Abstract
Imaging techniques, in particular magnetic resonance imaging (MRI), enable the visualization of changes in brain structure and function due to diseases of the central nervous system (CNS), and may thus contribute to a better understanding of the underlying mechanisms of associated behavioral impairments. Fatigue is a relatively broad concept with inconsistent definitions, and its severity is captured by different questionnaires (Kluger BM, Krupp LB, Enoka RM, Neurology. 80:409-16, 2013; Penner IK, Paul F, Nat Rev Neurol [Internet]. 13:662-75, 2017). Accordingly, the neurobiological causes of fatigue are complex, multifactorial, individually variable, and not yet well understood (Arm J, Ribbons K, Lechner-Scott J, Ramadan S, J Neurol Sci [Internet] 400:52-71, 2019). However, over the past 25 years, relevant structural and functional imaging correlates of primary (disease-related) fatigue in patients with multiple sclerosis (MS) have been identified, which will be presented and discussed in the following section.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Magnetic resonance imaging (MRI)
- Cortico-striato-thalamo-cortical loop
- White matter lesions
- Brain volume
- White matter integrity
- Brain activation
- Functional connectivity
1 Macrostructural Imaging Correlates of Primary Fatigue
1.1 Relevance of Inflammatory White Matter Lesions
MS-related focal inflammatory white matter changes can be visualized on MRI of the brain on T2-weighted sequences by hyperintense areas and are accordingly referred to as T2 lesions. In addition, hypointense areas on T1-weighted sequences (so-called chronic “black holes”) can indicate more pronounced focal tissue damage up to irreversible white matter tissue loss, whereas contrast enhancement of lesions on T1 weighted sequences reveals acute MS foci in the stage of blood-brain barrier disruption (Fig. 1).
The upper panel shows extensive, confluent, hyperintense and primarily periventricular localized white matter lesions after several years of MS (upper left, T2-FLAIR), some of these lesions are also visible on native T1-weighted images (upper right) by “dark” (hypointense) lesions (“black holes”) representing focal tissue damage. Note: age-related strong expansion of inner and moderate expansion of the outer ventricles representing cerebral atrophy. The lower panel shows native and contrast-enhanced T1-weighted images (“mit KM”), presenting an active lesion (so-called “wet black hole” due to formation of edema). This represents active disease
Although the majority of recent studies have failed to demonstrate an association between extent or volume of T1 or T2 lesions (Arm et al. 2019; Biberacher et al. 2017; Novo et al. 2018), previous studies have reported an association between higher white matter lesion volume and more pronounced fatigue (Calabrese et al. 2010; Mowry et al. 2009; Papadopoulou et al. 2013; Sepulcre et al. 2009; Tedeschi et al. 2007). However, a recent paper also concluded that global T2 lesion volume was increased in people with MS and prolonged fatigue, whereas patients with only a single episode of fatigue did not differ in global lesion volume. Consequently, the authors emphasized that the time course and evolution of fatigue should be better considered in future studies, as the pathophysiological mechanisms of persistent fatigue (due to irreversible white or gray matter damage) are likely to differ from those of fluctuating fatigue (e.g., due to inflammatory cytokines or hormonal changes) (Palotai et al. 2020).
Moreover, beyond these temporal dynamics, specific lesion localizations in the frontal (Morgante et al. 2011) and parieto-temporal white matter seem to be particularly associated with the occurrence of fatigue (Sepulcre et al. 2009; Altermatt et al. 2018; Palotai et al. 2019; Rocca et al. 2014). This suggests impairment or damage to pathways strategically relevant to the occurrence of fatigue, which play an essential role in cognitive processes such as task initiation, motivation, attention, and maintenance (Sepulcre et al. 2009).
1.2 Changes in Global and Regional Brain Volume
Some studies also showed an association between reduced global brain volume and the presence of fatigue (Biberacher et al. 2017; Mowry et al. 2009; Tedeschi et al. 2007). Interestingly, one study suggested an association between the presence of cognitive fatigue and a stronger decrease in brain volume over the subsequent 17 months (Sander et al. 2016). Whether fatigue is indeed a risk marker for disease progression should be investigated in further studies.
Importantly, regional analyses of cortical and subcortical gray matter suggest an association between neurodegeneration of striatal-thalamic-frontal regions and the presence of fatigue (Calabrese et al. 2010; Andreasen et al. 2010; Damasceno et al. 2016).
