Abstract
Methylobacterium spp. includes a group of stringently aerobic, Gram-negative, pink-pigmented, facultatively methylotrophs (PPFM) belonging to α-proteobacteria and are capable of growing on one-carbon compounds, such as formate, formaldehyde, methanol and methylamine or sometimes on multi-carbon compounds like diethyl ether and trimethyl amines. Significance of these bacteria for plant-growth promotion by the possible mechanisms include production of phytohormones, IAA, cytokinins, ACC-deaminase and perform nitrogen metabolism by means of bacterial urease, establish efficient nitrogen (N2)-fixing symbioses by nodulating legume roots; production of exopolysaccharides (EPS) and Poly-β-hydroxybutyrate (PHB) accumulation and abiotic stress endurance. These organisms induce systemic resistance by production of siderophores and proteins like phenylalanine ammonia lyase, peroxidase, chitinase and β-1,3-glucanase and phenolic compounds. On the other hand, they also promote the biodegradation of polycyclic aromatic hydrocarbon (PAH). In spite of their plant-growth promotional traits, commercialization of the Methylobacterium strains as bioinoculant have been hindered constantly.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Methylobacterium
- Pink-pigmented
- Facultatively methylotrophic
- Plant-growth promotion
- Biocontrol
- Quorum sensing
- Induced systemic resistance (ISR)
1 Introduction
The omnipresent occurrence of Methylobacterium on plant surfaces makes them a model for the study of plant-microbe interaction, and motivating approach for realizing the particular traits that these bacteria having on plant-growth promoting attribute. They utilize the gaseous methanol that is emitted by the plants through the stomata as carbon and energy sources, and promote the growth of their host through release of different metabolites. Methylobacterium strains have been localized as endosymbionts within cells in the buds. One species, Methylobacterium podarium is thought to be part of the natural human foot microflora. Methylobacterium have even been found living inside the human mouth. Actually, the members of PPFM are ubiquitous in nature and found in a variety of habitats including phyllosphere, rhizosphere, root nodules, dust, freshwater, drinking water, lake sediments, etc. (Corpe and Rheem 1989). Their association with more than 70 plant species makes them potential agents for plant-growth promotion and biocontrol against diseases (Holland and Polacco 1994).
The Methylotrophs are defined as those growing on C1 compounds like methanol, formaldehyde, formate and methylamine. Based on their utilization pattern, they are obligate methylotrophs, not able to grow on multi-carbon compounds but if grown on methanol or methylamine but not on methane they are strictly aerobic Gram-negative and classified under two genera, e.g. Methylophilus and Methylobacillus. In case if they utilize methane then they are called methanotrophs. Methanotrophs are Gram-negative bacteria and classified under five genera: Methylomonas, Methylobacter, Methylococcus, Methylosinus and Methylocystis . All methanotroph forms extensive intracellular membranes and resting cells, either as cysts or exospores. The intracellular membranes are hypothesizing to be concerned in methane oxidation. On the other hand, Methylotrophs growing on C1 compounds and multi-carbon compounds, such as trimethylamine, dimethyl ether, dimethyl carbonate are called facultative methylotroph. A number of Gram-positive and Gram-negative bacterial genera include Bacillus, Acetobacter, Mycobacterium, Arthrobacter, Hyphomicrobium, Methylobacterium and Nocardia. Further, most of the Methylobacterium species contain property of pigmentation (pink) which is extremely slow and nodulates Crotolaria podocarpa. These are capable of growing on one-carbon compounds, such as formate, formaldehyde, methanol and methylamine. Significance of these bacteria as plant-growth promotion by the possible mechanisms include production of phytohormones, such as indole-3-acetic acid (IAA), cytokinins, nitrogen metabolism, nitrogen (N2)-fixing, contains 1-aminocyclopropane-1-carboxylate (ACC) deaminase, secretes EPS, accumulates PHB and survives in abiotic stress. These beneficial soil bacteria can confer immunity against a wide range of foliar diseases by activating plant defenses, thereby reducing plant susceptibility to pathogen attack (van Loon et al. 1998). For many years, it was considered that beneficial microorganisms could increase plant yield when inoculated in crops; however, it is increasingly appreciated that classic and novel microbial signals may also directly participate in plant morphogenesis. Plant depends on bacteria for the removal of metabolic waste products generated during its growth (Holland 1997). Methanol, a waste product of plants, is a fitting example of this kind of relationship, degraded by PPFMs into simpler compounds, such as ammonium, which eventually return to the plant. Recently, different species of PPFM are reported to be able to benefit plant development using a wide range of mechanisms, including synthesizing compounds to promote plant growth and increasing the uptake of nutrients and acting as biocontrol agents by suppressing plant pathogens in the rhizosphere. Several species of Methylobacter namely Methylobacterium oryzae , M. funariae, Methylobacterium organophilum, Methylobacterium nodulans , Methylobacterium populi , Methylobacterium extorquens etc. are reported for plant-growth promotion.
2 Alliance of Methylobacterium with Plants
Methylobacterium are root-nodulating symbionts (Jaftha et al. 2002), endophytic (Van Aken et al. 2004) and epiphytic (Omer et al. 2004) on plant surfaces.
It has been considered that plant-Methylobacterium association is primeval and permanent (Fedorov et al. 2011), and that plant-associated Methylobacterium is a co-evolved phytosymbiont (Kutschera 2007) because of symbiotic interaction. In fact, more than 80 % of viable bacteria isolated from leaf surfaces are members of the genus Methylobacterium (Tani et al. 2012).
