Abstract
Under physiologic conditions, the brain is a microbiologically sterile site and is protected from infection by highly specialized barriers, including the hard bony skull, the tough dura mater, and the restrictive blood–brain barrier (BBB). Host defense mechanisms in the central nervous system (CNS) are limited and tightly regulated. Peripheral immune cells and plasma proteins are largely excluded from the brain parenchyma. Once they have breached the protective barriers and entered the CNS, bacteria multiply within the cerebrospinal fluid space (CSF) highly efficiently exhibiting similar kinetics as in vitro and reaching concentrations of up to 109 CFU/mL.
In response to the multiplying bacteria and their components, i.e., cell wall fragments, lipopolysaccharides, teichoic and lipoteichoic acids, peptidoglycans, bacterial DNA, and other cytosolic factors, resident cells in the perivascular space and the meninges release pro-inflammatory signaling molecules. Tumor necrosis factor-α, interleukin-1β, and IL-6 are released early on and trigger a cascade of other inflammatory mediators, including a variety of cytokines, chemokines, platelet-activating factor, antimicrobial peptides, prostaglandins, matrix metalloproteinases, nitric oxide, and reactive oxygen species initiating a self-perpetuating inflammatory cascade.
The immediate consequences of the intense inflammatory reaction are a massive influx of leukocytes, the breakdown of the blood–brain barrier with the formation of brain edema, and alterations of the cerebral blood flow. This overshooting inflammatory reaction to the invading pathogens causes damage to the brain parenchyma as collateral damage and is the driving pathophysiologic mechanism of inflammatory inner ear damage, brain cortical ischemic injury, and hippocampal apoptosis, the most frequent histopathological correlates of the neurofunctional sequelae of bacterial meningitis.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Meningitis is an inflammation of the membranes enclosing the brain and spinal cord. Meningitis may be caused by viral, fungal, or parasitic infection, but is frequently caused by bacteria capable of penetrating the barriers protecting the brain. The barrier function of the blood–brain barrier (BBB) and the blood–cerebrospinal fluid barrier (BCSFB) largely excludes peripheral immune cells and plasma proteins in normal physiological conditions, which led to the concept of the central nervous system (CNS) as a site of immune privilege. The lack of immediate and sufficient bactericidal activity in the cerebrospinal fluid (CSF) allows bacteria to multiply almost as efficiently as in vitro, to reach titers of up to 109 colony-forming units (CFU)/mL and to spread over the entire surface of the brain, the spinal cord, and along penetrating vessels. This explains that prior to the antibiotic era, bacterial meningitis almost always led to death.
To gain access to the CNS, bacteria must (a) colonize the host, (b) invade the bloodstream or cause focal infection in the vicinity of the brain, (c) survive in the bloodstream, and (d) cross the BBB or the BCSFB (Fig. 1). Bacterial meningitis can arise from bacteremia if pathogens colonizing the nasopharyngeal and intestinal mucosa invade the bloodstream or by continuous infection from nearby foci, e.g., sinusitis, mastoiditis. It may also follow a breach of the brain barriers by a foreign body (e.g., cerebrospinal fluid shunt, cochlear implant), a neurosurgical procedure, or trauma.
Once a pathogen has reached the CNS, a cascade of events follows until the full symptomatic manifestations of bacterial meningitis. These include the induction of cytokines and chemokines; activation of inflammatory mediators such as nitric oxide (NO), reactive oxygen species (ROS), or matrix metalloproteinases (MMP); recruitment of white blood cells to the site of infection; and cytotoxic events. In this chapter, the pathogenesis of bacterial meningitis and its neurological sequelae are discussed.
2 Pathogenesis of Bacterial Meningitis
2.1 Bacterial Invasion of the Host and Penetration of the Blood–Brain Barrier
Figure 2 shows the pathogenic steps involved in the development of bacterial meningitis.
Pathogenetic steps involved in the development of bacterial meningitis: (i) colonization of the nasopharynx and invasion of blood capillaries via crossing of the mucosal barrier, the epithelia and the vascular endothelium; (ii) bacteremia, survival in the blood and crossing of the blood–brain barrier (BBB); and (iii) bacterial multiplication in the CSF, induction of inflammation, BBB breakdown, and invasion of blood-derived neutrophils into the subarachnoid and ventricular space
Adherence of bacteria to the mucosal epithelium and colonization are affected by phase variation. Bacteria cross the mucosal barrier through or between epithelial cells. Once in the bloodstream, expression of a sufficiently thick capsule is necessary to protect the bacteria from circulating antibodies, complement-mediated bacterial killing, and neutrophil phagocytosis. Bacteria penetrate the BBB by (i) paracellular passage, (ii) transcellular passage, or (iii) invasion within white blood cells during diapedesis (intracellular pathogens). Neutrophilic pleocytosis occurs along a chemotactic gradient mainly created by chemokines locally expressed in the brain. Matrix metalloproteinases (MMP), such as MMP-8 and MMP-9, facilitate the process of extravasation by degrading extracellular matrix components of the brain microvasculature
2.1.1 Bacterial Colonization of the Host
Colonization of the host is a common first step of the major bacterial pathogens that cause meningitis (Table 1). S. pneumoniae, N. meningitidis, and H. influenzae type B colonize the nasopharynx and are transmitted from person to person by the respiratory route. Injury of the functional integrity of the respiratory tract mucosa by viral infections [2, 3] and physical damage to the mucosa [4] may increase the risk of invasive bacterial disease.
To colonize the nasopharyngeal mucosa, a pathogen first has to evade mucosal defense mechanisms like ciliary clearance [5–10], secretory immunoglobulin A (IgA) [11–13], and lysozyme [14–17]. Secondly, it has to prevail against other organisms in the aerobic environment of the upper respiratory tract [18–20]. Finally, it must adhere to the mucosal epithelium [10, 21–25].
Group B streptococcus (GBS) (S. agalactiae), E. coli, and L. monocytogenes colonize the gastrointestinal tract and are transmitted by the oral, vaginal, or fecal–oral route. They mainly cause meningitis at the vulnerable extremes of age and in immunocompromised persons. In neonatal meningitis, colonization of the intestinal tract of the infant follows similar principles as outlined for nasopharyngeal colonization [26–34]. The food-borne pathogen L. monocytogenes primarily affects immunocompromised patients and pregnant women and is transmitted to the fetus through the placenta or to the newborn during delivery [35, 36].
2.1.2 Invasion of the Bloodstream
To enter the bloodstream at the site of colonization, bacteria penetrate the mucosal barrier formed by epithelial cells and the lining of the blood vessels formed by the vascular endothelium (Fig. 2). Depending on the pathogen, the paracellular or transcellular route across the epithelial and endothelial layers is preferred.
Most known mechanisms by which meningeal pathogens invade human epithelial and endothelial cells via the transcellular route take advantage of the ubiquitous clathrin-mediated cellular endocytosis mechanism [37–39]. This then leads to the uptake into vacuoles that may either recycle back to the cell surface, fuse with lysosomes, or transit to the basolateral cell surface where the bacteria are released out of the cell [40–42].
Actin rearrangements further enhance bacterial internalization. Some transmembrane proteins of the epithelial cell that are targeted by the bacterial pathogen and initiate endocytosis are polymeric immunoglobulin receptor (pIgR) [43, 44], carcinoembryonic antigen-related cell adhesion molecule (CEACAM) proteins [45–47], and epithelial cadherin (E-cadherin) [48, 49].
Paracellular migration of bacteria across the epithelial barrier is facilitated by toll-like receptor (TLR)-dependent downregulation of tight junction protein expression [50] or extracellular degradation of junctional components mediated by bacterial enzymes or bacteria-bound plasmin [51–54].
The final barrier between epithelial cells and mesenchymal cells is the epithelial basement membrane. Meningeal pathogens may degrade the epithelial basement membrane by secreting proteolytic enzymes [51, 55], abuse the host’s plasminogen–plasmin system [53, 56–58], and cross the basement membrane in phagocytic cells [59, 60]. Finally, attachment to extracellular matrix (ECM) components is important in bacterial infection pathogenesis, and ECM adhesins have been identified in most meningeal pathogens (e.g., PavA of S. pneumoniae [21], Opc of N. meningitidis [61], fimbriae of H. influenzae [62], and ScpB of GBS [31]).
2.1.3 Intravascular Survival
Once in the bloodstream, meningeal pathogens are immediately exposed to the host’s immune defense. Clearance of S. pneumoniae, H. influenzae, and N. meningitidis strongly relies on antibody-mediated opsonization followed by activation of the complement system. S. pneumoniae and H. influenzae are then taken up by host leukocytes and killed intracellularly, while the membrane attack complex, formed by the terminal complement factors, is important for the killing of N. meningitidis [63]. Patients with impaired opsonization or complement activation (e.g., patients with sickle cell disease or complement deficiencies) have a higher susceptibility to invasive infections due to N. meningitidis and S. pneumoniae [64–67].
The polysaccharide capsule, although impeding adherence to the mucosal surface and entry into epithelial cells [10, 68], is the primary survival factor in the bloodstream [69]. It shields bacteria against circulating antibodies [70, 71], complement-mediated bacterial killing [72], and neutrophil phagocytosis [73]. Meningeal pathogens may also manipulate the complement cascade by enhancing the activity of regulatory proteins [74–79].
Intravascular survival of the intracellular pathogen L. monocytogenes depends mainly on its ability to evade intracellular killing in the phagolysosome [80]. Bacterial clearance by phagocytosis is an important early step in listeriosis, but the intracellular survival of L. monocytogenes facilitates evasion of innate immune mechanisms (“Trojan-horse” mechanism).
In summary, the intricate defense mechanisms of bacteria against the host immune system only need to protect few organisms, which may then cause meningitis [81].
2.1.4 Meningeal Invasion
The CNS is a tightly controlled environment. The boundary to the blood is provided by the endothelial BBB, the epithelial BCSFB, and the arachnoid [82]. Blood-borne pathogens must cross the BBB or the BCSFB to cause bacterial meningitis. Clinical observations and experimental studies link the magnitude of bacteremia with the risk of meningitis development for most meningeal pathogens [65, 83, 84]. High bacterial load in the blood alone, however, is not sufficient for the development of bacterial meningitis. The expression of selected bacterial adhesion and invasion factors is necessary for the invasion of the CNS [85].
Meningeal pathogens may migrate into the CSF by transcellular or paracellular migration [85]. The BCSFB may be more vulnerable to bacterial migration via the paracellular route because the tight junctions between the epithelial cells have a lower electrical resistance [82]. For L. monocytogenes, the previously described Trojan-horse mechanism has also been implicated in the invasion of the CNS [86–88].
Direct invasion of endothelial and epithelial cells by meningeal pathogens involves ligand–receptor interactions between bacteria and host cells followed by rearrangements of the actin cytoskeleton. This ultimately leads to the uptake of the pathogen in a vacuole and transcellular transport [42, 85, 89]. Attachment of meningeal pathogens to the tight endothelium of the BBB and the fenestrated endothelium of the BCSFB is facilitated by receptors on the apical side of the endothelial cells, some of which may be upregulated by inflammation. The 37/67-kDa laminin receptor is a shared target of cytotoxic necrotizing factor 1 (CNF1)-expressing E. coli K1, S. pneumoniae, N. meningitidis, and H. influenzae [90–94]. Bacterial adhesins mediate adhesion to the 37/67-kDa laminin receptor, including CNF1 in E. coli, choline-binding protein A (CbpA) in S. pneumoniae, secretin PilQ and porin PorA in N. meningitidis, and porin OmpP2 in H. influenzae [92, 93, 95]. S. pneumoniae may also bind to the cell adhesion molecule platelet endothelial cell adhesion molecule-1 (PECAM-1) and pIgR [96, 97].
Fimbriae and pili, hair-like structures frequently found on the surface of bacteria, are instrumental to bacterial adhesion. Type IV pili mediate attachment, bacterial aggregation, and persistence of N. meningitidis on brain endothelial cells [98]. Binding of E. coli K1 to brain endothelial cells is facilitated by FimH on type 1 fimbriae CD48 and the association of the outer membrane protein A (OmpA) with glycoproteins on the cell surface [85]. Attachment of GBS to brain endothelial cells is facilitated by several adhesins including fibrinogen receptor FbsA, laminin receptor Lmb, and the pilus tip adhesins PilA and Srr1 [99–103].
The exact mechanisms by which S. pneumoniae invades the BBB are not yet known. Partially conflicting in vitro and in vivo data indicate that binding of S. pneumoniae to transmembrane receptors pIgR and PAFr may trigger clathrin-mediated cellular endocytosis [41, 97, 104]. Specifically, the interaction of phosphorylcholine with PAFr and expression of NanA seem to be necessary for the invasion of the BBB by S. pneumoniae [40, 105–107].
E. coli invades the BBB following the induction of cytoskeletal rearrangements and actin condensations through the activation of the Rho family GTPases [85, 108, 109]. The interaction of several E. coli proteins with the BBB has been shown to facilitate cytoskeletal rearrangements culminating in the uptake of E. coli into a vacuole [94, 110–117].
For GBS, experimental data support an important role of the fibronectin-binding protein SfbA and pili in BBB invasion [101, 118]. The expression of fibronectin-binding protein OpC has also been shown to be instrumental to the invasion of the BBB by N. meningitidis [119]. N. meningitidis also form bacterial microcolonies on the luminal face of brain endothelial cells followed by the reorganization of the intracellular cytoskeleton. This may lead to the opening of intercellular junctions followed by paracellular penetration of the BBB [98]. Receptor-mediated signaling, as well as local inflammation with the recruitment of leucocytes, may further modify tight junctions and allow paracellular penetration [98, 101].
Conflicting evidence exists, whether the transcellular invasion of brain endothelial cells by L. monocytogenes may be mediated by InlA and InlB in a similar fashion as in the intestine and placenta [86, 120].
Bacteremia-independent invasion of the CNS may follow focal infections of structures close to the brain (e.g., otitis media, mastoiditis, and sinusitis) or after disruption of the integrity of the skull and meninges (e.g., malformations, trauma, neurosurgery). In a case series of 87 patients with pneumococcal meningitis, more than half of the patients had radiological signs consistent with otitis or sinusitis [121]. In a large observational study, otitis and sinusitis were reported as predisposing conditions in 25 % of patients [122]. This is supported by an experimental model of S. pneumoniae meningitis, in which a galU mutant and its parent pneumococcal strain both caused meningitis following otitis media infection in gerbils, despite the mutant’s impaired ability to disseminate to the bloodstream following infection [123].
For L. monocytogenes, S. pneumoniae and N. meningitidis retrograde access to the CNS via the neural route has also been documented [86, 124, 125]. For L. monocytogenes, however, this mechanism is likely only important in ruminants. These observations support the notion that meningeal pathogens can gain access to the CNS by several routes. The relative importance of the different routes of infection in bacterial meningitis is not exactly known.
3 Induction and Modulation of Inflammation
Our understanding of the cascade of events leading to brain inflammation during meningitis is mostly based on experimental models, in which bacteria are inoculated intranasally, but more often intracisternally or intracerebrally. These models do not fully replicate the development of meningitis as observed in patients, especially when considering the initial anatomical progression from the nasopharynx.
Early recognition of the pathogens involves brain vascular endothelial cells or perivascular macrophages [126], as well as ependymal cells. These may be aided by a trafficking population of central memory T cells which interact with local antigen-presenting cells and initiate inflammation. The very recent discovery of functional lymphatic vessels in the CNS, however, may considerably change the perception on the role of adaptive immunity in the brain [127]. Leukocytes, mainly neutrophils in the case of bacterial meningitis, are initially excluded from the CNS, being recruited later during infection [124–126]. Most pathogens responsible for bacterial meningitis have been shown to induce inflammation in brain microvascular endothelial cells in vitro, which facilitates their invasion through the blood–brain barrier. In experimental GBS meningitis, the early induction of tumor necrosis factor-α (TNF-α) and interleukin (IL)-1β mRNA expression has been documented in the ependyma and the meninges [127]. Little is known about the role of perivascular macrophages, but their depletion has been shown to worsen the clinical symptoms, to increase bacterial counts in the CSF, and to decrease leukocytes extravasation. However, since higher levels of CSF inflammation were still detected in depleted animals, a local production of inflammatory mediators in the parenchyma was also suggested [128].
During disease progression, cells in the parenchyma, especially next to the site of the initial inflammation, also express inflammatory factors, including TNF-α and IL-1β [127]. Microglia and astrocytes are sentinels in the brain parenchyma that are able to detect invading pathogens. It is postulated that, as the infection/inflammation progresses, diffusible bacterial products or host-derived factors penetrate the parenchyma, triggering a secondary response.
