Abstract
Objective
This study carried out the first patient-oriented survey on the practice of analgesia and sedation in German intensive care units, examining whether the goals of early spontaneous breathing and awake, cooperative patients are achieved.
Design
A postal survey was sent to 261 hospitals in Germany. Each hospital received three patient-oriented forms with questions regarding current agents and techniques for analgesia and sedation of a specific patient. Responses were obtained from 220 (84%) hospitals which returned 305 questionnaires.
Results
Patients' Ramsay sedation scale was significantly higher in all phases of analgesia and sedation, indicating that the patients were more deeply sedated than currently intended by the therapist. Propofol was used for most of the patients during short-term sedation (57%) and during weaning (48%). The preferred agent for sedation longer than 72 h was midazolam (66%).
Conclusion
The choice of agents and techniques for analgesia and sedation in the intensive care unit thus follows the German guidelines. The fact that the patients were more deeply sedated than intended by the therapist in all phases of sedation may be due to the low use of sedation scales and clinical practice guidelines or to the lack of training in using these techniques.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Critical care therapies such as ventilation, invasive procedures, and other measures inducing pain or stress require analgesia and sedation of the patient. Adequate analgesia and sedation is supposed to prevent stress-induced reactions [1, 2, 3] and to optimize patient comfort. Goal-oriented analgesia and sedation [4, 5] with the use of clinical practice guidelines, scoring systems, and targeted selection of agents for analgesia and sedation of the intensive care patient achieves an early spontaneous breathing [6], and with it a quick weaning and shorter stays in the intensive care unit [7]. German guidelines for analgesia and sedation recommendations for the different phases of analgesia and sedation are based upon the available literature [8]. Sedation that is too deep can have negative side effects [9, 10, 11] including increased risk of venous thrombosis, bowel motility problems, hypotension, and a prolonged stay in the intensive care unit (ICU), which result in increased costs [12, 13, 14]. The use of scoring systems is a substantial part of a patient-oriented analgesia and sedation. The administration of agents for analgesia and sedation can be properly controlled only by specifying the level of analgesia and sedation and monitoring it with scoring systems. The requirements for an ideal analgesia and sedation are the ability to sedate the patient deeply for necessary procedures, but with medication of short duration so that the patient can be quickly responsive and cooperative [4]. Martin et al. [15] showed in their survey about the practice of analgesia and sedation that only 8% of German ICUs use scoring systems routinely to monitor analgesia and sedation. Soliman et al. [16] conducted a survey in 16 European countries on the current practice of sedation. They found distinct differences between countries in the practice of analgesia and sedation. The most commonly used medication for continuous sedation in Europe is midazolam, whereas in the United States in 2002 the Society of Critical Care Medicine [17], recommended midazolam and propofol as the preferred medication for short-term sedation and lorazepam for long-term sedation. The preferred analgesics in English-speaking countries are morphine and fentanyl.
The goal of the present survey was to determine the current practice of analgesia and sedation in German ICUs run by anesthesiologists and compares these to the recommendations of the German evidence and consent-based guidelines. In contrast to all prior surveys, participants of the study were advised to collect the data at the patient's bedside. This makes it possible for the first time to assess whether the demands on modern analgesia and sedation in ICUs are achieved.
Materials and methods
We selected a simple random sample of one-third of the addresses (254 general hospitals and 15 university ICUs) from an address database of the German Society for Anesthesiology and Intensive Care Medicine (Deutsche Gesellschaft für Anästhesiologie und Intensivmedizin) that contains a total of 808 addresses of ICUs run by an anesthesiological department (45 university hospitals and 763 general hospitals). The letter proved undeliverable in the case of eight hospitals although it was sent up to four times to the hospitals between May and October 2002. Questions concerned the structure and procedures for analgesia and sedation for every department. Three patient-oriented questionnaires were included, and participants were asked arbitrarily to select one to three patients who were presently under sedation and to include them in the study, with the forms being filled in at the patient's bedside. This was left to the discretion of the physician. For organizational reasons neither the time nor the day of the data collection was predetermined. To maximize return rate the study was designed to leave it open to the participants of the survey.
