Abstract
With the growing success of halide perovskites as functional materials in solar cells, LEDs, and lasers, studies are progressing to reveal their optical and charge-carrier properties. This chapter describes the consequences of charge-carrier generation, stabilization, and recombination in perovskite nanocrystals, quantum dots, and their assemblies. The strong confinement of charge-carriers in perovskite quantum dots causes photoluminescence blinking with distinct ON and OFF events, which is due to photocharging and ultrafast Auger nonradiative recombination. Such a blinking with long OFF periods suppresses superoxide generation and oxidation of perovskites. When perovskite nanocrystals and quantum dots are closely-packed into superlattices, the carrier confinement is broken due to the narrowing of inter-particle energy levels and the formation of minibands that allow for carrier migration. This results in unexpectedly delayed photoluminescence at low intensities of excitation light, whereas at high intensities of excitation light, the ultrafast radiative recombination of charge-carriers occurs. These properties of quantum dots, nanocrystals, and assemblies of perovskites are important to be considered during the construction of devices such as solar cells, LEDs, and lasers.
Sushant Ghimire and Lata Chouhan: Equal contribution.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Halide perovskites are exciting materials for light-harvesting [1,2,3] and light-emitting [4,5,6] applications. Their success foots on the large absorption coefficient, high charge-carrier mobility, high photoluminescence (PL) quantum yield (QY), and cost-effective synthesis [1,2,3,4,5,6]. Further, the emission color from halide perovskites can cover the entire UV-visible-near infrared spectrum, which emanates from different band-gaps induced by changing the halogen compositions in the material [4,5,6]. Interestingly, these properties of halide perovskite are maintained irrespective of their size. Such attributes make halide perovskites highly promising semiconductor materials that can compete with the existing silicon technology for solar cells and quantum dot (QD) technology for light-emitting devices and displays. In addition to the chemical and physical stability of perovskites, the generation, stabilization, and binding energy of charge-carriers in these materials are also fundamental aspects to be considered for such device applications.
Charge-carrier dynamics in halide perovskites are complicated by the coexistence of bound electron-hole pairs (excitons) and free charge-carriers [7,8,9,10,11]. The coulombic binding between an electron and a hole in an exciton can be strong or weak, depending upon the exciton binding energy (Eb). Weakly and strongly bound excitons in semiconductors are shown schematically in Fig. 11.1. Weakly bound excitons are called Mott-Wannier excitons (Fig. 11.1a), where low Eb results in the larger delocalization of electron-hole pair, resulting in exciton Bohr radius (RB) much greater than the lattice constant (a) of the material. On the other hand, strongly bound exciton with high Eb is called Frenkel exciton whose RB is much smaller than the a of the material (Fig. 11.1b). In halide perovskites, Eb lies in between those of Mott-Wannier and Frenkel excitons and is either less than or comparable to the thermal energy. Hence, free charge-carriers dominate in the films and bulk single crystals of these materials. Studies have shown that the free charge-carriers are the consequence of dissociation of excitons when the associated binding energy is overcome by the thermal energy [8,9,10,11,12]. The free charge-carrier property of halide perovskites is associated with long carrier diffusion lengths and lifetimes, as observed in the case of bulk films and single crystals [2, 3]. Nevertheless, the carrier lifetime varies in the range of sub-nanoseconds to microseconds in different types of halide perovskites, depending upon their size, dimensionality, and composition [2, 3, 8,9,10,11,12,13,14,15].
Exciton binding energies in perovskite nanocrystals (PNCs) are higher than the corresponding bulk crystal. For example, Eb of 320 meV is reported for MAPbBr3 (MA = methylammonium, CH3NH3+) nanocrystals, which is more than three times higher (84 meV) than that for the corresponding bulk crystal [16]. Therefore, quantum confinement effects are observed in PNCs as well, when their physical size becomes smaller than the corresponding RB values [13, 15]. Additionally, the quantum confinement effects in PNCs are depended upon not only the size but also the dimensionality. For instance, blue-shift in absorption and PL spectra, which is associated with the increase in optical band-gap, is observed in the case of perovskite nanoplatelets when the layer thickness decreases below RB [15]. Despite the quantum confinement effects in PNCs, when compared to the conventional chalcogenide QDs, slow cooling and efficient extraction of hot carriers are reported in their colloidal solutions [17]. The intrinsic phonon bottleneck and Auger heating effects at low and high carrier densities, respectively, in PNCs account for such observations. Apart from this, the excellent optoelectronic properties of PNCs, which are comparable to the conventional QDs, are maintained even at the weak quantum confinement regime. While RB in lead halide perovskites lies in the range of 2–7 nm [13, 15, 16, 18, 19], most of the studies demonstrating the light-emitting [4, 6] and photovoltaic [17, 18] applications of PNCs show the nanocrystal size either only slightly smaller or larger than the corresponding RB value. This suggests that, compared to the chalcogenide QDs, the application of PNCs in light-harvesting and light-emitting device is diverse and immense.