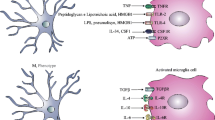
How the innate immune system recognizes invading pathogens is mostly determined by its ability to “sense” the bacterial surface. Bacterial pathogens causing meningitis usually are encapsulated microorganisms. The polysaccharide capsule is a virulence factor which is necessary for establishing invasive disease by preventing opsonophagocytosis (see above). In vivo, the pneumococcal capsule could also impair recognition by the innate immune system, in particular the toll-like receptor-mediated pathway [129]. The capsule can partially cover subcapsular bacterial components, depending on its thickness. For gram-positive bacteria, immunogenic components comprise teichoic (TA) and lipoteichoic acids (LTA) and peptidoglycan. For gram-negative bacteria, the endotoxin lipopolysaccharide (LPS) is recognized by the innate immune system. These components are released either by autolysis, when bacteria reach sufficient high density, or by the effect of bacteriolytic antibiotics and trigger the inflammatory reaction in bacterial meningitis. Specialized extracellular (i.e., toll-like receptors, TLRs) or intracytoplasmic (i.e., NOD-like receptors, NLRs) receptors can sense the major meningeal pathogens [130–133]. Most cells of the brain express these receptors, with microglia expressing the full repertoire of TLRs [134]. Brain endothelial cells and macrophages are also known to express these receptors. Negative outcome during bacterial meningitis has been associated with genetic variations in these pattern recognition receptors (PRR) [135–137]. TLR2 detects cell wall components of gram-positive bacteria, while TLR4 recognizes those of gram-negative bacteria. TLR9 recognizes bacterial DNA and other components. Scavenger receptors (SRs), initially described for their role in the identification and removal of modified lipoproteins, are found at the surface of perivascular and meningeal macrophages [138]. SRA-1 (CD204) and SRA-2 (MARCO) can detect N. meningitidis or S. pneumoniae [138]. The G-coupled receptor protein formyl peptide receptor-like-1 (FPRL-1), which increases the expression of antimicrobial peptide CRAMP, recognizes S. pneumoniae. Of note, it seems that the resident immune system in the brain parenchyma may already be activated during the initial presence of pneumococci in the blood in a bacteremia-derived meningitis model, even before the development of meningitis and the recruitment of neutrophils [139].
3.1 Cytokines and Chemokines
An increase in cytokines and chemokines expression levels has been documented in the brain parenchyma or the CSF of patients with bacterial meningitis and experimental models [140–143]. Cytokine and chemokine profiles vary in vitro and in experimental infection models in response to different meningeal pathogens. These differences are presumably related to pathogen-specific activation of PRR [144–147] and may in part explain the differences in mortality and morbidity observed in patients affected by the different pathogens.
The early release of TNF-α, IL-1β, and IL-6 in the course of bacterial meningitis is part of the initial innate immune answer. The expression of TNF-α and IL-1β is detected first in the ependyma and the meninges and later in the parenchyma in experimental meningitis [127]. The initial immune reaction induces a cascade of inflammatory mediators, including other cytokines and chemokines, antimicrobial peptides, prostaglandins, MMPs, NO, and ROS.
During bacterial meningitis, TNF-α activity is kept at a sustained high level in the CSF due to the continuous release of bacterial products into the CSF or by a positive feedback loop in the inflammatory cascade [148]. Similar to PPR engagement, binding of TNF-α to the TNF-α receptor leads to NF-kB activation resulting in the expression of further mediators described hereafter. Experimental administration of TNF-α into the CSF of rabbits or rats induces BBB breakdown, neutrophil influx, and increased cerebral blood flow (CBF), recapitulating pathophysiologic changes characteristic of bacterial meningitis in humans [149–154].
IL-1β is released by mononuclear phagocytes, glial cells, and endothelial cells in the CNS. Caspase-1 in the inflammasome complex activates the precursors of IL-1 or IL-18 [132, 155–157]. The CSF concentration of IL-1β significantly correlates with other inflammatory parameters and is a predictor of adverse disease outcome [158]. When experimentally injected into the CSF, IL-1 triggers a meningeal inflammation without detectable TNF-α activity, whereas the concomitant injection of TNF-α and IL-1 results in a synergistic increase in leukocyte influx into the CSF [159].
IL-6 is produced by monocytes, endothelial cells, and astrocytes, essentially in response to IL-1β and TNF-α. Its presence in the CSF of patients with bacterial meningitis is not correlated with any of the indices of meningeal inflammation or with disease severity [160]. IL-6 efficiently induces the expression of acute-phase proteins, fever, leukocytosis, and activation of the complement and clotting cascades [161]. IL-6 also possesses anti-inflammatory properties, by inhibiting TNF-α and IL-1β production in vitro and inducing IL-1 receptor antagonist [162].
Chemokine levels, including IL-8 (CXCL8), CXC5 (ENA-78), CXCL1 (GROα), and monocyte chemoattractant protein-1 (MCP-1, CCL2), MIP-1 (CCL3), and MIP-1 (CCL3), are elevated in the CSF of patients with bacterial meningitis [163, 164]. Cells shown to produce these chemokines upon stimulation include monocyte macrophages, polymorphonuclear leukocytes, endothelial cells, astrocytes, microglia, and neurons. Chemokines primarily activate and attract leukocytes to the site of inflammation. By enhancing neutrophil adhesion to endothelial cells, IL-8 regulates the migration of neutrophils toward the CNS during bacterial meningitis [142].
IL-10 is an anti-inflammatory cytokine that inhibits the production of TNF-α, IL-1, IL-6, and IL-8 in vitro. High levels of IL-10 have been found in the CSF of patients with bacterial meningitis. In experimental meningitis, application of IL-10 decreased brain edema and down-modulated subarachnoid space inflammation [165]. In addition to IL-10, transforming growth factor-β (TGF-β) also possesses anti-inflammatory properties, as demonstrated in experimental meningitis, where its application reduced cerebral edema, intracranial pressure, and CBF [166]. Similarly, IL-19 is produced by astrocytes in response to S. pneumoniae infection and acts as an anti-inflammatory modulator [167].
In bacterial meningitis, especially caused by S. pneumoniae, IFN-γ levels are increased in the CSF [157, 168]. IFN-γ stimulates nonspecific defense mechanisms such as phagocytosis or cytokine release by macrophages and polymorphonuclear leukocytes. In experimental pneumococcal meningitis in mice, the inflammasome complex initiates the production of IFN-γ [157]. IFN-γ inactivation results in increased survival, attenuation of pro-inflammatory cytokines in the CSF, less brain injury, and improved neurofunctional outcome [169].
3.2 Matrix Metalloproteinases and Related Proteins
MMPs are important role players to the pathogenesis of bacterial meningitis, participating in the breakdown of the BBB and the intrathecal production of cytokines, therefore contributing to the pleocytosis of neutrophils in the CSF [170, 171]. As endopeptidases are able to degrade ECM components, MMPs facilitate cell migration, tissue remodeling, and cytotoxicity. MMPs also modulate cytokine production and release. MMPs and related metalloproteinases, e.g., TNF-α converting enzymes (TACE/ADAM-17), transform membrane-bound cytokines, cytokine receptors, and adhesion molecules to their soluble forms by virtue of their sheddase activity. In return, cytokines such as TNF-α, IL-1, and IL-2 modulate the expression and regulation of MMPs. Most MMPs are not constitutively expressed but produced in response to increased levels of cytokines, eicosanoids, growth factors, and the presence of pathogens. Negative regulation of MMP activity is mediated by tissue inhibitors of metalloproteinases (TIMP), the specific endogenous inhibitors of MMPs. These inhibit MMPs by the formation of complexes with pro-peptide containing inactive and cleaved activated forms of MMPs. Similar to MMPs, TIMPs are also regulated in response to changing levels of a variety of signaling molecules [172].
Levels of MMP-1, MMP-3, MMP-7, MMP-8, MMP-9, and MMP-10 have been found to be elevated in the CSF of patients with bacterial meningitis. Modest increases in TIMP-1 and TIMP-2 levels have also been observed while TIMP-4 was concomitantly downregulated in the CSF of the same patient cohort [173–175]. In the brain tissue of patients with purulent meningoencephalitis, endothelial cells and infiltrating leukocytes have been shown to produce MMP-9, TIMP-1, and TIMP-2 [176].
In pediatric patients with bacterial meningitis, MMP-9 levels in the CSF fell by 90 % within 1 week, while TIMP-1 levels continued to rise by 51 % [177]. Persistent high levels of MMP-9 may be associated with a negative neurological outcome in childhood bacterial meningitis [177, 178].
In experimental pneumococcal meningitis, changes in MMP-9/TIMP-1 ratio and an increase in collagen degradation were observed, resulting in cortical brain damage [179]. MMP inhibitors prevented this effect [180, 181]. Also, combined inhibition of MMP and TACE led to a reduction of hippocampal apoptosis and preserved learning capacity of animals that recovered from bacterial meningitis [148, 182].
3.2.1 Neutrophil Invasion
Marked neutrophil pleocytosis in the CSF is a distinctive feature of bacterial meningitis. In response to chemotactic stimuli, neutrophils extravasate across the fenestrated endothelium followed by an inverse transmigration across the tight epithelial ependyma to accumulate in the CSF where they contribute to the deleterious effects of inflammation in the brain [183–185].
Inflammation induces E-selectin and P-selectin expression on the surface of endothelial cells. This allows the binding of neutrophils through P-selectin glycoprotein ligand-1 and other glycosylated ligands [186]. Deficiency in E-selectin or P-selectin expression resulted in almost complete inhibition of neutrophil influx when recombinant IL-1β and TNF-α were directly injected into the CSF of mice [187]. Blocking of selectins by the polysaccharide fucoidan also attenuates CSF neutrophil pleocytosis in experimental pneumococcal meningitis [188–190]. The firm adhesion of chemokine-activated leukocytes to the endothelium is mediated by the binding of integrins to members of the immunoglobulin-like superfamily on the endothelium, including ICAM-1 and vascular cell adhesion molecule-1 (VCAM-1). Antibody-mediated blocking of integrins or ICAM-1 decreases leukocyte influx in experimental meningitis models, confirming an important role for these molecules in assisting leukocyte entry into the inflamed neural tissue [191–193].
Bacteria-induced transmigration of leukocytes across the BBB or the BCSFB may occur by the paracellular or transcellular route as determined in in vitro models [194–197]. Intracellular signaling induced by the interaction of integrins on the leukocytes with endothelial cell adhesion molecules leads to structural changes in endothelial cells including remodeling of the actin cytoskeleton. This facilitates migration of leukocytes either transcellularly or paracellularly [198]. Interestingly, the early presence of leukocytes in the CSF does not affect bacterial multiplication in experimental meningitis models [199, 200].
3.2.2 Resolution of the Inflammation
Apart from the production of anti-inflammatory activity of IL-6, IL-10, and TGF-β, different mechanisms are initiated later during the disease course and participate in the resolution of inflammation. The clearance of invading neutrophils by apoptosis is necessary to avoid the excess release of neurotoxic molecules. Experimental inhibition of apoptosis in neutrophils by overexpression of the antiapoptotic factor bcl-2 prolonged the expression of pro-inflammatory cytokines in experimental pneumococcal meningitis in mice. On the contrary, treatment with roscovitine, a specific inducer of apoptosis in neutrophils, reduced damage and improved survival in vivo in the same model [201]. Similarly, TNF-related apoptosis-inducing ligand (TRAIL) released by microglia triggers leukocyte apoptosis. Elevated levels of TRAIL are documented in the CSF of patients with pneumococcal meningitis. The administration of recombinant TRAIL increased leukocyte apoptosis and decreased inflammation and neuronal apoptosis in experimental pneumococcal meningitis, while TRAIL knockout animals displayed a worse outcome than wild-type mice [202].
4 Sites of Damage in Meningitis
Derived from observations in patients and experimental models of bacterial meningitis, the cerebral vasculature, the brain parenchyma (cortex or hippocampus), and the inner ear are primarily injured by the disease. The morbidity observed in survivors reflects damage to these structures.
Figure 3 shows the histopathology of experimental pneumococcal meningitis and group B streptococcal meningitis.
Histopathology of experimental pneumococcal meningitis (a–c, e) or group B streptococcal meningitis (d). (a) Extensive cortical injury consisting of areas of cortical necrosis. A wedge-shaped distribution of reduced neuronal density, suggestive of ischemic damage, can be distinguished from neighboring more healthy cortical tissue (cresyl violet, magnification × 10). (b) Cortical neuronal loss on the right contains necrotic neurons characterized by cell swelling and fading of cytoarchitecture and is sharply demarcated from preserved neurons on the upper left (cresyl violet, magnification × 40). (c) Hippocampal dentate gyrus histology of an infant rat suffering from pneumococcal meningitis at 42 h after infection. Apoptotic cells are characterized by the presence of round or oval apoptotic bodies consisting of regularly shaped, often fragmented, dark chromatin clumps (upper left inset). Apoptotic cells are mostly observed in the inner rim (i.e., the subgranular zone) of the dentate gyrus (cresyl violet, magnification × 40). (d) Dentate gyrus of the hippocampus of an infant rat infected with group B streptococcus. Large clusters of cells with uniformly dense and shrunken (pyknotic) nuclear morphology (upper left inset) are observed throughout the entire blade. (e) Subarachnoid space inflammation consisting of bacteria and inflammatory cells (asterisk) extending into the Virchow-Robin space (arrowheads) around the penetrating cortical vasculature (cresyl violet, magnification × 63)
4.1 Cerebral Vasculature
The cerebral vasculature is one of the primary sites involved in the development of meningitis (Fig. 3e). The early activation of endothelial cells by inflammatory mediators, the disruption of endothelial function by meningeal pathogens, and the neutrophil influx into the CSF lead to several pathological changes. These include the disruption of the BBB, brain edema, loss of CBF autoregulation, and focal and global changes of CBF resulting in cerebral ischemia. Extensive cerebral infarction, resulting from vasculitis and coagulation disturbances, and circulatory failure due to septic shock may lead to acute death in bacterial meningitis [203].
4.1.1 Pathology
Subarachnoid space inflammation is a characteristic feature of acute bacterial meningitis. Arterial narrowing has been shown to be a predominant finding in patients with arterial complication. Vasculitis or vasospasm may lead to brain ischemia. Additionally, venous thrombosis has been observed in adult patients with pneumococcal meningitis [121, 204, 205]. In fatal cases of neonatal meningitis, inflammatory vasculitis is uniformly present, indicating that the cerebral vasculature of the neonate may be particularly susceptible to inflammatory damage, leading to notably severe structural damage to the neonatal brain [206].
4.1.2 Disruption of the Blood–Brain Barrier
In meningitis, the permeability of the BBB and BCSFB increases as the result of functionally relevant alterations induced by the disease process. Mainly paracellular leakage has been studied in this respect. The remodeling of the actin cytoskeleton, the reorganization of tight junctions, and the enzymatic degradation of tight junctions and basement membrane components participate in the permeabilization of the BBB [198, 207].
The meningeal pathogens are able to induce direct damage to endothelial or epithelial cells. In vitro cytotoxic damage to brain endothelial cells has been seen with S. pneumoniae, mainly mediated by pneumolysin [208–210], N. meningitidis, primarily mediated by NO [211], and E. coli K1 [212]. Pneumolysin was also shown to lead to astrocyte cytoskeleton remodeling in vitro [213], which might result in BBB disruption. In experimental GBS and S. pneumoniae meningitis, increased expression of the transcriptional repressor Snail1 has been found, leading to downregulation of tight junction components, like ZO-1, claudin-5, and occludin [214].
Signaling and effector molecules released during the inflammation process and changes caused by the interaction of inflammatory cells with the brain barriers also alter the function of cells constituting the BBB. A variety of cytokines and chemokines have been shown to induce tight junction and cytoskeleton rearrangements leading to BBB disruption [215–218].
MMPs increase BBB permeability by proteolytic breakdown of the basement membrane and tight junctions. In experimental meningococcal meningitis in rats, disruption of the BBB, increased intracranial pressure, and CSF pleocytosis were observed in parallel to an increase of MMP-9 activity in the CSF 6 h after infection [170]. Experimental inhibition of MMPs reduces occurrence of intracerebral bleeding and BBB breakdown in mice with meningococcal meningitis [219].
ROS and NO also contribute to BBB disruption via lipid peroxidation and oxidative damage to cell membrane proteins [220]. Neutralization of ROS and NO by radical scavengers in experimental meningitis prevented cerebral damage including BBB breakdown [221–224]. Additionally, ROS and NO lead to tight junction and cytoskeletal reorganization and MMP activation [225].
As a consequence of the increased BBB/BCSFB permeability, potentially harmful molecules found at high concentrations in the blood can accumulate in the CSF, unfavorably modifying the brain’s microenvironment. On the other hand, CSF penetration of antibiotics is facilitated by BBB opening, therefore improving the efficiency of therapy by increasing antibiotic CSF/serum ratio in comparison to noninflammatory conditions [226].
4.1.3 Alterations of Cerebral Blood Flow
Marked changes in CBF are observed during bacterial meningitis. Brain edema, obstructive hydrocephalus, meningitis-associated cerebritis, cerebral infarction and cerebral venous thrombosis, and status epilepticus may all lead to intracerebral hypertension [227, 228]. In the advanced disease stages, CBF is usually reduced [204, 229, 230]. Focal changes in the vasculature, loss of CBF autoregulation, intracranial pressure, and systemic hypotension perturb CBF in bacterial meningitis.
In humans, cerebrovascular changes in bacterial meningitis include segmental narrowing of vessels, irregularities of vessel walls, and arterial and venous thromboses [121, 231–233]. The middle and anterior cerebral arteries or the basilar artery are frequently affected by stroke associated with bacterial meningitis. Vessel narrowing results in an increased velocity of cerebral blood, an early predictor of cerebrovascular complications [121, 204]. Sequelae of vascular complications may result in hemiparesis or quadriparesis [206, 234, 235].
