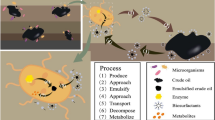
Abstract
Hydrocarbon contamination of soil is one of the major environmental problems today due to activities related to the petrochemical industry. Mechanical and chemical remediation and restoration to remove hydrocarbons from contaminated sites have limited effectiveness and costly. Bioremediation is the promising technology for the treatment of these contaminated sites since it is cost-effective and will lead to complete mineralization. Fungi and bacteria have been considered as highly effective in oil degradation. Several bacteria are even known to feed exclusively on hydrocarbons; Arthrobacter, Burkholderia, Mycobacterium, Pseudomonas, Sphingomonas, and Rhodococcus. Fungal genera, namely, Amorphoteca, Neosartorya, Talaromyces, and Graphium are proved to be the potential organisms for hydrocarbon degradation. Although laboratory experiments have indicated that the bacteria can ubiquitously degrade oil constituents, to date there is little convincing evidence that bioaugmentation (addition of more bacteria) significantly enhances the extent of oil biodegradation in soil. The potential benefits of using genetically modified bacteria represent a research frontier with significant results. However, many concerns are often raising due to the effectiveness of indigenous species, limited understanding of various phytoremediation mechanisms, including the regulation of enzyme systems that degrade pollutants. Thus, this chapter presents an updated overview of petroleum hydrocarbon degradation by microorganisms focusing biosurfactants and their mechanisms.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
9.1 Introduction
Oil hydrocarbon pollution originates from both natural and anthropogenic activities. Contamination due to exploration, draining off from drill sites, accidental spillage, refinery, and transportation has led to global environmental problems (Das and Chandran 2010). The physical and biological effects of oil in the aquatic environment include reduction of light transmission, reduction of dissolved oxygen, damage to water birds, and smothering of intertidal organisms. The toxic effects are exhibited mostly by the light portion (low boiling) of the oil due to the presence of poly aromatic hydrocarbon (PAH) (Atlas and Bartha 1992). The effects include cell damages and death of sensitive organisms and larval stages. The mitigation of oil pollution in sea, soil, and aquatic environment is a difficult task, and various efforts have been taken by the international community to curtail this problem.
In large-scale accidents, oil release into the environment harms the biological system due to the biomagnification of toxic compounds with toxic elements via food webs and food chains (Dillard et al. 1997; Head et al. 2006). The degradation of oil hydrocarbon in aquatic environment, sludge, soil, sediment, and costal ecosystem with natural phenomenon is great deal to overcome these types of problems arising due to the oil contamination. The xenobiotic nature of the oil products and its derivatives are high-risk factors of the environmental degradation due to the bioaccumulation (Head et al. 2006). Oil consists of different compositional ingredients with different chemical and physical properties (Hasanuzzaman et al. 2007). Once petroleum oil is spilled and leaked over the aquatic masses, it will disperse through the surface while releasing volatile compounds to atmosphere (Garrett et al. 2003).
Some hydrocarbons , such as PAH , are known as potent mutagens and carcinogens that pose serious human and environmental health risks. Therefore, the removal of hydrocarbons is essential to improve environmental health. Many different treatment techniques have been studied in order to restore and rehabilitate the soil. The main remediation methods practiced are containment, thermal desorption, incineration, and microbial degradation (Henner et al. 1997).
Cleanup technologies such as physical and chemical methods, volatilization, photooxidation, chemical oxidation, and bioaccumulation seem to be rarely successful on petroleum hydrocarbons due to the recalcitrant nature and not cost-effective when compared to microbial bioremediation (Prince 1997). Oil pollutants are slow in degradation under normal conditions due to the functional groups present and low water solubility (Desai et al. 2012; Hassanshahian et al. 2012). Therefore, it needs special specific conditions to enhance the degradation of particular oil pollutant at a considerable rate and has to reduce time taken to clean up the contaminated site. Degradation of oil depends on the nature of crude oil, composition, and genes responsible for the secretion of enzymes and ecological and environmental factors. Apart from that, efficiency of oil bioavailability is restricted by poor water solubility of oil contaminant (Banat et al. 2010). However, the mechanical removal of oily substances is very expensive and makes more harmful effects on animals and environment during the period of time (Urum et al. 2006). Recent studies have focused on bioremediation through the indigenous microorganisms in the oil-contaminated surroundings as a potential remediation measure. This chapter provides the background and updated information on microbial degradation of petroleum hydrocarbon contaminants towards the better understanding in bioremediation challenges.
9.2 Bioremediation of Oil Contaminants
Bacteria have long been considered as one of the predominant hydrocarbon-degrading agents found in the environment. The success of bioremediation technologies applied to hydrocarbon-polluted environments depends highly on the biodegrading capabilities of native microbial populations or exogenous microorganisms used as inoculants (Al-Wasify and Hamed 2014; Marchant and Banat 2012). Bioremediation is defined as the use of microorganisms in the biological system to detoxify or remove pollutants using their diverse metabolic capabilities. This process uses an evolving method for the removal and degradation of many environmental pollutants including the products of the petroleum industry (Tang et al. 2011).