2.1 Fate of C1 Compounds via Serine Cycle
Numerous studies have established that C1 metabolism plays the key role in the root colonization of Methylobacterium (Sy et al. 2005). Enzymes involved in methylotrophy of this microorganism have been identified and characterized in a metaproteomic study (Vorholt 2002). In this pathway, methanol is oxidized by methanol dehydrogenase (MDH) in the periplasmic space of the cell to produce formaldehyde (HCHO), which is then relocated into cytoplasm where part of the formaldehyde is oxidized to carbon dioxide (CO2) for energy generation, and rest is assimilated via the serine cycle (Fig. 1). The metabolism is characterized into three parts: Part 1 indicates bacterium which oxidizes methanol to formaldehyde is condensed with a tetrahydromethanopterin and further oxidized to formate. Formate reacts with tetrahydropterin and formyltetrahydrofolate is further converted to methylenetetrahydrofolate.
C1 metabolism of the methylotroph Methylobacterium extorquens AM1. Enzymes: 1 serine hydroxymethyl transferase; 2 serine-glyoxylate aminotransferase; 3 hydroxypyruvate reductase; 4 glycerate kinase; 5 enolase; 6 phosphoenolpyruvate carboxylase; 7 malate dehydrogenase; 8 malate-CoA ligase (malate thiokinase); 9, l-malyl-CoA/b-methylmalyl-CoA lyase; 10 β-ketothiolase; 11 acetoacetyl-CoA reductase; 12 crotonase; 13 crotonyl-CoA carboxylase reductase; 14 ethylmalonyl-CoA/methylmalonyl-CoA epimerase; 15 ethylmalonyl-CoA mutase; 16 methylsuccinyl-CoA dehydrogenase; 17 mesaconyl-CoA hydratase; 18 propionyl-CoA carboxylase; 19 methylmalonyl-CoA mutase; 20 methanol dehydrogenase. PHB polyhydroxybutyrate, Q quinone (Figure adapted from Smejkalova et al. 2010)
On the other hand, Part 2 involved metabolism during the serine cycle is used for the assimilation of formaldehyde plus bicarbonate and Part 3 contain Acetyl-CoA assimilation and conversion to glyoxylate proceeds via the ethylmalonyl-CoA pathway. The ethylmalonyl-CoA pathway, in connection with the serine cycle, represents an elegant solution of methanol assimilation, where methanol and carbon dioxide contribute nearly equal to cell carbon:
The assimilation of acetate through the ethylmalonyl-CoA pathway can be expressed by the following equation:
Methylobacterium nodulans is the causal organism of nodulation of the Crotalaria podocarpa (Jourand et al. 2004), possesses an mxa gene cluster for coding MDH.
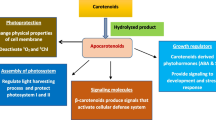
3 Mechanism for Plant-Growth Promotion
Methylobacterium spp. are ubiquitous in nature and colonize probably all land plants influencing the growth promotion by fixing atmospheric nitrogen, solubilizing insoluble phosphates, secreting hormones and producing antimicrobial compounds to combat phytopathogens. Induced systemic resistance (ISR), antibiosis, competition for nutrients, parasitism and production of metabolites (hydrogen cyanide, siderophores and enzymes) suppressive to deleterious rhizobacteria are some of the biocontrol mechanism that induce plant growth (Jha et al. 2010).
3.1 Phosphate Solubilization
Long back, Goldstein (2003) proposed direct oxidation of glucose to gluconic acid (GA) as a major mechanism for mineral phosphate solubilization (MPS) in Gram-negative bacteria. As a result of acidification of the surrounding medium, soluble orthophosphate ions (H2PO −14 and HPO −24 ) can be readily released. Nowadays, it is widely accepted that a large number of microbes produce a range of low-molecular weight organic acids, such as acetate, lactate, oxalate, tartarate, succinate, citrate, gluconate, ketogluconate, glycolate, etc., which are considered to solubilize insoluble mineral phosphates. It could be assumed that any gene involved in organic acid synthesis might have an effect on this character (Ahemad and Khan 2010). In Methylobacterium, by screening genomic libraries of mineral phosphate solubilization (MPS) bacteria for gluconic acid production traps Pyrroloquinoline quinine (PQQ) (Pyrroloquinoline quinine) biosynthesis genes which act as a prosthetic group of bacterial quinoprotein dehydrogenase. PQQ belongs to the family of quinone cofactors that has been recognized as the third class of redox cofactors following pyridine nucleotide and flavin-dependent cofactors (Liu et al. 1992). It is a prosthetic group required by several bacterial dehydrogenases, including methanol dehydrogenase (MDH) of Gram-negative methylotrophs and some glucose dehydrogenases. PQQ is derived from two amino acids, tyrosine and glutamic acid (Houck et al. 1991) but the pathway for its biosynthesis is unknown. Sequence analysis of this gene (Liu et al.1992) suggested its probable involvement in the synthesis of the enzyme PQQ synthase, which directs the synthesis of PQQ, a cofactor necessary for the formation of the holoenzyme glucose dehydrogenase (GDH)-PQQ. This enzyme catalyzes the formation of gluconic acid from glucose by the direct oxidation pathway (Goldstein 2003).
In M. extorquens AM1, the genes for PQQ synthesis are found in two clusters, pqqAB (C/D) E and pqqFG. These gene designations standardize the nomenclature with that of Klebsiella pneumoniae. These genes in Methylobacterium strains were formerly called pqqDGCBA. In M. extorquens AM1, pqqC and pqqD are not separate genes. Instead, they are fused into a single gene, pqqCD.
