Abstract
Uvulopalatopharyngoplasty (UPPP) was the first palatal surgery specifically developed for snoring and obstructive sleep apnea (OSA). To date, palatal surgery remains the paragon among various sleep surgeries and is the most commonly used surgery for OSA. During past decades, palatal surgery has evolved from the traditional UPPP, minimally invasive palatal surgery, to a functional reconstruction of the lateral pharyngeal wall and soft palate. The advancement of palatal surgery over the past 40 years has been a multi-dimensional evolution involving the concepts, surgical techniques, and postoperative care in treating sleep-disordered breathing (SDB). Conceptual evolution includes refinements of surgical indications, assessments of obstruction sites, and surgical limitations pertinent to clinical outcomes. Technical evolution comprises the suspension of muscle to maximize the velopharyngeal airspace and hybrid procedure to minimize the complications. Evolved postoperative care ensures better recovery after surgery and integrated treatment. In order to achieve comprehensive safety and efficacy in treating SDB patients, palatal surgery addressing the velopharyngeal obstruction continues to evolve.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
1.1 Introduction
Obstructive sleep apnea (OSA) is a chronic and prevalent disease associated with major adverse cardiac events, neurocognitive impairments, obesity, and other morbidities [1,2,3,4]. Mainstream therapy for OSA includes continuous positive airway pressure (CPAP), oral appliance, and surgery [5, 6]. CPAP is the first-line and gold standard for OSA. However, modest acceptance and low long-term compliance limit the application of CPAP in OSA patients [7]. Surgery is an alternative and salvage treatment for patients who are unwilling or intolerant to CPAP therapy [7]. Furthermore, surgery is the only treatment modality to improve OSA without the use of a device. Among various sleep surgeries, UPPP was the first surgical procedure specifically designed to treat snoring and OSA [6]. Traditional UPPP includes tonsillectomy and excision of the redundant pillars, soft palate, and uvula [6]. Although improvement in clinical symptoms, UPPP was criticized for its unacceptable pain, high complications, and low success rate [8]. Therefore, modification of UPPP has been undergoing in the past four decades. The first part of this article elaborates on the historical evolution of palatal surgery regarding key differences in surgical procedures from time to time. Conceptual evolution is then discussed in terms of surgical indication (why to treat), obstruction site (how to diagnose), and treatment endpoint (when to stop). Technical evolution dives into details of cutting-edge and state-of-the-art surgical techniques. Finally, the evolution of postoperative care is emphasized on enhanced recovery after surgery and integrated treatment to fulfill the quality of care and long-term outcomes.
1.2 Historical Evolution
-
1.
On the horizon. Resection of the soft palate triangle paramedian to the uvula has been implemented for snoring in the 1950s [9, 10]. Amputation of the uvula (uvulectomy) was performed to ameliorate snoring in the 1970s [11]. Theses techniques implied the critical relationship between the velum and snoring, which inspired following palatal surgeries.
-
2.
In-between. Tracheostomy via permanent stoma was the model treatment of Pickwickian syndrome and OSA with severe daytime sleepiness in the 1960s [12, 13]. However, related comorbidities and the development of non-invasive CPAP therapy prohibit its clinical use [14].
-
3.
Origin. UPPP developed by Fujita in the 1980s was the surgical milestone in treating snoring and OSA [6]. Traditional UPPP aimed to enlarge and stabilize the oropharyngeal airway and consequently prevented its collapse. Simmons advocated removing the soft palate as much as possible to maximize airspace and that became a stereotype of UPPP (classic UPPP) [15]. Clinical outcomes of UPPP revealed significant improvement of subjective symptoms in conjunction with incongruous changes in polysomnography [16].
-
4.
Mini-invasive procedure. Laser-assisted uvulopalatoplasty (LAUP), utilizing a CO2 laser to vaporize the vibrating uvula and soft palate, led to a series of office-based anti-snore procedures in the1990s [17]. Despite LAUP improved habitual snoring in the short term, the residual fibrotic palatal tissue narrowed the velopharyngeal airway and exacerbated OSA. Radiofrequency was introduced as interstitial thermotherapy to the soft palate without major complications in the late 1990s [18]. The caveat of RF was that it might require repetitive treatments to achieve desirable results due to decayed effects over time [19]. Pillar implant was introduced to stiffen the soft palate via a single application in the early 2000s [20]. It could reduce snoring in primary snorers; however, a comprehensive treatment approach was required to tackle more severe SDB patients [21].
