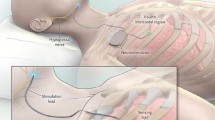
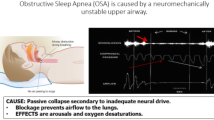
Abstract
Selective electrical neurostimulation of the twelfth cranial nerve, hypoglossal nerve stimulation (HNS), which innervates both the intrinsic and the extrinsic tongue muscles, during sleep to restore or maintain upper airway patency in patients with obstructive sleep apnea (OSA) has a long history of research and development of over 35 years.
The safety, feasibility and clinical effectiveness of different HNS systems have been explored in clinical trials over recent years. Currently, HNS using the Inspire IV device (Inspire Medical Systems, Minneapolis, MN, USA) remains the most evidence-based type of this innovative therapy in selected patients with moderate to severe OSA that are not able to use continuous positive airway pressure (CPAP) therapy. Other HNS systems are currently under investigation with the Genio device (Nyxoah SA, Mont-Saint-Guibert, Belgium) for bilateral neurostimulation of the terminal branches of the hypoglossal nerve submentally being studied in a FDA supervised pivotal trial, the DREAM study.
Evidence from the clinical trials clearly indicate that patient selection for HNS is crucial. This ground-breaking therapy should be offered to carefully selected patients in whom this non-CPAP therapy turns out to be highly effective. Apart from criteria on the severity of OSA and the degree of overweight and obesity, the absence of complete concentric collapse at the level of the palate during a drug-induced sleep endoscopy (DISE) seems to predict therapeutic success with implanted HNS therapy. Therefore, DISE is recommended as a patient selection tool for implantable neurostimulation therapy for the treatment of OSA.
This chapter provides a concise overview of the history and development of HNS for OSA over the years up to the current state of this innovative therapy anno 2021.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Cranial nerve
- Endoscopy
- Neurostimulation
- Pacemaker
- Sleep-related breathing disorders
- Surgical treatments
- Upper airway collapse
Electrical Stimulation of the Hypoglossal Nerve for Treatment of Obstructive Sleep Apnea: History, Current State and Future Perspectives
The research and development phases of the fascinating therapy of electrical neurostimulation of the hypoglossal nerve (nervus hypoglossus – twelfth cranial nerve – CN XII) for the treatment of sleep-disordered breathing spans a period of 30–35 years [1,2,3,4,5,6,7]. Since then the number of published scientific papers on this topic has steadily increased with a peak in the increase in amount of manuscripts over the last years (Fig. 4.1).
In their key paper, back in 1978, John Remmers et al. described the importance of the genioglossus (GG) muscle in maintaining upper airway patency during sleep in the counter balance with the loss of upper airway muscle tone during sleep [8]. The resulting hypothesis that artificial activation of the GG muscle might be effective in the treatment of obstructive sleep apnea (OSA) was first evaluated by Miki et al. as these authors could demonstrate that upper airway resistance decreased by electrical stimulation of the GG in anesthetized dogs [9]. The first human study was performed by the same research group assessing the efficacy of surface stimulation at the level of the submental region in six OSA patients during sleep [1]. The results of this experiment suggested a decrease in the frequency of apneic episodes and longest apnea duration, and an increase in arterial oxygen saturation [1]. The percutaneous electrical stimulation that was employed in this study was not causing arousal neither was it significantly affecting blood pressure or heart rate [10]. The findings suggested that submental electrical stimulation using the technology of apnea demand-type stimulator based on tracheal breath sounds could be a non-invasive and effective treatment for OSA [1, 10]. Subsequently, the observation was made that placement of electrodes for stimulation in the proximal half of the submental region could decrease supraglottic resistance during mouth breathing both in OSA patients and a control group, and that ideally the surface electrodes were 1 cm apart [11]. Apart from the influence of stimulation site, the effectiveness of submental stimulation turned out to be dependent on stimulation intensity with a frequency- and voltage dependency of the effect of stimulation [11]. After these first studies, results of other trials evaluation submental stimulation were published with mixed results [12]. A limited cure rate was reported in an abstract published by Verse et al. [13]: transcutaneous electrical stimulation was reported to reduce the apnea/hypopnea-index (AHI), snoring and daytime sleepiness significantly [13]. Both intraoral and subcutaneous submental stimulation are associated with time-linked arousals [12, 14]. In spite of these discouraging findings a recent flare of clinical trials on these concepts has been noted. After the publication of feasibility studies the protocol of a larger study that evaluates transcutaneous electrical stimulation for OSA was published in 2019 by He et al. [15]. Additionally a notable improvement in both snoring and mild OSA was reported by Kotecha et al. with a novel intraoral neuromuscular stimulation device using daytime awake neuromuscular electrical stimulation to induce toning of the tongue muscles [7]. This daytime neuromuscular electrical training appears to be well tolerated and effective at reducing snoring while improving sleep quality of both patient and his/her partner [16].
