Abstract
Mycotoxins are highly toxic secondary metabolites produced by fungi, which may contaminate a large variety of food and feed commodities. Among them, the aflatoxins, deoxynivalenol, zearalenone, fumonisins, and ochratoxins are the most common contaminants posing a serious threat to human and animal health.Given that traditional mycotoxin detection methods have been shown to be laborious and time consuming, interest in developing reliable and rapid mycotoxin detection methods has increased during recent decades. Herein, we review emerging nanotechnology-based methods, including gold nanoparticles, magnetic nanoparticles, and quantum dot-based sensors, which have been developed to reliably and efficiently detecting mycotoxins in food and feed commodities. We also summarize recent technologies used to remove mycotoxins via adsorption and photocatalytic degradation. As our review illustrates, the emerging use of nanotechnology offers a reliable and cost-effective means to prevent mycotoxin contamination in food and feed commodities, which could reduce health risks to consumers.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Mycotoxins
- Food contamination
- Nanotechnology
- Nanosensors
- Gold nanoparticles
- Quantum dots
- Magnetic nanoparticles
- Photocatalytic degradation
9.1 Introduction
Mycotoxins are highly toxic secondary metabolites produced by fungi. They can invade a wide variety of agricultural crops while still on the field or afterwards during their processing in food and feed production chains. The presence of mycotoxins in agricultural commodities is high, with an estimated 25% of the world’s crops being contaminated by molds or fungi, especially toxigenic species that belong to the genera Penicillium, Aspergillus, and Fusarium (Alshannaq and Yu 2017). At present, about 175 different mycotoxins have been identified in the food and feed commodities grown in outdoor and indoor environments (Bhat et al. 2010). Given their prevalence, mycotoxins have become a health hazard to both humans and animals worldwide (Fig. 9.1).
Based on possible dietary exposures together with their toxicity, aflatoxins (AFs), citrinin, patulin, ochratoxins A (OTA), zearalenone (ZEA), fumonisins, and trichothecenes are of a high concern due to their known health effects (Santos Pereira et al. 2019; Smith et al. 2016). Typical structures, source of contamination, food products contamination, health effects on mammalian system, and country-wise maximum tolerable levels of a wide range of mycotoxins are represented in Table 9.1. Risk assessments of mycotoxins in food are governed by the Joint Expert Committee of the Food and Agriculture Organization (JECFA) of the United Nations (FAO) and World Health Organization (WHO). The international standards are established by the Codex Alimentarius Commission, which lists maximum levels for various mycotoxins in foods (Jukes 2000; Organization 1999).
The traditional methods of mycotoxin analysis involve more complex sample preparation and longer detection times combined with the usage of highly sophisticated analytical instruments (Vidal et al. 2013; Zhang et al. 2018a). Nanotechnology has gone through a booming development since 1981. The usage of nanoparticles in sensing applications are accurate with high precision resulting in signal amplification due to their unique characteristics, such as high surface to volume ratio (Tothill 2011). Further the integration of nanomaterials not only enhances the sensitivity in detection of toxins, but they are highly selective, cost effective, and portable and a rapid response can be obtained (Malhotra et al. 2014). A wide-range of nanomaterials comprising gold nanoparticles , silver nanoparticles , quantum dots (QDs), magnetic nanoparticles, graphene oxide, carbon nanohorns, carbon nanotubes (CNTs), and TiO2 nanoparticles , have been used for the detection of different kinds of mycotoxins.
Till date many reviews have been published in sensing and detection of mycotoxins using nanotechnology (Goud et al. 2018; Horky et al. 2018; Ingle et al. 2020; Zeng et al. 2016). This is a comprehensive review including the various sensing applications with some of extensively used nanoparticles , such as gold, QDs, and magnetic nanoparticles. Here, we review advances from the last 5 years in the use of nanomaterials for the detection of mycotoxins in food and agriculture. This review also summarizes the recent technologies employed for the removal and the photocatalytic degradation and removal of mycotoxins. A literature search for the years from 2014 to 2019 was conducted in the PubMed (https://www.ncbi.nlm.nih.gov) and Scopus (https://www.scopus.com) databases using the keywords “nanoparticles , mycotoxin detection .” Additionally, three categories of nanomaterials were searched specifically: “gold nanoparticles ,” “magnetic nanoparticles,” and “quantum dots.”
9.2 Nano-sensors for the Detection of Mycotoxins
At present, there is an increasing attention on nanotechnology since it has generated novel advantages across the range of areas comprising the food industry. Accordingly, nanotechnology has revealed new tasks for novelty in food production at a quick rate. Quality and safety of foods are a key component of public health, and with growing public awareness, customers are now demanding foodstuffs that are devoid of any contaminants (Hamad et al. 2018; Pal 2017; Pathakoti et al. 2019; Pradhan et al. 2015).
Due to their staggeringly small size, nanomaterials show specific physical and chemical features. Nanosensors are described as any chemical, biological, or surgical sensory point utilized to expose the nanoparticles to the microscopic world (Yu et al. 2018). Nanosensors utilize several nanomaterials that can identify toxins in sustenance at precise low levels, in the midst of handling or processing of foods (Sonawane et al. 2014; Willner and Vikesland 2018). Thus, in nanosensors, nanomaterials are employed in an analytical device, which is an improved version of a chemical sensor or biosensor . A biosensor works on two basic principles, biological recognition, and sensing. In general, nanosensors or nanobiosensors enhance at the activity by the integration of nanostructures in the sensing component for enhanced output. The basic structure of nanosensor and working principal is schematically shown in Fig. 9.2. These nanostructures enhance the activity both at bioreceptor level and the transducer level. Moreover, the transducer aids in fabrications of sensor with the use of various nanoparticles , such as metal oxide nanoparticles , quantum dots, magnetic nanoparticles, and carbon nanoparticles . Due to their staggeringly small size, nanomaterials show specific physical and chemical features, such as superior optical, electrical, thermal properties, and high surface area, which improves the transducing capability to a large extent. Based on signal production, that is, based on transducer type, nanosensors can be three major types: (1) optical: as optical (colorimetry, fluorescence, luminescence, surface-enhanced Raman scattering (SERS), surface plasmon resonance (SPR ) and others), (2) electrochemical (amperometry, conductimetry, potentiometry, and voltammetry), and (3) Piezoelectric (quartz crystal microbalance).