Disturbance of CBF autoregulation, combined with systemic hypotension, may lead to cerebral ischemia [236–240]. Alternatively, systemic hypertension may augment vasogenic edema and intracranial pressure [240, 241], ultimately limiting CBF. In addition to global cerebral hypoperfusion, vasculitis of large and small arteries traversing the inflamed subarachnoid space leads to regional hypoperfusion. This form of ischemic damage (Fig. 3a, b) may be responsible for permanent neurologic sequelae following bacterial meningitis [235, 236].
NO plays a crucial albeit complex role in modulating CBF during meningitis. Early in the disease, the vasodilatory effect of NO contributes to the hyperemia induced by the subarachnoid space inflammation. Later, NO generated in the vasculature provides some protection against ischemia by attenuating the effects of the progressive decline in CBF due to the production of vasoconstrictive factors (see later discussion).
ROS play a critical role in modulating CBF during meningitis [221, 242–244]. Generation of ROS is localized primarily to the cells constituting the subarachnoid and ventricular inflammation and the cerebral vasculature, as shown in infant rats with experimental meningitis [221]. In this model, the cerebral vasculature showed evidence of marked oxidative alterations, whereas oxidative damage to the brain parenchyma itself was not documented conclusively [245]. Antioxidant treatment prevents oxidative vascular damage, hyperemia, CBF decline, and rise of ICP in experimental pneumococcal and GBS meningitis [221, 222, 243, 244].
The reaction of NO with ROS produces peroxynitrite, a strong oxidant that exerts cytotoxic effects [220, 246]. Nitrotyrosine residues, a reaction product of peroxynitrite with proteins, were detected in the meninges, the cortical blood vessels, and inflammatory cells in the brains of patients with bacterial meningitis and corresponding animal models [220, 247]. Treatment with urate, a peroxynitrite scavenger, reduced the meningeal inflammation, BBB disruption, and intracranial hypertension [247].
Increased endothelin levels are found in the CSF of patients with bacterial meningitis [248]. This potent vasoconstrictive peptide is produced in the CNS by vascular endothelial cells, astrocytes, and neurons and participates in CBF regulation. Endothelin synthesis is triggered by cytokines, i.e., TNF-α [249–251], and inhibited by NO [252]. In experimental pneumococcal meningitis, an endothelin antagonist (bosentan) significantly prevented the reduction of CBF and attenuated the extent of cerebral ischemia [253].
4.2 Inner Ear
Unilateral or bilateral sensorineural hearing loss (SNHL) is the most common neurologic sequelae following bacterial meningitis and is found in 5–30 % of survivors [254–257], with S. pneumoniae causing the highest rate of sensorineural hearing loss [258–261]. Hearing loss during bacterial meningitis is progressive rather than abrupt, and its magnitude depends on the duration of untreated infection [255]. Hearing loss is the direct result of inflammation in the inner ear during the acute phase of the disease. Magnetic resonance imaging (MRI) studies in humans with meningitis have confirmed the inflammatory involvement of the inner ear [262]. In survivors of meningitis, progressive cochlear ossification and spiral ganglion loss are observed years after the disease [263, 264].
4.2.1 Pathology
During the acute stage of meningitis, suppurative labyrinthitis is usually observed in the human temporal bone of patients, often accompanied by purulent infiltrate in the perilymphatic duct [265]. Studies in experimental meningitis have shown that bacteria and inflammatory cells are present in the cochlea in the earliest stages of pneumococcal infection [266, 267]. Inflammatory infiltration of the cochlea progresses via the cochlear aqueduct to the perilymphatic space (scala tympani) and via the spiral ligament to the endolymphatic space [267–269]. Damage to the blood–labyrinth barrier, the hair cells, and the spiral ganglion is observed first at the base and later at the apex of the cochlea, corresponding to hearing loss first at high then at low frequencies [270]. The first signs of damage and hearing loss are observed for hair cells with a partial reversibility of associated hearing loss [270]. Later during the course of the disease, irreparable ultrastructural inner ear damage, especially the loss of spiral ganglion neuronal cells, is associated with severe to profound deafness [266, 271]. Toxic effects of the meningeal pathogen (e.g., pneumolysin from S. pneumoniae) and inflammatory mediators appear to be responsible for the cytopathic effects [272, 273]. In particular, the production of ROS and NO has been involved in the pathogenesis of cochlear damage and hearing loss in bacterial meningitis [270]. Treatment with peroxynitrite scavengers or antioxidants attenuated hearing loss and protected spiral ganglion neuronal cells [274, 275].
4.3 Central Nervous Tissue
4.3.1 Neurologic Sequelae
Bacterial meningitis causes damage to both cortical and subcortical brain structures. The neurologic sequelae resulting from brain damage include hearing impairment (see previous discussion), obstructive hydrocephalus, focal sensory-motor deficits, mental retardation, seizure disorders, and cortical blindness. Behavioral and cognitive sequelae in children and adults after bacterial meningitis are common [276–281]. Morbidity in patients surviving bacterial meningitis is usually higher with S. pneumoniae as causative pathogen [122, 254, 282].
Follow-up studies revealed that neurologic sequelae of childhood meningitis persist for more than 10 years, impacting the school performance of affected children [277, 279, 283, 284]. In adults tested 0.5–13.5 years after acute bacterial meningitis, 32 % of 155 meningitis survivors suffered from relevant impairment of psychomotor performance, speed of cognitive processes, and concentration and memory functions [280].
4.3.2 Pathology
Histological changes are often seen in a focal pattern (Fig. 3a, b). Infarctions in the frontal, temporal, and parietal lobes, the basal ganglia, the thalamus, as well as periventricular white matter injuries are found in newborns or children affected by bacterial meningitis, leading to cerebral atrophy and hydrocephalus [205, 285–287]. Structural changes were also seen in the temporal lobe and the limbic system of adult patients [281]. Neuronal loss is associated with a marked activation of astrocytes and microglia [288], as well as axonal injury [289, 290]. Furthermore, meningitis induced by gram-negative bacteria is often characterized by the development of brain abscesses [291, 292].
Apoptotic cell death has been observed in the dentate gyrus of the hippocampus during meningitis [221, 293, 294] (Fig. 3c) with the presence of condensed, fragmented nuclei (Fig. 3c, inset), and the detection of fragmented DNA and active caspase-3, an effector enzyme involved in the execution of programmed cell death [295]. A second form of hippocampal neuronal damage with morphologically distinct features (uniformly shrunken nuclei, clusters of damaged cells) may also be found in the lower blade of the dentate gyrus cell, spanning the entire width of the dentate gyrus band (Fig. 3d). This form of hippocampal damage is reminiscent of ischemia-related neuronal damage [296] and is the preferential pattern of neuronal injury observed in meningitis caused by GBS [216, 294].
The neuronal injury in the hippocampus is of particular significance because experimental data suggest that it is related to learning impairment following meningitis [148, 297, 298]. The observation of cell death primarily of immature progenitor cells in the subgranular zone of the dentate gyrus, cells necessary for the acquisition of new memory, strengthens this hypothesis [294, 295, 299]. Cognitive impairment and learning disabilities following meningitis may thus be reflected by damage to the dentate gyrus of the hippocampus [148, 297, 298, 300].
Confirming the findings in animal models, brain sections of patients who died from bacterial meningitis showed apoptotic neurons with cells staining for the active form of caspase-3 in the dentate gyrus [301]. Also, volumetric measurements of the hippocampus by MRI techniques showed unilateral and bilateral hippocampal atrophy in patients surviving meningitis [302], potentially reflecting the apoptotic loss of neurons observed by histopathology.
4.3.3 Brain Edema and Cerebral Herniation
Cerebral edema in bacterial meningitis may be the combined result of vasogenic, cytotoxic, interstitial, or osmotic edema [296]. Vasogenic cerebral edema is the consequence of increased BBB permeability, with the extravasation of plasma proteins into the brain parenchyma (see earlier discussion) and mainly affects the white matter [296]. Cytotoxic edema is an increase in intracellular water due to the intracellular accumulation of osmotically effective ions (i.e., sodium, potassium, or glutamate). Cytotoxic mechanisms include ischemia and the effect of excitatory amino acids (EAA) [296, 303, 304]. Cytotoxic edema affects the gray and white matter. The increase of CSF influx due to a more sustained production (increased blood flow in the choroid plexus) or a decreased resorption (increased CSF outflow resistance across the arachnoid villi system of the sagittal sinus) is the cause of interstitial edema [305]. Finally, osmotic edema may be caused by inappropriate secretion of antidiuretic hormone during the course of bacterial meningitis. The observed hypoosmolality of serum results in a net influx of water into the brain [296, 306].
Brain edema is an important contributor to the fatal outcome of bacterial meningitis [121, 307, 308]. Extensive brain edema leads to increased intracranial pressure culminating in the herniation of brain tissue and compression of the brainstem.
Aquaporins (AQP) are pore-forming membrane proteins regulating water homeostasis of the brain [309]. In cytotoxic edema, the transcellular influx of water across the BBB is facilitated by AQP-4, while in vasogenic and interstitial brain edema, the absence of AQP-4 leads to more severe brain edema by limiting outflow of CSF [296, 310]. In a mouse model of pneumococcal meningitis, AQP-4 was significantly upregulated and associated with increased cytotoxic brain edema [311]. AQP-4 upregulation was also found in the brain of a patient with bacterial meningitis [312], but not in the CSF of patients with bacterial meningitis [313].
4.3.4 Mediators of Cell Death in Neuronal Tissue
EAAs including glutamate induce neuronal apoptosis and necrosis and appear to mediate cellular injury in a variety of brain disorders. In bacterial meningitis, ischemia and EAA may cause direct neuronal toxicity as suggested from experimental models or clinical studies [288, 304]. In patients and experimental pneumococcal meningitis in rabbits, CSF glutamate concentrations were significantly elevated and correlated with the severity of the disease [314, 315]. The cause for increased concentrations of EAA in the brain during meningitis is still poorly understood, but may be related to BBB disruption. Alternatively, pore-forming cytolysins, like pneumolysin, may cause the release of glutamate from astrocytes, leading to subsequent glutamate-dependent synaptic damage [316]. Kynurenic acid, a nonselective inhibitor of the neurotoxic effect of EAA acting on NMDA receptors, significantly attenuated brain injury, both in the cortex and in the hippocampus in an infant rat model of neonatal meningitis [288, 304].
The intensity and duration of the inflammatory response in bacterial meningitis correlates with the development of neuronal damage. Meningeal pathogens interfere with the programmed cell death of immune cells, possibly prolonging inflammation. For example, N. meningitidis inhibits apoptosis in neutrophils [317] or macrophages [318]. Similarly GBS interfere with TLR-2-mediated programmed cell death in macrophages and microglia [319].
The brain is one of the most metabolically active organ in the body, therefore requiring a high rate of oxygen consumption. But because it possesses a high level of polyunsaturated fatty acid prone to lipid peroxidation, the brain is particularly vulnerable to oxidative damage caused by ROS. This is also exacerbated by the high abundance of redox-active metals (iron or copper) and the relative low levels of endogenous antioxidant [320]. The high concentration of mitochondria in cerebrovascular endothelial cells might also account for the sensitivity of the BBB to oxidant stressors [321]. The prevention of BBB disruption, attenuation of lipid peroxidation, reduction of ischemia in the brain parenchyma, and the reduced neuronal injury observed with the use of radical scavengers in experimental meningitis underline the role of ROS [322]. Despite these observations, a direct neurotoxic effect of ROS in bacterial meningitis has not been documented conclusively. Most of the beneficial effects of ROS scavengers in experimental meningitis may be primarily due to a beneficial effect at the level of the cerebral vasculature [245] (see previous discussion).
NO may be either neuroprotective or neurotoxic in bacterial meningitis. In its oxidized form, it can inactivate glutamate receptors, therefore reducing EAA-mediated excitotoxicity. In its reduced form, it reacts with superoxide and forms peroxynitrite, which is highly reactive and causes damage to DNA, lipid membranes, and proteins [323]. Caspase-3-dependent hippocampal apoptosis was reduced during experimental pneumococcal meningitis in mice lacking iNOS expression [324]. In contrast, iNOS inhibition by aminoguanidine exacerbates neuronal injury in experimental GBS meningitis [325].
MMPs may exert direct neurotoxic effects by degrading perineuronal components of the ECM, like laminin [326, 327], or the neural cell adhesion molecule NCAM [328]. Indirectly, the shedding of death receptors ligands (FasL, TNF-α) by MMPs can induce death in cells that express these ligands, including neurons [329]. Given as adjuvant therapy, MMP inhibitors reduced the extent of cortical damage, and combined inhibition of MMP and TACE led to a reduction in hippocampal apoptosis [148, 180, 182]. MMP-TACE inhibitors also improved learning capacity after experimental pneumococcal meningitis in infant rats [148].
Bacteria themselves may also directly damage neurons. Pneumococci have been shown to induce cell death in microglia or neurons in vitro [330]. Pneumolysin and superoxide produced by S. pneumoniae have been identified as the responsible neurotoxic mediators. In vitro, these toxins cause neuronal death via damage to mitochondria with the subsequent release of the proapoptotic mitochondrial factors cytochrome c and AIF [331, 332]. In experimental pneumococcal meningitis in rabbits, pneumolysin co-localized with apoptotic neurons of the hippocampus, and infection with pneumococcal mutants deficient for pneumolysin and superoxide production caused significantly less damage [331]. Pneumolysin was also shown to induce cochlear hair cell death in the rat [333] and endothelial cell damage in vitro [209, 210]. Other bacterial pore-forming toxins such as the beta hemolysin/cytolysin of GBS [334] are able to directly induce neuronal damage.
5 Conclusion
The pathophysiology of bacterial meningitis involves a complex interplay between the invasive pathogen, the anatomical barriers of the CNS, and the immune defense system of the host. In that context, the BCSFB and BBB play a primordial role, by restricting access to the brain. Meningeal pathogens have evolved sophisticated strategies to cross the otherwise hard-to-reach CNS compartment. Doing so, the successfully invading pathogens initiate a catastrophic cascade of events, involving the permeabilization of the barrier membranes, the massive recruitment of neutrophils, and a deleterious inflammatory reaction. Protecting barrier integrity by targeting pathophysiological processes that lead to its impairment, as demonstrated from experimental evidence using pharmacologic interventions including, e.g., radical scavengers or MMP inhibitors, is a promising strategy with the potential to reduce neurofunctional impairment in survivors of the disease.
