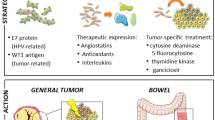
Abstract
Within the metaorganisms, cross-communication between commensal organisms and the host is crucial for the maintenance of physiological homeostasis. At times, microbiota becomes accountable for breaching homeostasis by creating a microenvironment favoring uncontrolled cell growth. Chronic infection and inflammation act as inciting factors for reactive oxygen species generation promoting damage to DNA, proteins, and lipids. Oxidative stress activates a variety of transcription factors that in turn control the expression of several inflammatory cytokines and chemokines. Consecutive changes including altered cell growth, genetic instability, and inhibition of apoptosis create a proper carcinogenic milieu. Apart from the complicated side effects associated with prevailing anticancer therapies, their safety, stability, and affordability are in question. At present, significant immunomodulating properties of probiotic bacteria are being considered as a promising tool in cancer treatment. In addition, to boost the innate defense mechanisms of hosts, probiotics can act as antigenotoxic, anticarcinogenic, and antioxidative agents. In this chapter, the role of pathogenic microbes in oxidative stress-induced cancer and the effectiveness of probiotics as a future therapeutic strategy in preventing cancer onset and progression through manipulating intestinal microflora are highlighted.
Similar content being viewed by others
Abbreviations
- 4-HNE:
-
4-Hydroxynonenal
- 8-OHdG:
-
8-Hydroxydeoxyguanosine
- 8-oxo-dG:
-
8-Oxo-2′-deoxyguanosine
- ADH:
-
Alcohol dehydrogenase
- AID:
-
Activation-induced cytidine deaminase
- AMPK:
-
Adenosine monophosphate-activated protein kinase
- AMPs:
-
Antimicrobial peptides
- APC:
-
Anaphase promoting complex
- APRIL:
-
A proliferation-inducing ligand
- AQP3:
-
Aquaporin 3
- ATP:
-
Adenosine triphosphate
- AvrA:
-
Avirulence protein A
- BAD:
-
BCL-2-associated death promoter
- BAFF:
-
B cell-activating factor
- BAX:
-
BCL-2 associated X
- BCL-2:
-
B-cell lymphoma 2
- BER:
-
Base excision repair
- BFT:
-
B. fragilis toxin
- BLIMP:
-
B lymphocyte-induced maturation protein 1
- cagA:
-
Cytotoxin-associated gene A
- CD:
-
Crohn’s disease
- CDKs:
-
Cyclin-dependent kinases
- CDT:
-
Cytolethal distending toxin
- CIDR:
-
Succeeding cysteine-rich interdomain region
- CLRs:
-
C-type lectin receptors
- CNF1:
-
Cytotoxic necrotizing factor 1
- COX:
-
Cyclooxygenase
- CRC:
-
Colorectal cancer
- CTCL:
-
Cutaneous T-cell lymphoma
- CTL:
-
Cytotoxic T lymphocytes
- CXCL:
-
C-X-C motif chemokine ligand
- CYP:
-
Cytochrome P450
- DC:
-
Dendritic cell
- DDR:
-
DNA damage responses
- DNA:
-
Deoxyribonucleic acid
- DNase I:
-
Deoxyribonuclease I
- DPPH:
-
2,2-Diphenyl-1-picrylhydrazyl
- DSBs:
-
Double-strand breaks
- EBV:
-
Epstein-Barr virus
- ERK:
-
Extracellular regulated kinase
- ETBF:
-
Enterotoxigenic B. fragilis
- Etk/BMX:
-
Epithelial and endothelial tyrosine kinase/bone marrow X
- FOXO3a:
-
Forkhead box O3a
- GEC:
-
Gingival epithelial cell
- GI:
-
Gastrointestinal
- GPRs:
-
G-protein-coupled receptors
- GPX1:
-
Glutathione peroxidase 1
- HCC:
-
Hepatocellular carcinoma
- HDAC:
-
Histone deacetylase
- HIF-1α:
-
Hypoxia-inducible factor 1α
- HNE:
-
4-Hydroxy-2-nonenal
- HRE:
-
Hypoxia response element
- HSP:
-
Heat shock protein
- IARC:
-
International Agency for Research on Cancer
- IBD:
-
Inflammatory bowel disease
- IFN-γ:
-
Interferon-γ
- IgA:
-
Immunoglobulin A
- IL:
-
