Abstract
Antinutrients are naturally occurring substances in plants, well known to block the absorption of beneficial or essential organic nutrients and inorganic minerals. Major antinutrients such as enzymes (lipase, amylase, and protease), trypsin inhibitors, phytate, polyphenols, lectins, glucosinolates, oxalates, and saponins have been characterized in different plant species. In general, these antinutrients are reported to have bitter taste and are unpalatable with bad odor and have a role in plant defenses. Antinutrients, which are known to bind the nutrients, severely affect the latter’s bio-accessibility and ultimately bioavailability. The current chapter briefly summarizes the various kinds of antinutrients and their role in limiting bioavailability of nutrients. Eventually, these antinutrients cause mineral deficiency and micronutrient malnutrition in humans. Hence, it is indispensable to reduce their contents in foods to a safe level for human consumption. Further, various strategies and concepts to tackle the deleterious effects of antinutrients are discussed. Many processing methods namely decortication, roasting, boiling, microwave heating, soaking, germination, autoclaving, fermentation, and extrusion technique are reported to decrease the antinutrient content in food crops. Further, the current understanding of the antinutrient’s health promoting effects such as reduction of blood glucose, cholesterol levels, prevention of cardiovascular diseases and cancer are also discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
8.1 Introduction
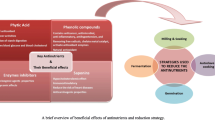
A World Health Organization (WHO) report reveals that more than two billion people are encountering the problem of micronutrient deficiency due to improper nutrition leading to mass malnutrition. This condition is more specifically encountered in developing countries. Vitamin A, zinc, and iron are the most essential micronutrients whose deficiencies cause serious health concerns (Gupta et al. 2015). Though nutrients and antinutrients both are naturally present in plants, former has positive effects and latter pose deleterious effect on human body (Popova and Mihaylova 2019). In order to improve the proper mineral intake, several countries have endorsed dietary intake guidelines toward consumption of the whole grains, as these plant foods are exceptional sources of almost all the essential nutrients. However, the presence of several antinutritional factors (ANF) (for example phytic acids) could severely hinder the immense nutritional benefits of plant-based foods (Samtiya et al. 2021). Antinutrients are phytochemicals acquired or developed during the process of plant evolution for their own defense with limited biological functions. Upon consumption, antinutrients reduce the optimal utilization of food nutrients especially proteins, vitamins, and minerals thereby decreasing the overall nutritional value of foods (Ijeoma and Adeyemi 2020). Various antinutrients present in plant foods are depicted in Fig. 8.1. ANFs inhibit the proper utilization of nutrients by forming complexes with these compounds which eventually reduce their absorption in the gastrointestinal (GI) tract (Nikmaram et al. 2017). ANFs are found in appreciable concentration in legumes, grains, beans, and nuts. However, they are also known to occur in fruits, leaves, and roots of certain types of plants. The anti-nutrients in plant foods are enzyme inhibitors, phytates, tannins, oxalates, saponins, lectins, etc. (Popova and Mihaylova 2019). ANFs considerably affect the nutritional values of foods; hence, reduction or elimination of these factors from plant foods is now a major goal to improve human nutrition. There are several processing methods such as dehulling, milling, soaking, germination, and fermentation that can be employed to reduce these ANFs (Samtiya et al. 2020). This chapter aims to present updated information about antinutrients, their positive and negative effects, and existing strategies to reduce ANFs. Figure 8.2 depicts a brief overview of possible health beneficial properties of antinutrients.
8.2 Types of Antinutritional Factors
8.2.1 Lipase Inhibitors
On the basis of chemical structures, plant lipase inhibitors can be grouped into the following categories: phenols, saponins, alkaloids, terpenes, glycosides, carotenoids, and polysaccharides (Singh et al. 2015). Lipase inhibitors reduce the biochemical functioning of enzyme lipases, which hydrolyze the triglyceride to monoglycerides, glycerol esters, fatty acids, and glycerol. Lipase inhibitors primarily affect fat metabolism, which inhibits lipid hydrolysis by macromolecules, reducing the absorption of small fat molecules and fat accumulation ultimately causing deficiency of fat-soluble vitamins (Liu et al. 2020; Yun 2010).
8.2.2 Amylase Inhibitors
Amylase inhibitors are the phytocompounds which bind to enzyme α-amylase and make them inactive. There are two main biological roles of α-amylase inhibitors in plants: protect the seeds against pests and microorganisms and inhibit mammalian α-amylases, insect α-amylases, and endogenous α-amylases (Popova and Mihaylova 2019; Wisessing and Choowongkomon 2012). Mammalian amylase enzyme is secreted by the pancreas into the small intestine, whereas α-glucosidase enzymes are located in the brush border of the small intestines. Further, amylases convert the carbohydrates into oligosaccharides (Barrett and Udani 2011), and α-glucosidase acts on starch to convert them into glucose molecules (Brás et al. 2018). Amylase inhibitors impair the activity of salivary and pancreatic amylase and affect the growth and metabolism of animals at appreciably high levels are ingested (Grant et al. 2003). However, their ability to inhibit the action of α-amylase enzymes resulting in reduction of starch hydrolysis shows positive effects on glycemic index control in diabetics (Bhat et al. 2011). Phaseolus species (kidney beans) have relatively high content of amylase inhibitors of about 4.3 g per kg (Grant et al. 2003).
8.2.3 Protease Inhibitors
Proteases are important class of enzymes found in all cells and tissues that regulate multiple cellular processes (Popova and Mihaylova 2019). Protease inhibitors are relatively small proteins commonly found in raw cereals, legumes, specifically soybeans, tubers, and also in aerial parts of some plants. The metabolic functioning of protease, i.e., breaking down of hydrolytic cleavage of peptide bonds in proteins is restricted by these inhibitors (Popova and Mihaylova 2019; Habib and Fazili 2007). Protease inhibition occurs due to the formation of an enzyme–inhibitory molecule complex thereby restricting the availability of enzyme to perform substrate hydrolysis (Paiva et al. 2013). This comprehension has led to the development of drugs; for example, protease inhibitors play an effective role in treating AIDS in the strategy of HAART, i.e., highly active antiretroviral therapy (Wang et al. 2015).
8.2.4 Trypsin Inhibitors
These are found in a variety of foods such as potato, eggplant, millet, barley, wheat, mung beans, soybeans, red kidney beans, chickpeas, and adzuki beans belonging to diverse families Solanaceae, Gramineae, and Leguminosae. It inhibits the activity of digestive enzymes (trypsin and chymotrypsin) by forming complexes and prevents the absorption of nutrients and digestion of protein (Tibe and Amarteifio 2010; Vanga et al. 2020). For example, their presence in human food causes improper growth and impairs protein digestion, pancreatic hyperplasia and disturbance of sulfur and amino acid utilization (Nikmaram et al. 2017). Peas, lentils, and soybean contain Bowman–Birk-like trypsin inhibitors which have a low molecular weight, i.e., 8–10 kDa and are double-headed to inhibit two enzyme molecules at a time (Grant et al. 2003).
