Abstract
Neuropsychiatric consequences, encompassing mood disorders and cognitive impairments, figure prominently among the ripple effects of traumatic brain injury (TBI). Biomarkers show promise in predicting and monitoring neuropsychiatric outcomes post TBI. This chapter explores a range of biomarkers in blood, cerebral spinal fluid (CSF), and neuroimaging, with notable contenders like GFAP and BDNF capable of identifying individuals with post-TBI neuropsychiatric changes, providing intriguing insights into the brain’s response to injury. Blood biomarkers such as Tau protein and GFAP showcase associations with cognitive impairment, TBI severity, and unfavorable cognitive outcomes. Further, neuroimaging techniques bring to light structural and functional changes in the brain following TBI. Exploring the strengths of varied neuroimaging modalities underscores the capacity to detect and typify neuropsychiatric symptoms and illnesses, forecast outcomes, and evaluate treatment responses. Despite advancements, noteworthy gaps and hurdles persist, including issues concerning the reliability and specificity of the ascertained biomarkers, uniformity of measurement methodologies, and the intrinsic molecular and cellular mechanisms contributing to these biomarkers’ ties with psychiatric disorders. Future inquiries should aim to refine and validate recognized biomarkers, uncover new ones, and craft personalized, targeted interventions for psychiatric conditions following TBI.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The interconnection between traumatic brain injury (TBI) and neuropsychiatric diseases finds its roots back in 1848 with the case of Phineas Gage, a railroad construction foreman who survived a severe TBI when an iron rod pierced his skull and brain [1]. The remarkable changes in Gage’s personality and behavior following the injury overshadowed his survival, providing an impetus for future explorations into the relationship between TBI and behavioral alterations [2].
Biomarkers serve as measurable indicators of biological processes and may provide instrumental tools to clarify the pathophysiology of a wide range of clinical symptoms and signs—including post-TBI neuropsychiatric symptoms. Such biomarkers potentially identify at-risk individuals, enhance diagnostic precision, and monitor disease progression and treatment response [3]. The field has seen exceptional advancements in recent years in identifying and validating biomarkers for post-TBI neuropsychiatric conditions, paving the path toward more personalized and targeted treatment approaches.
This chapter explores the present understanding of biomarkers for neuropsychiatric symptoms related to TBI. It discusses the primary sources of potential biomarkers, including blood and cerebral spinal fluid (CSF) (Fig. 1), and examines the challenges and opportunities with their clinical implementation. It also probes into the implications of biomarker-guided approaches for the management of post-TBI neuropsychiatric sequelae, as well as future research prospects in this field.
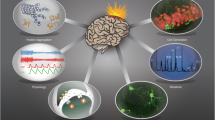
Potential blood and cerebrospinal fluid (CSF) biomarkers of traumatic brain injury (TBI)-associated neuropsychiatric disorders. Growing evidence has pointed out an association between blood levels of Neurofilament light chain (NFL), phos tau (p-tau), Glial Fibrillary Acidic Protein (GFAP), Ubiquitin C-Terminal Hydrolase L1 (UCHL-1), Interleukin-6 (IL-6), Tumor Necrosis Factor Alpha (TNF-α) and Brain-derived neurotrophic factor (BDNF) and TBI-related neuropsychiatric symptoms like anxiety, depression, post-traumatic stress disorder (PTSD) and cognitive deficits in the military population. In the general population, blood concentrations of soluble CD40L (sCD40L), Cathepsin-D, Interleukin-4 (IL-4), Neuropilin-1, Interferon alpha-2 (IFNα2) and Copeptin have been associated with impulsivity following mild TBI while the Interleukins IL-2 and IL-10 have been associated with depressive symptoms after a brain injury. Moreover, CSF levels of interleukins IL-7 and IL-8, soluble vascular cell adhesion molecule-1 (sVCAM-1), soluble intercellular adhesion molecule-1 (sICAM-1) as well as of the soluble form of Fas (sFas), an apoptosis-signaling receptor molecule, have been reported as potential biomarkers of depression secondary to TBI. (Created with biorender.com)
2 Neuropsychiatric and Cognitive Symptoms Associated with TBI
Clinically, a diverse landscape of neuropsychiatric syndromes and disorders – depression, anxiety, bipolar disorder, impulsivity, cognitive deficits, apathy, anhedonia, and aggressive behaviors – may manifest after TBI. Neuropsychiatric symptoms stemming from TBI can display persistent characteristics from months to years subsequent to the inciting injury. An intricate interplay of factors contributes to the severity and duration of these symptoms, key among them being the magnitude of the injury, the topographical location of the lesion, and any psychiatric history predating the injury. An enriched comprehension of the neurobiological apparatus that underscores these symptoms holds the potential to navigate the formulation of precisely targeted interventions, thereby amplifying the life quality of those affected with TBI [4].
