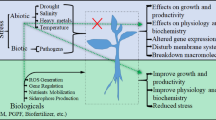
Abstract
Plants are constantly subjected to biotic and abiotic stress factors, from their planting time up to the harvesting, transport, storage and consumption of plant products. These stresses exert deleterious harmful effects on crop health as well as cause huge losses to their production worldwide. To combat these stress factors, researchers all around the globe are involved in procuring management practices ranging from traditional genetics and breeding techniques to present day available novel biotechnological tools. Use of microorganisms is one such method by which both abiotic and biotic stress can be tackled in an economical, ecofriendly and successful manner. Plant growth-promoting rhizobacteria (PGPR) are the bacteria living in rhizosphere region and promoting plant growth and suppressing stress components as well. Different microorganisms acquire different mechanisms to fight with these plant stresses. In this chapter, an effort has been made to impart the knowledge about the abiotic and biotic stress factors, their management in an efficient and novel way.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
6.1 Introduction
Stress is a physiological condition caused by factors that affect the equilibrium process (Gaspar et al. 2002). The pliability of normal processes develops reaction to the environmental fluctuations that can be predicted over daily and seasonal cycles, which means every change in a component from its normal range is not likely to cause stress. Stress affects the normal metabolic processes resulting in injury, disease or physiological changes. Plants are influenced by different environmental stresses like drought, low temperature, salt, flooding, heat, oxidative stress and heavy metal toxicity during their cultivation (Jaleel et al. 2009).
Agriculture is one of the highly unprotected sectors to climate deviation. Enhanced affect of abiotic and biotic stresses has evolved as an important cause for static crop production. There is considerable evidence of yield reductions of wheat and paddy in many regions of South Asia due to enhanced water stress, decreased number of rainy days and increased air temperature. The average temperature has increased by 0.57 °C in the last 100 years in the Indian sub-continent and it is expected to rise to a maximum of 2.5 °C by 2050 and 5.8 °C by 2100. Also the irrigation requirement in arid and semi-arid areas is estimated to rise by 10 % with every 1 °C increase in temperature. Besides high temperature, droughts, high CO2, increased rainfall, floods, cold and heat waves, and cyclones are the natural calamities that result in economic depletion and are consequences of global warming. These factors affect crops quantitatively and qualitatively and also put critical pressure on land and water resources (Grover et al. 2011).
Stresses caused by various environmental factors including light, UV, temperature extremes, freezing, drought, salinity, heavy metals and hypoxia result in substantial crop losses worldwide (Boyer 1982; Mahaian and Tuteja 2005; Mittler 2006). These abiotic stresses might increase in the near future owing to the global climate change. Plant growth and development is affected by the various environmental factors (Wahid et al. 2012). Abiotic stresses, including temperature, extremes salinity and drought, are serious intimidation to the sustainability and productivity of economic plants. Current climatic model predicts that global air temperature may increase by 1.1–6.4 °C with doubling of atmospheric CO2 (Kim et al. 2007; Lobell and Field 2007).
Around the world, abiotic and biotic stresses are largely affecting crop productivity. Due to imbalance in environmental conditions, stresses like drought, rains, floods, heat waves and frost damages can increase in future. To combat with these stresses wide range of modification plans are required. By well-planned use of available resources and crop improvement practices for producing better varieties, we can fight with abiotic stresses up to some level. But such strategies are time consuming and costly. We should formulate simple, effective and low-cost biological methods for managing abiotic stresses. Microorganisms possess qualities like endurance to extreme conditions, ubiquity, genetic diversity, relationship with plants and thus can play a pivotal role in this aspect. Through various modes of action like induction of osmoprotectants and heat shock proteins etc. in plant cells, microbes can affect plants’ response to abiotic stresses. Use of these microorganisms can diminish plant stresses and they can also be used as important models for becoming aware of stress tolerance, adaptation and response mechanisms that can be transferred into plants to combat with climate change because of plant stresses (Grover et al. 2011).
Plants exposed to various climatic factors, in order to sustain, have developed different mechanisms (Rejeb et al. 2014). Physiological changes in plants are due to exposure to many types of biotic and abiotic stresses (Heil et al. 2002, Swarbrick et al. 2006; Bolton 2009; Massad et al. 2012) which finally causes reduction in plant yield (Shao et al. 2008). Abiotic stress effects plant health and causes heavy losses. Biotic stress means harmful effects due to pathogen infection in plants (Strauss and Zangerl 2002; Maron and Crone 2006; Maron and Kauffman 2006; Mordecai 2011). Growth stage of plants (Zhang et al. 2013) and climatic factors (Liu et al. 2008) play an important role in plant’s reaction to abiotic and biotic stresses. Depending on the nature of abiotic stress and pathogen, defense mechanism gets altered in plants. Moreover, signaling compounds are increased when plants are exposed to both abiotic and biotic stress simultaneously, e.g. cross-tolerance.
6.2 Plant Stress
Plants’ sensitivity towards abiotic and biotic stresses causes yield loss and plants devise many kinds of modifications to adapt in stressed conditions (Rejeb et al. 2014).
6.2.1 Abiotic Stresses in Plants
Plants’ exposure to abiotic factors results in abiotic stresses reducing crop productivity (Heil and Bostock 2002) but it also affects ecological distribution of plants (Chaves et al. 2003). Abiotic stress examples are fluctuations in water, temperature, soil nutrients, toxic substances, light and soil texture (Versulues et al. 2006). Intergovernmental Panel on Climate Change 2012 (IPCC 2012) has predicted that various abiotic stresses like temperature extremes, drought, floods, climatic conditions and land-decline can cause huge losses in agriculture sector in many parts of developing countries (Field et al. 2012).
Among the various environmental conditions, cold, drought and salinity are most severely affecting plants resulting in heavy economic losses (Beck et al. 2007). Primary and secondary stresses are the result of primary and secondary damages; for example secondary stress and damage caused by ROS (reactive oxygen species, Allen 1995) is the consequence of electron transport rate fluctuations and the metabolic consumer activity of the reductive power. Similarly, the secondary stress occurs from primary stressors such as cold or excess of light energy (Huner et al. 1998).
The impact of natural and man-made issues (Eitzinger et al. 2010) can be seen in the form of average global temperature increase by 2–4 °C at the last of twenty-first century (IPCC 2007). One of the important causes of this temperature rise is the release of green house gases (GHG) (Maraseni et al. 2009; Smith and Olesen 2010). Due to this temperature fluctuation, various crops at different developmental stages are exposed to heat stress (Watanabe and Kume 2009).
6.2.2 Biotic Stresses in Plants
Apart from abiotic stress factors, plants are exposed to many kinds of pathogens including fungi, bacteria, viruses and nematodes and herbivores (Atkinson and Urwin 2012). The environmental conditions are likely to affect the habitual place of pests and pathogens. For instance, dispersal of pathogens is increased due to temperature extremes (Bale et al. 2002; Luck et al. 2011; Madgwick et al. 2011; Nicol et al. 2011). It is also reported that abiotic stress factors decrease the defense potential of plants and induce proneness to pathogen attack (Amtmann et al. 2008; Goel et al. 2008; Mittler and Blumwald 2010; Atkinson and Urwin 2012). In coming times, it is estimated that both abiotic and biotic stresses alone and in combination will attack crop plants with more power (Suzuki et al. 2014).