The clinical history of the more invasive direct method of hypoglossal nerve stimulation (HNS) therapy dates back to the four-centre trial evaluating unilateral HNS in eight patients with OSA [2]. This study converges with the first ever surgical procedure for implantable HNS therapy in a human OSA patient at the Antwerp University Hospital in Edegem, Belgium, in June 1996. The hypoglossal nerve stimulation system evaluated in this study (Inspire I stimulating system, Medtronic Inc., Minneapolis, MN, USA; Fig. 4.2) was designed to synchronize the delivery of the CN XII stimulation with the patient’s inspiration using an implantable intrathoracic pressure sensor, a programmable pulse-generating system, and a stimulating half-cuff silicone-insulated, guarded, bipolar platinum electrode placed around the main trunk of the nerve at the left side. Using an external programming unit the stimulation parameters and inspiratory sensing algorithms could be adjusted. Impulses were delivered to the hypoglossal nerve via a lead. A self-controlled programming unit was provided for patients to initiate and terminate electrical stimulation. The findings of this particular study could confirm the safety, feasibility and therapeutic potential of implanted HNS with a significant reduction of AHI [2].
Schematic overview of the different component of the Inspire I stimulating system (Medtronic Inc., Minneapolis, Minn, USA) and surgical access. Implantable pulse generator (IPG) placement in an infraclavicular pocket superficial to the pectoralis major muscle fascia. The nerve-electrode lead and pressure-transducer lead are tunnelled to the IPG pocket and connected to the IPG. (From Eisele [17]; with permission)
The learnings from that early experience in eight OSA patients included that implanted HNS synchronized with ventilatory effort during sleep may indeed provide a potential treatment for OSA and that improvements in OSA were related to CN XII dependent muscle recruitment and not to arousal. During the study, however, there were some technical issues related with stimulation hardware and software [2, 18].
The re-engineered Inspire II system (Inspire Upper Airway Stimulation (UAS) device, Inspire Medical Systems, Inc., Maple Grove, MN, USA; Fig. 4.3) got subsequently launched and evaluated in a clinical multi-centre trial enrolling patients from European and US centres [3]. The second generation implant included improved cuff electrode of the stimulation lead and demonstrated pressure sensing technology. During the surgical implantation the cuff section of the stimulation lead was placed on the medial division of the distal hypoglossal nerve aiming at selective stimulation of the protruding tongue muscles [4, 19].
Schematic overview of the different components of the Inspire II-IV therapy concept. The respiration-sensing lead located at the level of the fourth or fifth intercostal space detects in- and expiration of the patient during sleep thereby measuring the respiratory cycle. After conversion of the respiratory signal by the neurostimulator or IPG, placed in an infraclavicular pocket superficial to the pectoralis major muscle fascia at the right site, intermittent stimulating pulses are delivered to the hypoglossal nerve through the stimulation lead onto the cuff electrode around the protruding branches of the hypoglossal nerve. This means that a unilateral respiration-synchronized stimulation of the hypoglossal nerve generates a protrusion of the tongue. (With permission)
The findings of the study by Van de Heyning et al. evaluating the Inspire II system have become the evidence base for the current inclusion criteria in routine clinical practice for the application of HNS [3]. After all, the results have clearly demonstrated that this type of HNS therapy is safe and efficacious in selected OSA patients with moderate to severe disease who cannot or will not use CPAP as their primary treatment. In addition, patients turned out more eligible to respond when body mass index (BMI) is not greater than 35 kg/m2 and when no complete concentric palatal collapse (CCCp) is observed during drug-induced sleep endoscopy (DISE) [20]. The latter being subsequently confirmed in a larger set of OSA patients receiving HNS therapy with the Inspire II device [21].
