Abstract
Sorghum is a rich source of various phytochemicals including tannins, phenolic acids, anthocyanins, phytosterols and policosanols. These phytochemicals have potential to significantly impact on human health. Kissra is a naturally lactic acid bacteria- and yeast-fermented sorghum thin pancake-like flatbread, produced in Sudan from sorghum flour dough. Kissra has considerable potential as the basis for development of a gluten-free sandwich wrap. The Sudanese kissra is one of the types of popular bread that is loved by most of the people. Kissra bread has appreciable amounts of nutrients, vitamins, and minerals. Fermentation of sorghum flour improves the nutritional, therapeutic and digestibility of the resultant kissra product.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
26.1 Introduction
Sorghum bicolor (L.) Moench is a significant grain crop, especially on the planet’s semi-parched jungles. It is a significant food crop in sub-Saharan Africa and South Asia and is the staple nourishment for the most food-uncertain individuals on the planet (Bibi et al. 2010). It is the world’s fifth most significant oat, after wheat, rice, maize and grain (FAO 2010). In excess of 105 nations in Africa, Asia, Oceania and the Americas develop sorghum on 40 million hectares (Kumar et al. 2011), and 60% of this land is in Africa, where it keeps on playing a significant food security role (Assefa et al. 2020).
Sorghum bicolor is considered one of the important food and fodder crops for its use in feeding the livestock sector as grains with concentrates. It is also used as green fodder and in the manufacture of silage, as it is considered as a raw material for extracting starch, cellulose and making alcohol, in addition to the fact that some species have a high percentage of sugar (wcsp.science.kew.org 2020).
Sorghum bicolor exists in dry weather, and is greatly affected by frost, especially during flowering, so it is grown as a summer crop after the end of the frost period. For the success of germination and plant growth, temperatures should not drop below 8–10 ° C, as the appropriate temperature for its success is 30–32 ° C. It also needs rain rates of at least 250 mm / year and may be cultivated under irrigation. It is well cultivated in all types of soil and has the ability to withstand salinity and alkalinity, but fertile muddy lands are preferred at rates of 350 mm / year rain (data.nbn.org.uk. 2020).
The sorghum crop is considered one of the most important crops in Sudan and ranks first in terms of importance as food for the vast majority of Sudanese in terms of cultivated area and total productivity. Corn is grown in Sudan in the irrigated and rainy sectors, and corn is especially important for farms in irrigated projects in terms of securing The food for him, the labor he uses, and his animals. Due to the fluctuation of rains under rainfed conditions, irrigated corn has an important role in securing food for the country in general. Sudan grows about 24% of the corn area in Africa and produces 17% of the production. In addition, it contributes 70–85 to the volume of grain production in Sudan (Fig. 26.1).
Sorghum bicolour (L.) Monech plant. (Source: https://commons.wikimedia.org/)
26.2 Sorghum Production in Sudan
The sorghum crop is considered one of the most important crops in Sudan and ranks first in terms of importance as food for the vast majority of Sudanese in terms of cultivated area and total productivity. Sorghum is grown in Sudan in the irrigated and rainy sectors, and it is particularly important for farmer in irrigated projects in terms of securing the food for him, the labor he uses, and his animals. Due to the fluctuation of rains under rainfed conditions, irrigated sorghum has an important role in securing food for the country in general. Sudan grows about 24% of the corn area in Africa and produces 17% of the production. In addition, it contributes 70–85 to the volume of grain production in Sudan (http://www.tpsudan.gov.sd/index.php/ar/home/show_export/49).
Despite the importance of the corn crop and the existence of the basic ingredients for its production in irrigated projects, especially irrigation water, and the suitability of the climatic conditions for production, as Sudan is considered one of the habitats of corn. However, its productivity remained low and not commensurate with what was achieved in many countries that consider corn an alien crop, and the reason for the low productivity is the lack of or incomplete use of the technical packages recommended by the Agricultural Research Authority (http://www.ttea.gov.sd/sorghum.html) (Fig. 26.2).
Sorghum bicolor grains. (Source: https://commons.wikimedia.org/)
The cultivation of sorghum is good in all types of light and heavy soils, as well as tolerates salinity and alkalinity in relatively high degrees, and tolerates intense heat and intense thirst, as it continues dormant without any vital activity until we drop rain, so it continues to grow. Therefore, it is found that it is grown in all production areas in the country. In addition, its production is concentrated in all production areas in the country. It is production in the rain sector, both automatic and traditional, at about 78%, and the irrigated sector, about 22% of the total production, in an area that exceeds 40% of the total cultivated areas in Sudan. The country produced, on average, for the period from (97–2000) about 2.5 million tons of corn annually.
The most important uses of sorghum are thus summarized:
-
1-
Mixing white corn flour with wheat flour to make bread.
-
2-
It is used in poultry feeding and mixed with other feeds to feed dairy cattle. This is because of the closeness of the nutritional composition of this crop with the nutritional composition of yellow corn.
-
3-
Its green plants are used as fodder for livestock, provided that the plants are not less than 55 days old, given the toxicity of the leaves, and they are small because they contain toxic durin glucoside, provided that the leaves are dried for twelve hours before use.
-
4-
The plant residues are used after harvesting the crop to feed work animals and livestock.
-
5-
Sorghum is one of the most important agricultural products used in the production of glucose and starch sugar.
26.2.1 Chemical Composition and Nutritional Value of Sorghum
Sorghum plays an important role in the economy of Sudan, and it represents the main food for 65% of the population of the Sudan, particularly in rural areas in central and eastern Sudan, as sorghum is an important source of carbohydrates needed for human and animal food. It is used as fodder, fuel and as a building material in housing. Moreover, sorghum is used in the manufacture of starch and glucose (http://www.ttea.gov.sd/sorghum.html.)
Sorghum is considered as food with low nutritional value (Raihanatu et al. 2011). Further, sorghum contains anti-nutritional factors like tannin, cyanogenicglucoside, phytic acid, trypsin inhibitor, and oxalate (Etuk et al. 2012; Mohammed et al. 2011). Various researches have revealed that the processing condition decreased antinutritional factors and increased the bioavailability of other nutrient in cereals and legumes (Adegunwa et al. 2012; Mubarak 2005; Osman 2007; Yasmin et al. 2008; Ogbonna et al. 2012).