Interleukin
- JAK1/STAT3:
-
Janus kinase 1/signal transducer and activator of transcription 3
- JNK:
-
c-Jun N-terminal kinases
- LPS:
-
Lipopolysaccharide
- MALT:
-
Mucosa-associated lymphoid tissue
- MDA:
-
Malondialdehyde
- MDSC:
-
Myeloid-derived suppressor cells
- miRNA:
-
MicroRNA
- NDK:
-
Nucleoside diphosphate kinase
- NF-κβ:
-
Nuclear factor κ light-chain enhancer of activated B cells
- NK:
-
Natural killer
- NLRX1:
-
NOD-like receptor family member X1
- NO:
-
Nitric oxide
- NOX:
-
NADPH oxidase
- Nrf2:
-
Nuclear factor erythroid 2-related factor 2
- OSCC:
-
Oral squamous cell carcinoma
- PBMC:
-
Peripheral blood mononuclear cells
- PfEMP1:
-
P. falciparum erythrocyte membrane protein 1
- PGN:
-
Peptidoglycan
- PI3K/Akt:
-
Phosphatidylinositol 3-kinase/protein kinase B
- PKC:
-
Protein kinase C
- pro-MMP-9:
-
Pro-matrix metalloproteinase-9
- PTEN:
-
Phosphatase and tensin homolog deleted on chromosome 10
- RNI:
-
Reactive nitrogen intermediates
- RNS:
-
Reactive nitrogen species
- ROI:
-
Reactive oxygen intermediate
- ROS:
-
Reactive oxygen species
- SabA:
-
Sialic acid–binding adhesin
- SCFA:
-
Short-chain fatty acid
- SE:
-
Staphylococcal enterotoxin
- Sir2:
-
Silent information regulator 2
- SMO:
-
Spermine oxidase
- SNP:
-
Single-nucleotide polymorphism
- SOD2:
-
Superoxide dismutase 2
- T4SS:
-
Type IV secretion systems
- TER:
-
Transepithelial resistance
- TGF-β:
-
Tumor growth factor β
- TLR:
-
Toll-like receptor
- TNF-α:
-
Tumor necrosis factor α
- UC:
-
Ulcerative colitis
- VacA:
-
Vacuolating cytotoxin A
- WHO:
-
World Health Organization
- Wnt:
-
Wingless-related integration site
- ZO:
-
Zonula occludens
References
Abdul-Sater AA et al (2010) Enhancement of reactive oxygen species production and chlamydial infection by the mitochondrial Nod-like family member NLRX1. J Biol Chem 285:41637–41645. https://doi.org/10.1074/jbc.M110.137885
Adom D, Nie D (2013) Regulation of autophagy by short chain fatty acids in colon cancer cells. In: Bailly Y (ed) Autophagy - a double-edged sword. IntechOpen, Rijeka. https://doi.org/10.5772/54999
Ahire JJ, Mokashe NU, Patil HJ, Chaudhari BL (2013) Antioxidative potential of folate producing probiotic Lactobacillus helveticus CD6. J Food Sci Technol 50:26–34. https://doi.org/10.1007/s13197-011-0244-0
Al-hebshi NN, Al-Alimi A, Taiyeb-Ali T, Jaafar N (2015) Quantitative analysis of classical and new putative periodontal pathogens in subgingival biofilm: a case-control study. J Periodontal Res 50:320–329. https://doi.org/10.1111/jre.12210
Arthur JC et al (2012) Intestinal inflammation targets cancer-inducing activity of the microbiota. Science 338:120–123. https://doi.org/10.1126/science.1224820
Benamrouz S et al (2014) Cryptosporidium parvum-induced ileo-caecal adenocarcinoma and Wnt signaling in a mouse model. Dis Model Mech 7:693–700. https://doi.org/10.1242/dmm.013292
Bermudez-Brito M, Plaza-Diaz J, Munoz-Quezada S, Gomez-Llorente C, Gil A (2012) Probiotic mechanisms of action. Ann Nutr Metab 61:160–174. https://doi.org/10.1159/000342079
Bhattacharyya A, Chattopadhyay R, Mitra S, Crowe SE (2014) Oxidative stress: an essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol Rev 94:329–354. https://doi.org/10.1152/physrev.00040.2012
Binder Gallimidi A et al (2015) Periodontal pathogens Porphyromonas gingivalis and Fusobacterium nucleatum promote tumor progression in an oral-specific chemical carcinogenesis model. Oncotarget 6:22613–22623. https://doi.org/10.18632/oncotarget.4209