-
5.
Reconstruction of the lateral pharyngeal wall (pharyngoplasty). Several modifications of the UPPP technique were implemented to prevent airway collapse by stabilizing the lateral pharyngeal wall. Procedures such as lateral pharyngoplasty, expansion sphincter pharyngoplasty, Z-palatoplasty, and relocation pharyngoplasty had been published in the 2010s [22,23,24,25].
-
6.
Suspension technique. More palatal procedures attempted to further enlarge the velopharyngeal airspace by suspending the palatopharyngeus muscle to the pterygomandibular raphe. Barbed Roman blinds technique, barbed anterior pharyngoplasty, barbed reposition pharyngoplasty, suspension palatoplasty, barbed suspension pharyngoplasty, and omni-suspension were proposed in recent years [26,27,28,29,30,31,32].
1.3 Conceptual Evolution
Conceptual changes of palatal surgery for OSA could be discussed in three perspectives: the surgical aim (why to treat), obstruction site (how to diagnosis), and treatment endpoint (when to stop).
Surgical Aim
not only for OSA but holistic care of comorbidities
OSA is a chronic and age-related disease; surgical intervention for OSA is more likely to improve than cure the disease. Evidence showed that palatal surgery for OSA significantly alleviated clinical symptoms, prevented cardiovascular disease, improved disease severity but rarely cured it [8, 33,34,35]. Recent evidence revealed that OSA patients had a higher prevalence of vertigo, tinnitus, sudden deafness, normal-tension glaucoma, infection rate, Alzheimer’s disease than the matched non-OSA population [36,37,38,39]. Therefore, the purposes of palatal surgery are to improve quality of life, lessen disease severity, ameliorate comorbidities, and prevent major complications in OSA patients. In addition, morbid obesity may not be absolute surgical contraindication if combined airway and bariatric surgeries are considered to improve both OSA and obesity.
Obstruction Site Evaluation
not only DISE but interventional DISE
Traditional assessments of the airway obstruction are implemented in wakefulness, which cannot reflect the obstruction site during sleep [40]. Drug-induced sleep endoscopy (DISE) simulates the dynamic changes of the airway during pharmacologically induced sleep, and identification of obstruction sites of the airway becomes accessible. Therefore, DISE reasonably becomes one of the prerequisites in surgical planning for OSA patients [41]. Conversely, the effectiveness of palatal surgery in the setting of multi-level surgery for OSA remains unclear due to the complexity of aerodynamic interactions between different domains of the upper airway, such as velopharyngeal, oropharyngeal, and hypopharyngeal spaces. Interventional DISE by placing a nasopharyngeal tube during exam demonstrated that 74% of patients had partial improvement, and 35% had complete collapse resolution [42]. Reductions in the collapse were observed at sites of the lateral wall (86%), epiglottis (55%), and tongue base (50%) [42]. This finding supported that the patency of retropalatal airspace could potentially reduce negative pharyngeal pressure and alleviated other upper airway obstruction sites. As a result, interventional DISE can provide a tailor-made surgical plan for OSA patients with or without multi-level obstructions.
Surgical Endpoint
not only AHI but comprehensive and preventive effect
Typically, the success or failure of sleep surgery for OSA is judged by changes in AHI pre- and postoperatively [8]. However, OSA is a chronic disease associated with other comorbidities; thus, it is more realistic to intervene and improve sleep apnea in a timely fashion than pursue a statistical cure. Also, research has revealed that the change in AHI is not always consistent with the improvement of clinical symptoms, which are patients’ primary concerns. Therefore, the reasonable goal in treating OSA should be to improve the disease in terms of AHI reduction, ameliorating symptoms, and minimizing unfavorable clinical outcomes. In summary, the endpoint of palatal surgery for OSA is suggested as follows: (1) significant improvement of clinical symptoms (snoring and daytime sleepiness), (2) correction of OSA severity to “mild” (AHI < 15/h), (3) decreased profile of biomarkers indicating risks of coronary artery/cardiovascular diseases [43, 44], and (4) patient’s satisfaction of surgical results.