Both globally reduced cortex volume or thickness (Biberacher et al. 2017; Nourbakhsh et al. 2016; Nunnari et al. 2015; Nygaard et al. 2015) and specific reductions in frontal, insular (Sepulcre et al. 2009; Gonzalez Campo et al. 2019; Riccitelli et al. 2011), and parietal cortical volume (Hanken et al. 2016; Pellicano et al. 2010) have been related to the presence and severity of fatigue. Specifically, individuals with reduced cortical thickness of the primary motor cortex showed more severe motor fatigue, and there also tended to be a relationship between cognitive fatigue and cortical thickness in frontal and parietal regions responsible for attentional processes (Andreasen et al. 2019).
Also, decreased thalamic (Nourbakhsh et al. 2016; Bernitsas et al. 2017; Capone et al. 2019) and basal ganglia volumes (Damasceno et al. 2016; Bernitsas et al. 2017; Yarraguntla et al. 2018) have been frequently described in patients with fatigue. In addition, atrophy of the corpus callosum has been associated with more pronounced fatigue (Yaldizli et al. 2014; Yaldizli et al. 2011).
2 Microstructural Imaging Correlates of Primary Fatigue
Diffusion tensor imaging (DTI) allows the visualization and examination of nerve fiber connections of the brain in vivo. White matter integrity (“intactness”) using fractional anisotropy (FA) or the course of larger nerve fiber bundles using tractography can be assessed with this technique.
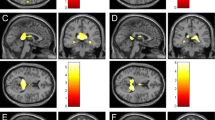
Reduced integrity of frontal nerve fiber connections (e.g., in the forceps minor) (Bisecco et al. 2016; Gobbi et al. 2014a; Pardini et al. 2010), in the cingulum (Bisecco et al. 2016; Pardini et al. 2015), in the corpus callosum (Sander et al. 2016; Bisecco et al. 2016), and in fronto-temporal connections, (e.g., in the fasciculus uncinatus) (Gobbi et al. 2014a), has been associated with more pronounced fatigue (Fig. 2).
Also, reduced integrity of subcortical connections, particularly towards the basal ganglia (especially to the caudate nucleus and the putamen (Palotai et al. 2019; Pardini et al. 2015)) and within the anterior thalamic radiation (Rocca et al. 2014; Bisecco et al. 2016; Gobbi et al. 2014a; Bester et al. 2013), as well as microstructural changes in the thalamus, basal ganglia, and frontal lobe (Wilting et al. 2016) have been described. Furthermore, in MS patients with fatigue, reduced integrity has been observed in the following structures: in the forceps major, the inferior fronto-occipital fascicle (Rocca et al. 2014), anterior capsula interna (Genova et al. 2013), and in nerve fiber connections from the hypothalamus to the brainstem (Hanken et al. 2015).
Interestingly, in contrast to the literature cited in the previous paragraph suggesting regionally selective processes, some DTI studies yielded findings suggesting that extensive white matter microstructural changes are related to the onset of fatigue independently from lesion burden and gray matter atrophy (Novo et al. 2018; Palotai et al. 2019).
Currently, it can be assumed that the continuous accumulation of focal lesions, independently or in concert with diffuse MS-related tissue damage, leads to an impairment of the cortical-subcortical brain connections, resulting in disturbances of the anatomical and functional connectivity. In particular, damage in cortico-striato-thalamo-cortical nerve fiber bundles (“cortico-striato-thalamo-cortical [CSTC] loop”) seems relevant for fatigue. In addition to the presented structural correlates of fatigue, potentially underlying changes in brain function are increasingly being investigated (Bertoli and Tecchio 2020).
3 Functional Imaging Correlates of Primary Fatigue
Functional magnetic resonance imaging (fMRI) of the brain can identify changes in cerebral tissue blood flow caused by the increased energy demand of active neurons and thus indirectly allows imaging of brain activity (e.g., which brain regions are active during a task) and functional connectivity (FC) (e.g., which brain regions work together and therefore exhibit synchronous changes in blood flow).
In one of the first functional imaging studies of fatigue in MS using positron emission tomography (PET), reduced glucose metabolism was found in the prefrontal cortex (especially in the lateral and medial prefrontal cortex and in pre- and supplementary motor areas) and in the basal ganglia (especially in the putamen and caudate nucleus) as well as an increased metabolism in the vermis cerebelli and the anterior cingulate in patients with MS and fatigue (Roelcke et al. 1997). Based on the findings of this study, it was hypothesized that changes in cortico-subcortical (mainly thalamus, basal ganglia) activation are associated with fatigue. These findings were corroborated by subsequent fMRI and further positron emission tomography (PET) studies (Derache et al. 2013; Filippi et al. 2002).