A next phase in the clinical evaluation of the second generation Inspire devices for upper airway stimulation therapy using unilateral HNS was the Stimulation Therapy for Apnea Reduction (STAR) Trial . This international multicenter, prospective, single-group trial with participants serving as their own controls evaluated the effectiveness of the therapy in a group of 126 patients with moderate to severe OSA with a history of nonadherence to CPAP. The analysis of the endpoints 12 months after implantation could illustrate that this unilateral stimulation of the hypoglossal nerve, synchronous with ventilation, resulted in significant and clinically relevant reductions in the severity of OSA and self-reported sleepiness, and, meaningful improvements in quality-of-life measures at 1 year [4]. Serious adverse events were uncommon and the side effects were not bothersome to most patients [4]. In addition, the results of a randomized, therapy-withdrawal part of the trial indicated that the reduction in the severity of OSA was maintained among those who continued the therapy illustrating that the therapy effect is due to stimulation [4].
The one-year follow-up findings of this STAR study published in the New England Journal of Medicine, are, up to this date, largely reassuring with regard to the clinical prescription of this innovative and emerging therapy to selected OSA patients.
DISE studies in a subset of patients that were under HNS therapy with the Inspire device, illustrated that HNS responders had larger retropalatal enlargement with electrical neurostimulation of the hypoglossal nerve as compared to non-responders and that the neurostimulation thus was able to increase both the retropalatal and retrolingual areas [22]. This observation of multilevel enlargement induced by upper airway neurostimulation therapy may provide an additional strength to the sustained reductions of OSA severity in selected patients receiving HNS therapy [4, 22].
Subsequently, the results of 5 year follow-up data on STAR trial were published in Otolaryngology–Head and Neck Surgery and confirm sustained improvements in sleepiness, quality of life, and respiratory outcomes during 5 years via a unilateral hypoglossal nerve implant in these patients with moderate to severe OSA who have failed nasal CPAP while serious adverse events were uncommon [5]. These results confirmed HNS therapy with the Inspire device as a nonanatomic surgical treatment with long-term benefit for these patients [5].
Meanwhile other systems for HNS therapy were evaluated for the treatment of OSA. Results of most of these trials have been published and confirm the safety and feasibility of implantable systems for HNS therapy for OSA [3, 23,24,25].
The HGNS system (Apnex Medical, Inc., St. Paul, MN, USA; Fig. 4.4) was evaluated at 12 months following implantation in up to 31 OSA patients [26]. The results revealed a significant decrease in the AHI when applying HGNS together with favorable safety and feasibility of this therapy [26]. The company Apnex Medical ceased operation in 2013.
Schematic overview of the implanted components of the HGNS system (Apnex) including a neurostimulator that delivers electrical stimulation to one hypoglossal nerve via a stimulation lead having a distal cuff electrode. Respiration sensing leads detect inspiration using bioimpedance, enabling the system to deliver stimulation synchronous with inspiration. (From Kezirian et al. [26]; with permission)
A next HNS system that has been under investigation is the Aura6000 system (Fig. 4.5) originally developed by ImThera Medical, Inc. (San Diego, CA, USA) and now owned by Liva Nova PLC (London, UK) [19]. The Aura6000 consists of six electrodes within a multi-electrode lead that are placed surgically around the trunk of the hypoglossal nerve [27, 28]. This so-called targeted hypoglossal neurostimulation (THN) uses cyclical neurostimulation to ensure no single nerve fiber is stimulated continuously to avoid muscle fatigue [19, 23]. The Aura6000 system provides a continuous neurostimulation onto the body of the proximal hypoglossal nerve via the multi-electrode lead, thereby obviating the need for respiration-sensing leads; whereas with the Inspire devices, the hypoglossal nerve stimulation is intermittent and synchronized with the respiration-sensing leads that measure the respiratory cycle. Therefore, this system, in contrast to Inspire and Apnex, does not utilize a sensing lead necessitating only two surgical incisions when implanting the Aura6000 system [27].