8.2.5 Phytates
Phytates are the salts of phytic acid, chemically known as myo-inositol hexaphosphate and its general molecular formula is C6H18O24P6 (Diouf et al. 2019). Phytate chelates micronutrients (calcium, zinc, iron, copper, and magnesium) and makes them less bioavailable to monogastric animals, including poultry and humans, due to absence or low activity of phytase enzyme in their digestive tract (Gupta et al. 2015). Based on the type of cation, the minerals may bind with one or more phosphate groups of a phytate or may bridge with two or more phytate molecules (Nikmaram et al. 2017). Phytates are also known as demineralizing agents because they form insoluble salts with divalent and trivalent cationic metal ions. Phytates also interact with protein residues to form phytate–protein and phytate–mineral–protein complexes which inhibit many reactions of digestive enzymatic (Betancur-Ancona et al. 2012). Plants synthesize phytate to store phosphorus and phytate may represent approx. 50–85% of total phosphorous in some plants. It occupies 1–5% weight in legumes, cereals, nuts, and oil seeds (Vats and Banerjee 2004; Reddy et al. 1982). Decortication, soaking, germination, and fermentation are some of the traditional methods followed to reduce the antinutrients (Eltayeb et al. 2007).
8.2.6 Polyphenols
Polyphenols are a group of secondary metabolites which abundantly occur in vegetables, fruits, herbs, spices, tea, wine, chocolate, other cocoa products, and extra virgin olive oil. Phenolics are classified into simple phenolics which include phenolic acids, flavones, isoflavones, etc., and complex phenolics which include catechins, tannins, lignans, etc. Dietary polyphenols show many beneficial effects, such as protection against oxidative stress, and degenerative diseases like diabetes, cancer, cardiovascular diseases (Han et al. 2007). Different classes of phenolic compounds include tannins, flavonoids, phenolic acids, lignans, and stilbenes (Shahidi et al. 2019). Tannins are the polyphenols known to form reversible or irreversible complexes with proteins, reducing latter’s digestibility and amino acid bioavailability (Lampart-Szczapa et al. 2003). Hydrolysable tannins mainly consist of phenolic acids or their derivatives attached to central unit of pentagalloylglucose with ester linkage. Gallotannins and ellagitanins are the two main classes of hydrolysable tannins, which consist of gallic acid and ellagic acid, respectively (Singh and Kumar 2019; Diouf et al. 2019). Condensed tannins are produced by the condensation of two or more flavanols units and have structure similar to flavonoids and they do not constitute sugar molecules (Hussain et al. 2019; Diouf et al. 2019). Dietary polyphenols show beneficial effects in reducing cancer, CVDs, diabetes, neurodegenerative diseases, and osteoporosis (Mattila et al. 2018; Vauzour et al. 2010). Polyphenolics are attributed with regulation of lipoprotein plasma concentrations and insulin resistance in a clinical trial involving men who have high risk of developing CVD (Chiva-Blanch et al. 2013).
8.2.7 Lectins
Lectins, known as phytohemagglutinins, are a glycoprotein compound that binds simple carbohydrates especially oligosaccharides and also agglutinates red blood cells (RBCs) in vitro (Klupšaitė and Juodeikienė 2015; Santos et al. 2014). Alternatively, lectins are known as carbohydrate-binding proteins found in plant foods like cereal grains and legumes (Van Buul and Brouns 2014). Lectins are synthesized at the time of seed development and are utilized by the plants as an amino acid source during germination as well as act as defensive compound against insects, fungi, and nematodes attack (Betancur-Ancona et al. 2012). Lectins are highly resistant to in vivo proteolytic degradation and survive while passing through the GI tract. Only if appropriate carbohydrate receptors present on epithelial cells of intestine, lectins can bind to them and are absorbed systematically. Thus, lectins can alter and modify many aspects of intestinal and systemic metabolism. The quantum of lectins in lentil and peas are comparable to that of soybean (2–4 g/kg) relatively low than what is reported in kidney bean (15–30 g/kg) (Grant et al. 2003).
8.2.8 Glucosinolates
Chemically, glucosinolates are S-β-thioglucoside N-hydroxysulfates which are found abundantly in cruciferous (Brassicaceae) plants but are also reported in other species like moringa, papaya, and capers. More than 120 different types of glucosinolates have been found in plants (Dinkova-Kostova 2012). Glucosinolates are water-soluble, nitrogen-containing compounds, having a glucopyranose moiety bound with sulfur in its molecular structure (Lopez-Rodriguez et al. 2020). Glucosinolates are the major secondary metabolites produced by plants, having crucial role in the physiology and defense of plants against various environmental stresses. Glucosinolates are enzymatically broken down by myrosinase (a thioglucosidase) whenever a plant tissue is damaged, to form glucose, sulfate, nitriles, indolic alcohols, isothiocyanates, thiocyanate, oxazolidinethions, amines, and epithionitriles. Glucosinolates along with their catabolized products play a vital role in plant defense (Miękus et al. 2020).
8.2.9 Oxalates
Oxalic acid forms potassium, sodium (soluble) or iron, calcium, magnesium (insoluble) salts, or esters called oxalates (Popova and Mihaylova 2019). By forming esters with these minerals, oxalates make them less available (Savage and Klunklin 2018). Compared to other ANFs, oxalates are found in less quantum in plants such as wheat, rye, millet, and barley (Siener et al. 2006). Foods rich in oxalate usually form a minor component of the human diet, but in some regions of the world, oxalates form an important ingredient of the seasonal diet. Calcium oxalate is responsible for the formation of kidney stones. People suffering from enteric and primary hyperoxaluria are advised to decrease the intake of oxalates because in the former more oxalates are absorbed and excreted via urine resulting in increased risk of kidney stone formation. In the latter condition, either liver does not produce adequate amount of enzymes to prevent the overproduction of oxalate or enzymes do not function properly (Popova and Mihaylova 2019; Bhasin et al. 2015).
8.2.10 Saponins
Saponins are a diverse group of chemicals and are called so because of their ability to form soap-like foams in aqueous solutions (Hill 2003). The structure of saponins contains a steroidal or triterpenoid aglycone, and one or more oligosaccharide moieties (Yildirım and Kutlu 2015). Saponins are found in broad beans, kidney beans, and lentils imparting bitter taste to food if present in high concentration hence low saponins foods are preferred (Shi et al. 2004). Saponins exhibit both the hypoglycemic and hypocholesteremic effects (Ikewuchi 2012; Barky et al. 2017).
8.2.11 Exorphins
The prolamins, i.e., alcohol-soluble proteins found in dairy products and cereal grains which are known as gliadins, can be degraded into opioid-like polypeptides in GI tract which are called as exorphins (Tatham and Shewry 2008; Pruimboom and de Punder 2015). Recent studies indicated that the epigenetic effects of opiate peptides derived from milk protein alpha-casein may contribute to GI dysfunction and inflammation in susceptible people (Trivedi et al. 2015). The bioactivity of food-derived exorphins can affect behavioral characteristics i.e. memory, spontaneity, and pain perception (Lister et al. 2015). On the basis of origin, opioid peptides are of two types: endogenous and exogenous. Endogenous peptides are synthesized by the organism which may function as hormones and neuromodulators (Garg et al. 2016) whereas exogenous peptides are formed when plant or animal precursor proteins are hydrolyzed in the digestive tract (Janecka et al. 2004; Nyberg and Hallberg 2013).
8.2.12 Contextual Antinutrients
Some food or food additives can also act as antinutrients depending on the context. For example, foods rich in calcium can stop the absorption of iron. Also a mutual antagonism exist between copper and zinc during the absorption process which occurs in the intestinal epithelium (Van Campen 1969). Scientific literature states that phospholipids and phytosterols can hinder cholesterol absorption when added to low-fat or non-fat foods (Ostlund 2002; Cohn et al. 2010). It has been also noticed that some foods can interfere with the absorption of medications (Genser 2008). Food–drug interaction is also very common. For example, if a drug is taken along with grapefruit, the latter inactivates the enzyme of drug metabolism as it contains bergamottin-a natural furanocoumarin. Studies have also demonstrated that the polyphenol compound resveratrol present in peanuts and red wine inhibits platelet aggregation, thus when taken with anticoagulant drugs, it can increase the risk of bleeding (Jin and Han 2010). Similarly, foods that contain tyramine (beer, wine, chocolate, and avocados) are inhibitors of monoamine oxidase inhibitors (MAOIs). Hence, the activity of MAOI drugs such as anti-depressant drugs is affected when consumed with foods rich in tyramine (Vaquero et al. 2010). Likewise, foods rich in coumadin and vitamin K such as broccoli and spinach exhibit antagonism with anticoagulant drugs.
