Abstract
The controls and trajectories of biogeochemical dynamics in chaparral dominated ecosystems are highly diverse. This breadth of diversity in chaparral biogeochemical dynamics is caused by a combination of diverse biogeophysical drivers, self-organization, and sensitivity to historical legacies. The high topographic heterogeneity and decoupling of moisture inputs and energy inputs in the winter precipitation dominated climates imposes unique ecohydrologic adaptations that influence plant and microbial metabolic activities and distributions throughout chaparral landscapes. Chaparral biogeochemical dynamics, especially interactions among carbon, nitrogen, and hydrologic cycling, may have important implications for the ecosystem services and disservices provided by these ecosystems, including greenhouse gas emissions and pollutants to air and water, and influence the vulnerability of chaparral biological communities to global change drivers. These ecosystems may have a large capacity for carbon storage and sustained carbon sequestration at rates comparable with those of old-growth forests. In other places, chaparral ecosystems may have low pools of carbon and low rates of sequestration. While commonly considered nitrogen limited, chaparral dominated ecosystems may also have large nitrogen emissions through atmospheric and hydrologic pathways. Future trajectories of chaparral biogeochemical dynamics are also variable. Unprecedented interactions among global change drivers, including climate, nitrogen deposition, fire frequency, and invasion risks, are pushing many of these systems to tipping points of reorganization and type-conversion to grass dominated states with uncertain biogeochemical consequences. Management for maintaining ecosystem services associated with biogeochemical dynamics can be improved with growing opportunities provided by a suite of environmental sensors and next generation modeling approaches.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
6.1 Introduction to Biogeochemical Cycling in Chaparral
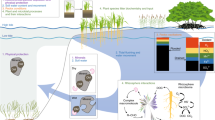
Biogeochemical dynamics in chaparral dominated ecosystems are unusually complex and provide a valuable testbed for evaluating general ecosystem theories. What is known suggests important differences between chaparral and most other terrestrial ecosystems . Notably, the rates of C and N accumulation , transformations, and losses in chaparral dominated ecosystems vary dramatically in time and space. As such, chaparral ecosystems have a potential for long-term carbon (C) accumulation (Luo et al. 2007) and high rates of nitrogen (N) losses (Homyak et al. 2014). However, the actual rates of C and N accumulation, transformations, and losses in chaparral dominated ecosystems vary considerably in time and space. The sources of chaparral biogeochemical variability, in part, depend on underlying biogeophysical drivers, historical legacies, and self-organizing processes connecting hourly to millennial time-scales and local to landscape spatial scales (Fig. 6.1). These interacting drivers dictate, to a large degree, the availability of moisture, which has an overriding influence on chaparral biogeochemical functioning .
The chaparral landscapes of California show extensive variation in their biogeophysical template; the combination of geology, climate, and available species that sets the stage for biogeochemical cycling. Important factors include the distribution of bedrock origins, climate gradients spanning coastal to inland and latitudinal gradients, and rugged topography . Distributions of biological traits of plants and microbes are another source of biogeochemical diversity that can moderate or accentuate variation from the geophysical template. Historical legacies, past conditions that influence current functioning , further influence biogeochemical variation and include dramatic events such as wildfire or variation in precipitation across event to decadal time-scales . Historical legacies can influence biological responses to the geophysical template and biological capacity. A distinguishing feature of biogeochemical cycling in chaparral, in part because of the diversity of environmental conditions and ecological responses, is a high diversity in the size of specific material pools (e.g., above-ground biomass , roots, litter, soil) and similarly high diversity of the controls to fluxes among these pools. Furthermore, other components of chaparral ecosystems can cause slowing or accelerating of the biogeochemical cycle. For example, ecosystem characteristics such as water availability during cooler periods and species with low rates of photosynthesis tend toward the slowing of biogeochemical cycles, while the rapid response of chaparral ecosystems to wetting and rapid soil movement tend toward accelerated biogeochemical cycling.
Future chaparral biogeochemical cycling will respond to inter-active global change drivers through several potentially interacting pathways. These drivers influence the biogeophysical template through changes in climate and nutrient inputs, biotic diversity, expansion of non-native species , and land use that removes and fragments chaparral communities. Furthermore, the combination of global change drivers may increase opportunities for community type change through conversion of native shrub to non-native grasses with unknown implications for the biogeochemical cycle. Understanding the spatial distribution and controls of biogeochemical dynamics in chaparral provides an essential basis for assessing future trajectories of these ecosystems and maximizing their services while providing valuable directions for improving the next generation of ecosystem and land surface models that include chaparral dynamics.
6.2 Controls of Biogeochemical Variation in Chaparral
6.2.1 Boundary Delineation of Chaparral Biogeochemistry
Ecosystem science operates from two often contrasting viewpoints: the Newtonian view where understanding broadly applicable general mechanisms is desired and the Darwinian view where understanding is derived from specific details of particular systems that are used to mechanistically build up from first principles (Harte 2002; Jenerette et al. 2012). In reconciling these alternate views, an ecosystems framework that relies on mass balanced fluxes of elements between different pools has been a fundamental perspective for examining biogeochemical dynamics that allows both for broad generality and detailed specificity (Odum 1983). A systems approach is used as a basis for biogeochemical and climate modeling and is leading to new developments in ecosystem theory (Wu and David 2002). Using a systems approach allows quantification of mass and energy balance and exchanges among distinct pools within the ecosystem and between the ecosystem and the environment (Odum 1983; Chapin et al. 2012). This framework allows an evaluation of how controls and general mechanisms vary among systems, while also facilitating detailed evaluation of individual systems.
An essential activity for any analysis within the ecosystem framework is boundary delineation. A fundamental challenge is that ecosystems are inherently open systems. Boundaries are necessarily imposed and can differ depending on the purpose of analysis. For chaparral biogeochemical cycling, boundaries can vary spatially, in both the vertical direction extending through the soil profile and vegetation and horizontally across the land surface, encompassing a diversity of landscape components. Boundaries also change temporally, spanning instantaneous snapshot to millennia. Vertically, boundaries typically include the upper few centimeters of soil profile to the top of the plant canopy that is readily accessible for measurements. However, extending the vertical profile deeper to the plant roots is desirable both to better characterize the active ecohydrological layer and its influence on plant dynamics, and the potential for storage of biogeochemical constituents. However, characterizing biogeochemical dynamics deeper in the soil poses logistical challenges and can be disruptive to the ecosystem. At the most complete scope, the critical zone—spanning the bottom of groundwater to the top of the trees, provides an increasingly used framework for bounding ecosystem processes (Amundson et al. 2007; Chorover et al. 2011). The full critical zone provides a comprehensive perspective for examining long-term biogeochemical dynamics resulting from coupled geomorphic and ecological dynamics. In the horizontal direction, several alternative boundary delineations are frequently used including watersheds, management units, and plant community extents (Fortin et al. 2000; Bailey 2004). Practically, much biogeochemical characterization occurs within individual sampling plots ranging from m2 to ha. Temporally, most analyses are bounded by only a few years of data, however, a limited number of studies of chaparral biogeochemistry span a few decades. The fine scale nature of ecological research compared to the expected time-scales of ecohydrological changes poses important limitations that need addressing. While boundaries are inherently arbitrary, imposing boundaries to chaparral biogeochemistry is an essential task for quantifying rates of biogeochemical cycling with important consequences for estimates of pool sizes and exchange rates.
6.2.2 Geophysical Template for Chaparral Biogeochemistry
While the fundamental components of biogeochemical cycling in chaparral are consistent with general ecosystem theory, the unique characteristics of chaparral ecosystems limit our understanding of how biogeochemical cycle components, their magnitudes, and their drivers vary. Chaparral dominated ecosystems occur in Mediterranean-type climate (MTC) regions that receive primarily winter precipitation . This imbalance between energy inputs in the summer months and moisture in the winter months imposes selective pressure for unique ecohydrologic adaptations that influence plant and microbial metabolic activities and distributions throughout the landscape (Baldocchi and Xu 2007; Pumo et al. 2008). Interacting with this general climate influence, the distribution of chaparral across latitudinal, coastal to inland, and elevation gradients has implications for the distributions of precipitation, temperature , and potential evaporation . Geologic variation also leads to differences in soil conditions, erosion, and deposition that can influence soil moisture distributions and nutrient availability. Through erosion and deposition processes chaparral ecosystems are characterized by high topographic heterogeneity, which further impacts water availability . The rugged topography leads to complex patterns in water availability, through large variation in insolation and upslope accumulation, influencing water loss and inputs respectively. Large rates of soil movement associated with the characteristically steep slopes in chaparral dominated regions also lead to large variation in the capacity of soil to hold water.
Precipitation in MTC regions imposes a key constraint on biological processes—these systems are by definition water-limited and changes in water availability have direct and indirect consequences to biogeochemical cycling (e.g., Biederman et al. 2016). The effects of precipitation can occur within minutes, however, it can leave legacies persisting across seasons, years, and potentially even decades (Jenerette et al. 2008; Scott et al. 2009; Shen et al. 2016). Total annual precipitation is commonly the principal variable used to quantify ecosystem responses to moisture. However, the partitioning of precipitation between hydrologically distinct flow paths such as transpiration , soil evaporation , or runoff can also have a large influence on how the ecosystem and rates of biogeochemical cycling respond to precipitation distributions. Independent of the total annual precipitation, the variation in timing, intensity, and distribution of precipitation can have large effects on moisture partitioning and lead to contrasting influences on biogeochemical cycling (Huxman et al. 2004; Shen et al. 2008).
With seasonal precipitation in chaparral ecosystems dominated by winter rainfall, plant growth, and many associated ecosystem dynamics are generally more closely connected to winter rainfall patterns. In contrast, moisture from summer rainfall in June–September (a total average of <10 mm in southern California and <15 mm in northern California chaparral) is less likely to contribute to plant growth and generally is rapidly lost through soil evaporation . Within the winter season, variation in the distribution of rainfall can have large influences on plant driven biogeochemical cycling. For example, high intensity rainfall events can lead to increased runoff as soil infiltration capacity is exceeded (see Chap. 7). Alternatively, clusters of rainfall events can lead to increased runoff losses as soil water holding capacity is filled through successive wetting. Both situations reduce precipitation -related moisture available in the ecosystem. In contrast, small low-intensity rainfall events may effectively only wet the leaf or soil surface and be primarily lost through evaporation. Large gaps between rainfall events may be associated with increased proportions of moisture lost through soil evaporation . The effects of contrasting precipitation distributions can influence plant uptake of C and N and subsequent litter contributions of C and N to the soil. Pulse responses to cycles of drying and re-wetting can have a large influence on biogeochemical cycling in chaparral dominated ecosystems (Miller et al. 2005; Jenerette and Chatterjee 2012; Jenerette et al. 2012). All of these components of precipitation can influence the effective moisture in the soil and the capacity of biogeochemical processes.
Across the range of chaparral vegetation , multiple geographic gradients further influence local energy balance and moisture availability. The large latitudinal gradient of chaparral, spanning most of California (Fig. 1.1), is further associated with a large variation in ecohydrologic dynamics, with more moisture and cooler temperatures in northern chaparral communities compared to southern communities. Landscape position along coastal to inland gradients are similarly associated with climate differences, with coastal chaparral communities generally wetter and cooler than more inland communities. Variation in elevation can also influence local climate, with drier and hotter conditions at lower elevation and cooler and wetter conditions at higher elevations. These geographic gradients lead to a complex mosaic of climate distributions influencing chaparral biogeochemical cycling.
Somewhat independent of the climate gradients is variation in underlying geology including bedrock and mountain formation. Bedrock mineralogy can influence many biogeochemical processes, for example, soils from parent materials dominated with non-crystalline materials, e.g., andesite, may have higher rates of soil C mineralization than soils from parent materials dominated by crystalline materials, e.g., granite (Rasmussen et al. 2006). An extreme of geological influence can been seen in chaparral found on serpentine soils. These ecosystems show characteristic responses to low nutrient and high metal content through communities with slow growth, dwarfed stature, and reduced cover (Kruckeberg 1984; Gough et al. 1989) (see Chap. 1).
6.2.3 Biotic Diversity as a Driver of Chaparral Biogeochemistry
While individual communities are generally dominated by only a few locally prevalent species, total plant species diversity in chaparral vegetation throughout California is high. Recent estimates suggest chaparral harbors 1177 vascular plant species, a substantial proportion of the plant diversity in California (Keeley 2005; Halsey and Keeley 2016). The biogeochemical diversity includes both the potential for high variation within individual sites and large differences among sites throughout the entire range of chaparral ecosystems (see Chap. 2). Species variation across broad geophysical gradients further influence biogeochemical diversity within chaparral.
In addition to taxonomic biodiversity, functional biodiversity, the breadth of traits represented in the community, also has a critical role on the influence of chaparral species and consequently biogeochemical cycling. The species comprising chaparral communities have a large breadth of functional diversity (Ackerly 2004; Pivovaroff et al. 2016). Typically, functional diversity has been considered along suites of potentially distinct axes such as leaf photosynthetic rate , size, parental investment, and hydraulic capacity (Westoby 1998; Li et al. 2015; Diaz et al. 2016). Leaf axes of functional variation are primarily arrayed along a leaf economic spectrum of high maximum rates of photosynthesis and short leaf lifespan versus lower maximum rates of photosynthesis and longer lifespans (Wright et al. 2004). Leaf traits further influence soil biogeochemical cycling as the shorter-lived leaves are also associated with more rapid decomposition (Cornwell et al. 2008). Species typically dominant in chaparral communities are generally associated with characteristics of the slow-end of the leaf economic spectrum with low N concentration , low maximum rates of photosynthesis, and slow to decay litter. Another key plant characteristic that varies among chaparral species is rooting depth, which influences the zone of biologically available water with concomitant changes to hydrology and plant functioning (Scott et al. 2014).