1.4 Technical Evolution
Through excision of the “redundant” soft palate, classical UPPP incurred severe postoperative pain and velopharyngeal insufficiency that further jeopardized salvage use of CPAP in surgical failure patients due to mouth leak [15, 45]. Studies showed that the lateral pharyngeal wall collapse was the crucial factor contributing to OSA [46]. To address this issue, the technical evolution of palatal surgery initially started as “functional reconstruction” of the lateral pharyngeal wall, which included lateral pharyngoplasty [22] and relocation pharyngoplasty [25], for example. However, suboptimal stabilization/expansion of the velopharynx resulted from the reconstructive-driven procedures because of the lack of structural support of the velum. Therefore, the evolution of palatal surgery transitioned to the concept of “suspension,” which suspended the pharyngeal muscle, especially the palatopharyngeal muscle, to the pterygomandibular raphe as the anchor [29]. The Barbed Roman blinds technique was first introduced [26, 27], and a series of Barbed variants were developed to cope with different patterns of palatal obstructions [28,29,30,31].
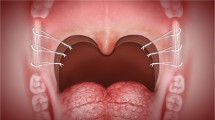
Furthermore, the thesis of tissue-specific hybrid surgery was postulated based on the suspension technique. Physiological hybrid palatal surgery could be constructed following histologic gradation: (1) mucosa: preservation, (2) lymphoid tissue: excision, (3) adipose tissue: ablation, and (4) muscle: suspension [47] (Fig. 1.1). Preservation of the mucosa is crucial to facilitate wound healing, lessen suture tension to reduce wound dehiscence, and maintain submucosal gland secretion to prevent dryness of the mouth. Tonsil is the only tissue to be excised to widen the oropharyngeal space and facilitate the lateral pharyngeal wall reconstruction. Also, deposition of redundant adipose tissue at the periuvular area and supratonsillar fossa can narrow the velopharyngeal airspace and impede the suspension procedure of pharyngeal muscles. Ablation of pharyngeal adipose tissue can be implemented in supratonsillar fossa after tonsillectomy via electrocautery (cutting mode) under assistance of endoscopy and in thick soft palate (thickness > 1 cm) via radiofrequency or coblation (ablation mode 5). Suspension of pharyngeal muscle via omni-suspension technique is pivotal in the hybrid reconstruction of palatal surgery. Omni-suspension involves stay suture of the palatopharyngeus muscle, figure-of-eight styled bundle suture of the pterygomandibular raphe, suspending the muscle to the raphe, and three suspension sutures toward anterior, middle, lateral directions [32] (Fig. 1.2). Noteworthy, omni-suspension is particularly helpful in OSA patients with concentric velopharyngeal collapse during DISE examination.
1.5 Postoperative Evolution
Postoperative inpatient care following palatal sleep surgery generally involves the delivery of humidified oxygen, positional therapy by elevating the cranial end of the bed, prophylactic antibiotics, analgesics, and ice packing on the submental area [48]. A comprehensive therapy program, Enhanced Recovery After Surgery (ERAS), has been proposed for head and neck surgery and sleep surgery to improve the quality of postoperative care [49, 50]. The ERAS for sleep surgery is a multidisciplinary measure composed of preoperative education, lifestyle adjustment (cessation of smoking and alcohol, if any), adequate nutritional support, evaluation of carbohydrate and body fluid intake, cooperation with anxiety, postoperative pain management of multimodal analgesics, and early swallowing rehabilitation and ambulation. Research showed a significantly lower overall complication rate and incidence of fever in ERAS-applied pediatric OSA patients undergoing adenotonsillectomy [50]. Furthermore, pediatric patients with ERAS reported less postoperative pain, had a better dietary intake, and were found with lower preoperative anxiety scores [50]. These suggested that the ERAS program could reduce physical and psychological burden perioperatively in OSA patients.
It is not uncommon that the clinical outcome of palatal surgery may be compromised over time owing to aging, circadian rhythm sleep disorders (delayed/ advanced sleep phase disorder, sleep deprivation, shift work sleep disorder) [51], open mouth breathing, supine sleeping position, and increased body weight. Therefore, integrated treatment is much needed to enhance short- and long-term outcomes. Avoidance of delayed sleep phase or sleep deprivation is essential to maintain the physiological muscle tone of the soft palate and tongue, which in turn counteracts the collapsibility during sleep. In addition to the effects of palatal suspension procedures, the retropalatal airspace is further stabilized by the constant airflow from the nasal cavity through the velopharynx during sleep. Therefore, it is crucial to control nasal obstruction, prevent open mouth breathing (possibly taping the mouth), and apply oropharyngeal myofunctional therapy to facilitate nocturnal nasal breathing [52, 53]. Positional therapy (lateral sleeping position) also can reduce snoring and sleep apnea by enlarging and maintaining the lateral dimension of the retropalatal space postoperatively [54]. Bodyweight reduction is beneficial for cardiopulmonary function, and the airspace can be less obstructive by the reduction of pharyngeal fat [55]. Figure 1.3 illustrates the overview of integrated treatment of palatal surgery for OSA: (1) baseline velopharyngeal obstruction, (2) hybrid reconstruction of the velopharyngeal airway, (3) restoration of the velopharyngeal airflow, and (4) rehabilitation of the oropharyngeal muscle.
