Abstract
Background
Anterior cruciate ligament (ACL) return-to-sport (RTS) test batteries are popular and are employed to test athletes’ sport performance and help ensure a safe return to sport.
Objective
To perform a systematic review and meta-analysis to determine: (1) the proportion of patients who passed RTS test batteries after ACL reconstruction, (2) whether passing RTS test batteries increased rates of return to play, and (3) whether passing RTS test batteries reduced subsequent rates of knee and ACL injury.
Methods
Five databases (PubMed, MEDLINE, Embase, CINAHL, and SPORTDiscus) were searched to identify relevant studies and data were extracted regarding the number of patients who passed the RTS test battery, as well as subsequent RTS rates and re-injury data when available. Results were combined using proportional and risk-ratio meta-analyses.
Results
Eighteen studies met eligibility criteria. Proportional meta-analysis showed that only 23% of patients passed RTS test batteries. One study showed that passing an RTS test battery led to greater RTS rates. Two studies showed passing RTS test batteries did not significantly reduce the risk of a further knee injury (risk ratio (RR) = 0.28 (95% CI 0.04–0.94), p = 0.09) and five studies showed that passing RTS test batteries did not reduce the risk for all subsequent ACL injuries (RR = 0.80 (95% CI 0.27–2.3), p = 0.7). However, passing an RTS test battery did significantly reduce the risk for subsequent graft rupture (RR = 0.40 (95% CI 0.23–0.69), p < 0.001], although it increased the risk for a subsequent contralateral ACL injury (RR = 3.35 (95% CI 1.52–7.37), p = 0.003].
Conclusion
These analyses shows that there are equivocal findings in terms of the validity of current RTS test batteries in relation to reduction of the risk of graft rupture and contralateral ACL injuries. These findings have implications for RTS advice given to patients based on the results of RTS test batteries, and further work is needed to validate the criteria currently used and determine the true value.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Current return-to-sport criteria do not appear to decrease the risk of subsequent anterior cruciate ligament (ACL) injury in athletes. |
Though passing return-to-sport criteria reduced the risk of subsequent graft rupture by 60%, it increased the risk of a contralateral ACL rupture by 235%. |
1 Introduction
Most athletes who undergo anterior cruciate ligament (ACL) reconstruction surgery plan to return to their pre-injury level of sport [1]. However, only approximately one-half at 1 year and two-thirds at 2 years achieve this goal and those who return have a high risk for further ACL injury [2, 3]. The rates of second ACL injury are highest in younger athletes [4,5,6,7,8], in whom rates of up to 35% have been reported [9].
There has consequently been marked interest and a rapid growth in studies that propose return-to-sport (RTS) criteria to reduce the risk of a second ACL injury. Typically, these are a set of criteria or “test battery” that is used to clear the athlete for return to sport at the final stage of rehabilitation [10]. Whilst the specific content of reported RTS test batteries has varied, overall, they are designed to incorporate a number of domains of risk factors. A systematic review and multidisciplinary consensus indicated that an RTS test battery should at least include a series of strength tests, hop tests and measures of quality of movement [11]. Thus, studies have attempted to cover a broad range of risk factors that has resulted in the inclusion of up to 15–20 different RTS tests [12, 13], which few patients pass [14, 15] and the validity of many of the included tests is unknown [16].
The true value of any RTS test battery is its ability to assess whether patients have returned to their prior level of sport at a high-performance level whilst also reducing the risk for a second ACL injury. There is no current systematic review or meta-analysis that has determined what proportion of patients pass RTS test batteries or whether passing an RTS test battery indeed reduces the risk for subsequent knee or second ACL injury. Thus, this review sought to answer three questions: (1) What proportion of patients pass RTS test batteries after ACL reconstruction? (2) Is passing RTS test batteries associated with increased rates of return to play? and 3) Is passing RTS test batteries associated with reduced rates of subsequent knee injury (all knee injuries and ACL injury)?
2 Methods
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [17] were used in preparing, conducting and reporting this systematic review.