The chemical composition and nutritional value of whole sorghum are similar to rice, corn, and wheat. The energy value of 100 g of sorghum grains varies between 296.1 and 356.0 kcal (Martino et al. 2012; U.S. Department of Agriculture 2012). The main components of sorghum are the polysaccharides (starch and non-starch), followed by proteins and lipids (Martino et al. 2012; U.S. Department of Agriculture 2012).
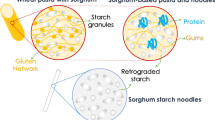
The content and composition of starch, the main polysaccharide of sorghum, are influenced by the genetic characteristics and growing conditions of the grain (Hill et al. 2012). In some varieties, starch ranges between 32.1 and 72.5 g/100 g and is composed mainly of amylopectin (81.0–96.5%) and amylase (3.5–19.0%) (Shegro et al. 2012; Udachan et al. 2012).
Sorghum has the lowest starch digestibility among cerealsbecause of the solid relationship between the starch granules and proteins and tannins (Barros et al. 2012; Rooney and Pflugfelder 1986). Generally the majority of the starch granules are gradually edible (30.0–66.2%) and the rest of quickly absorbable (15.3–26.6%) or safe (16.7–43.2%) (Sang et al. 2008; Mkandawire et al. 2013). The non-starch polysaccharides of sorghum (6.0 to 15.0 g/100 g) include insoluble fibers (75.0–90.0%), mainly arabinoxylans, and soluble fibers (10.0–25.0%) (Taylor and Emmambux 2010; Martino et al. 2012; U.S. Department of Agriculture 2012).
Sorghum proteins are classified as prolamins and not prolamins. Prolamins correspond on average to 79% (77–82%) of the total proteins (7 to 15 g/100 g) and the remainder is albumins, globulins, and glutelins (Belton et al. 2006; Martino et al. 2012; U.S. Department of Agriculture 2012; Afify et al. 2012b). The kafirins are the major prolamins of the sorghum and comprise three major classes: a-kafirins (66–84%), b kafirins (8–13%) and g-kafirins (9–21%) (Belton et al. 2006; Mokrane et al. 2010). Sorghum kafirins are stored in the endoplasmic reticulum in spherical protein bodies. The b and g-kafirins are located in the peripheral protein bodies region while a and d-kafirins are encapsulated in the inner region (Wu et al. 2013). This conformation determines the digestibility of sorghum proteins. Overall, the digestibility of sorghum proteins, especially after cooked, is lower than cereals like wheat and maize (Duodu et al. 2003; Mokrane et al. 2010; Afify et al. 2012b; Moraes et al. 2012b). Despite the reduction in protein digestibility of sorghum after cooking in wet heat, processing such as fermentation and germination may increase the digestibility up to 2 times (Correia et al. 2008; Wedad et al. 2008; ELKhier and Abd- ALRaheem 2011; Pranoto et al.; Afify et al. 2012b).
Sorghum has a reduced lipid content (1.24 to 3.07 g/100 g), which is mainly composed of unsaturated fatty acids (83–88%) (Afify et al. 2012a; Martino et al. 2012; U.S. Department of Agriculture 2012). In most of the varieties of sorghum the polyunsaturated fatty acids (PUFA) are higher than monounsaturated fatty acids (MUFA) (Mehmood et al. 2008; Hadbaoui et al. 2010; Afify et al. 2012a). The major fatty acids of sorghum are linoleic (45.6–51.1%), oleic (32.2–42.0%), palmitic(12.4–16.0%), and linolenic acids (1.4–2.8%) (Mehmood et al. 2008; Hadbaoui et al. 2010; Afify et al. 2012a).
Sorghum is a source of minerals (phosphorus, potassium, and zinc) whose content varies according to the place of cultivation (Martino et al. 2012; Shegro et al. 2012; Silva et al. 2012; U.S. Department of Agriculture 2012).
Information on the content of vitamins in sorghum is scarce. However, it is worth noting that it is a source of some B-complex vitamins (thiamine, riboflavin, and pyridoxine) and fatsoluble vitamins (D, E, and K) (Ochanda et al. 2010; Martino et al. 2012; U.S. Department of Agriculture 2012; Cardoso et al. 2014).
26.2.2 Bioactive Compounds of Sorghum
The phenolic compounds are the main bioactive compounds of sorghum and are present in all varieties of this cereal (Dykes and Rooney 2006). Almost all classes of phenolics are found in sorghum (Awika and Rooney 2004; Dykes et al. 2005); however, the classes of phenolic acids, tannins, and flavonoids are major. The profile and content of phenolic compounds in sorghum are more diverse and higher than those observed in wheat, barley, rice, maize, rye, and oats (Ragaee et al. 2006). Sorghum varieties resistant to biotic and abiotic stresses were found to have on average higher contents of proanthocyanidins, 3-deoxyanthocyanidins, and flavan-4-ols than susceptible varieties (Dicko et al. 2005).
The content of phenolic acids in some sorghum varieties ranged between 135.5 and 479.40 mg/g (Afify et al. 2012c; Chiremba et al. 2012).
Tannins, secondary metabolites found in many plant species, are phenolic compounds that often act as a defense mechanism against pathogens and predators (Kaufman et al. 2013). Overall, these compounds are absent in other major cereals, such as rice, wheat, and maize, but are present in sorghum varieties that have pigmented testa (Awika 2003; Dykes and Rooney 2006; Wu et al. 2012). The presence and content of condensedtannins in sorghum are controlled by the genes S and Tannin1, among others (Hahn and Rooney 1986; Wu et al. 2012).
26.2.3 Traditional Sorghum Use for Food
Sorghum is used in a variety of foods. The white food sorghums are processed into flour and other products, including expanded snacks, cookies and ethnic foods, and are gaining popularity in areas like Japan (United States Grains Council 2001; Rooney 2001).