Participants were asked about the agents used and the desired depth of sedation. Data regarding depth of sedation were to be specified on the Ramsay et al. [18] sedation scale (RSS) which was part of the assessment form; translation of the scale into German is published in the German S2e guidelines for analgesia and sedation [8]. Both the desired and the actual depth of sedation were to be determined, the latter at the patient's bedside. Only the desired depth of sedation for most of the day was asked. The length of sedation was categorized according to American [17] and German guidelines [8]: shorter than 24 h (“short-term sedation”), 24–72 h (“medium-term sedation”), longer than 72 h (“long-term sedation”), and during weaning from ventilation (“weaning sedation”). Table 1 presents patients' clinical characteristics in each of the categories of sedation. Age, sex, American Society of Anesthesiologists physical status classification, and number of renal replacement therapies did not differ significantly between groups (Table 1). Groups differed significantly in Sequential Organ Failure Assessment (SOFA) score [19]. Patients receiving long-term or weaning sedation had significantly more renal replacement therapies. Any interventions such as nursing activities were to be avoided during the monitoring of sedation. No daily wake-up trial was requested in the questionnaire. No hospital even mentioned this in the comments to the questionnaire.
Questionnaires were returned by 220 of the 261 departments (84%), and 305 patient-oriented forms were received. At this response rate the possibility of nonresponder bias is negligible (standard 2–2, NCES Statistical Standards; http://nces.ed.gov/statprog/2002/std2_2.asp). The data were collected in a Microsoft Access 97 database and analyzed with the programs Microsoft Excel 9.0 and SPSS for Windows version 10.07. Univariate statistical analyses were carried out depending on the scaling of the data using either the Mann–Whitney U test or the χ2 test. The analyses were to be understood as exploratory, and therefore no multiple adjustments were carried out. Statistical Significance (in the sense of exploratory analyses) was set at the p > 0.05 level.
Results
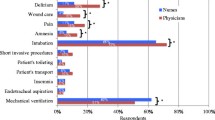
The actual depth of sedation was found to be significantly deeper than desired in each of the four groups (Fig. 1): in 62.4% of those under short-term sedation, 42.6% of those under medium-term sedation, 39.5% under long-term sedation, and 43.9% of those under weaning sedation (Fig. 2). Only in the long-term and weaning sedation groups was the RSS value in some of the patients lower than the desired value: 5.2% and 3.5%, respectively (Fig. 2). Among patients under long-term sedation the desired RSS value was 0, 2, or 3 in 44% of cases, but the desired value was achieved in only 28% of these; in 55% of cases the desired value was 4 or 5, but the desired value was achieved in only 68%; and in 1% of cases a value of 6 was desired, but this was achieved in only 6%.
As a continuous agent propofol (57%) was used significantly more often than midazolam for short-term sedation (36%; p = 0.005, χ2) and for weaning sedation (48% vs. 34%; p = 0.03, χ2), for long-term sedation midazolam was used more often (66.2%; p < 0.001, χ2), while in the group receiving medium-term sedation there was no difference (Table 2). For short-term analgesia piritramide was used significantly more often (50%) than fentanyl (10%) and sufentanil (24%). In all other groups sufentanil was administered more often than fentanyl and piritramide (p < 0.05, χ2; Table 2). Central neuroaxial blockade with an epidural catheter was used significantly more often during analgesia and short-term sedation (14%) than in long-term (1%) and weaning sedation (5%). There was also a significant difference between the groups receiving medium-term (12%) and long-term sedation (1%; p < 0.05; χ2 Table 2). As adjuvant technique for analgesia and sedation clonidine was used in 39% of patients under sedation during weaning, which differed significantly from the practice in the other groups (p < 0.05, χ2, Table 2). Nonsteroidal anti-inflammatory drugs were used significantly more often in short-term (17%) than long-term sedation (3%); there was no difference between the other groups. In the long-term sedation group ketamine (S) was administered significantly more often (12%) than in the short-term (5%) and weaning sedation patients (5%; p < 0.05, χ2 Table 2).
Discussion
As demonstrated by this study, the actual RSS in a substantial proportion of patients in all phases of analgesia and sedation is higher than which is aimed at, indicating a level of sedation that is deeper than desired. The setting of sedation goals contributes substantially to achieving a goal-oriented sedation [5, 20]. Recent studies show that close monitoring with the help of scoring systems lead to a reduction in the length of stay in the ICU and in hospital [20]. Technical methods such as the bispectral index and auditory evoked potential monitoring were not used in any patient in our survey [21, 22, 23]. The use of scoring systems is also not very well established in Germany; current studies cite a range of 8 to 49% for their use [15, 16]. A new finding of the present study is that even despite the use of scoring systems patients often do not achieve the desired sedation.