2.1 Depression
Among the psychiatric disorders ensuing from TBI, depression is the most common, with incidence rates oscillating between 25% and 50% within the initial year post injury [5, 6]. The onset of depression is often insidious, materializing several months after the primary injury [7]. While the pathophysiological mechanisms underlying TBI-induced depression remain to be fully discerned, researchers have postulated the involvement of monoamine neurotransmitter alterations, neuroinflammation, and injury to frontal and limbic structures [8].
2.2 Anxiety Disorders
TBI patient often grapple with anxiety disorders, a spectrum that includes generalized anxiety disorder, panic disorder, and phobias. The prevalence rates for post-TBI anxiety disorders oscillate between 11% and 37% [9]. It has been reported that a previous history of psychiatric conditions may increase the risk for the development of anxiety disorders following a TBI [7]. While the neurobiological basis of post-TBI-associated anxiety disorders remains to be fully clarified, a potential involvement of amygdala and prefrontal cortex damage has been proposed [10].
2.3 Posttraumatic Stress Disorder
PTSD is a mental health condition characterized by reexperiencing symptoms, avoidance, and alterations in arousal, cognition, and mood, triggered by a terrifying situation — either experiencing it or witnessing it. The increasing interest in investigating PTSD following TBI relies on the fact that the events that cause TBI can be often emotionally traumatic. Moreover, there is evidence that the physical brain damage resulting from a TBI might be an important risk factor for PTSD. Although the mechanisms underlying TBI-associated PTSD remain unclear, the physical impairment of neural circuits may hamper the regulation of fear responses, coping skills, and use of adaptive cognitive strategies contributing to PTSD [11, 12]. Higher rates of PTSD have been reported following TBI in military personnel varying from 27.3% to 43.9%. This might be explained, at least in part, by the fact that soldiers, apart from the physical trauma of TBI, often experience repeated psychological trauma through combat exposures [12]. Among civilians affected by TBI, the PTSD rates vary from 2.6% to 36% [13].
2.4 Bipolar Disorder and Mania
The incidence of bipolar disorder and mania among TBI patients is relatively modest, presenting prevalence rates spanning 1.7–9% [14]. The likelihood of post-TBI mania development increases with the presence of risk factors such as a familial history of bipolar disorder and lesions localized in the right hemisphere, particularly the orbitofrontal and basotemporal regions [14]. The post-TBI mania pathophysiology has been associated with dysregulated monoamine neurotransmitter systems, with dopamine playing a notable role [14].
2.5 Cognitive Impairment
TBI frequently causes cognitive impairments, a prevalent neuropsychiatric outcome that disrupts various cognitive domains spanning from attention and memory to executive function and processing speed. Tsai et al. (2021) demonstrated that mild TBI initially triggered memory and attention deficits in 31% and 20% of cases, respectively. This incidence slightly decreased in the subacute phase to 26% and 18%, respectively. Moderate-to-severe TBI exhibited a more pronounced subacute impact, with 49% experiencing memory deficits and 54% attention deficits. In the chronic phase, however, memory deficits reduced to 21%, while attention deficits remained high at 50% [15].
The intensity and duration of such cognitive impairment generally correlate with the severity of the trauma. This can result in a spectrum of effects: Mild TBI frequently triggers mild cognitive disturbances, whereas injuries of a higher magnitude often lead to enduring cognitive dysfunction [16]. The inflicted structural harm to the frontal and temporal lobes, coupled with widespread axonal injury, serves as a contributing factor to the cognitive deficits that ensue post TBI [16]. Associated with these structural changes, neuroinflammatory mechanisms, disruptions of neurotransmitter systems, such as the cholinergic and glutamatergic systems, also contribute to post-TBI cognitive impairment [4].