Microbial biodegradation is cost-effective, and it serves as highly efficient alternative compared to many types of approaches (Koutinas et al. 2007). Enhancement of crude oil degradation with the use of microorganisms has been identified as a novel approach to overcome oil contamination (Banat et al. 2010). Therefore, many studies have focused on the enhancement of biodegradation of oil contaminants to recover the oil pollution through bioremediation process to clean up the contaminated sites not cleaning up (Soberón-Chávez and Maier 2011). On the other hand, only few studies have concentrated on the generation of the toxic compounds during the bioremediation process by microorganism in some special remediation systems such as salt marshes and estuarine pools (Middaugh et al. 1998; Shelton et al. 1999). The efficiency of microbial degradation has been revealed by with the aerobic degradation (Das and Chandran 2010). According to the studies carried out to reveal the biodegradation of oil, it has aimed to isolate and identify the bacteria and other organisms having highly adapted nature to the contaminated sites to degrade contaminants (Zajic et al. 1977). Scientists have observed and reported isolated organisms from study sites which were chemically and physically stable and their metabolic activities are optimum at the particular environment (Zhang et al. 2010).
Recent researches have paid their attention on natural, safe, cost-effective materials to treat oil-contaminated sites (Van Hamme et al. 2003). Simple oil contaminants with simple structures are processed with different metabolic pathways based on the unique enzyme systems present in microorganisms (Van Hamme et al. 2003). Therefore, they have chosen biosurfactant as the best potential method of remediation of oil spills (Wei et al. 2005). Development of the possible eco-friendly tool to handle the contaminated sites using efficient strategies and an applicable model system is important (Franzetti et al. 2010a). Therefore it has been discovered that the use of naturally occurring bacteria for removal of oil contaminants have built up the new interpretations for the oil recoveries with the production of biosurfactants (Ron and Rosenberg 2002).
Biodegradation of oil can be influenced by the microorganisms present in the natural environment as well as by introducing improved microbial cultures (Table 9.1). Oil spill bioremediation has two main approaches: (a) bioaugmentation, where known oil-degrading bacteria are added to supplement the existing microbial population, and (b) biostimulation , where the growth of indigenous oil degraders are stimulated by the addition of nutrients or other growth-limiting substrates with the supplement of enriching factors of medium (Ojo 2005). Oil-degrading microbial consortia are used as the improved cultures, and it consists with one or more microorganisms showing an ability to degrade oil at a considerable rate (Rosenberg and Ron 1999). Several studies have proven with the evidence that the use of microorganisms to oil recovery is a desirable alternative to remove the oily contaminants from the environment (Banat et al. 2000) (Table 1).
Microorganisms should possess special metabolic activities responsible for the production of metabolites to utilize the oily compounds. Therefore, specific biochemical reactions of microorganisms are needed for the expression of the production of metabolites for the degradation of total petroleum products (Atlas and Bartha 1992). It is very important to know that the reasonable performance of microorganism during biodegradation depends on the enzymatic activity and its mechanism (Obayori et al. 2009).
But if an unexpectable oil drainage occurred, the naturally occurring microbial consortium is unable to degrade the spilled oil totally at considerable rate, because an already existing small proportion of microorganism consortium may not be enough to degrade the bulk of oil contaminant and some of them may inactive due to the toxicity nature of oil to them (Shelton et al. 1999). Therefore, it is necessary to use the best biotechnological approach to overcome this type of problems before dispersing oil over the surface. To fulfill this purpose, selection of the best biotechnological solution is very important as an innovative technique .
Exxon Valdez and BP Deepwater Horizon oil spill were the two large-scale oil spills (Atlas and Hazen 2011). The experimental evidence suggests that those are the worst oil spills in US history with svere environmental impacts of oil contamination (Bragg et al. 1994). Because petroleum hydrocarbons consist in crude oil with diverse derivatives of both aromatic and aliphatic hydrocarbons, is easily escape into groundwater reservoirs and surrounding environment. Generally petroleum hydrocarbons naturally exist in all marine environments with the compatible nature of naturally occurring microorganisms. Those microorganisms are capable of utilizing those contaminants by using oil as an energy source (Garon et al. 2002).
