Abstract
Anterior cruciate ligament (ACL) injuries typically occur during sports participation, and reconstruction surgery is commonly performed to enable the athlete to return to high-impact sports that involve landing, cutting, and pivoting movements. However, despite restoration of knee stability following surgery, many athletes do not return to their prior level of sport and of those that do the occurrence of second ACL injuries are of concern, particularly in younger athletes. Criteria-based return to sport rehabilitation has been shown to be of benefit to prepare athletes for a safe return to play and is discussed in this chapter. Such programs are conducted in stages, and criteria for progression from one phase to the next, which include return to sport, are proposed that target modifiable deficits. These typically include ligament, quadriceps, limb and trunk dominance patterns. Another approach to assess readiness to return to sport is the use of return to sport testing where a set of criteria or ‘test battery’ is given to the athlete for clearance to return to sport. This chapter also discusses the current knowledge of such return to sport testing.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
9.1 Introduction
Most anterior cruciate ligament (ACL) injuries occur during sports participation. Therefore, the goal for most athletes who undergo ACL reconstruction surgery is to return their preinjury sport at a similar level of performance and in the absence of further injury. However, it has become apparent that for many athletes, these goals are not always attained. Rates of return to sport are often lower than expected and younger athletes are at considerable risk for sustaining multiple ACL injuries. This chapter will explore current knowledge regarding return to sport after ACL reconstruction, with a focus on criteria-based rehabilitation and return to sport testing.
9.2 Return to Sport Rates and Factors that Influence Return to Sport
An initial systematic review that investigated return to sport rates after ACL reconstruction surgery showed that while 82% of patients returned to some form of sport, only 63% were participating at their pre-injury level at follow-up [1]. These return rates contrasted with the finding that 90% of patients rated normal or nearly normal on impairment-based measures. This review was subsequently updated, and in the update similar rates were noted; 65% returned to the pre-injury level of sport and 55% returned to competition sport [2]. Comparable findings have also been reported for reviews in which only patients with revision reconstruction have been included [3]. Perhaps not surprisingly, elite-level athletes have the highest return rates whereby 83% return to their pre-injury sport [4], followed by younger aged athletes, in whom it has been reported that 81% return to competition sport [5]. Despite higher return rates in some select groups, the overall message is that a return to sport is not guaranteed following ACL reconstruction surgery.
Many factors influence whether an individual will return to sport after ACL reconstruction, and include demographic and social characteristics, as well as surgical and rehabilitation factors [6]. There are empirical data to show that males have higher return rates, usually in the order of approximately 10%, and that younger athletes can have up to 30% higher return rates [1, 7]. A positive psychological response and higher levels of motivation during rehabilitation have also been associated with higher rates of returning to pre-injury sport [8,9,10,11]. It is also highly relevant to consider the role of patient expectations and work in this area has shown that for a first ACL injury, over 80% of patients expect to be able to return to their previous level of sport [12, 13]. As this is higher than what return rates are, many athletes will not realize their goals, and may therefore need support and advice to readjust their expectations.
9.3 Return to Sport and Second ACL Injury
When planning a return to sport after ACL reconstruction surgery patients are often concerned about the risk of sustaining the same injury again. This is logical, as return to sport puts the individual at risk of both ACL graft rupture and rupture of the contralateral ACL. Therefore, there has been much effort to determine predictors, such as patient and surgical variables, that can be used to identify at risk individuals. Numerous variables for re-injury have been considered, often with mixed findings [7]. However, most studies do report on sex and age.
Findings in relation to patient sex and re-injury are not straightforward. Sex as a risk factor for graft rupture has either shown no influence or males, particularly younger males have been shown to be at greatest risk [14,15,16]. In contrast, females appear at greatest risk for subsequent contralateral ACL injury [17, 18]. Why such sex difference may occur is not clear and requires further investigations that consider potential confounders such as the type of sport played as well as the amount of exposure.
There is a large amount of evidence to show that younger athletes are at significantly increased risk for both graft ruptures and contralateral ACL injuries [19]. Although the definition, or cut-off, for ‘younger’ has varied (usually from <16 years to <25 years), both large cohort studies and registry databases have confirmed this increased risk. The rate of second ACL injuries in the younger population is concerningly high, with cohort studies reporting that between 20% and 30% of younger athletes sustain a second ACL injury [16, 20,21,22]. It is therefore important to understand the reasons why younger patients are at such increased risk so that this can form part of any rehabilitation strategy or return to play decision making.
It is well accepted that it is unlikely that age itself is the risk factor, but rather a proxy for multiple factors [23]. In this regard, there are a few salient aspects where younger patients tend to differ from their ‘older’ counterparts. These are that younger patients are more likely to return to sport and they are also more likely to return earlier after surgery [24]. Furthermore, when they do return, the sports they play are high-risk sports for knee injury [25]. Recent studies have also shown that younger athletes resume these high-risk sports with strength and functional deficits [26, 27]. Data from a Swedish Rehabilitation Outcome Registry showed that 50% of adolescent patients (15–20 years) had resumed strenuous sports by 8 months post ACL reconstruction surgery despite only 29% having achieved satisfactory muscle function [26]. A similar study by Toole et al. [28] reported that only 14% of adolescent patients (mean age 17 years) met recommended strength and functional thresholds when cleared to return at 8 months post-surgery. As such, the rehabilitation of and timing for return to sport needs to be carefully considered in this group.
