Abstract
Air pollution is one of the most threatening challenges faced by humans. Rapid industrialization coupled with increase in vehicular emissions is responsible for degradation of air quality. Plants can be effectively used as bio-indicators for air quality. Air pollution tolerance index (APTI) is a parameter to assess the tolerance or sensitivity of a plant towards pollution. Ten common plant species have been selected from rural, urban and mine sites of Jharkhand. ANOVA showed no significant variation in APTI between different sites for monsoon and winter season at 0.5 level of significance. t-test showed significant seasonal variation in biochemical parameters of different species of a particular site. Anticipated performance index of the plants has also been calculated to assess the overall performance of a plant in a particular region. Azadirachta indica and Mangifera indica were found to be the good performers amongst the selected plants and Artocarpus heterophyllus to be the poor performer.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Air pollution can be defined as the introduction of chemicals, particulate matter and other biological matter into the atmosphere in such quantities and for such time duration that it causes harm and discomfort to humans, animals, plants and the surrounding environment [1]. Plants are an integral part of the ecosystem and are most likely to be affected by air pollution. Plants that are exposed to air pollution for an extended period of time show slow growth rate, and its physiology is affected thereby affecting the entire metabolism. Plants act as an effective indicator of air pollution. They absorb the gaseous air pollutants and act as natural cleansing agents of the environment. Bio-monitoring of air pollution by vegetation is a cost-effective method. The susceptibility of plants to pollution varies from species to species and can be measured by its biochemical parameters [2]. Air pollution tolerance index (APTI) and anticipated performance index (API) are two indexes which are useful to assess pollution tolerance of plants. The vegetation in urban areas has gained significance as it is an important indicator of air quality. Depending on the resistance of particular species to air pollutants, the level of pollution can be regulated by planting those specific plants in the affected area. It is possible that plants may store and absorb the pollutants and later release them. These processes include chemical transformation, accumulation and incorporation in the metabolic system [3]. Air pollution tolerance index (APTI) and anticipated performance index (API) methodology can be applied in any place in the world as they are based on biochemical parameters and universally relevant biological and socio-economic characters. Selected tree species are evaluated for biological and socio-economic parameters with various biochemical factors. These parameters are subjected to a grading scale to determine the anticipated performance index (API) of tree species [4]. This type of study will be essentially helpful for plantation purposes in urban and rural development programmes and green belt development. Plants with high APTI values indicate more tolerance towards pollution and hence can serve as sink of pollutants. Finally, plants also serve as bio-monitors of air quality which can be reflected from the APTI value of selected plants, without installing expensive monitoring equipment.
2 Materials and Methods
2.1 Sampling Sites
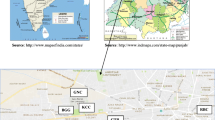
Jharkhand being a forest rich region has abundance of plant diversity. So far, air pollution tolerance index of plants of this region has not been reported. Three sites, namely Birla Institute of Technology campus in Mesra (rural site with lots of forest cover), Doranda region in Ranchi (urban site with high traffic density and one of the main commercial hub of the city) and Kuju region (coal mine site in Hazaribagh) have been taken for the study. The biochemical parameters studied were ascorbic acid, relative water content, pH, total chlorophyll. Reducing sugar and protein have also been studied for the winter season to conform the results (Fig. 1).
Leaves of ten common plant species were collected from all the three sites in early morning hours. The mature leaf samples of the most common plants in all the three regions such as Mangifera indica (mango), Ficus benghalensis (bargad), Dalbergia sissoo (sissoo), Shorea robusta (sal), Azadirachta indica (neem), Ficus religiosa (peepal), Bauhinia sp. (kachnar), Cassia fistula (amaltas), Neolamarckia cadamba (kadam) and Artocarpus heterophyllus (jackfruit) were taken. D. sissoo and N. cadamba were unavailable at the mine site, and the former was unavailable in the winter season due to leaf shedding.