Biswas P, Ghosh R, Das M, Pal S, Dam S (2021) Molecular characterization of EhAK6, an endonuclease V domain-containing aurora kinase protein from Entamoeba histolytica: protein-protein interaction, docking and functional aspect. Curr Res Biotechnol 3:225–234. https://doi.org/10.1016/j.crbiot.2021.07.003
Block K, Gorin Y (2012) Aiding and abetting roles of NOX oxidases in cellular transformation. Nat Rev Cancer 12:627–637. https://doi.org/10.1038/nrc3339
Boyer L et al (2004) Rac GTPase instructs nuclear factor-kappaB activation by conveying the SCF complex and IkBalpha to the ruffling membranes. Mol Biol Cell 15:1124–1133. https://doi.org/10.1091/mbc.e03-05-0301
Brzozowski T et al (2006) Effect of probiotics and triple eradication therapy on the cyclooxygenase (COX)-2 expression, apoptosis, and functional gastric mucosal impairment in Helicobacter pylori-infected Mongolian gerbils. Helicobacter 11:10–20. https://doi.org/10.1111/j.0083-8703.2006.00373.x
Candela M, Turroni S, Biagi E, Carbonero F, Rampelli S, Fiorentini C, Brigidi P (2014) Inflammation and colorectal cancer, when microbiota-host mutualism breaks. World J Gastroenterol 20:908–922. https://doi.org/10.3748/wjg.v20.i4.908
Chaturvedi AK et al (2010) Chlamydia pneumoniae infection and risk for lung cancer. Cancer Epidemiol Biomark Prev 19:1498–1505. https://doi.org/10.1158/1055-9965.EPI-09-1261
Cid TP, Fernandez MC, Benito Martinez S, Jones NL (2013) Pathogenesis of Helicobacter pylori infection. Helicobacter 18(Suppl 1):12–17. https://doi.org/10.1111/hel.12076
Costa Pereira C et al (2017) Association between polymorphisms in antioxidant genes and inflammatory bowel disease. PLoS One 12:e0169102. https://doi.org/10.1371/journal.pone.0169102
Dai Z et al (2019) The role of microbiota in the development of colorectal cancer. Int J Cancer 145:2032–2041. https://doi.org/10.1002/ijc.32017
Dam B, Misra A, Banerjee S (2019a) Role of gut microbiota in combating oxidative stress. In: Oxidative stress in microbial diseases, pp 43–82. https://doi.org/10.1007/978-981-13-8763-0_4
Dam S, Biswas P, Ghosh R (2019b) Oxidative stress in Entamoeba histolytica. In: Oxidative stress in microbial diseases, pp 257–280. https://doi.org/10.1007/978-981-13-8763-0_14
Davidsson S et al (2016) Frequency and typing of Propionibacterium acnes in prostate tissue obtained from men with and without prostate cancer. Infect Agent Cancer 11:26. https://doi.org/10.1186/s13027-016-0074-9
Dejea C, Wick E, Sears CL (2013) Bacterial oncogenesis in the colon. Future Microbiol 8:445–460. https://doi.org/10.2217/fmb.13.17
Diao Y et al (2014) Extracellular polysaccharide from Bacillus sp. strain LBP32 prevents LPS-induced inflammation in RAW 264.7 macrophages by inhibiting NF-kappaB and MAPKs activation and ROS production. Int Immunopharmacol 18:12–19. https://doi.org/10.1016/j.intimp.2013.10.021
Endo H, Niioka M, Kobayashi N, Tanaka M, Watanabe T (2013) Butyrate-producing probiotics reduce nonalcoholic fatty liver disease progression in rats: new insight into the probiotics for the gut-liver axis. PLoS One 8:e63388. https://doi.org/10.1371/journal.pone.0063388
Ferreira RM, Machado JC, Figueiredo C (2014) Clinical relevance of Helicobacter pylori vacA and cagA genotypes in gastric carcinoma. Best Pract Res Clin Gastroenterol 28:1003–1015. https://doi.org/10.1016/j.bpg.2014.09.004
Fijan S (2014) Microorganisms with claimed probiotic properties: an overview of recent literature. Int J Environ Res Public Health 11:4745–4767. https://doi.org/10.3390/ijerph110504745
