Abstract
The chapter encompasses the synthesis, chemical transformations, properties, and applications of fluorinated monocyclic 1,2,3-, 1,2,4-triazoles and tetrazoles as well as their fused analogs. The heterocycles directly bonded with a fluorine atom (N-F or C-F isomers) as well as trifluoromethyl, perfluoroalkyl, perfluoroaryl, SF5, NF2 groups and some other fluorinated fragments were considered.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Fluorinated polynitrogen heterocycles like 1,2,3-triazoles, 1,2,4-triazoles and tetrazoles differ considerably from the other related heterocyclic systems in the preparation methods and some characteristics. Heterocycles containing three or four endocyclic nitrogen atoms, including one, two, or three N-N bonds combined with a powerful electron-acceptor and energy-consuming substituent like а fluorine atom or perfluoroalkyl groups possess a relatively high enthalpy of formation and notably differ from the other azoles by their chemical properties. Some fluorinated tri- and tetrazoles are well known and find versatile applications [1]. These compounds are widely used in medicine, agriculture, and in material sciences. Thus, several commercially important drugs as well as a lot of bioactive compounds with different types of activity have in their composition fluorinated triazolyl or tetrazolyl groups. Among them there are well-known antidiabetic DPP-IV inhibitors, NK1 receptor antagonist, antifungal agents, herbicides, and some other compounds with useful properties [1–5]. On the other hand these compounds possess a number of unique properties providing a possibility to utilize them in quite different fields of technology. For instance, these compounds are efficient corrosion inhibitors, they can be used as components of energy-rich compounds, ionic liquids, semiconductors, etc. [1, 6]. The applications of fluorinated tri- and tetrazoles will be discussed in more detail in Sect. 6 of this chapter.
Fluorinated derivatives of azoles are known since over half century, the chemical methods of their synthesis and their certain chemical properties are described in many reviews and monographs [1, 6–15]. In general two different ways to incorporate fluorine or perfluoroalkyl groups into a heterocyclic system may be considered: The heterocyclization of fluorine-containing acyclic systems many various types of which are known, or direct introduction of fluorine or a perfluoroalkyl group into existing heterocyclic system [1, 7–15]. So far the problems of the synthesis of highly nitrogenated cyclic systems, especially of 1,2,3-triazoles and tetrazoles have been poorly understood. It should be noted that quite a big number of original publications has appeared in this field within the last decade.
Taking into account the essential difference in the properties and the preparation procedures we consider further in succession compounds having a fluorine atom directly bound to the heterocycle and another fluorine-containing substances, perfluoroalkyl and perfluoroaryl derivatives of 1,2,3-, 1,2,4-triazoles and tetrazoles. The research on the synthesis and the reactivity of tri- and tetrazoles containing as substituents at the endocyclic carbon atom CF3 or the other perfluoroalkyl groups have been developed since the 1960s. The last decade has been marked with a significant progress in this direction due to some of these compounds have important practical applications. Tri- and tetrazoles containing as substituents perfluoroaryl fragments, SF5 and NF2 groups, and also heterocycles involved into fluorobenzofused systems are considered separately.
2 Compounds Having Fluorine Bonded Directly to Endocyclic Atoms
2.1 N-F Derivatives
In the case of unsubstituted triazoles and tetrazoles the existence of two isomers is possible for each heterocyclic system. Such derivatives are very different in thermodynamic stability: 1Н- and 2Н-1,2,3-triazoles and tetrazoles, and also 1Н- and 4Н-1,2,4-triazoles. By now all parent N-F heterocycles 1–6 remain hypothetical. Among substituted derivatives all known N-F heterocycles are only 1Н-isomers.

In general the information on the synthesis and properties of the N-fluorinated derivatives of 1,2,3- and 1,2,4-triazoles is very scanty [6]. The data about N-fluorotetrazoles are totally absent today. We discuss below the results of the few publications considering the synthesis of N-fluoro-1,2,3- and 1,2,4-triazoles. This desultory information is difficult to classify since the most of these papers lack any experimental details on the synthesis of the compounds of this type and the proofs confirming the structure.
By now the synthesis of 1-fluoro-benzotriazole 7 is well established fact [16]. Gakh et al. carried out a direct replacement of the hydrogen attached to the nitrogen atom of the “pyrrole” type of benzotriazole by the fluorine. For this aim benzotriazole anion was fluorinated with cesium fluorooxysulfate (CEOX). The cesium fluorooxysulfate discovered by Appelman in 1979 is one of the soft and regioselective reagents for anions fluorination. It requires however a cautious handling for it is prone to spontaneous instant decomposition [8, 9].

An example of synthesis of N-fluoro-1,2,4-triazole was described in the patent of Strazdina and Grinstein [6, 17]. Here 1-fluoro-3,5-dibromo-1,2,4-triazole 8 was obtained by treating the heterocyclic substrate with the hypohalous acid or its derivatives at −40 to +50 °C in water or an organic solvent in the presence of bases. Langlet and Ostmark in a patent [18] have described the application of isomeric fluoro-triazolones 9, 10 as components of explosive compositions, but the data on the synthesis of these substances have been not reported.

Miroshnichenko et al. [19] published results on calorimetric investigation of N-F derivatives of various nitroazoles, in particular, of some N-fluoro derivatives of 1,2,3- and 1,2,4-triazoles 11–13 (the values of standard enthalpy of formation ∆H f o, kJ/mol are given in parentheses). Regretfully, the methods of the synthesis of N-F triazoles and the data confirming their structure and individuality were not reported in this article.

According to theoretical calculations the N-F bond in the molecules of N-fluoro-1,2,4-triazoles is rather strong, in contrast to N-Cl (Br, I) bonds [20]. Therefore, it remains possible to prepare a wide range of thermodynamically stable N-fluoro derivatives of 1,2,4-triazole which may be of a practical importance. Thus, using ab initio quantum-chemical methods, the structure of salts was investigated that were formed with N-F-1,2,4-triazolium cation and dinitramide anion which might be used for the preparation of exotic energy-rich ionic liquids [21].
2.2 C-F Derivatives
Taking into account that 1,2,3- and 1,2,4-triazole rings contain two carbon atoms, and the tetrazole cycle only a single one, the fluorine atom can be located in two different positions in the isomeric C-F triazoles with nonequivalent substituents 14–17 and in a single position in 5-fluorotetrazole 18. All types of these derivatives are known.

The direct incorporation of a fluorine atom in the ring essentially affects versatile physicochemical and chemical characteristics of the heterocyclic system, in particular, its tautomerism, dipole moment, and the acid-base properties. For instance, tautomeric equilibria involving NH-unsubstituted fluoro derivatives of 1,2,3-triazoles [22, 23], 1,2,4-triazoles [24], and tetrazoles [25–28] were explored in a series of theoretical studies. The acid-base properties of these heterocycles (in particular, the CH-acidity of some among them) were also studied [28–30]. These investigations showed that the fluorine atom attached to the heterocyclic system changed significantly the character of the electron density distribution and consequently the polarity of the heterocycle, strongly increased its acidity and decreased its basicity.
The synthetic procedures for the preparation of C-F derivatives of tri- and tetrazoles are more developed compared to the synthesis of the derivatives with the N-F bond. The preparation methods of C-F derivatives are known not only for 1,2,3- and 1,2,4-triazoles, but also for tetrazoles. The C-F bond is successfully formed by the replacement of hydrogen atom or various “leaving” groups under the action of the fluorination reagents. Along with the above methods diverse versions are applied of 1,3-dipolar cycloaddition of azides to dipolarophiles containing fluorine. Also very useful approach is oxidative cyclization of molecules with the linear structure containing a C-F bond.
The first report on the preparation of 1,2,4-triazoles fluorinated at the carbon atom of the ring appeared as early as 1973 [31]. In this study the corresponding 3-fluoro-1,2,4-triazoles 20 were obtained in good yields by treatment of 5-R-3-nitro-1,2,4-triazoles 19 with hydrogen fluoride. Nitro group substitution by fluorine under these conditions required a prolonged time (20–48 h) and heating at high temperature (100–150 °C).

The nucleophilic exchange of one bromine atom in 1-benzyl-3,5-dibromo-1,2,4-triazole 21 with fluorine under the action of CsF followed by the photoinitiated elimination of the benzyl protective group led to the formation of 3-bromo-5-fluoro-1Н-1,2,4-triazole 22 in 59 % overall yield. The alkylation of this substrate gave 1-alkyl-3-fluoro-1,2,4-triazoles for the first time. Both bromine atoms were substituted to provide the corresponding difluoro derivative 24 under the same reaction conditions, but using 1-(3,5-dimethoxybenzyl)-3,5-dibromo-1,2,4-triazole 23 as substrate for fluorination [32].

Cesium fluoride was also used in the synthesis of 2,3-di(p-tolyl)-5-fluorotetrazolium bromide 25 from the corresponding 5-triphenylphosphoniotetrazolium salt [33].

Another example of the direct introduction of the fluorine atom into a heterocycle was demonstrated in [34]. Here 3-fluoro-1,2,4-triazole 26 was obtained by the photochemical decomposition of hetaryldiazonium tetrafluoroborate in solution oversaturated with NaBF4.

A widely used method of the preparation of versatile 1,2,3-triazoles and tetrazoles consists in the reaction of azides with fluorine-containing compounds with multiple bonds. For example, this approach afforded N-substituted 4(5)-fluoro-1,2,3-triazoles 27, 28 by the 1,3-dipolar cycloaddition of perfluoropropadiene to phenyl azide [35]. In the course of the reaction a mixture of regioisomeric 1,2,3-triazoles was formed; 1-phenyl-4-fluoro-5-trifluoromethyl-1,2,3-triazole 27 was considerably prevailed. On the contrary, the reaction of phenyl azide with the perfluoropropyne resulted in predominantly the isomer that was minor in the previous scheme.


Reck et al. demonstrated that phenylsulfinic acid was eliminated in course of 1,3-dipolar cycloaddition involving 1-fluoro-1-(phenylsulfonyl)ethylene and (5R)-3-[4-(1,1-dioxo-3,6-dihydro-2H-thiopyran-4-yl)-3-fluorophenyl]-5-(azidomethyl)oxazolidin-2-one. As a result a mixture of regioisomeric 4- and 5-fluoro-1,2,3-triazoles 29, 30 in 7:1 ratio was formed in 28 % overall yield. Compounds 29, 30 may be regarded as effective antimicrobial agents [36].

Apparently the simplest 5-fluorotetrazole 18 and its derivatives can be synthesized by the reaction of 2 + 3 dipolar cycloaddition of azides to cyanogen fluoride or by some other method. Yet we failed to find the mention of such synthesis in available publications. The possibility of this reaction and its mechanism were assessed theoretically [37]. Publications are known where compound 18 is used as a component of the energy-rich compositions [38].
3 Trifluoromethyl and Perfluoroalkyl Derivatives
3.1 1,2,3-Triazoles
The most common synthetic procedure for the preparation of perfluoroalkylated 1,2,3-triazole derivatives is 1,3-dipolar cycloaddition of azides to diverse unsaturated dipolarophiles containing perfluoroalkyl substituents. Thus, in 1966 Carpenter et al. carried out the cycloaddition of benzyl azide to perfluoroalkyl substituted acetylene derivatives leading to the formation of trifluoromethyl-1,2,3-triazolines and 1,2,3-triazoles [39]. In this paper a synthesis of l-benzyl-4,5-bistrifluoromethyl-l,2,3-triazole 31 from hexafluoro-2-butyne was described.

The regioselectivity of 1,3-cycloaddition of benzyl azide to unsymmetrical acetylenes containing CF3 substituent at one carbon atom of the fragment С ≡ С and CO2Et group at the other carbon was analyzed [40]. The formation of regioisomeric mixtures of 1,2,3-triazoles 32, 33 was observed. This study was further developed recently. Zhang et al. published the results of a research on the 1,3-dipolar cycloaddition of benzyl azide and some other aryl azides to methyl perfluoroalkylalkynoate. It was shown that the ratio of regioisomers is governed by two factors: the orbital control (the role of the frontier orbitals was established) and also spatial interaction of the perfluoroalkyl and aryl (or benzyl) groups [41].

The influence of the steric effects on the regioselectivity of the cycloaddition was studied in detail by an example of the 1,3-dipolar cycloaddition of benzyl azide to trifluoromethylacetylene containing a TBDMS (tert-butyldimethylsilyl) protecting group. The regioselective formation in this case of the single isomer of 1,2,3-triazole 34 was explained by the presence in the structure of dipolarophile of a bulky substituent (TBDMS) [42]. Such an approach made it possible to perform a regioselective synthesis of nucleosides analogs containing in their molecular structure a trifluoromethyl-1,2,3-triazolyl fragment 35 [42].

The reaction of tert-butyl azidoacetate with diisopropyl-3,3,3-trifluoroprop-1-ynylphosphonate gave regioisomeric diisopropyl(1-tert-butoxycarbonylmethyl-4-trifluoromethyl-1H-l,2,3-triazol-5-yl)phosphonate 36 and disopropyl(l-tert-butoxycarbonylmethyl-5-fluoromethyl-lH-l,2,3-triazol-4-yl)phosphonate 37 in 75:25 ratio (90 % overall yield) [43].

Pentamethylcyclopentadienylrhodium (or iridium) azido complexes 38 react with ditrifluoromethylacetylene to give the corresponding 1,2,3-triazole rhodium complex 39. Subsequent treatment with NH4Cl opened the route to free NH-triazole 40 [44].