2.6 Apathy and Anhedonia
TBI regularly precipitates apathy, a state often delineated by diminished motivation, emotional numbness, and a reduction in goal-oriented behaviors. Prevalence estimates for this neuropsychiatric outcome ranges from 10.84% (without depression) to 60% (with depression) [17]. Moreover, TBI patients frequently exhibit anhedonia, a condition marked by the loss of capacity to derive satisfaction from activities traditionally deemed pleasurable. Both apathy and anhedonia are related to lesions of the frontal-subcortical circuits, with particular emphasis on the prefrontal and anterior cingulate regions and parieto-subcortical circuits [18].
2.7 Aggression
TBI is frequently associated with aggression, with its prevalence estimated to be in the range of 37–71% [19, 20]. The manifestation of this post-TBI syndrome often encompasses both verbal and physical aggression, coupled with a decrease in frustration tolerance and an increase in irritability. Post-TBI aggression is linked to cortical thinning of the orbitofrontal regions [21]. Beyond structural brain changes, researchers discern alterations in neurotransmitter systems as potential contributors to these behavioral transformations. Notably, they emphasize changes in the functioning of glutamate and cholinergic systems [22].
2.8 Impulsivity
Impulsivity is a multifaceted concept that is defined as a tendency to react rapidly without forethought of future negative consequences for oneself or others. It is a common complication following moderate to severe TBI [23]. There is evidence that approximately 35–38% of patients will present motor impulsivity, also known as response disinhibition, during acute recovering post TBI [24]. Impulsive behaviors map to dysfunctions in the frontal lobe, a cerebral area pivotal to the orchestration of decision-making and impulse control [25]. Hence, unraveling the intricacies of post-TBI impulsivity stands as a vital imperative for sculpting effective and tailored rehabilitation strategies.
3 TBI-Related Biomarkers for Neurobehavioral Symptoms and Disorders
3.1 Blood Biomarkers
Researchers are currently exploring the viability of blood biomarkers as tools of detection and progress monitoring to understand TBI’s subsequent behavioral and cognitive repercussions. In particular, molecular variations in TBI patients’ blood samples – cytokines, chemokines, and other biological markers could act as revealing signposts of TBI-related psychiatric disorders (Fig. 1).
Peltz et al. (2020) assessed neurofilament light chain (NFL), total tau, glial fibrillary acidic protein (GFAP), α-synuclein, β-amyloid 42 (Aβ42), phosphorylated tau (p-tau), along with cytokines like tumor necrosis factor–α (TNF-α) and interleukin-6 (IL-6) in serum and plasma of older veterans with a TBI history [26]. They observed discernable patterns of these markers when mapped against different post-TBI cognitive groups. These cognitive groups were defined by a set of assessment tools, including the Mini-Mental State Examination [MMSE], Auditory Verbal Learning Test [AVLT] Learning Trials, AVLT Delayed Recall, and Wechsler Adult Intelligence Scale [WAIS] Digit Symbol tests [26]. When comparing the TBI group with cognitive deficits and controls or the TBI group without cognitive impairment, p-tau, NFL, GFAP, IL-6, and TNF-α were increased. The cumulative integration of these markers wielded the power to differentiate the post-TBI cognitive groups with remarkable accuracy, boasting an area under the curve [AUC] score of 0.85 [26].
Lange et al. (2021) examined the correlation between serum biomarkers and neurobehavioral changes post-military-related TBI across a spectrum of injury severities and non-injured controls [27]. They identified tau, neurofilament light chain (NFL), glial fibrillary acidic protein (GFAP), and ubiquitin carboxy-terminal hydrolase L1 (UCHL-1) as crucial biomarkers. Intriguingly, increasing tau, NFL, and GFAP levels signaled worsening symptoms like anxiety, PTSD, and depression, among others [27]. UCHL-1 predicted an escalation in anxiety, somatic, and neurological symptoms [27]. In noninjured controls, however, these biomarkers did not correlate with symptom deterioration. This suggests that elevated levels of tau, NFL, GFAP, and UCHL-1 within the first year post injury might predict a persistent decline in neurobehavioral symptoms [27].
Vedantam et al. (2021) gathered data from patients diagnosed with mild traumatic brain injury (mTBI) and controls exhibiting orthopedic injuries (OI) across three trauma centers. The team drew blood samples from the patients within 24 h following the injury and then again 6 months later, providing them with measurements of plasma inflammatory cytokines. Neuropsychological tests took place at various intervals after the injury [28]. The study, involving mainly male participants associated with vehicle accidents, included 53 mTBI patients and 24 OI controls [28]. An early surge in plasma IL-2 levels showed a link with an escalation in post-concussive symptoms a week later. Moreover, 6 months after the injury, a spike in plasma IL-10 levels corresponded with more severe depression and PTSD symptoms [28]. These results underscore how inflammation and cytokine levels can influence the severity of post-concussive symptoms, PTSD, and depression following a mTBI [28].
