Abstract
Sedation is of paramount importance in the management of ARDS. Supporting the vital functions in the acute phase of acute respiratory failure management requires analgesic and tranquilizing drugs, both to permit the invasive procedures with the lowest iatrogenic damage and to control the body stress response.
Once clinical stabilization is obtained, the available scientific evidences underline the importance of managing pain, agitation, and delirium symptoms, while maintaining the mildest possible level of sedation. Compliance to mechanical ventilation, anxiety and agitation control, and oxygen consumption decrease are possible reasons to explain the role of sedative therapy in decreasing mortality and morbidity. Thus, sedation should aim to provide the best possible adaptation to the critical illness yet avoiding excessive unconsciousness or prolonged ICU stay. In this context, the key intervention is maintaining a continuous pain/agitation/delirium monitoring using validated tools to early manage the arising neurological dysfunctions according to locally agreed protocols. Careful neurologic monitoring is even more important than the choice of a particular sedative agent itself since the literature supports modest differences in outcomes by using non-benzodiazepine-based sedation protocols.
At a later stage, during the mechanical ventilation weaning, implementing early physical mobilization, as well as the family engagement, should be considered along with the earliest sedation interruption and the continuous monitoring and management of pain, agitation, and delirium.
Adoption of local sedation protocols is strongly advised by the most recent literature. They could be comprehensive of the daily interruption of short-term sedatives (propofol, dexmedetomidine), the analgesia-based sedation (morphine, fentanyl, or remifentanil), the use of inhalational halogenated in the earlier phase (sevoflurane), or the use of enteral approach during the subsequent phases. Whichever the local protocol, the international guidelines point out the necessity to keep patients “awake and cooperative” as soon as possible, then avoiding the iatrogenic risks of both under- and oversedation.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
- Intensive Care Unit
- Intensive Care Unit Patient
- Intensive Care Unit Stay
- Deep Sedation
- Spontaneous Breathing Trial
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
18.1 Introduction
The severity of the acute respiratory failure, the necessity of lifesaving invasive procedures, and the harsh intensive care unit (ICU) environment make sedation necessary in ARDS patients [1]. Such patients may experience a dangerous condition, both physical and psychological, and need the most appropriate and individualized neurological treatment. Among many other vital organ failures, the early and long-term consequences of ARDS may lead to severe brain dysfunction [2].
Pain, agitation, delirium, anxiety, and alteration of consciousness are common conditions in ARDS patients [3]. They are frequently triggered by treatable causes, like hypoxemia, hypercarbia, acidosis, hypoglycemia, hypo- or hypernatremia, sepsis, hypovolemia, and alcohol or drug withdrawal, or by lifesaving medical treatments comprehending mechanical ventilation, invasive procedures, forced body postures, and uninterrupted noise and light stimulation, together with their consequences like sleep deprivation or the impossibility of communicating with the staff [4]. International guidelines recommend to face and treat first all organic and metabolic causes of distress and to minimize environment-linked stressors [5, 6]. As a second step, they suggest the administration of analgesic, sedative, and antipsychotic drugs to ensure comfort, at all stages of the illness. Adequate levels of sedation, therefore, represent a primary target for managing ARDS patients. However, since neuroactive therapy is related to several important side effects like hemodynamic instability and cardiac dysrhythmias [7], sepsis [8], ileus, delirium [9], and lengthening of respiratory weaning [10], it is important to titrate it to the lowest effective amount [11].
Several authors have pointed out the need for light sedation [12, 13], due both to the detrimental effects of neuroactive therapy itself and to the clinical and economical costs associated with deeper-than-needed sedation levels [1, 14, 15]. Daily interruption of intravenous short-term sedative administration [16] in association with a spontaneous breathing trial [17] may decrease the mechanical ventilation length and reduce both complications and prevalence of delirium. Analgesia-based sedation could be another effective way to manage ARDS patients, relying on the opiates’ ability to maintain adaptation to mechanical ventilation while preserving patients’ consciousness [18]. However, the use of short-half-life analgesics (remifentanil) is only indicated for short-stay patients, while there is no difference among opiates for patients requiring mechanical ventilation longer than 3 days [19, 20]. In 2010, Strom et al. demonstrated that a protocol based only on morphine 5 mg boluses allows minimal sedation and provides better outcomes, rather than adding propofol to achieve deeper sedation levels [21].
The conscious sedation target is an innovation of the utmost relevance in the field of ICU care. Nonetheless, some intensivists tend to consider it unfeasible, considering the potential higher risk of ventilation-related lung damage, the risk of self-removal of invasive devices [22], and the possible stress/discomfort for patients [23]. From an ICU staff perspective, it also raises the issue of an increased workload. These beliefs, however, are at least partially unfounded [24]. Intensivists have to face the side effects of both pharmacological and physical “restraint” methods, continuously pursuing the best approach for patient security and healing.
Despite guideline suggestions and the fact that between 60 and 80% of ICUs use a specific score to evaluate the level of sedation [25], many physicians routinely maintain [26] a deeper than desired level of sedation [27], probably causing avoidable side effects. Pragmatically, it is important to underline the concept that it does not exist in one-and-only sedative treatment which can be always adequate; at the same time, a disproportionate fear to use sedative drugs is inappropriate. Even if the clinical course of each ARDS patient is different from the others, it is useful to distinguish at least two different scenarios, leading to the neuroactive drug prescription (Fig. 18.1) in ARDS patients. In the acute phase, intubation and placing of vascular catheters and other maneuvers devoted to clinical stabilization require a deep sedation target, similar to general anesthesia. On the other hand, in the subsequent phase, it is important to change the target toward a lighter sedation, by using the lowest amount of neuroactive drugs in order to only obtain a patient adaptation to critical illness. This second phase has to begin as soon as possible, frequently after the second/third ICU day. Other practices such as adjustment of the mechanical ventilation settings, of the drug therapies, and of the environment to the specific necessities of each specific patient can help in reducing discomfort.
18.2 The Key Importance of Continuous Neurological Assessment with Validated Tools
International guidelines clearly recommend systematic assessment of pain, agitation/sedation, and delirium in all ICU patients, with validated tools [4–6]. They provide repeatable and comparable measurements, able to adequately titrate the analgesic, sedative, and antipsychotic therapy. The goal is to control the stress response and the neurological symptoms together keeping ARDS patients awake, cooperative, and well adapted to the necessary invasive procedures as soon as possible [28]. An excessive use of neuroactive drugs [26] correlates with longer mechanical ventilation and ICU stay, as well as with an increased risk of serious neurological consequences in both the short- and long-term [10, 29].
Among the validated tools, the scales with the highest psychometric properties [30] are the Verbal Numeric Rating (VNR), the Behavioral Pain Scale (BPS) [31], or the Critical-Care Pain Observation Tool (CPOT) [32] for the pain assessment, the Richmond Agitation-Sedation Scale (RASS) [33] and the Sedation-Agitation Scale (SAS) [34] for the agitation/sedation assessment, and the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU) [35] and the Intensive Care Delirium Screening Checklist (ICDSC) [36] for the delirium assessment.
