Abstract
There is a growing need for alternative materials that could enhance the food nutritional value, dominating moisture and solute migration, prolong shelf life, and improve the quality of gas exchange and oxidative reaction rates. Besides, making the polymer-based plastics eco-friendly and biodegradable. Several bioactive compounds are implemented for the preparation of food active packaging including herbal extracts, metallic, and biological compounds. For the inclusion of these bioactive molecules, either synthetic polymers or natural polymers have been used. The encapsulation could be classified as microencapsulation and nanoencapsulation according to the carrier final diameter. Distinct methods were used to encapsulate bioactive substances like spray and freeze-drying, fluidized bed technologies, and molecular inclusion. Moreover, solid lipid nanoparticles and liposomes are lipid-based nanosystems that usually applied as a carrier of bioactive molecules and exhibited oxygen scavenging and antimicrobial properties. Hydrophobic phytochemicals molecules such as essential oils can be loaded in nanoemulsions and polymeric nanoparticles using emulsification–solvent evaporation, and supercritical fluid technologies. As well, Nanofibers are considered one of the promising materials in the inclusion of bioactive molecules in food packaging. This chapter sheds light on the methods used for inclusion, limitations, current trends, and regulatory issues, and biosafety in the encapsulation of bioactive components.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Active packaging
- Antimicrobial
- Antioxidant
- Bioactive molecules
- Encapsulation
- Liposomes
- Nanocomposites
- Nanoemulsions
- Nanofibers
- Nanostructures
1 Introduction
Food Packaging represents a vital role in the food sector. It maintains the food condition, extends shelf-life, and protects against environmental contamination such as temperature, microorganisms, and humidity (Han et al. 2018). Polymers play a substantial role in this regard, concerning their competitive properties. These include being cost-effective, easy to handle, and ideal mechanical properties, making them the perfect choice for food packaging. Some major type of polymers used as a matrix in the industry of food packaging involves Polyethylene terephthalate (PET), High-density polyethylene (HDPE), Low-density polyethylene (LDPE) (Siracusa and Blanco 2020). However, numerous different bioactive materials have recently been incorporated with those polymers to enhance the characteristics of the developed package material. For example, to improve food quality and safety, scientists have managed to delay oxidation, by creating packaging products with employed antioxidants and antimicrobial active components (Jouki et al. 2014). Others could include moisture absorbers, CO2 scavengers, oxygen scavengers, and emitters (Crisosto et al. 1993). Besides other packaging passive characteristics including thermal persistence, and mechanical capacity (Fernández-Pan et al. 2014). For instance, factors like discoloration, lack of aroma, and lipid oxidation can be controlled by comprising active additives to modify the surface of Low-density polyethylene (LDPE) and get rid of meat spoilage (Ben-Yehoshua. 1985).
1.1 Bioactive Compound Sources
Materials used in encapsulation should meet the safety guidelines according to the Food and Drug Administration (FDA). Most of these materials are biomolecules that are food-grade and biodegradable. The most utilized materials in food packaging industries could be natural or manufactured. For example, natural sources include polysaccharides, starch, and cellulose, besides plant extracts such as soluble soybean polysaccharides, pectins, Arabic gum, and galactomannans. While synthetic materials could be synthetic antioxidants, like ethylenediaminetetraacetic acid (EDTA) and poly(acrylic acid) (PAA) (https://reader.elsevier.com/reader/sd/pii/S0924224418302760?token=022AB9E1FF4BBDFD1F36E75608D69A35F711C9EAD3A6DAD330D36E5DB5D6887A3E7612D769D095AE946C531685EFDA1C) (Table 7.1).
1.2 Advantage of Nanotechnology in Food Packaging
Nanotechnology applications have opened new advances in the active food packaging industries. This extended till reach food safety and biosafety and innovation of active substances such as nanoparticles (https://www.ift.org/news-and-publications/food-technology-magazine/issues/2003/december/features/nanotechnology-a-new-frontier-in-food-science) (Fig. 7.1).
Many food packaging patents that utilized the applications of nanomaterials have been documented in different continents including the USA, Europe, and Asia, for which nano-clays and nano-silver were the most used ones. Recent studies have proved that the incorporation of allyl isothiocyanate and carbon nanotubes into the AP systems can reduce microbial activity and discoloration and regulate oxidation (Crisosto et al. 1993).
Nanoencapsulation has proved its competency in improving controlled release and provide accurate bioactive material delivery into targeted locations more efficiently than in bulky or microencapsulation techniques. This is due to the unique particle nanosize in the range of a nano diameter between 10 and 1000 nm that could be transported to various body organs in contrast to microencapsulation, in which the particles express a diameter of 3–800 μm (Hughes 2005).
Current efforts towards a “green environment” have put more focus on the fabrication of nanocomposite matrices and coatings through the utilization of biopolymers and bioactive components. The most recent example for the creation of nanocomposites is the use of polysaccharides synthetic matrices incorporated with inorganic materials. Fillers could be incorporated with biopolymer coatings to come up with bionanocomposite. For instance, nanoparticles can be used as fillers within biopolymer matrices to enhance the plastic properties and biodegradability (Uysal Unalan et al. 2014). Halloysite nanotubes (HNTs) are one of the interesting developments in the sector of food AP. They can act as nanofillers since they are hollow tubular clay nanoparticles. They have many excellent advantages in delaying food spoilage and fruit aging by absorbing the naturally generated ethylene gas. Polyethylene (PE) can be treated with such HNTs to provide more ethylene gas capturers with lower permeability to water vapor than in pure PE (Tas et al. 2017).
1.3 Nanoencapsulation for Improved Food Packaging Properties
Recent investigations showed the effectiveness of HNT/polyethylene (HNT/PE) nanocomposite films in delaying ripening process of bananas due to the ethylene scavenging features. Results showed a. HNT/PE nanocomposite films proved their abilities in preserving food safety and enhancing food products’ shelf life (Tas et al. 2017).
Moisture can reduce the quality of packed food and make it more liable to pathogenic microorganisms. Different moisture absorbers have been used in food packaging to reduce the moisture effect by combining desiccants substances like clay, zeolites, or silica, while others have tried bentonite and poly-acrylic acid sodium salts (Mahajan et al. 2008). To extend shelf life, ethylene scavengers have been used such as potassium permanganate, zeolite nanoparticles to absorb ethylene and prevent chlorophyll degradation. Essentially, carbon dioxide emitters are essential in inhibiting microbial invasion and expand the package shelf life. Conjugation of nanoparticles with antimicrobial properties like chitosan with gold, metal oxide, or silver nanoparticles has shown great versatility. Since they showed unparalleled antimicrobial effect when combined with other materials. For instance, silver nanoparticles have shown a typical attachment to the cell surface and penetration of microbial cells followed by DNA damage (Honarvar et al. 2016).
2 Encapsulation of Bioactive Molecules
Numerous studies have investigated bioactive encapsulation. Intensive studies have been made on food microbial spoilage and oxidation which affect consumer decisions in buying food. This usually happens to fruits including bananas, kiwi fruit, and tomatoes (Fernández-Pan et al. 2014). One study has used vacuum packaging to produce an oxygen-free environment in food packages (Dey and Neogi 2019). Others fabricated bionanocomposites packaging material using chitosan, zinc oxide nanoparticles, and carboxymethyl cellulose to prevent food spoilage by the effect of bacterial microorganisms (Winestrand et al. 2013). Anti-oxidants molecules are one of the crucial ones, especially in meat packaging. A study has utilized the antioxidant activity of rosemary extract and incorporated it into low-density polyethylene (LDPE) with the presence of α-tocopherol. Further, those materials were transformed into discs and came into contact with fresh beef. It has been reported over time that created a disc of rosemary were able to keep higher values of meat (redness) than control samples over a prolonged period (Moore et al. 2006) (Table 7.2).
3 Micro and Nano-encapsulation Techniques
The development of the micro and nano-encapsulated carriers that are needed for the entrapment of bioactive molecules into food packaging systems could be classified into two opposed approaches; (i) bottom-up, and (ii) top-down (Joye and McClements 2014). There is no optimum approach for encapsulation of all available bioactive compounds (Aguiar et al. 2016). The bottom-up one relies on small particles and elements association in a process that is controlled by the concentration of these monomers, pH of the media, a temperature that is needed for the self-assembly, and ionic strength. The bottom-up principle is found in several methods like electrospinning, spray drying, complex coacervation, and anti-solvent precipitation. This approach is characterized by its less energy consumption, and a good dominator on both shape and size of both micro and nanocarrier. Furthermore, the top-down principle is to focus on the downsizing of the large structure into a smaller one using external mechanical means. The top-down principle is found in techniques like emulsification, and extrusion processes. However, this approach shows less control over both the size and shape of micro and nanocarriers (Jia et al. 2016a). Different factors are affecting the selection of the optimum method for encapsulating the bioactive compounds for food packaging application such as particle size, solubility, and molecular weight of both bioactive compounds and encapsulating agents (Dias et al. 2017).
3.1 Spray Drying
One of the techniques that could be used for microencapsulating bioactive compounds is spray drying technique. It was established as a method of encapsulating different bioactive compounds such as; pumpkin seed oil (Ogrodowska et al. 2017), Agaricus bisporus extracts (Francisco et al. 2018), chia essential oil (Rodea-González et al. 2012), and Lippia sidoides oil (Abreu et al. 2012). It is characterized by its simplicity, rapid performance, as well as low-cost technique. The spray drying technique mode of action (Fig. 7.2) is based on introducing the bioactive compounds with the encapsulating material (solution, emulsion, or suspension in either aqueous or organic media), in form of fine mist or droplets of, in an atmosphere of hot air. The hot air is previously introduced through a tangential inlet generating as a centrifugation force inside the drying chamber. While spraying the liquid bioactive compounds in the hot atmosphere, the dehydration phase occurs, evaporating the exciting solvents forming a powder of bioactive compounds coated with the encapsulating agent. The drying process accomplished through the separation of the microencapsulated ingredients by the cyclone separator (Nesterenko et al. 2013a). The temperature inside the drying chamber could reach 130–180 °C. The main factor that infuences the encapsulation process is the solubility of the encapsulating material in the solvent. However, this technique suffers from the product low yield due to either adhesion of the dried powder on drying chamber wall or the degradability of the active compounds when they are facing this hot environment (Nesterenko et al. 2013a). The former disadvantage was controlled through generating ultrasound during the atomization phase that improves both encapsulation efficiency, product stability as well as the outcome yield. This is because the ultrasound waves produces less mechanical stress on the sprayed dried product (Tatar Turan et al. 2015). The latter disadvantage was minimized through using of the vacuum drying chamber, lowering the drying temperature to 40–60 °C (Islam et al. 2017).