The studies cited below frequently report increased brain activation or activation of additional brain areas in patients with MS and fatigue compared to patients with MS without fatigue or healthy controls during the performance of motor or cognitive tasks. When performing motor tasks, increased activations in the thalamus (Rocca et al. 2007), basal ganglia (Rocca et al. 2007; Specogna et al. 2012), frontal lobe (Rocca et al. 2007; Specogna et al. 2012; Pardini et al. 2013; Rocca et al. 2016), precuneus (Rocca et al. 2009), and cerebellum (Pardini et al. 2013; Rocca et al. 2009) were associated with fatigue.
In addition to these “overactivations,” two of the aforementioned studies were able to observe parallel reduced brain activation in partially adjacent regions, e.g., in the frontal lobe, middle temporal lobe, postcentral gyrus, and basal ganglia (Rocca et al. 2016; Rocca et al. 2009), which were related to the severity of fatigue. Similarly, complex altered activation patterns (i.e., activity increases/decreases in the cingulate, frontal lobe, primary sensory cortex, or insula) have been reported in patients with fatigue after manual or mental effort during the performance of motor tasks (Tartaglia et al. 2008; White et al. 2009).
Regarding cognitive task performance, increased activity in the thalamus (DeLuca et al. 2008), basal ganglia (DeLuca et al. 2008), frontal lobe (DeLuca et al. 2008; Huolman et al. 2011; Spiteri et al. 2017), parietal lobe (DeLuca et al. 2008; Engström et al. 2013), and substantia nigra (Engström et al. 2013) have been observed in MS patients with fatigue. One study also reported parallel reduced activity in the thalamus, basal ganglia, and frontal lobe (Engström et al. 2013).
It should be noted though that most of these studies defined fatigue as a trait. In studies that used the definition of transient fatigue, i.e., a state, increased brain activation in MS patients was also reported during a cognitive task in the frontal lobe, caudate nucleus, and cerebellum (Genova et al. 2013).
Comparability of these findings is difficult due to the different applied tasks (motor vs. cognitive; simple vs. complex), small and frequently heterogeneous samples, and variability in operationalization (use of different questionnaires), prompting the increasing use of resting-state functional MRI examinations expecting identification of possible changes in functional connectivity (FC).
Recent studies emphasize that reciprocal FC of the thalamus and basal ganglia and subcortical FC to the cortex are particularly relevant for the occurrence of fatigue (Capone et al. 2019; Chaudhuri and Behan 2000).
Patients with fatigue showed greater FC between the thalamus and the precentral gyrus, reduced FC between the thalamus and the parietal operculum and superior frontal gyrus, and reduced FC between the insula and posterior cingulate (Stefancin et al. 2019). In the presence of fatigue, patients showed higher FC between the thalamus and insula and reduced FC between the dorsolateral prefrontal cortex (DLPFC) and posterior cingulate compared to healthy controls (Lin et al. 2019). In agreement, increased FC of the superior frontal gyrus with cortical regions (frontal, temporal, occipital) and reduced FC to the thalamus have been reported in cognitive fatigue (Pravatà et al. 2016).
Examining FC of the thalamus (Hidalgo de la Cruz et al. 2017), the authors found specific changes in connectivity to the precuneus and cerebellum (cognitive fatigue), the sensorimotor network (SMN; motor fatigue), and the insula (psychosocial fatigue).
Greater expression of fatigue correlated with reduced FC of the striatum with the SMN and increased FC of the DLPFC to the inferior parietal gyrus and sensorimotor cortex (Jaeger et al. 2019). Consistent with these findings, more severe fatigue has been reported to correlate with stronger FC between the caudate nucleus and DLPFC (Wu et al. 2016). Greater fatigue severity correlated with reduced FC of the basal ganglia with the frontal and parietal cortex and increased FC between the caudate nucleus and motor cortex (Finke et al. 2015).
Patients with fatigue showed higher FC in the posterior cingulate and reduced FC in the anterior cingulate when investigating a so-called “resting network” (“default mode network” [DMN]). When focusing on the SMN, increased functional connectivity was observed in primary and supplementary motor areas (Cruz-Gómez et al. 2013), with changes in the DMN being more strongly associated with the occurrence of fatigue (Bisecco et al. 2018).
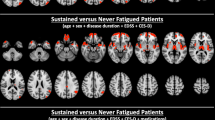
In general, FC, and thus the cooperation of brain areas responsible for maintaining and accomplishing sensorimotor function, motor planning, motivation, attention and executive function, as well as the resting network, appears to be disrupted in the context of MS in fatigue (Fig. 3).