Pain assessment has become culturally essential for intensivists, both nurses and physicians. Proper identification of pain symptoms in critically ill patients represents a challenge because of complexity in communication with patients needing endotracheal respiratory prostheses, because of altered state of consciousness or because of neuroactive drugs use. To address these issues, specific behavioral scales were designed and validated both in unconscious/sedated [37] and in conscious/awake patients [38] and established as a valid, reliable, and simple tool to be used in clinical practice.
Regarding sedative drugs, their under- or overuse may compromise clinical stability. Life-threatening side effects of untreated agitation and stress response are evident: self-removal of life-sustaining devices, tachypnea, tachycardia, hypertension, sustained hypoxemia, and hypercarbia due to uncoordinated mechanical ventilation [6]. At the same time, deeper-than-necessary sedation increases the length of ICU stay [5], the duration of mechanical ventilation [21], the sepsis severity [8], and the onset of new neurological failures both during hospitalization like delirium and after discharge [29] like the development of psychological reactions of traumatic nature (posttraumatic stress disorder, PTSD). In order to obtain the best sedative titration, the use of validated tools is mandatory. For example, the RASS describes ten levels of sedation/agitation through observation and verbal and physical stimulation. Scores range from −5 (unconscious, unresponsive to voice and physical stimuli) to + 4 (overtly combative, violent, immediate danger to staff), adequately describing the possible neurological condition needing an immediate intervention.
While regarding pain and agitation evaluation encouraging results are present in literature [39], delirium recognition [40] and assessment is more challenging, since its success is linked to an effective and lasting staff training [41]. Delirium has a very high prevalence in critically ill patients [42], with a direct relationship between increased morbidity and mortality and the duration (in days) of delirium [43, 44]. The presence and duration of delirium also correlates with a significant deterioration in the quality of life after ICU discharge [45]. Large literature recommends the use of validated tools [46], like the Confusion Assessment Method for the ICU (CAM-ICU) and the Intensive Care Delirium Screening Checklist (ICDSC). The CAM-ICU has excellent sensitivity and specificity and is based on only four items (acute modification of consciousness, inattention, disorganized thinking, altered level of consciousness). This allows a shorter training to teach ICU staff members a validated way to evaluate delirium.
All these measurements have to be reported in the clinical chart at least once per shift, together with an evaluation of adequacy of sedative treatment. The neurological monitoring plays a key role in ARDS patients, and it is quick to perform. A complete example of evaluations recommended by international guidelines is reported in Fig. 18.2. Physicians have to state the desired sedation target for that specific patient at that specific moment of clinical course; nurses, on their part, report the actual neurological state, describing pain, anxiety, agitation, sleep, need for physical restraints, and delirium, together with a comprehensive evaluation of sedative therapy, in order to get the most adequate treatment titration.
An example of very simple neurological monitoring card, used in a general ICU of the University Hospital (A.O. San Paolo, Milano), to be added within a dedicated spot in nursing sheets. For each shift – morning, afternoon, and night – physicians have to state the target level of sedation, while nurses have to monitor and record, through appropriate validated tools, neurological parameters, and also to evaluate the adequacy of sedative therapy as prescribed by physicians
18.3 Sedation Assessment
The appropriate target level of sedation primarily depends on a patient’s acute disease process and on the therapeutic and supportive interventions required. After the first few days of ICU stay, characterized by clinical stabilization and invasive procedures, the sedation target is a calm patient, awake during the day and asleep at night. The use of deep levels of sedation to facilitate mechanical ventilation or painful procedures should be minimized with ventilation setting optimization and adequate analgesia, rather than deepening unconsciousness [12, 47].
The appropriate balance of sedation and analgesia is difficult to be achieved and maintained. Without a rational agreement upon “target levels” of sedation, different members of the healthcare team will have disparate treatment goals, increasing the chance for iatrogenic complications and potentially delaying recovery [48].
The target level of sedation should be discussed and defined at the beginning of each staff shift and reevaluated regularly as the clinical condition of the patient changes. The pharmacological treatment should be planned with the appropriate flexibility to allow titration to the desired endpoint, anticipating fluctuations in sedation requirements throughout the day. Frequent monitoring with validated tools improves communication among clinicians and plays an important role in detecting and treating pain, agitation, and delirium while avoiding excessive or prolonged sedation [25].
18.3.1 Sedation Assessment with Objective Methods
Within several objective methods to sedation assessment proposed, none of them have fully yielded satisfying results. For example, bispectral index (BIS) monitor is a four-channel electroencephalographic (EEG) monitor which generates a single number that correlates with depth of consciousness during general anesthesia. The poor correlation between BIS and validated ICU sedation scales is related with BIS values variability at the awake/agitated levels and of the electromyography (EMG) interference [49]. Based on the analysis of EEG signal irregularity, the entropy monitor also utilizes the EMG signal, which may provide information useful for assessing whether a patient is responding to an external, painful stimulus, but without adding useful information to sedation assessment. In this context, some advantages could be offered by the responsiveness index [50]. Auditory evoked potentials (AEP) are electrophysiological responses of the nervous system to standard sensory stimulation transmitted through headphones. These methods may have a role in monitoring sedation levels only in patients needing deep sedation, or receiving neuromuscular blocking agents, as in this circumstance sedation scales cannot be used [49].
Monitoring blood drug values is useful only when there is a correlation between plasmatic concentration and pharmacological effect [51]. However, ARDS patients may be affected by renal and hepatic dysfunctions that impair their ability to metabolize and excrete drugs. Moreover, hypoxia, inflammatory mediators, and abnormal diets are common, too, and all affect enzymatic function. Thus, this method cannot be recommended for sedation monitoring [52, 53]. Spontaneous, non-propulsive lower esophageal contractility (LOC) is definitely stress related and increases in frequency as the dose of anesthetic is reduced. Deepening of anesthesia resulted in progressive suppression of LOC. However, LOC has great inter-variability and is affected by drugs such as atropine. The electromyogram responsiveness is not sufficiently sensitive to monitor sedation in ICU patients [51].
Actigraphy provides a continuous measure of body movements and was initially developed to measure sleep-wake cycles. This small electronic device containing an accelerometer continuously senses and records minimal movements, summarizing such data in numerical form. Wrist actigraphy provides useful nonspecific observations in ICU patients. Even if it does not discriminate the lack (or the excess) of analgesics and sedatives from other acute neurological dysfunctions, preliminary observations suggest that the measurement of body movements could provide a timely indication of acute changes in neurological status generating motor agitation or hypoactive behavior [54]. This objective method is relatively new in this context. It presents interesting properties, worthy of future investigation [55].
18.3.2 Sedation Assessment with Subjective Methods
Individual assessments of sedation, performed at the bedside by nurses or physicians, can be hampered by a lack of objectivity. Guidelines recommend establishing a sedation target and regularly redefining it for each patient, using a validated sedation assessment scale. The use of such a scale is a key component of sedation algorithms [1, 4–6]. It helps in managing agitation and establishing a target level of sedation for medication titration, in order to promptly detect oversedation when the target level is exceeded. All sedation algorithms recommend to use a sedation scale, such as Ramsay Sedation Scale (RSS), Richmond Agitation-Sedation Scale (RASS), and Riker Sedation-Agitation Scale (SAS) [56].