Cardoso et al. (2023) employed machine learning–based modeling to unearth potential biomarkers related to impulsivity following mTBI. They evaluated 21 mTBI patients within a month post injury and compared these findings to data from 19 healthy controls, using measures of impulsivity, executive functioning, episodic memory, self-reported cognitive failures, and blood biomarkers indicative of inflammation, vascular damage, and neuronal deterioration [23]. mTBI patients manifested significantly higher impulsivity than controls, both in terms of the overall Barratt Impulsiveness Scale (BIS) score and its subscales. Intriguingly, specific biomarkers, including sCD40L, Cathepsin D, IL-4, Neuropilin-1, IFN-α2, and Copeptin, showed associations with increased impulsivity in mTBI patients [22]. Hence, Cardoso et al. (2023) both validate the link between mTBI and elevated impulsivity in non-military populations and reveal novel pathophysiological pathways potentially implicated in mTBI-related impulsivity [23].
In the cross-sectional cohort study by Pattinson et al. (2020), researchers established a positive association between tau concentrations and symptom severity in military personnel and veterans, regardless of their TBI history. More specifically, significant correlations emerged between tau and specific subscales of the Neurobehavioral Symptom Inventory (NSI) for post-concussive symptoms, the PTSD Checklist self-report measure (PCL), and the patient health questionnaire (PHQ-9) for depressive symptoms [29]. Pattinson et al. (2020) also noted elevated NFL levels in individuals subjected to repetitive TBI. These findings suggest blood levels of tau and NFL as potential markers of persistent neurological and behavioral symptoms following TBI. These observations underscore the critical role of chronic biomarker measurements and indicate the need for future longitudinal studies following TBI [29].
Drestch et al. (2016) noted that the BDNF Val66 Met genotype significantly linked to the risk of sustaining mTBI and screening positive for traumatic stress. Predeployment traumatic stress, combat exposure, mTBI during deployment, and the BDNF Met/Met genotype collectively explained 22% of the variance in postdeployment PTSD scores [30]. However, predeployment traumatic stress alone accounted for 17% of the scores. These findings indicate that predeployment traumatic stress, genetics, and environmental factors play distinct roles in the development of combat-related traumatic stress among military service members [30].
Table 1 summarizes the association between blood biomarkers and behavioral/cognitive changes post-TBI across different studies.
Blood biomarkers carry immense promise in pinpointing individuals susceptible to neuropsychiatric and behavioral shifts following a TBI. The reviewed studies underscore the correlation of certain biomarkers, such as GFAP, with post-TBI cognitive impairment and PTSD. Despite these advancements, the role of blood biomarkers in tracking and predicting neuropsychiatric changes post TBI remains partially understood. The mechanisms that form these associations demand further investigation.
3.2 Cerebral Spinal Fluid Biomarkers
The CSF of people with TBI hosts an array of molecules, such as neurotransmitters, cytokines, and chemokines, among others, that can inform about clinical outcomes [31]. Fluctuations in the concentrations of these molecular components could serve as an informative gauge, potentially pinpointing individuals prone to post-TBI psychiatric disorders (Fig. 1) [31]. The notion of employing such markers not only for identification but also for progress monitoring, poses an avenue for future research.
Juengst et al. (2015) investigated the capacity of acute inflammation profiles as predictive factors for posttraumatic depression (PTD) risk within 6–12 months post traumatic brain injury [32]. Drawing from a prospective cohort design, they studied adults with moderate to severe traumatic brain injury from a university-associated level 1 trauma center, concentrating on participants with available acute serum and CSF levels [32]. By using the Patient Health Questionnaire-9 (PHQ-9) and an array of inflammatory biomarkers – including IL-1β, IL-4, IL-5, IL-6, IL-7, IL-8, IL-10, IL-12, TNF-α, sVCAM-1, sICAM-1, and sFAS – they found that elevated levels of CSF cytokine surface markers (sVCAM-1, sICAM-1, and sFAS) significantly increased PTD risk [32]. Notably, exceeding the 75th percentile in sICAM-1, sVCAM-1, or sFAS values elevated the likelihood of PTD risk at 6 months to 85.7%. Beyond this, the investigators discerned a potential link between inflammatory biomarkers IL-7 and IL-8 and PTD risk at the 12-month mark [32].