Even though the presented functional MRI findings are partly in agreement with the macro- and microstructural findings and provide further possible MRI markers of fatigue, the exemplary and diverse results presented above show how complex the underlying mechanisms of fatigue are likely to be.
4 Functional Imaging Correlates of Fatigability
In contrast to subjectively perceived fatigue, the objectively measurable decline in performance due to fatigue (“fatigability”) is increasingly being investigated by means of imaging. Although there is a positive correlation between fatigability and fatigue (Loy et al. 2017), distinct underlying mechanisms are suspected (Kluger et al. 2013).
Changes in brain activation associated with fatigue and fatigability in MS patients were specifically investigated. The findings revealed reduced activation in the insula, frontal and parietal lobes, basal ganglia and amygdala with higher cognitive fatigability, and increased activation in the anterior cingulate with increased fatigue (Spiteri et al. 2017). In contrast, increased activation in the caudate nucleus was found to be associated with higher cognitive fatigability (Berard et al. 2019).
5 Secondary Fatigue and Imaging Findings
Secondary fatigue describes the occurrence of severe fatigue that is not directly caused by MS, such as insomnia (Foschi et al. 2019), infections, chronic pain, medications, physical overexertion, and/or affective problems (especially depression). Even though fatigue and depression are distinct symptoms, both can overlap and lead to listlessness, exhaustion, and tiredness. It should be noted that the construct of secondary fatigue encompasses multifactorial etiologies and consequently correlations and specifically causalities can only be explored to a limited extent.
In this context, individual studies reported that regional cortical atrophy (Gobbi et al. 2014b) and cerebellar atrophy (Lazzarotto et al. 2020) in MS are related to both depression and fatigue. Another study demonstrated an association between reduced cortical thickness in the inferior parietal lobe and depression-independent fatigue, but cortical thickness provided little variance explanation for fatigue and depression, and the authors consequently emphasized the importance of subcortical mechanisms to study distinct brain markers (Hanken et al. 2016).
Regarding associated changes in white matter, damage to fronto-striatal and temporo-insular tracts appears to be crucial regarding the occurrence of persistent fatigue and independent of the presence of depression (Palotai et al. 2019). Reduced white matter integrity in frontal (forceps minor) and fronto-temporal (fasciculus uncinatus) fiber tracts has also been observed in fatigue, independent from depression (Gobbi et al. 2014a). Overlapping and distinct FC changes in the DMN were found for depression and fatigue (Høgestøl et al. 2019).
The identification of distinct and overlapping underlying brain mechanisms of depression and fatigue is only possible using a clear operationalization and comprehensive assessment of both impairments.
6 Summary and Outlook
Recent neuroimaging findings suggest that both macro- and microstructural as well as functional changes in specific brain regions (mainly frontal, parietal, temporal, and subcortical) and specific structural and functional connections (mainly cortico-striato-thalamo-cortical) are related to the occurrence of primary fatigue in people with MS (Arm et al. 2019; Palotai and Guttmann 2019) (Fig. 3).
Due to the diversity of fatigue assessment and the variety of operationalizations of this subjectively perceived impairment (Penner & Paul 2017), the identification of objective MRI markers of fatigue cohorts will continue to vary in the future. Furthermore, while the range of different analytical approaches (e.g., structural and functional, activation and connectivity, local changes vs. network analysis) allows for a more comprehensive investigation of underlying mechanisms, it also complicates the verification of “simple” markers, which are unlikely in this context. Currently, both structural and functional changes in the cortico-striato-thalamo-cortical network appear to be associated with fatigue. Consequently, multicenter and longitudinal studies with sufficient sample size are needed to assess objective MRI parameters and their specificity independently of other possible influencing factors (e.g., age, physical and cognitive impairment, depression).
To date, imaging techniques cannot contribute to the individual clinical diagnosis of fatigue, because imaging of individual patients with fatigue often does not yield obvious MRI correlates, and imaging markers of fatigue do not necessarily imply causality. However, MRI markers do provide important relevant information for developing and optimizing possible more targeted treatment approaches.
Exploring the underlying cerebral mechanisms of the complex concept of fatigue and associated constructs such as “fatigability” and secondary fatigue will continue to lead to interesting research findings in the future.