Bibliography
van de Beek, Cabellos et al (2016) ESCMID guideline: diagnosis and treatment of bacterial meningitis. Clin Microbiol Infect 22 Suppl 3:S37–S62. doi: 10.1016/j.cmi.2016.01.007
McCullers JA (2006) Insights into the interaction between influenza virus and pneumococcus. Clin Microbiol Rev 19(3):571–582. doi:10.1128/CMR.00058-05, 19/3/571 [pii]
Avadhanula V, Rodriguez CA, Devincenzo JP, Wang Y, Webby RJ, Ulett GC, Adderson EE (2006) Respiratory viruses augment the adhesion of bacterial pathogens to respiratory epithelium in a viral species- and cell type-dependent manner. J Virol 80(4):1629–1636. doi:10.1128/JVI.80.4.1629-1636.2006, 80/4/1629 [pii]
Greenwood BM (1987) The epidemiology of acute bacterial meningitis in tropical Africa. In: Bacterial meningitis. Academic press, London, p 61–91
Mook-Kanamori BB, Geldhoff M, van der Poll T, van de Beek D (2011) Pathogenesis and pathophysiology of pneumococcal meningitis. Clin Microbiol Rev 24(3):557–591. doi:10.1128/CMR.00008-11, 24/3/557 [pii]
Steinfort C, Wilson R, Mitchell T, Feldman C, Rutman A, Todd H, Sykes D, Walker J, Saunders K, Andrew PW et al (1989) Effect of Streptococcus pneumoniae on human respiratory epithelium in vitro. Infect Immun 57(7):2006–2013
Hirst RA, Sikand KS, Rutman A, Mitchell TJ, Andrew PW, O’Callaghan C (2000) Relative roles of pneumolysin and hydrogen peroxide from Streptococcus pneumoniae in inhibition of ependymal ciliary beat frequency. Infect Immun 68(3):1557–1562
Kadioglu A, Taylor S, Iannelli F, Pozzi G, Mitchell TJ, Andrew PW (2002) Upper and lower respiratory tract infection by Streptococcus pneumoniae is affected by pneumolysin deficiency and differences in capsule type. Infect Immun 70(6):2886–2890
Rao VK, Krasan GP, Hendrixson DR, Dawid S, St Geme JW 3rd (1999) Molecular determinants of the pathogenesis of disease due to non-typable Haemophilus influenzae. FEMS Microbiol Rev 23(2):99–129, S0168-6445(98)00039-4 [pii]
Merz AJ, So M (2000) Interactions of pathogenic neisseriae with epithelial cell membranes. Annu Rev Cell Dev Biol 16:423–457
Wani JH, Gilbert JV, Plaut AG, Weiser JN (1996) Identification, cloning, and sequencing of the immunoglobulin A1 protease gene of Streptococcus pneumoniae. Infect Immun 64:3967–3974
Poulsen K, Reinholdt J, Jespersgaard C, Boye K, Brown TA, Hauge M, Kilian M (1998) A comprehensive genetic study of streptococcal immunoglobulin A1 proteases: evidence for recombination within and between species. Infect Immun 66(1):181–190
Vitovski S, Read RC, Sayers JR (1999) Invasive isolates of Neisseria meningitidis possess enhanced immunoglobulin A1 protease activity compared to colonizing strains. FASEB J 13(2):331–337
Callewaert L, Van Herreweghe JM, Vanderkelen L, Leysen S, Voet A, Michiels CW (2012) Guards of the great wall: bacterial lysozyme inhibitors. Trends Microbiol 20(10):501–510. doi:10.1016/j.tim.2012.06.005, S0966-842X(12)00116-3 [pii]
Boneca IG, Dussurget O, Cabanes D, Nahori MA, Sousa S, Lecuit M, Psylinakis E, Bouriotis V, Hugot JP, Giovannini M, Coyle A, Bertin J, Namane A, Rousselle JC, Cayet N, Prevost MC, Balloy V, Chignard M, Philpott DJ, Cossart P, Girardin SE (2007) A critical role for peptidoglycan N-deacetylation in Listeria evasion from the host innate immune system. Proc Natl Acad Sci U S A 104(3):997–1002. doi:10.1073/pnas.0609672104, 0609672104 [pii]
Davis KM, Akinbi HT, Standish AJ, Weiser JN (2008) Resistance to mucosal lysozyme compensates for the fitness deficit of peptidoglycan modifications by Streptococcus pneumoniae. PLoS Pathog 4(12):e1000241. doi:10.1371/journal.ppat.1000241
Davis KM, Weiser JN (2011) Modifications to the peptidoglycan backbone help bacteria to establish infection. Infect Immun 79(2):562–570. doi:10.1128/IAI.00651-10, IAI.00651-10 [pii]
Shakhnovich EA, King SJ, Weiser JN (2002) Neuraminidase expressed by Streptococcus pneumoniae desialylates the lipopolysaccharide of Neisseria meningitidis and Haemophilus influenzae: a paradigm for interbacterial competition among pathogens of the human respiratory tract. Infect Immun 70(12):7161–7164
Bogaardt C, van Tonder AJ, Brueggemann AB (2015) Genomic analyses of pneumococci reveal a wide diversity of bacteriocins – including pneumocyclicin, a novel circular bacteriocin. BMC Genomics 16(1):554. doi:10.1186/s12864-015-1729-4
Margolis E, Yates A, Levin BR (2010) The ecology of nasal colonization of Streptococcus pneumoniae, Haemophilus influenzae and Staphylococcus aureus: the role of competition and interactions with host’s immune response. BMC Microbiol 10:59. doi:10.1186/1471-2180-10-59
Hammerschmidt S (2006) Adherence molecules of pathogenic pneumococci. Curr Opin Microbiol 9(1):12–20. doi:10.1016/j.mib.2005.11.001, S1369-5274(05)00193-1 [pii]
Anderton JM, Rajam G, Romero-Steiner S, Summer S, Kowalczyk AP, Carlone GM, Sampson JS, Ades EW (2007) E-cadherin is a receptor for the common protein pneumococcal surface adhesin A (PsaA) of Streptococcus pneumoniae. Microb Pathog 42(5–6):225–236. doi:10.1016/j.micpath.2007.02.003, S0882-4010(07)00021-6 [pii]
Rowe HA, Griffiths NJ, Hill DJ, Virji M (2007) Co-ordinate action of bacterial adhesins and human carcinoembryonic antigen receptors in enhanced cellular invasion by capsulate serum resistant Neisseria meningitidis. Cell Microbiol 9(1):154–168. doi:10.1111/j.1462-5822.2006.00775.x, CMI775 [pii]
Weber A, Harris K, Lohrke S, Forney L, Smith AL (1991) Inability to express fimbriae results in impaired ability of Haemophilus influenzae b to colonize the nasopharynx. Infect Immun 59(12):4724–4728
Cotter SE, Yeo HJ, Juehne T, St Geme JW 3rd (2005) Architecture and adhesive activity of the Haemophilus influenzae Hsf adhesin. J Bacteriol 187(13):4656–4664. doi:10.1128/JB.187.13.4656-4664.2005, 187/13/4656 [pii]
Le Bouguenec C (2005) Adhesins and invasins of pathogenic Escherichia coli. Int J Med Microbiol 295(6–7):471–478
Sokurenko EV, Chesnokova V, Dykhuizen DE, Ofek I, Wu XR, Krogfelt KA, Struve C, Schembri MA, Hasty DL (1998) Pathogenic adaptation of Escherichia coli by natural variation of the FimH adhesin. Proc Natl Acad Sci U S A 95(15):8922–8926
Antao EM, Wieler LH, Ewers C (2009) Adhesive threads of extraintestinal pathogenic Escherichia coli. Gut Pathog 1(1):22. doi:10.1186/1757-4749-1-22, 1757-4749-1-22 [pii]
Pizarro-Cerda J, Cossart P (2006) Bacterial adhesion and entry into host cells. Cell 124(4):715–727. doi:10.1016/j.cell.2006.02.012, S0092-8674(06)00187-5 [pii]
Tazi A, Disson O, Bellais S, Bouaboud A, Dmytruk N, Dramsi S, Mistou MY, Khun H, Mechler C, Tardieux I, Trieu-Cuot P, Lecuit M, Poyart C (2010) The surface protein HvgA mediates group B streptococcus hypervirulence and meningeal tropism in neonates. J Exp Med 207(11):2313–2322. doi:10.1084/jem.20092594, jem.20092594 [pii]
Maisey HC, Doran KS, Nizet V (2008) Recent advances in understanding the molecular basis of group B Streptococcus virulence. Expert Rev Mol Med 10:e27. doi:10.1017/S1462399408000811, S1462399408000811 [pii]
Rinaudo CD, Rosini R, Galeotti CL, Berti F, Necchi F, Reguzzi V, Ghezzo C, Telford JL, Grandi G, Maione D (2010) Specific involvement of pilus type 2a in biofilm formation in group B Streptococcus. PLoS One 5(2):e9216. doi:10.1371/journal.pone.0009216
Mengaud J, Ohayon H, Gounon P, Mege RM, Cossart P (1996) E-cadherin is the receptor for internalin, a surface protein required for entry of L. monocytogenes into epithelial cells. Cell 84(6):923–932, S0092-8674(00)81070-3 [pii]
Lecuit M (2005) Understanding how Listeria monocytogenes targets and crosses host barriers. Clin Microbiol Infect 11(6):430–436. doi:10.1111/j.1469-0691.2005.01146.x, CLM1146 [pii]
Posfay-Barbe KM, Wald ER (2009) Listeriosis. Semin Fetal Neonatal Med 14(4):228–233. doi:10.1016/j.siny.2009.01.006, S1744-165X(09)00006-7 [pii]
Lecuit M, Nelson DM, Smith SD, Khun H, Huerre M, Vacher-Lavenu MC, Gordon JI, Cossart P (2004) Targeting and crossing of the human maternofetal barrier by Listeria monocytogenes: role of internalin interaction with trophoblast E-cadherin. Proc Natl Acad Sci U S A 101(16):6152–6157. doi:10.1073/pnas.0401434101, 0401434101 [pii]
Conner SD, Schmid SL (2003) Regulated portals of entry into the cell. Nature 422(6927):37–44. doi:10.1038/nature01451, nature01451 [pii]
Veiga E, Guttman JA, Bonazzi M, Boucrot E, Toledo-Arana A, Lin AE, Enninga J, Pizarro-Cerda J, Finlay BB, Kirchhausen T, Cossart P (2007) Invasive and adherent bacterial pathogens co-Opt host clathrin for infection. Cell Host Microbe 2(5):340–351. doi:10.1016/j.chom.2007.10.001, S1931-3128(07)00247-8 [pii]
Gradstedt H, Iovino F, Bijlsma JJ (2013) Streptococcus pneumoniae invades endothelial host cells via multiple pathways and is killed in a lysosome dependent manner. PLoS One 8(6):e65626. doi:10.1371/journal.pone.0065626
Ring A, Weiser JN, Tuomanen EI (1998) Pneumococcal trafficking across the blood–brain barrier. Molecular analysis of a novel bidirectional pathway. J Clin Invest 102(2):347–360. doi:10.1172/JCI2406
Radin JN, Orihuela CJ, Murti G, Guglielmo C, Murray PJ, Tuomanen EI (2005) Beta-Arrestin 1 participates in platelet-activating factor receptor-mediated endocytosis of Streptococcus pneumoniae. Infect Immun 73(12):7827–7835. doi:10.1128/IAI.73.12.7827-7835.2005, 73/12/7827 [pii]
Kim KJ, Elliott SJ, Di Cello F, Stins MF, Kim KS (2003) The K1 capsule modulates trafficking of E. coli-containing vacuoles and enhances intracellular bacterial survival in human brain microvascular endothelial cells. Cell Microbiol 5(4):245–252, 271 [pii]
Zhang JR, Mostov KE, Lamm ME, Nanno M, Shimida S, Ohwaki M, Tuomanen E (2000) The polymeric immunoglobulin receptor translocates pneumococci across human nasopharyngeal epithelial cells. Cell 102(6):827–837
Elm C, Braathen R, Bergmann S, Frank R, Vaerman JP, Kaetzel CS, Chhatwal GS, Johansen FE, Hammerschmidt S (2004) Ectodomains 3 and 4 of human polymeric Immunoglobulin receptor (hpIgR) mediate invasion of Streptococcus pneumoniae into the epithelium. J Biol Chem 279(8):6296–6304. doi:10.1074/jbc.M310528200, M310528200 [pii]
Gray-Owen SD, Blumberg RS (2006) CEACAM1: contact-dependent control of immunity. Nat Rev Immunol 6(6):433–446. doi:10.1038/nri1864, nri1864 [pii]
Sadarangani M, Pollard AJ, Gray-Owen SD (2011) Opa proteins and CEACAMs: pathways of immune engagement for pathogenic Neisseria. FEMS Microbiol Rev 35(3):498–514. doi:10.1111/j.1574-6976.2010.00260.x
Griffiths NJ, Bradley CJ, Heyderman RS, Virji M (2007) IFN-gamma amplifies NFkappaB-dependent Neisseria meningitidis invasion of epithelial cells via specific upregulation of CEA-related cell adhesion molecule 1. Cell Microbiol 9(12):2968–2983. doi:10.1111/j.1462-5822.2007.01038.x, CMI1038 [pii]
Nikitas G, Deschamps C, Disson O, Niault T, Cossart P, Lecuit M (2011) Transcytosis of Listeria monocytogenes across the intestinal barrier upon specific targeting of goblet cell accessible E-cadherin. J Exp Med 208(11):2263–2277. doi:10.1084/jem.20110560, jem.20110560 [pii]
Disson O, Grayo S, Huillet E, Nikitas G, Langa-Vives F, Dussurget O, Ragon M, Le Monnier A, Babinet C, Cossart P, Lecuit M (2008) Conjugated action of two species-specific invasion proteins for fetoplacental listeriosis. Nature 455(7216):1114–1118. doi:10.1038/nature07303, nature07303 [pii]
Clarke TB, Francella N, Huegel A, Weiser JN (2011) Invasive bacterial pathogens exploit TLR-mediated downregulation of tight junction components to facilitate translocation across the epithelium. Cell Host Microbe 9(5):404–414. doi:10.1016/j.chom.2011.04.012, S1931-3128(11)00134-X [pii]
Zwijnenburg PJ, van der Poll T, Florquin S, van Deventer SJ, Roord JJ, van Furth AM (2001) Experimental pneumococcal meningitis in mice: a model of intranasal infection. J Infect Dis 183(7):1143–1146
Hirst RA, Kadioglu A, O’Callaghan C, Andrew PW (2004) The role of pneumolysin in pneumococcal pneumonia and meningitis. Clin Exp Immunol 138(2):195–201
Attali C, Durmort C, Vernet T, Di Guilmi AM (2008) The interaction of Streptococcus pneumoniae with plasmin mediates transmigration across endothelial and epithelial monolayers by intercellular junction cleavage. Infect Immun 76(11):5350–5356. doi:10.1128/IAI.00184-08, IAI.00184-08 [pii]
Soriani M, Santi I, Taddei A, Rappuoli R, Grandi G, Telford JL (2006) Group B Streptococcus crosses human epithelial cells by a paracellular route. J Infect Dis 193(2):241–250. doi:10.1086/498982, JID35189 [pii]
Kostyukova NN, Volkova MO, Ivanova VV, Kvetnaya AS (1995) A study of pathogenic factors of Streptococcus pneumoniae strains causing meningitis. FEMS Immunol Med Microbiol 10(2):133–137
Knaust A, Weber MV, Hammerschmidt S, Bergmann S, Frosch M, Kurzai O (2007) Cytosolic proteins contribute to surface plasminogen recruitment of Neisseria meningitidis. J Bacteriol 189(8):3246–3255. doi:10.1128/JB.01966-06, JB.01966-06 [pii]
Virkola R, Lahteenmaki K, Eberhard T, Kuusela P, van Alphen L, Ullberg M, Korhonen TK (1996) Interaction of Haemophilus influenzae with the mammalian extracellular matrix. J Infect Dis 173(5):1137–1147
Bergmann S, Rohde M, Preissner KT, Hammerschmidt S (2005) The nine residue plasminogen-binding motif of the pneumococcal enolase is the major cofactor of plasmin-mediated degradation of extracellular matrix, dissolution of fibrin and transmigration. Thromb Haemost 94(2):304–311. doi:10.1267/THRO05020304, 05080304 [pii]
Steukers L, Glorieux S, Vandekerckhove AP, Favoreel HW, Nauwynck HJ (2012) Diverse microbial interactions with the basement membrane barrier. Trends Microbiol 20(3):147–155. doi:10.1016/j.tim.2012.01.001, S0966-842X(12)00002-9 [pii]
Pron B, Boumaila C, Jaubert F, Berche P, Milon G, Geissmann F, Gaillard JL (2001) Dendritic cells are early cellular targets of Listeria monocytogenes after intestinal delivery and are involved in bacterial spread in the host. Cell Microbiol 3(5):331–340, cmi120 [pii]
Carbonnelle E, Hill DJ, Morand P, Griffiths NJ, Bourdoulous S, Murillo I, Nassif X, Virji M (2009) Meningococcal interactions with the host. Vaccine 27(Suppl 2):B78–B89. doi:10.1016/j.vaccine.2009.04.069, S0264-410X(09)00636-7 [pii]
Virkola R, Brummer M, Rauvala H, van Alphen L, Korhonen TK (2000) Interaction of fimbriae of Haemophilus influenzae type B with heparin-binding extracellular matrix proteins. Infect Immun 68(10):5696–5701
Walport MJ (2001) Complement. First of two parts. N Engl J Med 344(14):1058–1066. doi:10.1056/NEJM200104053441406
Overturf GD (2003) Indications for the immunological evaluation of patients with meningitis. Clin Infect Dis 36(2):189–194
Dietzman DE, Fischer GW, Schoenknecht FD (1974) Neonatal Escherichia coli septicemia – bacterial counts in blood. J Pediatr 85(1):128–130
Adriani KS, Brouwer MC, Geldhoff M, Baas F, Zwinderman AH, Paul Morgan B, Harris CL, van der Ende A, van de Beek D (2013) Common polymorphisms in the complement system and susceptiblity to bacterial meningitis. J Infect 66:255–262. doi:10.1016/j.jinf.2012.10.008, S0163-4453(12)00294-0 [pii]
