reactive protein as a biomarker of posttraumatic stress disorder risk. JAMA Psychiat. 2014;71(4):423–31.
Michopoulos V, Rothbaum AO, Jovanovic T, Almli LM, Bradley B, Rothbaum BO, et al. Association of CRP genetic variation and CRP level with elevated PTSD symptoms and physiological responses in a civilian population with high levels of trauma. Am J Psychiatry. 2015;172(4):353–62.
Miller AH, Raison CL. The role of inflammation in depression: from evolutionary imperative to modern treatment target. Nat Rev Immunol. 2016;16(1):22–34.
Maes M, Lambrechts J, Bosmans E, Jacobs J, Suy E, Vandervorst C, et al. Evidence for a systemic immune activation during depression: results of leukocyte enumeration by flow cytometry in conjunction with monoclonal antibody staining. Psychol Med. 1992;22(1):45–53.
Raison CL, Rutherford RE, Woolwine BJ, Shuo C, Schettler P, Drake DF, et al. A randomized controlled trial of the tumor necrosis factor antagonist infliximab for treatment-resistant depression: the role of baseline inflammatory biomarkers. JAMA Psychiat. 2013;70(1):31–41.
Dantzer R. Depression and inflammation: an intricate relationship. Biol Psychiatry. 2012;71(1):4–5.
Dowlati Y, Herrmann N, Swardfager W, Liu H, Sham L, Reim EK, et al. A meta-analysis of cytokines in major depression. Biol Psychiatry. 2010;67(5):446–57.
Krishnadas R, Cavanagh J. Depression: an inflammatory illness? J Neurol Neurosurg Psychiatry. 2012;83(5):495–502.
Dhabhar FS, Burke HM, Epel ES, Mellon SH, Rosser R, Reus VI, et al. Low serum IL-10 concentrations and loss of regulatory association between IL-6 and IL-10 in adults with major depression. J Psychiatr Res. 2009;43(11):962–9.
Muller N. Immunology of major depression. Neuroimmunomodulation. 2014;21(2–3):123–30.
Miller GE, Cole SW. Clustering of depression and inflammation in adolescents previously exposed to childhood adversity. Biol Psychiatry. 2012;72(1):34–40.
Leboyer M, Soreca I, Scott J, Frye M, Henry C, Tamouza R, et al. Can bipolar disorder be viewed as a multi-system inflammatory disease? J Affect Disord. 2012;141(1):1–10.
Kim HK, Andreazza AC, Elmi N, Chen W, Young LT. Nod-like receptor pyrin containing 3 (NLRP3) in the post-mortem frontal cortex from patients with bipolar disorder: a potential mediator between mitochondria and immune-activation. J Psychiatr Res. 2016;72:43–50.
Modabbernia A, Taslimi S, Brietzke E, Ashrafi M. Cytokine alterations in bipolar disorder: a meta-analysis of 30 studies. Biol Psychiatry. 2013;74(1):15–25.
Dargel AA, Godin O, Kapczinski F, Kupfer DJ, Leboyer M. C-reactive protein alterations in bipolar disorder: a meta-analysis. J Clin Psychiatry. 2015;76(2):142–50.
Mondelli V, Ciufolini S, Belvederi Murri M, Bonaccorso S, Di Forti M, Giordano A, et al. Cortisol and inflammatory biomarkers predict poor treatment response in first episode psychosis. Schizophr Bull. 2015;41(5):1162–70.
Miller BJ, Culpepper N, Rapaport MH. C-reactive protein levels in schizophrenia: a review and meta-analysis. Clin Schizophr Relat Psychoses. 2014;7(4):223–30.
Metcalf SA, Jones PB, Nordstrom T, Timonen M, Maki P, Miettunen J, et al. Serum C-reactive protein in adolescence and risk of schizophrenia in adulthood: a prospective birth cohort study. Brain Behav Immun. 2017;59:253–9.
Fernandes BS, Steiner J, Bernstein HG, Dodd S, Pasco JA, Dean OM, et al. C-reactive protein is increased in schizophrenia but is not altered by antipsychotics: meta-analysis and implications. Mol Psychiatry. 2016;21(4):554–64.
Passos IC, Vasconcelos-Moreno MP, Costa LG, Kunz M, Brietzke E, Quevedo J, et al. Inflammatory markers in post-traumatic stress disorder: a systematic review, meta-analysis, and meta-regression. Lancet Psychiatry. 2015;2(11):1002–12.
Cryan JF, Holmes A. The ascent of mouse: advances in modelling human depression and anxiety. Nat Rev Drug Discov. 2005;4(9):775–90.
Sanchez MM, Ladd CO, Plotsky PM. Early adverse experience as a developmental risk factor for later psychopathology: evidence from rodent and primate models. Dev Psychopathol. 2001;13(3):419–49.
