Abstract
This chapter examines the estuaries within the Southern Brazilian Marine Ecoregion (SBE), a coastline of some 850 km in the tropical-temperate transition zone of the extreme southern portion of Brazil. A particular attention is given to the ecology of benthic communities, considering their structure, functioning, impacts, and conservation issues. In a microtidal and wave dominated environment, there are tens of small coastal plain estuaries and lagoons along the SBE. Although there is much overlapping of characteristics between small estuaries and lagoons, differences in the freshwater influence, spatial organization, and variability of gradients constitute important drivers since they directly affect species composition, abundance, and dominance of ecological guilds. This is particularly true for estuaries with intermittently open inlets, which are dominant in SBE. El Nino Southern Oscillation (ENSO) has a strong influence on the regional climate, affecting interannual estuarine hydrodynamics, benthic fauna distribution and recruitment. The environmental quality status of 27 SBE estuarine systems according to the AZTI Marine Biotic Index (AMBI index) was largely classified as moderately disturbed (80%), whereas 20% of the sites were of slightly or undisturbed classifications. Different categories of marine protected areas (MPAs) are implemented in the estuaries in the SBE. Although MPAs, the Brazilian Coastal Management Plan as well as other policies could foster the sustainable use of coastal resources and ecosystem services, difficulties in coastal planning and in policy implementation still allow unsustainable practices, impacts, and habitat loss over all SBE estuaries.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
6.1 Introduction
The Southern Brazilian Marine Ecoregion (SBE) or Rio Grande Ecoregion (Spalding et al. 2007) encompasses about 850 km from the North of the Island of Santa Catarina (26°50′S), State of Santa Catarina, to the Chuí (33°44′S), on the extreme southern portion of Brazil, at the State of Rio Grande do Sul. The SBE has a typical microtidal environment along a highly embayed coast with predominance of rocky headlands in the north and extensive sand barriers in the south.
Estuaries of SBE typically have small catchments (except Patos Lagoon on the southern region) and are particularly well-endowed with small coastal plain estuaries and coastal lagoons. The small coastal plain estuaries typically have a funnel-shaped morphology and arise from marine flooding of the lower course of a myriad of small catchment rivers. Different from the sizeable estuaries of the East-Southeast Sector of Brazilian Coast, the small coastal plain estuaries have no granitic rocks at the entrance, but are fluvial valleys usually flourished by mangrove forests northward, and by saltmarshes southward of the SBE.
On the other hand, the southern Brazilian coastal lagoons are an evolving coastal landform that may go through a cycle from an open embayment to a partially back-barrier lagoon with progressive infilling, to a segmentation into small lagoons with unstable inlets, and then coastal lakes (Cooper 1994). Natural and gradual shifts from lagoons to temporarily open/closed lagoons and lakes (and vice versa) are long-term processes that result from large-scale (e.g., sea-level and climate changes) and local processes (e.g., sediment supply, alongshore drift, and coastal morphology). However, the environmental shift may also be hastened by anthropogenic activities at the ecological scale, such as hydrological management (Schock et al. 2014), artificially opening of lagoons (Netto et al. 2012), or modifications as a result of climate change (Chapman 2012). The combination of sediment accretion rates and sea level rise will determine the temporal volumetric capacity of the lagoon, its import/export status, and the resultant evolution. The relative importance of a particular process in a lagoon depends upon the local environmental setting in which the lagoon is located, and the evolutionary path followed by a lagoon depends upon the magnitude and relative importance of each of the operative processes. The dynamism of these forces promotes both long-term and short-term changes. In the long-term (seasons and years), it influences the connectivity with the sea, while in the short-term (tidal cycles), it affects the amount of seawater inflow.
Along the SBE there are 32 small coastal plain estuaries, 16 with mangrove forests occupying the quaternary fluvial facies, and 16 with no transitional land-sea vegetation or just sparse mangrove trees and patches of small beds of Spartina alterniflora saltmarsh limited to the mouth of the system (Fig. 6.1, Table 6.1). According to the present connectivity with the sea, the local coastal lagoons water bodies can roughly be divided into three major types: open (permanently connected to the sea), the intermittently open/closed (which includes seasonally or non-seasonally closed or those normally closed), and the closed (presently without connection with the sea, i.e., the coastal lakes). Today, along the SBE there are six lagoons permanently open to the sea (Barra Velha, Conceição, Laguna, Camacho, Tramandai-Armazém, and Patos), and seven with temporarily open/closed bars (Acaraí, Lagoinha do Leste, Ibiraquera, Urussanga-Velha, Garopaba, Sombrio, Peixe) (Fig. 6.1, Table 6.2), and tens of closed lagoons.
Estuaries of the Southern Brazilian Ecoregion (SBE). Capital letters are coastal lagoons and numbers are coastal plain estuaries. Coastal lagoons = A Acaraí, B Barra Velha, C Conceição, D Lagoinha do Leste, E Garopaba, F Ibiraquera, G Laguna, H Camacho, I Urussanga-Velha, J Sombrio, K Tramandaí-Armazém, L Peixe, M Patos. Coastal plain estuaries = 1 Itajubá, 2 Piçarras, 3 Gravatá, 4 Itajaí, 5 Camboriú, 6 Perequê, 7 Tijucas, 8 Inferninho, 9 Cachoeira-Camarão, 10 Biguaçu, 11 Serraria, 12 Três Henriques, 13 Büchele, 14 Maruím, 15 Curtume, 16 Aririu, 17 Cubatão Sul, 18 Praia do Pontal, 19 Praia de Fora, 20 Massiambu, 21 Ratones-Papaquara, 22 Veríssimo, 23 Saco Grande, 24 Itacorubi, 25 Tavares, 26 Tapera, 27 Ribeirão, 28 Tapera da Caicanga-Açu, 29 Madre-Paulo Lopes, 30 Araranguá, 31 Mampituba, 32 Barra do Chuí. See Tables 6.1 and 6.2 for details
Lagoons and small coastal plain estuaries share many common features including vegetation and fauna. However, differences in the freshwater influence and spatial organization of estuarine gradients may introduce major local variability and directly affect species composition, abundance, and dominance of ecological guilds (Pérez-Ruzafa et al. 2011). While coastal plain estuaries are drowned river valleys with hydrodynamics mainly conducted by river flux, with low residence time and clear gradients from freshwater to marine conditions, coastal lagoons are inland shallow and shore-parallel water bodies relatively more isolated from the open sea by low-lying sand barriers. This is particularly true for temporarily open/closed lagoons. Coastal lagoons present more mixed waters and their hydrodynamics is mainly controlled by the wind, higher residence time, and intra-lagoon gradients hidden by complex patterns in substrate properties and freshwater/marine inputs.
Contrary to the small coastal plain estuaries, the southern Brazilian coastal lagoons vary widely in their shape, size, ecological processes, and in the quantity and detail of research as well. In common they share social values and problems. Research on SBE lagoons largely focused at permanently open lagoons (e.g., Sierra de Ledo and Soriano-Sierra 1999; Seeliger et al. 1997; Odebrecht et al. 2017) as they are under more pressure due to larger urban developments. However, more than a half of the existing lagoons along southern Brazil are temporary open/closed estuaries which recently come under growing pressures, particularly artificial inlet managing (e.g., Netto et al. 2012; Crippa et al. 2013; Netto and Fonseca 2017). The value of the natural goods and services provided by coastal lagoons are among the highest of natural ecosystems, and can be categorized as suggested by Anthony et al. (2009) as pragmatic, scholarly, inspirational, and tacit. These provisions include: (a) ecosystem services that indirectly support human uses, such as nursery grounds for species that support commercial fisheries and protection from storms; (b) scientific inquiry and historical study seeking to extend knowledge, as to improve management, sustainable use, and tourism revenues; (c) inspiration to different sorts of artistic expressions; (d) recreation, visual and sonorous enjoyment of scenery, and sense of place. Despite their ecological and social relevance, SBE coastal lagoons are seriously threatened by pollution, eutrophication, inlet hydrological modifications, urbanization, aquaculture, and a variety of modification in their margins and watersheds, caused by fast and unplanned occupation practices within the lagoons’ floodplains.
6.2 Environmental Settings
The SBE is a transitional climatic zone between the South subtropical to humid temperate, or according to updated Brazilian climate map of Kӧppen (Alvares et al. 2014), it has Cfa type climate (temperate, without dry season, with hot summer). During the spring and summer months, the South Atlantic Tropical Anticyclone, a high-pressure center that produces a wet and warm air mass, predominates at the region, generating NE winds. During autumn and winter, the passage of cold fronts associated with the displacement of the Polar Migratory Anticyclone generates S/SE winds. The total annual precipitation in the ecoregion ranges from 1000 to 1500 mm and it is, in general, evenly distributed throughout the year. The mean air temperatures are lower than 18 °C in the winter, and in the summer lower than 27 °C (Bernardino et al. 2015). El Nino Southern Oscillation (ENSO) has a strong influence on the regional climate, with significant differences in precipitation rates, and on the intensity and direction of prevailing winds between years with positive and negative southern oscillation indices (Grimm et al. 2003). During El Nino events (positive ENSO phase), rainfall is often above normal and the water flow from the estuary towards the coast increases. On contrary, during La Nina (negative ENSO phase) rainfall is often below normal, favoring the input of salt water into the lagoons (Odebrecht et al. 2010). Thus, ENSO strongly affects interannual estuarine hydrodynamics (Fernandes et al. 2002), variability of planktonic and benthic primary producers (Odebrecht et al. 2010), benthic fauna distribution and recruitment (Colling et al. 2007), and many ecologically and commercially important estuarine species (Seeliger 2001; Garcia et al. 2003; Vieira et al. 2008).
Diverse barrier types and associated surficial landforms occur along the SBE (Hesp et al. 2009; Dillenburg et al. 2009). At the northern portion, from north of the Island of Santa Catarina to Santa Marta Cape, the coastline is characterized by a diversity of formations, such as small coastal bays and coves, lagoon inlets, large promontories, long and arched beaches, rocky shores and barriers dominated by transgressive dune fields. Southwards, the coastline changes direction and it is mostly a straight-line SW-NE orientated, characterized by long sand beaches and barriers dominated, in large scale, by active transgressive dune fields (see Hesp et al. 2009 and Short and Klein 2016 for details).