9.3 Mechanism of Microbial-Mediated Petroleum Hydrocarbon Degradation
Basically bioremediation expresses the mean of transformation and mineralization of toxic compounds via the endogenous or exogenous process of microorganisms (Franzetti et al. 2010b). In endogenous activity, carbon source has to go inside before it undergoes to the transformation event. Compared to endogenous process , exogenous process occurred in external environment. Therefore, it is essential to bind with carbon and the microbial cell before utilizing it (Mulligan and Gibbs 2004). The ability to form biosurfactants is found in numerous bacterial and fungal species (Desai and Banat 1997), for instance, Pseudomonas sp., Rhodococcus sp., Acinetobacter sp., Bacillus sp., Achromobacter sp., Brevibacillus sp., Lysinibacillus sp., Alcaligenes sp., Candida sp., and Torulopsis sp. (Desai and Banat 1997). Biosurfactants enables them to grow on hydrophobic substrate. However, due to low solubility of petroleum hydrocarbon , the biodegradation of contaminant by microorganisms is highly restricted (Banat et al. 2010), but with the presence of extracellular biosurfactants, these compounds are solubilized, increasing its bioavailability to microorganisms by providing contaminant as an effective energy source for them (Joshi et al. 2008). Studies on biosurfactants showed the degradation of contaminant depends on the length and nature of arrangement of the carbon chain. Aliphatic moieties are easily degradable than the rest of aromatic carbon structures (Setti et al. 1993), because aromatic fraction is a heavily branched compound and it is difficult to degrade. Hence, aromatic fraction may persist in the environment for a longer period without any change and will pose a significant harmful effect on an environment (Hasanuzzaman et al. 2007).
Most of the recent studies were focused on the degradation of crude oil . Diverse groups of microorganisms are able to synthesize various types of secondary metabolites (Banat 1995), which have an ability to produce biosurfactant effects to degrade pollutants in an efficient way. Especially the biosurfactants are commonly employed in crude oil recovery and have been explained (Banat et al. 2000).
9.4 Structure of the Biosurfactant
Biosurfactants are the naturally occurring structurally diverse groups showing surface-active mechanisms with amphiphilic properties. It consists with both hydrophobic and hydrophilic moieties with surface-active compounds (Fig. 9.1). Most biosurfactants are anionic or neutral, but few are cationic due to the presence of amine group. Biosurfactants are amphipathic molecules which accumulate at interfaces, decrease interfacial tensions, and form aggregates such as micelles. However, biosurfactants show unique biochemical properties which are highly important in biological applications. Biosurfactants are easily accessible and acceptable for the environmental application, especially for both land and sea (Cameotra et al. 2010).
9.5 Classification of Biosurfactants
Biosurfactants are classified based on their chemical structure, composition, molecular weight, physicochemical properties, mode of action, and the microbial origin that produced them (Zinjarde and Pant 2002) (Table 9.2). Its hydrophobic fraction may consist with the saturated or unsaturated fatty acid, and hydrophilic fraction may consist with the amino acids , peptides, and saccharides. Based on its chemical composition and chemical behavior, it can be divided into three major classes as follows (Desai and Banat 1997):
-
1.
Glycolipids (lipopeptides, lipoproteins, and phospholipids)
-
2.
Polymeric biosurfactants
-
3.
Particulate biosurfactants
In some cases, biosurfactants can be categorized based on its molecular weight as low- and high-molecular-weighted polymers (Rosenberg and Ron 1999). Low-molecular-mass biosurfactants include glycolipids, phospholipids, and lipopeptides, whereas high-molecular-mass surfactants include amphipathic polysaccharides, lipopolysaccharides, proteins, lipoproteins, and complex mixtures of biopolymers. Compared to the high-molecular-mass biosurfactants, low-molecular-weight compounds are efficient in lowering the surface and interfacial tension of contaminant, whereas high-molecular-mass compounds are effective at stabilizing the oil in water emulsion (Calvo et al. 2009).
9.6 Properties of Biosurfactants: Natural Choice for Bioremediation with Biosurfactants
Biosurfactants have been identified as eco-friendly, cost-effective, easily degradable substrate produce by microorganisms as their secondary metabolite, and they are producing mixture of biopolymers (Rosenberg and Ron 1999). It aims to mineralize and/or biotransform toxic contaminants to nontoxic or low-toxic compounds while utilizing it by microorganisms (Sanscartier et al. 2009). Due to high demand for the biosurfactants, the attention has been paid on artificially synthesizing this eco-friendly natural compound (Atlas and Bartha 1992). However, it has shown that synthetic chemicals had severe adverse effects on an environment over the use of natural surfactants. When the biosurfactant is ready to activate on any hydrophobic substrate to degrade it, biosurfactant accumulates at the interface between fluid and solid with the effective reduction of surface and interfacial tension. Hence, it is allowing those two dissimilar phases to mix and interact with each other more easily (Soberón-Chávez and Maier 2011).
