Abstract
Arsenic (As) is a ubiquitous and naturally occurring metalloid that poses significant carcinogenic and noncarcinogenic health risks to human. Apart from drinking water, food is the other major source of As exposure for humans. A principal source of As in diet for the general population living in the non-As endemic areas is rice. Rice is a staple food for the global population. The annual rice consumption per capita has been increasing over time. The issue of As accumulation in rice and its potential health impacts have become a global public health concern for several decades, as the rice consumption per capita is normally high and As is classified as group 1 carcinogen. Therefore, the Codex Alimentarius Commission, the Joint Food and Agriculture Organization (FAO) of the United Nations, and the World Health Organization (WHO) food standards program have established the maximum allowance level of As, especially inorganic As, in rice grain. This chapter draws attention to the most updated data on the total and inorganic As concentrations in the most popular types of rice sold in the markets of all the world regions. The bioaccessible As concentrations were also reported in this chapter, as this fraction is believed to represent the amount of As that is actually taken up by the human body. Furthermore, considerable attention is given to the field studies of As in rice grain which were cultivated in the As-contaminated areas.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Arsenic (As), the 52nd most abundant element in the earth’s crust, is a naturally occurring metalloid with a wide range of concentrations in different environmental media. It generally mobilizes in the environment through both natural processes and anthropogenic activities (Adriano 2001). Elevated As concentrations in the environment, especially in the air, soil, groundwater, and food, have become a global issue with respect to environmental and public health concerns (Chung et al. 2014; Flora 2015) due to their high toxicity and lethal effects. Acute exposure of As is quite rare and mainly occurs from the accidental ingestion of insecticides or pesticides and even less commonly from suicidal or homicidal ingestion (Ratnaike 2003; ASTDR 2014). Acute As poisoning depends on the dose of exposure, and its symptoms include nausea, vomiting, abdominal pain, gastrointestinal hemorrhage, and severe diarrhea (ASTDR 2014). Meanwhile, chronic As exposure has caused evident negative effects on health to a broad range of internal organs and systems in a human body, such as the skin, lungs, liver, kidney, bladder, and prostate, as well as the nervous, cardiovascular, respiratory, gastrointestinal, immune, and endocrine systems (Kapaj et al. 2006; Naujokas et al. 2013; Sohn 2014). The negative health outcomes of chronic As exposure can include noncarcinogenic and carcinogenic effects. The toxicity of As in terms of noncarcinogenic and carcinogenic health effects is well studied and extensively reported by many studies (Tchounwou et al. 2003; Kapaj et al. 2006; Hughes et al. 2011; Naujokas et al. 2013; Hong et al. 2014; Mazumder 2015).
Among the three environmental exposure pathways, a main route of chronic As exposure is ingestion. There is a strong relationship between chronic As ingestion and negative effects on health (Naujokas et al. 2013). Apart from drinking water, food is the other major source of As for human exposure (IARC 2012; Chung et al. 2014; Flora 2015). Approximately 40% of As in the human body comes from the food chain (Flora 2015). It was also reported that rice is a principal source of As, especially inorganic As exposure, in the general population living in the non-As endemic regions (Sohn 2014; Flora 2015; Lai et al. 2015).
As a source of dietary carbohydrates, micronutrients, vitamins, and amino acids, rice has become one of the world’s most popular foods (Food and Agriculture Organization of the United Nations 2004; Rohman et al. 2004). The theme “rice is life” launched by the Food and Agriculture Organization (FAO) of the United Nations in 2004 has, to date, truly confirmed the importance of rice as a basic food commodity for more than half of the world’s population (Food and Agriculture Organization of the United Nations 2004; OECD-FAO 2008; Jeong et al. 2017). Unfortunately, rice generally takes up and accumulates As concentrations approximately ten times higher than those in other food grains such as maize and barley (Williams et al. 2007; Sohn 2014). Thus, risks of As exposure and its poisoning through rice consumption have been a public health concern for several decades. To ensure the safe exposure of As from rice consumption, many scientific studies were conducted to determine the levels of As in rice grains collected from both local markets and paddy fields worldwide. This chapter, therefore, deals with the occurrence of As in rice grains from the perspectives of market basket and field studies.
2 Total Global Rice Consumption
Besides wheat, rice is another one of the world’s most important cereal grains. It is a staple food for more than half of the world’s population (IRIN 2010; Jeong et al. 2017). The global rice production has been annually increased by approximately 1.61% during the last 20 years (OECD-FAO 2008). In addition, it is expected that the total global rice consumption will increase from 439 million tons in 2010 to 555 million tons by 2035 (Jeong et al. 2017). In terms of consumption, the current percentage of global rice consumption to the major cereal grains is approximately 35% (Fig. 1a). Meanwhile, approximately 49% of the cereal grains consumed in Asia are rice. Figure 1b clearly confirms that rice is the most important staple food for the Asian population.
Percentages of major cereal grain consumption in the (a) world and (b) Asia. (Note: raw data on the amount of consumption was obtained from OECD-FAO (2017))
Figure 2 shows the average annual rice consumption in the world and Asia. It is projected that the global and Asian rice consumption per capita from 2017 to 2026 will increase by approximately 0.10–0.24% and 0.28–0.43%, respectively. The average annual rice consumption per capita in the Asian population is approximately 1.5 times higher than the global rice consumption. This high amount of rice consumption has caused a significant concern, especially when the rice is contaminated with highly toxic elements such as As. Therefore, several international and national organizations have established the maximum levels of As in rice and rice commodities to protect the population from the potential adverse consequences on public health.
Projections of the global and Asian rice consumption per capita. (Note: Raw data on the consumption per capita was obtained from OECD-FAO (2017))
3 International and National Standards of As in Rice
The toxicity of As in humans is a high concern of many international and national organizations that perform work related to food safety. The maximum levels of As in rice and some rice commodities were set to ensure the safe exposure level and reduce the potential negative health outcomes in the general population and some susceptible population such as infants, toddlers, and young children. The standard levels of As in rice and commodities are summarized in Table 1. Even though both organic (monomethylarsonic acid (MMA) and dimethylarsinic acid (DMA)) and inorganic (arsenite (As(III)) and arsenate (As(V))) As species are commonly present in rice, most of the standards generally regulate only the level of inorganic As in rice because inorganic As species are more toxic than the organic As species. In principle, the inorganic As is the summation of As(III) and As(V).
As seen from Table 1, those countries such as European countries, China, and South Korea where the analysis of inorganic As is reliable, the inorganic As level has been adopted into the regulation. In the case of countries with limitations to inorganic As analysis, the Codex Alimentarius Commission has agreed that they may use the total As concentration as a screening level for the inorganic As in rice. If the total As concentration is below the regulated maximum level for inorganic As, no further testing is required, and the sample is determined to be compliant with the Codex Alimentarius Commission regulation. If the total As concentration is above the regulated maximum level for inorganic As, follow-up testing should be conducted to determine if the inorganic As is above the maximum level.
4 Market Basket Studies of Total As in Rice
The total concentration of As that is equal to the concentrations of both organic and inorganic As in rice is more frequently reported and widely available, especially in less developed countries, since the determination of the total As concentration requires more simple digestion and extraction methods, a less advanced analytical technique, less time consumption, and more affordable costs of analysis. It is worth noting that this chapter mainly concentrates on the most updated official reports and scientific publications on the total As concentration in raw rice during the last decade (2009–2018), as shown in Table 2, to comply with the Codex standards for early public health protection. In addition, the concentration of As can be changed after various methods of cooking (Kollander and Sundstrom 2015); therefore, this part of the study is only focused on the total As content in raw rice prior to any preparation and cooking processes.