8.3 Health Beneficial Properties of Antinutritional Factors
Various health and beneficial properties of antinutritional factors derived from plant-based food are presented in Table 8.1. The health and beneficial effects of ANFs are known especially in the treatment of obesity, diabetes, antiviral treatment, neuroprotective effects, reduction in blood cholesterol levels, and inhibition of cancer.
8.4 Adverse Effects of Antinutritional Factors Relevance to Health
As stated below, the adverse effects of ANFs include reduced intestinal absorption of diet-derived nutrients, impaired digestion and growth, mal-absorption of mineral nutrients among others (Table 8.2).
8.5 Strategies for Reduction of Antinutrients in Plant Foods
Multiple studies have proven that antinutrients have negative effects on nutritional quality of food which eventually affect health. Several strategies are available to reduce antinutritional factors to great extent so as to improve food quality. Elimination of the antinutrients would be essential for effective utilization of nutrients like carbohydrates, proteins, and minerals in human nutrition (Ohiokpehai 2003; Tibe and Amarteifio 2010). Many traditional and modern techniques which include decortications, heating, soaking, germination, fermentation, and extrusion are available to reduce the concentration of antinutritional factors in food crops.
8.5.1 Decortication
Decortication or dehulling involves the removal of outer covering (pericarp) of grains. In the pre-mechanization era, dehulling was done using mortar and pestle but currently automatic rice huller/rice milling machines are available for efficient processing (Kulkarni et al. 2018). Decortication significantly reduces many antinutrient components present in bran and also lowers the concentration of condensed tannins (Lestienne et al. 2007; Pal et al. 2015). Pal et al. (2016) observed that around 52–56% reduction of phytic acid content in lentils is achieved by following the decortication process. In different varieties of lentils, the combination of decortication and germination techniques have shown appreciable reduction in antinutritional factors including tannins, phytate, trypsin inhibitors, total polyphenols, etc. (Pal et al. 2015). Ghavidel and Prakash (2007) found a reduction of 43–52% and 47–52% in tannin and phytic acid contents, respectively, in various legume seeds such as green gram, cowpea, lentil, and chickpea. Himanshu et al. (2018) reported that this method decreases phytic phosphorus content by 39% in little millet, 23% in barnyard millet, 25% in kodo millet, and 12% in common millet. The activity of antinutritional factors i.e. trypsin inhibitor, phytic acid, and tannins was reported to be reduced by 7.59%, 20.7%, and 33.3%, respectively, after dehulling of mung bean seed (Oghbaei and Prakash 2016).
8.5.2 Milling
Milling is a processing method which removes the outer layer, i.e., pericarp of the grain (Kulkarni et al. 2018). It is commonly employed to reduce phytic acid levels from grains but it also has some demerits as it removes dietary fibers and minerals from grains (Gupta et al. 2015). The process of milling can be of two types: (1) The whole grain is ground into flour without separating any parts or, (2) Differential milling to separate the grain into different parts (Oghbaei and Prakash 2016). Former results into grain flour whereas latter provides many products such as germ, bran, semolina, and refined wheat flour. Milled wheat flour is used to make chapattis which on heating decrease the phytic acid and polyphenol content and improve protein and starch digestion (Chowdhury and Punia 1997).
8.5.3 Heating
Heat from different sources is applied as processing method in roasting, cooking, boiling, and microwave heating. Adeyemo and Onilude (2013) found appreciable reduction in tannin (74.6%), phytate (28.4%), trypsin inhibitor (98.3%), and protease inhibitor (97.5%) after roasting soybean in comparison with cooking which eliminated tannin (42%), phytate (75.8%), trypsin inhibitor (95.8%), and protease inhibitor (95.8%). Microwave processing degrades the bioavailability of glucosinolates and significantly reduces the polyphenolic content in millets (Miękus et al. 2020; Hithamani and Srinivasan 2014). Vanga et al. (2020) reported that microwave heating can eliminate 70% of trypsin inhibitors in soy milk. Barba et al. (2016) reported that boiling decreases glucosinolates level more significantly than steaming. Ertop and Bektas (2018) stated that pressure cooking of black gram eliminates tannins and increases its protein digestibility. A comparative analysis of the effect of boiling and baking of taro leaves was conducted by Savage and Mårtensson (2010) which showed that boiling taro leaves for 40 min reduced 47% oxalate content whereas no such significant reduction of oxalate was obtained after baking. Many studies have demonstrated that autoclaving is best suited method to reduce levels of various antinutritional factors like tannin, phytic acid, and free phenolics compared to other processing treatments (Samtiya et al. 2020; Vijayakumari et al. 1996). The findings of Kadam et al. (1987) concluded that the methods like boiling and autoclaving improved the protein quality of winged beans by eliminating antinutrients.
8.5.4 Soaking
Soaking is a widely used process which generally precedes other treatments like germination, cooking, and fermentation. It is one of the easiest ways to reduce antinutrients (Kumari et al. 2014). Soaking is effective to eliminate tannins, phytic acid, total phenols, and trypsin inhibitor activity of many cereals, legumes, and millets (Oghbaei and Prakash 2016). It is also effective in reducing saponins on domestic as well as on industrial levels (Shi et al. 2004). Soaking for 12–18 h was very effective for the reduction of phytic acid and enzyme inhibitors in legumes (Handa et al. 2017). Phytate being water-soluble compound can be easily eliminated by soaking and discarding the soaked water (Greiner and Konietzy 2006). Soaking at room temperature for 24 h eliminated 16–21% of phytic acid levels in sorghum flour (Mahgoub and Elhag 1998). The combination of dehulling and soaking reduced 25% and 57% amylase inhibitors in Lathyrus sativa and Lathyrus beans, respectively (Srivastava 1994). Rehman and Shah (2001) found that soaking of black gram at 30 °C removed 18.45% of the tannins in 3 h; whereas 22.14% of the tannins were removed on soaking at 100 °C for 45 min.
8.5.5 Germination
Germination is suitable for minimizing the antinutrient components present in plant-based foods (Nkhata et al. 2018). During the process of germination, an increase in phytate-degrading enzyme activity was observed by Greiner and Konietzy (2006). About 40% of phytate content can be reduced by employing this technique (Masud et al. 2007) and 40.00–59.38% reduction in phytic acid contents was observed in lentils (Pal et al. 2016). Trypsin inhibitor content was eliminated to the range of 19.2–88.2% in different types of beans by germination (Avilés-Gaxiola et al. 2017). Gupta et al. (2015) observed that longer duration of germination has caused greater elimination of antinutrients. For instance, mungbean and kidney bean following germination revealed continuous decrease of phytic acid for 6 days (Rasha Mohamed et al. 2011).