While CSF biomarkers can potentially indicate long-term behavioral alterations due to TBI, their practical usage is limited [32]. The cost, invasive nature, and lack of availability in certain medical settings curtail the clinical feasibility of CSF sampling.
3.3 Neuroimaging Biomarkers
Different techniques, namely, magnetic resonance imaging (MRI), its functional variant (fMRI), positron emission tomography (PET), and magnetoencephalography (MEG), have emerged as invaluable investigative tools in TBI. Their coordinated application provides critical insights by identifying, delineating, and quantifying both structural and functional changes underlying brain dysfunction and, therefore, psychiatric manifestations.
Medeiros et al. (2022) reviewed potential neuroimaging biomarkers for post TBI. Combing through four databases, they curated 38 articles from 2035 citations, representing a diverse pool of 1793 subjects [33]. Predominantly, these studies used structural MRI and unveiled an intriguing correlation: Post-TBI depression links with reduced gray matter and increased white matter damage [33]. Consistencies emerged in gray matter reductions in specific regions like the rostral anterior cingulate cortex, pre-frontal cortex, and hippocampus, along with damage in five crucial white matter tracts including cingulum, internal capsule, superior longitudinal fasciculi, and anterior and posterior corona radiata [33]. This investigation, though not pinpointing a definitive neuroimaging biomarker for post-TBI depression, spotlights potential research paths, thus contributing significantly to the understanding of post-TBI depression.
Using fMRI, Raji et al. (2015) showed that TBI repercussions entailed diminished default mode network (DMN) connectivity as opposed to a healthy control group, whereas PTSD exhibited an opposite trend with enhanced DMN connectivity compared to both the control group and TBI cohort [34]. Another fMRI study by Mišić et al. (2016) revealed a marked reduction in neural activity variability, implying a potential constraining effect of PTSD on the brain dynamic range of neural activity after TBI [35].
A study by Todd et al. 2015, which employed MEG to probe the neural receptivity to combat-related cues in military personnel diagnosed with PTSD, observed an amplified neural response in these individuals when confronted with such stimuli, contrasting with their unaffected peers [36]. This implies a potential hypersensitivity within the PTSD-associated neural network to trauma-related instigators [36]. Nathan et al. confirmed persistent hyperconnectivity in emotion regulation in PTSD patients post TBI. Their research, informed by the application of FDG-PET, revealed that increased severity of PTSD symptoms was associated with heightened connectivity in the middle frontal, parahippocampal, and precuneus regions in military patients with TBI [37]. The heightened connectivity among these regions can be seen as a potential PTSD biomarker during post-TBI convalescence.
Essentially, exploiting neuroimaging techniques has allowed researchers to delve into the realms of structural and functional anomalies embedded within the brain, mapping critical connections between TBI and ensuing psychiatric disorders like PTSD. Pioneering studies have not only unveiled the stark differences in DMN connectivity between PTSD, TBI, and healthy individuals but have also exposed the potentially restricting influence of PTSD on neural activity variability. MEG, with its increasing prominence, has uncovered the hyperreactivity of PTSD-afflicted neural networks to combat-related cues, and the persistent emotional hyperconnectivity in PTSD patients post TBI, indicating a potential PTSD biomarker. While structural MRI studies have linked post-TBI depression to gray matter reductions and white matter damage, they haven’t identified a definitive neuroimaging biomarker. Nonetheless, they offer valuable leads for future research pathways.
4 Conclusion
The investigation and understanding of biomarkers, including CSF, blood biomarkers, and neuroimaging biomarkers, for psychiatric disorders following TBI are critical in advancing diagnosis, prognosis, and treatment.
The field of post-TBI psychiatric disorders has made significant advancements, but gaps and challenges persist. Longitudinal studies on a large scale are crucial to determine the reliability and specificity of CSF and blood biomarkers, such as BDNF and GFAP, for psychiatric conditions, particularly PTSD. These studies should consider factors like age, gender, ethnicity, and TBI severity to establish biomarkers’ generalizability and external validity in real-world settings. Understanding the underlying molecular and cellular mechanisms, exploring biomarker interactions, and standardizing measurement methods are essential. Integrating fluid-based biomarkers with neuroimaging findings can provide a comprehensive understanding, enhance diagnostics, and aid in developing targeted interventions. Longitudinal neuroimaging studies are also essential to track structural and functional changes. Additionally, Gotshall et al. (2021) discovered significant associations between sleep quality and chronic inflammation in chronic mTBI patients, indicating that sleep-focused interventions could potentially regulate inflammatory processes and impact neuropsychological outcomes [38]. Further research is needed to explore the bidirectional relationship between sleep and inflammation following mTBI and determine the therapeutic implications of addressing sleep disorders in these patients [38]. In sum, future perspectives should consider refining already identified biomarkers, discovering new ones, and personalized treatments for psychiatric disorders after TBI.