9.4 When Should Athletes Return to Sport?
Whatever surgical technique or rehabilitation program is used, perhaps the most difficult question to answer is when is it safe for the athlete to return to sport? There are two issues we are concerned about in relation to this question. The first is graft rupture/failure which was discussed in the previous section. Damage to the rest of the knee, both in the short and longer term, also should be considered.
Animal studies have shown that there are distinct phases of graft maturation with early graft necrosis and subsequent hypercellularity and revascularization being potential risk periods for reinjury [29]. While there is less information in humans, it has been suggested that there are the same phases, but that these occur over a slower time frame than in animals [30]. For example, the period of remodelling, where the graft is most at risk, corresponds roughly to the 4- to 12-month post-surgery time point. This also corresponds to the time when many athletes are considering a return to their pre-injury sport, which for most, includes pivoting and twisting movements. Data from hamstring tendon grafts also indicate that remodelling may take as long as 12–24 months [31]. This is the same time point where registry datasets have shown that the peak of second ACL injuries occurs.
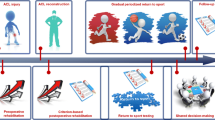
In addition to these biological considerations, it is common for athletes to demonstrate deficits in neuromuscular control and knee extension strength that do not reach baseline levels until at least 2 years after surgery. To avoid reinjury, such functional deficits should ideally be resolved before return to sport is considered. Therefore, Nagelli and Hewett [32] suggested that patients wait for 2 years before making a return to sport and Fig. 9.1 shows a schematic diagram of both biological and functional factors which are subject to change by having such a delay. While the theory behind this notion is evidence based, it is also recognized that there is yet to be evidence to confirm that such a delay would make a difference. It is also not clear whether athletes, parents or coaches would be willing to adhere to such long timeframes and these may significantly impact on career prospects for some young athletes. There is also wide individual variation in the recovery of ACL injury and reconstruction surgery and therefore an approach that is both time and criteria based may be preferable.
Schematic showing that the time of recovery of baseline joint health and function is not indicated until 2 years post ACL injury or reconstruction surgery. From Nagelli and Hewett [32]
9.5 Criteria-Based Rehabilitation
One approach to address readiness to return to sport is to target modifiable neuromotor deficits known to be associated with both first and second ACL injury in phased rehabilitation programs and set criteria for progression from one phase to the next, which include return to sport. These modifiable deficits include trunk, ligament, quadriceps, and limb/leg dominance patterns [33].
These neuromotor deficits of trunk, ligament, quadriceps, and limb/leg dominance are fortunately readily observable and serially measurable after ACL rupture and reconstruction and should be measured longitudinally during the return to sport (RTS) process [33]. The athlete should understand that although the surgical reconstruction of the ligament may have created a mechanically stable joint, they may not have a functionally stable knee during dynamic movements and that they must be serially tested for any potential functional deficits [34, 35] and these deficits need to be addressed before return to play is advised.
Well-controlled, valid and reliable longitudinal measurement of the aforementioned neuromotor imbalances of trunk, ligament, quadriceps and leg dominance are critical for the allowable progression of post-injury and post-operative progression of the injured athlete through both the early and late stages of rehabilitation. As such, coordination between active muscular and passive ligamentous control of tibiofemoral motion in all three planes of motion must be assessed and enhanced [33]. Fortunately, such neuromotor imbalances, trunk dominance, for example, are readily observable and measurable post ACL rupture and reconstruction. A dynamically stable knee joint, which is prepared to return to demands of competitive play, must coordinate passive mechanical function of the ACL graft with appropriate neuromuscular control of the hip, knee and entire kinetic chain, and most especially of the trunk or “core,” to provide muscular dampening of joint loads and reduce strain on the graft [34]. A progressive, functional testing and rehabilitation program is required to provide the athlete with an effective means of facilitation of positive adaptations to the much needed and often impaired proprioceptive function of the knee joint. Proper ACL return to play testing and training can provide the athlete with a dynamically functional joint that is prepared to respond to the extreme forces generated during athletic competition and reduce the risk of a second or even further subsequent injury and will optimally prepare even the young, highly active high-risk athlete to achieve pre-injury performance levels safely [36].
Neuromuscular control deficits, for example, quadriceps dominance, are readily observable and measurable after ACL rupture and well into the postoperative rehabilitation period [37]. Rehabilitation professionals should serially measure these deficits and focus rehabilitative interventions in these areas. Paterno et al. [38] demonstrated the observable and measurable presence of quadriceps dominance by a straight knee and a “stiffening” strategy on balance testing in athletes at high risk for a second ACL injury. Female athletes may have a greater loss of single-leg postural balance at return from a knee injury when compared with males. They may also return to their preinjury state slower than their male counterparts [37]. A standard level of proficiency in postural balance before return to sport is important to protect athletes from a second ACL injury. Athletes that do not demonstrate postural balance within two standard deviations of normal have a significantly higher risk of injury [39]. Balance and proprioceptive training past the acute postsurgical rehabilitation phase is a necessary requirement, not only for restoration of functionality, but for its prophylactic effect on ligament reinjury [37, 38, 40, 41].