8.5.6 Fermentation
Fermentation is a process where microorganisms effect some desirable biochemical changes in food material with the aid of their enzymes (Admassie 2018). It increases protein digestibility, reduces antinutritional factors, and enhances texture (Adewumi and Odunfa 2009). During the process of fermentation, metabolic activities of microorganisms modify the concentration of bioactive compounds present in the food crops. The cell wall of cereal grain ruptures as a result of fermentation which leads to production of several bioactive compounds. Some enzymes like amylases, proteases, and xylanases derived from cereals along with microorganisms modify the grain nutritional content (Đorđević et al. 2010). This process is really helpful in reduction of phytic acid and trypsin inhibitor activity in legumes (Oghbaei and Prakash 2016). Coulibaly et al. (2011) reported that 12- and 24-h fermentation of millet grains eliminates significant amount of tannins, phytates, and protease inhibitors. Villacrés et al. (2020) concluded that fermentation utilizing Rhizopus oligosporus significantly reduces the content of antinutrients such as phytic acid (70.06%), tannins (82.10%), and trypsin inhibitors (76.76%). Mohapatra et al. (2019) observed that fermentation eliminated 30% tannin from grain sorghum whereas Dlamini et al. (2007) reported even more (49–68%) reduction. Fermentation reduces antinutrients like oxalates and phytate content to a very large extent (Ijeoma and Adeyemi 2020). Mohapatra et al. (2019) concluded that during fermentation of whole-grain sorghum, the total phenolic content decreases by 28% and tannin content decreases by 30–39%.
8.5.7 Extrusion Cooking Technique
It is a food processing technique that combines different steps including mixing, kneading, cooking, shearing, shaping, and forming (Bordoloi and Ganguly 2014). It is one of the most widely used technique and modern food unit operation that converts plant-based foods, usually in a granular or powdered form, into partially cooked, low moisture, and highly shelf-stable food products. It results in enhanced functional properties of food (Ciudad-Mulero et al. 2020). Extrusion cooking technique is a high-temperature short-time (HTST) process that inactivates inherent enzymes and mitigates microbial contamination (Bordoloi and Ganguly 2014). This processing treatment shows significant reduction in antinutritional factors; for example, about 30% phytates are eliminated through extrusion (Chai and Liebman 2005; Harland et al. 2004). This technique completely inactivates lectin and trypsin inhibitors in peas and lentils (Grant et al. 2003).
8.5.8 Biotechnological Interventions
Several biotechnological approaches have been used to down-regulate or even eliminate the genes engaged in the metabolic pathways for reducing the production, aggregation, and/or inactivation of antinutrients. Low phytate rice and soybean with about 40–50% reduction in phytate without detrimental effects on their nutrient quality have been developed (Kumar et al. 2019; Punjabi et al. 2018; Larson et al. 2000). Single nucleotide substitution in rice Khira caused mutation in SPDT gene coding sequence which inhibited the accumulation of phytic acid in plant (Kumar et al. 2021). lpa mutants of rice have 45–95% less phytic acid content than wild-type seeds (Perera et al. 2018). To reduce phytic acid levels in rapeseed, ITPK gene encoding an enzyme that stimulates the last step of phytate synthesis was blocked using CRISPR/Cas9 (Liu et al. 2021). Shukla et al. (2009) utilized zinc-finger nucleases to mutate the IPK1, one of the phytic acid biosynthesis gene in Zea mays. Further, work has also been performed to reduce cyanogenic glycosides in cassava through expression of hydroxynitrile lyase enzyme in cassava roots (Siritunga et al. 2004). Likewise, the solanine content of potato has been reduced significantly using an antisense RNA approach, and efforts are ongoing to reduce the level of the other major potato glycoalkaloid and chaconine (McCue et al. 2003). In oilseed rape, Shashidhar et al. (2020) reported 35% reduction phytic acid by knocking out three essential BnITPK genes with an enhanced amount of phosphorus.
8.6 Current Trends in Antinutritional Factors
In the current scenario, our comprehension of dietary requirements of human body and associated complications has improved to great extent. Also, awareness regarding ANF in pulses and cereals has grown. Even though these ANFs cannot be detoxified by the human body, it can be diminished to an extent by adopting various processing methods. On the other hand, phenolic compounds especially flavonoids are added into beverages which make them more valuable to prevent early-stage diseases (i.e., hypertension and diabetes). It has been found that tannin can be used as therapeutic agent due to its laxative effect which helps in treating constipation (Youn Hwang 2019). In recent researches, tannins are introduced into cellulose nanofibrils to make a packaging material which has been proved to be sustainable, eco-friendly, non-toxic packaging material for pharmaceuticals as well as food. In addition, tannins showed antibacterial and antioxidant properties and are used as preservatives in food industries (Singh and Kumar 2019). Total pomegranate tannin (TPT) extract from Punica granatum L. when evaluated for anti-proliferative activity in vitro on human oral (KB, CAL27), prostate (RWPE-1, 22Rv1), and colon (HT-29, HCT116, SW480, SW620) tumor cells, and apoptotic effects suggest that it induces apoptosis and decreases the viable cell number of human oral, prostate, and colon tumor cells (Yildirım and Kutlu 2015). Flavonoids are associated with large number of health-promoting effects and are essential components in pharmaceutical, medicinal, nutraceutical, and cosmetic applications. Flavonoids have been shown to possess anti-cholinesterase activity which reduces the level of acetylcholinesterase (AChE) enzyme in the CNS and inhibition of it results in elevated levels of neural acetylcholine levels which is one of the therapeutic strategies to combat Alzheimer’s disease (Panche et al. 2016). Glucosinolates present in moringa leaves are extracted and used for decreasing the risk of developing non-communicable diseases (Lopez-Rodriguez et al. 2020). Also, glucosinolates have been experimented in the prevention of fungal and bacterial spoilage of food products by using advanced atmospheric packaging technology (MAP) that improves the shelf-life of these products (Melrose 2019).
8.7 Conclusions
Plant contains both nutrients and antinutrients. Antinutrients are a major area of concern for those whose diet predominantly comprises unprocessed plant-based foods. Plant foods especially cereals and grains can be processed to diversify their uses, and to enhance their nutritional value and consumer acceptability. Antinutritional factors can be easily removed by using traditional and advanced processing techniques. Traditional methods include soaking, boiling, germination, and fermentation whereas modern technique includes autoclaving, microwave heating, and extrusion technique. These techniques have been proved to reduce a great amount of antinutrients like phytates, oxalates, saponins, enzyme inhibitors, etc. Some antinutrients bind to different minerals and make them less bioavailable, some inhibit digestive enzymes, and some can cause health problems. Exorphins and contextual antinutrients are also present in foods. The former category of ANF is present in milk and cereals whereas the latter category interacts with other food and drugs resulting in its poor absorption in the digestive system which results in poor functioning of medicines. Therefore, they have to be eliminated from plant-based foods before consumption. Biotechnological interventions in plants by suppressing the gene responsible for the antinutrient production form an integral component of alternative strategies to reduce ANFs.