Biotic stress is a result of damage caused to the plants by other living organisms including bacteria, viruses, fungi, parasites, beneficial and harmful insects and weeds. Plants are under constant assault by biotic agents, including viral, bacterial and fungal pathogens, parasitic plants and insect herbivores, with enormous economic and ecological impact (Pimentel 2002). Biotic stress affects plant population dynamics and ecosystem nutrient cycling as well. Fungi, insects, viruses, bacteria and parasitic weeds can cause enormous loss to crop production (Mehta et al. 2012). The impact of aerial fungal diseases on crop yield differs with time and cropping areas. Rusts, downy mildews and powdery mildews are the major foliar diseases that have deleterious effects on crop production. For instance, species belonging to rust fungi can infect grains, e.g. Puccinia species, like Puccinia graminis on wheat rust, P. sorghi on maize and forage legumes; Uromyces species, like U. appendiculatus on common bean, lentil and U. vignae on cowpea. Different methods can be used for managing this disease as resistance sources are not available (Ramteke et al. 2004).
Root rot, caused by Aphanomyces euteiches, Rhizoctonia solani, Fusarium solani and wilt, caused by many formae speciales of Fusarium oxysporum are the most critical soil-borne diseases in pea, chickpea, lentil, fababean and lupin (Infantino et al. 2006). Damping-off, usually caused by either Rhizoctonia solani or Pythium spp., can cause about 80 % of plant demise (Wang et al. 2003). Fusarium root-rot (caused by Fusarium spp.) can too result in rigorous seedling fatalities particularly in tomato and lentils (Hamwieh et al. 2005). The production of tomato and lentil (Bayaa 1997) is majorly effected by Fusarium wilt (caused by F. oxysporum) where leaf chlorosis, wilting and death occurs at seedling and adult stage of plants. Similarly, southern stem rot (Sclerotium rolfsii) and white mold (Sclerotinia sclerotiorum) can result in seedling and pod rots in warm and cool climate respectively (Kolkman and Kelly 2003).
The co-evolution of plants and the pathogens results in development of defense mechanism in plants. Whenever plants are attacked by pathogens they have to balance between their developmental and defense requirements (Zangerl and Berenbaum 2003; Berger et al. 2007). With respect to food security, worldwide research focus is required to develop crops that can give sustainable yields along with the capability to survive harsh abiotic (Duque et al. 2013) as well as biotic stress situations.
6.3 Practices to Mitigate Plant Stresses
Diverse biotic and abiotic stresses are responsible for the badly affected production and yield of a number of crops. Massive financial fatalities are accountable globally due to these stresses. As biotic and abiotic stresses are affecting agriculture adversely, there is need to develop plants that can tolerate stress with high yields. For stress tolerant plant production, presently tissue culture based in vitro selection has been developed as an economic and effective method. Various substances like NaCl (for salt tolerance), PEG or mannitol (for drought tolerance) and pathogen culture filtrate, phytotoxin or pathogen itself (for disease resistance) are used in culture media for making stress tolerant plants. Stimulation of genetic distinction between cells, tissues or organs in cultured and regenerated plants is needed for in vitro selection. The selection of somaclonal variations appearing in the regenerated plants may be genetically established and useful in crop improvement. To endure under strain circumstances plants have developed numerous biochemical and molecular mechanisms such as ROS (reaction oxygen species) creation and elimination in plants (Rai et al. 2011).
Key for crop improvement is conventional breeding technologies and appropriate management practices. To stimulate stress tolerance in plants, traditional breeding programs are used to incorporate good genes of interest from inter crossing genera and species into the crops (Purohit et al. 1998).
6.3.1 Management Strategies to Reduce Abiotic Stress
Plants react to temperature changes at cellular, tissue and organ levels. The main survival responses to high temperature stress are photosynthetic acclimation to heat stress, production and buildup of primary and secondary metabolites, generation of stress proteins. Heat shock protein (hsp) genes, dehydrins (dhn), senescence-associated (sag) genes, stay green (sgr) genes are expressed in reaction to heat stress. Plants exhibit various adaptations like preservation of membrane strength, scavenging of ROS, production of enzymatic and non-enzymatic antioxidants and amendment of companionable solutes against heat stress. Mass screening and morphological and biochemical markers-assisted selection, recognition, and mapping of QTLs conferring heat resistance, conventional and molecular breeding, and exogenous use of osmoprotectants and stress-signaling agents can be used for heat tolerance in plants (Wahid et al. 2012). To overcome pH stress, it is significant to alter the nutrient accessibility as well as the soil properties to modify the pH of the soil. For example, pH of soil can be neutralized by addition of lime (calcium or magnesium carbonate) (Mehta et al. 2012).
In plants, drought stress causes changes like leaf size decrease, stems expansion and root propagation, disturbs plant water relations and reduces water-use effectiveness. CO2 assimilation by leaves is decreased by closing of stomata, membrane spoilage and disturbed action of enzymes like those of CO2 fixation and adenosine triphosphate synthesis. Plants exhibit a variety of mechanisms to endure drought stress, such as shortened water loss by amplified diffusive resistance, improved water uptake with plentiful and deep root systems and its efficient use, and smaller and tender leaves to lessen the transpirational loss. Nutrients are also helpful in this aspect, like potassium ions in osmotic regulation, silicon for improved root endodermal silicification and cell water equilibrium enhancement. Plant growth regulators like salicylic acid, auxins, gibberrellins, cytokinin and abscisic acid can also adjust the plant reaction towards drought. Enzymes like polyamines, citrulline behave as antioxidants and lessen the undesirable effects of water scarcity. Drought-responsive genes and transcription factors like dehydration-responsive element-binding gene, aquaporin, late embryogenesis abundant proteins and dehydrins have been reported. Mass screening and breeding, marker-assisted selection and exogenous application of hormones and osmoprotectants to seed or plants are the methods for overcoming the problem of drought stress (Farooq et al. 2009).
6.3.2 Management Strategies to Reduce Biotic Stress
In biological control, antagonistic microbes are employed to improve plant healthiness. Persistent demonstration of connections amongst the plant, the pathogen, the biocontrol agent, the microbial population on and in the region of the plant, and the physical surroundings is exhibited through disease inhibition by biocontrol agents. The use of biocontrol agents such as bacteria viz., Pseudomonas and Bacillus and the fungi Trichoderma symbolize an array of existence approaches and means of disease inhibition.