Ghadimi D, Vrese M, Heller KJ, Schrezenmeir J (2010) Effect of natural commensal-origin DNA on toll-like receptor 9 (TLR9) signaling cascade, chemokine IL-8 expression, and barrier integritiy of polarized intestinal epithelial cells. Inflamm Bowel Dis 16:410–427. https://doi.org/10.1002/ibd.21057
Guerra L, Cortes-Bratti X, Guidi R, Frisan T (2011) The biology of the cytolethal distending toxins. Toxins (Basel) 3:172–190. https://doi.org/10.3390/toxins3030172
Gulve N, Prusty BK, Rudel T (2019) Chlamydia trachomatis impairs host base excision repair by downregulating polymerase beta. Cell Microbiol 21:e12986. https://doi.org/10.1111/cmi.12986
Ha NH et al (2015) Prolonged and repetitive exposure to Porphyromonas gingivalis increases aggressiveness of oral cancer cells by promoting acquisition of cancer stem cell properties. Tumour Biol 36:9947–9960. https://doi.org/10.1007/s13277-015-3764-9
Han YW (2015) Fusobacterium nucleatum: a commensal-turned pathogen. Curr Opin Microbiol 23:141–147. https://doi.org/10.1016/j.mib.2014.11.013
Hurnik P et al (2019) Another case of coincidental Giardia infection and pancreatic cancer. Parasitol Int 71:160–162. https://doi.org/10.1016/j.parint.2019.04.013
Inaba H, Amano A, Lamont RJ, Murakami Y (2015) Involvement of protease-activated receptor 4 in over-expression of matrix metalloproteinase 9 induced by Porphyromonas gingivalis. Med Microbiol Immunol 204:605–612. https://doi.org/10.1007/s00430-015-0389-y
Jaiswal SS, Mehra R, Pattnaik MR (2015) Amoeboma-lest we forget!!! Indian J Surg 77:190–192. https://doi.org/10.1007/s12262-015-1267-y
Kamarajan P et al (2020) Periodontal pathogens promote cancer aggressivity via TLR/MyD88 triggered activation of integrin/FAK signaling that is therapeutically reversible by a probiotic bacteriocin. PLoS Pathog 16:e1008881. https://doi.org/10.1371/journal.ppat.1008881
Khan S, Imran A, Khan AA, Abul Kalam M, Alshamsan A (2016) Systems biology approaches for the prediction of possible role of Chlamydia pneumoniae proteins in the etiology of lung cancer. PLoS One 11:e0148530. https://doi.org/10.1371/journal.pone.0148530
Kostic AD et al (2012) Genomic analysis identifies association of Fusobacterium with colorectal carcinoma. Genome Res 22:292–298. https://doi.org/10.1101/gr.126573.111
Krejsgaard T et al (2014) Staphylococcal enterotoxins stimulate lymphoma-associated immune dysregulation. Blood 124:761–770. https://doi.org/10.1182/blood-2014-01-551184
Kumarasamy V, Kuppusamy UR, Samudi C, Kumar S (2013) Blastocystis sp. subtype 3 triggers higher proliferation of human colorectal cancer cells, HCT116. Parasitol Res 112:3551–3555. https://doi.org/10.1007/s00436-013-3538-5
Kurkivuori J et al (2007) Acetaldehyde production from ethanol by oral streptococci. Oral Oncol 43:181–186. https://doi.org/10.1016/j.oraloncology.2006.02.005
Landskron G, De la Fuente M, Thuwajit P, Thuwajit C, Hermoso MA (2014) Chronic inflammation and cytokines in the tumor microenvironment. J Immunol Res 2014:149185. https://doi.org/10.1155/2014/149185
Le Leu RK, Hu Y, Brown IL, Woodman RJ, Young GP (2010) Synbiotic intervention of Bifidobacterium lactis and resistant starch protects against colorectal cancer development in rats. Carcinogenesis 31:246–251. https://doi.org/10.1093/carcin/bgp197
LeBlanc JG et al (2011) Use of superoxide dismutase and catalase producing lactic acid bacteria in TNBS induced Crohn’s disease in mice. J Biotechnol 151:287–293. https://doi.org/10.1016/j.jbiotec.2010.11.008