Schematic representation of nanosensor and its working. modified from Goud et al. (2018), Copyright (2018) Elsevier
9.2.1 Gold Nanoparticles for the Detection of Mycotoxins
A variety of biochemical and electrochemical assays based on Au nanoparticles have been described for the detection of mycotoxins. The most important and advantageous properties of gold nanoparticles for their usage in designing the sensors to detect toxins include localized surface plasmon resonance (SPR) bands and enhanced scattering properties (Lara and Perez-Potti 2018). SPR imaging (SPRi) is an evolving label free semi-quantitative method for the sensitive detection of mycotoxins, which is highly reliable (Joshi 2017). Hossain and Maragos Hossain and Maragos (2018) described a gold nanoparticle based SPRi for the detection of Fusarium toxins such as DON, ZEA, and T-2 in wheat samples. This biosensor was amplified more than 12–90-fold with use of secondary antibody Ab2 with Au nanoparticles . Further, these antigen coated biochips are highly durable with short analysis time, are cost effective, and can be used for at least 46 cycles. In another study, Kong et al. (2016) have developed an Au nanoparticle-based immunochromatographic strip system for the detection of 20 different types of mycotoxins including ZEAs, DONs, T-2 toxins, AFs, and fumonisins (Fig. 9.3).
(A) Immunochromatographic strip structure (B) Negative sample (C) Positive sample; c1: ZEAs; c2: DONs; c3: T-2s Toxin; c4: Aflatoxins; c5: Fumonisin B. Copyright (2016) Elsevier, (Kong et al. 2016)
Similarly, AFs including B1, B2, M1, G1, and G2 in sustenance were identified depending upon a lateral flow strip (Santos et al. 2017). A monoclonal antibody (mB6 mAb), which has high specificity to AFs, was created to develop an immunochromatographic strip test, where the gold nanoparticles were coated with this antibody for detection . This strip consists of a control and test line, where the control line is coated with goat antimouse IgG and the test line containing AFB1 bovine serum albumin. In the strip, red gold nanoparticles form 1–2 pigmented lines on the membrane and the nanoparticles coated with anti-AFB1 as the signaling reagent for basic identification of AFB1.
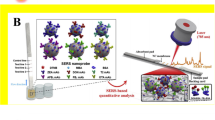
A surface-enhanced Raman scattering (SERS) aptamer-based sensor was developed by using the gold@gold-silver nanostructures for the selective detection of OTA (Fig. 9.4). Magnetic nanoparticles (Fe3O4) were used for separation of toxin from solution, which is reusable. After magnetic separation, Raman signals were collected and this SERS probe can be used for the real time detection of OTA (Shao et al. 2018). Table 9.2 represents a wide range of methodology for sensor development using gold nanomaterials for the detection of different types of mycotoxins.
Synthesis of Au@AuAg NNSs (A). Schematic illustration of SERS aptsensor-based on Au@AuAg NNSs-magnetic nanoparticles for OTA detection . Copyright (2018) Elsevier (Shao et al. 2018)
9.2.2 Magnetic Nanoparticles for the Detection of Mycotoxins
Superparamagnetism is one of the most important properties of magnetic nanoparticles. The superparamagnetic nanoparticles can be easily dispersed in aqueous solution, remain stable when coated with an appropriate layer, and are able to form ferrofluid. Wide ranges of applications of superparamagnetic nanoparticles are reported. Various methods of detection of mycotoxins by employing magnetic nanoparticles as sensors are represented in Table 9.3. A study by Gan et al. (2013) described the electrochemiluminescent immunoassay for the detection of mycotoxin , aflatoxin-M1 in milk samples. In their study, magnetic nanocomposites were prepared initially by immobilization of Fe3O4 nanoparticles onto graphene oxide, which is used as an adsorbent. Later on, aflatoxin-M1 antibody was attached to cadmium telluride quantum dots, which can be used as a signal tag. Finally, magnetic nanocomposites were used for the separation of aflatoxin-M1 from milk samples and detected using an immunoassay using an electrochemiluminescent signal. Further, this immunosensor is stable and can be regenerated after storing for two weeks at 4°C and its accuracy was about 95%.
9.2.3 Quantum Dots for the Detection of Mycotoxins
Physicochemical properties including enhanced fluorescence, narrow emission peaks, and high photostability of QDs are advantageous in developing the sensors for highly sensitive detection of mycotoxins. Some of the recent applications of quantum dots for mycotoxin detection are presented in Table 9.4. Fang et al. (2014) developed a novel molecularly imprinted optosensing material for selective detection of mycotoxin , ZON in cereal samples. The molecular imprinted polymer is based on ionic liquid stabilized cadmium selenium/ZnS QDs and the mechanism is based on fluorescence quenching.
A highly sensitive aptasensor for the detection of ochratoxin was developed with a combination of nanocomposites consisting of gold nanoparticles functionalized with silica coated Fe3O4 magnetic nanoparticles and CdTe QDs modified with graphene/gold nanoparticles . This novel aptasensor is ultrasensitive with detection limit at sub-picomolar level (Hao et al. 2016).
Zhang et al. (2017a) developed an optical sensor based on molecular imprinted polymer capped Mn-doped ZnS QD for detection of the mycotoxin , patulin, from apple juice. This nanosensor is based on a phosphorescence method, for determination of patulin from aqueous solutions, including the intended matrix, apple juice. Recently, Duan et al. (2019) developed multicolor CdSe/ZnS QD nanobeads with yellow, orange, and red luminescence for concurrent recognition of numerous mycotoxins such as ZEA, OTA, and fumonisin B1 from a corn matrix (Fig. 9.5). This is a qualitative method based on an immunochromatographic assay for visual detection of mycotoxins, which can be inferred with the naked eye under UV light with color differentiation.
Scheme 1. (a) Schematic representation for the synthesis of tricolor QB via using the emulsification evaporation method. (b) Schematic illustration of developed tricolor QB-based mICA, in which three T lines of T1, T2, and T3 were designed for the simultaneous detection of ZEN, OTA, and FB1, while one C line was set to indicate the validity of developed mICA, respectively. (C) Schematic presentation for the interpretation of test results. 1, negative; 2, ZEN (−), OTA (−), FB1 (+); 3, ZEN (−), OTA (+), FB1 (−); 4, ZEN (+), OTA (−), FB1 (−); 5, ZEN (−), OTA (+), FB1 (+); 6, ZEN (+), OTA (−), FB1 (+); 7, ZEN (+), OTA (+), FB1 (−); 8, ZEN (+), OTA (+), FB1 (+); 9, invalid. (“+” and “−” indicate the “positive” and “negative,” respectively). Copyright from (2019) Elsevier (Duan et al. 2019)
9.2.4 Miscellaneous Nanomaterials Employed in Developing Sensors for the Detection of Mycotoxins
Zinc oxide nanoparticles are recognized for applications in immunosensing, due to their distinct characteristics, such as a high isoelectric point and higher binding energy and moreover being cost-effective and biocompatible (Ansari et al. 2010). For OTA detection , ZnO nanofilm was immobilized with rabbit immunoglobulin antibodies placed onto glass plate made of Indium tin oxide and bovine serum albumin for blocking nonspecific binding of OTA. This nanozinc immunoelectrode has amplified electrochemical signal and has an application for OTA and other mycotoxins (Ansari et al. 2010).