2.1 Search Strategy
The electronic databases PubMed, MEDLINE, Embase, CINAHL, and SPORTDiscus were searched from the earliest possible dates through to 7 May 2018. Search terms were entered under two concepts; terms within each concept were combined with the OR Boolean operator, and the two concepts were combined with the AND Boolean operator. Where possible, terms were mapped to medical subject headings (MeSH) and searched using keywords; wildcards were also used. Examples of terms included in Concept 1 included ‘anterior cruciate ligament reconstruction’, and ‘ACL reconstruction’. Examples of terms included in Concept 2 included ‘return to sport’, ‘return to sport criteria’, ‘return to play’, ‘return to play criteria’, ‘functional testing’ and ‘return to athletic*’.
To supplement the electronic database search, the online contents pages and ‘articles in press’ lists of leading sports medicine journals were hand searched, and studies on the reference lists of the included studies were screened. Publication details from all identified studies in the literature search were exported to bibliographic software and duplicates removed.
2.2 Selection Criteria
Studies were included if they were: (1) published in the English language, (2) included participants who had undergone ACL reconstruction (primary or revision) surgery, (3) utilized a return-to-sport (RTS) test battery, and (4) reported the number of participants who passed the test battery or were cleared for return based on test battery results. There was no minimum number of tests that was required to be included in a test battery and the test could be from any domain; however, multiple domains needed to be represented. For example, studies that only measured strength (even if there were multiple strength tests used) or only measured function (i.e. only used hop tests) were excluded. Studies that only included the number of participants who passed single components of a test battery and not the full test battery were also excluded if the authors could not provide data for the full test battery. Similarly, studies that only included a cohort of patients who were selected because they had either failed or passed criteria were excluded. For studies that reported cut-off values for passing the RTS test battery they used there were no restrictions placed on what the actual cut-off value had to be. Conference proceedings, case studies, clinical commentaries and review studies were excluded.
The titles and abstracts were first screened for eligibility and those that did not meet the inclusion criteria, or had at least one exclusion criterion, were excluded. The full text versions of the remaining studies were retrieved, and the selection criteria were applied by two reviewers with any discrepancies discussed until consensus was reached.
2.3 Quality Assessment (Risk of Bias)
Included studies were assessed for methodological quality based on both the Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies and the Quality Assessment Tool for Case-Series Studies from the National Institutes of Health [18]. This was appropriate as the primary aims were to determine the proportion of patients passing RTS test batteries and whether passing an RTS test battery was related to return to sport or further knee injury. Therefore, there was no intervention or exposure. Items 1–5 from the Observational Cohort and Cross-Sectional Studies tool were combined with Items 5–9 from the Case-Series checklist to produce a 10-item list (Electronic Supplementary Material Table S1). The purpose of the assessment was to identify characteristics of study quality and design for all included studies and to provide a qualitative description of the study characteristics. Item 6 (Was the intervention described?) was assessed in reference to the RTS test battery. For studies that only reported percentage of participants who passed RTS criteria items 7 (Were outcome measures clearly defined?) and 8 (Was length of follow-up adequate?) were not applicable. Studies were assessed independently by two reviewers.
2.4 Data Extraction and Synthesis
Data from each of the studies were extracted using a standard form. For all studies, the number of patients who passed the RTS test battery was recorded. Pass rates were calculated from the number of patients who passed, out of the total number of patients, and were expressed as a percentage. Pooled pass rates were calculated using random-effects proportion meta-analyses (StatsDirect medical statistics software, Version 2.8, Cambridge, UK). This analysis was performed according to: (1) the proportion of patients who passed RTS criteria before return to sport and (2) the proportion of patients who passed RTS criteria after return to strenuous sports. Studies in which patients could not be classified as having returned to sport or not when the RTS testing was conducted were not included in the meta-analyses. Several studies with potential patient overlap were identified (i.e. same institution, same RTS test battery). For these studies, contact was made with the authors, and for studies in which 50% or more patient group overlap was identified, only the study with the largest number of patients was used for the meta-analysis.
Where available, the number of patients who passed RTS criteria and subsequently returned to sport was recorded along with the number of patients who had failed criteria but later returned to sport. Similarly, the number of patients who passed RTS criteria and sustained a subsequent knee and/or ACL injury was recorded along with the number of patients who failed RTS criteria and sustained a subsequent knee and/or ACL injury. These data were analysed with risk-ratio (RR) meta-analyses (RevMan V5.3; Copenhagen: The Nordic Cochrane Center, The Cochrane Collaboration, 2014). Separate analyses were conducted for all subsequent knee injuries and for ACL injuries. An RR value of less than 1 indicated reduced risk of subsequent injury if the patient passed RTS criteria.