The use of a scale to assess level of consciousness dates to the introduction of a six-point scale by Ramsay et al. (RSS) more than 40 years ago [57]. Nowadays, it continues to be a widely used scale for monitoring sedation in daily practice [58]. Some experts consider that it is more a scale of consciousness than a tool for the measurement of sedation. RSS has been extensively tested but it has never been validated. Moreover, it does not grade agitation. Consequently, this scale is excessively subjective and has poor validity. Many other scales have been proposed [59]; some of them are not validated. They are not recommended for clinical use.
The ideal scoring system should be easy, reliable, sensitive, and with minimal interobserver variability. Moreover, it should give no or minimal additional discomfort to the patient. Even though a complex scoring system is not suitable for the ICU, oversimplification brings risk of neglecting important information. Most of the proposed tools are a compromise between accuracy and time required for evaluation of sedation [60].
Recently developed scales often combine the sedation/arousal domain with an assessment of agitation, like the SAS, the RASS, the Motor Activity Assessment Scale (MAAS), the Observer’s Assessment of Alertness and Sedation (OAAS), the Nursing Instrument for the Communication of Sedation (NICS) [61], and the Bloomsbury Sedation Score (Fig. 18.3).
Unlike other validated instruments, the RASS separates verbal from physical stimulation so that the patient’s level of arousal may be graded according to the potency of the stimulus. Interestingly, RASS is validated also to assess patients’ sedation over time, both in spontaneously breathing/mechanically ventilated and in sedated/nonsedated critically ill patients.
Other multiple-item sedation scales are described in literature. The Adaptation to the Intensive Care Environment scale (ATICE) consists of five items [62]: awareness and comprehension combined in a “Conscious” domain; calmness, ventilator synchrony, and facial relaxation are combined in a “Tolerance” domain. As for ATICE, the Minnesota Sedation Assessment Tool (MSAT) evaluates the level of consciousness of patients receiving invasive mechanical ventilation. It measures arousability, spontaneous muscle activity, and global sedation quality. The Vancouver Interaction and Calmness Scale (VICS) consists of two five-item subscales quantifying separately calmness and interaction with operators. These more complex scoring systems are usually adopted in clinical trials to evaluate a new drug or a new objective tool for sedation assessment, whereas in daily practice, easily applied scores are usually preferred.
18.3.3 Choosing and Implementing an Evaluation Scale
Desirable features of sedation evaluation instruments should include rigorous multidisciplinary development; ease of administration, recall, and interpretation; well-designed discrete criteria for each level; sufficient sedation levels for effective drug titration; assessment of agitation; and demonstration of inter-rater reliability and validity in relevant patient populations [48]. Each ICU has to choose the best tools for its patient population and to plan specific intervention to introduce it in daily care [63].
Teaching protocols used for implementation of sedation scales have shown good results among ICU caregivers. Different methods have been used to implement evaluation tools in clinical practice. Typically, they are based on introductory in-service for nurses and operators followed by graded, staged educational interventions at regular intervals. Web-based, freely available teaching interventions have been also proposed (www.icudelirium.org, www.sedaicu.it).
Some emerging problems remain, particularly about the fluctuation of consciousness. ARDS patients are prone to sudden changes in their state of consciousness due to the effects of drugs, sleep disruption, organic and metabolic disease, or delirium. Assessment of sedation once a shift is indispensable but not sufficient. Among the different possibilities (minimal/maximal level, prevalent level, worst level), it is important to state the duration of each value within the observed shift. Sedation and agitation need to be reassessed both on a regular basis and during any clinical modification, to promptly capture all the modifications requiring intervention. Moreover, it is relatively common for patients to manifest sudden aggressive behavior when recovering from sedation and without fully awakening. For this reason, it is important to boost interdisciplinary communication between nurses and physicians in order to be aware of and prevent these problems.
Lastly, making a sedation assessment during the night is frequently challenging. Most analgesics and sedatives are known to make patients sleepy, but without reaching a restorative, physiological sleep [64]. If a critically ill patient appears calm and keeps his/her eyes closed during the night, he/she should not be stimulated just to make a sedation assessment. He/she could be observed during unavoidable procedures happening in the ICU during the night, in order to discriminate normal sleep (with arousals due to noise or light) from sedation or coma.
18.4 Clinical Practice Flowcharts for Pain, Agitation, and Delirium Management
Recognition that heavy sedation may increase mortality and morbidity has led to a new approach that maximizes the comfort of the patients while they remain awake, interactive, and oriented. This new approach relies on strategies such as daily interruptions of sedation, analgesia-based sedation, enteral sedation, avoidance of paralytic agents, early mobilization, and use of validated tools for sedation assessment [1]. In recent years, many guidelines have been proposed [4–6], representing a guide to symptom-oriented prevention, diagnosis, and treatment of delirium, anxiety, stress, and protocol-based analgesia, sedation, and sleep management. They comprehensively describe all attentions to be paid to perform the best neurological treatment. Nevertheless, even in guidelines with a high-quality rating, numerous recommendations have moderate or low levels of evidence [65].
Overall, the principal recommendation regarding analgesia is to evaluate pain and maintain its level ≤ 4/10, by beginning early than late the treatment of pain, and by using opioid drugs first, together with non-opioid and multimodal analgesia techniques. A simple and practical flowchart at the bedside is presented in Fig. 18.4.
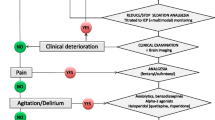
Regarding sedation, a target RASS of 0/−1 is recommended for all ICU patients, with the use of deep sedation reserved only for patients with specific indications (e.g., early ARDS patients, requiring neuromuscular blocking or prone positioning). In particular, intensivists should consider the specific indication and individual goal of sedation and the pharmacokinetics/pharmacodynamics of each drug used. Non-benzodiazepine drugs, such as propofol or dexmedetomidine, have to be preferred. The need for sedation varies widely among different patients and with the illness course, and it has to be defined among “deep sedation”, “cooperative/awake sedation,” and “no sedation,” always preferring superficial levels of sedation and promoting early mobilization. The use of containment measures in episodes of severe agitation has to be performed in this order: first verbal, then pharmacological, and finally physical, considering that neuroactive drugs should not be administered in excess as a form of “chemical immobilization.” In the sedation flowchart (Fig. 18.5), it is clear that defining a target and evaluating the actual level of sedation or agitation with validated tools are absolute priorities. The choice of the specific sedative drug, albeit important, comes only after a clinical reasoning focused on managing the organic/metabolic causes and the problems that may be caused by adjustable invasive devices.
Recommendations about delirium are prescribed first to identify modifiable risk factors (Fig. 18.6) and to detect regularly the appearance of delirium by using the CAM-ICU. In case of delirium appearance, it is indicated to use a non-pharmacological protocol first, along with the suspension or decreasing of deliriogenic therapies, and then to select the most appropriate drug (haloperidol, atypical antipsychotics, or dexmedetomidine; avoid the use of benzodiazepines) and titer its dose to the lowest effective dose.
18.5 Sedation Protocols Presented in Literature
Beyond the choice of the specific drug [66–68], the most frequently used method for administering sedatives is the continuous intravenous route, because of its pharmacokinetic properties [63]. Intravenous infusions present predictable and easy to handle onset/offset properties, justifying the search for an early goal-directed sedation strategy [69]. Although these characteristics are necessary in short ICU stays, they may be useless, or even dangerous, for patients requiring more than 3 days of mechanical ventilation [27]. When using potent drugs, it is easy to incur in overadministration albeit goals are established and adequate [25]. Moreover, daily awakening trials produce far-from-physiological neurological fluctuations [70], and continuous deep sedation does not permit patients to recall factual memories, which has been proven effective in preventing PTSD [71].