Similar to ZnO nanoparticles , nanostructured CeO2 is biocompatible due to its non-toxicity and has attracted interest in development of biosensors. In addition, nano-CeO2 has high chemical stability and mechanical strength. Kaushik et al. (2009a) described the fabrication of chitosan-nano-CeO2 nanocomposites film deposited on indium tin oxide-coated glass substrate for the immobilization of r-IgGs and BSA. This immunoelectrode has a large surface, including greater electron transport and potential application for OTA detection and other mycotoxins such as AFB1 and citrining.
Likewise, a study by Kaushik et al. (2009b) reported the detection of OTA by integrating nanosilicon-chitosan nanocomposites film for r-IgGs loading, and this immunoelectrode has enhanced sensing properties. This could be due to the presence of chitosan-nano-SiO2 composites due to greater surface area and higher electrochemical behavior. Similarly, a chitosan-TiO2-nanoparticle-based immunosensor was developed for the recognition of OTA from Aspergillus ochraceus (Khan and Dhayal 2008). Electrochemical impedance spectroscopy (EIS) was used to determine the electroactivity of the bioactive electrode. This matrix has several advantages in biosensor application . Chitosan is cost-effective and has higher mechanical strength and permeability. TiO2 provides the biocompatibility and thereby provides longer life and improved stability to the electrode. This technique of conjugating chitosan with nanoparticles also provided the fastest response (25 s) with the lowest detection limit.
Recently, Goftman et al. (2016) synthesized the silica coated CdSe quantum dots to detect DON by the microemulsion method. Further, to increase the bioapplicability of QD@SIO2 nanoparticles , they were modified using three diverse functional groups including amino, carboxyl, and epoxy groups and polyethylene glycol fragments. This developed fluorescence-labeled immunosorbent assay was used for rapid detection of mycotoxins, especially DON.
Carbon nanotubes (CNTs) are allotropes of carbon with a cylindrical nanostructure (Saifuddin et al. 2013). Among them, single-walled CNTs (SWCNTs) due to their exceptional characteristics such as greater surface area, high electrical conductivity, and mechanical strength provide a compatible environment to preserve the enzyme activity, thereby enhancing the electrochemical signal amplification (Guo 2013; Singh et al. 2009). Zhang et al. (2016) fabricated an electrochemical immunosensor for detection of AFB1 from corn flour using the SWCNTs and chitosan composites. As SWCNTs are not water soluble, addition of chitosan enhanced the dispersion of SWCNTs. This immunosensor was centered on an indirect competitive assay using the primary antibody anti-AFB1 and AFB1-BSA immobilized on glass carbon electrode.
9.3 Remediation of Mycotoxins From Food and Agriculture Using Nanotechnology
9.3.1 Removal of Mycotoxins by Using Adsorption
Traditional enzyme-based decontaminant methods for the removal of mycotoxins from food have several drawbacks, such that the toxin may remain in the food matrix or that it can also result in highly toxic secondary metabolites (Jard et al. 2011; Manubolu et al. 2018). Although nanotechnology has advanced substantially in the detection of mycotoxins, very few studies are available on the elimination or removal of mycotoxins from various food matrices. Among the various immobilization approaches, magnetic nanoparticles are the best solid carriers due to their exclusive high saturation magnetization for facile separation and reusability (Ansari and Husain 2012). Some of the recent applications for removal of mycotoxins from various biological matrices are described in this section (Table 9.5).
Surface active maghemite nanoparticles (SAMNS Fe2O3, 11 ± 2 nm) were used for the removal of citrinin in Monascus treated foods, which can be used in the food industry (Magro et al. 2016). The SAMN@citrinin conjugate complex was thoroughly characterized structurally and was magnetically isolated. The offered magnetic separation system was based on the citrinin binding on SAMNS surface and is based on the firm iron chelating keto-enol group on the toxin. In another study, Luo et al. (2017) reported the synthesis of chitosan coated Fe3O4 nanoparticles as a magnetic adsorbent for the patulin removal from fruit juice. Further, magnetic chitosan Fe3O4 nanoparticles coated with deactivated Candida utilis cells and used as a new biosorbent for patulin removal from fruit juice (Ge et al. 2017). Similarly, for patulin removal, magnetic multi-walled carbon nanotube (MWCNT) adsorbent was fabricated, which can be regenerated using NaOH solution for recycling usage up to four cycles (Zhang et al. 2019).
9.3.2 Degradation of Mycotoxins Using Nanotechnology
Recently, there has been a growing interest in photocatalytic degradation of many organic pollutants, including mycotoxins. Photocatalytic degradation has numerous advantages, such as being environmentally friendly, low–cost, and requiring mild conditions (Bhatkhande et al. 2002). Various photocatalysts, both under the UV light and visible light irradiation, are presented in Table 9.5. Nevertheless, as UV light accounts for only 4% of the total sunlight, visible light photocatalysts are more beneficial for practical degradation of organic pollutants in natural systems (Pathakoti et al. 2013, 2018).
Bai et al. (2017) reported the synthesis of graphene/ZnO hybrids using a single one-step hydrothermal method for the photodegradation of DON under UV irradiation. As shown in Scheme 1a, the proposed photodegradation mechanism is due to the formation of superoxide radicals at the conduction band. It is recognized that the graphene increases the adsorption capability and the ZnO increases the photocatalytic achievement by graphene hybridization (Fig. 9.6). Additionally, the degradation products of DON were analyzed by using ESI/MS analysis.
Schematic drawing illustrating synthetic route and the mechanism of charge separation and adsorption-photocatalytic process over graphene/ZnO hybrid photocatalysts under UV light irradiation. Obtained with permission from Elsevier, Ref: (Bai et al. 2017)
Recently, Mao et al. (2019) reported the photocatalytic inactivation of AFB1 at hypertoxic site C8=C9 under visible light treatment using the Z-schematic composite, clew-like WO3 adorned with CdS nanoparticles , synthesized by the microwave supported hydrothermal and precipitation procedure (Fig. 9.7). The reaction of hydroxyl radical formation with C8=C9 bond of AF for the photocatalytic inactivation was confirmed by radical trapping experiments and 18O isotope labeling. Further, the degraded products were identified by using High Resolution Mass Spectroscopy, and the theoretical calculations using the density functional theory were used to confirm the formation of hydroxyl radicals at the C9 site, thereby forming AFB1-9-hydroxy.