References
Altermatt A, Gaetano L, Magon S, Häring DA, Tomic D, Wuerfel J, et al. Clinical correlations of brain lesion location in multiple sclerosis: voxel-based analysis of a large clinical trial dataset. Brain Topogr [Internet]. 2018;31(5):886–94. Available from:. https://doi.org/10.1007/s10548-018-0652-9.
Andreasen A, Iversen P, Marstrand L, Siersma V, Siebner HR, Sellebjerg F. Structural and cognitive correlates of fatigue in progressive multiple sclerosis. Neurol Res [Internet]. 2019;41(2):168–76. https://doi.org/10.1080/01616412.2018.1547813.
Andreasen AK, Jakobsen J, Soerensen L, Andersen H, Petersen T, Bjarkam CR, et al. Regional brain atrophy in primary fatigued patients with multiple sclerosis. NeuroImage. 2010;50(2):608–15.
Arm J, Ribbons K, Lechner-Scott J, Ramadan S. Evaluation of MS related central fatigue using MR neuroimaging methods: scoping review. J Neurol Sci [Internet]. 2019;400(August 2018):52–71. https://doi.org/10.1016/j.jns.2019.03.007.
Berard JA, Fang Z, Walker LAS, Lindsay-Brown A, Osman L, Cameron I, et al. Imaging cognitive fatigability in multiple sclerosis: objective quantification of cerebral blood flow during a task of sustained attention using ASL perfusion fMRI. Brain Imaging Behav. 2019:1–12.
Bernitsas E, Yarraguntla K, Bao F, Sood R, Santiago-Martinez C, Govindan R, et al. Structural and neuronal integrity measures of fatigue severity in multiple sclerosis. Brain Sci. 2017;7(8):1–10.
Bertoli M, Tecchio F. Fatigue in multiple sclerosis: does the functional or structural damage prevail? Mult Scler J. 2020:1–7.
Bester M, Lazar M, Petracca M, Babb JS, Herbert J, Grossman RI, et al. Tract-specific white matter correlates of fatigue and cognitive impairment in benign multiple sclerosis. J Neurol Sci. 2013;330(1–2):61–6.
Biberacher V, Schmidt P, Selter R, Pernpeinter V, Kowarik M, Knier B, et al. Fatigue in multiple sclerosis: associations with clinical, MRI and CSF parameters. Mult Scler J. 2017:1–11.
Bisecco A, Caiazzo G, D’Ambrosio A, Sacco R, Bonavita S, Docimo R, et al. Fatigue in multiple sclerosis: the contribution of occult white matter damage. Mult Scler. 2016;22(13):1676–84.
Bisecco A, Di Nardo F, Docimo R, Caiazzo G, D’Ambrosio A, Bonavita S, et al. Fatigue in multiple sclerosis: the contribution of resting-state functional connectivity reorganization. Mult Scler J. 2018:1–10.
Calabrese M, Rinaldi F, Grossi P, Mattisi I, Bernardi V, Favaretto A, et al. Basal ganglia and frontal/parietal cortical atrophy is associated with fatigue in relapsing-remitting multiple sclerosis. Mult Scler. 2010;16(10):1220–8.
Capone F, Collorone S, Cortese R, Di Lazzaro V, Moccia M. Fatigue in multiple sclerosis: the role of thalamus. Mult Scler J. 2019:1–11.
Chaudhuri A, Behan PO. Fatigue and basal ganglia. J Neurol Sci. 2000;179(1–2):34–42.
Cruz-Gómez AJ, Ventura-Campos N, Belenguer A, Avila C, Forn C. The link between resting-state functional connectivity and cognition in MS patients. Mult Scler [Internet]. 2013;2013. http://www.ncbi.nlm.nih.gov/pubmed/23828871
Damasceno A, Damasceno BP, Cendes F. Atrophy of reward-related striatal structures in fatigued MS patients is independent of physical disability. Mult Scler J. 2016;22(6):822–9.
DeLuca J, Genova HM, Hillary FG, Wylie G. Neural correlates of cognitive fatigue in multiple sclerosis using functional MRI. J Neurol Sci. 2008;270(1–2):28–39.
Derache N, Grassiot B, Mézenge F, Emmanuelle Dugué A, Desgranges B, Constans JM, et al. Fatigue is associated with metabolic and density alterations of cortical and deep gray matter in relapsing-remitting-multiple sclerosis patients at the earlier stage of the disease: a PET/MR study. Mult Scler Relat Disord [Internet]. 2013;2(4):362–9. Available from:. https://doi.org/10.1016/j.msard.2013.03.005.
Engström M, Flensner G, Landtblom AM, Ek AC, Karlsson T. Thalamo-striato-cortical determinants to fatigue in multiple sclerosis. Brain Behav. 2013;3(6):715–28.