Lermontov et al. reported some successful reactions of two α,α-difluoroazides, namely, 2-hydroperfluoropropyl azide and the methyl ester of 3-azido-perfluoropropanoic acid, with various acetylene compounds, and described some properties of the resulting products [45]. Thus, phenylacetylene reacts with 2-hydroperfluoropropyl azide to give the corresponding 4-phenyl-1-(2H-perfluoropropyl)-1,2,3-triazole 41 and 5-phenyl-1-(2H-perfluoropropyl)-1,2,3-triazole 42 in 1/2 ratio. Disubstituted acetylenes also react with these azides to give the corresponding 1H-1,2,3-triazoles [45]. Thus in the case of triazole 43 the yield is close to the quantitative.

In the paper [46] of Wu, Chen et al. a method for the preparation of the fluoroalkylated 1,4-disubstituted-1,2,3-triazoles by the 1,3-dipolar cycloaddition of fluoroalkylated azides to terminal alkynes in the presence of Cu(I) salt as catalyst at room temperature was described. All these reactions were highly regioselective giving 1,4-disubstituted 44, 45, no 1,5-disubstituted products. The structure of key compounds was confirmed both by the NOSEY spectra and an X-ray diffraction study.

A series of fluoroalkylated amphiphilic 1,2,3-triazoles 46, 47, 48 was synthesized by efficient 1,3-dipolar cycloaddition of 2-perfluoroalkylethyl azides and acetylenic acids or esters [47].

Recently Yi et al. also utilized 2-perfluoroalkylethyl azides as 1,3-dipoles in the 1,3-dipolar cycloaddition to phenyl- or butylacetylenes [48]. As a result in the presence of copper(I) salt the corresponding 1-fluoroalkyl-4-substituted 1,2,3-triazoles were obtained in about 60 % yield. Note that in this case only anti- isomers were obtained: 1-fluoroalkyl-4-aryl- or 1-fluoroalkyl-4-butyl-1,2,3-triazoles. The authors do not explain the high selectivity of the process, although it may be attributed to the steric effect of the bulky substituents (aryl, butyl). Yi et al. note the relatively high efficiency of the fluoroalkyl 1,4-disubstituted-1,2,3-triazoles as catalysts of aldol condensation which may be easily recovered and reused [48]. Read et al. [49] virtually simultaneously with [48] published the results of their proper exploration of the copper salts catalyzed 1,3-dipolar cycloaddition of fluorinated alkyl azides to acetylenes. This research extended the methodology formerly suggested by Wu et al. [46] who were among the first to apply the catalytic system based on Cu(I). It was shown that practically for the generation of the Huisgen-Meldal catalyst the system CuSO4 5H2O – sodium ascorbate was preferable. Just under these reaction conditions a wide range of 1,2,3-triazoles 49 was obtained having the perfluoroalkyl groups at the atoms in the positions 1 and 4 [49]. It was also shown that the higher rate of the process and the higher yield of cycloaddition products were achieved both in the presence of copper salts and at the microwave acceleration.

1,3-Dipolar cycloaddition of fluoroalkyl azides to acetylenes in the version of the click-chemistry involving the above mentioned catalytic system (CuSO4 5H2O – sodium ascorbate) essentially extended the set of the known fluoroalkyl derivatives of 1,2,3-triazole; some 1,2,3-triazole derivatives previously regarded as exotic became relatively accessible. Read et al. recently reported on the results of research of the effect on the m-хylene surface tension of the additives of exotic surfactants based on fluoroalkyl derivatives of 1,2,3-triazoles, e.g., compound 50 [50]. The synthesis of fluoroalkyl-1,2,3-triazoles using the above mentioned procedure was also described in the earlier publication of the same team [51].

A convenient method for the preparation of 1,2,3-triazole-containing CF3-α-aminophosphonates 51 via copper-catalyzed (3 + 2)-cycloaddition of α-CF3-α-aminophosphonates bearing an alkynyl group at the α-carbon atom to different organic azides has been described [52].

A convenient and simple method for the synthesis based on copper-catalyzed 1,3-dipolar cycloaddition of azidopeptides to acetylenes of tetrapeptide surrogates containing CF3-alkyl-1,2,3-triazolyl moiety 52 having ester of phosphonates functionalities or have been developed by Nenajdenko et al. [53, 54].

Some approaches to the synthesis of trifluoromethyl-1,2,3-triazoles by the azidation of compounds containing not a triple but a double С=С bond were described. In this case apparently a functional group elimination occurs in situ to provide the formation of an additional carbon-carbon bond. For instance, Miethchen et al. described a synthesis of 4-trifluoromethyl-1,2,3-triazole 53 linked to the C6-atom of D-galactose and D-altrose. 1,3-Dipolar cycloaddition using the monosaccharide azides and the perfluoroalkyl- substituted phenylvinylsulfones was performed [55, 56].

A number of 5-fluoroalkylated 1H-1,2,3-triazoles 54 was synthesized in good yield by the 100 % regioselective 1,3-dipolar cycloaddition of (Z)-ethyl-3-fluoroalkyl-3-pyrrolidino-acrylates with aryl or benzyl azides [57].

1,3-Dipolar cycloaddition of aryl (or benzyl) azides to 1,1,1-trifluoro-4-ethoxy-3-butene-2-one proceeded smoothly by heating without solvent. As a result 1-substituted 4-trifluoroacetyl-1H-1,2,3-triazoles 55 were formed regioselectively in good yield [58]. These compounds were readily hydrated at air exposure.

Recently Nenaidenko et al. carried out reactions of a variety alkyl and aryl azides with 1-trifluoromethylated 1,3-dicarbonyl compounds what lead 100 % regioselectively to a single 4-acyl-5-trifluoromethyl-1,2,3-triazoles 56 isomer in good yields [59]. The reaction represents a general and highly selective method for the synthesis of 1,2,3-triazoles otherwise difficulty available. The observed regioselectivity can be explained by selective enolization of trifluoromethyl ketone fragment to form enolate with double bond conjugated to the CF3 group.

Fluoroalkanesulfonyl azides in reactions with alkenes are more reactive than alkyl azides because of strong electron-acceptor properties of the sulfonyl group. Thus, Zhu, He et al. shown that the reaction of vinyl ethers with 1-fluoroalkanesulfonyl azides proceeded in mild conditions to afford 1-fluoroalkanesulfonyl-5-alkoxy-1,2,3-triazolines in good yields (≈70 %) [60, 61]. The authors point out that the cycloaddition is extremely regioselective. Only the 5-alkoxy derivative of 1,2,3-triazoline 57 has been obtained, whereas the corresponding 1,4-isomer has not been detected.

According to Shreeve et al., 4-trifluoromethyl-1,2,3-triazole 58 may be synthesized as a result of 1,3-dipolar cycloaddition of TMSN3 to trifluoropropyne in the presence of Cu(I) [62]. The synthesis of perfluoroalkyl-1,2,3-triazoles was also described in the earlier cited paper of Taylor et al. [35]. In this study the reaction was investigated between perfluoropropadiene with phenyl azide resulting in regioisomeric 1,2,3-triazole containing at the endocyclic carbon atoms both the fluorine atom and the CF3-group.
Below we described some other, less wide spread methods as compared to 1,3-dipolar cycloaddition used in the preparation of perfluoroalkyl derivatives of 1,2,3-triazoles. For instance, Haszeldine et al. [63] developed an original method of the synthesis of trifluoromethyl-1,2,3-triazole about 40 years ago. The reaction of diazomethyltrimethylsilane with trifluoroacetonitrile led to the formation of an intermediate adduct rearranged into 2-trimethylsilyl-4-trifluoromethyl-1,2,3-triazole 59. This compound being treated with aqueous ethanol liberated the trimethylsilane to yield 4-trifluoromethyl-1,2,3-triazole 58.

Bargamov and Bargamova reported on new polyfluorinated 1-amino-1,2,3-triazoles 60 which were obtained by oxidation of the bis-hydrazones of aliphatic polyfluorinated α-dicarbonyl compounds with sulfuryl chloride, bromine, or selenium dioxide in an aprotic solvent [64, 65]. In a later article these authors described an oxidation of a dihydrazone of 1,1,1,5,5,5-hexafluoro-4-trifluoromethylpentane-2,3-dione in the presence of the H2SO4 – P2O5 mixture (molar ratio 3:1) to 4-(1,1,1,3,3,3-hexafluoroisopropyl)-5-trifluoromethyl-2H-1,2,3-triazole 61 (no yield was given) [66].

The reactions of 2-trifluoromethylchromones and 2-trifluoromethyl-4H-chromen-4-imines with sodium azide in the presence of acetic acid gave ketone (or imine) derivatives of 5(4)-trifluoromethyl-1,2,3-triazole 62 in high yields [67].

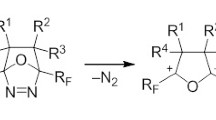
Furin et al. (2001) published a method for the synthesis of 4,4-bis(trifluoromethyl)-5-ethoxy-5-pentafluoroethyl-1,2,3-triazoline 63 based on the electrocyclic cyclization of linear vinyl azide as a precursor [68]. In the first stage of this process the perfluoro-2-methyl-2-pentene reacted with sodium azide in acetonitrile–ethanol mixture at −20 °C furnishing the vinyl azide, which at −10 °C underwent an intramolecular cyclization into an unstable intermediate 64. In the presence of ethanol a nucleophilic attack of ethoxy anion on С=N bond of intermediate 63 occurs to form a stable product 64 [68].

Bozkurt et al. demonstrated that 5-(perfluoroheptyl)-3H-1,2,3-triazole-4-carboxylate 65 is formed in a high yield by the intramolecular cyclization of ethyl 3-azido-3-(perfluorheptyl)propenoate 66 in presence of a significant excess of sodium azide [69].

An example of the “direct” incorporation of a perfluoroalkyl group into the structure of an already formed 1,2,3-triazole was given [70]. In this paper a method of synthesis of 1-trifuoromethyl-substituted benzotriazole 67 is described by the chlorination of the corresponding methyl 1-azoledithiocarboxylates 68 followed by the fluorination of the resulting 1-trichloromethyl derivatives 69 using anhydrous HF [70].

3.2 1,2,4-Triazoles
Perfluoroalkyl-1,2,4-triazoles are often formed by rearrangement of other heterocycles or by cyclization of carboxylic acid hydrazides or their analogues. In 1962 Brown and Cheng described for the first time the synthesis of 3,5-bis(perfluoroalky1)-1,2,4-triazoles 70 by treatment of bis(perfluoroalkyl)-1,3,4-oxadiazoles with methylamine [71]. The method for the synthesis of NH-unsubstituted 3,4-bis(perfluoropropyl)-l,2,4-triazole by the action of P2O5 on the corresponding hydrazine was also reported in this paper. Thirty years later Threadgill et al. [72] prepared 3,5-bis(trifluoromethyl)-4-(3-benzyloxypropyl)-4H-l,2,4-triazole by the reaction of 2,5-bis(trifluoromethyl)-l,3,4-oxadiazole with 3-benzyloxypropylamine in methanol (reflux, 9 days).

Brown et al. also suggested a version of the synthesis of 3,5-bis(perfluoroalkyl)-4H-1,2,4-triazoles 71 from perfluoroalkylhydrazides and from bis-(perfluoroalkyl)-1,2-dihydro-1,2,4,5-tetrazines [73].

Charushin et al. demonstrated that 4-substituted thiosemicarbazides react with di- and trifluoroacetic acids to give the corresponding 3-fluoroalkyl-4,5-dihydro-1,2,4-triazole-5(1H)-thiones 72 [74].

It should be noted that in 1998 El-Sayed and Khodairy recognized that thiones 72 are versatile substrates for the synthesis of fused and spiroheterocyclic systems [75]. Recently Chen et al. synthesized 4-amino-5-(trifluoromethyl)-4H-1,2,4-triazole-3-thiol 73 from thiocarbohydrazide [76]. The latter compound in its turn served as a reagent for the preparation of new functional triazole derivatives 74 and 75 [76].

Ivin et al. obtained 3-(perfluorohexyl)-7-phenyl-5H-[1, 2, 4]triazolo[3,4-b][1,3]thiazin-5-one 76 as a result of a reaction of methyl phenylpropynoate with a linear 1-(perfluoroheptanoyl)thiosemicarbazide 77 or a cyclic 5-(perfluorohexyl)triazole-3-thiol 78. In both cases the reaction conditions were practically identical, and the yields of compounds containing the trifluorohexyl group in the position 3 of the 1,2,4-triazole ring were comparable [77].

Lopyrev et al. developed an original method for the synthesis of 3-perfluoroalkyl-5-amino-1,2,4-triazoles 79 in nearly quantitative yields based on the cyclization of perfluoroacylaminoguanidines that in their turn were obtained in good yields (60–81 %) from hydrazides of perfluorocarboxylic acids and S-methylisothiourea [78].

New organotin(IV) compounds with 4-methyl-5-trifluoromethyl-4H-1,2,4-triazoline-3(2H)-thione fragments 80 have been synthesized and characterized [79]. The central tin atoms of complexes is five-coordinated with distorted trigonal bipyramidal geometry.

The synthesis of potentially biologically active substances whose structure contained a fragment of 5-trifluoromethyl-1,2,4-triazole linked to pyrazolo- or imidazolopyridine core 81 were presented in the article of Roberts et al. [80]. One of the typical schemes of the synthesis of such compounds includes a hydrazinolysis of the cyano group. Subsequent acylation with trifluoroacetic anhydride (TFAA) followed by the closure of the triazole ring afforded 81 (yields unknown).