Shallowness and microtidal regime determine that the wind and precipitation exert key roles in the circulation and sediment movement of coastal plain estuaries and coastal lagoons. Sediment infill of some elongated and shore-parallel lagoons, such as Conceição, Laguna, and Patos, involves the development of a series of cuspate divisions (septation) due to wind waves that build spits isolating the lagoon into separate basin (Woodroffe 2002). At Patos lagoon, in the southern part of SBE, large catchments and seasonal precipitation regulate variations of freshwater discharge, with higher values during the austral winter/spring (July to October) and lower during the summer/autumn (November to June) (Garcia 1997). Wind effects on circulation is particularly important for the small and medium sized lagoons located at the north of the SBE, characterized by small catchments, and where there is a lack in rainfall differences over the year. Predominant NE winds during summer and spring force lagoon water masses towards the southern margins (seaward), resulting in a decreased salinity and increase of suspended materials from continental origin. In contrast, during winter and autumn, periods of strong S-SE winds favor marine water intrusion into the lagoons resulting in lower turbidity and higher salinity values (Fig. 6.2; Eichler et al. 2006; Meurer and Netto 2007).
Schematic representation of wind influence on lagoon circulation, and influence on temperature and salinity values (modified from Castelão and Moller 2003)
The connectivity dynamics between the lagoons and the sea play a key role in their overall functioning, as intermittent closing (Fig. 6.3). The openness of the lagoon mouth is determined by the balance between scouring forces (primarily catchment runoff and tidal prism) and blocking forces (primarily onshore and longshore deposition of sediments; Whitfield and Bate 2007; McSweeney et al. 2017). Permanently open lagoons, such as Patos, Laguna, and Conceição, are primarily characterized by a wide spatiotemporal range in environmental conditions (e.g., salinity, temperature, oxygen), biological productivity, and movement of resources and consumers with other adjacent marine areas (Pérez-Ruzafa et al. 2011; Abreu et al. 2017). Temporarily closed lagoons (Fig. 6.3) are intermittently isolated from the sea by the formation of a sand berm across the estuary mouth, and typically present a small river catchment and high rates of longshore and onshore sediment transport (Cooper et al. 1999; Schallenberg et al. 2010). Once closed, depending on the freshwater inputs and the time of closure, the system may become gradually fresher or even more saline due to evaporation rates. An intermittent open lagoon may lead to remarkable changes in its physical-chemical environment over short time periods, which in turn triggers major biological responses in both pelagic and benthic compartments (Niekerk et al. 2005; Netto et al. 2012).
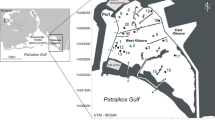
Ibiraquera lagoon, a typical temporarily open/closed coastal lagoon of Brazilian Southern Ecoregion (SBE): (a) during isolation from the sea, lagoon water level increases and disturbance from wind and water generated turbulence are minimal, offering a fairly homogeneous and stable environment; (b) connectivity during open phase permits the exchange of matter and energy between the lagoons and the sea, exposes sand banks and generates strong physical-chemical gradients that directly affect the biota
The mineralization of organic matter usually is higher in the inner sector (oligohaline) of the estuaries, where most of the materials are retained. In permanently stratified coastal lagoons, as the Conceição lagoon, or stratified systems, as the Madre-Paulo Lopes estuary, haloclines coincide with oxyclines due to the elevated rates of allochthonous and autochthonous organic matter mineralization in bottom waters (Fonseca and Braga 2006; Fontes and Abreu 2009). Heterotrophic conditions (e.g., hypoxic and anoxic waters) associated to high macrophyte biomass and high turbidity have been described to coastal plain with stratified (Madre-Paulo Lopes) and homogeneous (Ratones-Papaquara) water column. Anoxic and hypoxic events were observed in low hydrodynamics and deep regions due to the geomorphology isolation. Those events were enhanced during periods of low rainfall and high water residence time. High precipitation events boosted the exportation of eutrophic estuarine waters to the ocean, which improves the water quality of the SBE systems. The estuaries became dominated by continental freshwaters and it is expected an increase of nutrients in shallow continental shelf, that might relate with toxic algal blooms (e.g., red tide) (Fernandes and Brandini 2010; Proença 2004; Omachi et al. 2007). On the other hand, meteorological tides caused by the southerly winds can increase water residence time of coastal systems, enhancing physical and chemical stratification of the water column and the eutrophication process (Abreu et al. 2010).
The changes in nutrient concentrations are related to the uptake by primary producers, retention in sediment, recycling times and sources (river discharge, domestic and industrial effluents), as observed in Patos and Conceição lagoons (Fonseca 2004; Haraguchi et al. 2015; Silva et al. 2017). Denitrification and phosphorus adsorption by the benthic compartment are important biogeochemical processes, both removing dissolved nutrients from water column and regulating eutrophication in these coastal lagoons (Fonseca 2004). In a temporal perspective, some lagoons, such as Patos and Conceição, show an increase in the concentrations of nitrogen and phosphorous (Niencheski et al. 2006; Haraguchi et al. 2015; Lanari and Copertino 2017), which suggests an eutrophication trend. A significant N:P ratio increase was also shown, suggesting that changes in land use and increased urbanization in the watershed are altering the nutrient delivery, which may impact phytoplankton composition (Haraguchi et al. 2015; Silva et al. 2017). Although the phytoplankton biomass (chlorophyll a) do not present clear temporal changes (Abreu et al. 2010; Silva et al. 2017), significant trends of increasing biomass of cyanobacteria and dinoflagellates are observed at interannual scale (Haraguchi et al. 2015; Fontes et al. 2011; Fonseca and Braga 2006). At Patos lagoon, diatoms are the main phytoplankton group and in the seasonal scale, the chlorophyll a variation is closely related to these microalgae, influenced by the short water retention time and low light availability during winter (Haraguchi et al. 2015). The combined effects of climate (ENSO and rainfall) and hydrology (freshwater discharge and water physical-chemical parameters) drive the onset and magnitude of drift macroalgal blooms in the Patos Lagoon estuary, where the wind action further controls the persistence and possible occurrence of large green tides (Lanari and Copertino 2017).
Shoreline vegetation is dominated by plants of the genera Spartina, Scirpus, and Juncus in all SBE lagoons (Soriano-Sierra 1999; Costa and Davy 1992, Valgas 2009). Well-structured mangrove forests are found only in the coastal plain estuaries. The typical species from mangroves are Avicennia schaueriana, Laguncularia racemosa, and Rhizophora mangle. Isolated species typical from mangroves (A. schaueriana and L. racemosa) can be found at the inlet of Conceição Lagoon (Soriano-Sierra 1999) and at Laguna, the southernmost distribution of mangroves vegetation in America. A. schaueriana is the dominant mangrove species in SBE, with up to 4333 stems/ha, basal area ranging from 3.8 to 6.3 m2/ha, and height from 6 to 9 m in old-growth stands (Rovai et al. 2012). In the Itacorubi coastal plain estuary, the mangrove forests and saltmarsh productivity are 415 gC/m2/year and 711 gC/m2/year, respectively (Panitz 1986). In the southernmost lagoon, the mean net above- and belowground productivity of Spartina alterniflora ranges from 669 to 1928 gC/m2/year, respectively, while the aboveground Spartina densiflora values reach about 2260 gC/m2/year (Peixoto and Costa 2004; Cunha et al. 2005).
The euphotic zone of most SBE coastal lagoons reach the sediment as they are usually shallow (<2 m) with low particulate suspended material in water column, favoring the primary and secondary benthic production. Blooms of macroalgae Ulva sp. and Enteromorpha sp. are common along SBE lagoons. In Conceição lagoon, the productivity of those species may reach values up to 7000 gC/m2/year, followed by the macrophyte Ruppia sp. (3042 gC/m2/year) and the microphytobenthos (50 gC/m2/year) (dos Santos et al. 2017; Fonseca 2004). In the central region of Conceição lagoon, where the halocline is permanent, it was described high rates of phytoplankton production (1080 gC/m2/year) in deeper water. In this system, significative primary productions were measured under low solar radiance (438 gC/m2/year) and oxygen concentrations (360 gC/m2/year) which demonstrates the importance of carbon fixation by Cyanophyceae and anaerobic anoxygenic bacteria, respectively (Fontes et al. 2011; 2015). The phytoplankton production in the Patos Lagoon varies from 0.004 and 0.320 gC/m2/h (or 18 gC/m2/year and 1401 gC/m2/year; Abreu et al. 1994) and are largely determined by seasonality and the contribution of flagellates (Seeliger et al. 1997). In this lagoon, estimates on Ruppia maritima productivity indicate net annual values ranging from 39.6 to 43.2 gC/m2/year, and its variability is determined by species-specific seasonal growth cycles.
6.3 Benthic Fauna
Protected from the physical harshness of the open ocean, within calm, diverse, and plenty of food habitats, the benthic fauna thrives in estuaries. Large and productive populations of producers and primary consumers, maintained by the ability of estuaries to trap nutrients and food particles, support high biomass of benthic invertebrates. In contrast, changes in chemical conditions caused by local, marine, and riverine inputs have a relevant role in the structure of the estuarine benthic fauna. As natural stress levels increase with confinement, but also with the frequent oscillation between closeness and openness, estuarine benthic invertebrates are typically species poor when compared to adjacent seas.
Benthic invertebrates are an important component of any aquatic ecosystem, but shallowness amplifies their pivotal role in estuaries. They significantly contribute to energy flow and as diet of many other estuarine animals. The activity of some benthic organisms also increases the removal of fixed nitrogen from aquatic ecosystems by fostering nitrification and denitrification in the oxic–anoxic transition zone of the marine sediment (Bonaglia et al. 2014). Benthic organisms are largely used as biological indicators because they can provide information on environmental conditions either due to the sensitivity of assemblages or single species, or because of some general feature that makes them integrate environmental signals over a long period of time (Teixeira et al. 2010; Schratzberger and Ingels 2017).
6.3.1 Meiofauna
The meiobenthos or meiofauna comprises a particular group of microscopic invertebrates ranging from 0.03 mm to 0.5 mm in size. They represent the most abundant group of benthic metazoans. Along SBE estuaries, on average, their abundance ranges from 105 to 106 individuals per square meter, corresponding to a biomass of 1 to 2 g dry weight per square meter. The meiofauna is also more diverse than any other component of the marine biota with 24 phyla (out of a total of 35 known invertebrate phyla) with meiobenthic representatives. Despite of its importance, the current knowledge of the estuarine meiofauna along SBE is, so far, scarce and heterogeneous. Data on meiofauna with taxonomic resolution higher than phylum or class are only available for Santa Catarina state, northern portion of SBE.
Netto and Fonseca (2017) made the only attempt to comparatively examine the meiofauna of several coastal lagoons along the Brazilian coast. A large-scale sampling program was carried out from Florianopolis (Santa Catarina State) to Torres (Rio Grande do Sul State). The study investigated to what extent the differences in openness of coastal lagoons structure meiofauna communities. Using particular lagoon status (open or closed) as replicates of their evolution over time, they showed, using free-living nematodes as surrogate, that open and closed lagoons are mutually exclusive alternative states of equilibrium, and that temporarily open/closed lagoons are an intermediate or transition phase between them. The gradient in the structural connectivity between lagoons and the sea, due to their regime shifts, changes the movement of resources and consumers, and the internal physical-chemical gradients that directly affect the meiofaunal species diversity, abundance, and trophic status.