3.2 Freeze Drying
Freeze drying is a common method for that have beed used for microencapsulation process. Basically, it relies on using vacuum for converting the bioactive compounds (especially the thermosensitive substances) from the liquid state into encapsulated powder state. Its mode of action is depending on three main stages; (i) freezing of the polymer-bioactive fluid at a temperature of −50 °C, followed by (ii) primary drying step where solvent start to sublime forming a wet powder, and finally (iii) second drying step at which the remaining solvent on the surface of the powder starts to desorb. The second and third steps are achieved using a low temperature between 10 and 20 °C while applying vacuum till reaching the suitable moisture content (Fig. 7.3) (Abdul-Fattah et al. 2007). The main disadvantages of the freeze dryer is the long drying time that extends for days for a complete drying as well as its high cost (Fuciños et al. 2017a). However, there is another factor besides the drying time that can affect the encapsulation process which is the method of the preparation of the solution, suspension or emulsion that is needed to be freeze dried as it greatly affects the size and the morphology of the micro and nanosystems (Varshosaz et al. 2012). The freeze-drying techniques showed an efficiency in producing micro and nanosystems such as nano lipid carriers (Varshosaz et al. 2012), nanotubes (Fuciños et al. 2017a), and nanoparticles (Shetta et al. 2019; Attallah et al. 2020). Recently, spray freeze drying approach might be better alternative to the traditional freeze dryer by introducing an atomization means before starting the freezing phase (Ishwarya et al. 2015). Spray freeze drying decreases the drying time down to 8 h as well as increases the stability of the bioactive compounds (Hundre et al. 2015).
3.3 Complex Coacervation
One of the techniques that could be used for nano and microencapsulation is the complex coacervation approach. It is based on the ionic interaction between the core bioactive compounds and the coating material that are previously dissolved in immiscible solvents (Fig. 7.4) (Timilsena et al. 2017). After this interaction, the coating material starts to envelop the bioactive compounds after adjusting the pH of the media, concentration of both phases, and temperature of the solution. Finally, the resulted coacervate (complex) starts to solidify using thermal or chemical (cross-linking) means (Bakry et al. 2016). The coacervation process could be simple (by using one polymer), or complex (by using multiple polymeric materials) (Yuan et al. 2017a). There are many examples for encapsulating bioactive compounds using complex coacervation such as; anthocyanin (Arroyo-Maya and McClements 2015), avocado extract (Calderón-Oliver et al. 2020), algal oil (Yuan et al. 2017a) and β-carotene (Jain et al. 2016a). The complex coacervation technique could modify the release of the bioactive ingredientes, and provides high encapsulation efficiency (Rutz et al. 2017). Although, the complex coacervation needs high preparation cost, it shows difficulty in controlling both shape and size of micro and nanosystems (Jia et al. 2016a).
3.4 Emulsification
The emulsification technique is widely used for encapsulating bioactive ingredients. The emulsion produced could be either simple emulsion such as oil in water (o/w) or water in oil (w/o), or multiple emulsions such (w/o/w) or (o/w/o). The emulsification technique is used mainly for encapsulation of both polar and nonpolar bioactive ingredients (Gumus et al. 2017). The micro-emulsification process could be performed via low and high energy approach. High energy emulsification could be achieved by homogenization, ultrasonication, and high shear mixer (Fig. 7.5). The high energy approach is based on the formation of coarse emulsion by high shearing followed by the application of high energy to produce micro and nanoemulsions that could be achieved by high pressure (Piorkowski and McClements 2014) and ultrasonic energy (Jafari et al. 2007). There are so many examples for microencapsulation of active molecules such as roasted coffee oil (Freiberger et al. 2015), and D-limonene (Jafari et al. 2007). However, this technique requires sophisticated equipment that uses high energy (Joye and McClements 2014). In contrast, low energy emulsification techniques show simple and economic approach.
3.5 Antisolvent Precipitation
One of the main approaches for encapsulating bioactive compounds is the antisolvent precipitation method. It is a technique that is simple with low cost. The principle of this technique is relying on the dissolution of active molecules in binary solvent such as water and organic solvents. Then, a third solvent is added to act as antisolvent that decreases the solubility of the compounds leading to the formation of nanoparticles through the precipitation process (Fig. 7.6) (Jia et al. 2016a). One of the most common antisolvents used is supercritical carbon dioxide which is used for microencapsulation of active molecules such as curcumin molecules (Dias et al. 2017). Moreover, different bioactive molecules were encapsulated using the antisolvent precipitation such as epigallocatechin gallate (Donsì et al. 2017a), eugenol and thymol (Chen et al. 2015), and vitamins (David and Livney 2016). The antisolvent precipitation method could be improved through using of the ultrasound as it enhances the nucleation rates, and decreases agglomeration (Thorat and Dalvi 2012).
3.6 Extrusion
The passing of the mixture containing the bioactive compounds dispersed in the polymeric coating material across the nozzle, an area where gelling process occurs is called extrusion approach. It is a method that is used for the encapsulation of small as well as large molecules of both hydrophilic and hydrophobic nature (Fig. 7.7) (Han et al. 2018). Different materials could be encapsulated through this approach such as essential oil from marine resources (Bakry et al. 2016), gallic acid (Li et al. 2017), and riboflavin (He et al. 2015). However, the extrusion technique application is limited due to its inability to control the size. The reason behind that owing to the formation of a large and porous product during the extrusion and gelling steps. Several modifications were applied on the extrusion system to improve its properties such as applying the multi nozzle-system, co-extrusion, and melt extrusion (Rodriguez et al. 2016).
3.7 Electrospinning and Electro-Spraying
Electrospinning and electro-spraying are commonly used methods for encapsulating bioactive molecules. They are characterized by the formation of the nanofibers or small droplets from a dispersion containing bioactive molecules and the polymeric material (Fig. 7.8 a, and b). The principle of both techniques depends on applying a voltage over a liquid ejected from a nozzle (Faridi Esfanjani and Jafari 2016). The main difference between both techniques is the physical form produced, in case of the electrospinning techniques, the outcome is nanofibers owing to the high polymer concentration and the stability of the product while in electrospraying, the small droplets will be formed because of using loe polymer concentration and the instability of the product produced (Faridi Esfanjani and Jafari 2016). Moreover, electrospraying technique doesn’t require surfactant, while electrospinning technique requires surfactant (Tarhini et al. 2017). Different factors affect the nanofibers’ and the small droplets’ dimensions and shapes such as the applied voltage value, distance between the collector and syringe tip, the collection distance and the solution flow rate (Kegere et al. 2019; Wahbi et al. 2020). Both techniques are suitable for the production of micro and nanosystems (Elakkiya et al. 2014).
4 Various Forms of Micro and Nano-encapsulate Carriers Used in Food Packaging
Encapsulating technique is an inclusion approach of bioactive molecules or compounds into polymeric matrix or reservoir that is carried out to increase these compounds’ stability. Encapsulation processes mainly depend on making the first droplet of the bioactive molecules as solid (as powder), liquid or gas. Then, these droplets get enclosed by the carrier material through different techniques that include but are not limited to coacervation, co-extruding, emulsifications, fluidized bed coating, spray-drying, spray-cooling, melt injection, etc. (Dubey et al. 2009; Parra 2010; Sanguansri and Augustin 2007; Raybaudi-Massilia and Mosqueda-Melgar 2012).
4.1 Reservoir and Matrix
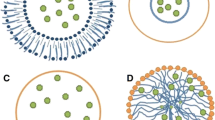
The encapsulating systems could possess different designs according to the nature and properties of the compounds incorporated as illustrated in Fig. 7.9. In addition, the design is controlled by the encapsulation method used to prepare the carriers either in the micro or nano scale. For instance, they can be classified into (a) reservoirs which can be either mono-core, where there is only a single chamber detected in the core of the capsule, or poly-core (multi-core), where there are more than one chamber inside the core that could be equal or different in shape and size, in addition to (b) matrix where the encapsulated compounds are scattered within the whole matrix of the carrier. In addition, the encapsulated compounds can be incorporated in their native form or as loaded nanoparticles or nanostructures (Dubey et al. 2009; Parra 2010).
4.2 Emulsions
4.2.1 Microemulsions
Microemulsions or traditional emulsions are colloidal systems that are made through mixing at least two immiscible liquids one of them is an aqueous phase while the another one is an oily phase (Brandelli et al. 2016). According to the type of dispersed material and dispersion medium, emulsions are classified into oil-in-water (O/W) emulsions and water-in-oil (W/O) emulsions as shown in (Fig. 7.10).
In O/W emulsions, oil is considered as the dispersed phase, while the continuous phase is the aqueous medium. On the contrary, the dispersed phase of the W/O emulsion contains the water droplets, while the oil constitutes the dispersion medium. Emulsions have to be stabilized through the addition of some amphiphilic macromolecules that act as emulsifying agents or stabilizers that act through increasing the steric hindrance, while minimizing the interfacial tension between the two immiscible media (Rayner et al. 2014). However, on long term duration, the dispersed droplets could coalesce again forming larger globules that could end up with complete separation of the two phases of microemulsions (Brandelli et al. 2016).
4.2.2 Nanoemulsions
Nanoemulsions are emulsified systems whose sizes range from 10 to 100 nm. They have the advantage of being more kinetically stable than microemulsions. Therefore, they require less surfactant concentrations as they are less susceptible to aggregation and coalescence. This returns back to the decrease in the attractive forces between the droplets after decreasing their size. Furthermore, stability of nanoemulsions was proven to be increased through addition of 5–10% alcohol as this amount was found to have an influence on decreasing the size of the droplets size owing to the interference with the interfacial tension among the droplets (Zeeb et al. 2014). Nanoemulsions can be prepared through different techniques either: (a) low energy techniques, or (b) high energy techniques (Gulotta et al. 2014; Ranjan et al. 2014). Table 7.3 summarizes some studies that involved the use of microemulsions and nanoemulsions as carriers incorporating bioactive molecules used in food applications.
