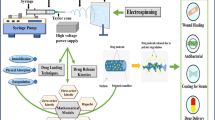
Abstract
Electrospun fibers have been very widely explored in the context of drug delivery. The rapid drying nature of the electrospinning process tends to result in amorphous solid dispersions, and hence the use of a hydrophilic filament-forming polymer can give significant increases in dissolution rate, apparent solubility, and bioavailability. Electrospun formulations thus have great potential to overcome the solubility challenges faced by > 70% of emerging drug candidates. Beyond this, by careful choice of the polymer carrier and the nanoscale architecture of the fibers (monolithic, core/shell, Janus, etc.) it is possible to precisely control both the drug release rate and location, and in a number of cases much sought after zero-order (constant rate) release has been obtained with electrospun systems. Fibers from electrospinning have thus been widely explored for drug delivery via a range of routes, including oral, transdermal, ocular, and implantation. In this chapter, we will review the body of literature in the area, focusing on the various types of release modality that can be obtained and the most exciting recent findings in the field. We will further consider issues of translation from bench to bedside, covering the great progress made in the scale-up of the electrospinning process in recent years, the need for production under Good Manufacturing Practice conditions, and evaluating how close electrospun formulations are to becoming marketed products.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
The ultimate goal of drug delivery systems is to deliver the drug locally, safely and in a controlled fashion in order to minimize side effects often associated with systemic administration of active pharmaceutical agents (APIs). To achieve the desired therapeutic effect, the correct API must be delivered to the required site of action, at the correct time, and in an appropriate amount. Inability to meet these criteria will render the medicine either ineffective or toxic to the patient. Although medicines can take a wide range of forms and be administered via numerous routes, for reasons of patient compliance and convenience the majority of APIs are given orally as solid dosage forms. This has a number of benefits: for instance, it is simple for a patient to take two tablets three times a day, and oral solid dosage forms can be administered by the patient themselves rather than requiring a healthcare professional (as is usually the case for injections).
There are also drawbacks to the use of oral solid dosage forms, however. In order to be active in the body, the API needs to be in solution. This requires a solid dosage form to dissolve into the fluids present in the gastrointestinal tract. In the process of dissolution, the large aggregates of molecules that make up a solid material are separated out, resulting in a solution in which each API molecule is surrounded by water molecules. Thus, API-API intermolecular interactions in the solid-state are replaced by API-water interactions. The dissolution rate and maximum concentration of the drug in solution (the solubility) are determined by the balance of these energies. Further, the majority of medicines given orally are intended to act systemically (throughout the body) rather than locally in the gastrointestinal tract. Thus, after it dissolves, the molecules from a solid dosage form need to pass through the lipid biomembranes which line the intestinal tract, so that they can reach the bloodstream. This process is termed permeation.
Some APIs have both high dissolution rates and solubilities and high permeability, but in most cases, there is a formulation challenge that needs to be overcome to improve at least one of these properties. The Biopharmaceutical Classification System (BCS) helps pharmaceutical scientists to classify APIs in terms of their properties: BCS class I drugs have high solubility and permeability; class II are poorly soluble but freely permeable; class III APIs are highly soluble but poorly permeable, and class IV materials are both poorly soluble and poorly permeable.
Beyond BCS considerations, pharmaceutical scientists need to consider carefully where in the body the drug will be released. For instance, many APIs cause irritation to the stomach lining, and thus it is desirable to avoid release there and instead target delivery to the latter stages of the gastrointestinal tract. The rate of release of a drug from a medicine also needs to be established, since this impacts both the therapeutic efficacy and the dosage frequency required. The stability of the API must additionally be established and understood. While small-molecule APIs such as paracetamol or ibuprofen are held together by covalent bonds and thus are generally stable in vivo, more complex biomolecules such as proteins rely heavily on weaker hydrogen and van der Waals interactions to sustain their 3D structure. A loss of 3D structure will result in a loss of activity, and since the interactions holding the structure together are weak proteins degrade easily. Great care must thus be taken in formulating, to ensure that the API delivered is therapeutically active.
Electrospinning offers routes to improve solubility and dissolution rate by processing the API into an amorphous solid dispersion (ASD), where there is a molecular dispersion of drug in polymer and thus no API-API interactions to overcome during the process of dissolution. The addition of a permeation enhancer into the formulation can improve permeability. Further, the choice of the polymer used as the filament-forming carrier can allow the drug release location and rate to be tuned. The fact that electrospinning does not require heat to evaporate the solvent can aid in maintaining the stability of protein APIs, but the use of organic solvents can be problematic in this regard. These topics will be discussed in more detail below, with illustrative examples presented. We will first consider small molecule APIs and the types of release profiles that can be attained. Later in the chapter, we will also consider the use of electrospinning to process more complex biologic-based therapeutics and consider the next steps in moving electrospun medicines towards the market.
2 Fiber Architecture
A range of fiber architectures can be achieved by using spinnerets with different needle arrangements, as shown in Fig. 7.1. The simplest experiment is mono-axial electrospinning, where a blunt-ended spinneret is used to generate monolithic fibers. These have a number of benefits, but their large surface area and the presence of drug at the fiber surface often results in an undesirable initial “burst” of release. To overcome this, co-axial spinning can be used to produce core/shell fiber architectures; if the drug is confined to the core then burst release can be reduced and/or release targeted to a particular part of the body. Tri-axial spinning allows the possibility of creating 3–layer fibers to provide further control over the drug release profile. Two-faced Janus fiber architectures can also be created using a side-by-side spinneret, resulting in two-compartment fibers where both sides can interact with their external environment.
Spinneret designs for different types of electrospinning, and the structures of the fibers produced. Reproduced from Williams G. R. et al. 2018. Nanofibres in Drug Delivery. London: UCL Press (https://doi.org/10.14324/111.9781787350182), under the terms of a CC BY 4.0 license
3 Pharmaceutical Polymers
Several aspects need to be considered when choosing the polymer for the electrospinning solution. Probably the most important consideration is the intended application of the product. The polymer degradation half-life and by-products, biocompatibility, and solubility will heavily influence the potential applications of the product. For example, for fast-release applications, a polymer with a relatively rapid dissolution/degradation rate and high solubility in aqueous solvents (such as polyvinylpyrrolidone (PVP)) would be preferred [1]. In contrast, when designing a long-term surgical implant, a hydrophobic polymer with slow degradation rates would be more suitable. In some cases, polymers with special characteristics such as thermo- or pH-sensitivity are of interest, aiding targeted delivery to the action site [2].
One of the most commonly used pharmaceutical polymers in electrospinning is poly(lactic-co-glycolic) acid (PLGA) (Fig. 7.2a). PLGA is a synthetic copolymer formed of polylactic (PLA) and polyglycolic (PGA) acids. PLGA degrades by hydrolysis of its ester linkages via bulk erosion in aqueous environments. The rate of degradation can be controlled, for example by adjusting the molecular weight of the polymer or the molar ratio of the monomer components. As an example, PLGA copolymers with a higher content of lactic acid will degrade more slowly due to the presence of hydrophobic methyl groups [3].
Another medically important polymer is poly(ε-caprolactone) (PCL; Fig. 7.2b), which is an aliphatic polyester composed of hexanoate repeat units. It is a semi-crystalline polymer widely used in the manufacturing of surgical sutures, drug delivery systems and tissue engineering scaffolds, and biodegrades through hydrolysis over a time period ranging from several months to years, depending on the molecular weight [4, 5].
Biomaterials manufactured from PLGA and PCL follow a similar triphasic breakdown profile. In the hydration step, water penetrates into the amorphous region of the material, thus disrupting the van der Waals forces. Subsequently, the polymer chain starts to degrade either by end-chain or random-chain scission and the cleavage of covalent bonds. This leads to a decrease in the molecular weight [6]. The degradation process is then autocatalysed by carboxylic end groups and the particle structure is severely disrupted by the breakdown of backbone covalent bonds. The resulting metabolite monomers incorporate into the Krebs cycle and can be safely eliminated from the body [7, 8].
