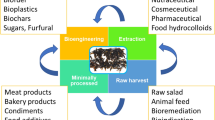
Abstract
Seaweeds have been consumed worldwide throughout coastal areas since the start of human civilization. During the last decades, the interest in seaweed consumption as a nutraceutical or functional food has witnessed an upsurge, concurrently research has evolved, and consumer awareness rose. Red macroalgae (phylum Rhodophyta) can be directly consumed, as raw ingredient. Or, alternatively, extractions can be performed in order to isolate some bioactive compounds (i.e.: polysaccharides, pigments, phenolic compounds, vitamins, lipids and proteins) with food industry applications, and present dietary benefits beyond their macro- and micronutrient content. In this book chapter, the reader can find a compilation of the nutritional characteristics of red seaweeds as a food resource and their health-promoting properties.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Seaweeds are currently seen as promising species for providing new biologically active compounds to produce novel foods due to the large variety of compounds they contain, thus contributing to the development of nutraceutical food products (Kim 2011; Villanueva et al. 2014; Shama et al. 2019). Previous studies have shown that several seaweed-based compounds can improve human health by reducing symptoms of several diseases like cancer, asthma, diabetes, autoimmune, ocular, or cardiovascular (Lopes et al. 2013; Alves et al. 2018; Tanna and Mishra 2018). Seaweeds have been seen as a feedstock for bioactive molecules that can be incorporated in the daily diet as a supplement to promote human health, thus being considered nutraceutical food products (Alwaleed 2019; Shama et al. 2019).
Among the algal phyla or classes, Chlorophyta (green), Phaeophyceae (brown) and Rhodophyta (red), the red seaweeds group contains great biodiversity (Torres et al. 2019). Red seaweeds contain pigments (i.e., phycoerythrin) that can be employed in food industry, as a natural substitute of synthetic food colorants, with antioxidant bioactivity (Ganesan and Shanmugam 2020). Besides that, these seaweeds present a rich nutritional profile, providing several important molecules, such as fatty acids, vitamins, essential amino acids, and minerals, through their direct incorporation in the daily diet. For instance, red seaweeds can contain up to 26 mg kg−1 of Vitamin E (Irene et al. 2018); and among the three phyla, Rhodophyta algal species exhibits the highest protein content (Černá 2011). Moreover, red seaweeds present several essential amino acids, exhibiting almost the same amount of plant-based protein sources (Barbier et al. 2019). For example, the species Pyropia/Porphyra has a score of 0.91 (on a scale between 0 and 1, whereas egg protein has a score of 1) (Murata and Nakazoe 2001). Furthermore, seaweeds lipidic profile, composed essentially by polyunsaturated fatty acids (ω-6 and ω-3), makes them an important source of these compounds, which humans acquire only through the daily diet (Belattmania et al. 2018).
However, their nutritional profile varies according to the harvest season, geolocation, and other biotic (i.e., temperature, salinity, light intensity, nutrients availability, and pollutants concentration) and abiotic parameters (i.e., predators). So, it is a current challenge to guarantee the safety and quality of seaweeds traded (Tanna and Mishra 2019; Cho and Rhee 2020; Rosa et al. 2020). For this reason, aquaculture techniques and technologies have evolved to tackle this need (Campbell et al. 2020). Currently, the most industrially exploited red seaweeds species are Gracilaria spp., Kappaphycus alvarezii, Porphyra/Pyropia spp. and Eucheuma denticulatum, being cultivated in several coastal countries.
Red seaweeds are pivotal for phycocolloids industry, for agar and carrageenan extraction, being widely employed in the food industry as thickener or emulsifier agent (Kraan 2012; Garcia-Vaquero et al. 2017). Furthermore, their physical capacity to emulsify and preserve water improves their technological functionality as food ingredients (Ruocco et al. 2016). The direct addition of seaweeds or their polysaccharides to meat and plant-based food products will improve nutritional, structural, antimicrobial, organoleptic, and shelf-life characteristics of these food products (Shannon and Abu-Ghannam 2019). Moreover, these phycocolloids can help to reduce glucose levels (BNF 2016). Besides the mentioned sulfated polysaccharides, porphyran can also be founded in some red seaweeds species, such as Porphyra/Pyropia sp., promoting health benefits, such as the reduction of cholesterol levels (Tsuge et al. 2004).
These polysaccharides, such as agar and carrageenan present several biological activities as it was forementioned, however their bioactivity varies according to their molecular weight, sulphation level and the quantity of sulphate esters groups present in the molecule (Tsuge et al. 2004; Cunha and Grenha 2016). Moreover, different seaweed species produce chemically distinct polysaccharides, and this can also be affected by the extraction method selected. Commonly, carrageenophytes contains a higher concentration of sulphate groups, in comparison to the agarophytes (Zhong et al. 2020). Still, carrageenan chemical structure is very heterogeneous, being the most economically important the carrageenan iota, lambda and kappa (Necas and Bartosikova 2013). So, what distinguish these different types of carrageenan is mainly their sulphate esters content and their position in the molecule (Sangha et al. 2015).
