Abstract
Glycerol fermentation redox routes by E. coli wild type batch cultures and effects of Ni2+, Fe2+ and Fe3+ ions of different concentration (0.01–0.3 mM) on cell growth and H2 production were investigated. Influences of aforementioned metal ions and Cu2+, as well as Fe2+ on H2 production rate in vitro assays were also studied in wild type and hyaB hybC double mutant (with defective hydrogenase (Hyd) 1 and Hyd -2). Cell growth was shown to be followed by decrease of pH and redox potential (ORP) measured by both titanium-silicate (Eh) and platinum electrodes (E′h). After 8 h growth, at pH 7.5, Eh dropped down negative value (−120 mV) and H2 production was observed at the middle log phase. Whereas at the same pH in the presence of 0.05 mM Fe2+ both Eh and Eh′ electrodes readings dropped to more negative values ~ −170 ± 10 and −450 ± 12 mV, respectively. All ions used at 0.05–0.1 mM concentrations stimulated bacterial growth ~1.2 to ~1.4 fold at different pHs. Ni2+ enhanced H2 formation in a concentration-dependent manner: maximal stimulation, up to 1.5 fold, was observed at 0.2 mM NiCl2 only at pH 7.5. Ni2+ also promoted to high H2 yield at pH 6.5 and 7.5 in vitro. In addition, 0.05–0.1 mM Fe2+ also affected on H2 production rate and increased it ~2 fold in vitro at pH 6.5 and 7.5 whereas, 0.05–0.1 mM Cu2+ had inhibitory effect on H2 production rate. In hyaB hybC mutant H2 production rate was decreased compared with wild type and Fe2+ had no effect on H2 production rate and yield both at pH 6.5 and 7.5.
The findings indicate the strengthening of reductive processes by E. coli during glycerol fermentation and point out the role of essential heavy metal ions in H2 production. Furthermore, enhanced H2 production by heavy metal ions probably depends on operation of Hyd- 1 and Hyd- 2 at pH 6.5 and 7.5.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
14.1 Introduction
Biohydrogen is known as ecologically-clean, renewable and abundant alternative energy source for the twenty-first century, because of its safety by-product (during its utilization produces only water) and high energy content (˜140 MJ/kg) [1, 2]. Nowadays many projects are focused on investigations of pathways for production and simulation of H2 during bacterial fermentative processes, besides, the use of cheap carbon sources for H2 production is more attractive and preferable. Gonzalez’s group [3] has discovered that Escherichia coli is able to utilize glycerol in a fermentative manner and produce ethanol with different organic acids as well as molecular hydrogen (H2). Crude glycerol is a main co-product of biodiesel, which is formed during the trans-etherification process of the triacylglycerols [4–6]. Biodiesel is derived from vegetable oil, algae, or animal fat. During the biodiesel production, for every 1 lb of oil converted, there will be approximately 0.1 lb of crude glycerol are produced as well [7]. So, with the increasing demand for biodiesel, the accumulation of glycerol as byproduct, made it cheaper than other carbon source like glucose. On the other hand, E. coli is the best-characterized bacterium and promising for glycerol utilization because it is one of the most commonly organisms used for metabolic engineering and industrial applications [6, 8–10].
H2 is established to be obtained from formate, by formate hydrogen lyase (FHL), with participation of formate dehydrogenase H (FDH-H) and hydrogenases (Hyd) [11]. The genome of E. coli encodes four membrane-associated [Ni-Fe]-Hyd enzymes [12]. Hyd-1 and Hyd-2 are thought hydrogen-oxidizing enzymes; on the other hand, Hyd-3 and Hyd-4 reduce protons to H2 and with the selenocysteine- and molybden cofactor-containing FDH-H and other electron-transferring components form the FHL complex. Participation of different Hyd enyzmes in H2 metabolism is dependent on medium properties and composition, especially pH and carbon source. Interestingly, H2 produced at acidic pH has negative impact on cell growth and glycerol fermentation [3, 8]. Recently it has been reported that, depending on medium composition, H2 could be also evolved at slightly alkaline pH [13, 14]. However the pH dependence of glycerol fermentation and H2 production is not clear.