ImThera Aura6000 Medical’s targeted hypoglossal neurostimulation system (Liva Nova). A pulse generator is implanted subcutaneously below the clavicle. A submandibular incision is used to expose the hypoglossal nerve trunk, and a lead cuff is placed around the nerve. The wire is passed subcutaneously using a canula and connected to the pulse generator. (From Friedman et al. [27]; with permission)
In a first study published by Mwenge et al. unilateral THN was implanted successfully in 13 out of 14 patients with moderate to severe OSA [23]. The study protocol included follow-up polysomnography 12 months after surgical implantation and a significant reduction in AHI as demonstrated together with improvements in oxygen desaturation index (ODI), arousal index and daytime sleepiness [23]. In a provisional last publication, the authors report the results of an open-label multicentre study suggesting that THN therapy being likely to be safe and effective in selected OSA patients with a baseline AHI lower than 65 per hour sleep and a baseline BMI not higher than 35 kg per square meter [27].
The concept of the Genio™ system for HNS therapy (Fig. 4.6 – Nyxoah SA, Mont-Saint-Guibert, Belgium) is quite different from the three other systems as it provides bilateral stimulation of the nerve and does not consist of an implantable pulse generator or IPG. The Genio™ system is implanted submentally and stimulates the terminal branches of the hypoglossal nerve bilaterally via an implanted neurostimulator that will be activated externally [25]. The external activation is provided by a disposable patch submentally on which the activation chip with its own rechargeable battery will be connected every night. A patented duty cycle algorithm will use the patient’s breathing frequency as a reference to make adjustments to the stimulation cycle.
Overview of the components of the Genio™ system for HNS therapy (Nyxoah SA, Mont-Saint-Guibert, Belgium). (a) the implanted stimulator straddling the genioglossus muscles and protruding branches of the hypoglossal nerve bilaterally and (b) the disposable patch and activation unit. (From Eastwood et al. [25]; with permission)
The results published in 2020 by Eastwood et al. report on 27 implanted patients with moderate to severe OSA. The data suggest that the relative non-invasiveness of Genio HNS therapy does not compromise its effectiveness relative to the other methods [25]. During the six-months post-implantation period no serious adverse events occurred and significant improvements in AHI, ODI, quality of life and snoring were noted [25].
Currently, several clinical trials that evaluate Genio therapy are enrolling. The Dual sided hypoglossal neRvE stimulAtion for the treatMent of obstructive sleep apnea (DREAM) study is an international multicentre FDA supervised pivotal trial with the target to implant 134 CPAP intolerant patients with moderate to severe OSA without CCCp on baseline DISE.
At this stage, the data in the literature on the use of Inspire HNS therapy is quite impressive with a proven track record of good adherence to the therapy and evidence on a remarkable clinical efficacy. Up to 30,000 patients did already undergo implantation with Inspire therapy with treatment reimbursement in an increasing amounts of countries worldwide.
The ADHERE Registry is an international multicentre prospective observational cohort study following outcomes of HNS Inspire therapy in patients who have failed CPAP therapy for OSA. The aim of this registry is to assess the outcomes of patients receiving this treatment of OSA in the “real world” setting outside of clinical trials. Up to February 2019 the registry had enrolled more than 1000 patients while an interim analysis on patients that completed their 12-month follow-up demonstrated that also in routine settings the HNS Inspire therapy effect is durable with significant improvements in both subjective and objective OSA outcomes and high adherence to the therapy [29].
Within and outside ADHERE the quest for the additional predictors for more successful HNS therapy is ongoing [29] with further analysis of, among others, the role of cross motor innervation of the hypoglossal nerve and other neuromuscular stimulation aspects of HNS, DISE phenotyping and polysomnographic pathophysiological endotypes [21, 30,31,32].
Currently, apart from an animal study reporting on electrical stimulation of the superior laryngeal nerve for apnea recovery [33], no evidence on electrical neurostimulation therapy for central sleep apnea can be demonstrated from a review of the existing literature.