3.2 Plant Hormone Production
Plant-growth promotion by Methylobacterium include synthesis of the major plant hormones IAA and cytokinin, besides breakdown of plant produced ethylene by production of ACC deaminase as stated by (Saraf et al. 2010). In the Methylobacterium, genes that encode enzymes related to auxin biosynthesis, such as amine oxidase, aldehyde dehydrogenase, cyanide hydratase, N-acyltransferase, nitrile hydratase, amidase have been reported (Kwak et al. 2014). Methylobacterium is able to produce IAA (Ivanova et al. 2001), suggesting that its inoculation can increase IAA accumulation in plants that leads to induce plant growth and development (Madhaiyan et al. 2006a).
Cytokinins can be produced by bacteria by at least two pathways. De novo synthesis involves the direct isopentenylation of AMP catalyzed by dimethyl alkyltransferase (DMAT), which was first characterized in Agrobacterium tumefaciens (Golberg et al. 1984) while the second pathway of bacterial cytokinin production involves turnover of modified tRNA which also operate in higher plants. The origin of cytokinins resulting from tRNA degradation involves isopentenylation of adenine by isopentenyl tRNA transferase, the product of the miaA gene. In Methylobacteria, this modified adenine is subsequently methylated or hydroxylated. It is hypothesized that upon turnover of tRNA the modified adenine residue is released as a free cytokinin. Methylobacteria prefer the second pathway for the production of cytokinins. Actually, tRNA is the source of low-level trans-zeatin (active and ubiquitous form of the naturally occurring cytokinins) production. Infact, M. extorquens produces the cytokinin trans-zeatin at low levels in pure culture and excrete it into the culture medium (Koenig et al. 2002). Earlier, Ivanova et al. (2000) reported the presence and expression of genes controlling the synthesis and secretion of cytokinins by the PPFM Methylobacterium mesophilicum VKM B-2143.
3.3 Nitrogen Fixation
The biological reduction of nitrogen to ammonia (NH3) can be performed only by some prokaryotes with the presence of the nitrogenase enzyme (Menna et al. 2006). M. nodulans was originally isolated from Crotalaria podocarpa (Sy et al. 2001) and it was the few nodulating Methylobacterium species reported so far (Kwak et al. 2014). M. nodulans ORS2060 was reported to contain the nifH gene (involved in nitrogen fixation) and to induce N2-fixing nodules on the 11 leguminous plants (Jourand et al. 2004). Kumar et al. (2009) reported that the ultimate aim of establishing endophytic interaction between diazotrophic bacteria and nonlegumes is to fix N2 which later transferred the fixed N2 to the plants. Azorhizobium caulinodans and Methylobacterium species were capable of N2-fixing in a free-living condition. It was anticipated that the intercellular colonization of rice might provide a niche for N2 fixation. They isolated Methylobacterium sp. NPFM-SB3 from Sesbania rostrata stem nodules possess nitrogenase activity and nodA genes.
3.4 1-Aminocyclopropane-1-Carboxylate (ACC) Deaminase Production
Rhizobacterial enzyme, ACC deaminase (EC 4.1.99.4), which regulates ethylene production by metabolizing ACC (an intermediate precursor of ethylene biosynthesis in higher plants) into α-ketobutyrate and NH3 (Shaharoona et al. 2007). The microbial enzyme ACC deaminase cleaves ACC irreversibly, this being the immediate precursor of ethylene in plants (Saraf et al. 2010). This enzyme facilitates plant growth as a consequence of the fact that it sequesters and cleaves plant produced ACC, thereby lowering the level of ethylene in the plant. In turn, decreased ethylene levels allow the plant to be more resistant to a wide variety of environmental stresses, all of which induce the plant to increase its endogenous level of ethylene; stress ethylene exacerbates the effects of various environmental stresses. ACC deaminase activity is quantified by monitoring the production of either NH3 or α-ketobutyrate, the products of ACC hydrolysis. However, at present, monitoring the amount of α-ketobutyrate is more widely used by researchers. The presence of ACC deaminase was also verified by Fourier Transform Infrared (FTIR) spectra. FTIR spectra clearly shows the peak at 1683 cm−1 which exhibits the presence of ketonic group (–C=O). Whereas, 3452 cm−1 peak shows the presence of amino group (–NH2) (Jha et al. 2012). Methylobacterium also carry the acdS gene that encodes ACC deaminase enzyme converts ACC into NH3 and α-ketobutyrate. An analysis of the genomes of Methylobacterium species, such as M. oryzae, M. nodulans and M. radiotolerans, contain this ACC deaminase gene (Kwak et al. 2014) and that M. nodulans and M. radiotolerans are able to use ACC as a nitrogen source by the actions of ACC deaminase, reducing ethylene levels (Fedorov et al. 2013) and consequently the stress ethylene response in the host plant. More recently, Joe et al. (2014) reported that the ACC deaminase-positive M. oryzae CBMB20 with Azospirillum brasilense CW903 strain reduced ethylene levels in plants.
3.5 Exoploysaccharides (EPS) Production
EPS play vital roles in a variety of processes among bacteria, such as formation of biofilm (Bhaskar and Bhosle 2005), protection of bacterial cell from desiccation (Pal et al. 1999), maintaining primary cellular functions and antibacterial activity against predators, gelling ability, pollutant degradation kinetics (Fusconi and Godinho 2002), bioremediation activity and plasma substituting capacity (Allison 1998). Breuer and Babel (1999) reported the production of EPS in M. rhodesiunum under ammonium limitation conditions. The high amount of PHB accumulation also observed in Methylobacterium strains (Alvarez et al. 1996) in some psychrophilic and psychrotrophic crude oil-utilizing marine bacteria, accumulate lipid storage compounds in the cytoplasm under nitrogen limiting conditions when the C:N ratio becomes high. Woo et al. (2012) had compared the growth pattern, floc yield, EPS production and PHB accumulation, resistance to osmotic and acid stress in Methylobacterium strains CBMB20, CBMB27, CBMB35 and CBMB110.