Besides, bioenergy is frequently stored and released by means of redox reactions. The ability of bacteria to carry out redox reactions depends on the redox state of the environment, or its redox potential (ORP) [15]. The latter itself depends on rate of redox processes. This simple relationship is hard to understand and to apply in biotechnology. On the other hand, ORP is suggested to be useful for monitoring changes in the metabolic state of bacterial cultures in biotechnology and for optimizing yield of fermentation products [16, 17]. Moreover, ORP has been shown can be applied to discriminate among species of bacteria [18, 19].
No doubt, redox state of environment and growth medium’s composition are important factors for bacterial growth and H2 production: it is known that traces of metal ions such us Fe are Ni are necessary for growth and metabolism of most microorganisms. Moreover, Fe and Ni can stimulate activity of metal-containing enzymes [20, 21]. In some bacteria Fe and Ni are required for a catalyzing Fe-S clusters biogenesis [22]. At the same time, abundance of free Fe and Ni are lethal for bacteria [23, 24].
Interestingly, it is shown that under anaerobic conditions Fe2+ is stable and more soluble than Fe3+. The latter is, therefore, inaccessible for living organisms. Thus, bacteria should have many mechanisms to satisfy the requirement of Fe, with the help of various transport systems. Multiple Fe transport system has been identified in E. coli under anaerobic conditions [25]. Among them Fe2+ uptake system, encoded by three feoABC genes, is probably ATP-driven primary transporter. The other ion – Fe3+ is also accumulated by E. coli but together with siderophore. The latter’s complex with Fe3+ is bonded to specific proteins to pass through the membrane into the cells; those proteins are components of ABC-type transporters [26].
Although various types of transporters can be involved in Ni uptake under certain conditions, the biosynthesis of Ni-dependent enzymes depends on highly specific transport systems with an affinity of Ni at the very low concentration (nM range). Two major types can be distinguished from each other: an ABC-type Ni transporter has been identified in E. coli [20, 23]. The majority of Ni transporters are independent of ATP hydrolysis; they form of novel class – the Ni/Co transporters family [20].
Cu2+ are also required for bacterial metabolism in low concentration [27]; but in a considerably higher concentration as an oxidizer they may cause inhibition of bacterial growth, Hyd activity and change H+ flux through the F0F1-ATPase as shown with E. coli in our laboratory [28, 29] as well as disrupt the membrane by inducing permeability. Cu2+ is found can affect the other bacteria – Enterococcus hirae growth through Eh or directly on proteins in bacterial membrane, probably F0F1 [30].
In this paper, the effects of Ni2+, Fe2+ and Fe3+ on growth of bacteria and kinetics of ORP, H2 production by E. coli wild type during glycerol fermentation were studied at different pHs (pH 5.5–7.5). It was shown that 0.05–0.1 mM Fe2+ and Ni2+ both stimulated bacterial growth at all pHs and enhanced H2 production rate and yield both at pH 6.5 and 7.5. Inhibitory effect of oxidizer Cu2+ on H2 production was also shown at all pHs. Moreover, Fe2+ did not stimulate H2 production in hyaB hybC mutant with defective Hyd-1 and Hyd-2 at pH 6.5 and 7.5.
14.2 Materials and Methods
14.2.1 Bacterial Strain and Growth, pH Determination
The E. coli BW25113 (lacl q rrnB T14 ΔlacZ W116 hsdR514 ΔaraBAD AH33 Δrha BAD LD78 ) wild-type and MW1000 (BW25113ΔhyaB ΔhybC) mutant with defective Hyd-1 and Hyd-2 [14] were used.
Bacteria were grown in batch culture under anaerobic conditions at 37 °C in peptone medium (20 g/L peptone, 2 g/L K2HPO4, 5 g/L NaCl) with glycerol 10 g/L at different pHs. The pH was measured using a pH-meter with selective pH-electrode (ESL-63-07, Gomel State Enterprise of Electrometric Equipment (GSEEE), Gomel, Belarus; or HJ1131B, HANNA Instruments, Portugal) and adjusted by 0.1 M NaOH and 0.1 N HCl. Bacterial growth was monitored by measuring optical density (OD) with a spectrophotometer at the wavelength of 600 nm.