The number of nematode genera and taxa diversity are generally higher in the open lagoons and coastal plain estuaries, intermediate in temporarily closed lagoons, and lower at permanently closed ones (Table 6.3 and Fig. 6.4). Density is normally higher in lagoons (open or temporally closed), decreasing in coastal plain estuaries and lower in closed lagoons. Differences within each estuarine typology increase with openness. These patterns are a result of combined effects of the input of marine species, the presence of environmental gradients, and higher environmental heterogeneity (Netto and Fonseca 2017). Overall, freshwater nematode assemblages are impoverished when compared to marine and brackish systems. In turn, the transitional structure of the nematode assemblages of temporarily open/closed lagoons reflects the intermediate pattern of isolation compared to lagoons and coastal lakes.
Mean (±SE) of nematode number of genera, diversity (H′), and density (inds.10/cm2) in inner and outer portions (columns) within closed lagoons, intermittently open/closed lagoons (ICOLL), open lagoons, and coastal plain estuary (CPE) in the Brazilian Southern Ecoregion (SBE). Black dots represent the total mean value (±SE) of the community descriptors in each type of estuary
Most of the nematode genera recorded in open lagoons are those typically found in coastal plain estuaries. The meiofauna of and the small coastal plain river estuaries (e.g., Ratones estuary; Netto and Gallucci 2003) shows relative similarity with SBE open lagoons, though the percentage of marine species is higher (Table 6.3). The proportion of brackish or marine species is reduced in open/closed lagoons with an increasing number of brackish/freshwater or freshwater genera. In closed lagoons, freshwater or brackish/freshwater genera largely dominate. Along the coastal lagoons of Santa Catarina, only five genera occurred in all the three types of lagoons, namely Anonchus (Aphanolaimidae), Anoplostoma (Anoplostomatidae), Desmodora (Desmodoridae), Dichromadora, and Hypodontolaimus (Chromadoridae) (Netto and Fonseca 2017). Besides, the percentage of exclusive genera (those found exclusively in only one type of lagoon) decreased with increasing connectivity (Table 6.3).
Changes in the structural connectivity between coastal lagoons and the sea affect their trophic status (Fig. 6.5). The impoverishment of the nematode assemblages and the substitution of brackish water species by freshwater species due to the decrease in the connectivity promotes a decrease of trophic diversity (an index based on the proportion of each feeding type; Heip et al. 1985). While closed lagoons were largely dominated by predator/omnivores, more opened lagoons were numerically dominated by nonselective deposit-feeders and epigrowth feeders (Fonseca and Netto 2006).
Mean value (±SE) of the index of trophic diversity and nematode variation in genera composition (total mean β-diversity, contribution of replacement and richness differences) in closed lagoons, intermittently open/closed lagoons, open lagoons, and coastal plain estuaries in the Brazilian Southern Ecoregion (SBE) (reproduced from Netto and Fonseca 2017)
Similarities of the nematode assemblages within and between lagoons of SBE also change according to the stable state (Netto and Fonseca 2017). While habitat connection and faunal exchange by open inlets increased similarity between more connected lagoons, with variations in the composition controlled by gradients, isolation increased variability of nematode assemblages among lagoons (β-diversity) with an increasing dominance of species replacement over richness (Fig. 6.5). At the same time, internal faunal variability (e.g., differences between outer and inner portions of lagoons) is higher within open lagoons than in closed lagoons, with temporarily open/closed lagoons assuming an intermediate position. This pattern may emerge because of the connectivity that modulates the degree to which the inlet state facilitates or impedes the exchange of matter, energy, and specimens among landscape elements. Besides, differences in structural connectivity can lead to internal homogeneity or strong physical-chemical gradients that directly affect species composition. While the low variability of nematode assemblages among lagoons is likely to be a result of faunal transport due to their physical link, the high dissimilarities of the assemblages between coastal lakes might be consequence of their spatially disconnection and exposure to discrete and variable surroundings. Thus, as the lagoons loose connectivity, gradually shifting the state, local processes become increasingly more important in structuring these communities than large-scale drivers.
Punctual studies focused on spatial variability of meiofaunal assemblages showed that the most important structuring variables on an individual lagoon scale are salinity and sediment characteristics, which are in turn largely modulated by the hydrodynamic conditions (Kapusta et al. 2002, 2004, 2005; Fonseca and Netto 2006). Besides, the amount of mangrove-derived detritus positively affects the diversity and density of the meiofauna in small coastal plain estuaries bordered by mangroves, indicating that nematodes may show a high degree of specificity in the microhabitat choice and high efficiency in exploiting these microhabitats originated by mangrove litter decomposition (Netto and Gallucci 2003).
6.3.2 Macrofauna
Connectivity of benthic macrofaunal communities (invertebrates retained by ≥0.5 mm mesh size) within and between lagoons and small coastal plain estuaries in SBE suggests that these organisms are exposed to similar structuring drivers. The dispersion and colonization of the macrofauna occurs primarily on the water column with a major passive component throughout large distances. The connections among the many small systems via adjacent ocean facilitate the exchange of individuals among them. As a result, benthic macrofaunal communities of local estuarine systems show similar species composition and richness, differing somewhat in both the spatiotemporal distribution of abundances and from the present closed lagoons.
The macrobenthic community at the inner portion of coastal plain estuaries is generally more abundant than at outer portions of estuaries (Rosa Filho and Bemvenuti 1998; Netto and Gallucci 2003; Pagliosa and Barbosa 2006). Species composition is therefore typically transitional, meaning that these regions have a key role in exchanges between the watershed and the marine system. In fact, the muddy sediments at inner estuarine portions represent a shifting gradient from the gross particles in freshwater habitats to finer sandy sediments in outward estuaries. Notwithstanding, the outer estuaries present the higher species richness, a pattern more related with the greater water renovation at the entrance of the system. Contrary to coastal plain estuaries, differences in species numbers and abundances along the main axis of coastal lagoons may present a more complex pattern, with intra-lagoon gradients (Bemvenuti and Netto 1998; Bemvenuti and Rosa Filho 2000; Fonseca and Netto 2006; Netto et al. 2012). Still, although such general patterns are variable among lagoons, many open and intermittently open/closed lagoons show higher species richness and/or densities at the outermost portions (i.e., Patos, Ibiraquera, Conceição, Laguna, and Lagoinha do Leste). Those spatial patterns are probably related to the site-specific inputs of salt water, presence of aquatic vegetation, and the heterogeneity of sedimentary habitats found within lagoons.
Polychaete annelids and oligochaetes are the richest taxonomical groups in SBE estuarine systems, followed by bivalves, gastropods, crustaceans, and insects. Altogether, 52 species of polychaetes, 19 of crustaceans, 14 of bivalves, 11 of oligochaetes, and 7 of gastropods, as well as 50 species of insects, have been reported for local lagoons and coastal plain estuaries. These numbers should yet be seen with caution because they represent a consensus among local experts in which just some of the previous taxa were well studied in terms of taxonomy, biology, and ecology. Species richness ranged from 41 to 74, but the mean richness by sample is very similar. The highest densities occur in the open lagoons while the lowest in the coastal plain estuaries, where the species diversity is high (Table 6.4, Fig. 6.6). The numerically dominant and most frequent macrofaunal species show similar patterns among open/closed lagoons, open lagoons, and coastal plain estuaries.
Mean (± SE) macrofaunal species richness, Shannon diversity (H′), and density (ind/m2) in inner and outer portions (columns) within closed lagoons, Intermittently Closed/Open Lagoons (ICOLL), open lagoons, and Coastal Plain Estuaries (CPE) in the Brazilian Southern Ecoregion (SBE). Black dots represent the total mean value (±SE) of the descriptors in each type of estuary
The gastropod Heleobia australis, a small opportunistic species (around 5 mm), occurs in all estuaries of SBE, including the freshwater closed lagoons. H. australis may be particularly abundant in vegetated sites, including drifting algae. This species displays efficient morphophysiological strategies of dispersion and occupation of non-colonized areas (i.e., fluctuation by retaining air inside their light shells) that allow them to numerically dominate most of the SBE lagoons. This species can be considered tolerant to pollution, reaching up to 300,000 ind/m2 near urban centers (Danulat et al. 2002).
In the three typical estuarine systems of SBE (coastal plain estuaries, open/closed lagoons, and permanently open lagoons), the polychaetes Heteromastus similis, Laeonereis acuta, and Nephtys fluviatilis are often recorded in high numbers. They inhabit the subtidal and intertidal zones in mangroves, salt marshes, and tidal flats. At the mangrove, the tentacle-crowned suspension-feeder polychaete Manayunkia brasiliensis is an endemic species in southern and southeastern Brazil with a highly patched distribution (Pagliosa et al. 2016). The polychaete Nephtys fluviatilis, usually found in less saline waters, is an active carnivorous and deposit feeder, predating on L. acuta and H. similis (Bemvenuti 1987a), meiofaunal ostracods and nematodes. The infaunal L. acuta and H. similis escape predation in deeper (approximately 20 cm) sediment strata. In superficial sediments, the juveniles of L. acuta are under stronger predation pressure, which controls species abundance (Bemvenuti 1992). The major predators of infaunal species in shallow mudflats are the small crab, Rhithropanopeus harrisii and juvenile crabs Cyrtograpsus angulatus and Callinectes sapidus and the shrimp Farfantepenaeus paulensis, that use shallow areas during warm months as nursery grounds (Bemvenuti 1987b; Bemvenuti 1997a).
The amphipod Monocorophium acherusicum and the tanaid Monokalliapseudes schubarti are very abundant in open lagoons and coastal plain estuaries. The tanaid shows at least six annual cohorts (Fonseca and D’Incao 2003), forming U-shaped tubes up to 15 cm deep in intertidal and shallow water muddy sands. It is a deposit and filter-feeder (Montagnolli et al. 2004; Freitas-Júnior et al. 2013), being the main prey for fishes (Micropogonias furnieri and Odontesthes bonarienses) and other crustaceans (F. paulensis, C. sapidus, and Neohelice granulata). The species also plays an essential role in estuarine trophic webs by transferring energy from the detritus compartment to higher trophic levels.
The bivalve Erodona mactroides occurs in coastal lagoons, from Laguna to southernmost of the SBE (Bemvenuti 1997b; Netto and Pereira 2008). They occur in subsurface sediments and can reach densities of more than 20,000 ind/m2 in shallow sublittoral areas. Their adult stocks are usually located at inner lagoon (or near sites with freshwater input) but the larvae can be carried following freshwater discharges, reaching the sheltered mudflats where they successfully recruit. In the upper estuary, all size classes are present (larger individuals reaching up to 30 mm) but at lower densities (461 ind/m2), reaching an average biomass of 281 g/m2, while in the lower estuary, despite the higher densities, the average biomass rarely exceeds 105 g/m2 (Bemvenuti et al. 1978). The key ecological role of this species is linked to its importance as one of the main prey for decapods such as Callinectes sapidus. Moreover, E. mactroides also influence the occurrence of the estuarine cirriped Amphibalanus improvisus, which in the absence of any other hard substrate, attaches to their shells (Bemvenuti 1997b).