4 Biocontrol Potentials
In recent decades, interaction studies have reflected that endophytic microorganisms may enhance plant protection against pathogen attacks. Biocontrol of pathogens can be achieved by several mechanisms viz: ISR, siderophore production, lytic enzyme production, etc. Ardanov et al. (2012) studied the ability of Methylobacterium sp. IMBG290 to induce resistance in potato (Solanum tuberosum L.) cultivars against Pectobacterium atrosepticum, Phytophthora infestans and Pseudomonas syringae pv. Tomato DC3000, as well as M. extorquens DSM13060 in pine (Pinus sylvestris L.) against Gremmeniella abietina. In earlier studies, Madhaiyan et al. (2006b) observed that seed treatment with Methylobacterium sp. induced significant protection against Aspergillus niger and Sclerotium rolfsii in groundnut. Further, the biocontrol potential of Methylobacterium spp. against several fungal pathogens, such as Fusarium udum, Fusarium oxysporum, Pythium aphanidermatum and Sclerotium rolfsii was evaluated in vitro by Poorniammal et al. (2009).
4.1 Induced Systemic Resistance (ISR)
The state of enhanced defensive capacity of plant’s innate immunity elicited by specific mechanism through Methylobacterium against biotic challenges is defined as ISR. Madhaiyan et al. (2004) reported that the treatment with Methylobacterium sp. strain PPFM-Os-07 increased activities of various defense-related enzymes like chitinase, Phenylalanine ammonia lyase, β-1,3-glucanase, peroxidase and PR-proteins, which accumulated in paddy with the onset of ISR. This enhanced state of resistance is effective against a broad range of pathogens and parasites (van Loon 2000). The two most clearly defined forms of induced resistance are: (1) SAR and (2) ISR, which can be differentiated on the basis of the nature of the elicitor. SAR is induced either upon infection by an avirulent pathogen or upon restricted infection by a virulent pathogen and depends on the synthesis of salicylic acid (SA) by the host. It is effective against pathogens that are restricted by SA-dependent basal resistance responses. On the other hand, ISR is triggered by selected strains of nonpathogenic rhizobacteria and does not require SA but does depend on the responsiveness of the plant to jasmonic acid (JA) and ethylene. Due to this reason, tolerance to abiotic stresses occurred in plants (Mantelin and Touraine 2004).
4.2 Siderophores Production
The genus Methylobacterium, as a member of PPFM, has ubiquitous occurrence in the environment and plays an important role in iron acquisition. Lacava et al. (2008) concluded that Methylobacterium spp. have no ability of producing catechol-type siderophores, but are capable to produce hydroxamate-type siderophores. In their study, in vitro growth of Xylella fastidiosa subsp. is stimulated by the presence of a supernatant siderophore of endophytic M. mesophilicum. Silva Stenico et al. (2005) also reported that a strain of M. extorquens isolated from Citrus sinensis was able to produce hydroxamate type of siderophore but negative for catechol type. Recently, Vaidehi and Sekar (2012) reported that Methylobacterium phyllosphaerae MB-5 and CBMB-27 contained hydroxamate type of siderophore during iron limitation.
4.3 Quorum Sensing
Quorum sensing (QS) systems use N-acyl-homoserine lactones (AHLs) as signaling molecules, commonly found in Gram-negative bacteria that live in association with plants (White and Winans 2007). QS system allows bacteria to function as multicellular organisms, because the extracellular concentration of autoinducer increases with bacteria population growth, after attaining a determinate number. This molecule disseminate back into the bacteria and regulate the transcription of different genes that may be related with the secretion of virulence factors, biofilm formation, sporulation, exchange of DNA and others (Zhu and Sun 2008). Although, several studies demonstrate the importance of the association between Methylobacterium plants (Dourado et al. 2012) and that members of the Methylobacterium genus produces AHL (Pomini et al. 2009). Recently, Dourado et al. (2013) reported the role of plant exudates and AHL on the expression of bacterial genes that are involved in bacterium plant-interaction. It was observed that AHL induces all analyzed genes mxaF, acdS, crtI and sss evade plant-microbe interactions. The gene sodium solute symporter (sss) is a transport gene responsible for the symport transport of solute with the sodium (Scier 1998). Genes crtI and acdS genes are associated with the stress response (Sandmann 2009) and plant metabolism (Hardoim et al. 2008). Phytoene dehydrogenase gene (crtI) codifies an enzyme that catalyzes the denaturation reaction resulting in the lycopene synthesis that protects the cell against oxidative damages and the acdS gene responsible for the degradation of ACC by ACC deaminase enzyme. Enhanced production of AHL corresponding to biofilms formation cannot be ruled out.
5 Applications
Methylobacterium spp. exhibited a vast range of biotechnological applications in the field of agriculture and industry. Recently, it was established as a potential bioinoculant for the sustainable agriculture.
5.1 Poly-β-Hydroxybutyrate (PHB) Accumulation
Poly-β-hydroxybutyrate (PHB) is an intracellular storage compound, which provides a reserve of carbon and energy in several microorganisms. It has been argued that methanol would appear as an alternative substrate for PHB production because of several advantages including low price and its complete water miscibility. Lopez-Cortes et al. (2008) suggested the presence of bright cytoplasmic inclusions as preliminary step for qualitative PHB determination. Methylobacterium sp. shows well-defined brightly refractile cytoplasmatic inclusions under phase contrast microscopy suggesting PHB accumulation. Zahra et al. (2009) reported the production of PHB using methanol by Methylobacterium extorquens DSMZ 1340.