14.2.2 ORP Measurements and H2 Production Assays
ORP was measured during bacterial growth in peptone medium and in bacterial suspension in vitro assays. In vitro ORP was measured in the assays mixture, which contained 150 mM Tris-phosphate buffer, 1 mM NaCl and 1 mM KCl, 0.4 mM MgSO4; pH was adjusted by titration of 1 M H3PO4.
ORP was determined by use of redox platinum (Pt) (EPB-1, GSEEE; or PT42BNC, HANNA Instruments, Portugal) and titanium-silicate (Ti-Si) electrodes (EO-02, GSEEE); Ag/AgCl (saturated by KCl) electrode was as reference electrode. In contrast to Pt electrode is sensitive to H2 (or oxygen), Ti-Si electrode measures the overall ORP and is not affected by the presence of H2 (or oxygen); this is allowing H2 detection under anaerobic conditions (in the absence of oxygen) [14, 31]. H2 production rate (VH2) is expressed as difference between Pt (Eh′) and Ti-Si (Eh) electrodes readings in mV in time per mg dry weight. The H2 yield is calculated by the decrease of Eh′ to low negative values as described by Piskarev et al. [32]; it is expressed in mol/l. Note Eh′ decrease by H2 evolution did not depend on salt content in water solution; pH was not affected by H2 supplemented [32].
Before assays ORP of two electrodes were checked in the control solution (the mixture of 0.049 M K3[Fe(CN)6] and 0.05 M K4[Fe(CN)6]· 3H2O, pH 6.86) according to the manufacturer’s instructions. Eh′ and Eh at 25 °C were of 245 ± 10 mV. Note no significant differences between Eh′ and Eh were detected during H2 assays in bacterial suspension without carbon source added; bacterial count alteration in the suspension by ~8–10-fold had no marked effect on Eh′ and Eh [14]. Moreover, the determination used is closed to the method with Clark-type electrode employed by Noguchi et al. [33].
14.2.3 Others, Reagents and Data Processing
All assays were done at 37 °C. Agar, K3[Fe(CN)6], K4[Fe(CN)6] were from Sigma (USA), casein peptone, glycerol, Tris were from Carl Roths (Germany), glycerol was from Unichem (China); other reagents used were of analytical grade. Data were averaged from duplicate or triplicate independent measurements, for which the standard errors do not exceed 3 % (if they do not indicated).
14.3 Results and Discussion
14.3.1 Heavy Metal Ions Effects on E. coli Growth, ORP Kinetics and H2 Production
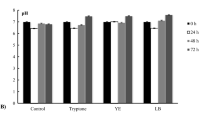
In this study, Ni2+, Fe2+ and Fe3+ effects on growth and ORP kinetics by E. coli BW25113 batch culture during glycerol fermentation at different pHs (5.5–7.5) were investigated. 0.01 up to 0.3 mM concentrations of metal ions were considered; optimal concentrations for bacterial growth were determined. 0.05–0.1 mM Ni2+, Fe2+ and Fe3+ in the growth medium was shown to stimulate bacterial growth up to ~1.2 to ~1.4 fold at different pHs (Fig. 14.1). These results are valuable, because glycerol fermentation by E. coli was relieved for the last years [3, 4] but its metabolism is complicated [34]; and medium composition optimal for bacterial growth are under the study.
The effects of Fe2+, Fe3+, Ni2+on E. coli BW25133 cell growth. Bacteria were grown under glycerol fermentation at pH 5.5–7.5. OD600 after 24 h bacterial growth was presented. Control was bacterial growth in the medium without metal ions added. 0.05 mM FeSO4, FeCl3 or NiCl2 were added into the growth medium (For details, see Sect. 2.2)
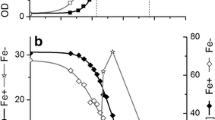
The lowering of medium pH and the drop of ORP was observed during bacterial growth with increase of bacterial count (OD). During glycerol fermentation by E. coli, Eh′ dropped down to −400 ± 12 mV (Fig. 14.2), −450 ± 10 mV and −350 ± 10 mV (not shown) when the culture was of 8 h growth at pH 7.5, 6.5 and 5.5, respectively. Eh with glycerol culture was of −50 to −100 ± 10 mV at pH 5.5–7.5 (not shown). The drop of ORP up to −550–600 mV during anaerobic growth of bacteria in the absence of external electron acceptors has been well demonstrated by many researchers [15, 19, 35–38]. This ORP drop might be a result of the secretion of redox active metabolites into the culture medium, leading to decrease of external pH [36, 37, 39] or might be connected with the processes on (in) the bacterial membranes [15, 40, 41]. Moreover, negative value of ORP with glycerol culture can be regarded by the properties of glycerol having highly reduced carbon and a higher production of reducing equivalents (NADH/NADPH/FADH2) during its fermentation compared to glucose [3, 4]. The reducing equivalents should have a profound effect on the whole metabolic network, when the higher NADH availability significantly changes the end products pattern under anaerobic conditions [4, 42].
