Abstract
Due to numerous human activities, organisms have been transported and either accidentally or deliberately introduced all around the globe. Biological invasions are now considered to be one of the main drivers of global change because many invasive plants have severe ecological, economic, and health consequences. Thus, there is an ever-growing need to better understand invasions to determine how specific plant species are able to establish in communities and, in many cases, expand their range. Here, we describe the invasion process and how it contributes to the invasion of plant communities. We present an invasion-factor framework (IFF) model that uses three factors (climate dynamics, ecosystem resistance, and invader fitness) to explain how each plays a role in the introduction of plants and their ultimate failure or success (i.e., becoming invasive). The invasion of plant communities starts with the uptake of propagules from the native range, followed by their transport to and release into a new territory, where they become established and can spread or expand. Propagule pressure, prior adaptation, anthropogenically induced adaptation to invade, and post-introduction evolution are several theories that have been posed to explain the establishment of invasive plants. Further, traits of invasive plants, either before (existing) or after (developed) introduction, provide a mechanistic understanding with direct ties to the three factors of the IFF. The IFF is a general guide with which to study the invasion process based on specific factors for individual invaders and their target communities. The IFF combines (a) climatic dynamics, analogous to environmental filters; (b) ecosystem resistance, which prevents invasive plants from becoming established even if they are able to overcome the climate factor; and (c) invader fitness, relating to the genetic diversity of invasive plants, which allows them to become established after overcoming climate and ecosystem resistance factors. Case studies from the literature provide examples of research investigating each of the three factors of the IFF, but none exist that describe all the factors at once for any given invasive plant species. The application of the IFF for management is most appropriate once an invasive plant has become established, as preventative measures before this point rely only on accurate identification (detection) and removal (response). The IFF model should be considered as a tool to establish research priorities and identify components in the invasion process and inform restoration efforts. We advocate that the IFF should be integrated into management practices to help in the decision-making process that contributes to more effective practices that reduce the occurrence and impacts of invasive plants in a range of communities.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Climate factor
- Ecosystem resistance factor
- Invader fitness factor
- Multistep invasion process
- Phenotypic trait diversity
- Propagule pressure
- Spatial scales
2.1 Introduction
In his book entitled The Ecology of Invasions by Animals and Plants, Charles Elton (1958) referred to biological invasions as “one of the great historical convulsions in the world’s flora and fauna.” Invasions are now recognized as one of the main drivers of global change (Vitousek et al. 1996; Sala et al. 2000). As a result of various human activities, the number and importance of invasions have only increased in recent times. Through migration, colonization, transport, and international commerce, human activities have moved plants to new regions for hundreds of years, and these events have contributed greatly to the introduction of a range of species around the globe (Mack et al. 2000; Crosby 2003; Bossdorf et al. 2005). Although many naturalized and invasive plants are the product of accidental introduction as contaminants in agricultural products (e.g., seed lots, hay, wool fleeces, etc.) and attached to cargo and machinery, the vast majority of naturalized and invasive plants have been deliberately introduced, either for food, horticultural purposes (i.e., ornamental plants), or medicinal uses (Mack and Lonsdale 2001; Reichard and White 2001; Mack and Erneberg 2002; van Kleunen et al. 2018). Deliberately introduced plants experience benefits not available to accidentally introduced plants, such as protection during transport from the native to the new range, introduction of seeds or propagules in large numbers, introduction at several entry points, and post-introduction protection in a habitat suitable for survival and growth (Novak and Mack 1995; Mack et al. 2000).
As plants become invasive in a new territory, they can have profoundly negative ecological, conservation, economic, and health consequences (D’Antonio and Vitousek 1992; Wilcove et al. 1998; Mooney and Hobbs 2000; Pimentel et al. 2005). Because of their negative impacts, invasive plants have been a focus of scientific research and a source of concern to natural resource managers (Mack et al. 2000). Much of the research has been aimed at predicting which plants will become invasive through identification of traits or characteristics associated with invasiveness (Richardson and Pyšek 2006, 2007; Pyšek et al. 2009; van Kleunen et al. 2010, 2011) and at the identification of the attributes or conditions that make communities susceptible to invasion (Shea and Chesson 2002; Jenkins and Pimm 2003; Rejmanek et al. 2005; Richardson and Pyšek 2006; Didham et al. 2007). Invasive species research has led to the development and testing of numerous ecological and demographic hypotheses (Henneman and Memmott 2001; Blumenthal 2005; Hierro et al. 2005; Jeschke 2014; Lau and Schulties 2015). Beyond providing basic ecological and evolutionary insights (Sax et al. 2005), the research also has value to public and private entities when it can be applied in the management of invasive plants, including restoration (Wittenberg and Cock 2005; Gaertner et al. 2012).
2.1.1 The Multistep Process of Invasion
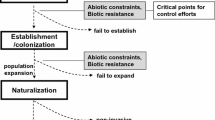
The invasion of plant communities occurs when organisms are introduced into a new range, where their descendants persist, proliferate, and spread (Mack et al. 2000; Colautti and MacIsaac 2004). The invasion process can be viewed as a series of steps in which propagules of a species (seeds, eggs, larvae, rhizome and stem fragments, mature individuals, etc.) are taken up from the native range, transported by a vector, released into a new area where they become established, and eventually spread beyond their points of introduction (Fig. 2.1) (Kolar and Lodge 2001; Sakai et al. 2001; Lockwood et al. 2005). However, according to the “tens rule” (Williamson and Fitter 1996), very few plants that are taken up in a native range and transported to a new territory will ever become invasive. That is because there is a high probability of mortality occurring at many points during the journey from a plants’ native range to its introduced range. First, organisms can die during transport, and following their release into a new area, invasive individuals may be extirpated because of climate mismatch (climate dynamics) or ecosystem resistance by native communities (see the Modeling section below). Alternatively, some invasive individuals survive and persist in these new locations and are said to be naturalized (i.e., they become incorporated within the resident community) (Richardson et al. 2000; Novak and Mack 2001). While many plants will remain in the naturalized category, a much smaller fraction of plants will go on to become invasive. At this point, the abundance of invasive plants has increased so that they are now prominent in the new range (and usually become more widespread or expand), and the plants’ negative ecological consequences are amplified, and the economic costs increase (Novak and Mack 2001).
The invasion process, illustrated as the area invaded over time (first x-axis) and possible management actions (second x-axis) that can be implemented depending on the area invaded. The point at which different filters in the invasion-factor framework (IFF) model are overcome is also included. Once the invasive species has become widespread and saturated across all suitable habitats, it may be possible to decrease the area occupied (dotted green line) by reestablishing one or more of the IFF factors
The transition from initial introduction of a plant species to it becoming invasive may occur relatively quickly, but this transition may also require an indefinite length of time (years to decades) (Fig. 2.1). The time delay in the transition to invasiveness is referred to as the lag phase. It is during the lag phase that extirpation of invasive populations is most likely to occur (see Application section). Alternatively, variation in the duration of the lag phase may be a result of multiple interacting factors. Mack et al. (2000) provide a detailed discussion of these factors, including (1) demographic lags during which the size of invasive populations increase slowly, (2) environmental and demographic stochasticity (which may signal the role of climate dynamics and ecosystem resistance), (3) additional introduction events that may occur during the lag phase but go undetected, and (4) the time required for post-introduction evolution to occur, based on invader fitness. If a plant species transitions to become invasive, the lag phase is followed by a period of rapid exponential proliferation in population size, population number, and the areal extent of the plant species in its new range: rapid range expansion is underway (Gurevitch et al. 2011). In time, the saturation phase occurs, and the geographical limits of an invasive plant species in its new range are realized and the population ceases to expand further (Fig. 2.1).
2.1.2 Native Range Dynamics
While a concatenation of events, including each step in the invasion process, determines whether plant communities are invaded, the first step of every invasion begins with the uptake of propagules from native populations. This means that the amount and distribution of phenotypic trait variability and genetic diversity within and among native populations can contribute to the likelihood of invasion. Therefore, the biogeographic and evolutionary history and ecological and biological characteristics of invasive plants in their native ranges can have a substantial impact on whether, or not, establishment followed by spread or expansion occurs (Novak 2007; Taylor and Keller 2007; Keller and Taylor 2008).
The probability that a plant species will become invasive appears to be influenced by the size of its native geographic range with those more widely distributed, more likely to become invasive (Pyšek et al. 2009; Jenkins and Keller 2011). If plants are broadly distributed in their native range, they have the ability to occupy more ecological habitats because they can tolerate a broader range of climate regimes (Pyšek et al. 2004, 2009). Thus, different populations of widely distributed native plants are more likely to possess prior adaptations that will contribute to fitness (survival and reproduction) and allow them to invade plant communities in the introduced range (see the General Theories section). Phenotypic and life history trait diversity (the invader fitness factor in the IFF model) also appear to be correlated with the distribution of plants in their native range: plants with a broad native geographic range size exhibit higher trait diversity (Jenkins and Keller 2011).
2.1.3 General Theories of Plant Community Invasion
The number of studies focused on invasive plants continues to increase as established hypotheses are tested, new hypotheses are proposed, ecological theories are applied, and empirical research is conducted (Enders et al. 2020). These theories include propagule pressure, prior adaptation, anthropogenically induced adaptation to invade (AIAI), and post-introduction evolution. They are important to touch on as they provide a basis for our current understanding and future research and are related to the conceptual framework of the IFF model.
Propagule pressure is defined as the number of individuals transported to and introduced into a new range or habitat (introduction effort) (Kolar and Lodge 2001; Lockwood et al. 2005; Simberloff 2009; Ricciardi et al. 2011; Blackburn et al. 2015). With high propagule pressure, invasive plants are buffered from the influence of stochastic events during all phases of the invasion process, and large founder populations and/or multiple introduction events will characterize the release of an invasive plant species in its new range (Simberloff 2009; Novak 2011). Propagule pressure not only holds demographic and ecological consequences for invasive plant introductions; it also has genetic consequences: high propagule pressure (compared to low propagule pressure) will likely increase the overall genetic and phenotypic diversity of populations in their new range, thus decreasing the potential for severe founder effects (Novak and Mack 2005; Simberloff 2009). In addition, high propagule pressure may lead to the formation of introduced populations that contain the genetic information of multiple native populations or admixtures (Kolbe et al. 2007; Novak 2011; van Boheemen et al. 2017). Propagule pressure can also influence the likelihood of invasion through the introduction of individual(s) with prior adaptations for invasiveness or by increasing the potential for post-introduction evolution.
