Abstract
The introduction of transpulmonary thermodilution technique allowed us to estimate extravascular lung water (EVLW) and pulmonary vascular permeability index (PVPI) quantitatively at the bedside. The estimation of EVLW by transpulmonary thermodilution has thoroughly been validated in humans against gravimetry which is regarded as the gold standard. EVLW correlates with the prognosis of patients suffering from acute respiratory distressed syndrome (ARDS), confirming it as a key pathophysiological component of this syndrome. Several studies suggest a normal EVLW value to be approximately 7 mL/kg and not exceeding 10 mL/kg. Thus, normal EVLW values can exclude the diagnosis of ARDS. Furthermore, in patients with elevated EVLW, PVPI may distinguish hydrostatic pulmonary edema from ARDS. Thus, PVPI <2 represents normal pulmonary permeability, and a PVPI of >3 suggests leaky lungs. Although the current diagnostic criteria of ARDS are controversial, EVLW and PVPI can quantitatively express the existence, severity, and nature of pulmonary edema at the bedside. Therefore, EVLW and PVPI may define ARDS and cardiogenic pulmonary edema, guiding the selection of the appropriate therapeutic strategy.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Acute respiratory distress syndrome
- Berlin definition
- Pulmonary edema
- Pulmonary permeability
- Transpulmonary thermodilution
1 Introduction
Pulmonary edema is one of the most common problems among critically ill patients with a profound effect on patient outcomes [1, 2]. Several publications have reported mortality reaching up to approximately 12% for cardiogenic [3] and 30% for non-cardiogenic pulmonary edema [1]. Thus, a better understanding and detailed evaluation of pulmonary edema is crucial for critical care management, especially fluid therapy. In this chapter, we will review the pathophysiology of pulmonary edema and the problems associated with it in clinical practice and describe several merits of evaluating pulmonary edema quantitatively using transpulmonary thermodilution-derived variables.
2 What Is Pulmonary Edema?
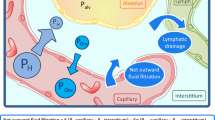
A pair of human lungs contain about 700 million alveoli [4]. Although alveoli are microscopically small, their overall superficial area is approximately 100 m2. Each alveolus consists of an epithelial layer, interstitium, and capillaries. The gaseous exchange of oxygen and carbon dioxide occurs between the inhaled air and the bloodstream in normal lungs (Fig. 14.1). The space outside the capillaries is known as the extravascular lung space with the fluid inside known as the extravascular lung water (EVLW).
Pulmonary edema is the accumulation of excess EVLW in the lungs [4] which impairs respiratory gas exchange, resulting in respiratory distress (Fig. 14.2) [5]. This pathological condition of the edema develops mainly by two mechanisms, an increase in the pulmonary capillary hydrostatic pressure (hydrostatic or cardiogenic pulmonary edema) and an increase in pulmonary capillary permeability (acute respiratory distress syndrome (ARDS)) [2]. An increase in the pulmonary capillary hydrostatic pressure is the main precursor of cardiogenic pulmonary edema. The elevated vascular pressure is usually accompanied by an increase in blood volume in the pulmonary vessels (e.g., fluid overload, untreated renal failure, or congestive heart failure). However, leaky lungs secondary to inflammatory mediators result in an increase in pulmonary capillary permeability, which is a representative type of non-cardiogenic pulmonary edema, including ARDS.
3 Evaluation of Pulmonary Edema
The question that arises is how can we evaluate the degree of pulmonary edema (i.e., EVLW amount) and differentiate cardiogenic from non-cardiogenic pulmonary edema in clinical practice?
The severity of pulmonary edema is evaluated by subjective methods (e.g., patient history, the presence of rales during physical examination, and chest X-ray findings) [2]. However, the interpretation of these methods is often limited due to subjectivity causing interobserver error, even among experts [6]. Several studies suggested only a moderate correlation between chest radiographic findings and EVLW amount [7, 8].
In addition, it is clinically difficult to discriminate between the edema caused by increased hydrostatic pressure in the course of cardiac disease, or by increased permeability associated with ARDS [2]. The latest Berlin definition of ARDS [9] basically consists of four main components: (1) acute onset, (2) chest radiography findings, (3) arterial blood gas results (PaO2/FiO2 ratio), and (4) the absence of cardiogenic pulmonary edema. Thus, the presence of cardiogenic pulmonary edema while diagnosing ARDS should be ruled out. The Berlin definition [10] panel agreed, in their conceptual model, of ARDS being a type of acute, diffuse, inflammatory lung injury leading to an increased pulmonary vascular permeability, increased lung weight, and loss of aerated lung tissue. Despite being the hallmarks of ARDS, none of the suggested criteria evaluated the increase in pulmonary vascular permeability and increased EVLW.