The ORP kinetics by E. coli BW25113 during glycerol fermentation at pH 7.5. Eh′, measured by platinum and Eh by titanium-silicate electrodes were expressed in mV (vs Ag/AgCl (saturated by KCl)). Control was bacterial growth in the medium without metal ions. 0.05 mM Fe2+ was added into the growth medium (For details, see Sect. 2.2)
Interesting situation was observed with ORP kinetics during E. coli growth at pH 7.5 (Fig. 14.2): in the presence of Fe2+ both Eh and Eh′ dropped to more negative value −170 ± 10 and −450 ± 12 mV, respectively, compared with the control. Probably this effect can be regarded with the reductive properties of Fe2+, which under alkaline pH has specific action: Fe2+ may reduce thiol groups on bacterial surface affecting on activity of membrane transport systems or stimulating activity of key enzymes in fermentation metabolism, such as Hyd or proton-transporting F0F1-ATPase [29].
Consequently, H2 production was observed at the middle of log growth phase (Fig. 14.2, Table 14.1). Ni2+ stimulated H2 formation after 24 h growth in a concentration-dependent manner: maximal stimulation (up to ~1.5 fold) was observed at 0.2 mM NiCl2.
14.3.2 Metal Ions Effects on H2 Production Rate by E. coli Upon Glycerol Fermentation in vitro
In order to investigate metal ions effects, bacterial culture transferred into the assays mixture (see Materials and methods) was treated 2 min with appropriate compounds; then glycerol was added and ORP kinetics was determined.
In the presence of glycerol Eh and Eh′ drops were observed and H2 production by E. coli was detected in vitro (Table 14.1). 0.1 and 0.05 mM Fe2+ stimulated H2 production rate ~1.7 and 2 fold at pH 6.5 and 7.5, respectively (Fig. 14.3). Fe3+ in 0.05–0.1 mM concentrations had no or opposite, inhibitory effect (Fig. 14.3). Similar discrimination between Fe2+ and Fe3+ has been observed with Rhodobacter sphaeroides: Fe2+ ions affected on ORP stimulating both H2 formation and bacterial growth, while Fe3+ had effect only on bacterial [43]. Moreover, it has been shown that Fe3+ increased Enterococcus hirae specific growth rate and the F0F1-ATPase activity of membrane vesicles, whereas Fe2+ had opposite effects [24]. It was supposed that Fe2+ may affect directly on Hyd activity or on ORP, which by-turn can regulate F0F1 operation. Fe2+ and Fe3+are reducers and oxidizers, respectively, thus, the opposite effects of these ions are expected.
H2 production rate (V) by E. coli BW25113 during glycerol fermentation in vitro. Bacteria were grown and VH2 was measured in vitro at pH 5.5 (a), 6.5 (b) and 7.5 (c) upon glycerol fermentation. Appropriate metal ions at the concentrations mentioned on the figures were added into the assays mixture. Control was H2 production rate without metal ions added (For details, see Sect. 2.2)
Furthermore, Ni2+ have not affected on H2 production rate, but both Fe2+ and Ni2+ stimulated H2 yield ~5 and ~3 fold at pH 6.5, and ~1.1 and ~1.6 fold at pH 7.5, respectively (Table 14.1). It has been reported by Trchounian’s group [31, 44] that during sugar fermentation, E. coli possesses membrane protein-protein complex composed from F0F1, K+ transporter TrkA and FHL systems. This complex is responsible for H2 production; it is formed and operated mainly at slightly alkaline pH. Moreover, the energy within the complex is suggested can be transferred through a dithiol-dosulfide interchange. So reducers could regulate the complex activity as well as H2 production. The complex is not supposed to operate in E. coli during glycerol fermentation although relationship of F0F1 with Hyd enzymes has been suggested [14]. Probably, Fe2+ and Ni2+ could directly affect on Hyd activity, which are [Fe-Ni] containing enzymes, or having reducing properties they may affect on thiol-dithiol interchange and stimulate H2 production.