The goal of this book chapter is to highlight the role of red seaweeds for food industry as a key element for the development of innovative products with nutraceutical properties for the food industry as human health promoters.
2 Red Seaweed Nutritional Characteristics
The red seaweeds nutritional values are very dispersed due to a high number of species, and due to the variation of abiotic factors (salinity, nutrients, pH, luminosity, water temperature, profundity). However, these seaweeds can have an internal ratio of the nutrient composition (Table 13.1). Several eastern countries traditionally consume seaweeds as food (mainly Japan, China and Korea) (Dawes 1995), due to its high nutritional value as a source of proteins, carbohydrates, vitamins and minerals (Pereira 2011; Leandro et al. 2020a). Moreover, the seaweeds have interesting secondary metabolites with nutraceutical properties (Table 13.2).
These bioactivities are observed in the most cultivated species of red seaweeds, according to Food and Agriculture Organization (FAO) data (FAO 2020): Gracilaria spp. , Kappaphycus alvarezii , Porphyra spp. /Porphyra tenera and Eucheuma denticulatum . Moreover, in 2018 32.4 million tons of seaweeds were produced. In 2016, the volume of aquatic plants collected or produced was 31.2 million tons, where practically all this production was related to seaweeds, with growth from 13.5 million tons in 1995 to over 30 million tons in 2016 (FAO 2018). Also, according to FAO (Ferdouse et al. 2018), some red seaweed species, such as Porphyra/Pyropia spp. (Rhodophyta) were produced in the East and Southeast Asia, are produced almost exclusively for direct human food consumption. Indonesia increased seaweed production from less than four million tons in 2010 to more than 11 million tons in 2015 and 2016, mainly in red seaweed species (FAO 2018). This exponential growth in the production of tropical red seaweeds species, such as Kappaphycus alvarezii and Eucheuma spp. was due to their commercial exploitation for carrageenan extraction and direct food consumption, which has been the main contributor to the growth in seaweed cultivation in recent years. The high economic interest is also justified by the growing demand for phycocolloids for different uses in the pharmaceutical, food, and cosmetics industries (Smit 2004; Leandro et al. 2020b; Morais et al. 2021), which has led several countries to cultivate seaweed (García-Poza et al. 2020).
2.1 Gracilaria sp. Nutritional Profile
Gracilaria sp. is a red seaweed that produces agar as main structural polysaccharide, and their content have interesting values for direct food consumption, due to high content in protein (10.86%) and carbohydrates (63.13%) (Rasyid et al. 2019).
The study of Kazir et al. (2019) aimed to study Gracilaria sp. protein content for the development of food products, obtaining interesting protein levels (25% dry weight (DW)). The authors also obtained, in the highest concentrations, the amino acids: glutamic acid (13.01%), aspartic acid (12.81%), arginine (10.32%) and alanine (10.03%). In order to develop a seaweed protein-based product, the authors employed the ion exchange technique, enabling a high protein yield (70%) and a reduced co-extraction of carbohydrates (1%) in the seaweed protein extract. Showing a potential of Gracilaria sp. as protein food alternative source as a raw or processed ingredient.
This potential is also demonstrated in lipidic profile (Table 13.3), where the content of lipids and polyunsaturated fatty acids (PUFAs) in several Gracilaria species exhibited a high content of PUFAs, mainly G. corticata and G. dura (Kumari et al. 2013).
Khan et al. (2019) studied the polysaccharides of Gracilaria chouae and found the presence of a heteropolysaccharide (agar). Agar contained a sulphate content of 7.9%, in addition to 52.63% total sugar (mainly galactose) and 9.62% galacturonic acid. Galactose and 3,6-anhydrogalactose were found in a molar ratio of 1.0: 0.6. On further analysis, this polysaccharide exhibited jellification and melting points at 41.3 and 71.7 °C, respectively, which makes it a suitable candidate for industrial processing where further heating is required and/or where the end product needs to have an extended shelf life in hot climate.
2.2 K. alvarezii Nutritional Profile
K. alvarezii is a red seaweed that produces kappa-carrageenan as a main polysaccharide, and its nutritional profile has interesting values of minerals (58%) and carbohydrates (38%) and lower content in lipids and proteins (Wanyonyi et al. 2017), which is similar to other Kappaphycus species (Adharini et al. 2019). Moreover, these seaweeds can have an interesting prebiotic effect as a food supplement (Wanyonyi et al. 2017).
Kumar et al. (2014) analyzed the protein content of K. alvarezii, grown on the west coast of India, and found that this species contained 62.3 ± 1.62%. To further exploit this high yield in the food industry, this protein content can be transformed into a concentrate (PC), by the increase of the pH up to 12 and adding a solution of NaCl (0.5 M in the final solution). The emulsifier and foaming properties of this PC varied with time and pH. Thus, the results obtained in this study suggest the possibility of this seaweed and their based PC as an inexpensive source of protein; thus, this PC could be incorporated into several value-added food products.