2 Photoluminescence Blinking
In quantum-confined semiconductor nanocrystals or QDs, the photogenerated electrons and holes are strongly correlated. Carrier dynamics in such strongly confined QDs is governed by the discrete energy states showing sharp excitonic band in the absorption spectrum, fast radiative recombination of charge-carriers, carrier multiplication or multi-exciton generation, and increased Auger recombination [20]. Moreover, the PL properties of these semiconductor nanocrystals at their single particle level are often intricated by the stochastic emission behavior with a sequence of bright ‘ON’ and dark ‘OFF’ events known as PL intermittency or blinking. This is attributed to the random charging by photoionization and neutralization or discharging of the QD. The blinking behavior in QDs is often described as A-type and B-type [21]. In A-type blinking, the ‘OFF’ state is associated with the decrease in PL lifetime, whereas the carrier-recombination rates are not affected by the ‘OFF’ or the ‘ON’ states in B-type blinking. The short PL lifetime observed during the ‘OFF’ state in A-type blinking is attributed to the formation of a charged excitonic state called a trion (X−) which promotes the mono-exponential nonradiative Auger-type carrier recombination in QDs. On the other hand, B-type blinking mostly depends on the activation and deactivation of the traps or its recombination centers which leads to the ‘ON’ and ‘OFF’ states, respectively.
Apart from the QDs, diverse stochastic emission behavior has been observed in the case of perovskite single nanocrystals, microcrystals, and thin films [22,23,24,25,26,27]. Some perovskite single-emitters have shown blinking with two-state fluctuations between ‘ON’ and ‘OFF’ events [24, 25, 27], which is similar to the case of chalcogenide QD and is ascribed to the process of photocharging and neutralization. Semiconductor nanocrystals or QDs show various interesting optoelectronic properties in the isolated forms. On the other hand, multi-state PL blinking has also been reported from the nanocrystals, microcrystals, and films of lead halide perovskite [22, 23, 26]. An early report on PL blinking in MAPbI3 PNCs and microcrystals has shown that the chemical and structural properties of material are responsible for the multi-state stochastic emission behavior [22]. This is explained through two mechanisms, namely the quenching-site and the emitting-site model, which is shown in Fig. 11.2. In emitting-site model, as shown in Fig. 11.2a, the emitters are at the either end of the nanocrystal which randomly get charged and discharged leading to the ‘ON’ and ‘OFF’ states, respectively. Quenching-site model (Fig. 11.2b) considers the whole nanocrystal as an emissive site, where the emission localization position and center of the nanocrystals lie at the same point. Quenchers in quenching-site model act as traps which are present at the either end of the nanocrystal and deactivate the electrons and holes during the diffusion, resulting in the ‘OFF’ state. An interesting behavior of PL blinking in lead halide perovskites is that, unlike lead chalcogenide QDs, the multi-state PL fluctuations in these semiconductor materials is suppressed under high-intensity of excitation, which on the other hand becomes more intense at low-intensity of excitation. Figure 11.3a shows the excitation laser intensity-dependent PL blinking in MAPbI3 PNCs at the ambient environment. The suppression of PL blinking in such perovskite samples at high-intensity excitation is attributed to the decrease in efficiency of active quenchers to trap large number of charges. Alternately, more emitting sites are generated at high-intensity excitation, resulting in the averaged PL intensity over many emitting sites. Nevertheless, as shown in Fig. 11.3a(i) and b(i), photobleaching cannot be ruled out at high-intensity excitation under the ambient atmosphere [22, 25, 27].

Reproduced with permission from Ref. [22]. Copyright 2015, American Chemical Society
Models of PL blinking in lead halide perovskites: a Quenching-site model and b emitting-site model.