The most likely reason for this inadequate use of the RSS is a training deficit. Indicative is the information that a RSS value between 1 and 2 is aimed at during short-term and weaning sedation. Among those receiving long-term sedation the desired RSS was 4 or less in 44% of patients; however, this value was actually achieved in only 28% of those patients. This is confirmed by the data reported by Otter et al. [24] and Kress et al. [20] that patients profit from lower RSS values even during long-term sedation. This information is worrying, considering that a sedation scale obviously cannot be used without training, and that the implementation of an adequate sedation is outcome relevant. Thus both oversedation [20] and undersedation may therefore have an impact on the complication rate, ICU length of stay, and lethality [25]. The German guidelines that show grade B for the use of scoring systems [8] are helpful in establishing evidence-based medicine, but we cannot assume that evidence-based guidelines may be put into action without training of technical and nontechnical skills [26].
Another aspect of poorly implemented sedation skills is indicated by the fact that only 20% of German ICUs have a written procedure instruction [15]. In the 1987 survey of ICUs in the United Kingdom by Bion et al. [27] 40% of respondents stated that they had established a formal procedure for sedation. Other surveys have shown that the use of clinical practice guidelines permits a decrease in the duration of sedation and ventilation and thus a reduction in costs [27, 28]. Mascia and coworkers [29] showed that duration of ventilation and length of ICU and overall hospital stay decrease only with the use of written clinical practice guidelines.
Implementing concepts for medication at a national level appears to be much easier. Propofol is recommended in the American [17] and German guidelines [8] for short-term sedation, and for long-term sedation lorazepam is recommended in the American guidelines and midazolam in the German guidelines. In our survey the most commonly used agent for short-term sedation and for weaning from ventilation was propofol. Midazolam was used mainly for long-term sedation. Lorazepam, recommended in the American guidelines [17] for long-term sedation, was not used by any department. Whereas the American guidelines recommend fentanyl, hydromorphone, and morphine for analgesia in all phases [17], we found piritramide to be used preferably for short-term analgesia. This German preference is based on the view that piritramide achieves a lower incidence of nausea and vomiting than morphine [30]. In all other phases sufentanil was preferred. While the United Kingdom survey [31] in 2000 found that alfentanil was very often used, this opioid did not play a role in German ICUs. Our results correspond to those of the European-wide survey by Soliman et al. [16] and the German survey by Martin et al. [15]. The actual practice of pharmacological analgesia conforms with the German guidelines [8].
Central neuroaxial techniques during sedation were most often used during short-term (14%) and medium-term sedation (12%). The studies by Brodner et al. [32] and by Beattie et al. [33] and the meta-analysis by Rodgers et al. [34] showed that perioperative use of epidural analgesia shortens ICU length of stay and reduces the incidence of cardiac and pulmonary events. The use of epidural analgesia during analgesia and sedation in the intensive care unit is recommended in the German guidelines. Clonidine as an adjuvant for sedation was preferably used in patients during weaning (39%). A reasonable use of this agent during weaning was shown by Walz et al. [35]. Bohrer and coworkers [36] reported that the requirements for opioids and sedation may be reduced with the use of clonidine. We found that ketamine (S) is most often used during long-term sedation. Only few studies have examined long-term sedation with ketamine, as demonstrated by Ostermann et al. [37] in their review. One important reason for the use of ketamine is the low effect on bowel motility [38].
Nonsteroidal anti-inflammatory drugs were used preferably during short-term sedation (17%). The low use of these agents can be explained by its spectrum of side effects [8]. Neuromuscular blocking agents were only rarely used in all phases of analgesia and sedation. The German guidelines provide no recommendation for the use of neuromuscular blocking agents in the scope of analgesia and sedation in ICUs [8]. Nasal intubation is only rarely performed. Our results show that the number of tracheotomized patients increases with the duration of ventilation. Rumbak et al. [39] showed that in patients who are ventilated for a prolonged time mortality is lower once they are tracheotomized than in patients who are intubated.
One of the limitations of this study was that the survey was carried out only in ICUs run by a department of anesthesiology. No data were collected from ICUs run by other specialties. Hack and colleagues [40] showed that most interdisciplinary ICUs in general hospitals in Germany are run by departments of anesthesiology.
Conclusion
For the first time in a survey on analgesia and sedation current patient data were collected which present a realistic view of the practice of analgesia and sedation. We found that German ICUs run by anesthesiologists administer mainly propofol as a short-acting agent, and that for long-term sedation the benzodiazepine midazolam is used. As a German preference for analgesia during short-term sedation piritramide is used and in the other phases the opioid sufentanil. The epidural analgesia is preferred postoperatively during short-term sedation. As an adjuvant agent during weaning clonidine is used most often. Nonsteroidal anti-inflammatory drugs play only a minor role for analgesia and sedation.