4.3 Lipid Based Carriers
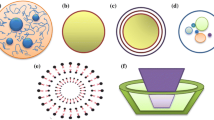
Lipid based carriers are fat-based encapsulating systems that are spherical structures capable of being dispersed in aqueous phase ranging from 0.1 to 100 um (Yalavarthi et al. 2014). Lipid based carriers can be mainly categorized into liposomes and solid lipid nanoparticles as shown in (Fig. 7.11).
4.3.1 Liposomes
Liposomes are spherical bilayer phospholipid colloidal systems. They are formed through dispersion of phospholipids in aqueous medium that self-assemble forming the vesicular structures. They are widely utilized as carriers for various bioactive molecules and nutraceuticals (Tan et al. 2013; Brandelli et al. 2017b; Thompson et al. 2006). Several approaches have been proven for their efficiency in preparation of liposomes. These approaches are classified into: (a) conventional techniques such as detergent removal, solvent evaporation & thin film hydration, and (b) emerging techniques such as modified electroformation, microfluidic devices and supercritical fluid procedures (Brandelli et al. 2016, 2017b; Patil and Jadhav 2014).
Liposomes bilayer phospholipid structure mimics the real structure of biomembranes. Therefore, they are commonly used as carriers. In addition, they have the advantage of masking the undesirable effects of the incorporated molecules as well as decreasing their toxicity (Chang et al. 2008). For example, liposomes reported high efficiency in increasing the stability of some incorporated thermo-labile bio-active compounds such as folic acid and many vitamins (Marsanasco et al. 2015). Some of the reported liposomal structure in food applications are summarized in Table 7.4.
4.3.2 Solid Lipid Nanoparticles
Solid lipid nanoparticles are spherical nanostructures composed of high food grade lipids capable of encapsulating hydrophobic compounds. They can be prepared by blending two lipid structures together differing in their melting points e.g. hydrogenated palm oil and cocoa butter. The pros of using solid lipid nanoparticles returns back to their capability of protecting the encapsulated compounds from chemical degradation in addition to their good physical stability. This is attributed to the strong steric repulsion among the particles generated by the non-ionic surfactants within the system (Brandelli et al. 2016; Qian et al. 2013).
4.4 Protein-Based Carriers
Proteins are natural biopolymers that could be extracted from either animal or plant sources. Proteins generally are insoluble in acidic medium while soluble in alkaline medium (Chen et al. 2006). Proteins are highly swellable materials and easily functionalized, therefore, they are considered as promising biomaterials for preparation of bioactive delivery systems for wide range of bioactive compounds such as fatty acids, fats, oils, and flavors (Jia et al. 2016b). Proteins extracted from animals include casein, collagen, gelatin, and whey proteins, while those obtained from plants include gliadin, soy and zein proteins (Tarhini et al. 2017). Compared to animal derived proteins, plant derived proteins have recently attracted many researches attention owing to their lower cost and minimized inflammatory properties (Nesterenko et al. 2013b). Many proteins have been reported as successful carrier systems for bioactive compounds.
4.4.1 Caseins
Caseins are the highest abundant proteins in milk. They are divided into αs1-casein, αs2- casein, β-casein, and κ-casein. They are characterized by possessing low solution viscosity, excellent emulsifying capabilities, in additional to their rich nutritional value. Hence, they are considered as promising materials for delivery systems preparation (Ho et al. 2017; Jain et al. 2016b).
Caseins are effective in encapsulating hydrophobic bioactive molecules such as β-carotene, vitamin D3, essential oils and naringenin (Jarunglumlert et al. 2015; Moeiniafshari et al. 2015; Chen et al. 2014).
Furthermore, caseinates salts of calcium and sodium are also obtained through their solubilization in alkaline medium. They have the advantage of being highly water soluble, thus enhancing the dispersibility of highly hydrophobic bioactive compounds (Jarunglumlert et al. 2015).
In addition, caseins can form micellar structures as colloidal particles that are formed from caseinates solutions upon the addition of some ions such as calcium, phosphate or citrate (Shishir et al. 2018).
4.4.2 Cereal Proteins
Zein proteins, the most important cereal proteins, are classified into alpha, beta, gamma, and delta. Zein proteins are hydrophobic water insoluble proteins that could self-assemble forming various structures according to the solvents used. Therefore, zein proteins are considered as promising candidates for preparing carrier systems for bioactive compounds (Dai et al. 2017; Donsì et al. 2017b).
Wheat proteins are composed of gliadin and glutenin. Although, they have low water solubility, high allergenic properties, and high susceptibility to celiac disease, they have been reported to be incorporated within encapsulation systems either alone or with polysaccharides. Therefore, wheat gluten has been modified through acid heating deamination in order to minimize its cytotoxicity and enhance their entire performance. For instance, succinic acid deamidated wheat gluten microspheres has been prepared to encapsulate fish oil through double emulsion technique. The produced deamidated wheat gluten microspheres proved their effeciency in controlling release and maintaining the stability of fish oil (Sun et al. 2009; Liao et al. 2012; Andreani et al. 2009).
Potato protein has been recently utilized as biomaterials for carriers preparation owing to its good antioxidant activity, high emulsifying ability, low cost, and low non-allergenic properties. Potato protein based nano carriers have been used for the efficient encapsulation of hydrophobic bioactive molecules such as vitamin D. The prepared potato protein based carriers protected vitamin D and extended its shelf-life when tested under different storage conditions (David and Livney 2016; Waglay et al. 2014).
4.4.3 Gelatins
Gelatins are animal derived proteins that could be obtained through partial either acid or alkaline hydrolysis of collagen. The hydrolysis could undergo enzymatically as well. Gelatins are classified into type A and type B according to the method of preparation. For instance, Gelatin type A is obtained from the skin of bovine, porcine or fish via acid hydrolysis procedure. However, gelatin type B is extracted from the bones of bovine, fish, or porcine through alkaline hydrolysis procedure (Shishir et al. 2018; Patel et al. 2008).
Gelatins have been reported as good biomaterials for preparation of carrier systems owing to their high biocompatibility, biodegradability as well as water retention ability. In addition, they are characterized by being non-carcinogenic and non-immunogenic biopolymer (Shishir et al. 2018; Chen et al. 2017).
Gelatin has proven their effeciency as biomaterials for the preparation of carrier systems incorporating wide range of bioactive compounds e.g. antimicrobial agents, antioxidants as well as some nutraceuticals (Chen et al. 2017; Gómez-Mascaraque et al. 2017).
4.4.4 Soy Proteins
Soy proteins are the highest portions of legume proteins. Soy proteins are considered to be promising biomaterials owing to their good physicochemical properties in terms of emulsification, fat absorption, gel formation, water binding capability, and antioxidant ability. Soy proteins consist of glycinin (11S globulin) and conglycinin (7S globulin). Carriers made of soy protein have been used to encapsulate diffferent bioactive compounds such as algal oil, curcumin, tomato oleoresin, paprika oleoresin, and lycopene (Ho et al. 2017; Dai et al. 2017; Lia et al. 2015).
In addition, soy proteins have been used with polysaccharides for preparation of blended encapsulate systems that showed enhanced stability and anti-oxidative properties. For instance, a study showed that a carrier system composed of soy protein and chitosan together was a successful encapsulate of algal oil with enhanced encapsulation efficiency reaching around 97.36% as well as high oxidative stability compared to soy protein alone. In addition, another study showed that the use of soy protein and gum acacia together to encapsulate tomato oleoresin showed better emulsifying features, higher biocompatibility as well as encapsulation efficiency than single soy protein carrier systems. Furthermore, conjugated soy protein with some polysaccharides revealed good protection ability of lycopene from light, temperature and humidity during storage conditions. In addition, the system showed higher capability to control the lycopene release within gastric conditions (Shishir et al. 2018; Lia et al. 2015; Yuan et al. 2017b).
4.4.5 Whey Proteins
Whey proteins are characterized by having superior biological characteristics especially emulsifying and gelling properties. Therefore, they are typically used in hydrogels preparations as well as nanoparticle systems through their conjugation with several polysaccharides. For instance, β-lactoglobulin was utilized in different forms i.e. pre-heat treated, cross-linked or even untreated form to incorporate various bioactive molecules e.g. sour cherries anthocyanins. It was found that cross-linked protein form reached the highest encapsulation efficiency up to around 64.69%. While both the cross-linked and pre-treated forms showed higher protection effeciency for anthocyanins against the gastric juice in the stomach compared to the untreated form. Consequently, this allowed the anthocyanins be released into the gut (Oancea et al. 2017). On the other hand, α-lactalbumin nanotubes were used successfully to incorporate caffeine with encapsulation efficiency of almost 100%. This proves the high capability of α-lactalbumin nanotubes to encapsulate bioactive compounds (Fuciños et al. 2017b).
4.5 Polysaccharide Micro and Nanocarriers
Polysaccharides, long polymeric chains of monosaccharides linked together by glycosidic linkages, can be used as edible encapsulating polymeric matrix through either microencapsulation or nanoencapsulation approaches according to the final dimensions of the prepared formulation. Polysaccharides have been reported as promising encapsulating systems for incorporating both additive and bioactive compounds used in food packaging systems. Among the most commonly used encapsulated additive and bioactive materials used in food packaging systems are: (a) natural nutraceuticals e.g. minerals, probiotics & vitamins, (b) antimicrobial agents, (c) antioxidants, and (d) anti-softening agents (Raybaudi-Massilia and Mosqueda-Melgar 2012; Tapia et al. 2007; Rojas-Graü et al. 2009).
Encapsulation of the above-mentioned compounds have the advantage of: (a) preserving & protecting them against the surrounding environment, (b) prolonging their shelf life, and (c) enhancing their safety and sensory features (Dubey et al. 2009; Parra 2010; Raybaudi-Massilia and Mosqueda-Melgar 2012). The efficiency of these encapsulated systems has been reported for wide range of various food products including dairy products, fresh fruit & vegetables cuts, as well as meat and poultry products (Olivas and Barbosa-Cánovas 2005; Min and Ahn 2005; Waghmare 2020; Cerqueira et al. 2009). Many polysaccharides have been reported for their efficiency in encapsulating various additives and bioactive agents mandatory in food packaging.