The significant number of official reports and scientific publications on the total As concentration in rice grain published in the last 10 years has strongly confirmed the importance of the As contamination in rice issue. It is getting more attention from the developed countries, and more efforts on the monitoring of As contamination in rice have been continuously raised in developing and less developed countries. Table 2 summarizes the results of the market basket surveys on the total As concentrations in rice without taking into account the variation in the crop production year, countries of production, collection, and distribution. All reported values are the levels of total As in the raw rice sold in grocery stores, local markets, supermarkets, commercial food stores, and online shopping stores in 36 countries covering all of the world regions. Two main types of rice for direct human consumption, namely, polished (white) rice and husked (brown) rice, were reported.
4.1 Total As Concentration in Polished Rice
The total As concentrations in the commercial polished (white) rice from different countries as shown in Table 2 varied greatly (nondetectable to 1.170 mg kg−1). The highest total As concentration was found in rice collected from Pakistan. Since the Codex Alimentarius Commission has agreed to the use of the total As concentration as a screening level of inorganic As in rice, the maximum and mean As concentrations in rice reported in Table 2 were compared to the Codex maximum standard of the As in polished rice (0.2 mg kg−1). Considering the maximum total As concentration, the As concentrations in the rice from most countries (30/36 countries) were higher than that of the Codex standard.
The range of the mean total As concentrations in the raw polished rice collected from all 36 countries around the globe ranged from 0.010 to 0.726 mg kg−1. The mean total As concentrations in the rice from Kazakhstan (0.266 mg kg−1), Myanmar (0.250 mg kg−1), India (0.283 mg kg−1), Pakistan (0.430 mg kg−1), Turkey (0.323 mg kg−1), the United Arab Emirates (0.726 mg kg−1), Finland (0.250 mg kg−1), and Italy (0.210 mg kg−1) were higher than the Codex standard. With the exception of the United Arab Emirates, the levels of As in rice from these countries fall within the category of high As concentration in rice (>0.202–0.357 mg kg−1) as prescribed in the study on global normal levels of total As in rice grain (>0.082–0.202 mg kg−1) by Zavala and Duxbury (2008). According to this previous study, rice collected in the United Arab Emirates contained unusually high As content (>0.357 mg kg−1). Zavala and Duxbury (2008) concluded that the levels of high (>0.202–0.357 mg kg−1) and unusually high (>0.357 mg kg−1) As in rice are indications of rice production in an As-contaminated environment. Similarly, to the sources of As contamination in the environment, As accumulation in rice can be caused by natural and anthropogenic sources. The natural sources of As in the rice production system are the levels of As in paddy soil and irrigation water (Sahoo and Kim 2013; Ruangwises et al. 2014), the addition of As through flooding and the wet and dry atmospheric deposition (Meharg and Zhao 2012). Meanwhile, the anthropogenic sources of As in paddy rice are point and nonpoint sources of industrial and urban pollution and the usage of As-bearing fertilizers and arsenical pesticides (Meharg and Zhao 2012). In the case of the United Arab Emirates (Shirwaikar et al. 2013) (Table 2), rice that contained unusually high As concentrations were imported from the United States (0.925 ± 0.017 mg kg−1), Egypt (0.786 ± 0.065 mg kg−1), and India (Kolkata, 0.808 ± 0.018 mg kg−1, and South India, 0.721 ± 0.056 mg kg−1). This finding is in accordance with Meharg and Zhao (2012), who stated that the higher mean total As concentration was generally found in rice cultivated from the more industrialized countries such as the United States. Meanwhile, the mean total As concentrations in rice from Egypt and India as shown in Table 2 were in contrast to the mean total As concentrations in rice from India (0.07 mg kg−1) and Egypt (0.05 mg kg−1) that were reported by Meharg et al. (2009). This outcome might be due to the significantly smaller number of rice samples collected from the United Arab Emirates (n = 3 for rice imported from each country) and those reported by Meharg et al. (2009) (n > 100), as well as the influence of samples with high As levels.
Apart from those eight countries (Kazakhstan, Myanmar, India, Pakistan, Turkey, the United Arab Emirates, Finland, and Italy) in which rice sold in the country contained higher total As concentrations than the global normal level, rice available in the markets of most remaining countries in Table 2 contained typical concentrations of As in rice grain (>0.082–0.202 mg kg−1). Zavala and Duxbury (2008) have compiled the total As concentrations in rice from different world regions and considered the level of As in rice of >0.082–0.202 mg kg−1 as the global normal As content in rice produced from the environment without As contamination. The levels of total As in rice in these particular countries (Table 2) are also in good agreement with those total As concentrations in the rice of the same countries reported during the last two decades (1990s–2009) (Rahman et al. 2014).
As clearly shown in Table 2, the total As in rice from the countries with less industrialization and urbanization processes such as Malawi, Nigeria, and Sri Lanka was relatively low (ranging from 0.01 to 0.059 mg kg−1). Zavala and Duxbury (2008) have classified the level of As in rice less than 0.082 mg kg1 as an unusually low As content in the grain.
4.2 Total As Concentration in Husked Rice
Total As concentrations ranging from <0.003 to 0.739 mg kg−1 were found in the commercial husked (brown) rice (Table 2). The maximum total As concentration in the rice collected from half of the countries that were surveyed exceeded the Codex maximum standard of the As in husked rice (0.35 mg kg−1). The mean total As concentrations in rice from most of the countries are in the category of high As in rice grain (>0.202–0.357 mg kg−1), as determined by Zavala and Duxbury (2008).
Comparing the total As concentration in polished and husked rice from the same country of study (Table 2), the concentrations of the total As in husked rice were approximately, on average, 1.7-fold higher than its total concentration in polished rice. The level of As in the husked rice collected in Malawi was even up to four times higher than that of polished rice. The localization of As, which is mainly distributed between the husk and the endosperm (starchy grain), is the main reason for the significantly higher As concentration in husked (brown) rice than the polished (white) rice. Meharg et al. (2008) and Lombi et al. (2009) found that the As in the rice grain usually localizes within the pericarp and aleurone layers, which are the layers that give the rice grain its brown color. Meharg et al. (2008) further concluded that As, especially As(III), in rice basically has a high affinity for the protein and nonprotein with the thiol (–SH) functional group, such as phytochelatin and glutathione. Thus, the higher content of As was found in the husked rice which normally contains approximately 14.5–24.1% higher protein than the polished rice (Juliano 1993). The examples of the thiol-containing protein in husked rice are cysteine and methionine, which account for up to 9.1% and 10.2%, respectively, of the amino acids content in rice. Thiamine is an example of a thiol-containing vitamin in husked rice grain (Juliano 1993).
5 Market Basket Studies of Inorganic As in Rice
Regarding its high and lethal toxicity, As is one of the WHO’s ten chemicals of major public health concern. It is well known that inorganic As (As(III) and As(V)) is more toxic than organic As (MMA and DMA). The International Agency for Research on Cancer (IARC) has classified As and its inorganic compounds as Group 1 human carcinogens. Although low levels of inorganic and organic As (generally less than 0.25 mg kg−1) have been found in most foodstuffs, one’s daily intake can be influenced by not only the level of As in food but also the quantity of the food consumed. The IARC (2012) has estimated that approximately 25% of daily dietary As intake is inorganic As. In addition, the ingestion of 70–180 mg of inorganic As could result in human death (Ruangwises et al. 2014). Since the daily rice consumption per capita is even as high as 422 g day−1 (OECD-FAO 2018), a concern regarding the exposure of As (especially inorganic As) to the level that may cause detrimental effects to an individual has been raised. This concern resulted in the establishment of the maximum level of inorganic As in rice, as summarized in Table 1.