The observed presence of significant leg dominance, as evidenced and measured by the absence of limb symmetry following ACL reconstruction, may be due to imbalances between muscular strength and joint kinematics between contralateral lower extremity measures in athletes at high risk for first and second ACL ruptures. For example, females may generate lower hamstrings torques on the non-dominant than in the dominant leg [42]. More specifically, adolescent female athletes have significant side-to-side differences in maximum knee valgus angle compared to males during a box drop vertical jump [43]. Side-to-side imbalances in muscular strength, flexibility and coordination are important predictors of increased injury risk [44,45,46]. Side-to-side balance in strength and flexibility is important for the prevention of injuries and when imbalances are present, the athlete may be more injury-prone [44]. Patients with muscle strength imbalances may exhibit a higher incidence of first and second ACL injury [45]. We [46] developed a model to predict ACL injury risk with high sensitivity and specificity. Half of the parameters in the predictive model were leg dominance indicators of side-to-side differences in lower limb kinematics and kinetics [46]. Side-to-side limb neuromotor imbalances likely increase risk for both limbs. Over-reliance on the non-ACL ruptured and reconstructed limb can put greater stress and torques on that knee, while the involved operated limb is also likely at increased risk due to an inability of the musculature on that side to effectively absorb the high forces associated with sporting activities.
A more advanced level of testing and training that should be utilized to observe, measure and target ligament, quadriceps, leg and trunk dominance patterns is the utilization of unanticipated cutting movements. Prior to teaching unanticipated landing and cutting, rehabilitation professionals, coaches and athletes should first consider the work of Paterno et al. [38]. Trunk dominance may be evidenced by the observation of a net negative hip internal rotation impulse on a drop vertical jump with 3D biomechanical testing or visually observable internal hip rotation. Ligament dominance can be observed by inward hip and knee collapse during the drop vertical jump. Quadriceps dominance by a straight knee and a “stiffening” strategy on balance testing. Leg dominance can be observed and measured by side-to side differences in relative quadriceps and hamstring activation via peak knee flexion/extension moments on a drop vertical jump during 3D biomechanical testing, peak torque on dynamometer testing or with standardized hop testing [33, 38, 47, 48].
Limb or leg dominance may not be fully corrected by simple single-faceted sagittal plane training and conditioning protocols that do not incorporate cutting manoeuvres will not provide similar levels of external varus/valgus or rotational loads that are seen during sport-specific cutting manoeuvres [49]. Testing and training programs that measure and teach safe levels of knee abduction/adduction moment dominated valgus and varus stresses may induce more muscle dominant neuromuscular adaptations [50]. Such adaptations can better prepare an athlete for more multi-directional sports movements that can improve their performance and reduce risk of second ACL injury [42, 51, 52]. High-risk athletes perform cutting techniques with decreased knee flexion and increased knee abduction/adduction valgus/varus angles [53].
Ligament dominance, as evidenced by increased “valgus” knee abduction loads, can double when performing unanticipated cutting manoeuvres similar to those utilized in sport [54]. Thus the endpoint of training designed to reduce knee abduction/adduction ACL loading via valgus and varus torques can be gained through training the athlete to use movement techniques that produce the low abduction and adduction knee joint moments [50]. Training that incorporates unanticipated movements can reduce knee joint loads [55]. In addition, training individuals to pre-activate the neuro-musculature that surrounds and controls the position of the knee joint prior to ground contact may facilitate appropriate kinematic adjustments and ACL loads may be reduced [54, 56]. Training the athlete to employ safe cutting techniques in unanticipated sport situations may instil technique adaptations that will more readily transfer onto the field of play. If achieved, the ligament dominant athlete may become muscular dominant, reducing their future risk of ACL injury [42, 52].
9.6 Return to Sport Testing
Another approach to assess readiness to return to sport that can be applied to either criteria or time-based rehabilitation is to use a set of criteria or ‘test battery’ to give the athlete clearance for return to sport. This is typically used at the final stages of rehabilitation and athletes who pass are cleared to return. Not surprisingly, the content of such testing is varied, and several consensus statements and clinical practice guidelines have been put forward. van Melick et al. [57] attempted to reach a consensus in regard to which criteria should be used to determine the moment of return to play. It was recommended that an extensive test battery for both quantity and quality of movement should be performed, and that the test battery itself should include a series of strength tests, hop tests and measurement of quality of movement. A limb symmetry index of greater than 90% was suggested as a pass criterion, but it was also suggested that this could be increased to 100% for patients planning a return to pivoting or contact sports. This seems a reasonable approach; however, as younger athletes are also more likely to meet criteria that are used to indicate readiness to return to sport, there may be a role for adjusting ‘pass’ thresholds for these criteria based on age as well.
An additional consensus statement on return to sport concluded that, for any injury, the return to sport decision should always use information gained from a battery of tests and should assess direction change and reactive agility, as well as psychological readiness [58]. In this regard, it is interesting to note that more recent studies have attempted to cover a broad range of risk factors, often suggesting that 15–20 different tests be used [59, 60]. This is likely due to uncertainty regarding what the most important factors for various outcomes, such as player performance or risk of further injury, are. However, this approach may cause an unnecessary burden and use of resources for both the patient and clinician. It has therefore been suggested that the focus should be on fewer but important factors and that five factors should be enough as any one factor would ideally account for at least 20% of the predictive variance [61].