3 Results
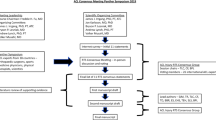
The electronic database search identified 3771 studies; eight were identified from the manual search of reference lists and relevant journals. After 2002 duplicates were removed, 1777 articles remained as the total yield. From reviewing titles and abstracts, 1664 studies were excluded, and the full text of the remaining 113 were downloaded for detailed assessment. Of these 95 were excluded (refer to Fig. 1 and Electronic Supplementary Material Table S2), 18 studies were included for qualitative analysis and 17 studies were included in one or more of the meta-analyses. The search process is described in Fig. 1.
The characteristics of the included studies are detailed in Table 1. Several studies were identified from the same institution and contact was made with the authors of these studies to identify any patient overlap. Krych et al. [19] and Souza et al. [20] were identified as the same patient population and were therefore not included in the same meta-analysis. Four studies from the Delaware-Oslo group (Logerstedt et al. [21], Grindem et al. [22], Nawasreh et al. [23] and Wellsandt et al. [24]) were identified as having a minimum of 50% patient overlap and were also not included in the same meta-analysis. Finally, Gokeler et al. [14] and Welling et al. [25] also had more than 50% patient overlap and were not included in the same meta-analysis.
A variety of RTS test batteries was used (Table 1), with the most common elements being quadriceps strength and hop tests for function. A limb symmetry index of ≥ 90 was the most common pass cut-off used. Some studies varied the cut-off according to the type of test or the level of sport that the patient aimed to return to [14, 25, 26]. Of the 18 studies, only five studies [20, 22, 24, 27, 28] had further injury data, and only one [23] assessed whether passing RTS criteria was associated with subsequent return to play. Grindem et al. [22] were contacted and provided subsequent ACL injury data as these raw data were not included in the published manuscript (only raw data for overall knee injuries were provided in the published paper). The most common time for RTS assessment was 6 months post-surgery.
3.1 Quality Assessment
Ten of the 18 studies adequately described the study population in terms of patient selection, age, sex, sports played and graft type used for the reconstruction surgery [19,20,21,22,23,24,25,26, 28, 29]. Only three studies included any form of sample size or power calculation [20, 30, 31]. Three studies did not report the individual elements and cut-off used for the RTS test battery in a way that could be easily replicated [19, 27, 29]. Three of the five studies that had injury data had detailed adequate follow-up of a minimum 2 years post-surgery [20, 22, 24] (see Electronic Supplementary Material Table S1 for assessment ratings).
3.2 Proportion of Patients Who Pass Return-to-Sport (RTS) Test Batteries Before Return to Sport
From eight studies with 876 patients, a proportional meta-analysis showed that 23% (95% CI 8–43%, I2 = 97.5%) passed RTS test batteries before return to sport (Fig. 2). There was heterogeneity amongst studies with pass rates that ranged from 0 to 79%. All studies that were included in the meta-analysis tested patients between 5 and 10 months post-surgery. If a study also tested patients at a later time point (i.e. 12 months) only the earlier time point was used in the analysis. For Herbst et al. [15] the pass rates for return to non-competitive sport were used and for Thomee et al. [33] data for the > 90 LSI cut-off was used as this was most consistent with the other papers.
3.3 RTS Test Battery Pass Rates in Patients Who Have Returned to Strenuous Sports
From three studies with 234 patients, a proportional meta-analysis showed that 23% (95% CI 18–29%, I2 = 0%) passed RTS test batteries despite having already returned to strenuous sports (Fig. 3).
3.4 Passing RTS Test Batteries and Rates of RTS
Only one study determined whether passing an RTS test battery was related to a subsequent return to sport. Nawasreh et al. [23] showed that patients who passed RTS criteria at 6 months were significantly more likely to have returned to play at both 12 and 24 months post-surgery. Specifically, in the group who had passed RTS testing, over 80% of that group had returned to sport at 12 months, whereas only 44% of the group who had not passed had returned at the same time. Sousa et al. [20] showed that there was no difference in the proportion of patients who met or exceeded their pre-injury Tegner score at a minimum 2-year follow-up, as 51% of the pass group and 52% of the fail group achieved this.