To diminish the biotic stress, biotechnological advances are also used. Many molecular marker-related methods have been used for managing biotic stresses like Random Amplified Polymorphic DNA (RAPD), Restriction Fragment Length Polymorphism (RFLP), Amplified Fragment Length Polymorphism (AFLP), and Simple Sequence Repeat (SSR). Because of these, genetic maps for several species were recognized in which impending resistance and/or tolerance loci or QTLs have been located. This also provides knowledge about the number, chromosomal location and individual or interactive effects of the QTLs involved that strengthens the genetic management of specific resistance and/or tolerance in many crops. These areas of expertise have recognized precise molecular markers, which may possibly be used in breeding plan through Marker-Assisted Selection (MAS) to augment biotic stress tolerance. Diers (2004) used the MAS for the breeding of resistant soybean to cyst nematode and similar markers have also been used by Mutlu et al. (2005), Yang et al. (2002) and Yang et al. (2004) for the resistance of pinto bean to common bacterial blight, resistant of narrow-leafed lupin (Lupinus angustifolius L.) to phomopsis stem blight and anthracnose. Besides, the gene pyramiding strategy aided by MAS can be a proficient technique when resistance is bestowed by single gene and/or easily conquered by novel pathogen races (Mehta et al. 2012).
Plant growth-promoting bacteria (PGPB) can encourage plant growth either directly or indirectly. Inhibition of plant disease (bioprotection), better nutrient accessibility (biofertilization), or construction of phytohormones (biostimulation) are numerous diverse strategies for promoting plant production (Saharan and Nehra 2011).
Directly these bacteria can regulate functioning of plants by mimicking production of plant hormones or those that make minerals and nitrogen further obtainable in the soil, e.g. the leguminous symbionts Rhizobium (Hirsch and Kapulnik 1998; Saharan and Nehra 2011). The siderophore production or volatiles (2, 3-butanediol and acetoin) or different antibiotic compounds, or induction of plant-mediated induced systemic resistance (ISR) are the indirect proponent of plant growth (Saharan and Nehra 2011).
6.3.3 Role and Mechanism of Microbes to Reduce/Conquer the Stress
Productivity of agricultural crops as well as the microbial activity in soil is being hampered by these stresses. The change in climatic conditions such as prolonged drought, intense rains, flooding, high temperatures, frost and low temperatures, which are expected to escalate in future, will significantly affect plants and soil microorganisms. The different stress factors have a significant influence over the performance of microorganisms. Mycorrhizal and/or endophytic fungi can interact with many plant species and thereby significantly contribute to the adaptation of these plants to a number of environmental stresses (Rodriguez et al. 2008). These conditions include drought, heat, pathogens, herbivores, or limiting nutrients.
Extensive research has been carried out on occurrence and functional diversity of agriculturally important microbes in stressed environments as reviewed by several authors (Grahm 1992; Venkateswarlu et al. 2008). The occurrence of Rhizobium, Bradyrhizobium, Azotobacter, Azospirillum, Pseudomonas and Bacillus has been reported from desert ecosystems, acid soils, saline and alkaline areas and highly eroded hill slopes of India (Tilak et al. 2005; Selvakumar et al. 2009; Upadhyay et al. 2009). Microorganisms could play an important role in adaptation strategies and increase of tolerance to abiotic stresses in agricultural plants. The impact of abiotic stresses (drought, low temperature, salinity, metal toxicity, and high temperatures) on plants can be minimized through the production of exopolysaccharates and biofilm formation by plant growth-promoting rhizobacteria (PGPR) which remain associated with plant roots. Different mechanisms like induction of osmoprotectors and heat shock proteins are mediated through their rhizospheric microorganisms when plants are exposed to stress conditions.
A variety of mechanisms have been proposed behind microbial elicited stress tolerance in plants (Table 6.1). The production of indole acetic acid, gibberellins and some unknown determinants by PGPR helps to increase the root length, root surface area number of root tips, leading to enhanced uptake of nutrients resulting in improved plant health under stress conditions (Egamberdieva and Kucharova 2009). In addition to this, PGPRs also help to enhance plant growth under saline conditions (Glick et al. 1997; Yildirim and Taylor 2005; Barassi et al. 2006).
The synthesis of cytokinin and antioxidants by the strains of PGPR can cause the building up of abscissic acid (ABA) and decomposition of reactive oxygen species (ROS). Oxidative stress tolerance has been found associated with the enhanced level of antioxidant enzymes (Stajner et al. 1997). There is effect of ethylene on different processes of plants and ethylene synthesis in plants is dependent on environmental factors and on various biotic and abiotic stresses (Hardoim et al. 2008). In the biosynthetic pathway of ethylene, S-adenosylmethionine (S-AdoMet) is converted by 1-aminocyclopropane-1-carboxylate synthase (ACS) to 1-aminocyclopropane-1-carboxylate (ACC), the immediate precursor of ethylene. The plant hormone such as ethylene which endogenously regulates plant homeostasis under stress conditions results in reduced root and shoots growth. Plants supplemented with nitrogen and energy are also prevented from harmful ethylene effect, improved plant stress due to the degeneration of ACC by ACC deaminase enzyme produced from bacterial cells (Glick 2007). Saleem et al. (2007) have reviewed the role of PGPR containing ACC deaminase, in stress agriculture. Inoculation with ACC deaminase containing bacteria induces longer roots which might be helpful in the uptake of relatively more water from deep soil under drought stress conditions, thus increasing water-use efficiency of the plants under drought conditions (Zahir et al. 2008).
The volatiles emitted by PGPR, down-regulate hkt1 (High Affinity K+ Transporter 1) expression in roots but upregulates it in shoots, orchestring lower Na+ levels and recirculation of Na+ in the whole plant under salt conditions (Zhang et al. 2008). By synthesis of the metabolite 2R, 3R-butanediol, the inoculation of Pseudomonas chlororaphis O6 in Arabidopsis thaliana roots resulted in increased abiotic and biotic stress tolerance. Studies with Arabidopsis mutant lines indicated that induced drought tolerance requires salicylic acid (SA), ethylene and jasmonic acid-signaling pathways (Cho et al. 2008).
Arbuscular mycorrhizal (AM) fungi alleviate the effects of drought and salinity stresses through osmoregulation and proline accumulation. AM symbiosis plays an important role in increasing the plant resistance against water deficit and drought stress through the alteration of plant physiology and the expression of plant genes (Subramanian and Charest 1998; Ruiz-Lozano and Azcon 2000). There are reports of AM-induced increases in drought tolerance, involving both increased dehydration and dehydration tolerance (Allen and Boosalis 1983). The role of abscissic acid (ABA) had been suggested behind AM-mediated stress response of plants (Aroca et al. 2008). In non-AM plants, it was observed that ABA content in the shoots increased as well as there was more expression of certain stress marker genes by the use of external source of ABA. However in AM plants such use of exogenic ABA reduced the ABA content in their shoots and did not result in increased expression of stress genes. Co-inoculation of lettuce with PGPR Pseudomonas mendocina and G. intraradices or G. mosseae augmented an antioxidative catalase under severe drought conditions, suggesting that they could be used in inoculants to alleviate the oxidative damage (Kohler et al. 2008).