1.6 Summary
In this article, the evolution of palate surgery was discussed in terms of concept, technique, and postoperative care. The palatal surgery remains the key procedure in treating snoring and OSA, and the suspension of the pharyngeal muscle enlarges and further stabilizes the velopharyngeal airspace. The chapter on palatal surgery is still evolving, rooting from physiological hybrid surgery and combined operation, multidisciplinary integrated treatment, and ultimately to the holistic care for OSA patients.
References
Young T, Palta M, Dempsey J, et al. The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med. 1993;328:1230–5.
Strollo PJ, Roger RM. Obstructive sleep apnea. N Engl J Med. 1996;334:99–104.
Yaggi HK, Concato J, Kernan WN, Lichtman JH, Brass LM, Mohsenin V. Obstructive sleep apnea as a risk factor for stroke and death. N Engl J Med. 2005;353:2034–41.
Bradley TD, Floras JS. Obstructive sleep apnoea and its cardiovascular consequences. Lancet. 2009;373:82–93.
Sullivan CE, Issa FG, Berthon-Jones M, et al. Reversal of obstructive sleep apnoea by continuous positive airway pressure applied through the nares. Lancet. 1981;1(8225):862–5.
Fujita S, Conway W, Zorick F, et al. Surgical correction of anatomic abnormalities in obstructive sleep apnea syndrome: Uvulopalatopharyngoplasty. Otolaryngol Head Neck Surg. 1981;89:923–34.
Li HY, Lee LA, Tsai MS, et al. How to manage continuous positive airway pressure (CPAP) failure -hybrid surgery and integrated treatment. Auris Nasus Larynx. 2020;2020(47):335–42.
Sher AE, Schechtman KB, Piccirrillo JF. The efficacy of surgical modifications of the upper airway in adults with obstructive sleep apnea syndrome. Sleep. 1996;19:156–77.
Yaremchuk K, Garcia-Rodriguez L. The history of sleep surgery. In: Lin HC, editor. Sleep-related breathing disorders. Advanced Otorhinolaryngology, vol. 80. Basel: Karger; 2017. p. 17–21.
Heinberg CJ. A surgical procedure for the relief of snoring. Eye Ear Nose Throat Mon. 1955;34:389.
Robin I. Snoring. Proc R Soc Med. 1968;61:575–82.
Weitzman ED, Pollack CP, Borowiecki B. Hypersomnia-sleep apnea due to micrognathia: reversal by tracheoplasty. Arch Neurol. 1978;35:392–5.
Fee WE Jr, Ward PH. Permanent tracheostomy: a new surgical technique. Ann Otol Rhinol Laryngol. 1977;86:635–8.
Conway WA, Victor LD, Magilligan DJ Jr, Fujita S, Zorick FJ, Roth T. Adverse effects of tracheostomy for sleep apnea. JAMA. 1981;246:347–50.
Simmons FB, Guilleminault C, Silvestri R. Snoring and some obstructive sleep apnea can be cured by oropharyngeal surgery. Arch Otolaryngol. 1983;109:503–7.
Li HY. Palatal surgery for obstructive sleep apnea: from ablation to reconstruction. Sleep Med Clin. 2019;14:51–8.
Kamami YV. Laser CO2 for snoring. Preliminary results. Acta Otorhinolaryngol Belg. 1990;44(4):451–6.
Powell NB, Riley RW, Troell RJ, et al. Radiofrequency volumetric tissue reduction of the palate in subjects with sleep-disordered breathing. Chest. 1998;113:1163–74.
Li KK, Powell NB, Riley RW, et al. Radiofrequency volumetric reduction of the palate: an extended follow-up study. Otolaryngol Head Neck Surg. 2000;122:410–4.