Reproduced with permission: (a) from Ref. [22]. Copyright 2015, American Chemical Society. (b) From Ref. [25]. Copyright 2019, Wiley-VCH
Excitation intensity- and local environment-dependent PL blinking in lead halide perovskites. a PL blinking in MAPbI3 PNCs under increasing intensity of the excitation laser: (bottom) 0.01 to (top) 0.5 Wcm−2. b PL blinking in MAPbI3 PNCs (i, iv) in air (ii) under continuous argon purging, and (iii) in polymer matrix
3 External Factors Affecting the Photoluminescence
Importantly, different environmental factors such as moisture, nitrogen, oxygen, polymer, and light have shown variable results on the PL properties of perovskites at their single particle level [24, 27]. The PL blinking trajectories of MAPbI3 nanocrystals at different local environment are shown in Fig. 11.3b. For example, different roles of oxygen on the PL properties of lead halide perovskites have been reported for different types of perovskite samples [22, 25, 27]. While PL brightening was observed as the consequence of photoinduced trap-filling in the case of MAPbI3 PNCs in presence of oxygen [22], PL bleaching was reported in CsPbI3 perovskite QDs under the similar environment [27]. Recently, a study carried out at the single particle level in MAPbI3 PNCs under air, argon, and polymer matrix (Fig. 11.3b) reveals its PL degradation mechanism [25]. The PNCs degrade or photobleach under the air atmosphere, whereas the PL intensity remains constant under the argon and the polymer atmosphere. The photostability of a PNC under the argon or the polymer atmosphere is the result of low reactivity of oxygen with the nanocrystal during its ‘ON’ state. Interestingly, as shown in Fig. 11.3b(iv), a recovery of PL intensity level is observed even at the air atmosphere after the long ‘OFF’ event, suggesting that there was no degradation of PNCs during the long ‘OFF’ duration that involves nonradiative recombination. Alternately, the PL degradation at the air atmosphere when the PNCs were more frequently in the ‘ON’ state or with the long-living ‘ON’ duration can be ascribed to the oxidation of nanocrystals by the generation of superoxide, which is discussed schematically in Fig. 11.4. Here, the neutral and ionized states are responsible for the ‘ON’ and ‘OFF’ events in the PL blinking trajectories of PNCs. When the nanocrystal is in excited state, it can either oxidize and transfer electron to the oxygen present in the atmosphere generating superoxide or get ionized. The generated superoxide can take the vacancies of the anion and deprotonate the methyl ammonium cation, thereby slowly degrading the PNCs. On the other hand, when the nanocrystal is in the ionized state, nonradiative recombination becomes faster than the electron transfer, and the reactivity of oxygen is suppressed during the long ‘OFF’ duration. In such ON-OFF dynamics and oxidation/antioxidation processes, the excitation light intensity plays a crucial role on the density and fate of charge-carriers. Apart from this, the confinement or diffusion of locally generated multiple charge-carriers depends upon the particle-particle interaction.

Reproduced with permission from Ref. [25]. Copyright 2019, Wiley-VCH
Oxidation in perovskite. Schematic representation of the PL blinking and the degradation of the MAPbI3 PNCs.
4 Carrier Migration in Perovskite Assemblies
When semiconductor nanocrystals or QDs are assembled into a superlattice, their individual properties transform to produce more unique optical, electronic, and thermal properties. This arises from the interaction between the individual particles in the assembly [28,29,30]. In a superlattice, when the QDs are very close to each other, the interaction between their electronic wavefunctions increases. At a point, when the inter-particle spacing is too short, the discrete energy states of individual nanocrystals or QDs split to form minibands. As the result, long-range energy and charge transport in these superlattices through these bands become possible [30]. Strong electronic coupling and the formation of minibands as a function of inter-particle distance in semiconductor nanocrystal superlattice is shown schematically in Fig. 11.5. Such properties of QD superlattice are highly applicable in electronic, optoelectronic and thermoelectric devices [30, 31]. Since the pioneering work of Bawendi and coworkers on chalcogenide QD superlattices [28], many researchers have carried out a successful coupling of QDs and migration of energy or excitons among them in their two- or three-dimensional superlattice structures [31,32,33]. Exciton diffusion is observed in a highly ordered CdSe QD superlattice with center-to-center distance less than 7 nm between the adjacent QDs [32]. In such QD superlattices, the exciton diffusion length as longer as 125 nm is reported. A faster exciton diffusion among highly interacting QDs can result in a faster exciton decay, which is attributed to the larger number of traps or defect sites on different QDs that the excitons may encounter within their lifetime. Besides, a highly ordered QD superlattice can result lower PLQY when compared to the disordered films and colloidal QD samples. This is ascribed to the faster energy transfer among closely-packed QDs. Similarly, directional transport of excitons is observed in long-range ordered and strongly coupled binary nanocrystal superlattice of PbSe consisting of two different sizes [33]. The surface-to-surface inter-particle spacing in such QD array can be reduced further by ligand exchange which results in the ultrafast exciton decay within few picoseconds, showing strong electronic coupling between QDs.