Within evolutionary biology, the term preadaptation describes when a trait that evolved in one environment attains a different function (Futuyma et al. 2005). Rather than preadaptation, we will use the term prior adaptation (sensu Hufbauer et al. 2011). Prior adaptations involve the chance sampling of genotypes (and phenotypes) that have evolved in one environment (the native range), with the subsequent release of these genotypes into new environments (the introduced range), where they fortuitously contribute to fitness (i.e., the invasion of plant communities) (Dietz and Edwards 2006; Bossdorf et al. 2008; Hufbauer et al. 2011). Prior adaptation is associated with niche conservatism in invasive plants (Broennimann et al. 2007). Additionally, phenotypic plasticity that occurs among individuals in native populations would represent another form of prior adaptation contributing to invasions (Richards et al. 2006). Thus, invasions are not only influenced by the attributes of plants and communities, but invasions may also be determined by the performance of certain genotypes/phenotypes under specific environmental conditions. Greater propagule pressure increases the likelihood that individuals with prior adaptations will arrive in areas with the appropriate ecological condition, thus increasing the potential for invasion (although the potential to invade may be population- and location-specific).
Hufbauer et al. (2011) described a specific form of prior adaptation: anthropogenically induced adaptation to invade (AIAI). With this mechanism, prior adaptations may take place through adaptation to human-altered (disturbed) habitats in the native range of a species. Populations with these prior adaptations that arise in human-altered habitats should therefore increase in abundance in other areas disturbed by human activities. In addition, because such areas are frequented by humans, there is increased potential for the uptake and transport of propagules to new ranges. If these propagules are introduced into similar disturbed habitats in the new range, they should have high fitness in the new range. Such a scenario is likely to result in a truncated lag phase that results in a faster transition from introduction to establishment to spread or expansion. Finally, because human alteration of habitats is ongoing and ubiquitous around the globe, AIAI is likely to contribute to increased rates of invasion in the future.
Post-introduction evolution is now widely recognized as an important mechanism contributing to invasion (Lee 2002; Cox 2004; Facon et al. 2006; Novak 2007; Prentis et al. 2008; Colautti and Lau 2015; Estoup et al. 2016) and has been demonstrated in a growing number of studies (e.g., Maron et al. 2004; Blair and Wolfe 2004; Lavergne and Molofsky 2007; Cano et al. 2008; Dlugosch and Parker 2008; Lachmuth et al. 2010; Xu et al. 2010). Post-introduction evolution in the new range of an invasive plant species occurs in association with the following events: generation of admixture populations, outcrossing or hybridization among individuals with different genotypes, genetic reshuffling and/or recombination that can generate novel genotypes and phenotypes, and natural selection on these novel phenotypes [e.g., the evolution of increased competitive ability hypothesis (Blossey and Notzold 1995), see Modeling section]. Clearly, sufficient genetic diversity (especially additive genetic variance) within introduced populations is a prerequisite for post-introduction adaptive evolution (Lee 2002; Prentis et al. 2008; Estoup et al. 2016). Increased propagule pressure increases the likelihood that high levels of genetic diversity will occur within invasive populations and therefore set the stage for spread or expansion through post-introduction evolution (Novak and Mack 2005). These findings concerning post-introduction evolution suggest that it may be more difficult than previously thought to predict whether an invasion will occur, and its timing, because for different plants this process can require variable amounts of time for different populations, in different habitats.
In addition to these theories, there is a need to develop models or theoretical frameworks for gaining a better understanding of the factors that contribute to the invasion of plant communities. Now we will introduce modeling efforts that describe the factors that create barriers to invasiveness and then provide case studies that illustrate them.
2.2 Modeling Efforts
We introduced invasion using a large-scale, global viewpoint that combines the uptake and transport process (from the native range) with the introduction and establishment process (into the introduced range) of a non-native species. Here, we parse the factors that are contributing specifically to the spread or expansion of invasive plants in target communities. Our focus is on plant traits in relation to the IFF model, which accounts for climate dynamics (climate matching), ecosystem resistance, and invader fitness (genetic factors). These factors may be viewed as similar to the abiotic and biotic filters described in community assembly theory and previously related to niche and invasion theories (Keddy 1992; MacDougall et al. 2009; Fukami 2015).
2.2.1 Invasion Factors and Plant Traits
In order for invasive plants to successfully establish and spread or expand in a resident community, they must be well suited to the climate, able to outcompete native plants, and able to successfully increase population size and range across the new region (Theoharides and Dukes 2007; Hellmann et al. 2008). Research into the first necessary factor, climate matching, has noted certain characteristics common to invasive species that enable them to establish and expand in many climates (Jones et al. 2019). Hypotheses regarding the ability of invasive plants to increase population sizes and expand their ranges often focus either on characteristics that are common to invasive plants or on factors that make resident communities more likely to be invaded (Perkins and Nowak 2013). Invasive plants are hypothesized to exhibit fast growth, high seed production, strong dispersal abilities, and low metabolic costs (Blumenthal 2005). Such hypotheses have led to numerous studies comparing traits of invasive plants to co-occurring natives (Grotkopp et al. 2002; Cavaleri and Sack 2010).
Compared to noninvasive plants, invasive plants usually exhibit higher diversity in traits and greater phenotypic values for plant growth characteristics (Leishman et al. 2007; Pyšek and Richardson 2007; van Kleunen et al. 2010, 2011; Godoy et al. 2011; Jenkins and Keller 2011), but these differences can vary by specific traits and environmental context and among plant species (Daehler 2003; Leffler et al. 2014). Invasive plants tend to be positioned more towards the “fast” end of the leaf economic spectrum (Wright et al. 2004), with higher values of traits associated with fast growth (e.g., relative growth rate, specific leaf area, leaf N) than noninvasives in the resident communities that they invade (Baruch and Goldstein 1999; Leishman et al. 2007; Feng et al. 2008). Van Kleunen et al. (2011) conducted a meta-analysis of 125 invasive and 196 noninvasive plants and concluded that the invaders generally had significantly higher (better) values than noninvasive plants for 6 performance-related trait categories: physiology (e.g., photosynthetic rate, transpiration, nitrogen use efficiency, and water-use efficiency), leaf area allocation (e.g., leaf area index, leaf area ratio, and specific leaf area), shoot allocation (e.g., shoot-root ratio), growth rate (e.g., increase in size or biomass over time), size (e.g., biomass of roots, shoot, and whole plants, plant height, and total leaf area), and fitness (e.g., traits associated with reproductive output, seed germination, and survival). In addition, invasive plants typically exhibit higher levels of phenotypic plasticity, or plastic response, when compared to noninvasive or co-occurring native plants (Daehler 2003; Richards et al. 2006; van Kleunen et al. 2011; Davidson et al. 2011). High levels of phenotypic plasticity allow invasive plants to establish and persist in a wide range of habitats and environmental conditions, especially disturbed sites, which often exhibit high habitat heterogeneity at small spatial scales.
Existing Traits
What prevents plants with existing traits (that enable invasion) from becoming highly abundant in their native range? The community in the native range may possess an entirely different suite of trait combinations than what the targeted or resident community has, leading to the evolution of more efficient resource use. In a study comparing traits of long-term native and newly established invasive plants in multiple Mediterranean climate systems, the invader occupied higher slopes in graphical representations of two plant traits in four of the five systems studied, suggesting that invasive plants were more efficient at using resources than the native members of the targeted or resident communities (Funk et al. 2017). Trade-offs, such as a high photosynthetic rate (advantage) offset by low water-use efficiency (disadvantage), may be critical to structuring resident communities and maintaining coexistence (Chesson 2000; Kimball et al. 2013). In some cases, these trade-offs promote rare plant advantages. Studies of trade-offs for high growth rate and stress tolerance indicate that successful invaders appear to have an ability to achieve high values of both types of traits (Kimball et al. 2014b; Valliere 2019). Perhaps trade-offs structure resident communities that slowly assemble over time, and invasive plants come from communities in their native range where higher values of both types of traits have been favored. Higher growth rates and stress tolerance traits would be a novel combination enabling plants to become invasive in a new range by outcompeting noninvasives (Fig. 2.2).
Hypothetical diagram indicating the trait values of native plants (filled circles) and invasive plants (open circles) in a community. Coexistence in the native community is maintained by an among-plants trade-off between traits related to stress tolerance or fast growth. Niche segregation during community assembly should prevent two plants with the same combination of traits from becoming established, so this community would be resistant to invasion. Invasive plants may be able to invade due to higher values of both traits, indicated by a higher slope on the trade-off plot
Developed Traits
Another possibility for the successful establishment and expansion of invasive plants is that they develop new traits following introduction, through rapid phenotypic evolution (Stockwell et al. 2003; Xu et al. 2010). Researchers have struggled to understand how newly introduced plants may undergo rapid evolution despite low genetic diversity that often corresponds with invasion, but one possibility is that multiple introductions increase genetic diversity (Lockwood et al. 2005; Novak and Mack 2005; Lavergne and Molofsky 2007). Under this scenario, strong pressure(s) allow for post-introduction selection, even when populations have low genetic diversity (Dlugosch and Parker 2008).
The evolution of increased competitive ability (EICA) hypothesis is based on the idea that invasive plants will experience reduced selection for defensive traits upon occupying a new habitat (introduced range) without their natural enemies (the Enemy Release Hypothesis) and are thus able to invest more in traits that increase their competitive ability, which contributes to invasion (Blossey and Notzold 1995). The development of this hypothesis has led to a flurry of research, yet many studies have not been able to find strong support for EICA, instead only revealing invasives with trait values favoring competitive abilities and no explanation of how the traits evolved (Colautti et al. 2004). This was a similar finding for the Enemy Release Hypothesis (Jeschke et al. 2012), emphasizing the need for studies on traits of the same plants in the native range compared to the introduced range to understand the evolution of traits related to establishment and expansion (Thebaud and Simberloff 2001; Bossdorf et al. 2005).