The chironomids Caladomya ortoni, Fissimentum sp., and Polypedilum are the most frequent species, and along with Aedokritus, Goeldichironomus, the crustacean Sinelobus stanfordi and the oligochaete Bothrioneurum numerically dominate the bottoms of closed lagoons. In general, chironomid species in coastal lakes presents a patchy distribution related with the food availability (Lemes-Silva et al. 2014, 2016). Macrofaunal predators are highly dependent on variations of prey abundance (like the ostracod species Stenocypris major and Cytheridella ilosvayi, other chironomids and copepods), while macrofaunal herbivores usually forage on the abundant resources found in soft-bottoms throughout the year (wood detritus, coarse and fine particulate organic matter, and microphytobenthos).
The macrofauna from SBE estuaries is predominantly composed of microphages (subsurface deposit-feeders, surface deposit-feeders, and suspension-feeders). Microphages handle food particles in the bulk using only part of the feeding apparatus, while macrophages are nimble organisms that move through sediments in search of their prey. Among the microphages, surface deposit-feeders and suspension-feeders are relatively more abundant in open lagoons, whereas subsurface deposit-feeders dominate in closed lagoons (Fig. 6.7). The occurrence patterns of feeding groups among estuarine types in SBE seem to be related with a fine scale composition and distribution of the resources in sediments, and with the organisms’ ability to handle food (Pagliosa et al. 2005). Surface deposit-feeders and suspension-feeders conduct food towards the mouth using their grooved palps or tentacular crowns with branched and ciliated lobes. In contrast, subsurface deposit-feeders swallow bulk particles with less handling of food. Microphages that extensively handle food tend to inhabit fine sands, feeding sites with high phytoplankton and microphytobenthos productivity. However, these patches of high food quality may be ephemeral, that is, environments where the nutritional status changes quickly due to continuous material flux at the sediment surface. On the other hand, microphages would occur in more protected sites. They consist of species that ingest sediments without much effort in sorting and only digest organic matter along with microorganisms associated to the sediment matrix.
6.4 Temporal Trends
Knowledge of seasonal fluctuation of benthic fauna is scanty along the SBE. In open lagoons, simultaneous comparison of seasonal variability between estuarine meiofauna and macrofauna indicates a clear temporal asynchrony in their variation. High abundance and richness of the meiofauna is evident during autumn and winter, whereas the macrofauna is more diversified and abundant during summer and spring periods (Fonseca and Netto 2006; Meurer and Netto 2007). Such divergent temporal trend exhibited by the meiofauna and macrofauna, also observed in some North American estuaries (e.g., Montagna and Kalke 1992), might result from a complex array of variables and processes, such as competition for food sources, predatory pressures, and differential response to disturbances, all ultimately linked to the contrasting mechanisms for diversity maintenance.
The reproduction and recruitment of macrobenthic species in SBE open lagoons are related to increases in temperature, benthic production, and sediment organic content during summer periods, which results in increased macrobenthic abundance and species number (Bemvenuti 1987a; Rosa and Bemvenuti 2006; Fonseca and Netto 2006; Meurer and Netto 2007). In the permanently open Patos and Laguna lagoons, for example, extreme values in total macrofauna density between winter and summer are 9000 and 17,000 ind/m2 in Patos and 10,600 and 18,900 ind/m2 in Laguna, respectively (Bemvenuti 1987a; Fonseca and Netto 2006). In contrast to permanently open lagoons, temporal oscillations of benthic communities in temporarily open/closed lagoons are primarily modulated by the inlet dynamics. Intermittent breaching of lagoon mouth leads to remarkable changes in its physical-chemical environment over short time periods (hours/days), which in turn triggers major biological responses in the benthic compartment. The barrier breaching results in strong advection and flushing into the sea. Together with sediment scouring from the lagoon, microphytobenthic biomass, sediment organic content, and benthic fauna numbers decrease. Following the shock produced by breaching, a new benthic fauna association slowly emerges (Netto et al. 2012). Many lagoons in South Africa and Australia experience seasonal opening and closure of inlets, with clear seasonal variations of streamflow and onshore sediment transport (Allanson and Baird 2008; Ranasinghe and Pattiaratchi 2003). This is not the case of SBE lagoons, even in the southern portion where rainy periods are more pronounced (Sbruzzi 2015). Despite species similarities among lagoons and coastal plain estuaries, the temporal dynamics of numerically dominant species seems to be quite different. At coastal plain systems, the population peaks in abundance (and greatest spatial variation of data within each estuary) of major species occurs in autumn-winter, while at lagoon systems they occur in summer-spring (Rosa Filho and Bemvenuti 1998).
Macrofaunal communities respond to environmental fluctuations at different time scales by changing their composition and structure (Morrisey et al. 1992). Seasonal cycles of benthic macrofauna (Alden et al. 1997; Ysebaert et al. 2005) and their interannual fluctuations are broadly associated with temperature, rainfall, and substrate alterations (Herman et al. 2001; Teske and Wooldridge 2003). El Niño Southern Oscillation (ENSO) has been extensively studied on the southern portion of SBE (e.g., Odebrecht et al. 2017 and references herein), where it has a major role in controlling the long-term variability of the composition and abundance of benthic invertebrates. In the last 20 years, large interannual variations of freshwater discharge (198 m3/s to 4021 m3/s) and four El Niño events (1997–1998; 2002–2003; 2009–2010; 2014–2015) affected the species composition by sustaining oligohaline and decreasing estuarine species numbers. During this same period, species numbers presented large interannual variability, with higher values (>28,000 ind/m2) observed during the polyhaline summer seasons of 1997, 1999, 2000, 2008, and 2013, and lower (<6000 ind/m2) related to the oligohaline periods influenced by El Niño events. However, low density values might also be related to extremely dry conditions (high salinity and low freshwater discharge) as observed in summer of 2005 (Fig. 6.8; Colling et al. 2010). In the main channel of the Patos Lagoon (>10 m deep), variations of abundance and species composition can also be attributed to a natural macrobenthic response related to environmental variations. Along the year 2000, for example, high salinity values decreased through seasons to almost a limnetic scenario in spring, reducing diversity and abundance of marine species. A contrasting situation was observed in 2006 when the maintenance of brackish waters throughout the year favored the input and survival of marine species from coastal adjacent areas inside the estuary, increasing macrobenthic diversity mainly by marine mollusks and polychaete worms (Pinotti et al. 2011).
The response of individual macrobenthic species to ENSO may differ. The El Niño events affected negatively the numerically dominant Monokalliapseudes schubarti, whose density values drop from a mean of higher than 5000 ind/m2 to less than 400 ind/m2 (Rosa and Bemvenuti 2006; Bemvenuti and Colling 2010) (Fig. 6.8). In contrast, intense recruitment of the bivalve Erodona mactroides recurrently occurs associated to El Niño events and pos-El Niño periods. Depending on synchronicity of reproduction and estuarine discharge, the larvae of E. mactroides generated by the adult reproductive stocks in the upper Patos Lagoon are carried by ebb tides during spring and summer, successfully recruiting in sheltered embayments within low estuarine regions (Bemvenuti 1997b; Colling et al. 2010).
6.5 Conservation, Impacts, and Management Issues
Four different categories of marine protected areas (MPAs) are present in estuaries from SBE: the Marine Extractive Reserve (Resex) of Pirajubaé and the Carijós Ecological Station, in the north, with small coastal plain estuaries bordered by mangroves; the Southern Right Whale Environmental Protection Area, in the center, that includes the inlet region of Garopaba and Ibiraquera temporarily open/closed lagoons, fragments of saltmarshes of Laguna, and the Camacho and Urussanga lagoons; and the Lagoa do Peixe National Park, in the south, that encompass total area of this temporarily open/closed lagoon. MPAs together with the Brazilian National Plan of Coastal Management (PNGC) and supported state and municipal plans, as well as several other legal, planning, and conservation measures could lead to a more sustainable management of coastal ecosystems and resources. However, despite almost three decades of establishing the PNGC, the overlap between laws, policies, plans, and provisions relevant to coastal planning and decision-making has not avoided unsustainable practices, impacts, and habitat loss all over the SBE estuaries. Even within the MPAs, SBE estuaries are under constant environmental pressure (e.g., artificial opening of Peixe and Ibiraquera lagoons, construction of roads in Resex Pirajubaé) that lead to losses of habitat and taxonomic and functional biodiversity. Although coastal management efforts in SBE have stimulated sound technical analysis, information gaps and inadequate exchange and coordination of information within and between government, civil society, private sector, and academia are commonplace.
Interactions between humans and SBE estuaries occur at least since the middle Holocene. Signs of pre-ceramic coastal populations can still be seen by some of the world’s largest shell mounds or “sambaquis” (in some cases reaching 30 m high and several hundred meters in diameter) predominantly composed by the estuarine carib pointed-venus or berbigão Anomalocardia flexuosa (DeBlasis et al. 2007; Colonese et al. 2017). Over the time, the lagoons mouths, particularly Patos Lagoon, became important sites of commerce, which magnified environmental changes (e.g., Seeliger and Odebrecht 2010). Human interactions with SBE estuaries have continued and accelerated over the past decades, so that at present no estuarine system is in its natural state.
A survey in the current literature of SBE accounted for the main causes of changes on sediment characteristics, the associated environmental impacts, and their consequences for the benthic fauna (Table 6.5):
-
1.
The growing urbanization in estuaries is primarily expressed by filling activities over the margins for the construction of roads, ports, commerce and households, promoting physical suffocation on the sediment surface, causing anoxia and habitat suppression. These practices usually result in the death of benthic organisms, losses of species, and assemblage functional diversity. A recent long-term local study showed that despite the potential for faunal colonization provided by the relative improvement of the conditions surrounding impacted areas by landfilling, the effective restoration did not occur once the stressors were not removed (i.e., pollutants in sediments and changes due to sediment refilling) (Pagliosa et al. 2016).
-
2.
The construction of infrastructure and the removal of coastal vegetation (Caruso 1990; Rovai et al. 2012) are among the causes of changes on the average grain size of sediments, which affect sorting, rates of sediment transport, and the local biogeochemistry cycles (Pagliosa et al. 2005, 2006b). The changes directly affect estuarine functions through alterations in the trophic guilds of benthic meiofauna and macrofauna (Pagliosa et al. 2012; Felix et al. 2015), mainly affecting filter-feeder, surface deposit-feeder, and burrower organisms. Those activities have caused the reduction of stocks of the bivalve Anomalocardia flexuosa, the white shrimp Litopenaeus schmitti, and the pink shrimp Farfantepenaeus brasiliensis, resources used by local fishing communities (Pezzuto and Echternacht 1999; Spínola et al. 2014; Pezzuto and Souza 2015). The coastal construction has attracted hard-bottom organism (Pagliosa et al. 2012a).