Samples were immediately kept in zipper packets to avoid moisture loss. The collected samples were analysed for ascorbic acid, chlorophyll, relative water content and leaf extract pH. The mean values of different parameters were used for computing the index.
The various biochemical parameters are analysed by the following procedure given below.
2.2 Relative Water Content
Relative water content (RWC) was determined by following the method of Sen and Bhandari [5]. Individual leaves of different plant species were cleaned and weighed immediately. They were dipped into water in a beaker overnight. Next day, the leaves were blotted dry and reweighed before being dried at 80 °C for 24 h and reweighed. The following formula was used to calculate the relative water content
$$ {\text{RWC}} = \frac{{{\text{Fresh weight}} - {\text{Dry weight}}}}{{{\text{Turgid weight}} - {\text{Dry weight}}}}*100. $$
2.3 Total Chlorophyll
Total chlorophyll was estimated by the method given by Arnon [6]. For analysis, 1-g-fresh leaves material was grounded to paste in a mortar and then extracted with 20 ml of 80% acetone. The liquid portion was decanted into another test tube and centrifuged at 2500 rpm for 5 min. The supernatant was then collected, and the absorbance taken at 645 and 663 nm using a spectrophotometer. Calculations were done using the formula below
where
- D x :
-
Absorbance of the extract at the wavelength x nm.
- V :
-
Total volume of the chlorophyll solution (ml).
- W :
-
Weight of the leaf extracted (g).
2.4 Leaf Extract pH
1 g of leaf material was ground to paste and dissolved in 50 ml of deionized water, and leaf extract pH was measured by using calibrated digital pH metre [7].
2.5 Ascorbic Acid Content
The ascorbic acid content (mg/g) was determined using the modified calorimetric 2,6-dichlorophenol indophenol method given by Sadasivam [8]. 1-g sample is extracted in 4% oxalic acid and made up to a known volume (20 ml) and centrifuged at 4500 rpm for 5 min. 5 ml of this supernatant is pipette out, and 10-ml oxalic acid is added and titrated against the dye.
where
- V 1 :
-
Volume of dye titrated against the working standard.
- V 2 :
-
Volume of dye titrated against the sample.
2.6 Reducing Sugar Estimation
The reducing sugar content of the samples was determined by dinitrosalicylic acid method as developed by Miller [9]. In this method, the sugar was first extracted from 100 mg of the sample with hot 80% ethanol twice (5 ml each time). The contents were cooled, and the intensity of the dark red colour was recorded at 510 nm.
2.7 Protein Estimation
“Lowry’s method” was used for protein determination [10]. 500 mg of leaf sample was extracted with 10 ml of 0.2M phosphate buffer (pH—7.0). The extract was centrifuged and the supernatant collected. Folin–ciocalteu reagent was used for colour development. Blue colour is developed, and the readings are taken at 660 nm.
2.8 Statistical Analysis
t-test was done in order to find the seasonal variation of the biochemical parameters of each species. ANOVA was done using Excel Analysis ToolPak version 15.
Quality control was achieved by using analytical grade chemicals and by estimating the parameters in triplicates. Double distilled water was used for preparation of the reagents.
2.9 Air Pollution Tolerance Index (APTI)
APTI of a species was determined by using the formula established by Singh and Rao [11].
where
- A :
-
Ascorbic acid content (mg/g).
- T :
-
Total chlorophyll (mg/g).
- P :
-
Leaf extract pH.
- R :
-
Relative water content (%).
2.10 Anticipated Performance Index
By combining the resultant APTI values with some significant biological and socio-economic characters (plant habit, canopy structure, type of plant, economic value and laminar structure), the API was calculated for different species. Based on these characteristics, different grades (+ or −) are allotted to plants. Different plants are scored according to their grades. The criteria used for calculating the API of various plant species are done by referring to the method given by Prajapati [3]. The percentage scoring based on which API grades are calculated is given by the formula
Maximum number of grades that can be allotted to a plant = 16.
