Abstract
A study has been conducted on the electrochemical conversion of CO2 into useful chemicals and PKL electricity. The experimental technique of CO2 production device for use in the electrochemical cell is found. Here, the chemical reaction is NaHCO3 + HCl = NaCl + H2O + CO2. The produced CO2 goes out through a tube. It is also found that the produced CO2 was supplied to a PKL Electrochemical cell for getting electricity for a long time duration. It is also found that the pH level of PKL extract remains almost constant during electricity generation. The produced CO2 is mixed with the H+ and formed HCO3, which maintains the constant pH level of the PKL extract during PKL electricity generation. Using this technique, the performance of PKL electrochemical cell has been studied. A comparative study has also been conducted between CO2-based and non-CO2-based PKL electrochemical cells. The studied parameters are self-discharge capacity, discharge capacity with load, internal resistance, etc. Mallic acid increased during night time. Performances are better in the night time for collected PKL than the day time collected PKL. Reactant ion (H+) increases in the night time. Cell voltage (Ecell) increases in the night time. The pH of the PKL extract decreases in the night time. This type of work has not been done before yet all over the world. This work will help in the sustainable PKL electricity generation in near future.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
- CO2 production
- Electrochemical conversion
- PKL electricity
- Useful chemicals
- CO2-based PKL electrochemical cell
- Galvanic cell
1 Introduction
The main theme should be the addressing global challenges in the new era through research, innovations, and strengthening connections (Hasan and Khan 2017, 2019, 2020). PKL electricity is produced mainly using Zn/Cu-based electrodes and PKL extract. The performances are gradually decreased within a few months (Hazrat Ali et al. 2019; Khan et al. 2016a, 2017). To remain sustain of this PKL electricity, several techniques have been developed like the use of nanoparticles, dynamic method, use of secondary salt, etc. The use of CO2 is a new technique for PKL electrochemical cells (Khan et al. 2016b, c, 2018a, b). To keep it in mind, the electrochemical conversion of CO2 into useful chemicals and PKL electricity has been designed and developed (Khan et al. 2019a, b). The PKL electricity is a new, renewable energy and innovative technique across the globe (Khan et al. 2019c). So that R&D work should be done regularly on PKL electricity. This research work will also be an excellent avenue for collaborative work for more innovative research for the uplifting of the community and industrial development. The nominal voltage of a galvanic cell is fixed by the electrochemical characteristics of the active chemicals used in the cell (Khan et al. 2020). The actual voltage generated at the cell terminals at a particular time depends on the load current and the internal impedance of the cell, and this also varies with the temperature, the state of charge, and with the age of the cell. A comparative study of the voltage of the normal voltaic/galvanic cell, traditional PKL electrochemical cell, and CO2-based PKL electrochemical cell has also been conducted (Ruhane et al. 2017a). The PKL electrochemical cell depends on some parameters (Ruhane et al. 2017b) which have been discussed here. The production and the use of CO2 is an innovative technology for an electrochemical cell or battery. The different parameters of the PKL electrochemical cells both with and without CO2 have been studied.
2 Methodology
In this section, the methodology of CO2 production has been discussed. The methodology of the preparation of PKL electrochemical cell has also been studied. Finally, the use of CO2 for PKL Electricity has been designed and fabricated.
2.1 Experimental Procedure
The experimental setup of the CO2 production device for practical utilization is shown (Fig. 1). The chemical reaction was NaHCO3 + HCl = NaCl + H2O + CO2. The produced CO2 goes out through a tube. Figure 1 also shows the collection of CO2 by a balloon. Figure 2 shows the utilization of CO2 in a PKL electrochemical cell for getting more electricity for a long time duration.
2.2 Application of CO2 Gas into the PKL Electrochemical Cell: Preparation of CO2 Gas
About 2.0 g of powdered NaHCO3 and 250 mL 0.1 M HCl solution were taken in a 500 mL conical flask. Then the mixture was heated very slowly, releasing CO2 gas which was dried by passing through silica gel.
The methods of CO2 gas preparation in the laboratory are shown in Fig. 3. Here, Fig. 3a shows the Schematic diagram of CO2 gas preparation and Fig. 3b shows the Experimental setup of CO2 gas preparation.