It is also worth discussing whether return to sport tests is designed to determine whether the patient is capable of returning to play or whether they are designed to determine whether it is safe. These are often used interchangeably, and the questions of safety and capability cannot necessarily be addressed in the same way [62]. Most return to sport testing is done with the aim of assessing safety, i.e., whether the patient can make a return without suffering a further ACL injury. However, the same or similar measurements can also be used as assessments of how much of their functional capacity a patient has regained and the likelihood of making a return to sport. For example, meeting return to sport criteria at 6 months post-operatively has been associated with higher rates of returning to preinjury levels of activity up to 2 years [63]. The remainder of this chapter will focus on return to sport testing to assess the safety of returning to sport.
9.6.1 Return to Sport Testing for Making a Safe Return to Play
As it is becoming increasingly common for return to sport testing to be used as part of the decision-making process for a safe return to sport following ACL reconstruction surgery, for such testing to be of value to both the clinician and the patient, its validity should be known, or at least scientifically assessed. Despite the many discussions about return to sport testing following ACL reconstruction, the evidence surrounding it is relatively limited and to some extent contradictory. However, one aspect for which there is consistency is that the proportion of patients who pass return to sport testing is actually rather low. This was highlighted in a recent systematic review which showed a 23% pass rate from 8 studies with 876 patients who were tested before returning to sport at between 5- and 10-month post-surgery [64]. Most of these studies used >90% limb symmetry as the threshold for a pass. Of potential concern, the same pass rate was also reported for three studies (234 patients) in whom the patients had already resumed playing strenuous sports.
The same systematic review also determined whether passing RTS test batteries reduced subsequent rates of any knee injury [64]. The combined results from two studies showed a 72% reduction in risk with passing RTS criteria. However, this reduction was not statistically significant, and 95% confidence intervals were large (from 93% reduction in risk to 21% increase in risk). One of the studies also reported that the risk of subsequent knee injury markedly reduced for each month an athlete delayed returning to sport, until the 9-month mark, and this has been frequently cited [65]. Of the individual components in the RTS test battery, having a quadriceps strength deficit prior to return to sport was the most significant predictor of further knee reinjury. However, since only 18 patients (out of 106) actually passed the RTS testing criteria, caution needs to be applied when interpreting these results and overall it is difficult to draw firm conclusions.
Regarding passing RTS testing and subsequent ACL injury, two reviews showed that passing test batteries did not reduce the risk for all subsequent ACL injuries [64, 66]. However, one review found that passing an RTS test battery did significantly reduce the risk for subsequent graft rupture by 60%, although it increased the risk for a subsequent contralateral ACL injury by 235% [64]. This highlights the conflicting findings that are reported in the literature. Of the included studies in these meta-analyses, only two showed significant results [67, 68]. These also had the largest patient sample sizes, and as such made the greatest contribution to the weighting of the meta-analyses. It is therefore relevant to have a closer look at both.
Kyritsis et al. [67] recorded graft ruptures in elite male athletes and reported that those who did not meet all 7 RTS criteria had a four times greater risk of graft rupture. The hamstring-to quadriceps ratio of the involved leg alone was also highly associated with graft rupture, with a ten times greater risk for every 10% difference in strength. Unfortunately, the contralateral side was not reported on. In comparison, Sousa et al. [68] did not find a reduced risk for graft rupture in their group who passed criteria at 6 months, but they did find a significantly increased risk for contralateral injuries. As the patients who passed RTS testing were cleared for an early return, compared to those who failed and were advised to wait, the authors suggested that this increase in contralateral ACL injuries may be related to an increased activity level in the patients who had passed the criteria. This is a logical assumption; however, a close look at the Kaplan-Meier survival curves presented in Sousa et al. shows that there were few early second ACL injuries in the cohort. For graft ruptures, the earliest occurred after 36 months from surgery and for contralateral ACL injury there are no differences between the RTS pass and fail groups until approximately 20–30 months from surgery. Therefore, despite one group (those who passed RTS testing at 6 months) being cleared for an earlier return this does not seem to have had an impact on the timing of subsequent ACL injuries. As such, the impact of any potential difference in early exposure may in fact be minimal and this needs further exploration [69].
Unfortunately, relatively few studies that investigate RTS testing and further ACL injury report return to sport rates along with re-injury rates. Even when they do, the definition of return to sport varies from one study to another. For example, Grindem et al. [65] classified any reported participation, including training, as having returned. If the patients who pass RTS test are not returning to comparable levels of activity or sport as those who fail testing, it is difficult to draw conclusions about the benefit of meeting the RTS criteria. It is also highly likely that other factors come into play in the time interval between when RTS testing is conducted and when further ACL injuries occur. It may not be meaningful to relate an injury that occurs back to an RTS test that occurred many years earlier. There has been little discussion in the literature as to what may be an appropriate follow-up time period following RTS testing [64].
The frequency of conducting RTS testing has also received little attention. A recent study did nonetheless conclude that it is pointless to undertake knee strength assessments that are closer than 2 months apart [70]. As knee strength outcomes are one variable which has more frequently been associated with reinjury [65, 67] such information can serve to reduce the pressures therapists may feel to provide continual testing. However, as this was a cross-sectional study much work is needed to validate this initial data and extend it to other types of tests.