3.5 Passing RTS Test Batteries and Re-injury
Five studies that utilized test batteries reported subsequent re-injury rates; two of these reported any subsequent knee injury in 114 patients and five reported ACL injuries in 565 patients (four studies [20, 22, 24, 27] reported all second ACL injuries and one [28] reported only graft ruptures). For all knee injuries, there was no significant reduction in risk for those who passed RTS criteria (RR = 0.28 (95% CI 0.04–0.94), p = 0.09; I2 = 13%; Fig. 4).
Meta-analysis showing the risks for subsequent knee injury after passing an RTS test battery. The risk ratio and 95% CI data from individual studies in addition to the pooled data are shown. RR < 1 indicates a reduced risk of re-injury if the patient passes an RTS test battery. RTS return to sport, M-H Mantel-Haenszel, RR risk ratio
Passing an RTS test battery had minimal effect on reduction of the risk of all subsequent ACL injuries (RR =0.80 (95% CI 0.27–2.3), p = 0.7; I2 = 79%; Fig. 5). However, for graft ruptures, those who passed a test battery had a significantly reduced risk of a subsequent graft rupture (RR=0.40 (95% CI 0.23–0.69), p < 0.001; I2 = 0%; Fig. 6), whereas for contralateral ACL injury, passing the test battery significantly increased the risk of a subsequent contralateral ACL injury (RR=3.35 (95% CI 1.52–7.37), p = 0.003; I2 = 0%; Fig. 7). Note that one patient in Graziano et al. [27] sustained a graft rupture at 3 months from an accidental playground injury and was excluded from all meta-analyses as they did not complete RTS testing.
Meta-analysis showing the risk for ACL injury after passing an RTS test battery. The risk ratio and 95% CI data from individual studies in addition to the pooled data are shown. RR < 1 indicates a reduced risk of injury if the patient passes an RTS test battery. RTS return to sport, M-H Mantel-Haenszel, RR risk ratio
Meta-analysis showing the risk for graft rupture after passing an RTS test battery. The risk ratio and 95% CI data from individual studies in addition to the pooled data are shown. RR < 1 indicates a reduced risk of graft rupture if the patient passes an RTS test battery. RTS return to sport; M-H Mantel-Haenszel, RR risk ratio
Meta-analysis showing the risk for contralateral ACL injury after passing an RTS test battery. The risk ratio and 95% CI data from individual studies in addition to the pooled data are shown. RR > 1 indicates an increased risk of contralateral ACL injury if the patient passes an RTS test battery. RTS return to sport, M-H Mantel-Haenszel, RR risk ratio
4 Discussion
This study summarizes the current evidence for the RTS test batteries that are frequently used to clear patients for a return to sport after ACL reconstruction surgery. Overall it was found that few patients passed test batteries and that there was limited evidence that passing an RTS test battery reduced the risk for any subsequent knee injury as well as all ACL injures. Interestingly, passing an RTS test battery was shown to significantly reduce the risk of subsequent graft rupture; however, passing RTS criteria also increased the risk of a contralateral ACL injury.
Whilst there was noted variation between studies, overall the proportion of patients who passed RTS test batteries was a low 23%. RTS testing was typically conducted between 5 and 10 months post-surgery, with the most common time being 6 months. Only one study had a pass rate above 50% [29]. For this study, in which the pass rate was 76%, the patient cohort were all 18 years and younger at surgery [29]. This is consistent with other recent research that has shown that younger patients have significantly higher pass rates for hop tests and other clinical outcomes than older patients [35]. However, the pass rate in another study that was included in the review, with a similar young athlete population, was only 14% [34]. One study in elite level athletes also had a high pass rate (73%) but was not included in the meta-analysis as it was unclear whether the RTS testing was conducted before all athletes had returned to play [28].
One problem with these test batteries is the ‘penalty’ of multiple tests [34]. With a test battery, multiple tests across a number of domains are required to be passed at a required pass rate, which was most often set at 90%. If athletes meet the pass rate for one test and a second test with a 90% pass requirement is added, the percentage of athletes who pass will almost certainly drop [34]. For example, even if 80% of athletes pass each test of a test battery, the overall pass rate for the test battery will be dependent on the total number of tests such that the pass rate for the first test will be 80%, but then only 64% (0.8 × 0.8) for two tests, 51% (0.64 × 0.8) for three, 40% (0.5 × 0.8) for four, and so on. Fortunately, this problem is correctable. Testing should be administered at multiple time points and once an athlete passes a test of the battery, that test pass requirement may be able to be dropped from the battery requirement. However, caution should still be exercised as athletes who pass a criterion at one time point may fail it at another [11].
