Abstract
Minimal change nephrotic syndrome (MCNS) is the most common cause of nephrotic syndrome in children and accounts for 10–25 % of nephrotic syndrome in adults. The etiology has been postulated as T lymphocytes (T cell) disorder; however, precise mechanisms still remain unknown. CD80 (also known as B7.1) is a transmembrane glycoprotein which is usually expressed on antigen-presenting cells (APC) and acts as a co-stimulatory signal for T cell activation. The role of CD80 expression in podocytes as a cause of proteinuric condition was first described by Reiser et al. in 2004. Administration of lipopolysaccharides (LPS) and puromycin aminonucleoside, knockout of alpha-3 integrin, and lupus nephritis were associated with CD80 induction in podocytes and concomitant proteinuria. Besides, LPS-induced proteinuria was not observed in CD80 knockout mice. In 2009, Garin et al. demonstrated that urinary CD80 levels are elevated in MCNS in relapse compared with those observed in remission and control subjects. Importantly, elevated urinary CD80 was suggested as a possible biomarker to distinguish MCNS and focal segmental glomerulosclerosis. Besides, polyinosinic:polycytidylic acid (polyI:C), ligand of Toll-like receptor 3 which mimics viral infection, induced CD80 in the podocytes. Thus, it is a possible explanation for the frequent relapses of MCNS after upper respiratory virus infections. LPS and polyI:C only induced transient proteinuria; therefore, impaired regulatory mechanisms after CD80 induction were postulated as a second hit cause of MCNS.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Co-stimulatory Pathway in the T Cell Activation
T cells require two signals to become fully activated. The first signal is an interaction of T cell receptor (TCR) with antigen displayed on the major histocompatibility complex (MHC) molecules on the surface of antigen-presenting cells (APC). The second signal is the co-stimulatory signal. Co-stimulatory molecule CD80 or CD86 (also known as B7.2) on the surface of APC binds CD28 or cytotoxic T-lymphocyte-associated protein 4 (CTLA-4, also known as CD152) expressed on the T cells. The first signal is antigen specific and the second one is antigen nonspecific. Here, the second signals differently act on T cells. (i) T cells are activated when CD80/CD86 on APC interact with CD28 on the T cells. (ii) T cells are inactivated when CD80/CD86 on APC interact with CTLA-4 on the T cells. (iii) The first signal without co-stimulatory signal leads to T cell anergy, namely, unresponsive state [1].
1.1 Co-stimulatory Molecule CD80 (B7.1)
CD80 and CD86 are members of immunoglobulin (Ig) superfamily with their extracellular regions consisting of two Ig-like domains, an NH2-terminal Ig variable-like domain (IgV) followed by a constant-like domain (IgC). Despite having only ~25 % sequence homology, CD80 and CD86 have similar receptor-binding properties [2]. Usually, CD80 is expressed on the activated B cells and APC such as monocytes, macrophages, and dendritic cells. Both molecules bind CTLA-4 and CD28 with much higher affinity with CTLA-4 [1].
There are several other co-stimulatory pathways. Inducible T cell co-stimulator (ICOS) on the T cell binds ICOS ligand on the APC, and programmed cell death protein-1 (PD-1) on the T cells binds programmed death-ligand 1 (PD-L1) or programmed death-ligand 2 (PD-L2) on APC. Thus, co-stimulatory pathway regulates the activation of T cells and prevents excess inflammation or autoreactive immune responses [3].
The expression of CD80 in the non-hematopoietic cells is relatively rare. In human keratinocytes, CD80 expression was upregulated by allergens or irritants [4]. Microglia in the brain and epidermal Langerhans cells also express CD80 on activation [5, 6].
Although the detailed difference between the role of CD80 and CD86 is not well elucidated, the role of CD80 has been postulated as a cause of proteinuric conditions, especially in MCNS [7–9].
1.2 Regulatory Mechanisms of CD80 Expression in the Immune Cells
The regulatory mechanisms of CD80 have been mostly investigated in the immune cells. Various immune stimuli such as anti-CD40 antibody, interferon (IFN)-γ, interleukin-2 (IL-2), IL-4, IL-5, and granulocyte-macrophage colony-stimulating factor (GM-CSF) and the binding of Toll-like receptor (TLR) with its ligands induce CD80 expression [10]. Meanwhile, IL-10 and CTLA-4 negatively regulate the expression of CD80 [5, 10, 11].
Regulatory T cells expressing forkhead box P3 (Foxp3) have an anti-inflammatory role by their enriched expression of CTLA-4 and by releasing anti-inflammatory cytokines such as IL-10 and transforming growth factor β (TGF-β) [12]. CTLA-4 competes with CD28 and preferentially binds with CD80/CD86, thus preventing T cell activation [12]. Besides, CTLA-4 suppresses the expression of CD80/CD86 in dendritic cells [13] and in B cells which leads to altered T cell-B cell interaction [14], CTLA-4 also has the ability to reduce CD80 in the opposing cells by trans-endocytosis [15]. CTLA-4 is expressed on the T cell; besides, soluble form (sCTLA-4) exists in the serum and has the ability to bind CD80/CD86 [11]. The expression of CTLA-4 becomes upregulated in the activated T cells; therefore, it is assumed that CTLA-4 helps the termination of the inflammation.
2 The Role of CD80 in the Proteinuric Conditions
2.1 Discovery of CD80 Induction in Podocytes as a Cause of Proteinuria
The role of CD80 in the proteinuric conditions was first described by Reiser et al. in 2004 [8]. α3β1-Integrin is thought to be largely responsible for the attachment of podocyte foot process to the glomerular basement membrane. α3β1-Integrin-deficient mice are unable to maintain normal podocyte structure [16]. The search for the changes in the α3 integrin-deficient podocytes unexpectedly led to the discovery of CD80 expression in the podocytes [17]. They demonstrated that CD80 can be induced in podocytes and transient proteinuria was observed concomitantly. In vivo administration of lipopolysaccharides (LPS) or puromycin aminonucleoside (PAN), genetic α3 integrin deficiency, and lupus nephritis in mice as well as in human were associated with increased CD80 expression in podocytes. Furthermore, CD80 expression in human lupus nephritis was associated with its severity. Interestingly, proteinuria was not induced in the CD80 knockout mice by LPS, but it was induced in the SCID mice which are immunodeficient lacking B cells and T cells, showing that CD80 is a key step, and this is independent of T cells and B cells. In vitro study using conditionally immortalized podocytes demonstrated that podocytes express CD14 and TLR4; both of them are the receptors which recognize LPS as ligands. Also, they described that LPS induced podocyte actin reorganization which is regarded as a change corresponding to the foot process effacement [8].
