Abstract
Globally escalating food demand and unpredictable global warming have threatened the humanity in jeopardy. Excessive use of commercial pesticides and chemical fertilizers is providing ease of handling, but their toxic nondegradable residues are known to exert negative impacts on the plants, microbes, and even soil health. The unpredictable climate change leads to enhanced incidence of abiotic and biotic stresses in plants. Several integral approaches of fungi, microbes, plant, and their derived metabolites are used to encounter the stresses for effective crop management. The secondary metabolites are proving their immense potential and have time and again being proven to cope up the unavoidable and unpredictable changes due to adverse environmental conditions. The chapter highlights the role of both microbial and plant-based flavonoids and carotenoids as key agents for managing biotic and abiotic stress tolerance in crop plants. Moreover, such feasible and efficient biological application using plant-based composition helps to mitigate the challenges and open new gates of sustainable agriculture.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Plants as sessile organisms are constantly being affected by changes in their environmental conditions. The unfavorable or stress conditions have forced plants to change the functional aspect of powerful signaling sensors that guide them towards attaining optimal growth via manipulating their biophysical, biochemical, and molecular events (Ingle and Padole 2019; Taïbi et al. 2016; Enebe and Babalola 2018). Unpredictable and unavoidable climatic changes enforce abiotic stresses as the prime limiting factors among diverse stresses threatening agriculture and leading to decline in productivity both qualitatively and quantitatively (Grayson 2013; Nabavi et al. 2020). Majorly, diverse environmental constraints affect crop lands worldwide and only 3.5% of the global land area remains unaffected. The abiotic stresses, viz., salinity, floods, nutrient starvation, drought, heat, light intensity, cold/chilling, anaerobiosis, and acidic conditions are known to decline agricultural productivity worldwide (Meena et al. 2017). In this regard, the enormous chemical compounds have been produced in the plants which can be used as taxonomic markers and known as secondary metabolites (Bennett and Wallsgrove 1994; Tabatabaei and Ehsanzadeh 2016; Kaab et al. 2020). Secondary metabolites are attracting interest of the scientific communities owing to their indirect biological role in the growth and development of organisms. Secondary metabolites are versatile compounds often demonstrating in plant physiological processes as protectants against abiotic and biotic stresses in plants. These play a central role in various associations of plant–rhizobia, plant–microbe, soil microbes, and arbuscular mycorrhizal symbiosis and boost plant growth via the production of important biological agents (Del Valle et al. 2020).
In the last decades, the secondary metabolites are proving their immense potential in the plant stress physiology. The immobile nature of plants diverted the sieve of selection to utilize the secondary metabolites to overcome the stress constrains and help them to survive in the changing environments (Ingle and Padole 2019). Rhizospheric microbes form an interacting association with crop plants which ultimately regulates the nutrient supply to plants through varied direct mechanisms (Rob et al. 2020; Kaab et al. 2020). Studies on plant-based microbial associations at physiological, biochemical, and molecular levels recognized that microbial interactions directly affect plant responses under various stress conditions (Farrar et al. 2014). For deciphering the mysterious interaction among connections and mechanisms regarding the functional role at molecular levels with the resistance responses against abiotic stresses. The priming stimulates on antioxidant function have been found to compare with enhanced transcript levels of glutathione and ascorbate biosynthesis, enzymatic antioxidants that representing complex transcriptional alteration of the antioxidant components (Savvides et al. 2016). Different technological advancements also accelerated understanding of mutant technology, RNAi-mediated gene silencing, metabolite profiling gene editing systems, and proteomic analysis to identify capacious molecular communication that helped in enlightening our awareness of microbial-based mitigation approaches toward abiotic stresses tolerance in plants. New advances in Multiomics have appeared as integrated, holistic, and other analytical approaches for the analysis of one of the major dynamic and complex system of plants-microbial interactions that regulating the consequences progressed in the different plants to improve them to tolerate stress conditions (Luan et al. 2015).
2 Classical Example of Secondary Metabolites in Plant Resistance
Pigmentation is hallmark of microbial colonies rich in flavonoids and carotenoids content. These compounds protect microbes from various adverse conditions including drought, heat, cold, salinity, UV protection, and other damages (Rob et al. 2020). Carotenoids represent an important group of natural occurring pigments that are chiefly abundant in both photosynthetic (algae and plants, cyanobacteria) and nonphotosynthetic organisms (bacteria and fungi). These are synthesized within the chloroplasts of algae and plants naturally and the lycopene cyclization is major branch point in their biosynthesis. Lycopene, the product is converted by the activity of lycopene β-cyclase into β-carotene-like derivatives that plays significant roles against oxidative burst, photoprotection, salinity, drought, and salinity damages in higher plants (Paliwal et al. 2017). Quercetin is the aglycone form of ubiquitous flavonoid glycosides such as quercetin and rutin, rich in the citrus fruits, buckwheat tea, celery, broccoli, lettuce, tomatoes, Ginkgo bioloba, and onions (Table 5.1). They have high antioxidant potential hence fall under the category of effective antioxidants under the UV radiation, drought, salinity, cold, nitrogen deficiency, and heavy metal stress (di Ferdinando et al. 2012; Zhan et al. 2019). The apigenin is naturally occurring metabolite available in 4′, 5, 7-trihydroxyflavone form, and is found to be present in large quantities in celery, chamomile, vine spinach, oregano, and artichoke in the dried form (Table 5.1). They also have high antioxidant potential induced under the drought and UV radiation (Shukla and Gupta 2010). Fisetins are the ubiquitously present glycosylated compounds having powerful antioxidative potential (Gonçalves et al. 2018). Kaempferols are known to regulate several proteins and their functional activities such as cell growth and differentiation (Peer and Murphy 2006; Pei et al. 2020). Naringenin is widely distributed in the bergamot, tomatoes, citrus, and other fruits, which functions to interfere with the activity of the phenylpropanoid pathway enzyme, 4-coumarate: CoA ligase. It causes allelopathic impact on the growth and lessens the lignin deposition in plants (Echinochloa oryzicola, Zea mays, and Oryza sativa) (Deng et al. 2004). Chrysin is involved in the presymbiotic growth, i.e., spore germination, hyphal growth, branching patterns, and the formation of secondary spores, of the various arbuscular mycorrhizal fungi (G. margarita, Gigaspora rosea, Glomus mosseae, and G. intraradices) (Scervino et al. 2005). Apigeninidin is the anthocyanidin produced in the sorghum under the pathogen attack (Mizuno et al. 2014). Rutin is the ubiquitously present flavone enhancing the defense responses against the number of abiotic stresses in plants (Samanta et al. 2011). Catechins are the anthocyanins produced against the pathogen attack in the variety of plants (Barry et al. 2002). Gallic acid is the simple phenolic acid observed in the rice under salt and osmotic stress promoting the antioxidant responses (de Klerk et al. 2011).
Similarly, Syringic acid was observed in the Vicia faba under the osmotic stress involved in the stomatal closure to maintain the turgidity of plants (Purohit et al. 1991). Vanillic acid is a phenolic acid which was observed to enhance rooting in the Malus plant (de Klerk et al. 2011). Hydroxycinnamic acid derivatives such as ferulic acid and caffeic acid are involved in the defense responses. Caffeic acid was observed in the legume plants under the saline and abiotic stresses enhancing the cell wall thickening, lignifications, and inducing plant defense mechanisms (Enebe and Babalola 2018). Similarly, ferulic acid increases the IAA oxidase activity in seedlings, gene overexpression in the ethylene and jasmonic acid biosynthesis, and inhibition of lateral roots and root hair formation (Chi et al. 2013). The resistance provided to organisms mediated by secondary metabolites, viz., carotenoids may have ecological significance allowing organisms to colonize certain environments, for example, invasion of wounds and colonization of leaf surfaces by pigmented heterobasidiomycetous yeasts. In microbes, the knowledge of the mechanisms underlying the control of singlet oxygen species is based on few model organisms, viz., Chlamydomonas, Rhodococcus sphaeroides, E. coli, Agrobacterium tumefaciens, and C. Crescentus. The response to any kind of stress appears to be tightly controlled at the transcriptional and post-transcriptional levels. It appears that the response of the microbes to stress involves a complex network of regulatory factors, which will make it difficult to unravel their individual functions. Recent research on chemical priming has provided further knowledge of the complex mode of action of specific signaling molecules in plant stress tolerance.