In a supplemental publication of the Berlin definition [10], expert panels (the ARDS Definition Task Force) presented typical examples of 12 chest radiographs, categorized into three groups, namely, consistent with, inconsistent with, and equivocal for ARDS. However, the interpretation of chest radiography is complicated and lacks objectivity. Sjoding et al. [8] recently reported clinicians showing only moderate interobserver agreement when diagnosing ARDS in patients with hypoxic respiratory failure under the Berlin criteria. The results were driven primarily by the low reliability of the interpretation of chest images [8]. This conclusion was supported by a recent multicenter prospective study of the inter-rater agreement, where 286 intensivists independently reviewed the same 12 chest radiographs developed by the panels, pre- and post-training. In the study, when the Berlin radiographic definition was used, radiographic diagnostic accuracy and inter-rater agreement were found to be poor and were not significantly improved by the training set of chest radiographs developed by the ARDS Definition Task Force [7].
Therefore, without any objective methods, the precise investigation of pulmonary edema regarding its existence, severity, and the nature of the disease (cardiogenic versus non-cardiogenic) is difficult.
4 Transpulmonary Thermodilution-Derived Extravascular Lung Water and Pulmonary Vascular Permeability
The last two decades have witnessed the evolution of the transpulmonary thermodilution technique for measuring EVLW and pulmonary vascular permeability index (PVPI). The details of the transpulmonary thermodilution technique were described elsewhere (Chap. 7).
The transpulmonary thermodilution-derived variables EVLW and PVPI are sensitive, specific, and conceptual markers for evaluating pulmonary edema [11,12,13,14,15,16,17,18]. The accuracy of the EVLW measurement was first validated against the gold standard gravimetric measurement in animal models [19]. The thermodilution measurement of EVLW values showed high accuracy in the normal lungs, cardiogenic pulmonary edema, and ARDS models. In a human autopsy study, a definite correlation was observed between EVLW and post-mortem lung weight from a wide range of normal and injured lungs [11]. More recently, Venkateswaran et al. [20] reported a close correlation of the EVLW with gravimetric measurements of lung water in human brain-dead donors. The most reliable pathophysiological feature of ARDS is the development of diffuse alveolar damage (DAD) with increased permeability [4], which results in the accumulation of water in the lungs, and designated as EVLW. We validated this relationship between EVLW and DAD in a pathological study using nationwide autopsy database [16].
Several studies suggest a normal EVLW value of 7 mL/kg and not exceeding 10 mL/kg (indexed by predicted body weight). Our clinical-pathological study showed the mean EVLW values of approximately 7.3 ± 2.8 mL/kg to be the normal reference range for humans [11]. This value was supported by Eichhorn et al. in a meta-analysis of clinical studies where they found a mean EVLW of 7.3 mL/kg (95% confidence interval, 6.8–7.6) in patients undergoing elective surgery, without pulmonary edema [21]. More recently, Wolf et al. [22] obtained a similar result (8 mL/kg, interquartile range of 7–9) in 101 elective brain tumor surgery patients.
Practical Advice
The normal reference range of extravascular lung water index for human is approximately 7 ± 3 mL/kg.
In addition, Japanese nationwide autopsy data (n = 1688) indicated that an EVLW of >9.8 mL/kg represented the optimal discrimination threshold for a diagnosis of pulmonary edema from the normal lungs, and an EVLW level of 14.6 mL/kg represents a 99% positive predictive value [16]. Several experts have proposed the inclusion of EVLW >10 mL/kg as an ideal criterion in a future definition of ARDS [23, 24]. According to pathological [16] and clinical [25] studies, EVLW values above 10 mL/kg represent higher-than-normal EVLW, and 15 mL/kg is the cutoff for severe pulmonary edema. By assessing EVLW, we can objectively evaluate the initial severity of pulmonary edema and subsequent changes quantitatively, thereby monitoring the ongoing therapeutic strategy.
Practical Advice
Extravascular lung water index of >10 mL/kg is a reasonable criterion for pulmonary edema and >15 mL/kg for a high degree of severity.