2.2 Urinary CD80 as a Possible Biomarker of MCNS
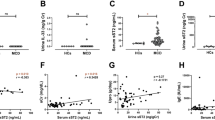
Then, Garin et al. suggested the role of urinary CD80 in MCNS for the first time in 2009 [7]. They showed that urinary CD80 levels are increased in MCNS in relapse compared with those observed in remission and control subjects. In patients with other glomerular diseases such as membranous nephropathy, lupus nephritis, IgA nephropathy, and focal segmental glomerulosclerosis (FSGS), urinary CD80 was not significantly increased compared with control subjects. They also showed that soluble CTLA-4 levels in the serum which usually regulate CD80 were not significantly altered, but the CD80/CTLA-4 ratio was increased in MCNS during relapse, with levels returning to normal range in remission. The serial measurements of urinary CD80 in recurrence and remission in the eight patients with MCNS revealed elevated levels of CD80 in recurrence and marked reduction in 1 month after remission.
In 2010, they showed that urinary CD80 is a useful biomarker to distinguish MCNS and FSGS [9]. Immunofluorescent staining showed that CD80 expression in the podocytes was positive in MCNS in relapse but not in remission nor in FSGS. Western blot of CD80 protein revealed that the molecular size of urinary CD80 was 53 kD, demonstrating its membrane-associated form. Soluble CD80 usually seen in the serum is 23 kD; therefore, it was suggested that urinary CD80 derives from podocytes.
3 Regulatory Mechanisms of CD80 Expression in the Podocytes
3.1 The Role of TLR in Innate Immunity
The host defense against microbial pathogens consists of innate immunity and acquired immunity. Innate immunity serves as the first-line defense which recognizes pathogen-associated molecular patterns (PAMPs). TLR is one of the pattern recognition receptor families that play a key role in innate immunity [18]. TLRs are expressed in various cell types, most abundantly in APC. Recognition of microbial pathogens by TLR results in downstream inflammatory responses such as activation of nuclear factor kappa B (NFκB) and production of type I interferon and inflammatory cytokines via activation of several adapter proteins such as myeloid differentiation primary response gene 88 (MyD88) and Toll/interleukin-1 receptor-domain-containing adapter-inducing interferon-β (TRIF). Of note, recognition of microbial pathogens by TLR enhances the expression of CD80/CD86 which is a critical step to the development of antigen-specific acquired immunity.
Recent advances of research revealed that TLRs recognize internal ligands as well as microbial pathogens which lead to various pathological conditions, such as ischemic acute kidney injury, organ transplant rejection, and various glomerulonephritis [19]. Among the 11 human and 13 mouse TLR receptors, TLR4 recognizes LPS, also known as endotoxin, which is a component of outer membrane of gram-negative bacteria, and TLR3 recognizes double-stranded RNA which is produced by most viruses during their replication [18].
3.2 Regulatory Mechanisms of CD80 in Podocyte
3.2.1 Polyinosinic:Polycytidylic Acid (PolyI:C)-Induced CD80 in Podocytes
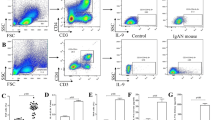
The expression of CD80 in the podocytes was further investigated in the in vitro study utilizing differentiated human podocytes [20]. Initially, Reiser et al. stimulated podocytes with LPS; however, it is related to septic condition, although many recurrences of MCNS are accompanied with subtle upper respiratory virus infections [21]. Therefore, polyI:C, synthesized double-stranded RNA, was used since it works as a ligand of TLR3 which mimics viral infection. It was demonstrated that podocytes express various TLRs under non-stimulated conditions. PolyI:C induced CD80 expression and actin reorganization in podocytes accompanied with augmented expressions of TLR4 and TLR3 [22]. In podocytes, PolyI:C induced the increased production of type I IFN as well as NFκB activation, consistent with the downstream changes seen in the stimulation of TLR by their ligands. NFκB inhibitor significantly reduced CD80 expression in the podocytes. It was an interesting finding since NFκB activation was indicated in the patients with MCNS in relapse [23, 24]. Besides, dexamethasone reduced polyI:C-induced CD80 expression and restored actin reorganization [22]. CD80 knockdown by interfering short hairpin RNA targeting CD80 effectively suppressed actin reorganization induced by polyI:C, thus suggesting the crucial role of CD80 in the phenotype change of the podocytes. Subsequently, Ishimoto et al. proved that systemic administration of polyI:C in mice induced CD80 in the podocytes and elevated CD80 levels in the urine and proteinuria [25].
3.2.2 Serum from the Patients with MCNS Induced CD80 in Podocytes
In MCNS, a circulating factor has been suggested for a long time. Therefore, Ishimoto et al. tested if the serum from patients with MCNS or the culture supernatant of their peripheral blood mononuclear cells (PBMCs) is capable of increasing CD80 in cultured podocytes [26]. They added the serum or the PBMC culture supernatant to the regular culture medium. They found that the serum from MCNS patients in relapse but not in remission increased CD80 expression in podocyte. In contrast, no such increase was observed when PBMC culture supernatant was added. They postulated that the circulating factor could be a virus particle, because it was reported that the existence of microbial products in upper respiratory virus infection is not limited to the respiratory mucosa, but viral fragments are detected in the circulation [27]. Interestingly, IL-13 was not detected in any of the serum samples from MCNS patients, but it was detected in the PBMC culture supernatant in one case in relapse and one case in remission. Thus, IL-13 is seemingly not an indispensable factor in MCNS.
These data indicate that virus-induced podocyte CD80 expression via TLR3 is a possible explanation for the frequent relapse of MCNS in the upper respiratory virus infection, whereas it is unclear if there are additional systemic factors in MCNS.
3.2.3 CTLA-4: Possible Regulator of CD80
CTLA-4 negatively regulates co-stimulatory pathways by the interruption of CD80-CD28 interaction, and CTLA-4 also downregulates the expression of CD80 in the immune cells. In contrast to the induction of CD80 in podocytes, the regulatory mechanisms in podocytes are not well established. Cara-Fuentes et al. performed simultaneous measurement of urinary CD80 and urinary CTLA-4 in MCNS patients [28]. Urinary CD80 levels are dramatically increased in relapse and significantly reduced in remission. Urinary CTLA-4 levels were significantly increased in relapse but were not correlated with the levels of urinary CD80. The increased urinary CTLA-4 may reflect the postinfectious systemic alterations. Ishimoto et al. showed the increased expression of IL-10 in the mice glomeruli after the systemic administration of polyI:C, and the expression of CTLA-4 tended to be increased, but there was no significance. It is unknown how local and systemic regulations work for CD80 in podocytes.