-
3.
The dredging and sediment deposition activities with overloading suspended material result in change of depth, coarser sediment deposits, increased turbidity due to resuspension of silt and clay (Schettini et al. 2000; Bemvenuti et al. 2005), and remobilization of metals. The increasing turbidity has implications for surface deposit-feeder and filter-feeder organisms, as well as for primary productivity. Changes in water compounds and in sediments have altered both nutrient dynamics (Pagliosa et al. 2005) and the relationships among several types of chemicals (Pagliosa et al. 2006a,b). Dredged areas can experience drastic decrease of both species richness and density, which requires efficient strategies of resilience of the dominant species to minimize the effects of these impact sources (Bemvenuti et al. 2005). In disposal sites at coastal marine zone, the absence of detrimental effects on benthic assemblages suggests that the resident biota can be adapted to dynamic sedimentary conditions, and that the dispersion of dredge material through the water column might minimize sediment deposition and damage to the benthic fauna (Angonesi et al. 2006).
-
4.
A recent study indicated that mangrove and saltmarshes are the most sensible areas to marine spills (Silva et al. 2008). Marine spill incidents and harbor/marina activities cause sink of pollutants, changes in coastal vegetation, habitat suppression, and changes in water and sediment biogeochemistry. A sulfuric acid spill within Patos Lagoon altered the water quality, causing physical damages in zooplankton (Montú and Gloeden 1998) and the decline of macrofaunal richness and abundance (Bemvenuti et al. 2003). Another impact related to harbor activities is the larvae transport by ship’s ballast water, which plays an important role in the dispersion of nonindigenous species. The occurrence of the decapod Rhithropanopeus harrisii (D’incao and Martins 1998) and the bivalve Limnoperna fortunei in SBE are attributed to this source of larval dispersion, being L. fortunei highly dependent of limnetic scenarios to their dispersion through estuaries (Capítoli et al. 2008).
-
5.
Along SBE, more than half of the coastal lagoons’ entrance channels become temporarily blocked by the buildup of marine sandbars. Artificial breaching of open/closed lagoons involves dredging and/or bulldozing the lagoon inlet at a level lower than natural breakout and has occurred for various reasons. While alleviating actual or perceived water quality is often cited as a trigger for artificial openings, the opening alone is not likely to significantly improve water quality. The limited tidal flushing and exchange efficiencies means that pollutants (particularly those entering from tributaries furthest from the entrance) may be moved around within the lagoon but may not be removed (e.g., Spurway et al. 2000). Artificial opening aiming to enhance fish and prawn recruitment and subsequent production is another reason always cited for dredging/bulldozing the lagoon, but its efficiency is largely unclear and virtually impossible to address without a detailed sampling and analysis of offshore and coastal larval populations. Finally, unplanned and irregular occupation within lagoon floodplains can further result in pressure for intervention to artificially breach lagoon inlet to avoid damage or inconvenience to low-lying properties. As inlet dynamics play a key role in the overall functioning coastal lagoons, intervention in the behavior of lagoon entrances is generally accompanied by negative environmental impacts (e.g., crash of the population of macrobenthic invertebrates, fish and vegetation and increase of sediment infill), potentially reducing lagoon resilience (Whitfield and Bate 2007; Crippa et al. 2013; Netto et al. 2012; Netto and Fonseca 2017). Monitoring, establishment of local estuarine management plan with short- and long-term goals, and permanent policy review would ensure that the most ecologically appropriate and cost-effective options are being implemented at any given location.
-
6.
The Center-North of SBE ecoregion is the main Brazilian area for aquaculture, being responsible for ~21,000 tons/year of molluscs and shrimps. The seabed enrichment as a by-product of aquaculture has large impacts on the availability of food for benthic fauna. Aquaculture activities reduce benthic diversity, increase anaerobic metabolism in the sediment, and increase both the rate of sedimentation and organic matter content in the sediment (Bonetti et al. 2007; Netto and Meurer 2007; Netto and Valgas 2007, 2010). Aquaculture in general is one of the ways by which cultured and associated species can be dispersed outward from their native regions. Particularly, the commercial oyster industry is responsible for accidental transport of many harmful shell-borer polychaetes as Polydora haswelli, P. ecuadoriana, P. carinhosa (Radashevsky et al. 2006), and Boccardiella bihamata (Junqueira et al. 2009). These worms bore into shells of live oysters Crassostrea brasiliana, C. gigas, C. rhizophorae, the barnacle Megabalanus and empty shells of the gastropods Pugilina morio, Stramonita haemastoma, Strombus pugilis, and Tegula viridula inhabited by hermit crabs Clibanarius vittatus, Paguristes tortugae and Pagurus brevidactylus.
-
7.
Fishery along SBE estuaries is a small-scale or artisanal activity. The most important estuarine fishery, the shrimp Penaeus paulensis, is overexploited or collapsed in the main fishery grounds, Patos and Laguna Lagoons (Sunye et al. 2014; Haimovici and Cardoso 2017). Local shrimp bottom tending gears may be either active (1—a hand light mini trawl, known as “berimbau”; 2—bag nets, known as coca; 3—motorized otter trawl, only in Patos Lagoon) or passive (fyke nets, normally used in groups, known as “aviãozinho”) (Netto and Perereira 2008; Haimovici and Cardoso 2017). Experimental evaluations of trawling on macrofauna of mud-bottoms (Angonesi and Bemvenuti 2004) and on meiofauna and macrofauna of sandy vegetated and unvegetated bottoms (Costa and Netto 2014) showed negative impacts on superficial benthic forms, such as the gastropod H. australis and the ostracod Cyprideis multidentate. The passive shrimp fishing gear “aviãozinho,” a very unselective gear (Vieira et al. 1996), is largely the actual dominant gear for shrimp in SBE lagoons. It is composed by a group of five to seven fyke nets (25 mm mesh size) set in contact to the bottom, fixed with stakes forming a cage-like structure (around 30 m2). The nets, kept in the same place during months, change sediment composition, decrease microphytobenthic biomass, and affect the trophic structure of the meiofauna (Netto and Pereira 2008).
-
8.
The rapid and unplanned urbanization without the appropriate treatment of residuals have changed the water and sediment estuarine properties (Pagliosa et al. 2005, Pagliosa et al. 2006a, 2006b), even promoting the release of emerging contaminants such as synthetic hormones and the herbicide glyphosate. Estuarine macrobenthic communities and populations, in turn, have shown altered responses in abundance, richness, size of individuals, biomass and secondary productivity, followed by DNA damage in the polychaete Laeonereis culveri (Pagliosa and Barbosa 2006; Weis et al. 2017).
The measurements of what might threaten estuarine outputs—namely impacts to the ecosystem itself as well as the social and economic systems to which they are linked—can be greatly facilitated by the use of efficient chemical markers of eutrophication and bioindicator tools. In this sense, the delivery of more integrated policy and management strategies is intrinsically linked to the conservation of estuarine functions, towards the preservation of complex mechanisms and processes that generate goods and services, even if not directly benefiting people at a first moment (Mahoney and Bishop 2017). Thus, assessments of estuarine quality might be highly improved when populations, species, or species guilds, with distinguishing properties and capabilities to support particular functions within an ecosystem, are used as real-world indicators. Productivity is also an important ecosystem function to consider because while it may not often be a direct service, it underpins many other kinds of output.
The environmental quality status along 27 SBE estuarine systems was analyzed based on the AZTI Marine Biotic Index (AMBI; Borja et al. 2000) and Trophic Index (TRIX; Vollenweider et al. 1998). According to the AMBI index, the environmental quality was largely classified as moderately disturbed (80%), whereas 20% of the sites were of slightly or undisturbed classifications (Fig. 6.9a). As estuaries are constantly subjected to natural sources of stress, they tend to be dominated by more tolerant taxa, and the indices based on macrofaunal indicator species in general tend to underestimate habitat quality assessments (Tweedley et al. 2016). However, regardless of natural background variability, consistent AMBI responses to contamination gradients have been found in Brazilian estuaries (Brauko et al. 2015, 2016), meaning that the AMBI levels of disturbance in the SBE estuaries might be even informative to some extent. Despite maintaining similar moderate ecological qualities, the proportion of ecological groups of fauna composing the AMBI values suggests community responses linked to salinity and hydrodynamic changes along the estuaries (see bar colors in Fig. 6.9). Freshwater communities of closed lagoons were dominated by ecological group III or tolerant species, followed by the absence of group I, composed of species more sensitive to disturbance. As the estuaries change with increasing salinity inputs from temporarily open/closed lagoons to open and coastal plain estuaries, the proportion of ecological groups I (of sensitive species) and IV (of opportunist species), in general, increases, while the relative contribution of group III decreases.
Environmental quality shown by: (a) AZTI Marine Biotic Index (AMBI) and percentage of ecological groups of macrofauna (I—sensitive; II—indifferent; III—tolerant; IV—second order opportunistic fauna; V—first order opportunistic fauna). Index values were calculated and their ecological status was therefore attributed from undisturbed to slightly disturbed and moderately disturbed. (b) Water trophic state estimated by trophic Index (TRIX) (mean ± SE, n = 160), from ultra-oligotrophic to eutrophic conditions in SBE estuaries. Indices were applied over inner and outer stations within closed, intermittently closed/open (ICOLL), and open lagoons, and coastal plain estuaries
Such natural subsequent changes in the physical-chemical settings and transitional communities along the estuarine systems have not yet led to inconsistencies in the habitat quality diagnosis shown by both AMBI and TRIX. These indices are based on distinct biological compartments, but agreed on their diagnosis of environmental stress despite the additional influence of natural background variability. Similarly, average values of TRIX indicated that the SBE estuarine waters are mesotrophic in general (Fig. 6.9b). However, TRIX values indicated that nearly 40% of open and closed estuarine waters were eutrophic, mostly driven by high nitrate and low oxygen concentrations within those systems. The proximity to urbanized areas and the lower resident water time (hours to weeks) also favor loads of served water.