5.1 Inorganic As Concentration in Polished Rice
Levels of inorganic As in the commercial polished rice grain (Table 3) from various countries were highly variable in the range of 0.002–0.699 mg kg−1. The highest inorganic As, which was approximately 3.5-folds higher than the Codex maximum standard of inorganic As in polished rice (0.2 mg kg−1), was detected in the Turkish polished rice grain. Apart from Turkish rice, the maximum inorganic As in rice from Canada (0.343 mg kg-1), Argentina (0.221 mg kg−1), Brazil (0.218 mg kg−1), Kazakhstan (0.550 mg kg−1), China (0.211 mg kg−1), Japan (0.221 mg kg−1), India (0.576 mg kg−1), and Finland (0.280 mg kg−1) was higher than the Codex standard. Interestingly, rice from those abovementioned countries from America and Asian regions that contained a maximum total As concentration (Table 2) higher than the Codex standard also contained inorganic As exceeding the Codex standard (Table 3). Nonetheless, the total As concentration in rice from all European countries was lower than the Codex standard, and only the maximum inorganic As in rice from Finland was found to be higher than the Codex standard. In general, Meharg and Zhao (2012) found a highly significant relationship between the total and inorganic As concentrations in rice (r2 = 0.768, p < 0.001). Therefore, it is possible to estimate the inorganic As concentration in grain as 0.01 + 0.54 times the total As concentration.
The mean concentrations of inorganic As in the polished rice grain ranged from 0.022 to 0.217 mg kg−1 (Table 3). Only the rice from Canada and Kazakhstan were found to have mean inorganic As concentrations higher than the Codex standard (0.2 mg kg−1). Table 3 shows that the inorganic As in rice grain varies between countries and regions. The rice grain from African countries such as Egypt and Nigeria had the lowest mean inorganic As concentrations. This outcome is in accordance with the low level of total As concentrations in rice from those countries, as reported in Table 2. By comparing the concentrations of inorganic As in rice from this study (Table 3) to those same countries reported during the last two decades (1990s–2009) (Rahman et al. 2014), similar ranges of inorganic As in rice from Italy, Spain, Thailand, and the United States were found. However, rice grain from China and India in this study contained 1.3- to 2.6-fold higher and 4.9- to 6.5-fold lower inorganic As concentrations than those reported by Rahman et al. (2014).
Regarding the percentage of inorganic As concentration (Table 3) to its total concentration (Table 2) in commercial rice, the percentages of inorganic As to total As varied from 0.4% to 100%. The lowest percentage of inorganic As to total As was found in Italian rice. This outcome was in good agreement with Williams et al. (2005) and Meharg et al. (2009), who reported great differences in the As speciation in the rice produced from different parts of the world. On average, 63.3% of the total As in rice grain in this study (Tables 2 and 3) is inorganic As. Meanwhile, organic As generally represents approximately 15.1–62.4% (mean: 36.7%) of the total concentration. Since it was reported by several researchers that DMA is the dominant organic As species in rice grain (Zavala et al. 2008; Meharg et al. 2009; Meharg and Zhao 2012; Rahman et al. 2014), rice can then be classified into two types: the DMA type and the inorganic As type. Rice with a concentration of inorganic As higher than DMA can be classified as the inorganic As type and vice versa.
It is clearly shown in Fig. 3 that rice from most countries comprise the inorganic As type with the average percentages of inorganic As to total As ranging from 50.3% to 84.9%. The percentages of inorganic As in rice grains were inconsistent with the countries and regions of this study. For example, rice from Kazakhstan, Japan, and Thailand contain 43.9–45.5% (Tattibayeva et al. 2016), 75–100% (Naito et al. 2015), and 42.5–96.1% (Ruangwises et al. 2012; Nookabkaew et al. 2013a), respectively, of inorganic As to total As. This outcome means that even though the rice is produced within one particular country, it is possible to find both types (DMA and inorganic As) in the study.
Regarding Fig. 3, rice from the United States, Brazil, China, and a few European countries including Italy and Spain can be classified as DMA rice type. Williams et al. (2005) and Meharg and Zhao (2012) found that the rice produced in the United States and European countries had high percentages of DMA. In contrast, Rahman et al. (2014) found that Australian and American rice mostly contained DMA, while European and Asian rice mainly contained inorganic As. Figure 3 accords with those previous studies in that the rice from the United States is predominant with DMA, while the rice from Asian and European countries and Australia are dominated by inorganic As. Zavala et al. (2008) concluded that the accumulation of different As species in rice grain is mainly controlled by the genetic features of rice rather than the environmental conditions, such as climate conditions, the water management regime, and the As speciation in the soil and soil solution.
5.2 Inorganic As Concentration in Husked Rice
The concentrations of inorganic As in commercial husked rice varied from 0.015 to 0.450 mg kg−1 (Table 3). The maximum inorganic As concentrations in Kazakh and Japanese rice were approximately 1.2 times higher than the Codex standard of inorganic As in husked rice of 0.35 mg kg−1. The mean inorganic As concentrations in rice from most of the countries surveyed except for the Kazakh rice were lower than the Codex standard. The concentrations of inorganic As in husked rice were higher than the corresponding inorganic As concentrations in the polished rice by approximately 1.8 times. This comparison has a similar magnitude to the total As concentration in the husked rice compared to that of the polished rice.
The ratios of inorganic As to total As in rice also varied widely. The highest ratio of inorganic total As in rice was found in Malawian rice (95%) (Adeyemi et al. 2017). The ratios of inorganic As in Asian rice were 90% for Kazakhstan (Tattibayeva et al. 2016), 68–88% for China (Liang et al. 2010; Li et al. 2015; Ma et al. 2017), 55.1–91.3% for South Korea (Yim et al. 2017; Lee et al. 2018), 40.5–83.7% for Thailand (Nookabkaew et al. 2013a), and 67.5–77.9% for Vietnam (Nookabkaew et al. 2013a). Asian rice usually contained higher inorganic As than American and European rice. By comparing it to the total As concentration, commercial husked rice from Belgium, Spain, France, and Switzerland had approximately 62.5%, 52.0%, 62%, and 75.6%, respectively, of inorganic As in its grain (Jitaru et al. 2016; Signes-Pastor et al. 2016; Ruttens et al. 2018). Meanwhile, the lowest percentages of inorganic As to total As were generally found in the rice from the American region including the United States (53.2–67.2%) (Trenary et al. 2012; U.S. Food and Drug Administration 2016) and Brazil (53.9–54%) (Batista et al. 2011).
The classification of husked rice regarding the presence of inorganic and organic As in rice is shown in Fig. 4. This result is in good agreement with the polished rice in that rice from the American region is generally the DMA type while Asian and European rice are considered as the inorganic rice type. A comparison between Fig. 4 and Fig. 3 agrees well with the results reported by Meharg et al. (2008) in which the higher inorganic As proportion is generally present in husked rice rather than polished rice. Carey et al. (2010) studied the spatial unloading of As species in the grain and found the dispersion of As(III), which is one of the inorganic As species, in the external parts of the grain (rice bran). Conversely, the DMA was generally dispersed in the endosperm (the starchy part of the grain) (Carey et al. 2012). Approximately 70–80% of As(III) was found in the pericarp and aleurone layer, which is called rice bran after polishing (Meharg et al. 2008). In contrast, about half of the total As in the polished rice was As(III) (Meharg et al. 2008).
6 Bioaccessible As in the Commercial Rice
The determination of total and inorganic As is of significant interest in terms of the food safety for countries around the world for several decades. However, during the past few years, the study of the bioaccessibility of As in rice grain has been under the spotlight because it is believed that both the total and inorganic As concentrations in rice grain could not represent the actual amount of As that is readily absorbed by the human body after passing through the digestive system. Therefore, the determination of the bioaccessible As using the in vitro digestion model has been introduced. This in vitro digestion, which is a simplified version of the digestive system of the human body, principally includes the sequential steps of food digestion in the oral cavity by saliva, the stomach by gastric juice, and the small intestine by intestinal fluid. The bioaccessibility refers to the maximum concentration of the contaminant that is released from the food into the gastrointestinal media and readily available for absorption into the bloodstream (Ruangwises et al. 2014).