3 Results and Discussions
Ambient air quality characteristics for all the three regions are different. The average (24 h) SO2 concentration for rural, mine and urban site are 2.27, 11.11 and 37.71 µg/m3, respectively. The average (24 h) NO x concentration values for rural, mine and urban site are 13.86, 23.44 and 68.7 µg/m3, respectively. PM2.5 has an average (24 h) value of 7.67, 178.25 and 21.25 µg/m3 for rural, mine and urban site, respectively, and PM10 has an average (24 h) value of 10.56, 230.75 and 37.71 µg/m3 for rural, mine and urban site, respectively. The NAAQS values for industrial, residential and rural sites for SO2 is 50 µg/m3, NO x is 40 µg/m3, PM10 is 60 µg/m3 and PM2.5 is 40 µg/m3. The values for NO x for urban region are beyond the permissible limit. PM10 and PM2.5 values for mine site are very high and exceed the permissible limit.
3.1 Relative Water Content
Moisture content for all the plants have shown higher values in the monsoon season and lower in the winter season (refer Figs. 2, 3 and 4). This can be explained by the high water content in the soil and lower rate of transpiration from leaves in monsoon season. High water content in plants facilitates the plant to maintain its physiological balance under high-stress conditions [12]. The values range from 70 to 97% amongst the various species. Higher values of water content indicate more tolerant plants as they are more resistant to extreme conditions [13].
For the rural site, maximum values were shown by A. indica (95.2%) and C. fistula (97.03%) while for the mine site M. indica (95.28%) and F. benghalensis (93.18%) showed the highest values. M. indica (96.13%) and F. religiosa (95.52%) showed the highest values in urban site exhibiting higher tolerance.
3.2 Leaf Extract pH
Leaf extract pH values for all the sites increased in the winter season (refer Figs. 5, 6 and 7), the slightest increase indicated by M. indica (5.74–6.16) and A. indica (6.95–7.04) for rural site; M. indica (5.96–6.61) and A. indica (6.44–6.98) in mine site, whereas in the urban site, the slightest increase was exhibited by M. indica (5.72–6.71) and A. indica (6.55–7.03). The values indicated increasing pH in the winter season owing to the fact that in winter season there are stable atmospheric conditions which inhibits dispersion and makes more SPM settle on the leaves. During the monsoon season, the same SPM is washed off lowering the pH value [14]. The increase in pH is supported by the findings of Das [15] for A. indica (5.3–6.6) and M. indica (4.6–5.3). Scholz and Reck [16] reported that the higher decline in pH indicates sensitive species. Hence, minimum decline in pH indicates tolerant species which are listed above. Leaf extract pH showed significant variation in monsoon and winter season. ANOVA (Fcritical = 2.99, F = 7.91) results indicated significant site-wise as well as species-wise variation at 0.05 level of significance. The pH is seen to be increasing with increased pollution. The suspended particulate matter settles on the plants and consequently dissolves in the cell sap thereby making it acidic [17].
3.3 Ascorbic Acid Content
Ascorbic acid is a reducing agent which is activated as soon as the plant is under stress. It plays an important role in the cell wall synthesis, defence mechanism and the also in photosynthetic carbon fixation. The ascorbic acid is the most important factor in calculating the APTI due to its pivotal role in defence and detoxification mechanism and thus used as a multiplication factor in the formula [14].