Brouwer MC, de Gans J, Heckenberg SG, Zwinderman AH, van der Poll T, van de Beek D (2009) Host genetic susceptibility to pneumococcal and meningococcal disease: a systematic review and meta-analysis. Lancet Infect Dis 9(1):31–44. doi:10.1016/S1473-3099(08)70261-5, S1473-3099(08)70261-5 [pii]
Albiger B, Johansson L, Jonsson AB (2003) Lipooligosaccharide-deficient Neisseria meningitidis shows altered pilus-associated characteristics. Infect Immun 71(1):155–162
Kim KS, Itabashi H, Gemski P, Sadoff J, Warren RL, Cross AS (1992) The K1 capsule is the critical determinant in the development of Escherichia coli meningitis in the rat. J Clin Invest 90(3):897–905
Hyams C, Camberlein E, Cohen JM, Bax K, Brown JS (2010) The Streptococcus pneumoniae capsule inhibits complement activity and neutrophil phagocytosis by multiple mechanisms. Infect Immun 78(2):704–715. doi:10.1128/IAI.00881-09, IAI.00881-09 [pii]
Kilian M, Reinholdt J, Lomholt H, Poulsen K, Frandsen EV (1996) Biological significance of IgA1 proteases in bacterial colonization and pathogenesis: critical evaluation of experimental evidence. APMIS 104(5):321–338
Vogel U, Hammerschmidt S, Frosch M (1996) Sialic acids of both the capsule and the sialylated lipooligosaccharide of Neisseria meningitis serogroup B are prerequisites for virulence of meningococci in the infant rat. Med Microbiol Immunol (Berl) 185(2):81–87
Peppoloni S, Ricci S, Orsi CF, Colombari B, De Santi MM, Messino M, Fabio G, Zanardi A, Righi E, Braione V, Tripodi S, Chiavolini D, Cintorino M, Zoli M, Oggioni MR, Blasi E, Pozzi G (2010) The encapsulated strain TIGR4 of Streptococcus pneumoniae is phagocytosed but is resistant to intracellular killing by mouse microglia. Microbes Infect 12(12–13):990–1001. doi:10.1016/j.micinf.2010.06.010
Agarwal V, Hammerschmidt S, Malm S, Bergmann S, Riesbeck K, Blom AM (2012) Enolase of Streptococcus pneumoniae binds human complement inhibitor C4b-binding protein and contributes to complement evasion. J Immunol 189(7):3575–3584. doi:10.4049/jimmunol.1102934, jimmunol.1102934 [pii]
Santi I, Scarselli M, Mariani M, Pezzicoli A, Masignani V, Taddei A, Grandi G, Telford JL, Soriani M (2007) BibA: a novel immunogenic bacterial adhesin contributing to group B Streptococcus survival in human blood. Mol Microbiol 63(3):754–767. doi:10.1111/j.1365-2958.2006.05555.x, MMI5555 [pii]
Zipfel PF, Skerka C, Hellwage J, Jokiranta ST, Meri S, Brade V, Kraiczy P, Noris M, Remuzzi G (2002) Factor H family proteins: on complement, microbes and human diseases. Biochem Soc Trans 30(Pt 6):971–978. doi:10.1042/bst0300971
Maruvada R, Prasadarao NV, Rubens CE (2009) Acquisition of factor H by a novel surface protein on group B Streptococcus promotes complement degradation. Faseb J 23(11):3967–3977. doi:10.1096/fj.09-138149, fj.09-138149 [pii]
Lewis LA, Carter M, Ram S (2012) The relative roles of factor H binding protein, neisserial surface protein A, and lipooligosaccharide sialylation in regulation of the alternative pathway of complement on meningococci. J Immunol 188(10):5063–5072. doi:10.4049/jimmunol.1103748, jimmunol.1103748 [pii]
Singh B, Su YC, Riesbeck K (2010) Vitronectin in bacterial pathogenesis: a host protein used in complement escape and cellular invasion. Mol Microbiol 78(3):545–560. doi:10.1111/j.1365-2958.2010.07373.x
Stavru F, Archambaud C, Cossart P (2011) Cell biology and immunology of Listeria monocytogenes infections: novel insights. Immunol Rev 240(1):160–184. doi:10.1111/j.1600-065X.2010.00993.x
Moxon ER, Murphy PA (1978) Haemophilus influenzae bacteremia and meningitis resulting from survival of a single organism. Proc Natl Acad Sci U S A 75(3):1534–1536
Redzic Z (2011) Molecular biology of the blood–brain and the blood-cerebrospinal fluid barriers: similarities and differences. Fluids Barriers CNS 8(1):3. doi:10.1186/2045-8118-8-3, 2045-8118-8-3 [pii]
Kim KS (2002) Strategy of Escherichia coli for crossing the blood–brain barrier. J Infect Dis 186(Suppl 2):S220–S224
Moxon ER, Ostrow PT (1977) Haemophilus influenzae meningitis in infant rats: role of bacteremia in pathogenesis of age-dependent inflammatory responses in cerebrospinal fluid. J Infect Dis 135(2):303–307
Kim KS (2008) Mechanisms of microbial traversal of the blood–brain barrier. Nat Rev Microbiol 6(8):625–634. doi:10.1038/nrmicro1952, nrmicro1952 [pii]
Drevets DA, Leenen PJ, Greenfield RA (2004) Invasion of the central nervous system by intracellular bacteria. Clin Microbiol Rev 17(2):323–347
Drevets DA, Jelinek TA, Freitag NE (2001) Listeria monocytogenes-infected phagocytes can initiate central nervous system infection in mice. Infect Immun 69(3):1344–1350. doi:10.1128/IAI.69.3.1344-1350.2001
Drevets DA, Dillon MJ, Schawang JS, Van Rooijen N, Ehrchen J, Sunderkotter C, Leenen PJ (2004) The Ly-6Chigh monocyte subpopulation transports Listeria monocytogenes into the brain during systemic infection of mice. J Immunol 172(7):4418–4424
Nassif X, Bourdoulous S, Eugene E, Couraud PO (2002) How do extracellular pathogens cross the blood–brain barrier? Trends Microbiol 10(5):227–232
Wang MH, Kim KS (2013) Cytotoxic necrotizing factor 1 contributes to Escherichia coli meningitis. Toxins (Basel) 5(11):2270–2280. doi:10.3390/toxins5112270
Kim BY, Kang J, Kim KS (2005) Invasion processes of pathogenic Escherichia coli. Int J Med Microbiol 295(6–7):463–470
Orihuela CJ, Mahdavi J, Thornton J, Mann B, Wooldridge KG, Abouseada N, Oldfield NJ, Self T, Ala’Aldeen DA, Tuomanen EI (2009) Laminin receptor initiates bacterial contact with the blood brain barrier in experimental meningitis models. J Clin Invest 119(6):1638–1646. doi:10.1172/JCI36759, 36759 [pii]
Abouseada NM, Assafi MS, Mahdavi J, Oldfield NJ, Wheldon LM, Wooldridge KG, Ala’Aldeen DA (2012) Mapping the laminin receptor binding domains of Neisseria meningitidis PorA and Haemophilus influenzae OmpP2. PLoS One 7(9):e46233. doi:10.1371/journal.pone.0046233
Chung JW, Hong SJ, Kim KJ, Goti D, Stins MF, Shin S, Dawson VL, Dawson TM, Kim KS (2003) 37-kDa laminin receptor precursor modulates cytotoxic necrotizing factor 1-mediated RhoA activation and bacterial uptake. J Biol Chem 278(19):16857–16862. doi:10.1074/jbc.M301028200
Kim KJ, Chung JW, Kim KS (2005) 67-kDa laminin receptor promotes internalization of cytotoxic necrotizing factor 1-expressing Escherichia coli K1 into human brain microvascular endothelial cells. J Biol Chem 280(2):1360–1368. doi:10.1074/jbc.M410176200
Iovino F, Molema G, Bijlsma JJ (2014) Platelet endothelial cell adhesion molecule-1, a putative receptor for the adhesion of Streptococcus pneumoniae to the vascular endothelium of the blood–brain barrier. Infect Immun 82(9):3555–3566. doi:10.1128/IAI.00046-14
Iovino F, Molema G, Bijlsma JJ (2014) Streptococcus pneumoniae interacts with pIgR expressed by the brain microvascular endothelium but does not co-localize with PAF receptor. PLoS One 9(5):e97914. doi:10.1371/journal.pone.0097914
Coureuil M, Join-Lambert O, Lecuyer H, Bourdoulous S, Marullo S, Nassif X (2012) Mechanism of meningeal invasion by Neisseria meningitidis. Virulence 3(2):164–172. doi:10.4161/viru.18639, 18639 [pii]
Maisey HC, Hensler M, Nizet V, Doran KS (2007) Group B streptococcal pilus proteins contribute to adherence to and invasion of brain microvascular endothelial cells. J Bacteriol 189(4):1464–1467. doi:10.1128/JB.01153-06, JB.01153-06 [pii]
Tenenbaum T, Bloier C, Adam R, Reinscheid DJ, Schroten H (2005) Adherence to and invasion of human brain microvascular endothelial cells are promoted by fibrinogen-binding protein FbsA of Streptococcus agalactiae. Infect Immun 73(7):4404–4409. doi:10.1128/IAI.73.7.4404-4409.2005, 73/7/4404 [pii]
Banerjee A, Kim BJ, Carmona EM, Cutting AS, Gurney MA, Carlos C, Feuer R, Prasadarao NV, Doran KS (2011) Bacterial Pili exploit integrin machinery to promote immune activation and efficient blood–brain barrier penetration. Nat Commun 2:462. doi:10.1038/ncomms1474, ncomms1474 [pii]
Seo HS, Mu R, Kim BJ, Doran KS, Sullam PM (2012) Binding of glycoprotein Srr1 of Streptococcus agalactiae to fibrinogen promotes attachment to brain endothelium and the development of meningitis. PLoS Pathog 8(10):e1002947. doi:10.1371/journal.ppat.1002947
Tenenbaum T, Spellerberg B, Adam R, Vogel M, Kim KS, Schroten H (2007) Streptococcus agalactiae invasion of human brain microvascular endothelial cells is promoted by the laminin-binding protein Lmb. Microbes Infect 9(6):714–720. doi:10.1016/j.micinf.2007.02.015
Asmat TM, Agarwal V, Saleh M, Hammerschmidt S (2014) Endocytosis of Streptococcus pneumoniae via the polymeric immunoglobulin receptor of epithelial cells relies on clathrin and caveolin dependent mechanisms. Int J Med Microbiol 304(8):1233–1246. doi:10.1016/j.ijmm.2014.10.001
Uchiyama S, Carlin AF, Khosravi A, Weiman S, Banerjee A, Quach D, Hightower G, Mitchell TJ, Doran KS, Nizet V (2009) The surface-anchored NanA protein promotes pneumococcal brain endothelial cell invasion. J Exp Med 206(9):1845–1852. doi:10.1084/jem.20090386, jem.20090386 [pii]
Banerjee A, Van Sorge NM, Sheen TR, Uchiyama S, Mitchell TJ, Doran KS (2010) Activation of brain endothelium by pneumococcal neuraminidase NanA promotes bacterial internalization. Cell Microbiol 12(11):1576–1588. doi:10.1111/j.1462-5822.2010.01490.x
Cundell DR, Gerard NP, Gerard C, Idanpaan-Heikkila I, Tuomanen EI (1995) Streptococcus pneumoniae anchor to activated human cells by the receptor for platelet-activating factor. Nature 377(6548):435–438
Reddy MA, Wass CA, Kim KS, Schlaepfer DD, Prasadarao NV (2000) Involvement of focal adhesion kinase in Escherichia coli invasion of human brain microvascular endothelial cells. Infect Immun 68(11):6423–6430
Prasadarao NV, Wass CA, Stins MF, Shimada H, Kim KS (1999) Outer membrane protein A-promoted actin condensation of brain microvascular endothelial cells is required for Escherichia coli invasion. Infect Immun 67(11):5775–5783
Khan NA, Kim Y, Shin S, Kim KS (2007) FimH-mediated Escherichia coli K1 invasion of human brain microvascular endothelial cells. Cell Microbiol 9(1):169–178. doi:10.1111/j.1462-5822.2006.00779.x
Maruvada R, Kim KS (2012) IbeA and OmpA of Escherichia coli K1 exploit Rac1 activation for invasion of human brain microvascular endothelial cells. Infect Immun 80(6):2035–2041. doi:10.1128/IAI.06320-11
Zou Y, He L, Huang SH (2006) Identification of a surface protein on human brain microvascular endothelial cells as vimentin interacting with Escherichia coli invasion protein IbeA. Biochem Biophys Res Commun 351(3):625–630. doi:10.1016/j.bbrc.2006.10.091
Zou Y, He L, Wu CH, Cao H, Xie ZH, Ouyang Y, Wang Y, Jong A, Huang SH (2007) PSF is an IbeA-binding protein contributing to meningitic Escherichia coli K1 invasion of human brain microvascular endothelial cells. Med Microbiol Immunol 196(3):135–143. doi:10.1007/s00430-006-0034-x
Teng CH, Tseng YT, Maruvada R, Pearce D, Xie Y, Paul-Satyaseela M, Kim KS (2010) NlpI contributes to Escherichia coli K1 strain RS218 interaction with human brain microvascular endothelial cells. Infect Immun 78(7):3090–3096. doi:10.1128/IAI.00034-10
Prasadarao NV, Wass CA, Huang SH, Kim KS (1999) Identification and characterization of a novel Ibe10 binding protein that contributes to Escherichia coli invasion of brain microvascular endothelial cells. Infect Immun 67(3):1131–1138
Hoffman JA, Badger JL, Zhang Y, Huang SH, Kim KS (2000) Escherichia coli K1 aslA contributes to invasion of brain microvascular endothelial cells in vitro and in vivo. Infect Immun 68(9):5062–5067
Parthasarathy G, Yao Y, Kim KS (2007) Flagella promote Escherichia coli K1 association with and invasion of human brain microvascular endothelial cells. Infect Immun 75(6):2937–2945. doi:10.1128/IAI.01543-06
Mu R, Kim BJ, Paco C, Del Rosario Y, Courtney HS, Doran KS (2014) Identification of a group B streptococcal fibronectin binding protein, SfbA, that contributes to invasion of brain endothelium and development of meningitis. Infect Immun 82(6):2276–2286. doi:10.1128/IAI.01559-13
Unkmeir A, Latsch K, Dietrich G, Wintermeyer E, Schinke B, Schwender S, Kim KS, Eigenthaler M, Frosch M (2002) Fibronectin mediates Opc-dependent internalization of Neisseria meningitidis in human brain microvascular endothelial cells. Mol Microbiol 46(4):933–946
Disson O, Lecuit M (2012) Targeting of the central nervous system by Listeria monocytogenes. Virulence 3(2):213–221. doi:10.4161/viru.19586, 19586 [pii]
Kastenbauer S, Pfister HW (2003) Pneumococcal meningitis in adults: spectrum of complications and prognostic factors in a series of 87 cases. Brain 126(Pt 5):1015–1025
van de Beek D, de Gans J, Spanjaard L, Weisfelt M, Reitsma JB, Vermeulen M (2004) Clinical features and prognostic factors in adults with bacterial meningitis. N Engl J Med 351(18):1849–1859
Marra A, Brigham D (2001) Streptococcus pneumoniae causes experimental meningitis following intranasal and otitis media infections via a nonhematogenous route. Infect Immun 69(12):7318–7325
van Ginkel FW, McGhee JR, Watt JM, Campos-Torres A, Parish LA, Briles DE (2003) Pneumococcal carriage results in ganglioside-mediated olfactory tissue infection. Proc Natl Acad Sci U S A 100(24):14363–14367. doi:10.1073/pnas.2235844100, 2235844100 [pii]
Sjolinder H, Jonsson AB (2010) Olfactory nerve – a novel invasion route of Neisseria meningitidis to reach the meninges. PLoS One 5(11):e14034. doi:10.1371/journal.pone.0014034
Konsman JP, Drukarch B, Van Dam AM (2007) (Peri)vascular production and action of pro-inflammatory cytokines in brain pathology. Clin Sci 112(1):1–25. doi:10.1042/CS20060043
Kim YS, Honkaniemi J, Sharp FR, Täuber MG (2004) Expression of proinflammatory cytokines tumor necrosis factor-alpha and interleukin-1beta in the brain during experimental group B streptococcal meningitis. Brain Res Mol Brain Res 128(1):95–102. doi:10.1016/j.molbrainres.2004.06.009
Polfliet MM, Zwijnenburg PJ, van Furth AM, van der Poll T, Dopp EA, Renardel de Lavalette C, van Kesteren-Hendrikx EM, van Rooijen N, Dijkstra CD, van den Berg TK (2001) Meningeal and perivascular macrophages of the central nervous system play a protective role during bacterial meningitis. J Immunol 167(8):4644–4650
de Vos AF, Dessing MC, Lammers AJ, de Porto AP, Florquin S, de Boer OJ, de Beer R, Terpstra S, Bootsma HJ, Hermans PW, van ’t Veer C, van der Poll T (2015) The polysaccharide capsule of Streptococcus pneumonia partially impedes MyD88-mediated immunity during pneumonia in mice. PLoS One 10(2):e0118181. doi:10.1371/journal.pone.0118181
Hanamsagar R, Hanke ML, Kielian T (2012) Toll-like receptor (TLR) and inflammasome actions in the central nervous system. Trends Immunol 33(7):333–342. doi:10.1016/j.it.2012.03.001, S1471-4906(12)00051-8 [pii]
Koedel U (2009) Toll-like receptors in bacterial meningitis. Curr Top Microbiol Immunol 336:15–40. doi:10.1007/978-3-642-00549-7_2