Despite relatively optimistic macrofaunal health scenarios of slight and moderate disturbance, previous evidence of diffuse pollution impacts highlight important local constrains for estuarine conservation. In particular, sediment and water chemical markers as well as benthic community parameters have responded to the increasing urbanization in estuaries of the SBE within the last decade (Pagliosa et al. 2005, Pagliosa and Barbosa 2006; Felix et al. 2015). In addition, more recent findings in Weis et al. (2017) showed that some of the more urbanized estuaries are experiencing loss of environmental quality linked to changes in a bioindicator estuarine species, the polychaete Laeonereis acuta (see details of comparisons between Urban and Non-urban estuaries in Fig. 6.10). There is very convincing evidence of molecular damage with increased micronuclei frequency, higher individual biomass and size, as well as losses in population production (P/B ratios) associated to a set of contaminants including total nitrogen, aluminum, copper, and lead contents (Fig. 6.10). The rapid sub-cellular or molecular changes provide the detection of early stages of pollution (from hours to days), and as responses progress to the higher biological levels of populations (e.g., biomass or body size) and communities (e.g., indicator species indexes) the responses take longer periods to emerge (from weeks to years). Eventual biomonitoring protocols of anthropic impacts could therefore benefit from the combination of rapid sub-cellular indicators along with biotic indices based on community responses of higher ecological and functional relevance.
Biomarkers of different levels of biological organization (micronuclei frequency, body size, individual biomass and P/B ratio) in Laeonereis acuta (mean ± SE) along urbanized and non-urbanized estuaries of the Southern Brazil Ecoregion (SBE). Adapted from Weis et al. (2017)
6.6 Final Considerations
Despite sensitive to anthropic impacts, the estuarine systems included in the Southern Brazilian Marine Ecoregion still present satisfactory conditions that sustain benthic biodiversity and ecosystem goods and services. Notwithstanding, the growing coastal urbanization intensified by agricultural and industrial activities may change the relatively optimistic scenario observed in the last decades.
As other impacts, climate change is expected to intensify environmental risks and amplify extreme weather events and coastal hazards. Although the slow-onset nature of climate changes, there is an urgent need to build institutional capacity to better understand and address climate change impacts in estuaries and chart adaptive pathways. Thus, the systematic application of long-term monitoring protocols (such as the Brazilian Monitoring Network for Coastal Benthic Habitats, ReBentos) within estuaries is essential to understand and predict changes. Besides, recommendations (see Polette et al. 2015) for building adaptive capacity, resilience, and sustainability in coastal communities should include: (1) raise public awareness about the coast and climate change through active social learning processes; (2) create opportunities for meaningful public participation in coastal management efforts; (3) integrate and mainstream coastal management, from pollution to disaster risk reduction, and climate change adaptation efforts.
References
Abreu PC, Odebrecht C, González A (1994) Particulate and dissolved phytoplankton production of the Patos lagoon estuary, southern Brazil: comparison of methods and influencing factors. J Plankton Res 16(7):737–753
Abreu PC, Bergesch M, Proença LA et al (2010) Short- and long-term chlorophyll a variability in the shallow microtidal Patos lagoon estuary, Southern Brazil. Estuar Coasts 33:554–569
Abreu PC, Marangoni JC, Odebrecht C (2017) So close, so far: differences in long-term chlorophyll a variability in three nearby estuarine-coastal stations. Mar Biol Res 13:9–21
Alden RW, Weisberg SB, Ranasinghe JA (1997) Optimizing temporal sampling strategies for benthic environmental monitoring programs. Mar Pollut Bull 34(11):913–922
Allanson BR, Baird D (eds) (2008) Estuaries of South Africa. Cambridge University Press, Cambridge
Alvares CA, Stape JL, Sentelhas PC et al (2014) Kӧppen’s climate classification map for Brazil. Meteorol Z 22(6):711–728
Angonesi LG, Bemvenuti CE (2004) Effects of shrimp trawling upon macrobenthic fauna in Patos lagoon estuary. J Coast Res SI 39:1368–1372
Angonesi LG, Bemvenuti CE, Gandra MS (2006) Effects of dredged sediment disposal on the coastal marine macrobenthic assemblage in Southern Brazil. Braz J Biol 66(2A):413–420
Anthony A, Atwood J, August P (2009) Coastal lagoons and climate change: ecological and social ramifications in U.S. Atlantic and Gulf coast ecosystems. Ecol Soc 14(1):8
Bemvenuti CE (1987a) Predation effects on a benthic community in estuarine soft sediments. Atlantica 9(1):5–32
Bemvenuti CE (1987b) Macrofauna bentônica da região estuarial da Lagoa dos Patos, RS – Brasil. Publ Acad Ciências do Estado de São Paulo 54(1):428–459
Bemvenuti CE (1992). Interações biológicas da macrofauna bentônica numa enseada estuarina da Lagoa dos Patos, RS, Brasil. Doctoral thesis, Universidade de São Paulo, São Paulo
Bemvenuti CE (1997a) Benthic invertebrates. In: Seeliger U, Odebrecht C, Castello JP (eds) Subtropical convergence environments: the coast and sea in the Southwestern Atlantic. Springer, Berlin, pp 43–47
Bemvenuti CE (1997b) Unvegetated intertidal flats and subtidal bottoms. In: Seeliger U, Odebrecht C, Castello JP (eds) Subtropical convergence environments: the coast and sea in the Southwestern Atlantic. Springer, Berlin, pp 78–85
Bemvenuti CE, Colling LA (2010) As comunidades de macroinvertebrados bentônicos. In: Seeliger U, Odebrecht C (eds) O estuário da Lagoa dos Patos, um século de transformações. Editora da FURG, Rio Grande, pp 101–114
Bemvenuti CE, Netto GA (1998) Distribution and seasonal patterns of the sublittoral benthic macrofauna of Patos lagoon (South Brazil). Rev Bras Biol 58:211–221
Bemvenuti CE, Rosa Filho JS (2000) Estrutura e dinâmica das associações de macroinvertebrados bentônicos dos ambientes estuarinos do Rio Grande do Sul: um estudo de caso. In: Anais do Workshop Avaliação e Ações Prioritárias para a Zona Costeira e Marinha, PROBIO, Federal University of Rio Grande, Rio Grande
Bemvenuti CE, Capítoli RR, Gianuca NM (1978) Estudos de ecologia bentônica na região estuarial da Lagoa dos Patos. II - Distribuição quantitativa do macrobentos infralitoral. Atlântica 3:23–32
Bemvenuti CE, Rosa Filho JS, Elliott M (2003) Changes in soft-bottom macrobenthic assemblages after a sulphuric acid spill in the Rio Grande Harbor (RS, Brazil). Braz J Biol 63(2):183–194
Bemvenuti CE, Angonesi LG, Gandra MS (2005) Effects of dredging operations on soft bottom macrofauna in a harbor in the Patos lagoon estuarine region of Southern Brazil. Braz J Oceanogr 65(4):573–581
Bernardino AF, Netto AS, Pagliosa PR et al (2015) Predicting ecological changes on benthic estuarine assemblages through decadal climate trends along Brazilian marine ecoregions. Estuar Coast Shelf Sci 166:74–82
Bonaglia S, Nascimento FJA, Bartoli M et al (2014) Meiofauna increases bacterial denitrification in marine sediments. Nat Commun 16:1–9
Bonetti C, Bonetti J, Barcellos RL (2007) Caracterização sedimentar e geoquímica de sistemas costeiros com ênfase na avaliação da influência de sítios de cultivo de moluscos. In: Barroso GF, Poersch LHS, Cavalli RO (eds) Sistemas de cultivos aquícolas na zona costeira do Brasil: recursos, tecnologias, aspectos ambientais e sócio econômicos. Museu Nacional, Rio de Janeiro, pp 139–150
Borja A, Franco J, Pérez V (2000) A marine biotic index to establish the ecological quality of soft-bottom benthos within European estuarine and coastal environments. Mar Pollut Bull 40:1100–1114
Brauko KM, Souza FM, Muniz P et al (2015) Spatial variability of three benthic indices for marine quality assessment in a subtropical estuary of Southern Brazil. Mar Pollut Bull 91:454–460
Brauko KM, Muniz P, Martins CC et al (2016) Assessing the suitability of five benthic indices for environmental health assessment in a large subtropical South American estuary. Ecol Indic 64:258–265
Capítoli RR, Colling LA, Bemvenuti CE (2008) Cenários de distribuição do mexilhão dourado Limnoperna fortunei (Mollusca – Bivalvia) sob distintas condições de salinidade no complexo lagunar Patos-Mirim, RS – Brasil. Atlantica 30(1):35–44
Caruso MML (1990) O desmatamento da Ilha de Santa Catarina de 1500 aos dias atuais. Editora da UFSC, Florianópolis
Castelão RM, Möller OO Jr (2003) Sobre a circulação tridimensional forçada por ventos na Lagoa dos Patos. Atlantica 25(2):91–106
Chapman PM (2012) Management of coastal lagoons under climate change. Estuar Coast Shelf Sci 110:32–35
Colling LA, Bemvenuti CE, Gandra MS (2007) Seasonal variability on the structure of sublittoral macrozoobenthic association in the Patos lagoon estuary, southern Brazil. Iheringia Ser Zool 97:257–263
Colling LA, Bemvenuti CE, Pinotti RM (2010) Temporal variability of the bivalve Erodona mactroides BOSC, 1802 during and after El Niño phenomenon (2002/2003) in a subtropical lagoon, Southern Brazil. Acta Limnol Bras 22(4):410–423
Colonese AC, Netto SA, Francisco AS et al (2017) Shell sclerochronology and stable isotopes of the bivalve Anomalocardia flexuosa (Linnaeus, 1767) from southern Brazil: implications for environmental and archaeological studies. Palaeogeogr Palaeoclimatol Palaeoecol 484:7. https://doi.org/10.1016/j.palaeo.2017.01.006
Cooper JAG (1994) Lagoons and microtidal coasts. In: Carter RWG, Woodroffe CD (eds) Coastal evolution. Late quaternary shoreline morphodynamics. Cambridge University Press, Cambridge, pp 219–265