Siedle et al. established that the hydrazinolysis of corresponding fluoroimine occurred via particular mechanism “nucleophilic addition – HF elimination” leading to the formation of 3,5-bis(heptafluoropropyl)-1,2,4-triazole 82 [81].

An original procedure of a “direct” incorporation of a trifluoromethyl group into molecules of heteroaromatic substrates was developed by Jamakawa et al. Using this procedure 3-amino-1,2,4-triazole was trifluoromethylated by trifluoromethyl iodide in DMSO in presence of hydrogen peroxide-ferrocene system [82] to obtain 3-amino-5-trifluoromethyl-1,2,4-triazole 83.

Yagupolskii et al. developed various versions of the alkylation of 1,2,4-triazolate anion with fluoroethylene [83] to form 1-(1,1,2,2-tetrafluoroethyl)-1,2,4-triazole 84 in a low yield.

The same authors developed an original method of the synthesis of 1-(1,2,2,2-tetrafluoroethyl)-1,2,4-triazole 85 by treatment of N-(2-chloro-1,1,2-trifluoro)-1,2,4-triazole with tetramethylammonium fluoride [83]. The assumed reaction mechanism consist of several steps. In the first stage elimination of HF and the formation of 2-chloro-1,2-difluoroethylene derivative takes place. Further chlorine atom is replaced by fluorine with the formation of 1,2,2-trifluoroethylene-1,2.4-triazole. Finally addition of HF gave the final product 85.

In extension of this study the sodium salt of 1,2,4-triazole was alkylated with 1,2-dibromotetrafluoroethane (Freon 114B2) to obtain N-(2-bromotetrafluoroethyl)-1,2,4-triazole 86. This compound was then subjected to photoinduced (UV-irradiation) reaction with thiophenol in liquid ammonia. Selective nucleophilic substitution of bromine with thiophenol residue was achieved under these conditions. As a result N-(2-phenylthiotetrafluoroethyl)-1,2,4-triazole 87 was obtained in a high yield [84, 85].

Lately Garg and Shreeve suggested a simple synthesis of trifluoromethanesulfonamide derivatives of 3-amino- and 3,5-diamino-1,2,4-triazoles 88, 89 from the corresponding aminoazoles and trifluoromethanesulfonyl fluoride [86].

New energetic materials were prepared from by perfluoroalkyl-1,2,4-triazoles. For instance, Shreeve et al. obtained 3-nitro-5-trifluoromethyl-1,2,4-triazole 90 by the diazotization of 3-amino-5-trifluoromethyl-1,2,4-triazole in the concentrated sulfuric acid [87]. In its turn 3-amino-1,2,4-triazole and 3-nitro-5-trifluoromethyl-1,2,4-triazole were used in the preparation of energetic 3-amino-1,2,4-triazolium 3-nitro-5-trifluoromethyl-1,2,4-triazolate 91. Energetic polymeric material 92 was synthesized containing in the monomer unit ions of 3-nitro-5-trifluoromethyl-1,2,4-triazolate. The yield of polymer is unknown [88].

The perfluoro-1,2,4-triazoles are known to be effective ligands. For instance, а number of charge neutral Os(II) pyridyl-1,2,4-triazolate complexes with either bis(diphenylphosphino)methane 93 or cis-1,2-bis(diphenylphosphino)ethene 94 chelates were synthesized, and their structural, electrochemical, photophysical properties and thermodynamic relationships were established [89, 90]. Chi, Carty et al. synthesized 6-(3-trifluoromethyl-1,2,4-triazolyl)-2,2’-bipyridine and built up on this basis tridentate 6-azolyl-2,2′-bipyridine chelate complex compounds of Ga and In 95 [91].

Among trifluoromethyl-derivatives of 1,2,4-triazole, an efficient inhibitors of dipeptidyl peptidaze IV (sitagliptin and its derivatives) have been found (see Sect. 6.1).
3.3 Tetrazoles
5-Trifluoromethyltetrazole 96 – the simplest of tetrazoles containing a perfluoroalkyl substituent – was prepared for the first time by Norris in 1962 by the cycloaddition of the azide-anion to the trifluoroacetonitrile [92]. This reaction occurred with a considerable heat evolution. The conversion of corresponding anion 97 into the neutral form required the use of concentrated mineral acids due to the relatively high NH-acidity of tetrazole 96 (pK BH+ 1.1) [28, 93]. It was shown later that the 5-trifluoromethyltetrazole 96 could be obtained reacting CF3CN with coordinated azides similarly to the synthesis of 4,5-bistrifluoromethyl-1,2,3-triazole 40 [44].

To explore the properties of some energetic salts 98, in a series of publications Shreeve et al. described the procedure of the synthesis of NH-5-difluoroaminodifluoromethyltetrazole by same manner for which F2NCF2CN was used as a precursor [94].

The 1-substituted 5-trifluoromethyltetrazoles may be also obtained by the azidation of imidoyl halides. For instance, the nucleophilic substitution of the chlorine for the azide group in the N-methylimidoyl chloride provided the corresponding imidoyl azide (azidoazomethine), which suffered a cyclization into 1-methyl-5-trifluoromethyltetrazole 99 [95]. Сarpenter et al. synthesized 1-benzyl-5-trifluoromethyltetrazole 100 by replacing the fluorine atom in N-benzyltrifluoroacetimidoyl fluoride by an azide group followed by electrocyclic cyclization of the intermediate imidoyl azide (azidoazomethine). The yield of purified product 100 is unknown [39].

1,3-Dipolar cycloaddition of perfluoroalkylethyl azides to isocyanates afforded 1-perfluoroalkyl-4-(n-Bu, phenyl or mesitylsulfonyl) tetrazol-5-ones 101 in good yields [96].

The synthesis of surfactants in the series of fluoroalkyltetrazoles 102, 103 and the study of their effect on the surface tension of m-xylene was described by Read et al. [50, 97].

The synthesis of N-fluoroalkyltetrazoles may be carried out also by the alkylation of the corresponding NH-unsubstituted tetrazoles. Jończyk et al. showed that 5-benzyl-1H-tetrazole reacted with chlorodifluoromethane in the presence of concentrated aqueous sodium hydroxide and a catalyst, benzyltriethylammonium chloride (TEBAC), in THF with the formation of regioisomeric N-difluoromethyl substituted derivatives 104, 105 [98].

An interesting approach to the synthesis of tetrazoles with fluorine atoms in the side chain was suggested by Fuchigami et al. who performed an anodic monofluorination of 1-substituted 5-tetrazolyl sulfides containing an α-electron-withdrawing group (EWG) leading to the formation of the corresponding C-F derivatives 105 [99].

Norris in his pioneering article described also some chemical transformations of 5-trifluoromethyltetrazole 96 [92]. For instance, the alkylation of anion 97 with methyl iodide resulted in regioisomeric N-methyl-5-trifluoromethyltetrazoles 106, 107 with 2-methyl isomer 107 prevailing. Also a direct halogenation was performed of the 5-trifluoromethyltetrazole sodium salt with the molecular chlorine furnishing the N-chloroderivative of 5-trifluoromethyltetrazole that was found an explosive extremely dangerous at handling [92].

The high regioselectivity of tetrazolate 97 alkylation in the environment of aqueous acetone giving predominantly the 2Н-isomer was later noted also by Spear et al. [100]. 5-Trifluoromethyltetrazole 96 slowly reacts with formaldehyde in water solution at pH 5 giving 2-hydroxymethyl-5-trifluoromethyltetrazole 108 [101]. Later the role of the electronic effects of the substituents at the endocyclic carbon of the tetrazole, and also the influence of the solvation effects on the alkylation regioselectivity of tetrazoles was treated in detail in quite a number of theoretic and experimental publications [28, 102].

Uncompromising regioselectivity of exhaustive alkylation of 2-substituted 5-trifluoromethyltetrazoles was exploited in a recently developed elegant procedure for the synthesis of 1-alkyltetrazoles starting from N-unsubstituted ones. This three-step reaction sequence utilizing an N2-regioselective t-butylation (the product is compound 109) in the first step, was reported to provide isomerically pure products 110 in high to nearly quantitative yields [103, 104].

2-(1-Methylvinyl)-5-trifluoromethyltetrazole 111 was obtained by the regioselective alkylation of 5-trifluoromethyltetrazole with 3-bromopropene in sulfuric acid followed by dehydrohalogenation of the intermediate products [105].

Generally, alcohols, readily generating carbenium cations in the presence of acidic catalysts, were found to react with NH-unsubstituted 5-trifluoromethyltetrazole 96 yielding N2-alkylated products 112 [28, 103]. The reaction can be carried out in neutral organic solvents (chloroform, dichloromethane, acetonitrile, nitromethane) in the presence of catalytic amounts of sulfuric or p-toluenesulfonic acids as well as Lewis acids like boron trifluoride etherate or zinc triflate.

Alkylation of 5-trifluoromethyltetrazole 96 with esters of 2-nitro-2-azapropanol in the presence of catalytic amounts of sulfuric acid was described [106]. Here the regioselectivity of the process was not assessed and a mixture of 1- and 2-alkyltetrazoles 113 (the ratio is unknown) was obtained.

Gaponik et al. found that the NH-unsubstituted 5-trifluoromethyltetrazole in the systems containing transition metal salts formed water-soluble polymeric complexes [107].
4 Perfluoroaryl and Fluorobenzo-Fused Heterocycles
4.1 1,2,3-Triazoles
Banks and Prakash were first to demonstrate the wide opportunities of the 1,3-dipolar cycloaddition of 1-azido-2,3,4,5,6-pentafluobenzene 114 to acetylenes as the general method of the synthesis of 1,2,3-triazoles 115–120 containing a perfluorophenyl group at the endocyclic nitrogen atom [108].

1,3-Dipolar cycloaddition of benzyl azide to 3-(2,3,4,5,6-pentafluorophenyl)propynenitrile resulted in the formation of isomeric 1,2,3-triazoles 121, 122 in 17 and 61 % yield respectively [109].

Swager et al. synthesized various Ir(III) complexes 123, among them complexes containing a perfluoroaryl substituent [110]. The method of building up these structures is underlain by the “click-reaction” leading to the formation of the corresponding Cu(I)-triazolide intermediate 124.

Schubert et al. subjected to a systematical examination reactions of 1,3-[3 + 2]-cycloaddition of substituted aromatic azides to trimethylsilylacetylenes in water affording regioisomeric 1,2,3-triazoles, in particular, those containing a perfluoroaryl substituent at endocyclic atoms of the heterocycle 125, 126 [111].

Recently, a number of chiral phosphoramidite ligands 127 containing 1,2,3-triazole ring at the 3,3′-positions of the binol scaffold were synthesized by McErlean et al. [112].

O’Mahony et al. [113] advanced an alternative version of cyclization resulting in perfluoroaryl-1,2,3-triazoles. These authors demonstrated that 2-(2,3,4,5,6-pentafluorophenyl)-4-methyl-5-methylthio-1,2,3-triazole 128 obtained by this procedure (yield is unknown) possessed considerable pesticide activity.

The paper of Frenna, Spinelli et al. contained data on the kinetics of the rearrangement of 3-benzoyl-5-phenyl-1,2,4-oxadiazole 129 into the corresponding 2-aryltriazoles 130 (Boulton–Katritzky reaction) [114].

Haszeldine in 1970 developed several routes for the synthesis of perfluorobenzo-fused 1,2,3-triazole 131 from decafluoroazoxybenzene 132, tetrafluoro-o-phenylenediamine 133, and 2,3,4,5,6-pentafluoronitrobenzene 134 [115]. More than a quarter of a century later Heaton et al. suggested a similar version of the synthesis of tetrafluorobenzotriazole 131 from 1,2,3,4-tetrafluoro-5,6-dinitrobenzene and 3,4,5,6-tetrafluoro-1,2-phenylenediamine that were regarded as versatile semiproducts in the synthesis of various tetrafluorobenzоheterocycles [116].

The synthesis of 1-phenyl-4,5,6,7-tetrafluorobenzotriazole 135 from tetrafluoroanthranilic acid through tetrafluorodehydrobenzene as intermediate is described by Yakobson et al. in 1967 [117].

Williams et al. reports that 1-(2,3,4,5-tetra-O-benzyl-β-D-glucosyl)-3,4,5,6-tetrafluoro-1–H-benzo[d][1,2,3]-triazole 136 can be prepared from available anomeric azide and 2-amino-3,4,5,6-tetrafluorobenzoic acid through “click” methodology [118].

Recently “click-chemistry” approach to the preparation of fluorobenzo-fused 1,2,3-triazole 137 was demonstrated in the paper of Larock et al. [119]. Zhang and Moses developed a special version of one-pot “click-chemistry” for the preparation of monofluoro derivatives of benzotriazole 138, 139 from p-methoxyaniline and o-fluoroanthranilic acid with two in situ generated intermediates: p-methoxyphenyl azide and fluorobenzyne [120].

Driver et al. explored the Ni-catalyzed C7-alkenylation of 6-fluorotriazolopyridine with diphenylacetylene applying bis-(1,5-cyclooctadiene)nickel(0) (Ni(COD)2) leading to the formation of fluorobenzotriazole 140 [121]. It was underlined in the article that the crucial importance for the high conversion of the reagents and the sufficient yields of the alkenylation products had the selection of the Lewis acid which was AlMe3 in this study.

Some isomers of fluorobenzotriazoles are capable of ring-chain rearrangements with the opening of the triazole ring. Elguero, Alkorta et al. performed a quantum-chemical investigation of the ring-chain isomerization of fluoro derivatives of 1,2,3-triazolopyridines 141 [122].