If continuous intravenous infusion is used, the daily interruption of sedatives and analgesics is recommended in order to reduce the total administered dosage and to perform a spontaneous breathing trial, if allowed by the respiratory condition of the patient. The purpose is to reduce the development of complications and the duration of mechanical ventilation [17]. This strategy may prove less effective if specific and ICU team-shared protocols are used [72].
Many quite different sedation protocols have been presented in literature [10, 73, 74]. Some of them essentially rely on the use of different drug doses [75], with the aim to different sedation levels [56, 76]. Other protocols are based on nursing-implemented algorithms [13, 77–79] or on analgesia-first sedation [21, 80]. Moreover, tested ways to optimize sedation management in ICU are patient-controlled sedation [81] or automated sedation in patients needing deep sedation [82], the use of inhaled halogenates [83, 84], or the enteral administration of drugs [28, 85]. All these protocols rely on the continuous and adequate neurological assessment made with validated tools, in order to measure not only pain, agitation, and delirium but also level of consciousness and patient mobilization.
The most promising protocol, in terms of efficacy, recommends to join the sedation strategies with early physiotherapy [86], mobilization [87], and occupational therapy [88, 89], also engaging patient families. Even if implementing such protocols is not simple [90], from the first presentation made in 2010 [91, 92], it has offered the best results in terms of effectiveness [93]. Briefly, following the acronym ABCDEF, the authors made these suggestions:
-
Assess, prevent, and manage pain.
-
Both spontaneous awakening trial and spontaneous breathing trial.
-
Choice of analgesia and sedation.
-
Delirium: assess, prevent, and manage.
-
Early mobility and exercise (goal-directed early mobilization).
-
Family engagement and empowerment.
18.6 Special Circumstances
Regarding ARDS patients needing extracorporeal life support systems (ECLS), there is a gray area about sedation, where safety aspects and the ability to positively influence recovery must be balanced. Patients on ECLS have numerous risk factors to develop both delirium during the ICU stay and PTSD after discharge. Hyperactive delirium or agitation can be life-threatening for these patients, so that a consequent monitoring and a symptomatic therapy of stress, anxiety, delirium, pain, and insomnia is essential to safely achieve a target RASS of 0. The higher level of alertness allows the patient to actively partake in physical exercises [94] that is considered a feasible and safe goal [95, 96]. International guidelines recommend a strict definition of sedation targets for patients on ECLS, including frequent clinical monitoring and continuous adjustment of the level of sedation required [4].
Positioning therapy has been demonstrated effective in ameliorating prognosis of severe ARDS patients. It is used for prophylaxis and treatment of respiratory dysfunctions and requires an individual sedation target. Changes of the position frequently represent a challenge for the symptomatic treatment of anxiety, stress, and pain. Therefore, a symptom-orientated therapy should be adapted for changing demands during positioning therapy. Though a deep sedation may be indicated for patient repositioning [97], an excessive sedation should be avoided through the use of objective tools based on EEG analysis, as previously described.
The use of non-depolarizing neuromuscular blockers in patients with severe ARDS is suggested during the first 48 ICU hours. Despite this practice, it is suggested not to routinely increase the dose of sedatives, when accompanied by an infusion of opiates, even in patients subjected to permissive hypercapnia [98]. To control respiratory rate, fentanyl is recommended as the analgesic of choice in patients with hemodynamic instability, bronchial asthma, or COPD, with respect to the other opiates. Methadone via the enteral route could be used in patients receiving opiates for more than 5 days, but still needing mechanical ventilation. Once tracheostomy is performed, it is advisable to consider a decrease in sedative and analgesic regimens.
After the hyperacute phase of respiratory failure, a defined sedation and analgesia monitoring and dose adjustment protocol is recommended, to shorten the weaning process. This protocol should include daily evaluation of sedation, an awakening test and a spontaneous breathing test. It is advisable not to use benzodiazepines in the withdrawal of MV. Dexmedetomidine is recommended in case of weaning difficulties, in patients with withdrawal syndrome, or after failed attempts of weaning secondary to agitation and delirium [67]. Low-dose remifentanil in continuous infusion is another effective alternative during weaning process. Music therapy is a possible non-pharmacological adjuvant to sedation [99]. Melatonin supplementation could be useful to decrease the need for sedative drugs, then shortening the ventilation length [100], and to restore the sleep-wake rhythm.
References
Sessler CN, Varney K (2008) Patient-focused sedation and analgesia in the ICU. Chest 133(2):552–565. doi:10.1378/chest.07-2026
Pandharipande PP, Girard TD, Jackson JC, Morandi A, Thompson JL, Pun BT, Brummel NE, Hughes CG, Vasilevskis EE, Shintani AK, Moons KG, Geevarghese SK, Canonico A, Hopkins RO, Bernard GR, Dittus RS, Ely EW (2013) Long-term cognitive impairment after critical illness. N Engl J Med 369(14):1306–1316. doi:10.1056/NEJMoa1301372
Fraser GL, Riker RR (2007) Sedation and analgesia in the critically ill adult. Curr Opin Anaesthesiol 20(2):119–123. doi:10.1097/ACO.0b013e32808255b4
Baron R, Binder A, Biniek R, Braune S, Buerkle H, Dall P, Demirakca S, Eckardt R, Eggers V, Eichler I, Fietze I, Freys S, Frund A, Garten L, Gohrbandt B, Harth I, Hartl W, Heppner HJ, Horter J, Huth R, Janssens U, Jungk C, Kaeuper KM, Kessler P, Kleinschmidt S, Kochanek M, Kumpf M, Meiser A, Mueller A, Orth M, Putensen C, Roth B, Schaefer M, Schaefers R, Schellongowski P, Schindler M, Schmitt R, Scholz J, Schroeder S, Schwarzmann G, Spies C, Stingele R, Tonner P, Trieschmann U, Tryba M, Wappler F, Waydhas C, Weiss B, Weisshaar G (2015) Evidence and consensus based guideline for the management of delirium, analgesia, and sedation in intensive care medicine. Revision 2015 (DAS-Guideline 2015) - short version. Ger Med Sci GMS E-Journal 13:Doc19. doi:10.3205/000223