Francis D, Diorio J, Liu D, Meaney MJ. Nongenomic transmission across generations of maternal behavior and stress responses in the rat. Science. 1999;286(5442):1155–8.
Harlow HF, Dodsworth RO, Harlow MK. Total social isolation in monkeys. Proc Natl Acad Sci U S A. 1965;54(1):90–7.
Hinde RA, Spencer-Booth Y. Effects of brief separation from mother on rhesus monkeys. Science. 1971;173(3992):111–8.
Levine S, Chevalier JA, Korchin SJ. The effects of early shock and handling on later avoidance learning. J Pers. 1956;24(4):475–93.
Suomi SJ. Early determinants of behaviour: evidence from primate studies. Br Med Bull. 1997;53(1):170–84.
Spitz RA. Hospitalism; an inquiry into the genesis of psychiatric conditions in early childhood. Psychoanal Study Child. 1945;1:53–74.
Bowlby J. Maternal care and mental health. Bull World Health Organ. 1951;3(3):355–533.
Kendler KS, Bulik CM, Silberg J, Hettema JM, Myers J, Prescott CA. Childhood sexual abuse and adult psychiatric and substance use disorders in women: an epidemiological and cotwin control analysis. Arch Gen Psychiatry. 2000;57(10):953–9.
Poole JC, Kim HS, Dobson KS, Hodgins DC. Adverse childhood experiences and disordered gambling: assessing the mediating role of emotion Dysregulation. J Gambl Stud. 2017;33(4):1187–200.
Widom CS, DuMont K, Czaja SJ. A prospective investigation of major depressive disorder and comorbidity in abused and neglected children grown up. Arch Gen Psychiatry. 2007;64(1):49–56.
Green JG, McLaughlin KA, Berglund PA, Gruber MJ, Sampson NA, Zaslavsky AM, et al. Childhood adversities and adult psychiatric disorders in the national comorbidity survey replication I: associations with first onset of DSM-IV disorders. Arch Gen Psychiatry. 2010;67(2):113–23.
Kessler RC, Davis CG, Kendler KS. Childhood adversity and adult psychiatric disorder in the US National Comorbidity Survey. Psychol Med. 1997;27(5):1101–19.
McLaughlin KA, Greif Green J, Gruber MJ, Sampson NA, Zaslavsky AM, Kessler RC. Childhood adversities and first onset of psychiatric disorders in a national sample of US adolescents. Arch Gen Psychiatry. 2012;69(11):1151–60.
Schaefer JD, Moffitt TE, Arseneault L, Danese A, Fisher HL, Houts R, et al. Adolescent victimization and early-adult psychopathology: approaching causal inference using a longitudinal twin study to rule out noncausal explanations. Clin Psychol Sci. 2018;6(3):352–71.
Gilbert R, Kemp A, Thoburn J, Sidebotham P, Radford L, Glaser D, et al. Recognising and responding to child maltreatment. Lancet. 2009;373(9658):167–80.
Varese F, Smeets F, Drukker M, Lieverse R, Lataster T, Viechtbauer W, et al. Childhood adversities increase the risk of psychosis: a meta-analysis of patient-control, prospective-and cross-sectional cohort studies. Schizophr Bull. 2012;38(4):661–71.
Widom CS. Posttraumatic stress disorder in abused and neglected children grown up. Am J Psychiatry. 1999;156(8):1223–9.
Mueller SC, Hardin MG, Korelitz K, Daniele T, Bemis J, Dozier M, et al. Incentive effect on inhibitory control in adolescents with early-life stress: an antisaccade study. Child Abuse Negl. 2012;36(3):217–25.
Goff B, Gee DG, Telzer EH, Humphreys KL, Gabard-Durnam L, Flannery J, et al. Reduced nucleus accumbens reactivity and adolescent depression following early-life stress. Neuroscience. 2013;249:129–38.
Mehta MA, Gore-Langton E, Golembo N, Colvert E, Williams SC, Sonuga-Barke E. Hyporesponsive reward anticipation in the basal ganglia following severe institutional deprivation early in life. J Cogn Neurosci. 2010;22(10):2316–25.
Brown GWHT. Social origins of depression: a study of psychiatric disorder in women. New York: The Free Press; 1978.
Danese A, Moffitt TE, Harrington H, Milne BJ, Polanczyk G, Pariante CM, et al. Adverse childhood experiences and adult risk factors for age-related disease: depression, inflammation, and clustering of metabolic risk markers. Arch Pediatr Adolesc Med. 2009;163(12):1135–43.
Agnew-Blais J, Danese A. Childhood maltreatment and unfavourable clinical outcomes in bipolar disorder: a systematic review and meta-analysis. Lancet Psychiatry. 2016;3(4):342–9.