In conclusion, many clinical trials and systematic reviews indicate that HNS therapy for selected OSA patients seems to be safe with high rates of compliance and therapy adherence, and, stable outcome results over several years of follow-up [5, 12, 18, 19, 24, 26, 29, 34,35,36,37]. A fascinating story of research and development for over 35 years has led to the clinical implementation of this innovative therapy while evaluation and further improvement of HNS therapy for OSA continues.
References
Miki H, Hida W, Inoue H, Takishima T. A new treatment for obstructive sleep apnea syndrome by electrical stimulation of submental region. Tohoku J Exp Med. 1988;154:91–2.
Schwartz AR, Bennett ML, Smith PL, De Backer W, Hedner J, Boudewyns A, Van de Heyning P, Ejnell H, Hochban W, Knaack L, Podszus T, Penzel T, Peter JH, Goding GS, Erickson DJ, Testerman R, Ottenhoff F, Eisele DW. Therapeutic electrical stimulation of the hypoglossal nerve in obstructive sleep apnea. Arch Otolaryngol Head Neck Surg. 2001;127:1216–23.
Van de Heyning PH, Badr MS, Baskin JZ, Cramer Bornemann MA, De Backer WA, Dotan Y, Hohenhorst W, Knaack L, Lin HS, Maurer JT, Netzer A, Odland RM, Oliven A, Strohl KP, Vanderveken OM, Verbraecken J, Woodson BT. Implanted upper airway stimulation device for obstructive sleep apnea. Laryngoscope. 2012;122:1626–33.
Strollo PJ Jr, Soose RJ, Maurer JT, de Vries N, Cornelius J, Froymovich O, Hanson RD, Padhya TA, Steward DL, Gillespie MB, Woodson BT, Van de Heyning PH, Goetting MG, Vanderveken OM, Feldman N, Knaack L, Strohl KP, Group ST. Upper-airway stimulation for obstructive sleep apnea. N Engl J Med. 2014;370:139–49.
Woodson BT, Strohl KP, Soose RJ, Gillespie MB, Maurer JT, de Vries N, Padhya TA, Badr MS, Lin HS, Vanderveken OM, Mickelson S, Strollo PJ Jr. Upper airway stimulation for obstructive sleep apnea: 5-year outcomes. Otolaryngol Head Neck Surg. 2018;159:194–202.
Lewis R, Petelle B, Campbell MC, MacKay S, Palme C, Raux G, Sommer JU, Maurer JT. Implantation of the nyxoah bilateral hypoglossal nerve stimulator for obstructive sleep apnea. Laryngoscope Investig Otolaryngol. 2019;4:703–7.
Kotecha B, Wong PY, Zhang H, Hassaan A. A novel intraoral neuromuscular stimulation device for treating sleep-disordered breathing. Sleep Breath = Schlaf Atmung. 2021;25(4):2083–90.
Remmers JE, de Groot WJ, Sauerland EK, Anch AM. Pathogenesis of upper airway occlusion during sleep. J Appl Physiol Respir Environ Exerc Physiol. 1978;44:931–8.
Miki H, Hida W, Shindoh C, Taguchi O, Inoue H, Takishima T. Dilation of upper airway by stimulation of genioglossus muscle in anesthetized dogs. Amer Rev Resp Dis. 1986;133:A306. (abstract).
Miki H, Hida W, Chonan T, Kikuchi Y, Takishima T. Effects of submental electrical stimulation during sleep on upper airway patency in patients with obstructive sleep apnea. Am Rev Respir Dis. 1989;140:1285–9.
Hida W, Okabe S, Miki H, Kikuchi Y, Kurosawa H, Takishima T, Shirato K. Submental stimulation and supraglottic resistance during mouth breathing. Respir Physiol. 1995;101:79–85.
Strohl K, Baskin J, Lance C, Ponsky D, Weidenbecher M, Strohl M, Yamauchic M. Origins of and implementation concepts for upper airway stimulation therapy for obstructive sleep apnea. Respir Investig. 2016;54:241–9.
Verse T, Schwalb J, Hormann K, Stuck BA, Maurer JT. submental transcutaneous electrical stimulation for obstructive sleep apnea. HNO. 2003;51:966–70.
Guilleminault C, Powell N, Bowman B, Stoohs R. The effect of electrical stimulation on obstructive sleep apnea syndrome. Chest. 1995;107:67–73.