In a QD superlattice, the coupling energy between the individual QDs is much smaller than the exciton binding energy. Therefore, the excitons do not dissociate into free charge-carriers in the superlattice, rather they diffuse or hop and undergo ultrafast recombination, or the nonradiative energy transfer takes place among the closely-packed QDs. On the other hand, if the inter-particle distance between the QDs is very small (for example, center-to-center distance <1 nm), it induces sintering and straining of QDs, leading to increased disorder [29, 32]. Owing to these limitations of QD solids, a superlattice of semiconductor nanocrystals where long-range diffusion of free charge-carriers, which is associated with delayed recombination, is largely required for their efficient applications in solar cells; and the organic-inorganic hybrid lead halide perovskites are promising candidates for this purpose.
Organic-inorganic hybrid PNCs (Fig. 11.6a) when assembled into close-packed film (Fig. 11.1c inset) show unexpectedly delayed PL (Fig. 11.6b, c, and d) under low-intensity excitation [34, 35], which are comparable to the free carrier recombination in single microcrystals and bulk films of perovskites [2, 3, 7, 14]. For instance, a PL lifetime >900 ns is observed for MAPbBr3 PNC film [34], which becomes much longer (>4 μs) in the case of FAPbBr3 (FA = formamidinium,CHN2H3+) perovskite nanocrystal PNC film [35]. Under the increasing excitation intensity, the radiative recombination becomes extremely fast, resulting in amplified emission. The excitation intensity-dependent PL decay profiles of MAPbBr3 PNC film and the temporally and spectrally resolved photocount maps of FAPbBr3 PNC film are shown in Fig. 11.6b, and d–g, respectively. The fast PL lifetimes at higher intensities of excitation are associated with the exponential rise in the number of photons emitted by the PNC film, which is discussed in Fig. 11.6c. On the other hand, a fast PL is obvious in the case of isolated PNCs in their colloidal solution, irrespective of the intensity of excitation. These observations point toward the long-range migration of excitonically unbound charge-carriers at low-intensity excitation and spatial confinement of these charge-carriers at higher intensities in PNC films. The amplified emission can be assigned to the radiative recombination of high-density electrons and holes that are generated and spatially confined among the close-packed PNCs in the film [34, 35].

Reproduced with permission from Ref. [34]. Copyright 2019, American Chemical Society
Excitation intensity-dependent PL and charge-carrier properties of PNC films. a TEM image of MAPbBr3 PNCs. The scale bar is 20 nm. b PL decay profiles of a MAPbBr3 PNC film under increasing intensity of excitation laser: (top) 0.0175 to (bottom) 170 MWcm−2. c Plots of the PL lifetime and the number of photons emitted as a function of excitation laser power for a MAPbBr3 PNC film. Inset: MAPbBr3 PNC film under UV light. d–g Temporally and spectrally resolved photocount maps of a FAPbBr3 PNC film with an increase in excitation laser power: d 0.17, e 1.7, f 17, g 170 MWcm−2.
The dynamics of charge-carriers generated in a PNC film at low- and high-intensity excitations is shown schematically in Fig. 11.7a and b, respectively. At high-intensity excitation, manifold charge-carriers are generated among different PNCs in the irradiated area (Fig. 11.7b). On the other hand, the concentration of photogenerated charge-carriers is less at low-intensity excitation, which reduces the carrier-carrier interactions and allows for the long-range diffusion in the film (Fig. 11.7a). Owing to such a long-range carrier diffusion, PNC films excited with low-intensity laser show unexpectedly delayed PL. On the other hand, the diffusion of photogenerated charge-carriers is spatially confined by increasing the intensity of excitation laser, which is the result of an increase in the carrier concentration. Accordingly, the rate of radiative recombination within the irradiated area controls over the diffusion, providing amplified emission.