Barr J, Fraser GL, Puntillo K, Ely EW, Gelinas C, Dasta JF, Davidson JE, Devlin JW, Kress JP, Joffe AM, Coursin DB, Herr DL, Tung A, Robinson BR, Fontaine DK, Ramsay MA, Riker RR, Sessler CN, Pun B, Skrobik Y, Jaeschke R (2013) Clinical practice guidelines for the management of pain, agitation, and delirium in adult patients in the intensive care unit. Crit Care Med 41(1):263–306. doi:10.1097/CCM.0b013e3182783b72
Celis-Rodriguez E, Birchenall C, de la Cal MA, Castorena Arellano G, Hernandez A, Ceraso D, Diaz Cortes JC, Duenas Castell C, Jimenez EJ, Meza JC, Munoz Martinez T, Sosa Garcia JO, Pacheco Tovar C, Palizas F, Pardo Oviedo JM, Pinilla DI, Raffan-Sanabria F, Raimondi N, Righy Shinotsuka C, Suarez M, Ugarte S, Rubiano S (2013) Clinical practice guidelines for evidence-based management of sedoanalgesia in critically ill adult patients. Med Intensiva/Sociedad Espanola de Medicina Intensiva y Unidades Coronarias 37(8):519–574. doi:10.1016/j.medin.2013.04.001
Ice CJ, Personett HA, Frazee EN, Dierkhising RA, Kashyap R, Oeckler RA (2016) Risk factors for dexmedetomidine-associated hemodynamic instability in noncardiac intensive care unit patients. Anesth Analg 122(2):462–469. doi:10.1213/ANE.0000000000001125
Nseir S, Makris D, Mathieu D, Durocher A, Marquette CH (2010) Intensive care unit-acquired infection as a side effect of sedation. Crit Care 14(2):R30
Pisani MA, Murphy TE, Araujo KL, Slattum P, Van Ness PH, Inouye SK (2009) Benzodiazepine and opioid use and the duration of intensive care unit delirium in an older population. Crit Care Med 37(1):177–183. doi:10.1097/CCM.0b013e318192fcf9
Jackson DL, Proudfoot CW, Cann KF, Walsh T (2010) A systematic review of the impact of sedation practice in the ICU on resource use, costs and patient safety. Crit Care 14(2):R59. doi:10.1186/cc8956
Riker RR, Fraser GL (2005) Adverse events associated with sedatives, analgesics, and other drugs that provide patient comfort in the intensive care unit. Pharmacotherapy 25(5 Pt 2):8S–18S
Cigada M, Corbella D, Mistraletti G, Forster CR, Tommasino C, Morabito A, Iapichino G (2008) Conscious sedation in the critically ill ventilated patient. J Crit Care 23(3):349–353. doi:10.1016/j.jcrc.2007.04.003
De Jonghe B, Bastuji-Garin S, Fangio P, Lacherade JC, Jabot J, Appere-De-Vecchi C, Rocha N, Outin H (2005) Sedation algorithm in critically ill patients without acute brain injury. Crit Care Med 33(1):120–127
Shehabi Y, Bellomo R, Reade MC, Bailey M, Bass F, Howe B, McArthur C, Seppelt IM, Webb S, Weisbrodt L (2012) Early intensive care sedation predicts long-term mortality in ventilated critically ill patients. Am J Respir Crit Care Med 186(8):724–731. doi:10.1164/rccm.201203-0522OC
Shehabi Y, Chan L, Kadiman S, Alias A, Ismail WN, Tan MA, Khoo TM, Ali SB, Saman MA, Shaltut A, Tan CC, Yong CY, Bailey M (2013) Sedation depth and long-term mortality in mechanically ventilated critically ill adults: a prospective longitudinal multicentre cohort study. Intensive Care Med 39(5):910–918. doi:10.1007/s00134-013-2830-2
Kress JP, Pohlman AS, O'Connor MF, Hall JB (2000) Daily interruption of sedative infusions in critically ill patients undergoing mechanical ventilation. N Engl J Med 342(20):1471–1477. doi:10.1056/NEJM200005183422002
Girard TD, Kress JP, Fuchs BD, Thomason JW, Schweickert WD, Pun BT, Taichman DB, Dunn JG, Pohlman AS, Kinniry PA, Jackson JC, Canonico AE, Light RW, Shintani AK, Thompson JL, Gordon SM, Hall JB, Dittus RS, Bernard GR, Ely EW (2008) Efficacy and safety of a paired sedation and ventilator weaning protocol for mechanically ventilated patients in intensive care (Awakening and Breathing Controlled trial): a randomised controlled trial. Lancet 371(9607):126–134. doi:10.1016/S0140-6736(08)60105-1
Breen D, Karabinis A, Malbrain M, Morais R, Albrecht S, Jarnvig IL, Parkinson P, Kirkham AJ (2005) Decreased duration of mechanical ventilation when comparing analgesia-based sedation using remifentanil with standard hypnotic-based sedation for up to 10 days in intensive care unit patients: a randomised trial [ISRCTN47583497]. Crit Care 9(3):R200–R210. doi:10.1186/cc3495
Spies C, Macguill M, Heymann A, Ganea C, Krahne D, Assman A, Kosiek HR, Scholtz K, Wernecke KD, Martin J (2011) A prospective, randomized, double-blind, multicenter study comparing remifentanil with fentanyl in mechanically ventilated patients. Intensive Care Med 37(3):469–476. doi:10.1007/s00134-010-2100-5
Tan JA, Ho KM (2009) Use of remifentanil as a sedative agent in critically ill adult patients: a meta-analysis. Anaesthesia 64(12):1342–1352. doi:10.1111/j.1365-2044.2009.06129.x
Strom T, Martinussen T, Toft P (2010) A protocol of no sedation for critically ill patients receiving mechanical ventilation: a randomised trial. Lancet 375(9713):475–480. doi:10.1016/S0140-6736(09)62072-9
Chevron V, Menard JF, Richard JC, Girault C, Leroy J, Bonmarchand G (1998) Unplanned extubation: risk factors of development and predictive criteria for reintubation. Crit Care Med 26(6):1049–1053
Guttormson JL, Chlan L, Weinert C, Savik K (2010) Factors influencing nurse sedation practices with mechanically ventilated patients: a U.S. national survey. Intensive Crit Care Nurs Off J Br Assoc Crit Care Nurses 26(1):44–50. doi:10.1016/j.iccn.2009.10.004
Goodwin H, Lewin JJ, Mirski MA (2012) ‘Cooperative sedation’: optimizing comfort while maximizing systemic and neurological function. Crit Care 16(2):217. doi:10.1186/cc11231
Martin J, Franck M, Fischer M, Spies C (2006) Sedation and analgesia in German intensive care units: how is it done in reality? Results of a patient-based survey of analgesia and sedation. Intensive Care Med 32(8):1137–1142. doi:10.1007/s00134-006-0214-6
Payen JF, Chanques G, Mantz J, Hercule C, Auriant I, Leguillou JL, Binhas M, Genty C, Rolland C, Bosson JL (2007) Current practices in sedation and analgesia for mechanically ventilated critically ill patients: a prospective multicenter patient-based study. Anesthesiology 106 (4):687–695; quiz 891-682. doi:10.1097/01.anes.0000264747.09017.da