The bioaccessibility of As in the certified reference material of rice (CRM-T07151QC) was studied by Chavez-Capilla et al. (2016). Approximately 80 ± 1% of the bioaccessible As to the total As in the rice after passing through the simulated gastrointestinal digestion was found. Regarding the total As concentration, approximately 50.0% and 35.7% of bioaccessible As were released from rice in the stomach and small intestine, respectively.
In comparison to the bioaccessibility of As in the reference material, He et al. (2012) purchased locally grown polished (n = 8) and husked (n = 9) rice samples from food stores in New York City and analyzed the bioaccessible As, and its speciation in those samples had a total As concentration higher than the Codex standard (0.2 mg kg−1). The in vitro digestion system in this study mimics the stomach and small intestine human digestion. The concentrations of bioaccessible As were 0.27–0.32 mg kg−1 in polished rice (n = 5; mean, 0.29 ± 0.05 mg kg−1) and 0.13–0.62 mg kg−1 in husked rice (n = 6; mean, 0.32 ± 0.05 mg kg−1). They found higher As bioaccessibility in the polished rice (92.6%) than in the reference material (80.0%) reported by Chavez-Capilla et al. (2016). The DMA was the predominant As species in the polished rice. Meanwhile, 66.2%, on average, of the total As in husked rice was bioaccessible As. The higher bioaccessible inorganic As was found in four husked rice samples. The ratios of bioaccessible inorganic As to total As in both types of rice (40.2 ± 7.4% for polished rice and 57.0 ± 15.7% for husked rice) were in the same magnitude of those ratios of inorganic As to total As in raw rice, as shown in Figs. 3 and 4.
On a contrary, Althobiti et al. (2018) studied the bioaccessible As in the polished and husked rice sold in Canada and found a lower mean level of bioaccessible As in polished (n = 7, 60.5 ± 25.6%) and husked (n = 3, 61.9 ± 21.8%) rice. Wide ranges of bioaccessible As to total As in both types of rice (16.0–93% in polished rice and 43.4–86.0% in husked rice) were reported. Compared to the stomach digestion, higher bioaccessible As species including As(III), As(V), and DMA were released into the saliva than the gastric juice. Interestingly, the most prevalent species was As(V).
In the case of the bioaccessible As in rice sold in Asia, Zhuang et al. (2016) analyzed the bioaccessible As in commercial and greenhouse-grown polished rice following two steps of gastric and small intestine digestion. High As bioaccessibilities were found in all rice samples with the highest bioaccessibility found in the greenhouse-grown rice (94.5%). The commercial rice sold in the local markets around the mining area contained higher bioaccessible As (88%) than that of the rice sold in the supermarket in South China (68.5%). An increase in bioaccessible As was found when comparing the bioaccessible As concentration between the gastric and gastrointestinal digestion steps. In contrast to Althobiti et al. (2018), this study concluded that the gastrointestinal phase plays an important role in the solubilization of As during the digestion process.
Meanwhile, Li et al. (2018) determined the As bioaccessibility in 70 Eastern Chinese rice samples after three digestive steps (oral, stomach, and intestine) and reported lower levels of bioaccessibility (n = 70, 37.9 ± 7.9%) than those levels in the rice collected in the United States (He et al. 2012) and Canada (Althobiti et al. 2018).
For Southeast Asian countries, Hensawang and Chanpiwat (2018) determined the bioaccessible As concentration in polished and husked rice sold in the local markets in Bangkok. Three simultaneous simulated human digestive steps (oral, stomach, and small intestine) were applied to dissolve the bioaccessible As from the rice grain. The concentrations of bioaccessible As in polished (n = 32, 0.026–0.134 mg kg−1) and husked (n = 17, 0.099–0.196 mg kg−1) rice were well below the Codex standards for both types of rice. Approximately 2.2–3.4 times and 1.8–2.5 times of the bioaccessible As concentrations were lower than the total As in polished and husked rice, respectively. In addition, 1.5- to 3.8-fold significantly higher concentrations of bioaccessible As were determined in the husked rice than the polished rice. Even though wide variations in the bioaccessibility of As in both types of rice were found (8.7–76.3% for polished rice and 29.4–74.0% for husked rice), a significant difference in the mean As bioaccessibilities in both types of rice could not be observed.
7 Field Studies of Arsenic in Rice Grain
In contrast to the market basket studies in which most of the studies are usually focused on the levels of total, inorganic, and bioaccessible As in the rice grain that was sold and consumed by the general population, the field studies of As in rice grain were mainly conducted in the As-contaminated areas to (i) investigate the levels of As in rice grain, (ii) determine the relationship between the As concentrations in soils and rice grains, and (iii) determine the soil factors that could affect the level of As in rice grain. Most of the study areas can be classified into two different types according to the sources of As contamination based on whether it is a natural or anthropogenic source. As clearly shown in the following summary, different levels of total As in rice grain according to the category of normal (>0.082–0.202 mg kg−1), high (>0.357 mg kg−1), and unusually high (>0.202–0.357 mg kg−1) As as classified by Zavala and Duxbury (2008) were found from different studies.
The paddy soil and rice grain grown in the geogenic or mining-derived As origin in the Iberian Peninsula were collected by Signes-Pastor et al. (2016). The range and mean total As concentrations were 2.3–17 mg kg−1 and 8.7 mg kg−1, respectively. The highest mean total As was found in the Portuguese soil (15 mg kg−1). However, the inorganic As concentrations in dehusked rice (0.052–0.161 mg kg−1) were lower than the Codex standard. The rice grown in this region was the inorganic As rice type. The range and mean percentage of inorganic As to total As were 41–97% and 85%, respectively. The relationship between the total As in soil and the inorganic As in rice followed a hyperbolic pattern and approached a maximum level of more than 0.100 mg kg−1. In other words, a moderate level of inorganic As in rice was found when rice was cultivated in the soil with a high total As. In addition, this study found positive correlations between the total and inorganic As concentrations in locally grown and commercial rice with the levels of 0.98 and 0.76, respectively.
Another study was conducted in the paddy fields located near the industrial zone comprised of printing, dyeing, electroplating, electronics, metallurgy, chemicals, pesticides, and livestock and poultry breeding farms in the Yangtze River Delta (Hang et al. 2009). A total of 155 samples of each paddy soil and the corresponding rice grains were collected. The total As concentrations in the soil ranged from 4.46 to 16.10 mg kg−1 (mean: 8.60 mg kg−1). The highest level of As in the soil was higher than the background value of As in soil (15 mg kg−1) that was regulated by the National Environmental Protection Agency. A wide variation in the total As in polished rice grain within the range of nondetectable to 0.587 mg kg−1 was observed. However, the mean total As concentration in rice (0.199 mg kg−1) was lower than the Codex standard. A bioaccumulation factor or a ratio between the total concentrations of As in rice grain and the corresponding soil in this study was approximately 0.025. A spatial distribution analysis of the As concentration in soil indicated that a high As concentration in soil was well matched to the locations of the industrial zone and agricultural area with pesticide applications. In contrast, high concentrations of As in the rice grain were distributed around the city. The different spatial distribution patterns of As in the soil and rice indicated that the As accumulation in rice was not only affected by concentration of As in the soil but also by the soluble concentration fraction in the deposited air pollutant.
In Brazil, rice samples were collected from the fields with high application rates of phosphate fertilizers for at least 8 years (Corguinha et al. 2015). The levels of As in the agricultural soils (11 ± 0.5 mg kg−1) were not significantly different from the noncultivated soils (12 ± 2.0 mg kg−1) and were lower than the Brazilian maximum allowable concentration in agricultural soils. Even though rice has a high potential to accumulate greater amounts of As than other crops, this study found a nondetectable level of total As (<0.015 mg kg−1) in all rice samples (n = 60).