Reproduced with permission from Ref. [34]. Copyright 2019, American Chemical Society
Diffusion and recombination of charge-carriers in a PNC film a, b Scheme of charge-carrier diffusion and recombination in a PNC film under a low-intensity and b high-intensity excitations. c PL spectra of FAPbBr3 PNC films with different PNC density. The corresponding PL lifetime values are shown in the line-symbol plot. d PL decay profiles of FAPbBr3 PNC films with different PNC density.
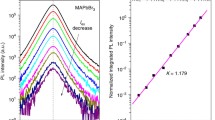
The degree of charge-carrier migration across PNCs in their close-packed assembly (or film) depends upon their packing density [34]. As seen in Fig. 11.7c, the PL spectra of a loose-packed PNC film is blue-shifted, whereas that of a close-packed film is red-shifted. Additionally, as shown in Fig. 11.7c and d, a red-shifted PL spectrum is associated with long PL lifetime. The low-energy photons which are associated with the delayed emission in PNC film point toward the diffusion of charge-carriers through closely-spaced energy states during which some energy is lost, resulting in long-lived low-energy photons. Such delayed PL, as a result of long-range migration of photogenerated charge-carriers, in close-packed assembly of PNCs is a promising property for the harvesting of charge-carriers in high-efficiency solar cells. Indeed, an appreciable rate of electron transfer (3.3 × 106 s−1) is achieved from the photoexcited close-packed assembly of PNCs to fullerene (C60), revealed by the fast PL decay of the PNC film doped with C60, when compared to a pristine film without C60, as shown in Fig. 11.8 [35].

Reproduced with permission from Ref. [35]. Copyright 2019, Royal Society of Chemistry
PL decay profiles of a PNC film excited with low-intensity laser (0.0175 µJ/pulse) (red) before and (blue) after doping with C60.
5 Conclusions
The optical and electronic properties of individual QDs modify when they are arranged into superlattices. The overlapping of QDs electronic wavefunctions in the lattice facilitates the long-range diffusion or hopping of excitons. Due to the high exciton binding energy in strongly quantum-confined systems, the diffusing excitons do not dissociate into free charge-carriers. Thus, the exciton recombination becomes ultrafast, and the isolated semiconductor nanocrystals or QDs show PL blinking. In contrast, metal halide perovskite nanocrystals and QDs arranged into superlattices exhibit long-range carrier migration and delayed radiative recombination. This is analogous to the long-range diffusion of free charge-carriers in bulk perovskites, suggesting a weak confinement of photogenerated electron-hole pairs. The formation of new inter-particle energy states in superlattices of PNCs assist in the diffusion of coulombically uncorrelated electrons and holes. Such freely diffusing electrons are effectively harvested using C60. Therefore, superlattices of PNCs are promising structures for the development of high-efficiency solar cells.
References
Kojima A, Teshima K, Shirai Y, Miyasaka T (2009) Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J Am Chem Soc 131:6050–6051
Dong Q, Fang NY, Shao Y, Mulligan P, Qiu J, Cao L, Huang J (2015) Electron-hole diffusion lengths > 175 µm in solution-grown CH3NH3PbI3 single crystals. Science 347:967–970
deQuilettes DW, Vorpahl SM, Stranks SD, Nagaoka H, Eperon GE, Ziffer ME, Snaith HJ, Ginger DS (2015) Impact of microstructure on local carrier lifetime in perovskite solar cells. Science 348:683–686
Zhang F, Zhong H, Chen C, Wu X, Hu X, Huang H, Han J, Zou B, Dong Y (2015) Brightly luminescent and color-tunable colloidal CH3NH3PbX3 (X = Br, I, Cl) quantum dots: potential alternatives for display technology. ACS Nano 9:4533–4542