Filippi M, Rocca MA, Colombo B, Falini A, Codella M, Scotti G, et al. Functional magnetic resonance imaging correlates of fatigue in multiple sclerosis. NeuroImage. 2002;15(3):559–67.
Finke C, Schlichting J, Papazoglou S, Scheel M, Freing A, Soemmer C, et al. Altered basal ganglia functional connectivity in multiple sclerosis patients with fatigue. Mult Scler. 2015;21(7):925–34.
Foschi M, Rizzo G, Liguori R, Avoni P, Mancinelli L, Lugaresi A, et al. Sleep-related disorders and their relationship with MRI findings in multiple sclerosis. Sleep Med. 2019;56:90–7.
Genova HM, Rajagopalan V, DeLuca J, Das A, Binder A, Arjunan A, et al. Examination of cognitive fatigue in multiple sclerosis using functional magnetic resonance imaging and diffusion tensor imaging. PLoS One. 2013;8(11):1–10.
Gobbi C, Rocca MA, Pagani E, Riccitelli GC, Pravatà E, Radaelli M, et al. Forceps minor damage and co-occurrence of depression and fatigue in multiple sclerosis. Mult Scler J. 2014a;20(12):1633–40.
Gobbi C, Rocca MA, Riccitelli G, Pagani E, Messina R, Preziosa P, et al. Influence of the topography of brain damage on depression and fatigue in patients with multiple sclerosis. Mult Scler. 2014b;20(2):192–201.
Gonzalez Campo C, Salamone PC, Rodríguez-Arriagada N, Richter F, Herrera E, Bruno D, et al. Fatigue in multiple sclerosis is associated with multimodal interoceptive abnormalities. Mult Scler J. 2019:9–12.
Hanken K, Eling P, Kastrup A, Klein J, Hildebrandt H. Integrity of hypothalamic fibers and cognitive fatigue in multiple sclerosis. Mult Scler Relat Disord. 2015;4(1):39–46.
Hanken K, Eling P, Klein J, Klaene E, Hildebrandt H. Different cortical underpinnings for fatigue and depression in MS? Mult Scler Relat Disord [Internet]. 2016;6:81–6. https://doi.org/10.1016/j.msard.2016.02.005.
Hidalgo de la Cruz M, Donaldson AIC, Myint PK. Abnormal functional connectivity of thalamic sub-regions contributes to fatigue in multiple sclerosis. Mult Scler Int. 2017:259–61.
Høgestøl EA, Nygaard GO, Alnæs D, Beyer MK, Westlye LT, Harbo HF. Symptoms of fatigue and depression is reflected in altered default mode network connectivity in multiple sclerosis. PLoS One. 2019;14(4):1–14.
Huolman S, Hämäläinen P, Vorobyev V, Ruutiainen J, Parkkola R, Laine T, et al. The effects of rivastigmine on processing speed and brain activation in patients with multiple sclerosis and subjective cognitive fatigue. Mult Scler J. 2011;17(11):1351–61.
Jaeger S, Paul F, Scheel M, Brandt A, Heine J, Pach D, et al. Multiple sclerosis–related fatigue: altered resting-state functional connectivity of the ventral striatum and dorsolateral prefrontal cortex. Mult Scler J. 2019;25(4):554–64.
Kluger BM, Krupp LB, Enoka RM. Fatigue and fatigability in neurologic inllnesses. Neurology. 2013;80:409–16.
Lazzarotto A, Margoni M, Franciotta S, Zywicki S, Riccardi A, Poggiali D, et al. Selective cerebellar atrophy associates with depression and fatigue in the early phases of relapse-onset multiple sclerosis. Cerebellum. 2020;19:192.
Lin F, Zivadinov R, Hagemeier J, Weinstock-Guttman B, Vaughn C, Gandhi S, et al. Altered nuclei-specific thalamic functional connectivity patterns in multiple sclerosis and their associations with fatigue and cognition. Mult Scler J. 2019;25(9):1243–54.
Loy BD, Taylor RL, Fling BW, Horak FB. Relationship between perceived fatigue and performance fatigability in people with multiple sclerosis: a systematic review and meta-analysis. J Psychosom Res. 2017;100:1–7.
Morgante F, Dattola V, Crupi D, Russo M, Rizzo V, Ghilardi MF, et al. Is central fatigue in multiple sclerosis a disorder of movement preparation? J Neurol. 2011;258(2):263–72.