References
Adewumi GA, Odunfa SA (2009) Effect of lactic fermentation on anti-nutritional factors in Nigerian Local Beans (Vigna unguiculata) varieties. Nigerian Food J 27(2):73–77
Adeyemo SM, Onilude AA (2013) Enzymatic reduction of anti-nutritional factors in fermenting soybeans by Lactobacillus plantarum isolates from fermenting cereals. Nigerian Food J 31(2):84–90
Adhikari B, Marasini BP, Rayamajhee B, Bhattarai BR, Lamichhane G, Khadayat K, Adhikari A, Khanal S, Parajuli N (2020) Potential roles of medicinal plants for the treatment of viral diseases focusing on COVID-19: a review. Phytother Res. https://doi.org/10.1002/ptr.6893
Admassie M (2018) A review on food fermentation and the biotechnology of lactic acid bacteria. World J Food Sci Technol 2(1):19. https://doi.org/10.11648/j.wjfst.20180201.13
Angeloni C, Hrelia S, Malaguti M (2017) Neuroprotective effects of glucosinolates. In: Mérillon JM, Ramawat K (eds) Glucosinolates. Reference series in phytochemistry. Springer, Cham. https://doi.org/10.1007/978-3-319-25462-3_20
Avilés-Gaxiola S, Chuck-Hernández C, Serna Saldívar SO (2017) Inactivation methods of trypsin inhibitor in legumes: a review. J Food Sci 83(1):17–29. https://doi.org/10.1111/1750-3841.13985
Barba FJ, Nikmaram N, Roohinejad S, Khelfa A, Zhu Z, Koubaa M (2016) Bioavailability of glucosinolates and their breakdown products: impact of processing. Front Nutr 3. https://doi.org/10.3389/fnut.2016.00024
Barky AR, Hussein SA, Alm-Eldeen A, Hafiz YA, Mohamed TM (2017) Saponins and their potential role in diabetes mellitus. Diab Manage 7(1):148–158
Barrett ML, Udani JK (2011) A proprietary alpha-amylase inhibitor from white bean (Phaseolus vulgaris): a review of clinical studies on weight loss and glycemic control. Nutr J 10(1). https://doi.org/10.1186/1475-2891-10-24
Betancur-Ancona D, Segura-Campos M, Rosado-Rubio JG, Franco LS, Chel-Guerrero L (2012) Chemical composition and anti-nutritional factors in five tropical legume seeds. Nutr Consum Health, Beans:117–141
Bhasin B, Ureki HM, Atta MG (2015) Primary secondary hyperoxaluria: understanding the enigma. World J Nephrol 4(2):235–244. https://doi.org/10.5527/wjn.v4.i2.235
Bhat M, Zinjarde SS, Bhargava SY, Kumar AR, Joshi BN (2011) Antidiabetic Indian plants: a good source of potent amylase inhibitors. Evid Based Complement Alternat Med 1–6. https://doi.org/10.1093/ecam/nen040
Bischoff KL (2016) Glucosinolates. Forum Nutr 551–554. https://doi.org/10.1016/b978-0-12-802147-7.00040-1
Bordoloi R, Ganguly S (2014) Extrusion technique in food processing and a review on its various technological parameters. Indian J Scientific Res Technol 2(1):1–3
Brandl T, Simic O, Skaanderup PR, Namoto K, Berst F, Ehrhardt C, Schiering N, Mueller I, Woelcke J (2016) Trypsin inhibitors for the treatment of pancreatitis. Bioorg Med Chem Lett 26(17):4340–4344. https://doi.org/10.1016/j.bmcl.2016.07.029
Brás NF, Santos-Martins D, Fernandes PA, Ramos MJ (2018) Mechanistic pathway on human α-glucosidase maltase-glucoamylase unveiled by QM/MM calculations. J Phys Chem B 122(14):3889–3899. https://doi.org/10.1021/acs.jpcb.8b01321
Chai W, Liebman M (2005) Oxalate content of legumes, nuts, and grain-based flours. J Food Compos Anal 18(7):723–729. https://doi.org/10.1016/j.jfca.2004.07.001
Chiva-Blanch G, Urpi-Sarda M, Ros E, Valderas-Martinez P, Casas R, Arranz S, Guillén M, Lamuela-Raventós RM, Llorach R, Andres-Lacueva C, Estruch R (2013) Effects of red wine polyphenols and alcohol on glucose metabolism and the lipid profile: a randomized clinical trial. Clin Nutr 32(2):200–206
Chowdhury S, Punia D (1997) Nutrient and anti-nutrient composition of pearl millet grains as affected by milling and baking. Food Nahrung 41(2):105–107
Ciudad-Mulero M, Fernández-Ruiz V, Cuadrado C, Arribas C, Pedrosa MM, Berrios JD, Pan J, Morales P (2020) Novel gluten-free formulations from lentil flours and nutritional yeast: evaluation of extrusion effect on phytochemicals and non-nutritional factors. Food Chem 315:126175. https://doi.org/10.1016/j.foodchem.2020.126175
Cohn JS, Kamili A, Wat E, Chung RW, Tandy S (2010) Dietary phospholipids and intestinal cholesterol absorption. Nutrients 2(2):116–127. https://doi.org/10.3390/nu2020116
Coulibaly A, Kouakou B, Chen J (2011) Phytic acid in cereal grains: structure, healthy or harmful ways to reduce phytic acid in cereal grains and their effects on nutritional quality. Am J Plant Nutr Fertil Technol 1(1):1–22. https://doi.org/10.3923/ajpnft.2011.1.22
De Mejía EG, Prisecaru VI (2005) Lectins as bioactive plant proteins: a potential in cancer treatment. Crit Rev Food Sci Nutr 45(6):425–445. https://doi.org/10.1080/10408390591034445
Dinkova-Kostova AT (2012) Chemoprotection against cancer by isothiocyanates: a focus on the animal models and the protective mechanisms. Top Curr Chem 329:179–201. https://doi.org/10.1007/128_2012_337
Diouf A, Sarr F, Sene B, Ndiaye C, Fall SM, Ayessou NC (2019) Pathways for reducing anti-nutritional factors: prospects for Vigna unguiculata. J Nutr Health Food Sci 7(2):1–10. https://doi.org/10.15226/jnhfs.2019.001157
Dlamini NR, Taylor JR, Rooney LW (2007) The effect of sorghum type and processing on the antioxidant properties of African sorghum-based foods. Food Chem 105(4):1412–1419. https://doi.org/10.1016/j.foodchem.2007.05.017
Đorđević TM, Šiler-Marinković SS, Dimitrijević-Branković SI (2010) Effect of fermentation on antioxidant properties of some cereals and pseudo cereals. Food Chem 119(3):957–963. https://doi.org/10.1016/j.foodchem.2009.07.049
Eltayeb MM, Babiker HA, Sulieman MA, Babiker EE (2007) Effect of processing followed by fermentation on antinutritional factors content of pearl millet (Pennisetum glaucum L.) cultivars. Pak J Nutr 6(5):463–467
Ertop MH, Bektas M (2018) Enhancement of bioavailable micronutrients and reduction of anti-nutrients in foods with some processes. Food Health 4(3):159–165. https://doi.org/10.3153/FH18016
Garg S, Nurgali K, Mishra VK (2016) Food proteins as source of opioid peptides: a review. Curr Med Chem 23(9):893–910
Gemede HF, Ratta N (2014) Antinutritional factors in plant foods: potential health benefits and adverse effects. Int J Nutr Food Sci 3(4):284–289. https://doi.org/10.11648/j.ijnfs.20140304.18
Genser D (2008) Food and drug interaction: consequences for the nutrition/health status. Ann Nutr Metab 52(Suppl. 1):29–32. https://doi.org/10.1159/000115345
Ghavidel RA, Prakash J (2007) The impact of germination and dehulling on nutrients, antinutrients, in vitro iron and calcium bioavailability and in vitro starch and protein digestibility of some legume seeds. LWT- Food Sci Technol 40(7):1292–1299. https://doi.org/10.1016/j.lwt.2006.08.002