4.2 1,2,4-Triazoles
Efficient enantioselective catalysts of various organic reactions were found among pentafluoroaryl-1,2,4-triazoles. Tetrafluoroborates of 2-(2,3,4,5,6-pentafluorophenyl)-1,2,4-triazolium 142–147 are practically useful compounds. The synthesis and application examples of these catalysts are presented in the series of publications [123–129].

As it was shown by Golding et al., 4-(2,3,4,5,6-pentafluorophenyl)-1,2,4-triazoline-3,5-dione 148 belonging to perfluoroaryl-1,2,4-triazoles is an efficient reagent for trapping volatile organic compounds (VOCs) included in the list of the most important environmental pollutants [130].

Shaaban showed the possibility to obtain under the microwave irradiation fused systems, 1,2,4-triazolo[1,5-a]pyrimidine 149 and 1,2,4-triazolo[3,4-c][1, 2, 4]triazine 150, containing a trifluoromethyl group as a substituent in the six-membered ring [131].

Saloutin, Chupakhin et al. applied a related condensation involving 1,2,4-triazolyldiazonium resulting in the formation of 6-benzoyl-7-hydroxy-7-trifluoromethyl-4,7-dihydro[1,2,4]triazolo[5,1-c][1,2,4]triazine 151 (59 %) [132]. Rusinov et al. reported on another successful example of this reaction involving lithium β-diketonates and 3-amino-1,2,4-triazole and affording 7-fluoromethyl-1,2,4-triazolo[1,5-a]pyrimidine 152 [133].

Dolzhenko et al. synthesized 12 new fluorinated 7-aryl-2-pyridyl-6,7-dihydro[1,2,4]triazolo[1,5-a][1,3,5]triazin-5-amines 153 via three-step procedure starting from (iso)nicotinic hydrazides [134].

Nenajdenko et al. obtained regioisomeric 5-CF3 or 7-CF3 triazolopyrimidines 154, 155 by the reaction of 1,1,1-trifluoro-4-sulfonyl-but-3-ene-2,2-diol with 3-amino-1,2,4-triazole [135].

4.3 Tetrazoles
Kim et al. showed that 1,3-dipolar cycloaddition of alkynyl Pd(II) azido complexes to perfluorobenzonitrile proceeded at room temperature affording the corresponding N-coordinated tetrazolato compound (trans-[Pd(C ≡ CPh)(N4C-C6F5)(PMe3)2]) 156 [136].

Klapötke et al. obtained recently by the intermolecular 1,3-dipolar cycloaddition of organomercury(II) azide to pentafluorocyanobenzene the corresponding (5-pentafluorophenyl-2H-tetrazol-2-yl)phenylmercury 157 (yield is unknown) [137].

The synthetic methods and photoluminescent properties of new dendrimers with an electron-deficient fluorinated starburst oxadiazole core were discussed in the publication of Chen, Fan et al. [138]. Here in the first, key stage the 1,3-dipolar cycloaddition occurred of 2,3,4,5,6-pentafluorobenzonitrile to NaN3 along the known procedure (Demko-Sharpless) giving 5-(perfluorophenyl)-1H-tetrazole 158. In the next stage 5-(perfluorophenyl)-1H-tetrazole was acylated by benzene-1,3,5-tricarbonyl trichloride to obtain 1,3,5-(5-perfluorophenyl-1,3,4-oxadiazol-2-yl)benzene 159.

The fused tetrazoles are prone to ring-chain isomerism. Cmoch and Korczak investigated the azidо-tetrazole equilibrium between two fluorine-containing tetrazoles: 6-fluorotetrazolopyridine 160 and 6-trifluoromethyl-8-chlorotetrazolopyridine 161 [139]. It turned out that the equilibrium in the case of compound 160 is completely shifted to the side of azide 160b, whereas for compound 161 the NMR spectra showed the presence of both tetrazole 161a and azide 161b forms.

Keith (2006) developed an original one-stage solvent-free method of preparation of fluorine-containing tetrazolo[1,5-a]pyridines 162, 163 forming in a plausible yield from the corresponding pyridine N-oxide and activated diphenylphosphorazidate (DPPA) in the presence of pyridine [140]. In this case the azido-tetrazole equilibrium is virtually totally shifted to the ring form.

In the above cited paper [135] it was also reported that the reaction of 1,1,1-trifluoro-4-sulfonylbut-3-ene-2,2-diol with 5-aminotetrazole furnished regioisomeric 5-CF3 or 7-CF3 tetrazolopyrimidines 164, 165. It was also shown that the ratio of the regioisomers (at equal overall yield of the products) was governed by the nature of the substituent R in the 1,1,1-trifluoro-4- sulfonylbut-3-ene-2,2-diol.

5 Other Types of Perfluorinated Substituents
5.1 SF5 Derivatives
Pentafluorosulfanyl (SF5) group is interesting as a stable moiety bearing simultaneously five fluorine atoms. In recent reviews [141, 142] it was stressed that this group is now widely applied in the organic synthesis and in industry. 1,2,3-Triazoles containing the SF5 group attached to the endocyclic carbon atoms became recently more accessible. A number of publications describes the acceleration of the 1,3-dipolar cycloaddition with the use of the catalytic system based on Cu(I) generated in situ from CuSO4 and sodium ascorbate. This procedure made it possible to obtain in relatively mild conditions a series of 1-R-4-pentafluorosulfanyl-1,2,3-triazoles 166 in “good yield” [62, 143].

It should be noted that the recent advances in the chemistry of trifluoromethyl- and pentafluorosulfanyl derivatives of tri- and tetrazole favor the applied studies of these unique objects of the chemistry of the fluoro-containing heterocycles. Thus a significant attention is paid to 1,2,3-triazoles and tetrazoles containing a pentafluorosulfanyl group. The introduction of this group into the heterocycle provides a possibility to prepare energetic compounds of high density [142]. Shreeve et al. investigated the 1,3-dipolar cycloaddition of alkyl azides containing a terminal pentafluorosulfanyl group to substituted acetylenes [144].

In this case the yield of pentafluorosulfanylalkyl-4-R-1,2,3-triazoles 167 crucially depends on the ratio of the initial reagents and on the reaction temperature. At the excess of the sodium azide and at a high temperature the replacing azidation occurs of both the tosyl and the pentafluorosulfanyl groups giving the corresponding diazide and further bis-1,2,3-triazoles 168 in good yields [144].
5.2 NF2 Derivatives
Tri- and tetrazoles containing an NF2 group at the endocyclic nitrogen or carbon atoms belong to an exotic group of highly energetic compounds. The combination in the same molecule of the polynitrogen heterocyclic ring and the electron-acceptor difluoroamine group enhanced the energy potential of these molecules. The prospect of application of the difluoroamino derivatives of 1,2,4-triazole as components of energetic ionic liquids was estimated by the ab initio quantum-chemical calculations [21]. In the evaluation of the application prospects of the N-difluoroazoles their high sensitivity to explosive decomposition caused by various effects (impact, friction, electric discharge, fire beam) should be taken into consideration [94, 145, 146]. In an original article Shevelev et al. [147] reported on the preparation of NF2 derivatives of 3-nitro-1,2,4-triazole 169 by the action of O-fluorosulfonyl-N,N-difluorohydroxylamine (F2NOSO2F) on NH-form of the corresponding 1,2,4-triazoles.

6 Applications
6.1 Medicine and Medicinal Chemistry
The medicinal applications of fluorinated tri- and tetrazoles grow steadily [148]. Some compounds containing in their molecular structure these heterocycles are included in the list of highly efficient modern drugs [149]. Many among fluorinated tri-and tetrazoles are regarded as promising antidiabetic, cardiological, fungicidal, antibacterial, and antiviral pharmaceuticals, drugs for the treatment of the central nervous system, etc. We give below some examples of these compounds exhibiting versatile kinds of biological activity.
6.1.1 Antidiabetics
Sitagliptin 170, the first inhibitor of dipeptidyl peptidase IV (DPP-4) approved by the FDA for the treatment of type 2 diabetes, has a trifluorophenyl group linked to a β-amino butanoyl moiety coupled to a triazolopiperazine [150, 151]. Many publications appeared treating the problem of the synthesis of this compound and its derivatives. For instance, a scheme of asymmetric synthesis of the sitagliptin phosphate from the precursor, α,β-enamine amide, is given in [152]. A wide search for effective antidiabetic agents was performed among the homologues of this compound; therewith the varied structural parameter was the substituent in the phenyl ring [153] and also the substituents in the piperazine fragment [154]. Biftu et al. based on the data of XRD analysis and computer simulation proposed a structure and synthesized a new analogous compound 171 that also proved to be an efficient inhibitor of DPP-4 (IC50 = 21 nM), showed high activity in vivo and possessed a feasible pharmacokinetic profile [155]. Note an interesting study of Chen et al. who synthesized and tested the biological activity of a series of Sitagliptin analogs in whose molecules the fragment of 3-trifluoromethyl-1,2,4-triazolopiperidine was replaced by 4R-1,2,3-triazolopiperidine moiety. The highest biological activity was found in compound 172 [156].

6.1.2 Fungicides
The following fluoroaryl derivatives of 1,2,4-triazole belong to the third generation fungicides: Fluconazole 173, Fosfluconazole (prodrug) 174, Voriconazole (UK 109496) 175, Itraconazole 176 [4]. All these compounds are the inhibitors of the fungal cytochrome P450 enzyme 14α-demethylase. Recently results were published of the investigation of the fungicidal activity of the derivatives of 3-trifluoromethyl-1,2,4-triazole-5-thione: one among these substances, 177, exhibited a high activity [157]. At present the research is carried out on the preparation of active pharmaceutical ingredients of new fungicides, Fluconazole analogs, containing alongside the 1,2,4-triazole ring fluoropyrimidine, tetrazole, and also the other heterocyclic fragments [158].

6.1.3 Antibacterials
Faidallah et al. showed that the derivatives of 3,5-di(trifluoromethyl)-1,2,4-triazolesulfonyl urea and thiourea exhibited a pronounced antimicrobial action [159]. McGeary et al. established a considerable inhibitory activity of 4-methyl-5-(trifluoromethyl)-4H-1,2,4-triazole-3-thiol and its derivatives with respect to β-lactamases [160].
6.1.4 Antiviral Agents
Girardet et al. synthesized and investigated the anti-HIV-1 activity of the derivatives of 3-trifluoromethyl-1,2,4-triazole-5-thiol 178 [161]. The anti-HIV action of compound 178 proved to be comparable with that of the efficient protease inhibitor Efavirenz. The authors of the article noted that the compound exhibited single-digit nanomolar activity against the Y188L mutant, with no cytotoxicity.

Known publications were cited in [162] concerning the structures of 1,2,3-triazole analogs of nucleosides containing a polyfluoroalkyl substituent in the heterocyclic core 179–181.

Nucleoside mimetics, N2-substituted derivatives of 4-tosyl-5-polyfluoroalkyl-1,2,3-triazoles containing fragments of 3-chlorotetrahydrofuran, 3-chloro-tetrahydropyran, tetrahydropyran, dihydrofuran, dihydropyran, or acyclic substituents, were also studied 182–186 [162]. It was demonstrated that some among the studied compounds exhibit a pronounced anti-Epstein-Barr virus (EBV) activity.

6.1.5 Hypotensive Drugs
Angiotensin II receptor antagonist like Losartan and its analogs retain strong positions on the pharmaceutical market of the hypotensive drugs [158]. Yagupolskii et al. have synthesized two fluorine-containing Losartan analogs with fluoroalkyl substituents 187–189, whose pharmacological activity is expected to be high [163].

6.1.6 CNS Therapy
Lebsack et al. produced a series of 1,2,3-triazolo[3,4-a]phthalazine derivatives, in particular, containing a trifluoromethyl substituent in the fused 1,2,4-triazole ring 190, as high-affinity ligands to the α2δ-1 subunit of voltage gated calcium channel [164]. These compounds are interesting as anticonvulsant drugs. The work of Williams et al. [165] consisted in the synthesis and the study of the biological activity of new antidepressants, 3-styryl[4.5]-spiroether and [4.5]-spiroether neurokinin-1 (NK1) antagonists 191, 192, containing in the molecular structure trifluoromethyltetrazol-1-yl fragment. The research concerning the pursuit of promising neurokinin-1 (NK1) antagonists, containing fluorinated tri- and tetrazoles is continued [166].

1,2,4-Triazol-3-yl-thiopropyl-tetrahydrobenzazepine 193 whose structure includes a nonfused 1,2,4-triazole-3-trifluoromethyl-5-thiol fragment separated by an alkyl bridge from the benzazepine framework was found to be a potent and selective dopamine D3 receptor antagonist [167]. Sugane et al. established the high biological activity in vitro of new glycine transporter 1 (GlyT1) inhibitors 3-biphenyl-4-yl-4-(2-fluorophenyl)-5-R-4H-1,2,4-triazole, in particular, of compound 194 [168].

Below two structural formulas of potent, orally active, long-acting morpholine acetal human NK-1 receptor antagonists 195, 196, are given. These compounds are tested as efficient drugs for the treatment of the Alzheimer disease [169]. The fluorine-containing biaryl-triazolopyridine 197 is tried as a potent and selective p38α inhibitor for stress relief [170].

6.1.7 Imaging Agents
One of the modern ways of CNS disorders diagnostics depends on the development of new radiolabeled NK1 receptor antagonists. Some examples of fluorine-18 labeling compounds are known [171, 172], effective radioligands for imaging brain neurokinin type-1 (NK1) receptors in clinical research and drug discovery with positron emission tomography, like [18F]SPA-RQ 198.