Traditionally, Africa has employed sorghum in both the malted and unmalted form in wide varieties of porridge and beverages, often using lactic and alcoholic fermentation to enhance their appeal. In the Sudan, sorghum is the staple food of the vast majority of the population and is produced mainly in the central clay plains of the Sudan under rain, with limited amount being produced in the irrigated schemes of Gezira, Rahad and New Half.
26.2.3.1 Sudanese Fermented Sorghum Based Foods
In general, two sorts of fermentation are practices: by addition of starter to the dough or by the natural flora activity of microflora. Sorghum fermentation is principally a lactic acid one (Abdel Gadir and Mohamed 1983; El Mahdi 1985; Mohammed et al. 1991). The raw material and its initial treatment will encourage the growth of an indigenous microbiota (Tamang and Fleet 2009).
It is well established that fermentation enhances the nutritional quality of foods and contributes to food safety particularly under conditions where refrigeration or other foods processing facilities are not available (Motarjemi 2002) such as in arid and semi-arid rural areas in Sudan. Fermentation also develops a new flavor and appearance in 21 food products, and is also utilized as a technique of preservation (Onwurafor et al. 2014).
26.3 Fermented Foods
Fermentation is one of the oldest biotechnology approaches of food processing and preservation that extensively applied in both developed and developing countries. Over thousands of years, the demands of producing and consuming fermented foods has extremely increased, so these products constituted a significant portion of the daily food globally (Elyas et al. 2015).
Fermentation enhances the nutritional quality of foods through the biosynthesis of vitamins, essential amino acids and proteins, improving protein and fiber digestibility, enhancing micronutrient bioavailability, and degrading anti-nutritional factors (Giraffa 2004). It also contributes to food safety and sustainability particularly under conditions where refrigeration or other foods processing facilities are not available such as in arid and semi-arid rural areas in Sudan (Elyas et al. 2015).
Food fermentation covers a wide scope of microbial and enzymatic processing of food and fixings to accomplish desirablecharacteristics such as prolonged shelf-life, improved safety, attractive flavour, nutritional enrichment, and health promotion (Giraffa 2004; Holzapfel 2002). Throughout the fermentation processes, microorganisms played a key role in the production of specific metabolites such as acids, alcohols, enzymes, antibiotics, carbohydrates, which contribute to the safety and nutritional quality of fermented foods. One of these metabolites, is lactic acid bacteria (LAB) , play a significant role in the majority of food fermentations and preservation, and a extensive variety of strains are routinely employed as starter cultures in the manufacture of dairy, meat, vegetable, and bakery products (Elyas et al. 2015; Giraffa 2004; Saeed et al. 2014). This is because of the fact that lactic acid bacteria has numerous essential technological properties, for example, acid production in various media and at different temperatures, proteinase and peptidase activities, autolysis, production of volatile compounds, resistance to bacteriophages and production of inhibitory compounds (Piraino et al. 2008). These properties are significant for the utilization of LAB as starters or assistants to keep up and improve the nourishing, tangible, and security characteristics of end results and their evaluation in the screening of proper starter culture from natural environments has been and still on the rise in recent years. However, these fermented foods are still mainly prepared at the household level under poor sanitary conditions and advertised through casual courses (Elyas et al. 2015; Saeed et al. 2014).In like manner, various polluting microorganism and additionally native microflora engaged with this fermentation processes could be anticipated. In addition, there is a lack of data on the technological properties of microorganisms involved and their metabolic impact on flavour, hygienic safety and shelf life of these products.
26.3.1 Fermented Cereal Foods
The well-documented fermented cereal foods of the world are sourdough of Europe, America, and Australia, selroti of India and Nepal (Yonzan and Tamang 2009), idli of India and Sri Lanka (Sridevi et al. 2010), dosa of India and Sri Lanka (Soni et al. 1986), mawè and gowé of Benin (Vieira-Dalodé et al. 2007), ben-saalga of Burkino Faso and Ghana (Humblot and Guyot 2009), kissra of Sudan (Hamad et al. 1997), kenkey of Ghana (Oguntoyinbo et al. 2011), togwa of Tanzania (Mugula et al. 2003), ting of Botswana (Sekwati-Monang and Gänzle 2011), ogi and kunu-zaki of Nigeria (Oguntoyinbo et al. 2011), and tarhana of Turkey, Cyprus and Greece (Sengun et al. 2009). Cereal fermentation is portrayed by a complex microbial environment, chiefly addressed by the types of LAB and yeasts (Corsetti and Settanni 2007), whose fermentation confers to the resulting bread its characteristic features such as palatability and high sensory quality (Blandino et al. 2003). The species of Enterococcus, Lactococcus, Lactobacillus, Leuconostoc, Pediococcus, Streptococcus, and Weissella are commonly associated with cereal fermentation (Guyot 2010). A native strain of Sacch. cerevisiae is the principal yeast of most bread fermentations (Hammes et al. 2005). Other non-Saccharomyces yeasts are involved in numerous cereal fermentations which include Candida, Debaryomyces, Hansenula, Pichia, Trichosporon, Yarrowia (Foschino et al. 2004, Vernocchi et al. 2006). Yeasts produce carbon dioxide and ethanol. Interactions between yeasts and lactobacilli are important for the metabolic activity of the sourdough. The changing conditions during fermentationcontribute to the activation of enzymes present, and adjustment ofpH selectively enhances performance of certain enzymes, such asamylases, proteases, hemicellulases and phytases. The enzymeinducedchanges, together with microbial metabolites, bring aboutthe technological and nutritional effects of fermented cereal foods .Sourdough fermentation can influence the nutritional quality bydecreasing or increasing levels of compounds, and enhancing orretarding the bioavailability of nutrients.
26.4 Kissra
Kissra can be defined in a number of ways (Abdualrahman and Ali 2012):
-
1-
Kissra is the staple Sudanese diet. It is a morsel or piece of bread prepared from fermented sorghum flour (Sulieman et al. 2003).
-
2-
Kisra is a naturally lactic acid bacteria- and yeast-fermented sorghum pancake-like flatbread(AwadElkareem and Taylor 2011).
-
3-
Kisra is an indigenous staple food of the majority of Sudanese people. It is pancake-like bread made from sorghum or millet flour.