This survey shows that the guidelines for pharmacological analgesia and sedation [8] are implemented as far as possible. However, a proportion of patients in all phases are more deeply sedated than desired. This is explained by the fact that only a small part of the hospitals have a written procedure instruction or a scoring system to monitor the analgesia and sedation established as the survey of Martin et al. [15] showed. Numerous publications have shown that the consistent use of these methods after prior training avoids sedation that is too deep and may reduce the time of ventilation and length of stay in the intensive care unit [20, 27, 30].
References
Koepke JP (1989) Effect of environmental stress on neural control of renal function. Miner Electrolyte MeTable 15:83–87
Bonica JJ (1987) Importance of effective pain control. Acta Anaesthesiol Scand Suppl 85:1–16
Lewis KS, Whipple JK, Michael KA, Quebbeman EJ (1994) Effect of analgesic treatment on the physiological consequences of acute pain. Am J Hosp Pharm 51:1539–1554
Tonner PH, Weiler N, Paris A, Scholz J (2003) Sedation and analgesia in the intensive care unit. Curr Opin Anaesthesiol 16:113–121
Schaffrath E, Kuhlen R, Tonner PH (2004) [Analgesia and sedation in intensive care medicine.]. Anaesthesist 53:1111–1130
Putensen C, Zech S, Wrigge H, Zinserling J, Stuber F, Von Spiegel T, Mutz N (2001) Long-term effects of spontaneous breathing during ventilatory support in patients with acute lung injury. Am J Respir Crit Care Med 164:43–49
Tung A, Rosenthal M (1995) Patients requiring sedation. Crit Care Clin 11:791–802
Martin J, Bäsell K, Bürkle H, Hommel J, Huth G, Kessler P, Kretz FJ, Putensen C, Quintel M, Tonner P et al. (2005) Analgesie und Sedierung in der Intensivmedizin-S2-Leitlinien der Deutschen Gesellschaft für Anästhesiologie und Intensivmedizin. Anasth Intensivmed 46:1–20
Merriman HM (1981) The techniques used to sedate ventilated patients. A survey of methods used in 34 ICUs in Great Britain. Intensive Care Med 7:217–224
Gast PH, Fischer A, Sear JW (1984) Intensive care sedation now. Lancet 8407:863–864
Miller-Jones CMH, Williams JH (1980) Sedation for ventilation. A retrospective study to ventilated patients. Anaesthesia 35:1104–1106
Burns AM, Shelly MP, Park GR (1992) The use of sedative agents in critically ill patients. Drugs 43:507–515
Kollef MH, Levy NT, Ahrens TS, Schaiff R, Prentice D, Sherman G (1998) The use of continuous i.v. sedation is associated with prolongation of mechanical ventilation. Chest 114:541–548
Durbin CG Jr (1994) Sedation in the critically ill patient. New Horiz 2:64–74
Martin J, Parsch A, Franck M, Wernecke KD, Fischer M, Spies C (2005) Practice of sedation and analgesia in German intensive care units: results of a national survey. Crit Care 9:R117–R123
Soliman H, Melot C, Vincent J (2001) Sedative and analgesic practice in the intensive care unit: the results of a European survey. Br J Anaesth 87:186–192
Jacobi J, Fraser GL, Coursin DB, Riker RR, Fontaine D, Wittbrodt ET, Chalfin DB, Masica MF, Bjerke HS, Coplin WM et al. (2002) Clinical practice guidelines for the sustained use of sedatives and analgesics in the critically ill adult. Crit Care Med 30:119–141
Ramsay MA, Savege TM, Simpson BR, Goodwin R (1974) Controlled sedation with alphaxalone-alphadolone. BMJ 2:656–659
Vincent JL, Moreno R, Takala J, Willatts S, De Mendonca A, Bruining H, Reinhart CK, Suter PM, Thijs LG (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 22:707–710
Kress JP, Pohlman AS, O' Connor M, Hall JB (2000) Daily interruption of sedative infusions in critically ill patients undergoing mechanical ventilation. N Engl J Med 342:1471–1477
Doi M, Morita K, Mantzaridis H, Sato S, Kenny GN (2005) Prediction of responses to various stimuli during sedation: a comparison of three EEG variables. Intensive Care Med 31:41–47
Andrews P, Azoulay E, Antonelli M, Brochard L, Brun-Buisson C, Dobb G, Fagon JY, Gerlach H, Groeneveld J, Mancebo J et al. (2006) Year in review in intensive care medicine, 2005. II. Infection and sepsis, ventilator-associated pneumonia, ethics, haematology and haemostasis, ICU organisation and scoring, brain injury. Intensive Care Med 32:380–390