6.2 Light-Emitting Diodes
Some complex compounds of fluorinated tri- and tetrazoles possess interesting photo- and electroluminescent properties. We have given above formulas of 93, 94, representatives of a new series of charge neutral Os (II) pyridyl 3-trifluoromethyl- 1,2,4-triazolate complexes with either bis(diphenylphosphino)methane or cis-1,2-bis(diphenylphosphino)ethene chelates. Their structural, electrochemical, photophysical properties and thermodynamic relationship were established [89, 90]. It was demonstrated that such coordination compounds can be used in the production of highly efficient white [173], blue [174], orange, and red organic light-emitting diodes (LEDs) [175].
The photo- and electroluminescence properties of a series of novel, heteroleptic, mer-cyclometallated iridium complexes have been fine-tuned from green to blue by changing the substituents on the 3-trifluoromethyl-1,2,4-triazolylpyridyl ring of the ligand [176, 177]. Chi, Chou, Wu et al. synthesized Ir(III) emissive complexes with 5-pyridyl-3-trifluoromethyl-1,2,4-triazole ligand which might be used in white OLEDs technologies [178].
6.3 Sorbents, Ion Liquids, and Surfactants
Omary et al. have demonstrated that fluorous metallorganic frameworks (FMOFs) 199, 200 are highly hydrophobic porous materials with a high capacity and affinity to C6-C8 hydrocarbons of oil components [179]. FMOFs exhibits reversible adsorption with a high capacity for n-hexane, cyclohexane, benzene, toluene, and p-xylene, with no detectable water adsorption even at near 100 % relative humidity drastically outperforming activated carbon and zeolite porous materials. The results suggest great promise for FMOFs in applications like removal of organic pollutants from oil spills or ambient humid air, hydrocarbon storage and transportation, water purification, etc. under practical working conditions

Read et al. synthesized surface-active derivatives of 1,2,3-triazole 49, 50 containing at the nitrogen atom perfluoroalkyl (lipophilic) substituents, and at the carbon atom perfluoroalkoxy or alkoxy (hydrophilic) groups. It is significant that the surfactant properties of these compounds can be purposefully regulated varying the length of the fluoroalkyl chain [49, 50].
The ionic liquids formed by the 1,2,4-triazolium cation and dinitramide anion as well as charge-diffuse tetrazolium cation with a variety of substituents coupled with various (usually oxygen containing) anions have been studied by ab initio quantum chemistry calculations [21, 180].
6.4 Ion and Electron Conductors
We cited above the study of Boskurt et al. who synthesized 5-(perfluoroheptyl)-3H-1,2,3-triazole-4-carboxylate 65 [69]. They also reported on the effect of the proton conductivity observed in the solutions of organic electrolytes with the additives of this fluoroalkylated 1,2,3-triazole. Recent research advances on conjugated polymers for photovoltaic devices have focused on creating low band gap materials, but a suitable band gap is only one of many performance criteria required for a successful conjugated polymer [181]. This work focuses on the design of two medium band gap copolymers for the use in photovoltaic cells which are designed to possess a high hole mobility, low highest occupied molecular orbital and lowest unoccupied molecular orbital energy levels. The resulting fluorinated polymer 201 is a viable candidate for the use in highly efficient tandem cells. It also highlights other performance criteria which contribute to high photovoltaic efficiency, besides a low band gap.

6.5 Ligands
Some fluorine-containing tri- and tetrazoles behave as polidentate ligands and form stable complex compounds with various metal ions. The coordination compounds of Os(II), Ir(III), Ga(III), In(III), Ag(I), Sn(IV) with 5-pyridyl-2-yl-3-trifluoromethyl-1,2,4-triazole ligands we have already mentioned.

Let us cite some other characteristic examples. Thus, based on 5-(6-methylpyridin-2-yl)-3-trifluoromethyl-1,2,4-triazole ligand platinum(II) complexes 202 were synthesized with spatially encumbered chelates, and their photophysical properties were also studied [182]. Compound 202 was found to be weakly emissive in both fluid and solid states at room temperature. Exotic coordination compounds are also known prepared from the mentioned bidentate ligands and the isoelectronic system Ir(I) 203 with distorted square-planar geometry [183]. Here the lowest absorption band consists of increased triplet dπ → π transitions of Ir (I) atom. Later Swager et al. obtained heteroleptic tris-cyclometallated Ir(III) complexes 204 based on 2-(1-perfluorophenyl-1,2,3-triazol-4-yl)pyridine [110]. Complex compounds 205 formed by two tridentate ligands coordinated to Os(II) or Ru(II) interesting as components of dye-sensitized solar cells (DSCs), were recently prepared by Chou et al. [184].
6.6 Organic Catalysis
Chiral N-pentafluorophenyl 1,2,4-triazolium salts (triazolium bicyclic catalysts) had been found to significantly influence reaction yields and enantiomeric ratios. The examples of the application of triazolium bicyclic catalysts 142, 144, 145, 147 whose structures we have already mentioned are listed in Table 1.
6.7 Energetic Compounds
The burning of the energetic compositions containing organofluorine derivatives affords products with relatively small molecular mass which favors the operating characteristics. Owing to these and many other useful properties the fluorine-containing heterocyclic compounds, first of all tri- and tetrazoles, are traditionally attractive energetic substances.
Klapotke et al. described the energetic properties of sodium 5-trifluoromethyltetrazolate, sodium 5-pentafluoroethyltetrazolate, and sodium 5-heptafluoropropyltetrazolate [185]. Based on DSC-thermograms the authors concluded that the 5-perfluoroalkyltetrazole salts are thermally and thermodynamically stable energy-rich substances. It was also indicated that the sodium salts of 5-perfluoroalkyltetrazoles showed relatively low sensitivity to impact and friction. The other publications of this scientific team reported on the calcium salts of 5-perfluoroalkylated tetrazoles as components of ternary mixtures with magnesium and Viton [186], and also on pyrolants containing magnesium and guanidinium 5-(perfluoropropyl)-5H-tetrazolate and ammonium 5-(perfluoropropyl)-H-tetrazolate [187] The subject of the energy-rich salts formed from nitro-1,2,4-triazol-5-one, 5-nitroaminotetrazole, and other nitro-substituted azoles, among them energetic polymer salts from 1-vinyl-1,2,4-triazole derivatives, was treated in a series of Shreeve et al. publications that we cited before [62, 86–88, 94]. The data on enthalpy of formation, density, detonation velocity (calculated value), and on the other parameters were published making it possible to regard these salts as interesting components of explosive compositions. As promising components of explosive compositions trifluoromethyl- or pentafluorosulfanyl-substituted poly-1,2,3-triazole compounds should be mentioned. Below are given the formulas of some representative of pentafluorosulfanyl derivatives of tri- and tetrazoles 206–210. It is noteworthy that the introduction of the SF5 group into the 1,2,3-triazole ring results in energetic compounds of high density (1.83–1.90 g/cm3) [142, 188], the most important quality for the energy-rich compounds and materials.
Let us turn again to the pioneering publication of Shevelev et al. [147] that has announced the synthesis of N-difluoroazoles, the representatives of the new series of N-substituted azole. Focusing our attention on NF2 derivative of 3-nitro-1,2,3-triazole containing the NF2 group 211, it may be stated that this extraordinary molecule may be regarded as a precursor of the hypothetic substance possessing exclusively high detonation parameters. Presumably, due to the high sensitivity to the mechanical treatment (impact, friction) similar compounds are very dangerous in handling.