Ho WK, Wei WI, Chung KF. Managing disturbing snoring with palatal implants: a pilot study. Arch Otolaryngol Head Neck Surg. 2004;130:753–8.
Friedman M, Vidyasagar R, Bliznikas D, et al. Patient selection and efficacy of pillar implant technique for treatment of snoring and obstructive sleep apnea/hypopnea syndrome. Otolaryngol Head Neck Surg. 2006;134:187–96.
Cahali MB. Lateral pharyngoplasty: a new treatment for obstructive sleep apnea hypopnea syndrome. Laryngoscope. 2003;113:1961–8.
Pang KP, Woodson BT. Expansion sphincter pharyngoplasty: a new technique for the treatment of obstructive sleep apnea. Otolaryngol Head Neck Surg. 2007;137:110–4.
Friedman M, Ibrahim HZ, Vidyasagar R, et al. Z-palatoplasty (ZPP): a technique for patients without tonsils. Otolaryngol Head Neck Surg. 2004;131:89–100.
Li HY, Lee LA. Relocation pharyngoplasty for obstructive sleep apnea. Laryngoscope. 2009;119:2472–7.
Mantovani M, Minetti A, Torretta S, Pincherle A, Tassone G, Pignataro L. The velo-uvulo-pharyngeal lift or “roman blinds” technique for treatment of snoring: a preliminary report. Acta Otorhinolaryngol Ital. 2012;32:48–53.
Mantovani M, Minetti A, Torretta S, Pincherle A, Tassone G, Pignataro L. The “Barbed Roman Blinds” technique: a step forward. Acta Otorhinolaryngol Ital. 2013;33:128.
Salamanca F, Costantini F, Mantovani M, Bianchi A, Amaina T, Colombo E, et al. Barbed anterior pharyngoplasty: an evolution of anterior palatoplasty. Acta Otorhinolaryngol Ital. 2014;34:434–8.
Vicini C, Hendawy E, Campanini A, et al. Barbed reposition pharyngoplasty (BRP) for OSAHS: a feasibility, safety, efficacy and teachability pilot study. “We are on the giant’s shoulders”. Eur Arch Otorhinolaryngol. 2015;272:3065–70.
Barbieri M, Missale F, Incandela F, et al. Barbed suspension pharyngoplasty for treatment of lateral pharyngeal wall and palatal collapse in patients affected by OSAHS. Eur Arch Otorhinolaryngol. 2019;276:1829–35.
Li HY, Lee LA, Kezirian EJ, Nakayama M. Suspension palatoplasty for obstructive sleep apnea—a preliminary study. Sci Rep. 2018;8:4224. https://doi.org/10.1038/s41598-018-22710-1.
Shen SC, Li HY. State-of-the art in reconstructive palatal surgery techniques for obstructive sleep apnea. Curr Sleep Med Rep. 2020;6:67–75.
Li HY, Chen NH, Shu YH, et al. Changes of quality of life and respiratory disturbance after extended uvulopalatal flap surgery in patients with obstructive sleep apnea. Arch Otolaryngol Head Neck Surg. 2004;130:195–200.
Li HY, Huang YS, Chen NH, et al. Mood improvement after surgery for obstructive sleep apnea. Laryngoscope. 2004;116:1098–102.
Li HY, Lee LA, Yu JF, et al. Changes of snoring sound after relocation pharyngoplasty for obstructive sleep apnoea: the surgery reduces mean intensity in snoring which correlates well to apnoea/hypopnoea index. Clin Otolaryngol. 2015;40:98–105.
Tsai MS, Lee LA, Tsai YT, et al. Sleep apnea and risk of vertigo: a nationwide population-based cohort study. Laryngoscope. 2018;128:763–8.
Li HY, Tsai MS, Huang CG, et al. Alterations in Alzheimer’s disease-associated gene expression in severe obstructive sleep apnea patients. J Clin Med. 2019;8:1361. https://doi.org/10.3390/jcm8091361.
Tsai MS, Li HY, Huang CG, et al. Risk of Alzheimer's disease in obstructive sleep apnea patients with or without treatment: real-world evidence. Laryngoscope. 2020;130:2292–8.
Ding MC, Hsu CM, Liu YC, et al. Deep neck infection risk in patients with sleep apnea: real-world evidence. Int J Environ Res Public Health. 2021;18:3191. https://doi.org/10.3390/ijerph18063191.