Illustration of the proposed mechanism over CdS/WO3 composites for inactivating the hypertoxic site [(C8=C9) in AFB1 under visible light irradiation. Obtained with permission from Elsevier, Ref. (Mao et al. 2019)
Similarly, another study reported the 10 mole% Scandanium doped SrTi0.7Fe0.3O3 (band gap, 1.58 eV) was synthesized for the photodegradation of afltoxinB1 under visible light irradiation (Jamil et al. 2017). Further, the degraded products were identified using gas chromatography-mass spectrometry (GC-MS), which are non-toxic to Vibrio fischeri organism. Hence, it is safe for water treatment, and moreover, both the catalyst and treated water can be reused for up to eight times. The mechanism of oxidation is due to rupture of short chain aliphatic alcohol, which leads to total degradation.
A study by Magzoub et al. (2019) reported the photocatalytic detoxification of AFB1 and AFB2 from its real sample matrix, peanut oil, using the TiO2 that was immobilized on a glass support under high pressure UV–vis irradiation. It is noteworthy that the TiO2 treatment did not alter the physicochemical parameters of the oil, such as iodine value, peroxide value, saponification value, fatty acid composition, and free fatty acids. Furthermore, the photocatalyst can be recycled up to ten cycles without reducing its effectiveness. The proposed mechanism is the formation of hydroxyl radicals and is comparable to previous reports (Jamil et al. 2017; Mao et al. 2019).
Likewise, Xu et al. (2019) reported the closed loop photocatalytic reactor comprising a glass tube holding TiO2 catalyst for the decontamination of AFB1 from peanut oil. Based on their experimental data on AF detoxification, a theoretical model, a Weibull distribution model, has been proposed, which offers a good depiction for the photocatalytic process kinetics.
A simple hydrothermal method was described by Sun et al. (2019) for the photocatalytic degradation of AFB1 using an activated charcoal/TiO2 composite under UV–vis irradiation. This newly synthesized composite had higher degradation efficiency when compared to the bare TiO2 and the catalyst can be regenerated and recycled up to four cycles with 80% degradation efficiency. Formation of hydroxyl radicals plays a vital role in the degradation of AF, whereas superoxide radicals do not have an effect.
Another effective approach is the improvement of the photocatalyst, dendritic-like α-Fe2O3, for the DON degradation under visible light irradiation (Wang et al. 2019). A simple hydrothermal has been proposed for dendritic-like α-Fe2O3, which has better photocatalytic activity (more than 90% in 2 h) than the commercial α-Fe2O3. After degradation the intermediate products formed were identified by high perrformance liquid chromatography-mass spectrophotometry (HPLC-MS) and the major toxicity group, epoxy group at C12 and C13 and hydroxyl groups are destroyed in DON, thereby providing an efficient and green technology for mycotoxin decontamination.
9.4 Summary and Conclusion
The occurrence of mycotoxins in the food chain is a major safety concern around the world. Control of mycotoxins commonly depends on the suitable care taken during pre- and post-harvest conditions. The potential and present uses of nanotechnology in farming and food industries offer various significant benefits to ensure the microbial food quality and safe of nourishment items. The use of gold, QDs, and magnetic nanoparticles has significantly enhanced the biosensor technology for the mycotoxin analyses. All the above discussed nano-based sensors developed for detection and sensing of mycotoxins can be easily applied in the fields and also can be operated by unskilled personnel. Further, the sensitive and early detection of mycotoxins in food will be beneficial in eliminating these toxins to enter into food chain and thereby protects human health. Although QDs have various advantages due to their optical properties in comparison to other nanoparticles in sensing applications, but they are not cost-effective and expensive till date. In order to overcome the overestimation, false positive or underestimation, usage of more than one technique such as multiple sensor devices, which can be facile to fulfill the rapid monitoring and control of mycotoxins is essential.
References
Abdul Kadir MK, Tothill IE (2010) Development of an electrochemical immunosensor for fumonisins detection in foods. Toxins 2:382–398
Adányi N et al (2018) Sensitivity enhancement for mycotoxin determination by optical waveguide lightmode spectroscopy using gold nanoparticles of different size and origin. Food Chem 267:10–14. https://doi.org/10.1016/j.foodchem.2018.04.089
Alshannaq A, Yu J-H (2017) Occurrence, toxicity, and analysis of major mycotoxins in food. Int J Environ Res Public Health 14:632. https://doi.org/10.3390/ijerph14060632
Anfossi L et al (2018) A lateral flow immunoassay for straightforward determination of fumonisin mycotoxins based on the quenching of the fluorescence of CdSe/ZnS quantum dots by gold and silver nanoparticles. Microchim Acta 185:94. https://doi.org/10.1007/s00604-017-2642-0
Ansari SA, Husain Q (2012) Potential applications of enzymes immobilized on/in nano materials: a review. Biotechnol Adv 30:512–523. https://doi.org/10.1016/j.biotechadv.2011.09.005
Ansari AA, Kaushik A, Solanki PR, Malhotra B (2010) Nanostructured zinc oxide platform for mycotoxin detection. Bioelectrochemistry 77:75–81
Arévalo FJ, Granero AM, Fernández H, Raba J, Zón MA (2011) Citrinin (CIT) determination in rice samples using a micro fluidic electrochemical immunosensor. Talanta 83:966–973
Authority EFS, Arcella D, Gergelova P, Innocenti ML, Steinkellner H (2017) Human and animal dietary exposure to T-2 and HT-2 toxin. EFSA J 15:e04972