7 Conclusions
The analysis of the literature published within the last decade easily demonstrates the essential intensification of applied research in the field of fluorinated derivatives of tri- and tetrazoles. The especially intensive development is observed in the directions of creating new biologically active substances based on the mentioned compounds, light-emitting diodes, polymer materials, sorbents, catalysts of chemical processes. The appearance of original and refinement of the known approaches to the synthesis of fluorine-containing tri- and tetrazoles is a natural response to these demands. It is easy to forecast in the near future a significant success in this field of the chemistry of fluorine-containing heterocyclic compounds.
References
Kirk KL (2009) Fluorinated five-membered nitrogen-containing heterocycles. In: Petrov VA (ed) Fluorinated heterocyclic compounds. Synthesis, chemistry and applications. Wiley, New York
Uneyama K, Sasaki K (2009) Pharmaceuticals containing fluorinated heterocyclic compounds. In: Petrov VA (ed) Fluorinated heterocyclic compounds. Synthesis, chemistry and applications. Wiley, New York
Hong W (2009) Agricultural products based on fluorinated heterocyclic compounds. In: Petrov VA (ed) Fluorinated heterocyclic compounds. Synthesis, chemistry and applications. Wiley, New York
Kirk KL (2006) Fluorine in medicinal chemistry: recent therapeutic applications of fluorinated small molecules. J Fluorine Chem 127:1013–1029. doi:10.1016/j.jfluchem.2006.06.007
Gakh AA, Kirk KL (2009) Fluorinated heterocycles, ACS symposium series. American Chemical Society, Washington, DC. doi:10.1021/bk-2009-1003.ch001
Pevzner MS (2000) Aromatic N-haloazoles. Adv Heterocycl Chem 75:1–77
Burger K, Wucherpfennig U, Brunner E (1994) Fluoro heterocycles with five-membered rings. Adv Heterocycl Chem 60:1–64
Furin GG, Fainzilberg AA (2000) Modern methods for fluorination of organic compounds. Nauka, Moscow
Furin GG, Chi K-W (2001) Synthesis methods for fluoroorganic compounds. University of Ulsan Press, Ulsan
Furin GG (2004) Fluorine-containing heterocycles. Part I. Synthesis by intramolecular cyclization. Adv Heterocycl Chem 86:129–224
Furin GG (2004) Fluorine-containing heterocycles. Part II. Synthesis of perfluoroalkyl heterocycles from carbonyl compounds. Adv Heterocycl Chem 87:273–383
Furin GG (2005) Fluorine-containing heterocycles. Part III: synthesis of perfluoroalkyl heterocycles using perfluoroolefins containing a reactive group at the double bond. Adv Heterocycl Chem 88:231–310
Pace A, Buscemi S, Vivona N (2005) The synthesis of fluorinated heteroaromatic compounds. Part 1. Five-membered rings with more than two heteroatoms. Org Prep Proced Int 37:447–506. doi:10.1080/00304940509354978
Dawood KM (2004) Electrolytic fluorination of organic compounds. Tetrahedron 60:1435–1451. doi:10.1016/j.tet.2003.11.017
Nenajdenko VG, Balenkova ES (2011) Preparation of unsaturated trifluoromethylketones and their application in the synthesis of heterocycles. Arkivoc 2011(1):246–328
Gakh AA, Romanko SV, Urgak BI, Fainzilberg AA (1991) N-fluorination with cesium fluoroxysulfate. Tetrahedron 47:7447–7458
Strazdina A, Grinsteins V (1968) Preparation of N-halogen derivatives of 1,2,4-triazole. SU203693
Langlet A, Ostmark H (1994) Explosive body. WO 94/024434
Miroshnichenko EA, Lebedev VP, Kostikova LM, Vorob’eva VP, Vorob’ev AB, Inozemtcev JO (2003) Book of Abstr Int Ann Conf ICT34. 122/1-122/7
Pevzner MS, Trifonov RE (1998) Theoretical study of the structure of N-halo-1,2,4-triazoles. Russ J Org Chem 34:742–743
Schmidt MW, Gordon MS, Boatz JA (2005) Triazolium-based energetic ionic liquids. J Phys Chem A 109:7285–7295
Oziminski WP, Dobrowolski JC, Mazurek AP (2003) DFT studies on tautomerism of C5-substituted 1,2,3-triazoles. J Mol Struct 651–653:697–704
Dabbagh HA, Rasti E, Chermahini AN (2010) Theoretical studies on tautomerism of triazole derivatives in the gas phase and solution. J Mol Struct (Theochem) 947:92–100
Oziminski WP, Dobrowolski JC, Mazurek AP (2004) DFT studies on tautomerism of C5-substituted 1,2,4-triazoles. J Mol Struct (Theochem) 680:107–115
Chermahini AN, Dabbagh HA, Teimouri A (2007) Relation between the substituent effect and aromaticity in tetrazoles, protonated tetrazoles and tetrazolate derivatives. J Mol Struct (Theochem) 822:33–37
Sadlej-Sosnowska N (2001) Application of natural bond orbital analysis to delocalization and aromaticity in C-substituted tetrazoles. J Org Chem 66:8737–8743
Chermahini AN, Nasr-Esfahani M, Dalirnasab Z, Dabbagh HA, Teimouri A (2007) Theoretical studies on tautomerism of tetrazole derivatives by polarisable continuum method (PCM). J Mol Struct (Theochem) 820:7–11
Ostrovskii VA, Koldobskii GI, Trifonov RE (2008) Tetrazoles. In: Katritzky AR, Ramsden CA, Scriven EFV, Taylor RJK (eds) Comprehensive heterocyclic chemistry III, vol 6. Elsevier, Oxford, pp 257–424
Murlowska K, Sadlej-Sosnowska N (2005) Absolute calculations of acidity of C-substituted tetrazoles in solutions. J Phys Chem A 109:5590–5595
Halauko YS, Matulis VE, Ivashkevich OA, Grigoriev YV, Gaponik PN (2010) DFT calculations of CH acidity of substituted triazoles and experimental study of their ability to undergo mercuration. Tetrahedron 66:3415–3420
Naik SR, Witkowski JT, Robins RK (1973) A novel route to 3(5)-fluoro-1,2,4-triazoles and 8-fluoropurines by displacement of the nitro group. J Org Chem 38:4353–4354
Zumbrunn A (1998) The first versatile synthesis of 1-alkyl-3-fluoro-1H-[1,2,4]-triazoles. Synthesis 1357–1361
Lowack RN, Weiss R (1990) In situ generation of 2,3-diaryltetrazolinylidenes: trapping experiments and ring opening to 1-cyanoazimines. J Am Chem Soc 112:333–338
Vilarrasa J, Galvez C, Calafell M (1975) Synthesis de Fluoroazoles. An Quim 71:631–632
Blackwell GB, Haszeldine RN, Taylor DR (1982) Polyhalogeno-allenes and -acetylenes. Part 15. Dipolar cycloadditions of N-phenylsydnone and aryl azides to perfluoropropadiene and perfluoropropyne. J Chem Soc, Perkin Trans 1: 2207–2210
Reck F, Zhou F, Girardot M, Kern G, Eyermann CJ, Hales NJ, Ramsay RR, Gravestock MB (2005) Identification of 4-substituted 1,2,3-trriazoles as novel oxazolidinone antibacterial agents with reduced activity against monoamine oxidase-a. J Med Chem 48:499–506
Himo F, Demko ZP, Noodleman L, Sharpless KB (2002) Mechanisms of tetrazole formation by addition of azide to nitriles. J Am Chem Soc 124:12210–12216
Lee LA, Baroody EE (1974) Carbon dioxide laser fuels. US Patent Appl US 530260 A0 19741206
Carpenter W, Haymaker A, Moore DW (1966) Fluorinated 1,2,3-triazolines. J Org Chem 31:789–792
Coats SI, Link JS, Gauthier D, Hlasta DJ (2005) Trimethylsilyl-directed 1,3-dipolar cycloaddition reactions in the solid-phase synthesis of 1,2,3-triazoles. Org Lett 7:1469–1472
Wei J, Chen J, Xu J, Cao L, Deng H, Sheng W, Zhang H, Cao W (2012) Scope and regioselectivity of the 1,3-dipolar cycloaddition of azides with methyl 2-perfluoroalkynoates for an easy, metal-free route to perfluoroalkylated 1,2,3-triazoles. J Fluorine Chem 133:146–154
Xiong Z, Qiu X-L, Huang Y, Qing F-L (2011) Regioselectivity synthesis of 5-trifluoromethyl-1,2,3-triazole nucleoside analogues via TBS-directed 1,3-dipolar cycloaddition reaction. J Fluorine Chem 132:166–174
Shen Y, Zheng J, Xin Y, Lin Y, Qi M (1995) Synthesis of perfluoroalkylated heterocyclic phosphonates. J Chem Soc Perkin Trans 1 997–999
Rigby W, Bailey PM, McCleverty JA, Maitlis PM (1979) Pentamethylcyclopentadienyl-rhodium and -iridium complexes 19. Preparation and reactions of azido-, cyanato-, thiocyanato-, nitrito-, and nitrato-rhodium complexes. J Chem Soc Dalton Trans 371–381
Lermontov SA, Shkavrov SV, Pushin AN (2000) The reaction of α, α-difluoroazides with acetylenic compounds. J Fluorine Chem 105:141–147
Wu Y-M, Deng J, Fang X, Chen Q-Y (2004) Regioselective synthesis of fluoroalkylated-1,2,3-triazoles by Huisgen cycloaddition. J Fluorine Chem 125:1415–1423
Mayot E, Gèrardin-Charbonnier C, Selve C (2005) Highly fluoroalkylated amphiphilic triazoles: regioselective synthesis and evaluation of physicochemical properties. J Fluorine Chem 126:715–720
Zhu Y-W, Yi W-B, Cai C (2011) A recyclable fluoroalkylated 1,4-disubstituted-1,2,3-triazole organocatalyst for aldol condensation of aldehydes and ketones. J Fluorine Chem 132:71–74
Francis DV, Miles DH, Mohammed AI, Read RW, Wang X (2011) Towards functional fluorous surfactants. Synthesis of hydrophilic fluorous 1,2,3-triazolylmethyl ethers and di(1,2,3-triazolylmethyl)ethers. J Fluorine Chem 132:898–906
Read RW, Wang X (2012) A structure–function study of the surface tension changes of m-xylene in the presence of fluorous 1H-1,2,3-triazoles and tetrazoles. J Fluorine Chem 135:25–32
Read RW, Wang X (2009) A conceptual design of fluorous surfactants based on a heterocyclic core, put into practice through synthesis of fluorous 1,2,3-triazoles. East-West J Math 36:247–257
Vorobyeva DV, Karimova NM, Vasilyeva TP, Osipov SN, Shchetnikov GT, Odinets IL, Röschenthaler G-V (2010) Synthesis of functionalized α-CF3-α-aminophosphonates via Cu(I)-catalyzed 1,3-dipolar cycloaddition. J Fluorine Chem 131:378–383
Vorobyeva DV, Sokolova NV, Nenajdenko VG, Peregudov AS, Osipov SN (2012) Synthesis of CF3-containing tetrapeptide surrogates via Ugi reaction/dipolar cycloaddition sequence. Tetrahedron 68:872–877
Sokolova NV, Vorobyeva DV, Osipov SN, Vasilyeva TP, Nenajdenko VG (2012) Synthesis of a-CF3-a-hydroxy acid-peptide conjugates via click chemistry. Synthesis 44:130–136
Hager C, Miethchen R, Reinke H (2000) Organofluorine compounds and fluorinating agents, part 26 New reversed nucleosides – perfluoroalkyl substituted 1,2,3-triazoles linked to D-galactose and D-altrose. J Fluorine Chem 104:135–142
Hager C, Miethchen R, Reinke H (2000) New trifluoromethyl substituted 1,2, 3-triazoles linked to D-galactose and D-gulose. J Prakt Chem 342:414–420
Peng W, Zhu S (2003) Efficient synthesis of 5-fluoroalkylated 1H-1,2,3-triazoles and application of the bromodifluoromethylated triazole to the synthesis of novel bicyclic gem-difluorinated 1H-pyrano[3,4-d][1,2,3]-triazol-4-one compounds. Tetrahedron 59:4395–4404
Peng W, Zhu S-Z (2002) 1,3-Dipolar cycloaddition of β-alkoxyvinyl trifluoromethylketones with aryl (or benzyl) azides. Synthesis of 4-trifluoroacetylated 1H-1,2,3-triazoles. J Fluorine Chem 116:81–86
Rozin YA, Leban J, Dehaen W, Nenaidenko VG, Muzalevskiy VM, Eltsov OS, Bakulev VA (2012) Regioselective synthesis of 5-trifluoromethyl-1,2,3-triazoles via CF3- directed cyclization of 1-trifluoromethyl-1,3-dicarbonyl compounds with azides. Tetrahedron 68:614–618
Zhu S-Z, He P, Zhao J-W, Cai X (2004) Reactions of fluoroalkanesulfonyl azides with vinyl ether and tetrakis(dimethylamino)ethylene. J Fluorine Chem 125:445–450
He P, Zhu S-Z (2005) Study on the reactions of fluoroalkanesulfonyl azides with indole derivatives. J Fluorine Chem 126:825–830
Ye C, Gard GL, Winter RW, Syvret RG, Twamley B, Shreeve JM (2007) Synthesis of pentafluorosulfanylpyrazole and pentafluorosulfanyl-1,2,3-triazole and their derivatives as energetic materials by click chemistry. Org Lett 9:3841–3844
Crossman JM, Haszeldine RN, Tipping AE (1973) Organosilicon chemistry. X. The reaction of and cyanogen halides to give triazoles. J Chem Soc (Dalton) 483–486
Bargamov GG, Bargamova MD (1994) Formation of 1-amino-1,2,3-triazole by the oxidation of 1,1,1,5,5,5-hexafluoro-4-(trifluoromethyl)pentane-2,3-dione dihydrazone. Izv Akad Nauk Ser Khim 1838–1839
Bargamov GG, Bargamova MD (1996) Polyfluoroalkyl- substituted 1-amino-1,2,3-triazoles. J Fluorine Chem 79:45–47
Bargamov GG, Bargamova MD (1998) Formation of 4- (1,1,1,3,3,3-hexafluo roisopropyl) – 5 – trifluoromethyl-2H–1,2,3-triazole by the oxidation of dihydrazone of 1,1,1,5,5,5 – hexafluoro- 4- trifluoromethylpentane – 2,3- dione. Russ Chem Bull 47:192
Sosnovskikh VY, Usachev BI (2002) A convenient synthesis of 4(5)-(2-hydroxyaroyl)-5(4)-trifluoromethyl-1,2,3-triazoles from 2-trifluoromethylchromones and chromen-4-imines. Mendeleev Commun 12:75–76
Furin GG, Gatilov YV, Bagryanskay IY, Zhurhgov EL (2001) Synthesis and transformations of perfluoro-2-methyl-3-azido-2-pentene. J Fluorine Chem 110:21–24