Cooper JAG, Wright CI, Mason TR (1999) Geomorphology and sedimentology of South African estuaries. In: Allanson BR, Baird D (eds) Estuaries of South Africa. Cambridge University Press, Cambridge, pp 5–25
Costa CSB, Davy AJ (1992) Coastal saltmarsh communities of Latin America. In: Seeliger U (ed) Coastal Plant Communities of Latin America. Academic Press, New York, pp 179–212
Costa KG, Netto SA (2014) Effects of small-scale trawling on benthic communities of estuarine vegetated and non-vegetated habitats. Biodivers Conserv 23:1041–1055
Crippa LB, Stenert C, Maltchik L (2013) Does the management of sandbar openings influence the macroinvertebrate communities in southern Brazil wetlands? A case study at Lagoa do Peixe National Park - Ramsar site. Ocean Coast Manag 71:26–32
Cunha SR, Asmus M, Costa CSB (2005) Production dynamics of Spartina alterniflora saltmarshes in the estuary of Patos lagoon (RS, Brazil): a simulation model approach. Brazil J Aquat Sci Technol 9:75–85
D’Incao F, Martins STS (1998) Occurrence of Rhithropanopeus harrisii (Gouls, 1841) in the southern coast of Brazil (Decapoda, Xanthidae). Nauplius 6:191–194
Danulat E, Muniz P, Garcıa-Alonso J et al (2002) First assessment of the highly contaminated harbour of Montevideo, Uruguay. Mar Pollut Bull 44(6):554–565
DeBlasis P, Kneip A, Scheel-Ybert R et al (2007) Sambaquis e paisagem: dinâmica natural e arqueologia regional no litoral do sul do Brasil. Arqueol Sudamer/Arqueol Sul-Amer 3(1):29–61
Dillenburg S, Barboza E, Tomazelli L et al (2009) The Holocene Coastal Barriers of Rio Grande do Sul. In: Dillenburg SR, Hesp PA (eds) Geology and Geomorphology of Holocene Coastal Barriers of Brazil. Springer, Berlin, pp 53–91
Dos Santos AC, Fonseca ALDO, Koerich G (2017) Caracterização do estado trófico e da produção primária dos sistemas estuarinos e lagunares de Santa Catarina (Brasil) e sua relação com a atividade antrópica. XVII Congresso Latino-Americano de Ciências do Mar, Camboriú, SC, Brasil, p 1465–1468
Eichler PPB, Casteão GP, Pimenta FM, Eichler BB (2006) Avaliação da saúde ecológica do Sistema Estuarino de Laguna (SC) baseado nas espécies de foraminíferos e tecamebas. Pesq Geocien 33:101–115
Felix G, Cândido TF, Silveira CB et al (2015) Resposta de associações bênticas ao lançamento de resíduos de drenagem urbana. Brazil J Aquat Sci Technol 19(2):47–57
Fernandes LF, Brandini FP (2010) The potentially toxic diatom Pseudo-nitzschia H. Peragallo in the Paraná and Santa Catarina states, southern Brazil. Iheringia Ser Bot 65:47–62
Fernandes EHL, Dyer KR, Möller OO et al (2002) The Patos lagoon hydrodynamics during an El Nino event (1998). Cont Shelf Res 22(11):1699–1713
Fonseca ALO (2004) Variação sazonal e espacial das características hidroquímicas, dos fluxos de nutrientes e do metabolismo na interface água-sedimento da Lagoa da Conceição (SC, Brasil). Doctoral thesis, University of São Paulo, São Paulo
Fonseca A, Braga ES (2006) Spatial and seasonal variation of dissolved inorganic nutrients and phytoplankton biomass in the pelagic system of the Conceição lagoon, Southern Brazil. J Coast Res 39:1229–1233
Fonseca DB, D’Incao F (2003) Growth and reproductive parameters of Kalliapseudes schubartii in the estuarine region of the Lagoa dos Patos (southern Brazil). J Mar Biol Assoc UK 83:931–935
Fonseca G, Netto SA (2006) Shallow sublittoral benthic communities of the Laguna estuarine system, South Brazil. Braz J Oceanogr 54(1):41–54
Fontes MLS, Abreu PA (2009) Spatiotemporal variation of bacterial assemblages in a shallow subtropical coastal lagoon in Southern Brazil. Microb Ecol 58(1):140–153
Fontes MLS, Suzuki MT, Cottrell MT et al (2011) Primary production in a subtropical stratified coastal lagoon, contribution of anoxygenic phototrophic bacteria. Microb Ecol 61:223–237
Freitas-Júnior F, Christoffersen ML, Araújo JP et al (2013) Spatiotemporal distribution and population structure of Monokalliapseudes schubarti (Tanaidacea: Kalliapseudidae) in an estuary in Southern Brazil. Sci World J 2013:1. https://doi.org/10.1155/2013/363187
Garcia CAE (1997) Hydrographic characteristics. In: Seeliger U, Odebrecht C, Castello JP (eds) Subtropical convergence environments: the coast and sea in the Southwestern Atlantic. Springer, Berlin, pp 18–20
Garcia AM, Vieira JP, Winemiller KO (2003) Effects of 1997-1998 El Niño on the dynamics of the shallow-water fish assemblage of the Patos lagoon estuary (Brazil). Estuar Coast Shelf Sci 57(3):489–500
Grimm AM, Barros VR, Doyle ME (2003) Climate variability in Southern South America associated with El Niño and La Niña events. J Clim 1:35–38
Haimovici M, Cardoso LG (2017) Long-term changes in the fisheries in the Patos lagoon estuary and adjacent coastal waters in Southern Brazil. Mar Biol Res 13:135–150
Haraguchi L, Carstensen J, Abreu PC et al (2015) Long-term changes of the phytoplankton community and biomass in the subtropical shallow Patos lagoon estuary, Brazil. Estuar Coast Shelf Sci 162:76–87
Heip C, Vincx M, Vranken G (1985) The ecology of marine nematodes. Oceanogr Mar Biol Ann Rev 23:399–489
Herman PM, Middelburg JJ, Heip CH (2001) Benthic community structure and sediment processes on an intertidal flat: results from the ECOFLAT project. Cont Shelf Res 21(18):2055–2071
Hesp PA, Giannini P, Martinho C et al (2009) The Holocene barrier systems of the Santa Catarina coast, southern Brazil. In: Dillenburg SR, Hesp PA (eds) Geology and geomorphology of Holocene coastal barriers of Brazil. Springer-Verlag, Berlin Heidelberg, pp 93–133
Junqueira AOR, Tavares MDS, Schaeffer-Noveli I et al (2009) Zoobentos. In: Lopes RM, Coradin L, Pombo VB et al (eds) Informe Sobre as Espécies Exóticas Invasoras Marinhas no Brasil. Ministério do Meio Ambiente, Brasília, pp 145–371
Kapusta SC, Würdig NL, Bemvenuti CE (2002) Distribuição vertical da meiofauna, inverno e verão, no estuário de Tramandaí- Armazém, RS – Brasil. Acta Limnol Bras 14(1):81–90
Kapusta SC, Bemvenuti CE, Würdig NL (2004) Meiofauna spatial-temporal distribution in a subtropical estuary of southern coast Brazil. J Coastal Res SI 39:1238–1242
Kapusta SC, Würdig NL, Bemvenuti CE et al (2005) Meiofauna structure in Tramandaí-Armazém estuary (South Brazil). Acta Limnol Bras 17(4):349–359
Lanari M, Copertino MS (2017) Drift macroalgae in the Patos lagoon estuary (Southern Brazil): effects of climate, hydrology and wind action on the onset and magnitude of blooms. Mar Biol Res 13:1–12
Lemes-Silva AL, Pagiosa PR, Petrucio MM (2014) Inter- and intra-guild patterns of food resource utilization by chironomid larvae in a subtropical coastal lagoon. Limnology 15:1–12
Lemes-Silva AL, Pires JR, Pagiosa PR et al (2016) Distribution of aquatic macroinvertebrate assemblages in a subtropical coastal lake: response to environmental parameters. Fund Appl Limnol 188:113–127
Mahoney PC, Bishop MJ (2017) Assessing risk of estuarine ecosystem collapse. Ocean Coast Manag 140:46–58
McSweeney SL, Kennedy DM, Rutherfurd ID et al (2017) Intermittently closed/open lakes and lagoons: their global distribution and boundary conditions. Geomorphology 292:142. https://doi.org/10.1016/j.geomorph.2017.04.022
Meurer AZ, Netto SA (2007) Seasonal dynamics of benthic communities in a shallow sublitoral site of Laguna estuarine system (south, Brazil). Braz J Aquat Sci Technol 1(2):53–62
Montagna PA, Kalke RD (1992) The effect of freshwater inflow on meiofaunal and macrofaunal populations in the Guadalupe and Nueces estuaries, Texas. Estuaries 15:307–326
Montagnolli W, Zamboni A, Luvizotto-Santos R et al (2004) Acute effects of Microcystis aeruginosa from the Patos lagoon estuary, Southern Brazil, on the microcrustacean Kalliapseudes schubartii (Crustacea: Tanaidacea). Arch Environ Cont Toxicology 46(4):463–469
Montú M, Gloeden IM (1998) O derramamento ácido do navio NT Bahamas e as primeiras consequências sobre o zooplâncton do estuário da Lagoa dos Patos (Rio Grande, RS). Nauplius 6:195–197
Morrisey DJ, Underwood AJ, Howitt L et al (1992) Temporal variation in soft-sediment benthos. J Exp Mar Biol Ecol 164:233–245
Netto SA, Fonseca GF (2017) Regime shifts in coastal lagoons: evidence from free-living marine nematodes. PLoS One 12(2):e0172366
Netto SA, Gallucci F (2003) Meiofauna and macrofauna communities in a mangrove from the Island of Santa Catarina, South Brazil. Hydrobiologia 505:159–170
Netto SA, Meurer AZ (2007) Influência de efluentes de cultivo de camarão na estrutura das comunidades bênticas do Sistema Estuarino de Laguna (SC, Brasil). In: Barroso GF, Poersch LHS, Cavalli RO (eds) Sistemas de Cultivos aquícolas na zona costeira do Brasil: recursos, tenologias, aspectos ambientais e sócio-economicos. Museu Nacional, Rio de Janeiro, pp 129–138
Netto SA, Pereira TJ (2008) Benthic community response to a passive fishing gear in a coastal lagoon (South Brazil). Aquat Ecol 43(2):521–538
Netto SA, Valgas IS (2007) Efeitos de cultivos de mexilhões sobre a estrutura da meiofauna. In: Barroso GF, Poersch LHS, Cavalli RO (eds) Sistemas de Cultivos aquícolas na zona costeira do Brasil: recursos, tenologias, aspectos ambientais e sócio-economcos. Museu Nacional, Rio de Janeiro, pp 119–128
Netto SA, Valgas IS (2010) The response of nematode assemblages to intensive mussel farming in coastal sediments (Southern Brazil). Environ Monit Assess 162:81–93
Netto SA, Domingos AM, Kurtz M (2012) Effects of artificial breaching of a temporarily open/closed estuary on benthic macroinvertebrates (Camacho lagoon, southern Brazil). Estuar Coasts 35:1069–1081
Niekerk L, Merwe JH, Huizinga P (2005) The hydrodynamics of the Bot River Estuary revisited. Water SA 31(1):73–85
Niencheski LFH, Zepka-Baumgarten MG, Cabrera L et al (2006) Patos lagoon: indicators of organic pollution. J Coast Res 39:1356–1359