Mowry EM, Beheshtian A, Waubant E, Goodin DS, Cree BA, Qualley P, et al. Quality of life in multiple sclerosis is associated with lesion burden and brain volume measures. Neurology. 2009;72(20):1760–5.
Nourbakhsh B, Nunan-Saah J, Maghzi AH, Julian LJ, Spain R, Jin C, et al. Longitudinal associations between MRI and cognitive changes in very early MS. Mult Scler Relat Disord. 2016;5:47–52.
Novo AM, Batista S, Alves C, Dalmeida OC, Marques IB, Macario C, et al. The neural basis of fatigue in multiple sclerosis. A multimodal MRI approach. Neurol Clin Pract. 2018;8(6):492–500.
Nunnari D, De Cola MC, D’Aleo G, Rifici C, Russo M, Sessa E, et al. Impact of depression, fatigue, and global measure of cortical volume on cognitive impairment in multiple sclerosis. Biomed Res Int. 2015;2015:11–3.
Nygaard GO, Walhovd KB, Sowa P, Chepkoech JL, Bjørnerud A, Due-Tønnessen P, et al. Cortical thickness and surface area relate to specific symptoms in early relapsing-remitting multiple sclerosis. Mult Scler J. 2015;21(4):402–14.
Palotai M, Cavallari M, Healy BC, Guttmann CRG. A novel classification of fatigue in multiple sclerosis based on longitudinal assessments. Mult Scler J. 2020;26:725.
Palotai M, Cavallari M, Koubiyr I, Morales Pinzon A, Nazeri A, Healy BC, et al. Microstructural fronto-striatal and temporo-insular alterations are associated with fatigue in patients with multiple sclerosis independent of white matter lesion load and depression. Mult Scler J. 2019:1–11.
Palotai M, Guttmann CRG. Brain anatomical correlates of fatigue in multiple sclerosis. Mult Scler J. 2019;
Papadopoulou A, Müller-Lenke N, Naegelin Y, Kalt G, Bendfeldt K, Kuster P, et al. Contribution of cortical and white matter lesions to cognitive impairment in multiple sclerosis. Mult Scler J. 2013;19(10):1290–6.
Pardini M, Bonzano L, Bergamino M, Bommarito G, Feraco P, Murugavel A, et al. Cingulum bundle alterations underlie subjective fatigue in multiple sclerosis. Mult Scler J. 2015;21(4):442–7.
Pardini M, Bonzano L, Mancardi GL, Roccatagliata L. Frontal networks play a role in fatigue perception in multiple sclerosis. Behav Neurosci. 2010;124(3):329–36.
Pardini M, Bonzano L, Roccatagliata L, Mancardi GL, Bove M. The fatigue-motor performance paradox in multiple sclerosis. Sci Rep. 2013;3:10–3.
Pellicano C, Gallo A, Li X, Ikonomidou VN, Evangelou IE, Ohayon JM, et al. Relationship of cortical atrophy to fatigue in patients with multiple sclerosis. Arch Neurol. 2010;67(4):447–53.
Penner IK, Paul F. Fatigue as a symptom or comorbidity of neurological diseases. Nat Rev Neurol [Internet]. 2017;13(11):662–75. https://doi.org/10.1038/nrneurol.2017.117.
Pravatà E, Zecca C, Sestieri C, Caulo M, Riccitelli GC, Rocca MA, et al. Hyperconnectivity of the dorsolateral prefrontal cortex following mental effort in multiple sclerosis patients with cognitive fatigue. Mult Scler. 2016;22(13):1665–75.
Riccitelli G, Rocca MA, Forn C, Colombo B, Comi G, Filippi M. Voxelwise assessment of the regional distribution of damage in the brains of patients with multiple sclerosis and fatigue. Am J Neuroradiol. 2011;32(5):874–9.
Rocca MA, Agosta F, Colombo B, Mezzapesa DM, Falini A, Comi G, et al. fMRI changes in relapsing-remitting multiple sclerosis patients complaining of fatigue after IFNβ-1a injection. Hum Brain Mapp. 2007;28(5):373–82.
Rocca MA, Gatti R, Agosta F, Broglia P, Rossi P, Riboldi E, et al. Influence of task complexity during coordinated hand and foot movements in MS patients with and without fatigue : AA kinematic and functional MRI study. J Neurol. 2009;256(3):470–82.