Bai X et al (2017) Photocatalytic degradation of deoxynivalenol using graphene/ZnO hybrids in aqueous suspension. Appl Catal B Environ 204:11–20
Barthelmebs L, Hayat A, Limiadi AW, Marty J-L, Noguer T (2011) Electrochemical DNA aptamer-based biosensor for OTA detection, using superparamagnetic nanoparticles. Sensors Actuators B Chem 156:932–937
Beloglazova N et al (2014) Novel multiplex fluorescent immunoassays based on quantum dot nanolabels for mycotoxins determination. Biosens Bioelectron 62:59–65
Beloglazova NV et al (2016) Sensitive QD@ SiO2-based immunoassay for triplex determination of cereal-borne mycotoxins. Talanta 160:66–71
Beloglazova NV, Sobolev AM, Tessier MD, Hens Z, Goryacheva IY, De Saeger S (2017) Fluorescently labelled multiplex lateral flow immunoassay based on cadmium-free quantum dots. Methods 116:141–148
Bhat R, Rai RV, Karim AA (2010) Mycotoxins in food and feed: present status and future concerns. Compr Rev Food Sci Food Saf 9:57–81
Bhatkhande DS, Pangarkar VG, Beenackers AACM (2002) Photocatalytic degradation for environmental applications. J Chem Technol Biotechnol 77:102–116
Chauhan R, Singh J, Solanki PR, Manaka T, Iwamoto M, Basu T, Malhotra B (2016) Label-free piezoelectric immunosensor decorated with gold nanoparticles: kinetic analysis and biosensing application. Sensors Actuators B Chem 222:804–814
Chehri K, Godini R (2017) Detection of nivalenol and deoxynivalenol chemotypes produced by Fusarium graminearum species complex isolated from barley in Iran using specific PCR assays. J Plant Prot Res 57:237–242
chuan Li S, hua Chen J, Cao H, sheng Yao D, ling Liu D (2011) Amperometric biosensor for aflatoxin B1 based on aflatoxin-oxidase immobilized on multiwalled carbon nanotubes. Food Control 22:43–49
Duan H, Chen X, Xu W, Fu J, Xiong Y, Wang A (2015) Quantum-dot submicrobead-based immunochromatographic assay for quantitative and sensitive detection of zearalenone. Talanta 132:126–131
Duan H, Li Y, Shao Y, Huang X, Xiong Y (2019) Multicolor quantum dot nanobeads for simultaneous multiplex immunochromatographic detection of mycotoxins in maize. Sensors Actuators B Chem 291:411–417. https://doi.org/10.1016/j.snb.2019.04.101
Erdoğan A, Ghimire D, Gürses M, Çetin B, Baran A (2018) Patulin contamination in fruit juices and its control measures. Eur J Sci Technol:39–48
Fang G, Fan C, Liu H, Pan M, Zhu H, Wang S (2014) A novel molecularly imprinted polymer on CdSe/ZnS quantum dots for highly selective optosensing of mycotoxin zearalenone in cereal samples. RSC Adv 4:2764–2771
Flajs D, Peraica M (2009) Toxicological properties of citrinin. Arch Ind Hyg Toxicol 60:457–464
Foubert A, Beloglazova NV, De Saeger S (2017) Comparative study of colloidal gold and quantum dots as labels for multiplex screening tests for multi-mycotoxin detection. Anal Chim Acta 955:48–57. https://doi.org/10.1016/j.aca.2016.11.042
Gan N, Zhou J, Xiong P, Hu F, Cao Y, Li T, Jiang Q (2013) An ultrasensitive electrochemiluminescent immunoassay for Aflatoxin M1 in milk, based on extraction by magnetic graphene and detection by antibody-labeled CdTe quantumn dots-carbon nanotubes nanocomposite. Toxins 5:865–883. https://doi.org/10.3390/toxins5050865
Ge N, Xu J, Li F, Peng B, Pan S (2017) Immobilization of inactivated microbial cells on magnetic Fe3O4@CTS nanoparticles for constructing a new biosorbent for removal of patulin in fruit juice. Food Control 82:83–90. https://doi.org/10.1016/j.foodcont.2017.06.027
Ghali R, Ghorbel H, Hedilli A (2009) Fumonisin determination in tunisian foods and feeds. ELISA and HPLC methods comparison. J Agric Food Chem 57:3955–3960
Goftman VV et al (2016) Synthesis, modification, bioconjugation of silica coated fluorescent quantum dots and their application for mycotoxin detection. Biosens Bioelectron 79:476–481
González-Sálamo J, Socas-Rodríguez B, Hernández-Borges J, Rodríguez-Delgado MÁ (2017) Core-shell poly(dopamine) magnetic nanoparticles for the extraction of estrogenic mycotoxins from milk and yogurt prior to LC–MS analysis. Food Chem 215:362–368. https://doi.org/10.1016/j.foodchem.2016.07.154
Goud KY, Kailasa SK, Kumar V, Tsang YF, Lee SE, Gobi KV, Kim K-H (2018) Progress on nanostructured electrochemical sensors and their recognition elements for detection of mycotoxins: A review. Biosens Bioelectron 121:205–222. https://doi.org/10.1016/j.bios.2018.08.029
Gu C, Yang L, Wang M, Zhou N, He L, Zhang Z, Du M (2019) A bimetallic (Cu-Co) Prussian Blue analogue loaded with gold nanoparticles for impedimetric aptasensing of ochratoxin a. Mikrochim Acta 186:343. https://doi.org/10.1007/s00604-019-3479-5
Guo X (2013) Single-molecule electrical biosensors based on single-walled carbon nanotubes. Adv Mater 25:3397–3408
Hamad AF, Han J-H, Kim B-C, Rather IA (2018) The intertwine of nanotechnology with the food industry. Saudi J Biol Sci 25:27–30. https://doi.org/10.1016/j.sjbs.2017.09.004
Hao N, Jiang L, Qian J, Wang K (2016) Ultrasensitive electrochemical Ochratoxin A aptasensor based on CdTe quantum dots functionalized graphene/Au nanocomposites and magnetic separation. J Electroanal Chem 781:332–338
Hendrickson O, Chertovich J, Petrakova A, Zherdev A, Dzantiev B (2016) Immunoenzyme assay of zearalenone using magnetic nanoparticles and chemiluminescent detection. Int J Appl Chem 12:613–627
Heurich M, Kadir MKA, Tothill IE (2011) An electrochemical sensor based on carboxymethylated dextran modified gold surface for ochratoxin A analysis. Sensors Actuators B Chem 156:162–168
Horky P, Skalickova S, Baholet D, Skladanka J (2018) Nanoparticles as a solution for eliminating the risk of mycotoxins. Nanomaterials (Basel, Switzerland) 8. https://doi.org/10.3390/nano8090727
Hossain MZ, Maragos CM (2018) Gold nanoparticle-enhanced multiplexed imaging surface plasmon resonance (iSPR) detection of Fusarium mycotoxins in wheat. Biosens Bioelectron 101:245–252. https://doi.org/10.1016/j.bios.2017.10.033