4.5.1 Cellulose & Cellulose Derivatives
Cellulose, the first most abundant polymer in nature, as well as its derivatives including carboxymethyl cellulose (CMC), hydroxylpropyl methyl cellulose (HPMC), and methyl cellulose (MC) have been reported as efficient polysaccharides for encapsulating antimicrobial agents in food packaging to prevent the growth of microorganisms and prolong the shelf life of packaged food. For example, cellulose based systems incorporating various antimicrobial compounds e.g. nisin and Pediocin, an antimicrobial peptide produced by Pediococcus sp., have shown good antimicrobial potency against L. monocytogenes, L. innocua, and Salmonella sp. This has been proven when tested on fresh fruits cuts such as strawberry and sliced meat products (Nguyen et al. 2008; Park et al. 2005).
In addition, CMC and HPMC based carriers of some antimicrobial agents such as sodium benzoate, sodium propionate, potassium sorbate, etc. have shown a high inhibitory effect of fungal and mold growth such as Penicillum digitatum and Penicilllum italicum when tested with fresh pistachio and oranges (Sayanjali et al. 2011; Valencia-Chamorro et al. 2009). Furthermore, HPMC based carrier systems incorporating some antioxidants that have shown good inhibitory effects on lipid oxidation of almond.
4.5.2 Chitosan
Another example for polysaccharides involved in encapsulation of antimicrobials in food packaging applications, is chitosan, which is obtained from chitin. Chitin is counted as the second most abundant natural polysaccharide after cellulose. Chitosan has been reported for its ability to encapsulate different antimicrobial agents during food packaging e.g. acetic acid, sodium benzoate, sodium diacetate, sodium lactate, potassium sorbate, propionic acid, lauric acid, cinnamaldehyde, vanillin, lysozymes, nisin, etc. (Dutta et al. 2009; Ouattara et al. 2000; Duan et al. 2007; Ye et al. 2008). Chitosan based systems have shown high efficiency in protecting salmon, meat products and fresh fruit cuts against various microorganisms. These include bacteria, fungi and molds such as Lactobacillus sakei, Serratia liquefaciens, L. monocytogenes, Pseudomonas Fluorescens, Saccharomyces cerevisiae, Escherichia coli, Cladosporium sp., Rhizopus sp, among others (Ojagh et al. 2010; Fajardo et al. 2010; Siripatrawan and Noipha 2012).
4.5.3 Alginate
Alginates, anionic polysaccharide chains obtained from brown algae cell walls, have been used as encapsulating systems for some wide range of antimicrobial agents such as nisin, lysozymes, sodium diacetate, sodium lactate, enterocins A and B, etc., to protect different meat and poultry products against wide range of microorganisms such as Brochothrix thermosphacta, Salmonella enterica, Staphylococcus aureus, Listeria monocytogenes (Natrajan and Sheldon 2000; Millette et al. 2007; Datta et al. 2008; Neetoo et al. 2010; Marcos et al. 2008).
Furthermore, alginate based encapsulating systems have been reported as promising carriers for some essential oils as well as their active ingredients such as Spanish oregano, Chinese cinnamon, winter savory, lemongrass, oregano, vanillin, palmarose, eugenol, geraniol and citral. They showed high efficiency in protecting meat & poultry products as well as fresh fruit cuts in refrigerators against wide range of microorganisms such as Salmonella enterica, Listeria innocua, Escherichia coli, native flora, in addition to mesophilic and psychotropic bacteria, molds and yeasts (Oussalah et al. 2006, 2007; Rojas-Graü et al. 2007; Raybaudi-Massilia et al. 2008a; Olivas et al. 2007; Seol et al. 2009).
4.5.4 Starch
Starch, composed of polysaccharide chains consisting of glucose monomers linked together via α 1,4 linkages, based carriers systems incorporating antioxidants agents such as rosemary oleoresin, tocopherols, stearic acid, etc., reported inhibitory effect on lipid oxidation and moisture loss in food packaging (Wu et al. 2000, 2001; Hargens-Madsen et al. 1995). Tables 7.5 and 7.6 summarize the use of polysaccharides as carriers.
5 Technological Challenges, Food Integrity and Regulatory Manifestations in Encapsulation
5.1 Technological Challenges in Preparation of Micro and Nanoencapsuled Systems
Although micro- and nanoencapsulation techniques have been widely used to prepare successful carrier systems for bioactive compounds, not all of them have been applied on large scale for pharmaceutical and nutraceutical applications. This returns back to the necessity of more long term and extensive studies towards these techniques to assure their credibility to be used on large scale. The used techniques should be tested again in terms of their impact on the chemical and physical stabilities of both the encapsulating systems and encapsulated compounds. For instance, concerning the newly emerged techniques, only freeze drying and spray drying are the most extensively used techniques in the industrial production (de Souza et al. 2017; Đorđević et al. 2016).
Even the traditional techniques still need more investigations to overcome their drawbacks and maximize their benefits. For example, it is advisable to discover more biomaterials that could be used in the encapsulate development. Furthermore, studies should be conducted to investigate the limitation of each materials and study how these could be improved (Shishir et al. 2018).
For instance, it is highly challengeable to incorporate liposomes in food owing to the high semi-permeability while low physical stability of the membranes. For successful preparation of liposomal structures as carrier systems in food packaging, it is mandatory to understand deeply the stability and solubilization features of the encapsulated compounds as well as their effect on the structure and properties of bilayer membrane of lipids in terms of fluidity, molecule dynamics and micropolarity (Brandelli et al. 2016; Frenzel and Steffen-Heins 2015; Yoshimoto et al. 2007).
Another technological challenge in encapsulate system is the tailoring of the ratio between oily and aqueous phases during the preparation of emulsions. In addition, the selection of the appropriate type of surfactant or the emulsifying agent as well as the proper required concentrations are considered as challenging aspects (Brandelli et al. 2016; Rayner et al. 2014).
5.2 Safety Aspects
Nanotechnology applications have gained high popularity in encapsulation approach of bioactive compounds in food and pharmaceutical industry. Although they showed promising and interesting solutions to overcome the drawbacks of using the bare bioactive compounds, some environmental and safety concerns have been raised during the past few years. The raised fears are related to the effect of nano- sized delivery systems on both the environment and the human health. The point of view for many researchers is attributed to the nanoscale systems potential toxicity risk on both the human hygiene and the surrounding environment (Katouzian et al. 2017).
Therefore, safety information concerning the size of the nanoencapsulated systems should be furtherly assessed especially their long-term toxicity (Katouzian et al. 2017; He and Hwang 2016; Ezhilarasi et al. 2013).
For instance, the small size of nanoparticles allows them to cross the biological barriers easily leading to probable interference with some biological reaction resulting in possible toxicological effects. Furthermore, the high surface area and consequently the high reactivity of the small sized nanoparticles could lead to acute inflammation and irritation (Jafari et al. 2017; Zhang et al. 2007; Fröhlich and Roblegg 2012).
Finally, the chemicals such as surfactants, stabilizing agents, etc. could have some toxicological effects on cells. Therefore, it is advisable to use green and naturally derived and FDA approved biomaterials during the preparation procedures. Moreover, it has been proven through cell viability assessments that coating of the prepared nanoparticles leads to decreasing the adverse effects on the cells (Shishir et al. 2018; He and Hwang 2016; Cruz et al. 2015; El Badawy et al. 2011; Seeli and Prabaharan 2016).
5.3 Regulatory Aspects
Unfortunately, the regulatory aspect concerning the use of nanoparticle systems in food and drug industry has not been firmly legislated globally till now. Most of the countries do not have particular roles to estimate the risk of the encapsulated nano-products or even to limit or minimize their risk factors (de Souza et al. 2017; Katouzian et al. 2017).
However, only the European Union (EU) has started recently to initiate clear regulatory vision for the use of nano- materials in industry (de Souza et al. 2017). Furthermore, Regulation (EU) no. 10/2011 states that nanoparticles cold lead to different toxicological features. Therefore, they need to be assessed on a case-by-case basis. For instance, nanoparticles can be utilized only if they have been approved or mentioned in Annex I of Regulation (EU) no. 10/2011.
The US Food and Drug Administration (FDA) does not possess precise principle relating the nanomaterials that could be used in the food and drug industry. However, it has issued guidelines concerning the regulatory and safety manifestations in new food industry technological approaches entitled “Draft Guidance for Industry” (Duvall and Knight 2011). The guidance stated some responsibilities that should be considered by the industrial sectors which include: (a) monitoring the changes that could occur to the food materials in terms of appearance of impurities or alteration in physicochemical properties, (b) evaluating the food products safety after being modified, (c) submitting a regulatory assessment to US FDA, and (d) specifying a regulatory concern for the new food products consumption. USA FDA states that the current law protocols are proper for the assessment of nanomaterials safety (Shishir et al. 2018; Katouzian et al. 2017).
6 Future Trends
Active food packaging through nano and micro-encapsulation is a method that has been gradually integrated into food industries to improve the quality and safety of food. Numerous gaps are still existing and hinder the development of this widely growing field. It needs to be easily accessible by industry and consumers. Safety and regulatory matters are being improved by the national organizations to establish convenient management for the progression of active food packaging.
The great challenge is to come with a product that is safe, high in quality, with extended shelf life, and able to prevent active microorganisms, moisture, gases, and mechanical forces. Thus, the process of active packaging and intelligent packaging has progressed to overcome those challenges for safer and healthier food.
Food pathogenic microorganisms present one of the serious problems that shortened the product shelf life and lead to food spoilage, and so reducing its quality. Multiple methods have come with promising ways to incorporate bioactive materials (e.g., essential oils) that have antimicrobial activity. Nevertheless, an urgent need to develop new substances with higher features is essential to get the most use of that active materials in the food industry. In this regard, the role of nanotechnology is an attractive system to improve and fulfill the different related issues.
The utilization of antimicrobial materials is remarkable since it allows the control of the pathogenic microorganisms by delaying or terminating their actions. This method can guarantee microbiologically safe food with no chemical additives and prolonged shelf-life. Multiple plant extracts showed magnificent antimicrobial behavior without negatively affecting the organoleptic properties of the food and so-called as Generally Recognized As Safe” (GRAS).