For those natural sources of As contamination in the paddy fields, the As accumulation in rice grains as a result of cultivation with As-contaminated irrigation water and soil was reported. The total As concentrations in the rice grain cultivated in the western part of Bangladesh varied from 0.03 to 1.84 mg kg−1 (Rajmohan and Prathapar 2014). High As in the rice was generally correlated with high As in the soil. The levels of As concentrations in rice were in the order of husk>bran>husked rice>polished rice. Approximately 44–86% of the total As in Bangladeshi rice was inorganic As.
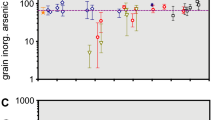
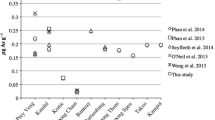
Phan et al. (2013) investigated the total As concentration in the paddy rice irrigated with groundwater from the As-contaminated areas in the Kandal and Kampong Cham provinces in Cambodia. There were significantly higher total As concentrations in both paddy soil and rice collected from Kandal (soil, 12.858 ± 10.430 mg kg−1, rice, 0.247 ± 0.187 mg kg−1) than those from Kampong Cham (soil, 0.794 ± 0.088 mg kg−1, rice, 0.029 ± 0.024 mg kg−1). The mean total As concentration in rice from Kandal (n = 8, 0.247 ± 0.187 mg kg−1) was greater than the Codex standard of 0.2 mg kg−1 because rice in Kandal was irrigated by As-contaminated groundwater (n = 46, 846 ± 298 μg L−1). Meanwhile, the groundwater (n = 18) in Kampong Cham contained only 1.28 ± 0.58 μg L−1 of total As. A high significant relationship between levels of As in the soil and rice grain was observed (r2 = 0.826, p < 0.01). In 2014, Seyfferth et al. (2014) investigated the As concentrations in paddy soil and rice in the major rice-growing regions of Cambodia (Kandal, Prey Veng, Battambang, Banteay Meanchey, and Kampong Thom). The total As concentrations in husked rice ranged from 0.10 to 0.37 mg kg−1, which exceeded the Codex maximum level of As in husked rice of 0.35 mg kg−1. However, the average As concentration (0.20 ± 0.06 mg kg−1) in rice grain was within the Codex standard. The rice produced from Banteay Meanchey contained significant higher As concentrations than those rice that were grown in the other areas. It was interesting to see that the total As in rice grain grown in Kandal, which is one of the most severely As-impacted areas in Cambodia, was not significantly higher than the rice grown in other areas, even though total As concentrations in the soil from Kandal and Prey Veng were significantly higher than the soil collected from Banteay Meanchey and Battambang. The total As concentration in the soil ranged from 0.8 to 18 mg kg−1. In contrast to a significant relationship between the As in the soil and rice grain reported by Phan et al. (2013), Seyfferth et al. (2014) could not observe significant relationships between either the total or CaCl2 extractable As concentrations, which are readily available concentrations for plants, to the As level in rice grain. However, they found that the As concentrations in husk and straw were important predictors of the As in rice grain.
In the case of Bolan et al. (2017), a study was conducted under either the greenhouse or field conditions to study the As accumulation in rice. Rice was irrigated with water containing various As (supplied as Na2HAsO4·7H2O) levels. The total As contents in rice ranged from 0.456 to 1.095 mg kg−1. Rice cultivation with As-contaminated water was a reason for the total As in rice exceeding the Codex standard. The total As concentrations in rice grains were significantly correlated with its concentrations in both irrigation water and soil. This study further determined the levels of gastric bioaccessible As in rice grain. The percentage of As bioaccessibility ranged from 23.2% to 32.3%.
References
Adeyemi JA, Adedire CO, Martins-Junior AC, Paulelli AC, Awopetu AF, Segura FR, de Oliveira-Souza VC, Batista BL, Barbosa F (2017) Arsenic speciation in rice consumed in south-western Nigeria, and estimation of dietary intake of arsenic species through rice consumption. Toxicol Environ Chem 99(5–6):999–1006
Adomako EE, Williams PN, Deacon C, Meharg AA (2011) Inorganic arsenic and trace elements in Ghanaian grain staples. Environ Pollut 159:2435–2442
Adriano DC (2001) Trace elements in terrestrial environments: biogeochemistry, bioavailability, and risk of metals, 2nd edn. Springer-Verlag, New York, p 867
Agency for Toxic Substances & Disease Registry (ASTDR) (2014) Toxic substances portal: arsenic trioxide. https://www.atsdr.cdc.gov/MMG/MMG.asp?id=1200&tid=279. Accessed 20 Jul 2018
Ahmed MK, Shaheen N, Islam MS, Habibullah-Al-Mamun M, Islam S, Islam MM, Kundu GK, Bhattacharjee L (2016) A comprehensive assessment of arsenic in commonly consumed foodstuffs to evaluate the potential health risk in Bangladesh. Sci Total Environ 544:125–133
Althobiti RA, Sadiq NW, Beauchemin D (2018) Realistic risk assessment of arsenic in rice. Food Chem 257:230–236
Antoine JMR, Fung LAH, Grant CN, Dennis HT, Lalor GC (2012) Dietary intake of minerals and trace elements in rice on the Jamaican market. J Food Compos Anal 26:111–121
Batista BL, Souza JMO, Souza SSD, Barbosa FJ (2011) Speciation of arsenic in rice and estimation of daily intake of different arsenic species by Brazilians through rice consumption. J Hazard Mater 191(1–3):342–348
Bolan S, Kunhikrishnan A, Chowdhury S, Seshaadri B, Naidu R, Ok YS (2017) Comparative analysis of speciation and bioaccessibility of arsenic in rice grains and complementary medicines. Chemosphere 182:433–440
Canadian Food Inspection Agency (2014) Food safety action plan report (2011–2013 Targeted surveys – chemistry): arsenic speciation in selected foods. Food Safety Division, Canadian Food Inspection Agency, Ottawa
Cano-Lamadrid M, Munera-Pizaco S, Burlo F, Hojjati M, Carbonell-Barrachina AAC (2015) Total and inorganic arsenic in Iranian rice. J Food Sci 80(5):T1129–T1135
Carey AM, Scheckel KG, Lombi E, Newville M, Choi Y, Norton NJ, Charnock JM, Feldmann J, Price AH, Meharg AA (2010) Grain unloading of arsenic species in rice. Plant Physiol 152:309–319
Carey AM, Lombi E, Donner E, de Jonge MD, Punshon T, Jackson BP, Guerinot ML, Price AH, Meharg AA (2012) A review of recent developments in the speciation and location of arsenic and selenium in rice grain. Anal Bioanal Chem 402:3275–3286
Chaleshtori FS, Kopaei MR, Chaleshtori RS (2016) A review of heavy metals in rice (Oryza sativa) in Iran. Toxin Rev 36(2):147–153
Chavez-Capilla T, Beshai M, Maher W, Kelly T, Foster S (2016) Bioaccessibility and degradation of naturally occurring arsenic species from food in the human gastrointestinal tract. Food Chem 212:189–197
Chung JY, Yu SD, Hong YS (2014) Environmental source of arsenic exposure. J Prev Med Public Health 47:253–257