Hu W et al (2014) Sensitive detection of multiple mycotoxins by SPRi with gold nanoparticles as signal amplification tags. J Colloid Interface Sci 431:71–76. https://doi.org/10.1016/j.jcis.2014.06.007
Huang X, Zhan S, Xu H, Meng X, Xiong Y, Chen X (2016) Ultrasensitive fluorescence immunoassay for detection of ochratoxin A using catalase-mediated fluorescence quenching of CdTe QDs. Nanoscale 8:9390–9397
Ingle AP, Gupta I, Jogee P, Rai M (2020) Chapter 2 - Role of nanotechnology in the detection of mycotoxins: a smart approach. In: Rai M, Abd-Elsalam KA (eds) Nanomycotoxicology. Academic Press, pp 11–33. https://doi.org/10.1016/B978-0-12-817998-7.00002-1
Jamil TS, Abbas H, Nasr RA, El-Kady AA, Ibrahim MI (2017) Detoxification of aflatoxin B1 using nano-sized Sc-doped SrTi0. 7Fe0. 3O3 under visible light. J Photochem Photobiol A Chem 341:127–135
Jard G, Liboz T, Mathieu F, Guyonvarc’h A, Lebrihi A (2011) Review of mycotoxin reduction in food and feed: from prevention in the field to detoxification by adsorption or transformation. Food Addit Contam A 28:1590–1609
Jiang W, Beloglazova NV, Luo P, Guo P, Lin G, Wang X (2017) A dual-color quantum dots encoded frit-based immunoassay for visual detection of aflatoxin M1 and pirlimycin residues in milk. J Agric Food Chem 65:1822–1828. https://doi.org/10.1021/acs.jafc.6b05337
Joshi S (2017) Nanostructured imaging surface plasmon resonance biosensing. Wageningen University
Jukes D (2000) The role of science in international food standards. Food Control 11:181–194
Kalarestaghi A, Bayat M, Hashemi SJ, Razavilar V (2015) Highly sensitive FRET-based fluorescence immunoassay for detecting of aflatoxin B1 using magnetic/silica core-shell as a signal intensifier. Iran J Biotechnol 13:25
Kaushik A, Solanki P, Pandey M, Ahmad S, Malhotra B (2009a) Cerium oxide-chitosan based nanobiocomposite for food borne mycotoxin detection. Appl Phys Lett 95:173703–173703. https://doi.org/10.1063/1.3249586
Kaushik A, Solanki PR, Sood K, Ahmad S, Malhotra BD (2009b) Fumed silica nanoparticles–chitosan nanobiocomposite for ochratoxin-A detection. Electrochem Commun 11:1919–1923
Khan R, Dhayal M (2008) Nanocrystalline bioactive TiO2–chitosan impedimetric immunosensor for ochratoxin-A. Electrochem Commun 10:492–495. https://doi.org/10.1016/j.elecom.2008.01.013
Kim S, Lim HB (2015) Chemiluminescence immunoassay using magnetic nanoparticles with targeted inhibition for the determination of ochratoxin A. Talanta 140:183–188. https://doi.org/10.1016/j.talanta.2015.03.044
Ko J, Lee C, Choo J (2015) Highly sensitive SERS-based immunoassay of aflatoxin B1 using silica-encapsulated hollow gold nanoparticles. J Hazard Mater 285:11–17. https://doi.org/10.1016/j.jhazmat.2014.11.018
Kong D et al (2016) A gold nanoparticle-based semi-quantitative and quantitative ultrasensitive paper sensor for the detection of twenty mycotoxins. Nanoscale 8:5245–5253. https://doi.org/10.1039/C5NR09171C
Lara S, Perez-Potti A (2018) Applications of nanomaterials for immunosensing. Biosensors 8:104
Li W-k, H-x Z, Y-p S (2018a) Simultaneous determination of aflatoxin B1 and zearalenone by magnetic nanoparticle filled amino-modified multi-walled carbon nanotubes. Anal Methods 10:3353–3363. https://doi.org/10.1039/C8AY00815A
Li Y, Sun M, Mao X, You Y, Gao Y, Yang J, Wu Y (2018b) Mycotoxins contaminant in kelp: a neglected dietary exposure pathway. Toxins 10:481. https://doi.org/10.3390/toxins10110481
Li Z, Sheng W, Liu Q, Li S, Shi Y, Zhang Y, Wang S (2018c) Development of a gold nanoparticle enhanced enzyme linked immunosorbent assay based on monoclonal antibodies for the detection of fumonisin B1, B2, and B3 in maize. Anal Methods 10:3506–3513. https://doi.org/10.1039/C8AY01036F
Liang Y, Huang X, Yu R, Zhou Y, Xiong Y (2016) Fluorescence ELISA for sensitive detection of ochratoxin A based on glucose oxidase-mediated fluorescence quenching of CdTe QDs. Anal Chim Acta 936:195–201. https://doi.org/10.1016/j.aca.2016.06.018
Lin Y, Zhou Q, Tang D, Niessner R, Yang H, Knopp D (2016) Silver nanolabels-assisted ion-exchange reaction with CdTe quantum dots mediated exciton trapping for signal-on photoelectrochemical immunoassay of mycotoxins. Anal Chem 88:7858–7866
Liu M et al (2019) Patulin removal from apple juice using a novel cysteine-functionalized metal-organic framework adsorbent. Food Chem 270:1–9
Lu L, Gunasekaran S (2019) Dual-channel ITO-microfluidic electrochemical immunosensor for simultaneous detection of two mycotoxins. Talanta 194:709–716. https://doi.org/10.1016/j.talanta.2018.10.091
Lu Z, Chen X, Wang Y, Zheng X, Li CM (2015) Aptamer based fluorescence recovery assay for aflatoxin B1 using a quencher system composed of quantum dots and graphene oxide. Microchim Acta 182:571–578
Lu T et al (2018) Fluorescence ELISA based on CAT-regulated fluorescence quenching of CdTe QDs for sensitive detection of FB1. Anal Methods 10:5797–5802. https://doi.org/10.1039/C8AY02065E
Luan Y, Chen J, Li C, Xie G, Fu H, Ma Z, Lu A (2015a) Highly sensitive colorimetric detection of ochratoxin A by a label-free aptamer and gold nanoparticles. Toxins 7:5377–5385
Luan Y et al (2015b) Rapid visual detection of aflatoxin B1 by label-free aptasensor using unmodified gold nanoparticles. J Nanosci Nanotechnol 15:1357–1361. https://doi.org/10.1166/jnn.2015.9225
Luo Y, Zhou Z, Yue T (2017) Synthesis and characterization of nontoxic chitosan-coated Fe3O4 particles for patulin adsorption in a juice-pH simulation aqueous. Food Chem 221:317–323
Luo L et al (2019) Monitoring zearalenone in corn flour utilizing novel self-enhanced electrochemiluminescence aptasensor based on NGQDs-NH2-Ru@SiO2 luminophore. Food Chem 292:98–105. https://doi.org/10.1016/j.foodchem.2019.04.050