5.2 Nitogen-Fixing Biofertilizers
Methylobacterium play a vital role by mediating nutrient transformation from the soil to plants. Rekadwad (2014) reported a thermophilic M. organophilum (N2)-fixing species isolated from hotspring originated mud is able to fix dinitrogen at elevated temperature. During last decade, a first report appeared in the year 2001 by a group of scientists about the symbiotic association of M. nodulans to that of Crotolaria podocarpa. This root-nodulating bacterium fixes nitrogen in symbiosis with legumes (Sy et al. 2001)
5.3 Seed Germination and Plant-Growth Promotion
The seed resident Methylobacterium is a contributing factor to vigor and seed viability. The cytokinins produced by these PPFMs are responsible for their stimulatory effect on germination (Freyermuth et al. 1996). Role of some other compounds to contribute for the enhancement of germination and growth of plants cannot be ruled out. Anitha (2010) reported that increase in maize seeds germination increased by 86 % to those seeds treated with 0.5 mg/l of benzyl adenine and 0.5 mg/l of zeatin (Holland and Polacco 1992). Wei et al. (2014) reported that germination energy and the germination rate decreased with increasing phenanthrene concentrations in wheat. Since date back in 1995, Holland and Polacco granted a patent. They coated seeds with at least one PPFM to improve seed germination, affirming that PPFM can be used to produce cytokinin. Verginer et al. (2010) observed that M. extorquens DSM 21961 increase the production of two furanoid compounds, 2,5-dimethyl-4-hydroxy-2H-furanone (DMHF) and 2,5-dimethyl-4-methoxy-2H-furanone in vitro, which are responsible for strawberry flavor.
5.4 Induced Pathogenesis
Methylobacteria induces systemic resistance against diseases due to siderophores and enzymes like phenylalanine ammonia lyase, peroxidase, chitinase and β-1,3-glucanase and phenolic compounds. Methylobacterium induce defense programs, such as SAR and ISR, thus reducing phytotoxic microbial communities. Further, bacteria also elicit induced systemic tolerance (IST) to abiotic stress. Proposed signal molecules for PGP by Methylobacterium include synthesis of IAA and cytokinin besides breakdown of plant-produced ethylene by bacterial production of ACC deaminase. Although low-molecular weight plant volatiles, such as terpenes, jasmonates and green leaf components have been identified as potential signal molecules for plants and organisms of other trophic levels (Farag and Pare 2002). Additional signals from microbes have been found to play a role in plant morphogenetic processes, including the N-acyl-l-homoserine lactones (AHLs) and volatile organic compounds (VOCs).
5.5 Plant-Growth Promotion
Methylobacteria a significant organism used for plant-growth promotion by producing promising mechanisms which include production of phytohormones, establish efficient (N2)-fixing symbioses by nodulating legume roots, ACC deaminase-production, exopolysaccharides (EPS) Production, PHB (Poly-β-hydroxybutyrate) accumulation and abiotic stress endurance. In addition, certain isolated Methylobacterium strains which produce vitamin B12 suggested stimulating plant development. Methylotrophic bacteria are being associated with plant nitrogen metabolism through bacterial urease production (Holland and Polacco 1994). However, the overall nature of their relationship with plants is as yet poorly understood, and the biological significance of these bacterial species is still under infancy and yet to be fully explored (Abanda-Nkpwatt et al. 2006).
5.6 Bioremediation
Methylobacterium sp. contributes to the bioremediation process via multiple modes of action, because these microorganisms can degrade and mineralize organic xenobiotic compounds allowing them to serve directly as contaminant degraders. The synergistic action of both Methylobacterium and the plants lead to increased availability of hydrophobic compounds, affecting their degradation. Ventorino et al. (2014) reported the biodegradation of polycyclic aromatic hydrocarbon (PAH) by M. populi VP2, a plant growth promoters. Methylobacterium is capable of the metabolism of monochlorinated, dichlorinated and aliphatic substrate. Jing et al. (2008) reported that Methylobacterium sp. HJ1 is able to degrade the herbicide 2,2-dichloropropionic acid by removal of the halogen and subsequent metabolism of the product for energy. d,l-2-chloropropionate also supported good growth of the organism, but 3-chloropropionate, monochloroacetate and dichloroacetate were not utilized. Cell-free extracts of the 2,2-dichloropropionate-grown bacteria converted 2,2-dichloropropionate into pyruvate with the release of two chloride ions for each molecule of pyruvate formed.
5.7 A Model Gene Expression System for Recombinant Protein
Research suggests that the Methylobacterium is proved as a model organism or an interesting candidate for overexpression of recombinant proteins. Marx and Lidstrom (2001) developed a series of new expression vectors for M. extorquens AM1 enabling efficient expression of reporter genes. One of the expression vectors, pCM110, is a 5.8 kb IncP-derived plasmid possessing the strong M. extorquens native promoter of methanol dehydrogenase (MDH) (PmxaF). When compared to a similar vector containing only the lacZ promoter (PlaC), PmxaF led to a 50-fold increase in the expression of the reporter gene xylE (Marx and Lidstrom 2001). Belanger et al. (2004) observed the usefulness of two distinct vectors (pRK310 and pCM110) and promoters (PlaC and PmxaF) for heterologous expression in a high cell density for fed-batch fermentation process using M. extorquens ATCC 55366.