6.4 Advantages of Microbes over Other Practices
A group of beneficial microbes has been reported by the various/different researchers from different agro ecosystem in the past. Some of these microbes are playing an important role in stimulating the plant growth and increasing the crop yields during adverse environmental conditions. Plant growth-promoting bacteria (PGPB) are able to promote the plant growth, production and nutrient availability through various mechanisms. For example, certain bacteria can cause elevation of plant growth by increasing nutrient uptake from soil or by production of some substances similar to plant hormones. The PGPR can affect plant growth and development in direct, indirect or collective manner (Joseph et al. 2007; Yasmin et al. 2007). For instance, few PGPR are known to alleviate growth of Arabidopsis thaliana by exudation of compounds like 2, 3-butanediol and acetoin (Ryu et al. 2003). The inoculation of diazotroph bacteria in cotton resulted in promotion of the seed cotton yield, plant height and population of soil microorganisms (Anjum et al. 2007). Similarly in apple, it has been found that the strength and quality of rooting is increased due to collective use of IBA, bacteria and carbohydrates (Karakurt et al. 2009).
Many bacteria present in rhizoplane are able to utilize root exudates efficiently. Increased fertilizer use efficiency and lower fertilizer rates can be achieved by using PGPRs alone or in combination with AMF (Adesemoye et al. 2009). In rice, increased growth was observed with the inoculation of PGPR isolates (Ashrafuzzaman et al. 2009). In chickpea also, better development and production occurred due to the use of PGPRs as biofertilizers (Rokhzadi et al. 2008). There are two different kinds (direct and indirect) of effect of PGPR on plant growth. Directly PGPR can make available their synthesized products to the plant or they can help plants in taking up nutrients (Glick 1995). Indirectly PGPR can reduce or block the attack of harmful plant pathogens and thus enhance the growth of plants. Bacteria like Pseudomonas fluorescens and P. putida produce siderophores, which bind iron and facilitate its transport from the environment into the microbial cell (Fig. 6.1).
Direct and indirect (plant-mediated) effects of rhizosphere- and plant-associated organisms on interactions between Pseudomonas fluorescens and host plant defenses (Adopted from Hol et al. 2013)
Rice (Oryza sativa) is one of the important crops grown globally and specially in Asian continent as noted by Kumar et al. (2011). For the proper growth and development of rice, there is more need of nitrogen (Sahrawat 2000). In Vietnam, rise in growth rate and production of rice was observed with the use of PGPR-based commercial product BioGro (Nguyen et al. 2003; Nguyen 2008). Similarly in India, the commercial PGPR formulation Ecomonas was found to decrease the incidence of rice sheath blight caused by the fungus Rhizoctonia solani over the control treatment by 37.7 % and a significant increase in yield was also noticed. In chickpea an increase in plant height, dry weight, number of pods and nutrient content was reported by the inoculation of vesicular arbuscular mycorrhizal fungi (Glomus mosseae, G. fasciculatum, Acaulospora laevis and Gigaspora gilmorei) in India (Kumar et al. 2009).
In another study, to access the role of PGPRs on nutrient uptake two rhizospheric Pseudomonas spp. were taken and their bioassociative effect with root nodulating symbiotic nitrogen fixer Rhizobium leguminosarum-PR1 on plant growth and nutrients uptake by lentil (Lens culinaris L.), was studied under greenhouse conditions. In Pseudomonas treated plants, more vigorous vegetative growth with increase in nodulation, leg-hemoglobin content, physiologically available iron, total iron, chlorophyll content, P uptake and N uptake was observed. Co-inoculation of Pseudomonas with R. leguminosarum recorded maximum increase in the nodulation, leg-hemoglobin content, total iron, total chlorophyll content, N uptake and P uptake over the plants treated with R. leguminosarum alone suggesting a strong synergistic relationship between Pseudomonas sp. and R. leguminosarum (Mishra et al. 2011).
In another experiment, Methylobacterium oryzae and three AMF were evaluated for nutrient uptake on red pepper (Capsicum annum L.). The co-inoculation of M. oryzae and AMF significantly increased various plant growth parameters like root and shoot length, fresh and dry weight and chlorophyll content compared to uninoculated controls. Also nitrogen and phosphorus content of the plants increased; in addition, Zn, Cu, Fe and Mn content of the inoculated plants also increased by almost 1.5 times that of uninoculated control in most of the inoculation treatments. The results obtained suggest that apart from affecting plant growth and nutrient uptake individually, microorganisms can also form mutualistic relationships thereby benefiting the plant (Kim et al. 2010).
Therefore in natural systems, plant pathogens co-exist with host plants and other microorganisms; also biological control entails any reduction in the incidence and severity of the pathogen achieved through any biological mechanism.
6.5 Conclusion
Successful management of plant stress requires a complex range of interactions. Understanding these interactlions between plants and microbes through different molecular and biochemical techniques will improve their stress management mechanism. Application of genetic analysis to microorganisms involved in stress management has resulted in significant advancement in understanding the microbial metabolites and regulatory genes involved in stress management. Ecological analyses have begun to describe the responses of microbial communities towards introduction of biocontrol agents. The integrated use of genetic, molecular and ecological approaches will form the basis for significant future advances in stress management research.
The development of stress tolerant crop varieties is a time-consuming effort, while microbial inoculation to manage stresses in plants could be a more economical and ecofriendly alternative which would be available in shorter time duration. In the future intensive research is required on field evaluation and application of potential microorganisms. Increasing concerns over environmental issues gives microbial biocontrol an exciting perspective. Therefore, by the application of naturally occurring soil microbes instead of deleterious chemicals can give a very promising substitute for plant stress management.