The kissra is called ‘al-Rahafa’ and the way it was prepared has not changed for centuries, but its presence on the table in Sudan has declined due to the availability of bread in various forms, after it was scarce. There are many types of corn used in the making of the Sudanese kissra, including (fretrita - yellow hybrid, dr Aker, al-Fahl, and al-Zrizira) (Fig. 26.3).
Today, with growing urbanization, kisra is becoming a commercial home-based industry in Sudan. Internationally, because of the apparent increase in the incidence of celiac disease and intolerance to wheat, interest in gluten-free cereal products is increasing rapidly (Kelly et al. 2008).
Kissra seems to have significant potential as the basis for development of a gluten-free sandwich wrap. As of late, it has been shown that Lactobacillus and Saccharomyces cultures can be used to reduce the fermentation time from 19 to 4 hr. (Ali and Mustafa 2009), which would be useful for commercial production.
26.4.1 Kissra Preparation
Kissra fermentation is a conventional process, whereby sorghum or millet flour is blendid with water in a proportion of about 1:2 (w/v), generally a starter is added by a back-slopping utilizing mother batter from a past fermentation as a starter at a degree of about 10%. Fermentation is finished in around 12–19 hours by which time the pH drops from around six to less than four. Because of the tedious process of kissra perparation, the greater part of the populace deserted kissra utilization and moved to bread (Ali and Mustafa 2009).
The process begins with fermenting the flour at night in a closed container, and in the morning the rest of the flour is added to it, kneaded and reduced until it becomes coherent and ready to make the kissra. The process of making kissra is called “Awassa,” and the woman or girl who is preparing it is called “Awassa”. Awassa” process starts by pouring of a small amount of dough on the saj (hot plate), which is placed on a slow fire of charcoal on a canon (wood stove). In the past, the “douka” made of pottery in a flat form was used. The Awassa saj is often wiped with oil or taouk (animal marrow), which is the material of the spinal cord. This is because it helps to pull out the fracture quickly so that it does not stick to the saj and burn.
After that, the kissra is placed on the plate one by one, and the plate is an airtight, flat round container that can be stored in it for several days.
Nowadays, with developing urbanization, kissra is turning into a business locally situated industry in Sudan. Globally, due to the clear expansion in the frequency of celiac infection and prejudice to wheat, interest in gluten-free cereal items is expanding quickly (Kelly et al. 2008) (Fig. 26.4).
26.4.2 Nutritive Value of Kissra
Abubaker et al., (2019) investigated the chemical compositionof Kissra. They found that contents of moisture, ash, protein, fats, fibres and carbohydrates of kissra ranged between 5.17–5.02%, 1.32–1.29%, 10.01–9.95%, 1.66–1.89%and 79.09–79.26%, respectively. They Furthermore, they found that kisra fermentation improved levels of the protein, fiber, fat, ash, and minerals contents. Moreover, maximum amino acids contents were found in Kisra prepared from Tabat sorghum flour compared with that prepared with Wad-Ahmed sorghum flour.Kissra fermentation also resulted in increasing ascorbic acid content, in vitro protein (IVPD) and in vitro starch digestibilities (IVSD) of Kissra from both sorghum cultivars, with a concomitant decrease in phytate and tannin contents (Figs. 26.5 and 26.6).
Kawthar et al. (2018) analyzed six samples of kissra from Khartoum Bahry markets. Three were made from pure mixed cluture of lactic acid bacteria (LAB) and yeast (Saccharomyces cerevisiae). The proximate composition of kissra results showedsignificant (P < 0.05) difference in moisture content of most of the various kissra samples.The control showed the highest level of moisture content (8.76%) which was higherthan the values reported for the recommended dietary allowance (RDA) value(<5%) for older infants and young children (FAO/WHO 1991). The lowermoisture content is an indication of the better quality of the products with thelonger shelf life. On the other hand, Mohammed et al. (2017) reported a value of 3.13% for moisture content of kissra.
According to Kawthar et al. (2018), ash content ranged between (1.710–1.333%) which than the values reported by Mohammed et al. (2017) which was (2.86%), butagreed with the recommended dietary allowance (RDA) value (< 3%) for olderinfants and young children (FAO/WHO 1991).
Higher protein contents of kissra was reported by both Kawthar et al. (2018) and Mohammed et al. 2017) who reported an average of 10.90–13.37%.
Elkhalifa et al. (2005) announced that customary Sudanese technique for fermentation prompts an expansion in the protein solubility of sorghum flour in the acidic reach (pH 2–4). Additionally they detailed that fermented sorghum flour had a least gelation concentration of 6% after 16 h of maturation, while it was 18% for unfermented sorghum.
Fermentation likewise expanded oil binding capacity, emulsifying capacity and emulsifying strength, while it diminished the water-binding capacity. Sorghum flour, fermented or unfermented showed no frothing capacity (Elkhalifa et al. 2005). According to study by Kawthar et al. (2018) All types of kissra showed significant difference in fibre content, the kissra which have the highest levels of (1.267 and 1.287%), and the lowest level (0.617%). The fibre content of all kissra was less than the result obtained by Mohammed et al. (2017) which was (2.41%) but agreed with the recommended dietary allowance (RDA) value (< 5%) for older infants and young children (FAO/WHO 1991). The reduction in fiber content of kissra would enable the children to utilized sufficient amounts, giving an opportunity to meet their daily energy and other vital nutrient requirements.
Fat and carbohydrates content the kissra ranged 1.133% - 5.52% and 73. 63–78.34%, respectively (Kawther et al. 2018; Mohammed et al. 2017). However, these values were higher than that reported (FAO/WHO 1991).
Contents of thiamin (vitamin B1) and riboflavin (vitamin B2) in kissra prepared from two sorghum cultivars, dabar and feterita were investigated by Salah et al., (1998). They found that contents of both vitamins improved as a result of fermentation process. The riboflavin contents of the two sorghum cultivars were almost the same (1.08 mg/g for dabar and 1.07 mg/g for fetarita). The difference in the thiamine content of the two cultivars (3.92 mg/g for dabar and 3.47 mg/g for fetarita) was insignificant (p < 0.01). On the other hand, germination of fetarita grains for 6 days caused significant effects on the levels of thiamine and riboflavin which increased by 70.6% and 42%, respectively (Fig. 26.7).