4 Small Molecule Drugs
There exist a range of drug release profiles which find utility in medicines development (Fig. 7.3). These range from very rapid release, aiming to deliver a drug cargo within a few minutes of administration, to formulations seeking to prolong release over days or weeks. Complex multi-stage profiles can also be desirable, for instance to provide an initial “loading” dose for rapid relief of pain followed by a maintenance dose to keep the concentration in the patient’s bloodstream at a therapeutic level. Electrospun formulations have been explored for many of these modalities of small molecule API delivery, and we will discuss a series of examples below.
4.1 Fast Dissolving Drug Delivery Systems
Fast-dissolving drug delivery systems (FD-DDSs) offer a variety of advantages such as very rapid onset of action and enhanced bioavailability. This type of release is ideal for situations where instant drug dissolution is required to achieve rapid therapeutic effects. Electrospinning can be used to generate ASDs with very high surface area to volume ratios, and thus is ideal to achieve fast dissolution of otherwise poorly water-soluble drugs. Such electrospun FD-DDSs are commonly designed as solid films that undergo instant wetting by saliva to dissolve/disintegrate in the patient’s mouth. These formulations are beneficial to administer drugs to children and patients with swallowing difficulties, as they can be used without consuming liquid or chewing [9].
Numerous electrospun FD-DDSs have been reported in the literature, often based on the hydrophilic and fast-dissolving PVP. For instance, fibers containing paracetamol and caffeine were generated by Illangakoon et al. The fiber mats disintegrated completely within a very short time (< 0.5 s) when added to simulated saliva solution and released the drug cargo within around 150 s in a dissolution test, giving notably faster dissolution compared to the pure drugs [10]. PVP fibers have also been used to enhance the dissolution rate of mebeverine hydrochloride [11], ketoprofen (KET) [12], indomethacin [13], ornidazole [14], borneol [15], emodin [16], sildenafil citrate [17], and loratadine [18]. Caution must be taken in designing the formulations, however: the drug loading in the system has been shown to be crucial in determining the release properties. In a very recent report, PVP was employed for the preparation of KET loaded fibers. ASDs were obtained, but while fibers with low KET loading (9% w/w) disintegrated quickly and hastened KET dissolution, fibers with 23% w/w drug did now show these benefits (Fig. 7.4) [12].
Digital camera images of the disintegration of PVP fibers containing 9% w/w KET. Reprinted with permission from Geng et al. [12]
While PVP has attracted the most attention, other hydrophilic polymers can also be used to generate electrospun FD-DDSs. For instance, polyvinyl alcohol (PVA) fibers have been prepared loaded with caffeine and riboflavin [19]. Electrospun blends of polyethylene oxide are also reported to provide fast release of drugs such as lidocaine and diclofenac [20]. More recently, the natural polymers jelly fig polysaccharide and pullulan have been investigated for fast delivery of hydrophobic drugs such as ampicillin and dexamethasone [21].
4.2 Extended Release
Extended release systems (also termed sustained, controlled, prolonged, or slow-release) are designed to release their drug cargo over a prolonged period of time in the body, with the goal of maintaining a therapeutically potent dose over this period of time and reducing the required dosing frequency. For instance, electrospun (ES) mats capable of delivering analgesics or anti-inflammatory medications in a sustained manner could be very beneficial for patients suffering from long term diseases such as rheumatic arthritis. Thus, many publications report ES matrices for sustained release of analgesics over periods of hours (for oral delivery) or days (for wound care or cancer therapy), containing APIs such as diclofenac [22], naproxen [23], tramadol [24], ketoprofen [25], lidocaine [26], and steroidal drugs [27]. In addition to pain relief, ES fiber systems have been generated for sustained release of antibacterial agents (e.g. tetracyclines [28], gentamicin [29]), anti-viral drugs (e.g. acyclovir [30]), and anti-cancer APIs (e.g. niclosamide [31], hydroxycamptothecin [32], CPT-11 and SN-38 [33], oxaliplatin [34], 5-fluorouracil [35], paclitaxel [36], and doxorubicin [37, 38]). In the latter case, the formulations are often designed as implants to be placed for instance in a tumor resection site.
Suitable choice of polymer is key to develop an effective sustained-release ES formulation. Appropriate polymers are hydrophobic; they can be stable (in which case drug release occurs by diffusion out of the matrix) or degrade slowly into soluble monomers. Commonly explored biodegradable polymers include PLA [27], PGA, PLGA, and PCL [39, 40] while the non-degradable polyurethane [32], polycarbonate [32], and nylon-6 [41, 42] have also been investigated. Sustained release can additionally be achieved by using natural polymers like silk [43, 44], collagen [45, 46], gelatin [47, 48], alginate [49, 50], cellulose derivatives [23, 51], and chitosan [52, 53], either spinning them alone, or by blending with other components [54,55,56,57,58,59].
A widely observed phenomenon in fibers from mono-axial ES is an initial burst of API release due to the high surface area-to-volume ratio of the fibers and the presence of drug at or near the fiber surface. While this is not an issue with FD-DDSs (where release is intended to be rapid), it can be problematic when seeking to develop sustained release systems. Researchers worldwide have thus probed various strategies to overcome this effect. One strategy is to generate core/shell fibers, where drug loaded within the core has to diffuse through a drug-free polymer shell before reaching the release media [60,61,62,63]. Designing composite ES fibers with drug-loaded nanoparticles can also be used to reduce the initial burst release. Silica nanoparticle (NP)-loaded PLGA fibers [64] and chitosan NPs in PCL fibers have both been explored to this end [65].
Core/shell systems can also be applied to deliver multiple APIs at different rates. Heydari et al. developed core/shell fibers composed of polyglycerol-sebacate as the shell and poly (hydroxybutyrate) elastomer as the core. Simvastatin, a drug known for accelerating diabetic wound healing by enhancing angiogenesis and lymphangiogenesis, was loaded into the fiber core, and the antibacterial drug ciprofloxacin included in the shell [66]. This resulted in a smart wound-healing system, since the fibers gave 60% ciprofloxacin release in 24 h, but simvastatin was released much more slowly [66].
4.3 Zero-Order Release
Therapeutic systems that can release an API at a constant rate are much sought after in order to minimize variations in plasma drug concentrations and help maintain the drug concentration within the therapeutic range [67]. Various strategies have been applied to achieve zero-order release using electrospun formulations. One of the earliest reports of this developed drug-loaded cellulose acetate (CA) nanofibers using a modified coaxial electrospinning process [61]. Core/shell fibers were produced with both compartments made of CA and the model drug KET confined to the core, and were able to provide zero-order release over 96 h [61]. Subsequent approaches using alternative core/shell fiber formulations have also been shown to yield zero-order release profiles [60, 61]. Three-layer fibers from triaxial spinning have additionally been investigated. By preparing a triaxial system with all three compartments made of ethyl cellulose (EC) and with an increasing w/w drug loading moving from the exterior to the core compartment, it was possible to achieve zero-order release of KET over 20 h (Fig. 7.5) [63].
Data on trilayer EC/KET fibers prepared by Yu et al. a Transmission electron micrograph showing the three-layer architecture of the fibers. b The KET release profile. Reprinted with permission from Yu et al. [63]
4.4 Targeted Release
There exist a variety of different pH environments in the body, which can be used to target drug release. For instance, the stomach is acidic (ca. pH 1–2) whereas the small intestine is much closer to neutral. pH-responsive polymers can thus be used to target API release to the small intestine. Polymers such as Eudragit L-100 (EL100) or Eudragit L100-55 (EL100-55) have been exploited for such delayed-release delivery of many drugs. The Eudragits are methacrylate based copolymers; they contain myriad COOH groups which are unionized at low pH, rendering the polymer insoluble. As the pH increases the carboxylate groups ionize to COO−, and the solubility of the polymer increases. Through careful control of the polymer chemical structure, systems can be developed with dissolve at certain distinct pHs: thus, EL100-55 is soluble at pH 5.5 or greater, while EL100 becomes soluble at pH 6.0. DDSs composed of Eudragit are insoluble in the stomach and release the drug only when it reaches the small intestine. Sustained release DDSs targeting delivery of mebeverine hydrochloride (MB-HCl) to the colon have been prepared by electrospinning EL100-55 [11]. Diclofenac sodium-loaded EL100 fibers have also been reported [68].