Measurement of biosurfactant activity has been done by the measurement of surface and interfacial tension, stabilization of emulsion, and hydrophilic-lipophilic balance (HLB) of the surfactants and contaminant (Oberbremer et al. 1990). Surface tension was measured at the air-water and oil/water interface with the use of tensiometer (Salamanca et al. 2001). The high molecular weight surfactants are less effective in reducing interfacial tension, but are important at coating the oil droplets and preventing their coalescence (Fig. 9.2). These are highly efficient emulsifiers that work at low concentrations (Ron and Rosenberg 2002). Emulsification activity is measured by emulsification assay. It is based on the ability of the surfactant to regenerate droplets in particular aqueous assay system (Kim et al. 2000). Biosurfactants can increase the bound substrates by desorbing them from surfaces or by increasing their apparent water solubility.
9.7 Relationship Between Surface Tension and Critical Micelle Concentration (CMC)
The efficiency and effectiveness of biosurfactant depends on the formation of critical micelle concentration of the surface-active compound with the collection of sufficient surface-active molecules on the substrate. At the concentration above CMC, biosurfactant molecules gathered to form micelles, vesicles, and lamellae on the substrate as a continuous bilayer (Whang et al. 2008). Formation of micelle enables to increase the solubility and bioavailability on hydrophobic contaminant to make easy degradation while reducing the surface tension (Desai and Banat 1997). However, an efficient biosurfactant has the lower CMC value; therefore, less amount of biosurfactant is required to decrease the surface tension (Nguyen et al. 2008) (Fig. 9.3). It has direct relationship with the formation of microemulsion layer (Desai and Banat 1997). That allows the contaminant to adjust to the hydrophobic environment leading the formation of stable liquid mixture of water and oily particles by the formation of droplets dispersed on the liquid phase (Soberón-Chávez and Maier 2011).
Relationship between surface tension and CMC is expressed in the semilogarithmic plot of the surface tension of a solution against the surfactant concentration as mentioned below (Fig. 9.4). HLB indicates the promotion of surfactant activity in water-in-oil or oil-in-water to emulsify the contaminant easier. If HLB value is less than 6, it is preferred to emulsify water-in-oil ; if the HLB value is in between 10–18, it favors the emulsification of contaminant in oil-in-water (Oberbremer et al. 1990).
In practical approaches, HLB value can be measured as follows:
HLB value = 20 * Mh/M.
Mh—molecular weight of the hydrophilic fraction
M—molecular weight of the whole molecule
HLB value of 0 corresponds to a completely lipophilic/hydrophobic molecule, and a value of 20 corresponds to a completely hydrophilic/lipophobic molecule. Therefore, HLB values of surfactants are useful in predicting the surfactant properties of the surface-active compounds.
If the two moieties of biosurfactant are not arranged in an equal way, this formula does not apply (Vollbrecht et al. 1999).
Ex-HLB value of glycolipid in the system containing water and hydrophobic phase consists with soybean and cyclohexane mixture. For this system, HLB value is measured as follows:
A = (HLBneeded – HLBB)/(HLBA – HLBB)
A = % of cyclohexane
HLBA—HLB of cyclohexane (−15)
HLBB—HLB of soybean (−6)
Using this formula, most stable emulsification index can be determined for the selected system.
9.8 Effect of pH and Temperature on Biosurfactant Stability
The activity of biosurfactant decreases at extreme pH values (Champion et al. 1995). However, biosurfactants are stable at high temperatures (Banat 1995). Biosurfactant was stable during exposure to high salinity (10 % NaCl), elevated temperatures (120 °C for 15 min), and within a wide pH range (4.0–10.0) (Shavandi et al. 2011). The activity of the biosurfactant was enhanced optimally at NaCl concentration of 5 %, pH of 8.0, and temperature of 40 °C. The biosurfactant retained 77 % of its original activity after 120 min of exposure to heat at a temperature of 100 °C (Ilori et al. 2005).
9.9 Mechanisms of Biosurfactants in Biodegradation
The use of biosurfactants to remove oil contaminants is a promising method that can improve the effectiveness of bioremediation in contaminated environments. They can enhance hydrocarbon bioremediation by two mechanisms. The first mechanism includes the induction of substrate bioavailability for microorganisms, while the other involves interaction with the cell surface which increases the hydrophobicity of the surface allowing hydrophobic substrates to associate more easily with bacterial cells (Mulligan and Gibbs 2004). By reducing surface and interfacial tensions, biosurfactants increase the surface areas of insoluble compounds leading to increased mobility and bioavailability of hydrocarbons. Addition of biosurfactants to the polluted site through the process of bioaugmentation is capable of degrading contaminants. This process is expected to enhance the hydrocarbon biodegradation by mobilization, solubilization, or emulsification of contaminants efficiently (Nguyen et al. 2008).
Usually microbial aerobic degradation of alkanes follows intracellular enzymatic pathway (Morgan and Watkinson 1990). Aerobic degradation is the most rapid and complete degradation of organic pollutants by microorganisms (Fig. 9.5). Organic pollutants including various oil pollutants and its derivatives are initially attacked by microorganisms through an oxidative process. Hereafter, activation and incorporation of pollutant into cell is essential before utilizing it. Those two processes are catalyzed by two enzymes named as oxygenase and peroxidase . Then intermediates are formed via the peripheral degradation pathway, Eg–TCA cycle (Fig. 9.6).