3 Flavonoids and Carotenoids: New Contenders of Plant Resistance in Twenty-First Century
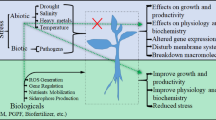
Flavonoids and carotenoids both of plant and microbe origin are well-known contenders providing tolerance response to higher plants in abiotic stresses (Fig. 5.1). Additionally, long-reported responses as protectors of damaging solar radiation both have been known for key roles as antioxidants by reducing the production of reactive oxygen species (ROS) after formation at primary response in stressed plant. Flavonoids are abundantly distributed in various cell organelles including chloroplast, nucleus, small vesicles endoplasmic reticulum, vacuole, cytosol, and extracellular space. Flavonoids such as flavones, flavonols, anthocyanins, and proanthocyanidins have been identified as antioxidants and function to protect plants from cellular damage under stress conditions (Clayton et al. 2018). The chloroplast-associated metabolites scavenge free radicals to maintain cellular redox. Kaempferol and Quercetin glycosides were reported abundant in chloroplasts during fungal attack, ultraviolet (UV) irradiation, salinity, drought, and function as protective antioxidants systems under stress conditions (Rob et al. 2020). According to Agati et al. (2009) flavonoids have long been accounted to be present in the cell vacuoles, epidermal cells wall, glandular, and nonsecretary trichome structures and hence have been expected mainly to have the role of mitigating short ranges of solar wavelengths. Although anthocyanins have been well known to store in the vacuoles (mesophyll cells) in most of the plant species, and strongly supported by in vitro studies (Agati and Tattini 2010). However, studies confined that flavonoids may help in ROS scavenging within or towards the production sites. Microalgae species belonging to genus Chlorella, Dunaliella, Scenedesmus, and Haematococcus are considered as the key producers of commercially vital carotenoids. Many microorganisms have been reported to produce secondary metabolites such as astaxanthin, lutein, and b-carotene, etc. (Table 5.2).
Rutin accumulation was significantly lowered in salt susceptible species. High antioxidant (O−2 and H2O2) contents were detected in both types of rutin-reduced transgenic plants under high salt stress. Moreover, exogenous application of rutin effectively enhances the ROS scavenging and ultimately gains the salinity tolerance. The environmental stresses have multiple and complicated effect on plant growth and development. The environmental stresses altered either incrementally or rapidly. Essentially plants recognize and respond differentially to biological signals appropriately with the required pace of survival (Takahashi and Shinozaki 2019). The sophisticated networks of physiological processes in plants arise due to involvement of diverse molecular and cellular level of regulatory mechanisms required for both short- and long-term responses varying from cellular to whole plant level (Rob et al. 2020). It has been accepted that flavonoids also control movements of phytohormones like auxin, and a nuclear allocation of chalcone isomerase (CHI) and chalcone synthase (CHS) is consistent with regulation employed by flavonoid compounds on the genes transcription mediated for development changes, facilitated via the auxin-transporting facilitator proteins (ATFP) (Agati and Tattini 2010).
4 Stress Tolerance Responses by Flavonoids and Carotenoids
4.1 Drought Tolerance
Under the drought conditions, the osmotic and ROS balance are disturbed which stressed the cellular metabolism and structure (Rao et al. 2020). Plants develop array of mechanisms such as compatible solute production, antioxidant production, and other entities to withstand the unavoidable stressed conditions. The concentration of ascorbic acid and α-tocopherol and epigallocatechin gallate (flavonoids) were higher in the Cistus clusii grown under the drought condition (Hernandez et al. 2004). In the A. thaliana seedlings, the total flavonoids and the quercetin, and kaemferol (flavanols) were higher in roots (Shojaie et al. 2016). Similarly, the Jatropha seedlings showed higher concentration of flavonoids under the drought conditions (Lama et al. 2016). Carotenoids have ability to neutralize various forms of ROS and can quench the triplet form of chlorophyll in the leaves (Ramel et al. 2012). Under the drought conditions, the carotenoid levels increased in the olive trees, beans, and alpine plants which were considered to be associated with the xanthophyll cycle activation (Doupis et al. 2013; Buchner et al. 2017; Schweiggert et al. 2017). The increase in the zeaxanthin content was also suggested to play important role in the different abiotic stress tolerance (Wu et al. 2015). However, it has been also reported that under moderate drought conditions, carotenoid content tends to decrease (Sudrajat et al. 2015). Applications of Rhizobium cultures enhance N-supply and fixation process in cowpea, soybean, red-clover, pea, and alfalfa crops. The symbiosis between Bradyrhizobium and Rhizobium sp. effectively supports the nutritional strategies, ensuring an efficient N-supply in legumes, pastures, and grasses (Nagpal et al. 2020; Kumawat et al. 2019). The N-fixation, nodulation number, and growth in Medicago sativa improved with R. meliloti strain along with increased yield under water- deficient environment. In another study, Rhizobium strains effectively enhanced the nodulation rate in M. sativa, V. faba, P. sativum, and P. vulgaris, whereas reports support the effectiveness of plant–microbe interactions to improve drought stress in legumes via degrading phosphorus (Rob et al. 2020).
4.2 Heat or UV Tolerance
UV radiations are naturally imparted on the plants along with the sunlight. To which plants have to sense and respond on the basis of damage caused which depends upon the repair and acclimation response. Flavonoids provide themselves as absorbing cushion for UV radiation nullifying the ROS- induced cellular damages. The activation of photoreceptors induces the transcription factors to trigger the transcription of flavanoid biosynthetic genes. It has been observed that UV-B photoreceptor mutants (uvr8-1) and Arabidopsis wild plant under continuous 4 h of low-dose UV radiation depict the activation of chalcone synthase stimulation (enzyme involved in flavanoid biosynthesis) (Jenkins 2013). In several plant species, the ratio of luteolin to apigenin and quercetin to kaempferol has been enhanced under the UV light (di Ferdinando et al. 2012). The quercetin and luteolin glycosides aid in reducing the ROS production by effectively chelating with Cu and Fe ions (Brown et al. 1998; Berli et al. 2010). In the pepper plant, the compounds for UV-B absorption and antioxidants such as carotenoids have been induced under the UV-B radiation exposure (León-Chan et al. 2017). Similarly, in the tobacco plant, the carotenoid content and antioxidant capacity have been intensified after the low and high UV-B exposure (Shen et al. 2017). Moreover, the antioxidants accumulated in apical epidermal layer of leaves and stems to protect from the injuries from internal tissues against UV-B radiation. In A. thaliana, the kaempferol synthesis is deficient in flavone (chalcone) isomerase mutant shows much sensitivity to UV radiation. The worthy role of flavonoids is not exceptional functional of UV-protection via inducing antioxidant molecules, signal molecules, secretion of defensive compounds, and allelochemicals during exposure of UV radiation in plant. Moreover, flavonoids that accumulate in upper epidermis layer specially absorb a large amount of light ranging from 280 to 340 nm wavelengths.
Xanthophyll cycle was activated during intensive light conditions and helps in biosynthesis of zeaxanthin and proteins. According to Couso et al. (2012), the cytochrome-dependent hydroxylases and carotenes enzymes were evaluated to be up-regulated that control the xanthophyll cycle during light stress. Accumulation of zeaxanthin and antheraxanthin, and corresponding decrease in violaxanthin amounts were correlated with high light intensity resistance. This interconversion of zeaxanthin and violaxanthin is associated to a promising photoprotective role in the photosynthetic machineries indulging in dissipation of excessive light energy when exposed to intensive light stress.