2.2.2 Invasion-Factor Framework Model
Abiotic environmental conditions, the attributes of native ecosystems, and the fitness of invasive plants all determine whether or not plant communities will be invaded (Young et al. 2017). Evaluating the importance of all three factors (climate dynamics, ecosystem resistance, and invader fitness) for any invasive plant can help in better understanding how establishment and expansion have occurred and how to prevent and/or control it in the future. We present a conceptual model, the invasion-factor framework (IFF), which expands on Young et al. (2017), to integrate and evaluate the role and importance of the factors that act to prevent an invasive plants from establishing and expanding in resident communities (Fig. 2.3).
Conceptual diagram indicating potential filters or barriers to the success of invasive plants following dispersal to novel habitats. (a) Traits of invasives interact with climate conditions to determine whether the plants will be able to survive (climate factor). (b) Composition and functional traits of the existing biotic community interact with traits of the invasive plants to determine whether the plants will become invasive (ecosystem resistance factor). (c) Plasticity of invader traits, the number of dispersal events, resulting genetic diversity of the invader population, and the ability to adapt to changing climate and biotic conditions determine invader fitness. (d) If all filters are successfully overcome, then the plants will become invasive
Each factor in the IFF (Fig. 2.3) can be visualized as a static feature that acts sequentially, as this is useful for conceptual purposes. However, we expect these factors to be dynamic, even operating simultaneously, thus allowing for feedbacks and interactions. For example, climate dynamics may be based on long-term averages for precipitation and temperature, but extreme weather events are likely to lead to greater environmental stochasticity and the association with other climate parameters. Further, the factors of climate dynamics and ecosystem resistance may be more associated for broadly distributed invasive plant generalists making them more likely to be better established than invasive plants with specific characteristics (Pyšek et al. 2009; Saarinen et al. 2019). Ecological niche models have been used to map the potential distribution of invasive plants based on current and future conditions. Such models demonstrate that the range of many invasives are expected to increase with changing climates (Jarnevich et al. 2018). However, an understanding of how climate change has influenced current (and future) invasions requires the incorporation of ecosystem resistance and invader fitness factors into these models (Young et al. 2017).
Climate Dynamics
The climate factor (Fig. 2.3a) is analogous to an environmental filter that prevents non-native or introduced plants with traits not well suited to the new environmental conditions from establishing. Many invasive plants are generalists and can withstand a wide range of environmental conditions (Qian and Ricklefs 2006). Traits that allow for high growth rates may enable plants to be successful in resident communities that experience disturbance(s) and nutrient addition (Blumenthal 2006). Disturbances that lead to reduced or complete removal of vegetation create vacant niches that often favor invasive plants. Ruderal types are the most frequent and can quickly occupy these open spaces, thus altering the environmental conditions through priority effects (Hess et al. 2019). The anthropogenically induced adaptation to invade (AIAI) hypothesis, introduced in the previous section, suggests that plants adapted to human-disturbed habitats are able to easily colonize new locations across the globe as humans disturb and homogenize them (Hufbauer et al. 2011). Invasive plant trait values determine resource use under different environmental conditions; thus the traits, in combination with climate dynamics, can be used to help explain their ability to establish and (potentially) expand.
An additional stress for resident plant communities is climate change, which can hasten and in some cases cause composition and structure alterations, making them more vulnerable to invasion. A continuous disturbance, such as drought-induced mortality of native plants, will reduce ecosystem resistance and create vacant niches to be filled by invasives (Kimball et al. 2014a). An increase in fire frequency that is related to climate change can promote “fire-adapted” invasive plants (Garcia-Duro et al. 2019). Cheatgrass (Bromus tectorum) , an introduced annual grass in western North America, invades post-fire and increases the frequency of fires through positive feedback mechanisms, such as early phenology and increased fuel loads and connectivity. These mechanisms along with an annual life cycle favor cheatgrass in extreme drought and fire-prone conditions and ultimately lead to further expansion and prevention of native recovery (D’Antonio and Vitousek 1992; Mack et al. 2000; Brooks 2003).
The availability of abiotic resources influences the climate factor, such that ecosystems with limited resources tend to be less invaded than those with high resource levels (Colautti et al. 2006). Atmospheric N and other anthropogenic disturbances have added to soil nutrients, which frequently lead to increases in the abundance of invasive plants (Brooks 2003; Kimball et al. 2014a). Valliere (2019) found that native plants exhibited increased growth when grown alone with higher levels of N and water but were quickly outcompeted under high resource conditions when grown with invasive plants. The fluctuating resource theory predicts that invasion of resident communities increases with greater amounts of unused resources (Davis et al. 2000). The theory incorporates increases in available resources due to disturbances that either decrease resource use by native plants or increase abiotic resources.
Ecosystem Resistance
The ecosystem resistance factor (Fig. 2.3b), incorporating both biotic resistance and abiotic resistance properties, prevents invasive plants from establishing and expanding their range, even if they have traits that enable them to overcome the climate factor. Trait values of the resident community, as an example of biotic resistance, will influence what may happen to an invasive plant. Niche theory, competitive exclusion, and limiting similarity all support the idea that invasive plants would not be able to establish in a resident community with native plants possessing the same trait values (Feng et al. 2019; Walder et al. 2019). According to prior adaptions theory (see previous section), populations evolve to be better adapted to certain environmental conditions based on their traits. This would also occur for invasives with trait also occur for invasives with trait values similar to the resident community, allowing for their increased establishment and (potential) expansion. While climate factors may allow invasive plants with the ability to tolerate abiotic conditions to become established and expand, ecosystem resistance may either prevent invasive plants from being successful (Adler et al. 2013) or allow for coexistence.
Research on biotic resistance has more recently shifted to focus on resident community-level trait values. Community-weighted mean traits (CWM), calculated as the sum of the mean plant trait values multiplied by the mean biomass of each plant, and functional diversity (FD), a measure of the amount of dispersion in the trait values of plants in the resident community, have been used to estimate ecosystem processes, including biotic resistance (Diaz and Cabido 2001; Garnier et al. 2004; Mason and de Bello 2013). Resident communities with greater diversity appear to be more resistant to invasion (Fargione and Tilman 2005). In a study by Catford et al. (2019), CWM traits were a stronger measure of invasion risk than the trait values of an individual plant.
The ecosystem resistance factor includes all members of the resident community, including soil microbes, herbivores, pathogens, and top predators. Healthy, diverse, and fully functioning resident communities are less likely to become invaded. Disruptions to the resident community, such as disturbances due to habitat destruction, overgrazing, and agricultural activities, degrade ecosystem resistance factors and render these communities more vulnerable to invasion (Jauni et al. 2015). Invasive plants can disrupt food webs, reducing resident community resistance by altering nutrient cycling processes (Young et al. 2010). In addition, invasive plants may have the ability to produce molecules (e.g., allelopathic chemicals) that native soil microbes are not able to tolerate (the Novel Weapons Hypothesis), thus altering soil conditions and disrupting ecosystem resistance factors (Hierro and Callaway 2003). Invasive plants may promote the growth of soil pathogens that are especially harmful to native plants (Eppinga et al. 2006; Mangla and Inderjit 2008) or that attack the mycorrhizal fungi associated with them (Stinson et al. 2006). The symbiotic relationships with mycorrhizal fungi that are necessary for the growth of many native plants may not be required by invasive plants (Pringle et al. 2009). These non-mycorrhizal invasive plants can alter the soil community by decreasing the abundance and availability of arbuscular mycorrhizal fungi (Aslani et al. 2019).
Invader Fitness
The invader fitness factor (Fig. 2.3c) prevents invasive plants from establishing and expanding due to low fitness, even after overcoming the climate and ecosystem resistance factors. Invasive plants may have more phenotypic plasticity than naturalized or native plants, allowing them to achieve higher survival and reproductive success across a range of conditions, including “unfavorable” environments (Richards et al. 2006). One frequently discussed paradox in invasion science is the phenomenon of resident plant communities being invaded by individuals lacking the genetic variation representing the source populations (i.e., founder effects discussed in previous section), yet still able to quickly adapt and have high fitness in their new environment (Allendorf and Lundquist 2003; Schrieber and Lachmuth 2017). Multiple introductions from genetically distinct source populations can result in high genetic diversity within invasive population, genetic admixtures, and hybridization, which could explain increased invader fitness through the production of novel genotypes (Novak and Mack 1993; Novak and Mack 2001; Ellstrand and Schierenbeck 2006). Even without multiple introductions, founder effects do not appear to prevent invasive plants from becoming established in their introduced range, despite the reduction in genetic variation expected with such events (Dlugosch and Parker 2008). With sufficiently high adaptive genetic diversity, invasive plants are able to achieve high fitness and become established and expand (Dlugosch et al. 2015).
There are feedbacks and interactions among the factors of invader fitness, ecosystem resistance, and climate dynamics. For example, invasive plants may be better suited to adapt to ongoing climate change because they are already undergoing rapid phenotypic evolution enabling them to overcome the invader fitness factor (Nguyen et al. 2016). Evolution of earlier phenology would lead to seed production prior to competition with natives when resources become limited, especially during drought conditions (Franks et al. 2007; Alexander and Levine 2019). An example of a possible interaction between the ecosystem resistance and the invader fitness factors is the adaptation to open habitats with high light levels, which can increase invader fitness in disturbed areas (Corliss and Sultan 2016). In this case, the “fast growth traits” of the invasive plants, as described earlier (e.g., high growth rates, high leaf nitrogen content), allow for them to overcome ecosystem resistance factors, which have the potential to lead to higher rates of nutrient cycling. Certain invasive plants, such as cheatgrass, can actually have an effect on the climate factor by changing abiotic conditions such as soil moisture content at the microsite scale, in a positive feedback mechanism (Ehrenfeld 2003).
2.2.3 Application of Spatial Scales
Spatial scale influences whether and how these three factors contribute to preventing invasion. For example, the ecosystem resistance factor, at local spatial scales, results in a negative relationship between plant richness and risk of invasion. Conversely, at regional scales, there may be a positive relationship between native and invasive plant richness due to climate factors or habitat heterogeneity (Kennedy et al. 2002; Davies et al. 2005). Spatial scales may also influence the degree to which invasive plants are phylogenetically related to natives. At local scales, invasive plants that are phylogenetically related to natives would be prevented from becoming established because they are more likely to occupy similar ecological niches. This suggests that a phylogenetic component may be somewhat associated with ecosystem resistance of communities. At regional scales, neutral processes and dispersal limitations could allow phylogenetically similar plants to establish (Thuiller et al. 2010). The effect of disturbance on invasion is also thought to be scale-dependent, with greater effects of disturbance on ecosystem resistance factors at local spatial scales and over longer temporal scales (Jauni et al. 2015). Being mindful of the spatial scale of interest will help researchers to parse the relative contribution of the three factors in the IFF to an invasion. Essentially, our model can be used to assess the role of multiple processes, including interactive effects and feedbacks, across spatial scales, when assessing plant invasions.