Devlin JW (2008) The pharmacology of oversedation in mechanically ventilated adults. Curr Opin Crit Care 14(4):403–407. doi:10.1097/MCC.0b013e32830280b3
Mistraletti G, Mantovani ES, Cadringher P, Cerri B, Corbella D, Umbrello M, Anania S, Andrighi E, Barello S, Di Carlo A, Martinetti F, Formenti P, Spanu P, Iapichino G (2013) Enteral vs. intravenous ICU sedation management: study protocol for a randomized controlled trial. Trials 14:92. doi:10.1186/1745-6215-14-92
Granja C, Gomes E, Amaro A, Ribeiro O, Jones C, Carneiro A, Costa-Pereira A (2008) Understanding posttraumatic stress disorder-related symptoms after critical care: the early illness amnesia hypothesis. Crit Care Med 36(10):2801–2809. doi:10.1097/CCM.0b013e318186a3e7
Robinson BR, Berube M, Barr J, Riker R, Gelinas C (2013) Psychometric analysis of subjective sedation scales in critically ill adults. Crit Care Med 41(9 Suppl 1):S16–S29. doi:10.1097/CCM.0b013e3182a16879
Payen JF, Bru O, Bosson JL, Lagrasta A, Novel E, Deschaux I, Lavagne P, Jacquot C (2001) Assessing pain in critically ill sedated patients by using a behavioral pain scale. Crit Care Med 29(12):2258–2263
Gelinas C, Fillion L, Puntillo KA, Viens C, Fortier M (2006) Validation of the critical-care pain observation tool in adult patients. Am J Crit Care Off Publ Am Assoc Crit-Care Nurses 15(4):420–427
Sessler CN, Gosnell MS, Grap MJ, Brophy GM, O'Neal PV, Keane KA, Tesoro EP, Elswick RK (2002) The Richmond Agitation-Sedation Scale: validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med 166(10):1338–1344. doi:10.1164/rccm.2107138
Riker RR, Picard JT, Fraser GL (1999) Prospective evaluation of the Sedation-Agitation Scale for adult critically ill patients. Crit Care Med 27(7):1325–1329
Ely EW, Margolin R, Francis J, May L, Truman B, Dittus R, Speroff T, Gautam S, Bernard GR, Inouye SK (2001) Evaluation of delirium in critically ill patients: validation of the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU). Crit Care Med 29(7):1370–1379
Bergeron N, Dubois MJ, Dumont M, Dial S, Skrobik Y (2001) Intensive Care Delirium Screening Checklist: evaluation of a new screening tool. Intensive Care Med 27(5):859–864
Aissaoui Y, Zeggwagh AA, Zekraoui A, Abidi K, Abouqal R (2005) Validation of a behavioral pain scale in critically ill, sedated, and mechanically ventilated patients. Anesth Analg 101(5):1470–1476. doi:10.1213/01.ANE.0000182331.68722.FF
Ahlers SJ, van der Veen AM, van Dijk M, Tibboel D, Knibbe CA (2010) The use of the behavioral pain scale to assess pain in conscious sedated patients. Anesth Analg 110(1):127–133. doi:10.1213/ANE.0b013e3181c3119e
Pun BT, Gordon SM, Peterson JF, Shintani AK, Jackson JC, Foss J, Harding SD, Bernard GR, Dittus RS, Ely EW (2005) Large-scale implementation of sedation and delirium monitoring in the intensive care unit: a report from two medical centers. Crit Care Med 33(6):1199–1205
Morandi A, Pandharipande P, Trabucchi M, Rozzini R, Mistraletti G, Trompeo AC, Gregoretti C, Gattinoni L, Ranieri MV, Brochard L, Annane D, Putensen C, Guenther U, Fuentes P, Tobar E, Anzueto AR, Esteban A, Skrobik Y, Salluh JI, Soares M, Granja C, Stubhaug A, de Rooij SE, Ely EW (2008) Understanding international differences in terminology for delirium and other types of acute brain dysfunction in critically ill patients. Intensive Care Med 34(10):1907–1915. doi:10.1007/s00134-008-1177-6
Spronk PE, Riekerk B, Hofhuis J, Rommes JH (2009) Occurrence of delirium is severely underestimated in the ICU during daily care. Intensive Care Med 35(7):1276–1280. doi:10.1007/s00134-009-1466-8
Vasilevskis EE, Han JH, Hughes CG, Ely EW (2012) Epidemiology and risk factors for delirium across hospital settings. Best Pract Res Clin Anaesthesiol 26(3):277–287. doi:10.1016/j.bpa.2012.07.003
Pisani MA, Kong SY, Kasl SV, Murphy TE, Araujo KL, Van Ness PH (2009) Days of delirium are associated with 1-year mortality in an older intensive care unit population. Am J Respir Crit Care Med 180(11):1092–1097. doi:10.1164/rccm.200904-0537OC
Ely EW, Shintani A, Truman B, Speroff T, Gordon SM, Harrell FE Jr, Inouye SK, Bernard GR, Dittus RS (2004) Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. JAMA J Am Med Assoc 291(14):1753–1762. doi:10.1001/jama.291.14.1753
Mistraletti G, Pelosi P, Mantovani ES, Berardino M, Gregoretti C (2012) Delirium: clinical approach and prevention. Best Pract Res Clin Anaesthesiol 26(3):311–326. doi:10.1016/j.bpa.2012.07.001
Devlin JW, Fong JJ, Schumaker G, O’Connor H, Ruthazer R, Garpestad E (2007) Use of a validated delirium assessment tool improves the ability of physicians to identify delirium in medical intensive care unit patients. Crit Care Med 35 (12):2721–2724; quiz 2725
Strom T, Toft P (2011) Time to wake up the patients in the ICU: a crazy idea or common sense? Minerva Anestesiol 77(1):59–63
Sessler CN, Grap MJ, Ramsay MA (2008) Evaluating and monitoring analgesia and sedation in the intensive care unit. Crit Care 12(Suppl 3):S2. doi:10.1186/cc6148
Haenggi M, Ypparila-Wolters H, Buerki S, Schlauri R, Korhonen I, Takala J, Jakob SM (2009) Auditory event-related potentials, bispectral index, and entropy for the discrimination of different levels of sedation in intensive care unit patients. Anesth Analg 109(3):807–816. doi:10.1213/ane.0b013e3181acc85d
Walsh TS, Lapinlampi TP, Ramsay P, Sarkela MO, Uutela K, Viertio-Oja HE (2011) Responsiveness of the frontal EMG for monitoring the sedation state of critically ill patients. Br J Anaesth 107(5):710–718. doi:10.1093/bja/aer228
Mondello E, Siliotti R, Gravino E, Coluzzi F, David T, Sinardi AU (2005) Sedation monitoring in ICU. Minerva Anestesiol 71(9):487–496
LeBlanc JM, Dasta JF, Pruchnicki MC, Gerlach A, Cook C (2012) Bispectral index values, sedation-agitation scores, and plasma Lorazepam concentrations in critically ill surgical patients. Am J Crit Care Off Publ Am Assoc Crit Care Nurses 21(2):99–105. doi:10.4037/ajcc2012777