In algal species, carotenoid accumulation was reported to enhance with temperature that activates cellular response or antioxidant mechanism related to enhanced activity of biosynthetic enzyme, free oxygen radical formation, and damaging effects observed at elevated temperatures. The enhanced threshold levels and carotenoid content by two-folds were reported by Juneja et al. (2013) in Haematococcus pluvialis and Chlorococcum sp., with temperature rise from 20 to 30 °C. Dunaliella salina (Halophytic green alga) has been noted to accumulate huge concentration of β-carotene when exposed to excessive water salt, light, and nutrient amounts in extreme conditions (Llorente et al. 2017).
The effect of ultraviolet-B radiation affected the chlorophyll content by degrading them and induced the accumulation carotenoids in Capsicum annuum (León-Chan et al. 2017). Enhanced level of UV-B rapidly elevates carotenoid content compared with control plants and the production of those compounds. The increased carotenoids and ROS at high UV-B radiation were reported in Nicotiana tabacum L. leaves play important role in tolerating the oxidative damage in plant caused by high UV-B conditions (Shen et al. 2017).
4.3 Salinity Tolerance
Excessive accumulation of ROS is primary cause of salinity stress usually resulting in deleterious effects on plants (Kang et al. 2018). Flavonoids and carotenoids play an important protective role against oxidative burst and injuries caused by salt stress. In Lupinus albus seedling, the Chryseobacterium balustinum Aur9 elevated total nitrogen content, root surface, germination rate, aerial growth, N-absorption, and biological N-fixation. It also promoted the systematic induced responses and helped to overcome the salt stress in soybean and Arabidopsis species. Under high salinity condition, strains of Sinorhizobium fredii were used to enhance the nitrogen fixation rate via eliciting the nodule number in significant manner (Rodríguez-Navarro et al. 2002). In Rhizobium tropici, salt-tolerant wild-strain, the increased antioxidant enzymes activities including dehydroascorbate reductase and superoxide dismutase resulted in salinity tolerance and were correlated to enhanced antioxidant enzymes in nodules (Tejera et al. 2004). The various nitrogen-fixing bacteria tolerant to high concentration of NaCl are Rhizobium meliloti, R. leguminosarum, and rhizobia from Vigna unguiculata whereas Rhizobium fredii, R. meliloti, and Sinorhizobium fredii show regulatory mechanism for K+ strictly but control Mg2+ flux under salt stress condition. The R. meliloti derived from xerophytic tree species formed a symbiotic association with M. sativa and confirmed effective salt tolerance (Zahran 1999).
Several metabolic and physiological changes are affected by the salinity stress. On the basis of duration and intensity, the salinity stress can negatively affect the growth and development of plants. According to Zhao and Dixon (2009), a positive correlation was observed between the flavanoid biosynthesis level and the Glutathione-S-transferase content (role in flavonoids transportation to vacuole). Similarly, the biosynthesis of luteolin 7-O-glycosides was significantly enhanced under the salinity conditions (Agati et al. 2011). The seedling of Solanum nigrum, having lower dry biomass of leaves and roots along with the higher flavanoid contents (Abdallah et al. 2016). The accumulation of carotenoids such as β-carotene and lutein in the sweet potatoes were observed under the salinity stress. The IbZDS gene encoding β-Carotene desaturase is related with the carotenoid accumulation for tolerance whose expression induced for carotenoid biosynthesis under salinity condition (Li et al. 2017). In the Solanum nigrum, the expression of phytoene desaturases, zeta carotene desaturases, phytoene synthase, capsanthin/capsorubin synthase, and lycopene b-cyclase was down-regulated under the higher salt concentration in the salt sensitive Capsicum plants (Maurya et al. 2015).
According to Li et al. (2017), the expression of carotenoids can be up-regulated or down-regulated by overexpression or suppression of genes inducing activity of f-Carotene desaturases like enzymes and their biosynthetic pathways. The overexpression of IbZDS (Ipommea batata) gene is associated with ZDS, resulted in significant increases in β-carotene and lutein-related accumulation improved salinity tolerance sweet potatoes. The suppression of lycopene-cyclase (α-LCY) and lycopene β-cyclase (β-LCY) genes by RNA interference resulted in enhanced concentrations of total carotenoids, β-carotene, and ultimately stress tolerance cells of sweet potatoes (Kim et al. 2015). The findings were also supported by Kang et al. (2017) in sweet potatoes, where CHY-b suppression up-regulates the carotenoid level and enhances tolerance to stress. According to Jin et al. (2015), salinity stress tolerance observed to be associated with LCY-β and α-LCY genes in Nicotiana tabacum in which suppression of α-LCY and overexpression of LCY-β play role for enhanced salinity tolerance. The variations reported in species level depend upon the complexity in regulation of carotenoids. However, the transcriptional control regulating genes interaction that is all involved in regulating level of carotenes compounds during stress tolerance. The expression of phytoene synthase 2 and LCYB genes was associated with the enhanced level of lutein and carotene observed in Solanum nigrum. The expression of lycopene β-cyclase, zeta carotene desaturases, phytoene desaturases, phytoene synthase, and capsorubin/capsanthin synthase in capsicum sp. was down-regulated with increase in salinity level (Maurya et al. 2015; Ben Abdallah et al. 2016).
Elevated levels of copper induce the production of ROS and carotenoids formation; in Vicia faba, subjected to elevating the amount of copper were reported to be concentration-dependent. The increased carotenoid levels could be result of enhanced activity of nonenzymatic antioxidant system related to secondary metabolic compounds (Abdel-Latef and Abu-Alhmad 2013). In Cicer arietinum, cadmium (Cd) stress caused significant enhancement in level of carotenoids. The carotenoid synthesis lowered ROS damage induced by cadmium toxicity (Ahmad et al. 2016) whereas, phosphorus (P) or nitrogen (N) repletion was observed to elevate the carotenoid levels in algae and increased levels of thiobarbituric acid (TBA) and H2O2 reactive species along with decreased SOD activity (Moussa et al. 2017). In microalgae, Zhang et al. (2017) studied enhanced carotenoid accumulation in unfavorable conditions. The findings showed that nitrogen depletion facilitated the synthesis of carotenoids, compared to chlorophylls and demonstrated that carotenoids were highly sensitive to N stress. Comparatively declined chlorophyll content was observed with progressed nitrogen stress. These findings showed that the carotenoid content in Chlorella vulgaris varied over time with different salinity environmental conditions (Zhang et al. 2017).
4.4 Chilling Stress
The low temperature can affect the plant growth cycle with each stage from its germination to the maturity. Plants respond differentially based on their genetic potential to tolerate chilling stress. Many plant species either tropical or subtropical (rice, maize, and tobacco) fail to survive under chilling temperature; however, some overwintering cereals and Arabidopsis can continue to survive and grow (Liu et al. 2018). The increase in flavonoid content was noticed with the lowering in temperature reported in the epidermal cells of many plant species (Bilger et al. 2007). In the A. thaliana accessions, the positive correlation was recorded between the cold tolerance and the level of flavonoid content (Korn et al. 2008). Many biosynthetic genes such as chalcone synthase, dihydroflavonol 4-reductase, flavonol synthase, and chalcone isomerase for flavonoids were expressed higher in the cold tolerant plants (having higher anthocynin and quercetin derivative content) than the sensitive ones (Hannah et al. 2006). The higher flavonoids in the fruits of Phoenix dactylifera L. were reported at the 0 °C temperature condition along with the no any kind of chilling injury symptoms observed (El-Rayes 2009). The low temperature resulted in decline in chlorophyll content and signaled to accumulation of carotenoids in the Capsicum annuum (León-Chan et al. 2017).