2.3 Euchema sp. Nutritional Profile
Eucheuma sp. is a red seaweed that produces iota-carrageenan as main polysaccharide, which is mainly exploited from these species (Naseri et al. 2020). From an industrial perspective, the seaweed powder can be added into the food products to preserve and add nutritional value (Huang and Yang 2019). However, there is a lack of nutritional studies in this genus.
The red macroalgae Eucheuma denticulatum , also known by the common name “Spinosum”, develops naturally in coral reefs with moderately strong currents in tropical and subtropical regions. This species has a high commercial value, as it contains iota-carrageenan, a compound widely used in the nutraceutical and manufacturing industries. Due to the high demand, the cultivation of Eucheuma denticulatum has significantly expanded (Othman et al. 2019).
Balasubramaniam et al. (2020) studied E. denticulatum carotenoids (mg/100 g of DW extract) and detected in various samples maximum content of each of the pigments viz. lutein 87.7, zeaxanthin 21.3, coxanthin 4.0, β-cryptoxanthin 3.6, canthaxanthin <0.001, astaxanthin 3.0, and β-carotene 4.7. These results indicate that E. denticulatum has an excellent carotenoid profile (as vitamin A) composition and hence rich in antioxidant potential.
De Corato et al. (2017) evaluated E. denticulatum composition in terms of concentration of fatty acids, polysaccharides and phenolic compounds. E. denticulatum presented in its composition 20.5 ± 0.5% of lipids, water-soluble polysaccharides 16 ± 0.6% and phenolic compounds 0.2 ± 0.001%, demonstrating that the species presents a good percentage of fatty acids and polysaccharides, with potential in several applications as direct food source and as ingredient for food industry.
2.4 Porphyra/Pyropia/Neopyropia/Neoporphyra Nutritional Profile
The genus Porphyra is evolving and modified/divided into four different genera due to the genetic analysis (Yang et al. 2020; Kavale et al. 2021).
Porphyra/Pyropia/Neopyropia/Neoporphyra sp. is a red seaweed that produces porphyran, as main polysaccharide, and actually is the seaweed most consumed in the world, due to their presence in the Japanese cuisine as “nori” (Levine and Sahoo 2010; Bito et al. 2017) Nori can ameliorate the deficiency of iron and vitamin B12 in vegan diet. Thus, Porphyra/Pyropia/Neopyropia/Neoporphyra sp. are one of the most economically important species, as it has functional bioactivities such as porphyrans, dietary fibers, PUFAs, minerals, phycoerythrin, mycosporine-like Amino Acids and vitamins (Bito et al. 2017). Porphyran and oligo-porphyran have a range of biological functions, such as antioxidant, anticancer, anti-aging, anti-allergic, immunomodulatory, hypoglycemic and hypolipemic effects. Consequently, these species’ demonstrate several potential applications in the food, medicinal and cosmetic fields (Qiu et al. 2021). The most consumed and cultivated seaweeds are Porphyra tenera, Neopyropia tenera, Neopyropia yezoensis, Neoporphyra dentata and Neoporphyra haitanensis (Levine and Sahoo 2010; Niu et al. 2010). In fact, for these genera cultivation was of the value US Dollar 0.9 billion (Kim et al. 2017) mainly for direct food consumption.
Kim et al. (2018), studied Neopyropia tenera, determining the composition of carbohydrates, lipids, and proteins of N. tenera procured from a local market. The composition ratio (% DW) of carbohydrates, lipids, and proteins in N. tenera was 41.4%, 1.7% and 39.6%, respectively were determined. The study showed similar levels of carbohydrates and proteins, but with a low lipid content.
Holdt and Kraan (2011) and Rioux and Turgeon (2015), evaluated several seaweed species of the genus Porphyra/Pyropia/Neopyropia/Neoporphyra, and studied their bioactive compounds with economic importance. Among the determined compounds, the total polysaccharides had higher concentrations, with 40 and 76%, while the lipid content was 0.12 and 2.8% and protein of 7 and 50%. The results demonstrate that the Porphyra/Pyropia/Neopyropia/Neoporphyra genus, due to its higher concentration of polysaccharides, namely the hybrid porphyran/carrageenan/agar, are essential compounds in the food industry, as they are characterized by their solubility, gelation, viscosity, stability, reactivity with proteins and thixotropy properties (Hongfeng et al. 1993; Sasuga et al. 2017; Wahlström et al. 2018).
Larrea-Marín et al. (2010), evaluated the macro and trace elements in commercially grown Porphyra from four different countries (Table 13.4). The 12 elements determined were Al, Ba, Ca, Cu, Fe, K, Mg, Mn, Na, P, Sr and Zn, in the seaweed used as human food. However, differential compositions were due to the origin of the seaweeds, and impact of seawater mineral composition.