Grant G, Duncan M, Alonso R, Marzo F (2003) Peas and lentils. Encycl Food Sci Nutr:4433–4440. https://doi.org/10.1016/b0-12-227055-x/00899-3
Greiner R, Konietzny U (2006) Phytase for food application. Food Technol Biotechnol 44(2):125–140
Gupta RK, Gangoliya SS, Singh NK (2015) Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. J Food Sci Technol 52(2):676–684. https://doi.org/10.1007/s13197-013-0978-y
Habib H, Fazili KM (2007) Plant protease inhibitors: a defense strategy in plants. Biotechnol Mol Biol Rev 2(3):068–085
Han X, Shen T, Lou H (2007) Dietary polyphenols and their biological significance. Int J Mol Sci 8(9):950–988. https://doi.org/10.3390/i8090950
Handa V, Kumar V, Panghal A, Suri S, Kaur J (2017) Effect of soaking and germination on physicochemical and functional attributes of horsegram flour. J Food Sci Technol 54(13):4229–4239. https://doi.org/10.1007/s13197-017-2892-1
Harland BF, Smikle-Williams S, Oberleas D (2004) High performance liquid chromatography analysis of phytate (IP6) in selected foods. J Food Compos Anal 17(2):227–233. https://doi.org/10.1016/j.jfca.2003.08.005
Hassan S, Faiza N, Farooq MA, Egbuna C, Habib-ur-Rehman, Olatunde A (2020) Role of nutraceuticals in maternal nutrition. In: Functional foods and nutraceuticals. Springer, Cham, pp 527–541. https://doi.org/10.1007/978-3-030-42319-3_24
Hill GD (2003) Plant antinutritional factors | characteristics. In: Encyclopedia of food sciences and nutrition. Elsevier, pp 4578–4587. https://doi.org/10.1016/b0-12-227055-x/01318-3
Himanshu CM, Sonawane SK, Arya SS (2018) Nutritional and nutraceutical properties of millets: a review. Clin J Nutr Diet 1(1):1–10
Hithamani G, Srinivasan K (2014) Effect of domestic processing on the polyphenol content and bioaccessibility in finger millet (Eleusine coracana) and pearl millet (Pennisetum glaucum). Food Chem 164:55–62. https://doi.org/10.1016/j.foodchem.2014.04.107
Hussain G, Huang J, Rasul A, Anwar H, Imran A, Maqbool J, Razzaq A, Aziz N, Konuk M, Sun T (2019) Putative roles of plant-derived tannins in neurodegenerative and neuropsychiatry disorders: an updated review. Molecules 24(12):2213. https://doi.org/10.3390/molecules24122213
Ijeoma O, Adeyemi OO (2020) Effect of fermentation on some minerals and antinutrient content of Telfaira Occidentalis and Gnetum Africanum leaves. Int J Adv Sci Res Eng 6(2):6. https://doi.org/10.31695/IJASRE.2020.33703
Ikewuchi CC (2012) Hypocholesterolemic effect of an aqueous extract of the leaves of Sansevieria senegambica Baker on plasma lipid profile and atherogenic indices of rats fed egg yolk supplemented diet. Exp Clin Sci J (EXCLI J) 11:346–356
Janecka A, Fichna J, Janecki T (2004) Opioid receptors and their ligands. Curr Top Med Chem 4(1):1–17. https://doi.org/10.2174/1568026043451618
Jin MJ, Han HK (2010) Effect of piperine, a major component of black pepper, on the intestinal absorption of fexofenadine and its implication on food-drug interaction. J Food Sci 75(3):H93–H96. https://doi.org/10.1111/j.1750-3841.2010.01542.x
Kadam SS, Smithard RR, Eyre MD, & Armstrong DG (1987) Effects of heat treatments of antinutritional factors and quality of proteins in winged bean. J Sci Food Agric 39(3): 267–275doi: https://doi.org/10.1002/jsfa.2740390310
Klupšaitė D, Juodeikienė G (2015) Legume: composition, protein extraction and functional properties. A review. Chem Technol 1(66). https://doi.org/10.5755/j01.ct.66.1.12355
Kulkarni DB, Sakhale BK, Giri NA (2018) A potential review on millet grain processing. Int J Nutr Sci 3(1)
Kumar A, Kumar V, Krishnan V, Hada A, Marathe A, Jolly M, Sachdev A (2019) Seed targeted RNAi-mediated silencing of GmMIPS1 limits phytate accumulation and improves mineral bioavailability in soybean. Sci Rep 9(1):7744. https://doi.org/10.1038/s41598-019-44255-7
Kumar A, Nayak S, Ngangkham U, Sah RP, Lal MK, Tp A, Behera S, Swain P, Behera L, Sharma S (2021) A single nucleotide substitution in the SPDT transporter gene reduced phytic acid and increased mineral bioavailability from Rice grain (Oryza sativa L.). J Food Biochem 45(7):e13822. https://doi.org/10.1111/jfbc.13822
Kumari S, Sachdeva A, Krishnan V (2014) Impact of soaking and germination durations on antioxidants and antinutrients of black and yellow soybean (Glycine max. L) varieties. J Plant Biochem Biotechnol 24(3):355–358. https://doi.org/10.1007/s13562-014-0282-6
Lampart-Szczapa E, Siger A, Trojanowska K, Nogala-Kalucka M, Malecka M, cholek B. (2003) Chemical composition and antibacterial activities of lupin seeds extracts. Nahrung/Food 47(5):286–290
Larson SR, Rutger N, Young KA, Raboy V (2000) Isolation and genetic mapping of a non-lethal rice (Oryza sativa L.) low phytic acid 1 mutation. Crop Sci J 40:1397–1405. https://doi.org/10.2135/cropsci2000.4051397x
Lestienne I, Buisson M, Lullien-Pellerin V, Picq C, Trèche S (2007) Losses of nutrients and anti-nutritional factors during abrasive decortication of two pearl millet cultivars (Pennisetum glaucum). Food Chem 100(4):1316–1323. https://doi.org/10.1016/j.foodchem.2005.11.027
Lister J, Fletcher PJ, Nobrega JN, Remington G (2015) Behavioral effects of food-derived opioid-like peptides in rodents: implications for schizophrenia? Pharmacol Biochem Behav 134:70–78. https://doi.org/10.1016/j.pbb.2015.01.020
Liu T-T, Liu X-T, Chen Q-X, Shi Y (2020) Lipase inhibitors for obesity: a review. Biomed Pharmacother 128. https://doi.org/10.1016/j.biopha.2020.110314
Liu Q, Yang F, Zhang J, Liu H, Rahman S, Islam S, Ma W, She M (2021) Application of CRISPR/Cas9 in crop quality improvement. Int J Mol Sci 22(8):4206. https://doi.org/10.3390/ijms22084206
Lopez-Rodriguez NA, Gaytán-Martínez M, de la Luz R-VM, Loarca-Piña G (2020) Glucosinolates and isothiocyanates from Moringa oleifera: chemical and biological approaches. Plant Foods Hum Nutr. https://doi.org/10.1007/s11130-020-00851-x
Lunagariya NA, Patel NK, Jagtap S, Bhutani KK (2014) Inhibitors of pancreatic lipase: state of the art and clinical perspectives. Exp Clin Sci J (EXCLI J) 13:897–921. https://doi.org/10.17877/DE290R-6941
Mahgoub SE, Elhag SA (1998) Effect of milling, soaking, malting, heat-treatment and fermentation on phytate level of four Sudanese sorghum cultivars. Food Chem 61(1–2):77–80. https://doi.org/10.1016/s0308-8146(97)00109-x
Malar DS, Devi KP (2014) Dietary polyphenols for treatment of Alzheimer’s disease--future research and development. Curr Pharm Biotechnol 15(4):330–342. https://doi.org/10.2174/1389201015666140813122703