6 Concluding Remarks
Microbes being an integral component of any soil ecosystem provide life to the soil. Methylotrophs are a polyphyletic group of microorganisms capable of utilizing C1 compounds as electron donor and of the most abundant bacteria, which is able to grow on methanol as well as on multi-carbon compounds as sole carbon and energy source. Continued research with colonization and biofilm formation by these bacterial genera also holds potential for developing biofertilizer and biocontrol agents that may be self-perpetuating within the colonizing host plants. Focusing research in these areas may also be aimed to establish Methylobacterium sp. as promising plant-growth promoter and a model bioremediator.
References
Abanda-Nkpwatt D, Musch M, Tschiersch J, Boettner M, Schwab W (2006) Molecular interaction between Methylobacterium extorquens and seedlings: growth promotion, methanol consumption, and localization of the methanol emission site. J Exp Bot 57(15):4025–4032
Ahemad M, Khan MS (2010) Plant growth promoting activities of phosphate-solubilizing Enterobacter asburiae as influenced by fungicide. Eur Asian J Biosci 4:88–95
Allison AG (1998) Exopolysaccharide production in bacterial biofilm. Biofilm J 3(2):1–19
Alvarez MI, Sueldo RJ, Barassi CA (1996) Effect of Azospirillum inoculation on coleoptile growth in wheat seedlings under water stress. Cereal Res Commun 24:101–107
Anitha KG (2010) Enhancing seed germination of Mono and Dicotyledons through IAA production of PPFM. Trends Soil Sci Plant Nutr J 1(1):14–18
Ardanov P, Sessitsch A, Haggman H, Kozyrovska N, Pirttila AM (2012) Methylobacterium-induced endophyte community changes correspond with protection of plants against pathogen attack. PLoS ONE 7:10 Article ID e46802
Belanger L, Figueira MM, Bourque D, Morel L, Beland M, Larame L, Groleau D, Miguez DB (2004) Production of heterologous protein by Methylobacterium extorquens in high cell density fermentation. FEMS Microbiol Lett 231:197–204
Bhaskar PV, Bhosle NB (2005) Microbial extracellular polymeric substances in marine biogeochemical processes. Curr Sci 88(1):45–53
Breuer U, Babel W (1999) Methylobacterium rhodesianum produces poly-3-hydroxybutyrate and after mutagenesis in addition exopolysaccharides. Acta Biotechnol 19:779–786
Corpe WA, Rheem S (1989) Ecology of the methylotrophic bacteria on living leaf surfaces. FEMS Microbiol Ecol 62:243–250
Dourado MN, Andreote FD, Dini-Andreote F, Conti R, Araújo JM, Araújo WL (2012) Analysis of 16S rRNA and mxaF genes revealing insights into Methylobacterium niche specific plant association. Genet Mol Biol 35:142–148
Dourado MN, Bogas AC, Pomini AM, Andreote FD, Quecine MC, Marsaioli AJ, Araújo WL (2013) Methylobacterium-plant interaction genes regulated by plant exudate and quorum sensing molecules. Brazilian J Microbiol 44(4):1331–1339
Farag MA, Pare PW (2002) C6-green leaf volatiles trigger local and systemic VOC emissions in tomato. Phytochemistry 61:545–554
Fedorov DN, Doronina NV, Trotsenko YA (2011) Phytosymbiosis of aerobic methylobacteria: new facts and views. Microbiology 80(4):443–454
Fedorov DN, Ekimova GA, Doronina NV, Trotsenko YA (2013) 1-aminocyclopropane-1-carboxylate (ACC) deaminases from Methylobacterium radiotolerans and Methylobacterium nodulans with higher specificity for ACC. FEMS Microbiol Lett 343(1):70–76
Freyermuth SK, Long RL, Mathur S, Holland MA, Holstford TP, Stebbins NE, Morris RO, Polacco JC (1996) Metabolic aspects of plant interaction with commensal methylotrophs. In: Lindstorm M, Tabita R (eds) Microbial growth on C1 compounds. Kluwer Academic Publishers, New York, pp 21–134
Fusconi R, Godinho MJL (2002) Screening for exopolysaccharide producing bacteria from sub-tropical polluted groundwater. Brazilian J Biol 62(2):363–369
Golberg SB, Flick JS, Rogers SG (1984) Nucleotide sequence of the tmrlocus of Agrobacterium tumefaciens pTiT37 T-DNA. Nucl Acids Res 12:4665–4677
Goldstein AH, Lester T, Brown J (2003) Research on the metabolic engineering of the direct oxidation pathway for extraction of phosphate from ore has generated preliminary evidence for PQQ biosynthesis in Escherichia coli as well as a possible role for the highly conserved region of quinoprotein dehydrogenases. Biochimica Biophysica Acta 1647:266–271
Hardoim PR, Overbeek LSV, Elsas JDV (2008) Properties of bacterial endophytes and their proposed role in plant growth. Trends Microbiol 16:463–471
Holland MA (1997) Occams razor applied to hormonology. Are cytokinins produced by plants? Plant Physiol 115:865–868
Holland MA, Polacco JC (1992) Urease null and hydrogenase null phenotypes of a phylloplane bacterium reveal altered nickel metabolism in two soybean mutants. Plant Physiol 98:942–948
Holland MA, Polacco JC (1994) PPFMs and other contaminants: is there more to plant physiology than just plant. Annu Rev Plant Physiol Plant Mol Biol 45:197–209
Houck DR, Hanners JL, Unkefer CJ (1991) Biosynthesis of pyrroloquinoline quinone. Biosynthetic assembly from glutamate and tyrosine. J Am Chem Soc 113:3162–3166
Ivanova EG, Doronina NV, Shepeliakovskaia AO, Laman AG, Brovko FA, TrotsenkoIu A (2000) Facultative and obligate aerobic methylobacteria synthesize cytokinins. Mikrobiologia 69(6):764–769
Ivanova EG, Doronina NV, Trotsenko YA (2001) Aerobic methylobacteria are capable of synthesizing auxins. Microbiology 70(4):392–397