Walsh TS, Ramsay P, Kinnunen R (2004) Monitoring sedation in the intensive care unit: can “black boxes” help us? Intensive Care Med 30:1511–1513
Otter H, Martin J, Bäsell K, von Heymann C, Hein O, Böllert P, Jänsch P, Behnisch I, Wernecke K-D, Konertz W et al. (2005) Validity and reliability of the DDS for severity of delirium in the ICU. Neurocrit Care 2:150–158
Ely EW, Shintani A, Truman B, Speroff T, Gordon SM, Harrell FE Jr, Inouye SK, Bernard GR, Dittus RS (2004) Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. JAMA 291:1753–1762
Blum RH, Raemer DB, Carroll JS, Sunder N, Felstein DM, Cooper JB (2004) Crisis resource management training for an anaesthesia faculty: a new approach to continuing education. Med Educ 38:45–55
Bion JF, Ledingham IM (1987) Sedation in intensive care-a postal survey. Intensive Care Med 13:215–216
MacLaren R, Plamondon JM, Ramsay KB, Rocker GM, Patrick WD, Hall RI (2000) A prospective evaluation of empiric versus protocol-based sedation and analgesia. Pharmacotherapy 20:662–672
Mascia MF, Koch M, Medicis JJ (2000) Pharmacoeconomic impact of rational use guidelines on the provision of analgesia, sedation, and neuromuscular blockade in critical care. Crit Care Med 28:2300–2306
Breitfeld C, Peters J, Vockel T, Lorenz C, Eikermann M (2003) Emetic effects of morphine and piritramide. Br J Anaesth 91:218–223
Murdoch S, Cohen A (2000) Intensive care sedation: a review of current British practice. Intensive Care Med 26:922–928
Brodner G, Van Aken H, Hertle L, Fobker M, Von Eckardstein A, Goeters C, Buerkle H, Harks A, Kehlet H (2001) Multimodal perioperative management-combining thoracic epidural analgesia, forced mobilization, and oral nutrition-reduces hormonal and metabolic stress and improves convalescence after major urologic surgery. Anesth Analg 92:1594–1600
Beattie WS, Badner NH, Choi P (2001) Epidural analgesia reduces postoperative myocardial infarction: a meta-analysis. Anesth Analg 93:853–858
Rodgers A, Walker N, Schug S, McKee A, Kehlet H, van Zundert A, Sage D, Futter M, Saville G, Clark T et al. (2000) Reduction of postoperative mortality and morbidity with epidural or spinal anaesthesia: results from overview of randomised trials. BMJ 321:1493
Walz M, Mollenhoff G, Muhr G (1999) Verkürzung der Weaningphase nach maschineller Beatmung durch kombinierte Gabe von Clonidin und Sufentanil. Chirurg 70:66–73
Bohrer H, Bach A, Layer M, Werning P (1990) Clonidine as a sedative adjunct in intensive care. Intensive Care Med 16:265–266
Ostermann ME, Keenan SP, Seiferling RA, Sibbald WJ (2000) Sedation in the intensive care unit: a systematic review. JAMA 283:1451–1459
Zielmann S, Grote R (1995) Auswirkungen der Langzeitsedierung auf die intestinale Funktion. Anaesthesist 44 Suppl 3:549–558
Rumbak MJ, Newton M, Truncale T, Schwartz SW, Adams JW, Hazard PB (2004) A prospective, randomized, study comparing early percutaneous dilational tracheotomy to prolonged translaryngeal intubation (delayed tracheotomy) in critically ill medical patients. Crit Care Med 32:1689–1694
Hack G, Götz E, Sorgatz H, van Eimeren W, Wulff A (2000) Umfrage zur Situation der Anästhesiologie in Deutschland. Anasth Intensivmed 41:535–541
Author information
Authors and Affiliations
Corresponding author
Additional information
J.M. has been employed by GlaxoSmithKline and B.Braun to present educational talks on therapy for analgesia and sedation and economics. The authors declare that they have no competing or financial interests.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Martin, J., Franck, M., Fischer, M. et al. Sedation and analgesia in German intensive care units: how is it done in reality? Results of a patient-based survey of analgesia and sedation. Intensive Care Med 32, 1137–1142 (2006). https://doi.org/10.1007/s00134-006-0214-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-006-0214-6