3.3 The Role of Allergy in MCNS
The link between MCNS and atopic disorders has been suggested for a long time. The frequency is generally 30–40 %, and elevated IgE levels are sometimes seen even in the absence of allergic symptoms [29]. Usually, allergic tendency was associated with type 2 helper T cell activation, and increased IL-4 and IL-13 trigger the class switch of B cells for production of IgE [29]. The direct link between the allergy and MCNS has not proven, and it seems that allergy is not indispensable for MCNS; however, the recurrence sometimes occurs right after exposure to allergens such as pollens, mold, poison oak, bee stings, and vaccinations, suggesting causal relationship in some cases. The elevated levels of IL-13 in the serum have been suggested in MCNS [30, 31], although the data vary among reports. Importantly, Lai et al. demonstrated that overexpression of IL-13 in rat induced MCNS-like nephrotic syndrome [32]. They produced the IL-13 overexpression model in Wistar rats through transfection of a mammalian expression vector cloned with the rat IL-13 gene into the quadriceps by in vivo electroporation. The IL-13-transfected rats showed significant albuminuria, hypoalbuminemia, and hypercholesterolemia when compared with control rats. No significant histologic changes were seen in glomeruli by periodic acid-Schiff stain. However, electron microscopy showed foot process fusion up to 80 % of podocytes. Glomerular gene expression was significantly upregulated for CD80, IL-4 receptor-α, and IL-13 receptor-α2 but downregulated for nephrin, podocin, and dystroglycan. The strength of this study was that their model has close similarity to human MCNS compared with other proteinuric models which only induce transient proteinuria. They demonstrated that podocytes express IL-13 receptor and IL-13 induces CD80 in podocytes for the first time. Whereas there are possible problems because this model has not been reproduced in mice, probably, genetic background is crucial for the induction of nephrotic syndrome by IL-13. Besides, the levels of IL-13 in the patients with MCNS did not correlate with disease status [29, 33].
4 Two-Hit Hypothesis: Is MCNS a CD80 Podocytopathy?
CD80 induction was shown as a cause of proteinuria; however, in vivo models by LPS or polyI:C induced only transient proteinuria [8, 25]. Therefore, it was postulated that MCNS is a two-hit disorder [29, 34, 35]. The initial hit is the induction of CD80 in podocytes by various causes such as allergen exposure, microbes, or probably imbalanced cytokines. Then, a shape change in podocytes occurs, namely, actin reorganization which is a corresponding change to foot process effacement. In a healthy subject, it is presumed that CD80 induction is spontaneously resolved; thus, the second hit is the impairment of sensing or regulatory mechanism to terminate CD80 expression, or persistent stimulation for CD80 induction may result in persistent CD80 expression and nephrotic syndrome. Impairment of CTLA-4, a negative regulator of CD80 which is abundantly expressed by regulatory T cells, is a candidate of the second hit. Actually, Araya et al. suggested the impaired regulatory function in the patients with MCNS, although the number of regulatory T cells was not altered significantly [36]. CTLA-4+49GG genotype is associated with susceptibility of nephrotic syndrome [37]. It was also shown that dexamethasone enhances CTLA-4 expression [38]. Anti-inflammatory cytokines, such as IL-10, could be another candidate for the second hit. Nevertheless, it is not clear if the second hit is a systemic disorder or a podocyte disorder which is regulated in an autocrine or paracrine fashion.
5 Classic View of MCNS: T Cell Disorder Hypothesis and CD80
How can we interpret Shalhoub’s hypothesis from the viewpoint of “CD80 podocytopathy”? In 1974, Shalhoub proposed that proteinuria in MCNS is due to a circulation factor released by T cells [39]. It was based on the observations that (i) remission commonly occurs with measles infection which causes cell-mediated immunosuppression; (ii) MCNS sometimes occur with Hodgkin’s disease, which is a known T cell disorder; (iii) unlike many glomerular disorders, there is an absence of humoral immune components (immunoglobulin and complement) in glomeruli; and (iv) MCNS responds to steroids and cyclophosphamide, the agents commonly used to suppress cell-mediated immunity.
Possible explanation could be as follows: (i) in the measles infection, it was shown that regulatory T cells are highly enriched following infection [40]. Besides, there is an interesting data that measles virus caused suppression of CD80 expression in dendritic cells [41]. It is not known if there is any direct effect of measles virus on the podocytes. (ii) In Hodgkin’s disease, the Reed-Sternberg cells can secrete IL-13 and work as an autocrine growth factor [42], which may possibly have a role in the CD80 induction in podocytes. Recently, in the cases of concomitant occurrence of Hodgkin’s disease and MCNS, overexpression of c-maf inducing protein (c-mip) was demonstrated, and a causal relationship was suggested [43]. Recent knowledge suggests that Reed-Sternberg cells are mostly of B cell origin [44]. Besides, there are cases with non-Hodgkin lymphoma and MCNS, and many of them were of B cell origin [45]. Therefore, the relationship between lymphoma and MCNS may not necessarily suggest “T cell disorder.” (iii) In many glomerulonephritis, immune complexes and complement activations are observed. Instead, CD80 induction is a direct cause of actin reorganization of podocytes and the cause of proteinuria in MCNS. (iv) Corticosteroid was able to suppress the expression of CD80 and actin reorganization [22]. Thus, it seems that CD80 induction by polyI:C (mimicry of virus infection) and its resolution by corticosteroid could be an in vitro model of MCNS. The effects of cyclophosphamide and other immunosuppressants on the CD80 expression in the podocytes are not known.
6 Novel View of MCNS
6.1 Is MCNS a CD80 Podocytopathy?
The etiology of idiopathic nephrotic syndrome is still far from being fully elucidated, although recent advances in the podocyte biology have provided us the novel views of nephrotic syndrome as podocytopathies [46].
In MCNS, massive foot process effacement which is a reversible phenotype change of podocytes, namely, actin reorganization, occurs, and the number of podocyte per glomerulus does not change. Corticosteroid reverses this condition. Although there are several other candidates as a cause of MCNS, evidence for the role of elevated urinary CD80 induction in the patients with MCNS is now reported from different research groups [7, 47], suggesting a certain role, at least to some extent. Insufficient regulatory system presumably due to the impaired regulatory T cell function leads to prolonged CD80 expression, although it requires more evidences [29, 34, 35]. Of note, in the very rare cases of renal transplantations, MCNS could relapse when a patient with MCNS received a kidney [48]. On the other hand, when a patient with MCNS donates kidneys, there was no recurrence of proteinuria [49]. Thus, MCNS cannot be solely explained by podocyte disorder, but the circulating factors are suggested.