In MTC regions , diverse trait combinations have evolved to address fundamental constraints of water limitation but also take advantage of the large ecohydrological diversity within chaparral dominated ecosystems. Drought adaptation strategies among chaparral species are diverse and include strategies that look to maximize one or combinations of vulnerabilities to cavitation, sapwood capacitance, deciduousness, photosynthetic stems, photosynthetic responses to leaf water potential, and hydraulic architecture (Ackerly 2004; Pivovaroff et al. 2016). Within chaparral ecosystems, many distinct plant community associations can be identified in addition to the dominant shrubs including grass, conifer forest , and riparian communities. These individual communities also vary in height, leaf, and rooting depth of the dominant organism. In addition, subdominant plant species also contribute to functional diversity of plant traits . For example, shrub communities have a breadth of plant heights spanning low growing herbs and taller trees and are characterized by long-lived leaves and low rates of photosynthesis. In contrast, many other inter-stitial members of the plant community, including annual grasses, have much shorter-lived leaves and higher rates of photosynthesis. Chaparral species also express extensive variation in rooting depth (Ichii et al. 2009), ability to form symbioses allowing N fixation (Delwiche et al. 1965), and resprouting capacity after a fire (Bell 2001). Together the combinations of different traits contribute to diverse biogeochemical dynamics in chaparral ecosystems.
6.2.4 Ecohydrological Self-Organization and Chaparral Biogeochemical Cycling
In the context of the biogeophysical drivers, a system of ecohydrological processes and non-linear feedbacks can mute or amplify environmental sensitivities through processes whose outcomes cannot be understood through independent evaluation of the parts. This “self-organizing” (for a general introduction see Levin 2000) process, for example specific trajectory of hydrologic flow-paths, species distributions , and fire dynamics, lead to complex biogeochemical dynamics spanning scales of individual site to entire chaparral landscapes. Variation in chaparral biogeochemical cycling and potential future trajectories are defined by both dynamics of individual component processes and system level feedbacks and interactions. Systems are understood through a web of inter-connected relationships between many distinct components, where the connections between components can be as important as dynamics of individual components. Feedbacks , resulting from the web of inter-connected relationships between many distinct system components (Ashby 1956; Forrester 1961), are important aspects of system dynamics that can both magnify changes through positive feedbacks or moderate change through negative feedbacks. Feedbacks can lead to emergent properties through self-organization of a system in concert with its environment (Wu and Loucks 1995; Levin 1998; Holling and Gunderson 2002). Through self-organization biogeochemical dynamics are not predictable from individual component processes in isolation. Recognition of the importance of emergent properties has increased with growing evidence of alternative stable states and tipping points between regime changes occurring within ecosystems (Scheffer et al. 2001; Scheffer and Carpenter 2003), where a system can rapidly change between contrasting configurations. These “state changes” occur with only limited changes in the environment at a critical threshold, while otherwise the system is largely insensitive to environmental variation away from such thresholds. Thus, for the same underlying biogeophysical drivers contrasting biogeochemical patterns can develop in response to ecohydrological dynamics and historical legacies. Evaluating biogeochemical variation in the context of self-organization systems theory provides both a framework for identifying the relationships between discrete components and examining dynamics spanning from of steady-state stabilizing mechanisms to self-organized and adaptive responses to the environment that can lead to large and potentially rapid changes in biogeochemical dynamics .
Self-organizing ecohydrological processes that influence chaparral biogeochemical variation are primarily associated with changing geomorphological processes, ecological processes, and in the context of high rates of endemism within chaparral evolutionary processes (Jenerette et al. 2012). Geomorphological processes occur in the context of changes in underlying geophysical and climate drivers and the characteristics of precipitation and meteorological dynamics. Geomorphological processes contribute to the rugged topography of chaparral ecosystems and influence water storage and movement as well as soil formation and transport. The influence of aspect in response to geomorphology has a strong influence on biogeochemical cycling, with wetter north-facing slopes typically harboring more biomass than corresponding drier south-facing slopes (Parker et al. 2016). Hydrologic routing can also have strong influences on soil biogeochemical dynamics, where locations with large amounts of upslope contributing area, such as riparian areas, are often wetter than locations with low upslope contribution areas, such as ridges (Riveros-Iregui et al. 2012). Geomorphological dynamics are further coupled with plant and microbial communities that influence chaparral biogeochemical dynamics through ecological and evolutionary changes. Ecological dynamics lead to assembly of communities and distribution of functional traits. The resulting ecological interactions between biotic communities and abiotic environment, structure nutrient cycles , disturbance , and physical organization can have large influences on biogeochemical dynamics. Interactions among traits and ecosystem feedbacks can further influence biogeochemical cycling, imposing constraints to processes varying in space and time and directly influencing rates of nutrient transformation.
Disturbance dynamics, especially in context of fire, can rapidly change community composition and capacity. Fire distributions are closely related to self-organizing processes associated with fuel load accumulation and decay. Fire frequency distributions often follow power-law distributions distinct from the distributions of the drivers of fire occurrence, a key characteristic of self-organization processes (Malamud et al. 1998). Fire has long been associated with chaparral as a major disturbance that resets the ecosystem and maintains shrub dominated communities. By burning biomass, litter, and topsoil, fires emit a large amount of particulate material that can have relatively high carbon content of variable chemical composition. Intense burning of biomass coupled with high burn temperatures results in greater transportation and transformation of nutrients. While all fires alter nutrient cycling in chaparral, the extent to which nutrient transformations occur depends on the fire severity , extent, and return interval. Notably, if seedbanks are not destroyed, high severity fires can induce germination of many chaparral seedlings and promote quick recovery times of native herb and shrub species, allowing nutrient storage to begin again (Thanos and Rundel 1995).
6.2.5 Historical Legacies and Their Influence on Chaparral Biogeochemistry
Arising from dynamics of biogeophysical drivers and ecohydrological self-organization are legacies of past processes that can further influence chaparral biogeochemistry. Historical ecosystem legacies are processes that occurred in the past and have a discernable influence on current ecosystem functioning , including biogeochemistry. Disturbances , such as fire, are prominent events whose legacy on ecosystem functioning has immediate consequences (Moreno et al. 2013) and can propagate for decades. Much of the biogeochemical sensitivity to precipitation variation such as timing and distribution, reflects an influence of precipitation legacies from individual events to potentially decadal temporal scales on ecosystem functioning (Scott et al. 2009; Shen et al. 2016). For example, the length of time between precipitation events is a direct quantification of legacy effects. Such effects can propagate across seasons and between years, with increasing moisture in wet years potentially priming production or reducing nutrient availability (Jenerette et al. 2010; Shen et al. 2016). At inter-decadal scales, variation in precipitation may influence ecosystem C storage and elevate rates of C loss in subsequent dry periods (Scott et al. 2009; Shen et al. 2016). However, our understanding of how historical legacy effects influence chaparral biogeochemical cycling is currently limited and more research is needed.
6.2.6 Synthesis of Variation in Chaparral Biogeochemical Cycling
The magnitudes of material storage, transformation, and loss associated with biogeochemical cycling in chaparral vary dramatically in response to both differences in biogeophysical drivers, ecohydrological self-organization, and the influence of historical legacies (Fig. 6.1). Biogeochemical variation in chaparral ecosystems is high at both local and regional scales (Chatterjee and Jenerette 2011; Dahlin et al. 2013). The suite of biogeophysical drivers associated with below-ground geology, above-ground climate, and a diverse suite of biological species all provide constraints and capacities for biogeochemical dynamics . These drivers intersect with self-organizing ecohydrological processes that influence geomorphology, ecology, and evolutionary dynamics that have a strong direct influence on chaparral biogeochemical cycling. Historical contingencies arising from both dynamics of biogeophysical drivers and ecohydrological self-organization further impose constraints to biogeochemical cycling. A recurring thread through these processes is the resulting distribution and sensitivities to soil moisture conditions. In general, greater moisture availability, either through greater inputs from winter rains or reduced evaporative demands, will be associated with greater biogeochemical pools. Microenvironments created by geomorphology and organismal effects can contribute to variations in total pools of C, N, and other elements and fluxes. Interacting drivers on biogeochemical variation can be complex with non-linear effects that have large uncertainties. Recently, and increasingly in the future, anthropogenic influences are altering the dynamics of biogeophysical drivers, ecohydrological self-organization, and leaving legacies that may remain for millennia.
6.3 Key Biogeochemical Cycles in Chaparral: Carbon and Nitrogen
6.3.1 Overview of Biogeochemical Dynamics
Biogeochemical dynamics of chaparral ecosystems are characterized by continuous and discontinuous responses to environmental conditions, known as pulse dynamics, which are both associated with soil moisture variation. A biogeochemical pulse occurs when wetting of previously dry soil acts as a trigger for biogeochemical processes and then subsequent dynamics are decoupled from soil moisture availability but depend on subsequently limiting resources and biological capacity. Rates of biogeochemical cycling decline with lower water availability and through direct desiccation and reduced rates of resource supply to microbes. In contrast, higher water availabilities can also reduce biogeochemical cycling through reduced oxygen availability. Biogeochemical cycling may respond to isolated precipitation events that act as a trigger, but then subsequent dynamics may be then regulated by substrate availability and lead to a decoupling of biogeochemical dynamics from actual moisture availability. Pulse dynamics are frequently observed in dryland ecosystems and are particularly relevant to chaparral dominated landscapes (Baldocchi et al. 2006; Jarvis et al. 2007; Jenerette and Chatterjee 2012). Because of pulse dynamics and the importance of wetting dry soils, small precipitation events can have disproportionately large influences on soil biogeochemical cycling and thus the timing and distribution of wetting is also important in addition to the total amount of precipitation (Huxman et al. 2004; Jenerette et al. 2008).
Another overarching influence on chaparral biogeochemical dynamics is the distinct characteristic time-scales between microbial and plant dynamics, with microbial dynamics changing much faster than plant dynamics. These contrasts are most evident in pulse dynamics, where microbial processes may respond within hours and much more dramatically than plants, which typically respond at the scale of days. As a consequence, rain in summer seasons or the initiation of winter season precipitation may have a much larger influence on chaparral soil-driven biogeochemical cycling than wetting patterns favoring plant growth (Miller et al. 2005; Jenerette and Chatterjee 2012). The contrasting influence of microbes and plants on nutrients can lead to complex dynamics that appear to favor nutrient losses and recycling simultaneously, in contrast to general ecosystem expectations.
In the context of the drivers and processes influencing chaparral biogeochemical dynamics, C and N cycles are closely connected to many ecosystem processes and are directly related to multiple ecosystems services, including climate regulation and provisioning of clean water. These two cycles include components of biomass accumulation , photosynthesis, and what is commonly considered its main limiting nutrient following water, N. Likewise, C availability can influence some N cycling pathways and thus leads to two-way coupling between biogeochemical cycles and opportunities for feedbacks.
6.3.2 Carbon Cycling in Chaparral Ecosystems
Within an ecosystem many transformations of C occur between its initial fixation and eventual return to the atmosphere. In chaparral dominated ecosystems, C cycling features uptake through photosynthesis, transformations within the ecosystem, and losses through respiration with opportunities for deposition or erosion to bring new C into, or remove it from, the ecosystem (Fig. 6.2, see Box 6.1). Dominant pools of C include soil and plant components, with additional C contained in litter. The identification of long-term C sequestration, or positive Net Ecosystem Production (NEP), has become increasingly of interest, as a result effort has been directed to identifying mechanisms that promote the stabilization of C and prevent the return of C to the atmosphere as a greenhouse gas (GHG).
Synthesis of existing literature reporting pools and fluxes of C in chaparral dominated ecosystems. The values are not exhaustive but represent an initial review of the range of variation in key C cycle pools and fluxes. All pools (above-ground biomass, root biomass, soil, litter are in units g C m−2. All fluxes (litter mineralization, litter fall, NPP , root respiration root growth litter incorporation into soil) are in units g C m−1 year−1. Units are re-scaled for comparison amongst different studies when units differed. Data were derived from published results (Harrison et al. 1971; Rundel and Parsons 1979; Mooney and Rundel 1979; Gray and Schlesinger 1981; Dunn et al. 1988; Ulery et al. 1995; Quideau et al. 1998; Miller et al. 2005; Li et al. 2006; Smith et al. 2007; Grünzweig et al. 2007; Pasquini and Vourlitis 2010; Drenovsky et al. 2013; Dickens and Allen 2014; Homyak et al. 2014; Hanan et al. 2016a; Vourlitis and Hentz 2016)
C uptake at the leaf is the point of entry of C into chaparral ecosystems . The total amount of photosynthesis, or gross primary production, is regulated by physical and biochemical characteristics of individual leaves and their extent and distribution within an ecosystem. Plants also respire C and the balance between photosynthesis and respiration is Net Primary Production (NPP) or the amount of plant growth. In response to regular water limitation , root allocation is a large component of plant C, which leads to deeper C inputs into the soil. Recent interest has been directed to C in plants that is readily mobilized and not used in structures , such as plant Non-Structural Carbohydrates (NSC) (Dietze et al. 2014). Increasing evidence from many woody plants suggests increasing NSC content is linked to increased growth, seed production, disease resistance, and drought resistance. Within some chaparral shrubs plant age may have a role in the distribution and allocation rates of the NSC pool (Sparks et al. 1993), however the dynamics and distribution of chaparral NSC is an emerging research need.
The transfer of plant C to soil heterotrophs and potential long-term sequestration occur through several pathways associated with both structural and non-structural C . Leaf and stem litter drop to the soil surface. Root death and root exudates provide plant C within the soil. The litter itself can be directly consumed or incorporated into the soil. In MTC ecosystems, as in most dryland ecosystems, processes of litter decomposition may be greatly accelerated by UV induced photodegradation (Austin and Vivanco 2006; Gliksman et al. 2017). However, with a frequently closed canopy, the availability of UV at the litter layer may be limited. Living plant C and litter C can have turnover times of seasons to decades. In this sense, increasing C in living biomass or rapidly cycling litter may represent a short-term C sink—this C is primarily respired and does not contribute to long-term C sequestration. Stabilization, the conversion of organic C to a form highly resistant to decomposition, may have low rates but contribute to long-term C sequestration (Six et al. 2002). C stabilization can occur through physical and chemical processes, such as the formation of soil aggregates or humification. Through aggregate formation, soil C is physically protected within the soil matrix such that it is inaccessible to microbial degradation (Schmidt et al. 2011). Humification, conversion of soils along a continuum from chemically simple to large and complex organic molecules, is an alternate pathway of ecosystem C stabilization (Lehmann and Kleber 2015). Sequestration rates may be influenced by vegetation, as chaparral communities dominated by Ceanothus spp. sequestered more C than Adanostoma spp. dominated chaparral (Ulery et al. 1995; Quideau et al. 1998).