Although , these four genera are being cultivated and consumed, there is a lack of nutritional values of these seaweeds, which are understandable, due to the variation and influence of extrinsic factor that make seaweed composition fluctuate (García-Poza et al. 2020; Leandro et al. 2020b). However, the principal traits are normally identical as demonstrated above. Still, Porphyra/Pyropia/Neopyropia/Neoporphyra are the most exploited seaweeds for food and not for polysaccharide extraction, mainly due to the presence of a hybrid polysaccharide.
3 Red Seaweeds Consumption and Commercial Based Products
Seaweeds and their components already hold a market positioning worldwide (Ścieszka and Klewicka 2019; Rahikainen and Yang 2020). Still, several seaweeds remain as undiscovered resources, showing a very promising potential for the food industry, with nutraceutical properties (Pereira et al. 2020; John et al. 2020).
For instance, the red seaweeds dulse (Palmaria palmata) and nori (Porphyra/Pyropia/Neopyropia/Neoporphyra) are the major commercial products in the nutraceutical industry, as a food ingredient. These can be marketed fresh or dried, in a form of sheet, powder or capsule (Griffiths et al. 2016). The incorporation of milled seaweeds, such as Porphyra umbilicalis in meat products can indeed enrich these products in phenolic compounds and other nutrients likewise manganese, calcium and magnesium, which are pivotal for the homeostasis of the human organism (López-López et al. 2009). Besides, the bioactive and nutritional components of this seaweed species are associated with several health benefits, such as anticancer, cardiovascular disease prevention, antioxidant and anti-inflammatory (Cho and Rhee 2020). Moreover, the enrichment of cereal-based products (i.e., bread or pasta) with dried and milled seaweeds, such as Kappaphycus alvarezii was found to improve the nutritional profile of commercial noodles (Kumoro et al. 2016). However, heavy metals, toxic isotopes, dioxins, or pesticides are all risks associated with the whole seaweed intake. To address these drawbacks, thorough testing of seaweed for food application is needed prior to its use (Garcia-Vaquero and Hayes 2016).
In counterpart, industrial phycocolloids are under strict regulation, and several analyses are required for their incorporation as food additives in commercially available products (Mortensen et al. 2016; Younes et al. 2018). Thus, red seaweeds phycocolloids are already exploited and employed in several commercial products in food and nutraceutical industries. For instance, WavePure is a product based on Gracilaria sp., whereas the phycocolloid (agar) is extracted in order to be commercialized for food proposes as a gelling and thickening agent for desserts confection (Cargill 2021a). While carrageenan is a key element of the products Satiagel™, Satiagum™, Aubygel™ and Seabrid™, which can be used in a wide range of food products, such as dairy, fruit meat, ice-creams, powder products, pharmaceuticals and nutraceuticals (Cargill 2021b). Moreover, the vegetable jelly sold by Condi (based on carrageenan), shown to be also a nutraceutical promoter, due to its anti-cholesterolemic properties (Valado et al. 2020).
4 Conclusions and Future Perspectives
As novel foods and nutraceutical products demand for red seaweeds has risen. From the seaweeds belonging to the phylum Rhodophyta, only a few of them are industrially exploited. Thus, red seaweeds constitute a pool of unexplored biodiversity in several areas; for food and nutraceutical implying that a wide range of innovative products can be developed with these seaweeds.
Abbreviations
- FAO:
-
Food and Agriculture Organization
- PC:
-
Protein concentrate
- PUFAs:
-
Polyunsaturated fatty acids
References
Adharini RI, Suyono EA, Suadi, Jayanti AD, Setyawan AR (2019) A comparison of nutritional values of Kappaphycus alvarezii, Kappaphycus striatum, and Kappaphycus spinosum from the farming sites in Gorontalo Province, Sulawesi, Indonesia. J Appl Phycol 31:725–730. https://doi.org/10.1007/s10811-018-1540-0
Alves C, Silva J, Pinteus S, Gaspar H, Alpoim MC, Botana LM, Pedrosa R (2018) From marine origin to therapeutics: the antitumor potential of marine algae-derived compounds. Front Pharmacol 9:777. https://doi.org/10.3389/fphar.2018.00777