Masud T, Mahmood T, Latif A, Sammi S, Hameed T (2007) Influence of processing and cooking methodologies for reduction of phytic acid content in wheat (Triticum aestivum) varieties. J Food Process Preserv 31:583–594
Mattila PH, Pihlava JM, Hellström J, Nurmi M, Eurola M, Mäkinen S, Jalava T, Pihlanto A (2018) Contents of phytochemicals and antinutritional factors in commercial protein-rich plant products. Food Qual Safety 2(4):213–219. https://doi.org/10.1093/fqsafe/fyy021
McCue KF, Allen PV, Rockhold DR, Maccree MM, Belknap WR, Shephard LV, Davies H, Joyce PE, Corsini DL, Moehs CP (2003) Reduction of total steroidal glycoalkaloids in potato tubers using antisense constructs of a gene encoding a solanidine glucosyl transferase. Acta Horticulturae 619:77–86. https://doi.org/10.17660/ActaHortic.2003.619.9
Melrose (2019) The glucosinolates: a sulphur glucoside family of mustard anti-tumour and antimicrobial phytochemicals of potential therapeutic application. Biomedicine 7(3):62. https://doi.org/10.3390/biomedicines7030062
Miękus N, Marszałek K, Podlacha M, Iqbal A, Puchalski C, Świergiel AH (2020) Health benefits of plant-derived sulfur compounds, glucosinolates, and organosulfur compounds. Molecules 25(17):3804. https://doi.org/10.3390/molecules25173804
Mohan VR, Tresina PS, Daffodil ED (2016) Antinutritional factors in legume seeds: characteristics and determination. Encycl Food Health:211–220. https://doi.org/10.1016/B978-0-12-384947-2.00036-2
Mohapatra D, Patel AS, Kar A, Deshpande SS, Tripathi MK (2019) Effect of different processing conditions on proximate composition, anti-oxidants, anti-nutrients and amino acid profile of grain sorghum. Food Chem 271:129–135. https://doi.org/10.1016/j.foodchem.2018.07.196
Nikmaram N, Leong SY, Koubaa M, Zhu Z, Barba FJ, Greiner R, Oey I, Roohinejad S (2017) Effect of extrusion on the anti-nutritional factors of food products: an overview. Food Control 79:62–73. https://doi.org/10.1016/j.foodcont.2017.03.027
Ning W, Wang Y, Zhang F, Wang H, Wang F, Wang X, Tang H, Liang S, Shi X, Liu Z (2013) Beneficial effects of trypsin inhibitors derived from a spider venom peptide in l-arginine-induced severe acute pancreatitis in mice. PLoS One 8(4):e61049. https://doi.org/10.1371/journal.pone.0061049
Nkhata SG, Ayua E, Kamau EH, Shingiro JB (2018) Fermentation and germination improve nutritional value of cereals and legumes through activation of endogenous enzymes. Food Sci Nutr 6(8):2446–2458
Nyberg FAC, Hallberg M (2013) Casomorphins/hemorphins. In: Kastin A (ed) Handbook of biologically active peptides. USA, Academic Press, pp 1550–1555
Oghbaei M, Prakash J (2016) Effect of primary processing of cereals and legumes on its nutritional quality: a comprehensive review. Cogent Food Agric 2(1). https://doi.org/10.1080/23311932.2015.1136015
Ohiokpehai O (2003) Promoting the nutritional goodness of traditional food products. Pak J Nutr 2(4):267–270
Ostlund RE Jr (2002) Phytosterols in human nutrition. Annu Rev Nutr 22:533–549. https://doi.org/10.1146/annurev.nutr.22.020702.075220
Paiva PMG, Pontual E, Coelho L, Napoleão TH (2013) Protease inhibitors from plants: biotechnological insights with emphasis on their effects on microbial pathogens. In: Microbial pathogens and strategies for combating them: science, technology and education, vol 1, pp 641–649
Pal RS, Bhartiya A, ArunKumar R, Kant L, Aditya JP, Bisht JK (2015) Impact of dehulling and germination on nutrients, antinutrients, and antioxidant properties in horsegram. J Food Sci Technol 53(1):337–347. https://doi.org/10.1007/s13197-015-2037-3
Pal RS, Bhartiya A, Yadav P, Kant L, Mishra KK, Aditya JP, Pattanayak A (2016). Effect of dehulling, germination and cooking on nutrients, anti-nutrients, fatty acid composition and antioxidant properties in lentil (Lens culinaris). J Food Sci Technol 54(4):909–920. doi:https://doi.org/10.1007/s13197-016-23
Panche AN, Diwan AD, Chandra SR (2016) Flavonoids: an overview. J Nutr Sci 5. https://doi.org/10.1017/jns.2016.41
Perera I, Seneweera S, Hirotsu N (2018) Manipulating the phytic acid content of rice grain toward improving micronutrient bioavailability. Rice 11(1):4. https://doi.org/10.1186/s12284-018-0200-y
Popova A, Mihaylova D (2019) Antinutrients in plant-based foods: a review. Open Biotechnol J 13:68–76. https://doi.org/10.2174/1874070701913010068
Possenti M, Baima S, Raffo A, Durazzo A, Giusti AM, Natella F (2016) Glucosinolates in food. In: Glucosinolates. Springer, Cham, pp 1–46. https://doi.org/10.1007/978-3-319-26479-0_4-1
Prieto MA, López CJ, Simal-Gandara J (2019) Glucosinolates: molecular structure, breakdown, genetic, bioavailability, properties and healthy and adverse effects. Adv Food Nutr Res. https://doi.org/10.1016/bs.afnr.2019.02.008
Pruimboom L, de Punder K (2015) The opioid effects of gluten exorphins: asymptomatic celiac disease. J Health Popul Nutr 33(24). https://doi.org/10.1186/s41043-015-0032-y
Punjabi M, Bharadvaja N, Jolly M, Dahuja A, Sachdev A (2018) Development and evaluation of low Phytic acid soybean by siRNA triggered seed specific silencing of inositol polyphosphate 6-/3-/5-kinase gene. Front Plant Sci 9:804. https://doi.org/10.3389/fpls.2018.00804
Rasha Mohamed K, Abou-Arab EA, Gibriel AY, Rasmy NM, Abu-Salem FM (2011) Effect of legume processing treatments individually or in combination on their phytic acid content. Afr J Food Sci Technol 2(2):036–046
Reddy NR, Sathe SK, Salunkhe DK (1982) Phytates in legumes and cereals. Adv Food Res 28:1–92. https://doi.org/10.1016/s0065-2628(08)60110-x
Rehman Z, Shah WH (2001) Tannin contents and protein digestibility of black grams (Vigna mungo) after soaking and cooking. Plant Foods Hum Nutr 56:265–273
Samtiya M, Aluko RE, Dhewa T (2020) Plant food anti-nutritional factors and their reduction strategies: an overview. Food Prod Process Nut 2(6). https://doi.org/10.1186/s43014-020-0020-5
Samtiya M, Aluko RE, Puniya AK, Dhewa T (2021) Enhancing micronutrients bioavailability through fermentation of plant-based foods: a concise review. Fermentation 7(2):63. https://doi.org/10.3390/fermentation7020063
Santos AFS, da Silva MDC, Napoleão TH, Paiva PMG, Correia MTS, Coelho LCBB (2014) Lectins: function, structure, biological properties and potential applications. Curr Top Peptide Protein Res 15:41–62
Sashidhar N, Harloff HJ, Potgieter L, Jung C (2020) Gene editing of three BnITPK genes in tetraploid oilseed rape leads to significant reduction of phytic acid in seeds. Plant Biotechnol J 18(11):2241–2250. https://doi.org/10.1111/pbi.13380