Studying biomarkers for post-TBI psychiatric disorders, including CSF, blood, and neuroimaging biomarkers, poses significant potential for improving the quality of life for affected individuals. Future research will contribute to a more comprehensive understanding of TBI-related psychiatric disorders and ultimately pave the way for better diagnosis, prognosis, and treatment options.
References
Harlow JM. Passage of an iron rod through the head. 1848. J Neuropsychiatry Clin Neurosci. 1999;11(2):281–3. https://doi.org/10.1176/jnp.11.2.281.
Damasio H, Grabowski T, Frank R, Galaburda AM, Damasio AR. The return of Phineas Gage: clues about the brain from the skull of a famous patient. Science (New York, NY). 1994;264(5162):1102–5. https://doi.org/10.1126/science.8178168.
Strimbu K, Tavel JA. What are biomarkers? Curr Opin HIV AIDS. 2010;5(6):463–6. https://doi.org/10.1097/COH.0b013e32833ed177.
Masel BE, DeWitt DS. Traumatic brain injury: a disease process, not an event. J Neurotrauma. 2010;27(8):1529–40. https://doi.org/10.1089/neu.2010.1358.
Bombardier CH, Fann JR, Temkin NR, Esselman PC, Barber J, Dikmen SS. Rates of major depressive disorder and clinical outcomes following traumatic brain injury. JAMA. 2010;303(19):1938–45. https://doi.org/10.1001/jama.2010.599.
Seel RT, Kreutzer JS, Rosenthal M, Hammond FM, Corrigan JD, Black K. Depression after traumatic brain injury: a National Institute on Disability and Rehabilitation Research Model Systems multicenter investigation. Arch Phys Med Rehabil. 2003;84(2):177–84. https://doi.org/10.1053/apmr.2003.50106.
Fann JR, Burington B, Leonetti A, Jaffe K, Katon WJ, Thompson RS. Psychiatric illness following traumatic brain injury in an adult health maintenance organization population. Arch Gen Psychiatry. 2004;61(1):53–61. https://doi.org/10.1001/archpsyc.61.1.53.
Jorge RE, Robinson RG, Arndt S. Are there symptoms that are specific for depressed mood in patients with traumatic brain injury? J Nerv Ment Dis. 1993;181(2):91–9. https://doi.org/10.1097/00005053-199302000-00004.
Osborn AJ, Mathias JL, Fairweather-Schmidt AK. Prevalence of anxiety following adult traumatic brain injury: a meta-analysis comparing measures, samples and postinjury intervals. Neuropsychology. 2016;30(2):247–61. https://doi.org/10.1037/neu000022.
Kraus MF, Susmaras T, Caughlin BP, Walker CJ, Sweeney JA, Little DM. White matter integrity and cognition in chronic traumatic brain injury: a diffusion tensor imaging study. Brain. 2007;130(Pt 10):2508–19. https://doi.org/10.1093/brain/awm216.
Williamson JB, Jaffee MS, Jorge RE. Posttraumatic stress disorder and anxiety-related conditions. Continuum (Minneapolis, Minn). 2021;27(6):1738–63. https://doi.org/10.1212/CON.0000000000001054.
Howlett JR, Nelson LD, Stein MB. Mental health consequences of traumatic brain injury. Biol Psychiatry. 2022;91(5):413–20. https://doi.org/10.1016/j.biopsych.2021.09.024.
Van Praag DLG, Cnossen MC, Polinder S, Wilson L, Maas AIR. Post-traumatic stress disorder after civilian traumatic brain injury: a systematic review and meta-analysis of prevalence rates. J Neurotrauma. 2019;36(23):3220–32. https://doi.org/10.1089/neu.2018.5759.
Jorge RE, Arciniegas DB. Mood disorders after TBI. Psychiatr Clin North Am. 2014;37(1):13–29. https://doi.org/10.1016/j.psc.2013.11.005.