Beyond the Eudragit materials, a range of other polymers such as hydroxypropyl methyl cellulose phthalate and hydroxypropyl methylcellulose acetate succinate have been explored for targeted release, for instance of erythromycin [69] and theophylline [70]. Natural macromolecules can also be applied to give targeted release. Zein and shellac are both naturally occurring polymers with environment-responsive solubility. Shellac is insoluble under acid conditions but soluble in a neutral environment. Yang et al. observed that electrospun fibers with a shellac shell and diclofenac sodium/PVP core could resist acid erosion and thereby potentially deliver the API specifically to the colon [71].
Electrospun fibers have been extensively explored for applications in cancer therapy [72, 73]. Typically, the tumor microenvironment is at a slightly lower pH than the general physiological pH, and this difference can be used to target drug delivery. Electrospun fibers composed of PVA/PCL [74], and poly (N-isopropylacrylamide-co-methacrylic acid-co-vinylpyrrolidone) [P(NIPAAm-MAA-VP) [75] have been studied for delivery of doxorubicin (DOX) and keratin/PLA fibers for 5-fluorouracil [76]. Cellulose nanocrystal-poly[2-(dimethylamino)ethyl methacrylate] reinforced poly(3-hydroxybutyrate-co-3-hydroxy valerate) systems facilitating both pH and temperature sensitive drug release are additionally reported to have potential for cancer treatment [77, 78]. pH-sensitivity can also be imparted to fibers by the inclusion of additional components with polymers which are not themselves stimuli-responsive. For instance, incorporation of sodium bicarbonate has been employed to generate electrospun systems giving accelerated release under acidic conditions, using polymers such as gelatin/PCL [79] and PLA [80].
4.5 Multi-stage Release
Multi-stage drug release can also be of great benefit, and there are a range of examples of ES formulations which have been developed to provide this. The core–shell nanofiber design can be used to obtain biphasic drug release, using a hydrophilic polymer shell to give an initial burst release of the drug and a hydrophobic core to give a sustained maintenance dose. Using this strategy, Qian et al. were able to develop core/shell EC/PVP fibers with biphasic drug release [81]. In other work, pulsatile release (with bursts of “on” and “off” release) has been reported using blend fibers composed of poly(ethylene oxide), sodium alginate and sodium ibuprofen [82]. These fibers give two stage pulsatile release at pH 3, with the extent of release in the “on” phases and the lag time between them tunable by dint of varying the w/w ratio of the ingredients in the formulation.
5 Electrospinning of Biologicals
Biological products, or ‘biologics’ are a potent group of APIs that include vaccines, cell and gene therapy, and recombinant therapeutic proteins [83]. With recent advancements in biotechnology, biological treatments are becoming key in therapeutic protocols for multiple conditions – most notably perhaps in cancer immunotherapy and tissue engineering. In tandem, interest in electrospun biologic formulations has accelerated in the past few years.
5.1 Proteins
The complex and fragile structures of many therapeutic proteins require advanced delivery methods and careful optimization of formulation and manufacturing conditions. Quite often the protein integrity is compromised by the route of administration, leading to denaturation, aggregation or misfolding. For example, the loss of fragile tertiary structures of proteins, defined by non-covalent interactions, may result in reduced biological activity. Currently, parenteral administration of proteins remains the gold standard as other delivery routes are limited or not yet clinically feasible [9, 84, 85].
The field of pharmaceutical technology has long sought possible solutions to protein formulation challenges, leading to the development of multiple inorganic, lipid or polymer-based formulations. Many biomaterial fabrication processes may negatively impact the stability of the protein being formulated. Typical conventional encapsulation methods, such as spray-drying or hot melt extrusion, require harsh conditions (e.g. application of heat or prolonged contact with solvents) that can potentially cause protein misfolding or aggregation, and consequent loss of activity [9]. Electrospinning could offer a simple route to the fabrication of biocompatible protein-loaded scaffolds that can be easily implanted at the desired site of action. Multiple approaches have been proposed for the incorporation of therapeutic proteins within electrospun materials, including monoaxial and co-axial electrospinning [9]. While monoaxial spinning is attractive in its simplicity, and has been used to develop a number of protein-loaded formulations [86], prolonged exposure to the organic solvent typically required for electrospinning may cause misfolding or aggregation of labile biomolecules like proteins, leading to loss of activity. For this reason, the encapsulation of therapeutic proteins using blend electrospinning is used with caution.
Controlled and sustained protein delivery can be achieved using emulsion electrospinning, where a core–shell structure is formed by electrospinning of a water-in-oil emulsion formed from an aqueous phase (protein) and an organic phase (polymer). When subjected to electric charge, the organic phase (typically containing a volatile solvent) rapidly evaporates. This causes the migration of the aqueous phase (with protein) to the center of the jet, forming core–shell structures with favorable localization of proteins within the center of the fibers/particles, minimizing the risk of the initial burst release of therapeutic cargo. Although the relative simplicity of emulsion electrospinning is attractive from the manufacturability point of view, the protein-polymer blend composition must be carefully optimized to ensure there is no loss of bioactivity resulting from accidental exposure of the biomolecule to the organic solvent. A non-ionic surfactant is often incorporated in the formulation to decrease the contact between protein and organic solvents by forming reverse micelles (emulsions), therefore avoiding denaturation or aggregation [9]. In 2017, Frizzell et al. [2] developed enzyme-loaded electrospun fibers for peroral delivery using surfactant-assisted emulsion electrospinning. Horseradish peroxidase and alkaline phosphatase were blended with EL100 in the presence of the surfactant Tween 20. The nanofibers showed pH-dependent enzyme release patterns, with ~ 5% release at pH 2 and almost 100% at pH 7. Use of EL100 ensured dissolution of the fibers only occurred in neutral and basic pH conditions, leading to protected transit through the stomach and preventing acid- medicated protein degradation.
An alternative to emulsion electrospinning is the co-axial approach. As both solutions are physically separated until the formation of the fiber, protein exposure to organic solvents is limited and therefore accidental degradation can be minimized. The resulting fiber usually holds the protein in the core with a hydrophobic polymeric shell allowing for the gradual release of the cargo at the site of action. A notable example of protein encapsulation within electrospun fibers using this approach has been recently published by Angkawinitwong et al. [87], who explored coaxial electrospinning in the fabrication of bevacizumab-loaded PCL nanofibers for the treatment of age-related macular degeneration in the eye (Fig. 7.6). The study highlighted the importance of the pH of the protein solution. It was found that electrospinning protein solutions at the isoelectric point (pI) of the antibody (where it is uncharged) significantly enhances its in-process stability. Bevacizumab released from the optimal core–shell fibers remained intact and biologically active, confirming the appropriateness of the approach for sustained and localized delivery of antibodies. However, the encapsulation efficiency was negatively affected by electrospinning at the pI of bevacizumab, which was attributed to the lack of electrostatic interactions driving the migration of protein towards the grounded electrode when the antibody is in its uncharged state.
A schematic diagram showing the effect of bevacizumab migration during electrospinning on the fiber properties. a When the protein solution is at pH 6.2, the protein carries a net positive charge; this causes it to migrate uncontrollably from the aqueous core to the polymer shell and become unfolded upon exposure to the shell solvent. The resultant distribution of both intact bevacizumab and unfolded bevacizumab within the core and shell leads to first-order release behavior. b In contrast, when working at pH 8.3, the pI of bevacizumab, the protein is neutral and does not interact with the electric field. This results in zero-order release of intact bevacizumab. Reprinted with permission from Angkawinitwong et al. [87]
5.2 Cells and Extracellular Vesicles
Cell therapies are gaining real momentum in the current landscape of medical innovations, but their clinical translation is often impeded by manufacturing constraints or poor cell survival post-transplantation. Biomaterials are often explored for example to aid stem cell differentiation or trafficking into the desired therapeutic location [88]. Delivering cells directly to the site of action within implantable and biocompatible scaffolds could be an attractive approach for a variety of conditions, such as cardiac repair [89]. Electrospinning has been shown to have significant potential in the generation of such scaffolds.