Odebrecht C, Abreu PC, Bemvenuti CE et al (2010) The Patos lagoon estuary, Southern Brazil. Biotic responses to natural and anthropogenic impacts in the last decades (1979-2008). In: Kennish MJ, Paerl HW (eds) Coastal Lagoons, Critical Habitats of Environmental Change. CRC Press, Boca Raton, pp 433–455
Odebrecht C, Secchi ER, Abreu PC et al (2017) Biota of the Patos lagoon estuary and adjacent marine coast: long-term changes induced by natural and human-related factors. Mar Biol Res 13:3–8
Omachi CY, Tamanaha MS, Proença LAO (2007) Bloom of Alexandrium fraterculus in coastal waters of Itajaí, SC, Southern Brazil. Braz J Oceanogr 55(1):57–61
Pagliosa PR, Barbosa FAR (2006) Assessing the environment–benthic fauna coupling in protected and urban areas of southern Brazil. Biol Conserv 129:408–417
Pagliosa PR, Fonseca A, Bosquilha GE et al (2005) Phosphorus dynamics in water and sediments in urbanized and non-urbanized rivers in Southern Brazil. Mar Pollut Bull 50:965–974
Pagliosa PR, Fonseca A, Barbosa FAR (2006a) Evidence of systemic changes in trace metal concentrations in subtropical estuarine sediments as a result of urbanization. J Coast Res 39:1078–1083
Pagliosa PR, Fonseca A, Barbosa FAR et al (2006b) Urbanization impact on subtropical estuaries: a comparative study of water properties in urban areas and in protected areas. J Coast Res 39:731–735
Pagliosa PR, Cantor M, Scherner F et al (2012) Influence of piers on functional groups of benthic primary producers and consumers in the channel of a subtropical coastal lagoon. Braz J Oceanogr 60(1):65–73
Pagliosa PR, Oortman MS, Rovai AS et al (2016) Is mangrove planting insufficient for benthic macrofaunal recovery when environmental stress is persistent? Ecol Eng 95:290–301
Panitz CMN (1986) Produção e decomposição de serapilheira no mangue do rio Itacorubi, Ilha de Santa Catarina, Florianópolis, Brasil. Doctoral thesis, Federal University of São Carlos, São Carlos
Peixoto AE, Costa CSB (2004) Produção primária líquida aérea de Spartina densiflora Brong (Poaceae) no estuário da laguna dos Patos, Rio Grande do Sul, Brasil. Iheringia Ser Bot 59:27–34
Pérez-Ruzafa A, Marcos C, Pérez-Ruzafa IM, Pérez-Marco M (2011) Coastal lagoons: “transitional ecosystems” between transitional and coastal waters. J Coast Conserv 15:369–392
Pezzuto PR, Echternacht AM (1999) Avaliação de impactos da construção da via expressa SC-sul sobre o berbigão Anomalocardia brasiliana (Gmelin, 1791) (Mollusca: Pelecypoda) na reserva extrativista marinha do Pirajubaé (Florianópolis, SC - Brasil). Atlantica 21:105–119
Pezzuto PR, Souza DS (2015) A pesca e o manejo do berbigão (Anomalocardia brasiliana) (Bivalvia: Veneridae) na Reserva Extrativista Marinha do Pirajubaé, SC, Brasil. Desenv Meio Amb 34:169–189
Pinotti RM, Colling LA, Bemvenuti CE (2011) Temporal dynamics of deep inffralittoral macrobenthic fauna inside a subtropical estuarine environment. Braz J Aquat Sci Technol 15(1):226–241
Polette M, Muehe D, Soares MLG, Glavovic BC (2015) The promise of coastal management in Brazil in times of global climate change. In: Glavovic BC, Kelly M, Kay R, Travers A (eds) Climate change and the coast: building resilient communities. CRC Press, Boca Raton, pp 379–413
Proença LAO (2004) A red water caused by Mesodinium rubrum on the coast of Santa Catarina, Southern Brazil. Braz J Oceanogr 52(2):153–161
Radashevsky VI, Lana PC, Nalesso RC (2006) Morphology and biology of Polydora species (Polychaeta: Spionidae) boring into oyster shells in South America, with the description of a new species. Zootaxa 1353:1–37
Ranasinghe R, Pattiaratchi C (2003) The seasonal closure of tidal inlets: causes and effects. Coast Eng J 45(04):601–627
Rosa LC, Bemvenuti CE (2006) Temporal variability of the estuarine macrofauna of the Patos lagoon, Brazil. Rev Biol Mar Ocean 41(1):1–9
Rosa Filho JS, Bemvenuti CE (1998) Caracterización de las comunidades macrobentónicas de fondos blandos en regiones estuarinas de Rio Grande do Sul (Brasil). Thalassas 18:43–56
Rovai AS, Soriano-Sierra EJ, Pagliosa PR et al (2012) Secondary succession impairment inrestored mangroves. Wetl Ecol Manag 20:447–459
Sbruzzi JB (2015) Análise da dinâmica da embocadura da lagoa do Peixe– RS utilizando dados de sensoriamento remoto orbital. Dissertação de mestrado, Universidade Federal do Rio Grande do Sul, Porto Alegre
Schallenberg M, Larned S, Hayward S et al (2010) Contrasting effects of managed opening regimes on water quality in two intermittently closed and open coastal lakes. Estuar Coast Shelf Sci 86:587–597
Schettini CAF, Pereira FOJ, Spillere L (2000) Caracterização oceanográfica e biogeoquímica dos estuários dos rios Tavares e Defuntos, Reserva Extrativista de Pirajubaé, Florianópolis, SC. Not Técn Fac Ciênc Mar 4:11–28
Schock NT, Murry BA, Uzarski DG (2014) Impacts of agricultural drainage outlets on Great Lakes coastal wetlands. Wetlands 24:297–307
Schratzberger M, Ingels J (2017) Meiofauna matters: the roles of meiofauna in benthic ecosystems. J Exp Mar Biol Ecol. https://doi.org/10.1016/j.jembe.2017.01.007
Seeliger U (2001) The Patos lagoon estuary. In: Seeliger U, Kjerfve B (eds) Coastal marine ecosystems of Latin America. Springer, New York, pp 167–183
Seeliger U, Odebrecht C (2010) O Estuário da Lagoa dos Patos: um século de transformações. Editora da FURG, Rio Grande
Seeliger U, Odebrecht C, Castello JP (1997) Subtropical convergence environments: the coast and sea in the Southwestern Atlantic. Springer, Berlin
Short AD, Klein AHF (eds) (2016) Brazilian beach systems. Springer, Berlin
Sierra de Ledo B, Soriano-Sierra EJ (eds) (1999) O ecossitema da lagoa da Conceição. SDM-FEPEMA, Florianópolis
Silva AF, Klein AHF, Petermann RM et al (2008) Índice de sensibilidade do litoral (ISL) ao derramamento de óleo, para a Ilha de Santa Catarina e áreas do entorno. Braz J Aquat Sci Technol 12:73–89
Silva VEC, Fonseca A, Franco D et al (2017) Space time evolution of the trophic state of a subtropical lagoon, Lagoa da Conceição, Florianópolis Island of Santa Catarina. Brazil Rev Bras Rec Hid. https://doi.org/10.1590/2318-0331.011716027
Soriano-Sierra EJ (1999) Ecossistema de marismas da lagoa da Conceição. II. A fitocenosis. In: Sierra de Ledo B, Soriano-Sierra EJ (eds) O ecossitema da lagoa da Conceição. SDM-FEPEMA, Florianópolis, pp 171–183
Spalding MD, Fox HE, Allen GR et al (2007) Marine ecoregions of the world: a bioregionalization of coastal and shelf areas. Bioscience 57:573e583
Spínola JL, Teixeira CF, Andriguetto Filho JM (2014) Desafios à cogestão: os impactos da Via Expressa Sul sobre o extrativismo na RESEX Marinha do Pirajubaé. Desenv Meio Amb 32:139–150
Spurway P, Roper T, Stephens K (2000) Community consultation, conservation, crustaceans, calculations and consents – Entrance Management Policies for ICOLLs. In: Proceedings 10th Annual NSW Coastal Conference, Yamba, 2000
Sunye PS, Pereira TJ, Russo A, Netto SA (2014) A pesca do camarão-rosa no Sistema Estuarino de Laguna, SC: história e acasos das políticas de manejo. In: Haimovici M, Andriguetto JM, Sunye PS (eds) A pesca marinha e estuarino no Brasil: estudos de caso multidisciplinares. Editora da Furg, Rio Grande, pp 75–85
Teixeira H, Borja A, Weisberg SB et al (2010) Assessing coastal benthic macrofauna community condition using best professional judgement - developing consensus across North America and Europe. Mar Pollut Bull 60:589–600
Teske P, Wooldridge T (2003) What limits the distribution of subtidal macrobenthos in permanently open and temporally open/closed south African estuaries? Salinity vs. sediment particle size. Estuar Coast Shelf Sci 57:225–238
Tweedley JR, Hallett CS, Warwick RM et al (2016) The hypoxia that developed in a microtidal estuary following an extreme storm produced dramatic changes in the benthos. Mar Fresh Res 67(3):327–341
Valgas, IS (2009) As marismas de Spartina alterniflora e os taboais de Typha domingensis do Sistema Estuarino de Laguna (Santa Catarina, Brasil): distribuição espacial, estrutura e macrofauna associada. Master thesis, Universidade Federal do Paraná, Pontal do Paraná
Vieira JP, Vasconcellos MCA, Silva RES, Fischer LGF (1996) Rejeição da pesca do camarão-rosa (Penaeus paulensis) no estuário da Lagoa dos Patos, RS, Brasil. Atlantica 18:123–142
Vieira JP, Garcia AM, Grimm AM (2008) Preliminary evidences of El Niño effects on the mullet fishery of Patos lagoon estuary (Brazil). Braz Arch Biol Technol 51:433–440
Vollenweider RA, Giovanardi F, Montanari G, Rinaldi A (1998) Characterization of the trophic conditions of marine coastal waters with special reference to the NW Adriatic Sea: proposal for a trophic scale, turbidity and generalized water quality index. Environmetrics 9:329–357
Weis WA, Soares CHL, Quadros DPC et al (2017) Urbanization effects on different biological organization levels of an estuarine polychaete tolerant to pollution. Ecol Indic 73:698–707
Whitfield A, Bate G (2007) A review of information on temporarily open/closed estuaries in the warm and cool temperate biogeographic regions of South Africa, with particular emphasis on the influence of river flow on these systems. Water Research Commission Report, Pretoria
Woodroffe CD (2002) Coasts: form, process and evolution. Cambridge University Press, Cambridge
Ysebaert T, Fettweis M, Meire P et al (2005) Benthic variability in intertidal soft-sediments in the mesohaline part of the Schelde estuary. Hydrobiologia 540:197–216
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Netto, S.A., Pagliosa, P.R., Colling, A., Fonseca, A.L., Brauko, K.M. (2018). Benthic Estuarine Assemblages from the Southern Brazilian Marine Ecoregion. In: Lana, P., Bernardino, A. (eds) Brazilian Estuaries. Brazilian Marine Biodiversity . Springer, Cham. https://doi.org/10.1007/978-3-319-77779-5_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-77779-5_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-77778-8
Online ISBN: 978-3-319-77779-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)