Whether clinical measures can be used to identify groups at high risk of reinjury has also been investigated [71, 72]. While some risk factors have been identified, such as increased knee laxity and performance on hop for distance tests, no matter what testing is done there are still athletes who suffer a further injury without the presence of risk factors and don’t fit into a ‘high-risk classification’. Therefore, although some athletes may be able to be identified who go onto have a second injury, the accuracy of the prediction is low, and many athletes who are considered to be at low risk also sustain further injuries. Therefore, despite the increasing popularity surrounding RTS testing, there are many questions that remain unanswered. Until further research data are available, caution should be exercised when using information from RTS testing to provide advice to patients—particularly at an individual patient level—regarding their risk for subsequent injury, if or when they choose to return to sport.
9.6.2 The Psychological Aspect
While much emphasis has been placed on tests for physical function and strength, as previously noted, the importance of addressing psychological factors has been recognized. Although there has been much less empirical work in this area, what is available shows promise. Two initial pilot studies showed that fear of reinjury was associated with a second ACL injury. In the first, Tagesson and Kvist [73] conducted a battery of assessments in a cohort of 19 patients before surgery and at 5 weeks after ACL reconstruction which included; fear of reinjury, knee confidence, patient-reported function, activity levels, static and dynamic tibial translation and muscle strength. They followed the group for 5 years and found that those who went on to have a second ACL injury had greater fear of reinjury and greater static tibial translation in both knees compared to those who remained uninjured. In the second pilot study, Paterno et al. [74] assessed 40 patients who had been cleared to return to sport and tracked them for 12 months to identify second ACL injuries. Patients with a greater fear of injury were 13 times more likely to suffer a second ACL injury. These authors suggested that it may be important to measure self-reported fear of movement and incorporate this into return to sport discharge criteria to reduce the risk of further ACL injury.
In a subsequent larger study, McPherson et al. [75] had 429 athletes complete the anterior cruciate ligament return to sport after injury (ACL-RSI) scale, a measure of psychological readiness for returning to sport, both before and at 12 months after ACL reconstruction, and then followed the cohort for a minimum 2 years to determine further injury. Given that most ACL injuries occur in the context of sport, only those who had made a return to sport were included in the final analysed cohort of 329 patients. For this group, when measured at 12 months, younger injured patients (≤20 years) had significantly lower psychological readiness than younger non-injured patients. A follow-up study in the same young cohort showed that those who re-injured had little improvement in ACL-RSI scores from preoperative to 12 months post-surgery, whereas those not injured showed a 20-point increase [76]. While this work needs further validation and replication, it would appear prudent to consider a psychological aspect alongside any physical return to sport assessment.
9.7 Conclusions
Many athletes do not return to their prior level of sport following ACL reconstruction surgery. Of those that do, a proportion will suffer a second ACL injury and younger patients who return to strenuous sports are at high risk of this. Criteria-based return to sport rehabilitation has been shown to be of significant benefit and should be preferred over solely time-based rehabilitation. Despite much interest in return to sport testing there is currently insufficient evidence that it can be used to provide advice to individual patients regarding their risk for further ACL injury.
ACL rupture and reconstruction should not be a career-ending injury; however, there are considerable and serious obstacles to overcome during rehabilitation and prior to return to sport. Surgical management with appropriate early physical rehabilitation can bring an athlete back to baseline functional level which allows the athlete to safely return to competitive play. However, the neuromotor imbalances of trunk, ligament, quadriceps, leg dominance may continue to exist and be observable and longitudinally measurable during dynamic sports manoeuvres out to and even beyond 2 years in competitive athletes if not appropriately assessed and treated. Therefore, additional focus on longitudinal measurement and maximization of strength along with minimization of the aforementioned neuromotor imbalances is necessary to ensure successful rehabilitative management and return to sport following ACL reconstruction of the young, highly active high-risk competitive athlete.
References
Ardern CL, Webster KE, Taylor NF, Feller JA. Return to sport following anterior cruciate ligament reconstruction surgery: a systematic review and meta-analysis of the state of play. Br J Sports Med. 2011;45(7):596–606.
Ardern CL, Taylor NF, Feller JA, Webster KE. Fifty-five per cent return to competitive sport following anterior cruciate ligament reconstruction surgery: an updated systematic review and meta-analysis incluing aspects of physcial functioning and contextual factors. Br J Sports Med. 2014;48(21):1543–52.
Grassi A, Zaffagnini S, Marcheggiani Muccioli GM, Neri MP, Della Villa S, Marcacci M. After revision anterior cruciate ligament reconstruction, who returns to sport? A systematic review and meta-analysis. Br J Sports Med. 2015;49(20):1295–304.
Lai CCH, Ardern CL, Feller JA, Webster KE. Eighty-three per cent of elite athletes return to preinjury sport after anterior cruciate ligament reconstruction: a systematic review with meta-analysis of return to sport rates, graft rupture rates and performance outcomes. Br J Sports Med. 2018;52(2):128–38.
Kay J, Memon M, Marx RG, Peterson D, Simunovic N, Ayeni OR. Over 90% of children and adolescents return to sport after anterior cruciate ligament reconstruction: a systematic review and meta-analysis. Knee Surg Sports Traumatol Arthrosc. 2018;26(4):1019–36.
Feller JA, Webster KE. Return to sport following anterior cruciate ligament reconstruction. Int Orthop. 2013;37(2):285–90.
Webster KE, Feller JA, Leigh W, Richmond A. Younger patients are at increased risk for graft rupture and contralateral injury after anterior cruciate ligament reconstruction. Am J Sports Med. 2014;42(3):641–7.