PVPI allows the distinction between the pulmonary edemas [19, 26] and can be calculated from the relationship between EVLW and pulmonary blood volume (Fig. 14.3). If the EVLW is elevated without a corresponding increase in PVPI, the patient has cardiogenic pulmonary edema. However, an increase in EVLW along with an increase in PVPI corresponds to the patient having permeability pulmonary edema. Previous studies indicated the use of PVPI to differentiate cardiogenic and non-cardiogenic (ARDS) pulmonary edema [27, 28]. PVPI, along with EVLW, correlates with the level of a biological mediator which is related to the increased pulmonary vascular permeability and the accumulation of lung water [13, 29].
Pulmonary vascular permeability index (PVPI). PVPI is calculated as the ratio of extravascular lung water (EVLW) and pulmonary blood volume. For example, if the EVLW is elevated without a corresponding increase in PVPI, the patient has cardiogenic, pulmonary edema. In contrast, an increase in EVLW along with an increase in PVPI means that the patient has ARDS
Monnet et al. [28] first showed the differentiation of hydrostatic pulmonary edema from permeability pulmonary edema, with a cutoff PVPI value of 3. A large-scale, prospective multicenter study from Japan found almost the same results, with a PVPI cutoff value between 2.6 and 2.85 (specificity, 0.90 and 0.95, respectively) providing a definitive diagnosis of ARDS, and a value of <1.7 (specificity, 0.95) ruled out an ARDS diagnosis [27]. Another study evaluating patients with either normal cardiac function or chronic cardiac dysfunction found a PVPI of <3 in all studied patients [30]. Collectively, PVPI <2 may represent normal pulmonary permeability, and PVPI >3 indicates high permeability of the lungs.
Practical Advice
Pulmonary vascular permeability index >3 suggests increased vascular permeability. Pulmonary vascular permeability index <2 can rule out high vascular permeability.
Several clinical studies conducted with ARDS patients suggest the correlation of both EVLW and PVPI with the disease severity [31] and risk factors of mortality [15, 32]. The landmark study by Sakka et al. [31] showed the degree of initial EVLW on admission to the intensive care unit correlated with mortality, with a significant cutoff point of 14 mL/kg. The relationship between EVLW and prognosis was also clearly demonstrated in a systematic review of literature [32] and a recent large-scale study [15]. The results of our multicenter study suggested the decrease in EVLW during the first 48 h associated with a 28-day survival in ARDS [33]. Therefore, the initial absolute value of EVLW is useful for the diagnosis of ARDS, along with the subsequent changes in clinical practice [33].
5 Diagnostic Framework for Pulmonary Edema Using Extravascular Lung Water and Pulmonary Vascular Permeability Index
Accurate and objective diagnoses can be made for pulmonary edema using the following diagnostic framework by EVLW and PVPI (Fig. 14.4) [24]. For diagnosing the existence of pulmonary edema, EVLW >10 mL/kg may be reasonable. EVLW >15 mL/kg indicates severe pulmonary edema. After quantitative diagnosis as pulmonary edema by EVLW >10 mL/kg, PVPI should be examined. PVPI <2 represents normal pulmonary permeability, suggesting cardiogenic pulmonary edema. PVPI >3 (with EVLW >10 mL/kg) represents permeability pulmonary edema or ARDS. PVPI >3 and EVLW >15 suggest severe ARDS. Even though the initial EVLW and PVPI are high and indicate a high probability of mortality, with the improvement in values over time (especially during the first 48 h), there may be hope for a better outcome.
Diagnostic framework for pulmonary edema. Pulmonary edema: extravascular lung water (EVLW) >10 mL/kg. Cardiogenic pulmonary edema: EVLW >10 mL/kg and pulmonary vascular permeability index (PVPI) <2.0. ARDS: EVLW >10 mL/kg and PVPI >3.0. Combined pulmonary edema (e.g., cardiogenic pulmonary edema, reduced cardiac function or fluid overload, and permeability lung injury secondary to the generation of inflammatory mediators): EVLW >10 mL/kg and PVPI of 2.0–3.0. (Reproduced from [24] with permission)
6 Conclusions
The transpulmonary thermodilution-derived variables EVLW and PVPI can quantitatively express its existence, severity, and the nature of pulmonary edema at the bedside. The accuracy is validated compared to the gold standard method, and the precision is clinically acceptable. EVLW >10 mL/kg is a reasonable criterion for inferring the existence of pulmonary edema and EVLW >15 mL/kg for a severe condition. PVPI <2 may represent normal pulmonary permeability, and PVPI >3 suggests leaky lungs. EVLW and PVPI may be better alternatives to define management algorithms for pulmonary edema patients.
