Abstract
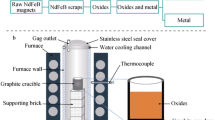
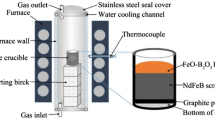
For recovery of rare earth elements from the NdFeB based permanent magnet scraps, a high temperature process was used. Permanent magnets scraps were firstly oxidized in air. The optimal oxidation conditions under air are at temperature of 700°C holding for 30min. After the oxidation process, the oxides were reduced by graphite crucible at 1500°C under Ar atmosphere. By this way, the rare earth elements were recovered in the form of oxides, and Fe was separated to the metal phase. Boron distributed in both the rare earth oxides (REO) containing slag and the metal phase. In order to reduce the boron contents in the rare earth oxides, the carbon saturated iron bath and NdFeB added carbon saturated iron bath were used to refine the slag. By this process, the rare earth oxides with a comparatively high purity of 98.2% were gotten.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
S. Doris et al., Study on Rare Earths and Their Recycling, Final Report for The Greens/EFA Group in the European Parliament, (2011). 3–6.
H. George C, L. Jinfang, G. Alexander, and M. Melania, “Current Status of Rare Earth Permanent Magnet Research in USA,” Proceedings of 19th International Workshop on Rare Earth Permanent Magnets & Their Applications, (2006), 12–22.
B. Koen, J. Peter Tom, B. Bart, G. Tom Van, Y. Yongxiang, W. Allan, and B. Matthias, “Recycling of Rare Earths a Critical Review,” Journal of Cleaner Production, 51 (2013), 1–22.
L. Ran, B. Matthias, D. Stefanie, M. Andreas, M. Cornelia, and S. Doris, “Application of Rare Earths in Consumer Electronics and Challenges for Recycling,” iEEE International Conference on Consumer Electronics (Berlin Germany), (2011), 286–290.
M. Stefania and R. Marcello, “Rare Earth Elements as Critical Raw Materials: Focus on International Markets and Future Strategies,” Resources Policy, 38 (2013), 36–43.
D. Xiaoyue and T.E. Graedel, “Global In-Use Stocks of the Rare Earth Elements: A First Estimate,” Environmental Science & Technology, 45 (9) (2011), 4096–4101.
J. Nan, D. Han, M. Yang, M. Cui, and X. Hou, “Recovery of metal values from a mixture of spent lithium-ion batteries and nickel-metal hydride batteries,” Hydrometallurgy, 84 (1–2) (2006), 75–80.
S. Nishihama, N. Sakaguchi, T. Hirai, and I. Komasawa, “Extraction and Separation of Rare Earth Metals Using Microcapsules Containing Bis (2-Ethylhexyl) Phosphinic Acid,” Hydrometallurgy, 64 (1) (2002), 35–42.
F. Yang, F. Kubota, Y. Baba, N. Kamiya, and M. Goto, “Selective extraction and recovery of rare earth metals from phosphor powders in waste fluorescent lamps using an ionic liquid system,” Journal of Hazardous Materials, 254–255 (2013), 79–88.
I. Masahiro, M. Koji, and M. Ken-ichi, “Novel Rare Earth Recovery Process on NdFeB Magnet Scraps by Selective Chlorination using NH4Cl,” Journal of Alloys and Compounds, 477 (1–2) (2009), 484–487.
A.T. Dinsdale, “SGTE data for pure elements,” Calphad, 15 (1991), 317–425.
J.M. Le Breton, J. Teillet, P.J. McGuiness, D.S. Edgley, and R. Harris, “The oxidation of Nd-Fe-B permanent magnet at 400oC: a SEM, microhardness and Mössbauer study,” Magnetics, IEEE Transactions on, 28 (5) (1992), 2157–2159.
Y. Li, H.E. Evans, I.R. Harris, and I.P. Jones, “The Oxidation of NdFeB Magnets,” Oxidation of Metals, 59 (2003), 167–182.
G.W. Warren, G. Gao, and Q. Li, “Corrosion of NdFeB Permanent Magnet Materials,” Journal of Applied Physics, 70 (10) (1991), 6609–6611.
M. Nakamoto, K. Kubo, Y. Katayama, T. Tanaka, and T. Yamamoto, “Extraction of Rare Earth Elements as Oxides from a Neodymium Magnetic Sludge,” Metal. Mater. Trans B, 43 (3) (2011), 468–476.
T. Saito, H. Sato, S. Ozawa, J. Yu, and T. Motegi, “The Extraction of Nd from Waste NdFeB Alloys by the Glass Slag Method,” Journal of Alloys and Compounds, 353 (11) (2003), 189–193.
U. Tetsuya, “Recovery of Rare Earths from Magnet Sludge by FeCl2,” Materials Transactions, 43 (1) (2002), 55–62.
T. Osamu, H. Toru, Okabe, and U. Yoshiaki, “Recovery of Neodymium from a Mixture of Magnet Scrap and Other Scrap,” Journal of Alloys and Compounds, 408–412 (2006), 387–390.
O. Takeda, T.H. Okabe, Y. Umetsu, “Phase Equilibrium of the System Ag-Fe-Nd, and Nd Extraction from Magnet Scraps using Molten Silver,” Journal of Alloys and Compounds, 379 (1–2) (2004), 305–313.
A.M. Martinez, O. Kjos, E. Skybakmoen, A. Solheim, and G.M. Haarberg, “Extraction of Rare Earth Metals from Nd-based Scrap by Electrolysis from Molten Slag,” ECS Transactions, 50 (2012), 453–461.
K. Miura, M. Itoh, and K.-I. Machida, “Extraction and Recovery Characteristics of Fe Element from Nd-Fe-B Sintered Magnet Powder Scrap by Carbonylation,” Journal of Alloys and Compounds, 466 (1–2) (2008), 228–232.
D.S. Edgley, J.M. Le Breton, S. Steyaert, F.M. Ahmed, I.R. Harris, and J. Teillet, “Characterisation of High Temperature Oxidation of Nd-Fe-B Magnets,” Journal of Magnetism and Magnetic Materials, 173 (1–2) (1997) 29–42.
S.C. Parida, S. Dash, Z. Singh, R. Prasad, K.T. Jacob, and V. Venugopal, “Thermodynamic Studies on NdFeO3(s), Journal of Solid State Chemistry,” 164 (1) (2002), 34–41.
A. Roine, Outolumpu HSC Thermochemistry for Windows, Ver.5.1 (Ourokumpu Research Oy, Pori, Finland).
S.K. Thomas and C.J. Buckley, “Metastable States in Alumina-Rich La2O3-Al2O3 and Nd2O3-Al2O3,” Journal of American Ceramic Society, 88 (1) (2005) 191–195.
G. Goller, C. Toy, A. Tekin, and C.K. Gupta, “The Production of Boron Barbide by Carbothermic Reduction,” High Temperature Materials and Processes, 15 (1–2) (1996) 117–122.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 TMS (The Minerals, Metals & Materials Society)
About this chapter
Cite this chapter
Bian, Y. et al. (2015). Recovery of Rare Earth Elements from NdFeB Magnet Scraps by Pyrometallurgical Processes. In: Neelameggham, N.R., Alam, S., Oosterhof, H., Jha, A., Dreisinger, D., Wang, S. (eds) Rare Metal Technology 2015. Springer, Cham. https://doi.org/10.1007/978-3-319-48188-3_27
Download citation
DOI: https://doi.org/10.1007/978-3-319-48188-3_27
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-48606-2
Online ISBN: 978-3-319-48188-3
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)