As mentioned above Cu2+ as oxidizers inhibited Hyd activity in bacteria [28, 45], as well as bacterial growth affecting the lag phase duration and decreasing specific growth rate [30]. Moreover, it has been established [28] that the addition of 0.1 mM to 2 mM Cu2+ into the E. coli growth medium results in a delayed decrease of Eh although a drop in Eh is less for rather 2 mM than 0.1 mM. All these findings can be taken into consideration to explain oxidizers effects mechanisms on E. coli. Thus, it was shown, that 0.05–0.1 mM Cu2+ suppressed H2 production rate at all pHs during glycerol fermentation in vitro (Fig 14.3). The obtained effect may be resulted by action of Cu2+ on ORP or directly affect on proteins in bacterial membrane, probably on Hyd or the F0F1-ATPase as was proposed [29].
It is of interest, that in hyaB hybC mutant during glycerol fermentation H2 production rate was ~2 fold lower (~1.1 mV/min/mg dry weight), compared with wild type, and Fe2+ did not stimulate H2 production rate and yield both in vitro at pH 6.5 and 7.5. Thus, it was supposed, that enhanced H2 formation observed depends on the operation of Hyd-1 and Hyd- 2, which is consistent with the results that during glycerol fermentation at alkaline pH for H2 formation is responsible Hyd-2 mainly and Hyd-1 [14].
14.4 Conclusions
Ni2+, Fe2+ and Fe3+ were shown to stimulate bacterial growth on glycerol. Ni2+ and Fe2+ enhanced H2 production rate and yield both by E. coli during glycerol fermentation especially at pH 6.5 and 7.5. In the presence of Fe2+ upon glycerol fermentation more reductive processes are induced, which by turn may affect on H2 formation. Increasing H2 production by heavy metal ions probably is regarded by the action of Hyd-1 or Hyd-2.
The results might be taken into account for optimizing fermentation processes on glycerol and developing H2 production biotechnology. The coupling of H2 production to utilization of waste materials containing high concentrations of glycerol may simultaneously provide economic and environmental benefits.
References
Momirlan M, Veziroglu TN (2005) The properties of hydrogen as fuel tomorrow in sustainable energy system for a cleaner planet. Int J Hydrog Energy 33:795–802
Maeda T, Sanches-Torres V, Wood TK (2008) Metabolic engineering to enhance bacterial hydrogen. Microb Biotechnol 1:30–39
Dharmadi Y, Murarka A, Gonzalez R (2006) Anaerobic fermentation of glycerol by Escherichia coli: a new platform for metabolic engineering. Biotechnol Bioeng 94:821–828
Yazdani S, Gonzalez R (2007) Anaerobic fermentation of glycerol, a path to economic viability for the biofuels industry. Curr Opin Biotechnol 18:213–221
da Silva GP, Mack M, Contiero J (2009) Glycerol: a promising and abundant carbon source for industrial microbiology. Biotechnol Adv 27:30–39
Khanna S, Goyal A, Moholkar VS (2012) Microbial conversion of glycerol: present status and future prospects. Crit Rev Biotechnol 32(3):235–262. doi:10.3109/07388551.2011. 604839
Dasari MA, Kiatsimkul PP, Sutterlin WR, Suppes GJ (2005) Low-pressure hydrogenolysis of glycerol to propylene glycol. Appl Cat Gen 281:225–231
Murarka A, Dharmadi Y, Yazmandi S, Gonzalez R (2008) Fermentative utilization of glycerol by Escherichia coli and its implications for the production of fuels and chemicals. Appl Environ Microbiol 74:1124–1135
Hu H, Wood TK (2010) An evolved Escherichia coli strain for producing hydrogen and ethanol from glycerol. Biochem Biophys Res Commun 391:1033–1038
Sarma SJ, Brar SK, Sydney EB, Bihan YL, Buelna G, Soccol CR (2012) Microbial hydrogen production by bioconversion of crude glycerol: a review. Int J Hydrog Energy 37:6473–6490