5 Biotic Stress Tolerance: A Feasible and Efficient Biological Application
The microbial inoculants and their secondary metabolites are proved to be efficient agents and act as biofungicide, bioherbicides, and biopesticides. Microbial inoculants play important role as biocontrol approach could be driven by elevating phosphorus solubilization, improve biological nitrogen fixation, nutrient absorption at rhizospheric region, systemic acquired resistance, and production of synergetic secondary metabolites in low concentration provides toxicity to pathogens. Moreover, the toxic effect could be overcome by production of regenerative hormones (ethylene and auxin) at damage site. Several fungi and microbe inoculation of Aspergillus niger, Penicillium citrinum, Aspergillus fumigatus, Penicillium funiculosum, Penicillium aurantiogriseum, Trichoderma koningii, Phytophthora infestans, Sporolactobacillus inulinus, Amphibacillus xylanus, Bacillus amyloliquefaciens, Microbacterium oleovorans release inhibitory compounds against fungal pathogens (Nesci et al. 2005; Pereira et al. 2007; Etcheverry et al. 2009). Strains of Bacillus spp. secrete inhibitory compounds and used as biological controlling agent in wide range of fungal infections in plants. Bacillus subtilis was used to be control production of aflatoxin and Aspergillus flavus in field condition, whereas Pseudomonads and rhizobia are effectively known to suppress the growth of Fusarium and Pythium sp. (Antoun and Prévost 2005; Cepeda 2012; Alori and Babalola 2018). Moreover, both microbes and plants employed alternative mechanism in form of antibiotics or phytoalexins containing flavonols, carotenoids, alkaloids substance to control the plant pathogens. The compounds show antagonistic effect at rhizosphere and exhibit biocidal properties on soil-borne pathogens. The properties can be altered by formulation, cultivation, and application methods of PGPR under stress responses (Glick 2012, 2015; Raaijmakers and Mazzola 2012; Grobelak et al. 2015; Bhattacharyya et al. 2016; Olanrewaju et al. 2017). Certain classical examples for PGPR producing bactericidal effects are Pseudomonas spp. and Bacillus that have been identified to liberate aerugine, azomycin, bacilysin, butyrolactones, bacillaene, cepaciamide A, cepafungins, chlorotetain, 2,4-diacetyl phloroglucinol, fengycin, tas A, karalicin, phenazine-1-carboxylic acid, rhamnolipids, iturin, pseudomonic subtilin, subtilosin, sublancin, oomycin A, zwittermycin A, kanosamine, and pyrrolnitrin (Alori and Babalola 2018). The total phenols, carotenoids, and anthocyanin content observed to be enhanced in lettuce via exogenous application of inoculation containing Glomus fasciculatum and Azotobacter chroococcum (Baslam et al. 2011). Similarly, flavonoid content was reported to be increased with inoculation of Glomus mosseae and G. fasciculatum in lettuce (Baslam et al. 2011). Arbuscular mycorrhizal fungi (AMF) speed up antioxidant biosynthesis upto 75% significantly through rhizobacterial inoculation in soybean seedlings. The above findings are well supported by the inoculation studies of Bacillus megaterium and Pseudomonas fluorescens in catharanthus under stress conditions (Alori and Babalola 2018).
5.1 Fungal Tolerance
Different bacterial strains of PGPR including Bacillus subtilis, Bacillus licheniformis, Chryseobacterium, Balustinum, and Pseudomonas sp. when combined enhance significant level of resistance against soil-borne diseases in tomato and pepper under in vitro conditions. The genes isolated from Lycium chinensis such as carotene isomerase (LcCRTISO), phytoene desaturase (LcPDS), and β-carotene desaturase (LcZDS) via carotenoid biosynthesis are significantly enhanced making the tobacco plant salinity resistant (Lattanzio et al. 2006; Domenech et al. 2006). Pseudomonas mendocina, Nitrinicola lacisaponensis, Bacillus pumilus, Halomonas sp., and Arthrobacter sp., isolated from salinity conditions showed siderophore, indole acetic acid (IAA), ammonia production, P-solubilization, and other plant growth-effecting traits. Studies on bacterial inoculation of above isolates enhanced presence of flavonoid quercetin and phenolics compounds cinnamic, ferulic, gallic, vanillic, syringic, and caffeic, acids in the rhizosphere soil. The high concentration of phenolics, quercetin, and IAA in rhizosphere soil zone exhibited a cumulative synergistic function that supported elevated salinity stress tolerance in wheat (Venkidasamy et al. 2018; Pathan et al. 2020; Tiwari et al. 2011; Mahmoudi et al. 2019).
5.2 Bacterial Tolerance
Bacillus cereus (strain BS107) against Xanthomonas axonopodis pv. vesicatoria in pepper leaves shows induction of induced systematic response where, ISR-elicited responses of Bacillus spp. was also reported against root-knot nematodes, viral, bacterial, and fungal pathogens. Yang and Yen (2000) reported molecular evidences in favor of priming effect of Rhizobacterium strains on the up-regulation of ISR and defense genes in pepper plant.
5.3 Insect and Nematode Tolerance
The flavonoids and isoflavonoids are lethal to the insect pests by altering the behavior, growth, and development of target insects (War et al. 2013; Melo et al. 2017). The flavones 5-hydroxyisoderricin, 5-methoxyisoronchocarpin, and 7-methoxy-8- (3- methylbutadienyl)-flavanone are present in Tephrosia villosa (L.), T. vogelii, and T. purpurea (L.), respectively, which can be used to repell Spodoptera exempta and S. littoralis (Simmonds et al. 1990). The overexpression of transcriptional factors required for flavonoid production confers the resistance in Arabidopsis against the Spodoptera frugiperda (Johnson and Dowd 2004). The chickpea plant was showing antifeedent activity against the Helicoverpa armigera by mean of maackiain, judaicin, 2-methoxyjudaicin, and judaicin-7-O-glucoside isoflavonoid productions (Simmonds and Stevenson 2001). The alliarinoside, cyanopropenyl glycoside, and isovitexin-600-D-β-glucopyranoside interfere with the feeding activity of Pieris napi oleracea (native American butterfly) (Renwick et al. 2001). The overexpression of CCD1 leads to the β-ionone (carotenoid) in Arabidopsis plant increases the emission of β-ionone to prevent the damage induced by the insect attack (Wei et al. 2010).
6 Recent Advancement in Functional Aspect of Flavonoids and Carotenoids in Crop Improvement
Recently, many reports published on the functional aspect of flavonoids and carotenoids in term of stress tolerance in crop plants (Liu et al. 2015; Kudoyarova et al. 2019). The sulfoquinovosyl transferase enzyme induces glycosylation of flavonoid that elevates tolerance to osmotic stress in rice (Zhan et al. 2019). Application of soil – organic matter enhances the efficacy of secondary metabolites including flavonoids and microbes- based communication (Del Valle et al. 2020). Identification of allelochemicals and phytotoxic activity induce tolerance from Schumannianthus dichotomus observed by Rob et al. (2020). The overexpression of the cytochrome CYP82A3 gene derived from Glycine max improves drought and salinity tolerance and also proves a strong resistance against Botrytis cinerea and Phytophthora parasitica in transgenic tobacco (Nicotiana benthamiana) species (Yan et al. 2016; Tian et al. 2017). Rhizosphere acts as hotspot for allelochemicals, PGPR, and secondary metabolites production based on soil-plant –microbe interaction inducing cycling of nitrogen and carbon in soil (Del Valle et al. 2020). Enhanced level of Arbuscular mycorrhiza (AMF) induces production of root flavonoids, shows colonization in invasive tree species reported by Pei et al. (2020). Screening of tunisian plant extract formulation shows bioherbicide properties and in Cynara cardunculus (Kaab et al. 2020). In sweet potato, enhanced lutein and β-carotene level through suppression or down-regulation of the β-carotene hydroxylase gene and increase abiotic stress tolerance in transgenic stress tolerance (Li et al. 2017). Similarly, findings reported by Kea et al., showed enhanced β-carotene amount, lycopene β-cyclase gene (IbLCYB2), and suppression of lycopene ε-cyclase expression in sweet potatoes (Li et al. 2017; Kang et al. 2018). Multiple stress conditions impose more beneficial impacts on plants compared to that posed in presence of individual stress alone (Liu et al. 2015). Combination of stresses ultimately reduces the detrimental effect of each other thereby, increasing the probability of better survival of plants. The cumulative impact of drought and accumulation of ozone (O3) in plants resulted in better tolerance. The combined affect was attributed to decreased values of stomatal conductance. Elevated concentration of reduced glutathione and ascorbic acid effectively scavenge ROS, thereby causing a considerable drop in the total ROS content. Several mechanisms highlighting the role of microbes in abiotic stress alleviation have been proposed (Kumar et al. 2019; Ke et al. 2019; Khan et al. 2020). Soil-inhabiting microbes belonging to genera Achromobacter, Azospirillum, Variovorax, Bacillus, Enterobacter, Azotobacter, Aeromonas, Klebsiella, and Pseudomonas have been shown to enhance plant growth even under unfavorable environmental conditions (Kaushal and Wani 2016; Sorty et al. 2016) (Fig. 5.2).