Even when RTS testing was conducted over a longer time frame (1–2 years) [21] and once the patient had returned to sport [26, 30, 32], the pass rate was still only 23%. This is of concern from an injury prevention perspective as it highlights that many patients may have returned without acceptable knee function and control. Beischer et al. [30] showed that only 29% of adolescent (15–20 years) patients achieved a limb symmetry index of > 90% on five tests of muscle function despite having already returned to strenuous sports at 8 months. When these findings are considered in parallel with the high rates of reinjury that have been reported in younger patients, it is reasonable to suggest that poor knee function combined with high exposure was a contributing cause of reinjury in this younger patient group. From a practical point of view, the overall low pass rates should also lead us to question how such tests can be utilized if the majority of patients fail (i.e. the test batteries have large floor effects). A fundamental question is whether RTS test batteries are designed to determine whether the patient is capable of return to play at a specific performance level or are they designed to determine whether return is safe.
In terms of capability of return to play, passing an RTS test battery at 6 months post-surgery was shown to lead to significantly higher RTS rates at both 12 and 24 months in the only study included that investigated the relationship between passing RTS criteria and subsequent return rates [23]. The group that passed had signifiantly more male patients and was overall of younger age. However, Sousa et al. [20] showed that a similar proportion of patients who had passed RTS testing at 6 months compared to those who failed, reached or exceded their pre-injury Tegner activity score at a mid-term follow-up (minimum 2 years). Despite this, the pass group had an overall higher activity level at mid-term and also had significantly better knee function. Therefore these combined results indicate that 6-month RTS test results may be indicative of latter-term function.
With regard to whether RTS testing can determine whether it is safe to return to sport, five studies were included in this review that investigated subsequent injury [20, 22, 24, 27, 28]. Two of these recorded all knee injuries and showed that passing an RTS test battery resulted in an overall 72% reduction in risk for any subsequent knee injury [22, 27]. However, this reduction was not statistically significant, and the risk estimate should be considered imprecise due to a large 95% CI (from 93% reduction in risk to 21% increase in risk), mainly due to the low number of studies and small subject numbers (only 114 patients in total). Nonetheless, as both studies showed the same trend, further research is both encouraged and required to confirm this finding. In these two studies, Grindem et al. [22] noted that patients should wait at least 9 months before return to play as the risk for further knee injury was significantly reduced for each month the athlete delayed return until the 9-month mark. Graziano et al. [27] similarly noted that none of their young patient cohort was ready to return before 9 months.
Passing an RTS test battery led to a significant 60% reduction in risk for graft rupture; however, it also led to a significant 235% increase in risk of contralateral ACL injury compared to not passing the test battery. Therefore, passing a test battery did not change the risk for all subsequent ACL injuries, which is similar to findings of a recent review of four studies that showed a nonsignificant 3% reduction in risk [36].These current findings highlight the need to consider the outcome and rehabilitation of both knees as, for the athlete, any further ACL injury is a devastating outcome. Of the five studies included in this analysis there were two with significant results, and these were the only two studies with a total sample size of more than 100 patients. Kyritsis et al. [28] recorded graft ruptures in elite male athletes and reported that those who did not meet all RTS criteria had a four times greater risk of graft rupture. The hamstring-to-quadriceps ratio of the involved leg alone was also highly associated with graft rupture, with a ten times greater risk for every 10% difference in strength. In comparison, Sousa et al. [20] did not find a reduced risk for graft rupture in their group who passed RTS criteria; they did, however, find a significantly increased risk for contralateral injuries. The authors suggested that this may be related to an increased activity level in their patients as they had been cleared for an earlier return to play. In addition, biomechanical and epidemiological findings demonstrated increased loading of the contralateral limb at the time of return to sport and beyond [37, 38]. This increased loading of the contralateral limb may also account for the increased risk in contralateral injury post-release to return to play.