Jaftha JB, Strijdom BW, Steyn PL (2002) Characterization of pigmented methylotrophic bacteria which nodulate Lotononis bainesii. Syst Appl Microbiol 25(3):440–449
Jha CK, Patel D, Rajendran N, Saraf M (2010) Combinatorial assessment on dominance and informative diversity of PGPR from rhizosphere of Jatropha curcas L. J Basic Microbiol 50:211–217
Jha CK, Annapurna K, Saraf M (2012) Isolation of rhizobacteria from jatropha curcas and characterization of produced ACC deaminase. J Basic Microbiol 52:285–295
Jing NH, Taha AM, Pakingking RV, Wahabs RAB, Huyop F (2008) Dehalogenase from Methylobacterium sp. HJ1 induced by the herbicide 2,2-dichloropropionate (Dalapon). Afr J Microbiol Res 2:32–36
Joe MM, Saravanan VS, Islam MR, Sa T (2014) Development of alginate-based aggregate inoculants of Methylobacterium sp. and Azospirillum brasilense tested under in vitro conditions to promote plant growth. J App Microbiol 116(2):408–423
Jourand P, Giraud E, Béna G, Sy A, Willems A, Gillis M, Dreyfus B, De Lajudie P (2004) Methylobacterium nodulans sp. nov. for a group of aerobic, facultatively methylotrophic, legume root-nodule-forming and nitrogen-fixing bacteria. Int J Syst Evol Microbiol 54:2269–2273
Koenig RL, Morris RO, Polacco JC (2002) tRNA is the source of low-level trans-Zeatin production in Methylobacterium spp. J Bacteriol 184(7):1832–1842
Kutschera U (2007) Plant-associated methylobacteria as co-evolved phytosymbionts. Plant Signal Behav 2:74–78
Kwak MJ, Jeong H, Madhaiyan M, Lee Y, Sa TM, Oh TK, Kim JF (2014) Genome information of Methylobacterium oryzae, a plant-probiotic methylotroph in the phyllosphere. PLoS ONE 9(9):106–110
Lacava PT, Silva-Stenico ME, Araújo WL, Simionato AC, Carrilho E, Tsai SM, Azevedo JL (2008) Detection of siderophores in endophytic bacteria Methylobacterium spp. associated with Xylella fastidiosa subsp. Pauca. Pesqagropec Bras Brasília 43(4):521–528
Liu TS, Lee LY, Tai CY, Hung CH, Chang YS, Wolfram JH, Rogers R, Goldstein AH (1992) Cloning of an Erwinia carotovora gene necessary for gluconic acid production and enhanced mineral phosphate solubilization in Escherichia coli HB101: Nucleotide sequence and probable involvement in biosynthesis of the coenzyme pyrroloquinoline quinine. J Bacteriol 174:5814–5819
Lopez-Cortes A, Lanz-landazuri A, Garcia-Maldonado JQ. (2008) Screening and isolation of PHB producing bacteria in a polluted marine microbial mat. Microb. Ecol. 56:112–120
Madhaiyan M, Poonguzhali S, Senthilkumar M, Sundaram S, Chung H, Yang J, Sundaram S, Sa T (2004) Growth promotion and induction of systemic resistance in rice cultivar Co-47 (Oryza sativa L.) by Methylobacterium spp. Bot Bull Acad Sin 45:315–324
Madhaiyan M, Poonguzhali S, Ryu JH, Sa TM (2006a) Regulation of ethylene levels in canola (Brassica campestris) by 1-aminocyclopropane-1-carboxylate deaminase-containing Methylobacterium fujisawaense. Planta 224(2):268–278
Madhaiyan M, Reddy BVS, Anandham R (2006b) Plant growth-promoting Methylobacterium induces defense responses in groundnut (Arachis hypogaea L.) compared with rot pathogens. Curr Microbiol 53(4):270–276
Mantelin S, Touraine B (2004) Plant growth-promoting bacteria and nitrate availability: impacts on root development and nitrate uptake. J Exp Bot 55:27–34
Marx CJ, Lidstrom ME (2001) Development of improved versatile broad-host-range vectors for use in methylotrophs and other Gram-negative bacteria. Microbiology 147:2065–2075
Menna P, Hungria M, Barcellos FG, Bangel EV, Hess PN, Martínez-Romero E (2006) Molecular phylogeny based on the 16S rRNA gene of elite rhizobial strains used in Brazilian commercial inoculants. Syst Appl Microbiol 29:315–332
Omer ZS, Tombolini R, Gerhardson B (2004) Plant colonization by pink-pigmented facultative methylotrophic bacteria (PPFMs). FEMS Microbiol Ecol 47(3):319–326
Pal S, Manna A, Paul AK (1999) Production of (poly-hydroxy butyric acid) and exopolysacharide by Azotobacte rbeijerinckii WDN—01. World J Microbiol Biotechnol 15:15–21
Pomini AM, Cruz PLR, Gai C, Araújo WL, Marsaioli AJ (2009) Long-chain Acyl-Homoserine lactones from Methylobacterium mesophilicum: synthesis and absolute configuration. J Nat Prod 72:2130–2134
Poorniammal R, Sundaram SP, Kumutha K (2009) In vitro biocontrol activity of Methylobacterium extorquens against fungal pathogens. Int J Plant Prot 2:59–62
Rekadwad BN (2014) Growth promotion of crop plants by Methylobacterium organophilum: efficient bioinoculant and bio-fertilizer isolated from mud. Res Biotechnol 5(5):01–06
Sandmann G (2009) Evolution of carotene desaturation: the complication of a simple pathway. Arch Biochem Biophys 483:169–174
Saraf M, Jha CK, Patel D (2010) The role of ACC deaminase producing PGPR in sustainable agriculture. In: Maheshwari DK (ed) Plant growth and health promoting bacteria. Springer, Berlin Heidelberg, pp 365–385
Scier MH (1998) Proposed independent evolution of different channel and carrier families. In: Poole RK (ed) Advances in microbial physiology. Academic Press, London, pp 81–136
Senthil Kumar M, Madhaiyan M, Sundaram SP, Kannaiyan S (2009) Intercellular colonization and growth promoting effects of Methylobacterium sp. with plant-growth regulators on rice (Oryza sativa L. Cv CO-43). Microbes Res. 164(1):92–104.