2.3 Case Studies
The realization that invasive plants negatively impact native biodiversity and the environment, economies, and human health and well-being has resulted in research aimed at improving the understanding of the process and trying to lessen the impacts (Kumar and Singh 2020). Thus, an ever-growing body of research examining various aspects of invasion of resident plant communities, in particular, has been produced with useful insights. We conducted a systematic search of the literature and chose the most relevant and descriptive papers that used a “case study” format with a model plant being the focus. We examined the subcategories described by Young et al. (2017) for each factor (climate dynamics, ecosystem resistance, invader fitness) in the IFF model.
Case Studies: Climate Dynamics
Historical climate conditions (based on 30-year averages), extreme high/low precipitation and temperature, duration of extremes, and rising atmospheric [CO2] are the subcategories for climate dynamics that were described by Young et al. (2017) that would potentially allow for the establishment and expansion of invasive plants.
Adaptations to climate, and now extreme weather events, by invasive plants result in new community assemblages. Sheppard et al. (2016) report that Australian palm (Archontophoenix cunninghamiana), guava (Psidium guajava) , and umbrella tree (Schefflera actinophylla) are introduced and now naturalized plants in New Zealand. The potential for these invasive plants to expand is predicted to increase with climate change, and there is an increasing need to improve and/or incorporate best practice modeling, surveillance, and well-managed citizen science. In oak forests of northwest Spain, the evergreen shrub, silver wattle (Acacia dealbata) , and blue gum tree (Eucalyptus globulus) have adapted to altered climatic conditions and expanded their range, as the native flora are slower to respond to the new weather patterns (González-Muñoz et al. 2014). The authors project that basal area increment (BAI) or the average area occupied by tree stems, will be positive (indicating an increase in tree size) for natives and negative (indicating an increase in number of trees) for silver wattle and bluegum. These findings are different from earlier studies , which did not account for climate change. In Hawai’i Volcanoes National Park, plants that have the C4 photosynthetic pathway, including both invasive and native, are now found at higher elevations due to the climate change and an alteration of the fire regime (Angelo and Daehler 2013).
Invasive plants that have become established and are expanding under climate change often exhibit phenotypic plasticity, as described in the previous sections. In Chile, seed traits, specifically seed coat thickness and germination, of common dandelion (Taraxacum officinale) were evaluated in relation to climate variation (Molina-Montenegro et al. 2018). Thickness of seed coat decreased with latitude, while germination was highest in seeds originating from the southernmost part of the country. Bermudagrass (Cynodon dactylon) , South African lovegrass (Eragrostis plana) , Madagascar ragwort (Senecio madagascariensis) , and common gorse (Ulex europaeus) are the most important invasive plants infesting grasslands of southern Brazil (Guido et al. 2016). The strongest correlation for all four invaders was with decreasing water due to climate change. Less correlated were road density (landscape structure) and land use (human activity). Packer et al. (2017) conducted a global assessment of the biology of common reed (Phragmites australis), which is particularly invasive in North and Central America, and projected declining populations in the British Isles and along the east coasts of the United States and Panama. The latter due to rising sea levels associated with climate change.
Case Studies: Ecosystem Resistance
The main features that comprise a plant community include species composition, macro- and micro-fauna, available nutrients, and soil type (Young et al. 2017). These have been examined in biological and physical contexts to assess ecosystem resistance to invasive plants (Levine et al. 2004).
Establishment of and resistance to non-native plants are two of the most common research topics in invasion plant biology. In a classic ecosystem resistance study, Houseman et al. (2014) assessed the invasion of sericea lespedeza (Lespedeza cuneata) in temperate grasslands of North America by creating models that included propagule pressure, soil fertility, and disturbance in relation to risk of invasion. Not surprisingly, they found that invasibility decreased as the soil fertility was enhanced and disturbance lessened. In an experimental grassland in Europe, increasing native plant richness reduced the above-ground growth of field scabious (Knautia arvensis ), an invasive herbaceous perennial (Scherber et al. 2010). Compared to a species-poor community, the species rich one was more efficient in using resources and thus better able to resist invasion. Two more recently established invasive annuals, barbed goatgrass (Aegilops triuncialis) and medusahead (Elymus caput-medusae), in California grasslands, are effecting soil dynamics by reducing available nitrates in resident plant communities and lowering microbial biomass (Carey et al. 2017). Similar effects on soil N have been predicted to occur by increasing numbers of invasive symbiotic nitrogen-fixing lupines (Lupinus spp.) and birch (Alnus spp.) genera in the Boreal, subarctic, and upper montane temperate regions (Hiltbrunner et al. 2014).
In forest systems of the warm tropics to cold temperate regions, a large amount of research has documented many categories of ecosystem resistance. For example, eastern deciduous forests of New York and Connecticut, USA, were effective at resisting invasion by Norway maple (Acer platanoides) if there was an intact and closed canopy. Dispersal and growth of maple stands were hampered by the lack of sunlight penetrating through the canopy. False brome (Brachypodium sylvaticum), an invasive grass in temperate forests of the Pacific Northwest, USA, was more common with soil disturbance and forest community structure: conifer forests being more invaded than deciduous forests (Taylor and Cruzan 2015). In tropical and subtropical forests of East Africa and China, respectively, an invasive tree (Cinnamomum verum) and two invasive herbs, mile-a-minute (Mikania micrantha) and eupatorium (Eupatorium catariu) , were reported to effect soil microbes and fertility (Kueffer et al. 2007; Chen et al. 2017). The presence of soil mycorrhizal fungi was found to be more beneficial in resisting invasion of the two herbs in later succession forests, and nutrient poor soils were more conducive to invasion by C. verum.
Wetlands and riparian areas are very common systems in which ecosystem resistance to invasions has been tested, specifically the diversity of habitats and soil properties. In comparing invasive common reed (Phragmites australis) to native cordgrass (Spartina spp.) , Allen et al. (2018) found no direct impact from soil biota, interspecific competition, or lack of nutrient availability on the invader. Renöfält et al. (2005) found no relationship between richness and invasiveness with the introduction of common sunflower (Helianthus annuus) in the Vindel River of Sweden; only substrate was important in limiting the invader. In wetlands of Zurich, Switzerland, invasive goldenrod (Solidago gigantea) affected the soil microbes and nutrients to the detriment of the resident plant community.
Case Studies: Invader Fitness
The subcategories of plasticity, genetic mutation, phenological adaptation, and genetic selection compose the invader fitness factor in the IFF model (Young et al. 2017). These have been highlighted in the following case studies as plant hybridization, population genetics, and phenotypic plasticity.
Genome size, ploidy level, and mode of reproduction are topics that provide an assessment of the role of genetic diversity in plant invasions (Burns et al. 2011; Te Beest et al. 2012; Suda et al. 2015). Giant goldenrod (Solidago gigantea) occurs as a diploid, tetraploid, and hexaploid in its native range of North America, while in its introduced range (Europe), only tetraploid populations are known (Schlaepfer et al. 2010). These cytotypes exhibit differences in the habitats they occupy and segregate geographically. In a global study, Nagy et al. (2018) assessed the performance of hexaploids in the introduced range of giant goldenrod and found that compared with tetraploids, they did not present a greater invasion risk. In Australia, two separate studies were conducted on plants from the Cucurbitaceae and Boraginaceae families to assess the role of invasive plant genetic diversity in establishment (Shaik et al. 2016). In the first study, paddy melon (Cucumis myriocarpus) and camel melon (Citrullus lanatus) were found to consist of a single genotype, suggesting one introduction event for each species. Desert gourd (Citrullus colocynthis) , a related summer weed, was genetically diverse and thought to originate from multiple introductions. In the second study with two similar congeneric species (Shaik et al. 2016), Paterson’s curse (Echium plantagineum) was found to be genetically diverse and highly invasive, whereas the other, common viper’s bugloss (Echium vulgare) , exhibited less genetic diversity and occupied a more limited ecological niche.
In addition to the level of genetic diversity within introduced populations, as described above, phenotypic plasticity can contribute to establishment success and increase the likelihood of invasion. For example, the invasion of smooth cordgrass (Spartina alterniflora) across much of China has apparently occurred due to plasticity of plant phenotypes that have matched local abiotic conditions, instead of adaptive evolution (Liu et al. 2016). Tufted knotweed (Polygonum cespitosum) , a summer annual and recent invader in the Northeastern United States from Asia, has been found to exhibit high amounts of phenotypic plasticity, which would suggest an increased potential for invasiveness (Sultan and Matesanz 2015). The researchers detected higher fitness in all populations regardless of whether the habitat was dominated by shade, sun, wet, or dry conditions. Similarly, Lamarque et al. (2013) found that boxelder maple (Acer negundo) genotypes did not increase trait plasticity but displayed genetic differentiation in southern and Eastern Europe, where it is highly invasive.
Hybridization, which is a form of genetic selection, is a routine occurrence among plants and has implications in terms of fitness of an invader and their establishment. In one study conducted in the salt marshes of San Francisco, California, invasive cordgrass (S. alterniflora) has been shown to hybridize with California cordgrass (S. foliosa) , a native (Anttila et al. 1998). The invader produces greater amounts of pollen, which readily fertilizes the native. The resulting hybrids are more similar to the invader in trait characteristics and ability to adapt to changing conditions. In addition to habitat loss and decline in the native populations, the introgression through backcrossing has led to genetic pollution of the native.
While these case studies represent a large body of work, nonexistent are those that address all factors of the IFF simultaneously for any invasive plant (but see Young et al. 2017). Additional research, empirical and/or theoretical, is needed to comprehensively assess the effect of climate dynamics, ecosystem resistance, and invader fitness of invasive plants and those with the potential to become invasive. Such efforts will lead to a more comprehensive understanding of the invasion process and contribute to mitigating their negative effects through improved management.