The highest ascorbic acid values have been shown by C. Fistula, A. indica and M. indica in all the three sites (Figs. 8, 9 and 10). As pollution load is the lowest in the rural site, the ascorbic acid values have also shown lower values—5.27, 2.23, 5.33 mg/g, respectively. The maximum values for these plants in the urban site being 21.48, 7.48 and 9.71 mg/g, respectively. The mine site values have shown lower values indicating that these plants are not able to perform very well in that area owing to the very heavy pollution load with the values being 3.91, 2.25 and 2.85 mg/g, respectively. Ascorbic acid shows a positive correlation with SO2 and NO x . The production of reactive oxygen species (ROS) under pollution stress increases the production of ascorbic acid, which in turn prevents plants from oxidative stress by scavenging the ROS [18]. This might be one of the possible reasons of increased ascorbic acid levels in urban site as the SO2 and NO x concentration are much higher there as compared to the mine and rural site. Increasing ascorbic acid content with increasing pollution load is in agreement with the findings of [16, 19] for A. indica (5.91–11.33 mg/g) and M. indica (0.69–7.5 mg/g).
3.4 Total Chlorophyll Content
Table 1 shows the chlorophyll values in both monsoon and winter season. The maximum values have been shown by C. fistula and A. indica in all the rural and mine sites with the values being 4.16 and 3.51 mg/g in the rural site, 1.21 and 5.89 mg/g in the mine site, respectively. In the urban site Bauhinia sp. (2.71 mg/g) and M. indica (2.78 mg/g) showed the maximum values. t-test for seasonal variation showed variation indicating higher values in monsoon season and lower in winter season. ANOVA results did not show any significant variation species-wise and site-wise for monsoon and winter season. The lower values in winter season are due to the fact that dust is accumulated on the moist leaf surface during winter thereby inhibiting chlorophyll synthesis. Higher total chlorophyll values for monsoon season is supported by the findings of [3, 16, 20] for M. indica (1.49–2.41 mg/g), C. fistula (3.87 mg/g) and A. indica (6.8 mg/g) Also, the chlorophyll content was seen to be decreasing for the mine and urban site. This is because of the fact that chloroplast is the attacked by the air pollutants thereby decreasing its synthesis [21]. Higher chlorophyll values are an indicator of tolerance in plants [22].
3.5 Reducing Sugar and Protein Content
Reducing sugar is a source of energy for plants and is indicative of the physiological activity of plants [21]. Studies reveal that reducing sugar is accumulated in plants due to increase in pollution stress [23]. The values increased with increasing pollution load for F. religiosa, C. fistula, F. benghalensis, N. cadamba, S. robusta and M. indica (refer Figs. 11 and 12) in mine and urban site as compared to the rural site. Increased amounts in polluted sites in this study suggest that the plant is under stress. ANOVA (Fcritical = 3.88, F = 5.10) results for reducing sugar show significant variation site-wise. Protein shows increasing and decreasing trends when plants are under stress, and it depends upon the inherent capacity of the species [22]. In this study, the protein content showed increasing trend with increasing pollution load. One possible reason to this can be proline accumulation. Proline is accumulated when plants are subjected to stress. Due to this, protein degradation is slowed down [24]. Higher protein and reducing content in polluted site are also reported by Bakiyaraj and Ayyappan [15].
3.6 Air Pollution Tolerance Index
t-test showed significant seasonal variation in the APTI values as shown in Table 7. The values are seen to be increasing with the increase in the pollution load. The APTI value is highest for C. fistula and M. indica and lowest for A. heterophyllus and Bauhinia sp. The values for the APTI have been shown in Table 2. Higher APTI values with increase in pollution are in agreement with the researches of [19, 22, 25].
3.7 Anticipated Performance Index
API is a broader term which takes into consideration not only the biochemical parameters but also the socio-economic parameters giving a holistic perspective to it. Various plant parameters such as the leaf size, canopy structure also regulate the plant capacity to abate pollution. Greater leaf surface area expedites dust interception by direct impaction, and rough texture facilitates the dust particles to be captured by the leaves making the plant more efficient to function as a pollutant sink. According to the average APTI values of winter and monsoon season, the API values for the plants have been calculated for the urban and mine site. Table 6 shows which plant performs better which may be used for plantation purpose for developing an effective green belt around these highly polluted regions [22] (Tables 3, 4 and 5).