26.4.3 Fermentation Effects on Antinutritional Components
Sorghum is considered as food with low nutritional value (Raihanatu et al. 2011). Poor digestibility of sorghum and limited product diversification compared to other cereals limit the use of sorghum (Mella 2011). Further, sorghum contains anti-nutritional factors like tannin, cyanogenicglucoside, phytic acid, trypsin inhibitor, and oxalate (Etuk et al. 2012; Mohammed et al. 2011). Due to these and other reasons, sorghum is categorized as of low nutritional value and a food for the poor. Low protein digestibility and mineral absorption are also associated with the presence of antinutritional factors (Mohammed et al. 2011). Different investigates have uncovered that the handling condition reduced antinutritional factors and expanded the bioavailability of other supplement in cereals and vegetables (Adegunwa et al. 2012; Mubarak 2005; Osman 2007; Yasmin et al. 2008; Ogbonna et al. 2012).
According to the study conducted by Pravin et al. 2017 to observe the effect of malting and fermentation on antinutritional component and functional characteristics of sorghum flour, they found that, the lower yield of sorghum flour was obtained compared to whole and malted sorghum flour. Germination of sorghum decreased phytate, tannin, and oxalate by 40%, 16.12% and 49.1%, respectively, whereas fermentation of sorghum flour reduced these compounds by 77%, 96.7% and 67.85%, respectively. Furthermore, in their study on nutritional value, protein quality and antioxidant activity of Sudanese sorghum-based kissra bread fortified with bambara groundnut, Mohamed et al. (2019) indicated that tannins, phytic acid and trypsin inhibitor activity were significantly reduced (P < 0.05) (Fig. 26.8).
26.4.4 Microbiology of Kissra Bread
According to many investigations on sorghum flour lactic acid bacteria (LAB) and yeasts dominated fermentation. Mohammed et al., (1991) demonstrated that bacterial burden expanded with fermentation time. Besides the microbial populace during the 24 h of fermentation comprised of bacteria (Pediococcus pentosaceus, Lactobacillus confusus, Lactobacillus brevis, Lactobacillus sp., Erwinia ananas, Klebsiella pneumoniae, and Enterobacter cloacae), yeasts (Candida intermedia and Debaryomyces hansenii), and molds (Aspergillus sp., Penicillium sp., Fusarium sp., and Rhizopus sp.). P. pentosaceus was the dominant microorganism toward the end of the 24-h fermentation.
. Lactic acid bacteria were the dominant microflora and their number increased during fermentation of three sorghum varieties, in the fermentation of the three varieties and at each interval the number of lactic acid bacteria was greater than the number of yeasts and molds and they dominated till the end of fermentation. This fact implied that Kisra fermentation is mainly a lactic acid fermentation (Abdel Rahman et al. 2010).
Yagoub et al., (2009) indicated significant drop in pH during fermentation of sorghum dough. Microbial examination of sorghum varieties before fermentation showed that the total bacterial counts and the counts of yeast and molds expanded altogether after fermentation time (24 hours), while, there was a reduction in staphylococcal counts. The microbiological examination additionally uncovered that E. coli counts exceeded 2.400 cfu/g in the raw sorghum flour but the counts were very low in the fermented dough. Salmonella was identified in the three sorghum varieties vanished in the fermented dough after 24 h fermentation.
In their study on the investigation on the microbiological qualities, germinated sorghum (malt) and fermented dough at different timeframes, Sulieman et al., (2015) indicated that three samples out of ten were devoid of any lactic acid bacteria (LAB)development, while the other seven samples indicated development of bacteria, thus, a total of 140 isolates of these bacteria were examined to identify their bacterial genera. Their outcomes demonstrated that a portion of the isolates were cocci while others were rods in shape, not many of the isolates were hetrofermentative, and most of them were homofermentative, all isolates were grow at 15 °C and some growth at 10 °C and 45 °C. And the LAB isolated bacteria recognized in this examination, have a place with five genera; Streptocpccus, Lactobacillus, Leuconostoc, Enrococcus and pediococcuswith various rates .
The concurrence among LAB and yeasts affirmed the synergistic connection between the organisms in a fermenting food matrix (Wood 2004). Yeast is significant for or acceptable batter and raising, while LAB produce acids and other metabolites which hinder the development of spoilage organisms. Lactic acid bacteria (LAB) strains are able to improve the time span of usability of few food items (Lopez et al. 2001; Di Cagno et al. 2004) (Table 26.1).
26.5 Conclusion
The traditional Sudanese method of fermentation of sorghum significantly improved the functional properties of sorghum flour. Traditional fermentation in Kisra bread is desirable because of their role in taste and flavour of the final product. In conclusion, sorghum flour and its fermented foods can be affected by fermentation and the method of preparation. A reduction of some nutritional parameters (carbohydrate and oil) and an increase in others (protein) were observed. In spite of the losses in some nutritional compounds, the fermented sorghumbase items, kissra, was found to have obvious nutritious qualities.
References
Abdel Gadir MA, Mohamed M (1983) Sudanese kisra. In: Handbook of indigenous fermented foods. Steinkraus KH (ed). Microbiology Series 9. Marcel Dekker Inc., New York, pp 175–179
Abdel Rahman IE, Hamad SH, Osman MA, Dirar HA (2010) Characterization and distribution of microorganisms associated with Kisra bread preparation from three Sorghum varieties in Sudan. Curr Res Bacteriol 3:138–147
Abdualrahman MAY, Ali AO (2012) Effect of full and partial substitution of sesame seeds with Bambara groundnuts and peanuts on the quality of Sudanese traditional food (Khemiss-tweria). Am J Food Technol 7(7):420–428
Adegunwa MO, Adebowale AA, Solano EO (2012) Effect of thermal processing on the biochemical composition, antinutritional factors and functional properties of beniseeds (Sesamumindicum) flour. American Journal of Biochemistry and Molecular Biology 2:175–182 https://doi.org/10.3923/ajbmb.2012.175.182
Afify A, El-Beltagi H, ABD EL-SALAM SM, Azza A, Omran AA (2012) Effect of Soaking, Cooking, Germination and Fermentation Processing on Proximate Analysis and Mineral Content of Three White Sorghum Varieties (Sorghum bicolor L. Moench). November 2012.Notulae Botanicae Horti Agrobotanici Cluj-Napoca 40(2):92–98. https://doi.org/10.15835/nbha4027930
Ali AA, Muna M, Mustafa MM (2009) Isolation, characterization and identification of lactic acid bacteria from fermented sorghum dough Used in Sudanese Kisra Preparation. Pak J Nutr 8(11).