6.2 Diagnosis of FSGS and MCNS
Recent view of podocytopathy revealed that detachment or death of podocytes leads to glomerulosclerosis in FSGS [46]. It is a heterogeneous disease [50]. In addition to the circulating factors which have been suggested, genetic defects in the important component of podocytes lead to FSGS in some familial or sporadic cases. The “primary FSGS” in a narrow sense is presumably attributed by the circulating factors. In these cases, plasmapheresis is often effective and posttransplant recurrence rates are high, and recurrences sometimes occur very quickly, supporting the idea that circulating factors exist, whereas in the cases with gene mutations, posttransplant recurrences occur less [51].
It is suggested that the levels of soluble urokinase-type plasminogen activator receptor (suPAR) in the serum are elevated in primary FSGS, and it serves as a possible biomarker [52]. Although there are some conflicting data and heated debate, it is based on the scientific explanation that suPAR increases glomerular permeability and activates alphaV beta3 integrin on podocytes causing foot process detachment of glomerular basement membrane [52]. Ling et al. performed the measurement of urinary CD80 in MCNS in relapse, remission, FSGS, and control subjects. They demonstrated that urinary CD80 was significantly higher in the patient with recurrent MCNS compared with other groups. Here, four out of 27 FSGS patients had values near or exceeding the cutoff value, and histological findings in these patients were three cases of tip lesions and one case of not otherwise specified (NOS). Tip lesions could be observed in MCNS patient with heavy proteinuria, and no one had the collapsing type, which has the worst prognosis [47]. In the patients with MCNS and FSGS, Cara-Fuentes et al. measured urinary suPAR, suPAR in the serum, and urinary CD80 simultaneously. They confirmed the elevated urinary CD80 in MCNS and elevated suPAR in the serum in FSGS. The levels of suPAR in the urine were just correlated with urinary protein both in MCNS and FSGS [53].
Recently, Cara-Fuentes et al. reported a case of nephrotic syndrome with NPHS2 mutation which usually causes FSGS [54]. The patient was a 5-year-old Caucasian boy, and the findings in the biopsy were mild mesangial expansion and cellularity which could be early signs of FSGS, at the same time not inconsistent with MCNS with robust proteinuria. In this case, the expression of CD80 in the podocytes was shown by immunofluorescent staining, and increased urinary CD80 was also demonstrated by western blotting. At the same time, mildly elevated suPAR in the urine and serum was observed. Theoretically, if we consider MCNS and FSGS as different disease entities, the hybrid cases are possible. Or we can interpret these data that urinary CD80 is consistent with the initial idea that Reiser et al. showed that genetic defect leads to CD80 induction in the podocytes [8].
The future studies will further elucidate the diagnostic value of urinary CD80 and suPAR in the serum. It would be of great clinical value if the combinations of these parameters are helpful to distinguish MCNS, primary FSGS, and FSGS with gene mutations.
6.3 Novel View of T Cell Disorder
Previously, CD4+ T cell subset was regarded as type 1 helper T cell (Th1)/Th2 binary system. Th1 cells produce IFN-γ and are involved in cell-mediated immunity. Th2 cells produce IL-4 and IL-13 and contribute to humoral immunity and are involved in allergic tendency. Since MCNS is frequently accompanied with allergies, it was postulated as Th2 disease [29]. The recent advances in the immunology expanded Th1/Th2 paradigm to include Th17 and regulatory T cells (Treg) [12]. Indeed, Liu et al. showed that there are Th17/Treg imbalances in MCNS [55]. It is very intriguing that in the very rare cases of congenital T cell immune disorder, several cases of MCNS are reported in immunodysregulation polyendocrinopathy enteropathy X-linked (IPEX) syndrome which is caused by mutation of Foxp3, the key transcription factor of Treg [56], and hyper IgE syndrome which is caused by mutations of signal transducer and activator of transcription (STAT) 3, the indispensable factor for the Th17 cell differentiation [57]. GATA-3, STAT-5, and IL-13 mRNA levels are increased in the PBMC from patients with MCNS in relapse [58]. Allergy as well could be interpreted as an imbalance of CD4+ T cells. In contrast to the genetic mutations in the podocyte components in FSGS, various types of T cell imbalances are observed in the patients with MCNS. These T cell disorders may be related to the impaired regulatory mechanism to terminate CD80 expression via insufficient function of CTLA-4 or other mechanisms, although the proof for the direct relationship between CD80 and these T cell imbalances is lacking.
7 Novel Immunosuppressive Therapies in MCNS
7.1 How to Explain the Effect of Rituximab?
Recent reports which suggest the effectiveness of rituximab in the difficult-to-treat nephrotic patients with frequent relapses or who are steroid dependent [59, 60] compelled us to think about the relationship between CD80 and the effect of rituximab. Rituximab is a monoclonal antibody against CD20. It binds to CD20 and destroys B cells through antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC). CD20 is widely expressed on B cells, from early pre-B cells to later in differentiation, but it is absent on terminally differentiated plasma cells [61]. Superior response to rituximab was associated with older age of children at diagnosis and longer duration until the reconstitution of circulating CD20 [65], whereas the benefit seems to be limited in steroid-resistant patients, especially those with underlining FSGS [62, 63]. Precise mechanisms of anti-proteinuric effect of rituximab still remain unknown. The direct relationship between CD80 and rituximab is not known as well. Podocytes do not express CD20, although Fornoni et al. showed that rituximab colocalized with sphingomyelin phosphodiesterase, acid-like 3B (SMPDL3B) in podocytes and prevented the podocyte damage via the preservation of SMPDL3B and acid sphingomyelinase (ASM) activity [64]. Interestingly, rituximab-treated dendritic cells had decreased expression of CD80 [65], and rituximab treatment induced increase in the number of Treg and augmented their function [66].
7.2 Access the Effect of Cytotoxic T-Lymphocyte-associated Protein 4 (CTLA-4) Immunoglobulin (Ig)
CTLA-4-Ig is a novel immunosuppressive drug which is the fusion protein of the extracellular domain of CTLA-4 and a fragment of the Fc domain of human IgG1. Like the intrinsic CTLA-4, the fusion protein binds to CD80/CD86 on the APC with much higher affinity than CD28, thus interfering with the T cell activation. The first agent abatacept was approved for rheumatoid arthritis in 2005. Then second-generation CTLA-4-Ig belatacept which has superior binding with CD80/CD86 was approved for the maintenance immunosuppression in renal transplantation.