Instrumentation and Apparatus
Model CHI 660E electrochemical workstation, Graphite pencil electrode (GPE) as working electrode (Livo), GCE, modified GCE, carbon-based paper electrode and activated GPE electrode as working electrode, Pt wire auxiliary electrode, Ag/AgCl reference electrode, pH meter (Hanna), Digital Multimeter, Zn, Cu, Al, Ag, Fe electrodes, and Magnetic Stirrer.
Chemicals and Reagents.
Carbon dioxide (CO2), Sodium bicarbonate solution (NaHCO3), Hydrochloric acid (HCl), PBS solutions for various pH, Deionized water or double-distilled water, Graphite powder, Activated charcoal (Sigma Aldese), Nujol, Alumina powder, Silica gel, and Highly pure N2 gas (Linde BD Ltd.).
2.3 Zn and Cu Electrodes Preparation
To prepare a PKL (Pathor Kuchi Leaf) electrochemical cell, a cathode and an anode are needed. In our research article, it is shown that Zn (Zinc) plate is used as a cathode and the Cu (Copper) plate is used as an anode. The Zn and Cu plates are available in the local market. The price of the Zn plate is of low cost compared to the Cu plate. On the other hand, the longevity of the Cu plate is much more than the Zn plate. Moreover, the Zn plate is called a sacrificial element rather than the other metal plates. That is why the performance Zn/Cu-based PKL electrochemical cell is better than the other metals. The preparation technique of the Zn/Cu-based PKL electrochemical cell for practical utilization is shown in Fig. 4.
Figure 4a shows the Zn and Cu for a cell (Zn:Cu = 1:1). Figure 4b shows the keeping of Zn plate first (Zn:Cu = 2:2). Figure 4c shows the keeping of Cu plate first (Zn:Cu = 2:2). Figure 4d shows the keeping of the Zinc plate first (Zn:Cu = 3:2). Figure 4e shows the keeping of the Zinc plate first (Zn:Cu = 3:23:2). Figure 4f shows the design and fabrication of 1 KW PKL Power and, finally, Fig. 4 g shows the Practical application of 1 KW PKL Power.
2.4 Chemical Reactions
Since Zn is a sacrificial element, we have
Adding (1) + (2), \({\text{Zn }} + {\text{ 2e}}^{ - } + {\text{Cu}}^{{{2} + }} = {\text{Zn}}^{{{2} + }} + {\text{2e}}^{ - } + {\text{ Cu}}\)
where Cu2+ = Reactant Ions and Zn2+ = Product Ions.
Adding (4) + (5), \({\text{Zn }} + {\text{ 2e}}^{ - } + {\text{H}}^{ + } = {\text{Zn}}^{{{2} + }} + {\text{2e}}^{ - } + {\text{ H}}_{{2}}\)
where H+ = Reactant Ions and Zn2+ = Product Ions.
Here, NaCl and H2O are the useful chemicals and CO2 is the conversion/production product of electricity.
From Eq. (7), it is shown that the produced CO2 can be used in the dead PKL electrochemical cell. The pH of the dead PKL electrochemical cell decreased than that of starting pH of the PKL electrochemical cell. When the produced CO2 is applied to the dead PKL electrochemical cell, then the following chemical equation has occurred:
The HCO3 will be produced in the dead PKL electrochemical cell. It is shown that the pH of the dead PKL electrochemical cell has been increased and then the dead PKL electrochemical cell has become reactive to generate electricity. In this case, H2 gas will be produced as a byproduct.
2.5 Advantages of Electrochemical Reduction of CO2
It is an easy process and the rate of electrochemical reduction of CO2 is so much higher than the chemical reduction of CO2. The advantages are as follows: (1) In presence of CO2, it can easily convert into HCOOH (formic acid), CO (carbon monoxide), CH3OH (methanol), (COOH)2 (oxalic acid), C2H4 (ethylene), CH4 (methane), etc. (which are useful products). (2) During the electrochemical conversion of CO2 without aqueous catholyte (electrolyte), other’s reagent is not necessary as the chemical conversion of CO2 into the same product. (3) Higher temperature and pressure is not necessary as the chemical conversion of CO2:

(4) In this process very low current (micro amp level) is needed to get the product. Because in this process, an electron is necessary (which is available here and obtained easily) for reduction. So this process is not expensive. (5) In this process, the reaction is carried out in the solution phase. Generally, the solution phase reaction is more easy than gas phase reaction.