He B, Al-Sherif M, Nido M, Tas R, Beach M, Schwarz EI, Cheng M, Ishak A, Lee K, Shah N, Kent B, Eze-John P, Ratneswaran C, Rafferty G, Williams AJ, Hart N, Luo Y, Moxham J, Pengo M, Steier J. Domiciliary use of transcutaneous electrical stimulation for patients with obstructive sleep apnoea: a conceptual framework for the tesla home programme. J Thorac Dis. 2019;11:2153–64.
Baptista PM, Martinez Ruiz de Apodaca P, Carrasco M, Fernandez S, Wong PY, Zhang H, Hassaan A, Kotecha B. Daytime neuromuscular electrical therapy of tongue muscles in improving snoring in individuals with primary snoring and mild obstructive sleep apnea. J Clin Med. 2021;10:1883.
Eisele DW, et al. Tongue neuromuscular and direct hypoglossal nerve stimulation for obstructive sleep apnea. Op Tech Otolaryngol Head Neck Surg. 2000;11:59–65.
Kezirian EJ, Boudewyns A, Eisele DW, Schwartz AR, Smith PL, Van de Heyning PH, De Backer WA. Electrical stimulation of the hypoglossal nerve in the treatment of obstructive sleep apnea. Sleep Med Rev. 2010;14:299–305.
Mashaqi S, Patel SI, Combs D, Estep L, Helmick S, Machamer J, Parthasarathy S. The hypoglossal nerve stimulation as a novel therapy for treating obstructive sleep apnea-a literature review. Int J Environ Res Public Health. 2021;18:1642.
Dieltjens M, Vanderveken OM, Hamans E, Verbraecken JA, Wouters K, Willemen M, De Backer WA, Van de Heyning PH, Braem MJ. Treatment of obstructive sleep apnea using a custom-made titratable duobloc oral appliance: A prospective clinical study. Sleep Breath = Schlaf Atmung. 2013;17:565–72.
Vanderveken OM, Maurer JT, Hohenhorst W, Hamans E, Lin HS, Vroegop AV, Anders C, de Vries N, Van de Heyning PH. Evaluation of drug-induced sleep endoscopy as a patient selection tool for implanted upper airway stimulation for obstructive sleep apnea. J Clin Sleep Med. 2013;9:433–8.
Safiruddin F, Vanderveken OM, de Vries N, Maurer JT, Lee K, Ni Q, Strohl KP. Effect of upper-airway stimulation for obstructive sleep apnoea on airway dimensions. Eur Respir J. 2015;45(1):129–38.
Mwenge GB, Rombaux P, Dury M, Lengele B, Rodenstein D. Targeted hypoglossal neurostimulation for obstructive sleep apnoea: a 1-year pilot study. Eur Respir J. 2013;41:360–7.
Eastwood PR, Barnes M, Walsh JH, Maddison KJ, Hee G, Schwartz AR, Smith PL, Malhotra A, McEvoy RD, Wheatley JR, O’Donoghue FJ, Rochford PD, Churchward T, Campbell MC, Palme CE, Robinson S, Goding GS, Eckert DJ, Jordan AS, Catcheside PG, Tyler L, Antic NA, Worsnop CJ, Kezirian EJ, Hillman DR. Treating obstructive sleep apnea with hypoglossal nerve stimulation. Sleep. 2011;34:1479–86.
Eastwood PR, Barnes M, MacKay SG, Wheatley JR, Hillman DR, Nguyen XL, Lewis R, Campbell MC, Petelle B, Walsh JH, Jones AC, Palme CE, Bizon A, Meslier N, Bertolus C, Maddison KJ, Laccourreye L, Raux G, Denoncin K, Attali V, Gagnadoux F, Launois SH. Bilateral hypoglossal nerve stimulation for treatment of adult obstructive sleep apnoea. Eur Respir J. 2020;55:1901320.
Kezirian EJ, Goding GS Jr, Malhotra A, O’Donoghue FJ, Zammit G, Wheatley JR, Catcheside PG, Smith PL, Schwartz AR, Walsh JH, Maddison KJ, Claman DM, Huntley T, Park SY, Campbell MC, Palme CE, Iber C, Eastwood PR, Hillman DR, Barnes M. Hypoglossal nerve stimulation improves obstructive sleep apnea: 12-month outcomes. J Sleep Res. 2014;23:77–83.