Cell electrospinning, first introduced by Jayasinghe in 2006 [90], refers to the incorporation of cell suspensions directly into electrospun fibers using a modified co-axial electrospinning apparatus. To ensure sterility, the experiments are often conducted in laminar flow hoods. Probably the most important modification in the setup arises from the fact that cell-containing fiber product cannot be allowed to dehydrate, as this would negatively influence cell viability. For this reason, traditional metal collecting substrates are unsuitable and instead fibers are collected directly into a dish containing cell culture media maintained at 37 ℃. Cell electrospinning has now been tested on a variety of cell lines, and in all cases if the operating parameters are optimized it is possible to maintain cell integrity after fiber generation. It seems that exposure to a high voltage at low current (nanoamperes) during electrospinning does not affect the viability [90, 91] and functionality [89] of processed cells.
It is also important to note that the polymers traditionally used for standard electrospinning often require processing in solvents such as tetrahydrofuran, chloroform or acetone. Such solvents, however, are toxic to living organisms and therefore unsuitable for cell electrospinning. Natural polymers such as alginates, collagens and cellulose may appear more appropriate, but are rarely used due to their poor mechanical properties. Instead, water-soluble synthetic polymers such as PVA or poly(dimethylsiloxane) appear to be more effective for the development of cell-loaded mats [92].
Recently, Trindade et al. [93] tackled the unexplored concept of incorporating extracellular vesicles (EVs) into electrospun patches. EVs are cell-derived nano-sized vesicles enclosed within a lipid bilayer [94, 95]. They are less immunogenic and easier to produce and process than stem cells and therefore hold great potential in tissue engineering applications [93]. High encapsulation efficiencies (> 70%) were obtained, and the EVs observed to retain their therapeutic potency after processing. This suggests that electrospinning could be an effective way of preserving EVs for long-term release formulations.
6 Translation
This chapter has presented a wide range of electrospun drug delivery systems and sought to highlight some of the key benefits of the approach. However, the vast majority of the work performed to date has been undertaken on the lab scale, and the throughput of such experiments is low: typical yields might be 100 mg per hour [96, 97]. This is clearly insufficient for industrial applications, and for electrospun formulations to make it to the market production on the ton scale is required. Recent years have seen rapid development of large-scale electrospinning, and are discussed elsewhere in this volume. Researchers in the pharmaceutical technology field have been quick to explore such high-throughput systems, and it is encouraging to see that, although increasing the production rate tends to result in more irregular fiber morphologies, the functional performance is unaffected [97, 98].
Most electrospun drug delivery systems have been developed using solution spinning. For translation, great attention must be paid to choosing the solvent system. First, there could be health and safety issues pertaining to the use of volatile solvents in large scale production [99]. Second, it is important to ensure that the final product is solvent free, as certain organic solvents (e.g. dimethylformamide or tetrahydrofuran) may result in harmful toxic effects if any residual solvent remains in the fibers [100,101,102]. Batch-to-batch consistency must additionally be assured, to ensure therapy is safe and effective.
Work has begun to implement the quality control procedures required for pharmaceutical applications (so called Good Manufacturing Practice, GMP), and industrial facilities now exist where it is possible to prepare large (ton) amounts of materials under GMP conditions. These recent developments auger well for the future translation of electrospun systems from the lab bench to the clinic [97, 103]. It should be noted however that while high-throughput monoaxial electrospinning is now possible, the scale-up of co-axial and other multi-fluid processes lags significantly behind. Thus, the first electrospun formulations to make it to market are likely to be those which exploit the ASD nature of the fibers to accelerate dissolution rates.
7 Conclusions
This chapter presents a brief overview of the applications of electrospun fibers in drug delivery. We have shown that it is possible to process both small molecule and biological active ingredients without any loss of functionality. A wide range of release profiles can be obtained. Many reports in the literature explore formulations designed to offer very rapid release of a drug cargo, using a hydrophilic and water soluble polymer to overcome problems of low API solubility and provide rapid relief of symptoms. Others use insoluble or slow-dissolving or degrading polymers to give extended release of an API, meaning there is the potential to reduce dosing frequency and improve patient compliance. Zero-order, targeted, and pulsatile release can also be achieved through judicious choice of the polymer carrier. The high stability of small molecule drugs makes them relatively straightforward to incorporate into fibers, while there is significantly more challenge with biologics owing to their instability, particularly upon exposure to organic solvents. Nevertheless, a range of potent electrospun formulations containing proteins and even cells have been developed over recent years.
Elecrospinning thus has versatile applications in drug delivery. However, there are a number of challenges still to be addressed. More work is required to develop methods and provide quality control for high-throughput fiber production, particularly when it comes to multi-fluid processing. Maintaining batch-to-batch consistency and putting in place the quality control guidelines needed to comply with regulatory frameworks will be key for clinical applications. There are a number of researchers, both in academia and industry, who are working on these challenges, and at the time of writing several electrospun products have reached clinical trials (e.g. NCT02409628 or NCT03686800). In the authors’ view therefore, the future for electrospun formulations is bright, and we confidently expect electrospun medicines to emerge in the market in the next few years.
References
Williams, G. R., Raimi-Abraham, B. T., & Luo, C. J. (2018). Nanofibres in drug delivery. UCL Press
Frizzell, H., Ohlsen, T. J., & Woodrow, K. A. (2017). Protein-loaded emulsion electrospun fibers optimized for bioactivity retention and PH-controlled release for peroral delivery of biologic therapeutics. International Journal of Pharmaceutics, 533(1), 99–110. https://doi.org/10.1016/j.ijpharm.2017.09.043
Anderson, J. M., & Shive, M. S. (2012). Biodegradation and biocompatibility of PLA and PLGA microspheres. Advanced Drug Delivery Reviews, 64, 72–82. https://doi.org/10.1016/j.addr.2012.09.004
Chiu, Y.-J., Zhang, Z., Dziemidowicz, K., Nikoletopoulos, C.-G., Angkawinitwong, U., Chen, J.-T., & Williams, R. G. (2020). The effect of solvent vapor annealing on drug-loaded electrospun polymer fibers. Pharmaceutics. https://doi.org/10.3390/pharmaceutics12020139
Labet, M., & Thielemans, W. (2009). Synthesis of polycaprolactone: A review. Chemical Society Reviews, 38(12), 3484–3504. https://doi.org/10.1039/B820162P
Fu, K., Pack, D. W., Klibanov, A. M., & Langer, R. (2000). Visual evidence of acidic environment within degrading poly(lactic-co-glycolic acid) (PLGA) microspheres. Pharmaceutical Research, 17(1), 100–106. https://doi.org/10.1023/A:1007582911958
Houchin, M. L., & Topp, E. M. (2008). Chemical degradation of peptides and proteins in PLGA: A review of reactions and mechanisms. Journal of Pharmaceutical Science, 97 (7), 2395–2404. https://doi.org/10.1002/jps.21176
Castilla-Cortázar, I., Más-Estellés, J., Meseguer-Dueñas, J. M., Escobar Ivirico, J. L., Marí, B., & Vidaurre, A. (2012). Hydrolytic and enzymatic degradation of a poly(ε-caprolactone) network. Polymer Degradation Stability, 97 (8), 1241–1248. https://doi.org/10.1016/j.polymdegradstab.2012.05.038