Lv X et al (2015) Electrochemiluminescence modified electrodes based on RuSi@Ru(bpy)32+ loaded with gold functioned nanoporous CO/Co3O4 for detection of mycotoxin deoxynivalenol. Biosens Bioelectron 70:28–33. https://doi.org/10.1016/j.bios.2015.03.020
Magro M et al (2016) Citrinin mycotoxin recognition and removal by naked magnetic nanoparticles. Food Chem 203:505–512. https://doi.org/10.1016/j.foodchem.2016.01.147
Magzoub R, Yassin A, Abdel-Rahim A, Gubartallah E, Miskam M, Saad B, Sabar S (2019) Photocatalytic detoxification of aflatoxins in Sudanese peanut oil using immobilized titanium dioxide. Food Control 95:206–214
Mahdi M, Mansour B, Afshin M (2016) Competitive immunoassay for Ochratoxin a based on FRET from quantum dot-labeled antibody to rhodamine-coated magnetic silica nanoparticles. Microchim Acta 183:3093–3099. https://doi.org/10.1007/s00604-016-1951-z
Majdinasab M, Sheikh-Zeinoddin M, Soleimanian-Zad S, Li P, Zhang Q, Li X, Tang X (2015) Ultrasensitive and quantitative gold nanoparticle-based immunochromatographic assay for detection of ochratoxin A in agro-products. J Chromatogr B 974:147–154. https://doi.org/10.1016/j.jchromb.2014.10.034
Malhotra BD, Srivastava S, Ali MA, Singh C (2014) Nanomaterial-based biosensors for food toxin detection. Appl Biochem Biotechnol 174:880–896. https://doi.org/10.1007/s12010-014-0993-0
Manubolu M, Goodla L, Pathakoti K, Malmlöf K (2018) Enzymes as direct decontaminating agents—mycotoxins. In: Enzymes in human and animal nutrition. Elsevier, pp 313–330
Mao J et al (2019) Insights into photocatalytic inactivation mechanism of the hypertoxic site in aflatoxin B1 over clew-like WO3 decorated with CdS nanoparticles. Appl Catal B Environ 248:477–486
Maragos C (2012) Signal amplification using colloidal gold in a biolayer interferometry-based immunosensor for the mycotoxin deoxynivalenol. Food Addit Contam A 29:1108–1117
Molinero-Fernández Á, Jodra A, Moreno-Guzmán M, López MÁ, Escarpa A (2018) Magnetic reduced graphene oxide/nickel/platinum nanoparticles micromotors for mycotoxin analysis. Chem Eur J 24:7172–7176
Niazi S et al (2018) A novel bioassay based on aptamer-functionalized magnetic nanoparticle for the detection of zearalenone using time resolved-fluorescence NaYF4: Ce/Tb nanoparticles as signal probe. Talanta 186:97–103. https://doi.org/10.1016/j.talanta.2018.04.013
Organization WH (1999) The application of risk communication to food standards and safety matters: report of a joint FAO/WHO expert consultation, Rome, 2–6 February 1998. vol 70. Food & Agriculture Org
Pal M (2017) Nanotechnology: a new approach in food packaging. J Food e Microbiol Safety Hyg 2:121
Panini NV, Bertolino FA, Salinas E, Messina GA, Raba J (2010) Zearalenone determination in corn silage samples using an immunosensor in a continuous-flow/stopped-flow systems. Biochem Eng J 51:7–13
Pathakoti K, Morrow S, Han C, Pelaez M, He X, Dionysiou DD, Hwang H-M (2013) Photoinactivation of Escherichia coli by sulfur-doped and nitrogen–fluorine-codoped TiO2 nanoparticles under solar simulated light and visible light irradiation. Environ Sci Technol 47:9988–9996
Pathakoti K, Manubolu M, Hwang H-M (2018) Nanotechnology applications for environmental industry. In: Handbook of nanomaterials for industrial applications. Elsevier, pp 894–907
Pathakoti K, Goodla L, Manjunath M, Hwang H-M (2019) Nanoparticles and its potential applications in the agriculture, biological therapies, food, biomedical and pharmaceutical industries: a review. https://doi.org/10.1201/9780429425660-3
Peltomaa R, Amaro-Torres F, Carrasco S, Orellana G, Benito-Peña E, Moreno-Bondi MC (2018) Homogeneous quenching immunoassay for fumonisin B1 based on gold nanoparticles and an epitope-mimicking yellow fluorescent protein. ACS Nano 12:11333–11342
Pfohl-Leszkowicz A, Hadjeba-Medjdoub K, Ballet N, Schrickx J, Fink-Gremmels J (2015) Assessment and characterisation of yeast-based products intended to mitigate ochratoxin exposure using in vitro and in vivo models. Food Addit Contam A 32:604–616
Pirouz A, Selamat J, Iqbal S, Mirhosseini H, Karjiban RA, Bakar FA (2017) The use of innovative and efficient nanocomposite (magnetic graphene oxide) for the reduction on of Fusarium mycotoxins in palm kernel cake. Sci Rep 7:12453
Pleadin J, Vulić A, Babić J, Šubarić D (2018) The incidence of T-2 and HT-2 toxins in cereals and methods of their reduction practice by the food industry. In: Fusarium – plant diseases, pathogen diversity, genetic diversity, resistance and molecular markers. IntechOpen, London, pp 41–64
Pradhan N, Singh S, Ojha N, Shrivastava A, Barla A, Rai V, Bose S (2015) Facets of nanotechnology as seen in food processing, packaging, and preservation industry. Biomed Res Int 2015:365672–365672
Saifuddin N, Raziah AZ, Junizah AR (2013) Carbon nanotubes: a review on structure and their interaction with proteins. J Chem 2013:18. https://doi.org/10.1155/2013/676815
Santos Pereira C, C Cunha S, Fernandes JO (2019) Prevalent mycotoxins in animal feed: occurrence and analytical methods. Toxins 11:290. https://doi.org/10.3390/toxins11050290
Santos VO et al (2017) Development and validation of a novel lateral flow immunoassay device for detection of aflatoxins in soy-based foods. Anal Methods 9:2715–2722. https://doi.org/10.1039/C7AY00601B
Shao B, Ma X, Zhao S, Lv Y, Hun X, Wang H, Wang Z (2018) Nanogapped Au(core) @ Au-Ag(shell) structures coupled with Fe3O4 magnetic nanoparticles for the detection of Ochratoxin A. Anal Chim Acta 1033:165–172. https://doi.org/10.1016/j.aca.2018.05.058
Singh P, Campidelli S, Giordani S, Bonifazi D, Bianco A, Prato M (2009) Organic functionalisation and characterisation of single-walled carbon nanotubes. Chem Soc Rev 38:2214–2230
Smith M-C, Madec S, Coton E, Hymery N (2016) Natural co-occurrence of mycotoxins in foods and feeds and their in vitro combined toxicological effects. Toxins 8(94)