Coṣgun S, Celik Ṻ, Özden S, Tüysüz S, Bozkurt A, Gèrardin-Charbonnier C (2010) Proton conductivity properties of acid doped fluoroalkylated 1,2,3-triazole. J Fluorine Chem 131:776–779
Yagupolskii LM, Fedyuk DV, Petko KI, Troitskaya VI, Rudyk VI, Rudyuk VV (2000) N-Trihalomethyl derivatives of benzimidazole, benzotriazole and indazole. J Fluorine Chem 106:181–187
Brown HC, Cheng MT (1962) Nucleophilic аttack on the 2,5-bis(perfluoroalkyl)-1,3,4-oxadiazoles. I. Synthesis of 3,5-bis(perfluoroalky1)-1,2,4-triazoles and 4-methyl-1,2,4-triazoles. J Org Chem 27:3240–3241
Jones BG, Branch SK, Thompson AS, Brian GJ, Branch SK, Thompson AS, Threadgill MD (1996) Synthesis of a series of trifluoromethylazoles and determination of pK, of acidic and basic trifluoromethyl heterocycles by I9F NMR spectroscopy. J Chem Soc Trans 1 2685–2691
Brown HC, Gisler HJ, Cheng MT Jr (1966) Nucleophilic attack on the 2,5-bis(perfluoroalkyl)-1,3,4-oxadiazoles, II. Synthesis of perfluoroalkyl-substituted 1,2,4,5-tetrazines, 1,2-dihydro-1,2,4,5-tetrazines, and 4-amino-1,2,4-triazoles. J Org Chem 31:781–783
Vasil’eva EB, Filyakova VI, Sidorova LP, Filatov IE, Charushin VN (2005) Ambident properties of 4-substituted thiosemicarbazides in condensations with fluoroacetic acids. Russ J Org Chem 41:1522–1525
El-Sayed AM, Khodairy A (1998) Synthesis of new fused and spiroheterocycles derived from 4-amino-5-mercapto-3-trifluoromethyl-1,2,4-triazole. Phosphorus Sulfur Silicon 132:41–52
Chen M, Wang X-F, Wang S-S, Feng Y-X, Chen F, Yang C-L (2012) Synthesis, characterization and fungicidal activities of novel fluorinated 3,5-disubstituted-4H-1,2,4-triazol-4-amines. J Fluorine Chem 135:323–329
Danilkina NA, Vershilov SV, Ganina MB, Mikhailov LE, Ivin BA (2004) Synthesis of perfluoroalkyl[1,2,4]triazolo[1,3]thiazinones. Russ J Gen Chem 74:472–474
Lopyrev VA, Sidorova LP, Netsetskaya OA, Grinblat MP (1969) Reactions of hydrazides of perfluoro acids. I. Preparation of perfluoroacylamidoguanidines and their cyclization. Zh Obshch Khim 39:2525–2528
Ma C, Li J, Zhang R, Qiu L (2007) Organotin(IV) derivatives of a triazoline thione ligand. Dimeric and self-assembled supramolecular helical structures. J Mol Struct 830:1–7
Roberts R, Bradley PA, Bunnage ME, England KS, Fairman D, Fobian YM, Fox DNA, Gymer GE, Heasley SE, Molette J, Smith GL, Schmidt MA, Tones MA, Dack KN (2011) Acidic triazoles as soluble guanylate cyclase stimulators. Bioorg Med Chem Lett 21:6515–6518
Siedle AR, Webb RJ, Newmark RA, Brostrom M, Weil DA, Erickson K, Behr FE, Young VG Jr (2003) Solvolyis reactions of perfluoro-5-aza-4-nonene, C3F7–CF=N–C4F9. J Fluorine Chem 122:175–182
Kino T, Nagase Y, Ohtsuka Y, Yamamoto K, Uraguchi D, Tokuhisa K, Yamakawa T (2010) Trifluoromethylation of various aromatic compounds by CF3I in the presence of Fe(II) compound, H2O2 and dimethylsulfoxide. J Fluorine Chem 131:98–105
Rudyuk VV, Fedyuk DV, Yagupolskii LM (2004) N-Polyfluoroethyl and N-2-chlorodifluorovinyl derivatives of azoles. J Fluorine Chem 125:1465–1471
Petko KI, Sokolenko TM, Bezdudny AV, Yagupolskii LM (2005) N-(2-bromotetrafluoroethyl) derivatives of five-membered nitrogen-containing heterocycles. J Fluorine Chem 126:1342–1346
Dmowski W (2012) 1,2-Dibromotetrafluoroethane (Freon 114B2) as a building block for fluorine compounds. J Fluorine Chem 142:6–13
Garg S, Shreeve JM (2011) Trifluoromethanesulfonamide derivatives of azoles. J Fluorine Chem 132:241–243
Xue H, Gao H, Twamley B, Shreeve JM (2007) Energetic salts of 3-nitro-1,2,4-triazole-5-one, 5-nitroaminotetrazole, and other nitro-substituted azoles. Chem Mater 19:1731–1739
Xue H, Gao H, Shreeve JM (2008) Energetic polymer salts from 1-vinyl-1,2,4-triazole derivatives. J Polym Sci (A) 46:2414–2421
Tung Y-L, Lee S-W, Chi Y, Tao Y-T, Chien C-H, Cheng Y-M, Chou P-T, Peng S-M, Liu C-S (2005) Organic light-emitting diodes based on charge-neutral Os(II) emitters: generation of saturated red emission with very high external quantum efficiency. J Mater Chem 15:460–464
Cheng Y-M, Lee G-H, Chou P-T, Chen L-S, Chi Y, Yang C-H, Song Y-H, Chang S-Y, Shih P-I, Shu C-F (2008) Rational design of chelating phosphine functionalized Os(II) emitters and fabrication of orange polymer light-emitting diodes using solution process. Adv Funct Mater 18:183–194
Song Y-H, Chiu Y-C, Chi Y, Chou P-T, Cheng Y-M, Lin C-W, Lee G-H, Carty AJ (2008) Synthesis, characterization, and photophysical properties of luminescent gallium and indium complexes constructed using tridentate 6-azolyl-2,2′-bipyridine chelates. Organometallics 27:80–87
Norris WP (1962) 5-Trifluoromethyltetrazole and its derivatives. J Org Chem 27:3248–3251
Trifonov RE, Ostrovskii VA (2006) Protolytic equilibria in tetrazoles. Russ J Org Chem 42:1585–1605
Ye C, Gao H, Shreeve JM (2007) Synthesis and thermochemical properties of NF2-containing energetic salts. J Fluorine Chem 128:1410–1415
Uneyama K (1999) Chemistries of trifluoroacetimidoyl halides: preparation, reactions, and applications for the synthesis of fluorinated heterocycles. J Fluorine Chem 97:11–25
Mekni N, Baklouti A (2008) Synthesis of new 1-substituted 4-perfluoroalkyl tetrazol-5-ones. J Fluorine Chem 129:1073–1075
Huang Y, Read RW, Wang X (2010) Efficient alkylation methods for the synthesis of hybrid fluorocarbon-hydrocarbon tetrazoles as potential fluorinated surfactants. Aust J Chem 63:802–807
Jończyk A, Nawrot E, Kisielewski M (2005) Reactions of some nitrogen heterocycles with chlorodifluoromethane under conditions of phase-transfer catalysis. J Fluorine Chem 126:1587–1591
Zagipa B, Nagura H, Fuchigami T (2007) Electrolytic partial fluorination of organic compounds 89: regioselective anodic fluorination of tetrazolyl sulfides. J Fluorine Chem 128:1168–1173
Spear RJ (1984) Positional selectivity of the methylation of 5-substituted tetrazolate anions. Aust J Chem 37:2453–2568
Tselinskii IV, Mel’nikov AA, Varyagina LG, Zhigadlova IG (1983) Synthesis of N-oxymethylderivatives for 5-substituted tetrazoles. Chem Heterocycl Compd 19:341–344
Trifonov RE, Alkorta I, Ostrovskii VA, Elguero J (2004) A theoretical study of the tautomerism and ionization of 5-substituted NH-tetrazoles. J Mol Struct (Theochem) 668:123–132
Koren AO, Gaponik PN (1990) Regioselective of tetrazole and 5-substituted tetrazoles N2 alkylation by alcohols. Chem Heterocycl Compd 26:1366–1370
Koren AO, Gaponik PN, Ivashkevich OA, Kovalyova TB (1995) A new route to 1-alkyltetrazoles: via 2-tert-butyltetrazoles. Mendeleev Commun 5:10–11
Voitekhovich SV, Gaponik PN, Koren AO (1997) Selective synthesis of 2-(1-methylvinyl)tetrazoles. Mendeleev Commun 7:41–42
Gareev GA, Kirillova LP, Shulgina VM, Buzilova SR, Vologdina LV, Vereshchagin LI (1988) Reaction of some derivatives of 2-nitro-2-azapropanol with azoles. Russ J Org Chem 24:2221–2226
Gaponik PN, Ivashkevich OA, Krasitskii VA, Tuzik AA, Lesnikovich AI (2002) Water-soluble polymeric complexes of cobalt(II) and nickel(II) with azolate anions. Russ J Gen Chem 72:1457–1462
Banks RE, Prakash A (1974) Studies in azide chemistry. Part VI. Some reactions of perfluoro-azidobenzene and perfluoro-4-azidotoluene. J Chem Soc Perkin Trans 1 1365–1371
Rama Rao VVVNS, Lingaiah BPV, Yadla R, Shanthan Rao P, Ravikumar K, Swamy GYSK, Narsimuluc K (2006) Synthesis of fluorine containing N-benzyl-1,2,3-triazoles and N-methyl-pyrazoles from conjugated alkynes. J Heterocycl Chem 43:673–679
Liu S, Müller P, Takase MK, Swager TM (2011) “Click” synthesis of heteroleptic tris-cyclometalated iridium(III) complexes: Cu(I) triazolide intermediates as transmetalating. Inorg Chem 50:7598–7609
Kloss F, Köhn U, Jahn BO, Hager MD, Görls H, Schubert US (2011) Metal-free 1,5-regioselective azide–alkyne [3 + 2]-cycloaddition. Chem Asian J 6:2816–2824
Recsei C, McErlean CSP (2012) Synthesis of modified binol-phosphoramidites. Tetrahedron 68:464–480
Boddy IK, Briggs GG, Harrison RP, Jones TH, O’Mahony MJ, Marlow ID, Roberts BG, Willis RJ, Bardsley R, Reid J (1996) The synthesis and insecticidal activity of a series of 2-aryl- l,2,3-triazoles. Pest Sci 48:189–196
D’Anna F, Ferroni F, Frenna V, Guernelli S, Lanza CZ, Macaluso G, Pace V, Petrillo G, Spinelli D, Spisani R (2005) On the application of the extended Fujita–Nishioka equation to polysubstituted systems. A kinetic study of the rearrangement of several poly-substituted Z-arylhydrazones of 3-benzoyl-5-phenyl-1,2,4-oxadiazole into 2-aryl-4-benzoylamino-5-phenyl-1,2,3-triazoles in dioxane/water. Tetrahedron 61:167–178
Birchall JM, Haszeldine RN, Kemp JEG (1970) Polyfluoroarenes. XII. Some azoxy-compounds and benzotriazoles. J Chem Soc (C) 1519–1523
Heaton A, Hill M, Drakesmith F (1997) Polyhalogenonitrobenzenes and derived compounds. 5. Improved preparations of 1,2,3,4-tetrafluoro-5,6-dinitrobenzene and 3,4,5,6-tetrafluoro-1,2-phenylenediamine, and the use of the latter for the synthesis of tetrafluorobenzheterocycles. J Fluorine Chem 81:133–138
Petrova TD, Savchenko TI, Yakobson GG (1967) Formation of tetrafluorodehydrobenzene from tetrafluoroanthranilic acid. Zhurn Obshch Khim 37:1170
Watt JA, Gannon CT, Loft KJ, Dinev Z, Williams SJ (2008) Click’ Preparation of carbohydrate 1-benzotriazoles, 1,4-disubstituted, and 1,4,5-trisubstituted triazoles and their utility as glycosyl donors. Aust J Chem 61:837–846
Shi F, Waldo JP, Chen Y, Larock RC (2008) Benzyne click chemistry: synthesis of benzotriazoles from benzynes and azides. Org Lett 10:2409–2412
Zhang F, Moses JE (2009) Benzyne click chemistry with in situ generated aromatic azides. Org Lett 11:1587–1590
Liu S, Sawicki J, Driver TG (2012) Ni-Catalyzed alkenylation of triazolopyridines: synthesis of 2,6-disubstituted pyridines. Org Lett 14:3744–3747
Blanco F, Alkorta I, Elguero J, Cruz V, Abarca B, Ballesteros R (2008) [1,2,3]triazolo[1,5-a]pyridines. A theoretical (DFT) study of the ring–chain isomerization. Tetrahedron 64:11150–11158
Mennen SM, Miller SJ (2007) Development of a bio-inspired acyl-anion equivalent macrocyclization and synthesis of a trans-resorcylide precursor. J Org Chem 72:5260–5269
Vora HU, Moncecchi JR, Epstein O, Rovis T (2008) Nucleophilic carbene catalyzed synthesis of 1,2 amino alcohols via azidation of epoxy aldehydes. J Org Chem 73:9727–9731
Joann MU, DiRocco DA, Noey EL, Rovis T, Houk KN (2011) Quantum mechanical investigation of the effect of catalyst fluorination in the intermolecular asymmetric stetter reaction. J Am Chem Soc 133:11249–11254
Rong Z-Q, Jia M-Q, You S-L (2011) Enantioselective N-heterocyclic carbene-catalyzed Michael addition to α, β-unsaturated aldehydes by redox oxidation. Org Lett 13:4080–4083
Rong Z-Q, Li Y, Yang G-Q, You S-L (2011) D-Camphor-derived triazolium salts for enantioselective intramolecular stetter reactions D-camphor-derived triazolium salts for stetter reactions. Synlett 1033–1037
Jia M-Q, You S-L (2012) Desymmetrization of cyclohexadienones via D-camphor-derived triazolium salt catalyzed intramolecular Stetter reaction. Chem Commun 6363–6365
De Vreese R, D’hooghe M (2012) N-Heterocyclic carbene/brønsted acid cooperative catalysis as a powerful tool in organic synthesis. Beilstein J Org Chem 8:398–402
Henderson AP, Mutlu E, Leclercq A, Bleasdale C, Clegg W, Henderson RA, Golding BT (2002) Trapping of benzene oxide-oxepin and methyl-substituted derivatives with 4-phenyland 4-pentafluorophenyl-1,2,4-triazoline-3,5-dione. Chem Commun 1956–1957
Shaaban MR (2008) Microwave-assisted synthesis of fused heterocycles incorporating trifluoromethyl moiety. J Fluorine Chem 129:1156–1161
Khudina OG, Shchegol’kov EV, Burgart YV, Kodess MI, Kazheva ON, Chekhlov AN, Shilov GV, Dyachenko OA, Saloutin VI, Chupakhin ON (2005) Synthesis and the reactions of trifluoromethylated 1,2,3-triketones 2-(het)arylhydrazones and 4,7-dihydroazolo[5,1-c]triazines. J Fluorine Chem 126:1230–1238
Kuznetsova OA, Filyakova VI, Pashkevich KI, Ulomskii EN, Plekhanov PV, Rusinov GL, Kodess MI, Rusinov VL (2003) Fluoroalkyl containing lithium β-diketonates in the synthesis of 1,2,4-triazolo[1,5-a]pyrimidines. Russ Chem Bull Int Ed 52:1190–1194
Dolzhenko AV, Tan BJ, Dolzhenko AV, Chiu GNC, Chui WK (2008) Synthesis and biological activity of fluorinated 7-aryl-2-pyridyl-6,7-dihydro[1,2,4]triazolo[1,5-a][1,3,5]triazin-5-amines. J Fluorine Chem 129:429–434
Krasovsky AL, Moiseev AM, Nenajdenko VG, Balenkova ES (2002) Synthesis of new fluorine-containing triazolo- and tetrazolopyrimidines. Synthesis 901–905