Dziemidowicz, K., Sang, Q., Wu, J., Zhang, Z., Zhou, F., Lagaron, J. M., Mo, X., Parker, G. J. M., Yu, D.-G., Zhu, L.-M., & Williams, G. R. (2021). Electrospinning for healthcare: Recent advancements. Journal of Materials Chemistry B, 9, 939–951. https://doi.org/10.1039/D0TB02124E
Illangakoon, U. E., Gill, H., Shearman, G. C., Parhizkar, M., Mahalingam, S., Chatterton, N. P., & Williams, G. R. (2014). Fast dissolving paracetamol/caffeine nanofibers prepared by electrospinning. International Journal of Pharmaceutics, 477(1–2), 369–379. https://doi.org/10.1016/j.ijpharm.2014.10.036
Illangakoon, U. E., Nazir, T., Williams, G. R., & Chatterton, N. P. (2014). Mebeverine-loaded electrospun nanofibers: Physicochemical characterization and dissolution studies. Journal of Pharmaceutical Sciences, 103, 283–292. https://doi.org/10.1002/jps.23759
Geng, Y., Zhou, F., Williams, G. R. (2021). Developing and Scaling up fast-dissolving electrospun formulations based on poly(Vinylpyrrolidone) and Ketoprofen. Journal of Drug Delivery Science and Technology, 61, 102138. https://doi.org/10.1016/j.jddst.2020.102138
Rasekh, M., Karavasili, C., Soong, Y. L., Bouropoulos, N., Morris, M., Armitage, D., Li, X., Fatouros, D. G., & Ahmad, Z. (2014). Electrospun PVP-indomethacin constituents for transdermal dressings and drug delivery devices. International Journal of Pharmaceutics, 473(1–2), 95–104. https://doi.org/10.1016/j.ijpharm.2014.06.059
Tort, S., Yıldız, A., Tuğcu-Demiröz, F., Akca, G., Kuzukıran, Ö., & Acartürk, F. (2019). Development and characterization of rapid dissolving ornidazole loaded PVP electrospun fibers. Pharmaceutical Development and Technology, 24(7), 864–873. https://doi.org/10.1080/10837450.2019.1615088
Li, X. Y., Wang, X., Yu, D. G., Ye, S., Kuang, Q. K., Yi, Q. W., & Yao, X. Z. (2012). Electrospun borneol-PVP nanocomposites. Journal of Nanomaterials, 2012, 731382. https://doi.org/10.1155/2012/731382
Dai, X. Y., Nie, W., Wang, Y. C., Shen, Y., Li, Y., & Gan, S. J. (2012). Electrospun emodin polyvinylpyrrolidone blended nanofibrous membrane: A novel medicated biomaterial for drug delivery and accelerated wound healing. Journal of Materials Science. Materials in Medicine, 23(11), 2709–2716. https://doi.org/10.1007/s10856-012-4728-x
Torres-Martínez, E. J., Vera-Graziano, R., Cervantes-Uc, J. M., Bogdanchikova, N., Olivas-Sarabia, A., Valdez-Castro, R., Serrano-Medina, A., Iglesias, A. L., Pérez-González, G. L., Cornejo-Bravo, J. M., & Villarreal-Gómez, L. J. (2020). Preparation and characterization of electrospun fibrous scaffolds of either PVA or PVP for fast release of sildenafil citrate. E-Polymers, 20(1), 746–758. https://doi.org/10.1515/epoly-2020-0070
Akhgari, A., Dezfuli, A. G., Rezaei, M., Kiarsi, M., & Abbaspour, M. (2016). The design and evaluation of a fast-dissolving drug delivery system for Loratadine using the electrospinning method. Jundishapur Journal of Natural Pharmaceutical Products, 11(2), e33613. https://doi.org/10.17795/jjnpp-33613
Li, X., Kanjwal, M. A., Lin, L., & Chronakis, I. S. (2013). Electrospun polyvinyl-alcohol nanofibers as oral fast-dissolving delivery system of caffeine and riboflavin. Colloids Surfaces B Biointerfaces, 103, 182–188. https://doi.org/10.1016/j.colsurfb.2012.10.016
Maver, T., Kurečič, M., Smrke, D. M., Kleinschek, K. S., & Maver, U. (2016). Electrospun nanofibrous CMC/PEO as a part of an effective pain-relieving wound dressing. Journal of Sol-Gel Science and Technology, 79(3), 475–486. https://doi.org/10.1007/s10971-015-3888-9
Ponrasu, T., Chen, B. H., Chou, T. H., Wu, J. J., & Cheng, Y. S. (2021). Fast dissolving electrospun nanofibers fabricated from jelly fig polysaccharide/pullulan for drug delivery applications. Polymers (Basel), 13(2), 1–17. https://doi.org/10.3390/polym13020241
El-newehy, M. H., El-naggar, M. E., Alotaiby, S., El-hamshary, H., Moydeen, M., & Al-deyab, S. (2016). Preparation of biocompatible system based on electrospun CMC/PVA nanofibers as controlled release carrier of diclofenac sodium. Pure and Applied Chemistry, 53(9), 566–573. https://doi.org/10.1080/10601325.2016.1201752
Godakanda, V. U., Li, H., Alquezar, L., Zhao, L., Zhu, L., De Silva, R., Nalin De Silva, K. M., & Williams, G. R. (2019). Tunable drug release from blend poly(vinyl pyrrolidone)—ethyl cellulose nanofibers. International Journal of Pharmaceutics, 562, 172–179. https://doi.org/10.1016/j.ijpharm.2019.03.035
Giannetti, R., Abraham, G. A., & Rivero, G. (2019). The role of emulsion parameters in tramadol sustained-release from electrospun mats. Materials Science & Engineering C, 99, 1493–1501. https://doi.org/10.1016/j.msec.2019.02.085
Hu, J., Li, H. Y., Williams, G. R., Yang, H. H., Tao, L., & Zhu, L. M. (2016). Electrospun poly(n-isopropylacrylamide)/ethyl cellulose nanofibers as thermoresponsive drug delivery systems. Journal of Pharmaceutical Sciences, 105(3), 1104–1112. https://doi.org/10.1016/S0022-3549(15)00191-4
Tseng, Y. Y., Liao, J. Y., Chen, W. A., Kao, Y. C., & Liu, S. J. (2014). Biodegradable poly([D, L]-lactide-co-glycolide) nanofibers for the sustainable delivery of lidocaine into the epidural space after laminectomy. Nanomedicine, 9(1), 77–87. https://doi.org/10.2217/nnm.13.42
Alves, P. E., Soares, B. G., Lins, L. C., Livi, S., & Santos, E. P. (2019). Controlled delivery of dexamethasone and betamethasone from PLA electrospun fibers: A comparative study. European Polymer Journal, 117, 1–9. https://doi.org/10.1016/j.eurpolymj.2019.05.001
Cheng, M., Qin, Z., Hu, S., Dong, S., Ren, Z., & Yu, H. (2017). Achieving long-term sustained drug delivery for electrospun biopolyester nanofibrous membranes by introducing cellulose nanocrystals. ACS Biomaterials Science & Engineering, 3(8), 1666–1676. https://doi.org/10.1021/acsbiomaterials.7b00169
Pisani, S., Dorati, R., Chiesa, E., Genta, I., Modena, T., Bruni, G., Grisoli, P., & Conti, B. (2019). Release profile of gentamicin sulfate from polylactide-co-polycaprolactone electrospun nanofiber matrices. Pharmaceutics, 11(4), 161. https://doi.org/10.3390/pharmaceutics11040161
Wang, M., Hou, J., Yu, D. G., Li, S., Zhu, J., & Chen, Z. (2020). Electrospun tri-layer nanodepots for sustained release of acyclovir. Journal of Alloys and Compounds, 846, 156471. https://doi.org/10.1016/j.jallcom.2020.156471
Dubey, P., & Gopinath, P. (2016). Fabrication of electrospun poly(ethylene oxide)-poly(capro lactone) composite nanofibers for co-delivery of niclosamide and silver nanoparticles exhibits enhanced anti-cancer effects in vitro. Journal of Materials Chemistry B, 4(4), 726–742. https://doi.org/10.1039/c5tb02351c
Luo, X., Xie, C., Wang, H., Liu, C., Yan, S., & Li, X. (2012). Antitumor activities of emulsion electrospun fibers with core loading of hydroxycamptothecin via intratumoral implantation. International Journal of Pharmaceutics, 425(1–2), 19–28. https://doi.org/10.1016/j.ijpharm.2012.01.012
Yohe, S. T., Herrera, V. L. M., Colson, Y. L., & Grinstaff, M. W. (2012). 3D superhydrophobic electrospun meshes as reinforcement materials for sustained local drug delivery against colorectal cancer cells. Journal of Controlled Release, 162(1), 92–101. https://doi.org/10.1016/j.jconrel.2012.05.047
Zhang, J., Wang, X., Liu, T., Liu, S., & Jing, X. (2016). Antitumor activity of electrospun polylactide nanofibers loaded with 5-fluorouracil and oxaliplatin against colorectal cancer. Drug Delivery, 23(3), 794–800. https://doi.org/10.3109/10717544.2014.916768
Hadjianfar, M., Semnani, D., & Varshosaz, J. (2018). Polycaprolactone/chitosan blend nanofibers loaded by 5-fluorouracil: An approach to anticancer drug delivery system. Polymers for Advanced Technologies, 29(12), 2972–2981. https://doi.org/10.1002/pat.4417
Xie, J., & Wang, C. H. (2006). Electrospun micro- and nanofibers for sustained delivery of paclitaxel to treat c6 glioma in vitro. Pharmaceutical Research, 23(8), 1817–1826. https://doi.org/10.1007/s11095-006-9036-z
Habibi, M., Hosseini, S., & Mahboobnia, K. (2019). Simultaneous controlled release of 5-FU, DOX and PTX from chitosan/PLA/5-FU/g-C3N4-DOX/g-C3N4-PTX triaxial nano fibers for breast cancer treatment in vitro. Colloids and surfaces B: Biointerfaces, 179 (November 2018), 495–504.