The primary mechanism of immunosuppressive effect of CTLA-4-Ig is the binding with CD80/CD86 and preventing CD80/CD86-CD28 interaction. In addition, it seems that CTLA-4 reduces the expression of CD80 in B cells or APC. In rheumatoid arthritis, CD80 expression in the synovium tissue was reduced by abatacept [67].
In the podocytes, especially in the settings of MCNS where infiltrated inflammatory cells are not observed in glomeruli, it is not known if there is a direct podocyte T cell interaction. Although, there is a soluble form of CTLA-4 which exists in the blood and has the ability of binding with CD80/CD86. Actually, Yu et al. demonstrated that in in vitro study, abatacept had the ability to suppress podocyte migration in podocytes. Since podocyte migration is suggested as the sign of podocyte disorder [68], anti-proteinuric effect of CTLA-4-Ig was theoretically expected in MCNS.
Garin et al. reported a case series of nephrotic syndrome treated with abatacept therapy. One patient with MCNS, one patient with primary FSGS, and three patients with recurrent FSGS posttransplantation were treated with abatacept. In a patient with MCNS, abatacept effectively reduces urinary CD80 and proteinuria, although there was a quick reappearance of urinary CD80 and proteinuria. In a patient with primary FSGS, urinary CD80 was further reduced from normal range, although proteinuria persisted. In the case of immediately recurrent FSGS in an 8-year-old boy post renal transplantation, elevated urinary CD80 and proteinuria were observed immediately, and abatacept infusion and plasmapheresis were initiated on the postsurgery day 6. Urinary CD80 was decreased immediately, but proteinuria persisted another week; thus, urinary CD80 levels were not correlated with the levels of proteinuria in these settings. Subsequently, the repeated plasmapheresis and abatacept infusions result in partial remission [69]. In the patients with lupus nephritis, the administration of abatacept reduced the amount of proteinuria by 20–30 %, only in the patient with nephritic range proteinuria. It is not clear if the effect is related to CD80 [70]. Thus, there is only limited proof for anti-proteinuric effect of CTLA-4-Ig. Future studies are required to access the efficacy of these drugs as a novel therapy of MCNS.
8 Other Renal Diseases and CD80
8.1 Lupus Nephritis
CD80 induction was initially described in lupus nephritis. It was observed in murine model of lupus nephritis and in human as well, fluorescent immunostaining demonstrated CD80 expression in the podocytes, and it was correlated with the severity of nephritis and the severity of proteinuria [8]. However, later studies did not find significant elevation of urinary CD80, the levels of urinary CD80 in lupus nephritis were not significantly elevated [7], and urinary CD80 was elevated in only one of five lupus patients [47]. Of note, there is an argument about the false-positive staining in CD80 immunofluorescent staining especially in membranous nephropathy [71, 72]. Positive staining without the measurement of urinary CD80 may need to be carefully interpreted.
8.2 Diabetic Nephropathy
Recently, the role of CD80 in diabetic nephropathy was demonstrated by Fiorina et al. [73]. In the kidney biopsy obtained from patients with type 2 diabetes with nephropathy, CD80 was expressed in 47 % of those patients shown by immunofluorescent staining. In vitro study showed that high glucose condition induced CD80 and subsequent podocyte actin reorganization. It was phosphatidylinositol-3-kinases (PI3K) dependent and resolved with CTLA-4-IgG. In vivo study also showed that CD80 was induced in podocytes and CTLA-4-IgG reduced urinary albumin excretion in db/db mice and streptozotocin-administered C57BL/6 mice.
It is intriguing that CTLA-4-Ig was effective in the podocyte disorder with increased CD80 expression. However, several questions remain. Dexamethasone is known to downregulate CD80 in the podocytes [22], although, in contrast to MCNS, proteinuria in diabetic nephropathy is not dramatically resolved with corticosteroid. Therefore, it is not clear how CD80 induction in MCNS and diabetic nephropathy are related. Besides, infection is the common serious complication in diabetic patients; therefore, it may lead to limitation of clinical use of CTLA-4-IgG which is an immunosuppressant.
8.3 MCNS as a Posttransplant Complication
Recently, accumulating case reports revealed that nephrotic syndrome rarely occurs as a complication of hematopoietic transplant. Among them, membranous nephropathy and MCNS are common and suggested as rare phenotype of chronic graft versus host disease (GVGD), because they are often accompanied with other symptoms of GVHD and sometimes occur right after the cessation of immunosuppressive drugs [74]. Huskey et al. reported a case of biopsy-proven MCNS after allogeneic stem cell transplant for T cell prolymphocytic leukemia. The patient was a 61-year-old female. She developed nephrotic syndrome 3 weeks after she stopped all the immunosuppressive drugs. Markedly elevated 53 kD urinary CD80 protein was shown by western blotting, demonstrating that it had the same molecular size with the urinary CD80 in the primary MCNS. Proteinuria almost disappeared following remission by the treatment with corticosteroid [75].
De novo MCNS is also reported in renal transplant in case original diagnosis was not MCNS nor FSGS [76, 77].
Together with the recent findings that podocytes are as capable as professional antigen-presenting cells [78], posttransplant CD80-uria may be related to the immunological phenomena in the specific circumstances of posttransplantation. Therefore, posttransplant CD80-uria may need to be discussed separately with the argument over the differential diagnosis between MCNS and FSGS.
9 Conclusion
In conclusion, the etiology of MCNS is still not fully elucidated. Recently, it was revealed that CD80 induction in podocytes is closely related to proteinuric conditions, and urinary CD80 derived from podocytes is a possible biomarker to distinguish MCNS and FSGS. Suppression of CD80 could be a novel therapeutic target, and CTLA-4-Ig is one of the candidates; however, more evidences are required.
References
Alegre ML, Frauwirth KA, Thompson CB. T-cell regulation by CD28 and CTLA-4. Nat Rev Immunol. 2001;1(3):220–8. Epub 2002/03/22.
Peach RJ, Bajorath J, Naemura J, Leytze G, Greene J, Aruffo A, et al. Both extracellular immunoglobulin-like domains of CD80 contain residues critical for binding T cell surface receptors CTLA-4 and CD28. J Biol Chem. 1995;270(36):21181–7. Epub 1995/09/08.
Greenwald RJ, Freeman GJ, Sharpe AH. The B7 family revisited. Annu Rev Immunol. 2005;23:515–48. Epub 2005/03/18.