Ardern CL, Taylor NF, Feller JA, Whitehead TS, Webster KE. Psychological responses matter in returning to preinjury level of sport after anterior cruciate ligament reconstruction surgery. Am J Sports Med. 2013;41(7):1549–58.
Kvist J, Ek A, Sporrstedt K, Good L. Fear of re-injury: a hindrance for returning to sports after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2005;13(5):393–7.
Langford JL, Webster KE, Feller JA. A prospective longitudinal study to assess psychological changes following anterior cruciate ligament reconstruction surgery. Br J Sports Med. 2009;43(5):377–81.
Webster KE, Feller JA, Lambros C. Development and preliminary validation of a scale to measure the psychological impact of returning to sport following anterior cruciate ligament reconstruction surgery. Phys Ther Sport. 2008;9(1):9–15.
Webster KE, Feller JA. Expectations for return to Preinjury sport before and after anterior cruciate ligament reconstruction. Am J Sports Med. 2019;47(3):578–83.
Feucht MJ, Cotic M, Saier T, Minzlaff P, Plath JE, Imhoff AB, et al. Patient expectations of primary and revision anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2016;24(1):201–7.
Bourke HE, Gordon DJ, Salmon LJ, Waller A, Linklater J, Pinczewski LA. The outcome at 15 years of endoscopic anterior cruciate ligament reconstruction using hamstring tendon autograft for ‘isolated’ anterior cruciate ligament rupture. J Bone Joint Surg. 2012;94B(5):630–7.
Shelbourne KD, Gray T, Haro M. Incidence of subsquent injury to either knee within 5 years after anterior cruciate ligament reconstruction with patellar tendon autograft. Am J Sports Med. 2009;37(2):246–51.
Webster KE, Feller JA. Exploring the high Reinjury rate in younger patients undergoing anterior cruciate ligament reconstruction. Am J Sports Med. 2016;44(11):2827–32.
Maletis GB, Inacio MC, Funahashi TT. Risk factors associated with revision and contralateral anterior cruciate ligament reconstructions in the Kaiser Permanente ACLR registry. Am J Sports Med. 2015;43(3):641–7.
Snaebjornsson T, Hamrin Senorski E, Sundemo D, Svantesson E, Westin O, Musahl V, et al. Adolescents and female patients are at increased risk for contralateral anterior cruciate ligament reconstruction: a cohort study from the Swedish National Knee Ligament Register based on 17,682 patients. Knee Surg Sports Traumatol Arthrosc. 2017;25(12):3938–44.
Wiggins AJ, Grandhi RK, Schneider DK, Stanfield D, Webster KE, Myer GD. Risk of secondary injury in younger athletes after anterior cruciate ligament reconstruction: a systematic review and meta-analysis. Am J Sports Med. 2016;44(7):1861–76.
Morgan MD, Salmon LJ, Waller A, Roe JP, Pinczewski LA. Fifteen-year survival of endoscopic anterior cruciate ligament reconstruction in patients aged 18 years and younger. Am J Sports Med. 2016;44(2):384–92.
Paterno MV, Rauh MJ, Schmitt LC, Ford KR, Hewett TE. Incidence of second ACL injuries 2 years after primary ACL reconstruction and return to sport. Am J Sports Med. 2014;42(7):1567–73.
Dekker TJ, Godin JA, Dale KM, Garrett WE, Taylor DC, Riboh JC. Return to sport after pediatric anterior cruciate ligament reconstruction and its effect on subsequent anterior cruciate ligament injury. J Bone Joint Surg Am. 2017;99(11):897–904.
Grindem H, Engebretsen L, Axe M, Snyder-Mackler L, Risberg MA. Activity and functional readiness, not age, are the critical factors for second anterior cruciate ligament injury - the Delaware-Oslo ACL cohort study. Br J Sports Med. 2020; https://doi.org/10.1136/bjsports-2019-100623.
Webster KE, Feller JA. A research update on the state of play for return to sport after anterior cruciate ligament reconstruction. J Orthop Traumatol. 2019;20(1):10.
Webster KE, Feller JA, Whitehead TS, Myer GD, Merory PB. Return to sport in the younger patient with anterior cruciate ligament reconstruction. Orthop J Sports Med. 2017;5(4):2325967117703399.
Beischer S, Hamrin Senorski E, Thomee C, Samuelsson K, Thomee R. Young athletes return too early to knee-strenuous sport, without acceptable knee function after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2018;26(7):1966–74.
Hamrin Senorski E, Svantesson E, Beischer S, Thomee C, Thomee R, Karlsson J, et al. Low 1-year return-to-sport rate after anterior cruciate ligament reconstruction regardless of patient and surgical factors: a prospective cohort study of 272 patients. Am J Sports Med. 2018;46(7):1551–8.
Toole AR, Ithurburn MP, Rauh MJ, Hewett TE, Paterno MV, Schmitt LC. Young athletes cleared for sports participation after anterior cruciate ligament reconstruction: how many actually meet recommended return-to-sport criterion cutoffs? J Orthop Sports Phys Ther. 2017;47(11):825–33.
Scheffler SU, Unterhauser FN, Weiler A. Graft remodeling and ligamentization after cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2008;16(9):834–42.