2.4 Application of the IFF
We conclude this chapter with a discussion regarding how the three factors of the IFF model (climate dynamics, invader fitness, and ecosystem resistance), singly and synergistically, can be integrated into management decision-making. The IFF model provides a way to help determine what contributes to the establishment of non-native species and their range expansion during invasion. Such an assessment provides not only a better understanding of the invasion of native plant communities but also a useful way to help improve management practices. The generalized invasion curve can be used to depict introduction, establishment, and expansion for invasive plants, but this curve can also be used to show how management approaches must adapt to changes in the population size and distribution of invasive plants (Fig. 2.1).
Pre-introduction
The most ecologically and cost-effective management approach for curtailing plant invasions is preventing their introduction in the first place. This can only happen prior to the inflection point – the instant a propagule drops to the ground, attaches to a tree, or slips away into the water. Barriers, whether anthropogenic or natural, are the most sure way to curtail the invasion process. While border check stations and points of entry can halt human transport of invasive plant propagules, a lack of the ability to enforce laws to prohibit other routes (e.g., ocean vessels) and markets (e.g., internet) makes for a very porous system (Pyšek et al. 2020). The prevention of the introduction of invasive plants is ideal but in practice is nearly impossible to achieve.
Post-introduction
Early detection rapid response (EDRR) is an approach for controlling non-native plants. The EDRR approach has been defined by the US Department of the Interior as “…a coordinated set of actions to find and eradicate potential invasive plants before they spread and cause harm…” (DOI 2016). Alternatively, Reaser et al. (2020) define EDRR as a “…a guiding principle for minimizing the impact of invasive plants in an expedited yet effective and cost efficient manner, where ‘detection’ is the process of observing and documenting an invasive plants, and ‘response’ is the process of reacting to the detection once the organism has been authoritatively identified and response options have been assessed.” The use of EDRR, while proposed by many (e.g., Maxwell et al. 2009; Pyšek and Richardson 2010; Littell et al. 2012; Antunes and Schamp 2017), has not been shown to be entirely effective. Largely, the lack of success is knowing exactly how to implement “detection” and what the term “early” really means. Similarly, responses can vary and range from complete eradication of plants and propagules to partial or limited control of the target plant (Reaser et al. 2020). An EDRR approach has little need of understanding invasive plants beyond recognition and application of a removal technique.
Even before a non-native species has been introduced and EDRR applied, risk assessments of plant species can help determine potential negative impacts (e.g., costs) that occur during all phases of the generalized invasion curve (Meyers et al. 2020). Effectively assessing non-native plants for their risk is largely inadequate due to lack of data, transparency and repeatability, and uncertainty (Kumar and Singh 2020). However, risk assessments continue to be used by federal agencies when non-native species are initially detected, the status of a non-native species changes, and a potential pathway of introduction has been identified (Meyers et al. 2020). In California, USA, and likely other arid regions, the public is encouraged to use drought-tolerant plants in order to reduce water use. The result is an abundance of plants with drought-tolerant characteristics, which over time have an increasing likelihood of becoming invasive (Pemberton and Liu 2009).
Establishment and Range Expansion
During the establishment phase of an invasion, as population size and/or population number increases, eradication may still be possible. But, as populations increase in size and expand their distribution, other approaches, such as containment and “asset-based protection,” are required (the latter focuses on the protection of farmlands, industries, recreational areas, and natural ecosystems). Each of the three factor of the IFF must be overcome for a non-native species to expand its range. Thus, it is critically important to better understanding of how these factors can influence non-native establishment and range expansion (e.g., invasion process) to predict which plant communities are likely to be invaded in the future. This means that management strategies should rapidly shift from eradication, to containment, to asset-based protection, as the pace of an invasion quickens.
Employing the IFF
Any one of the three factors of the IFF model can terminate the potential that an invasion will occur, so all of them should be taken into account by researchers or managers. Using the IFF model, a qualitative and quantitative snapshot about a potential or current plant invasion can be generated based on published research (Young et al. 2017). The IFF model should be considered a tool to establish research priorities and identify components in the invasion process that can be used to facilitate eradication, containment, or asset-based protection. Several recent papers have identified a “knowledge gap” or disconnect between research and practice in the management of invasive plants (Funk et al. 2020; Pyšek et al. 2020; Young and Kettenring 2020).
Additionally, the IFF model can be used to inform the removal of invasive species and the restoration of native communities. In the restoration of a site dominated by invasive plants, the first step (i.e., site preparation phase) frequently involves the physical and/or chemical removal of the invaders (Rowe 2010). Immediately afterwards, native plants are seeded or planted to fill vacant niches and prevent subsequent reinvasion by the same or other invasive plants (Masters and Sheley 2001; Hulme 2006). The overall goal of these efforts is to assemble an invasion-resistant community, so practitioners involved in ecological restoration should establish native plant species with a diversity of phenotypic traits, including those that have unique phenology, growth rate, and stress-tolerant abilities (Kimball et al. 2016). For problematic invasive plants, which have, by definition, overcome all three factors of the IFF model, restoration efforts should include plant species that have specific ecological niches to establish native communities with different ecosystem resistance characteristics, compared to the community that was initially invaded. In this manner, it may be possible to reestablish the ecosystem resistance factor (dotted line in Fig. 2.1).
When speed of invasion, pathways of distribution, and evolutionary changes of invasive plants are not well known, information from an IFF model can be used to improve the effectiveness of management programs and tools. Funk et al. (2020) listed soil seedbank dynamics, life history traits, and the effects of ongoing climate change as important plant invasion ecology knowledge gaps identified by land managers that researchers should fill. The three factors of the IFF model address each of these gaps: ecosystem resistance (additions to and loss of seeds in soil seedbanks), invader fitness (adaptations linked to life history trait evolution), and climate dynamics (the range of climates, including extreme weather events, that an invasive species can tolerate). Young et al. (2017) used musk thistle (Carduus nutans) as an example of how the IFF could be applied to help focus research to answer basic questions and address management challenges. Musk thistle, a problematic invasive plant around the globe (Shea and Kelly 2004), “failed” to become established in a native perennial grassland of the Central United States (Young 2015). Using the IFF model, empirical studies conducted on musk thistle (see Young et al. 2017) provided evidence to suggest that musk thistle did not exhibit phenotypic plasticity (invader fitness) in these perennial grasslands during extreme drought (climate dynamics), leading to the inability of the plant to become established in this region.
2.5 Conclusions
By conducting research using the IFF model, a better understanding of how climate dynamics, ecosystem resistance, and invader fitness influence the eventual fate of non-native plants can be obtained. This knowledge can be used to improve decision-making by land managers to help mitigate the negative consequences of invasive plants, especially the invasion of native plant communities.
References
Adler PB, Fajardo A, Kleinhesselink AR et al (2013) Trait-based tests of coexistence mechanisms. Ecol Lett 16:1294–1306. https://doi.org/10.1111/ele.12157
Alexander JM, Levine JM (2019) Earlier phenology of a nonnative plant increases impacts on native competitors. Proc Natl Acad Sci U S A 116:6199–6204. https://doi.org/10.1073/pnas.1820569116
Allen WJ, Meyerson LA, Flick AJ et al (2018) Intraspecific variation in indirect plant–soil feedbacks influences a wetland plant invasion. Ecology 99:1430–1440
Allendorf FW, Lundquist LL (2003) Introduction: population biology, evolution, and control of invasive species. Conserv Biol 17:24–30. https://doi.org/10.1046/j.1523-1739.2003.02365.x
Angelo CL, Daehler CC (2013) Upward expansion of fire-adapted grasses along a warming tropical elevation gradient. Ecography 36:551–559
Anttila CK, Daehler CC, Rank NE et al (1998) Greater male fitness of a rare invader (Spartina alterniflora, Poaceae) threatens a common native (Spartina foliosa) with hybridization. Am J Bot 85:1597–1601
Antunes PM, Schamp B (2017) Constructing standard invasion curves from herbarium data - toward increased predictability of plant invasions. Inv Plant Sci Manag 10:293–303
Aslani F, Juraimi AS, Ahmad-Hamdani MS et al (2019) The role of arbuscular mycorrhizal fungi in plant invasion trajectory. Plant Soil 441:1–14