Hoegen T, Tremel N, Klein M, Angele B, Wagner H, Kirschning C, Pfister HW, Fontana A, Hammerschmidt S, Koedel U (2011) The NLRP3 inflammasome contributes to brain injury in pneumococcal meningitis and is activated through ATP-dependent lysosomal cathepsin B release. J Immunol 187(10):5440–5451. doi:10.4049/jimmunol.1100790, jimmunol.1100790 [pii]
Neal JW, Gasque P (2013) How does the brain limit the severity of inflammation and tissue injury during bacterial meningitis? J Neuropathol Exp Neurol 72(5):370–385. doi:10.1097/NEN.0b013e3182909f2f
Kigerl KA, de Rivero Vaccari JP, Dietrich WD, Popovich PG, Keane RW (2014) Pattern recognition receptors and central nervous system repair. Exp Neurol 258:5–16. doi:10.1016/j.expneurol.2014.01.001
Geldhoff M, Mook-Kanamori BB, Brouwer MC, Valls Seron M, Baas F, van der Ende A, van de Beek D (2013) Genetic variation in inflammasome genes is associated with outcome in bacterial meningitis. Immunogenetics 65(1):9–16. doi:10.1007/s00251-012-0653-x
Sanders MS, van Well GT, Ouburg S, Morre SA, van Furth AM (2012) Toll-like receptor 9 polymorphisms are associated with severity variables in a cohort of meningococcal meningitis survivors. BMC Infect Dis 12:112. doi:10.1186/1471-2334-12-112, 1471-2334-12-112 [pii]
van Well GT, Sanders MS, Ouburg S, van Furth AM, Morre SA (2012) Polymorphisms in toll-like receptors 2, 4, and 9 are highly associated with hearing loss in survivors of bacterial meningitis. PLoS One 7(5):e35837. doi:10.1371/journal.pone.0035837, PONE-D-11-19179 [pii]
Canton J, Neculai D, Grinstein S (2013) Scavenger receptors in homeostasis and immunity. Nat Rev Immunol 13(9):621–634. doi:10.1038/nri3515
Iovino F, Orihuela CJ, Moorlag HE, Molema G, Bijlsma JJ (2013) Interactions between blood-borne Streptococcus pneumoniae and the blood–brain barrier preceding meningitis. PLoS One 8(7):e68408. doi:10.1371/journal.pone.0068408
Täuber MG, Moser B (1999) Cytokines and chemokines in meningeal inflammation: biology and clinical implications. Clin Infect Dis 28(1):1–11; quiz 12
Kastenbauer S, Angele B, Sporer B, Pfister HW, Koedel U (2005) Patterns of protein expression in infectious meningitis: a cerebrospinal fluid protein array analysis. J Neuroimmunol 164(1–2):134–139
Zwijnenburg PJ, van der Poll T, Roord JJ, van Furth AM (2006) Chemotactic factors in cerebrospinal fluid during bacterial meningitis. Infect Immun 74(3):1445–1451. doi:10.1128/IAI.74.3.1445-1451.2006, 74/3/1445 [pii]
Klein M, Paul R, Angele B, Popp B, Pfister HW, Koedel U (2006) Protein expression pattern in experimental pneumococcal meningitis. Microbes Infect 8(4):974–983. doi:10.1016/j.micinf.2005.10.013, S1286-4579(05)00381-3 [pii]
Mogensen TH, Paludan SR, Kilian M, Ostergaard L (2006) Live Streptococcus pneumoniae, Haemophilus influenzae, and Neisseria meningitidis activate the inflammatory response through toll-like receptors 2, 4, and 9 in species-specific patterns. J Leukoc Biol 80(2):267–277. doi:10.1189/jlb.1105626, jlb.1105626 [pii]
Fowler MI, Weller RO, Heckels JE, Christodoulides M (2004) Different meningitis-causing bacteria induce distinct inflammatory responses on interaction with cells of the human meninges. Cell Microbiol 6(6):555–567
Tietze K, Dalpke A, Morath S, Mutters R, Heeg K, Nonnenmacher C (2006) Differences in innate immune responses upon stimulation with gram-positive and gram-negative bacteria. J Periodontal Res 41(5):447–454. doi:10.1111/j.1600-0765.2006.00890.x, JRE890 [pii]
Diab A, Zhu J, Linquist L, Wretlind B, Bakhiet M, Link H (1997) Haemophilus influenzae and streptococcus pneumoniae induce different intracerebral mRNA cytokine patterns during the course of experimental bacterial meningitis. Clin Exp Immunol 109:233–241
Leib SL, Clements JM, Lindberg RL, Heimgartner C, Loeffler JM, Pfister LA, Tauber MG, Leppert D (2001) Inhibition of matrix metalloproteinases and tumour necrosis factor alpha converting enzyme as adjuvant therapy in pneumococcal meningitis. Brain 124(Pt 9):1734–1742
Tureen J (1995) Effect of recombinant human tumor necrosis factor-alpha on cerebral oxygen uptake, cerebrospinal fluid lactate, and cerebral blood flow in the rabbit: role of nitric oxide. J Clin Invest 95(3):1086–1091
Rosenberg GA, Estrada EY, Dencoff JE, Stetler-Stevenson WG (1995) Tumor necrosis factor-alpha-induced gelatinase B causes delayed opening of the blood–brain barrier: an expanded therapeutic window. Brain Res 703(1–2):151–155
Andersson PB, Perry VH, Gordon S (1992) Intracerebral injection of proinflammatory cytokines or leukocyte chemotaxins induces minimal myelomonocytic cell recruitment to the parenchyma of the central nervous system. J Exp Med 176(1):255–259
Liu T, Clark RK, McDonnell PC, Young PR, White RF, Barone FC, Feuerstein GZ (1994) Tumor necrosis factor-alpha expression in ischemic neurons. Stroke 25(7):1481–1488
Glabinski A, Krajewski S, Rafalowska J (1998) Tumor necrosis factor-alpha induced pathology in the rat brain: characterization of stereotaxic injection model. Folia Neuropathol 36(1):52–62
Candelario-Jalil E, Taheri S, Yang Y, Sood R, Grossetete M, Estrada EY, Fiebich BL, Rosenberg GA (2007) Cyclooxygenase inhibition limits blood–brain barrier disruption following intracerebral injection of tumor necrosis factor-alpha in the rat. J Pharmacol Exp Ther 323(2):488–498. doi:10.1124/jpet.107.127035, jpet.107.127035 [pii]
Costa A, Gupta R, Signorino G, Malara A, Cardile F, Biondo C, Midiri A, Galbo R, Trieu-Cuot P, Papasergi S, Teti G, Henneke P, Mancuso G, Golenbock DT, Beninati C (2012) Activation of the NLRP3 inflammasome by group B streptococci. J Immunol 188(4):1953–1960. doi:10.4049/jimmunol.1102543, jimmunol.1102543 [pii]
Opitz B, Puschel A, Schmeck B, Hocke AC, Rosseau S, Hammerschmidt S, Schumann RR, Suttorp N, Hippenstiel S (2004) Nucleotide-binding oligomerization domain proteins are innate immune receptors for internalized Streptococcus pneumoniae. J Biol Chem 279(35):36426–36432
Mitchell AJ, Yau B, McQuillan JA, Ball HJ, Too LK, Abtin A, Hertzog P, Leib SL, Jones CA, Gerega SK, Weninger W, Hunt NH (2012) Inflammasome-dependent IFN-gamma drives pathogenesis in Streptococcus pneumoniae meningitis. J Immunol 189(10):4970–4980. doi:10.4049/jimmunol.1201687, jimmunol.1201687 [pii]
Waage A, Halstensen A, Shalaby R (1998) Local production of tumor necrosis factor, interleukin-l, and interleukin-6 in meningococcal meningitis. J Exp Med 170:1859–1864
Ramilo O, Saez-Llorens X, Mertsola J, Jafari H, Olsen KD, Hansen EJ, Yoshinaga M, Ohkawara S, Nariuchi H, McCracken GH Jr (1990) Tumor necrosis factor alpha/cachectin and interleukin 1 beta initiate meningeal inflammation. J Exp Med 172(2):497–507
Rusconi F, Parizzi F, Garlaschi L, Assael BM, Sironi M, Ghezzi P, Mantovani A (1991) Interleukin 6 activity in infants and children with bacterial meningitis. The collaborative study on meningitis. Pediatr Infect Dis J 10(2):117–121
Hirano T, Akira S, Taga T, Kishimoto T (1990) Biological and clinical aspects of interleukin-6. Immunol Today 11:443–445
Cohen MC, Cohen S (1996) Cytokine function, a study in biologic diversity. Am J Clin Pathol 105:589–598
Spanaus KS, Nadal D, Pfister HW, Seebach J, Widmer U, Frei K, Gloor S, Fontana A (1997) C-X-C and C-C chemokines are expressed in the cerebrospinal fluid in bacterial meningitis and mediate chemotactic activity on peripheral blood-derived polymorphonuclear and mononuclear cells in vitro. J Immunol 158(4):1956–1964
Zwijnenburg PJ, de Bie HM, Roord JJ, van der Poll T, van Furth AM (2003) Chemotactic activity of CXCL5 in cerebrospinal fluid of children with bacterial meningitis. J Neuroimmunol 145(1–2):148–153
Paris MM, Hickey SM, Trujillo M, Ahmed A, Olsen K, McCracken GH Jr (1997) The effect of interleukin-10 on meningeal inflammation in experimental bacterial meningitis. J Infect Dis 176(5):1239–1246
Pfister HW, Frei K, Ottnad B, Koedel U, Tomasz A, Fontana A (1992) Transforming growth factor beta 2 inhibits cerebrovascular changes and brain edema formation in the tumor necrosis factor alpha-independent early phase of experimental pneumococcal meningitis. J Exp Med 176(1):265–268
Cooley ID, Chauhan VS, Donneyz MA, Marriott I (2014) Astrocytes produce IL-19 in response to bacterial challenge and are sensitive to the immunosuppressive effects of this IL-10 family member. Glia 62(5):818–828. doi:10.1002/glia.22644
Grandgirard D, Gaumann R, Coulibaly B, Dangy JP, Sie A, Junghanss T, Schudel H, Pluschke G, Leib SL (2013) The causative pathogen determines the inflammatory profile in cerebrospinal fluid and outcome in patients with bacterial meningitis. Mediators Inflamm 2013:312476. doi:10.1155/2013/312476
Too LK, Ball HJ, McGregor IS, Hunt NH (2014) The pro-inflammatory cytokine interferon-gamma is an important driver of neuropathology and behavioural sequelae in experimental pneumococcal meningitis. Brain Behav Immun 40:252–268. doi:10.1016/j.bbi.2014.02.020
Kieseier BC, Paul R, Koedel U, Seifert T, Clements JM, Gearing AJ, Pfister HW, Hartung HP (1999) Differential expression of matrix metalloproteinases in bacterial meningitis. Brain 122(Pt 8):1579–1587
Paul R, Lorenzl S, Koedel U, Sporer B, Vogel U, Frosch M, Pfister HW (1998) Matrix metalloproteinases contribute to the blood–brain barrier disruption during bacterial meningitis. Ann Neurol 44(4):592–600
Leppert D, Lindberg RL, Kappos L, Leib SL (2001) Matrix metalloproteinases: multifunctional effectors of inflammation in multiple sclerosis and bacterial meningitis. Brain Res Brain Res Rev 36(2–3):249–257
Green JA, Thi Hong Chau T, Farrar JJ, Friedland JS, Thwaites GE (2011) CNS infection, CSF matrix metalloproteinase concentrations, and clinical/laboratory features. Neurology 76(6):577–579. doi:10.1212/WNL.0b013e31820b7600, 76/6/577 [pii]
Tsai HC, Shi MH, Lee SS, Wann SR, Tai MH, Chen YS (2011) Expression of matrix metalloproteinases and their tissue inhibitors in the serum and cerebrospinal fluid of patients with meningitis. Clin Microbiol Infect 17(5):780–784. doi:10.1111/j.1469-0691.2010.03393.x
Lindberg RL, Sorsa T, Tervahartiala T, Hoffmann F, Mellanen L, Kappos L, Schaad UB, Leib SL, Leppert D (2006) Gelatinase B [matrix metalloproteinase (MMP)-9] and collagenases (MMP-8/-13) are upregulated in cerebrospinal fluid during aseptic and bacterial meningitis in children. Neuropathol Appl Neurobiol 32(3):304–317
Sulik A, Chyczewski L (2008) Immunohistochemical analysis of MMP-9, MMP-2 and TIMP-1, TIMP-2 expression in the central nervous system following infection with viral and bacterial meningitis. Folia Histochem Cytobiol 46(4):437–442. doi:10.2478/v10042-008-0058-8, 468Q020105H97207 [pii]
Roine I, Pelkonen T, Lauhio A, Lappalainen M, Cruzeiro ML, Bernardino L, Tervahartiala T, Sorsa T, Peltola H (2015) Changes in MMP-9 and TIMP-1 concentrations in cerebrospinal fluid after 1 week of treatment of childhood bacterial meningitis. J Clin Microbiol 53(7):2340–2342. doi:10.1128/JCM.00714-15
Leppert D, Leib SL, Grygar C, Miller KM, Schaad UB, Hollander GA (2000) Matrix metalloproteinase (MMP)-8 and MMP-9 in cerebrospinal fluid during bacterial meningitis: association with blood–brain barrier damage and neurological sequelae. Clin Infect Dis 31(1):80–84
Sellner J, Leib SL (2006) In bacterial meningitis cortical brain damage is associated with changes in parenchymal MMP-9/TIMP-1 ratio and increased collagen type IV degradation. Neurobiol Dis 21(3):647–656
Meli DN, Loeffler JM, Baumann P, Neumann U, Buhl T, Leppert D, Leib SL (2004) In pneumococcal meningitis a novel water-soluble inhibitor of matrix metalloproteinases and TNF-alpha converting enzyme attenuates seizures and injury of the cerebral cortex. J Neuroimmunol 151(1–2):6–11. doi:10.1016/j.jneuroim.2004.01.026
Liechti FD, Bachtold F, Grandgirard D, Leppert D, Leib SL (2015) The matrix metalloproteinase inhibitor RS-130830 attenuates brain injury in experimental pneumococcal meningitis. J Neuroinflammation 12:43. doi:10.1186/s12974-015-0257-0
Liechti FD, Grandgirard D, Leppert D, Leib SL (2014) Matrix metalloproteinase inhibition lowers mortality and brain injury in experimental pneumococcal meningitis. Infect Immun 82(4):1710–1718. doi:10.1128/IAI.00073-14
Tunkel AR, Scheld WM (1993) Pathogenesis and pathophysiology of bacterial meningitis. Clin Microbiol Rev 6(2):118–136
Korthuis RJ, Anderson DC, Granger DN (1994) Role of neutrophil-endothelial cell adhesion in inflammatory disorders. J Crit Care 9(1):47–71
Täuber MG, Sande MA (1984) Pathogenesis of bacterial meningitis: contributions by experimental models in rabbits. Infection 12(Suppl 1):S3–S10
Ley K, Laudanna C, Cybulsky MI, Nourshargh S (2007) Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat Rev Immunol 7(9):678–689. doi:10.1038/nri2156, nri2156 [pii]
Tang T, Frenette PS, Hynes RO, Wagner DD, Mayadas TN (1996) Cytokine-induced meningitis is dramatically attenuated in mice deficient in endothelial selectins. J Clin Invest 97(11):2485–2490
Granert C, Raud J, Xie X, Lindquist L, Lindbom L (1994) Inhibition of leukocyte rolling with polysaccharide fucoidan prevents pleocytosis in experimental meningitis in the rabbit. J Clin Invest 93(3):929–936
Ostergaard C, Yieng-Kow RV, Benfield T, Frimodt-Moller N, Espersen F, Lundgren JD (2000) Inhibition of leukocyte entry into the brain by the selectin blocker fucoidan decreases interleukin-1 (IL-1) levels but increases IL-8 levels in cerebrospinal fluid during experimental pneumococcal meningitis in rabbits. Infect Immun 68(6):3153–3157
Brandt CT, Lundgren JD, Frimodt-Moller N, Christensen T, Benfield T, Espersen F, Hougaard DM, Ostergaard C (2005) Blocking of leukocyte accumulation in the cerebrospinal fluid augments bacteremia and increases lethality in experimental pneumococcal meningitis. J Neuroimmunol 166(1–2):126–131
Weber JR, Angstwurm K, Burger W, Einhaupl KM, Dirnagl U (1995) Anti ICAM-1 (CD 54) monoclonal antibody reduces inflammatory changes in experimental bacterial meningitis. J Neuroimmunol 63(1):63–68
Saez-Llorens X, Jafari HS, Severien C, Parras F, Olsen KD, Hansen EJ, Singer II, McCracken GH Jr (1991) Enhanced attenuation of meningeal inflammation and brain edema by concomitant administration of anti-CD18 monoclonal antibodies and dexamethasone in experimental Haemophilus meningitis. J Clin Invest 88(6):2003–2011
Tuomanen EI, Prasad SM, George JS, Hoepelman AI, Ibsen P, Heron I, Starzyk RM (1993) Reversible opening of the blood–brain barrier by anti-bacterial antibodies. Proc Natl Acad Sci U S A 90(16):7824–7828
Steinmann U, Borkowski J, Wolburg H, Schroppel B, Findeisen P, Weiss C, Ishikawa H, Schwerk C, Schroten H, Tenenbaum T (2013) Transmigration of polymorphonuclear neutrophils and monocytes through the human blood-cerebrospinal fluid barrier after bacterial infection in vitro. J Neuroinflammation 10:31. doi:10.1186/1742-2094-10-31