2.6 Application of CO2 Gas into the PKL Electrochemical Cell
During the preparation of the PKL electrochemical cell, the PKL extract was mixed with H2O. In this case, O2 was dissolved in water. So that O2 should be neutralized before being applied to CO2 gas into the PKL electrochemical cell.
3 Results and Discussion
3.1 Comparative Studies on Performance of Traditional Electrolytic Cells/Batteries
The variation of the voltages for five different typical electrolytic cells/batteries has been studied. It is also shown the comparison of the variation of the voltages with the variation of percentage of capacity discharged among the five different typical electrolytic cells/battery like Zn/MnO2, Ni–Cd, Ni–Zn, Lead Acid, and Lithium-Ion battery.
Figure 5 shows that the variation of the voltages with the variation of percentage of capacity discharged for Zn/MnO2 electrolytic cells/battery. The change of voltage was (1.50–1.10 V) 0.40 V up to 100% of Capacity Discharged. Figure 6 shows that the voltages vary with the percentage of capacity discharged for Ni–Cd electrolytic cells/batteries. The change of voltage was (1.48–1.15 V) 0.33 V up to 100% of capacity discharged.
Figure 7 shows that the voltages vary with the variation of percentage of capacity discharged for Ni–Zn electrolytic cells/batteries. The change of voltage was (1.60–1.55 V) 0.05 V up to 100% of Capacity Discharged. Figure 7 shows that the voltages vary with the percentage of capacity discharged for lead acid electrolytic cells/batteries. The change of voltage was (2.01–1.73 V) 0.24 V up to 100% of Capacity Discharged (Figs. 8 and 9).
Figure 7 shows that the voltages vary with the percentage of capacity discharged for lithium-ion electrolytic cells/batteries. The change of voltage was (4.10–3.80 V) 0.30 V up to 100% of Capacity Discharged.
Figure 10 shows that the Cell voltages (V) vary with the percentage capacity of discharged for Lithium-Ion, Lead Acid, Ni–Zn, Ni–Cd, and Zn/MnO2 electrolytic cells together.
3.2 Discharge Characteristics with Load for PKL Electrochemical Cell
The discharge characteristics of PKL cell/module has been measured carefully. The fabricated PKL module was 6 V, which was used as a load for practical utilizations. A Light Emitting Diode (LED) lamp was used as a load of 6 V. For simplicity, it has been shown that the PKL module system is as shown in Fig. 11.
The observed values of load voltage, load current, and load power are tabulated. Taking the load tests for 120 h has been continued. It may be pointed out here that initially when we connected the load, a substantial voltage drop occurs and this drop is 0.48 V which is around 8% of system voltage.
Voltage, Current, and power of PKL module under load conditions have been studied. The data was taken up to 120 h. The data has been tabulated very carefully. The discharge characteristics of a PKL electrochemical cell with the load are also shown. The Voltage, Current, and power without CO2-based PKL module under load condition. It is shown that the change of load voltage for 120 h is 0.34 V, that the change of load current for 120 h is 0.32 A, and that the change of load power for 120 h is 1.9 W (Fig. 12).
Figure 13 shows that the load voltage varies with the time duration. As per this graph, it is shown that the load voltage is reducing gradually as time duration. If we compare it with other cells, we find this change is relatively rapid. Figure 14 shows the variation of load current with the variation of time duration. As per this graph, it is seen that the load current is reducing gradually as time passes. If we compare it with other traditional cells, we find this change is relatively rapid.
Figure 15 shows the load power graph for the same system. According to the definition of load power, we have Power, P = V × I Watts, where V = Voltage (in volts) and I = Current (in Amp). Since voltage and current are reducing with time duration, therefore, as per this graph, it is seen that the load power is also reducing gradually as time passes.
Figure 16 shows the observed results which have been plotted for three characteristics of PKL cells together. The variations of load voltage, load current, and load power with time duration have been discussed.