Anaraki, N. A., Rad, L. R., Irani, M., & Haririan, I. (2015). Fabrication of PLA/PEG/MWCNT electrospun nanofibrous scaffolds for anticancer drug delivery. Journal of Applied Polymer Science, 132(3), 1–9. https://doi.org/10.1002/app.41286
Zhang, A. Q., Li, Y., Yuan, Z., Lin, W., Wong, K. K. Y., Lin, M., Yildirimer, L., Zhao, X., & William, Z. Y. (2017). Electrospun polymeric micro/nanofibrous scaffolds for long-term drug release and their biomedical applications. Drug Discovery Today, 22 (9), 1351–1366. https://doi.org/10.1016/j.drudis.2017.05.007
Budak, K., Sogut, O., & Sezer, U. A. (2020). A review on synthesis and biomedical applications of polyglycolic acid. Journal of Polymer Research, 27, 208. https://springerlink.bibliotecabuap.elogim.com/article/10.1007/s10965-020-02187-1
Sohrabi, A., Shaibani, P. M., Etayash, H., Kaur, K., & Thundat, T. (2013). Sustained drug release and antibacterial activity of ampicillin incorporated poly(methyl methacrylate)-nylon6 core/shell nanofibers. Polymer, 54(11), 2699–2705. https://doi.org/10.1016/j.polymer.2013.03.046
El-Newehy, M. H., Al-Deyab, S. S., Kenawy, E. R., & Abdel-Megeed, A. (2011). Nanospider technology for the production of nylon-6 nanofibers for biomedical applications. Journal of Nanomaterials, 2011, 626589. https://doi.org/10.1155/2011/626589
Farokhi, M., Mottaghitalab, F., Reis, R. L., Ramakrishna, S., & Kundu, S. C. (2020). Functionalized silk fibroin nanofibers as drug carriers: Advantages and challenges. Journal of Controlled Release, 321, 324–347. https://doi.org/10.1016/j.jconrel.2020.02.022
Dadras Chomachayi, M., Solouk, A., Akbari, S., Sadeghi, D., Mirahmadi, F., & Mirzadeh, H. (2018). Electrospun nanofibers comprising of silk fibroin/gelatin for drug delivery applications: Thyme essential oil and doxycycline monohydrate release study. Journal of Biomedical Materials Research—Part A, 106 (4), 1092–1103. https://doi.org/10.1002/jbm.a.36303
Zhang, M., Li, Z., Liu, L., Sun, Z., Ma, W., Zhang, Z., Zhang, R., Sun, D. (2016). Preparation and characterization of vancomycin-loaded electrospun Rana Chensinensis skin collagen/poly(L-lactide) nanofibers for drug delivery. Journal of Nanomaterials, 2016, 9159364. https://doi.org/10.1155/2016/9159364
Huo, P., Han, X., Zhang, W., Zhang, J., Kumar, P., Liu, B. (2021). Electrospun nanofibers of polycaprolactone/collagen as a sustained-release drug delivery system for artemisinin. Pharmaceutics, 13(8), 1228. https://doi.org/10.3390/pharmaceutics13081228
Meng, Z. X., Xu, X. X., Zheng, W., Zhou, H. M., Li, L., Zheng, Y. F., & Lou, X. (2011). Preparation and characterization of electrospun PLGA/gelatin nanofibers as a potential drug delivery system. Colloids Surfaces B Biointerfaces, 84(1), 97–102. https://doi.org/10.1016/j.colsurfb.2010.12.022
Dongargaonkar, A. A., Bowlin, G. L., & Yang, H. (2013). Electrospun blends of gelatin and gelatin-dendrimer conjugates as a wound-dressing and drug-delivery platform. Biomacromolecules, 14(11), 4038–4045. https://doi.org/10.1021/bm401143p
De Silva, R. T., Dissanayake, R. K., Mantilaka, M. M. M. G. P. G., Wijesinghe, W. P. S. L., Kaleel, S. S., Premachandra, T. N., Weerasinghe, L., Amaratunga, G. A. J., & De Silva, K. M. N. (2018). Drug-loaded halloysite nanotube-reinforced electrospun alginate-based nanofibrous scaffolds with sustained antimicrobial protection. ACS Applied Materials & Interfaces, 10(40), 33913–33922. https://doi.org/10.1021/acsami.8b11013
Wongkanya, R., Chuysinuan, P., Pengsuk, C., Techasakul, S., Lirdprapamongkol, K., Svasti, J., & Nooeaid, P. (2017). Electrospinning of alginate/soy protein isolated nanofibers and their release characteristics for biomedical applications. Journal of Science: Advanced Materials and Devices, 2(3), 309–316. https://doi.org/10.1016/j.jsamd.2017.05.010
Khoshnevisan, K., Maleki, H., Samadian, H., Shahsavari, S., Sarrafzadeh, M. H., Larijani, B., Dorkoosh, F. A., Haghpanah, V., Khorramizadeh, M. R. (2018). Cellulose acetate electrospun nanofibers for drug delivery systems: Applications and recent advances. Carbohydrate Polymers, 198, 131–141. https://doi.org/10.1016/j.carbpol.2018.06.072
Jain, R., Shetty, S., Yadav, K. S. (2020). Unfolding the electrospinning potential of biopolymers for preparation of nanofibers. Journal of Drug Delivery Science and Technology, 57, 101604. https://doi.org/10.1016/j.jddst.2020.101604
Farshi Azhar, F., & Olad, A. (2014). A study on sustained release formulations for oral delivery of 5-fluorouracil based on alginate-chitosan/montmorillonite nanocomposite systems. Applied Clay Science, 101, 288–296. https://doi.org/10.1016/j.clay.2014.09.004
Reshmi C. R, Menon, T., Binoy, A., Mishra, N., Elyas K. K., & Sujith, A. (2017). Poly (L-lactide-co-caprolactone)/collagen electrospun mat: potential for wound dressing and controlled drug delivery. International Journal of Polymeric Materials and Polymeric Biomaterials, 148 (13), 148–162. https://doi.org/10.1080/00914037.2016.1252357
López-Calderón, H. D., Avilés-Arnaut, H., Galán Wong, L. J., Almaguer-Cantú, V., Laguna-Camacho, J. R., Calderón-Ramón, C., Escalante-Martínez, J. E., & Arévalo-Niño, K. (2020). Cellulose acetate bi-layer scaffold loaded with gentamicin as possible wound dressing. Polymers, 12(2311), 1–12.