Claes S, Verdonk P, Forsyth R, Bellemans J. The “ligamentization” process in anterior cruciate ligament reconstruction: what happens to the human graft? A systematic review of the literature. Am J Sports Med. 2011;39:2476–83.
Pauzenberger L, Syre S, Schurz M. “Ligamentization” in hamstring tendon grafts after anterior cruciate ligament reconstruction: a systematic review of the literature and a glimpse into the future. Arthroscopy. 2013;29(10):1712–21.
Nagelli CV, Hewett TE. Should return to sport be delayed until 2 years after anterior cruciate ligament reconstruction? Biol Funct Considerat Sports Med. 2017;47(2):221–32.
Hewett TE, Ford KR, Hoogenboom BJ, Myer GD. Understanding and preventing acl injuries: current biomechanical and epidemiologic considerations. 2010. N Am J Sports Phys Ther. 2010;5(4):234–51.
Laskowski ER, Newcomer-Aney K, Smith J. Refining rehabilitation with proprioceptive training: expediting return to play. Phys Sportsmed. 1997;25:89–102.
Lephart SM, Fu FH. Proprioception and neuromuscular control in joint stability. Champaign, IL: Human Kinetics; 2000. p. 439.
Griffin LY, Agel J, Albohm MJ, Arendt EA, Dick RW, Garrett WE, et al. Noncontact anterior cruciate ligament injuries: risk factors and prevention strategies. J Am Acad Orthop Surg. 2000;8(3):141–50.
Hewett TE, Paterno MV, Myer GD. Strategies for enhancing proprioception and neuromuscular control of the knee. Clin Orthop Relat Res. 2002;402:76–94.
Paterno MV, Schmitt LC, Ford KR, Rauh MJ, Myer GD, Huang B, et al. Biomechanical measures during landing and postural stability predict second anterior cruciate ligament injury after anterior cruciate ligament reconstruction and return to sport. Am J Sports Med. 2010;38(10):1968–78.
Tropp H, Odenrick P. Postural control in single-limb stance. J Orthop Res. 1988;6(6):833–9.
Caraffa A, Cerulli G, Projetti M, Aisa G, Rizzo A. Prevention of anterior cruciate ligament injuries in soccer. A prospective controlled study of proprioceptive training. Knee Surg Sports Traumatol Arthrosc. 1996;4(1):19–21.
Tropp H, Ekstrand J, Gillquist J. Factors affecting stabilometry recordings of single limb stance. Am J Sports Med. 1984;12(3):185–8.
Hewett TE, Stroupe AL, Nance TA, Noyes FR. Plyometric training in female athletes. Decreased impact forces and increased hamstring torques. Am J Sports Med. 1996;24(6):765–73.
Ford KR, Myer GD, Hewett TE. Valgus knee motion during landing in high school female and male basketball players. Med Sci Sports Exerc. 2003;35(10):1745–50.
Knapik JJ, Bauman CL, Jones BH, Harris JM, Vaughan L. Preseason strength and flexibility imbalances associated with athletic injuries in female collegiate athletes. Am J Sports Med. 1991;19(1):76–81.
Baumhauer J, Alosa D, Renstrom A, Trevino S, Beynnon B. A prospective study of ankle injury risk factors. Am J Sports Med. 1995;23(5):564–70.
Hewett TE, Myer GD, Ford KR, Heidt RS Jr, Colosimo AJ, McLean SG, et al. Neuromuscular control and valgus loading of the knee predict ACL injury risk in female athletes. Am J Sports Med. 2005;33(4):492–501.
Myer GD, Paterno MV, Ford KR, Hewett TE. Neuromuscular training techniques to target deficits before return to sport after anterior cruciate ligament reconstruction. J Strength Cond Res. 2008;22(3):987–1014.
Myer GD, Paterno MV, Ford KR, Quatman CE, Hewett TE. Rehabilitation after anterior cruciate ligament reconstruction: criteria-based progression through the return-to-sport phase. J Orthop Sports Phys Ther. 2006;36(6):385–402.
Lloyd DG, Buchanan TS. Strategies of muscular support of varus and valgus isometric loads at the human knee. J Biomech. 2001;34(10):1257–67.
Lloyd DG. Rationale for training programs to reduce anterior cruciate ligament injuries in Australian football. J Orthop Sports Phys Ther. 2001;31(11):645–54.
Cahill BR, Griffith EH. Effect of preseason conditioning on the incidence and severity of high school football knee injuries. Am J Sports Med. 1978;6(4):180–4.
Hewett TE, Riccobene JV, Lindenfeld TN, Noyes FR. The effect of neuromuscular training on the incidence of knee injury in female athletes: a prospective study. Am J Sports Med. 1999;27(6):699–706.
Malinzak RA, Colby SM, Kirkendall DT, Yu B, Garrett WE. A comparison of knee joint motion patterns between men and women in selected athletic tasks. Clin Biomech. 2001;16(5):438–45.
Besier TF, Lloyd DG, Ackland TR, Cochrane JL. Anticipatory effects on knee joint loading during running and cutting maneuvers. Med Sci Sports Exerc. 2001;33(7):1176–81.
Myer GD, Ford KR, Palumbo JP, Hewett TE. Neuromuscular training improves performance and lower-extremity biomechanics in female athletes. J Strength Cond Res. 2005;19(1):51–60.