Certal VF, Pratas R, Guimaraes L, et al. Awake examination versus DISE for surgical decision making in patients with OSA: a systemic review. Laryngoscope. 2016;126:768–74.
Kezirian EJ, Hohenhorst W, de Vries N. Drug-induced sleep endoscopy: the VOTE classification. Eur Arch Otorhinolaryngol. 2011;268:1233–6.
Victores AJ, Olson K, Takashima M, Alford BR. Interventional drug-induced sleep endoscopy: a novel technique to guide surgical planning for obstructive sleep apnea. J Clin Sleep Med. 2017;13:169–74.
Lee LA, Chen NH, Huang CG, Lin SW, Fang TJ, Li HY. Patients with severe obstructive sleep apnea syndrome and elevated high-sensitivity C-reactive protein need priority treatment. Otolaryngol Head Neck Surg. 2010;143:72–7.
Lee LA, Huang CG, Chen NH, Wang CL, Fang TJ, Li HY. Severity of obstructive sleep apnea syndrome and high-sensitivity C-reactive protein reduced after relocation pharyngoplasty. Otolaryngol Head Neck Surg. 2011;144:632–8.
Mortimore IL, Bradley PA, Murray JA, et al. UPPP may compromise nasal CPAP therapy in sleep apnea syndrome. Am J Respir Crit Care Med. 1996;154:1759–62.
Schwab RJ, Gupta KB, Gefter WB, et al. Upper airway and soft tissue anatomy in normal subjects and patients with sleep disordered breathing: significance of the lateral pharyngeal walls. Am J Respir Crit Care Med. 1995;152:1673–89.
Li HY, Lee LA, Hsin LJ, et al. Intrapharyngeal surgery with integrated treatment for obstructive sleep apnea. Biom J. 2019;42:84–92.
Li HY. Uvulopalatopharyngoplasty: patient selection and effects on airway. Chapter 33. In: Sleep apnea and snoring. 2nd ed; 2019. p. 191–7.
Jandali DB, Vaughan D, Eggerstedt M, et al. Enhanced recovery after surgery in head and neck surgery: reduced opioid use and length of stay. Laryngoscope. 2020;130:1227–32.
Zhang Y, Liu DW, Chen XM, Ma JH, Song XC. An enhanced recovery program improves the comfort and outcomes in children with obstructive sleep apnea undergoing adenotonsillectomy: a retrospective historical control study. Clin Otolaryngol. 2021;46:249–55.
Butler MP, Smales C, Wu H, et al. The circadian system contributes to apnea lengthening across the night in obstructive sleep apnea. Sleep. 2015;38:1179–801.
Camacho M, Guilleminault C, Wei JM, et al. Oropharyngeal and tongue exercises (myofunctional therapy) for snoring: a systematic review and meta-analysis. Eur Arch Otorhinolaryngol. 2018;275:849–55.
Camacho M, Certal V, Abdullatif J, et al. Myofunctional therapy to treat obstructive sleep apnea: a systematic review and meta-analysis. Sleep. 2015;38:669–75.
Li HY, Cheng WN, Chuang LP, et al. Positional dependency and surgical success of relocation pharyngoplasty among patients with severe obstructive sleep apnea. Otolaryngol Head Neck Surg. 2013;149:506–12.
Romero-Corral A, Caples SM, Lopez-Jimenez F, Somers VK. Interaction between obesity and obstructive sleep apnea. Chest. 2010;137:711–9.
Acknowledgments
The author would like to express the deepest gratitude to Linkou-Chang Gung Memorial Hospital for long-term support by grants in our sleep research. The authors also thank Dr. Yi-Chan Lee, Dr. Chien-Han Tsao, and Dr. Ying-Shuo Hsu for their extensive consultation to this chapter.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Li, HY., Lee, LA., Tsai, MS., Shen, SC. (2022). Evolution of Palatal Surgery for Sleep-Disordered Breathing. In: Vicini, C., Salamanca, F., Iannella, G. (eds) Barbed Pharyngoplasty and Sleep Disordered Breathing. Springer, Cham. https://doi.org/10.1007/978-3-030-96169-5_1
Download citation
DOI: https://doi.org/10.1007/978-3-030-96169-5_1
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-96168-8
Online ISBN: 978-3-030-96169-5
eBook Packages: MedicineMedicine (R0)