Baruch Z, Goldstein G (1999) Leaf construction cost, nutrient concentration, and net CO2 assimilation of native and invasive species in Hawaii. Oecologia 121:183–192
Blackburn TM, Lockwood JL, Cassey P (2015) The influence of numbers on invasion success. Mol Ecol 24:1942–1953
Blair AC, Wolfe LM (2004) The evolution of an invasive plant: an experimental study with Silene latifolia. Ecology 85:3035–3042
Blossey B, Notzold R (1995) Evolution of increased competitive ability in invasive nonindigenous plants: a hypothesis. J Ecol 83:887–889. https://doi.org/10.2307/2261425
Blumenthal DM (2005) Interrelated causes of plant invasion. Science 310:243–244. https://doi.org/10.1126/science.1114851
Blumenthal DM (2006) Interactions between resource availability and enemy release in plant invasion. Ecol Lett 9:887–895. https://doi.org/10.1111/j.1461-0248.2006.00934.x
Bossdorf O, Auge H, Lafuma L et al (2005) Phenotypic and genetic differentiation between native and introduced plant populations. Oecologia 144:1–11
Bossdorf O, Lipowsky A, Prati D (2008) Selection of preadapted populations allowed Senecio inaequidens to invade Central Europe. Div Dist 14:676–685
Broennimann O, Treier UA, Muller-Scharer H et al (2007) Evidence of climatic niche shift during biological invasion. Ecol Lett 10:701–709
Brooks ML (2003) Effects of increased soil nitrogen on the dominance of alien annual plants in the Mojave Desert. J Appl Ecol 40:344–353. https://doi.org/10.1046/j.1365-2664.2003.00789.x
Burns JH, Ashman TL, Steets JA et al (2011) A phylogenetically controlled analysis of the roles of reproductive traits in plant invasions. Oecologia 166:1009–1017
Cano L, Escarre J, Fleck I et al (2008) Increased fitness and plasticity of an invasive species in its introduced range: a study using Senecio pterophorus. J Ecol 96:468–476
Carey CJ, Blankinship JC, Eviner VT et al (2017) Invasive plants decrease microbial capacity to nitrify and denitrify compared to native California grassland communities. Biol Invasions 19:2941–2957
Catford JA, Smith AL, Wragg PD et al (2019) Traits linked with species invasiveness and community invasibility vary with time, stage and indicator of invasion in a long-term grassland experiment. Ecol Lett 22:593–604
Cavaleri MA, Sack L (2010) Comparative water use of native and invasive plants at multiple scales: a global meta-analysis. Ecology 91:2705–2715. https://doi.org/10.1890/09-0582.1
Chen BM, Li S, Liao HX et al (2017) Do forest soil microbes have the potential to resist plant invasion? A case study in Dinghushan biosphere reserve (South China). Acta Oecol 81:1–9
Chesson P (2000) Mechanisms of maintenance of species diversity. Annu Rev Ecol Syst 31:343–366. https://doi.org/10.1146/annurev.ecolsys.31.1.343
Colautti RI, Lau JA (2015) Contemporary evolution during invasion: evidence for differentiation, natural selection, and local adaptation. Mol Ecol 24:1999–2017
Colautti RI, MacIsaac HJ (2004) A neutral terminology to define “invasive” species. Div Dist 10:135–141
Colautti RI, Ricciardi A, Grigorovich IA et al (2004) Is invasion success explained by the enemy release hypothesis? Ecol Lett 7:721–733. https://doi.org/10.1111/j.1461-0248.2004.00616.x
Colautti RI, Grigorovich IA, MacIsaac HJ (2006) Propagule pressure: a null model for biological invasions. Biol Invasions 8:1023–1037
Corliss CT, Sultan SE (2016) Evolutionary potential for increased invasiveness: high-performance Polygonum cespitosum genotypes are competitively superior in full sun. Am J Bot 103:348–354. https://doi.org/10.3732/ajb.1500306
Cox GW (2004) Alien species and evolution. Island Press, Washington, DC
Crosby AW (2003) The Columbian exchange: biological and cultural consequences of 1492, 30th anniversary edition. Praeger Publishers, Westport
D’Antonio CM, Vitousek PM (1992) Biological invasions by exotic grasses, the grass/fire cycle, and global change. Annu Rev Ecol Evol Syst 13:63–87
Daehler CC (2003) Performance comparisons of co-occurring native and alien invasive plants: implications for restoration and conservation. Annu Rev Ecol Evol Syst 34:183–211. https://doi.org/10.1146/annurev.ecolsys.34.011802.132403
Davidson AM, Jennions M, Nicotra AB (2011) Do invasive species show higher phenotypic plasticity than native species and, if so, is it adaptive? A meta-analysis. Ecol Lett 14:419–431
Davies KF, Chesson P, Harrison S et al (2005) Spatial heterogeneity explains the scale dependence of the native-exotic diversity relationship. Ecology 86:1602–1610. https://doi.org/10.1890/04-1196
Davis MA, Grime JP, Thompson K (2000) Fluctuating resources in plant communities: a general theory of invasibility. J Ecol 88:528–534
Diaz S, Cabido M (2001) Vive la difference: plant functional diversity matters to ecosystem processes. Tren Ecol Evol 16:646–655
Didham RK, Tylianakis JM, Gemmel NJ et al (2007) Interactive effects of habitat modification and species invasion on native species decline. Trends Ecol Evol 22:489–496
Dietz H, Edwards PJ (2006) Recognition that causal processes change during plant invasion helps explain conflicts in evidence. Ecology 87:1359–1367
Dlugosch KM, Parker IM (2008) Invading populations of an ornamental shrub shows rapid life history evolution despite genetic bottlenecks. Ecol Lett 11:701–709. https://doi.org/10.1111/j.1461-0248.2008.01181.x
Dlugosch KM, Anderson SR, Braasch J et al (2015) The devil is in the details: genetic variation in introduced populations and its contributions to invasion. Mol Ecol 24:2095–2111. https://doi.org/10.1111/mec.13183
DOI (2016) Safeguarding America’s lands and waters from invasive species: a national framework for early detection and rapid response. US Department of the Interior, Washington, DC
Ehrenfeld JG (2003) Effects of exotic plant invasions on soil nutrient cycling processes. Ecosystems 6:503–523. https://doi.org/10.1007/s10021-002-0151-3
Ellstrand NC, Schierenbeck KA (2006) Hybridization as a stimulus for the evolution of invasiveness in plants? Euphytica 148:35–46. https://doi.org/10.1007/s10681-006-5939-3
Elton CS (1958) The ecology of invasions by animals and plants. Metheun, London
Enders M, Havemann F, Ruland F et al (2020) A conceptual map of invasion biology: integrating hypotheses into a consensus network. Glob Ecol Biogeogr 29:978–991. https://doi.org/10.1111/geb.13082
Eppinga MB, Rietkerk M, Dekker SC (2006) Accumulation of local pathogens: a new hypothesis to explain exotic plant invasions. Oikos 114:168–176
Estoup A, Ravigne V, Hufbauer R et al (2016) Is there a genetic paradox of biological invasions? Annu Rev Ecol Evol Syst 47:51–72
Facon B, Genton J, Shykoff J et al (2006) A general eco-evolutionary framework for understanding bioinvasions. Trends Ecol Evol 21:130–135
Fargione JE, Tilman D (2005) Diversity decreases invasion via both sampling and complementarity effects. Ecol Lett 8:604–611
Feng YL, Fu GL, Zheng YL (2008) Specific leaf area relates to the differences in leaf construction cost, photosynthesis, nitrogen allocation, and use efficiencies between invasive and noninvasive alien congeners. Planta 228:383–390. https://doi.org/10.1007/s00425-008-0732-2
Feng YH, Fouqueray TD, van Kleunen M (2019) Linking Darwin's naturalisation hypothesis and Elton's diversity-invasibility hypothesis in experimental grassland communities. J Ecol 107:794–805. https://doi.org/10.1111/1365-2745.13061
Franks SJ, Sim S, Weis AE (2007) Rapid evolution of flowering time by an annual plant in response to a climate fluctuation. Proc Natl Acad Sci U S A 104:1278–1282. https://doi.org/10.1073/pnas.0608379104
Fukami T (2015) Historical contingency in community assembly: integrating niches, species pools, and priority effects. Annu Rev Ecol Evol Syst 46:1–23. https://doi.org/10.1146/annurev-ecolsys-110411-160340
Funk JL, Nguyen MA, Standish RJ et al (2017) Global resource acquisition patterns of invasive and native plant species do not hold at the regional scale in Mediterranean type ecosystems. Biol Invasions 19:1143–1151. https://doi.org/10.1007/s10530-016-1297-9
Funk JL, Parker IM, Matzek V et al (2020) Keys to enhancing the value of invasion ecology research for management. Biol Invasions 22:2431–2445. https://doi.org/10.1007/s10530-020-02267-9
Futuyma DJ, Edwards SV, John R (2005) Evolution. Sinauer Associates, Sunderland
Gaertner M, Fisher JL, Sharma GP et al (2012) Insights into invasion and restoration ecology: time to collaborate towards a holistic approach to tackle biological invasions. NeoBiota 12:57–75. https://doi.org/10.3897/neobiota.12.2123
Garcia-Duro J, Cruz O, Casal M et al (2019) Fire as driver of the expansion of Paraserianthes lophantha (Willd.) I. C. Nielsen in SW Europe. Biol Invasions 21:1427–1438. https://doi.org/10.1007/s10530-018-01910-w
Garnier E, Cortez J, Billes G et al (2004) Plant functional markers capture ecosystem properties during secondary succession. Ecology 85:2630–2637
Godoy O, Saldana A, Fuentes N et al (2011) Forests are not immune to plant invasions: phenotypic plasticity and local adaptation allow Prunella vulgaris to colonize a temperate evergreen rainforest. Biol Invasions 13:1615–1625. https://doi.org/10.1007/s10530-010-9919-0
González-Muñoz N, Linares JC, Castro-Díez P et al (2014) Predicting climate change impacts on native and invasive tree species using radial growth and twenty-first century climate scenarios. Eur J For Res 133:1073–1086
Grotkopp E, Rejmanek M, Rost TL (2002) Toward a causal explanation of plant invasiveness: seedling growth and life-history strategies of 29 pine (Pinus) species. Am Nat 159:396–419. https://doi.org/10.1086/338995
Guido A, Vélez-Martin E, Overbeck GE et al (2016) Landscape structure and climate affect plant invasion in subtropical grasslands. Appl Veg Sci 19:600–610
Gurevitch J, Fox GA, Wardle GM et al (2011) Emergent insights from the synthesis of conceptual frameworks for biological invasions. Ecol Lett 14:407–418
Hellmann JJ, Byers JE, Bierwagen BG et al (2008) Five potential consequences of climate change for invasive species. Conserv Biol 22:534–543. https://doi.org/10.1111/j.1523-1739.2008.00951.x