Nanotechnology has shown outstanding progression in numerous sectors including food technology. This extended to involve different aspects of active food packaging (e.g. controlled release of the delivery systems, nano-sensors for food safety, and nanoencapsulation or emulsion for low stability bioactive. Although nanotechnology strategy is promising in many sectors, food is not progressed as like as other fields (e.g., cosmetics). Extensive research studies are substantially needed to evaluate the efficiency and biosafety of such novel packaging in food through regulator bodies (e.g., the Food and Drug Administration). Scientists still required a research’s increase and knowledge in the field of active food packaging by investigating the control of the release kinetics and the development of products that economically feasible.
7 Conclusion
The encapsulation of the bioactive compounds helps in preserving their activities, enhancing the stability, and improving the release profile. Different materials could be used for the microencapsulataion of the bioactive ingredients such as polysaccharides, proteins, and lipids. The successful encapsulation in either micro or nano-forms relies on different properties of both encapsulating agents and bioactive compounds. Different encapsulating techniques such as spray drying, molecular inclusion, freeze-drying, and emulsification are used to prepare both micro and nano-systems as well. Nanofibers are considered one of the main platforms that could be used to to encapsulate the bioactive ingredients in food packaging. Future studies are needed to solve any problems regarding micro and nano encapsulation techniques to enhance their efficiency as well as scalability.
References
A concise guide to active agents for active food packaging | Elsevier Enhanced Reader [Internet]. [cited 2020 Nov 30]. Available from: https://reader.elsevier.com/reader/sd/pii/S0924224418302760?token=022AB9E1FF4BBDFD1F36E75608D69A35F711C9EAD3A6DAD330D36E5DB5D6887A3E7612D769D095AE946C531685EFDA1C
Abdul-Fattah AM, Kalonia DS, Pikal MJ. The challenge of drying method selection for protein pharmaceuticals: product quality implications. J Pharm Sci. 2007;96(8):1886–916.
Abreu FO, Oliveira EF, Paula HC, de Paula RC. Chitosan/cashew gum nanogels for essential oil encapsulation. Carbohydr Polym. 2012;89(4):1277–82.
Aguiar J, Estevinho BN, Santos L. Microencapsulation of natural antioxidants for food application – the specific case of coffee antioxidants – a review. Trends Food Sci Technol. 2016;58:21–39.
Ali MR, Said RME. Assessment of the potential of Arabic gum as an antimicrobial and antioxidant agent in developing vegan “egg-free” mayonnaise. J Food Saf. 2020;40(2):e12771.
Amenta V, Aschberger K, Arena M, Bouwmeester H, Botelho Moniz F, Brandhoff P, et al. Regulatory aspects of nanotechnology in the agri/feed/food sector in EU and non-EU countries. Regul Toxicol Pharmacol. 2015;73(1):463–76.
Andreani L, Cercená R, Ramos BGZ, Soldi V. Development and characterization of wheat gluten microspheres for use in a controlled release system. Mater Sci Eng C. 2009;29(2):524–31.
Arroyo-Maya IJ, McClements DJ. Biopolymer nanoparticles as potential delivery systems for anthocyanins: fabrication and properties. Food Res Int. 2015;69:1–8.
Atarés L, Pérez-Masiá R, Chiralt A. The role of some antioxidants in the HPMC film properties and lipid protection in coated toasted almonds. J Food Eng. 2011;104(4):649–56.
Attallah OA, Shetta A, Elshishiny F, Mamdouh W. Essential oil loaded pectin/chitosan nanoparticles preparation and optimization via Box–Behnken design against MCF-7 breast cancer cell lines. RSC Adv. 2020;10(15):8703–8.
Bakry AM, Abbas S, Ali B, Majeed H, Abouelwafa MY, Mousa A, et al. Microencapsulation of oils: a comprehensive review of benefits, techniques, and applications. Compr Rev Food Sci Food Saf. 2016;15(1):143–82.
Baldwin EA, Nisperos MO, Chen X, Hagenmaier RD. Improving storage life of cut apple and potato with edible coating. Postharvest Biol Technol. 1996;9(2):151–63.
Ben-Yehoshua. S. Individual seal-packaging of fruit and vegetables in plastic film-a new postharvest technique. HortScience. 1985;20(1):32–7.
Bhat M, Sharma A, Rao NN. Carrageenan-based edible biodegradable food packaging: a review. J Food Sci Nutr. 2020;(5):69–75.
Brandelli A, Brum LFW, dos Santos JHZ. Nanobiotechnology methods to incorporate bioactive compounds in food packaging. 2016. p. 27–58.
Brandelli A, Ritter AC, Veras FF. Antimicrobial activities of metal nanoparticles. In: Mahendra R, Ranjita S, editors. Metal nanoparticles in pharma [Internet]. Cham: Springer; 2017a. p. 337–63. [cited 2020 Dec 1]. Available from: https://doi.org/10.1007/978-3-319-63790-7_15.
Brandelli A, Brum LFW, dos Santos JHZ. Nanostructured bioactive compounds for ecological food packaging, Environmental Chemistry Letters, vol. 15. Springer; 2017b. p. 193–204.
Calderón-Oliver M, Escalona-Buendía HB, Ponce-Alquicira E. Effect of the addition of microcapsules with avocado peel extract and nisin on the quality of ground beef. Food Sci Nutr. 2020;8(3):1325–34.
Cerqueira MA, Lima ÁM, Souza BWS, Teixeira JA, Moreira RA, Vicente AA. Functional polysaccharides as edible coatings for cheese. J Agric Food Chem. 2009;57(4):1456–62.
Chang CC, Liu DZ, Lin SY, Liang HJ, Hou WC, Huang WJ, et al. Liposome encapsulation reduces cantharidin toxicity. Food Chem Toxicol. 2008;46(9):3116–21.
Chang SK, Alasalvar C, Shahidi F. Superfruits: phytochemicals, antioxidant efficacies, and health effects – a comprehensive review. Crit Rev Food Sci Nutr. 2019;59(10):1580–604.
Chen L, Remondetto GE, Subirade M. Food protein-based materials as nutraceutical delivery systems, Trends in Food Science and Technology, vol. 17. Elsevier; 2006. p. 272–83.
Chen H, Zhang Y, Zhong Q. Physical and antimicrobial properties of spray-dried zein-casein nanocapsules with co-encapsulated eugenol and thymol. J Food Eng. 2014;144:93–102.
Chen H, Zhang Y, Zhong Q. Physical and antimicrobial properties of spray-dried zein–casein nanocapsules with co-encapsulated eugenol and thymol. J Food Eng. 2015;144:93–102.
Chen H, Yang M, Shan Z, Mansouri S, May BK, Chen X, et al. On spray drying of oxidized corn starch cross-linked gelatin microcapsules for drug release. Mater Sci Eng C. 2017;74:493–500.
Chiu PE, Lai LS. Antimicrobial activities of tapioca starch/decolorized hsian-tsao leaf gum coatings containing green tea extracts in fruit-based salads, romaine hearts and pork slices. Int J Food Microbiol. 2010;139(1–2):23–30.
Crisosto CH, Garner D, Doyle J, Day KR. Relationship between fruit respiration, bruising susceptibility, and temperature in sweet cherries. HortScience. 1993;28(2):132–5.
Cruz Z, García-Estrada C, Olabarrieta I, Rainieri S. Lipid nanoparticles: delivery system for bioactive food compounds. In: Microencapsulation and microspheres for food applications. Elsevier Science Ltd.; 2015. p. 313–31.
da Silva MP, Daroit DJ, Brandelli A. Food applications of liposome-encapsulated antimicrobial peptides, Trends in Food Science and Technology, vol. 21. Elsevier; 2010. p. 284–92.
Dai L, Sun C, Li R, Mao L, Liu F, Gao Y. Structural characterization, formation mechanism and stability of curcumin in zein-lecithin composite nanoparticles fabricated by antisolvent co-precipitation. Food Chem. 2017;237:1163–71.
Dasgupta N, Ranjan S, Mundra S, Ramalingam C, Kumar A. Fabrication of food grade vitamin E nanoemulsion by low energy approach, characterization and its application. Int J Food Prop. 2016;19(3):700–8.
Datta S, Janes ME, Xue QG, Losso J, La Peyre JF. Control of Listeria monocytogenes and Salmonella anatum on the surface of smoked salmon coated with calcium alginate coating containing oyster lysozyme and nisin. J Food Sci. 2008;73(2):M67–71.
David S, Livney YD. Potato protein based nanovehicles for health promoting hydrophobic bioactives in clear beverages. Food Hydrocoll. 2016;57:229–35.
de Mello MB, da Silva MP, Brandelli A, da Silveira NP, Jantzen MM, de Souza da Motta A. Characterization and Antilisterial effect of phosphatidylcholine Nanovesicles containing the antimicrobial peptide Pediocin. Probiotics Antimicrob Proteins. 2013;5(1):43–50.
de Souza SL, Madalena DA, Pinheiro AC, Teixeira JA, Vicente AA, Ramos ÓL. Micro- and nano bio-based delivery systems for food applications: in vitro behavior, Advances in Colloid and Interface Science, vol. 243. Elsevier B.V; 2017. p. 23–45.
Devi N, Sarmah M, Khatun B, Maji TK. Encapsulation of active ingredients in polysaccharide–protein complex coacervates, Advances in Colloid and Interface Science, vol. 239. Elsevier B.V; 2017. p. 136–45.
Dey A, Neogi S. Oxygen scavengers for food packaging applications: a review. Trends Food Sci Technol. 2019;90:26–34.
Dias DR, Botrel DA, Fernandes RVDB, Borges SV. Encapsulation as a tool for bioprocessing of functional foods. Curr Opin Food Sci. 2017;13:31–7.
Donsì F, Voudouris P, Veen SJ, Velikov KP. Zein-based colloidal particles for encapsulation and delivery of epigallocatechin gallate. Food Hydrocoll. 2017a;63:508–17.
Donsì F, Voudouris P, Veen SJ, Velikov KP. Zein-based colloidal particles for encapsulation and delivery of epigallocatechin gallate. Food Hydrocoll. 2017b;63:508–17.
Đorđević V, Paraskevopoulou A, Mantzouridou F, Lalou S, Pantić M, Bugarski B, et al. Encapsulation technologies for food industry, Food Engineering Series. Springer; 2016. p. 329–82.
Duan J, Park SI, Daeschel MA, Zhao Y. Antimicrobial chitosan-lysozyme (CL) films and coatings for enhancing microbial safety of mozzarella cheese. J Food Sci. 2007;72(9):M355–62.