Maercker A, Brewin CR, Bryant RA, Cloitre M, van Ommeren M, Jones LM, et al. Diagnosis and classification of disorders specifically associated with stress: proposals for ICD-11. World Psychiatry. 2013;12(3):198–206.
Galea I, Bechmann I, Perry VH. What is immune privilege (not)? Trends Immunol. 2007;28(1):12–8.
Yirmiya R, Goshen I. Immune modulation of learning, memory, neural plasticity and neurogenesis. Brain Behav Immun. 2011;25(2):181–213.
Gillespie CFGS, Binder EB, Schatzberg AF, Nemeroff CB. In: Schatzberg AF, Nemeroff CB, editors. Textbook of psychopharmacology. New York: America Psychiatric Publishing; 2009.
Maes M, Leonard BE, Myint AM, Kubera M, Verkerk R. The new '5-HT' hypothesis of depression: cell-mediated immune activation induces indoleamine 2,3-dioxygenase, which leads to lower plasma tryptophan and an increased synthesis of detrimental tryptophan catabolites (TRYCATs), both of which contribute to the onset of depression. Prog Neuro-Psychopharmacol Biol Psychiatry. 2011;35(3):702–21.
Raison CL, Dantzer R, Kelley KW, Lawson MA, Woolwine BJ, Vogt G, et al. CSF concentrations of brain tryptophan and kynurenines during immune stimulation with IFN-alpha: relationship to CNS immune responses and depression. Mol Psychiatry. 2010;15(4):393–403.
Tavares RG, Tasca CI, Santos CE, Alves LB, Porciuncula LO, Emanuelli T, et al. Quinolinic acid stimulates synaptosomal glutamate release and inhibits glutamate uptake into astrocytes. Neurochem Int. 2002;40(7):621–7.
Hardingham GE, Fukunaga Y, Bading H. Extrasynaptic NMDARs oppose synaptic NMDARs by triggering CREB shut-off and cell death pathways. Nat Neurosci. 2002;5(5):405–14.
Chao CC, Hu S, Molitor TW, Shaskan EG, Peterson PK. Activated microglia mediate neuronal cell injury via a nitric oxide mechanism. J Immunol. 1992;149(8):2736–41.
Kehrer JP. Cause-effect of oxidative stress and apoptosis. Teratology. 2000;62(4):235–6.
Uttara B, Singh AV, Zamboni P, Mahajan RT. Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol. 2009;7(1):65–74.
Beal MF. Bioenergetic approaches for neuroprotection in parkinson's disease. Ann Neurol. 2003;53(Suppl 3):S39–47. discussion S-8
Christen Y. Oxidative stress and Alzheimer disease. Am J Clin Nutr. 2000;71(2):621S.
Mahadik SP, Mukherjee S. Free radical pathology and antioxidant defense in schizophrenia: a review. Schizophr Res. 1996;19(1):1–17.
Perry VH, Newman TA, Cunningham C. The impact of systemic infection on the progression of neurodegenerative disease. Nat Rev Neurosci. 2003;4(2):103–12.
Breder CD, Hazuka C, Ghayur T, Klug C, Huginin M, Yasuda K, et al. Regional induction of tumor necrosis factor alpha expression in the mouse brain after systemic lipopolysaccharide administration. Proc Natl Acad Sci U S A. 1994;91(24):11393–7.
Monje ML, Toda H, Palmer TD. Inflammatory blockade restores adult hippocampal neurogenesis. Science. 2003;302(5651):1760.
Weaver IC, Cervoni N, Champagne FA, D'Alessio AC, Sharma S, Seckl JR, et al. Epigenetic programming by maternal behavior. Nat Neurosci. 2004;7(8):847–54.
Klengel T, Mehta D, Anacker C, Rex-Haffner M, Pruessner JC, Pariante CM, et al. Allele-specific FKBP5 DNA demethylation mediates gene-childhood trauma interactions. Nat Neurosci. 2013;16(1):33–41.
Miller GE, Chen E. Harsh family climate in early life presages the emergence of a proinflammatory phenotype in adolescence. Psychol Sci. 2010;21(6):848–56.
Gregor MF, Hotamisligil GS. Inflammatory mechanisms in obesity. Annu Rev Immunol. 2011;29:415.
Gregory AM, Sadeh A. Annual research review: sleep problems in childhood psychiatric disorders—a review of the latest science. J Child Psychol Psychiatry. 2016;57(3):296–317.
Kajeepeta S, Gelaye B, Jackson CL, Williams MA. Adverse childhood experiences are associated with adult sleep disorders: a systematic review. Sleep Med. 2015;16(3):320–30.
Noll JG, Trickett PK, Susman EJ, Putnam FW. Sleep disturbances and childhood sexual abuse. J Pediatr Psychol. 2006;31(5):469–80.