Neptune RR, Wright IC, van den Bogert AJ. Muscle coordination and function during cutting movements. Med Sci Sports Exerc. 1999;31(2):294–302.
van Melick N, van Cingel RE, Brooijmans F, Neeter C, van Tienen T, Hullegie W, et al. Evidence-based clinical practice update: practice guidelines for anterior cruciate ligament rehabilitation based on a systematic review and multidisciplinary consensus. Br J Sports Med. 2016;50(24):1506–15.
Ardern CL, Glasgow P, Schneiders A, Witvrouw E, Clarsen B, Cools A, et al. Consensus statement on return to sport from the First World Congress in Sports Physical Therapy, Bern. Br J Sports Med. 2016;50:14853–64.
Panariello R, Stump T, Allen A. Rehabilitation and return to play following anterior cruciate ligament reconstruction. Oper Tech Sports Med. 2017;26:181–93.
Ellman MB, Sherman SL, Forsythe B, LaPrade RF, Cole BJ, Bach BR Jr. Return to play following anterior cruciate ligament reconstruction. J Am Acad Orthop Surg. 2015;23(5):283–96.
Hewett TE, Webster KE, Hurd WJ. Systematic selection of key logistic regression variables for risk prediction analyses: a five-factor maximum model. Clin J Sport Med. 2019;29(1):78–85.
Feller JA, Webster KE. Where are we with return-to-sport testing following ACL reconstruction? Orthop Traumatol Surg Res. 2019;105(6):1037–8.
Nawasreh Z, Logerstedt D, Cummer K, Axe M, Risberg MA, Snyder-Mackler L. Functional performance 6 months after ACL reconstruction can predict return to participation in the same preinjury activity level 12 and 24 months after surgery. Br J Sports Med. 2018;52(6):375.
Webster KE, Hewett TE. What is the evidence for and validity of return-to-sport testing after anterior cruciate ligament reconstruction surgery? A systematic review and meta-analysis. Sports Med. 2019;49(6):917–29.
Grindem H, Snyder-Mackler L, Moksnes H, Engebretsen L, Risberg MA. Simple decision rules can reduce reinjury risk by 84% after ACL reconstruction: the Delaware-Oslo ACL cohort study. Br J Sports Med. 2016;50(13):804–8.
Losciale JM, Zdeb RM, Ledbetter L, Reiman MP, Sell TC. The association between passing return-to-sport criteria and second anterior cruciate ligament injury risk: a systematic review with meta-analysis. J Orthop Sports Phys Ther. 2019;49(2):43–54.
Kyritsis P, Bahr R, Landreau P, Miladi R, Witvrouw E. Likelihood of ACL graft rupture: not meeting six clinical discharge criteria before return to sport is associated with a four times greater risk of rupture. Br J Sports Med. 2016;50(15):946–51.
Sousa PL, Krych AJ, Cates RA, Levy BA, Stuart MJ, Dahm DL. Return to sport: does excellent 6-month strength and function following ACL reconstruction predict midterm outcomes? Knee Surg Sports Traumatol Arthrosc. 2017;25(5):1356–63.
Webster KE, Hewett TE. Return-to-sport testing following ACL reconstruction revisited. Br J Sports Med. 2020;54(1):2–3.
Bodkin SG, Rutherford MH, Diduch DR, Brockmeier SF, Hart JM. How much time is needed between serial “return to play” assessments to achieve clinically important strength gains in patients recovering from anterior cruciate ligament reconstruction? Am J Sports Med. 2020;48(1):70–7.
Paterno MV, Huang B, Thomas S, Hewett TE, Schmitt LC. Clinical factors that predict a second ACL injury after ACL reconstruction and return to sport: preliminary development of a clinical decision algorithm. Orthop J Sports Med. 2017;5(12):2325967117745279.
Webster KE, Feller JA. Clinical tests can be used to screen for second anterior cruciate ligament injury in younger patients who return to sport. Orthop J Sports Med. 2019;7(8):2325967119863003.
Tagesson S, Kvist J. Greater fear of re-injury and increased tibial translation in patients who later sustain an ACL graft rupture or a contralateral ACL rupture: a pilot study. J Sports Sci. 2016;34(2):125–32.
Paterno MV, Flynn K, Thomas S, Schmitt LC. Self-reported fear predicts functional performance and second ACL injury after ACL reconstruction and return to sport: a pilot study. Sports Health. 2018;10(3):228–33.
McPherson AL, Feller JA, Hewett TE, Webster KE. Psychological readiness to return to sport is associated with second anterior cruciate ligament injuries. Am J Sports Med. 2019;47(4):857–62.
McPherson AL, Feller JA, Hewett TE, Webster KE. Smaller s. Am J Sports Med. 2019;47(5):1209–15.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 ISAKOS
About this chapter
Cite this chapter
Webster, K.E., Hewett, T.E. (2022). Return to Sport After Anterior Cruciate Ligament Reconstruction: Criteria-Based Rehabilitation and Return to Sport Testing. In: Nakamura, N., Marx, R.G., Musahl, V., Getgood, A., Sherman, S.L., Verdonk, P. (eds) Advances in Knee Ligament and Knee Preservation Surgery. Springer, Cham. https://doi.org/10.1007/978-3-030-84748-7_9
Download citation
DOI: https://doi.org/10.1007/978-3-030-84748-7_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-84747-0
Online ISBN: 978-3-030-84748-7
eBook Packages: MedicineMedicine (R0)