Wewer C, Seibt A, Wolburg H, Greune L, Schmidt MA, Berger J, Galla HJ, Quitsch U, Schwerk C, Schroten H, Tenenbaum T (2011) Transcellular migration of neutrophil granulocytes through the blood-cerebrospinal fluid barrier after infection with Streptococcus suis. J Neuroinflammation 8:51. doi:10.1186/1742-2094-8-51
Che X, Chi F, Wang L, Jong TD, Wu CH, Wang X, Huang SH (2011) Involvement of IbeA in meningitic Escherichia coli K1-induced polymorphonuclear leukocyte transmigration across brain endothelial cells. Brain Pathol 21(4):389–404. doi:10.1111/j.1750-3639.2010.00463.x
von Wedel-Parlow M, Schrot S, Lemmen J, Treeratanapiboon L, Wegener J, Galla HJ (2011) Neutrophils cross the BBB primarily on transcellular pathways: an in vitro study. Brain Res 1367:62–76. doi:10.1016/j.brainres.2010.09.076, S0006-8993(10)02142-6 [pii]
Wittchen ES (2009) Endothelial signaling in paracellular and transcellular leukocyte transmigration. Front Biosci 14:2522–2545, 3395 [pii]
Ernst JD, Decazes JM, Sande MA (1983) Experimental pneumococcal meningitis: role of leukocytes in pathogenesis. Infect Immun 41(1):275–279
Tuomanen EI, Saukkonen K, Sande S, Cioffe C, Wright SD (1989) Reduction of inflammation, tissue damage, and mortality in bacterial meningitis in rabbits treated with monoclonal antibodies against adhesion-promoting receptors of leukocytes. J Exp Med 170(3):959–969
Koedel U, Frankenberg T, Kirschnek S, Obermaier B, Hacker H, Paul R, Hacker G (2009) Apoptosis is essential for neutrophil functional shutdown and determines tissue damage in experimental pneumococcal meningitis. PLoS Pathog 5(5):e1000461. doi:10.1371/journal.ppat.1000461
Hoffmann O, Priller J, Prozorovski T, Schulze-Topphoff U, Baeva N, Lunemann JD, Aktas O, Mahrhofer C, Stricker S, Zipp F, Weber JR (2007) TRAIL limits excessive host immune responses in bacterial meningitis. J Clin Invest 117(7):2004–2013. doi:10.1172/JCI30356
McMillan DA, Lin CY, Aronin SI, Quagliarello VJ (2001) Community-acquired bacterial meningitis in adults: categorization of causes and timing of death. Clin Infect Dis 33(7):969–975
Klein M, Koedel U, Pfefferkorn T, Zeller G, Woehrl B, Pfister HW (2011) Arterial cerebrovascular complications in 94 adults with acute bacterial meningitis. Crit Care 15(6):R281. doi:10.1186/cc10565, cc10565 [pii]
Jan W, Zimmerman RA, Bilaniuk LT, Hunter JV, Simon EM, Haselgrove J (2003) Diffusion-weighted imaging in acute bacterial meningitis in infancy. Neuroradiology 45(9):634–639. doi:10.1007/s00234-003-1035-8
Takeoka M, Takahashi T (2002) Infectious and inflammatory disorders of the circulatory system and stroke in childhood. Curr Opin Neurol 15(2):159–164
Weiss N, Miller F, Cazaubon S, Couraud PO (2009) The blood–brain barrier in brain homeostasis and neurological diseases. Biochim Biophys Acta 1788(4):842–857. doi:10.1016/j.bbamem.2008.10.022, S0005-2736(08)00348-9 [pii]
Zysk G, Schneider-Wald BK, Hwang JH, Bejo L, Kim KS, Mitchell TJ, Hakenbeck R, Heinz HP (2001) Pneumolysin is the main inducer of cytotoxicity to brain microvascular endothelial cells caused by Streptococcus pneumoniae. Infect Immun 69(2):845–852
N’Guessan PD, Schmeck B, Ayim A, Hocke AC, Brell B, Hammerschmidt S, Rosseau S, Suttorp N, Hippenstiel S (2005) Streptococcus pneumoniae R6x induced p38 MAPK and JNK-mediated caspase-dependent apoptosis in human endothelial cells. Thromb Haemost 94(2):295–303. doi:10.1267/THRO05020295, 05080295 [pii]
Bermpohl D, Halle A, Freyer D, Dagand E, Braun JS, Bechmann I, Schroder NW, Weber JR (2005) Bacterial programmed cell death of cerebral endothelial cells involves dual death pathways. J Clin Invest 115(6):1607–1615. doi:10.1172/JCI23223
Constantin D, Ala’Aldeent D, Murphy S (2002) Transcriptional activation of nitric oxide synthase-2, and NO-induced cell death, in mouse cerebrovascular endothelium exposed to Neisseria meningitidis. J Neurochem 81(2):270–276
Khan NA, Iqbal J, Siddiqui R (2012) Escherichia coli K1-induced cytopathogenicity of human brain microvascular endothelial cells. Microb Pathog 53(5–6):269–275. doi:10.1016/j.micpath.2012.07.001
Hupp S, Heimeroth V, Wippel C, Fortsch C, Ma J, Mitchell TJ, Iliev AI (2012) Astrocytic tissue remodeling by the meningitis neurotoxin pneumolysin facilitates pathogen tissue penetration and produces interstitial brain edema. Glia 60(1):137–146. doi:10.1002/glia.21256
Kim BJ, Hancock BM, Bermudez A, Del Cid N, Reyes E, van Sorge NM, Lauth X, Smurthwaite CA, Hilton BJ, Stotland A, Banerjee A, Buchanan J, Wolkowicz R, Traver D, Doran KS (2015) Bacterial induction of Snail1 contributes to blood–brain barrier disruption. J Clin Invest 125(6):2473–2483. doi:10.1172/JCI74159
Daneman R (2012) The blood–brain barrier in health and disease. Ann Neurol 72(5):648–672. doi:10.1002/ana.23648
Stamatovic SM, Keep RF, Andjelkovic AV (2008) Brain endothelial cell-cell junctions: how to “open” the blood brain barrier. Curr Neuropharmacol 6(3):179–192. doi:10.2174/157015908785777210
Paul R, Koedel U, Winkler F, Kieseier BC, Fontana A, Kopf M, Hartung HP, Pfister HW (2003) Lack of IL-6 augments inflammatory response but decreases vascular permeability in bacterial meningitis. Brain 126(Pt 8):1873–1882
Stamatovic SM, Dimitrijevic OB, Keep RF, Andjelkovic AV (2006) Inflammation and brain edema: new insights into the role of chemokines and their receptors. Acta Neurochir Suppl 96:444–450
Ricci S, Grandgirard D, Wenzel M, Braccini T, Salvatore P, Oggioni MR, Leib SL, Koedel U (2014) Inhibition of matrix metalloproteinases attenuates brain damage in experimental meningococcal meningitis. BMC Infect Dis 14:726. doi:10.1186/s12879-014-0726-6
Kastenbauer S, Koedel U, Becker BF, Pfister HW (2002) Oxidative stress in bacterial meningitis in humans. Neurology 58(2):186–191
Leib SL, Kim YS, Chow LL, Sheldon RA, Tauber MG (1996) Reactive oxygen intermediates contribute to necrotic and apoptotic neuronal injury in an infant rat model of bacterial meningitis due to group B streptococci. J Clin Invest 98(11):2632–2639. doi:10.1172/JCI119084
Auer M, Pfister LA, Leppert D, Tauber MG, Leib SL (2000) Effects of clinically used antioxidants in experimental pneumococcal meningitis. J Infect Dis 182(1):347–350
Koedel U, Pfister HW (1997) Protective effect of the antioxidant N-acetyl-L-cysteine in pneumococcal meningitis in the rat. Neurosci Lett 225(1):33–36
Kastenbauer S, Koedel U, Becker BF, Pfister HW (2001) Experimental meningitis in the rat: protection by uric acid at human physiological blood concentrations. Eur J Pharmacol 425(2):149–152
Pun PB, Lu J, Moochhala S (2009) Involvement of ROS in BBB dysfunction. Free Radic Res 43(4):348–364. doi:10.1080/10715760902751902, 909050919 [pii]
Owens HM, Destache CJ, Dash AK (1999) Simple liquid chromatographic method for the analysis of the blood brain barrier permeability characteristics of ceftriaxone in an experimental rabbit meningitis model. J Chromatogr B Biomed Sci Appl 728(1):97–105
Fishman RA (1975) Brain edema. N Engl J Med 293(14):706–711. doi:10.1056/NEJM197510022931407
Brown LW, Feigin RD (1994) Bacterial meningitis: fluid balance and therapy. Pediatr Ann 23(2):93–98
Lu CH, Chang HW, Lui CC, Huang CR, Chang WN (2006) Cerebral haemodynamics in acute bacterial meningitis in adults. QJM 99(12):863–869. doi:10.1093/qjmed/hcl119, 99/12/863 [pii]
Ries S, Schminke U, Fassbender K, Daffertshofer M, Steinke W, Hennerici M (1997) Cerebrovascular involvement in the acute phase of bacterial meningitis. J Neurol 244(1):51–55
Pfister HW, Borasio GD, Dirnagl U, Bauer M, Einhaupl KM (1992) Cerebrovascular complications of bacterial meningitis in adults. Neurology 42(8):1497–1504
Pfister HW, Feiden W, Einhaupl KM (1993) Spectrum of complications during bacterial meningitis in adults. Results of a prospective clinical study. Arch Neurol 50(6):575–581
Vergouwen MD, Schut ES, Troost D, van de Beek D (2010) Diffuse cerebral intravascular coagulation and cerebral infarction in pneumococcal meningitis. Neurocrit Care 13(2):217–227. doi:10.1007/s12028-010-9387-5
Perry JR, Bilbao JM, Gray T (1992) Fatal basilar vasculopathy complicating bacterial meningitis. Stroke 23(8):1175–1178
Ment LR, Ehrenkranz RA, Duncan CC (1986) Bacterial meningitis as an etiology of perinatal cerebral infarction. Pediatr Neurol 2(5):276–279
Koedel U, Bernatowicz A, Paul R, Frei K, Fontana A, Pfister HW (1995) Experimental pneumococcal meningitis: cerebrovascular alterations, brain edema, and meningeal inflammation are linked to the production of nitric oxide. Ann Neurol 37(3):313–323
Tureen J, Liu Q, Chow L (1996) Near-infrared spectroscopy in experimental pneumococcal meningitis in the rabbit: cerebral hemodynamics and metabolism. Pediatr Res 40(5):759–763
Täuber MG (1989) Brain edema, intracranial pressure and cerebral blood flow in bacterial meningitis. Pediatr Infect Dis J 8(12):915–917
Pedersen M, Brandt CT, Knudsen GM, Ostergaard C, Skinhoj P, Frimodt-Moller N, Moller K (2007) Cerebral blood flow autoregulation in early experimental S. pneumoniae meningitis. J Appl Physiol 102(1):72–78. doi:10.1152/japplphysiol.00697.2006, 00697.2006 [pii]
Tureen JH, Dworkin RJ, Kennedy SL, Sachdeva M, Sande MA (1990) Loss of cerebrovascular autoregulation in experimental meningitis in rabbits. J Clin Invest 85(2):577–581
Pfister HW, Koedel U, Haberl RL, Dirnagl U, Feiden W, Ruckdeschel G, Einhaupl KM (1990) Microvascular changes during the early phase of experimental bacterial meningitis. J Cereb Blood Flow Metab 10(6):914–922
Lorenzl S, Koedel U, Frei K, Bernatowicz A, Fontana A, Pfister HW (1995) Protective effect of a 21-aminosteroid during experimental pneumococcal meningitis. J Infect Dis 172(1):113–118
Pfister HW, Kodel U, Dirnagl U, Haberl RL, Ruckdeschel G, Einhaupl KM (1992) Effect of catalase on regional cerebral blood flow and brain edema during the early phase of experimental pneumococcal meningitis. J Infect Dis 166(6):1442–1445
Hoffmann OM, Becker D, Weber JR (2007) Bacterial hydrogen peroxide contributes to cerebral hyperemia during early stages of experimental pneumococcal meningitis. J Cereb Blood Flow Metab 27(11):1792–1797. doi:10.1038/sj.jcbfm.9600474, 9600474 [pii]
Schaper M, Gergely S, Lykkesfeldt J, Zbaren J, Leib SL, Tauber MG, Christen S (2002) Cerebral vasculature is the major target of oxidative protein alterations in bacterial meningitis. J Neuropathol Exp Neurol 61(7):605–613
Szabo C (2003) Multiple pathways of peroxynitrite cytotoxicity. Toxicol Lett 140–141:105–112
Kastenbauer S, Koedel U, Becker BF, Pfister HW (2002) Pneumococcal meningitis in the rat: evaluation of peroxynitrite scavengers for adjunctive therapy. Eur J Pharmacol 449(1–2):177–181
Koedel U, Gorriz C, Lorenzl S, Pfister HW (1997) Increased endothelin levels in cerebrospinal fluid samples from adults with bacterial meningitis. Clin Infect Dis 25(2):329–330
Skopal J, Turbucz P, Vastag M, Bori Z, Pek M, deChatel R, Nagy Z, Toth M, Karadi I (1998) Regulation of endothelin release from human brain microvessel endothelial cells. J Cardiovasc Pharmacol 31(Suppl 1):S370–S372
Sibson NR, Blamire AM, Perry VH, Gauldie J, Styles P, Anthony DC (2002) TNF-alpha reduces cerebral blood volume and disrupts tissue homeostasis via an endothelin- and TNFR2-dependent pathway. Brain 125(Pt 11):2446–2459
Sury MD, Frese-Schaper M, Muhlemann MK, Schulthess FT, Blasig IE, Tauber MG, Shaw SG, Christen S (2006) Evidence that N-acetylcysteine inhibits TNF-alpha-induced cerebrovascular endothelin-1 upregulation via inhibition of mitogen- and stress-activated protein kinase. Free Radic Biol Med 41(9):1372–1383. doi:10.1016/j.freeradbiomed.2006.07.016, S0891-5849(06)00469-2 [pii]
Brunner F, Bras-Silva C, Cerdeira AS, Leite-Moreira AF (2006) Cardiovascular endothelins: essential regulators of cardiovascular homeostasis. Pharmacol Ther 111(2):508–531. doi:10.1016/j.pharmthera.2005.11.001, S0163-7258(05)00262-7 [pii]
Pfister LA, Tureen JH, Shaw S, Christen S, Ferriero DM, Tauber MG, Leib SL (2000) Endothelin inhibition improves cerebral blood flow and is neuroprotective in pneumococcal meningitis. Ann Neurol 47(3):329–335
Edmond K, Clark A, Korczak VS, Sanderson C, Griffiths UK, Rudan I (2010) Global and regional risk of disabling sequelae from bacterial meningitis: a systematic review and meta-analysis. Lancet Infect Dis 10(5):317–328. doi:10.1016/S1473-3099(10)70048-7, S1473-3099(10)70048-7 [pii]
Kesser BW, Hashisaki GT, Spindel JH, Ruth RA, Scheld WM (1999) Time course of hearing loss in an animal model of pneumococcal meningitis. Otolaryngol Head Neck Surg 120(5):628–637
Woolley AL, Kirk KA, Neumann AM Jr, McWilliams SM, Murray J, Freind D, Wiatrak BJ (1999) Risk factors for hearing loss from meningitis in children: the Children’s Hospital experience. Arch Otolaryngol Head Neck Surg 125(5):509–514
Oostenbrink R, Maas M, Moons KG, Moll HA (2002) Sequelae after bacterial meningitis in childhood. Scand J Infect Dis 34(5):379–382
Edmond K, Dieye Y, Griffiths UK, Fleming J, Ba O, Diallo N, Mulholland K (2010) Prospective cohort study of disabling sequelae and quality of life in children with bacterial meningitis in urban Senegal. Pediatr Infect Dis J 29(11):1023–1029. doi:10.1097/INF.0b013e3181e598ea
Adachi N, Ito K, Sakata H (2010) Risk factors for hearing loss after pediatric meningitis in Japan. Ann Otol Rhinol Laryngol 119(5):294–296
Douglas SA, Sanli H, Gibson WP (2008) Meningitis resulting in hearing loss and labyrinthitis ossificans – does the causative organism matter? Cochlear Implants Int 9(2):90–96. doi:10.1002/cii.344
Kutz JW, Simon LM, Chennupati SK, Giannoni CM, Manolidis S (2006) Clinical predictors for hearing loss in children with bacterial meningitis. Arch Otolaryngol Head Neck Surg 132(9):941–945. doi:10.1001/archotol.132.9.941, 132/9/941 [pii]
Dichgans M, Jager L, Mayer T, Schorn K, Pfister HW (1999) Bacterial meningitis in adults: demonstration of inner ear involvement using high-resolution MRI. Neurology 52(5):1003–1009
Caye-Thomasen P, Dam MS, Omland SH, Mantoni M (2012) Cochlear ossification in patients with profound hearing loss following bacterial meningitis. Acta Otolaryngol 132(7):720–725. doi:10.3109/00016489.2012.656323
Nadol JB Jr, Hsu WC (1991) Histopathologic correlation of spiral ganglion cell count and new bone formation in the cochlea following meningogenic labyrinthitis and deafness. Ann Otol Rhinol Laryngol 100(9 Pt 1):712–716
Merchant SN, Gopen Q (1996) A human temporal bone study of acute bacterial meningogenic labyrinthitis. Am J Otol 17(3):375–385
Klein M, Koedel U, Pfister HW, Kastenbauer S (2003) Morphological correlates of acute and permanent hearing loss during experimental pneumococcal meningitis. Brain Pathol 13(2):123–132
Caye-Thomasen P, Worsoe L, Brandt CT, Miyazaki H, Ostergaard C, Frimodt-Moller N, Thomsen J (2009) Routes, dynamics, and correlates of cochlear inflammation in terminal and recovering experimental meningitis. Laryngoscope 119(8):1560–1570. doi:10.1002/lary.20260