3.3 Discharge Characteristics with CO2-Based PKL Electrochemical Cell for Load
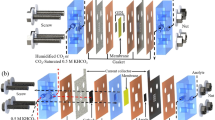
Figure 17 shows the methods of CO2 Production for the PKL module. Figure 18 shows the method of CO2-based PKL module for electricity production. Figure 19 shows the Measurement technique of pH of the PKL extract of a CO2-based PKL unit cell. Figure 20 shows the appliance of CO2-based PKL electricity with the load.
The Voltage, Current, and power of CO2-based PKL (Bryophillum pinnatum Leaf) module under load conditions have been studied. It is shown that the change of load voltage for 120 h is 0.04 V, that the change of load current for 120 h is 0.02 A, and that the change of load power for 120 h is 0.14 W.
Figure 21 shows that the Load voltage (V) for CO2-based PKL cell varies with the time duration (h). It is shown that the load voltage was constant up to 15 h and then it decreased up to 0.01 V up to 45 h and then after it decreased 0.01 V up to 65 h and then it decreased 0.01 V up to 100 h, and finally, it decreased 0.01 V up to 120 h. Figure 22 shows that the Load current (A) for CO2-based PKL electrochemical cell varies with the time duration (h). It shows that the load current was constant up to 35 h and then it decreased up to 0.01 A up to 100 h and finally, it decreased 0.01 A up to 120 h.
Figure 23 shows that the Load power (W) for CO2-based PKL electrochemical cell varies with the time duration (h). It shows that the load power was constant up to 15 h and then it decreased up to 0.06 W up to 35 h and then after it decreased 0.01 W up to 65 h and then it decreased 0.06 W up to 100 h, and finally, it decreased 0.01 W up to 120 h.
Figure 24 shows the variation of the voltage, current, and power of CO2-based PKL module under load conditions. It is shown that the change of load voltage is 0.04 V, that the change of load current for 120 h is 0.02 A, and that the change of load power for 120 h is 0.14 W for 120 h.
3.4 Self-discharge Characteristics of a CO2-Based PKL Electrochemical Cell
Self-discharge characteristics of a CO2-based PKL electrochemical cell are found. Using the Self-discharge characteristics, some data for open-circuit voltage (Voc), Short circuit current (Isc), maximum power (Pmax), and internal resistance (rin) has been collected and then tabulated carefully.
Figure 25 shows that the open-circuit voltage Voc (V) varies with and without CO2-based PKL electrochemical cell with the time duration (h). It is found that the open-circuit voltage (Voc) is better with CO2-based PKL electrochemical cell than without CO2-based PKL electrochemical cell. Furthermore, the open-circuit voltage is more steady with CO2-based PKL electrochemical cell than without CO2-based PKL electrochemical cell. So that it can be said that the longevity has been increased for use in CO2 with the PKL extract.
Figure 26 shows that the short circuit current Isc (A) with and without CO2-based PKL electrochemical cell varies with the time duration (h). It is found that the short circuit current (Isc) is better with CO2-based PKL electrochemical cell than without CO2-based PKL electrochemical cell. Furthermore, the short circuit current (Isc) is more with CO2-based PKL electrochemical cell than without CO2-based PKL electrochemical cell. So that it can be said that the performance has been increased for use in CO2 with the PKL extract.
Figure 27 shows that the maximum power Pmax (W) with and without CO2-based PKL electrochemical cell varies with the time duration (h). It is found that the maximum power Pmax (W) is better with CO2-based PKL electrochemical cell than without CO2-based PKL electrochemical cell. Furthermore, the maximum power Pmax (W) is more with CO2-based PKL electrochemical cell than without CO2-based PKL electrochemical cell. So that it can be said that the performance has been increased for use in CO2 with the PKL extract.
Figure 28 shows that the internal resistance, rin (ohm) with and without CO2-based PKL electrochemical cell varies with the time duration (h). It is found that the internal resistance, rin (ohm) is better with CO2-based PKL electrochemical cell than without CO2-based PKL electrochemical cell. Furthermore, the internal resistance, rin (ohm) is less with CO2-based PKL electrochemical cell than without CO2-based PKL electrochemical cell. So that it can be said that the performance has been increased for using CO2 gas with the PKL extract .