Rocca MA, Meani A, Riccitelli GC, Colombo B, Rodegher M, Falini A, et al. Abnormal adaptation over time of motor network recruitment in multiple sclerosis patients with fatigue. Mult Scler [Internet]. 2016;22(9):1144–53. http://www.ncbi.nlm.nih.gov/pubmed/26493126
Rocca MA, Parisi L, Pagani E, Copetti M, Rodegher M, Colombo B, et al. Regional but not global brain damage contributes to fatigue in multiple sclerosis. Radiology. 2014;273(2):511–20.
Roelcke U, Kappos L, Lechner-Scott J, Brunnschweiler H, Huber S, Ammann W, et al. Reduced glucose metabolism in the frontal cortex and basal ganglia of multiple sclerosis patients with fatigue: a 18F-fluorodeoxyglucose positron emission tomography study. Neurology. 1997;48(6):1566–71.
Sander C, Eling P, Hanken K, Klein J, Kastrup A, Hildebrandt H. The impact of MS-related cognitive fatigue on future brain parenchymal loss and relapse: a 17-month follow-up study. Front Neurol. 2016;7(SEP)
Sepulcre J, Masdeu JC, Goñi J, Arrondo G, Vélez de Mendizábal N, Bejarano B, et al. Fatigue in multiple sclerosis is associated with the disruption of frontal and parietal pathways. Mult Scler. 2009;15(3):337–44.
Specogna I, Casagrande F, Lorusso A, Catalan M, Gorian A, Zugna L, et al. Functional MRI during the execution of a motor task in patients with multiple sclerosis and fatigue. Radiol Med. 2012;117(8):1398–407.
Spiteri S, Hassa T, Claros-Salinas D, Dettmers C, Schoenfeld M. Neural correlates of effort-dependent and effort-independent cognitive fatigue components in patients with multiple sclerosis. MSJ. 2017;9:1–11.
Stefancin P, Govindarajan ST, Krupp L, Charvet L, Duong TQ. Resting-state functional connectivity networks associated with fatigue in multiple sclerosis with early age onset. Mult Scler Relat Disord [Internet]. 2019;31(March):101–5. https://doi.org/10.1016/j.msard.2019.03.020.
Tartaglia MC, Narayanan S, Arnold DL. Mental fatigue alters the pattern and increases the volume of cerebral activation required for a motor task in multiple sclerosis patients with fatigue. Eur J Neurol. 2008;15(4):413–9.
Tedeschi G, Dinacci D, Lavorgna L, Prinster A, Savettieri G, Quattrone A, et al. Correlation between fatigue and brain atrophy and lesion load in multiple sclerosis patients independent of disability. J Neurol Sci. 2007;263(1–2):15–9.
White AT, Lee JN, Light AR, Light KC. Brain activation in multiple sclerosis: a BOLD fMRI study of the effects of fatiguing hand exercise. Mult Scler. 2009;15(5):580–6.
Wilting J, Rolfsnes HO, Zimmermann H, Behrens M, Fleischer V, Zipp F, et al. Structural correlates for fatigue in early relapsing remitting multiple sclerosis. Eur Radiol. 2016;26(2):515–23.
Wu L, Zhang Y, Zhour F, Gao L, He L, Zeng X, et al. Altered intra- and interregional synchronization in relapsing—remitting multiple sclerosis : a resting-state fMRI study. Neuropsychiatr Dis Treat. 2016;12:853–62.
Yaldizli Ö, Glassl S, Sturm D, Papadopoulou A, Gass A, Tettenborn B, et al. Fatigue and progression of corpus callosum atrophy in multiple sclerosis. J Neurol. 2011;258(12):2199–205.
Yaldizli Ö, Penner IK, Frontzek K, Naegelin Y, Amann M, Papadopoulou A, et al. The relationship between total and regional corpus callosum atrophy, cognitive impairment and fatigue in multiple sclerosis patients. Mult Scler J. 2014;20(3):356–64.
Yarraguntla K, Seraji-Bozorgzad N, Lichtman-Mikol S, Razmjou S, Bao F, Sriwastava S, et al. Multiple sclerosis fatigue: a longitudinal structural MRI and diffusion tensor imaging study. J Neuroimaging. 2018;28(6):650–5.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Pinter, D., Enzinger, C. (2023). Neuroimaging and Fatigue. In: Penner, IK. (eds) Fatigue in Multiple Sclerosis. Springer, Cham. https://doi.org/10.1007/978-3-031-13498-2_11
Download citation
DOI: https://doi.org/10.1007/978-3-031-13498-2_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-13497-5
Online ISBN: 978-3-031-13498-2
eBook Packages: Behavioral Science and PsychologyBehavioral Science and Psychology (R0)