Fires have immediate and legacy effects on chaparral C cycling . Fire itself results in an immediate loss of C from the ecosystem, primarily as a fraction of above-ground plant biomass , litter, and organic C in the upper horizons of the soil. C losses from fire include particulate material and a variety of C trace gasses including CO2, CO , and CH4. Recently fire mediated charcoal production has been noted as a potential sequestration pathway, which converts comparatively labile plant C forms with residence times of decades to centuries to a form with residence times of millennia or longer (Heckman et al. 2013). Incompletely burned plant C can be returned to the litter layer and soil as black carbon, which can be highly stabilized and serve as an important pathway of long-term sequestered C. May et al. (2014) propose that chaparral fires emit the highest amounts of refractory black carbon compared to other ecosystems, further supporting the potential for this pathway for chaparral ecosystem to sequester C . Further, periodic fires may also increase the rate of soil C inputs within chaparral communities by producing large quantities of wood ash and charred woody material from which carbon in the form of calcite readily leaches into the soil (Ulery et al. 1995). Fire can lead to short-term reductions in soil C, although these can return to prefire levels within a year (Graham et al. 2016). While fire does lead to immediate losses of C through combustion, the long-term consequences of fire may primarily serve as mechanism for C sequestration. Thus, while typically fire is associated with large emissions of C from an ecosystem, perhaps surprisingly fire may also be associated with long-term sequestration of C into highly recalcitrant C or black C. The mechanisms of organic C transformation into long-term stable forms are highly uncertain and are a current area of active research (Stewart et al. 2008; Schmidt et al. 2011).
Box 6.1 Case Studies of Chaparral Carbon Dynamics
At present, the majority of information about chaparral has been obtained from widely distributed individual studies and concerted evaluation of specific sites and landscapes. The Deep Canyon transect in southern California, which spans a 2500 m (8202 ft) elevation gradient has been used for more than 4 decades of research in soil biogeochemistry across a transect of chaparral vegetation bounded at the lower end by a creosote dominated desert ecosystem and at the higher end by coniferous dominated subalpine forests (Hanawalt and Whittaker 1976; Jenerette and Chatterjee 2012). Exploring the local and regional variation in chaparral soil C emissions was the focus of a series of studies conducted along this transect. With increasing elevation, the climate is characterized by decreasing temperatures and increasing precipitation . Soil organic matter and nutrient content in the chaparral components of the transect generally increase with elevation (Hanawalt and Whittaker 1976; Chatterjee and Jenerette 2015). The spatial structure of soil organic matter and soil CO2 emissions also changes throughout the transect. The microenvironment effects of local shrub canopies on distributions of organic matter, C, and CO2 emission potentials decrease in magnitude, but increase with higher elevations (Chatterjee and Jenerette 2011). Throughout the transect, maximum rates of soil CO2 emissions from laboratory incubations were consistently above 35 °C and in chaparral ecosystem were above 45 °C. While soil C emissions generally increased with elevation, the pulse sensitivity of CO2 emissions to wetting generally decreased with elevation, which led to a large scale negative relationship between soil C and pulse emissions. However, within sites at the same elevation, soil C emission pulses were positively related with soil C and pulse emissions. Thus, across an elevation gradient in chaparral dominated ecosystems a contrasting scale-effect of C and pulse dynamics were observed (Jenerette and Chatterjee 2012). This case study, demonstrates the large variation in chaparral biogeochemistry and the varying potential of pulse driven biogeochemical fluxes.
A second case study of chaparral C cycling examines whole ecosystem fluxes and net ecosystem C exchange within a single site in southern California. The Sky Oaks field station, featuring both old-growth chaparral and some recently burned areas, has served as a model system of chaparral with several connected long-term studies into C cycling. An evaluation of this well-studied site presents a valuable lesson about the complexity of chaparral C cycling. Data from the Sky Oaks site provides the most comprehensive evaluation of chaparral ecosystem C cycling variation and has led to improved understanding of the distinct controls on chaparral biogeochemical dynamics and the large variability of C in chaparral ecosystems. In an old-growth area at Sky Oaks, whole ecosystem CO2 exchanges known as Net Ecosystem Exchange (NEE), have been measured through the eddy covariance approach and showed high C uptake rates, up to 155 g C m−2 year−1 (Luo et al. 2007). These rates are comparable with uptake rates of other old-growth forests distributed globally. However, in dry years NEE led to no uptake or even net emissions back to the atmosphere at the annual scale. Within years, whole ecosystem C exchange showed large daily variation, spanning effectively 0 g C m–2 to above 4.5 g C m−2 year−1 in two distinct chaparral communities (Stylinski et al. 2002; Sims et al. 2006). Spatially mapping CO2 fluxes suggests equally large spatial variation within local landscapes of chaparral dominated ecosystems (Fuentes et al. 2006). The complex temporal and spatial dynamics of chaparral C at this single site highlights both the large potential for C uptake, especially from old-growth stands, to net emissions depending on moisture availability. While likely other chaparral communities show similar magnitudes of variation in C exchanges, the Sky Oaks site provides a novel window into C dynamics through the long-term records that are not available elsewhere.
6.3.3 Nitrogen Cycling in Chaparral Ecosystems
While N is the most abundant element in the atmosphere it occurs in the highly stable form of dinitrogen (N2, Schlesinger and Bernhardt 2013). Reactive N that can be used by organisms is comparatively scarce. As with many dryland ecosystems, chaparral productivity and many biogeochemical processes have been historically considered secondarily N limited after water limitation . N limited systems are characterized by several factors promoting nutrient conservation, including long leaf life-times, nutrient-poor sclerophyllic tissues, and nutrient resorption during senescence. The resulting low-quality litter increases N immobilization during decomposition and lower rates of mineralization. Slow soil cycling of N and increased losses contribute to poor soil fertility and tighter N recycling within plants. For example, many evergreen chaparral shrubs uptake N during the fall or winter and store in old tissues for new growth in the spring (Mooney and Rundel 1979). Internal cycling and outputs reflect processes associated with N limitation , although they also suggest processes, including potential for high rates of N losses that contrast with predictions of N limitations (Fig. 6.3). Resolving this apparent dichotomy has been an important source of recent progress in chaparral biogeochemistry.
Synthesis of existing literature reporting pools and fluxes of N in chaparral dominated ecosystems. The values are not exhaustive but represent an initial review of the range of variation in key C cycle pools and fluxes. All pools (above-ground N, root N, soil, litter N, are in units g C m−2. All fluxes (litter ammonification, nitrification, immobilization, denitrification, leaching are in units g C m−1 year−1. Units are re-scaled for comparison amongst different studies when units differed. Data were derived from published results (Kummerow et al. 1978; Mooney and Rundel 1979; Gray and Schlesinger 1981; DeBano et al. 1984; Riggan et al. 1985; Bytnerowicz et al. 1987; Ulery et al. 1995; Fenn et al. 1996; Quideau et al. 1998; Fenn et al. 2003; Miller et al. 2005; Li et al. 2006; Meixner et al. 2006; Grünzweig et al. 2007; Pasquini and Vourlitis 2010; Vourlitis and Fernandez 2012; Drenovsky et al. 2013; Dickens and Allen 2014; Homyak et al. 2014; Hanan et al. 2016a; Vourlitis and Hentz 2016)
Inputs of reactive N, including mineralized and organic forms, are a key step in chaparral N cycling. Reactive N constitutes the pools of N critical to ecosystem dynamics and is distributed through many pools in chaparral dominated ecosystems analogous to C pools (Fig. 6.3). An important source of chaparral N is the direct conversion of N2 into the bioavailable form of ammonia (NH3 +) through the process of nitrogen fixation , mainly through the action of symbiotic bacteria and to a lesser extent by free-living bacteria or lightning. Several chaparral species, including Ceanothus spp., western redbud (Cercis occidentalis), desert false indigo (Amorpha fruticose), and chaparral pea (Pickeringia montana) form N fixing symbioses and contribute to N inputs to chaparral ecosystems (Delwiche et al. 1965; Ulery et al. 1995). N fixation in addition to atmospheric inputs, which have historically been low but are now elevated for many chaparral ecosystems, set the stage for chaparral N biogeochemical cycling. Rates of N deposition to chaparral ecosystems can vary widely and may depend on both atmospheric concentrations of reactive N as well as endogenous factors of species traits and biomass. For the same atmospheric reactive N concentrations, deposition increases with both increases in leaf-specific deposition velocity and increased leaf area (Bytnerowicz et al. 2015). Thus, while atmospheric reactive N concentrations contribute to N inputs, ecosystem processes including fixation and deposition also have roles in determining N inputs.
Following fixation or atmospheric inputs, if not directly incorporated into plant biomass, NH3 + is then converted to nitrate (NO3 −) via nitrite (NO2 −) in the two-step process of nitrification carried out by aerobic chemoautotrophs. Both NH3 +and NO3 − are available for assimilation by plants and soil microorganisms into organic matter. The process of mineralization returns organic N to plant and microbial available N pools by converting soil organic matter back to ammonia. Simultaneously, soil conditions in chaparral tend to be coarse-textured, quickly draining, and aerobic, with high base cation saturation and low organic matter, which generally lead to high rates of nitrification in relation to mineralization (Fenn et al. 2003). Nevertheless, in some sites, such as a coastal old-growth sites, low rates of net nitrification have been observed (Hanan et al. 2016a) further highlighting the diversity of chaparral biogeochemistry. Species traits, such as higher leaf N content and low concentrations of secondary compounds, can increase N inputs to the soil and rates of biogeochemical cycling through litter interactions with soil microbes. Some shrubs (e.g., chamise [Adenostema fasciculatum]) promote higher concentrations of nitrogenous compounds in soil than others (e.g., desert ceanothus [Ceanothus greggii]), presumably because some shrubs cultivate nitrifying bacteria more than others (Fenn et al. 1993).
Mineral N in the soil that is not immobilized in microbial or plant biomass may be lost from the ecosystem primarily through leaching, volatilization , and trace-gas emission . Nitrate is highly mobile in soil, and coupled with low rates of biotic activity during the winter rainy season, considerable leaching loss can occur. Nitrate leaching becomes a significant form of N export from chaparral dominated ecosystems at deposition rates above 25 kg N ha−1 year−1 (Fenn et al. 1996). While leaching is relatively low in undisturbed and unpolluted chaparral, losses are generally highest following the first winter rains. N can also be lost to the atmosphere through the process of denitrification where oxidized nitrogen is used as an alternative electron donor in heterotrophic anaerobic respiration resulting in the sequential production of nitric oxide (NO), nitrous oxide (N2O) and N2. The emissions of NO and N2O are conceptualized by the hole-in-the-pipe model developed by Firestone and Davidson (1989), where the size of the pipe describes the total amount of nitrogen moving through the system and the “holes” in the pipe represent the relative amount of NO or N2O lost through gaseous production. The hole size is controlled mainly by soil water content, influencing both oxygen availability and diffusion of gases from the soil, facilitating production or consumption. NO and presumably N2O emissions from chaparral soil are generally are low but can be large upon wetting of dry soils (Homyak and Sickman 2014).
Fire can lead to immediate N losses to the atmosphere and have longer term influences on chaparral N biogeochemistry. Following fires, stream export of N can be elevated up to 40 times that of unburned areas (Riggan et al. 1994), suggesting both increased mobility of N and reduced plant uptake. In the first growing season after fire, new leaf and stem growth contains higher N content than growth before fire or in subsequent growing seasons owing to higher mineral N availability in the soil (Franco-Vizcaino and Sosa-Ramirez 1997). Spikes in nitrogen can also induce germination of certain chaparral seeds postfire (Thanos and Rundel 1995; Keeley and Fotheringham 1997). However, elevated N cycling has been shown to dissipate within 7 months of the fire (de Koff et al. 2006). High severity fires can also result in higher emissions of volatilized N , NOx and N2O into the atmosphere (DeBano and Conrad 1978). Although soil N is reduced postfire, burned biomass can lead to a spike of soil NO3 − (Christensen 1973). This spike subsequently increases nitrification rates and results in a net loss of N from chaparral systems as NOx and N2O emissions into the atmosphere and NO3 − into streamflow . Char legacies from previous fires may increase rates of N immobilization into microbial biomass (Hanan et al. 2016b). As with soil C, fire can lead to short-term reductions in soil N, which also can return to prefire levels within a year (Graham et al. 2016).
Because chaparral exists under the unique constraints of a Mediterranean-type climate, soils are subject to long periods of hot and dry conditions. As a consequence of this regular drying, pulse-driven dynamics can also contribute to nitrogen cycling and associated losses via leaching and trace-gas emissions (Fierer and Schimel 2002; Miller et al. 2005; Oikawa et al. 2015; Liang et al. 2016). Soil C and N accumulate during the dry summer months when microbial and plant activity is limited by soil moisture. Precipitation results in a large flush of C and N resources that can produce a substantial pulse in soil gaseous emissions. These wetting-induced pulses can contribute to the annual budget of C and N chaparral in arid systems. For example, NO emissions from chaparral ecosystem can be large, exceeding 350 ng m−2 s−1 compared to unwetted values in the summer (near zero) (Homyak and Sickman 2014). Thus, in contrast to an expectation of N limitation , the potential for chaparral ecosystems to also exhibit high N leakiness, typically associated with N saturation, is also evident (Fenn et al. 1996; Homyak et al. 2014).
The potential for high N losses have led to revisions of standard ecosystem concepts of nutrient limitation and nutrient saturation in chaparral dominated ecosystems. Microbial activity can more rapidly immobilize N at the beginning of the rain season, but plant uptake can outcompete microbial nitrogen cycling towards the end of the rain season, resulting in a transition from nitrogen loss to nitrogen retention (Homyak et al. 2014, 2016). The often low N input rates and presence of N conserving strategies by chaparral communities, including plant nutrient reabsorption, litter N immobilization, deep rooting , and supporting N fixation, which conserve N within chaparral ecosystems, suggests N limitation . In contrast, the regular large emissions suggest N saturation. Working towards a more comprehensive theory of ecosystem N cycling that allows for simultaneous indicators of both N limitation and saturation has been a source of recent interest. Recent findings suggest N cycling rates and the importance of immobilization and loss pathways may dominate following initial rain at the beginning of the winter season but then plant activity immobilizes N later in the season with the initiation of plant growth (Homyak et al. 2016).
In addition to the nutrient consequences for chaparral ecosystems, N losses to gaseous production of NO and N2O can have multiple environmental consequences. NO contributes to the production of ozone, representing a significant health concern for people and plants and contributes to acid rain and N redeposition to other ecosystems. N2O is a potent greenhouse gas, which is ~300 times more effective than CO2 at depleting ozone in the upper atmosphere. NO3 − losses to streamflow can lead to deterioration of downstream water quality (Fenn et al. 1998). In addition, many of the chaparral ecosystems in California, primarily inland areas, are subjected to elevated rates of anthropogenic nitrogen deposition , leading to stream eutrophication , and the hydrologic export of nitrate , all of which are indicative of nitrogen saturation in these systems.
6.4 Anthropogenic Influences and Future Trajectory of Chaparral Biogeochemistry
6.4.1 Nitrogen Deposition and Chaparral Biogeochemistry
Anthropogenic activities have resulted in inputs of N into natural ecosystems that have surpassed biological fixation (Galloway et al. 2008). In the western United States and the regions of chaparral dominated ecosystems in North America, the largest sources of N deposition are transportation , agriculture , and industry, typically resulting in deposition rates of 1–4 kg N ha−1 year−1 (Fenn et al. 2003). This number can be as high as 30–90 kg N ha−1 year−1 in chaparral dominated communities near urban and agricultural areas (Fenn et al. 2003; Bytnerowicz et al. 2015). This dramatic increase in N deposition can result in extensive consequences to chaparral ecosystems, such as species composition shifts , soil acidification , and eutrophication of aquatic systems (see Box 6.2). In southern California, which experiences some of the highest rates of N deposition in North America (Bytnerowicz et al. 2015), research has linked N deposition with an increase in the establishment of non-native annual plant species and consequently, greater risk of fire (Fenn et al. 2003; Rao et al. 2010). While N deposition can lead to increases in chaparral ecosystem C and N pools (Vourlitis et al. 2007b), the effects are complex. Critical loads of N deposition for chaparral have been assessed at 10 kg N ha−1 year−1 for small catchments (4–10 ha or 9.9–24.7 acres) and 14 kg N ha−1 year−1 for intermediate and large catchments (>10 ha or 24.7 acres) (Fenn et al. 2010). Currently, ~15% of chaparral land area in California is in exceedance of 10 kg N and 3.3% of chaparral land for the 14 kg N thresholds. In contrast, if a critical load of 5.5 kg N ha−1 year−1 is used, the level identified leading to a shift in chaparral lichen communities to eutrophic communities, then 53% of chaparral areas exceed this threshold (Fenn et al. 2010). The impact of changes in N can be seen in the modification of lichen communities which experience an increasing dominance by nitrophytes. Increases in N deposition have been associated with more than three orders of magnitude greater NO3 − export from chaparral ecosystems (Riggan et al. 1985), with stream [NO3 −] reaching 1120 μmolc L−1 from catchments dominated by chaparral in a high deposition region after a recent fire (Riggan et al. 1994) compared to very low detection levels in pristine streams. While chaparral communities are historically considered resistant to the effects of atmospheric pollution (Allen et al. 2005a; Vourlitis and Pasquini 2009), increased atmospheric N deposition may significantly alter the soil chemistry, resulting in a modification of the competitive balance between chaparral shrubs and non-native herbs (Rao et al. 2009; Fenn et al. 2010; Vourlitis 2017). The effects of increased deposition can influence the biogeophysical drivers of chaparral and further influence self-organizational processes within the ecosystem.
Box 6.2: Case Study of Nitrogen Fertilization Experiment
One of the most comprehensive and long-term evaluations of the effects of experimental N fertilization to shrublands of southern California, including the chaparral dominated Sky Oaks field site , has been conducted by Vourlitis and colleagues (Vourlitis et al. 2007a; Vourlitis and Hentz 2016) (Fig. 6.4). This experiment has shown complex biogeochemical responses to high rates of N amendments that continue to change over more than a decade of N additions (Vourlitis and Hentz 2016). Immediately following N amendments, large increases in mineral N was observed (Vourlitis et al. 2007a), however the mineral N increases did not translate into immediate changes to total ecosystem N or C content. Several complex biogeochemical pathways lead to multiple indirect responses to N addition in chaparral dominated ecosystems (Vourlitis and Fernandez 2015). For example, soils from the experimental plots showed patterns of reduced rates of litter decomposition and N demand, which may affect C storage and N availability following increased N deposition (Biudes and Vourlitis 2012). Alternatively, increases in N trace gas emissions following N deposition may reflect an elevated N loss pathway counteracting the effects of N deposition to the ecosystem (Vourlitis et al. 2015). The high N leakage potentials determined from laboratory incubations are consistent with findings after 4 years following of N additions that no changes in either ecosystem C inputs or N storage was observed (Vourlitis et al. 2009).
Overlaying the experimental manipulation , the effects of N addition on biogeochemical cycling was strongly coupled to precipitation patterns, with greater effects observed in wetter years further complicated by historical legacy effects (Vourlitis 2012). After 4 years of fertilization, increases in above-ground C and N were observed and the ecosystem continues responding to N additions up to a decade following N amendments (Vourlitis and Hentz 2016). Throughout this period, N loss pathways were also elevated, again highlighting the complex N dynamics in chaparral ecosystems that show characteristics both of N limitation and N saturation. Further examination of plant leaf and hydraulic responses have shown large differences among species in their responses, with nitrogen addition leading to increased leaf N content and increasing hydraulic conductivity, stomatal conductance, and leaf carbon isotopic composition while decreasing wood density in several species (Pivovaroff et al. 2016). These plant responses to nitrogen generally increase photosynthetic rates and water movement through the organism and have clear implications for ecosystem drought sensitivity and may influence future community assembly. This prediction has been supported through recent findings of species shifts in response to fertilization and increasing the potential of non-native grass invasion (Pasquini and Vourlitis 2010; Vourlitis 2017). This long-term experiment shows that even after a decade of elevated N, chaparral dominated ecosystems can continue exhibiting dynamics of both N limitation , through positive effects on C accumulation , and N saturation, through high potential rates of N leakage. Future biogeochemical trajectories may depend on community reorganization dependent on nutrient availability and drought tolerances .
6.4.2 Grazing and Chaparral Biogeochemistry
The influence of grazing on C and N has long been recognized. Studies of grazing in chaparral indicate that soil C and N contents can be significantly reduced by grazing (Brejda 1997; Milchunas 2006). Grazing induced C and N reductions are consistent with other disturbances such as fire. Nevertheless, grazing has also, in some cases, been found to increase soil C and N (Brejda 1997), contributing to the biogeochemical diversity of chaparral dominated ecosystems. Grazing-induced changes in soil N and C primarily occur in litter and surface soil layers due to interception of foliage by grazers before leaf senescence (Severson and DeBano 1991). However, studies of grazing associated reductions in soil N undertaken in chaparral shrublands in Arizona found reductions were greatest under the canopy of nitrogen-fixing Ceanothus shrubs than under other shrubs, likely due to preferential browsing of that taxa by goats and other grazers (Sidahmed et al. 1981; Knipe 1983; Severson and DeBano 1991). While goats typically exhibit grazing preferences similar to native grazers and cattle (Milchunas 2006), the preferences of different grazer species can differ substantially, with some grazing heavily on Ceanothus (Knipe 1983). Thus, the effects of grazing on N fixing species, and associated impacts on N cycling and storage, may depend on the preferences of local ungulates.
In addition to the direct impacts on soil chemistry through browsing and litter reduction, heavy grazing has been observed to reduce shrub cover while also preventing colonization of the subsequent bare soil by perennial forbs and grasses. In the absence of this re-colonization, previous shrub cover is converted to an annual dominated cover that is more capable of withstanding heavy grazing pressures (Severson and DeBano 1991). Nevertheless, examination of different levels of grazing in a watershed in Arizona detected no difference in runoff or erosion (Rich and Reynolds 1963). Thus grazing can have direct influence on above-ground C and N pools through consumption, and may have multiple indirect effects through changes in soil structure and plant species composition. Yet, many aspects of chaparral biogeochemistry may be resilient to grazing. The diversity of chaparral responses to grazing may depend on moisture availability, topography , local species characteristics, and historical legacies.
6.4.3 Fire and Chaparral Biogeochemistry
Fires and fire regimes are an interaction between biophysical drivers and ecohydrological self-organization that creates potentially long-lived legacies. In recent times, chaparral fires are influenced by human activities both through increased fire suppression and ignition sources with a net effect of reducing the fire interval (Syphard et al. 2007). More frequent fires can influence plant community composition by first reducing the competitive abilities of obligate seeders compared to resprouters (Franklin et al. 2005) which, in turn, may have large indirect effects on biogeochemical cycling. In addition to the direct loss of biomass and litter pools through burning , differences in fire history and fire characteristics can have large consequences to chaparral biogeochemistry. Connected to fire, fuel management practices such as the creation of fuelbreaks and fire breaks are formed by crushing or otherwise denuding strips of land of shrub cover (Green 1977), to slow fires and facilitate access by firefighters and equipment into otherwise inaccessible areas. Such disturbances can alter soil chemistry substantially. Firebreaks are also colonized predominantly by non-native species and may then function as sources for further invasion into adjacent areas, with a variety of impacts on local biogeochemistry (Zink et al. 1995; Keeley et al. 2005; Mayberry 2011; Syphard et al. 2014).
6.4.4 Land Use and Development
In recent decades, urban development has increasingly encroached on chaparral landscapes (Syphard et al. 2007). The conversion and fragmentation of chaparral has multiple implications, both due to the direct loss of chaparral habitat and due to the downstream effects of developed landscapes on adjacent chaparral. As chaparral habitat is developed, the removal of deep-rooted shrubs and replacement by largely impermeable surfaces typically results in significant increases in runoff and stormwater discharge (White and Greer 2006; Warrick and Rubin 2007, see Chap. 7). Ironically, urbanization can lead to some improvements in water quality , as sediment discharge often remains similar to pre-development levels, while water discharge increases dramatically, thereby diluting suspended sediment loads (Warrick and Rubin 2007). In addition, as housing development encroaches into chaparral vegetation , the natural fire cycle has increasingly become a major hazard for human safety and property loss (Rodrigue 1993), leading to a variety of management strategies intended to mitigate risks to life and property (Gardner et al. 1987; Gill and Stephens 2009). Many of these strategies may exacerbate the effects of such development on native chaparral vegetation, and also impact the biogeochemistry of the surrounding landscape through denudation and conversion of native vegetation , leaching of heavy metals, and other processes.
Associated with encroaching urbanization into chaparral systems, is the creation of roads for access and alternative evacuation routes in case of fire (Cova 2005). Roads have been found to act as corridors for non-native plant species and increases the susceptibility of adjacent areas to invasion (Lambrinos 2006; Davies et al. 2013), which may in turn alter soil sequestration of both C and N. In addition, roadside soils have been observed to exhibit increased deposition of reactive N owing to automobile exhaust (Fenn et al. 1996; Rossi et al. 2015) as well as increased heavy metal content (Rossi et al. 2015).
6.4.5 Atmospheric CO2 Concentrations and Chaparral Biogeochemistry
Another important influence on the biogeochemistry of chaparral systems is the direct effect of rising CO2 concentrations which not only influence climate but also have a direct effect on many biogeochemical processes. To experimentally evaluate C cycle sensitivities to CO2 concentrations in chaparral dominated landscapes a Free Air CO2 Enrichment (FACE) experiment was established in 1995 at the Sky Oaks field site (Roberts et al. 1998). Within 3.5 years, the chaparral ecosystems had increased rates of CO2 uptake and increased storage of soil C (Treseder et al. 2003). The effect of increased CO2 in particular led to increases in the C content of water stable aggregates, which was hypothesized to have long turnover periods and form a sequestration pathway. In contrast microbial turnover may have been faster and potentially limited by N availability (Allen et al. 2005b). Nevertheless, after 6 years of elevated CO2, a surprising result showed that increasing CO2 concentrations led to reductions in soil C content (Del Galdo et al. 2006). This response was associated with decreases in the fraction of large particles within the soil, bound in part by organic materials (macroaggregates) at the highest CO2 levels, which implied reduced physical protection of soil organic matter. Based on these findings, future CO2 atmospheric concentrations may impose a positive feedback from chaparral soils as they will concomitantly increase emissions (Del Galdo et al. 2006; Trueman et al. 2009).
6.5 Potential Chaparral Type-Conversion and Biogeochemical Cycles
Historically, much effort has been directed to type-converting chaparral either to grass dominated communities for grazing or coniferous forest for timber harvesting and more recently, a variety of factors are causing type-conversion of chaparral to non-native grasslands. Evidence suggests these regime shifts of chaparral communities from shrub dominated to grass dominated may have a large influence on biogeochemical cycling (Park et al. in press).
Coniferous forests , while unlikely to encroach into chaparral under normal circumstances (Burns and Sauer 1992), have invaded into chaparral following extensive fire suppression (Nagle and Taylor 2005; Lauvaux et al. 2016). Further, conifers have been planted in areas dominated by chaparral after fires, primarily to increase the area of conifer forest for future timber harvest on federal lands (Burns and Sauer 1992). Comparisons of soil C sequestration between chaparral and coniferous forests have shown mixed conclusions regarding the effects of such vegetation conversion on C and N storage. Some studies indicate chaparral sequesters more C and N within the soil A-horizon than coniferous forests (Ulery et al. 1995). Similarly, comparisons of chaparral to oak or conifer dominated forests also found that chaparral sequestered greater amounts of soil C and N than pine forests (Ulery et al. 1995; Quideau et al. 1998). In contrast to these findings, other studies on shrublands in Israel (Grünzweig et al. 2007) found far less above- and below-ground carbon sequestration than pine and juniper forests. Evidence suggests a combination of increased N use efficiency, enhanced Soil Organic Carbon (SOC) protection, and reduced decomposition rates following an increase in forest species. This latter finding implies the potential for increases in biogeochemical pools when chaparral shrublands are converted to coniferous forests.
In contrast, the potential for non-native grass invasions is posing a much greater threat to chaparral ecosystems . Although chaparral has historically been considered resistant to plant invasions (Minnich and Bahr 1995; Allen et al. 2005b), increasing drought intensity and anthropogenic disturbances such as altered fire regimes , encroaching urbanization , and increased dissection of chaparral landscapes may push many of these systems to tipping points of shrub mortality and possible type-conversion to invaded grasslands (Hamilton 1997; Keeley and Brennan 2012; Syphard et al. 2013; Meng et al. 2014). Roadsides, firebreaks, and other anthropogenic features within chaparral are also typically dominated by non-native herbs (Zink et al. 1995; Giessow and Zedler 1996; Lambrinos 2006; Davies et al. 2013). In the past, managers have attempted to forcibly convert chaparral to grasslands in efforts to increase water yield, improve fire control, or increase grazing habitat (Bentley 1967; Cable 1975; Anderson et al. 1976). These conversion efforts, completed through the extensive use of physical removal, active seeding, and chemical application, have been somewhat successful and have now contributed to the invasion of non-native annual grasses . Grazing has also been used as a mechanism to impose type-conversion of chaparral into grasslands (Knipe 1982, 1983; Severson and DeBano 1991).
Conversion of coastal sage scrub (CSS) communities into non-native grasslands has been documented to increase sequestration of both N and C (Wolkovich et al. 2010), in contrast, chaparral dominated communities have experienced substantial reductions in soil C sequestration after type-conversion to non-native grasslands (Williamson et al. 2004a). The reductions are in part associated with greater plant cover and depth of rooting in chaparral species than grasses, along with different phenology , and litter chemistry. Soil C reductions occur not only at soil depths below the root horizon of the shallower-rooted grasses, but also in the shallower soil layers in which grasses may be expected to deposit the majority of soil C. Such increases in surface C may increase rates of decomposition through greater exposure to photodegradation and sensitivity to small rainfall events.
In addition to reductions in C and N sequestration with the invasion of non-native grasses, these grasses also intercept far less moisture than chaparral shrubs (Rowe 1963; Corbett and Crouse 1968; Williamson et al. 2004a) and do not capture water at depths below their roots (Williamson et al. 2004b), which are shallow in comparison to many chaparral shrub species. As a consequence, chaparral sites have also been observed to exhibit large increases in throughflow and downstream soil moisture (Hibbert 1971; Davis 1984) as well as possible groundwater recharge (Williamson et al. 2004b) after conversion into invaded grasslands. In aggregate, the hydrologic effects of conversion can even convert streams from intermittent to perennial (Orme and Bailey 1970). The increase in throughflow leads to reduced water available for plants to use in conjunction with photosynthesis and can directly reduce total ecosystem C inputs. Further, when coupled with a reduced ability of grasses to anchor soils, invaded grassland invasion often leads to substantial increases in erosion and soil slippage compared to intact chaparral (Rice et al. 1969). Increased erosion associated with the conversion of chaparral into grasslands may further exacerbate the reduced ability of grass cover to sequester C into the soil by exposing lower soil layers to weathering, and increasing the rate of C cycling from the deeper soil layers back into the atmosphere.
Conversion of chaparral to grasslands has also been observed to increase the nitrate content of stream runoff (Longstreth and Patten 1975). Runoff nitrate content has been observed to increase by up to 100-fold in the years immediately following conversion (Davis and DeBano 1986; Davis 1987), likely due to the decomposition of above- and below-ground organic matter from the remnants of displaced shrubs. Such effects are also long-lasting with studies estimating nitrate runoff to remain 14-fold or greater above pre-conversion levels a decade after conversion (Davis 1984).
Increases in downslope moisture availability after conversion of chaparral watersheds to grasses has also been observed to produce substantial effects on downstream vegetation. Examinations of riparian areas downstream of formerly chaparral sites that had been recently converted were found to have tripled the density of their riparian vegetation due to increased streamflow and flow duration (DeBano et al. 1984). Moisture availability is also a critical component in determining the success of postfire chaparral re-colonization, with low moisture benefitting non-native herbs and grasses over native shrubs (Frazer and Davis 1988; Keeley et al. 2005). Thus, conversion of upstream watersheds from chaparral into grasslands possibly will increase resistance and resilience of downstream chaparral to invasion.
6.6 Toward Improved Monitoring and Modeling of Chaparral Biogeochemical Cycles
6.6.1 Environmental Sensing Approaches for Assessing Chaparral dominated Biogeochemical Cycles
The ability to measure and model the dynamics and trajectories of chaparral biogeochemical cycles is rapidly increasing in sophistication. Many new in-situ and remote sensing tools are becoming available that allow multiple temporal scale resolutions—from minutes to decades—of many important biogeochemical dynamics . These new monitoring technologies are providing extensive information to improve models and their prediction capabilities. Historically, the primary approach for quantifying ecosystem biogeochemical dynamics were through measurements of changes in the dominant pools, typically at annual scales for production and longer scales for soils. This research in chaparral dominated ecosystems requires extensive fieldwork in environments that are challenging to sample due to the continuous low level woody coverage, rugged topography, and deep rocky soils. Advanced environmental sensing platforms, both remote imaging and in-situ sensors, are providing new methods for quantifying variation in many factors closely connected to biogeochemical cycling in chaparral, although deep soil characterization remains a challenge.
Remote imaging platforms allow new methods for mapping chaparral biogeochemical variation for C, N, and potentially other elements using hyperspectral approaches from airborne and satellite sensors which record information across hundreds of spectral bands (Serrano et al. 2002; Dahlin et al. 2013). Resulting hyperspectral imagery have been used to develop estimates of carbon exchange in chaparral dominated ecosystems (Fuentes et al. 2006). Detailed information on canopy cover and vertical biomass distributions can also be collected through active remote sensing approaches using Light Detection and Ranging Laser (LIDAR) imagery (Garcia et al. 2015). Finally, thermal imaging of land surfaces is providing new opportunities to evaluate metabolic activity and water dynamics, especially evapotranspiration (Sims et al. 2008; Kustas and Anderson 2009), which is currently the focus of studies in chaparral, forest, and grassland ecosystems. When compared with field measurements, thermal imaging provides Other satellite-based sensors, such as the Ozone Monitoring Instrument (OMI) allow the characterization of atmospheric pollution including ozone and NO2 (Levelt et al. 2006) that may affect chaparral biogeochemistry. In the future, opportunities for the remote characterization of chaparral will likely expand through efforts such as the combined imaging spectrometer and multi-band thermal sensors in the HyspIRI mission (Roberts et al. 2015) and the combination of different instruments such as Sentinal (European Space Agency) and Landsat (US National Aeronautical and Space Administration) to create a virtual satellite constellation providing multiple viewing angles and imaging times.
The advances in remote sensing are concomitant with advancements in developing automated in-situ sensor measurements. In-situ environmental sensors can now provide minute and hourly scale resolution information on many components of biogeochemical cycling (Rundel et al. 2009). Repeating ground-based cameras are providing extensive information on the dynamics of fine scale heterogeneity occurring both above-ground (Sonnentag et al. 2012) and in the soil (Allen and Kitajima 2013). Embedded sensors are also allowing the evaluation of many biogeochemical transformations, including whole ecosystem carbon and energy exchanges, soil metabolism, and whole plant water fluxes. Expanded applications of environmental sensor approaches in chaparral dominated ecosystems is a clear research priority for improved understanding of biogeochemical dynamics in this unusual ecosystem.
6.6.2 Looking Toward the Next Generation of Chaparral Biogeochemical Models
To synthesize understanding of chaparral dominated biogeochemical interactions and improve predictive skill of biogeochemical trajectories new modeling approaches are necessary. The performance of current ecosystem, land surface, and biogeochemistry models are generally applied to forested ecosystems and minimally evaluated for chaparral dominated ecosystems. Broadly used ecosystem models applicable to global ecosystem distributions generally perform poorly in regions of extensive water limitation (Niu et al. 2014) and likely even more so in MTC’s where energy and moisture are disconnected. Even when specifically parameterized for chaparral dominated ecosystems model performance is limited (Li et al. 2006) or not evaluated (Tague et al. 2009). Ecosystem and land surface models generally miss important pulse driven dynamics and have poor representation of precipitation -driven phenology . Current models also have limited abilities to explicitly incorporate the large biological and geomorphological diversity characteristic of chaparral dominated ecosystems that can lead to spatial hotspots and temporal pulses of biogeochemical cycling.
To improve biogeochemical models , lessons can be learned from other dryland specific models. Ecosystem models developed specifically for drylands can obtain reasonable matches with field data (Shen et al. 2008, 2016) although even dryland specific models have not been tested in chaparral dominated ecosystems. Pulse driven biogeochemical cycling has characteristics that are distinct from non-pulse dynamics and generally excluded from ecosystem or land surface models. Simplified models of pulse driven biogeochemical dynamics can be effective using either theoretical (Jenerette et al. 2008) or empirical (Li et al. 2010) justifications, which can then provide pulse functionality within more general models. Recently, more complex pulse models have expanded in their representation of underlying mechanisms, including the formation and consumption of a labile C pool, and predictive abilities (Oikawa et al. 2014) and fit within broader modeling goals of a better microbial representation within ecosystem models. Addressing two research priorities will help advance chaparral modelling efforts. One is the accurate inclusion of precipitation and its relationship with phenology (Jenerette et al. 2010). The underlying controls on phenology are not well understood and the variation among species in phenological timing and responsiveness to meteorological dynamics can be large. Second, an improved representation of topographic heterogeneity is needed to better account for water dynamics and fine scale hotspots of biogeochemical cycling within chaparral dominated regions (Tague et al. 2009).
New biogeochemical models that build from theoretical underpinnings and combine data from new remote sensing techniques with extensive empirical evidence will provide a roadmap for improved understanding of chaparral dominated biogeochemical dynamics . The large uncertainties in models currently applied to chaparral ecosystems suggest a need for improved models that can better predict interactive element cycles in response to multiple global change drivers. At the same time the uniqueness of chaparral ecosystems provides an important testbed for a more comprehensive theory of ecosystem dynamics.
6.7 Conclusion
The breadth of chaparral biogeochemical diversity in part contributes to its high potential for continued biogeochemical sequestration storage over long time periods as well as rapid fluxes over short periods. Interactions among biophysical drivers, self-organization, and historical legacies generate large diversity in chaparral ecosystem dynamics. The consequences of ecosystem diversity are broad distributions in the rates of biogeochemical cycling. Current and future distributions of chaparral biogeochemical cycling will likely respond to multiple interactive anthropogenic drivers including increased nitrogen inputs, grazing , increased fire frequencies , more fragmented landscapes, and increasing CO2 concentrations. These changes can influence the biogeophysical template, self-organization, and the influence of historical legacies through direct and indirect pathways. To better quantify the ecosystem services provided by chaparral and their potential future trajectories, new measurement and modeling opportunities are becoming available. Monitoring through in-situ and remote sensing techniques substantially increases the amount of data in both spatial and temporal resolution and extent than can be obtained from time-intensive field sampling. New modeling approaches are reducing uncertainties in extending findings spatially and improving projections of future biogeochemical cycling. Validating sensor measurements and model projections will still require direct field measurements, but this work will allow a larger source of information than fieldwork alone. A greater emphasis on measuring biogeochemical cycles in chaparral in combination with traditional fieldwork, new sensor tools , and advanced modeling can provide information to resolve uncertainties in ecosystem theory and to assess current and future provisioning of chaparral ecosystem services, such as climate change moderation through C sequestration or clean water provisioning through limited NO3 losses to streams.
References
Ackerly, D. 2004. Functional strategies of chaparral shrubs in relation to seasonal water deficit and disturbance. Ecological Monographs 74:25-44.
Allen, M. F., and K. Kitajima. 2013. In situ high-frequency observations of mycorrhizas. New Phytologist 200:222-228.
Allen, M. F., J. N. Klironomos, K. K. Treseder, and W. C. Oechel. 2005a. Responses of soil biota to elevated CO2 in a chaparral ecosystem. Ecological Applications 15:1701-1711.
Allen, E. B., A. G. Sirulnik, L. Egerton-Warburton, S. N. Kee, A. Bytnerowicz, P. E. Padgett, P. J. Temple, M. E. Fenn, M. A. Poth, and T. Meixner. 2005b. Air pollution and vegetation change in southern California coastal sage scrub: a comparison with chaparral and coniferous forest. Pages 79-95 in B. E. Kus and J. L. Beyers, editors. Planning for biodiversity: bringing research and management together. General Technical Report PSW-GTR-195. Pacific Southwest Research Station, USDA Forest Service, Albany, California, USA.
Amundson, R., D. D. Richter, G. S. Humphreys, E. G. Jobbagy, and J. Gaillardet. 2007. Coupling between biota and earth materials in the Critical Zone. Elements 3:327-332.
Anderson, H. W., M. D. Hoover, and K. G. Reinhart. 1976. Forests and water: effects of forest management on floods, sedimentation, and water supply. General Technical Report GTR-PSW-018. USDA Forest Service, Pacific Southwest Forest and Range Experiment Station, Berkeley, California, USA.
Ashby, W. R. 1956. An introduction to cybernetics. Chapman and Hall Ltd., New York, New York, USA.
Austin, A. T., and L. Vivanco. 2006. Plant litter decomposition in a semi-arid ecosystem controlled by photodegradation. Nature 442:555-558.
Bailey, R. G. 2004. Identifying ecoregion boundaries. Environmental Management 34:S14-S26.
Baldocchi, D., J. W. Tang, and L. K. Xu. 2006. How switches and lags in biophysical regulators affect spatial-temporal variation of soil respiration in an oak-grass savanna. Journal of Geophysical Research: Biogeosciences 111:G02008.
Baldocchi, D. D., and L. K. Xu. 2007. What limits evaporation from Mediterranean oak woodlands—the supply of moisture in the soil, physiological control by plants, or the demand by the atmosphere? Advances in Water Resources 30:2113-2122.
Bell, D. T. 2001. Ecological response syndromes in the flora of southwestern Western Australia: fire resprouters versus reseeders. Botanical Review 67:417-440.
Bentley, J. R. 1967. Conversion of chaparral areas to grassland: techniques used in California. USDA Agriculture Handbook number 328. USDA, Washington D.C., USA.
Biederman, J. A., R. L. Scott, M. L. Goulden, R. Vargas, M. E. Litvak, T. E. Kolb, E. A. Yepez, W. C. Oechel, P. D. Blanken, T. W. Bell, J. Garatuza-Payan, G. E. Maurer, S. Dore, and S. P. Burns. 2016. Terrestrial carbon balance in a drier world: the effects of water availability in southwestern North America. Global Change Biology 22:1867-1879.
Biudes, M. S. and G. L. Vourlitis. 2012. Carbon and nitrogen mineralization of a semiarid shrubland exposed to experimental nitrogen deposition. Soil Science Society of America Journal 76:2068-2073.
Brejda, J. T. 1997. Soil changes following 18 years of protection from grazing in Arizona chaparral. Southwestern Association of Naturalists 42:478-487.
Burns, C., and J. Sauer. 1992. Resistance by natural vegetation in the San Gabriel Mountains of California to invasion by introduced conifers. Global Ecology and Biogeography Letters 2:46-51.
Bytnerowicz, A., R. F. Johnson, L. Zhang, G. D. Jenerette, M. E. Fenn, S. L. Schilling, and I. Gonzalez-Fernandez. 2015. An empirical inferential method of estimating nitrogen deposition to Mediterranean-type ecosystems: the San Bernardino Mountains case study. Environmental Pollution 203:69-88.
Bytnerowicz, A., P. R. Miller, D. M. Olszyk, P. J. Dawson, and C. A. Fox. 1987. Gaseous and particulate air-pollution in the San Gabriel Mountains of southern California. Atmospheric Environment 21:1805-1814.
Cable, D. R. 1975. Range management in the chaparral type and its ecological basis: the status of our knowledge. Research Paper RM-155. USDA Forest Service, Rocky Mountain Forest and Range Experiment Station, Fort Collins, Colorado, USA.
Chapin, F. S., P. A. Matson, and H. A. Mooney. 2012. Principles of terrestrial ecosystem ecology. Springer, New York, New York, USA.
Chatterjee, A., and G. D. Jenerette. 2011. Spatial variability of soil metabolic rate along a dryland elevation gradient. Landscape Ecology 26:1111-1123.
Chatterjee, A., and G. D. Jenerette. 2015. Variation in soil organic matter accumulation and metabolic activity along an elevation gradient in the Santa Rosa Mountains of Southern California, USA. Journal of Arid Land 7:814-819.
Chorover, J., P. A. Troch, C. Rasmussen, P. D. Brooks, J. D. Pelletier, D. D. Breshears, T. E. Huxman, S. A. Kurc, K. A. Lohse, J. C. McIntosh, T. Meixner, M. G. Schaap, M. E. Litvak, J. Perdrial, A. Harpold, and M. Durcik. 2011. How water, carbon, and energy drive Critical Zone evolution: the Jemez-Santa Catalina Critical Zone Observatory. Vadose Zone Journal 10:884-899.
Christensen, N. L. 1973. Fire and the nitrogen cycle in California chaparral. Science 181:66-67.
Corbett, E. S., and R. P. Crouse. 1968. Rainfall intrception by annual grass and chaparral: losses compared. PSW-RP-48. USDA Forest Service, Pacific Southwest Forest and Range Experiment Station, Berkeley, California, USA.
Cornwell, W. K., J. H. C. Cornelissen, K. Amatangelo, E. Dorrepaal, V. T. Eviner, O. Godoy, S. E. Hobbie, B. Hoorens, H. Kurokawa, N. Perez-Harguindeguy, H. M. Quested, L. S. Santiago, D. A. Wardle, I. J. Wright, R. Aerts, S. D. Allison, P. van Bodegom, V. Brovkin, A. Chatain, T. V. Callaghan, S. Diaz, E. Garnier, D. E. Gurvich, E. Kazakou, J. A. Klein, J. Read, P. B. Reich, N. A. Soudzilovskaia, M. V. Vaieretti, and M. Westoby. 2008. Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecology Letters 11:1065-1071.
Cova, T. J. 2005. Public safety in the urban-wildland interface: should fire-prone communities have a maximum occupancy? Natural Hazards Review 6:99-108.
Dahlin, K. M., G. P. Asner, and C. B. Field. 2013. Environmental and community controls on plant canopy chemistry in a Mediterranean-type ecosystem. Proceedings of the National Academy of Sciences of the United States of America 110:6895-6900.
Davies, K. W., A. M. Nafus, and M. D. Madsen. 2013. Medusahead invasion along unimproved roads, animal trails, and random transects. Western North American Naturalist 73:54-59.
Davis, E. A. 1984. Conversion of Arizona chaparral to grass increases water yield and nitrate loss. Water Resources Research 20:1643-1649.
Davis, E. A. 1987. Chaparral conversion and streamflow: nitrate increase is balanced mainly by a decrease in bicarbonate. Water Resources Research 23:215-224.
Davis, E. A., and L. F. DeBano. 1986. Nitrate increases in soil water following conversion of chaparral to grass. Biogeochemistry 2:53-65.
DeBano, L. F., J. J. Brejda, and J. H. Brock. 1984. Enhancement of riparian vegetation following shrub control in Arizona chaparral. Journal of Soil and Water Conservation 39:317-320.
DeBano, L. F., and C. E. Conrad. 1978. The effect of fire on nutrients in a chaparral ecosystem. Ecology 59:489–497.
de Koff, J. P., R. C. Graham, K. R. Hubbert, and P. M. Wohlgemuth. 2006. Prefire and postfire erosion of soil nutrients within a chaparral watershed. Soil Science 171.12:915-928.
Del Galdo, I., W. C. Oechel, and M. F. Cotrufo. 2006. Effects of past, present and future atmospheric CO2 concentrations on soil organic matter dynamics in a chaparral ecosystem. Soil Biology and Biochemistry 38:3235-3244.
Delwiche, C. C., P. J. Zinke, and C. M. Johnson. 1965. Nitrogen fixation by ceanothus. Plant Physiology 40:1045-1047.
Diaz, S., J. Kattge, J. H. C. Cornelissen, I. J. Wright, S. Lavorel, S. Dray, B. Reu, M. Kleyer, C. Wirth, I. C. Prentice, E. Garnier, G. Bonisch, M. Westoby, H. Poorter, P. B. Reich, A. T. Moles, J. Dickie, A. N. Gillison, A. E. Zanne, J. Chave, S. J. Wright, S. N. Sheremet'ev, H. Jactel, C. Baraloto, B. Cerabolini, S. Pierce, B. Shipley, D. Kirkup, F. Casanoves, J. S. Joswig, A. Gunther, V. Falczuk, N. Ruger, M. D. Mahecha, and L. D. Gorne. 2016. The global spectrum of plant form and function. Nature 529:167-173.
Dickens, S. J. M., and E. B. Allen. 2014. Exotic plant invasion alters chaparral ecosystem resistance and resilience pre- and post-wildfire. Biological Invasions 16:1119-1130.
Dietze, M. C., A. Sala, M. S. Carbone, C. I. Czimczik, J. A. Mantooth, A. D. Richardson, and R. Vargas. 2014. Nonstructural carbon in woody plants. Annual Review of Plant Biology 65:667-687.
Drenovsky, R. E., C. E. Koehler, K. Skelly, and J. H. Richards. 2013. Potential and realized nutrient resorption in serpentine and non-serpentine chaparral shrubs and trees. Oecologia 171:39-50.
Dunn, P. H., S. C. Barro, W. G. Wells II, M. A. Poth, P.M. Wohlgemuth, and C. G. Colver. 1988. The San Dimas experimental forest: 50 years of research. General Technical Report PSW-104. Pacific Southwest Region, USDA Forest Service, Berkeley, California, USA.
Fenn, M. E., E. B. Allen, S. B. Weiss, S. Jovan, L. H. Geiser, G. S. Tonnesen, R. F. Johnson, L. E. Rao, B. S. Gimeno, F. Yuan, T. Meixner, and A. Bytnerowicz. 2010. Nitrogen critical loads and management alternatives for N-impacted ecosystems in California. Journal of Environmental Management 91:2404-2423.
Fenn, M. E., J. S. Baron, E. B. Allen, H. M. Rueth, K. R. Nydick, L. Geiser, W. D. Bowman, J. O. Sickman, T. Meixner, D. W. Johnson, and P. Neitlich. 2003. Ecological effects of nitrogen deposition in the western United States. Bioscience 53:404-420.
Fenn, M. E., M. A. Poth, J. D. Aber, J. S. Baron, B. T. Bormann, D. W. Johnson, A. D. Lemly, S. G. McNulty, D. E. Ryan, and R. Stottlemyer. 1998. Nitrogen excess in North American ecosystems: Predisposing factors, ecosystem responses, and management strategies. Ecological Applications 8:706–733.
Fenn, M. E., M. A. Poth, P. H. Dunn, and S. C. Barro. 1993. Microbial N and biomass, respiration, and N-mineralization in soils beneath 2 chaparral species along a fire-induced age gradient. Soil Biology and Biochemistry 25:457-466.
Fenn, M. E., M. A. Poth, and D. W. Johnson. 1996. Evidence for nitrogen saturation in the San Bernardino Mountains in southern California. Forest Ecology and Management 82:211-230.
Fierer, N., and J. P. Schimel. 2002. Effects of drying-rewetting frequency on soil carbon and nitrogen transformations. Soil Biology and Biochemistry 34:777-787.
Firestone, M. K., and E. A. Davidson. 1989. Microbiological basis of NO and N20 production and consumption in soil. Pages 7-21 in M. O. Andreae and D. S. Schimel. Exchange of trace gases between terrestrial ecosystems and the atmosphere. John Wiley and Sons, New York, New York, USA.
Forrester, J. W. 1961. Industrial dynamics. MIT Press, Cambridge, Massachusetts, USA.
Fortin, M. J., R. J. Olson, S. Ferson, L. Iverson, C. Hunsaker, G. Edwards, D. Levine, K. Butera, and V. Klemas. 2000. Issues related to the detection of boundaries. Landscape Ecology 15:453-466.
Franco-Vizcaino, E., and J. Sosa-Ramirez. 1997. Soil properties and nutrient relations in burned and unburned Mediterranean-climate shrublands of Baja California, Mexico. Acta Oecologica-International Journal of Ecology 18:503-517.
Franklin, J., A. D. Syphard, H. S. He, and D. J. Mladenoff. 2005. Altered fire regimes affect landscape patterns of plant succession in the foothills and mountains of southern California. Ecosystems 8:885-898.
Frazer, J. M., and S. D. Davis. 1988. Differential survival of chaparral seedlings during the first summer drought after wildfire. Oecologia 76:215-221.
Fuentes, D. A., J. A. Gamon, Y. F. Cheng, H. C. Claudio, H. L. Qiu, Z. Y. Mao, D. A. Sims, A. F. Rahman, W. Oechel, and H. Y. Luo. 2006. Mapping carbon and water vapor fluxes in a chaparral ecosystem using vegetation indices derived from AVIRIS. Remote Sensing of Environment 103:312-323.
Galloway, J. N., A. R. Townsend, J. W. Erisman, M. Bekunda, Z. C. Cai, J. R. Freney, L. A. Martinelli, S. P. Seitzinger, and M. A. Sutton. 2008. Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320:889-892.
Garcia, M., J. Gajardo, D. Riano, K. G. Zhao, P. Martin, and S. Ustin. 2015. Canopy clumping appraisal using terrestrial and airborne laser scanning. Remote Sensing of Environment 161:78-88.
Gardner, P. D., H. J. Cortner, and K. Widaman. 1987. The risk perceptions and policy response toward wildland fire hazards by urban home-owners. Landscape and Urban Planning 14:163-172.
Giessow, J., and P. H. Zedler. 1996. The effects of fire frequency and firebreaks on the abundance and species richness of exotic species in coastal sage scrub. In J. E. Lovich, J. Randall, and M. D. Kelly, editors. Proceedings of the 1996 California Exotic Pest Council 1996 Symposium, Vol 2, San Diego, California 1996. Available from: http://www.cal-ipc.org/symposia/archive
Gill, A. M., and S. L. Stephens. 2009. Scientific and social challenges for the management of fire-prone wildland-urban interfaces. Environmental Research Letters 4:034014.
Gliksman, D., A. Rey, R. Seligmann, R. Dumbur, O. Sperling, Y. Navon, S. Haenel, P. De Angelis, J. A. Arnone, and J. M. Grunzweig. 2017. Biotic degradation at night, abiotic degradation at day: positive feedbacks on litter decomposition in drylands. Global Change Biology 23:1564-1574.
Gough, L. P., G. R Meadows, L. L. Jackson, and S. Dudka. 1989. Biogeochemistry of a highly serpentinized, chromite-rich ultramafic area, Tehama County, California. US Geological Survey Bulletin 1901. The US Government Printing Office, Washington D.C., USA.
Graham, R. C., L. M. Egerton-Warburton, P. F. Hendrix, P. J. Shouse, J. L. Johnson-Maynard, S. A. Quideau, P. D. Sternberg, J. A. Jobes, and J. M. Breiner. 2016. Wildfire effects on soils of a 55-year-old chaparral and pine biosequence. Soil Science Society of America Journal 80:376-394.
Gray, J. T., and W. H. Schlesinger. 1981. Nutrient cycling in Mediterranean-type ecosystems. Pages 259-285 in P. C. Miller, editor. Resource use by chaparral and matorral: a comparison of vegetation function in two Mediterranean-type ecosystems. Springer, Berlin, Germany.
Green, L. R. 1977. Fuel reduction without fire—current technology and ecosystem impact. Pages 163-171 in Proceedings of the symposium of environmental consequences of fire and fuel management in Mediterranean ecosystems. General Technical Report WO-3. USDA Forest Service, Washington D.C., USA.
Grünzweig, J. M., I. Gelfland, and D. Yakir. 2007. Biogeochemical factors contributing to enhanced carbon storage following afforestation of a semi-arid shrubland. Biogeosciences Discussions, European Geosciences Union 2111-2145.
Halsey, R. W., and J. E. Keeley. 2016. Conservation issues: California chaparral. In S. A. Elias, D. Alterton, J. K. Cochran, D. A. Dellasala, F. Funiciello, M. I. Goldstein, K. Lajtha, S. Marshall, and P. Mineau. Reference module in earth systems and environmental sciences. Elsevier Publications Inc., Amsterdam, Netherlands. https://doi.org/10.1016/B978-0-12-409548-9.09584-1.
Hamilton, J. G. 1997. Changing perceptions of pre-European grasslands in California. Madroño 44:311-333.
Hanan, E. J., C. M. D'Antonio, D. A. Roberts, and J. P. Schimel. 2016a. Factors regulating nitrogen retention during the early stages of recovery from fire in coastal chaparral ecosystems. Ecosystems 19:910-926.
Hanan, E. J., J. P. Schimel, K. Dowdy, and C. M. D’Antonio. 2016b. Effects of substrate supply, pH, and char on net nitrogen mineralization and nitrification along a wildfire-structured age gradient in chaparral. Soil Biology and Biochemistry 95:87-99.
Hanawalt, R. B., and R. H. Whittaker. 1976. Altitudnally coordinated patterns of soils and vegetation in San Jacinto Mountains, California. Soil Science 121:114-124.
Harrison, A. A. T., E. Small, and H. A. Mooney. 1971. Drought relationships and distribution of two Mediterranean-climate California plant communities. Ecology 52:869-875.
Harte, J. 2002. Toward a synthesis of the Newtonian and Darwinian worldviews. Physics Today 55:29-34.
Heckman, K., J. Campbell, H. Powers, B. Law, and C. Swanston. 2013. The influence of fire on the radiocarbon signature and character of soil organic matter in the Siskiyou National Forest, Oregon, USA. Fire Ecology 9:40-56.
Hibbert, A. R. 1971. Increases in streamflow after converting chaparral to grass. Water Resources Research 7:71-80.
Holling, C. S., and L. Gunderson. 2002. Resilience and adaptive cycles. Pages 25-62 in L. Gunderson and C. S. Holling, editors. Panarchy: understanding transformations in human and natural systems. Island Press, Washington D.C., USA.
Homyak, P. M., J. C. Blankinship, K. Marchus, D. M. Lucero, J. O. Sickman, and J. P. Schimel. 2016. Aridity and plant uptake interact to make dryland soils hotspots for nitric oxide (NO) emissions. Proceedings of the National Academy of Sciences of the United States of America 113:E2608-E2616.
Homyak, P. M., and J. O. Sickman. 2014. Influence of soil moisture on the seasonality of nitric oxide emissions from chaparral soils, Sierra Nevada, California, USA. Journal of Arid Environments 103:46-52.
Homyak, P. M., J. O. Sickman, A. E. Miller, J. M. Melack, T. Meixner, and J. P. Schimel. 2014. Assessing nitrogen-saturation in a seasonally dry chaparral watershed: limitations of traditional indicators of N-saturation. Ecosystems 17:1286-1305.
Huxman, T. E., K. A. Snyder, D. Tissue, A. J. Leffler, K. Ogle, W. T. Pockman, D. R. Sandquist, D. L. Potts, and S. Schwinning. 2004. Precipitation pulses and carbon fluxes in semiarid and arid ecosystems. Oecologia 141:254-268.
Ichii, K., W. L. Wang, H. Hashimoto, F. H. Yang, P. Votava, A. R. Michaelis, and R. R. Nemani. 2009. Refinement of rooting depths using satellite-based evapotranspiration seasonality for ecosystem modeling in California. Agricultural and Forest Meteorology 149:1907-1918.
Jarvis, P., A. Rey, C. Petsikos, L. Wingate, M. Rayment, J. Pereira, J. Banza, J. David, F. Miglietta, M. Borghetti, G. Manca, and R. Valentini. 2007. Drying and wetting of Mediterranean soils stimulates decomposition and carbon dioxide emission: the "Birch effect". Tree Physiology 27:929-940.
Jenerette, G. D., G. A. Barron-Gafford, A. J. Guswa, J. J. McDonnell, and J. C. Villegas. 2012. Organization of complexity in water limited ecohydrology. Ecohydrology 5:184-199.
Jenerette, G. D., and A. Chatterjee. 2012. Soil metabolic pulses: water, substrate, and biological regulation. Ecology 93:959-966.
Jenerette, G. D., R. L. Scott, and T. E. Huxman. 2008. Whole ecosystem metabolic pulses following precipitation events. Functional Ecology 22:924-930.
Jenerette, G. D., R. L. Scott, and A. R. Huete. 2010. Functional differences between summer and winter season rain assessed with MODIS-derived phenology in a semiarid region. Journal of Vegetation Science 21:16-30.
Keeley, J. E. 2005. Fire as a threat to biodiversity in fire-type shrublands. Pages 97-106 in B. E. Kus and J. L. Beyers, technical coordinators. Planning for biodiversity—bringing research and management together. General Technical Report PSW-GTR-195. US Forest Service, Pacific Southwest Research Station, Albany, California, USA.
Keeley, J. E., M. Baer-Keeley, and C. J. Fotheringham. 2005. Alien plant dynamics following fire in mediterranean-climate california shrublands. Ecological Applications 15:2109-2125.
Keeley, J. E., and T. J. Brennan. 2012. Fire-driven alien invasion in a fire-adapted ecosystem. Oecologia 169:1043-1052.
Keeley, J. E., and C. J. Fotheringham. 1997. Trace gas emissions and smoke-induced seed germination. Science 276:1248-1250.
Knipe, O. D. 1982. Angora goats for conversion of Arizona chaparral: early results. General Technical Report PSW-GTR-59. USDA Forest Service, Pacific Southwest Forest and Range Experiment Station, Berkeley, California, USA.
Knipe, O. D. 1983. Effects of Angora goat browsing on burned-over Arizona chaparral. Rangelands 5:252-255.
Kruckeberg, A. R. 1984. California serpentines, flora, vegetation, geology, soils, and management problems. University of California Press, Berkeley, California, USA.
Kummerow, J., J. V. Alexander, J. W. Neel, and K. Fishbeck. 1978. Symbiotic nitrogen-fixation in Ceanothus roots. American Journal of Botany 65:63-69.
Kustas, W. and M. Anderson. 2009. Advances in thermal infrared remote sensing for land surface modeling. Agricultural and Forest Meteorology 149:2071-2081.
Lambrinos, J. G. 2006. Spatially variable propagule pressure and herbivory influence invasion of chaparral shrubland by an exotic grass. Oecologia 147:327-334.
Lauvaux, C. A., C. N. Skinner, and A. H. Taylor. 2016. High severity fire and mixed conifer forest-chaparral dynamics in the southern Cascade Range, USA. Forest Ecology and Management 363:74–85
Lehmann, J., and Kleber, M. 2015. The contentious nature of soil organic matter. Nature 528:60-68.
Levelt, P. F., G. H. J. Van den Oord, M. R. Dobber, A. Malkki, H. Visser, J. de Vries, P. Stammes, J. O. V. Lundell, and H. Saari. 2006. The ozone monitoring instrument. Ieee Transactions on Geoscience and Remote Sensing 44:1093-1101.
Levin, S. A. 1998. Ecosystems and the biosphere as complex adaptive systems. Ecosystems 1:431-436.
Levin, S. A. 2000. Fragile dominion: complexity and the commons. Perseus Books, New York, New Yok, USA.
Li, L., M. L. McCormack, C. G. Ma, D. L. Kong, Q. Zhang, X. Y. Chen, H. Zeng, U. Niinemets, and D. L. Guo. 2015. Leaf economics and hydraulic traits are decoupled in five species-rich tropical-subtropical forests. Ecology Letters 18:899-906.
Li, X. Y., T. Meixner, J. O. Sickman, A. E. Miller, J. P. Schimel, and J. M. Melack. 2006. Decadal-scale dynamics of water, carbon and nitrogen in a California chaparral ecosystem: DAYCENT modeling results. Biogeochemistry 77:217-245.
Li, X. Y., A. E. Miller, T. Meixner, J. P. Schimel, J. M. Melack, and J. O. Sickman. 2010. Adding an empirical factor to better represent the rewetting pulse mechanism in a soil biogeochemical model. Geoderma 159:440-451.
Liang, L. L., D. A. Grantz, and G. D. Jenerette. 2016. Multivariate regulation of soil CO2 and N2O pulse emissions from agricultural soils. Global Change Biology 22:1286-1298.
Longstreth, D. T., and D. T. Patten. 1975. Conversion of chaparral to grass in Central Arizona: Effects on selected ions in watershed runoff. American Midland Naturalist 93:25-34.
Luo, H. Y., W. C. Oechel, S. J. Hastings, R. Zulueta, Y. H. Qian, and H. Kwon. 2007. Mature semiarid chaparral ecosystems can be a significant sink for atmospheric carbon dioxide. Global Change Biology 13:386-396.
Malamud, B. D., G. Morein, and D. L. Turcotte. 1998. Forest fires: an example of self-organized critical behavior. Science 281:1840-1842.
May, A. A., G. R. McMeeking, T. Lee, J. W. Taylor, J. S. Craven, I. Burling, A. P. Sullivan, S. Akagi, J. L. Collett, Jr., M. Flynn, H. Coe, S. P. Urbanski, J. H. Seinfeld, R. J. Yokelson, and S. M. Kreidenweis. 2014. Aerosol emissions from prescribed fires in the United States: a synthesis of laboratory and aircraft measurements. Journal of Geophysical Research: Atmospheres 119:11826-11849.
Mayberry, J. A. 2011. Community wildfire preparedness: balancing community safety and ecosystem sustainability in southern California chaparral. Humboldt State University.
Meixner, T., M. E. Fenn, P. Wohlgemuth, M. Oxford, and P. Riggan. 2006. N saturation symptoms in chaparral catchments are not reversed by prescribed fire. Environmental Science and Technology 40:2887-2894.
Meng, R., P. E. Dennison, C. M. D’Antonio, and M. A. Moritz. 2014. Remote sensing analysis of vegetation recovery following short-interval fires in Southern California shrublands. PLoS ONE 9:e110637.
Milchunas, D. G. 2006. Responses of plant communities to grazing in the southwestern United States. General Technical Report RMRS-GTR-169. USDA Forest Service, Rocky Mountain Research Station, Fort Collins, Colorado, USA.
Miller, A. E., J. P. Schimel, T. Meixner, J. O. Sickman, and J. M. Melack. 2005. Episodic rewetting enhances carbon and nitrogen release from chaparral soils. Soil Biology and Biochemistry 37:2195-2204.
Minnich, R. A., and C. J. Bahr. 1995. Wildland fire and chaparral succession along the California Baja-California boundary. International Journal of WIldland Fire 5:13-24.
Mooney, H. A., and P. W. Rundel. 1979. Nutrient relations of the evergreen shrub, Adenostoma fasciculatum, in the California chaparral. Botanical Gazette 140:109-113.
Moreno, J. M., I. Torres, B. Luna, W. C. Oechel, and J. E. Keeley. 2013. Changes in fire intensity have carry-over effects on plant responses after the next fire in southern California chaparral. Journal of Vegetation Science 24:395-404.
Nagle, T. A., and A. H. Taylor. 2005. Fire and persistence of montane chaparral in mixed conifer forest landscapes in the northern Sierra Nevada, Lake Tahoe Basin, California, USA. Journal of the Torrey Botanical Society 132:442-457.
Niu, G.-Y., C. Paniconi, P. A. Troch, R. L. Scott, M. Durcik, X. Zeng, T. Huxman, and D. C. Goodrich. 2014. An integrated modelling framework of catchment-scale ecohydrological processes: 1. Model description and tests over an energy-limited watershed. Ecohydrology 7:427-439.
Odum, H. T. 1983. Systems ecology: an introduction. John Wiley & Sons, New York, New York, USA.
Oikawa, P. Y., C. Ge, J. Wang, J. R. Eberwein, L. L. Liang, L. A. Allsman, D. A. Grantz, and G. D. Jenerette. 2015. Unusually high soil nitrogen oxide emissions influence air quality in a high-temperature agricultural region. Nature Communications 6:1-10.
Oikawa, P. Y., D. A. Grantz, A. Chatterjee, J. E. Eberwein, L. A. Allsman, and G. D. Jenerette. 2014. Unifying soil respiration pulses, inhibition, and temperature hysteresis through dynamics of labile soil carbon and O2. Journal of Geophysical Research: Biogeosciences 119:521-536.
Orme, A. R., and R. G. Bailey. 1970. The effect of vegetation conversion and flood discharge on stream channel geometry: a case of southern California watersheds. Proceedings of the American Association of Geographers 2:101-106.
Park, I. W., J. Hooper, J. M. Flegal, and G. D. Jenerette. 2018. Drivers of herbaceous cover throughout chaparral shrublands: Insights from a novel remote sensing method. Diversity and Distributions, in press.
Parker, V.T., R. B. Pratt, and J. E. Keeley. 2016. Chaparral. Pages 479-507 in H. Mooney and E. Zavaleta, editors. Ecosystems of California. University of California Press, Berkeley, California, USA.
Pasquini, S. C., and G. L. Vourlitis. 2010. Post-fire primary production and plant community dynamics in chaparral stands exposed to varying levels of nitrogen deposition. Journal of Arid Environments 74:310-314.
Pivovaroff, A. L., L. S. Santiago, G. L. Vourlitis, D. A. Grantz, and M. F. Allen. 2016. Plant hydraulic responses to long-term dry season nitrogen deposition alter drought tolerance in a Mediterranean-type ecosystem. Oecologia 181:721-731.
Pumo, D., F. Viola, and L. V. Noto. 2008. Ecohydrology in Mediterranean areas: a numerical model to describe growing seasons out of phase with precipitations. Hydrology and Earth System Sciences 12:303-316.
Quideau, S. A., R. C. Graham, O. A. Chadwick, and H. B. Wood. 1998. Organic carbon sequestration under chaparral and pine after four decades of soil development. Geoderma 83:227–242.
Rao, L. E., E. B. Allen, and T. Meixner. 2010. Risk-based determination of critical nitrogen deposition loads for fire spread in southern California deserts. Ecological Applications 20:1320-1335.
Rao, L. E., D. R. Parker, A. Bytnerowicz, and E. B. Allen. 2009. Nitrogen mineralization across an atmospheric nitrogen deposition gradient in Southern California deserts. Journal of Arid Environments 73:920-930.
Rasmussen, C., R. J. Southard, and W. R. Horwath. 2006. Mineral control of organic carbon mineralization in a range of temperate conifer forest soils. Global Change Biology 12:834-847.
Rice, R. M., E. S. Corbett, and R. G. Bailey. 1969. Soil Slips related to vegetation, topography, and soil in southern California. Water Resources Research 5:647-659.
Rich, L. R., and H. G. Reynolds. 1963. Grazing in relation to runoff and erosion on some chaparral watersheds of central Arizona. Society for Range Management 16:322-326.
Riggan, P. J., R. N. Lockwood, and E. N. Lopez. 1985. Deposition and processing of airborne nitrogen pollutants in Mediterranean-type ecosystems of southern California. Environmental Science and Technology 19:781-789.
Riggan, P. J., R. N. Lockwood, P. M. Jacks, C. G. Colver, F. Weirich, L. F. Debano, and J. A. Brass. 1994. Effects of fire severity on nitrate mobilization in watersheds subject to chronic atmospheric deposition. Environmental Science and Technology 28:369-375.
Riveros-Iregui, D. A., B. L. McGlynn, R. E. Emanuel, and H. E. Epstein. 2012. Complex terrain leads to bidirectional responses of soil respiration to inter-annual water availability. Global Change Biology 18:749-756.
Roberts, D. A., P. E. Dennison, K. L. Roth, K. Dudley, and G. Hulley. 2015. Relationships between dominant plant species, fractional cover and land surface temperature in a Mediterranean ecosystem. Remote Sensing of Environment 167:152-167.
Roberts, S. W., W. C. Oechel, P. J. Bryant, S. J. Hastings, J. Major, and V. Nosov. 1998. A field fumigation system for elevated carbon dioxide exposure in chaparral shrubs. Functional Ecology 12:708-719.
Rodrigue, C. M. 1993. Home with a view: chaparral fire hazard and the social geographies of risk and vulnerability. California Geographical Society 33:29-42.
Rossi, R. J., D. J. Bain, G. D. Jenerette, L. W. Clarke, and K. Wilson. 2015. Responses of roadside soil cation pools to vehicular emission deposition in southern California. Biogeochemistry 124:131-144.
Rowe, P. B. 1963. Streamflow increases after removing woodland-riparian vegetation from a southern california watershed. Journal of Forestry 61:365-370.
Rundel, P. W., E. A. Graham, M. F. Allen, J. C. Fisher, and T. C. Harmon. 2009. Environmental sensor networks in ecological research. New Phytologist 182:589-607.
Rundel, P. W., and D. J. Parsons. 1979. Structural changes in chamise (Adenostoma fasciculatum) along a fire-induced age gradient. Journal of Range Management 32:462-466.
Scheffer, M., S. Carpenter, J. A. Foley, C. Folke, and B. Walker. 2001. Catastrophic shifts in ecosystems. Nature 413:591-596.
Scheffer, M., and S. R. Carpenter. 2003. Catastrophic regime shifts in ecosystems: linking theory to observation. Trends in Ecology and Evolution 18:648-656.
Schlesinger, W. H., and E. S. Bernhardt. 2013. Biogeochemistry: an analysis of global change. Academic Press, Waltham, Massachusetts, USA.
Schmidt, M. W. I., M. S. Torn, S. Abiven, T. Dittmar, G. Guggenberger, I. A. Janssens, M. Kleber, I. Kogel-Knabner, J. Lehmann, D. A. C. Manning, P. Nannipieri, D. P. Rasse, S. Weiner, and S. E. Trumbore. 2011. Persistence of soil organic matter as an ecosystem property. Nature 478:49-56.
Scott, R. L., T. E. Huxman, G. A. Barron-Gafford, G. D. Jenerette, J. M. Young, and E. P. Hamerlynck. 2014. When vegetation change alters ecosystem water availability. Global Change Biology 20:2198-2210.
Scott, R. L., G. D. Jenerette, D. L. Potts, and T. E. Huxman. 2009. Effects of seasonal drought on net carbon dioxide exchange from a woody-plant-encroached semiarid grassland. Journal of Geophysical Research: Biogeosciences 114:G04004.
Serrano, L., J. Penuelas, and S. L. Ustin. 2002. Remote sensing of nitrogen and lignin in Mediterranean vegetation from AVIRIS data: decomposing biochemical from structural signals. Remote Sensing of Environment 81:355-364.
Severson, K. E., and L. F. Debano. 1991. Influence of Spanish goats on vegetation and soils in Arizona chaparral. Journal of Range Management 44:111-117.
Shen, W., G. D. Jenerette, D. Hui, R. P. Phillips, and H. Ren. 2008. Effects of changing precipitation regimes on dryland soil respiration and C pool dynamics at rainfall event, seasonal, and interannual scales. Journal of Geophysical Research: Biogeosciences 113:G03024.
Shen, W., G. D. Jenerette, D. Hui, and R. L. Scott. 2016. Precipitation legacy effects on dryland ecosystem carbon fluxes: direction, magnitude and biogeochemical carryovers. Biogeosciences 13:425-439.
Sidahmed, A. E., J. G. Morris, and S. R. Radosevich. 1981. Summer diet of Spanish goat grazing chaparral. Journal of Range Management 34:1981.
Sims, D. A., H. Y. Luo, S. Hastings, W. C. Oechel, A. F. Rahman, and J. A. Gamon. 2006. Parallel adjustments in vegetation greenness and ecosystem CO2 exchange in response to drought in a Southern California chaparral ecosystem. Remote Sensing of Environment 103:289-303.
Sims, D. A., A. F. Rahman, V. D. Cordova, B. Z. El-Masri, D. D. Baldocchi, P. V. Bolstad, L. B. Flanagan, A. H. Goldstein, D. Y. Hollinger, L. Misson, R. K. Monson, W. C. Oechel, H. P. Schmid, S. C. Wofsy, and L. Xu. 2008. A new model of gross primary productivity for North American ecosystems based solely on the enhanced vegetation index and land surface temperature from MODIS. Remote Sensing of Environment 112:1633-1646.
Six, J., R. T. Conant, E. A. Paul, and K. Paustian. 2002. Stabilization mechanisms of soil organic matter: implications for C-saturation of soils. Plant and Soil 241:155-176.
Smith, S. V, S. H. Bullock, A. Hinojosa-Corona, E. Franco-Vizcaino, M. Escoto-Rodriguez, T. G. Kretzschmar, L. M. Farfan, and J. M. Salazar-Cesena. 2007. Soil erosion and significance for carbon fluxes in a mountainous Mediterranean-climate watershed. Ecological Applications 17:1379-1387.
Sonnentag, O., K. Hufkens, C. Teshera-Sterne, A. M. Young, M. Friedl, B. H. Braswell, T. Milliman, J. O'Keefe, and A. D. Richardson. 2012. Digital repeat photography for phenological research in forest ecosystems. Agricultural and Forest Meteorology 152:159-177.
Sparks, S. R., W. C. Oechel, and Y. Mauffette. 1993. Photosynthate allocation patterns along a fire-induced age sequence in 2 shrub species from the California chaparral. International Journal of Wildland Fire 3:21-30.
Stewart, C. E., A. F. Plante, K. Paustian, R. T. Conant, and J. Six. 2008. Soil carbon saturation: linking concept and measurable carbon pools. Soil Science Society of America Journal 72:379-392.
Stylinski, C. D., J. A. Gamon, and W. C. Oechel. 2002. Seasonal patterns of reflectance indices, carotenoid pigments and photosynthesis of evergreen chaparral species. Oecologia 131:366-374.
Syphard, A. D., T. J. Brennan, and J. E. Keeley. 2014. The role of defensible space for residential structure protection during wildfires. International Journal of WIldland Fire 23:1165-1175.
Syphard, A. D., K. C. Clarke, and J. Franklin. 2007. Simulating fire frequency and urban growth in southern California coastal shrublands, USA. Landscape Ecology 22:431-445.
Syphard, A. D., H. M. Regan, J. Franklin, R. M. Swab, and T. C. Bonebrake. 2013. Does functional type vulnerability to multiple threats depend on spatial context in Mediterranean-climate ecosystems? Diversity and Distributions 19:1263-1274.
Tague, C., L. Seaby, and A. Hope. 2009. Modeling the eco-hydrologic response of a Mediterranean type ecosystem to the combined impacts of projected climate change and altered fire frequencies. Climatic Change 93:137-155.
Thanos, C. A., and P. W. Rundel. 1995. Fire-followers in chaparral: nitrogenous compounds trigger seed-germination. Journal of Ecology 83:207-216.
Treseder, K. K., L. M. Egerton-Warburton, M. F. Allen, Y. F. Cheng, and W. C. Oechel. 2003. Alteration of soil carbon pools and communities of mycorrhizal fungi in chaparral exposed to elevated carbon dioxide. Ecosystems 6:786-796.
Trueman, R. J., L. Taneva, M. A. Gonzalez-Meler, W. C. Oechel, and H. BassiriRad. 2009. Carbon losses in soils previously exposed to elevated atmospheric CO2 in a chaparral ecosystem: potential implications for a sustained biospheric C sink. Journal of Geochemical Exploration 102:142-148.
Ulery, A. L., R. C. Graham, O. A. Chadwick, and H. B. Wood. 1995. Decadal-scale changes in of soil carbon, nitrogen, and exchangable cations under chaparral and pine. Geoderma 65:121-134.
Vourlitis, G. L. 2012. Aboveground net primary production response of semi-arid shrublands to chronic experimental dry-season N input. Ecosphere 3(3):22.
Vourlitis, G. L. 2017. Chronic N enrichment and drought alter plant cover and community composition in a Mediterranean-type semi-arid shrubland. Oecologia 184:267-277.
Vourlitis, G. L., C. DeFotis, and W. Kristan. 2015. Effects of soil water content, temperature and experimental nitrogen deposition on nitric oxide (NO) efflux from semiarid shrubland soil. Journal of Arid Environments 117:67-74.
Vourlitis, G. L., and J. S. Fernandez. 2012. Changes in the soil, litter, and vegetation nitrogen and carbon concentrations of semiarid shrublands in response to chronic dry season nitrogen input. Journal of Arid Environments 82:115-122.
Vourlitis, G. L., and J. S. Fernandez. 2015. Carbon and nitrogen mineralization of semi-arid shrubland soils exposed to chronic nitrogen inputs and pulses of labile carbon and nitrogen. Journal of Arid Environments 122:37-45.
Vourlitis, G. L., and C. S. Hentz. 2016. Impacts of chronic N input on the carbon and nitrogen storage of a postfire Mediterranean-type shrubland. Journal of Geophysical Research: Biogeosciences 121:385-398.
Vourlitis, G. L., S. Pasquini, and G. Zorba. 2007a. Plant and soil N response of southern Californian semi-arid shrublands after 1 year of experimental N deposition. Ecosystems 10:263-279.
Vourlitis, G. L., and S. C. Pasquini. 2009. Experimental dry-season N deposition alters species composition in southern Californian mediterranean-type shrublands. Ecology 90:2183-2189.
Vourlitis, G. L., S. C. Pasquini, and R. Mustard. 2009. Effects of dry-season N input on the productivity and N storage of Mediterranean-type shrublands. Ecosystems 12:473-488.
Vourlitis, G. L., G. Zorba, S. C. Pasquini, and R. Mustard. 2007b. Chronic nitrogen deposition enhances nitrogen mineralization potential of semiarid shrubland soils. Soil Science Society of America Journal 71:836-842.
Warrick, J. A., and D. M. Rubin. 2007. Suspended-sediment rating curve response to urbanization and wildfire, Santa Ana River, California. Journal of Geophysical Research 112:F02018.
Westoby, M. 1998. A leaf-height-seed (LHS) plant ecology strategy scheme. Plant and Soil 199:213-227.
White, M. D., and K. A. Greer. 2006. The effects of watershed urbanization on the stream hydrology and riparian vegetation of Los Peñasquitos Creek, California. Landscape and Urban Planning 74:125-138.
Williamson, T. N., R. C. Graham, and P. J. Shouse. 2004a. Effects of a chaparral-to-grass conversion on soil physical and hydrologic properties after four decades. Geoderma 123:99-114.
Williamson, T. N., B. D. Newman, R. C. Graham, and P. J. Shouse. 2004b. Regolith water in zero-order chaparral and perennial grass watersheds four decades after vegetation conversion. Vadose Zone Journal 3:1007-1016.
Wolkovich, E. M., D. A. Lipson, R. A. Virginia, K. L. Cottingham, and D. T. Bolger. 2010. Grass invasion causes rapid increases in ecosystem carbon storage in a semiarid shrubland. Global Change Biology 16:1351-1365.
Wright, I. J., P. B. Reich, M. Westoby, D. D. Ackerly, Z. Baruch, F. Bongers, J. Cavender-Bares, T. Chapin, J. H. C. Cornelissen, M. Diemer, J. Flexas, E. Garnier, P. K. Groom, J. Gulias, K. Hikosaka, B. B. Lamont, T. Lee, W. Lee, C. Lusk, J. J. Midgley, M. L. Navas, U. Niinemets, J. Oleksyn, N. Osada, H. Poorter, P. Poot, L. Prior, V. I. Pyankov, C. Roumet, S. C. Thomas, M. G. Tjoelker, E. J. Veneklaas, and R. Villar. 2004. The worldwide leaf economics spectrum. Nature 428:821-827.
Wu, J. G., and J. L. David. 2002. A spatially explicit hierarchical approach to modeling complex ecological systems: theory and applications. Ecological Modelling 153:7-26.
Wu, J. G., and O. L. Loucks. 1995. From balance of nature to hierarchical patch dynamics: a paradigm shift in ecology. Quarterly Review of Biology 70:439-466.
Zink, T. A., M. F. Allen, B. Heinde-Tenhunen, and E. B. Allen. 1995. The effect of a disturbance corridor on an ecological reserve. Restoration Ecology 3:304-310.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Jenerette, G.D., Park, I.W., Andrews, H.M., Eberwein, J.R. (2018). Biogeochemical Cycling of Carbon and Nitrogen in Chaparral Dominated Ecosystems. In: Underwood, E., Safford, H., Molinari, N., Keeley, J. (eds) Valuing Chaparral. Springer Series on Environmental Management. Springer, Cham. https://doi.org/10.1007/978-3-319-68303-4_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-68303-4_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-68302-7
Online ISBN: 978-3-319-68303-4
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)