4 Conclusions
There are a lot of research papers on the electrochemical conversion of CO2 into useful chemicals. But there was no use of CO2 for electricity generation. This time the produced CO2 has been used for electricity production. It is found that the performance of PKL electrochemical cell during electricity generation has been increased for using the produced CO2. The variation of the voltage, current, and power of CO2-based PKL module under load conditions has been studied. It is shown that the change of load voltage is 0.04 V, that the change of load current for 120 h is 0.02 A, and that the change of load power for 120 h is 0.14 W for 120 h. The Voltage, Current, and power without CO2-based PKL module under load condition were found. It is shown that the change of load voltage for 120 h is 0.34 V, that the change of load current for 120 h is 0.32 A, and that the change of load power for 120 h is 1.9 W. Finally, it is also found that the performances have been increased after adding CO2 with the PKL extract. The life cycle of the PKL electrochemical cell has been increased. For a bigger size of the PKL electric plant, a CO2 gas cylinder is needed. By using this CO2 gas technology, the dead PKL electrochemical cell will get life for the generation of electricity. In this research work, the produced H2 gas was a byproduct. Mallic acid increases during night time, Performances are better for night time collected PKL than the day time collected leaf. Reactant ion (H + ) increases in the night time.
References
M. Hasan, K.A. Khan, Dynamic model of Bryophyllum pinnatum leaf fueled BPL cell: a possible alternate source of electricity at the off-grid region in Bangladesh. Microsystem Technologies (2018). Micro and Nanosystems Information Storage and Processing Systems, Springer, ISSN 0946–7076, Microsyst. Technol. (2017). https://doi.org/10.1007/s00542-018-4149-y
M. Hasan, K.A. Khan, Experimental characterization and identification of cell parameters in a BPL electrochemical device. SN Appl. Sci. 1, 1008 (2019). https://doi.org/10.1007/s42452-019-1045-8
L. Hassan, K.A. Khan, A study on harvesting of PKL electricity. J. Microsyst. Technol. 26, 1031–1041 (2020). https://doi.org/10.1007/s00542-019-04625-7. 26(3), 1032–1041 (2019)
M. Hazrat Ali, U. Chakma, D. Howlader, M. Tawhidul Islam, K.A. Khan, Studies on performance parameters of a practical transformer for various utilizations. Microsyst. Technol. (2019). https://doi.org/10.1007/s00542-019-04711-w
M.K.A. Khan, M.S. Rahman, T. Das, M.N. Ahmed, K.N. Saha, S. Paul, Investigation on parameters performance of Zn/Cu Electrodes of PKL, AVL, tomato and lemon juice based electrochemical cells: a comparative study, in Electrical Information and Communication Technology (EICT), 2015 3rd International Conference on (IEEE, 2017), pp. 1–6
K.A. Khan, A. Rahman, M.S. Rahman, A. Tahsin, K.M. Jubyer, S. Paul, Performance analysis of electrical parameters of PKL electricity (An experimental analysis on discharge rates, capacity & discharge time, pulse performance and cycle life & deep discharge of PathorKuchi Leaf (PKL) electricity cell), in Innovative Smart Grid Technologies-Asia (ISGT-Asia), 2016 IEEE (IEEE, 2016a), pp. 540–544
M.K.A. Khan, S. Paul, M.S. Rahman, R.K. Kundu, M.M. Hasan, M. Moniruzzaman, M.A. Mamun, A study of performance analysis of PKL electricity generation parameters: (An experimental analysis on voltage regulation, capacity and energy efficiency of pathorkuchi leaf (PKL) electricity cell), in Power India International Conference (PIICON), 2016 IEEE 7th (IEEE, 2016b), pp. 1–6