Sawers RG (2005) Formate and its role in hydrogen production in Escherichia coli. Biochem Soc Transact 33:42–46
Trchounian K, Poladyan A, Vassilian A, Trchounian A (2012) Multiple and reversible hydrogenases for hydrogen production by Escherichia coli: dependence on fermentation substrate, pH and the F0F1-ATPase. Crit Rev Biochem Mol Biol 47:236–249
Trchounian K, Trchounian A (2009) Hydrogenase 2 is most and hydrogenase 1 is less responsible for H2 production by Escherichia coli under glycerol fermentation at neutral and slightly alkaline pH. Int J Hydrog Energy 34:8839–8845
Trchounian K, Sanchez-Torres V, Wood TK, Trchounian A (2011) Escherichia coli hydrogenase activity and H2 production under glycerol fermentation at low pH. Int J Hydrog Energy 36:4323–4331
Vassilian A, Trchounian A (2009) Environment oxidation-reduction potential and redox sensing by bacteria. In: Trchounian A (ed) Bacterial membranes. Ultrastructure, bioelectrochemistry, bioenergetics and biophysics. Research Signpost, Trivandrum, pp 163–195
Kwong SCW, Rao G (1992) Effect of reducing agents in anaerobic amino acid fermentation. Biotechnol Bioeng 40:851–857
Riondet C, Cachon R, Waché Y, Alcaraz G, Divies C (2000) Extracellular oxidoreduction potential modifies carbon and electron flow in Escherichia coli. J Bacteriol 182:620–624
Brasca M, Morandi R, Lodi R, Tamburini A (2006) Redox potential to discriminate among species of lactic acid bacteria. J Appl Microbiol 103:1516–1524
Soghomonyan D, Akopyan K, Trchounian A (2011) pH and oxidation-reduction potential change of environment during growth of lactic acid bacteria: effect of oxidizers and reducers. Appl Biochem Microbiol 47:33–38
Andreini C, Bertini I, Cavallaro G, Holliday GL, Thornton JM (2008) Metal ions in biological catalysis: from enzyme databases to general principles. J Biol Inorg Chem 13:1205–1218
Mulrooney SB, Hausinger RP (2003) Nickel uptake and utilization by microorganisms. FEMS Microbiol Rev 27:239–261
Touati D (2000) Iron and oxidative stress in bacteria. Arch Biochem Biophys 373:1–6
Wu L-F, Navarro C, Pina K, Quenard M, Mandrand M-A (1994) Antagonistic effect of nickel on the fermentative growth of Escherichia coli K-12 and comparison of nickel and cobalt toxicity on the aerobic and anaerobic growth. Environ Health Perspect 102:297–300
Vardanyan Z, Trchounian A (2012) Fe (III) and Fe (II) ions different effects on Enterococcus hirae cell growth and membrane-associated ATPase activity. Biochem Biophys Res Commun 417:541–545
Kammler M, Schön C, Hantke K (1993) Characterization of ferrous iron uptake system of Escherichia coli. J Bacteriol 175:6212–6219
Ouyang Z, Isaacson R (2006) Identification and characterization of a novel ABC iron transport system in Escherichia coli. Infect Immun 74:6949–6956
Rensing C, Grass G (2003) Escherichia coli mechanisms of copper homeostasis in a changing environment. FEMS Microbiol Rev 27:197–213
Kirakosyan G, Trchounian A (2007) Redox sensing by Escherichia coli: effects of copper ions as oxidizers on proton-coupled membrane transport. Bioelectrochemistry 70:58–63
Kirakosyan G, Trchounian K, Vardanyan Z, Trchounian A (2008) Copper (II) ions affect Escherichia coli membrane vesicles’ SH-groups and a disulfide-dithiol interchange between membrane proteins. Cell Biochem Biophys 51:45–50
Vardanyan Z, Trchounian A (2010) The effects of copper (II) ions on Enterococcus hirae cell growth and the proton-translocating F0F1 ATPase activity. Cell Biochem Biophys 57:19–26
Poladyan A, Trchounian A (2009) Production of molecular hydrogen by mixed-acid fermentation in bacteria and its energetic. In: Trchounian A (ed) Bacterial membranes. Ultrastructure, bioelectrochemistry, bioenergetics and biophysics. Research Signpost, Trivandrum, pp 197–231