Instead of oxygenase degradation pathway being used for the degradation of oil derivatives, it is linked with the many other enzymatic pathways. The activity of cytochrome P450 alkane hydroxylase enzyme is the well-studied enzyme on this event (van Beilen and Funhoff 2007). Degradation depends on the change of length, and the enzyme system efficiency is more important to initiate the degradation (Table 9.3).
Apart from that, another enzyme system has been discussed by van Beilen and Funhoff (2005). The enzyme is di-iron methane monooxygenase showing membrane-bound copper-containing monooxygenase responsible for the degradation of oil with elementary constituent in toxic level (van Beilen and Funhoff 2007).
9.10 Attachment of Microorganism to Substrate as a Mechanism to Obtain the Degradation of Contaminant
In the latest literature, it has explained the interaction between microorganisms and hydrocarbon which is used to utilize as a substrate by microorganisms (Van Hamme et al. 2003). However, by modulating of cell surface, hydrophobicity may increase the surface permeability allowing microorganisms to make direct contact with the oil drops (Patist et al. 2000). However, researchers have reported the low hydrophobicity modulation by microorganism on cell surface permits their adhesion to micelles of emulsified oil. Based on their studies, pseudo-solubilization of substrate by microorganism was used to make direct contact of microbial cells with large oil droplets. Also it has been revealed that at the different growth stages of microorganisms, their capability of producing different secondary metabolites has an ability with changing hydrocarbon accession modes (Singh et al. 2006). An increment of bioavailability of oil to the bacterial has been exhibited by the biosurfactant-coated contaminant (Cameotra and Makkar 2004). This process is involving internalization of hydrocarbon inside the cell for subsequent degradation. It is similar in the process called active pinocytosis (Cameotra and Singh 2009).
9.11 Conclusions and Future Perspectives
Continuous release of oil pollutants through either natural or industrial processes is occurring as one of the leading environmental problems. Hence, the bioremediation as a green technology is an acceptable solution due to the cost and effort involved. Therefore, the use of biology systems is playing an important role in this novel strategy. Hence, currently the demand for biological system in various industries is steadily increasing. In this case, biosurfactants play an important role on oil hydrocarbon degradation. However, the commercial success of biosurfactant is still limited because it has to pay high production cost. It is necessary to find out more economically feasible methods to produce biosurfactant in large scale. Hence, future research must focus on producing low-cost biosurfactants and test. Application of biosurfactant and other microbial degradation methods is still in under experimental level. Therefore, the use of these experimental processes in the field scale is important in order to validate the laboratory results. Most of the studies carried out under in vitro condition are needed to move to field scale evaluating its effectiveness.
The development of microbial degradation of contaminant with improvements in genetic expression via the manipulation of gene expression is very important. Application of this technology benefits in large-scale application of microbial degradation and provides us with a cleaner environment in a safe way. The genetically modified (GM) bacteria give a novel approach in biotechnology. However, still there is a limited understanding on their enzyme regulation systems that degrade pollutants. Studies must focus on degrading both aliphatic and aromatic compounds effectively at the same time, and in this case, the biotechnology may play a rigorous role. Thus, the integration of biological engineering and biotechnology is possible and profoundly needed in order to achieve high yields and low costs on biosurfactant production. High-throughput analysis , predictive computational modeling, or simulation and experimental perturbation can be combined to generate new knowledge in order to design strategies for efficient reactions.