Kim Y-J, Lee S-H, Lee S-H, Jeon SI, Lim MS, Lee SW (2005) Reactivity of alkynyl Pd(II) azido complexes toward organic isocyanides, isothiocyanates, and nitriles. Inorg Chim Acta 358:650–658
Klapцtke TM, Krumm B, Moll R (2011) Convenient room-temperature, mercury-assisted synthesis of tetrazoles by1,3-dipolar cycloaddition. Eur J Inorg Chem 422–428
Zhao Z-H, Jin H, Zhang Y-X, Shen Z, Zou D-C, Fan X-H (2011) Synthesis and properties of dendritic emitters with a fluorinated starburst oxadiazole core and twisted carbazole dendrons. Macromolecules 44:1405–1413
Cmoch P, Korczak H, Stefaniak L, Webb GA (1999) 1H, 13C and 15 N NMR and IR studies of halogen-substituted tetrazolo[1,5-a]pyridines. J Phys Org Chem 12:470–478
Keith JM (2006) One-step conversion of pyridine N-oxides to tetrazolo[1,5-a]pyridines. J Org Chem 71:9540–9543
Altomonte S, Zanda M (2012) Synthetic chemistry and biological activity of pentafluorosulphanyl (SF5) organic molecules. J Fluorine Chem 143:57–93
Martinez H, Zheng Z, Dolbier WR Jr (2012) Energetic materials containing fluorine. Design, synthesis and testing of furazan-containing energetic materials bearing a pentafluorosulfanyl group. J Fluorine Chem 143:112–122
Rostovtsev VV, Green LG, Fokin VV, Sharpless KB (2002) A stepwise Huisgen cycloaddition process: copper(I)-catalyzed regioselective ligation of azides and terminal alkynes. Angew Chem Int Ed 41:2596–2599
Huang Y, Gard GL, Shreeve JM (2010) One-pot syntheses of 1,2,3-triazoles containing a pentafluorosulfanylalkyl group via click chemistry. Tetrahedron Lett 51:6951–6954
Politzer P, Lane P (1997) Computational studies of structures and properties of energetic difluoramines. Adv Mol Struct 3:269–285
Ostrovskii VA, Pevzner MS, Kofman TP, Shcherbinin MB, Tselinskii IV (1999) Energetic 1,2,4-triazoles and tetrazoles. Targets Heterocycl Syst 3:467–526
Dalinger IL, Vinogradov VM, Shevelev SA, Kuz’min VS (1996) N-(Difluoroamino)azoles – a new class of N-substituted azoles. Mendeleev Commun 6:13–15
Hagmann WK (2008) The many roles for fluorine in medicinal chemistry. J Med Chem 51:4359–4369
Ismail FMD (2002) Important fluorinated drugs in experimental and clinical use. J Fluorine Chem 118:7–33
Lyseng-Williamson KA (2007) Sitagliptin. Drugs 67:587–597
Desai AA (2011) Sitagliptin manufacture: a compelling tale of green chemistry, process intensification, and industrial asymmetric catalysis. Angew Chem Int Ed 50:1974–1976
Clausen AM, Dziadul B, Cappuccio KL, Kaba M, Starbuck C, Hsiao Y, Dowling TM (2006) Identification of ammonium chloride as an effective promoter of the asymmetric hydrogenation of α, β-enamine amide. Org Proc Res Dev 10:723–726
Kim D, Kowalchick JE, Edmondson SD, Mastracchio A, Xu J, Eiermann GJ, Leiting B, Wu JK, Pryor KD, Patel RA, He H, Lyons KA, Thornberry NA, Weber AE (2007) Triazolopiperazine-amides as dipeptidyl peptidase IV inhibitors: close analogs of JANUVIATM (sitagliptin phosphate). Bioorg Med Chem Lett 17:3373–3377
Kim D, Kowalchick JE, Brockunier LL, Parmee ER, Eiermann GJ, Fisher MH, He H, Leiting B, Lyons K, Scapin G, Patel SB, Petrov A, Pryor K-AD, Roy RS, Wu JK, Zhang X, Wyvrat MJ, Zhang BB, Zhu L, Thornberry NA, Weber AE (2008) Discovery of potent and selective dipeptidyl peptidase IV inhibitors derived from β-aminoamides bearing substituted triazolopiperazines. J Med Chem 51:589–602
Biftu T, Scapin G, Singh S, Feng D, Becker JW, Eiermann G, Huaibing H, Lyons K, Patel S, Petrov A, Sinha-Roy R, Zhang B, Wu J, Zhang X, Doss GA, Thornberry NA, Weber AE (2007) Rational design of a novel, potent, and orally bioavailable cyclohexylamine DPP-4 inhibitor by application of molecular modeling and X-ray crystallography of sitagliptin. Bioorg Med Chem Lett 17:3384–3387
Shan Z, Peng M, Fan H, Lu Q, Lu P, Zhao C, Chen Y (2011) Discovery of potent dipeptidyl peptidase IV inhibitors derived from β-aminoamides bearing substituted [1,2,3]-triazolopiperidines for the treatment of type 2 diabetes. Bioorg Med Chem Lett 21:1731–1735
Wang B-L, Liu X-H, Zhang X-L, Zhang J-F, Song H-B, Li Z-M (2011) Synthesis, structure and biological activity of novel 1,2,4-triazole mannich bases containing a substituted benzylpiperazine moiety. Chem Biol Drug Des 78:42–49
Ostrovskii VA, Trifonov RE, Popova EA (2012) Medicinal chemistry of tetrazoles. Russ Chem Bull Int Ed 61:768–780
Faidallah HM, Khan KA, Asiri AM (2011) Synthesis and biological evaluation of new 3,5-di(trifluoromethyl)-1,2,4-triazolesulfonyl urea and thiourea derivatives as antidiabetic and antimicrobial agents. J Fluorine Chem 132:870–877
Faridoon, Hussein WM, Vella P, Islam NU, Ollis DL, Schenk G, McGeary RP (2012) 3-mercapto-1,2,4-triazoles and N-acylated thiosemicarbazidesas metallo-β-lactamase inhibitors. Bioorg Med Chem Lett 22:380–386
De La Rosa M, Kim HW, Gunic E, Jenket C, Boyle U, Koh Y, Korboukh I, Allan M, Zhang W, Chen H, Xu W, Nilar S, Yao N, Hamatake R, Lang SA, Hong Z, Zhang Z, Girardet J-L (2006) Tri-substituted triazoles as potent non-nucleoside inhibitors of the HIV-1 reverse transcriptase. Bioorg Med Chem Lett 16:4444–4449
Kanishchev OS, Gudz GP, Shermolovich YG, Nesterova NV, Zagorodnya SD, Golovan AV (2011) Synthesis and biological activity of the nucleoside analogs based on polyfluoroalkyl-substituted 1,2,3-triazoles. Nucleosides Nucleotides Nucleic Acids 30:768–783
Yagupolskii LM, Maletina II, Petko KI, Fedyuk DV, Handrock R, Shavaran SS, Klebanov BM, Herzig S (2001) New fluorine-containing hypotensive preparations. J Fluorine Chem 109:87–94
Lebsack AD, Gunzner J, Wang B, Pracitto R, Schaffhauser H, Santini A, Aiyar J, Bezverkov R, Munoz B, Liuc W, Venkatramana S (2004) Identification and synthesis of [1,2,4]triazolo[3,4-a]phthalazine derivatives as high-affinity ligands to the α2δ-1 subunit of voltage gated calcium channel. Bioorg Med Chem Lett 14:2463–2467
Williams BJ, Cascieri MA, Chicchi GG, Andrew TH, Owens P, Owen SN, Rupniak NMJ, Tattersall DF, Williams A, Swain CJ (2002) Spirocyclic NK1 antagonists II: [4.5]-spiroethers. Bioorg Med Chem Lett 12:2719–2722
Shirai J, Yoshikawa T, Yamashita M, Yamamoto Y, Kawamoto M, Tarui N, Kamo I, Hashimoto T, Ikeura Y (2011) Design, structure–activity relationship, and highly efficient asymmetric synthesis of 3-phenyl-4-benzylaminopiperidine derivatives as novel neurokinin-1 receptor antagonists. Bioorg Med Chem 19:6430–6446
Micheli F, Bonanomi G, Blaney FE, Braggio S, Capelli AM, Checchia A, Curcuruto O, Damiani F, Di Fabio R, Donati D, Gentile G, Gribble A, Hamprecht D, Tedesco G, Terreni S, Tarsi L, Lightfoot A, Stemp G, MacDonald G, Smith A, Pecoraro M, Petrone M, Perini O, Piner J, Rossi T, Worby A, Pilla M, Valerio E, Griffante C, Mugnaini M, Wood M, Scott C, Andreoli M, Lacroix L, Schwarz A, Gozzi A, Bifone A, Ashby CR Jr, Hagan JJ, Heidbreder C (2007) 1,2,4-triazol-3-yl-thiopropyl-tetrahydrobenzazepines: a series of potent and selective dopamine D3 receptor antagonists. J Med Chem 50:5076–5089
Sugane T, Tobe T, Hamaguchi W, Shimada I, Maeno K, Miyata J, Suzuki T, Kohara A, Kimizuka T, Morita T, Doihara H, Saita K, Aota M, Furutani M, Shimada Y, Hamada N, Sakamoto S, Tsukamoto S-I (2011) Synthesis and biological evaluation of 3-biphenyl-4-yl-4-phenyl-4H-1,2,4-triazoles as novel glycine transporter 1 inhibitors. J Med Chem 54:387–391
Hale JJ, Mills SG, MacCoss M, Finke PE, Cascieri MA, Sadowski S, Ber E, Chicchi GG, Kurtz M, Metzger J, Eiermann G, Tsou NN, Tattersall FD, Rupniak NMJ, Williams AR, Rycroft W, Hargreaves R, MacIntyre DE (1998) Structural optimization affording 2-(R)-(1-(R)-3,5-bis(trifluoromethyl)phenylethoxy)-3-(S)-(4-fluoro)phenyl-4-(3-oxo-1,2,4-triazol-5-yl)methylmorpholine, a potent, orally active, long-acting morpholine acetal human NK-1 receptor antagonist. J Med Chem 41:4607–4614
Aiguadé J, Balagué C, Carranco I, Caturla F, Dominguez M, Eastwood P, Esteve C, González J, Lumeras W, Orellana A, Preciado S, Roca R, Vidal L, Vidal B (2012) Novel triazolopyridylbenzamides as potent and selective p38a inhibitors. Bioorg Med Chem Lett 22:3431–3436
Hamill TG, Burns HD (2004) The synthesis of tritium labelled neurokinin-1 receptor ligands. J Label Compd Radiopharm 47:099–106
Chin FT, Morse CL, Shetty HU, Pike VW (2006) Automated radiosynthesis of [18 F] SPA-RQ for imaging human brain NK1 receptors with PET. J Label Compd Radiopharm 49:17–31
Zhang Y, Huang F, Chi Y, Jen AKY (2008) Highly efficient white polymer light-emitting diodes based on nanometer-scale control of the electron injection layer morphology through solvent processing. Adv Mater 20:1565–1570
Cheng Y-M, Li EY, Lee G-H, Chou P-T, Lin S-Y, Shu C-F (2007) Strategic design and synthesis of osmium(II) complexes bearing a single pyridyl azolate π-chromophore: achieving high-efficiency blue phosphorescence by localized excitation. Inorg Chem 46:10276–10286
Chien C-H, Hsu F-M, Shu C-F, Chi Y (2009) Efficient red electrophosphorescence from a fluorene-based bipolar host material. Org Electron 10:871–876
Mak CSK, Hayer A, Pascu SI, Watkins SE, Holmes AB, Kцhler A, Friend RH (2005) Blueto-Green electrophosphorescence of iridium-based cyclometallated materials. Chem Commun 4708–4710
Yang C-H, Li S-W, Chi Y, Cheng Y-M, Yeh Y-S, Chou P-T, Lee G-H, Wang C-H, Shu C-F (2005) Heteroleptic cyclometalated iridium(III) complexes display blue phosphorescence in solution and solid state at room temperature. Inorg Chem 44:7770–77780
Hung J-Y, Lin C-H, Chi Y, Chung M-W, Chen Y-J, Lee G-H, Chou P-T, Chen C-C, Wu C-C (2010) Phosphorescent Ir(III) complexes bearing double benzyldiphenylphosphine cyclometalates; strategic synthesis, fundamental and integration for white OLED fabrication. J Mater Chem 20:7682–7693
Yang C, Kaipa U, Mather QZ, Wang X, Nesterov V, Venero AF, Omary MA (2011) Fluorous metal organic frameworks with superior adsorption and hydrophobic properties toward oil spill cleanup and hydrocarbon storage. J Am Chem Soc 133:18094–18097
Zorn DD, Boatz JA, Gordon MS (2006) Electronic structure studies of tetrazolium-based ionic liquids. J Phys Chem B 110B:11110–11119
Price SC, Stuart AC, Yang L, Zhou H, You W (2011) Fluorine substituted conjugated polymer of medium band gap yields 7 % efficiency in polymer-fullerene solar cells. J Am Chem Soc 133:4625–4631
Hsieh H-Y, Lin C-H, Tu G-M, Chi Y, Lee G-H (2009) Platinum(II) complexes with spatially encumbered chelates, syntheses, structure and photophysics. Inorg Chim Acta 362:4734–4739
Fang C-H, Chen Y-L, Yang C-H, Chi Y, Yeh Y-S, Li EY, Cheng Y-M, Hsu C-J, Chou P-T, Chen C-T (2007) Iridium(I) pyridyl azolate complexes with saturated Red metal-to-ligand charge transfer phosphorescence; fundamental and potential applications in organic light-emitting diodes. Chem Eur J 13:2686–2694
Wu K-L, Ho S-T, Chou C-C, Chang Y-C, Pan H-A, Chi Y, Chou P-T (2012) Engineering of osmium(II)-based light absorbers for dye-sensitized solar cells. Angew Chem Int Ed 51:5642–5646
Crawford M-J, Klapotke TM, Radies H (2008) Synthesis and characterization of perfluorinated nitriles and the corresponding sodium 5-perfluoroalkyltetrazolate salts. J Fluorine Chem 129:1199–1205
Koch E-C, Klapotke TM, Radies H, Lux K, Hahma A (2011) Metal-forocarbon prolants. XII: Calcium salts of 5-prfluoroalkylated tetrazoles –synthesis, characterization and performance evaluation as oxidizers in ternary mixtures with magnesium and Viton. Z Naturforsch 66b:378–386
Koch E-C, Hahma A, Klapotke TM, Radies H (2010) Metal – fluorocarbon pyrolants: XI. Radiometric performance of pyrolants based on magnesium, perfluorinated tetrazolates, and Viton A. Propell Explos Pyrotech 35:248–253
Garg S, Shreeve JM (2011) Trifluoromethyl- or pentafluorosulfanyl-substituted poly-1,2,3-triazole compounds as dense stable energetic materials. J Mater Chem 21:4787–4795
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Ostrovskii, V.A., Trifonov, R.E. (2014). Fluorinated Triazoles and Tetrazoles. In: Nenajdenko, V. (eds) Fluorine in Heterocyclic Chemistry Volume 1. Springer, Cham. https://doi.org/10.1007/978-3-319-04346-3_11
Download citation
DOI: https://doi.org/10.1007/978-3-319-04346-3_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-04345-6
Online ISBN: 978-3-319-04346-3
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)