Codex Alimentarius Commission (1995) Codex standard 198–1995L standard for rice. http://www.fao.org/fao-who-codexalimentarius/sh-proxy/ru/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FStandards%252FCODEX%2BSTAN%2B198-1995%252FCXS_198e.pdf. Accessed 2 May 2018
Codex Alimentarius Commission (2014) Joint FAO/WHO food standards programme (Codex Alimentarius Commission) 37th session Geneva, Switzerland, 14–18 July 2014 and report of the eighth session of the Codex committee on contaminants in foods, The Hague, The Netherlands, 31 March-4 April 2014. http://www.codexalimentarius.org/download/report/906/REP14_CFe.pdf. Accessed 2 May 2018
Codex Alimentarius Commission (2016) Joint FAO/WHO food standards programme (Codex Alimentarius Commission) 39th session Rome, Italy, 27 June–1 July 2016 and report of the 10th session of the Codex committee on contaminants in foods, Rotterdam, The Netherlands, 4–8 April 2016. http://www.fao.org/fao-who-codexalimentarius/sh-proxy/zh/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FMeetings%252FCX-735-10%252FReport%252FREP16_CFe.pdf. Accessed 2 May 2017
Codex Alimentarius Commission (2017) Joint FAO/WHO food standards programme (Codex Alimentarius Commission) 40th session ICCG, Geneva, Switzerland, 17–22 July 2017 and report of the 11th session of the Codex committee on contaminants in foods, Rio de Janeiro, Brazil, 3–7 April 2017. https://www.livsmedelsverket.se/globalassets/om-oss/samarbeten/codex-alimentarius/rapport-fran-cccf-mote-april-2017-.pdf. Accessed 3 May 2017
Corguinha APB, de Souza GA, Goncalves VC, Carvalho CA, de Lima WEA, Martins FAD, Yamanaka CH, Francisso EAB, Chulherme LRG (2015) Assessing arsenic, cadmium, and lead contents in major crops in Brazil for food safety exposure. J Food Compos Anal 37:143–150
EU Commission Regulation (2015) Commission regulation (EU) 2015/1006 of 25 June 2015 amending regulation (EC) No. 1881/2006 as regards maximum levels of inorganic arsenic in foodstuffs. http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32015R1006. Accessed 2 May 2018
Fang Y, Sun X, Yang W, Ma N, Xin Z, Fu J, Liu X, Liu M, Mariga AM, Zhu X, Hu Q (2014) Concentrations and health risks of lead, cadmium, arsenic, and mercury in rice and edible mushrooms in China. Food Chem 147:147–151
Flora SJS (2015) Arsenic: chemistry, occurrence, and exposure. In: SJS F (ed) Handbook of arsenic toxicology, 1st edn. Academic, San Diego, pp 1–49
Food and Agriculture Organization of the United Nations (2004) Rice is life. http://www.fao.org/newsroom/en/focus/2004/36887/index.html. Accessed 2 Apr 2018
Food Standards Australia New Zealand (2017) Arsenic. http://www.foodstandards.gov.au/consumer/chemicals/arsenic/Pages/default.aspx. Accessed 2 May 2018
Fransisca Y, Small DM, Morrison PD, Spencer MJS, Ball AS, Jones OAH (2015) Assessment of arsenic in Australian grown and imported rice varieties on sale in Australia and potential links with irrigation practices and soil geochemistry. Food Chem 147:147–151
Ghazanfarirad N, Dehghan K, Fakhernia M, Rahmanpour F, Bolouki M, Zeynali F, Asadzadeh J, Bahmani M (2014) Determination of lead, cadmium and arsenic metals in imported rice into the west Azerbaijan province, northwest of Iran. J Nov Appl Sci 3(5):452–456
Gilbert PJ, Polya DA, Cooke DA (2015) Arsenic hazard in Cambodian rice from a market-based survey with a case study of Preak Russey village, Kandal Province. Environ Geochem Health 37(4):757–766
Guillod-Magnin R, Bruschweiler BJ, Aubert R, Haldimann M (2018) Arsenic species in rice and rice-based products consumed by toddlers in Switzerland. Food Addit Contam Part A 35(6):1164–1178
Halder D, Biswas A, Slejkovec Z, Chatterjee D, Nriagu J, Jacks G, Bhattacharya P (2014) Arsenic species in raw and cooked rice: implications for human health in rural Bengal. Sci Total Environ 497–498:200–208
Hang Z, Wang H, Zhou J, Ma C, Du C, Chen X (2009) Risk assessment of potentially toxic element pollution in soils and rice (Oryza sativa) in a typical area of the Yangtze river delta. Environ Pollut 157:2542–2549
He Y, Pedigo CE, Lam B, Cheng Z, Zheng Y (2012) Bioaccessibility of arsenic in various types of rice in an in vitro gastrointestinal fluid system. J Environ Sci Health B 47:74–80
Hensawang S, Chanpiwat P (2017) Health impact assessment of arsenic and cadmium intake via rice consumption in Bangkok, Thailand. Environ Monit Assess 189(11):599
Hensawang S, Chanpiwat P (2018) Analysis and probabilistic risk assessment of bioaccessible arsenic in polished and husked jasmine rice sold in Bangkok. Chemosphere 207:637–648
Hong YS, Song KH, Chung JY (2014) Health effects of chronic arsenic exposure. J Prev Med Public Health 47(5):245–252
Hughes MF, Beck BD, Chen Y, Lewis AS, Thomas DJ (2011) Arsenic exposure and toxicology: a historical perspective. Toxicol Sci 123(2):305–332
IARC (2012) IARC monographs on the evaluation of carcinogenic risks to humans: volume 100C (arsenic and arsenic compounds). http://monographs.iarc.fr/ENG/Monographs/vol100C/mono100C-6.pdf. Accessed 9 Jan 2018
IRIN (2010) Key facts about rice. http://www.irinnews.org/report/91012/asia-key-facts-about-rice. Accessed 2 May 2018
Islam MS, Ahmed MK, Habibullah-Al-Mamun M, Raknuzzaman M (2015) The concentration, source and potential human health risk of heavy metals in the commonly consumed foods in Bangladesh. Ecotoxicol Environ Saf 122:462–469
Islam MS, Ahmed MK, Habibullah-Al-Mamun A, Eaton DW (2017a) Arsenic in the food chain and assessment of population health risks in Bangladesh. Environ Syst Decis 37(3):344–352
Islam MS, Ahmed MK, Habibullah-Al-Mamun M, Eaton DW (2017b) Geographical variation and age-related dietary exposure to arsenic in rice from Bangladesh. Sci Total Environ 601–602:122–131
Jafari A, Kamrehie B, Ghaderpoori M, Khoshnamvand N, Birjandi M (2018) The concentration data of heavy metals in Iranian grown and imported rice and human health hazard assessment. Data Brief 16:453–459
Jeong JM, Kim EC, Venkatanagappa S, Lee JS (2017) Review of rice: production, trade, consumption and future demand in Korea and worldwide. Kor J Crop Sci 62(3):157–165
Jitaru P, Millour S, Roman M, Koulali KE, Noel L, Guerin T (2016) Exposure assessment of arsenic speciation in different rice types depending on the cooking mode. J Food Compos Anal 54:37–47
Joy EJM, Ander EL, Broadley MR, Young SD, Chilimba ADC, Hamilton EM, Watts MJ (2017) Elemental composition of Malawian rice. Environ Geochem Health 39:835–845
Juliano BO (1993) Rice in human nutrition. The International Rice Research Institute and Food and Agriculture Organizations of the United States, Rome. http://www.fao.org/docrep/t0567e/T0567E00.htm#Contents. Accessed 2 May 2018
Kapaj S, Peterson H, Liber K, Bhattacharya P (2006) Human health effects from chronic arsenic poisoning–a review. J Environ Sci Health A 41:2399–2428