Kim Y-H, Cho H, Heo JH, Kim T-S, Myoung N, Lee C-L, Im SH, Lee T-W (2015) Multicolored organic/inorganic hybrid perovskite light-emitting diodes. Adv Mater 27:1248–1254
Yakunin S, Protesescu L, Krieg F, Bodnarchuk MI, Nedelcu G, Humer M, De Luca G, Fiebig M, Heiss W, Kovalenko MV (2015) Low-threshold amplified spontaneous emission and lasing from colloidal nanocrystals of caesium lead halide perovskites. Nat Commun 6:8056
Saba M, Cadelano M, Marongiu D, Chen F, Sarritzu V, Sestu N, Figus C, Aresti M, Piras R, Lehmann AG, Cannas C, Musinu A, Quochi F, Mura A, Bongiovanni G (2014) Correlated electron-hole plasma in organometal perovskites. Nat Commun 5:5049
Yang Y, Yang M, Li Z, Crisp R, Zhu K, Beard MC (2015) Comparison of recombination dynamics in CH3NH3PbBr3 and CH3NH3PbI3 perovskite films: influence of exciton binding energy. J Phys Chem Lett 6:4688–4692
Jha A, Duan H-G, Tiwari T, Nayak PK, Snaith HJ, Thorwart M, Miller RJD (2018) Direct observation of ultrafast exciton dissociation in lead iodide perovskite by 2D electronic spectroscopy. ACS Photonics 5:852–860
Phuong LQ, Yamada Y, Nagai M, Maruyama N, Wakamiya A, Kanemitsu Y (2016) Free carriers versus excitons in CH3NH3PbI3 perovskite thin films at low temperatures: charge transfer from the orthorhombic phase to the tetragonal phase. J Phys Chem Lett 7:2316–2321
Blancon J-C, Tsai H, Nie W, Stoumpos CC, Pedesseau L, Katan C, Kepenekian M, Soe CMM, Appavoo K, Sfeir MY, Tretiak S, Ajayan PM, Kanatzidis MG, Even J, Crochet JJ, Mohite AD (2017) Extremely efficient internal exciton dissociation through edge states in layered 2D perovskites. Science 355:1288–1292
Miyata A, Mitioglu A, Plochocka P, Portugall O, Wang JT, Stranks SD, Snaith HJ, Nicholas RJ (2015) Direct measurement of the exciton binding energy and effective masses for charge carriers in organic-inorganic tri-halide perovskites. Nat Phys 11:582–587
Butkus J, Vashishtha P, Chen K, Gallaher JK, Prasad SKK, Metin DZ, Laufersky G, Gaston N, Halpert JE, Hodgkiss JM (2017) The Evolution of quantum confinement in CsPbBr3 perovskite nanocrystals. Chem Mater 29:3644–3652
Shi D, Adinolfi V, Comin R, Yuan M, Alarousu E, Buin A, Chen Y, Hoogland S, Rothenberger A, Katsiev K, Losovyj Y, Zhang X, Dowben PA, Mohammed OF, Sargent EH, Bakr OM (2015) Low trap-state density and long carrier diffusion in organolead trihalide perovskite single crystals. Science 347:519–522
Levchuk I, Osvet A, Tang X, Brandl M, Perea JD, Hoegl F, Matt GJ, Hock R, Batentschuk M, Brabec CJ (2017) Brightly luminescent and color-tunable formamidinium lead halide perovskite FAPbX3 (X = Cl, Br, I) colloidal nanocrystals. Nano Lett 17:2765–2770
Zheng K, Zhu Q, Abdellah M, Messing ME, Zhang W, Generalov A, Niu Y, Ribaud L, Canton SE, Pullerits T (2015) Exciton binding energy and the nature of emissive states in organometal halide perovskites. J Phys Chem Lett 6:2969–2975
Li M, Bhaumik S, Goh TW, Kumar MS, Yantara N, Grätzel M, Mhaisalkar S, Mathews N, Sum TC (2017) Slow cooling and highly efficient extraction of hot carriers in colloidal perovskite nanocrystals. Nat Commun 8:14350
Li M, Begum R, Fu J, Xu Q, Koh TM, Veldhuis SA, Grätzel M, Mathews N, Mhaisalkar S, Sum TC (2018) Low threshold and efficient multiple exciton generation in halide perovskite nanocrystals. Nat Commun 9:4197
Tanaka K, Takahashi T, Ban T, Kondo T, Uchida K, Miura N (2003) Comparative study on the excitons in lead-halide-based perovskite-type crystals CH3NH3PbBr3 CH3NH3PbI3. Solid State Commun 127:619–623
Ghimire S, Biju V (2018) Relations of exciton dynamics in quantum dots to photoluminescence, lasing, and energy harvesting. J Photochem Photobiol C 34:137–151