References
Al-Wasify RS, Hamed SR (2014) Bacterial biodegradation of crude oil using local isolates. Int J Bacteriol 2014:863272
Appanna VD, Finn H, Pierre MS (1995) Exocellular phosphatidylethanolamine production and multiple-metal tolerance in Pseudomonas fluorescens. FEMS Microbiol Lett 131:53–56
Atlas RM, Bartha R (1992) Hydrocarbon biodegradation and oil spill bioremediation. Adv Microb Ecol 12:287–338
Atlas RM, Hazen TC (2011) Oil biodegradation and bioremediation: a tale of the two worst spills in US history. Environ Sci Technol 45:6709–6715
Banat IM (1995) Biosurfactants production and possible uses in microbial enhanced oil recovery and oil pollution remediation: a review. Bioresour Technol 51:1–12
Banat IM, Makkar RS, Cameotra S (2000) Potential commercial applications of microbial surfactants. Appl Microbiol Biotechnol 53:495–508
Banat IM, Franzetti A, Gandolfi I, Bestetti G, Martinotti MG, Fracchia L, Smyth TJ, Marchant R (2010) Microbial biosurfactants production, applications and future potential. Appl Microbiol Biotechnol 87:427–444
Bozo-Hurtado L, Rocha CA, Malavé R, Suárez P (2012) Biosurfactant production by marine bacterial isolates from the Venezuelan Atlantic Front. Bull Environ Contam Toxicol 89:1068–1072
Bragg JR, Prince RC, Harner EJ, Atlas RM (1994) Effectiveness of bioremediation for the Exxon Valdez oil spill. Nature 368:413–418
Bundy BM, Campbell AL, Neidle EL (1998) Similarities between the antABC-encoded anthranilate dioxygenase and the benABC-encoded benzoate dioxygenase of Acinetobacter sp. strain ADP1. J Bacteriol 180:4466–4474
Calvo C, Manzanera M, Silva-Castro G, Uad I, González-López J (2009) Application of bioemulsifiers in soil oil bioremediation processes: future prospects. Sci Total Environ 407:3634–3640
Cameotra SS, Makkar RS (2004) Recent applications of biosurfactants as biological and immunological molecules. Curr Opin Microbiol 7:262–266
Cameotra SS, Singh P (2009) Synthesis of rhamnolipid biosurfactant and mode of hexadecane uptake by Pseudomonas species. Microb Cell Fact 8:16
Cameotra SS, Makkar RS, Kaur J, Mehta S (2010) Synthesis of biosurfactants and their advantages to microorganisms and mankind. In: Sen R (ed) Biosurfactants. Springer, New York, pp 261–280
Champion JT, Gilkey JC, Lamparski H, Retterer J, Miller RM (1995) Electron microscopy of rhamnolipid (biosurfactant) morphology: effects of pH, cadmium, and octadecane. J Colloid Interface Sci 170:569–574
Cooper D, Macdonald C, Duff S, Kosaric N (1981) Enhanced production of surfactin from Bacillus subtilis by continuous product removal and metal cation additions. Appl Environ Microbiol 42:408–412
Das K, Mukherjee AK (2007) Crude petroleum-oil biodegradation efficiency of Bacillus subtilis and Pseudomonas aeruginosa strains isolated from a petroleum-oil contaminated soil from North-East India. Bioresour Technol 98:1339–1345
Das N, Chandran P (2010) Microbial degradation of petroleum hydrocarbon contaminants: an overview. Biotechnol Res Int 2011:1–13
Desai JD, Banat IM (1997) Microbial production of surfactants and their commercial potential. Microbiol Mole Biol Rev 61:47–64
Desai H, Mehdi TH, Cappello S (2012) Isolation and characterization of two crude oil-degrading yeast strains, Yarrowia lipolytica PG-20 and PG-32, from the Persian Gulf. Mar Pollut Bull 64:1386–1391
Dillard LA, Essaid HI, Herkelrath WN (1997) Multiphase flow modeling of a crude-oil spill site with a bimodal permeability distribution. Water Resour Res 33:1617–1632
El-Sheshtawy H, Khalil N, Ahmed W, Abdallah R (2014) Monitoring of oil pollution at Gemsa Bay and bioremediation capacity of bacterial isolates with biosurfactants and nanoparticles. Mar Pollut Bull 87:191–200
Franzetti A, Gandolfi I, Bestetti G, Banat I (2010a) Biosurfactant and bioremediation, successes and failures. In: Plaza G (ed) Trends in bioremediation and phytoremediation. Research Signpost, Kerala, pp 145–156
Franzetti A, Gandolfi I, Bestetti G, Smyth TJ, Banat IM (2010b) Production and applications of trehalose lipid biosurfactants. Eur J Lipid Sci Technol 112:617–627
Fujii T, Narikawa T, Sumisa F, Arisawa A, Takeda K, Kato J (2006) Production of α, ω-alkanediols using Escherichia coli expressing a cytochrome P450 from Acinetobacter sp. OC4. Biosci Biotechnol Biochem 70:1379–1385
Gao B, Yang L, Wang X, Zhao J, Sheng G (2000) Influence of modified soils on the removal of diesel fuel oil from water and the growth of oil degradation micro-organism. Chemosphere 41:419–426