References
Adesemoye A, Torbert H, Kloepper J (2009) Plant growth-promoting Rhizobacteria allow reduced application rates of chemical fertilizers. Microb Ecol 58(4):921–929
Ait Bakra E, Nowak J, Clement C (2006) Enhancement of chillingesistance of inoculated grapevine plantlets with a plant growth promoting rhizobacterium, Burkholderia phytofirmans strain PsJN. Appl Environ Microbiol 72(11):7246–7252
Alami Y, Achouak W, Marol C, Heulin T (2000) Rhizosphere soil aggregation and plant growth promotion of sunflowers by exopolysaccharide producing Rhizobium sp. strain isolated from sunflower roots. Appl Environ Microbiol 66:3393–3398
Ali SZ, Sandhya V, Grover M, Kishore N, Rao LV, Venkateswarlu B (2009) Pseudomonas sp. strain AKM-P6 enhances tolerance of sorghum seedlings to elevated temperatures. Biol Fert Soils 46:45–55
Allen MF, Boosalis MG (1983) Effects of two species of VA mycorrhizal fungi on drought tolerance of winter wheat. New Phytol 93:67–76
Allen R (1995) Dissection of oxidative stress tolerance using transgenic plants. Plant Physiol 107:1049–1054
Amellal N, Burtin G, Bartoli F, Heulin T (1998) Colonization of wheat rhizosphere by EPS producing Pantoea agglomerans and its effect on soil aggregation. Appl Environ Microbiol 64:3740–3747
Amtmann A, Troufflard S, Armengaud P (2008) The effect of potassium nutrition on pest and disease resistance in plants. Physiol Plant 133:682–691
Anjum MA, Sajjad MR, Akhtar N, Qureshi MA, Iqbal A, Jami AR, Hasan M (2007) Response of cotton to plant growth promoting Rhizobacteria (PGPR) inoculation under different levels of nitrogen. J Agric Res 45(2):135–143
Aroca R, Vernieri P, Ruiz-Lozano JM (2008) Mycorrhizal and non- mycorrhizal Lactuca sativa plants exhibit contrasting responses to exogenous ABA during drought stress and recovery. J Exp Bot 59(8):2029–2041
Arshad M, Sharoona B, Mahmood T (2008) Inoculation with Pseudomonas spp. containing ACC deaminase partially eliminate the effects of drought stress on growth, yield and ripening of pea (Pisum sativum L.). Pedosphere 18:611–620
Ashraf M, Berge SH, Mahmood OT (2004) Inoculating wheat seedling with exopolysaccharide-producing bacteria restricts sodium uptake and stimulates plant growth under salt stress. Biol Fert Soils 40:157–162
Ashrafuzzaman M, Hossen FA, Ismail MR, Hoque MA, Islam MZ, Shahidullah SM, Meon S (2009) Efficiency of plant growthpromoting Rhizobacteria (PGPR) for the enhancement of rice growth. Afr J Biotechnol 8(7):1247–1252
Atkinson NJ, Urwin PE (2012) The interaction of plant biotic and abiotic stresses: from genes to the field. J Exp Bot 63:3523–3543
Bale JS, Masters GJ, Hodkinson ID, Awmack C, Bezemer TM, Brown VK, Butterfield J, Buse A, Coulson JC, Farrar J (2002) Herbivory in global climate change research: direct effects of rising temperature on insect herbivores. Glob Chang Biol 8:1–16
Barassi CA, Ayrault G, Creus CM, Sueldo RJ, Sobero MT (2006) Seed inoulation with Azospirillum mitigates NaCl effects on lettuce. Sci Hortic (Amsterdam) 109:8–14
Bayaa B (1997) Screening lentil for resistance to Fusarium wilt: methodology and sources of resistance. Euphytica 98:69–74
Beck EH, Fettig S, Knake C, Hartig K, Bhattarai T (2007) Specific and unspecific responses of plants to cold and drought stress. J Biosci 32:501–510
Berger S, Sinha AK, Roitsch T (2007) Plant physiology meets phytopathology: plantprimary metabolism and plant-pathogen Interactions. J Exp Bot 58:4019–4026
Bolton MV (2009) Primary metabolism and plant defense-fuel for the fire. Mol Plant Microbe Interact 22:487–497
Boyer JS (1982) Plant productivity and environment. Science 218:443–448
Chang WS, van de Mortel M, Nielsen L, de Guzman GN, Li X, Halverson LJ (2007) Alginate production by Pseudomonas putida creates a hydrated microenvironment and contributes to biofilm architecture and stress tolerance under water-limiting conditions. J Bacteriol 189:8290–8299
Chaves MM, Maroco JP, Pereira JS (2003) Understanding plant responses to drought-from genes to the whole plant. Funct Plant Biol 30:239–264
Cho K, Toler H, Lee J, Ownley B, Stutz JC, Moore JL, Auge RM (2006) Mycorrhizal symbiosis and response of sorghum plants to combined drought and salinity stresses. J Plant Physiol 163:517–528
Cho SM, Kang BR, Han SH, Anderson AJ, Park J-Y, Lee Y-H, Cho BH, Yang K-Y, Ryu C-M, Kim YC (2008) 2R,3R-butanediol, a bacterial volatile produced by Pseudomonas chlororaphis O6, is involved in induction of systemic tolerance to drought in Arabidopsis thaliana. Mol Plant Microbe Interact 21:1067–1075
Creus CM, Sueldo RJ, Barassi CA (2004) Water relations and yield in Azospirillum-inoculated wheat exposed to drought in the field. Can J Bot 82(2):273–281
Diers B (2004) Soybean genetic improvement through conventional and molecular based strategies In: 5th European conference on grain legumes, Djion, France, 7–11 June 2004, AEP, pp 147–148
Dodd IC, Belimov AA, Sobeih WY, Safronova VI, Grierson D, Davies WJ (2005) Will modifying plant ethylene status improve plant productivity in water-limited environments? 4th international crop science congress. http://www.cropscience.org.au/
Duque AS, Almeida AM, Bernardes da Silva A, Marques da Silva J, Farinha AP, Santos D, Fevereiro P, Araújo SS (2013) Abiotic stress responses in plants: unraveling the complexity of genes and networks to survive. In: Kourosh Vahdati K, Leslie C (eds) Abiotic stress—plant responses and applications in agriculture. InTech, Rijeka, pp 49–101
Egamberdieva D, Kucharova Z (2009) Selection for root colonizing bacteria stimulating wheat growth in saline soils. Biol Fert Soils 45:563–571
Egamberdiyeva D (2007) The effect of plant growth promoting bacteria on growth and nutrient uptake of maize in two different soils. Appl Soil Ecol 36:184–189
Eitzinger J, Orlandini S, Stefanski R, Naylor REL (2010) Climate change and agriculture: introductory editorial. J Agric Sci Cambridge 148:499–500
Farooq M, Wahid A, Kobayashi N, Fujita D, Basra SMA (2009) Plant drought stress: effects, mechanisms and management. Agron Sustain Dev 29:185–212
Field CB, Barros V, Stocker TF, Qin D, Dokken DJ, Ebi KL, Mastrandrea MD, Mach KJ, Plattner G-K, Allen SK, Tignor M, Midgley PM (2012) In: Intergovernmental Panel on Climate Change (IPCC) (ed) Managing the risks of extreme events and disasters to advance climate change adaptation. Cambridge University Press, Cambridge