Henneman ML, Memmott J (2001) Infiltration of a Hawaiian community by introduced biological control agents. Science 293:1314–1316
Hess MC, Mesleard F, Buisson E (2019) Priority effects: emerging principles for invasive plant species management. Ecol Eng 127:48–57. https://doi.org/10.1016/j.ecoleng.2018.11.011
Hierro JL, Callaway RM (2003) Allelopathy and exotic plant invasion. Plant Soil 256:29–39. https://doi.org/10.1023/a:1026208327014
Hierro JL, Maron JL, Callaway RM (2005) A biogeographical approach to plant invasions: the importance of studying exotics in their introduced and native range. J Ecol 93:5–15
Hiltbrunner E, Aerts R, Bühlmann T et al (2014) Ecological consequences of the expansion of N2-fixing plants in cold biomes. Oecologia 176:11–24
Houseman GR, Foster BL, Brassil CE (2014) Propagule pressure-invasibility relationships: testing the influence of soil fertility and disturbance with lespedeza cuneate. Oecologia 174:511–520
Hufbauer RA, Facon B, Ravigne V et al (2011) Anthropogenically induced adaptation to invade (AIAI): contemporary adaptation to human-altered habitats within the native range can promote invasions. Evol Appl 5:89–101. https://doi.org/10.1111/j.1752-4571.2011.00211.x
Hulme PE (2006) Beyond control: wider implications for the management of biological invasions. Appl Ecol 43:835–847. https://doi.org/10.1111/j.1365-2664.2006.01227.x
Jarnevich CS, Young NE, Talbert M et al (2018) Forecasting an invasive species' distribution with global distribution data, local data, and physiological information. Ecosphere 9:12. https://doi.org/10.1002/ecs2.2279
Jauni M, Gripenberg S, Ramula S (2015) Non-native plant species benefit from disturbance: a meta-analysis. Oikos 124:122–129. https://doi.org/10.1111/oik.01416
Jenkins C, Keller SR (2011) A phylogenetic comparative study of preadaptation for invasiveness in the genus Silene (Caryophyllaceae). Biol Invasions 13:1471–1486
Jenkins CN, Pimm SL (2003) How big is the global weed patch? Ann Missouri Bot Gard 90:172–178
Jeschke JM (2014) General hypotheses in invasion ecology. Divers Dist 20:1229–1234
Jeschke J, Aparicio LG, Haider S et al (2012) Support for major hypotheses in invasion biology is uneven and declining. NeoBiota 14:1–20. https://doi.org/10.3897/neobiota.14.3435
Jones EJ, Kraaij T, Fritz H et al (2019) A global assessment of terrestrial alien ferns (Polypodiophyta): Species' traits as drivers of naturalisation and invasion. Biol Invasions 21:861–873. https://doi.org/10.1007/s10530-018-1866-1
Keddy PA (1992) Assembly and response rules: two goals for predictive community ecology. J Veg Sci 3:157–164
Keller SR, Taylor DR (2008) History, chance, and adaptation during biological invasion: separating stochastic phenotypic evolution from response to selection. Ecol Lett 11:852–866
Kennedy TA, Naeem S, Howe KM et al (2002) Biodiversity as a barrier to ecological invasion. Nature 417:636–638. https://doi.org/10.1038/nature00776
Kimball S, Gremer JR, Huxman TE et al (2013) Phenotypic selection favors missing trait combinations in coexisting annual plants. Am Nat 182:191–207. https://doi.org/10.1086/671058
Kimball S, Goulden ML, Suding KN et al (2014a) Altered water and nitrogen input shifts succession in a southern California coastal sage community. Ecol Appl 24:1390–1404. https://doi.org/10.1890/13-1313.1
Kimball S, Gremer JR, Barron-Gafford GA et al (2014b) High water-use efficiency and growth contribute to success of non-native Erodium cicutarium in a Sonoran Desert winter annual community. Conserv Phys 2(1):cou006. https://doi.org/10.1093/conphys/cou006
Kimball S, Funk JL, Sandquist DR et al (2016) Ecophysiological considerations for restoration. In: Palmer MA, Falk DA, Zedler JB (eds) Foundations of restoration ecology II. Island Press, Washington DC, pp 153–181
Kolar CS, Lodge DM (2001) Progress in invasion biology: predicting invaders. Trends Ecol Evol 16:199–204
Kolbe JJ, Larson A, Losos JB (2007) Differential admixture shapes morphological variation among invasive populations of the lizard Anolis sagrei. Mol Ecol 16:1579–1591
Kueffer C, Schumacher E, Fleischmann K et al (2007) Strong below-ground competition shapes tree regeneration in invasive Cinnamomum verum forests. J Ecol 95:273–282
Kumar RP, Singh JS (2020) Invasive alien plant species: their impact on environment, ecosystem services and human health. Ecol Indic 111:106020. https://doi.org/10.1016/j.ecolind.2019.106020
Lachmuth S, Durka W, Schurr FM (2010) The making of a rapid plant invader: genetic diversity and differentiation in the native and invaded range of Senecio inaequidens. Mol Ecol 19:3952–3967
Lamarque LJ, Porté AJ, Eymeric C (2013) A test for pre-adapted phenotypic plasticity in the invasive tree Acer negundo L. PLoS One 8(9):e74239
Lau JA, Schulties EH (2015) When two invasion hypotheses are better than one. New Phytol 205:958–960
Lavergne S, Molofsky J (2007) Increased genetic variation and evolutionary potential drive the success of an invasion. Proc Natl Acad Sci U S A 104:3883–3888. https://doi.org/10.1073/pnas.0607324104
Lee CE (2002) Evolutionary genetics of invasive species. Trends Ecol Evol 17:386–391
Leffler AJ, James JJ, Monaco TA et al (2014) A new perspective on trait differences between native and invasive exotic plants. Ecology 95:298–305
Leishman MR, Haslehurst T, Ares A et al (2007) Leaf trait relationships of native and invasive plants: community- and global-scale comparisons. New Phytol 176:635–643. https://doi.org/10.1111/j.1469-8137.2007.02189.x
Levine JM, Adler PB, Yelenik SG (2004) A meta-analysis of biotic resistance to exotic plant invasions. Ecol Lett 7:975–989
Littell JS, Peterson DL, Millar CI et al (2012) US National Forests adapt to climate change through science-management partnerships. Clim Chang 110:269–296
Liu W, Maung-Douglass K, Strong DR et al (2016) Geographical variation in vegetative growth and sexual reproduction of the invasive Spartina alterniflora in China. J Ecol 104:173–181
Lockwood JL, Cassey P, Blackburn T (2005) The role of propagule pressure in explaining species invasions. Trends Ecol Evol 20:223–228. https://doi.org/10.1016/j.tree.2005.02.004
MacDougall AS, Gilbert B, Levine JM (2009) Plant invasions and the niche. J Ecol 97:609–615
Mack RN, Erneberg M (2002) The United States naturalized flora: largely the product of deliberate introductions. Ann Missouri Bot Gard 89:176–189
Mack RN, Lonsdale WM (2001) Humans as global plant dispersers: getting more than we bargained for. Bioscience 51:95–102
Mack RN, Simberloff D, Lonsdale WM et al (2000) Biotic invasions: causes, epidemiology, global consequences, and control. Ecol Appl 10:689–710. https://doi.org/10.1890/1051-0761(2000)010[0689:bicegc]2.0.co;2
Mangla S, Inderjit CRM (2008) Exotic invasive plant accumulates native soil pathogens which inhibit native plants. J Ecol 96:58–67. https://doi.org/10.1111/j.1365-2745.2007.01312.x
Masters RA, Sheley RL (2001) Principles and practices for managing rangeland invasive plants. J. Range Manag 54:502–517
Maron JL, Vila M, Bommarco R et al (2004) Rapid evolution of an invasive plant. Ecol Monogr 74:261–280
Mason NH, de Bello F (2013) Functional diversity: a tool for answering challenging ecological questions. J Veg Sci 24:777–780
Maxwell BD, Lehnhoff E, Rew LJ (2009) The rationale for monitoring invasive plant populations as a crucial step for management. Invasive Plant Sci Manage 2:1–9
Meyers N, Reaser JK, Hoff MH (2020) Instituting a national early detection and rapid response program: needs for building federal risk screening capacity. Biol Invasions 22:53–65. https://doi.org/10.1007/s10530-019-02144-0
Molina-Montenegro MA, Acuña-Rodríguez IS, Flores TM et al (2018) Is the success of plant invasions the result of rapid adaptive evolution in seed traits? Evidence from a latitudinal rainfall gradient. Front Plant Sci 9:208
Mooney H, Hobbs R (2000) Invasive species in a changing world. Island Press, Washington, DC
Nagy DU, Stranczinger S, Godi A et al (2018) Does higher ploidy level increase the risk of invasion? A case study with two geo-cytotypes of Solidago gigantea Aiton (Asteraceae). J Plant Ecol 11:317–327
Nguyen MA, Ortega AE, Nguyen KQ et al (2016) Evolutionary responses of invasive grass species to variation in precipitation and soil nitrogen. J Ecol 104:979–986. https://doi.org/10.1111/1365-2745.12582
Novak SJ (2007) The role of evolution in the invasion process. Proc Natl Acad Sci USA 104:3671–3672
Novak SJ (2011) Geographic origins and introduction dynamics. In: Simberloff D, Rejmanek M (eds) Encyclopedia of biological invasions. University of California Press, Berkeley, pp 273–280
Novak SJ, Mack RN (1993) Genetic variation in Bromus tectorum (Poaceae): comparison between native and introduced populations. Heredity 71:167–176. https://doi.org/10.1038/hdy.1993.121
Novak SJ, Mack RN (1995) Allozyme diversity in the apomictic vine Bryonia alba (Cucurbitaceae): potential consequences of multiple introductions. Am J Bot 82:1153–1162
Novak SJ, Mack RN (2001) Tracing plant introduction and spread: genetic evidence from Bromus tectorum (Cheatgrass). Bioscience 51:114–122. https://doi.org/10.1641/0006-3568(2001)051[0114:tpiasg]2.0.co;2
Novak SJ, Mack RN (2005) Genetic bottlenecks in alien species: influence of mating systems and introduction dynamics. In: Sax DF, Stachowicz JJ, Gaines SD (eds) Species invasions: insights into ecology, evolution and biogeography. Sinauer, Sunderland, pp 210–228
Packer JG, Meyerson LA, Skálová H et al (2017) Biological flora of the British Isles: Phragmites australis. J Ecol 105:1123–1162
Pemberton RW, Liu H (2009) Marketing time predicts naturalization of horticultural plants. Ecology 90:69–80. https://doi.org/10.1890/07-1516.1
Perkins LB, Nowak RS (2013) Invasion syndromes: hypotheses on relationships among invasive species attributes and characteristics of invaded sites. J Arid Land 5:275–283. https://doi.org/10.1007/s40333-013-0161-3
Pimentel D, Zuninga R, Morrison D (2005) Update on the environmental and economic costs associated of with alien-invasive species in the United States. Ecol Econ 52:273–288