Lee DK et al (2009) Lactic acid bacteria affect serum cholesterol levels, harmful fecal enzyme activity, and fecal water content. Lipids Health Dis 8:21. https://doi.org/10.1186/1476-511X-8-21
Lee CU et al (2015) Redox modulation of PTEN phosphatase activity by hydrogen peroxide and bisperoxidovanadium complexes. Angew Chem Int Ed Eng 54:13796–13800. https://doi.org/10.1002/anie.201506338
Liu X, Lu R, Xia Y, Wu S, Sun J (2010) Eukaryotic signaling pathways targeted by Salmonella effector protein AvrA in intestinal infection in vivo. BMC Microbiol 10:326. https://doi.org/10.1186/1471-2180-10-326
Lu R et al (2014) Enteric bacterial protein AvrA promotes colonic tumorigenesis and activates colonic beta-catenin signaling pathway. Oncogene 3:e105. https://doi.org/10.1038/oncsis.2014.20
Moodley Y et al (2012) Age of the association between Helicobacter pylori and man. PLoS Pathog 8:e1002693. https://doi.org/10.1371/journal.ppat.1002693
Mroczynska M, Galecka M, Szachta P, Kamoda D, Libudzisz Z, Roszak D (2013) Beta-glucuronidase and Beta-glucosidase activity in stool specimens of children with inflammatory bowel disease. Pol J Microbiol 62:319–325
Ng SC, Hart AL, Kamm MA, Stagg AJ, Knight SC (2009) Mechanisms of action of probiotics: recent advances. Inflamm Bowel Dis 15:300–310. https://doi.org/10.1002/ibd.20602
Pan C, Xu X, Tan L, Lin L, Pan Y (2014) The effects of Porphyromonas gingivalis on the cell cycle progression of human gingival epithelial cells. Oral Dis 20:100–108. https://doi.org/10.1111/odi.12081
Pinchuk IV, Beswick EJ, Reyes VE (2010) Staphylococcal enterotoxins. Toxins (Basel) 2:2177–2197. https://doi.org/10.3390/toxins2082177
Prusty BK, Krohne G, Rudel T (2013) Reactivation of chromosomally integrated human herpesvirus-6 by telomeric circle formation. PLoS Genet 9:e1004033. https://doi.org/10.1371/journal.pgen.1004033
Rao RK, Samak G (2013) Protection and restitution of gut barrier by probiotics: nutritional and clinical implications. Curr Nutr Food Sci 9:99–107. https://doi.org/10.2174/1573401311309020004
Robbiani DF et al.,. (2015). Plasmodium infection promotes genomic instability and AID-dependent B cell lymphoma. Cell 162:727–737. https://doi.org/10.1016/j.cell.2015.07.019
Rodriguez-Rivera LD, Bowen BM, den Bakker HC, Duhamel GE, Wiedmann M (2015) Characterization of the cytolethal distending toxin (typhoid toxin) in non-typhoidal Salmonella serovars. Gut Pathogens 7:19. https://doi.org/10.1186/s13099-015-0065-1
Rubinstein MR, Wang X, Liu W, Hao Y, Cai G, Han YW (2013) Fusobacterium nucleatum promotes colorectal carcinogenesis by modulating E-cadherin/beta-catenin signaling via its FadA adhesin. Cell Host Microbe 14:195–206. https://doi.org/10.1016/j.chom.2013.07.012
Satuman S et al (2021) The effect of acute and chronic infection-induced by AvrA protein of salmonella typhimurium on radical oxygen species, phosphatase and tensin homolog, and cellular homolog expression during the development of colon cancer. Open Access Macedonian J Med Sci 9:343–351. https://doi.org/10.3889/oamjms.2021.4945
Schlee M, Wehkamp J, Altenhoefer A, Oelschlaeger TA, Stange EF, Fellermann K (2007) Induction of human beta-defensin 2 by the probiotic Escherichia coli Nissle 1917 is mediated through flagellin. Infect Immun 75:2399–2407. https://doi.org/10.1128/IAI.01563-06
Sellers TA et al (2001) Dietary folate intake, alcohol, and risk of breast cancer in a prospective study of postmenopausal women. Epidemiology 12:420–428. https://doi.org/10.1097/00001648-200107000-00012