Wakem P, Burns Jr RP, Ramirez F, Zlotnick D, Ferbel B, Haidaris CG, et al. Allergens and irritants transcriptionally upregulate CD80 gene expression in human keratinocytes. J Invest Dermatol. 2000;114(6):1085–92. Epub 2000/06/09.
Kawamura T, Furue M. Comparative analysis of B7-1 and B7-2 expression in Langerhans cells: differential regulation by T helper type 1 and T helper type 2 cytokines. Eur J Immunol. 1995;25(7):1913–7. Epub 1995/07/01.
Satoh J, Lee YB, Kim SU. T-cell costimulatory molecules B7-1 (CD80) and B7-2 (CD86) are expressed in human microglia but not in astrocytes in culture. Brain Res. 1995;704(1):92–6. Epub 1995/12/15.
Garin EH, Diaz LN, Mu W, Wasserfall C, Araya C, Segal M, et al. Urinary CD80 excretion increases in idiopathic minimal-change disease. J Am Soc Nephrol. 2009;20(2):260–6. Epub 2008/12/06.
Reiser J, von Gersdorff G, Loos M, Oh J, Asanuma K, Giardino L, et al. Induction of B7-1 in podocytes is associated with nephrotic syndrome. J Clin Invest. 2004;113(10):1390–7. Epub 2004/05/18.
Garin EH, Mu W, Arthur JM, Rivard CJ, Araya CE, Shimada M, et al. Urinary CD80 is elevated in minimal change disease but not in focal segmental glomerulosclerosis. Kidney Int. 2010;78(3):296–302. Epub 2010/05/21.
Lenschow DJ, Walunas TL, Bluestone JA. CD28/B7 system of T cell costimulation. Annu Rev Immunol. 1996;14:233–58. Epub 1996/01/01.
Vogt B, Warncke M, Micheel B, Sheriff A. Lentiviral gene transfer of CTLA4 generates B cells with reduced costimulatory properties. Autoimmunity. 2009;42(4):380–2. Epub 2009/10/09.
Ochs HD, Oukka M, Torgerson TR. TH17 cells and regulatory T cells in primary immunodeficiency diseases. J Allergy Clin Immunol. 2009;123(5):977–83. quiz 84–5. Epub 2009/05/05.
Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T, Miyara M, Fehervari Z, et al. CTLA-4 control over Foxp3+ regulatory T cell function. Science. 2008;322(5899):271–5. Epub 2008/10/11.
Cutolo M, Nadler SG. Advances in CTLA-4-Ig-mediated modulation of inflammatory cell and immune response activation in rheumatoid arthritis. Autoimmun Rev. 2013;12(7):758–67. Epub 2013/01/24.
Qureshi OS, Zheng Y, Nakamura K, Attridge K, Manzotti C, Schmidt EM, et al. Trans-endocytosis of CD80 and CD86: a molecular basis for the cell-extrinsic function of CTLA-4. Science. 2011;332(6029):600–3. Epub 2011/04/09.
Kreidberg JA. Functions of alpha3beta1 integrin. Curr Opin Cell Biol. 2000;12(5):548–53. Epub 2000/09/09.
Reiser J, Mundel P. Danger signaling by glomerular podocytes defines a novel function of inducible B7-1 in the pathogenesis of nephrotic syndrome. J Am Soc Nephrol. 2004;15(9):2246–8. Epub 2004/09/02.
Takeda K, Akira S. Toll-like receptors in innate immunity. Int Immunol. 2005;17(1):1–14. Epub 2004/12/09.
Shirali AC, Goldstein DR. Tracking the toll of kidney disease. J Am Soc Nephrol. 2008;19(8):1444–50. Epub 2008/06/27.
Saleem MA, O’Hare MJ, Reiser J, Coward RJ, Inward CD, Farren T, et al. A conditionally immortalized human podocyte cell line demonstrating nephrin and podocin expression. J Am Soc Nephrol. 2002;13(3):630–8. Epub 2002/02/22.
MacDonald NE, Wolfish N, McLaine P, Phipps P, Rossier E. Role of respiratory viruses in exacerbations of primary nephrotic syndrome. J Pediatr. 1986;108(3):378–82. Epub 1986/03/01.
Shimada M, Ishimoto T, Lee PY, Lanaspa MA, Rivard CJ, Roncal-Jimenez CA, et al. Toll-like receptor 3 ligands induce CD80 expression in human podocytes via an NF-kappaB-dependent pathway. Nephrol Dial Transplant. 2012;27(1):81–9. Epub 2011/05/28.
Sahali D, Pawlak A, Le Gouvello S, Lang P, Valanciute A, Remy P, et al. Transcriptional and post-transcriptional alterations of IkappaBalpha in active minimal-change nephrotic syndrome. J Am Soc Nephrol. 2001;12(8):1648–58. Epub 2001/07/20.
Sahali D, Pawlak A, Valanciute A, Grimbert P, Lang P, Remy P, et al. A novel approach to investigation of the pathogenesis of active minimal-change nephrotic syndrome using subtracted cDNA library screening. J Am Soc Nephrol. 2002;13(5):1238–47. Epub 2002/04/19.
Ishimoto T, Shimada M, Gabriela G, Kosugi T, Sato W, Lee PY, et al. Toll-like receptor 3 ligand, polyIC, induces proteinuria and glomerular CD80, and increases urinary CD80 in mice. Nephrol Dial Transplant. 2013;28(6):1439–46. Epub 2012/12/25.
Ishimoto T, Cara-Fuentes G, Wang H, Shimada M, Wasserfall CH, Winter WE, et al. Serum from minimal change patients in relapse increases CD80 expression in cultured podocytes. Pediatr Nephrol. 2013;28(9):1803–12. Epub 2013/05/22.
Xatzipsalti M, Kyrana S, Tsolia M, Psarras S, Bossios A, Laza-Stanca V, et al. Rhinovirus viremia in children with respiratory infections. Am J Respir Crit Care Med. 2005;172(8):1037–40. Epub 2005/07/05.
Cara-Fuentes G, Wasserfall CH, Wang H, Johnson RJ, Garin EH. Minimal change disease: a dysregulation of the podocyte CD80-CTLA-4 axis? Pediatr Nephrol. 2014;29(12):2333–40. Epub 2014/07/16.
Abdel-Hafez M, Shimada M, Lee PY, Johnson RJ, Garin EH. Idiopathic nephrotic syndrome and atopy: is there a common link? Am J Kidney Dis. 2009;54(5):945–53. Epub 2009/06/27.