Duan J, Cherian G, Zhao Y. Quality enhancement in fresh and frozen lingcod (Ophiodon elongates) fillets by employment of fish oil incorporated chitosan coatings. Food Chem. 2010;119(2):524–32.
Dubey R, Shami TC, Rao B. Microencapsulation technology and applications. Def Sci J. 2009;59(1):82–95.
Dutta PK, Tripathi S, Mehrotra GK, Dutta J. Perspectives for chitosan based antimicrobial films in food applications. Food Chem. 2009;114(4):1173–82.
Duvall MN, Knight K. FDA regulation of nanotechnology. Food Drug Agency FDA USA; 2011.
El Badawy AM, Silva RG, Morris B, Scheckel KG, Suidan MT, Tolaymat TM. Surface charge-dependent toxicity of silver nanoparticles. Environ Sci Technol. 2011;45(1):283–7.
Elakkiya T, Malarvizhi G, Rajiv S, Natarajan TS. Curcumin loaded electrospun Bombyx mori silk nanofibers for drug delivery. Polym Int. 2014;63(1):100–5.
Ezhilarasi PN, Karthik P, Chhanwal N, Anandharamakrishnan C. Nanoencapsulation techniques for food bioactive components: a review, Food and Bioprocess Technology, vol. 6. Springer; 2013. p. 628–47.
Fajardo P, Martins JT, Fuciños C, Pastrana L, Teixeira JA, Vicente AA. Evaluation of a chitosan-based edible film as carrier of natamycin to improve the storability of Saloio cheese. J Food Eng. 2010;101(4):349–56.
Faridi Esfanjani A, Jafari SM. Biopolymer nano-particles and natural nano-carriers for nano-encapsulation of phenolic compounds. Colloids Surf B Biointerfaces. 2016;146:532–43.
Fernández-Pan I, Carrión-Granda X, Maté JI. Antimicrobial efficiency of edible coatings on the preservation of chicken breast fillets. Food Control. 2014;36(1):69–75.
Francisco CRL, Heleno SA, Fernandes IPM, Barreira JCM, Calhelha RC, Barros L, et al. Functionalization of yogurts with Agaricus bisporus extracts encapsulated in spray-dried maltodextrin crosslinked with citric acid. Food Chem. 2018;245:845–53.
Freiberger EB, Kaufmann KC, Bona E, Hermes de Araújo PH, Sayer C, Leimann FV, et al. Encapsulation of roasted coffee oil in biocompatible nanoparticles. LWT Food Sci Technol. 2015;64(1):381–9.
Frenzel M, Steffen-Heins A. Impact of quercetin and fish oil encapsulation on bilayer membrane and oxidation stability of liposomes. Food Chem. 2015;185:48–57.
Fröhlich E, Roblegg E. Models for oral uptake of nanoparticles in consumer products. Vol. 291, toxicology. Elsevier; 2012. p. 10–7.
Fuciños C, Míguez M, Fuciños P, Pastrana LM, Rúa ML, Vicente AA. Creating functional nanostructures: encapsulation of caffeine into α-lactalbumin nanotubes. Innov Food Sci Emerg Technol. 2017a;40:10–7.
Fuciños C, Míguez M, Fuciños P, Pastrana LM, Rúa ML, Vicente AA. Creating functional nanostructures: encapsulation of caffeine into α-lactalbumin nanotubes. Innov Food Sci Emerg Technol. 2017b;40:10–7.
Gaikwad KK, Singh S, Negi YS. Ethylene scavengers for active packaging of fresh food produce. Environ Chem Lett. 2020;18(2):269–84.
Gómez-Mascaraque LG, Casagrande Sipoli C, de La Torre LG, López-Rubio A. Microencapsulation structures based on protein-coated liposomes obtained through electrospraying for the stabilization and improved bioaccessibility of curcumin. Food Chem. 2017;233:343–50.
Gulotta A, Saberi AH, Nicoli MC, McClements DJ. Nanoemulsion-based delivery systems for polyunsaturated (ω-3) oils: formation using a spontaneous emulsification method. J Agric Food Chem. 2014;62(7):1720–5.
Gülseren I, Guri A, Corredig M. Encapsulation of tea polyphenols in Nanoliposomes prepared with Milk phospholipids and their effect on the viability of HT-29 human carcinoma cells. Food Dig. 2012;3(1–3):36–45.
Gumus CE, Decker EA, McClements DJ. Gastrointestinal fate of emulsion-based ω-3 oil delivery systems stabilized by plant proteins: lentil, pea, and faba bean proteins. J Food Eng. 2017;207:90–8.
Gutiérrez L, Escudero A, Batlle R, Nerín C. Effect of mixed antimicrobial agents and flavors in active packaging films. J Agric Food Chem. 2009;57(18):8564–71.
Han C, Zhao Y, Leonard SW, Traber MG. Edible coatings to improve storability and enhance nutritional value of fresh and frozen strawberries (Fragaria x ananassa) and raspberries (Rubus ideaus). Postharvest Biol Technol. 2004;33(1):67–78.
Han J-W, Ruiz-Garcia L, Qian J-P, Yang X-T. Food packaging: a comprehensive review and future trends. Compr Rev Food Sci Food Saf. 2018;17(4):860–77.
Hargens-Madsen M, Schnepf M, Hamouz F, Weller C, Roy S. Use of edible films and tocopherols in the control of warmed over flavor. J Am Diet Assoc. 1995;95(9 Suppl):A41.
He X, Hwang HM. Nanotechnology in food science: functionality, applicability, and safety assessment. J Food Drug Anal. Elsevier Taiwan LLC. 2016;24:671–81.
He Z, Zhang X, Qi W, Huang R, Su R. Alginate-casein microspheres as bioactive vehicles for nutrients. Trans Tianjin Univ. 2015;21(5):383–91.
Hernández-Muñoz P, Almenar E, Ocio MJ, Gavara R. Effect of calcium dips and chitosan coatings on postharvest life of strawberries (Fragaria x ananassa). Postharvest Biol Technol. 2006;39(3):247–53.
Ho KKHY, Schroën K, San Martín-González MF, Berton-Carabin CC. Physicochemical stability of lycopene-loaded emulsions stabilized by plant or dairy proteins. Food Struct. 2017;12:34–42.
Honarvar Z, Hadian Z, Mashayekh M. Nanocomposites in food packaging applications and their risk assessment for health. Electron Physician. 2016;8(6):2531–8.
Hu Q, Fang Y, Yang Y, Ma N, Zhao L. Effect of nanocomposite-based packaging on postharvest quality of ethylene-treated kiwifruit (Actinidia deliciosa) during cold storage. Food Res Int. 2011;44(6):1589–96.
Hughes GA. Nanostructure-mediated drug delivery. Nanomed Nanotechnol Biol Med. 2005;1(1):22–30.
Hundre SY, Karthik P, Anandharamakrishnan C. Effect of whey protein isolate and β-cyclodextrin wall systems on stability of microencapsulated vanillin by spray–freeze drying method. Food Chem. 2015;174:16–24.
Ishwarya SP, Anandharamakrishnan C, Stapley AGF. Spray-freeze-drying: a novel process for the drying of foods and bioproducts. Trends Food Sci Technol. 2015;41(2):161–81.
Islam MZ, Kitamura Y, Kokawa M, Monalisa K, Tsai F-H, Miyamura S. Effects of micro wet milling and vacuum spray drying on the physicochemical and antioxidant properties of orange (Citrus unshiu) juice with pulp powder. Food Bioprod Process. 2017;101:132–44.
Jafari SM, He Y, Bhandari B. Production of sub-micron emulsions by ultrasound and microfluidization techniques. J Food Eng. 2007;82(4):478–88.
Jafari SM, Esfanjani AF, Katouzian I, Assadpour E. Release, characterization, and safety of Nanoencapsulated food ingredients. In: Nanoencapsulation of food bioactive ingredients. Elsevier; 2017. p. 401–53.
Jain A, Thakur D, Ghoshal G, Katare OP, Shivhare US. Characterization of microcapsulated β-carotene formed by complex coacervation using casein and gum tragacanth. Int J Biol Macromol. 2016a;87:101–13.
Jain A, Thakur D, Ghoshal G, Katare OP, Shivhare US. Characterization of microcapsulated β-carotene formed by complex coacervation using casein and gum tragacanth. Int J Biol Macromol. 2016b;87:101–13.
Jarunglumlert T, Nakagawa K, Adachi S. Influence of aggregate structure of casein on the encapsulation efficiency of β-carotene entrapped via hydrophobic interaction. Food Struct. 2015;5:42–50.
Jia Z, Dumont M-J, Orsat V. Encapsulation of phenolic compounds present in plants using protein matrices. Food Biosci. 2016a;15:87–104.
Jia Z, Dumont MJ, Orsat V. Encapsulation of phenolic compounds present in plants using protein matrices, Food Bioscience, vol. 15. Elsevier Ltd.; 2016b. p. 87–104.
Jiang Z, Neetoo H, Chen H. Efficacy of freezing, frozen storage and edible antimicrobial coatings used in combination for control of listeria monocytogenes on roasted Turkey stored at chiller temperatures. Food Microbiol. 2011a;28(7):1394–401.
Jiang Z, Neetoo H, Chen H. Control of listeria monocytogenes on cold-smoked Salmon using chitosan-based antimicrobial coatings and films. J Food Sci. 2011b;76(1):M22–6.
Jouki M, Yazdi FT, Mortazavi SA, Koocheki A. Quince seed mucilage films incorporated with oregano essential oil: physical, thermal, barrier, antioxidant and antibacterial properties. Food Hydrocoll. 2014;36:9–19.
Joye IJ, McClements DJ. Biopolymer-based nanoparticles and microparticles: fabrication, characterization, and application. Curr Opin Colloid Interface Sci. 2014;19(5):417–27.
Katouzian I, Faridi Esfanjani A, Jafari SM, Akhavan S. Formulation and application of a new generation of lipid nano-carriers for the food bioactive ingredients, Trends in Food Science and Technology, vol. 68. Elsevier Ltd.; 2017. p. 14–25.
Kegere J, Ouf A, Siam R, Mamdouh W. Fabrication of poly(vinyl alcohol)/chitosan/Bidens pilosa composite electrospun nanofibers with enhanced antibacterial activities. ACS Omega. 2019;4(5):8778–85.