Tsai YC, Liu CJ, Huang HC, Lin JH, Chen PY, Su YK, Chen CT, Chiu HY. A meta-analysis of dynamic prevalence of cognitive deficits in the acute, subacute, and chronic phases after traumatic brain injury. J Neurosci Nurs. 2021;53(2):63–8. https://doi.org/10.1097/JNN.0000000000000570.
Wolf JA, Koch PF. Disruption of network synchrony and cognitive dysfunction after traumatic brain injury. Front Syst Neurosci. 2016;10:43. https://doi.org/10.3389/fnsys.2016.00043.
Kant R, Duffy JD, Pivovarnik A. Prevalence of apathy following head injury. Brain Inj. 1998;12(1):87–92. https://doi.org/10.1080/026990598122908.
Worthington A, Wood RL. Apathy following traumatic brain injury: a review. Neuropsychologia. 2018;S0028393218301532 https://doi.org/10.1016/j.neuropsychologia.2018.04.012.
Grafman J, Schwab K, Warden D, Pridgen A, Brown HR, Salazar AM. Frontal lobe injuries, violence, and aggression: a report of the Vietnam Head Injury Study. Neurology. 1996;46(5):1231–8. https://doi.org/10.1212/wnl.46.5.1231.
Alderman N, Knight C, Henman C. Aggressive behaviour observed within a neurobehavioural rehabilitation service: utility of the OAS-MNR in clinical audit and applied research. Brain Inj. 2002;16:469–89. https://doi.org/10.1080/02699050110118458.
Epstein DJ, Legarreta M, Bueler E, King J, McGlade E, Yurgelun-Todd D. Orbitofrontal cortical thinning and aggression in mild traumatic brain injury patients. Brain Behav. 2016;6(12):e00581. https://doi.org/10.1002/brb3.581.
McAllister TW. Neurobiological consequences of traumatic brain injury. Dialogues Clin Neurosci. 2011;13(3):287–300. https://doi.org/10.31887/DCNS.2011.13.2/tmcallister.
Cardoso MGF, de Barros JLV, de Queiroz RAB, Rocha NP, Silver C, da Silva AS, da Silva EWM, Roque IG, Carvalho JL, Dos Santos LF, Cota LB, Lemos LM, Miranda MF, Miranda MF, Vianna PP, Oliveira RA, de Oliveira FT, Soares TSS, Pedroso VSP, Faleiro RM, Minas Gerais’ Traumatic Brain Injury Study Group. Potential biomarkers of impulsivity in mild traumatic brain injury: a pilot study. Behav Brain Res. 2023;449:114457. https://doi.org/10.1016/j.bbr.2023.114457.
Ozga JE, Povroznik JM, Engler-Chiurazzi EB, Haar CV. Executive (dys)function after traumatic brain injury: special considerations for behavioral pharmacology. Behav Pharmacol. 2018;29(7):617–37. https://doi.org/10.1097/FBP.0000000000000430.
Wood RL, Worthington A. Neurobehavioral abnormalities associated with executive dysfunction after traumatic brain injury. Front Behav Neurosci. 2017;11:195. https://doi.org/10.3389/fnbeh.2017.00195.
Peltz CB, Kenney K, Gill J, Diaz-Arrastia R, Gardner RC, Yaffe K. Blood biomarkers of traumatic brain injury and cognitive impairment in older veterans. Neurology. 2020;95(9):e1126–33. https://doi.org/10.1212/WNL.0000000000010087.
Lange RT, Lippa S, Brickell TA, Gill J, French LM. Serum tau, neurofilament light chain, glial fibrillary acidic protein, and ubiquitin carboxyl-terminal hydrolase L1 are associated with the chronic deterioration of neurobehavioral symptoms after traumatic brain injury. J Neurotrauma. 2023;40(5–6):482–92. https://doi.org/10.1089/neu.2022.0249.
Vedantam A, Brennan J, Levin HS, McCarthy JJ, Dash PK, Redell JB, Yamal JM, Robertson CS. Early versus late profiles of inflammatory cytokines after mild traumatic brain injury and their association with neuropsychological outcomes. J Neurotrauma. 2021;38(1):53–62. https://doi.org/10.1089/neu.2019.6979.