Keynotes
-
Extravascular lung water and pulmonary vascular permeability index can be measured by the transpulmonary thermodilution technique.
-
Extravascular lung water index of >10 mL/kg is a reasonable criterion for pulmonary edema and extravascular lung water index of >15 mL/kg for a high degree of severity.
-
In addition to extravascular lung water index of >10 mL/kg, pulmonary vascular permeability index of >3 suggests an increased vascular permeability (i.e., ARDS), and pulmonary vascular permeability index <2 represents normal vascular permeability (i.e., cardiogenic pulmonary edema).
References
Sweeney RM, McAuley DF. Acute respiratory distress syndrome. Lancet. 2016;388(10058):2416–30.
Ware LB, Matthay MA. Clinical practice. Acute pulmonary edema. N Engl J Med. 2005;353(26):2788–96.
Edoute Y, Roguin A, Behar D, Reisner SA. Prospective evaluation of pulmonary edema. Crit Care Med. 2000;28(2):330–5.
Corrin B, Nicholoson A. Pathology of the lungs. Edinburgh: Churchill Livingstone; 2011.
Assaad S, Kratzert WB, Shelley B, Friedman MB, Perrino A Jr. Assessment of pulmonary edema: principles and practice. J Cardiothorac Vasc Anesth. 2018;32(2):901–14.
Meade MO, Cook RJ, Guyatt GH, Groll R, Kachura JR, Bedard M, et al. Interobserver variation in interpreting chest radiographs for the diagnosis of acute respiratory distress syndrome. Am J Respir Crit Care Med. 2000;161(1):85–90.
Peng JM, Qian CY, Yu XY, Zhao MY, Li SS, Ma XC, et al. Does training improve diagnostic accuracy and inter-rater agreement in applying the Berlin radiographic definition of acute respiratory distress syndrome? A multicenter prospective study. Crit Care. 2017;21(1):12.
Sjoding MW, Hofer TP, Co I, Courey A, Cooke CR, Iwashyna TJ. Interobserver reliability of the Berlin ARDS definition and strategies to improve the reliability of ARDS diagnosis. Chest. 2018;153(2):361–7.
Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, Fan E, et al. Acute respiratory distress syndrome: the Berlin definition. JAMA. 2012;307(23):2526–33.
Ferguson ND, Fan E, Camporota L, Antonelli M, Anzueto A, Beale R, et al. The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med. 2012;38(10):1573–82.
Tagami T, Kushimoto S, Yamamoto Y, Atsumi T, Tosa R, Matsuda K, et al. Validation of extravascular lung water measurement by single transpulmonary thermodilution: human autopsy study. Crit Care. 2010;14(5):R162.
Monnet X, Persichini R, Ktari M, Jozwiak M, Richard C, Teboul JL. Precision of the transpulmonary thermodilution measurements. Crit Care. 2011;15(4):R204.
Tagami T, Kushimoto S, Tosa R, Omura M, Yonezawa K, Akiyama G, et al. Plasma neutrophil elastase correlates with pulmonary vascular permeability: a prospective observational study in patients with pneumonia. Respirology. 2011;16(6):953–8.
Zhang Z, Lu B, Ni H. Prognostic value of extravascular lung water index in critically ill patients: a systematic review of the literature. J Crit Care. 2012;27(4):420.e1–8.
Jozwiak M, Silva S, Persichini R, Anguel N, Osman D, Richard C, et al. Extravascular lung water is an independent prognostic factor in patients with acute respiratory distress syndrome. Crit Care Med. 2013;41(2):472–80.
Tagami T, Sawabe M, Kushimoto S, Marik PE, Mieno MN, Kawaguchi T, et al. Quantitative diagnosis of diffuse alveolar damage using extravascular lung water*. Crit Care Med. 2013;41(9):2144–50.
Gavelli F, Teboul JL, Azzolina D, Beurton A, Taccheri T, Adda I, et al. Transpulmonary thermodilution detects rapid and reversible increases in lung water induced by positive end-expiratory pressure in acute respiratory distress syndrome. Ann Intensive Care. 2020;10(1):28.
Tagami T, Kushimoto S, Tosa R, Omura M, Hagiwara J, Hirama H, et al. The precision of PiCCO measurements in hypothermic post-cardiac arrest patients. Anaesthesia. 2012;67(3):236–43.