Whilst this review has shown that RTS test batteries currently have limited validity in the reduction of overall second ACL injury risk, we cannot conclude that they have no benefit. They can be used to provide the patient with important feedback with regard to their rehabilitation progress and may also, for example, boost confidence for when the patient returns to play. However, these test batteries have the potential to decrease confidence as well. Due to the heterogeneity of tests that were conducted in the studies included in this review, as well as those in the wider literature, it is apparent that there is still a high level of uncertainly as to what tests are best to include, the value of any specific test and when they should be used.
4.1 Limitations
There are a number of limitations that need to be considered. There was only one study that assessed whether passing RTS criteria was associated with subsequent return to play, so it is difficult to draw firm conclusions from the data. The definition of RTS has also varied and is not always clearly defined in any one study. A recent consensus statement has attempted to provide a working definition [39], but this was not adopted in any of the included studies. The sample sizes in most of the studies were limited and the inclusion criteria were broad. For example, the age limits typically ranged from 14 to 50 years, some studies had greater than 50% allografts [23, 24], and there was a mixture of the types of sports played. All these factors have been related to second ACL injury and have not been specifically controlled for in the included studies. In addition, no study reported on player exposure, so it is unclear whether the group who passed RTS criteria actually had a higher level of exposure as suggested by Sousa et al. [20], and also whether this affects the risk of subsequent injury. Measurement of exposure is of course a challenging undertaking. Finally, whilst a minimum 2-year follow-up time was used in the studies that reported further injury, and this length of follow-up is considered a strength for capture of all further injuries, it also means that over this time frame factors other than passing an RTS test battery come into play. There has been little discussion in the literature as to what may be an appropriate follow-up time period following RTS testing. For instance, if a patient is reinjured during his/her first couple of games/matches after return to play it would be logical to see if he/she had passed RTS testing or not. However, if a player has made a successful return to play and played for at least two full seasons, it may not be meaningful to relate an injury that occurs after this time point back to an RTS test that occurred many years earlier.
5 Summary and Conclusions
In conclusion, this review showed that less than a quarter (23%) of patients pass RTS test batteries irrespective of whether or not they have already returned to sport, though patients who pass test batteries earlier may have better RTS outcomes in terms of participation. Passing RTS test batteries did not significantly reduce the risk for further knee injuries in general or ACL injuries specifically. A highly interesting finding was observed in terms of graft rupture and contralateral ACL injury, where passing significantly decreased the risk for graft rupture by 60%, but significantly increased the risk for contralateral ACL injury by 235%. Therefore, given these mixed and equivocal results, the information that can be gained from these current RTS test batteries may be hard to apply in clinical practice as there is a high level of uncertainly as to their validity for providing advice to patients regarding their risk for subsequent injury should or when they choose to return to play.
Data availability
All the data in these studies are available in the figures and tables.
References
Feucht MJ, Cotic M, Saier T, Minzlaff P, Plath JE, Imhoff AB, et al. Patient expectations of primary and revision anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2016;24(1):201–7. https://doi.org/10.1007/s00167-014-3364-z.
Ardern CL, Taylor NF, Feller JA, Webster KE. Fifty-five per cent return to competitive sport following anterior cruciate ligament reconstruction surgery: an updated systematic review and meta-analysis including aspects of physical functioning and contextual factors. Br J Sports Med. 2014;48(21):1543–52.
Ardern CL, Webster KE, Taylor NF, Feller JA. Return to sport following anterior cruciate ligament reconstruction surgery: a systematic review and meta-analysis of the state of play. Br J Sports Med. 2011;45(7):596–606.
Dekker TJ, Godin JA, Dale KM, Garrett WE, Taylor DC, Riboh JC. Return to sport after pediatric anterior cruciate ligament reconstruction and its effect on subsequent anterior cruciate ligament injury. J Bone Jt Surg Am. 2017;99(11):897–904.
Morgan MD, Salmon LJ, Waller A, Roe JP, Pinczewski L. Fifteen-year survival of endoscopic anterior cruciate ligament reconstruction in patients aged 18 years and younger. Am J Sports Med. 2016;44(2):384–92.
Paterno MV, Rauh MJ, Schmitt LC, Ford KR, Hewett TE. Incidence of second ACL injuries 2 years after primary ACL reconstruction and return to sport. Am J Sports Med. 2014;42(7):1567–73.
Webster KE, Feller JA, Leigh W, Richmond AK. Younger patients are at increased risk for graft rupture and contralateral injury after anterior cruciate ligament reconstruction. Am J Sports Med. 2014;42(3):641–7.