Alwaleed EA (2019) Biochemical composition and nutraceutical perspectives Red Sea seaweeds. Am J Appl Sci 16:346–354. https://doi.org/10.3844/ajassp.2019.346.354
Balasubramaniam V, June Chelyn L, Vimala S, Mohd Fairulnizal MN, Brownlee IA, Amin I (2020) Carotenoid composition and antioxidant potential of Eucheuma denticulatum, Sargassum polycystum and Caulerpa lentillifera. Heliyon 6:e04654. https://doi.org/10.1016/j.heliyon.2020.e04654
Barbier M, Charrier B, Araujo R, Holdt SL, Jacquemin B, Rebours C (2019) PEGASUS—PHYCOMORPH European guidelines for a sustainable aquaculture of seaweeds, Roscoff, France. https://doi.org/10.21411/2c3w-yc73
Belattmania Z, Engelen AH, Pereira H, Serrão EA, Custódio L, Varela JC, Zrid R, Reani A, Sabour B (2018) Fatty acid composition and nutraceutical perspectives of brown seaweeds from the Atlantic coast of Morocco. Int Food Res J 25:1520–1527
Bito T, Teng F, Watanabe F (2017) Bioactive compounds of edible purple laver Porphyra sp. (nori). J Agric Food Chem 65:10685–10692. https://doi.org/10.1021/acs.jafc.7b04688
BNF (2016) Nutrition requirements: reference nutrient intakes for minerals
Campbell I, Kambey CSB, Mateo JP, Rusekwa SB, Hurtado AQ, Msuya FE, Stentiford GD, Cottier-Cook EJ (2020) Biosecurity policy and legislation for the global seaweed aquaculture industry. J Appl Phycol 32:2133–2146. https://doi.org/10.1007/s10811-019-02010-5
Cargill (2021a) WavePure® ADG for gelled dairy desserts
Cargill (2021b) Carrageenan for innovative textures
Černá M (2011) Chapter 24 - Seaweed proteins and amino acids as nutraceuticals. In: Kim S-K (ed) Advances in food and nutrition research. Academic, San Diego, pp 297–312
Cho TJ, Rhee MS (2020) Health functionality and quality control of laver (Porphyra, Pyropia): current issues and future perspectives as an edible seaweed. Mar Drugs 18:1–31. https://doi.org/10.3390/md18010014
Cotas J, Leandro A, Monteiro P, Pacheco D, Figueirinha A, Gonçalves AMM, da Silva GJ, Pereira L (2020a) Seaweed phenolics: from extraction to applications. Mar Drugs 18:384. https://doi.org/10.3390/md18080384
Cotas J, Leandro A, Pacheco D, Gonçalves AMMM, Pereira L (2020b) A comprehensive review of the nutraceutical and therapeutic applications of red seaweeds (Rhodophyta). Life 10:19. https://doi.org/10.3390/life10030019
Cunha L, Grenha A (2016) Sulfated seaweed polysaccharides as multifunctional materials in drug delivery applications. Mar Drugs 14:42. https://doi.org/10.3390/md14030042
Dawes CJ (1995) Marine Botany. John Wiley & Sons, New York
De Corato U, Salimbeni R, De Pretis A, Avella N, Patruno G (2017) Antifungal activity of crude extracts from brown and red seaweeds by a supercritical carbon dioxide technique against fruit postharvest fungal diseases. Postharvest Biol Technol 131:16–30. https://doi.org/10.1016/j.postharvbio.2017.04.011
FAO (2018) The state of the world fisheries and aquaculture—meeting the sustainable development goals, vol 3. FAO, Rome
FAO (2020) The state of world fisheries and aquaculture. Sustainability in action. Rome
Ferdouse F, Holdt SL, Smith R, Murúa P, Yang Z, et al (2018) The global status of seaweed production, trade and utilization. FAO Globefish Research Programme 124, p 120
Ganesan AR, Shanmugam M (2020) Isolation of phycoerythrin from Kappaphycus alvarezii: a potential natural colourant in ice cream. J Appl Phycol 32:4221–4233. https://doi.org/10.1007/s10811-020-02214-0
García-Poza S, Leandro A, Cotas C, Cotas J, Marques JC, Pereira L, Gonçalves AMMM (2020) The evolution road of seaweed aquaculture: cultivation technologies and the industry 4.0. Int J Environ Res Public Health 17(8):6528. https://doi.org/10.3390/ijerph17186528
Garcia-Vaquero M, Hayes M (2016) Red and green macroalgae for fish and animal feed and human functional food development. Food Rev Int 32:15–45. https://doi.org/10.1080/87559129.2015.1041184
Garcia-Vaquero M, Rajauria G, O’Doherty JV, Sweeney T (2017) Polysaccharides from macroalgae: recent advances, innovative technologies and challenges in extraction and purification. Food Res Int 99:1011–1020. https://doi.org/10.1016/j.foodres.2016.11.016
Griffiths M, Harrison STL, Smit M, Maharajh D (2016) Major commercial products from micro- and macroalgae. In: Bux F, Chisti Y (eds) Algae biotechnology. Green energy and technology. Springer, Cham, pp 269–300. https://doi.org/10.1007/978-3-319-12334-9_14