7 Conclusion and Future Remarks
Presently, greatest interest is towards new prospective of microbial and their secondary products including carotenoids and flavonoids that can be used in plant stress tolerance and crop improvement under climate change scenario. Looking into application of crop production without compromising yield loss due to severity of biotic and abiotic using microbial-based inoculants become more efficient and effective. Moreover, the PGPR, microbial and plants-based metabolites having both biological as well as ecofriendly approaches in term of sustainable crop improvement, stress management, and promoting soil health are common. Enhanced flavonoids and carotenoids level in plants are significantly relayed to the elevated light-dependant oxidative damages and promoting antioxidant systems in plants. Morphological structures including specialized glandular trichomes and stomatal guard cells may be conveniently noted to investigate the functional-localization interaction of flavonoids in photoprotection systems. It is evident that high concentration of carotenoids and flavonoids in rhizosphere increases bacterial-mediated growth-promoting responses in root exudates and leaves confirmed growth and resistance in plants. These play a central role in various associations of plant–rhizobia, plant–microbe, soil microbes, and arbuscular mycorrhizal symbiosis and boost plant growth via the production of important biological agents. These microbial-based inoculations may lead to the identification of new signaling molecules and decipher the pathways responsible for triggering stress resistance in challenging environment.
References
Abdallah SB, Aung B, Amyot L, Lalin I, Lachaal M, Karray-Bouraoui N, Hannoufa A (2016) Salt stress (NaCl) affects plant growth and branch pathways of carotenoid and flavonoid biosyntheses in Solanum nigrum. Acta Physiol Plant 38:72–84
Abdel-Latef AAH, Abu-Alhmad MF (2013) Strategies of copper tolerance in root and shoot of broad bean (Vicia faba L.). Pak J Agri Sci 50:223–328
Agati G, Tattini M (2010) Multiple functional roles of flavonoids in photoprotection. New Phytol 186(4):786–793
Agati G, Stefano G, Biricolti S, Tattini M (2009) Mesophyll distributionof antioxidant flavonoids in Ligustrum vulgare leaves under contrastingsunlight irradiance. Ann Bot 104:853–861
Agati G, Biricolti S, Guidi L, Ferrini F, Fini A, Tattini M (2011) The biosynthesis of flavonoids is enhanced similarly by UV radiation and root zone salinity in L. vulgare leaves. J Plant Physiol 168:204–212
Ahmad P, Latef AAA, Abdallah EF, Hashem A, Sarwat M, Anjum NA, Gucel S (2016) Calcium and potassium supplementation enhanced growth, osmolyte secondary metabolite production, and enzymatic antioxidant machinery in cadmium-exposed chickpea (Cicer arietinum L.). Front Plant Sci 7:513
Akcin A, Yalcin E (2016) Effect of salinity stress on chlorophyll, carotenoid content, and proline in Salicornia prostrata Pall. and Suaeda prostrata Pall. subsp. prostrata (Amaranthaceae). Braz J Bot 39:101–106
Alori ET, Babalola (2018) Microbial inoculants for improving crop quality and human health in Africa. Front Microbiol 9:1–12
Antoun H, Prévost D (2005) Ecology of plant growth-promoting rhizobacteria PGPR. In: Siddiqui ZA (ed) Biocontrol and biofertilization. Springer, Berlin, pp 1–38. https://doi.org/10.1007/1-4020-4152-7_1
Barry KM, Davies NW, Mohammed CL (2002) Effect of season and different fungi on phenolics in response to xylem wounding and inoculation in Eucalyptus nitens. For Pathol 32:163–178
Baslam M, Garmendia I, Goicoechea N (2011) Arbuscular mycorrhizal fungi (AMF) improved growth and nutritional quality of greenhouse-grown lettuce. J Agric Food Chem 59:5504–5515
Ben Abdallah S, Aung B, Amyot L, Lalin I, Lachâal M, Karray-Bouraoui N, Hannoufa A (2016) Salt stress (NaCl) affects plant growth and branch pathways of carotenoid and flavonoid biosyntheses in Solanum nigrum. Acta Physiol Plant 38:72
Bennett RN, Wallsgrove RM (1994) Secondary metabolites in plant defense mechanisms. Tansley review no. 72. New Phytol 127:618
Berli FJ, Moreno D, Piccoli P, Hespanhol-Viana L, Silva MF, Bressan-Smith R, Cavagnaro JB, Bottini R (2010) Abscisic acid is involved in the response of grape (Vitis vinifera L.) cv. Malbec leaf tissues to ultraviolet-B radiation by enhancing ultraviolet-absorbing compounds, antioxidant enzymes and membrane sterols. Plant Cell Environ 33:1–10
Bhattacharyya PN, Goswami MP, Bhattacharyya LH (2016) Perspective of beneficial microbes in agriculture under changing climatic scenario: a review. J Phytology 8:26–41. https://doi.org/10.19071/jp.2016.v8.3022
Bhutia LP, Chakraborty BN, Chakraborty U (2012) Management of charcoal stump rot disease using AMF and PGPR in Temi Tea Estate. Sikkim J Mycol Plant Pathol 42(1):1–12
Bilger W, Rolland M, Nybakken L (2007) UV screening in higher plants induced by low temperature in the absence of UV-B radiation. Photochem Photobiol Sci 6:190–195
Boué SM, Carter CH, Ehrlich KC, Cleveland TE (2000) Induction of the soybean phytoalexins coumestrol and glyceollin by Aspergillus. J Agric Food Chem 48:2167–2172
Brown JE, Khodr H, Hider RC, Rice-Evans CA (1998) Structural dependence of flavonoid interactions with Cu2+ ions: implication for their antioxidant properties. Biochem J 330:1173–1178
Buchner O, Roach T, Gertzen J, Schenk S, Karadar M, Stöggl W, Kranner I (2017) Drought affects the heat-hardening capacity of alpine plants as indicated by changes in xanthophyll cycle pigments, singlet oxygen scavenging, α-tocopherol and plant hormones. Environ Exp Bot 133:159–175
Cepeda MV (2012) Effects of microbial inoculants on biocontrol and plant growth promotion. Plant Pathology. Master of Science, Ohio State University, Columbus, OH, p 102
Cesari A, Paulucci N, López-Gómez M, Hidalgo-Castellanos J, Plá CL, Dardanelli MS (2019) Restrictive water condition modifies the root exudates composition during peanut-PGPR interaction and conditions early events, reversing the negative effects on plant growth. Plant Physiol Biochem 142:519–527
Chi WC, Chen YA, Hsiung YC et al (2013) Autotoxicity mechanism of Oryza sativa: transcriptome response in rice roots exposed to ferulic acid. BMC Genomics 14:351. https://doi.org/10.1186/1471-2164-14-351
Clayton WA, Albert NW, Thrimawithana AH, McGhie TK, Deroles SC, Schwinn KE, Davies KM (2018) UVR8-mediated induction of flavonoid biosynthesis for UVB tolerance is conserved between the liverwort Marchantia polymorpha and flowering plants. Plant J 96(3):503–517