Figueiredo MVB, Burity HA, Martinez CR, Chanway CP (2008) Alleviation of drought stress in common bean (Phaseolus vulgaris L.) by co-inoculation with Paenibacillus polymyxa and Rhizobium tropici. Appl Soil Ecol 40:182–188
Fougnies L, Renciot S, Muller F, Plenchette C, Prin Y, de Faria SM, Bouvet JM, Sylla SND, Dreyfus B, Bâ AM (2007) Arbuscular mycorrhizal colonization and nodulation improve tolerance in Pterocarpus officinalis Jacq. seedlings. Mycorrhiza 17:159–166
Gaspar T, Franck T, Bisbis B, Kevers C, Jouve L, Hausman JF, Dommes J (2002) Concepts in plant stress physiology. Application to plant tissue cultures. Plant Growth Regul 37:263–285
Glick BR (1995) The enhancement of plant growth by free-living bacteria. Can J Microbiol 41:109–117
Glick BR (2007) Promotion of plant growth by bacterial ACC deaminase. Crit Rev Plant Sci 26:227–242
Glick BR, Liu C, Ghosh S, Dumbrof EB (1997) Early development of canola seedlings in the presence of plant growth promoting rhizobacterium Pseudomonas putida GR 12–2. Soil Biol Biochem 29:1233–1239
Goel AK, Lundberg D, Torres MA, Matthews R, Akimoto-Tomiyama C, Farmer L, Dangl JL, Grant SR (2008) The Pseudomonas syringae type III effector HopAM1 enhances virulence on water-stressed plants. Mol Plant Microbe Interact 21:361–370
Grahm PH (1992) Stress tolerance in Rhizobium, Bradyrhizobium and nodulation under adverse soil conditions. Can J Microbiol 38:475–484
Grichko VP, Glick BR (2001) Amelioration of flooding stress by ACC deaminase containing plant growth promoting bacteria. Can J Microbiol 47:77–80
Grover M, Ali SZ, Sandhya V, Rasul A, Venkateswarlu B (2011) Role of microorganisms in adaptation of agriculture crops to abiotic stresses. World J Microbiol Biotechnol 27:1231–1240
Gupta DK, Rai UN, Sinha S, Tripathi RD, Nautiyal BD, Rai P, Inouhe M (2004) Role of Rhizobium (CA-1) inoculation in increasing growth and metal accumulation in Cicer arietinum L. growing under fly-ash stress condition. Bull Environ Contam Toxicol 73:424–431
Hamdia ABE, Shaddad MAK, Doaa MM (2004) Mechanisms of salt tolerance and interactive effects of Azospirillum brasilense inoculation on maize cultivars grown under salt stress conditions. Plant Growth Regul 44:165–174
Hamwieh A, Udupa SM, Choumane W, Sarkar A, Dreyer F, Jung C, Baum M (2005) A genetic linkage map of lentil based on microsatellite and AFLP markers and localization of fusarium vascular wilt resistance. Theor Appl Genet 110:669–677
Hardoim PR, van Overbeek SV, van Elsas JD (2008) Properties of bacterial endophytes and their proposed role in plant growth. Trends Microbiol 16:463–471
Heil M, Bostock RM (2002) Induced systemic resistance (ISR) against pathogens in the context of induced plant defences. Ann Bot 89:503–512
Hirsch AM, Kapulnik Y (1998) Signal transduction pathways in mycorrhizal associations: comparisons with the rhizobium–legume symbiosis. Fungal Genet Biol 23(3):205–212
Hol WHG, Bezemer TM, Biere A (2013) Getting the ecology into interactions between plants and the plant growth-promoting bacterium Pseudomonas fluorescens. Front Plant Sci 4:81
Huner NPA, Öquist G, Sarhan F (1998) Energy balance and acclimation to light and cold. TIPS 3:224–230
Infantino A, Kharrat M, Riccioni L, Coyne CJ, McPhee KE, Grünwald NJ (2006) Screening techniques and sources of resistance to root diseases in cool season food legumes. Euphytica 147:201–221
IPCC (Intergovernmental Panel on Climate Change) (2007) Climate change and its impacts in the near and long term under different scenarios. In: The Core Writing Team, Pachauri RK, Reisinger A (eds) Climate change 2007: synthesis report. IPCC, Geneva, pp 43–54
IPCC (Intergovernmental Panel on Climate Change) (2012) Managing the risks of extreme events and disasters to advance climate change adaptation. In: Field CB, Barros V, Stocker TF, Qin D, Dokken DJ, Ebi KL, Mastrandrea MD, Mach KJ, Plattner GK, Allen SK, Tignor M, Midgley PM. Cambridge University Press, Cambridge
Jaleel CA, Manivannan P, Wahid A, Farooq M, Somasundaram R, Panneerselvam R (2009) Drought stress in plants: a review on morphological characteristics and pigments composition. Int J Agric Biol 11:100–105
Joseph B, Patra RR, Lawrence R (2007) Characterization of plant growth promoting Rhizobacteria associated with chickpea (Cicer arietinum L). Int J Plant Prod 1(2):141–152
Karakurt H, Aslantas R, Ozkan G, Guleryuz M (2009) Effects of indol–3-butyric acid (IBA), plant growth promoting rhizobacteria (PGPR) and carbohydrates on rooting of hardwood cutting of MM106 Apple rootstock. Afr J Agric Res 4(2):060–064
Kim SH, Dennis CG, Richard CS, Jeffrey TB, Dennis JT, Vangimalla RR (2007) Temperature dependence of growth, development and photosynthesis in maize under elevated CO2. Environ Exp Bot 61:224–236
Kim K, Yim W, Trivedi P, Madhaiyan M, Boruah HPD, Islam MR, Lee G, Sa T (2010) Synergistic effects of inoculating arbuscular mycorrhizal fungi and Methylobacterium oryzae strains on growth and nutrient uptake of red pepper (Capsicum annuum L.). Plant Soil 327:429–440
Kohler J et al (2008) Plant growth promoting rhizobacteria and arbuscular mycorrhizal fungi modify alleviation biochemical mechanisms in water stressed plants. Funct Plant Biol 35:141–151
Kolkman JM, Kelly JD (2003) QTL conferring resistance and avoidance to white mold in common bean. Crop Sci 43:539–548
Kumar VK, Raju SK, Reddy MS, Kloepper JW, Lawrence KS, Groth DE, Miller ME, Sudini H, Binghai D (2009) Evaluation of commercially available PGPR for control of rice sheath blight caused by Rhizoctonia solani. J Pure Appl Microbiol 3(2):485–488
Kumar VK, Reddy MS, Kloepper JW, Lawrence KS, Zhou XG, Groth DE, Zhang S, Sudhakara Rao R, Wang Q, Raju MRB, Krishnam R, Dilantha Fernando WG, Sudini H, Du B, Miller ME (2011) Commercial potential of microbial inoculants for sheath blight management and yield enhancement of rice. In: Maheshwari DK (ed) Bacteria in agrobiology: crop ecosystems. Springer, Berlin, pp 237–264
Liu C, Ruan Y, Lin Z, Wei R, Peng Q, Guan C, Ishii H (2008) Antagonism between acibenzolar-S-methyl-induced systemic acquired resistance and jasmonic acid-induced systemic acquired susceptibility to Colletotrichum orbiculare infection in cucumber. Physiol Mol Plant Pathol 72:141–145
Lobell DB, Field CB (2007) Global scale climate-crop yield relationships and the impacts of recent warming. Environ Res Lett 2:1–7