Yap HK, Cheung W, Murugasu B, Sim SK, Seah CC, Jordan SC. Th1 and Th2 cytokine mRNA profiles in childhood nephrotic syndrome: evidence for increased IL-13 mRNA expression in relapse. J Am Soc Nephrol. 1999;10(3):529–37. Epub 1999/03/12.
Cheung W, Wei CL, Seah CC, Jordan SC, Yap HK. Atopy, serum IgE, and interleukin-13 in steroid-responsive nephrotic syndrome. Pediatr Nephrol. 2004;19(6):627–32. Epub 2004/04/06.
Lai KW, Wei CL, Tan LK, Tan PH, Chiang GS, Lee CG, et al. Overexpression of interleukin-13 induces minimal-change-like nephropathy in rats. J Am Soc Nephrol. 2007;18(5):1476–85. Epub 2007/04/13.
Tain YL, Chen TY, Yang KD. Implications of serum TNF-beta and IL-13 in the treatment response of childhood nephrotic syndrome. Cytokine. 2003;21(3):155–9. Epub 2003/04/17.
Ishimoto T, Shimada M, Araya CE, Huskey J, Garin EH, Johnson RJ. Minimal change disease: a CD80 podocytopathy? Semin Nephrol. 2011;31(4):320–5. Epub 2011/08/16.
Shimada M, Araya C, Rivard C, Ishimoto T, Johnson RJ, Garin EH. Minimal change disease: a “two-hit” podocyte immune disorder? Pediatr Nephrol. 2011;26(4):645–9. Epub 2010/11/06.
Araya C, Diaz L, Wasserfall C, Atkinson M, Mu W, Johnson R, et al. T regulatory cell function in idiopathic minimal lesion nephrotic syndrome. Pediatr Nephrol. 2009;24(9):1691–8. Epub 2009/06/06.
Spink C, Stege G, Tenbrock K, Harendza S. The CTLA-4 +49GG genotype is associated with susceptibility for nephrotic kidney diseases. Nephrol Dial Transplant. 2013;28(11):2800–5. Epub 2013/08/27.
Xia M, Gasser J, Feige U. Dexamethasone enhances CTLA-4 expression during T cell activation. Cell Mol Life Sci. 1999;55(12):1649–56. Epub 1999/10/20.
Shalhoub RJ. Pathogenesis of lipoid nephrosis: a disorder of T-cell function. Lancet. 1974;2(7880):556–60. Epub 1974/09/07.
Sellin CI, Jegou JF, Renneson J, Druelle J, Wild TF, Marie JC, et al. Interplay between virus-specific effector response and Foxp3 regulatory T cells in measles virus immunopathogenesis. PLoS One. 2009;4(3), e4948. Epub 2009/03/26.
Servet-Delprat C, Vidalain PO, Bausinger H, Manie S, Le Deist F, Azocar O, et al. Measles virus induces abnormal differentiation of CD40 ligand-activated human dendritic cells. J Immunol. 2000;164(4):1753–60. Epub 2000/02/05.
Skinnider BF, Kapp U, Mak TW. Interleukin 13: a growth factor in Hodgkin lymphoma. Int Arch Allergy Immunol. 2001;126(4):267–76. Epub 2002/01/30.
Audard V, Zhang SY, Copie-Bergman C, Rucker-Martin C, Ory V, Candelier M, et al. Occurrence of minimal change nephrotic syndrome in classical Hodgkin lymphoma is closely related to the induction of c-mip in Hodgkin-Reed Sternberg cells and podocytes. Blood. 2010;115(18):3756–62. Epub 2010/03/05.
Kuppers R, Rajewsky K. The origin of Hodgkin and Reed/Sternberg cells in Hodgkin’s disease. Annu Rev Immunol. 1998;16:471–93. Epub 1998/05/23.
Kofman T, Zhang SY, Copie-Bergman C, Moktefi A, Raimbourg Q, Francois H, et al. Minimal change nephrotic syndrome associated with non-Hodgkin lymphoid disorders: a retrospective study of 18 cases. Medicine. 2014;93(24):350–8. Epub 2014/12/17.
Barisoni L, Schnaper HW, Kopp JB. Advances in the biology and genetics of the podocytopathies: implications for diagnosis and therapy. Arch Pathol Lab Med. 2009;133(2):201–16. Epub 2009/02/07.
Ling C, Liu X, Shen Y, Chen Z, Fan J, Jiang Y, et al. Urinary CD80 levels as a diagnostic biomarker of minimal change disease. Pediatr Nephrol. 2015;30(2):309–16. Epub 2014/08/22.
Mauer SM, Hellerstein S, Cohn RA, Sibley RK, Vernier RL. Recurrence of steroid-responsive nephrotic syndrome after renal transplantation. J Pediatr. 1979;95(2):261–4. Epub 1979/08/01.
Ali AA, Wilson E, Moorhead JF, Amlot P, Abdulla A, Fernando ON, et al. Minimal-change glomerular nephritis. Normal kidneys in an abnormal environment? Transplantation. 1994;58(7):849–52. Epub 1994/10/15.
Fogo AB. Causes and pathogenesis of focal segmental glomerulosclerosis. Nat Rev Nephrol. 2015;11(2):76–87. Epub 2014/12/03.
Shimizu A, Higo S, Fujita E, Mii A, Kaneko T. Focal segmental glomerulosclerosis after renal transplantation. Clin Transplant. 2011;25 Suppl 23:6–14. Epub 2011/06/03.
Wei C, El Hindi S, Li J, Fornoni A, Goes N, Sageshima J, et al. Circulating urokinase receptor as a cause of focal segmental glomerulosclerosis. Nat Med. 2011;17(8):952–60. Epub 2011/08/02.
Cara-Fuentes G, Wei C, Segarra A, Ishimoto T, Rivard C, Johnson RJ, et al. CD80 and suPAR in patients with minimal change disease and focal segmental glomerulosclerosis: diagnostic and pathogenic significance. Pediatr Nephrol. 2014;29(8):1363–71. Epub 2013/11/23.
Cara-Fuentes G, Araya C, Wei C, Rivard C, Ishimoto T, Reiser J, et al. CD80, suPAR and nephrotic syndrome in a case of NPHS2 mutation. Nefrologia. 2013;33(5):727–31. Epub 2013/10/04.
Liu LL, Qin Y, Cai JF, Wang HY, Tao JL, Li H, et al. Th17/Treg imbalance in adult patients with minimal change nephrotic syndrome. Clin Immunol. 2011;139(3):314–20. Epub 2011/04/01.
Mishra K, Batra VV, Basu S, Rath B, Saxena R. Steroid-resistant nephrotic syndrome associated with steroid sulfatase deficiency-x-linked recessive ichthyosis: a case report and review of literature. Eur J Pediatr. 2012;171(5):847–50. Epub 2012/03/16.