Kollander B, Sundstrom B (2015) Inorganic arsenic in rice and rice products on the Swedish market 2015. Part I –a survey of inorganic arsenic (Rapport 16–205). Livsmedelsverket National Food Agency https://www.livsmedelsverket.se/globalassets/publikationsdatabas/rapporter/2015/a-survey-of-inorganic-arsenic-in-rice-and-rice-products-on-the-swedish-market-2015%2D%2D-part-1.pdf. Accessed 19 Jul 2018
Lai PY, Cottingham KL, Steinmaus C, Karaga MR, Miller MD (2015) Arsenic and rice: translating research to address health care providers’ needs. J Pediatr 167(4):797–803
Lee SG, Kim DH, Lee YS, Cho SY, Chung MS, Cho MJ, Kang YW, Kim HJ, Kim DS, Lee KW (2018) Monitoring of arsenic contents in domestic rice and human risk assessment for daily intake of inorganic arsenic in Korea. J Food Compos Anal 69:25–32
Li X, Xie K, Yue B, Gong Y, Shao Y, Shang X, Wu Y (2015) Inorganic arsenic contamination of rice from Chinese major rice-producing areas and exposure assessment in Chinese population. Science China Chem 58(12):1898–1905
Li T, Song Y, Yuan X, Li J, Ji J, Fu X, Zhang Q, Guo S (2018) Incorporating bioaccessibility into human health risk assessment of heavy metals in rice (Oryza sativa L.): a probabilistic-based analysis. J Agric Food Chem 66(2):5683–5690
Liang F, Li Y, Zhang G, Tan M, Lin J, Liu W (2010) Total and speciated arsenic levels in rice from China. Food Addit Contam Part A 27(6):810–816
Lombi E, Scheckel KG, Pallon J, Carey AM, Zhu YG, Meharg AA (2009) Speciation and distribution of arsenic and localization of nutrients in rice grains. New Phytol 184:193–201
Ma L, Wang L, Jia Y, Yang Z (2016) Arsenic speciation in locally grown rice grains from Hunan Province, China: spatial distribution and potential health risk. Sci Total Environ 557–558:438–444
Ma L, Wang L, Tang J, Yang Z (2017) Arsenic speciation and heavy metal distribution in polished rice grown in Guangdong Province, Southern China. Food Chem 233:110–116
Magamage C, Waidyaratna WHMCU, Dhanapala WPAP (2017) Determination of heavy metals in rice available in Kandy district, Sri Lanka. Ann Sri Lanka Dep Agric 19:351–368
Mazumder DNG (2015) Health effects chronic arsenic toxicity. In: SJS F (ed) Handbook of arsenic toxicology, 1st edn. Academic, San Diego, pp 137–177
Meharg AA, Zhao FJ (2012) Arsenic & rice, 1st edn. Springer, New York
Meharg AA, Lombi E, Williams PN, Scheckel KG, Feldmann J, Raab A, Zhu Y, Islam R (2008) Speciation and localization of arsenic in white and brown rice grains. Environ Sci Technol 42:1051–1057
Meharg AA, Williams PN, Adomako E, Lawgali YY, Deacon C, Villada A, Cambell RCJ, Sun G, Zhu YG, Feldmann J, Raab A, Zhao FJ, Islam R, Hossain S, Yanai J (2009) Geographical variation in total and inorganic arsenic content of polished (white) rice. Environ Sci Technol 43:1612–1617
Mohamed H, Haris PI, Brima EI (2017) Estimated dietary intakes of toxic elements from four staple foods in Najran city, Saudi Arabia. Int J Environ Res Public Health 14:1575
Mwale T, Rahman MM, Mondal D (2018) Risk and benefit of different cooking methods on essential elements and arsenic in rice. Int J Environ Res Public Health 15:1056
Naito S, Matsumoto E, Shinhdoh K, Nishimura T (2015) Effects of polishing, cooking, and storing on total arsenic and arsenic species concentrations in rice cultivated in Japan. Food Chem 168:294–301
Naujokas MF, Anderson B, Ahsan H, Aposhian HV, Graziano JH, Thompson C, Suk WA (2013) The broad scope of health effects from chronic arsenic exposure: update on a worldwide public health problem. Environ Health Perspect 121(3):295–302
Nawab J, Farooqi S, Xiaoping W, Khan S, Khan A (2018) Levels, dietary intake, and health risk of potentially toxic metals in vegetables, fruits, and cereal crops in Pakistan. Environ Sci Pollut Res 25(6):5558–5571
Nemati S, Mosaferi M, Ostadrahimi A, Mohammadi A (2014) Arsenic intake through consumed rice in Iran: markets role or government responsibility. Health Promot Perspect 4(2):180–186
Nookabkaew S, Rangkadilok N, Mahidol C, Promsuk G, Satayavivad S (2013a) Determination of arsenic species in rice from Thailand and other Asian countries using simple extraction and HPLC-ICP-MS analysis. J Agric Food Chem 61:6991–6998
Nookabkaew S, Rangkadilok N, Akib CA, Tuntiwigit N, Saehun J, Satayavivad J (2013b) Evaluation of trace elements in selected foods and dietary intake by young children in Thailand. Food Addit Contam Part B 6(1):55–67
OECD-FAO (2008) OECD-FAO agricultural outlook 2008–2017. http://argus.iica.ac.cr/Esp/organizacion/LTGC/Documentacion/BibliotecaVenezuela/Boletines/2008/n11/OECD-FAO-Agricultural%20Outlook.pdf. Accessed 2 Apr 2018
OECD-FAO (2017) OECD-FAO agricultural outlook 2017–2026, by commodity. http://stats.oecd.org/Index.aspx?datasetcode=HIGH_AGLINK_2017. Accessed 1 Apr 2018
OECD-FAO (2018) OECD-FAO agricultural outlook 2018–2027. https://stats.oecd.org/Index.aspx?datasetcode=HIGH_AGLINK_2018 Accessed 30 Jul 2018
Pasias IN, Thomaidis NS, Piperaki EA (2013) Determination of total arsenic, total inorganic arsenic and inorganic arsenic species in rice and rice flour by electrothermal atomic absorption spectrometry. Microchem J 108:1–6
Petursdottir AH, Friedrich N, Musil S, Raab A, Gunnlaugsdottir H, Krupp EM, Feldman J (2014) Hydride generation ICP-MS as a simple method for determination of inorganic arsenic in rice for routine biomonitoring. Anal Methods 6:5392–5396
Phan K, Sthiannopkao S, Heng S, Huoy L, Wong MH, Kim KW (2013) Arsenic contamination in the food chain and its risk assessment of populations residing in the Mekong river basin of Cambodia. J Hazard Mater 262:1064–1071
Raab A, Ducos SM (2016) Determination of inorganic arsenic in rice using IC-ICP-MS (application note 43255). Thermo Scientific. https://pdfs.semanticscholar.org/160a/0c72f5f2f308f8865808354143e020eff6cd.pdf. Accessed 20 Jul 2018
Rahman MA, Rahman MM, Naidu R (2014) Arsenic in rice: sources and human health risk. In: Watson RR, Preedy VR, Zibadi S (eds) Wheat and rice in disease prevention and health, 1st edn. Academic, San Diego, pp 365–375
Rajmohan N, Prathapar SA (2014) Extent of arsenic contamination and its impacts on the food chain and human health in the eastern Ganges Basin: a review. International Water Management Institute (IWMI)., 47 p. (IWMI Working Paper 161), Colombo. https://doi.org/10.5337/2014.224. Accessed 20 Jul 2018
Ratnaike RN (2003) Acute and chronic arsenic toxicity. Postgrad Med J 79(933):391–396
Rintala EM, Ekholm P, Koivisto P, Peltonen K, Venalainen ER (2014) The intake of inorganic arsenic from long grain rice and rice-based baby food in Finland – low safety margin warrants follow up. Food Chem 150:199–205
Rohman A, Helmiyati S, Hapsari M, Setyaningrum DL (2004) Rice in health and nutrition. Int Food Res J 21(1):13–24
Rowell C, Kuiper N, Al-Saad K, Nriagu J, Shomar B (2014) A market basket survey of As, Zn and Se in rice imports in Qatar: health implications. Food Chem Toxicol 70:33–39
Roya AQ, Ali MS (2017) Heavy metals in rice samples on the Torbat-Heidarieh market, Iran. Food Addit Contam Part B 10(1):59–63