Holdt SL, Kraan S (2011) Bioactive compounds in seaweed: functional food applications and legislation. J Appl Phycol 23:543–597. https://doi.org/10.1007/s10811-010-9632-5
Hongfeng G, Minghou J, Wenda C (1993) Comparative studies on structural feature of agar polysaccharides from Porphyra haitanensis grown in south and North China. Chin J Oceanol Limnol 11:25–33. https://doi.org/10.1007/BF02850725
Huang M, Yang H (2019) Eucheuma powder as a partial flour replacement and its effect on the properties of sponge cake. LWT 110:262–268. https://doi.org/10.1016/j.lwt.2019.04.087
Irene B, Ikram B, Christian BG, Nina LS, Rune W, Heidi A, Svenja H, Erik-Jan L (2018) Chemical characterization of 21 species of marine macroalgae common in Norwegian waters: benefits of and limitations to their potential use in food and feed. J Sci Food Agric 98:2035–2042. https://doi.org/10.1002/jsfa.8798
John OD, du Preez R, Panchal SK, Brown L (2020) Tropical foods as functional foods for metabolic syndrome. Food Funct 11:6946–6960. https://doi.org/10.1039/D0FO01133A
Kavale MG, Kazi MA, Brodie J (2021) Phycocalidia species (Bangiales, Rhodophyta), from the warm west coast of India. Eur J Phycol 56(3):337–347. https://doi.org/10.1080/09670262.2020.1829714
Kazir M, Abuhassira Y, Robin A, Nahor O, Luo J, Israel A, Golberg A, Livney YD (2019) Extraction of proteins from two marine macroalgae, Ulva sp. and Gracilaria sp., for food application, and evaluating digestibility, amino acid composition and antioxidant properties of the protein concentrates. Food Hydrocoll 87:194–203. https://doi.org/10.1016/j.foodhyd.2018.07.047
Khan BM, Qiu H-M, Wang X-F, Liu Z-Y, Zhang J-Y, Guo Y-J, Chen W-Z, Liu Y, Cheong K-L (2019) Physicochemical characterization of Gracilaria chouae sulfated polysaccharides and their antioxidant potential. Int J Biol Macromol 134:255–261. https://doi.org/10.1016/j.ijbiomac.2019.05.055
Kim S (2011) In: Kim S-K (ed) Handbook of marine macroalgae. John Wiley & Sons, Chichester. https://doi.org/10.1002/9781119977087
Kim JK, Yarish C, Hwang EK, Park M, Kim Y (2017) Seaweed aquaculture: cultivation technologies, challenges and its ecosystem services. Algae 32:1–13. https://doi.org/10.4490/algae.2017.32.3.3
Kim Y-M, Han TU, Lee B, Watanabe A, Teramae N, Kim J-H, Park Y-K, Park H, Kim S (2018) Analytical pyrolysis reaction characteristics of Porphyra tenera. Algal Res 32:60–69. https://doi.org/10.1016/j.algal.2018.03.003
Kraan S (2012) Algal polysaccharides, novel applications and outlook. In: Carbohydrates—comprehensive studies on glycobiology and glycotechnology. InTech, London. https://doi.org/10.5772/51572
Kumari P, Bijo AJ, Mantri VA, Reddy CRK, Jha B (2013) Fatty acid profiling of tropical marine macroalgae: an analysis from chemotaxonomic and nutritional perspectives. Phytochemistry 86:44–56. https://doi.org/10.1016/j.phytochem.2012.10.015
Kumoro AC, Johnny D, Alfilovita D (2016) Incorporation of microalgae and seaweed in instant fried wheat noodles manufacturing: nutrition and culinary properties study. Int Food Res J 23:715–722
Larrea-Marín MT, Pomares-Alfonso MS, Gómez-Juaristi M, Sánchez-Muniz FJ, Ródenas de la Rocha S (2010) Validation of an ICP-OES method for macro and trace element determination in Laminaria and Porphyra seaweeds from four different countries. J Food Compos Anal 23:814–820. https://doi.org/10.1016/j.jfca.2010.03.015
Leandro A, Pacheco D, Cotas J, Marques JC, Pereira L, Gonçalves AMM (2020a) Seaweed’s bioactive candidate compounds to food industry and global food security. Life 10:140. https://doi.org/10.3390/life10080140
Leandro A, Pereira L, Gonçalves AMMM (2020b) Diverse applications of marine macroalgae. Mar Drugs 18:17. https://doi.org/10.3390/md18010017
Levine IA, Sahoo D (2010) Porphyra: harvesting gold from the sea. Journal of Chemical Information and Modeling. I. K. International Publishing House Pvt. Ltd., New Delhi
Lopes G, Sousa C, Valentão P, Andrade PB (2013) Sterols in algae and health. In: Bioactive compounds from marine foods: plant and animal sources. Wiley, Oxford, pp 173–191. https://doi.org/10.1002/9781118412893.ch9
López-López I, Bastida S, Ruiz-Capillas C, Bravo L, Larrea MTT, Sánchez-Muniz F, Cofrades S, Jiménez-Colmenero F (2009) Composition and antioxidant capacity of low-salt meat emulsion model systems containing edible seaweeds. Meat Sci 83:492–498. https://doi.org/10.1016/j.meatsci.2009.06.031