Savage G, Klunklin W (2018) Oxalates are found in many different European and Asian foods—effects of cooking and processing. J Food Res 7(3):76. https://doi.org/10.5539/jfr.v7n3p76
Savage GP, Mårtensson L (2010) Comparison of the estimates of the oxalate content of taro leaves and corms and a selection of Indian vegetables following hot water, hot acid and in vitro extraction methods. J Food Compos Anal 23(1):113–117. https://doi.org/10.1016/j.jfca.2009.07.001
Shahidi F, Ramakrishnan VV, Oh WYJ (2019) Bioavailability and metabolism of food bioactives and their health effects: a review. J Food Bioactives 8:6–41. https://doi.org/10.31665/JFB.2019.8204
Shi J, Arunasalam K, Yeung D, Kakuda Y, Mittal G, Jiang Y (2004) Saponins from edible legumes: chemistry, processing, and health benefits. J Med Food 7(1):67–78. https://doi.org/10.1089/109662004322984734
Shukla VK, Doyon Y, Miller JC, DeKelver RC, Moehle EA, Worden SE, Mitchell JC, Arnold NL, Gopalan S, Meng X, Choi VM (2009) Precise genome modification in the crop species Zea mays using zinc-finger nucleases. Nature 459(7245):437–441. https://doi.org/10.1038/nature07992
Siener R, Hönow R, Voss S, Seidler A, Hesse A (2006) Oxalate content of cereals and cereal products. J Agric Food Chem 54(8):3008–3011. https://doi.org/10.1021/jf052776v
Singh AP, Kumar S (2019) Applications of tannins in industry. In: Tannins-structural properties, biological properties and current knowledge. https://doi.org/10.5772/intechopen.85984
Singh G, Suresh S, Bayineni VK, Kadeppagari RK (2015) Lipase inhibitors from plants and their medical applications. Int J Pharmacy Pharm Sci 7:1–5
Siritunga D, Arias-Garzon D, White W, Sayre RT (2004) Over-expression of hydroxynitrile lyase in cassava roots accelerates cyanogenesis and detoxification. Plant Biotechnol J 2(1):37–43. https://doi.org/10.1046/j.1467-7652.2003.00047.x
Soundararajan P, Kim J (2018) Anti-carcinogenic glucosinolates in cruciferous vegetables and their antagonistic effects on prevention of cancers. Molecules 23(11):2983. https://doi.org/10.3390/molecules23112983
Sridhar SNC, George G, Verma A, Paul AT (2019) Natural products-based pancreatic lipase inhibitors for obesity treatment. In: Natural bio-active compounds. Springer, Singapore, pp 149–191. https://doi.org/10.1007/978-981-13-7154-7_6
Srivastava S (1994). Study on the effects of processing on nutrient and non-nutrient composition of Lathyrus sativus. M.Sc. Thesis, CCS Haryana Agricultural University, Hisar, India.
Tanito M, Masutani H, Kim Y-C, Nishikawa M, Ohira A, Yodoi J (2005) Sulforaphane induces thioredoxin through the antioxidant-responsive element and attenuates retinal light damage in mice. Invest Ophthalmol Vis Sci 46:979–987
Tatham AS, Shewry PR (2008) Allergens to wheat and related cereals. Clin Exp Allergy 38(11):1712–1726. https://doi.org/10.1111/j.13652222.2008.03101.x
Tibe O, Amarteifio J (2010) Trypsin inhibitor activity and condensed tannin content in Bambara Groundnut (Vigna Subterranea (L.) Verdc) Grown in Southern Africa. J Appl Sci Environ Manag 11(2). https://doi.org/10.4314/jasem.v11i2.55021
Trivedi MS, Hodgson NW, Walker SJ, Trooskens G, Nair V, Deth RC (2015) Epigenetic effects of casein-derived opioid peptides in SH-SY5Y human neuroblastoma cells. Nutr Metab 12:54. https://doi.org/10.1186/s12986-015-0050-1
Tungmunnithum D, Thongboonyou A, Pholboon A, Yangsabai A (2018) Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: an overview. Medicines 5(3):93
Van Buul VJ, Brouns FJPH (2014) Health effects of wheat lectins: a review. J Cereal Sci 59(2):112–117. https://doi.org/10.1016/j.jcs.2014.01.010
Van Campen DR (1969) Copper interference with the intestinal absorption of zinc-65 by rats. J Nutr 97(1):104–108. https://doi.org/10.1093/jn/97.1.104
Vanga SK, Wang J, Raghavan V (2020) Effect of ultrasound and microwave processing on the structure, in-vitro digestibility and trypsin inhibitor activity of soymilk proteins. LWT 131:109708. https://doi.org/10.1016/j.lwt.2020.109708
Vaquero MP, Muniz FS, Redondo SJ, Oliván PP, Higueras FJ, Bastida S (2010) Major diet-drug interactions affecting the kinetic characteristics and hypolipidaemic properties of statins. Nutr Hosp 25(2):193–206
Vats P, Banerjee UC (2004) Production studies and catalytic properties of phytases (myo-inositol-hexakis-phosphate phosphohydrolases): an overview. Enzyme Microb Technol 35:3–14. https://doi.org/10.1016/s0141-0229(04)00087-0
Vauzour D, Rodriguez-Mateos A, Corona G, Oruna-Concha MJ, Spencer JP (2010) Polyphenols and human health: prevention of disease and mechanisms of action. Nutrients 2(11):1106–1131. https://doi.org/10.3390/nu2111106
Vijayakumari K, Siddhuraju P, Janardhanan K (1996) Effect of different post-harvest treatments on antinutritional factors in seeds of the tribal pulse, Mucuna pruriens (L.) DC. Int J Food Sci Nutr 47(3):263–272. https://doi.org/10.3109/09637489609012587
Villacrés E, Quelal MB, Fernández E, Garcìa G, Cueva G, Rosell CM (2020) Impact of debittering and fermentation processes on the antinutritional and antioxidant compounds in Lupinus mutabilis sweet. LWT 131:109745. https://doi.org/10.1016/j.lwt.2020.109745
Wang Y, Lv Z, Chu Y (2015) HIV protease inhibitors: a review of molecular selectivity and toxicity. HIV/AIDS: Res Palliative Care 7:95. https://doi.org/10.2147/hiv.s79956
Wisessing A, Choowongkomon K (2012) Amylase inhibitors in plants: structures, functions and application. Funct Plant Sci Biotechnol 6(1):31–41
Yildirım I, Kutlu T (2015) Anticancer agents: saponin and tannin. Int J Biol Chem 9:332–340. https://doi.org/10.3923/ijbc.2015.332.340
Youn Hwang D (2019) Therapeutic role of natural products containing tannin for treatment of constipation. Constipation. https://doi.org/10.5772/intechopen.81837
Yun JW (2010) Possible anti-obesity therapeutics from nature—a review. Phytochemistry 71(14-15):1625–1641. https://doi.org/10.1016/j.phytochem.2010.07.011
Zhang L, Li J, Han F, Ding Z, Fan L (2017) Effects of different processing methods on the antioxidant activity of 6 cultivars of foxtail millet. J Food Qual:1–9.: Article ID 8372854. https://doi.org/10.1155/2017/8372854
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Soni, K., Samtiya, M., Krishnan, V., Dhewa, T. (2022). Antinutritional Factors: Nutrient Bioavailability and Health Beneficial Effects. In: S. V., R., Praveen, S. (eds) Conceptualizing Plant-Based Nutrition. Springer, Singapore. https://doi.org/10.1007/978-981-19-4590-8_8
Download citation
DOI: https://doi.org/10.1007/978-981-19-4590-8_8
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-4589-2
Online ISBN: 978-981-19-4590-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)