Assefa A, Bezabih A, Girmay G, Alemayehu T, Lakew A (2020) Evaluation of sorghum (Sorghum bicolor (L.) Moench) variety performance in the lowlands area of wag lasta, north eastern Ethiopia. Cogent Food Agric 6:1778603
Awika JM, Rooney LW (2004) Sorghum phytochemicals and their potential aspects on human health. Phytochemistry 65:1199–1221
Awadelkareem A, Taylor JRN (2011) Protein quality and physical characteristics of Kisra (fermented sorghum pancake-like flatbread) made from tannin and non-tannin sorghum cultivars. Cereal Chem 88:44–348
Barros R, Isidoro D, Aragüés R (2012) Three study decades on irrigation performance and salt concentrations and loads in the irrigation return flows of La Violada irrigation district (Spain). Agric. Ecosyst. Environ. 151: 44–52
Belton P, Delgadillo I, Halford N, Shewr PR (2006) Kafirin structure and functionality. November 2006. Journal of Cereal Science 44(3):272–286. https://doi.org/10.1016/j.jcs.2006.05.004
Bibi A, Sadaqat HA, Akram HM, Mohammed MI (2010) Physiological markers for screening sorghum (Sorghum bicolor) germplasm under stress condition. Int J Agric Biol Sci 12(3):451–455
Blandino A, Al-Aseeri M, Pandiella SS et al (2003) Cereal-based fermented foods and beverages. Int Food Res J 36(6):527–543
Cardoso LdM, Montini TA, Pinheiro SS, Pinheiro Sant’Ana HM, Martino HSD, and Moreira AVB (2014) Effects of processing with dry heat and wet heat on the antioxidant profile of sorghum (Sorghum bicolor L.). Food Chem. 152:201–217
Clarke CI, Arendt E (2005) A review of the application of sourdough technology to wheat breads. Adv Food Nutr Res 49:137–161
Corsetti A, Settanni L (2007) Lactobacilli in sourdough fermentation. Int Food Res J 40:539–558
Antónia Correia, Miguel Moital, Carlos Ferreira da Costa and Rita Peres (2008) The determinants of gastronomic tourists’ satisfaction: a second-order factor analysis. Journal compilation © 2008, Blackwell Publishing Journal of Foodservice, 19, pp. 164–176 https://doi.org/10.1111/j.1745-4506.2008.00097.x
data.nbn.org.uk. data.nbn.org.uk. Archived from the original on July 23, 2020
Di Cagno R, Tamborrino A et al. (2004) Uses of mares’ milk in manufacture of fermented milk. Int Dairy J 14(9):767–775
Duodu KG, Taylor JRN, Belton PS and Hamaker BR (2003) Factors affecting sorghum protein digestibility, J. Cereal Sci. 38: 117–131
ELKhier MKS, and Abd-ALRaheem AA (2011) Effect of fermentation period on the chemical composition, in vitro protein digestibility and tannin content in two Sorghum cultivars (Dabar and Tabat) in Sudan. J. Appl. Biosci. 39:2602–2606
Elyas YYA, Yousif NME, Mohamed AIA (2015) Screening of lactic acid bacteria from Sudanese fermented foods for bacteriocin production. J Microbial Biotechnol Food Sci 4:373–378
El Mahdi ZM (1985) Microbial and biochemical characteristics of legume protein supplemented kisra. M.Sc. Thesis, University of Sudan
Elkhalifa AO, Schiffler B, Bernhardt R (2005) Effect of fermentation on the functional properties of sorghum flour. Food Chem 92:1–5
Etuk EB, Okeudo NJ, Esonu BO, Udedibie ABI (2012) Anti-nutritional factors in sorghum: chemistry, mode of action and effects on livestock and poultry. Online J Animal Feed Res 2:113–119
FAO/WHO (1991) Protein quality evaluation : report of the Joint FAO/WHO Expert Consultation, Bethesda, Md., USA 4-8 December 1989
FAO (2006) Official Agricultural statistics, 18th edn. Canada, Foodand Agricultural Organization, pp 1002–1004
FAO (2010) FAOSTAT. Retrieved December 23, 2012, from http://www.faostat.fao.org
Fenster C( 2003) White food sorghum in the American diet. In: US Grains Council 43rd Board of Delegates’ Meeting, July 2003, Minneapolis, MN. Available from http://www.grains.org/news/latest_news/43_delegates_meeting_updates.html
Finney PL (1983) Eect of germination on cereal and legume nutrient changes and food or feed value: a comprehensive review. Recent Adv Phytochem 17:229–305
Food and Agricultural Organization (FAO) (1995) Sorghum and millet inhuman nutrition. FAO Food and Nutrition Series No. 27. ISBN 92–5–103381-1. Consulted on 10 September 2005
Food and Agricultural Organization (FAO) (2005) FAOSTAT
Foschino R, Gallina S, Andrighetto C, Rossetti L, Galli A (2004) Comparison of cultural methods for the identification and molecular investigation of yeasts from sourdoughs for Italian sweet baked products. FEMS Yeast Res 4:609–618
Giraffa G (2004) Studying the dynamics of microbial populations during food fermentation. FEMS Microbiol Rev 28:251–260
Grains Council 43rd Board of Delegates’ Meeting, July 2003
Guyot JP (2010) Fermented cereal products. In: Tamang JP, Kailasapathy K (eds) Fermented foods and beverages of the world. CRC Press, Taylor and Francis Group, New York, NY, pp. 247–261
Hahn DH, Rooney LW (1986) Effect of genotype on tannins and phenols of sorghum. Cereal Chem 63:4–8
Hamad SH, Dieng MC, Ehrmann MA, Vogel RF (1997). Characterization of the bacterial flora of Sudanese sorghum flour and sorghum sour dough. J Appl Microbiol 83:764–770
Hammes WP, Brandt MJ, Francis KL, Rosenheim J, Seitter MFH, Vogelmann SA (2005) Microbial ecology of cereal fermentations. Trends Food Sci Technol 16:4–11