Miyazaki K, Miyazawa T, Sugimoto K, Fujita S, Yanagida H, Okada M, et al. An adolescent with marked hyperimmuno-globulinemia E showing minimal change nephrotic syndrome and a STAT3 gene mutation. Clin Nephrol. 2011;75(4):369–73. Epub 2011/03/24.
Komatsuda A, Wakui H, Iwamoto K, Togashi M, Masai R, Maki N, et al. GATA-3 is upregulated in peripheral blood mononuclear cells from patients with minimal change nephrotic syndrome. Clin Nephrol. 2009;71(6):608–16. Epub 2009/05/29.
Munyentwali H, Bouachi K, Audard V, Remy P, Lang P, Mojaat R, et al. Rituximab is an efficient and safe treatment in adults with steroid-dependent minimal change disease. Kidney Int. 2013;83(3):511–6. Epub 2013/01/18.
Ravani P, Ponticelli A, Siciliano C, Fornoni A, Magnasco A, Sica F, et al. Rituximab is a safe and effective long-term treatment for children with steroid and calcineurin inhibitor-dependent idiopathic nephrotic syndrome. Kidney Int. 2013;84(5):1025–33. Epub 2013/06/07.
Leget GA, Czuczman MS. Use of rituximab, the new FDA-approved antibody. Curr Opin Oncol. 1998;10(6):548–51. Epub 1998/11/18.
Kronbichler A, Bruchfeld A. Rituximab in adult minimal change disease and focal segmental glomerulosclerosis. Nephron Clin Pract. 2014;128(3–4):277–82. Epub 2014/11/18.
Yabu JM, Ho B, Scandling JD, Vincenti F. Rituximab failed to improve nephrotic syndrome in renal transplant patients with recurrent focal segmental glomerulosclerosis. Am J Transplant. 2008;8(1):222–7. Epub 2007/11/06.
Fornoni A, Sageshima J, Wei C, Merscher-Gomez S, Aguillon-Prada R, Jauregui AN, et al. Rituximab targets podocytes in recurrent focal segmental glomerulosclerosis. Sci Transl Med. 2011;3(85):46–85. Epub 2011/06/03.
Xu GF, Zhang LS, Li LJ, Yi LC, Zeng PY, Wu CY. The immune effects of rituximab on dendritic cells derived from patients with primary immune thrombocytopenia. Zhonghua xue ye xue za zhi = Zhonghua xueyexue zazhi. 2012;33(3):207–10. Epub 2012/07/12.
Vigna-Perez M, Hernandez-Castro B, Paredes-Saharopulos O, Portales-Perez D, Baranda L, Abud-Mendoza C, et al. Clinical and immunological effects of Rituximab in patients with lupus nephritis refractory to conventional therapy: a pilot study. Arthritis Res Ther. 2006;8(3):R83. Epub 2006/05/09.
Kanbe K, Chiba J, Nakamura A. Immunohistological analysis of synovium treated with abatacept in rheumatoid arthritis. Rheumatol Int. 2013;33(7):1883–7. Epub 2012/01/04.
Mundel P, Reiser J. Proteinuria: an enzymatic disease of the podocyte? Kidney Int. 2010;77(7):571–80. Epub 2009/11/20.
Garin EH, Reiser J, Cara-Fuentes G, Wei C, Matar D, Wang H, et al. Case series: CTLA4-IgG1 therapy in minimal change disease and focal segmental glomerulosclerosis. Pediatr Nephrol. 2015;30(3):469–77. Epub 2014/09/23.
Furie R, Nicholls K, Cheng TT, Houssiau F, Burgos-Vargas R, Chen SL, et al. Efficacy and safety of abatacept in lupus nephritis: a twelve-month, randomized, double-blind study. Arthritis Rheum. 2014;66(2):379–89. Epub 2014/02/08.
Yu CC, Fornoni A, Weins A, Hakroush S, Maiguel D, Sageshima J, et al. Abatacept in B7-1-positive proteinuric kidney disease. N Engl J Med. 2013;369(25):2416–23. Epub 2013/11/12.
Benigni A, Gagliardini E, Remuzzi G. Abatacept in B7-1-positive proteinuric kidney disease. N Engl J Med. 2014;370(13):1261–3. Epub 2014/03/29.
Fiorina P, Vergani A, Bassi R, Niewczas MA, Altintas MM, Pezzolesi MG, et al. Role of podocyte B7-1 in diabetic nephropathy. J Am Soc Nephrol. 2014;25(7):1415–29. Epub 2014/03/29.
Hu SL. The role of graft-versus-host disease in haematopoietic cell transplantation-associated glomerular disease. Nephrol Dial Transplant. 2011;26(6):2025–31. Epub 2010/10/22.
Huskey J, Rivard C, Myint H, Lucia S, Smith M, Shimada M, et al. Minimal change disease in graft versus host disease: a podocyte response to the graft? Clin Nephrol. 2013;80(6):469–73. Epub 2012/09/26.
Nongnuch A, Assanatham M, Sumethkul V, Chalermsanyakorn P, Kitiyakara C. Early posttransplant nephrotic range proteinuria as a presenting feature of minimal change disease and acute T cell-mediated rejection. Transplant Proc. 2014;46(1):290–4. Epub 2012/12/27.
Zafarmand AA, Baranowska-Daca E, Ly PD, Tsao CC, Choi YJ, Suki WN, et al. De novo minimal change disease associated with reversible post-transplant nephrotic syndrome. A report of five cases and review of literature. Clin Transplant. 2002;16(5):350–61. Epub 2002/09/13.
Goldwich A, Burkard M, Olke M, Daniel C, Amann K, Hugo C, et al. Podocytes are nonhematopoietic professional antigen-presenting cells. J Am Soc Nephrol. 2013;24(6):906–16. Epub 2013/03/30.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Japan
About this chapter
Cite this chapter
Shimada, M., Ishimoto, T., Johnson, R.J. (2016). Co-stimulatory Molecule CD80 (B7.1) in MCNS. In: Kaneko, K. (eds) Molecular Mechanisms in the Pathogenesis of Idiopathic Nephrotic Syndrome. Springer, Tokyo. https://doi.org/10.1007/978-4-431-55270-3_4
Download citation
DOI: https://doi.org/10.1007/978-4-431-55270-3_4
Publisher Name: Springer, Tokyo
Print ISBN: 978-4-431-55269-7
Online ISBN: 978-4-431-55270-3
eBook Packages: MedicineMedicine (R0)