Wiggins AJ, Grandhi RK, Schneider DK, Stanfield D, Webster KE, Myer GD. Risk of secondary injury in younger athletes after anterior cruciate ligament reconstruction: a systematic review and meta-analysis. Am J Sports Med. 2016;44(7):1861–76. https://doi.org/10.1177/0363546515621554.
Webster KE, Feller JA. Exploring the high reinjury rate in younger patients undergoing anterior cruciate ligament reconstruction. Am J Sports Med. 2016;44(11):2827–32.
Dingenen B, Gokeler A. Optimization of the return-to-sport paradigm after anterior cruciate ligament reconstruction: a critical step back to move forward. Sports Med. 2017;47(8):1487–500.
van Melick N, van Cingel RE, Brooijmans F, Neeter C, van Tienen T, Hullegie W, et al. Evidence-based clinical practice update: practice guidelines for anterior cruciate ligament rehabilitation based on a systematic review and multidisciplinary consensus. Br J Sports Med. 2016;50(24):1506–15.
Ellman MB, Sherman SL, Forsythe B, LaPrade RF, Cole BJ, Bach BR Jr. Return to play following anterior cruciate ligament reconstruction. J Am Acad Orthop Surg. 2015;23(5):283–96.
Panariello RA, Stump TJ, Allen AA. Rehabilitation and return to play following anterior cruciate ligament reconstruction. Oper Tech Sports Med. 2017;25(3):181–93.
Gokeler A, Welling W, Zaffagnini S, Seil R, Padua D. Development of a test battery to enhance safe return to sports after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2017;25(1):192–9.
Herbst E, Hoser C, Hildebrandt C, Raschner C, Hepperger C, Pointner H, et al. Functional assessments for decision-making regarding return to sports following ACL reconstruction. Part II: clinical application of a new test battery. Knee Surg Sports Traumatol Arthrosc. 2015;23(5):1283–91.
Davies GJ, McCarty E, Provencher M, Manske RC. ACL return to sport guidelines and criteria. Curr Rev Musculoskelet Med. 2017;10:307–14.
Moher D, Liberati A, Tetzlaff J, Altman D, the PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151(4):264–9.
Quality assessment tool for observational cohort and cross-sectional studies. 2014. https://www.nhlbi.nih.gov/health-pro/guidelines/in-develop/cardiovascular-risk-reduction/tools/cohort.
Krych A, Woodcock J, Morgan J, Levy B, Stuart M, Dahm D. Factors associated with excellent 6-month functional and isokinetic test results following ACL reconstruction. Knee Surg Sports Traumatol Arthrosc. 2015;23(4):1053–9.
Sousa PL, Krych AJ, Cates RA, Levy BA, Stuart MJ, Dahm DL. Return to sport: Does excellent 6-month strength and function following ACL reconstruction predict midterm outcomes? Knee Surg Sports Traumatol Arthrosc. 2017;25(5):1356–63.
Logerstedt D, Di Stasi S, Grindem H, Lynch A, Eitzen I, Engebretsen L, et al. Self-reported knee function can identify athletes who fail return-to-activity criteria up to 1 year after anterior cruciate ligament reconstruction: a Delaware-Oslo ACL cohort study. J Orthop Sports Phys Ther. 2014;44(12):914–23.
Grindem H, Snyder-Mackler L, Moksnes H, Engebretsen L, Risberg MA. Simple decision rules can reduce reinjury risk by 84% after ACL reconstruction: the Delaware-Oslo ACL cohort study. Br J Sports Med. 2016;50(13):804–8.
Nawasreh Z, Logerstedt D, Cummer K, Axe M, Risberg MA, Snyder-Mackler L. Functional performance 6 months after ACL reconstruction can predict return to participation in the same preinjury activity level 12 and 24 months after surgery. Br J Sports Med. 2018;52(6):375.
Wellsandt E, Failla MJ, Snyder-Mackler L. Limb symmetry indexes can overestimate knee function after anterior cruciate ligament injury. J Orthop Sports Phys Ther. 2017;47(5):334–8.
Welling W, Benjaminse A, Seil R, Lemmink K, Zaffagnini S, Gokeler A. Low rates of patients meeting return to sport criteria 9 months after anterior cruciate ligament reconstruction: a prospective longitudinal study. Knee Surg Sports Traumatol Arthrosc. 2018. https://doi.org/10.1007/s00167-018-4916-4.