K.A. Khan, A. Rahman, M. Siddikur Rahman, A. Tahsin, K.M. Jubyer, S. Paul, Performance analysis of electrical parameters of PKL electricity (An experimental analysis on discharge rates, capacity & discharge time, pulse performance and cycle life & deep discharge of Pathor Kuchi Leaf (PKL) electricity cell), in Innovative Smart Grid Technologies-Asia (ISGT-Asia), 2016 IEEE (IEEE, 2016c), pp. 540–544
K.A. Khan, L. Hassan, A.K.M. Obaydullah, S.M. Azharul Islam, M.A. Mamun, T. Akter, M. Hasan, M. Shamsul Alam, M. Ibrahim, M. Mizanur Rahman, M. Shahjahan, Bioelectricity: a new approach to provide the electrical power from vegetative and fruits at off-grid region. Microsyst. Technol. (2018a). https://doi.org/10.1007/s00542-018-3808-3
K.A. Khan, M.S. Bhuyan, M.A. Mamun, M. Ibrahim, L. Hasan, M.A. Wadud, Organic electricity from Zn/Cu-PKL electrochemical cell, in Contemporary Advances in Innovative and Applicable Information Technology, Advances in Intelligent Systems and Computing, by ed. J.K. Mandal et al., vol. 812, Chapter 9, pp. 75–90 (Springer Nature Singapore Pvt. Ltd., 2018b)
K.A. Khan, M. Hazrat Ali, A.K.M. Obaydullah, M.A. Wadud, Production of candle using solar thermal technology. Microsystem Technologies Micro- and Nanosystems Information Storage and Processing Systems, Springer. ISSN 0946–7076, Microsyst. Technol. 25(12) (2019a). https://doi.org/10.1007/s00542-019-04390-7(2019)
K.A. Khan, S.R. Rasel, M. Ohiduzzaman, Homemade PKL electricity generation for use in DC fan at remote areas, Microsystem Technologies Micro- and Nanosystems Information Storage and Processing Systems. ISSN 0946–7076. Microsyst. Technol. 25(12) (2019b). https://doi.org/10.1007/s00542-019-04422-2
K.A. Khan, M.A. Mamun, M. Ibrahim, M. Hasan, M. Ohiduzzaman, A.K.M. Obaydullah, M.A. Wadud, M. Shajahan, PKL electrochemical cell: physics and chemistry. SN Appl. Sci. 1, 1335 (2019c). https://doi.org/10.1007/s42452-019-1363-x
K.A. Khan, M. Hazrat Ali, M.A. Mamun, M. Mahbubul Haque, A.K.M. Atique Ullah, M.N. Islam Khan, L. Hassan, A.K.M. Obaydullah, M.A. Wadud, Bioelectrical characterization and production of nanoparticles (NPs) using PKL extract for electricity generation, Received: 31 July 2018/Accepted: 4 February 2020. Microsyst. Technol. (2020). https://doi.org/10.1007/s00542-020-04774-0
T.A. Ruhane, M. Tauhidul Islam, M. Saifur Rahman, M.M.H. Bhuiyah, J.M.M. Islam, T.I. Bhuiyah, K.A. Khan, M.A. Khan,Impact of photo electrode thickness annealing temperature on natural dye sensitized solar cell. Sustain. Energy Technol. Assess. (2017a). https://doi.org/10.1016/j.seta.2017.01.012
T.A. Ruhane, M. Tauhidul Islam, M. Saifur Rahaman, M.M.H. Bhuiyan, J.M.M. Islam, M.K. Newaz, K.A. Khan, M.A. Khan, Photo current enhancement of natural dye sensitized solar cell by optimizing dye extraction and its loading period. Optik Int. J. Light Electron Optics (2017b)
Acknowledgements
The authors are grateful to the GARE (Grant of Advanced Research in Education) project, Ministry of Education, GoB for providing the financial support during the research work (Project/User ID: PS2019949).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this paper
Cite this paper
Khan, K.A., Mamun, M.A., Adal, M.I., Mia, S., Ali, M.H. (2022). Electrochemical Conversion of CO2 into Useful Chemicals and PKL Electricity. In: Chanda, C.K., Szymanski, J.R., Sikander, A., Mondal, P.K., Acharjee, D. (eds) Advanced Energy and Control Systems. Lecture Notes in Electrical Engineering, vol 820. Springer, Singapore. https://doi.org/10.1007/978-981-16-7274-3_5
Download citation
DOI: https://doi.org/10.1007/978-981-16-7274-3_5
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-7273-6
Online ISBN: 978-981-16-7274-3
eBook Packages: EnergyEnergy (R0)