Irwin MR, Wang M, Campomayor CO, Collado-Hidalgo A, Cole S. Sleep deprivation and activation of morning levels of cellular and genomic markers of inflammation. Arch Intern Med. 2006;166(16):1756–62.
Miller MA, Kandala NB, Kivimaki M, Kumari M, Brunner EJ, Lowe GD, et al. Gender differences in the cross-sectional relationships between sleep duration and markers of inflammation: Whitehall II study. Sleep. 2009;32(7):857–64.
Felger JC, Mun J, Kimmel HL, Nye JA, Drake DF, Hernandez CR, et al. Chronic interferon-alpha decreases dopamine 2 receptor binding and striatal dopamine release in association with anhedonia-like behavior in nonhuman primates. Neuropsychopharmacology. 2013;38(11):2179–87.
Dowell NG, Cooper EA, Tibble J, Voon V, Critchley HD, Cercignani M, et al. Acute changes in striatal microstructure predict the development of interferon-alpha induced fatigue. Biol Psychiatry. 2016;79(4):320–8.
Haroon E, Woolwine BJ, Chen X, Pace TW, Parekh S, Spivey JR, et al. IFN-alpha-induced cortical and subcortical glutamate changes assessed by magnetic resonance spectroscopy. Neuropsychopharmacology. 2014;39(7):1777–85.
Harrison NA, Cercignani M, Voon V, Critchley HD. Effects of inflammation on hippocampus and substantia nigra responses to novelty in healthy human participants. Neuropsychopharmacology. 2015;40(4):831–8.
Harrison NA, Voon V, Cercignani M, Cooper EA, Pessiglione M, Critchley HD. A Neurocomputational account of how inflammation enhances sensitivity to punishments versus rewards. Biol Psychiatry. 2016;80(1):73–81.
Harrison NA, Brydon L, Walker C, Gray MA, Steptoe A, Dolan RJ, et al. Neural origins of human sickness in interoceptive responses to inflammation. Biol Psychiatry. 2009;66(5):415–22.
Slavich GM, Way BM, Eisenberger NI, Taylor SE. Neural sensitivity to social rejection is associated with inflammatory responses to social stress. Proc Natl Acad Sci U S A. 2010;107(33):14817–22.
Eisenberger NI, Lieberman MD. Why rejection hurts: a common neural alarm system for physical and social pain. Trends Cogn Sci. 2004;8(7):294–300.
Muscatell KA, Dedovic K, Slavich GM, Jarcho MR, Breen EC, Bower JE, et al. Greater amygdala activity and dorsomedial prefrontal-amygdala coupling are associated with enhanced inflammatory responses to stress. Brain Behav Immun. 2015;43:46–53.
Inagaki TK, Muscatell KA, Irwin MR, Cole SW, Eisenberger NI. Inflammation selectively enhances amygdala activity to socially threatening images. NeuroImage. 2012;59(4):3222–6.
Bilbo SD, Schwarz JM. Early-life programming of later-life brain and behavior: a critical role for the immune system. Front Behav Neurosci. 2009;3:14.
Knuesel I, Chicha L, Britschgi M, Schobel SA, Bodmer M, Hellings JA, et al. Maternal immune activation and abnormal brain development across CNS disorders. Nat Rev Neurol. 2014;10(11):643–60.
Patterson PH. Immune involvement in schizophrenia and autism: etiology, pathology and animal models. Behav Brain Res. 2009;204(2):313–21.
Short SJ, Lubach GR, Karasin AI, Olsen CW, Styner M, Knickmeyer RC, et al. Maternal influenza infection during pregnancy impacts postnatal brain development in the rhesus monkey. Biol Psychiatry. 2010;67(10):965–73.
Khandaker GM, Zimbron J, Lewis G, Jones PB. Prenatal maternal infection, neurodevelopment and adult schizophrenia: a systematic review of population-based studies. Psychol Med. 2013;43(2):239–57.
Mednick SA, Machon RA, Huttunen MO, Bonett D. Adult schizophrenia following prenatal exposure to an influenza epidemic. Arch Gen Psychiatry. 1988;45(2):189–92.
Danese A, McCrory E. Child maltreatment. In: Rutter’s textbook of child & adolescent psychiatry. 6th ed. London: Wiley-Blackwell; 2015.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Shin, S.H., Kim, YK. (2023). Early Life Stress, Neuroinflammation, and Psychiatric Illness of Adulthood. In: Kim, YK. (eds) Neuroinflammation, Gut-Brain Axis and Immunity in Neuropsychiatric Disorders. Advances in Experimental Medicine and Biology, vol 1411. Springer, Singapore. https://doi.org/10.1007/978-981-19-7376-5_6
Download citation
DOI: https://doi.org/10.1007/978-981-19-7376-5_6
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-7375-8
Online ISBN: 978-981-19-7376-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)