Katzenelson R, Perel A, Berkenstadt H, Preisman S, Kogan S, Sternik L, et al. Accuracy of transpulmonary thermodilution versus gravimetric measurement of extravascular lung water. Crit Care Med. 2004;32(7):1550–4.
Venkateswaran RV, Dronavalli V, Patchell V, Wilson I, Mascaro J, Thompson R, et al. Measurement of extravascular lung water following human brain death: implications for lung donor assessment and transplantation. Eur J Cardiothorac Surg. 2013;43(6):1227–32.
Eichhorn V, Goepfert MS, Eulenburg C, Malbrain ML, Reuter DA. Comparison of values in critically ill patients for global end-diastolic volume and extravascular lung water measured by transcardiopulmonary thermodilution: a meta-analysis of the literature. Med Intensiva. 2012;36(7):467–74.
Wolf S, Riess A, Landscheidt JF, Lumenta CB, Schurer L, Friederich P. How to perform indexing of extravascular lung water: a validation study. Crit Care Med. 2013;41(4):990–8.
Michard F, Fernandez-Mondejar E, Kirov MY, Malbrain M, Tagami T. A new and simple definition for acute lung injury. Crit Care Med. 2012;40(3):1004–6.
Tagami T, Ong MEH. Extravascular lung water measurements in acute respiratory distress syndrome: why, how, and when? Curr Opin Crit Care. 2018;24(3):209–15.
Sakka SG, Klein M, Reinhart K, Meier-Hellmann A. Prognostic value of extravascular lung water in critically ill patients. Chest. 2002;122(6):2080–6.
Kirov MY, Kuzkov VV, Kuklin VN, Waerhaug K, Bjertnaes LJ. Extravascular lung water assessed by transpulmonary single thermodilution and postmortem gravimetry in sheep. Crit Care. 2004;8(6):R451–8.
Kushimoto S, Taira Y, Kitazawa Y, Okuchi K, Sakamoto T, Ishikura H, et al. The clinical usefulness of extravascular lung water and pulmonary vascular permeability index to diagnose and characterize pulmonary edema: a prospective multicenter study on the quantitative differential diagnostic definition for acute lung injury/acute respiratory distress syndrome. Crit Care. 2012;16(6):R232.
Monnet X, Anguel N, Osman D, Hamzaoui O, Richard C, Teboul JL. Assessing pulmonary permeability by transpulmonary thermodilution allows differentiation of hydrostatic pulmonary edema from ALI/ARDS. Intensive Care Med. 2007;33(3):448–53.
O'Kane CM, McKeown SW, Perkins GD, Bassford CR, Gao F, Thickett DR, et al. Salbutamol up-regulates matrix metalloproteinase-9 in the alveolar space in the acute respiratory distress syndrome. Crit Care Med. 2009;37(7):2242–9.
Hilty MP, Franzen DP, Wyss C, Biaggi P, Maggiorini M. Validation of transpulmonary thermodilution variables in hemodynamically stable patients with heart diseases. Ann Intensive Care. 2017;7(1):86.
Kushimoto S, Endo T, Yamanouchi S, Sakamoto T, Ishikura H, Kitazawa Y, et al. Relationship between extravascular lung water and severity categories of acute respiratory distress syndrome by the Berlin definition. Crit Care. 2013;17(4):R132.
Huber W, Findeisen M, Lahmer T, Herner A, Rasch S, Mayr U, et al. Prediction of outcome in patients with ARDS: a prospective cohort study comparing ARDS-definitions and other ARDS-associated parameters, ratios and scores at intubation and over time. PLoS One. 2020;15(5):e0232720.
Tagami T, Nakamura T, Kushimoto S, Tosa R, Watanabe A, Kaneko T, et al. Early-phase changes of extravascular lung water index as a prognostic indicator in acute respiratory distress syndrome patients. Ann Intensive Care. 2014;4:27.
Conflict of Interest
Takashi Tagami is a member of the Medical Advisory Board of Pulsion/Gettinge.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Tagami, T. (2021). Extravascular Lung Water. In: Kirov, M.Y., Kuzkov, V.V., Saugel, B. (eds) Advanced Hemodynamic Monitoring: Basics and New Horizons. Springer, Cham. https://doi.org/10.1007/978-3-030-71752-0_14
Download citation
DOI: https://doi.org/10.1007/978-3-030-71752-0_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-71751-3
Online ISBN: 978-3-030-71752-0
eBook Packages: MedicineMedicine (R0)