Lazić V, Vivod V, Peršin Z, Stoiljković M, Ratnayake IS, Ahrenkiel PS, et al. Dextran-coated silver nanoparticles for improved barrier and controlled antimicrobial properties of nanocellulose films used in food packaging. Food Packag Shelf Life. 2020;26:100575.
Li M, Ioannidis N, Gogos C, Bilgili E. A comparative assessment of nanocomposites vs. amorphous solid dispersions prepared via nanoextrusion for drug dissolution enhancement. Eur J Pharm Biopharm. 2017;119:68–80.
Lia C, Wang J, Shi J, Huang X, Peng Q, Xueb F. Encapsulation of tomato oleoresin using soy protein isolate-gum aracia conjugates as emulsifier and coating materials. Food Hydrocoll. 2015;45:301–8.
Liao L, Luo Y, Zhao M, Wang Q. Preparation and characterization of succinic acid deamidated wheat gluten microspheres for encapsulation of fish oil. Colloids Surf B Biointerfaces. 2012;92:305–14.
Mahajan PV, Rodrigues FAS, Motel A, Leonhard A. Development of a moisture absorber for packaging of fresh mushrooms (Agaricus bisporous). Postharvest Biol Technol. 2008;48(3):408–14.
Marcos B, Aymerich T, Monfort JM, Garriga M. High-pressure processing and antimicrobial biodegradable packaging to control Listeria monocytogenes during storage of cooked ham. Food Microbiol. 2008;25(1):177–82.
Marsanasco M, Márquez AL, Wagner JR, Chiaramoni NS, Del V, Alonso S. Bioactive compounds as functional food ingredients: characterization in model system and sensory evaluation in chocolate milk. J Food Eng. 2015;166:55–63.
Mehmood T. Optimization of the canola oil based vitamin E nanoemulsions stabilized by food grade mixed surfactants using response surface methodology. Food Chem. 2015;183:1–7.
Millette M, Le Tien C, Smoragiewicz W, Lacroix M. Inhibition of Staphylococcus aureus on beef by nisin-containing modified alginate films and beads. Food Control. 2007;18(7):878–84.
Min B, Ahn DU. Mechanism of lipid peroxidation in meat and meat products – a review. Food Sci Biotechnol. 2005;14(1):152–63.
Moeiniafshari AA, Zarrabi A, Bordbar AK. Exploring the interaction of naringenin with bovine beta-casein nanoparticles using spectroscopy. Food Hydrocoll. 2015;51:1–6.
Moore ME, Han I, Acton J, Ogale A, Barmore C, Dawson P. Effects of antioxidants in polyethylene film on fresh beef color. J Food Sci. 2006;68:99–104.
Mosquera M, Giménez B, Da Silva IM, Boelter JF, Montero P, Gómez-Guillén MC, et al. Nanoencapsulation of an active peptidic fraction from sea bream scales collagen. Food Chem. 2014;156:144–50.
Nanotechnology: a New Frontier in Food Science [Internet]. [cited 2020 Nov 27]. Available from: https://www.ift.org/news-and-publications/food-technology-magazine/issues/2003/december/features/nanotechnology-a-new-frontier-in-food-science
Natrajan N, Sheldon BW. Inhibition of Salmonella on poultry skin using protein- and polysaccharide-based films containing a nisin formulation. J Food Prot. 2000;63(9):1268–72.
Neetoo H, Ye M, Chen H. Bioactive alginate coatings to control Listeria monocytogenes on cold-smoked salmon slices and fillets. Int J Food Microbiol. 2010;136(3):326–31.
Nesterenko A, Alric I, Silvestre F, Durrieu V. Vegetable proteins in microencapsulation: a review of recent interventions and their effectiveness. Ind Crop Prod. 2013a;42:469–79.
Nesterenko A, Alric I, Silvestre F, Durrieu V. Vegetable proteins in microencapsulation: a review of recent interventions and their effectiveness, Industrial Crops and Products, vol. 42. Elsevier; 2013b. p. 469–79.
Nguyen VT, Gidley MJ, Dykes GA. Potential of a nisin-containing bacterial cellulose film to inhibit Listeria monocytogenes on processed meats. Food Microbiol. 2008;25(3):471–8.
Ni S, Sun R, Zhao G, Xia Q. Quercetin loaded nanostructured lipid carrier for food fortification: preparation, characterization and in vitro study. J Food Process Eng. 2015;38(1):93–106.
Oancea AM, Aprodu I, Ghinea IO, Barbu V, Ioniţă E, Bahrim G, et al. A bottom-up approach for encapsulation of sour cherries anthocyanins by using β-lactoglobulin as matrices. J Food Eng. 2017;210:83–90.
Ogrodowska D, Tańska M, Brandt W. The influence of drying process conditions on the physical properties, bioactive compounds and stability of encapsulated pumpkin seed oil. Food Bioprocess Technol. 2017;10(7):1265–80.
Ojagh SM, Rezaei M, Razavi SH, Hosseini SMH. Effect of chitosan coatings enriched with cinnamon oil on the quality of refrigerated rainbow trout. Food Chem. 2010;120(1):193–8.
Olivas GI, Barbosa-Cánovas GV. Edible coatings for fresh-cut fruits. Crit Rev Food Sci Nutr. 2005;45:657–70.
Olivas GI, Mattinson DS, Barbosa-Cánovas GV. Alginate coatings for preservation of minimally processed “Gala” apples. Postharvest Biol Technol. 2007;45(1):89–96.
Oms-Oliu G, Soliva-Fortuny R, Martín-Belloso O. Edible coatings with antibrowning agents to maintain sensory quality and antioxidant properties of fresh-cut pears. Postharvest Biol Technol. 2008;50(1):87–94.
Ouattara B, Simard RE, Piette G, Bégin A, Holley RA. Inhibition of surface spoilage bacteria in processed meats by application of antimicrobial films prepared with chitosan. Int J Food Microbiol. 2000;62(1–2):139–48.
Oussalah M, Caillet S, Salmiéri S, Saucier L, Lacroix M. Antimicrobial effects of alginate-based film containing essential oils for the preservation of whole beef muscle. J Food Prot. 2006;69(10):2364–9.
Oussalah M, Caillet S, Salmiéri S, Saucier L, Lacroix M. Antimicrobial effects of alginate-based films containing essential oils on Listeria monocytogenes and Salmonella typhimurium present in bologna and ham. J Food Prot. 2007;70(4):901–8.
Pant A, Sängerlaub S, Müller K. Gallic acid as an oxygen scavenger in bio-based multilayer packaging films. Materials. 2017;10(5):489.
Park SI, Stan SD, Daeschel MA, Zhao Y. Antifungal coatings on fresh strawberries (Fragaria × ananassa) to control mold growth during cold storage. J Food Sci. 2005;70(4):202–7.
Parra RR. Microencapsulación de Alimentos. Rev Fac Nac Agron. 2010;63(2):5669–84.
Patel ZS, Yamamoto M, Ueda H, Tabata Y, Mikos AG. Biodegradable gelatin microparticles as delivery systems for the controlled release of bone morphogenetic protein-2. Acta Biomater. 2008;4(5):1126–38.
Patil YP, Jadhav S. Novel methods for liposome preparation, Chemistry and Physics of Lipids, vol. 177. Elsevier Ireland Ltd.; 2014. p. 8–18.
Piorkowski DT, McClements DJ. Beverage emulsions: recent developments in formulation, production, and applications. Food Hydrocoll. 2014;42:5–41.
Qian C, Decker EA, Xiao H, McClements DJ. Impact of lipid nanoparticle physical state on particle aggregation and β-carotene degradation: potential limitations of solid lipid nanoparticles. Food Res Int. 2013;52(1):342–9.
Ranjan S, Dasgupta N, Chakraborty AR, Melvin Samuel S, Ramalingam C, Shanker R, et al. Nanoscience and nanotechnologies in food industries: opportunities and research trends. J Nanoparticle Res. Kluwer Academic Publishers. 2014;16:1–23.
Raybaudi-Massilia R, Mosqueda-Melgar H. Polysaccharides as carriers and protectors of additives and bioactive compounds in foods. In: The complex world of polysaccharides. InTech; 2012. p. 27.
Raybaudi-Massilia RM, Rojas-Graü MA, Mosqueda-Melgar J, Martín-Belloso O. Comparative study on essential oils incorporated into an alginate-based edible coating to assure the safety and quality of fresh-cut Fuji apples. J Food Prot. 2008a;71(6):1150–61.
Raybaudi-Massilia RM, Mosqueda-Melgar J, Martín-Belloso O. Edible alginate-based coating as carrier of antimicrobials to improve shelf-life and safety of fresh-cut melon. Int J Food Microbiol. 2008b;121(3):313–27.
Rayner M, Marku D, Eriksson M, Sjöö M, Dejmek P, Wahlgren M. Biomass-based particles for the formulation of Pickering type emulsions in food and topical applications. Colloids Surf Physicochem Eng Asp. 2014;458(1):48–62.
Rodea-González DA, Cruz-Olivares J, Román-Guerrero A, Rodríguez-Huezo ME, Vernon-Carter EJ, Pérez-Alonso C. Spray-dried encapsulation of chia essential oil (Salvia hispanica L.) in whey protein concentrate-polysaccharide matrices. J Food Eng. 2012;111(1):102–9.
Rodriguez J, Martín MJ, Ruiz MA, Clares B. Current encapsulation strategies for bioactive oils: from alimentary to pharmaceutical perspectives. Food Res Int. 2016;83:41–59.
Rojas-Graü MA, Raybaudi-Massilia RM, Soliva-Fortuny RC, Avena-Bustillos RJ, McHugh TH, Martín-Belloso O. Apple puree-alginate edible coating as carrier of antimicrobial agents to prolong shelf-life of fresh-cut apples. Postharvest Biol Technol. 2007;45(2):254–64.
Rojas-Graü MA, Soliva-Fortuny R, Martín-Belloso O. Edible coatings to incorporate active ingredients to fresh-cut fruits: a review. Trends Food Sci Technol. 2009;20(10):438–47.
Rutz JK, Borges CD, Zambiazi RC, Crizel-Cardozo MM, Kuck LS, Noreña CPZ. Microencapsulation of palm oil by complex coacervation for application in food systems. Food Chem. 2017;220:59–66.
Salvia-Trujillo L, Rojas-Graü A, Soliva-Fortuny R, Martín-Belloso O. Physicochemical characterization and antimicrobial activity of food-grade emulsions and nanoemulsions incorporating essential oils. Food Hydrocoll. 2015;43:547–56.