Couso I, Vila M, Vigara J, Cordero B, Vargas M, Rodríguez H, León R (2012) Synthesis of carotenoids and regulation of the carotenoid biosynthesis pathway in response to high light stress in the unicellular microalga Chlamydomonas reinhardtii. Eur J Phycol 47:223–232
Dashti NH, Smith DL, Cherian VM (2014) PGPR to alleviate the stress of suboptimal root zone temperature on leguminous plant growth. In: Use of microbes for the alleviation of soil stresses, vol 1. Springer, New York, NY, pp 111–137
de Klerk GJ, Guan H, Huisman P, Marinova S (2011) Effects of phenolic compounds on adventitious root formation and oxidative decarboxylation of applied indoleacetic acid in Malus ‘Jork 9’. Plant Growth Regul 63:175–185
Del Valle I, Webster TM, Cheng HY, Thies JE, Kessler A, Miller MK, Ball ZT, MacKenzie KR, Masiello CA, Silberg JJ et al (2020) Soil organic matter attenuates the efficacy of flavonoid-based plant-microbe communication. Sci Adv 6:eaax8254
Deng F, Aoki M, Yogo Y (2004) Effect of naringenin on the growth and lignin biosynthesis of gramineous plants. Weed Biol Manag 4:49–55
di Ferdinando M, Brunetti C, Fini A, Tattini M (2012) Flavonoids as antioxidants in plants under abiotic stresses. In: Ahmad P, Prasad MNV (eds) Abiotic stress responses in plants: metabolism, productivity and sustainability. Springer, pp 159–179
Domenech J, Reddy MS, Kloepper JW, Ramos B, Gutierrez-Mañero J (2006) Combined application of the biological product LS213 with Bacillus, Pseudomonas or Chryseobacterium for growth promotion and biological control of soil-borne diseases in pepper and tomato. BioControl 51:245–258
Doupis G, Bertaki M, Psarras G, Kasapakis I, Chartzoulakis K (2013) Water relations, physiological behavior and antioxidant defence mechanism of olive plants subjected to different irrigation regimes. Scient Horticul 153:150–156
El-Rayes DA (2009) Effect of carbon dioxide-enriched atmosphere during cold storage on limiting antioxidant losses and maintaining quality of ‘Barhy’ date fruits. J Meteorol Environ Arid Land Agric Sci 20(1):3–22
Enebe MC, Babalola OO (2018) The influence of plant growth-promoting rhizobacteria in plant tolerance to abiotic stress: a survival strategy. Appl Microbiol Biotechnol 102(18):7821–7835
Etcheverry MG, Scandolara A, Nesci A, Vilas B, Ribeiro MS, Pereira P et al (2009) Biological interactions to select biocontrol agents against toxigenic strains of Aspergillus flavus and Fusarium verticillioides from maize. Mycopathologia 167:287–295. https://doi.org/10.1007/s11046-008-9177-1.
Farrar K, Bryant D, Cope-Selby N (2014) Understanding and engineering beneficial plant–microbe interactions: plant growth promotion in energy crops. Plant Biotechnol J 12:1193–1206
Glick BR (2012) Plant growth-promoting bacteria: mechanisms and applications. Scientifica. https://doi.org/10.6064/2012/963401
Glick BR (2015) Resource acquisition- beneficial plant-bacterial interactions. Springer, New York, NY, pp 29–63. https://doi.org/10.1007/978-3-319-13921-0
Gonçalves AC, Bento C, Jesus F, Alves G, Silva LR (2018) Sweet cherry phenolic compounds: identification, characterization, and health benefits. In: Studies in natural products chemistry, vol 59. Elsevier, pp 31–78
Grayson M (2013) Agriculture and drought. Nature 501:S1. https://doi.org/10.1038/501S1a
Grobelak A, Napora A, Kacprzak M (2015) Using plant growth-promoting rhizobacteria (PGPR) to improve plant growth. Ecol Engr 84:22–28. https://doi.org/10.1016/j.ecoleng.2015.07.019
Hamayun M, Hussain A, Khan SA, Kim HY, Khan AL, Waqas M, Lee IJ (2017) Gibberellins producing endophytic fungus Porostereum spadiceum AGH786 rescues growth of salt affected soybean. Front Micro 8:686
Hannah MA, Weise D, Freund S, Fiehn O, Heyer AG, Hincha DK (2006) Natural genetic variation of freezing tolerance in Arabidopsis. Plant Physiol 142:98–112
Hernandez I, Alegre L, Munne-Bosch S (2004) Drought-induced changes in flavonoids and other low- molecular weight antioxidants in Cistus clusii grown under Mediterranean field conditions. Tree Physiol 24:1303–1311
Ingle K, Padole D (2019) Secondary metabolites for plant growth promotion and plant protection. Adv Life Sci 5(23):10888–10891
Jenkins GI (2013) Photomorphogenic responses of plants to UV-B radiation. Am Soc Photobiol. http://photobiology.info/Jenkins.html.
Jin C, Ji J, Zhao Q, Ma R, Guan C, Wang G (2015) Characterization of lycopene b-cyclase gene from Lycium chinense conferring salt tolerance by increasing carotenoids synthesis and oxidative stress resistance in tobacco. Mol Breed 35:228
Johnson ET, Dowd PF (2004) Differentially enhanced insect resistance, at a cost, in Arabidopsis thaliana constitutively expressing a transcription factor of defensive metabolites. J Agric Food Chem 52:5135–5138
Juneja A, Ceballos R, Murthy G (2013) Effects of environmental factors and nutrient availability on the biochemical composition of algae for biofuels production: a review. Energies 6:4607–4638
Kaab SB, Rebey IB, Hanafi M, Hammi KM, Smaoui A, Fauconnier ML, De Clerck C, Jijakli MH, Ksouri R (2020) Screening of Tunisian plant extracts for herbicidal activity and formulation of a bioherbicide based on Cynara cardunculus. S Afr J Bot 128:67–76
Kang L, Ji CY, Kim SH, Ke Q, Park SC, Kim HS, Lee HU, Lee JS, Park WS, Ahn MJ, Lee HS, Deng X, Kwak SS (2017) Suppression of the b-carotene hydroxylase gene increases b-carotene content and tolerance to abiotic stress in transgenic sweet potato plants. Plant Physiol Biochem 117:24–33
Kang C, Zhai H, Xue L, Zhao N, He S, Liu Q (2018) A lycopene β-cyclase gene, IbLCYB2, enhances carotenoid contents and abiotic stress tolerance in transgenic sweet potato. Plant Sci 272:243–254
Kaushal M, Wani SP (2016) Plant growth-promoting rhizobacteria: drought stress alleviators to ameliorate crop production in drylands. Ann Microbiol 66:35–42
Ke Q, Kang L, Kim HS, Xie T, Liu C, Ji CY, Kwak SS (2019) Down-regulation of lycopene ε-cyclase expression in transgenic sweet potato plants increases the carotenoid content and tolerance to abiotic stress. Plant Sci 281:52–60
Khan N, Bano A, Ali S, Babar MA (2020) Crosstalk amongst phytohormones from planta and PGPR under biotic and abiotic stresses. Plant Growth Regul 90:189–203
Kim HJ, Park WS, Bae JY, Kang SY, Yang MH, Lee S, Ahn MJ (2015) Variations in the carotenoid and anthocyanin contents of Korean cultural varieties and home-processed sweet potatoes. J Food Compos Anal 41:188–193
Korn M, Peterek S, Petermock H, Heyer AG, Hincha DK (2008) Heterosis in the freezing tolerance, and sugar and flavonoid contents of crosses between Arabidopsis thaliana accessions of widely varying freezing tolerance. Plant Cell Environ 31:313–327
Kousar B, Bano A, Khan N (2020) PGPR modulation of secondary metabolites in tomato infested with Spodoptera litura. Agron 10(6):778