Faltstrom A, Hagglund M, Kvist J. Functional performance among active female soccer players after unilateral primary anterior aruciate ligament reconstruction compared with knee-healthy controls. Am J Sports Med. 2017;45(2):377–85.
Graziano J, Chiaia T, de Mille P, Nawabi DH, Green DW, Cordasco FA. Return to sport for skeletally immature athletes after ACL reconstruction: preventing a second injury using a quality of movement assessment and quantitative measures to address modifiable risk factors. Orthop J Sports Med. 2017;5(4):2325967117700599.
Kyritsis P, Bahr R, Landreau P, Miladi R, Witvrouw E. Likelihood of ACL graft rupture: not meeting six clinical discharge criteria before return to sport is associated with a four times greater risk of rupture. Br J Sports Med. 2016;50(15):946–51.
Luo TD, Ashraf A, Dahm DL, Stuart MJ, McIntosh AL. Femoral nerve block is associated with persistent strength deficits at 6 months after anterior cruciate ligament reconstruction in pediatric and adolescent patients. Am J Sports Med. 2015;43(2):331–6.
Beischer S, Senorski EH, Thomee C, Samuelsson K, Thomee R. Young athletes return too early to knee-strenuous sport, without acceptable knee function after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2018;26(7):1966–74.
Di Stasi SL, Logerstedt D, Gardinier ES, Snyder-Mackler L. Gait patterns differ between ACL-reconstructed athletes who pass return-to-sport criteria and those who fail. Am J Sports Med. 2013;41(6):1310–8.
Ebert JR, Edwards P, Yi L, Joss B, Ackland T, Carey-Smith R, et al. Strength and functional symmetry is associated with post-operative rehabilitation in patients following anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2018;13(3):389–400.
Thomee R, Neeter C, Gustavsson A, Thomee P, Augustsson J, Eriksson B, et al. Variability in leg muscle power and hop performance after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc. 2012;20(6):1143–51.
Toole AR, Ithurburn MP, Rauh MJ, Hewett TE, Paterno MV, Schmitt LC. Young athletes cleared for sports participation after anterior cruciate ligament reconstruction: how many actually meet recommended return-to-sport criterion cutoffs? J Orthop Sports Phys Ther. 2017;47(11):825–33.
Webster KE, Feller JA. Return to level I sports after anterior cruciate ligament reconstruction: Evaluation of age, sex, and readiness to return criteria. Orthop J Sports Med. 2018;6(8):2325967118788045.
Losciale JM, Zdeb RM, Ledbetter L, Reiman MP, Sell TC. The association between passing return-to-sport criteria and second anterior cruciate ligament injury risk: a systematic review with meta-analysis. J Orthop Sports Phys Ther. 2018;2018:1–52. https://doi.org/10.2519/jospt.2019.8190.
Paterno MV, Ford KR, Myer GD, Heyl R, Hewett TE. Limb asymmetries in landing and jumping 2 years following anterior cruciate ligament reconstruction. Clin J Sport Med. 2007;17(4):258–62.
Paterno MV, Rauh MJ, Schmitt LC, Ford KR, Hewett TE. Incidence of contralateral and ipsilateral anterior cruciate ligament (ACL) injury after primary ACL reconstruction and return to sport. Clin J Sport Med. 2012;22(2):116–21.
Ardern CL, Glasgow P, Schneiders A, Witvrouw E, Clarsen B, Cools A, et al. 2016 Consensus statement on return to sport from the First World Congress in Sports Physical Therapy, Bern. Br J Sports Med. 2016;50(14):853–64.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No sources of funding were used to assist in the preparation of this article
Conflicts of Interest
Kate Webster and Timothy Hewett declare that they have no conflicts of interest relevant to the content of this review.
Author Contributions
Both authors contributed to the design, analyses and reporting for this manuscript. Both authors read and approved the final submitted manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Webster, K.E., Hewett, T.E. What is the Evidence for and Validity of Return-to-Sport Testing after Anterior Cruciate Ligament Reconstruction Surgery? A Systematic Review and Meta-Analysis. Sports Med 49, 917–929 (2019). https://doi.org/10.1007/s40279-019-01093-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-019-01093-x