Table 6 shows the plant performance at various sites. M. indica is seen to be the best performer amongst the selected plant species. A. indica, F. religiosa and N. cadamba are good performers. It can be seen from the result that the same plant is performing differently in different sites which is due to the pollution characteristics of the area. As the mine site is very heavily polluted, the only good performer seen here is M. indica. More plant species can be assessed for this area apart from the selected ones for better performance.
From the study, it is evident that the overall performance of a plant does not depend on the biochemical parameters alone but the socio-economic parameters also play an important role. For example, C. fistula has the highest APTI values for both the regions but due to its socio-economic parameters, it has been categorized as a poor, very poor and moderate and performer in rural, mine and urban site, respectively. From Table 6, it can be seen that for the rural site M. indica and F. religiosa are the best performers and for the mine and urban site M. indica is the best performer and is recommended for plantation.
3.8 Statistical Analysis
Seasonal variation for the parameters has been statistically evaluated by the t-test (refer Table 7). All the parameters have shown significant variation. The parameter-wise seasonal variation has been explained earlier for all the parameters. APTI values have also shown significant variation. The APTI has increased for almost all the plants in the mine site and the rural site. The urban site values have shown decreasing trend from monsoon to winter season. M. indica, Bauhinia sp. and C. fistula exhibited higher values in the monsoon season than in winter season. All the other plant species showed higher values in the winter season owing to increased pollution concentration due to stable atmospheric conditions.
4 Conclusions
This study concluded that for any region APTI and API can prove to be very effective for development of green belt and mitigating the effect of pollution depending on the pollution characteristics of the area. APTI value was highest for C. fistula and M. indica and lowest for A. heterophyllus and Bauhinia sp. API assessment showed M. indica as the best performer and A. indica, F. religiosa and N. cadamba as good performers in all the three sites. The above species are recommended for social forestry, mine reclamation projects and urban planning as these have high APTI values as well as good socio-economic uses. C. fistula has high APTI value but due to its poor socio-economic parameters (type of plant, texture, economic use), the API is low for this species. Species with good API values provide good canopy cover and rough leaf texture for dust capturing, better habitat for wildlife. This method can prove to be an effective strategy for air pollution management without any expensive equipments.
References
Bernatzky, A.: Tree ecology and preservation. In: Development in Agricultural and Management Forest Ecology, vol. 2. Elsevier Scientific Publishing Co., New York (1978)
Singh, S.K., Rao, D.N., Agrawal, M., Pandey, J., Narayan, D.: Air pollution tolerance index of plants. J. Environ. Manage. 32, 45–55 (1991)
Prajapati, S.K., Tripathi, B.D.: Anticipated performance index of some tree species considered for green belt development in and around an urban area: a case study of Varanasi city, India. J. Environ. Manag. 88, 1343–1349 (2007)
Dhankhar, R., Mor, V., Lilly, S., Chopra, K., Khokhar, A.: Evaluation of anticipated performance index of some tree species of Rohtak city, Haryana, India. Int. J. Recent Sci. Res. 6(3), 2890–2896 (2015)
Sen, D.N., Bhandari, M.C.: Ecological and water relation to two Citrullus spp. In: Althawadi, A.M. (ed.) Indian Arid Zone, Environmental Physiology and Ecology of Plants, pp. 203–228 (1978)