Simone O, Bejarano MT, Pierce SK, Antonaci S, Wahlgren M, Troye-Blomberg M, Donati D (2011) TLRs innate immunereceptors and Plasmodium falciparum erythrocyte membrane protein 1 (PfEMP1) CIDR1alpha-driven human polyclonal B-cell activation. Acta Trop 119:144–150. https://doi.org/10.1016/j.actatropica.2011.05.005
Sulzyc-Bielicka V et al (2018) Colorectal cancer and Cryptosporidium spp. infection. PLoS One 13:e0195834. https://doi.org/10.1371/journal.pone.0195834
Takahashi N, Nyvad B (2011) The role of bacteria in the caries process: ecological perspectives. J Dent Res 90:294–303. https://doi.org/10.1177/0022034510379602
Takaki A, Yamamoto K (2015) Control of oxidative stress in hepatocellular carcinoma: helpful or harmful? World J Hepatol 7:968–979. https://doi.org/10.4254/wjh.v7.i7.968
Tang Y, Chen Y, Jiang H, Robbins GT, Nie D (2011) G-protein-coupled receptor for short-chain fatty acids suppresses colon cancer. Int J Cancer 128:847–856. https://doi.org/10.1002/ijc.25638
Thirugnanam S, Rout N, Gnanasekar M (2013) Possible role of Toxoplasma gondii in brain cancer through modulation of host microRNAs. Infect Agent Cancer 8:8. https://doi.org/10.1186/1750-9378-8-8
Tjalsma H, Boleij A, Kato I (2012) Streptococcus bovis and colorectal cancer. In: Bacteria and cancer, pp 61–78. https://doi.org/10.1007/978-94-007-2585-0_3
Uitto VJ et al (2005) Fusobacterium nucleatum increases collagenase 3 production and migration of epithelial cells. Infect Immun 73:1171–1179. https://doi.org/10.1128/IAI.73.2.1171-1179.2005
Varet H et al (2018) Enteric bacteria boost defences against oxidative stress in Entamoeba histolytica. Sci Rep 8:9042. https://doi.org/10.1038/s41598-018-27086-w
Yong X et al (2016) Helicobacter pylori upregulates Nanog and Oct4 via Wnt/beta-catenin signaling pathway to promote cancer stem cell-like properties in human gastric cancer. Cancer Lett 374:292–303. https://doi.org/10.1016/j.canlet.2016.02.032
Yu Q, Yuan L, Deng J, Yang Q (2015) Lactobacillus protects the integrity of intestinal epithelial barrier damaged by pathogenic bacteria. Front Cell Infect Microbiol 5:26. https://doi.org/10.3389/fcimb.2015.00026
Yusof AM, Kumar S (2012) Phenotypic ‘variant’ forms of Trichomonas vaginalis trophozoites from cervical neoplasia patients. Exp Parasitol 131:267–273. https://doi.org/10.1016/j.exppara.2012.03.015
Zhou Y et al (2015) Noncanonical activation of beta-catenin by Porphyromonas gingivalis. Infect Immun 83:3195–3203. https://doi.org/10.1128/IAI.00302-15
Acknowledgments
The financial assistance from DAE-BRNS, Government of India (Project No. 37 (1)/20/15/2014-BRNS), and CSIR, Government of India (Project No. 27(0322)/17/EMR-II) is thankfully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Section Editor information
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this entry
Cite this entry
Biswas, P., Pal, S., Das, M., Dam, S. (2022). Microbe-Induced Oxidative Stress in Cancer Development and Efficacy of Probiotics as Therapeutics in Preventing Its Onset and Progression. In: Chakraborti, S. (eds) Handbook of Oxidative Stress in Cancer: Therapeutic Aspects. Springer, Singapore. https://doi.org/10.1007/978-981-16-1247-3_159-1
Download citation
DOI: https://doi.org/10.1007/978-981-16-1247-3_159-1
Received:
Accepted:
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-1247-3
Online ISBN: 978-981-16-1247-3
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences