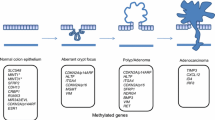
Abstract
Colorectal polyps are small clumps of cells and they are classified on the basis of their histological characteristics. Until recently, they have been divided into two major groups: neoplastic and non-neoplastic mucosal polyps. Adenomatous polyps contain epithelial neoplasia and their size and histology correlate with the risk of progression to carcinoma. Non-neoplastic polyps, instead, can be divided into several distinct and unrelated categories including hyperplastic, mucosal, juvenile, Peutz–Jeghers, and inflammatory. Adenomas are recognized as the precursor lesions for colorectal carcinoma and recently also some hyperplastic lesions, with serrated morphology, have been reported to display a significant risk of neoplastic progression through the so-called serrated pathway.
The sequence adenoma-carcinoma is caused by different molecular pathways. The most frequent are those of chromosomal instability pathway; the microsatellite instability pathway; the serrated pathway. Molecular complexity can explain the morphological heterogeneity and the timing of neoplastic progression.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1.1 Introduction
Intestinal polyps are projections from the mucosal surface that bulge into the visceral lumen and they are classified on the basis of their clinico-pathological qualities (i.e., neoplastic versus non-neoplastic) (Table 1.1) and/or their histopathological characteristics (Table 1.2). Adenomas are recognized as the precursor lesions for colorectal carcinoma [1]. Endoscopically, any superficial intestinal lesion may be described as follows: polypoid type (pedunculated, sessile, semi-pedunculated lesions), non-polypoid type (slightly elevated, flat, slightly depressed lesions) [2, 3].
Recently, it has been recognized that some hyperplastic lesions, with serrated morphology, can exhibit a significant risk of neoplastic progression, through the so-called serrated pathway [1].
1.2 Histological features of adenoma
1.2.1 Adenoma
Adenomatous polyps contain epithelial neoplasia account for approximately 10% of polyps [2, 4].
From a histological point of view, three types of adenomas are defined: tubular adenomas, villous adenomas, tubulovillous adenomas depending on their predominant glandular pattern [5]. The difference between tubular and tubulovillous adenomas depends on the percentage of volume of adenoma that is villous (20% in tubulovillous, 80% in villous) [2]. Tubular adenomas are small and present mild dysplasia whereas large adenomas often exhibit villous architecture and are associated with more severe degree of dysplasia [5]. The highest morbidity and mortality rates are associated with villous adenomas.
1.3 Adenoma Size
Adenomas can be classified by size in three classes: diminutive (1–5 mm diameter); small [6, 9], to and large (more than 10 mm) [4]. Size and histology of adenomas correlate with the risk of progression to carcinoma.
Adenomas of 10 mm of diameter or more are considered advanced, instead adenomas which are less than 1 cm are considered advanced when contain more at least 25% villous features, high grade dysplasia or carcinoma [4]. Diminutive polyps are commonly encountered during endoscopy and less than 1% of them are villous or contain a focus of high grade dysplasia.
1.4 Adenoma Carcinoma Sequence
Molecular studies have reported that several signaling pathways are involved in the carcinogenesis of Colorectal Cancer (CRC) [6, 7]. Molecular complexity can explain the morphological heterogeneity of CRC, in terms of site, grade, and type of the tumor [6, 8, 9]. Studies have centered on genetic changes of the three main categories of genes: (1) Tumor suppressor genes (TSG), such as APC, DCC, TP53, SMAD2, SMAD4, and p16INK4α; (2) protooncogenes, such as K-ras, N-ras, and (3) DNA repair genes, such as MMR and MUTYH [10,11,12]. The first elucidated is suppressor or chromosomal instability, the classical pathway recognized in the Fearon and Vogelstein genetic model [6, 10, 13]. It includes FAP tumors and 80% of sporadic colorectal carcinomas. This model proposes that mutations in concrete genes, in particular TSGs, cause the histopathological sequence of the progression of CRC [10]. Changes start with the mutation or loss of APC gene, followed by KRAS mutations, TP53 and DCC mutations [10]. 70–80% of colorectal tumors present APC gene mutations, instead K-ras mutations are found in 40% of tumors [10, 14,15,16]. The Fearon–Vogelstein model is even now considered valid for illustrating the concept of multiple steps of tumor progression. The sequences of proposed changes are the result of a statistical analysis so not all individuals have to show all of the changes [10].
The second elucidated pathway is the mutating or microsatellite instability (MIN) pathway that is responsible for the development of hypermutating carcinomas [6]. It includes the Lynch syndrome tumors and approximately 15% of sporadic tumors [10]. Lynch syndrome is caused by a mutation in one of the genes encoding proteins involved in mismatch repair (MMR) [6, 17]. Mutation on MMR genes provoke genomic instability that leads to hypermutator phenotype known as microsatellite instability determining an accelerated progression to carcinoma [10, 18].
The third, alternative, mechanism of carcinogenesis has been described as an epigenetic process. A frequently encountered mechanism responsible for silencing of TSGs is the hypermethylation of gene promoter associated CpG Island (CIMP), involved in the regulation of transcription [6, 19]. These epigenetic changes create instability in the genes as a result of the inactivation of TGS or MSI or CIN repair genes. The causes that activate the hypermethylation process are not clear. Environmental factors that could be involved are lesion due to chemotherapeutic agents, the ingestion of folates but the reasons or genetics that regulate this phenomenon are not well known [10, 20].
1.5 Serrated Lesions
Serrated polyps are defined as epithelial lesions with a serrated or “saw-toothed appearance” on histologic section due to infolding of crypt epithelium [21]. Serrated lesions vary from hyperplastic polyps (HP), sessile serrated adenomas (SSAs), dysplastic serrated polyps (traditional serrated polyps), or mixed lesion/polyps [2, 21].
1.5.1 Hyperplastic Polyps
HPs are highly prevalent diminutive sessile polyps that occur in the distal part of the colon and rectum. HPs are also the most frequent serrated polyps (80–90%) and they represent the 29–40% of all the polyps [22]. They usually have a diameter less than 5 mm and microscopically they exhibit distinct surface patterns. They show elongated crypts with serrated architecture in the upper half part of crypts or sometimes in the upper third and on the surface of the crypts leading to an irregular distension and a serration in the upper half of crypts [1, 21]. These surface patterns correspond to the histological subtypes: microvesicular serrated polyps (MVSPs), goblet cell serrated polyps (GCSPs), and mucin poor type (very rare) [1, 21]. Nuclei, in cells of the basal part are small, uniform, and basally oriented and nuclei in the upper part of the crypts are not crowed and there is no hyperchromasia [1, 2].
There is no cytologic dysplasia or intraepithelial neoplasia, no structural or architectural changes [1]. The two different histological subtypes differ also in molecular profiles. MVSPs show BRAF mutation and an increased level of susceptibility to aberrant methylation at promoter regions. GCSPs show KRAS mutation which is not shown in SSAs but there is some evidence that large GCSPs can progress to SSAs. So classifying HPs has no clinical importance [21].
Recently, it has been discovered that HPs could possess some malignant potential in the setting of hyperplasic polyposis syndrome. Patients presenting hyperplastic polyposis syndrome present: five or more serrated polyps proximal to the sigmoid colon with two or more larger than 10 mm in diameter; a total of more than 20 polyps, or a serrated polyp proximal to the sigmoid colon and a first-degree relative with the syndrome [23, 24]. The risk of malignant progression for most of the small distally located HPs in the colorectum is very low [1]. Polyps larger than 10 mm, instead, should be removed because some studies demonstrated that they could have malignant potential [1].
1.5.2 Sessile Serrated Adenomas
Sessile serrated polyps are heterogeneous lesions usually found in proximal colon (75%). They are less common than HP and have been suggested to account around 15–25% of all serrated polyps and 1.7–9% of all polyps [22]. SSAs are characterized by hyperserrations with rectangular dilatation of the whole crypts with or without the presence of mucus, T and L branching at the crypt base, and pseudoinvasion into the mucosal layer. Distortion in the bases of the colonic crypts is often present with an increased number of goblet cells, slightly enlarged vesicular nuclei with prominent nucleoli, and proliferation zone in the middle third of the crypts [1, 2, 4].
SSAs often produce excessive extracellular mucin, and stain positively for MUC5AC and MUC2 which are present in the surface of the polyp [22].
Endoscopically, SSAs appear as flat, sessile, or slightly elevated lesions, malleable, and often covered by a thin layer of yellowish mucus and usually they are larger than 5 mm in diameter [1, 21].
Their surface is generally smooth or granular and often their borders are irregular and poorly defined [21].
SSAs have two defining molecular genetic characteristics that indicate their relationship to MVSPs and to sporadic colon rectal cancer with high levels of microsatellite instability (MSI-H), namely BRAF-mut and high levels of CpG island methylation [21, 25, 26]. However, some contradictory studies highlight that they differ for anatomical distribution.
1.5.3 Dysplastic Serrated Polyps
Dysplastic serrated polyps are less common than conventional adenomas or HP, accounting for only 1–2% of all polyps [21]. Dysplastic serrated polyps show neoplastic crypts with serrated structures [2].
They share histological features as serrated gland component and the presence of eosinophilic dysplastic epithelium [2, 21]. Dysplastic serrated polyps can also be divided into two categories: SSA with dysplasia (SSAD) and traditional serrated adenoma (TSA). SSADs exhibit inverted T L shaped crypt bases, crypt branching and dilatation, presence of mature goblet cells in the crypt base or they show SSA characteristic next to an area of serrated or conventional dysplasia [21]. TSAs, instead, resemble conventional adenomas [21]. TSAs usually locate in the distal colon, and present a cytoplasmic eosinophilia and tubulovillous architectures [21].
Most SSADs present BRAF mutation. TSAs, instead, are more frequently associated with KRAS-mutation and likely give rise to microsatellite stable (MSS) cancer through hypermethylation of DNA repair gene MGMT [22]. Dysplasia is found in SSAs that present hyper-methylation of various genes (CIMP) including p16 and MLH1 resulting in early invasive cancer, more aggressive than conventional adenoma of proximal colon [22].
Several studies have put light on the fact that SSADs are at greater risk of progression to colorectal cancer than TSAs, marking the necessity of an adequate surveillance [21]. Dysplasia is a necessary step for progression to malignancy [21]. High grade dysplasia or intramucosal carcinomas have been found in KRAS-mut TSAs [21, 27]. The combination of serrated pathway features like CpG island methylation and the characteristics of the conventional pathway like chromosomal deletion and p53 mutation have been called “fusion pathway polyps” [21, 26, 28].
1.5.4 Mixed Polyps
The World Health Organization defined mixed polyps lesions which have a combination of hyperplastic, classical adenomatous or TSA or sessile serrated lesion components with different grades of intraepithelial neoplasia [1, 2]. The mixed polyps can contain components of SSA and TSA, SSA and conventional adenomas, TSA and conventional adenoma, and rarely HP and conventional adenoma [1]. The different histopathological type must be indicated in the diagnosis [2].
1.6 Mucosal Polyps
Mucosal polyps are small polyps where the submucosa has elevated the normal tissue. They have no clinical significance [3].
1.7 Juvenile Polyps
Juvenile polyps are solitary, spherical polyps that usually are located in the rectum of children [2, 3]. They show an excess of lamina propria and have cystic dilated glands [2]. In juvenile polyps, neoplasia is rare but they often show hyperplasia [2]. They are also known as retention polyps for the distended, mucus-filled glands, inflammatory cells, and edematous lamina propria [3, 29].
1.8 Peutz-Jeghers’s polyps
Peutz–Jeghers polyps are usually present in Peutz–Jeghers syndrome and rarely as single polyps [2]. Polyps have a very organized structure consisting in a central core smooth muscle with conspicuous branching covered by hyperplastic colorectal mucosa [2].
Differently from juvenile polyps, in Peutz–Jeghers polyps the lamina propria is normal and the abnormal muscle tissue confer the characteristic architecture of the lesion [3].
1.9 Inflammatory Polyps
Inflammatory polyps are associated with inflammatory bowel disease, diverticulosis, and mucosal prolapse [2]. Inflammatory polyps are thought to originate after mucosal inflammation with full thickness ulceration and tissue regeneration [30]. Inflammatory polyps might represent island of inflamed edematous mucosa with granulation tissue in the middle of mucosal ulceration [30]. These polyps have no intrinsic neoplastic potential, but they often appear in diseased colons that are at high risk to develop colon cancer [30].
1.10 Malignant Polyps
Malignant polyps are adenomatous growth containing transformed cells that have invaded the submucosa [31]. Such malignant polyps are found in 0.2–9% of endoscopically removed adenomatous polyps [32].
Higher rates of malignancy have been reported in villous adenomas (10–18%) compared with tubulovillous (6–8%) and tubular (2–3%) types [33].
Endoscopically, the presence of depressed ulceration, irregular contour, deformity, a short and immobile stalk, and the inability to elevate sessile polyp upon submucosa bleb formation must put in alert endoscopist [31].
The diagnosis of malignancy is ultimately histological [31]. There are several histological features which have been suggested to associate with higher probability of residual disease or recurrent carcinoma [32, 33]. The diagnosis and the oncological risk are defined by these elements: histological grade, lymphovascular invasion, tumor budding, margin of resection, and microstadiation [33]. There are four different levels of tumor differentiation from G1 (well differentiated) to G4 (undifferentiated). For therapeutic purposes, these levels are further divided into low-grade malignant polyps (G1 and G2) and high-grade malignant polyps (G3 and G4) [34]. Lymphatic invasion by cancer is defined as tumor cells visible within a true endothelial channel in absence of erythrocytes [33]. CD31 or CD34 marker could help in assessing vascular invasion especially if there are fixation artifacts in paraffin sections [33]. Tumor budding describes the presence of isolated single cancer cells or small cluster of cancer cells (less than 5 cells) at the advancing edge of the tumor [33, 34]. Tumors are positive for budding if they present five or more buds per 20 power fields [33]. There is increasing evidence that the presence of tumor budding reflects clinical aggressiveness of colon cancer and it is associated with lymph node metastasis and other adverse outcomes [33, 35]. Ensuring a histologically assessed resection margin free of cancer is very important. Cancer cells present near (less than a 1 mm) the resection margin or within diathermocoagulation line on histological examination increase the risk for an adverse outcome [33, 34]. Finally, microstadiation permits to recognize lesions of different metastatic potential. Histologically, polyps are classified by different factors but probably the most important characteristic is the depth of invasion [35]. There are two schemes to evaluate tumor invasion: Haggitt and Kikuchi scheme. Haggitt classification system for pedunculated and sessile polyps is an important prognostic factor. Pedunculated polyps can be classified as level 0–4.
-
Level 0 suggests that cancer cells are restricted to the mucosa and do not enter the muscularis mucosae [35, 36].
-
Levels 1 indicates cancer cells invading into the submucosa but limited to the head of the polyps [33, 35, 36].
-
Level 2 is present when carcinoma invades the level of the neck (the junction of the head and stalk) [33, 35, 36].
-
Level 3 signifies that cancer cells invade any part of the stalk [33, 35, 36].
-
Level 4 indicates that cancer cells invade into the submucosa of the bowel wall below the stalk but above the muscularis propria of the polyps [33, 35, 36].
Sessile adenomas with any degree of invasion that give rise to invasive cancer are defined as level 4 [33, 35, 36].
The submucosal invasion of adenocarcinoma in sessile polyps was further classified by Kudo [37] and Kikuchi [38]. They classified submucosal invasion into three levels: Sm1 in case of invasion of the upper third of submucosa; Sm2 invasion into the middle third of the mucosa; Sm3 invasion into the lower third of the mucosa [35]. Kikuchi and colleagues further modified the classification deepening Sm1 classification: SM1a less than a quarter of the width of the tumor invading the submucosa; SM1b between a quarter and a half of the width of the tumor invading the submucosa; SM1c more than a half of the width of the tumor invading the submucosa [33]. This classification is difficult to use if histological samples do not include a significant portion of the submucosa or some of the muscularis propria [35] (Fig. 1.1). On the basis of these criteria it is possible to differentiate malignant polyps at high or low grade of metastasis risk (Table 1.3).
(a) Macroscopic appearance of tubular adenoma; (b) macroscopic appearance of polypoid sessile adenocarcinoma in tubular adenoma; (c) macroscopic appearance of pedunculated lipomatous polypoid lesion with lobulated yellow cut surface; (d) macroscopic appearance of residual mucosa after polyp endoscopic removal; (e) histological features of tubulovillous pedunculated adenoma; (f) low-grade epithelial dysplasia in adenomatous polyp; (g) malignant polyps: adenocarcinoma in tubular adenoma. The malignant neoplastic proliferation invades the upper third of submucosa; (h) micrometric evaluation of distance of neoplastic cells from the resection margin
References
Aust DE, Baretton GB, Members of the Working Group GI-pathology of the German Society of Pathology. Serrated polyps of the colon and rectum (hyperplastic polyps, sessile serrated adenomas, traditional serrated adenomas, and mixed polyps)-proposal for diagnostic criteria. Virchows Arch. 2010;457(3):291–7.
Segnan N, Patnick J, von Karsa L, editors. European Guidelines for quality assurance in colorectal cancer screening and diagnosis. 1st ed. Luxembourg, Pubblication Office of the European Union; 2010. p. 386.
The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon: November 30 to December 1, 2002. Gastrointest Endosc. 2003;58(6 Suppl):S3–43. Review.
Strum WB. Colorectal adenomas. N Engl J Med. 2016;374(11):1065–75.
Konishi F, Morson BC. Pathology of colorectal adenomas: a colonoscopic survey. J Clin Pathol. 1989;35:830.
Bosman F, Yan P. Molecular pathology of colorectal cancer. Pol J Pathol. 2014;65(4):257–66.
Fearon ER, Vogelstein B. A genetic model for colorectal carcinogenesis. Cell. 1996;87:159–70.
De Sousa E Melo F, Wang X, Jansen M, et al. Poor-prognosis colon cancer is defined by a molecularly distinct subtype and develops from serrated precursor lesions. Nat Med. 2013;19:614–8.
Budinska E, Popovici V, Tejpar S, et al. Gene expression patterns unveil a new level of molecular heterogeneity in colorectal cancer. J Pathol. 2013;231:63–76.
Arvelo F, Sojo F, Cotte C. Biology of colorectal cancer. Ecancermedicalscience. 2015;9:520.
Worthley DL, Leggett BA. Colorectal cancer: molecular features and clinical opportunities. Clin Biochem Rev. 2010;31(2):31–8.
Fleming NI, Jorissen RN, Mouradov D, et al. SMAD2, SMAD3 and SMAD4 mutations in colorectal cancer. Crit Rev Oncol Hematol. 2011;79:1–16.
Fearon ER, Vogelstein B. A genetic model for colorectal tumorigenesis. Cell. 1990;61(5):759–67.
Laurent-Puig P, Agostini J, Maley K. Colorectal oncogenesis bull. Cancer. 2010;97:13.
Arrington AK, Heinrich EL, Lee W, Duldulao M, Patel S, Sanchez J, Garcia-Aguilar J, Kim J. Prognostic and predictive roles of KRAS mutation in colorectal cancer. Int J Mol Sci. 2012;13(10):12153–68.
Powell SM, Zilz N, Beazer-Barclay Y, et al. APC mutations occur early during colorectal tumorigenesis. Nature. 1992;359:235–7.
Nyström-Lahti M, Parsons R, Sistonen P, et al. Mismatch repair genes on chromosomes 2p and 3p account for a major share of hereditary nonpolyposis colorectal cancer families evaluable by linkage. Am J Hum Genet. 1994;55:659–65.
Loeb LA. Cancer cells exhibit a mutator phenotype. Adv Cancer Res. 1998;72:25–56.
Cunningham JM, Christensen ER, Tester DJ, et al. Hypermethylation of the hMLH1 promoter in colon cancer with microsatellite instability. Cancer Res. 1998;58:3455–60.
Beggs AD, et al. Whole-genome methylation analysis of benign and malignant colorectal tumours. J Pathol. 2013;229(5):697–704.
Huang CS, Farraye FA, Yang S, O’Brien MJ. The clinical significance of serrated polyps. Am J Gastroenterol. 2011;106(2):229–40.
Okamoto K, Kitamura S, Kimura T, Nakagawa T, Sogabe M, Miyamoto H, Muguruma N, Takayama T. Clinicopathological characteristics of serrated polyps as precursors to colorectal cancer: current status and management. J Gastroenterol Hepatol. 2017;32(2):358–67.
Spring KJ, Zhao ZZ, Karamatic R, Walsh MD, Whitehall VL, Pike T, Simms LA, Young J, James M, Montgomery GW, Appleyard M, Hewett D, Togashi K, Jass JR, Leggett BA. High prevalence of sessile serrated adenomas with BRAF mutations: a prospective study of patients undergoing colonoscopy. Gastroenterology. 2006;131(5):1400–7.
Leggett B, Whitehall V. Role of the serrated pathway in colorectal cancer pathogenesis. Gastroenterology. 2010;138(6):2088–100.
Jass JR. Serrated adenoma of the colorectum and the DNA-methylator phenotype. Nat Clin Pract Oncol. 2005;2:398–405.
Snover DC. Update on the serrated pathway to colorectal carcinoma. Hum Pathol. 2011;42:1–10.
Kim KM, Lee EJ, Kim YH, Chang DK, Odze RD. KRAS mutations in traditional serrated adenomas from Korea herald an aggressive phenotype. Am J Surg Pathol. 2010;34(5):667–75.
Jass JR, Baker K, Zlobec I, Higuchi T, Barker M, Buchanan D, Young J. Advanced colorectal polyps with the molecular and morphological features of serrated polyps and adenomas: concept of a ‘fusion’ pathway to colorectal cancer. Histopathology. 2006;49(2):121–31.
Calva D, Howe JR. Hamartomatous polyposis syndromes. Surg Clin North Am. 2008;88(4):779–817.
Syal G, Budhraja V. Recurrent obstructive giant inflammatory polyposis of the colon. ACG Case Rep J. 2016;3(4):e89.
Hassan C, Zullo A, Winn S, Eramo A, Tomao S, Rossini FP, Morini S. The colorectal malignant polyp: scoping a dilemma. Dig Liver Dis. 2007;39(1):92–100.
Netzer P, Forster C, Biral R, Ruchti C, Neuweiler J, Stauffer E, Schönegg R, Maurer C, Hüsler J, Halter F, Schmassmann A. Risk factor assessment of endoscopically removed malignant colorectal polyps. Gut. 1998;43(5):669–74.
Williams JG, Pullan RD, Hill J, Horgan PG, Salmo E, Buchanan GN, Rasheed S, McGee SG, Haboubi N, Association of Coloproctology of Great Britain and Ireland. Management of the malignant colorectal polyp: ACPGBI position statement. Colorectal Dis. 2013;15(Suppl 2):1–38.
Marini M, Lazzi S. Il polipo cancerizzato. Giorn Ital End Dig. 2014;37:263–8.
Aarons CB, Shanmugan S, Bleier JI. Management of malignant colon polyps: current status and controversies. World J Gastroenterol. 2014;20(43):16178–83.
Haggitt RC, Glotzbach RE, Soffer EE, Wruble LD. Prognostic factors in colorectal carcinomas arising in adenomas: implications for lesions removed by endoscopic polypectomy. Gastroenterology. 1985;89:328–36.
Kudo S. Endoscopic mucosal resection of flat and depressed types of early colorectal cancer. Endoscopy. 1993;25:455–61.
Kikuchi R, Takano M, Takagi K, Fujimoto N, Nozaki R, Fujiyoshi T, Uchida Y. Management of early invasive colorectal cancer. Risk of recurrence and clinical guidelines. Dis Colon Rectum. 1995;38:1286–95.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG
About this chapter
Cite this chapter
Cannizzaro, R., Magris, R., Maiero, S., Fornasarig, M., Canzonieri, V. (2018). Classification of Colon Polyps and Risk of Neoplastic Progression. In: Facciorusso, A., Muscatiello, N. (eds) Colon Polypectomy. Springer, Cham. https://doi.org/10.1007/978-3-319-59457-6_1
Download citation
DOI: https://doi.org/10.1007/978-3-319-59457-6_1
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-59456-9
Online ISBN: 978-3-319-59457-6
eBook Packages: MedicineMedicine (R0)