Kudoyarova G, Arkhipova TN, Korshunova T, Bakaeva M, Loginov O, Dodd IC (2019) Phytohormone mediation of interactions between plants and non-symbiotic growth-promoting bacteria under edaphic stresses. Front Plant Sci 10:1368
Kumar A, Patel JS, Meena VS, Ramteke PW (2019) Plant growth-promoting rhizobacteria: strategies to improve abiotic stresses under sustainable agriculture. J Plant Nutr 42:1402–1415
Kumawat KC, Sharma P, Sirari A, Singh I, Gill BS, Singh U, Saharan K (2019) Synergism of Pseudomonas aeruginosa (LSE-2) nodule endophyte with Bradyrhizobium sp. (LSBR-3) for improving plant growth, nutrient acquisition and soil health in soybean. W J Microbiol Biotechnol 35:1–17
Lama AD, Kim J, Martiskainen O, Klemola T, Salminen JP, Tyystjarvi E, Niemeka P, Vuorisalo T (2016) Impacts of simulated drought stress and artificial damage on concentrations of flavonoids in Jatropha curcas (L.), a biofuel shrub. J Plant Res 129:1141–1150
Lattanzio V, Lattanzio VM, Cardinali A (2006) Role of phenolics in the resistance mechanisms of plants against fungal pathogens and insects. Phytochem Adv Res 661:23–67
León-Chan R, López-Meyer M, Osuna-Enciso T, Sañudo-Barajas J, Heredia J, León-Félix J (2017) Low temperature and ultraviolet-B radiation affect chlorophyll content and induce the accumulation of UV-B-absorbing and antioxidant compounds in bell pepper (Capsicum annuum) plants. Environ Exp Bot 139:143–151
Li R, Kang C, Song X, Yu L, Liu D, He S, Liu Q (2017) A ζ-carotene desaturase gene, IbZDS, increases β-carotene and lutein contents and enhances salt tolerance in transgenic sweet potato. Plant Sci 262:39–51
Liu RQ, Xu XJ, Wang S, Shan CJ (2015) Lanthanum improves salt tolerance of maize seedlings. Photosynthetica 54:148–151
Liu X, Zhou Y, Xiao J, Bao F (2018) Effects of chilling on the structure, function and development of chloroplasts. Front Plant Sci 9:1715
Llorente B, Martinez-Garcia J, Stange C, Rodriguez-Concepcion M (2017) Illuminating colors: regulation of carotenoid biosynthesis and accumulation by light. Curr Opin Plant Biol 37:49–55
Luan Y, Cui J, Zhai J, Li J, Han L, Meng J (2015) High-throughput sequencing reveals differential expression of miRNAs in tomato inoculated with Phytophthora infestans. Planta 241:1405–1416
Mahmoudi TR, Yu JM, Liu S, Pierson IIILS, Pierson EA (2019) Drought-stress tolerance in wheat seedlings conferred by phenazine-producing rhizobacteria. Front Microbiol 10:1590
Maurya KV, Srinvasan R, Ramesh N, Anbalagan M, Gothandam KM (2015) Expression of carotenoid pathway genes in three capsicum varieties under salt stress. Asian J Crop Sci 7:286–294
Meena KK, Sorty AM, Bitla UM, Choudhary K, Gupta P, Pareek A, Singh DP, Prabha R, Sahu PK, Gupta VK, Singh HB, Krishanani KK, Minhas PS (2017) Abiotic stress responses and microbe-mediated mitigation in plants: the omics strategies. Front Plant Sci 8:172
Mekawy AMM, Abdelaziz MN, Ueda A (2018) Apigenin pretreatment enhances growth and salinity tolerance of rice seedlings. Plant Physiol Biochem 130:94–104. https://doi.org/10.1016/j.plaphy.2018.06.036
Melo HF, de Souza ER, Duarte HHF, Cunha JC, Santos HRB (2017) Gas exchange and photosynthetic pigments in bell pepper irrigated with saline water. Revist Brasil Engen Agrícola Ambient 21:38–43
Mizuno H, Yazawa T, Kasuga S, Sawada Y, Ogata J, Ando T, Kanamori H, Yonemaru JI, Wu J, Hirai MY, Matsumoto T (2014) Expression level of a flavonoid 3′-hydroxylase gene determines pathogen-induced color variation in sorghum. BMC Res Notes 7(1):1–12
Moussa ID, Chtourou H, Karray F, Sayadi S, Dhouib A (2017) Nitrogen or phosphorus repletion strategies for enhancing lipid or carotenoid production from Tetraselmis marina. Biorese Technol 238:325–332
Nabavi SM, Samec D, Tomczyk M, Milella L, Russo D, Habtemariam S, Suntar I, Rastrelli L, Daglia M, Xiao J et al (2020) Flavonoid biosynthetic pathways in plants: versatile targets for metabolic engineering. Biotechnol Adv 38:107316
Nagpal S, Sharma P, Sirari A, Gupta RK (2020) Coordination of Mesorhizobium sp. and endophytic bacteria as elicitor of biocontrol against Fusarium wilt in chickpea. Eur J Plant Pathol. https://doi.org/10.1007/s10658-020-02062-1
Nesci A, Bluma R, Etcheverry M (2005) In vitro selection of maize rhizobacteria to study potential biological control of Aspergillus section flavi and aflatoxin production. Eur J Plant Pathol 113:1–13. https://doi.org/10.1007/s10658-005-5548-3.
Olanrewaju OO, Glick BR, Babalola OO (2017) Mechanisms of action of plant growth-promoting bacteria. World J Microbiol Biotechnol 33:197. https://doi.org/10.1007/s11274-017-2364-9
Paliwal C, Mitra M, Bhayani K, Bharadwaj S, Ghosh T, Dubey S, Mishra S (2017) Abiotic stresses as tools for metabolites in microalgae. Bioresour Technol 244:1216–1226
Passari AK, Mishra VK, Singh G, Singh P, Kumar B, Gupta VK, Singh BP (2017) Insights into the functionality of endophytic actinobacteria with a focus on their biosynthetic potential and secondary metabolites production. Sci Rep 7(1):1–17
Pathan SI, Ceccherini MT, Sunseri F, Lupini A (2020) Rhizosphere as hotspot for plant-soil-microbe interaction. In: Carbon and nitrogen cycling in soil. Springer, Berlin/Heidelberg, pp 17–43
Peer WA, Murphy AS (2006) Flavonoids as signal molecules. In: Grotewold E (ed) The science of flavonoids. Springer, New York, NY, pp 239–267
Pei Y, Siemann E, Tian B, Ding J (2020) Root flavonoids are related to enhanced AMF colonization of an invasive tree. AoB Plants 12(1):plaa002
Pereira P, Nesci A, Etcheverry M (2007) Effects of biocontrol agents on Fusarium verticillioides count and fumonisin content in the maize agroecosystem: impact on rhizospheric bacterial and fungal groups. Biol Control 42:281–287. https://doi.org/10.1016/j.biocontrol.2007.05.015
Purohit S, Laloraya MM, Bharti S (1991) Effect of phenolic compounds on abscisic acid-induced stomatal movement: structure—activity relationship. Physiol Plant 81:79–82. https://doi.org/10.1111/j.1399-3054.1991.tb01716.x
Raaijmakers JM, Mazzola M (2012) Diversity and natural functions of antibiotics produced by beneficial and plant pathogenic bacteria. Annu Rev Phytopathol 50:403–424. https://doi.org/10.1146/annurev-phyto-081211-172908
Ramel F, Birtic S, Cuine S, Triantaphylides C, Ravanat JL, Havaux M (2012) Chemical quenching of singlet oxygen by carotenoids in plants. Plant Physiol 158:1267–1278
Rao MJ, Xu Y, Tang X, Huang Y, Liu J, Deng X, Xu Q (2020) CsCYT75B1, a citrus cytochrome P450 gene, is involved in accumulation of antioxidant flavonoids and induces drought tolerance in transgenic Arabidopsis. Antioxidants 9(2):161