Morais T, Cotas J, Pacheco D, Pereira L (2021) Seaweeds compounds: an ecosustainable source of cosmetic ingredients? Cosmetics 8:8. https://doi.org/10.3390/cosmetics8010008
Mortensen A, Aguilar F, Crebelli R, Di Domenico A, Frutos MJ, Galtier P, Gott D et al (2016) Re-evaluation of agar (E 406) as a food additive. EFSA J 14:e04645. https://doi.org/10.2903/j.efsa.2016.4645
Murata M, Nakazoe JI (2001) Production and use of marine algae in Japan. Jpn Agric Res Q 35(4):281–290. https://doi.org/10.6090/jarq.35.281
Naseri A, Jacobsen C, Sejberg JJP, Pedersen TE, Larsen J, Hansen KM, Holdt SL (2020) Multi-extraction and quality of protein and carrageenan from commercial spinosum (Eucheuma denticulatum). Foods 9:1072. https://doi.org/10.3390/foods9081072
Necas J, Bartosikova L (2013) Carrageenan: a review. Vet Med 58:187–205. https://doi.org/10.17221/6758-VETMED
Niu J-F, Chen Z-F, Wang G-C, Zhou B-C (2010) Purification of phycoerythrin from Porphyra yezoensis Ueda (Bangiales, Rhodophyta) using expanded bed absorption. J Appl Phycol 22:25–31. https://doi.org/10.1007/s10811-009-9420-2
Othman R, Rasib AAA, Ilias MA, Murthy S, Ismail N, Hanafi NM (2019) Transcriptome data of the carrageenophyte Eucheuma denticulatum. Data Brief 24:103824. https://doi.org/10.1016/j.dib.2019.103824
Pereira L (2011) A review of the nutrient composition of selected edible seaweeds. In: Seaweed: ecology, nutrient composition and medicinal uses. Nova Science, Hauppauge, pp 15–47
Pereira SA, Kimpara JM, Valenti WC (2020) A bioeconomic analysis of the potential of seaweed Hypnea pseudomusciformis farming to different targeted markets. Aquacult Econ Manag 24:507–525. https://doi.org/10.1080/13657305.2020.1803445
Qiu Y, Jiang H, Fu L, Ci F, Mao X (2021) Porphyran and oligo-porphyran originating from red algae Porphyra: preparation, biological activities, and potential applications. Food Chem 349:129209. https://doi.org/10.1016/j.foodchem.2021.129209
Rahikainen M, Yang B (2020) Macroalgae as food and feed ingredients in the Baltic Sea region—regulation by the European Union. Finland
Rasyid A, Ardiansyah A, Pangestuti R (2019) Nutrient composition of dried seaweed Gracilaria gracilis. ILMU KELAUTAN Indones J Mar Sci 24(1):1–16. https://doi.org/10.14710/ik.ijms.24.1.1-6
Rioux L, Turgeon SL (2015) Chapter 7 - Seaweed carbohydrates. In: Seaweed sustainability: food and non-food applications. Elsevier, Amsterdam. https://doi.org/10.1016/B978-0-12-418697-2/00007-6
Rosa J, Lemos MFL, Crespo D, Nunes M, Freitas A, Ramos F, Pardal MÂ, Leston S (2020) Integrated multitrophic aquaculture systems—potential risks for food safety. Trends Food Sci Technol 96:79–90. https://doi.org/10.1016/j.tifs.2019.12.008
Ruocco N, Costantini S, Guariniello S, Costantini M (2016) Polysaccharides from the marine environment with pharmacological, cosmeceutical and nutraceutical potential. Molecules 21(5):551. https://doi.org/10.3390/molecules21050551
Sangha JS, Kandasamy S, Khan W, Bahia NS, Singh RP, Critchley AT, Prithiviraj B (2015) λ-Carrageenan suppresses tomato chlorotic dwarf viroid (TCDVd) replication and symptom expression in tomatoes. Mar Drugs 13:2875–2889. https://doi.org/10.3390/md13052875
Sasuga K, Yamanashi T, Nakayama S, Ono S, Mikami K (2017) Optimization of yield and quality of agar polysaccharide isolated from the marine red macroalga Pyropia yezoensis. Algal Res 26:123–130. https://doi.org/10.1016/j.algal.2017.07.010
Ścieszka S, Klewicka E (2019) Algae in food: a general review. Crit Rev Food Sci Nutr 59:3538–3547. https://doi.org/10.1080/10408398.2018.1496319
Shama A, Joyce SG, Mari FD, Ranga Rao A, Ravishankar GA, Hudaa N (2019) Macroalgae and microalgae: novel sources of functional food and feed. In: Ravishankar GA, Rao AR (eds) Handbook of algal technologies and phytochemicals: Volume I: food, health and nutraceutical applications. CRC Press, Boca Raton, pp 207–219
Shannon E, Abu-Ghannam N (2019) Seaweeds as nutraceuticals for health and nutrition. Phycologia 58:563–577. https://doi.org/10.1080/00318884.2019.1640533
Smit AJ (2004) Medicinal and pharmaceutical uses of seaweed natural products: a review. J Appl Phycol 16:245–262. https://doi.org/10.1023/B:JAPH.0000047783.36600.ef
Sonani RR (2016) Recent advances in production, purification and applications of phycobiliproteins. World J Biol Chem 7:100. https://doi.org/10.4331/wjbc.v7.i1.100