Sangsuwan J, Rattanapanone N, Rachtanapun P. Effect of chitosan/methyl cellulose films on microbial and quality characteristics of fresh-cut cantaloupe and pineapple. Postharvest Biol Technol. 2008;49(3):403–10.
Sanguansri L, Augustin M-A. Microencapsulation and delivery of Omega-3 fatty acids. In: Functional food ingredients and nutraceuticals processing technologies. Taylor & Francis; 2007. p. 297–327.
Sayanjali S, Ghanbarzadeh B, Ghiassifar S. Evaluation of antimicrobial and physical properties of edible film based on carboxymethyl cellulose containing potassium sorbate on some mycotoxigenic Aspergillus species in fresh pistachios. LWT Food Sci Technol. 2011;44(4):1133–8.
Seeli DS, Prabaharan M. Guar gum succinate as a carrier for colon-specific drug delivery. Int J Biol Macromol. 2016;84:10–5.
Seol KH, Lim DG, Jang A, Jo C, Lee M. Antimicrobial effect of κ-carrageenan-based edible film containing ovotransferrin in fresh chicken breast stored at 5 °C. Meat Sci. 2009;83(3):479–83.
Shetta A, Kegere J, Mamdouh W. Comparative study of encapsulated peppermint and green tea essential oils in chitosan nanoparticles: encapsulation, thermal stability, in-vitro release, antioxidant and antibacterial activities. Int J Biol Macromol. 2019;126:731–42.
Shishir MRI, Xie L, Sun C, Zheng X, Chen W. Advances in micro and nano-encapsulation of bioactive compounds using biopolymer and lipid-based transporters. Trends Food Sci Technol. 2018;78:34–60.
Siracusa V, Blanco I. Bio-polyethylene (bio-PE), bio-polypropylene (bio-PP) and bio-poly(ethylene terephthalate) (bio-PET): recent developments in bio-based polymers analogous to petroleum-derived ones for packaging and engineering applications. Polymers. 2020;12(8):1641.
Siripatrawan U, Noipha S. Active film from chitosan incorporating green tea extract for shelf life extension of pork sausages. Food Hydrocoll. 2012;27(1):102–8.
Song Y, Liu L, Shen H, You J, Luo Y. Effect of sodium alginate-based edible coating containing different anti-oxidants on quality and shelf life of refrigerated bream (Megalobrama amblycephala). Food Control. 2011;22(3–4):608–15.
Sun S, Song Y, Zheng Q. Rheological behavior of heat-induced wheat gliadin gel. Food Hydrocoll. 2009;23(3):1054–6.
Suppakul P, Miltz J, Sonneveld K, Bigger SW. Active packaging technologies with an emphasis on antimicrobial packaging and its applications. J Food Sci. 2003;68(2):408–20.
Tan C, Xia S, Xue J, Xie J, Feng B, Zhang X. Liposomes as vehicles for lutein: preparation, stability, liposomal membrane dynamics, and structure. J Agric Food Chem. 2013;61(34):8175–84.
Tapia M. Using polysaccharide-based edible coatings to maintain quality of fresh-cut Fuji apples. LWT Food Sci Technol. 2008;41:139–47.
Tapia MS, Rojas-Graü MA, Rodríguez FJ, Ramírez J, Carmona A, Martin-Belloso O. Alginate- and gellan-based edible films for probiotic coatings on fresh-cut fruits. J Food Sci. 2007;72(4):E190–6.
Tapia MS, Rojas-Graü MA, Carmona A, Rodríguez FJ, Soliva-Fortuny R, Martin-Belloso O. Use of alginate- and gellan-based coatings for improving barrier, texture and nutritional properties of fresh-cut papaya. Food Hydrocoll. 2008;22(8):1493–503.
Tarhini M, Greige-Gerges H, Elaissari A. Protein-based nanoparticles: from preparation to encapsulation of active molecules. Int J Pharm Elsevier BV. 2017;522:172–97.
Tas CE, Hendessi S, Baysal M, Unal S, Cebeci FC, Menceloglu YZ, et al. Halloysite nanotubes/polyethylene nanocomposites for active food packaging materials with ethylene scavenging and gas barrier properties. Food Bioprocess Technol. 2017;10(4):789–98.
Tatar Turan F, Cengiz A, Kahyaoglu T. Evaluation of ultrasonic nozzle with spray-drying as a novel method for the microencapsulation of blueberry’s bioactive compounds. Innov Food Sci Emerg Technol. 2015;32:136–45.
Taylor TM, Davidson PM, Bruce BD, Weiss J. Ultrasonic spectroscopy and differential scanning calorimetry of liposomal-encapsulated nisin. J Agric Food Chem. 2005;53(22):8722–8.
Thompson AK, Hindmarsh JP, Haisman D, Rades T, Singh H. Comparison of the structure and properties of liposomes prepared from milk fat globule membrane and soy phospholipids. J Agric Food Chem. 2006;54(10):3704–11.
Thorat AA, Dalvi SV. Liquid antisolvent precipitation and stabilization of nanoparticles of poorly water soluble drugs in aqueous suspensions: recent developments and future perspective. Chem Eng J. 2012;181–182:1–34.
Timilsena YP, Wang B, Adhikari R, Adhikari B. Advances in microencapsulation of polyunsaturated fatty acids (PUFAs)-rich plant oils using complex coacervation: a review. Food Hydrocoll. 2017;69:369–81.
Tsironi T, Ntzimani A, Gogou E, Tsevdou M, Semenoglou I, Dermesonlouoglou E, et al. Modeling the effect of active modified atmosphere packaging on the microbial stability and shelf life of Gutted Sea Bass. Appl Sci. 2019;9(23):5019.
Uysal Unalan I, Cerri G, Marcuzzo E, Cozzolino CA, Farris S. Nanocomposite films and coatings using inorganic nanobuilding blocks (NBB): current applications and future opportunities in the food packaging sector. RSC Adv. 2014;4(56):29393–428.
Valencia-Chamorro SA, Pérez-Gago MB, del Río MÁ, Palou L. Effect of antifungal hydroxypropyl methylcellulose (HPMC)-lipid edible composite coatings on postharvest decay development and quality attributes of cold-stored “Valencia” oranges. Postharvest Biol Technol. 2009;54(2):72–9.
Varshosaz J, Eskandari S, Tabbakhian M. Freeze-drying of nanostructure lipid carriers by different carbohydrate polymers used as cryoprotectants. Carbohydr Polym. 2012;88(4):1157–63.
Waghmare RB. Encapsulation techniques for delivery of bioactive compounds in Milk and dairy products – a review. J Dairy Res Technol. 2020;3(1):1–9.
Waglay A, Karboune S, Alli I. Potato protein isolates: recovery and characterization of their properties. Food Chem. 2014;142:373–82.
Wahbi W, Siam R, Kegere J, El-Mehalmey WA, Mamdouh W. Novel inulin electrospun composite nanofibers: prebiotic and antibacterial activities. ACS Omega. 2020;5(6):3006–15.
Winestrand S, Johansson K, Järnström L, Jönsson LJ. Co-immobilization of oxalate oxidase and catalase in films for scavenging of oxygen or oxalic acid. Biochem Eng J. 2013;72:96–101.
Wu Y, Rhim JW, Weller CL, Hamouz F, Cuppett S, Schnepf M. Moisture loss and lipid oxidation for precooked beef patties stored in edible coatings and films. J Food Sci. 2000;65(2):300–4.
Wu Y, Weller CL, Hamouz F, Cuppett S, Schnepf M. Moisture loss and lipid oxidation for precooked ground-beef patties packaged in edible starch-alginate-based composite films. J Food Sci. 2001;66(3):486–93.
Yalavarthi P, Dudala T, Mudumala N, Pasupati V, Thanniru J, Vadlamudi H, et al. A perspective overview on lipospheres as lipid carrier systems. Int J Pharm Investig. 2014;4(4):149.
Ye M, Neetoo H, Chen H. Control of Listeria monocytogenes on ham steaks by antimicrobials incorporated into chitosan-coated plastic films. Food Microbiol. 2008;25(2):260–8.
Yoshimoto M, Sakamoto H, Yoshimoto N, Kuboi R, Nakao K. Stabilization of quaternary structure and activity of bovine liver catalase through encapsulation in liposomes. Enzym Microb Technol. 2007;41(6–7):849–58.
Youssef Ahmed M, EL-Sayed SM, EL-Sayed HS, Salama HH, Dufresne A. Enhancement of Egyptian soft white cheese shelf life using a novel chitosan/carboxymethyl cellulose/zinc oxide bionanocomposite film. Carbohydr Polym. 2016;151:9–19.
Yuan Y, Kong Z-Y, Sun Y-E, Zeng Q-Z, Yang X-Q. Complex coacervation of soy protein with chitosan: constructing antioxidant microcapsule for algal oil delivery. LWT Food Sci Technol. 2017a;75:171–9.
Yuan Y, Kong ZY, Sun YE, Zeng QZ, Yang XQ. Complex coacervation of soy protein with chitosan: constructing antioxidant microcapsule for algal oil delivery. LWT Food Sci Technol. 2017b;75:171–9.
Zeeb B, Herz E, McClements DJ, Weiss J. Impact of alcohols on the formation and stability of protein-stabilized nanoemulsions. J Colloid Interface Sci. 2014;433:196–203.
Zhang X, Sun H, Zhang Z, Niu Q, Chen Y, Crittenden JC. Enhanced bioaccumulation of cadmium in carp in the presence of titanium dioxide nanoparticles. Chemosphere. 2007;67(1):160–6.
Zhang L, Hayes DG, Chen G, Zhong Q. Transparent dispersions of milk-fat-based nanostructured lipid carriers for delivery of β-carotene. J Agric Food Chem. 2013;61(39):9435–43.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Shetta, A., Ali, I.H., Elshishiny, F., Mamdouh, W. (2022). Different Approaches for the Inclusion of Bioactive Compounds in Packaging Systems. In: Jafari, S.M., Silva, A.S. (eds) Releasing Systems in Active Food Packaging. Food Bioactive Ingredients. Springer, Cham. https://doi.org/10.1007/978-3-030-90299-5_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-90299-5_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-90298-8
Online ISBN: 978-3-030-90299-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)