Luck J, Spackman M, Freeman A, Tre˛bicki P, Griffiths W, Finlay K, Chakraborty S (2011) Climate change and diseases of food crops. Plant Pathol 60:113–121
Madgwick J, West J, White R, Semenov M, Townsend J, Turner J, Fitt BL (2011) Impacts of climate change on wheat anthesis and Fusarium ear blight in the UK. Eur J Plant Pathol 130:117–131
Madhaiyan M, Poonguzhali S, Sa T (2007) Metal tolerating methylotrophic bacteria reduces nickel and cadmium toxicity and promotes plant growth of tomato (Lycopersicon esculentum L.). Chemosphere 69:220–228
Mahaian S, Tuteja N (2005) Cold, salinity and drought stresses: an overview. Arch Biochem Biophys 444:139–158
Maraseni TN, Mushtaq S, Maroulis J (2009) Greenhouse gas emissions from rice farming inputs: a cross-country assessment. J Agric Sci Cambridge 147:117–126
Maron JL, Crone E (2006) Herbivory: effects on plant abundance, distribution and population growth. Proc R Soc B 273(2):575–2584
Maron JL, Kauffman M (2006) Habitat-specific consumer impacts on plant population dynamics. Ecology 87:113–124
Marquez LM, Redman RS, Rodriguez RJ, Roosinck MJ (2007) A virus in a fungus in a plant: three-way symbiosis required for thermal tolerance. Science 315(5811):513–515
Marulanda A, Porcel R, Barea JM, Azcon R (2007) Drought tolerance and antioxidant activities in lavender plants colonized by native drought tolerant or drought sensitive Glomus species. Microb Ecol 54(3):543–552
Massad TJ, Dyer LA, Vega CG (2012) Cost of defense and a test of the carbon-nutrient balance and growth-differentiation balance hypotheses for two co-occurring classes of plant defense. PLoS One 7, e7554
Mayak S, Tirosh T, Glick BR (2004a) Plant growth promoting bacteria that confer resistance to water stress in tomato and pepper. Plant Sci 166:525–530
Mayak S, Tirosh T, Glick BR (2004b) Plant growth-promoting bacteria confer resistance in tomato plants to salt stress. Plant Physiol Biochem 42:565–572
McLellan CA, Turbyville TJ, Wijeratne K, Kerschen A, Vierling E, Queitsch C, Whiteshell L, Gunatilaka AAL (2007) A rhizosphere fungus enhances Arabidopsis thermotolerance through production of an HSP90 inhibitor. Plant Physiol 145:174–182
Mehta CM, Gupta V, Singh S, Srivastava R, Sen E, Romantschuk M, Sharma AK (2012) Role of microbiologically rich compost in reducing biotic and abiotic stresses. In: Satyanarayana T, Johri BN, Prakash A (eds) Microorganisms in environmental management. Springer, Dordrecht, pp 113–134
Mishra PK, Bisht SC, Ruwari P, Joshi GK, Singh G, Bisht JK, Bhatt JC (2011) Bioassociative effect of cold tolerant Pseudomonas spp. and Rhizobium leguminosarum-PR1 on iron acquisition, nutrient uptake and growth of lentil (Lens culinaris L.). Eur J Soil Biol 47:35–43
Mittler R (2006) Abiotic stress, the field environment and stress combination. Trends Plant Sci 11:15–18
Mittler R, Blumwald E (2010) Genetic engineering for modern agriculture: challenges and perspectives. Annu Rev Plant Biol 61:443–462
Mordecai EA (2011) Pathogen impacts on plant communities: unifying theory, concepts, and empirical work. Ecol Monogr 81:429–441
Mutlu N, Miklas P, Reiser J, Coyne D (2005) Backcross breeding for improved resistance to common bacterial blight in pinto bean (Phaseolus vulgaris L.). Plant Breed 124:282–287
Nguyen TH (2008) The product BioGro and improvements in its performance. In: Kennedy IR, Choudhury ATMA, Kecskés ML, Rose MT (eds) Efficient nutrient use in rice production in Vietnam achieved using inoculant biofertilisers. In: Proceedings of a project (SMCN/2002/073) workshop held in Hanoi, Vietnam, 12–13 Oct 2007, pp 15–24
Nguyen TH, Deaker R, Kennedy IR, Roughley RJ (2003) The positive yield response of field grown rice to inoculation with a multi-strain biofertiliser in the Hanoi area, Vietnam. Symbiosis 35:231–245
Nicol JM, Turner SJ, Coyne DL, Nijs L, Hockland S, Maafi ZT (2011) Current nematode threats to world agriculture. In: Jones J, Gheysen G, Fenoll C (eds) Genomics and molecular genetics of plant–nematode interactions. Springer, Amsterdam, pp 21–43
Pimentel D (2002) Biological invasions: economic and environmental costs of alien plant, animal and microbe species. CRC Press, Boca Raton, p 384
Purohit M, Srivastava S, Srivastava PS (1998) Stress tolerant plants through tissue culture. In: Srivastava PS (ed) Plant tissue culture and molecular biology: application and prospects. Narosa Publishing House, New Delhi, pp 554–578
Rai MK, Kalia RK, Singh R, Gangola MP, Dhawan AK (2011) Developing stress tolerant plants through in vitro selection—an overview of the recent progress. Environ Exp Bot 71:89–98
Ramteke R, Gupta GK, Joshi OP (2004) Evaluation of rust resistance in soybean (Glycine max) under field condition. Indian J Agric Sci 74:623–624
Rejeb IB, Pastor V, Mauch-Mani B (2014) Plant responses to simultaneous biotic and abiotic stress: molecular mechanisms. Plants 3:458–475
Rodriguez AA, Stella AM, Storni MM, Zulpa G, Zaccaro MC (2006) Effect of cyanobacterial extracellular products and gibberellic acid on salinity tolerance in Oryza sativa L. Saline Sys 2:7
Rodriguez RJ, Henson J, Van Volkenburgh E, Hoy M, Wright L, Beckwith F, Kim YO, Redman RS (2008) Stress tolerance in plants via habitat-adapted symbiosis. ISME J 2:404–416
Rokhzadi A, Asgharzadeh A, Darvish F, Nour-Mohammadi G, Majidi E (2008) Influence of plant growth promoting Rhizobacteria on dry matter accumulation of Chickpea (Cicer arietinum L) under field conditions. J Agric Environ Sci 3(2):253–257
Ruiz-Lozano JM, Azcon R (2000) Symbiotixc efficiency and infectivity of an autochthonous arbuscular mycorrhizal Glomus sp. from saline soils and Glomus deserticola under salinity. Mycorrhiza 10:137–143
Ryu C, Farag MA, Hu C, Reddy MS, Wei H, Paré PW, Kloepper JW (2003) Bacterial volatiles promote growth in Arabidopsis. Proc Natl Acad Sci U S A 100(8):4927–4932
Saharan BS, Nehra V (2011) Plant growth promoting rhizobacteria: a critical review. Life Sci Med Res 21:1–30
Sahrawat KL (2000) Macro and micronutrients removed by upland and lowland rice cultivars in West Africa. Commun Soil Sci Plant Anal 31:717–723
Saleem M, Arshad M, Hussain S, Bhatti AS (2007) Perspective of plant growth promoting rhizobacteria (PGPR) containing AC deaminase in stress agriculture. J Ind Microbiol Biotechnol 34:635–648