Ruangwises S, Saipan P, Tengjaroenkul B, Ruangwises N (2012) Total and inorganic arsenic in rice and rice bran purchased in Thailand. J Food Prot 75(4):771–774
Ruangwises S, Saipan P, Ruangwises N (2014) Inorganic arsenic in rice and rice bran: health implications. In: Watson RR, Preedy VR, Zibadi S (eds) Wheat and rice in disease prevention and health, 1st edn. Academic, Cambridge, MA, pp 393–399
Ruttens A, Cheyns K, Blanpain AC, Temmerman LD, Waegeneers N (2018) Arsenic speciation in food in Belgium. Part 2: cereals and cereal products. Food Chem Toxicol 118:32–41
Sadee BA, Foulkes ME, Hill SJ (2016) An evaluation of extraction techniques for arsenic in staple diets (fish and rice) utilizing both classical and enzymatic extraction methods. Food Addit Contam Part A 33(3):433–441
Sahoo PK, Kim K (2013) A review of the arsenic concentration in paddy rice from the perspective of geoscience. Geosci J 17(1):107–122
Segura FR, Souza JMO, Paula ESD, Martin ACJ, Paulelli ACCP, Barbosa FJ, Babista BL (2016) Arsenic speciation in Brazilian rice grains organically and traditionally cultivated: is there any difference in arsenic content? Food Res Int 89(1):169–176
Seyfferth AL, McCurdy S, Schaefer MV, Fendorf S (2014) Arsenic concentrations in paddy soil and rice and health implications for major rice-growing regions of Cambodia. Environ Sci Technol 48:4699–4706
Shirwaikar A, Jacob S, Salman YG, Hamoudi NM, Chandrasekharan VR (2013) Preliminary investigation of arsenic in rice consumed in United Arab Emirates. Gulf Med J 2(S1):S146–S153
Shraim AM (2017) Rice is a potential dietary source of not only arsenic but also other elements like lead and chromium. Arab J Chem 10(S2):S3434–S3443
Signes-Pastor AJ, Carey M, Carbonell-Barrachina AA, Moreno-Jimenez E, Green AJ, Meharg AA (2016) Geographical variation in inorganic arsenic in paddy field samples and commercial rice from the Iberian Peninsula. Food Chem 202:356–363
Sirgist M, Hilbe N, Brusa L, Campagnoli D, Beldomenico H (2016) Total arsenic in selected food samples from Argentina: estimation of their contribution to inorganic arsenic dietary intake. Food Chem 210(1):96–101
Sohn A (2014) Contamination: the toxic side of arsenic. Nature 514:S62–S63
Sommella A, Deacon C, Norton G, Pigna M, Violante A, Meharg AA (2013) Total arsenic, inorganic arsenic, and other elements concentrations in Italian rice grain varies with origin and type. Environ Pollut 181:38–43
Tattibayeva D, Nebot C, Miranda JM, Cepeda A, Mateyev E, Erkebaev M, Franco CM (2016) A study on toxic and essential elements in rice from the Republic of Kazakhstan: comparing the level of contamination in rice from the European community. Environ Geochem Health 38(1):85–98
Tchounwou PB, Patlolla AK, Centeno JA (2003) Carcinogenic and systematic health effects associate with arsenic exposure – a critical review. Toxicol Pathol 31:575–588
Trenary HR, Creed PA, Young AR, Mantha M, Schwegel CA, Xue J, Kohan MJ, Herbin-Davis K, Thomas DJ, Caruso JA, Creed JT (2012) An in vitro assessment of bioaccessibility of arsenicals in rice and the use of this estimate within a probabilistic exposure model. J Expo Environ Epidemiol 22:369–375
U.S. Food and Drug Administration (FDA) (2016) Arsenic in rice and rice products risk assessment report. Center for Food Safety and Applied Nutrition, Food and Drug Administration, U.S. Department of Health and Human Services. https://www.fda.gov/downloads/food/foodscienceresearch/risksafetyassessment/ucm486543.pdf. Accessed 23 Jul 2018
United Nations (2018) Methodology: standard country or area codes for statistical use (M49). https://unstats.un.org/unsd/methodology/m49/. Accessed 20 Jul 2018
United States Department of Agriculture (2011) USDA foreign agricultural service (global agricultural information network: Israel food safety–heavy metals in foodstuffs. https://gain.fas.usda.gov/Recent%20GAIN%20Publications/Food%20Safety%20%E2%80%93%20Heavy%20Metals%20in%20Foodstuffs_Tel%20Aviv_Israel_1-18-2011.pdf. Accessed 3 May 2018
United States Department of Agriculture (2014) USDA foreign agricultural service (global agricultural information network: Peoples Republic of China’s maximum levels for contaminants in foods. https://gain.fas.usda.gov/Recent%20GAIN%20Publications/Maximum%20Levels%20of%20Contaminants%20in%20Foods%20_Beijing_China%20-%20Peoples%20Republic%20of_12-11-2014.pdf. Accessed 3 May 2018
Wang HS, Sthiannopkao S, Chen ZJ, Man YB, Du J, Xing GH, Kim KW, Mohamed Yasin M, Hashim JH, Wong MH (2013) Arsenic concentration in rice, fish, meat and vegetables in Cambodia: a preliminary risk assessment. Environ Geochem Health 35(6):745–755
Williams PN, Price AH, Raab A, Hossain SA, Feldmann J, Meharg AA (2005) Variation in arsenic speciation and concentration in paddy rice related to dietary exposure. Environ Sci Technol 39:5531–5540
Williams PN, Villada A, Deacon C, Raab A, Figuerola J, Green AJ, Feldmann J, Meharg AA (2007) Greatly enhanced arsenic shoot assimilation in rice leads to elevated grain levels compared to wheat and barley. Environ Sci Technol 41:6854–6859
Wong WWK, Chung SWC, Chan BTP, Ho YY, Xiao Y (2013) Dietary exposure to inorganic arsenic of the Hong Kong population: results of the first Hong Kong total diet study. Food Chem Toxicol 51:379–385
Yim SR, Park GY, Lee KW, Chung MS, Shim SM (2017) Determination of total arsenic content and arsenic speciation in different types of rice. Food Sci Biotechnol 26(1):293–298
Zavala YJ, Duxbury JM (2008) Arsenic in rice: I. Estimating normal levels of total arsenic in rice grain. Environ Sci Technol 42:3856–3860
Zavala YJ, Gerards R, Gurleyuk H, Duxbury JM (2008) Arsenic in rice: II. Arsenic speciation in USA grain and implications for human health. Environ Sci Technol 42:3861–3866
Zhuang P, Zhang C, Li Y, Zou B, Mo H, Wu K, Wu J, Li Z (2016) Assessment of influences of cooking on cadmium and arsenic bioaccessibility in rice, using an in vitro physiologically-based extraction test. Food Chem 213:206–214
Acknowledgments
This study was supported by the Office of International Affairs, the Chulalongkorn University Scholarship for Short-Term Research, and the Ministry of Science and Technology in the Republic of Korea through the International Environmental Research Institute (IERI) of Gwangju Institute of Science and Technology (GIST).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Chanpiwat, P., Stiannopkao, S., Ko, MS., Kim, KW. (2020). Global Market and Field Studies of Arsenic Accumulation in Rice. In: Srivastava, S. (eds) Arsenic in Drinking Water and Food. Springer, Singapore. https://doi.org/10.1007/978-981-13-8587-2_7
Download citation
DOI: https://doi.org/10.1007/978-981-13-8587-2_7
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-8586-5
Online ISBN: 978-981-13-8587-2
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)