Tsekova, P. B., Spasova, M. G., Manolova, N. E., Markova, N. D., & Rashkov, I. B. (2017). Electrospun curcumin-loaded cellulose acetate/polyvinylpyrrolidone fibrous materials with complex architecture and antibacterial activity. Materials Science and Engineering C, 73, 206–214. https://doi.org/10.1016/j.msec.2016.12.086
Zandi, N., Lotfi, R., Tamjid, E., Shokrgozar, M. A., & Simchi, A. (2020). Core-sheath gelatin based electrospun nanofibers for dual delivery release of biomolecules and therapeutics. Materials Science and Engineering C, 108, 110432. https://doi.org/10.1016/j.msec.2019.110432
Bazhban, M., Nouri, M., Mokhtari, J. (2013). Electrospinning of cyclodextrin functionalized chitosan/PVA nanofibers as a drug delivery system. Chinese Journal of Polymer Science (English Ed.) 31 (10), 1343–1351. https://doi.org/10.1007/s10118-013-1309-5
Cui, Z., Zheng, Z., Lin, L., Si, J., Wang, Q., Peng, X., & Chen, W. (2018). Electrospinning and crosslinking of polyvinyl alcohol/chitosan composite nanofiber for transdermal drug delivery. Advances in Polymer Technology, 37(6), 1917–1928. https://doi.org/10.1002/adv.21850
Yang, G. Z., Li, J. J., Yu, D. G., He, M. F., Yang, J. H., & Williams, G. R. (2017). Nanosized sustained-release drug depots fabricated using modified tri-axial electrospinning. Acta Biomaterialia, 53, 233–241. https://doi.org/10.1016/j.actbio.2017.01.069
Yu, D. G., Li, X. Y., Wang, X., Chian, W., Liao, Y. Z., & Li, Y. (2013). Zero-order drug release cellulose acetate nanofibers prepared using coaxial electrospinning. Cellulose, 20(1), 379–389. https://doi.org/10.1007/s10570-012-9824-z
Liu, X., Yang, Y., Yu, D. G., Zhu, M. J., Zhao, M., & Williams, G. R. (2019). Tunable zero-order drug delivery systems created by modified triaxial electrospinning. Chemical Engineering Journal, 356, 886–894. https://doi.org/10.1016/j.cej.2018.09.096
Yu, D. G., Li, X. Y., Wang, X., Yang, J. H., Bligh, S. W. A., & Williams, G. R. (2015). Nanofibers fabricated using triaxial electrospinning as zero order drug delivery systems. ACS Applied Materials & Interfaces, 7(33), 18891–18897. https://doi.org/10.1021/acsami.5b06007
Song, B., Wu, C., Chang, J. (2012). Controllable delivery of hydrophilic and hydrophobic drugs from electrospun poly(lactic-co-glycolic acid)/mesoporous silica nanoparticles composite mats. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 100(8), 2178–186. https://doi.org/10.1002/jbm.b.32785
Wang, Y., Wang, B., Qiao, W., Yin, T. (2010). A novel controlled release drug delivery system for multiple drugs based on electrospun nanofibers containing nanoparticles. 99 (12), 4805–4811. https://doi.org/10.1002/jps
Heydari, P., Varshosaz, J., Zargar Kharazi, A., & Karbasi, S. (2018). Preparation and evaluation of poly glycerol sebacate/poly hydroxy butyrate core-shell electrospun nanofibers with sequentially release of ciprofloxacin and simvastatin in wound dressings. Polymers for Advanced Technologies, 29(6), 1795–1803. https://doi.org/10.1002/pat.4286
Laracuente, M. L., Yu, M. H., & McHugh, K. J. (2020). Zero-order drug delivery: state of the art and future prospects. Journal of Controlled Release, 327, 834–856. https://doi.org/10.1016/j.jconrel.2020.09.020
Yu, D. G., Xu, Y., Li, Z., Du, L. P., Zhao, B. G., Wang, X. (2014). Coaxial electrospinning with mixed solvents: from flat to round Eudragit L100 nanofibers for better colon-targeted sustained drug release profiles. Journal of Nanomaterials, 2014, 967295. https://doi.org/10.1155/2014/967295
Wang, M., Wang, L., Huang, Y. (2007). Electrospun hydroxypropyl methyl cellulose phthalate (HPMCP)/erythromycin fibers for targeted release in intestine. Journal of Applied Polymer Science, 106 (4), 2177–2184. https://doi.org/10.1002/app.25666
Liu, M., Wang, X., Wang, Y., & Jiang, Z. (2020). Controlled stimulation-burst targeted release by PH-sensitive HPMCAS/theophylline composite nanofibers fabricated through electrospinning. Journal of Applied Polymer Science, 137(7), 1–7. https://doi.org/10.1002/app.48383
Yang, Y. Y., Liu, Z. P., Yu, D. G., Wang, K., Liu, P., & Chen, X. (2018). Colon-specific pulsatile drug release provided by electrospun shellac nanocoating on hydrophilic amorphous composites. International Journal of Nanomedicine, 13, 2395–2404. https://doi.org/10.2147/IJN.S154849
Khodadadi, M., Alijani, S., Montazeri, M., Esmaeilizadeh, N., Sadeghi-Soureh, S., & Pilehvar-Soltanahmadi, Y. (2020). Recent advances in electrospun nanofiber-mediated drug delivery strategies for localized cancer chemotherapy. Journal of Biomedical Materials Research, Part A, 108(7), 1444–1458. https://doi.org/10.1002/jbm.a.36912
Abid, S., Hussain, T., Raza, Z. A., Nazir, A. (2019). Current applications of electrospun polymeric nanofibers in cancer therapy. Materials Science and Engineering C, 97, 966–977. https://doi.org/10.1016/j.msec.2018.12.105
Yan, E., Jiang, J., Yang, X., Fan, L., Wang, Y., An, Q., Zhang, Z., Lu, B., Wang, D., & Zhang, D. (2020). pH-sensitive core-shell electrospun nanofibers based on polyvinyl alcohol/polycaprolactone as a potential drug delivery system for the chemotherapy against cervical cancer. Journal of Drug Delivery Science and Technology, 55, 101455. https://doi.org/10.1016/j.jddst.2019.101455
Salehi, R., Irani, M., Eskandani, M., Nowruzi, K., Davaran, S., & Haririan, I. (2014). Interaction, controlled release, and antitumor activity of doxorubicin hydrochloride from PH-sensitive P(NIPAAm-MAA-VP) nanofibrous scaffolds prepared by green electrospinning. International Journal of Polymeric Materials and Polymeric Biomaterials, 63(12), 609–619. https://doi.org/10.1080/00914037.2013.854234
Zhang, J., Li, J., Xu, C., Xi, Z., Ren, Y., Song, Q., & Lu, S. (2020). Novel PH-sensitive drug-loaded electrospun nanofibers based on regenerated keratin for local tumor chemotherapy. Textile Research Journal, 90(19–20), 2336–2349. https://doi.org/10.1177/0040517520919920
Chen, Y., Abdalkarim, S. Y. H., Yu, H. Y., Li, Y., Xu, J., Marek, J., Yao, J., & Tam, K. C. (2020). Double stimuli-responsive cellulose nanocrystals reinforced electrospun PHBV composites membrane for intelligent drug release. International Journal of Biological Macromolecules, 155, 330–339. https://doi.org/10.1016/j.ijbiomac.2020.03.216
Zhao, X., Yuan, Z., Yildirimer, L., Zhao, J., Lin, Z. Y., Cao, Z., Pan, G., & Cui, W. (2015). Tumor-triggered controlled drug release from electrospun fibers using inorganic caps for inhibiting cancer relapse. Small, 11(34), 4284–4291. https://doi.org/10.1002/smll.201500985
Sang, Q., Li, H., Williams, G., Wu, H., & Zhu, L. M. (2018). Core-shell poly(lactide-Co-ε-caprolactone)-gelatin fiber scaffolds as PH-sensitive drug delivery systems. Journal of Biomaterials Applications, 32(8), 1105–1118. https://doi.org/10.1177/0885328217749962