Blank AL, Davis GL, VanDeWater TR, Ruben RJ (1994) Acute Streptococcus pneumoniae meningogenic labyrinthitis. An experimental guinea pig model and literature review. Arch Otolaryngol Head Neck Surg 120(12):1342–1346
Bhatt S, Halpin C, Hsu W, Thedinger BA, Levine RA, Tuomanen E, Nadol JB Jr (1991) Hearing loss and pneumococcal meningitis: an animal model. Laryngoscope 101(12 Pt 1):1285–1292
Klein M, Koedel U, Kastenbauer S, Pfister HW (2008) Nitrogen and oxygen molecules in meningitis-associated labyrinthitis and hearing impairment. Infection 36(1):2–14. doi:10.1007/s15010-007-7153-1
Rappaport JM, Bhatt SM, Kimura RS, Lauretano AM, Levine RA (1999) Electron microscopic temporal bone histopathology in experimental pneumococcal meningitis. Ann Otol Rhinol Laryngol 108(6):537–547
Brandt CT, Caye-Thomasen P, Lund SP, Worsoe L, Ostergaard C, Frimodt-Moller N, Espersen F, Thomsen J, Lundgren JD (2006) Hearing loss and cochlear damage in experimental pneumococcal meningitis, with special reference to the role of neutrophil granulocytes. Neurobiol Dis 23(2):300–311. doi:10.1016/j.nbd.2006.03.006, S0969-9961(06)00060-X [pii]
Winter AJ, Comis SD, Osborne MP, Tarlow MJ, Stephen J, Andrew PW, Hill J, Mitchell TJ (1997) A role for pneumolysin but not neuraminidase in the hearing loss and cochlear damage induced by experimental pneumococcal meningitis in guinea pigs. Infect Immun 65(11):4411–4418
Klein M, Koedel U, Pfister HW, Kastenbauer S (2003) Meningitis-associated hearing loss: protection by adjunctive antioxidant therapy. Ann Neurol 54(4):451–458. doi:10.1002/ana.10684
Ge NN, Brodie HA, Tinling SP (2008) Long-term hearing loss in gerbils with bacterial meningitis treated with superoxide dismutase. Otol Neurotol 29(8):1061–1067. doi:10.1097/MAO.0b013e31818b6479
Bedford H, de Louvois J, Halket S, Peckham C, Hurley R, Harvey D (2001) Meningitis in infancy in England and Wales: follow up at age 5 years. BMJ 323(7312):533–536
Anderson V, Anderson P, Grimwood K, Nolan T (2004) Cognitive and executive function 12 years after childhood bacterial meningitis: effect of acute neurologic complications and age of onset. J Pediatr Psychol 29(2):67–81
Schmidt H, Heimann B, Djukic M, Mazurek C, Fels C, Wallesch CW, Nau R (2006) Neuropsychological sequelae of bacterial and viral meningitis. Brain 129(Pt 2):333–345
Grimwood K, Anderson P, Anderson V, Tan L, Nolan T (2000) Twelve year outcomes following bacterial meningitis: further evidence for persisting effects. Arch Dis Child 83(2):111–116
Hoogman M, van de Beek D, Weisfelt M, de Gans J, Schmand B (2007) Cognitive outcome in adults after bacterial meningitis. J Neurol Neurosurg Psychiatry 78(10):1092–1096. doi:10.1136/jnnp.2006.110023, jnnp.2006.110023 [pii]
Focke NK, Kallenberg K, Mohr A, Djukic M, Nau R, Schmidt H (2013) Distributed, limbic gray matter atrophy in patients after bacterial meningitis. AJNR Am J Neuroradiol 34:1164–1167. doi:10.3174/ajnr.A3351, ajnr.A3351 [pii]
Grimwood K, Nolan TM, Bond L, Anderson VA, Catroppa C, Keir EH (1996) Risk factors for adverse outcomes of bacterial meningitis. J Paediatr Child Health 32(5):457–462
Libster R, Edwards KM, Levent F, Edwards MS, Rench MA, Castagnini LA, Cooper T, Sparks RC, Baker CJ, Shah PE (2012) Long-term outcomes of group B streptococcal meningitis. Pediatrics 130(1):e8–e15. doi:10.1542/peds.2011-3453, peds.2011-3453 [pii]
Bargui F, D’Agostino I, Mariani-Kurkdjian P, Alberti C, Doit C, Bellier N, Morin L, Galli Gibertini G, Smail A, Zanin A, Lorrot M, Dauger S, Neve M, Faye A, Armoogum P, Bourrillon A, Bingen E, Mercier JC, Bonacorsi S, Nigrovic LE, Titomanlio L (2012) Factors influencing neurological outcome of children with bacterial meningitis at the emergency department. Eur J Pediatr 171(9):1365–1371. doi:10.1007/s00431-012-1733-5
Hernandez MI, Sandoval CC, Tapia JL, Mesa T, Escobar R, Huete I, Wei XC, Kirton A (2011) Stroke patterns in neonatal group B streptococcal meningitis. Pediatr Neurol 44(4):282–288. doi:10.1016/j.pediatrneurol.2010.11.002, S0887-8994(10)00491-1 [pii]
Jorens PG, Parizel PM, Wojciechowski M, Laridon A, De Weerdt A, Mertens G, Ceulemans B (2008) Streptococcus pneumoniae meningoencephalitis with unusual and widespread white matter lesions. Eur J Paediatr Neurol 12(2):127–132. doi:10.1016/j.ejpn.2007.06.007, S1090-3798(07)00112-2 [pii]
Shah DK, Daley AJ, Hunt RW, Volpe JJ, Inder TE (2005) Cerebral white matter injury in the newborn following Escherichia coli meningitis. Eur J Paediatr Neurol 9(1):13–17. doi:10.1016/j.ejpn.2004.09.002, S1090-3798(04)00189-8 [pii]
Täuber MG, Kim YS, Leib SL (1997) Neuronal injury in meningitis. In: Peterson PK, Remington JS (eds) In defense of the brain. Blackwell Science, Malden, MA, pp 124–143
Nau R, Gerber J, Bunkowski S, Bruck W (2004) Axonal injury, a neglected cause of CNS damage in bacterial meningitis. Neurology 62(3):509–511
Gerber J, Seitz RC, Bunkowski S, Bruck W, Nau R (2009) Evidence for frequent focal and diffuse acute axonal injury in human bacterial meningitis. Clin Neuropathol 28(1):33–39, 5350 [pii]
Renier D, Flandin C, Hirsch E, Hirsch JF (1988) Brain abscesses in neonates. A study of 30 cases. J Neurosurg 69(6):877–882. doi:10.3171/jns.1988.69.6.0877
Cone LA, Leung MM, Byrd RG, Annunziata GM, Lam RY, Herman BK (2003) Multiple cerebral abscesses because of Listeria monocytogenes: three case reports and a literature review of supratentorial listerial brain abscess(es). Surg Neurol 59(4):320–328, S0090301903000569 [pii]
Zysk G, Bruck W, Gerber J, Bruck Y, Prange HW, Nau R (1996) Anti-inflammatory treatment influences neuronal apoptotic cell death in the dentate gyrus in experimental pneumococcal meningitis. J Neuropathol Exp Neurol 55(6):722–728
Bifrare YD, Gianinazzi C, Imboden H, Leib SL, Tauber MG (2003) Bacterial meningitis causes two distinct forms of cellular damage in the hippocampal dentate gyrus in infant rats. Hippocampus 13(4):481–488
Gianinazzi C, Grandgirard D, Imboden H, Egger L, Meli DN, Bifrare YD, Joss PC, Tauber MG, Borner C, Leib SL (2003) Caspase-3 mediates hippocampal apoptosis in pneumococcal meningitis. Acta Neuropathol 105(5):499–507. doi:10.1007/s00401-003-0672-7
Nag S, Manias JL, Stewart DJ (2009) Pathology and new players in the pathogenesis of brain edema. Acta Neuropathol 118(2):197–217. doi:10.1007/s00401-009-0541-0
Loeffler JM, Ringer R, Hablutzel M, Tauber MG, Leib SL (2001) The free radical scavenger alpha-phenyl-tert-butyl nitrone aggravates hippocampal apoptosis and learning deficits in experimental pneumococcal meningitis. J Infect Dis 183(2):247–252. doi:10.1086/317921
Wellmer A, Noeske C, Gerber J, Munzel U, Nau R (2000) Spatial memory and learning deficits after experimental pneumococcal meningitis in mice. Neurosci Lett 296(2–3):137–140
Grandgirard D, Bifrare YD, Pleasure SJ, Kummer J, Leib SL, Tauber MG (2007) Pneumococcal meningitis induces apoptosis in recently postmitotic immature neurons in the dentate gyrus of neonatal rats. Dev Neurosci 29(1–2):134–142
Leib SL, Heimgartner C, Bifrare YD, Loeffler JM, Taauber MG (2003) Dexamethasone aggravates hippocampal apoptosis and learning deficiency in pneumococcal meningitis in infant rats. Pediatr Res 54(3):353–357. doi:10.1203/01.PDR.0000079185.67878.72
Nau R, Soto A, Bruck W (1999) Apoptosis of neurons in the dentate gyrus in humans suffering from bacterial meningitis. J Neuropathol Exp Neurol 58(3):265–274
Free SL, Li LM, Fish DR, Shorvon SD, Stevens JM (1996) Bilateral hippocampal volume loss in patients with a history of encephalitis or meningitis. Epilepsia 37(4):400–405
Derugin N, Wendland M, Muramatsu K, Roberts TP, Gregory G, Ferriero DM, Vexler ZS (2000) Evolution of brain injury after transient middle cerebral artery occlusion in neonatal rats. Stroke 31(7):1752–1761
Leib SL, Kim YS, Ferriero DM, Täuber MG (1996) Neuroprotective effect of excitatory amino acid antagonist kynurenic acid in experimental bacterial meningitis. J Infect Dis 173(1):166–171
Scheld WM, Dacey RG, Winn HR, Welsh JE, Jane JA, Sande MA (1980) Cerebrospinal fluid outflow resistance in rabbits with experimental meningitis. Alterations with penicillin and methylprednisolone. J Clin Invest 66:243–253
Kaplan SL, Feigin RD (1978) The syndrome of inappropriate secretion of antidiuretic hormone in children with bacterial meningitis. J Pediatr 92:758–761
Winkler F, Kastenbauer S, Yousry TA, Maerz U, Pfister HW (2002) Discrepancies between brain CT imaging and severely raised intracranial pressure proven by ventriculostomy in adults with pneumococcal meningitis. J Neurol 249(9):1292–1297
Lindvall P, Ahlm C, Ericsson M, Gothefors L, Naredi S, Koskinen LO (2004) Reducing intracranial pressure may increase survival among patients with bacterial meningitis. Clin Infect Dis 38(3):384–390. doi:10.1086/380970, CID31898 [pii]
Arcienega II, Brunet JF, Bloch J, Badaut J (2010) Cell locations for AQP1, AQP4 and 9 in the non-human primate brain. Neuroscience 167(4):1103–1114. doi:10.1016/j.neuroscience.2010.02.059, S0306-4522(10)00287-3 [pii]
Saadoun S, Papadopoulos MC (2010) Aquaporin-4 in brain and spinal cord oedema. Neuroscience 168(4):1036–1046. doi:10.1016/j.neuroscience.2009.08.019, S0306-4522(09)01339-6 [pii]
Papadopoulos MC, Verkman AS (2005) Aquaporin-4 gene disruption in mice reduces brain swelling and mortality in pneumococcal meningitis. J Biol Chem 280(14):13906–13912. doi:10.1074/jbc.M413627200, M413627200 [pii]
Saadoun S, Papadopoulos MC, Krishna S (2003) Water transport becomes uncoupled from K+ siphoning in brain contusion, bacterial meningitis, and brain tumours: immunohistochemical case review. J Clin Pathol 56(12):972–975
Blocher J, Eckert I, Elster J, Wiefek J, Eiffert H, Schmidt H (2011) Aquaporins AQP1 and AQP4 in the cerebrospinal fluid of bacterial meningitis patients. Neurosci Lett 504(1):23–27. doi:10.1016/j.neulet.2011.08.049, S0304-3940(11)01234-1 [pii]
Spranger M, Krempien S, Schwab S, Maiwald M, Bruno K, Hacke W (1996) Excess glutamate in the cerebrospinal fluid in bacterial meningitis. J Neurol Sci 143(1–2):126–131
Guerra-Romero L, Tauber MG, Fournier MA, Tureen JH (1992) Lactate and glucose concentrations in brain interstitial fluid, cerebrospinal fluid, and serum during experimental pneumococcal meningitis. J Infect Dis 166(3):546–550
Wippel C, Maurer J, Fortsch C, Hupp S, Bohl A, Ma J, Mitchell TJ, Bunkowski S, Bruck W, Nau R, Iliev AI (2013) Bacterial cytolysin during meningitis disrupts the regulation of glutamate in the brain, leading to synaptic damage. PLoS Pathog 9(6):e1003380. doi:10.1371/journal.ppat.1003380
Recher M, Malipiero U, Schaer DJ, Koedel U, Pfister HW, Birchler T, Petrausch U, Claus H, Gast H, Fontana A (2013) Inhibition of meningitis-associated neutrophil apoptosis by TNF-alpha depends on functional PI3-kinase in monocytes. J Leukoc Biol 93(2):259–266. doi:10.1189/jlb.0511218, jlb.0511218 [pii]
Tunbridge AJ, Stevanin TM, Lee M, Marriott HM, Moir JW, Read RC, Dockrell DH (2006) Inhibition of macrophage apoptosis by Neisseria meningitidis requires nitric oxide detoxification mechanisms. Infect Immun 74(1):729–733
Wennekamp J, Henneke P (2008) Induction and termination of inflammatory signaling in group B streptococcal sepsis. Immunol Rev 225:114–127. doi:10.1111/j.1600-065X.2008.00673.x, IMR673 [pii]
Kim GH, Kim JE, Rhie SJ, Yoon S (2015) The role of oxidative stress in neurodegenerative diseases. Exp Neurobiol 24(4):325–340. doi:10.5607/en.2015.24.4.325
Grammas P, Martinez J, Miller B (2011) Cerebral microvascular endothelium and the pathogenesis of neurodegenerative diseases. Expert Rev Mol Med 13:e19. doi:10.1017/S1462399411001918
Klein M, Koedel U, Pfister HW (2006) Oxidative stress in pneumococcal meningitis: a future target for adjunctive therapy? Prog Neurobiol 80(6):269–280. doi:10.1016/j.pneurobio.2006.11.008, S0301-0082(06)00143-2 [pii]
Simon RP, Beckman JS (2002) Why pus is bad for the brain. Neurology 58(2):167–168
Braun J (2009) Inducible nitric oxide synthase mediates hippocampal caspase-3 activation in pneumococcal meningitis. Int J Neurosci 119(4):455–459. doi:10.1080/00207450802479970, 908868698 [pii]
Leib SL, Kim YS, Black SM, Tureen JH, Tauber MG (1998) Inducible nitric oxide synthase and the effect of aminoguanidine in experimental neonatal meningitis. J Infect Dis 177(3):692–700
Guo Z, Sun X, He Z, Jiang Y, Zhang X (2010) Role of matrix metalloproteinase-9 in apoptosis of hippocampal neurons in rats during early brain injury after subarachnoid hemorrhage. Neurol Sci 31(2):143–149. doi:10.1007/s10072-009-0192-x
Murase S, McKay RD (2012) Matrix metalloproteinase-9 regulates survival of neurons in newborn hippocampus. J Biol Chem 287(15):12184–12194. doi:10.1074/jbc.M111.297671, M111.297671 [pii]
Fujita-Hamabe W, Tokuyama S (2012) The involvement of cleavage of neural cell adhesion molecule in neuronal death under oxidative stress conditions in cultured cortical neurons. Biol Pharm Bull 35(4):624–628, JST.JSTAGE/bpb/35.624 [pii]
Rosenberg GA (2009) Matrix metalloproteinases and their multiple roles in neurodegenerative diseases. Lancet Neurol 8(2):205–216. doi:10.1016/S1474-4422(09)70016-X, S1474-4422(09)70016-X [pii]
Braun JS, Novak R, Murray PJ, Eischen CM, Susin SA, Kroemer G, Halle A, Weber JR, Tuomanen EI, Cleveland JL (2001) Apoptosis-inducing factor mediates microglial and neuronal apoptosis caused by pneumococcus. J Infect Dis 184(10):1300–1309
Braun JS, Sublett JE, Freyer D, Mitchell TJ, Cleveland JL, Tuomanen EI, Weber JR (2002) Pneumococcal pneumolysin and H(2)O(2) mediate brain cell apoptosis during meningitis. J Clin Invest 109(1):19–27
Braun JS, Hoffmann O, Schickhaus M, Freyer D, Dagand E, Bermpohl D, Mitchell TJ, Bechmann I, Weber JR (2007) Pneumolysin causes neuronal cell death through mitochondrial damage. Infect Immun 75(9):4245–4254. doi:10.1128/IAI.00031-07, IAI.00031-07 [pii]
Beurg M, Hafidi A, Skinner L, Cowan G, Hondarrague Y, Mitchell TJ, Dulon D (2005) The mechanism of pneumolysin-induced cochlear hair cell death in the rat. J Physiol 568(Pt 1):211–227. doi:10.1113/jphysiol.2005.092478, jphysiol.2005.092478 [pii]
Reiss A, Braun JS, Jager K, Freyer D, Laube G, Buhrer C, Felderhoff-Muser U, Stadelmann C, Nizet V, Weber JR (2011) Bacterial pore-forming cytolysins induce neuronal damage in a rat model of neonatal meningitis. J Infect Dis 203(3):393–400. doi:10.1093/infdis/jiq047, jiq047 [pii]
Acknowledgments
This work was supported by grant no. 162583 (to SLL) from the Swiss National Science Foundation.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Agyeman, P., Grandgirard, D., Leib, S.L. (2017). Neuroinflammation in Bacterial Meningitis. In: Lyck, R., Enzmann, G. (eds) The Blood Brain Barrier and Inflammation. Progress in Inflammation Research. Springer, Cham. https://doi.org/10.1007/978-3-319-45514-3_10
Download citation
DOI: https://doi.org/10.1007/978-3-319-45514-3_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-45512-9
Online ISBN: 978-3-319-45514-3
eBook Packages: MedicineMedicine (R0)