Garon D, Krivobok S, Wouessidjewe D, Seigle-Murandi F (2002) Influence of surfactants on solubilization and fungal degradation of fluorene. Chemosphere 47:303–309
Garrett RM, Rothenburger SJ, Prince RC (2003) Biodegradation of fuel oil under laboratory and arctic marine conditions. Spill Sci Technol Bull 8:297–302
Hasanuzzaman M, Ueno A, Ito H, Ito Y, Yamamoto Y, Yumoto I, Okuyama H (2007) Degradation of long-chain n-alkanes (C 36 and C 40) by Pseudomonas aeruginosa strain WatG. Int Biodeterior Biodegrad 59:40–43
Hassanshahian M, Emtiazi G, Cappello S (2012) Isolation and characterization of crude-oil-degrading bacteria from the Persian Gulf and the Caspian Sea. Mar Pollut Bull 64:7–12
Head IM, Jones DM, Röling WF (2006) Marine microorganisms make a meal of oil. Nature Rev Microbiol 4:173–182
Henner P, Schiavon M, Morel JL, Lichtfouse E (1997) Polycyclic aromatic hydrocarbon (PAH) occurrence and remediation methods. Analusis 25:M56–M59
Ilori M, Amobi C, Odocha A (2005) Factors affecting biosurfactant production by oil degrading Aeromonas spp. isolated from a tropical environment. Chemosphere 61:985–992
Joshi S, Bharucha C, Jha S, Yadav S, Nerurkar A, Desai AJ (2008) Biosurfactant production using molasses and whey under thermophilic conditions. Bioresour Technol 99:195–199
Kim SH, Lim EJ, Lee SO, Lee JD, Lee TH (2000) Purification and characterization of biosurfactants from Nocardia sp. L-417. Biotechnol Appl Biochem 31:249–253
Koutinas M, Baptista II, Meniconi A, Peeva LG, Mantalaris A, Castro PM, Livingston AG (2007) The use of an oil–absorber–bioscrubber system during biodegradation of sequentially alternating loadings of 1,2-dichloroethane and fluorobenzene in a waste gas. Chem Engi Sci 62:5989–6001
Marchant R, Banat IM (2012) Microbial biosurfactants: challenges and opportunities for future exploitation. Trends Biotechnol 30:558–565
Middaugh D, Shelton M, McKenney C Jr, Cherr G, Chapman P, Courtney L (1998) Preliminary observations on responses of embryonic and larval Pacific herring, Clupea pallasi, to neutral fraction biodegradation products of weathered Alaska North Slope oil. Arch Environ Contam Toxicol 34:188–196
Morgan P, Watkinson RJ (1990) Assessment of the potential for in situ biotreatment of hydrocarbon-contaminated soils. Water Sci Technol 22:63–68
Mulligan CN, Gibbs BF (2004) Types, production and applications of biosurfactants. Proc Ind Natl Sci Acad Part B 70:31–56
Nguyen TT, Youssef NH, McInerney MJ, Sabatini DA (2008) Rhamnolipid biosurfactant mixtures for environmental remediation. Water Res 42:1735–1743
Obayori OS, Ilori MO, Adebusoye SA, Oyetibo GO, Omotayo AE, Amund OO (2009) Degradation of hydrocarbons and biosurfactant production by Pseudomonas sp. strain LP1. World J Microbiol Biotechnol 25:1615–1623
Oberbremer A, Müller-Hurtig R, Wagner F (1990) Effect of the addition of microbial surfactants on hydrocarbon degradation in a soil population in a stirred reactor. Appl Microbiol Biotechnol 32:485–489
Ojo O (2005) Petroleum-hydrocarbon utilization by native bacterial population from a wastewater canal Southwest Nigeria. Afr J Biotechnol 5:333–337
Pasumarthi R, Chandrasekaran S, Mutnuri S (2013) Biodegradation of crude oil by Pseudomonas aeruginosa and Escherichia fergusonii isolated from the Goan coast. Mar Pollut Bull 76:276–282
Patist A, Bhagwat S, Penfield K, Aikens P, Shah D (2000) On the measurement of critical micelle concentrations of pure and technical-grade nonionic surfactants. J Surfactants Deterg 3:53–58
Prince RC (1997) Bioremediation of marine oil spills. Trends Biotechnol 15:158–160
Ron EZ, Rosenberg E (2002) Biosurfactants and oil bioremediation. Curr Opin Biotechnol 13:249–252
Rosa CFC, Michelon M, JFdM B, SJ K, CAV B (2010) Production of a rhamnolipid-type biosurfactant by Pseudomonas aeruginosa LBM10 grown on glycerol. Afr J Biotechnol 9(53):9012–9017
Rosenberg E, Ron E (1999) High-and low-molecular-mass microbial surfactants. Appl Microbiol Biotechnol 52:154–162
Salamanca J, Ciampi E, Faux D, Glover P, McDonald P, Routh A, Peters A, Satguru R, Keddie J (2001) Lateral drying in thick films of waterborne colloidal particles. Langmuir 17:3202–3207
Sanscartier D, Reimer K, Koch I, Laing T, Zeeb B (2009) An investigation of the ability of a 14C-labeled hydrocarbon mineralization test to predict bioremediation of soils contaminated with petroleum hydrocarbons. Bioremediation J 13:92–101