Hill SE, Rodeheffer CD, Griskevicius V, Durante K, & White AE (2012) Boosting beauty in an economic decline: Mating, spending, and the lipstick effect. Journal of Personality and Social Psychology, 103(2), 275–291. https://doi.org/10.1037/a0028657
Hind Mokrane H, Houria A, Naima B, Christophe M, Courtin, Jan A Delcour, Nadjemi B (2010) Assessment of Algerian sorghum protein quality [Sorghum bicolor (L.) Moench] using amino acid analysis and in vitro pepsin digestibility. August 2010 Food Chemistry 121(3):719–723 https://doi.org/10.1016/j.foodchem.2010.01.020
Holzapfel WH (2002) Appropriate starter culture technologies for small-scale fermentation in developing countries. Int J Food Microbiol 75:197–212
http://faostat.fao.org/faostat/
http://www.tpsudan.gov.sd/index.php/ar/home/show_export/49
http://www.ttea.gov.sd/sorghum.html
https://doi.org/10.1080/23311932.2020.1778603
https://doi.org/10.3923/ajbmb.2012.175.182
Humblot C, Guyot JP (2009) Pyrosequencing of tagged 16S rRNA gene amplicons for rapid deciphering of the microbiomes of fermented foods such as pearl millet slurries. Appl Environ Microbiol 75:4354–4361
Kawthar MA, Hanan BE, Yousif FHE, Ahmed EE (2018) Molecular characterization of lactic acid bacteria isolated from starter dough of Sudanese sorghum fermented flat bread (Kissra). Pak J Nutr 17(2):57–63
Kelly JA, Van der Linde TC, Anderson GS (2008) The influence of clothing and wrapping on carcass decomposition and arthropod succession: A winter study in Central South Africa. Can Soc Forensic Sci J 41(3):135–147
Kumar AA, Reddy BVS, Sharma HC, Hash CT, Rao PS, Ramaia B, Reddy PS (2011) Rancement research at ICRISAT. Am J Plant Sci 2(4):589–600. https://doi.org/10.4236/ajps.2011.24070
Lopez PP, Santaren JF, Ruiz MF, Esponda P, Sanchez L (2001) The Drosophila melanogaster X-Linked mfs(1)6E locus is required for production of normal seminal fluid by the male accessory glands. Exp Cell Res 267(1):1–12
Martino H, Alves Tomaz P, Lopes Conceição L et. al. (2012) Chemical characterization and size distribution of sorghum genotypes for human consumption. Revista do Instituto Adolfo Lutz 71(2):337–44
Mehmood S, Orhan I, Ahsan Z, Aslan S, and Gulfraz M (2008) Fatty acid composition of seed oil of different Sorghum bicolor varieties. Food Chem. 109:855–859
Mella ONO (2011) Effects of malting and fermentation on the composition and functionality of sorghum flour, M.Sc Thesis, The University of Nebraska, USA
Mohammed SI, Steenson LR, Kirleis AW (1991) Isolation and characterization of microorganisms associated with the traditional sorghum fermentation for production of Sudanese Kisra. Appl Environ Microbiol 57:2529–2533
Mohammed NA, Mohammed IE, Barbiker EF (2011) Nutritionalevaluation of sorghum flour (Sorghum bicolour L. Moench) duringprocessing of injera. Int J Biol Biomol Agric Food Biotechnol Eng 5:99–103
Mohammed A, O’Hare MB, Warley A, Tear G, Tuxworth RI (2017) In vivo localization of the neuronal ceroid lipofuscinosis proteins, CLN3 and CLN7, at endogenous expression levels. Neurobiol Disease 103:123–132
Moraes DJ, A. Zoccal DB, Machado BH (2012b) Sympatho-excitation during chemoreflex active expiration is mediated by L-glutamate in RVLM/Botzinger complex of rats J. Neurophysiol. 108 (2), 610–623
Motarjemi Y (2002) Impact of small scale fermentation technology on food safety in developing countries. Int J Food Microbiol 75(3):213–229
Mubarak AE (2005) Nutritional composition and antinutritional factorsof mung bean seeds (Phaseolusaureus) as affected by some home traditionalprocesses. Food Chem 89:489–495
Mugula JK, Ninko SAM, Narvhus JA, Sorhaug T (2003) Microbiological and fermentation characteristics of togwa, a Tanzanian fermented food. Int J Food Microbiol 80:187–199. https://doi.org/10.1016/S0168-1605(02)00141-1
Ochanda SO, Onyango C, Mwasaru A, Ochieng J, and Mathooko F (2010) Effects of malting and fermentation treatments on group Bvitamins of red sorghum, white sorghum and pearl millets in Kenya. J. Appl. Biosci. 34:2128–2134
Ogbonna AC, Abuajah CI, Ide EO, Udofia US (2012) Effect of malting conditions on the nutritional and anti-nutritional factors of sorghum grist. Food Tech 36(2):64–72
Oguntoyinbo FA, Tourlomousis P, Gasson MJ, Narbad A (2011) Analysis of bacterial communities of traditional fermented West African cereal foods using culture independent methods. Int J Food Microbiol 145:205–210
Ojha P, Sigdel A, Karki R et al. (2017) Physiochemical and bioactive characteristics of osmo-air dried mulberry fruit. Nepalese Horticulture 12
Onwurafor EU, Onweluzo JC, Ezeoke AM (2014) Effect of fermentation methods on chemical and microbial properties of Mung Bean (Vigna radiata) Flour. Niger Food J 32(1)
Okoli IC, Okparaocha CO, Chinweze CE, Udedibie ABI (2012) Physicochemical and hydrogen cyanide content of three processed cassava products used for feeding poultry in Nigeria. Asian J Anim Vet Adv 7(4):334–340
Osman AM (2007) Effect of different processing methods on nutrient composition, anti-nutritionalfactors and in vitro protein digestibility on Dolichos lablab bean (Lablab purpureus (L) sweet). Pak J Nutr 6:299–303