Sandhya V, Ali SZ, Grover M, Reddy G, Venkateswarlu B (2009a) Alleviation of drought stress effects in sunflower seedlings by exopolysaccharides producing Pseudomonas putida strain P45. Biol Fert Soils 46:17–26
Sandhya V, Ali SZ, Grover M, Kishore N, Venkateswarlu B (2009b) Pseudomonas sp. strain P45 protects sunflowers seedlings from drought stress through improved soil structure. J Oilseed Res 26:600–601
Saravanakumar D, Samiyappan R (2007) Effects of 1-aminocyclopropane-1-carboxylic acid (ACC) deaminase from Pseudomonas fluorescence against saline stress under in vitro and field conditions in groundnut (Arachis hypogea) plants. J Appl Microbiol 102:1283–1292
Selvakumar G, Joshi P, Mishra PK, Bisht JP, Gupta HS (2009) Mountain aspects influence the genetic clustering of psychrotolerant phosphate solubilizing Pseudomonads in the Uttarakhand Himalayas. Curr Microbiol 59:432–438
Shao HB, Chu LY, Jaleel CA, Zhao CX (2008) Water-deficit stress—induced anatomical changes in higher plants. C R Biol 331:215–225
Smith P, Olesen JE (2010) Synergies between the mitigation of, and adaptation to, climate change in agriculture. J Agric Sci Cambridge 148:543–552
Stajner D, Kevresan S, Gasic O, Mimica-Dukic N, Zongli H (1997) Nitrogen and Azotobacter chroococcum enhance oxidative stress tolerance in sugar beet. Biol Plantarum 39(3):441–445
Strauss SY, Zangerl AR (2002) Plant-insect interactions in terrestrial ecosystems. In: Herrera CM, Pellmyr O (eds) Plant-animal interactions: an evolutionary approach. Blackwell Science, Oxford, pp 77–106
Subramanian KS, Charest C (1998) Arbuscular mycorrhizae and nitrogen assimilation in maize after drought and recovery. Physiol Plant 102:285–296
Suzuki N, Rivero RM, Shulaev V, Blumwald E, Mittler R (2014) Abiotic and biotic stress combinations. New Phytol 203:32–43
Swarbrick PJ, Schulze-Lefert P, Scholes JD (2006) Metabolic consequences of susceptibility and resistance in barley leaves challenged with powdery mildew. Plant Cell Environ 29:1061–1076
Sziderics AH, Rasche F, Trognitz F, Sessitsch A, Wilhelm E (2007) Bacterial endophytes contribute to abiotic stress adaptation in pepper plants (Capsicum annuum L.). Can J Microbiol 53:1195–1202
Tilak KVBR, Ranganayaki N, Pal KK, De R, Saxena AK, Nautiyal CS, Mittal S, Tripathi AK, Johri BN (2005) Diversity of plant growth and soil health supporting bacteria. Curr Sci 89(1):135–150
Timmusk S, Wagner EGH (1999) The plant growth-promoting rhizobacterium Paenibacillus polymyxa induces changes in Arabidopsis thaliana n gene expression: a possible connection between biotic and abiotic stress responses. Mol Plant Microbe Interact 12:951–959
Upadhyay SK, Singh DP, Saikia R (2009) Genetic diversity of plant growth promoting rhizobacteria from rhizospheric soil of wheat under saline conditions. Curr Microbiol 59(5):489–496
Venkateswarlu B, Desai S, Prasad YG (2008) Agriculturally important microorganisms for stressed ecosystems: challenges in technology development and application. In: Khachatourians GG, Arora DK, Rajendran TP, Srivastava AK (eds) Agriculturally important microorganisms, vol 1. Academic World, Bhopal, pp 225–246
Versulues PE, Agarwal M, Katiyar-Agarwal S, Zhu J, Zhu JK (2006) Methods and concepts in quantifying resistance to drought, salt and freezing, abiotic stresses that affect plant water status. Plant J 45(4):523–539
Wahid A, Farooq M, Hussain I, Rasheed R, Galani S (2012) Responses and management of heat stress in plants. In: Ahmad P, Prasad MNV (eds) Environmental adaptations and stress tolerance of plants in the era of climate change, pp 135–57
Waller F, Achatz B, Baltruschat H, Fodor J, Becker K, Fischer M, Heier T, Hückelhoven R, Neumann C, Wettstein D (2005) The endophytic fungus Piriformospora indica reprograms barley to salt-stress tolerance, disease resistance, and higher yield. Proc Natl Acad Sci U S A 102:13386–13391
Wang MC, Bohmann D, Jasper H (2003) JNK signaling confers tolerance to oxidative stress and extends lifespan in Drosophila. Dev Cell 5:811–816
Watanabe T, Kume T (2009) A general adaptation strategy for climate change impacts on paddy cultivation: special reference to the Japanese context. Paddy Water Environ 7:313–320
Yang H, Shankar M, Buirchell BJ, Sweetingham MW, Caminero C, Smith PMC (2002) Development of molecular markers using MFLP linked to a gene conferring resistance to Diaporthe toxica in narrow-leafed lupin (Lupinus angustifolius L.). Theor Appl Genet 105:265–270
Yang H, Boersma JG, You MP, Buirchell BJ, Sweetingham MW (2004) Development and implementation of a sequence specific PCR marker linked to a gene conferring resistance to anthracnose disease in narrow-leafed lupin (Lupinus angustifolius L.). Mol Breed 14:145–151
Yasmin F, Othman R, Saad MS, Sijam K (2007) Screening for beneficial properties of Rhizobacteria isolated from sweet potato rhizosphere. J Biotechnol 6(1):49–52
Yildirim E, Taylor AG (2005) Effect of biological treatments on growth of bean plans under salt stress. Ann Rep Bean Improv Coop 48:176–177
Zahir ZA, Munir A, Asghar HN, Arshad M, Shaharoona B (2008) Effectiveness of rhizobacteria containing ACC-deaminase for growth promotion of peas (Pisum sativum) under drought conditions. J Microbiol Biotech 18(5):958–963
Zangerl AR, Berenbaum MR (2003) Phenotype matching in wild parsnip and parsnip webworms. causes and consequences. Evolution 57:806–815
Zhang H et al (2008) Soil bacteria confer plant salt tolerance by tissue-specific regulation of sodium transporter HKT1. Mol Plant Microbe Interact 21:737–744
Zhang Y, Butelli E, de Stefano R, Schoonbeek HJ, Magusin A, Pagliarani C, Wellner N, Hill L, Orzaez D, Granell A (2013) Anthocyanins double the shelf life of tomatoes by delaying over ripening and reducing susceptibility to graymold. Curr Biol 23:1094–1100
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer India
About this chapter
Cite this chapter
Sirari, K., Kashyap, L., Mehta, C.M. (2016). Stress Management Practices in Plants by Microbes. In: Singh, D., Singh, H., Prabha, R. (eds) Microbial Inoculants in Sustainable Agricultural Productivity. Springer, New Delhi. https://doi.org/10.1007/978-81-322-2644-4_6
Download citation
DOI: https://doi.org/10.1007/978-81-322-2644-4_6
Published:
Publisher Name: Springer, New Delhi
Print ISBN: 978-81-322-2642-0
Online ISBN: 978-81-322-2644-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)