Arnon, D.I.: Copper enzymes in isolated chloroplast polyphenol oxidase in Beta vulgaris. Plant Physiol. 24, 1–15 (1949)
Liu, Y.J., Ding, H.: Variation in air pollution tolerance index of plants near a steel factory: implications for landscape-plant species selection for industrial areas. WSEAS Trans. Environ. Dev. 4, 24–32 (2008)
Sadasivam, S., Manickam, A.: Biochemical Methods, 2nd edn. New Age International (P) Limited, New Delhi (1996)
Miller, G.L.: Use of DNS reagent for the determination of glucose. Anal. Chem. 31, 426–428 (1972)
Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J.: Protein measurement with the Folin phenol reagent. J. Biol. Chem, 293–265 (1951)
Singh, S.K., Rao, D.N.: Evaluation of plants for their tolerance to air pollution. Proceeding of Symposium on Air Pollution Control, vol. 1, pp. 218–224. Indian Association of Air Pollution Control, New Delhi (1983)
Agarwal, S., Tiwari, S.L.: Susceptibility level of few plants on the basis of air pollution tolerance index. Indian Forester, 319–322 (1997)
Keller, T.: The electrical conductivity of Norway spruce needle diffusate as affected by air pollutants. Tree Physiol. 1, 85–94 (1986)
Keller and Schwager: Air pollution and ascorbic acid. Eur. J. For. Pathol. 7, 338–350 (1977)
Bakiyaraj, R., Ayyappan, D.: Air pollution tolerance index of some terrestrial plants around an industrial area. Int. J. Mod. Res. Rev. 2(1), 1–7 (2014)
Das, S., Prasad, P.: Seasonal variation of air pollution tolerance indices and selection of plant species for industrial areas of Rourkela. IJEP 30(12), 978–988 (2010)
Scholz, F., Reck, S.: Effect of acids on forest trees as measured by titration in vitro, inheritance of buffering capacity in Piceaabies. Water Air Soil Pollut. 8, 41–45 (1977)
Bhattacharya, T., Kriplani, L., Chakraborty, S.: Seasonal variation in air pollution tolerance index of various plant species of Baroda city. Univ. J. Environ. Res. Technol. 3(2), 199–208 (2013)
Lakshmi, P.S., Sravanti, K.L., Srinivas, N.: Air pollution tolerance index of various plant species growing in industrial areas. Ecoscan 2, 203–206 (2008)
Rai, P.K., Panda, L.S.: Roadside plants as bio indicators of air pollution in an industrial region, Rourkela, India. Int. J. Adv. Res. Technol. 4(1) (2015). ISSN 2278-7763
Tripathi, A.K., Gautam, M.: Biochemical parameters of plants as indicators of air pollution. J. Environ. Biol. 28(1), 127–132 (2007)
Mondal, D., Gupta, S., Datta, J.K.: Anticipated performance index of some tree species considered for green belt development in an urban area. Int. Res. J. Plant Sci. 2(4), 099–106 (2011). ISSN: 2141-5447
Prado, F.E., Boero, C., Gallarodo, M., Gonzalez, J.A.: Effect of NaCl on germination, growth and soluble sugar content in Chenopodium quinoa wild seeds. Bot. Bull. Acad. Sin. 41, 27–34 (2000)
Thomas, H.: Accumulation and consumption of solutes in swards of Loliumperenne during drought and after rewatering. New Phytol. 118, 35–48 (1991)
Agbaire, P.O., Akporhonor, E.E.: The effects of air pollution on plants around the vicinity of the delta steel company, Ovwian-Aladja, Delta State, Nigeria. IOSR J. Environ. Sci. Toxicol. Food Technol. 8(7), 61–65 (2014). e-ISSN: 2319-2402, p-ISSN: 2319-2399
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Singapore Pte Ltd.
About this paper
Cite this paper
Jain, S., Bhattacharya, T., Chakraborty, S. (2019). Comparison of Plant Tolerance Towards Air Pollution of Rural, Urban and Mine Sites of Jharkhand: A Biochemical Approach to Identify Air Pollutant Sink. In: Kalamdhad, A., Singh, J., Dhamodharan, K. (eds) Advances in Waste Management . Springer, Singapore. https://doi.org/10.1007/978-981-13-0215-2_9
Download citation
DOI: https://doi.org/10.1007/978-981-13-0215-2_9
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-0214-5
Online ISBN: 978-981-13-0215-2
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)