Masica AL, Girard TD, Wilkinson GR, Thomason JW, Truman Pun B, Nair UB, Light RW, Canonico AE, Dunn J, Pandharipande P, Shintani AK, Ely EW (2007) Clinical sedation scores as indicators of sedative and analgesic drug exposure in intensive care unit patients. Am J Geriatr Pharmacother 5(3):218–231. doi:10.1016/j.amjopharm.2007.10.005
Mistraletti G, Taverna M, Sabbatini G, Carloni E, Bolgiaghi L, Pirrone M, Cigada M, Destrebecq AL, Carli F, Iapichino G (2009) Actigraphic monitoring in critically ill patients: preliminary results toward an “observation-guided sedation”. J Crit Care 24(4):563–567. doi:10.1016/j.jcrc.2009.05.006
Grap MJ, Hamilton VA, McNallen A, Ketchum JM, Best AM, Arief NY, Wetzel PA (2011) Actigraphy: analyzing patient movement. Heart Lung J Crit Care 40(3):e52–e59. doi:10.1016/j.hrtlng.2009.12.013
Patel SB, Kress JP (2012) Sedation and analgesia in the mechanically ventilated patient. Am J Respir Crit Care Med 185(5):486–497. doi:10.1164/rccm.201102-0273CI
Ramsay MA, Savege TM, Simpson BR, Goodwin R (1974) Controlled sedation with alphaxalone-alphadolone. Br Med J 2(5920):656–659
Reschreiter H, Maiden M, Kapila A (2008) Sedation practice in the intensive care unit: a UK national survey. Crit Care 12(6):R152. doi:10.1186/cc7141
De Jonghe B, Cook D, Appere-De-Vecchi C, Guyatt G, Meade M, Outin H (2000) Using and understanding sedation scoring systems: a systematic review. Intensive Care Med 26(3):275–285
McGrane S, Pandharipande PP (2012) Sedation in the intensive care unit. Minerva Anestesiol 78(3):369–380
Mirski MA, LeDroux SN, Lewin JJ 3rd, Thompson CB, Mirski KT, Griswold M (2010) Validity and reliability of an intuitive conscious sedation scoring tool: the nursing instrument for the communication of sedation. Crit Care Med 38(8):1674–1684. doi:10.1097/CCM.0b013e3181e7c73e
De Jonghe B, Cook D, Griffith L, Appere-de-Vecchi C, Guyatt G, Theron V, Vagnerre A, Outin H (2003) Adaptation to the Intensive Care Environment (ATICE): development and validation of a new sedation assessment instrument. Crit Care Med 31(9):2344–2354. doi:10.1097/01.CCM.0000084850.16444.94
Brush DR, Kress JP (2009) Sedation and analgesia for the mechanically ventilated patient. Clin Chest Med 30 (1):131–141, ix. doi:10.1016/j.ccm.2008.09.001
Brown EN, Lydic R, Schiff ND (2010) General anesthesia, sleep, and coma. N Engl J Med 363(27):2638–2650. doi:10.1056/NEJMra0808281
Girardis M, Cantaroni C, Savoia G, Melotti R, Conti G (2016) A critical appraisal of the quality of analgosedation guidelines in critically ill patients. Minerva Anestesiol 82(2):230–235
Pandharipande PP, Pun BT, Herr DL, Maze M, Girard TD, Miller RR, Shintani AK, Thompson JL, Jackson JC, Deppen SA, Stiles RA, Dittus RS, Bernard GR, Ely EW (2007) Effect of sedation with dexmedetomidine vs lorazepam on acute brain dysfunction in mechanically ventilated patients: the MENDS randomized controlled trial. JAMA 298(22):2644–2653. doi:10.1001/jama.298.22.2644
Riker RR, Shehabi Y, Bokesch PM, Ceraso D, Wisemandle W, Koura F, Whitten P, Margolis BD, Byrne DW, Ely EW, Rocha MG (2009) Dexmedetomidine vs midazolam for sedation of critically ill patients: a randomized trial. JAMA 301(5):489–499. doi:10.1001/jama.2009.56
Jakob SM, Ruokonen E, Grounds RM, Sarapohja T, Garratt C, Pocock SJ, Bratty JR, Takala J (2012) Dexmedetomidine vs midazolam or propofol for sedation during prolonged mechanical ventilation: two randomized controlled trials. JAMA 307(11):1151–1160. doi:10.1001/jama.2012.304
Shehabi Y, Bellomo R, Reade MC, Bailey M, Bass F, Howe B, McArthur C, Murray L, Seppelt IM, Webb S, Weisbrodt L (2013) Early goal-directed sedation versus standard sedation in mechanically ventilated critically ill patients: a pilot study*. Crit Care Med 41(8):1983–1991. doi:10.1097/CCM.0b013e31828a437d
Svenningsen H, Egerod I, Videbech P, Christensen D, Frydenberg M, Tonnesen EK (2013) Fluctuations in sedation levels may contribute to delirium in ICU patients. Acta Anaesthesiol Scand 57(3):288–293. doi:10.1111/aas.12048
Jones C, Griffiths RD, Humphris G, Skirrow PM (2001) Memory, delusions, and the development of acute posttraumatic stress disorder-related symptoms after intensive care. Crit Care Med 29(3):573–580
Mehta S, Burry L, Cook D, Fergusson D, Steinberg M, Granton J, Herridge M, Ferguson N, Devlin J, Tanios M, Dodek P, Fowler R, Burns K, Jacka M, Olafson K, Skrobik Y, Hebert P, Sabri E, Meade M (2012) Daily sedation interruption in mechanically ventilated critically ill patients cared for with a sedation protocol: a randomized controlled trial. JAMA 308(19):1985–1992
Schweickert WD, Kress JP (2008) Strategies to optimize analgesia and sedation. Crit Care 12(Suppl 3):S6. doi:10.1186/cc6151
Sessler CN, Pedram S (2009) Protocolized and target-based sedation and analgesia in the ICU. Crit Care Clin 25 (3):489–513, viii. doi:10.1016/j.ccc.2009.03.001
Zhou Y, Jin X, Kang Y, Liang G, Liu T, Deng N (2014) Midazolam and propofol used alone or sequentially for long-term sedation in critically ill, mechanically ventilated patients: a prospective, randomized study. Crit Care 18(3):R122. doi:10.1186/cc13922
Samuelson KA, Lundberg D, Fridlund B (2008) Light vs. heavy sedation during mechanical ventilation after oesophagectomy–a pilot experimental study focusing on memory. Acta Anaesthesiol Scand 52(8):1116–1123. doi:10.1111/j.1399-6576.2008.01702.x
de Wit M, Gennings C, Jenvey WI, Epstein SK (2008) Randomized trial comparing daily interruption of sedation and nursing-implemented sedation algorithm in medical intensive care unit patients. Crit Care 12(3):R70. doi:10.1186/cc6908
Quenot JP, Ladoire S, Devoucoux F, Doise JM, Cailliod R, Cunin N, Aube H, Blettery B, Charles PE (2007) Effect of a nurse-implemented sedation protocol on the incidence of ventilator-associated pneumonia. Crit Care Med 35(9):2031–2036
Arias-Rivera S, Sanchez-Sanchez Mdel M, Santos-Diaz R, Gallardo-Murillo J, Sanchez-Izquierdo R, Frutos-Vivar F, Ferguson ND, Esteban A (2008) Effect of a nursing-implemented sedation protocol on weaning outcome. Crit Care Med 36(7):2054–2060. doi:10.1097/CCM.0b013e31817bfd60
Wilhelm W, Kreuer S (2008) The place for short-acting opioids: special emphasis on remifentanil. Crit Care 12(Suppl 3):S5. doi:10.1186/cc6152
Chlan LL, Weinert CR, Skaar DJ, Tracy MF (2010) Patient-controlled sedation: a novel approach to sedation management for mechanically ventilated patients. Chest 138(5):1045–1053. doi:10.1378/chest.09-2615