Rashid MI, Fareed MI, Rashid H, Aziz H, Ehsan N, Khalid S, Ghaffar I, Ali R, Gul A, Hakeem KR (2019) Flavonoids and their biological secrets. In: Plant and human health, vol 2. Springer, Cham, pp 579–605
Renwick JAA, Zhang W, Haribal M, Attygalle AB, Lopez KD (2001) Dual chemical barriers protect a plant against different larval stages of an insect. J Chem Ecol 27:1575–1583
Rob MM, Hossen K, Iwasaki A, Suenaga K, Kato-Noguchi H (2020) Phytotoxic activity and identification of phytotoxic substances from schumannianthus dichotomus. Plan Theory 9:102
Rodríguez-Navarro DN, Bellogín R, Camacho M, Daza A, Medina C, Ollero FJ, Santamaría C, Ruíz-Saínz JE, Vinardell JM, Temprano FJ (2002) Field assessment and genetic stability of Sinorhizobium fredii strain SMH12 for commercial soybean inoculants. Eur J Agron 19:299–309
Samanta A, Das G, Das SK (2011) Roles of flavonoids in plants. Carbon 100(6):12–35
Savvides A, Ali S, Tester M, Fotopoulos V (2016) Chemical priming of plants against multiple abiotic stresses: mission possible? Trends Plant Sci 21(4):329–340
Scervino JM, Ponce MA, Erra-Bassells R et al (2005) Flavonoids exhibit fungal species and genus specific effects on the presymbiotic growth of Gigaspora and Glomus. Mycol Res 109(7):789–794
Schweiggert RM, Ziegler JU, Metwali EM, Mohamed FH, Almaghrabi OA, Kadasa NM, Carle R (2017) Carotenoids in mature green and ripe red fruits of tomato (Solanum lycopersicum L.) grown under different levels of irrigation. Arch Biol Sci 69(2):305–314
Shen J, Jiang C, Yan Y, Liu B, Zu C (2017) Effect of increased UV-B radiation on carotenoid accumulation and total antioxidant capacity in tobacco (Nicotiana tabacum L.) leaves. Genet Mol Res 16(1):1–11
Shojaie B, Mostajerani A, Mustafa Ghannadian M (2016) Flavonoid dynamic responses to different drought conditions: amount, type, and localization of flavonols in roots and shoots of Arabidopsis thaliana L. Turk J Biol 40:612–622
Shukla S, Gupta S (2010) Apigenin: a promising molecule for cancer prevention. Pharm Res 27:962–978
Simmonds MSJ, Stevenson PC (2001) Effects of isoflavonoids from Cicer on larvae of Helicoverpa armigera. J Chem Ecol 27:965–977
Simmonds MSJ, Blaney WM, Fellows LE (1990) Behavioural and electrophysiological study of antifeedant mechanisms associated with polyhydroxyalkaloids. J Chem Ecol 16:3167–3196
Sorty AM, Meena KK, Choudhary K, Bitla UM, Minhas PS, Krishnani KK (2016) Effect of plant growth-promoting bacteria associated with halophytic weed (Psoralea corylifolia L.) on germination and seedling growth of wheat under saline conditions. Appl Biochem Biotechnol 180:872–882
Sudrajat DJ, Siregar IZ, Khumaida N, Siregar UJ, Mansur I (2015) Adaptability of white jabon (Anthocephalus cadamba MIQ.) seedling from 12 populations to drought and water logging. Agri 37:130–143
Tabatabaei S, Ehsanzadeh P (2016) Photosynthetic pigments, ionic and antioxidative behaviour of hulled tetraploid wheat in response to NaCl. Photosynthetica 54:340–350
Taïbi K, Taïbi F, Abderrahim LA, Ennajah A, Belkhodja M, Mulet JM (2016) Effect of salt stress on growth, chlorophyll content, lipid peroxidation and antioxidant defence systems in Phaseolus vulgaris L. South Afr J Bot 105:306–312
Takahashi F, Shinozaki K (2019) Long-distance signaling in plant stress response. Curr Opin Plant Biol 47:106–111
Tejera NA, Campos R, Sanjuán J, Lluch C (2004) Nitrogenase and antioxidant enzyme activities in Phaseolus vulgaris nodules formed by Rhizobium tropici isogenic strains with varying tolerance to salt stress. J Plant Physiol 161:329–338
Tian F, Wang W, Liang C, Wang X, Wang G, Wang W (2017) Over accumulation of glycine betaine makes the function of the thylakoid membrane better in wheat under salt stress. Crop J 5:73–82
Tiwari S, Singh P, Tiwari R, Meena KK, Yandigeri M, Singh DP, Arora DK (2011) Salt-tolerant rhizobacteria-mediated induced tolerance in wheat (Triticum aestivum) and chemical diversity in rhizosphere enhance plant growth. Biol Fertil Soils 47:907–916
Venkidasamy B, Rajendran V, Sathishkumar R (2018) Flavonoids (antioxidants systems) in higher plants and their response to stresses. Springer International Publishing AG Antioxidants and Antioxidant Enzymes in Higher. Plan Theory 12:253–268
War AR, Paulraj MG, Hussain B, Buhroo AA, Ignacimuthu S, Sharma HC (2013) Effect of plant secondary metabolites on Helicoverpa armigera. J Pest Sci 86:399–408
Wei J, Xu M, Zhang D, Mi H (2010) The role of carotenoid isomerase in maintenance of photosynthetic oxygen evolution in rice plant. Acta Biochim Biophys Sin 42:457–463
Wu J, Ji J, Wang G, Wu G, Diao J, Li Z, Chen X, Chen Y, Luo L (2015) Ecotopic expression of the Lyciumbarbarum b-carotene hydroxylase gene (chyb) enhances drought and salt stress resistance by increasing xanthophyll cycle pool in tobacco. Plant Cell Tissue Organ Cult 121:559–569
Yan Q, Cui X, Lin S, Gan S, Xing H, Dou D (2016) GmCYP82A3, a soybean cytochrome P450 family gene involved in the jasmonic acid and ethylene signaling pathway, enhances plant resistance to biotic and abiotic stresses. PLoS One 11:e0162253
Yang J, Yen HE (2000) Early salt stress effects on the changes in chemical composition in leaves of ice plant and Arabidopsis- a fourier transform infrared spectroscopy study. Plant Physiol 130:1032–1042
Yu O, Jung W, Shi J, Croes RA, Fader GM, McGonigle B, Odell JT (2000) Production of the isoflavones genistein and daidzein in non-legume dicot and monocot tissues. Plant Physiol 124(2):781–794
Zahran HH (1999) Rhizobium-legume symbiosis and nitrogen fixation under severe conditions and in an arid climate. Micro Mol Biol Rev 63(4):968–989
Zhan X, Shen Q, Chen J, Yang P, Wang X, Hong Y (2019) Rice sulfoquinovosyltransferase SQD2. 1 mediates flavonoid glycosylation and enhances tolerance to osmotic stress. Plant Cell Environ 42(7):2215–2230
Zhang P, Li Z, Lu L, Xiao Y, Liu J, Guo J, Fang F (2017) Effects of stepwise nitrogen depletion on carotenoid content, fluorescence parameters and the cellular stoichiometry of Chlorella vulgaris. Spectrochim Acta Part a Mol Biomol Spectr 181:30–38
Zhao J, Dixon RA (2009) The ‘ins’ and ‘outs’ of flavonoid transport. Trend Plant Sci 14:72–80
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Kumar, A., Nagpal, S., Choudhary, A. (2021). Carotenoids and Flavonoids in Plant Stress Management. In: Singh, H.B., Vaishnav, A., Sayyed, R. (eds) Antioxidants in Plant-Microbe Interaction. Springer, Singapore. https://doi.org/10.1007/978-981-16-1350-0_5
Download citation
DOI: https://doi.org/10.1007/978-981-16-1350-0_5
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-1349-4
Online ISBN: 978-981-16-1350-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)