Piraino P, Zotta T, Ricciardi A, PLH MS, Parente E (2008) Acid production, proteolysis, autolytic and inhibitory properties of lactic acid bacteria isolated from pasta filata cheeses: a multivariate screening study. Inter Dairy J 18:81–92
Raihanatu MB, Modu S, Falmata AS, Shettima YA, Heman M (2011) Effect of processing (sprouting and fermentation) of five local varieties ofsorghum on some biochemical parameters. Biokemistri 23:91–96
Rooney LW (2001) Food and nutritional quality of sorghum and millet. INTSORMIL 2001 Annual Report, Project TAM-226, pp. 105–114. Available from: http://intsormil.org/2001anlrpt/ 2001TAM-226.pdf
Rooney LW, Pflugfelder RL (1986) Factors affecting starch digestibility with special emphasis on sorghum and corn. J Anim Sci. 1986 Nov;63(5):1607–23. https://doi.org/10.2527/jas1986.6351607x. PMID: 3539904
Saeed RM, YYA E, NME Y, Eltayeb MM, Mohamed AIA (2014) Incidence of antibiotic resistance of lactic acid Bacteria (LAB) isolated from various Sudanese fermented foods. J Food Nutr Disor 3:6
Sang Y, Bean SR, Serib PA, Pederson JF, Shi YC (2008) Structural and functional of Sorghum starches differing in amylose content. J. Agric. food chem. 56, 6680–6685
Sekwati-Monang B, Gänzle MG (2011) Microbiological and chemical characterisation of ting, a sorghumbased. Int J Food Microbiol 150:115–121
Sengun IY, Nielsen DS, Karapinar M, Jakobsen M (2009) Identification of lactic acid bacteria isolated from Tarhana, a traditional Turkish fermented food. Int J Food Microbiol 135, 105–111
Abe Shegro A, Nemera GS, Angeline van Biljon, Maryke TL (2012) Diversity in Starch, Protein and Mineral Composition of Sorghum Landrace Accessions from Ethiopia. J. Crop Sci. Biotech. 2012 (December) 15 (4): 275. https://doi.org/10.1007/s12892-012-0008-z
Silva CSd, Queiroz VAV, Simeone MLF, Guimar~aes CdC, Schaffert RE, Rodrigues JAS, and Miguel RdA (2012) Teores de minerais em linhagens de sorgo para uso na alimentac¸~ao humana. In XXIX Congresso Nacional de Milho e Sorgo, (pp.2776–2782). Agua de Lindoia
Sulieman AME, Osman Y, Yousif H (2003) Nutritional aspects of cow pea (Vigna unguiculata) fortified cookies. Agri Sci 11(3):10–13
Sulieman AME, Mudawi EAM, Abdelgadir WS (2015) Isolation and identification of lactic acid from different stages of hul-ulmur fermentation. J Adv Food Sci Technol 2(1):9–15
Sridevi J, Halami PM, Vijayendra SVN (2010) Selection of starter cultures for idli batter fermentation and their effect on quality of idli. J. Food Sci Technol 47:557–563
Soni SK, Sandhu DK, Vilkhu KS, Kamra N (1986) Microbiological studies on Dosa fermentation. Food Microbiol 3:45–53
Tamang JP, Fleet GH (2009) Yeasts diversity in fermented foods and beverages. In: Satyanarayana T, Kunze G (eds) Yeasts biotechnology: diversity and applications. Springer, New York, pp 169–198
Taylor IRN and Emmambux MN (2010) Developments in our understanding of sorghum polysaccharides and their health benefits. Cereal chemistry, 87, 263–271
Udachan, Iranna S, Sahoo AK. and Hend GM (2012) Extraction and characterization of sorghum (Sorghum bicolor L. Moench) starch. International Food Research Journal 19(1): 315–319
United States Grains Council (2001) Sorghum production and usage data. Available from: http://www.grains.org/grains/sorghum.html
U.S. Department of Agriculture, National Agricultural Statistics Service (USDA, NASS). 2012. Acreage
Vernocchi P, Valmorri S, Dalai I et al. (2006) Characterization of the yeast population involved in the production of a typical Italian bread. J Food Sci 69(7):182–186
Vieira-Dalodé G, Jespersen L, Hounhouigan J, Moller PL, Nago CM, Jakobsen M (2007) Lated with gowé production from sorghum in Bénin. J Appl Microbiol 103:342–349
Wedad H, Abdelhaleem, Abdullahi H. El (2008) Tinay, Abdelmoneim I. Mustafa and Elfadil E. Babiker. Effect of Fermentation, Malt-Pretreatment and Cooking on Antinutritional Factors and Protein Digestibility of Sorghum Cultivars. Pakistan Journal of Nutrition 7 (2): 335–341, 2008 ISSN 1680-5194
wcsp.science.kew.org. 2020
Wu M et al. (2012) Identification of drug targets by chemogenomic and metabolomic profiling in yeast. Pharmacogenet Genomics 22(12):877–886
Yagoub AE, Suleiman AME, Abdel Gadir WS (2009) Effect of fermentation on the nutritional and microbiological quality of dough of different sorghum varieties. J Sci Technol 10(3):109–119
Yasmin A, Zeb A, Khalil AW, Paracha GM, Khattak AB (2008) Effect of processing on anti-nutritionalfactors of red kidney beangrains. Food Bioprocess Technol 1:415–419
Yonzan H, Tamang J (2009) Traditional processing of Selroti―A cereal based ethnic fermented food of the Nepalis. Indian J Tradit Knowl 8:110–114
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Sulieman, A.M.E., Ali, E. (2022). Production and Quality Assessment of Kissra, a Sudanese Fermented Sorghum Product. In: Elhadi Sulieman, A.M., Adam Mariod, A. (eds) African Fermented Food Products- New Trends. Springer, Cham. https://doi.org/10.1007/978-3-030-82902-5_26
Download citation
DOI: https://doi.org/10.1007/978-3-030-82902-5_26
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-82901-8
Online ISBN: 978-3-030-82902-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)