Le Guen M, Liu N, Bourgeois E, Chazot T, Sessler DI, Rouby JJ, Fischler M (2013) Automated sedation outperforms manual administration of propofol and remifentanil in critically ill patients with deep sedation: a randomized phase II trial. Intensive Care Med 39(3):454–462. doi:10.1007/s00134-012-2762-2
Mesnil M, Capdevila X, Bringuier S, Trine PO, Falquet Y, Charbit J, Roustan JP, Chanques G, Jaber S (2011) Long-term sedation in intensive care unit: a randomized comparison between inhaled sevoflurane and intravenous propofol or midazolam. Intensive Care Med 37(6):933–941. doi:10.1007/s00134-011-2187-3
Migliari M, Bellani G, Rona R, Isgro S, Vergnano B, Mauri T, Patroniti N, Pesenti A, Foti G (2009) Short-term evaluation of sedation with sevoflurane administered by the anesthetic conserving device in critically ill patients. Intensive Care Med 35(7):1240–1246. doi:10.1007/s00134-009-1414-7
Wanzuita R, Poli-de-Figueiredo LF, Pfuetzenreiter F, Cavalcanti AB, Westphal GA (2012) Replacement of fentanyl infusion by enteral methadone decreases the weaning time from mechanical ventilation: a randomized controlled trial. Crit Care 16(2):R49. doi:10.1186/cc11250
Balas MC, Burke WJ, Gannon D, Cohen MZ, Colburn L, Bevil C, Franz D, Olsen KM, Ely EW, Vasilevskis EE (2013) Implementing the awakening and breathing coordination, delirium monitoring/management, and early exercise/mobility bundle into everyday care: opportunities, challenges, and lessons learned for implementing the ICU Pain, Agitation, and Delirium Guidelines. Crit Care Med 41(9 Suppl 1):S116–S127. doi:10.1097/CCM.0b013e3182a17064
Morris PE, Goad A, Thompson C, Taylor K, Harry B, Passmore L, Ross A, Anderson L, Baker S, Sanchez M, Penley L, Howard A, Dixon L, Leach S, Small R, Hite RD, Haponik E (2008) Early intensive care unit mobility therapy in the treatment of acute respiratory failure. Crit Care Med 36(8):2238–2243. doi:10.1097/CCM.0b013e318180b90e
Llano-Diez M, Renaud G, Andersson M, Marrero HG, Cacciani N, Engquist H, Corpeno R, Artemenko K, Bergquist J, Larsson L (2012) Mechanisms underlying ICU muscle wasting and effects of passive mechanical loading. Crit Care 16(5):R209. doi:10.1186/cc11841
Schweickert WD, Pohlman MC, Pohlman AS, Nigos C, Pawlik AJ, Esbrook CL, Spears L, Miller M, Franczyk M, Deprizio D, Schmidt GA, Bowman A, Barr R, McCallister KE, Hall JB, Kress JP (2009) Early physical and occupational therapy in mechanically ventilated, critically ill patients: a randomised controlled trial. Lancet 373(9678):1874–1882. doi:10.1016/S0140-6736(09)60658-9
Hodgson C, Bellomo R, Berney S, Bailey M, Buhr H, Denehy L, Harrold M, Higgins A, Presneill J, Saxena M, Skinner E, Young P, Webb S (2015) Early mobilization and recovery in mechanically ventilated patients in the ICU: a bi-national, multi-centre, prospective cohort study. Crit Care 19:81. doi:10.1186/s13054-015-0765-4
Vasilevskis EE, Ely EW, Speroff T, Pun BT, Boehm L, Dittus RS (2010) Reducing iatrogenic risks: ICU-acquired delirium and weakness–crossing the quality chasm. Chest 138(5):1224–1233. doi:10.1378/chest.10-0466
Pandharipande P, Banerjee A, McGrane S, Ely EW (2010) Liberation and animation for ventilated ICU patients: the ABCDE bundle for the back-end of critical care. Crit Care 14(3):157. doi:10.1186/cc8999
Balas MC, Vasilevskis EE, Olsen KM, Schmid KK, Shostrom V, Cohen MZ, Peitz G, Gannon DE, Sisson J, Sullivan J, Stothert JC, Lazure J, Nuss SL, Jawa RS, Freihaut F, Ely EW, Burke WJ (2014) Effectiveness and safety of the awakening and breathing coordination, delirium monitoring/management, and early exercise/mobility bundle. Crit Care Med 42(5):1024–1036. doi:10.1097/CCM.0000000000000129
Rahimi RA, Skrzat J, Reddy DR, Zanni JM, Fan E, Stephens RS, Needham DM (2013) Physical rehabilitation of patients in the intensive care unit requiring extracorporeal membrane oxygenation: a small case series. Phys Ther 93(2):248–255. doi:10.2522/ptj.20120336
Del Sorbo L, Pisani L, Filippini C, Fanelli V, Fasano L, Terragni P, Dell'Amore A, Urbino R, Mascia L, Evangelista A, Antro C, D'Amato R, Sucre MJ, Simonetti U, Persico P, Nava S, Ranieri VM (2015) Extracorporeal Co2 removal in hypercapnic patients at risk of noninvasive ventilation failure: a matched cohort study with historical control. Crit Care Med 43(1):120–127. doi:10.1097/CCM.0000000000000607
Fuehner T, Kuehn C, Hadem J, Wiesner O, Gottlieb J, Tudorache I, Olsson KM, Greer M, Sommer W, Welte T, Haverich A, Hoeper MM, Warnecke G (2012) Extracorporeal membrane oxygenation in awake patients as bridge to lung transplantation. Am J Respir Crit Care Med 185(7):763–768. doi:10.1164/rccm.201109-1599OC
Kredel M, Bischof L, Wurmb TE, Roewer N, Muellenbach RM (2014) Combination of positioning therapy and venovenous extracorporeal membrane oxygenation in ARDS patients. Perfusion 29(2):171–177. doi:10.1177/0267659113502834
Cheng IW, Eisner MD, Thompson BT, Ware LB, Matthay MA (2005) Acute effects of tidal volume strategy on hemodynamics, fluid balance, and sedation in acute lung injury. Crit Care Med 33(1):63–70 discussion 239-240
Chlan LL, Weinert CR, Heiderscheit A, Tracy MF, Skaar DJ, Guttormson JL, Savik K (2013) Effects of patient-directed music intervention on anxiety and sedative exposure in critically ill patients receiving mechanical ventilatory support: a randomized clinical trial. JAMA 309(22):2335–2344. doi:10.1001/jama.2013.5670
Mistraletti G, Umbrello M, Sabbatini G, Miori S, Taverna M, Cerri B, Mantovani ES, Formenti P, Spanu P, D'Agostino A, Salini S, Morabito A, Fraschini F, Reiter RJ, Iapichino G (2015) Melatonin reduces the need for sedation in ICU patients: a randomized controlled trial. Minerva Anestesiol 81(12):1298–1310
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Mistraletti, G., Formenti, P., Sabbatini, G. (2017). Sedation. In: Chiumello, D. (eds) Acute Respiratory Distress Syndrome. Springer, Cham. https://doi.org/10.1007/978-3-319-41852-0_18
Download citation
DOI: https://doi.org/10.1007/978-3-319-41852-0_18
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-41850-6
Online ISBN: 978-3-319-41852-0
eBook Packages: MedicineMedicine (R0)