Tanna B, Mishra A (2018) Metabolites unravel nutraceutical potential of edible seaweeds: an emerging source of functional food. In: Comprehensive reviews in food science and food safety. Blackwell, Malden. https://doi.org/10.1111/1541-4337.12396
Tanna B, Mishra A (2019) Nutraceutical potential of seaweed polysaccharides: structure, bioactivity, safety, and toxicity. Compr Rev Food Sci Food Saf 18:817–831. https://doi.org/10.1111/1541-4337.12441
Torres P, Nagai A, Teixeira DIA, Marinho-Soriano E, Chow F, dos Santos DYACAC (2019) Brazilian native species of Gracilaria (Gracilariales, Rhodophyta) as a source of valuable compounds and as nutritional supplements. J Appl Phycol 31:3163–3173. https://doi.org/10.1007/s10811-019-01804-x
Tsuge K, Okabe M, Yoshimura T, Sumi T, Tachibana H, Yamada K (2004) Dietary effects of Porphyran from Porphyra yezoensis on growth and lipid metabolism of Sprague-Dawley rats. Food Sci Technol Res 10:147–151. https://doi.org/10.3136/fstr.10.147
Valado A, Pereira M, Caseiro A, Figueiredo JP, Loureiro H, Almeida C, Cotas J, Pereira L (2020) Effect of carrageenans on vegetable jelly in humans with hypercholesterolemia. Mar Drugs 18:19. https://doi.org/10.3390/md18010019
Villanueva MJ, Morcillo M, Tenorio MD, Mateos-Aparicio I, Andrés V, Redondo-Cuenca A (2014) Health-promoting effects in the gut and influence on lipid metabolism of Himanthalia elongata and Gigartina pistillata in hypercholesterolaemic Wistar rats. Eur Food Res Technol 238:409–416. https://doi.org/10.1007/s00217-013-2116-5
Wahlström N, Harrysson H, Undeland I, Edlund U (2018) A strategy for the sequential recovery of biomacromolecules from red macroalgae Porphyra umbilicalis Kützing. Ind Eng Chem Res 57:42–53. https://doi.org/10.1021/acs.iecr.7b03768
Wanyonyi S, du Preez R, Brown L, Paul N, Panchal S (2017) Kappaphycus alvarezii as a food supplement prevents diet-induced metabolic syndrome in rats. Nutrients 9:1261. https://doi.org/10.3390/nu9111261
Yang L-E, Deng Y-Y, Xu G-P, Russell S, Lu Q-Q, Brodie J (2020) Redefining Pyropia (Bangiales, Rhodophyta): four new genera, resurrection of Porphyrella and description of Calidia pseudolobata sp. nov. from China. Edited by K. Müller. J Phycol 56:862–879. https://doi.org/10.1111/jpy.12992
Younes M, Aggett P, Aguilar F, Crebelli R, Filipič M, Frutos MJ, Galtier P et al (2018) Re-evaluation of carrageenan (E 407) and processed Eucheuma seaweed (E 407a) as food additives. EFSA J 16(4):e05238. https://doi.org/10.2903/j.efsa.2018.5238
Zhong H, Gao X, Cheng C, Liu C, Wang Q, Han X (2020) The structural characteristics of seaweed polysaccharides and their application in gel drug delivery systems. Mar Drugs 18:658. https://doi.org/10.3390/md18120658
Acknowledgments
This work is financed by national funds through FCT—Foundation for Science and Technology, I.P., within the scope of the projects UIDB/04292/2020 granted to MARE—Marine and Environmental Sciences Centre and UIDP/50017/2020 + UIDB/50017/2020 (by FCT/MTCES) granted to CESAM—Centre for Environmental and Marine Studies. João Cotas thanks to the European Regional Development Fund through the Interreg Atlantic Area Program, under the project NASPA (EAPA_451/2016). Diana Pacheco thanks to PTDC/BIA-CBI/31144/2017-POCI-01 project-0145-FEDER-031144-MARINE INVADERS, co-financed by the ERDF through POCI (Operational Program Competitiveness and Internationalization) and by the Foundation for Science and Technology (FCT, IP). Ana M.M. Gonçalves acknowledges University of Coimbra for the contract IT057-18-7253, and also thanks to the project MENU—Marine Macroalgae: Alternative recipes for a daily nutritional diet (FA_05_2017_011), funded by the Blue Fund under Public Notice No. 5—Blue Biotechnology, which partially financed this research.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Pacheco, D., Araújo, G., Silva, J.W.A., Cotas, J., Gonçalves, A.M.M., Pereira, L. (2022). Red Seaweeds: Their Use in Formulation of Nutraceutical Food Products. In: Ranga Rao, A., Ravishankar, G.A. (eds) Sustainable Global Resources of Seaweeds Volume 2. Springer, Cham. https://doi.org/10.1007/978-3-030-92174-3_13
Download citation
DOI: https://doi.org/10.1007/978-3-030-92174-3_13
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-92173-6
Online ISBN: 978-3-030-92174-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)