Setti L, Lanzarini G, Pifferi P, Spagna G (1993) Further research into the aerobic degradation of n-alkanes in a heavy oil by a pure culture of a Pseudomonas sp. Chemosphere 26:1151–1157
Shao Z (2011) Trehalolipids, biosurfactants. Springer, New York, pp. 121–143
Shavandi M, Mohebali G, Haddadi A, Shakarami H, Nuhi A (2011) Emulsification potential of a newly isolated biosurfactant-producing bacterium, Rhodococcus sp. strain TA6. Colloids Surf B: Biointerfaces 82:477–482
Shelton M, Chapman P, Foss S, Fisher W (1999) Degradation of weathered oil by mixed marine bacteria and the toxicity of accumulated water-soluble material to two marine crustacea. Arch Environ Contam Toxicol 36:13–20
Shigematsu T, Hanada S, Eguchi M, Kamagata Y, Kanagawa T, Kurane R (1999) Soluble methane monooxygenase gene clusters from trichloroethylene-degrading Methylomonas sp. strains and detection of methanotrophs during in situ bioremediation. Appl Environ Microbiol 65:5198–5206
Singh R, Paul D, Jain RK (2006) Biofilms: implications in bioremediation. Trends Microbiol 14:389–397
Soberón-Chávez G, Maier RM (2011) Biosurfactants: a general overview, biosurfactants. Springer, New York, pp. 1–11
Tabuchi T, Nakamura I, Higashi E, Kobayashi H (1977) Factors affecting the production of the open-ring acid of spiculisporic acid by Penicillium spiculisporum. J Ferment Technol 55:43–49
Tang J, Wang M, Wang F, Sun Q, Zhou Q (2011) Eco-toxicity of petroleum hydrocarbon contaminated soil. J Environ Sci 23:845–851
Urum K, Grigson S, Pekdemir T, McMenamy S (2006) A comparison of the efficiency of different surfactants for removal of crude oil from contaminated soils. Chemosphere 62:1403–1410
Van Beilen JB, Funhoff EG (2005) Expanding the alkane oxygenase toolbox: new enzymes and applications. Curr Opin Biotechnol 16:308–314
Van Beilen JB, Funhoff EG (2007) Alkane hydroxylases involved in microbial alkane degradation. Appl Microbiol Biotechnol 74:13–21
Van Bogaert IN, Demey M, Develter D, Soetaert W, Vandamme EJ (2009) Importance of the cytochrome P450 monooxygenase CYP52 family for the sophorolipid-producing yeast Candida bombicola. FEMS Yeast Res 9:87–94
Van Hamme JD, Singh A, Ward OP (2003) Recent advances in petroleum microbiology. Microbiol Mol Biol Rev 67:503–549
Vollbrecht E, Rau U, Lang S (1999) Microbial conversion of vegetable oils into surface-active di-, tri-, and tetrasaccharide lipids (biosurfactants) by the bacterial strain Tsukamurella sp. Lipid/Fett 101:389–394
Wei Q, Mather R, Fotheringham A (2005) Oil removal from used sorbents using a biosurfactant. Bioresour Technol 96:331–334
Whang LM, Liu PWG, Ma CC, Cheng SS (2008) Application of biosurfactants, rhamnolipid, and surfactin, for enhanced biodegradation of diesel-contaminated water and soil. J Hazard Mater 151:155–163
Whyte L, Smits T, Labbe D, Witholt B, Greer C, van Beilen J (2002) Gene cloning and characterization of multiple alkane hydroxylase systems in Rhodococcus strains Q15 and NRRL B-16531. Appl Environ Microbiol 68:5933–5942
Wu JY, Yeh K, Lu WB, Lin CL, Chang JS (2008) Rhamnolipid production with indigenous Pseudomonas aeruginosa EM1 isolated from oil-contaminated site. Bioresour Technol 99:1157–1164
Yan P, Lu M, Yang Q, Zhang HL, Zhang ZZ, Chen R (2012) Oil recovery from refinery oily sludge using a rhamnolipid biosurfactant-producing Pseudomonas. Bioresour Technol 116:24–28
Zajic J, Guignard H, Gerson D (1977) Properties and biodegradation of a bioemulsifier from Corynebacterium hydrocarboclastus. Biotechnol Bioeng 19:1303–1320
Zhang Z, Gai L, Hou Z, Yang C, Ma C, Wang Z, Sun B, He X, Tang H, Xu P (2010) Characterization and biotechnological potential of petroleum-degrading bacteria isolated from oil-contaminated soils. Bioresour Technol 101:8452–8456
Zinjarde SS, Pant A (2002) Emulsifier from a tropical marine yeast, Yarrowia lipolytica NCIM 3589. J Basic Microbiol 42:67–73
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Wijesekara, S., Seneviratne, M., Vithanage, M. (2017). Role of Biosurfactants on Microbial Degradation of Oil-Contaminated Soils. In: Singh, J., Seneviratne, G. (eds) Agro-Environmental Sustainability. Springer, Cham. https://doi.org/10.1007/978-3-319-49727-3_9
Download citation
DOI: https://doi.org/10.1007/978-3-319-49727-3_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-49726-6
Online ISBN: 978-3-319-49727-3
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)