Prentis PJ, Wilson JR, Dormontt EE et al (2008) Adaptive evolution in invasive species. Trends Plant Sci 13:288–294
Pringle A, Bever JD, Gardes M et al (2009) Mycorrhizal symbioses and plant invasions. Ann Rev Ecol Evo Syst 40:699–715
Pyšek P, Richardson DM (2007) Traits associated with invasiveness in alien plants: where do we stand? Biol Invasions 9:97–126
Pyšek P, Richardson DM (2010) Invasive species, environmental change and management, and health. Ann Rev Environ Res 35:25–55
Pyšek P, Richardson DM, Williamson M (2004) Predicting and explaining plant invasions through analysis of source area floras: some critical considerations. Div Dist 10:179–187
Pyšek P, Jarosik V, Pergl J et al (2009) The global invasion success of central European plants is related to distribution characteristics in their native range and species traits. Div Dist 15:891–903
Pyšek P, Hulme PE, Simberloff D et al (2020) Scientists' warning on invasive alien species. Biol Rev 95:1511–1534. https://doi.org/10.1111/brv.12627
Qian H, Ricklefs RE (2006) The role of exotic species in homogenizing the North American flora. Ecol Lett 9:1293–1298. https://doi.org/10.1111/j.1461-0248.2006.00982.x
Reaser JK, Burgiel SW, Kirkey J et al (2020) The early detection of and rapid response (EDRR) to invasive species: a conceptual framework and federal capacities assessment. Biol Invasions 22:1–19
Reichard SH, White P (2001) Horticulture as a pathway of invasive plant introductions in the United States. Bioscience 51:148–153
Rejmanek M, Richardson DM, Higgins SI et al (2005) Ecology of invasive plants: state of the art. In: Mooney HA, Mack RN, McNeely JA, Neville LE, Schei PJ, Waage JK (eds) Invasive alien species, a new synthesis. Island Press, Washington, DC, pp 104–161
Renöfält BM, Jansson R, Nilsson C (2005) Spatial patterns of plant invasiveness in a riparian corridor. Landsc Ecol 20:165–176
Ricciardi A, Jones LA, Kestrup AM et al (2011) Expanding the propagule pressure concept to understand the impact of biological invasions. In: Richardson DM (ed) Fifty years of invasion ecology: the legacy of Charles Elton, Oxford, pp 225–236
Richards CL, Bossdorf O, Muth NZ et al (2006) Jack of all trades, master of some? On the role of phenotypic plasticity in plant invasions. Ecol Lett 9:981–993. https://doi.org/10.1111/j.1461-0248.2006.00950.x
Richardson DM, Pyšek P (2006) Plant invasions: merging the concepts of species invasiveness and community invisibility. Prog Phys Geogr 30:409–431
Richardson D, Pyšek P (2007) Elton CS 1958: the ecology of invasions by animals and plants. Prog Phys Geogr 31:659–666
Richardson DM, Pyšek P, Rejmanek M et al (2000) Naturalization and invasion of alien plants: concepts and definitions. Biodiv Res 6:93–107
Rowe HI (2010) Tricks of the trade: techniques and opinions from 38 experts in tallgrass prairie restoration. Restor Ecol 18:253–262. https://doi.org/10.1111/j.1526-100X.2010.00663.x
Saarinen K, Lindstrom L, Ketola T (2019) Invasion triple trouble: environmental fluctuations, fluctuation-adapted invaders and fluctuation-mal-adapted communities all govern invasion success BMC. Evol Biol 19(1):42. https://doi.org/10.1186/s12862-019-1348-9
Sakai AK, Allendorf FW, Holt JS et al (2001) The population biology of invasive species. Annu Rev Ecol Evol Syst 32:305–332
Sala OE, Chapin FS, Armesto JJ et al (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–1774
Sax DF, Stachowicz JJ, Gaines SD (2005) Species invasions, insights into ecology, evolution, and biogeography. Sinauer, Sunderland
Scherber C, Mwangi PN, Schmitz M et al (2010) Biodiversity and belowground interactions mediate community invasion resistance against a tall herb invader. J Plant Ecol 3:99–108
Schlaepfer DR, Edwards PJ, Billeter R (2010) Why only tetraploid Solidago gigantea (Asteraceae) became invasive: a common garden comparison of ploidy levels. Oecologia 163:661–673. https://doi.org/10.1007/s00442-010-1595-3
Schrieber K, Lachmuth S (2017) The genetic paradox of invasions revisited: the potential role of inbreeding x environment interactions in invasion success. Biol Rev 92:939–952. https://doi.org/10.1111/brv.12263
Shaik RS, Zhu X, Clements DR et al (2016) Understanding invasion history and predicting invasive niches using genetic sequencing technology in Australia: case studies from Cucurbitaceae and Boraginaceae. Conserv Physiol 4(1):cow030. https://doi.org/10.1093/conphys/cow030
Shea K, Chesson P (2002) Community ecology theory as a framework for biological invasions. Trends Ecol Evol 17:170–176
Shea K, Kelly D (2004) Modeling for management of invasive species: musk thistle (Carduus nutans) in New Zealand. Weed Tech 18:1338–1341
Sheppard CS, Burns BR, Stanley MC (2016) Future-proofing weed management for the effects of climate change: is New Zealand underestimating the risk of increased plant invasions? N Z J Ecol 40:398–405
Simberloff D (2009) The role of propagule pressure in biological invasions. Annu Rev Ecol Evol Syst 40:81–102
Stinson KA, Campbell SA, Powell JR et al (2006) Invasive plant suppresses the growth of native tree seedlings by disrupting belowground mutualisms. PLoS Biol 4:727–731. https://doi.org/10.1371/journal.pbio.0040140
Stockwell CA, Hendry AP, Kinnison MT (2003) Contemporary evolution meets conservation biology. Trends Ecol Evol 18:94–101. https://doi.org/10.1016/s0169-5347(02)00044-7
Suda J, Meyerson LA, Leitch IJ et al (2015) The hidden side of plant invasions: the role of genome size. New Phytol 205:994–1007
Sultan SE, Matesanz S (2015) An ideal weed: plasticity and invasiveness in Polygonum cespitosum. Ann N Y Acad Sci 1360:101–119
Taylor LV, Cruzan MB (2015) Propagule pressure and disturbance drive the invasion of perennial false-brome (Brachypodium sylvaticum). Invasive Plant Sci Manage 8:169–180
Taylor DR, Keller SR (2007) Historical range expansion determines the phylogenetic diversity introduced during contemporary invasion. Evolution 61:334–345
Te Beest M, Le Roux JJ, Richardson DM et al (2012) The more the better? The role of polyploidy in facilitating plant invasions. Ann Bot 109:19–45
Thebaud C, Simberloff D (2001) Are plants really larger in their introduced ranges? Am Nat 157:231–236. https://doi.org/10.1086/318635
Theoharides KA, Dukes JS (2007) Plant invasion across space and time: factors affecting nonindigenous species success during four stages of invasion. New Phytol 176:256–273. https://doi.org/10.1111/j.1469-8137.2007.02207.x
Thuiller W, Gallien L, Boulangeat I et al (2010) Resolving Darwin's naturalization conundrum: a quest for evidence. Div Dist 16:461–475. https://doi.org/10.1111/j.1472-4642.2010.00645.x
Valliere JM (2019) Tradeoffs between growth rate and water-use efficiency in seedlings of native perennials but not invasive annuals. Plant Ecol 220:361–369. https://doi.org/10.1007/s11258-019-00919-y
van Boheemen LA, Lombaert E, Nurkowski KA et al (2017) Multiple introductions, admixtures, and bridgehead invasion characterize the introduction history of Ambrosia artemisiifolia in Europe and Australia. Mol Ecol 26:5421–5434
van Kleunen M, Weber E, Fischer M (2010) A meta-analysis of trait differences between invasive and non-invasive plant species. Ecol Lett 13:235–245
van Kleunen M, Schlaepfer DR, Glaetti M et al (2011) Preadapted for invasiveness: do species traits or their plastic response to shading differ between invasive and non-invasive plant species in their native range? J Biogeogr 38:1294–1304. https://doi.org/10.1111/j.1365-2699.2011.02495.x
van Kleunen M, Essl F, Pergi J et al (2018) The changing role of ornamental horticulture in alien plant invasions. Biol Rev 93:1421–1437
Vitousek PM, D’Antonio CM, Loop LL et al (1996) Biological invasions as global environmental change. Am Sci 84:468–478
Walder M, Armstrong JE, Borowicz VA (2019) Limiting similarity, biotic resistance, nutrient supply, or enemies? What accounts for the invasion success of an exotic legume? Biol Invasions 21:435–449. https://doi.org/10.1007/s10530-018-1835-8
Wilcove DS, Rothstein D, Dubow J et al (1998) Quantifying threats to imperiled species in the United States. Bioscience 48:607–615
Williamson M, Fitter A (1996) The varying success of invaders. Ecology 77:1661–1666
Wittenberg R, Cock MW (2005) Best practices for the prevention and management of invasive alien species. In: Mooney HA, Mack RN, McNeely JA, Neville LE, Schei PJ, Waage JK (eds) Invasive alien species, a new synthesis. Island Press, Washington, DC, pp 209–232
Wright I, Reich P, Westoby M et al (2004) The worldwide leaf economics spectrum. Nature 428:821–827. https://doi.org/10.1038/nature02403
Xu C-Y, Julien MH, Fatemi M et al (2010) Phenotypic divergence during the invasion of Phyla canescens in Australia and France: evidence for selection-driven evolution. Ecol Lett 13:32–44
Young SL (2015) When an invasive plant fails to invade. Front Ecol Environ 13:450–451. https://doi.org/10.1890/1540-9295-13.8.450
Young SL, Kettenring KM (2020) The social-ecological system driving effective invasive plant management: two case studies of non-native Phragmites. J Environ Manag 267:110612. https://doi.org/10.1016/j.jenvman.2020.110612
Young HS, McCauley DJ, Dunbar RB et al (2010) Plants cause ecosystem nutrient depletion via the interruption of bird-derived spatial subsidies. Proc Natl Acad Sci U S A 107:2072–2077. https://doi.org/10.1073/pnas.0914169107
Young SL, Clements DR, DiTommaso A (2017) Climate dynamics, invader fitness, and ecosystem resistance in an invasion-factor framework. Invasive Plant Sci Manag 10:215–231. https://doi.org/10.1017/inp.2017.28
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Young, S.L., Kimball, S., Novak, S.J. (2022). Invasion of Plant Communities. In: Clements, D.R., Upadhyaya, M.K., Joshi, S., Shrestha, A. (eds) Global Plant Invasions. Springer, Cham. https://doi.org/10.1007/978-3-030-89684-3_2
Download citation
DOI: https://doi.org/10.1007/978-3-030-89684-3_2
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-89683-6
Online ISBN: 978-3-030-89684-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)