Zhao, J., Liu, S., Li, B., Yang, H., Fan, C., & Cui, W. (2013). Stable acid-responsive electrospun biodegradable fibers as drug carriers and cell scaffolds. Macromolecular Bioscience, 13(7), 885–892. https://doi.org/10.1002/mabi.201200452
Qian, W., Yu, D. G., Li, Y., Liao, Y. Z., Wang, X., & Wang, L. (2014). Dual drug release electrospun core-shell nanofibers with tunable dose in the second phase. International Journal of Molecular Sciences, 15(1), 774–786. https://doi.org/10.3390/ijms15010774
Kaassis, A. Y. A., Young, N., Sano, N., Merchant, H. A., Yu, D. G., Chatterton, N. P., & Williams, G. R. (2014). Pulsatile drug release from electrospun poly(ethylene oxide)-sodium alginate blend nanofibres. Journal of Materials Chemistry B, 2(10), 1400–1407. https://doi.org/10.1039/c3tb21605e
What are biologics? FAQ. https://www.fda.gov/about-fda/center-biologics-evaluation-and-research-cber/what-are-biologics-questions-and-answers
Zhang, Y., Davis, D. A., AboulFotouh, K., Wang, J., Williams, D., Bhambhani, A., Zakrewsky, M., Maniruzzaman, M., Cui, Z., Williams, R. O. (2021). Novel formulations and drug delivery systems to administer biological solids. Advanced Drug Delivery Reviews, 172, 183–210. https://doi.org/10.1016/j.addr.2021.02.011
Durán-Lobato, M., López-Estévez, A. M., Cordeiro, A. S., Dacoba, T. G., Crecente-Campo, J., Torres, D., Alonso, M. J. (2021). Nanotechnologies for the delivery of biologicals: Historical perspective and current landscape. Advanced Drug Delivery Reviews, 176, 113899. https://doi.org/10.1016/j.addr.2021.113899
Moreira, A., Lawson, D., Onyekuru, L., Dziemidowicz, K., Angkawinitwong, U., Costa, P. F., Radacsi, N., Williams, G. R. (2021). Protein encapsulation by electrospinning and electrospraying. Journal of Controlled Release, 329, 1172–1197. https://doi.org/10.1016/j.jconrel.2020.10.046
Angkawinitwong, U., Awwad, S., Khaw, P. T., Brocchini, S., & Williams, G. R. (2017). Electrospun formulations of bevacizumab for sustained release in the eye. Acta Biomaterialia, 64, 126–136. https://doi.org/10.1016/j.actbio.2017.10.015
Hashemzadeh, M. R., Taghavizadeh Yazdi, M. E., Amiri, M. S., Mousavi, S. H. (2021). Stem cell therapy in the heart: Biomaterials as a key route. Tissue and Cell, 71, 101504. https://doi.org/10.1016/j.tice.2021.101504
Ehler, E., & Jayasinghe, S. N. (2014). Cell electrospinning cardiac patches for tissue engineering the heart. The Analyst, 139(18), 4449–4452. https://doi.org/10.1039/C4AN00766B
Townsend-Nicholson, A., & Jayasinghe, S. N. (2006). Cell electrospinning: A unique biotechnique for encapsulating living organisms for generating active biological microthreads/scaffolds. Biomacromolecules, 7(12), 3364–3369. https://doi.org/10.1021/bm060649h
Jayasinghe, S. N., Irvine, S., & McEwan, J. R. (2007). Cell electrospinning highly concentrated cellular suspensions containing primary living organisms into cell-bearing threads and scaffolds. Nanomedicine, 2(4), 555–567. https://doi.org/10.2217/17435889.2.4.555
Jayasinghe, S. N. (2013). Cell electrospinning: A novel tool for functionalising fibres, scaffolds and membranes with living cells and other advanced materials for regenerative biology and medicine. The Analyst, 138(8), 2215–2223. https://doi.org/10.1039/C3AN36599A
Trindade, R. P., Renault, N., El Harane, N., Menasché, P., Williams, G. R. (2021). Extracellular vesicles can be processed by electrospinning without loss of structure or function. Materials Letters, 282, 128671. https://doi.org/10.1016/j.matlet.2020.128671
Vader, P., Mol, E. A., Pasterkamp, G., Schiffelers, R. M. (2016). Extracellular vesicles for drug delivery. Advanced Drug Delivery Reviews, 106, 148–156. https://doi.org/10.1016/j.addr.2016.02.006
Elsharkasy, O. M., Nordin, J. Z., Hagey, D. W., de Jong, O. G., Schiffelers, R. M., Andaloussi, S. E. L., Vader, P. (2020). Extracellular vesicles as drug delivery systems: Why and how? Advanced Drug Delivery Reviews, 159, 332–343. https://doi.org/10.1016/j.addr.2020.04.004
Radacsi, N., Giapis, K. P., Ovari, G., Szabó-Révész, P., & Ambrus, R. (2019). Electrospun nanofiber-based niflumic acid capsules with superior physicochemical properties. Journal of Pharmaceutical and Biomedical Analysis, 166, 371–378. https://doi.org/10.1016/j.jpba.2019.01.037
Vass, P., Szabó, E., Domokos, A., Hirsch, E., Galata, D., Farkas, B., Démuth, B., Andersen, S. K., Vigh, T., Verreck, G., Marosi, G., & Nagy, Z. K. (2020). Scale-up of electrospinning technology: Applications in the pharmaceutical industry. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology, 12(4), e1611. https://doi.org/10.1002/wnan.1611
Nagy, Z. K., Balogh, A., Démuth, B., Pataki, H., Vigh, T., Szabó, B., Molnár, K., Schmidt, B. T., Horák, P., Marosi, G., Verreck, G., Van Assche, I., & Brewster, M. E. (2015). High speed electrospinning for scaled-up production of amorphous solid dispersion of itraconazole. International Journal of Pharmaceutics, 480(1–2), 137–142. https://doi.org/10.1016/j.ijpharm.2015.01.025
Zhang, B., Yan, X., He, H. W., Yu, M., Ning, X., Long, Y. Z. (2017). Solvent-free electrospinning: Opportunities and challenges. Polymer Chemistry, 8, 333–352. https://doi.org/10.1039/c6py01898j
Li, H., Zhang, Z., Godakanda, V. U., Chiu, Y. J., Angkawinitwong, U., Patel, K., Stapleton, P. G., de Silva, R. M., de Silva, K. M. N., Zhu, L. M., & Williams, G. R. (2019). The Effect of collection substrate on electrospun ciprofloxacin-loaded poly(vinylpyrrolidone) and ethyl cellulose nanofibers as potential wound dressing materials. Materials Science and Engineering C, 104, 1–27. https://doi.org/10.1016/j.msec.2019.109917
Russo, F., Ursino, C., Avruscio, E., Desiderio, G., Perrone, A., Santoro, S., Galiano, F., & Figoli, A. (2020). Innovative poly(vinylidene fluoride) (PVDF) electrospun nanofiber membrane preparation using DMSO as a low toxicity solvent. Membranes, 10(3), 36. https://doi.org/10.3390/membranes10030036
Shawon, J., & Sung, C. (2004). Electrospinning of polycarbonate nanofibers with solvent mixtures THF and DMF. Journal of Materials Science, 9(39), 4605–4613. https://doi.org/10.1023/B:JMSC.0000034155.93428.ea
Omer, S., Forgách, L., Zelkó, R., & Sebe, I. (2021). Scale-up of electrospinning: Market overview of products and devices for pharmaceutical and biomedical purposes. Pharmaceutics, 13(2), 286. https://doi.org/10.3390/pharmaceutics13020286
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Godakanda, V.U., Dziemidowicz, K., de Silva, R.M., de Silva, K.M.N., Williams, G.R. (2022). Electrospun Fibers in Drug Delivery. In: Vaseashta, A., Bölgen, N. (eds) Electrospun Nanofibers. Springer, Cham. https://doi.org/10.1007/978-3-030-99958-2_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-99958-2_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-99957-5
Online ISBN: 978-3-030-99958-2
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)