Shaharoona B, Arshad M, Khalid A (2007) Differential response of etiolated pea seedling to 1-aminocyclopropane-1-carboxylate and/or L-methionine utilizing rhizobacteria. J Microbiol 45(1):15–20
Silva Stenico ME, Pacheco FTH, Rodriques JLM, Carrilho E, Tsai SM (2005) Growth and siderophore production of Xylella fastidiosa under iron-limited conditions. Microbiol Res 160(1):429–436
Smejkalova H, Erb TJ, Fuchs G (2010) Methanol assimilation in Methylobacterium extorquens AM1: demonstration of all enzymes and their regulation. PLoS ONE 5(10):e13001
Sy A, Giraud E, Jourand P, Garcia N, Willems A, De Lajudie P, Prin Y, Neyra M, Gillis M, Boivin-Masson C, Dreyfus B (2001) Methylotrophic Methylobacterium bacteria nodulate and fix nitrogen in symbiosis with legumes. J Bacteriol 183:214–220
Sy A, Timmers ACJ, Knief C, Vorholt JA (2005) Methylotrophic metabolism is advantageous for Methylobacterium extorquens during colonization of Medicago truncatula under competitive conditions. App Environ Microbiol 71(11):7245–7252
Tani A, Sahin N, Matsuyama Y, Enomoto T, Nishimura N, Yokota A, Kimbara K (2012) High-throughput identification and screening of novel Methylobacterium species using whole-cell MALDI-TOF/MS analysis. PLoS ONE 7(7):1–13
Vaidehi K, Sekar C (2012) Amino acid conjugated hydroxamate type of siderophore production in Methylobacterium phyllosphaerae MB-5. CIBtech J microbiol 1(1):25–30
Van Aken B, Yoon JM, Schnoor JL (2004) Biodegradation of nitro-substituted explosives 2,4,6-trinitrotoluene, hexahydro-1,3,5-trinitro-1,3,5-triazine, an octahydro-1,3,5,7-tetranitro-1,3,5-tetrazocine by a phytosymbiotic Methylobacterium sp. associated with poplar tissues (Populus deltoides nigra DN34). App. Environ Microbiol 70(1):508–517
Van Loon LC (2000) Systemic induced resistance. In: Slusarenko AJ, Fraser RSS, Van Loon LC (eds) Mechanisms of resistance to plant diseases. Kluwer, Dordrechet, pp 521–574
Van Loon LC, Bakker PAHM, Pieterse CMJ (1998) Systemic resistance induced by rhizosphere 660 bacteria. Annu Rev Phytopathol 36:453–483
Ventorino V, Sannino F, Piccolo A, Cafaro V, Carotenuto R, Pepe O (2014) Methylobacterium populi VP2: plant growth-promoting bacterium isolated from a highly polluted environment for polycyclic aromatic hydrocarbon (PAH) biodegradation. Sci World J Article ID 931793, 11 pages
Verginer M, Siegmund B, Cardinale M et al (2010) Monitoring the plant epiphyte Methylobacterium extorquens DSM21961 by real-time PCR and its influence on the strawberry flavor. FEMS Microbiol Ecol 74:136–145
Vorholt JA (2002) Cofactor-dependent pathways of formaldehyde oxidation in methylotrophic bacteria. Arch Microbiol 178(4):239–249
Wei H, Song S, Tian H, Liu T (2014) Effects of phenanthrene on seed germination and some physiological activities of wheat seedling. Compets Rendus Biol 337(2):95–100
White EW, Winans SC (2007) Cell-cell communication in the plant pathogen Agrobacterium tumefaciens. Phil Trans R Soc B 362:1135–1148
Woo S, Subramanian P, Ramasamy K, Joe MM, Sa T (2012) EPS production, PHB accumulation and abiotic stress endurance of plant growth promoting Methylobacterium strains grown in a high carbon concentration. Korean J Soil SciFert 45(4):572–581
Zahra BMH, Ebrahim VF, Abbas SS, Ramin K (2009) Media selection for poly hydroxybutyrate production from methanol by Methylobacterium extorquens DSMZ 1340. Iran J Chem Eng 28(3):45–52
Zhu H, Sun SJ (2008) Inhibition of bacterial quorum sensing regulated behaviors by Tremella fuciformis extract. Curr Microbiol 57:418–422
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Jha, C.K., Maheshwari, D.K., Saraf, M. (2015). Emergence of Methylobacterium spp. as Potential Organism in Agroecosystems. In: Maheshwari, D. (eds) Bacterial Metabolites in Sustainable Agroecosystem. Sustainable Development and Biodiversity, vol 12. Springer, Cham. https://doi.org/10.1007/978-3-319-24654-3_3
Download citation
DOI: https://doi.org/10.1007/978-3-319-24654-3_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-24652-9
Online ISBN: 978-3-319-24654-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)