Galland C, Ghosh Y, Steinbrück A, Sykora M, Hollingsworth JA, Klimov VI, Htoon H (2011) Two types of luminescence blinking revealed by spectroelectrochemistry of single quantum dots. Nature 479:203–207
Tian Y, Merdasa A, Peter M, Abdellah M, Zheng K, Ponseca CS Jr, Pullerits T, Yartsev A, Sundström V, Scheblykin IG (2015) Giant photoluminescence blinking of perovskite nanocrystals reveals single-trap control of luminescence. Nano Lett 15:1603–1608
Gerhard M, Louis B, Camacho R, Merdasa A, Li J, Kiligaridis A, Dobrovolsky A, Hofkens J, Scheblykin IG (2019) Microscopic insight into nonradiative decay in perovskite semiconductors from temperature-dependent luminescence blinking. Nat Commun 10:1698
Park Y-S, Guo S, Makarov NS, Klimov VI (2015) Room temperature single-photon emission from individual perovskite quantum dots. ACS Nano 9:10386–10393
Chouhan L, Ghimire S, Biju V (2019) Blinking beats bleaching: the control of superoxide generation by photo-ionized perovskite nanocrystals. Angew Chem Int Ed 58:4875–4879
Li C, Zhong Y, Luna CAM, Unger T, Deichsel K, Gräser A, Köhler J, Köhler A, Hildner R, Huettner S (2016) Emission enhancement and intermittency in polycrystalline organolead halide perovskite films. Molecules 21:1081
Yuan G, Ritchie C, Ritter M, Murphy S, Gómez DE, Mulvaney P (2018) The degradation and blinking of single CsPbI3 perovskite quantum dots. J Phys Chem C 122:13407–13415
Murray CB, Kagan CR, Bawendi MG (1995) Self-organization of CdSe nanocrystallites into three-dimensional quantum dot superlattices. Science 270:1335–1338
Collier CP, Vossmeyer T, Heath JR (1998) Nanocrystal superlattices. Annu Rev Phys Chem 49:371–404
Kagan CR, Murray CB (2015) Charge transport in strongly coupled quantum dot solids. Nat Nanotechnol 10:1013–1026
Harman TC, Taylor PJ, Walsh MP, LaForge BE (2002) Quantum dot superlattice thermoelectric materials and devices. Science 297:2229–2232
Yoon SJ, Guo Z, dos Santos Claro PC, Shevchenko EV, Huang L (2016) Direct imaging of long-range exciton transport in quantum dot superlattices by ultrafast microscopy. ACS Nano 10:7208–7215
Wu Y, Li S, Gogotsi N, Zhao T, Fleury B, Kagan CR, Murray CB, Baxter JB (2017) Directional carrier transfer in strongly coupled binary nanocrystal superlattice films formed by assembly and in situ ligand exchange at a liquid-air interface. J Phys Chem C 121:4146–4157
Ghimire S, Chouhan L, Takano Y, Takahashi K, Nakamura T, Yuyama K, Biju V (2019) Amplified and multicolor emission from films and interfacial layers of lead halide perovskite nanocrystals. ACS Energy Lett. 4:133–141
Ghimire S, Nair VC, Muthu C, Yuyama K, Vacha M, Biju V (2019) Photoinduced photoluminescence enhancement in self-assembled clusters of formamidinium lead bromide perovskite nanocrystals. Nanoscale 11:9335–9340
Acknowledgements
S. G. acknowledges the MEXT scholarship for doctoral research, and L. C. acknowledges the JICA scholarship for doctoral research. V. B. acknowledges financial support (Grants 15H01099 and 17H05243) from MEXT under the JSPS Grant-in-Aid for Scientific Research on Innovative Areas “Photosynergetics.”
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Ghimire, S., Chouhan, L., Biju, V. (2020). The Confinement and Migration of Charge-Carriers in Lead Halide Perovskites. In: Miyasaka, H., Matsuda, K., Abe, J., Kawai, T. (eds) Photosynergetic Responses in Molecules and Molecular Aggregates. Springer, Singapore. https://doi.org/10.1007/978-981-15-5451-3_11
Download citation
DOI: https://doi.org/10.1007/978-981-15-5451-3_11
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-5450-6
Online ISBN: 978-981-15-5451-3
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)