Pattinson CL, Shahim P, Taylor P, Dunbar K, Guedes VA, Motamedi V, Lai C, Devoto C, Peyer J, Roy MJ, Gill JM. Elevated tau in military personnel relates to chronic symptoms following traumatic brain injury. J Head Trauma Rehabil. 2020;35(1):66–73. https://doi.org/10.1097/HTR.0000000000000485.
Dretsch MN, Williams K, Emmerich T, Crynen G, Ait-Ghezala G, Chaytow H, Mathura V, Crawford FC, Iverson GL. Brain-derived neurotropic factor polymorphisms, traumatic stress, mild traumatic brain injury, and combat exposure contribute to postdeployment traumatic stress. Brain Behav. 2015;6(1):e00392. https://doi.org/10.1002/brb3.392.
Blennow K, Hardy J, Zetterberg H. The neuropathology and neurobiology of traumatic brain injury. Neuron. 2012;76(5):886–99. https://doi.org/10.1016/j.neuron.2012.11.021.
Juengst SB, Kumar RG, Failla MD, Goyal A, Wagner AK. Acute inflammatory biomarker profiles predict depression risk following moderate to severe traumatic brain injury. J Head Trauma Rehabil. 2015;30(3):207–18. https://doi.org/10.1097/HTR.0000000000000031.
Medeiros GC, Twose C, Weller A, Dougherty JW 3rd, Goes FS, Sair HI, Smith GS, Roy D. Neuroimaging correlates of depression after traumatic brain injury: a systematic review. J Neurotrauma. 2022;39(11–12):755–72. https://doi.org/10.1089/neu.2021.0374.
Raji CA, Willeumier K, Taylor D, Tarzwell R, Newberg A, Henderson TA, Amen DG. Functional neuroimaging with default mode network regions distinguishes PTSD from TBI in a military veteran population. Brain Imaging Behav. 2015;9(3):527–34. https://doi.org/10.1007/s11682-015-9385-5.
Mišić B, Dunkley BT, Sedge PA, Da Costa L, Fatima Z, Berman MG, Doesburg SM, McIntosh AR, Grodecki R, Jetly R, Pang EW, Taylor MJ. Post-traumatic stress constrains the dynamic repertoire of neural activity. J Neurosci. 2016;36(2):419–31. https://doi.org/10.1523/JNEUROSCI.1506-15.2016.
Todd RM, MacDonald MJ, Sedge P, Robertson A, Jetly R, Taylor MJ, Pang EW. Soldiers with posttraumatic stress disorder see a world full of threat: magnetoencephalography reveals enhanced tuning to combat-related cues. Biol Psychiatry. 2015;78(12):821–9. https://doi.org/10.1016/j.biopsych.2015.05.011.
Nathan DE, Bellgowan JAF, French LM, Wolf J, Oakes TR, Mielke J, Sham EB, Liu W, Riedye G. Assessing the impact of post-traumatic stress symptoms on the resting-state default mode network in a military chronic mild traumatic brain injury sample. Brain Connect. 2017;7(4):236–49. https://doi.org/10.1089/brain.2016.0433.
Gottshall JL, Guedes VA, Pucci JU, Brooks D, Watson N, Sheth P, Gabriel A, Mithani S, Leete JJ, Lai C, Qu BX, Devoto C, Gill JM, Kenney K, Werner JK. Poor sleep quality is linked to elevated extracellular vesicle-associated inflammatory cytokines in warfighters with chronic mild traumatic brain injuries. Front Pharmacol. 2021;12:762077. https://doi.org/10.3389/fphar.2021.762077.
Acknowledgments
The authors would like to thank FAPEMIG (Fundação de Amparo à Pesquisa do Estado de Minas Gerais, Brazil #APQ-02556-17), CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico, Brazil), and CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) for financial support.
Conflict of Interest
The author(s) declare(s) that there is no conflict of interest regarding the publication of this chapter.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
de Barros, J.L.V.M., dos Santos, L.F., de Lima Carvalho, J., de Oliveira Miranda dos Santos, S.L., de Miranda, A.S. (2023). Biomarkers of Traumatic Brain Injury and Related Neuropsychiatric Symptoms and Disorders. In: Teixeira, A.L., Rocha, N.P., Berk, M. (eds) Biomarkers in Neuropsychiatry. Springer, Cham. https://doi.org/10.1007/978-3-031-43356-6_19
Download citation
DOI: https://doi.org/10.1007/978-3-031-43356-6_19
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-43355-9
Online ISBN: 978-3-031-43356-6
eBook Packages: MedicineMedicine (R0)