Piskarev M, Ushkanov VA, Aristova NA, Likhachev PP, Myslivets TS (2010) Establishment of the redox potential of water saturated with hydrogen. Biophysics 55:19–24
Noguchi K, Riggins DP, Eldahan KG, Kitko RD, Slonczewski JL (2010) Hydrogenase -3 contributes to anaerobic acid resistance of Escherichia coli. PLoS One 5:e10132
Cintolesi A, Clomburg JM, Rigou V, Zygourakis K, Gonzalez R (2012) Quantitative analysis of the fermentative metabolism of glycerol in Escherichia coli. Biotechnol Bioeng 109:187–198
Oblinger JL, Kraft AA (1973) Oxidation reduction potential and growth of Salmonella and Pseudomonas fluorescens. J Food Sci 38:1108–1112
Bagramyan K, Galstyan A, Trchounian A (2000) Redox potential is a determinant in the Escherichia coli anaerobic fermentative growth and survival: effects of impermeable oxidant. Bioelectrochemistry 51:151–156
Bagramyan K, Trchounian A (1997) Decrease of redox potential in the anaerobic growing Escherichia coli suspension and proton-potassium exchange. Bioelecrochem Bioenerg 43:129–134
Waché Y, Riondet C, Diviès C, Cachon R (2002) Effect of reducing agents on the acidification capacity and the proton motive force of Lactococcus lactis ssp. cremoris resting cells. Bioelectrochemistry 57:113–118
Oktyabrski ON, Sirnova GV (2012) Redox potentials in bacterial cultures under stress conditions. Microbiology 81:131–142
Michelon D, Abraham S, Ebel B, De Coninck J, Husson F, Feron G, Gervais P (2010) Contribution of exofacial thiol groups in the reducing activity of Lactococcus lactis. FEBS J 277:2282–2290
Kirakosyan G, Bagramyan K, Trchounian A (2004) Redox sensing by Escherichia coli: effects of dithiothreitol, a redox reagent reducing disulphides, on bacterial growth. Biochem Biophys Res Commun 325:803–806
Berrios-Rivera SJ, Sanchez AM, Bennett GN, San KY (2004) Effect of different levels of NADH availability on metabolite distribution in Escherichia coli fermentation in minimal and complex media. Appl Microbiol Biotechnol 65:26–432
Hakobyan L, Gabrielyan L, Trchounian A (2012) Discrimination between Fe (II) and Fe (III) effects on redox potential and biohydrogen photoproduction by Rhodobacter sphaeroides. ASM 112th General Meeting. San Fransico, USA, Q-2557
Trchounian A (2004) Escherichia coli proton-translocating F0F1-ATP synthase and its association with solute secondary transpopters and/or enzymes of anaerobic oxidation-reduction under fermentation. Biochem Biophys Res Commun 315:1051–1057
Sapra R, Bagramyan K, Adams MWW (2003) A simple energy-conserving system: proton reduction coupled to proton translocation. Proc Natl Acad Sci 100:7545–7550
Acknowledgments
The authors thank to Prof. T.K. Wood (Department of Chemical Engineering, Texas A & M University, College Station, TX, USA) for supplying strains and advice. This study was done in frame of Research grant to AT (#11-F-202) from Ministry of Education and Sciences of Armenia.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media Dordrecht
About this paper
Cite this paper
Poladyan, A., Trchounian, K., Minasyants, M., Trchounian, A. (2013). Glycerol Fermentation and Molecular Hydrogen Production by Escherichia Coli Batch Cultures Affected by Some Reducing Reagents and Heavy Metal Ions. In: Veziroğlu, A., Tsitskishvili, M. (eds) Black Sea Energy Resource Development and Hydrogen Energy Problems. NATO Science for Peace and Security Series C: Environmental Security. Springer, Dordrecht. https://doi.org/10.1007/978-94-007-6152-0_14
Download citation
DOI: https://doi.org/10.1007/978-94-007-6152-0_14
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-007-6151-3
Online ISBN: 978-94-007-6152-0
eBook Packages: EnergyEnergy (R0)