Friedman M, Jacobowitz O, Hwang MS, Bergler W, Fietze I, Rombaux P, Mwenge GB, Yalamanchali S, Campana J, Maurer JT. Targeted hypoglossal nerve stimulation for the treatment of obstructive sleep apnea: six-month results. Laryngoscope. 2016;126:2618–23.
Zaidi FN, Meadows P, Jacobowitz O, Davidson TM. Tongue anatomy and physiology, the scientific basis for a novel targeted neurostimulation system designed for the treatment of obstructive sleep apnea. Neuromodulation. 2013;16:376–86; discussion 386.
Thaler E, Schwab R, Maurer J, Soose R, Larsen C, Stevens S, Stevens D, Boon M, Huntley C, Doghramji K, Waters T, Kominsky A, Steffen A, Kezirian E, Hofauer B, Sommer U, Withrow K, Strohl K, Heiser C. Results of the adhere upper airway stimulation registry and predictors of therapy efficacy. Laryngoscope. 2020;130:1333–8.
Heiser C, Vanderveken OM, Edenharter GM, Hofauer B. Cross motor innervation of the hypoglossal nerve-a pilot study of predictors for successful opening of the soft palate. Sleep Breath Schlaf Atmung. 2020;25(1):425–31.
Op de Beeck S, Wellman A, Dieltjens M, Strohl KP, Willemen M, Van de Heyning PH, Verbraecken JA, Vanderveken OM, Sands SA, STAR Trial Investigators. Endotypic mechanisms of successful hypoglossal nerve stimulation for obstructive sleep apnea. Am J Respir Crit Care Med. 2020;203(6):746–55.
Huyett P, Kent DT, D'Agostino MA, Green KK, Soose RJ, Kaffenberger TM, Woodson BT, Huntley C, Boon MS, Heiser C, Birk A, Suurna MV, Lin HS, Waxman JA, Kezirian EJ. Drug-induced sleep endoscopy and hypoglossal nerve stimulation outcomes: a multicenter cohort study. Laryngoscope. 2021;131:1676–82.
Lawson EE. Recovery from central apnea: effect of stimulus duration and end-tidal CO2 partial pressure. J Appl Physiol Respir Environ Exerc Physiol. 1982;53:105–9.
Certal VF, Zaghi S, Riaz M, Vieira AS, Pinheiro CT, Kushida C, Capasso R, Camacho M. Hypoglossal nerve stimulation in the treatment of obstructive sleep apnea: a systematic review and meta-analysis. Laryngoscope. 2014;125(5):1254–64.
Costantino A, Rinaldi V, Moffa A, Luccarelli V, Bressi F, Cassano M, Casale M, Baptista P. Hypoglossal nerve stimulation long-term clinical outcomes: a systematic review and meta-analysis. Sleep Breath Schlaf Atmung. 2020;24(2):399–411.
Woodson BT, Soose RJ, Gillespie MB, Strohl KP, Maurer JTM, de Vries N, Steward DL, Baskin JZ, Badr MS, Lin HS, Padhya TA, Mickelson S, Anderson WM, Vanderveken OM, Strollo PJ Jr, Investigators ST. Three-year outcomes of cranial nerve stimulation for obstructive sleep apnea: the star trial. Otolaryngol Head Neck Surg. 2016;154:181–8.
Strohl MM, Yamauchi M, Peng Z, Strohl KP. Insights since fda approval of hypoglossal nerve stimulation for the treatment of obstructive sleep apnea. Curr Sleep Med Rep. 2017;3:133–41.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Van de Heyning, P., Vanderveken, O.M. (2022). History of Electrical Stimulation in Sleep Apnea. In: Heiser, C., de Vries, N. (eds) Upper Airway Stimulation in Obstructive Sleep Apnea . Springer, Cham. https://doi.org/10.1007/978-3-030-89504-4_4
Download citation
DOI: https://doi.org/10.1007/978-3-030-89504-4_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-89503-7
Online ISBN: 978-3-030-89504-4
eBook Packages: MedicineMedicine (R0)