Socas-Rodríguez B, Hernández-Borges J, Herrera-Herrera AV, Rodríguez-Delgado MÁ (2018) Multiresidue analysis of oestrogenic compounds in cow, goat, sheep and human milk using core-shell polydopamine coated magnetic nanoparticles as extraction sorbent in micro-dispersive solid-phase extraction followed by ultra-high-performance liquid chromatography tandem mass spectrometry. Anal Bioanal Chem 410:2031–2042
Sonawane SK, Arya SS, LeBlanc JG, Jha N (2014) Use of nanomaterials in the detection of food contaminants. Eur J Nutr Food Safety 4:301–317
Sun S, Zhao R, Feng S, Xie Y (2018) Colorimetric zearalenone assay based on the use of an aptamer and of gold nanoparticles with peroxidase-like activity. Microchim Acta 185:535. https://doi.org/10.1007/s00604-018-3078-x
Sun S, Zhao R, Xie Y, Liu Y (2019) Photocatalytic degradation of aflatoxin B1 by activated carbon supported TiO2 catalyst. Food Control 100:183–188
Sunday CE, Masikini M, Wilson L, Rassie C, Waryo T, Baker PG, Iwuoha EI (2015) Application on gold nanoparticles-dotted 4-nitrophenylazo graphene in a label-free impedimetric deoxynivalenol immunosensor. Sensors (Basel) 15:3854–3871. https://doi.org/10.3390/s150203854
Tian J, Wei W, Wang J, Ji S, Chen G, Lu J (2018) Fluorescence resonance energy transfer aptasensor between nanoceria and graphene quantum dots for the determination of ochratoxin A. Anal Chim Acta 1000:265–272
Tothill I (2011) Biosensors and nanomaterials and their application for mycotoxin determination. World Mycotoxin J 4:361–374
Urusov A, Petrakova A, Vozniak M, Zherdev A, Dzantiev B (2014) Rapid immunoenzyme assay of aflatoxin B1 using magnetic nanoparticles. Sensors 14:21843–21857
Urusov AE, Petrakova AV, Kuzmin PG, Zherdev AV, Sveshnikov PG, Shafeev GA, Dzantiev BB (2015) Application of gold nanoparticles produced by laser ablation for immunochromatographic assay labeling. Anal Biochem 491:65–71. https://doi.org/10.1016/j.ab.2015.08.031
Vidal JC, Bonel L, Ezquerra A, Hernández S, Bertolín JR, Cubel C, Castillo JR (2013) Electrochemical affinity biosensors for detection of mycotoxins: A review. Biosens Bioelectron 49:146–158
Wang H, Zhou X-J, Liu Y-Q, Yang H-M, Guo Q-L (2011) Simultaneous determination of chloramphenicol and aflatoxin M1 residues in milk by triple quadrupole liquid chromatography− tandem mass spectrometry. J Agric Food Chem 59:3532–3538
Wang B et al (2016a) Aptamer induced assembly of fluorescent nitrogen-doped carbon dots on gold nanoparticles for sensitive detection of AFB1. Biosens Bioelectron 78:23–30. https://doi.org/10.1016/j.bios.2015.11.015
Wang Q, Chen M, Zhang H, Wen W, Zhang X, Wang S (2016b) Enhanced electrochemiluminescence of RuSi nanoparticles for ultrasensitive detection of ochratoxin A by energy transfer with CdTe quantum dots. Biosens Bioelectron 79:561–567
Wang C et al (2017) Magneto-controlled aptasensor for simultaneous electrochemical detection of dual mycotoxins in maize using metal sulfide quantum dots coated silica as labels. Biosens Bioelectron 89:802–809. https://doi.org/10.1016/j.bios.2016.10.010
Wang Y, Lu M, Tang D (2018) Novel photoluminescence enzyme immunoassay based on supramolecular host-guest recognition using L-arginine/6-aza-2-thiothymine-stabilized gold nanocluster. Biosens Bioelectron 109:70–74. https://doi.org/10.1016/j.bios.2018.03.007
Wang H, Mao J, Zhang Z, Zhang Q, Zhang L, Li P (2019) Photocatalytic degradation of deoxynivalenol over dendritic-like α-Fe2O3 under visible light irradiation. Toxins 11:105
Willner MR, Vikesland PJ (2018) Nanomaterial enabled sensors for environmental contaminants. J Nanobiotechnol 16:95–95. https://doi.org/10.1186/s12951-018-0419-1
Wu S, Duan N, Zhu C, Ma X, Wang M, Wang Z (2011) Magnetic nanobead-based immunoassay for the simultaneous detection of aflatoxin B1 and ochratoxin A using upconversion nanoparticles as multicolor labels. Biosens Bioelectron 30:35–42
Wu L, Yan H, Li G, Xu X, Zhu L, Chen X, Wang J (2019) Surface-imprinted gold nanoparticle-based surface-enhanced raman scattering for sensitive and specific detection of patulin in food samples. Food Anal Methods:1–10
Xiong S, Zhou Y, Huang X, Yu R, Lai W, Xiong Y (2017) Ultrasensitive direct competitive FLISA using highly luminescent quantum dot beads for tuning affinity of competing antigens to antibodies. Anal Chim Acta 972:94–101. https://doi.org/10.1016/j.aca.2017.03.039
Xu C, Ye S, Cui X, Song X, Xie X (2019) Modelling photocatalytic detoxification of aflatoxin B1 in peanut oil on TiO2 layer in a closed-loop reactor. Biosyst Eng 180:87–95
Yang M, Liu G, Mehedi HM, Ouyang Q, Chen Q (2017) A universal sers aptasensor based on DTNB labeled GNTs/Ag core-shell nanotriangle and CS-Fe3O4 magnetic-bead trace detection of Aflatoxin B1. Anal Chim Acta 986:122–130
Ye Y, Zhou Y, Mo Z, Cheng W, Yang S, Wang X, Chen F (2010) Rapid detection of aflatoxin B1 on membrane by dot-immunogold filtration assay. Talanta 81:792–798
Yu H, Park J-Y, Kwon CW, Hong S-C, Park K-M, Chang P-S (2018) An overview of nanotechnology in food science: preparative methods, practical applications, and safety. J Chem 2018:10. https://doi.org/10.1155/2018/5427978
Zeng Y, Zhu Z, Du D, Lin Y (2016) Nanomaterial-based electrochemical biosensors for food safety. J Electroanal Chem 781:147. https://doi.org/10.1016/j.jelechem.2016.10.030
Zhan S, Huang X, Chen R, Li J, Xiong Y (2016) Novel fluorescent ELISA for the sensitive detection of zearalenone based on H2O2-sensitive quantum dots for signal transduction. Talanta 158:51–56
Zhang X et al (2016) A novel electrochemical immunosensor for highly sensitive detection of aflatoxin B1 in corn using single-walled carbon nanotubes/chitosan. Food Chem 192:197–202. https://doi.org/10.1016/j.foodchem.2015.06.044
Zhang W, Han Y, Chen X, Luo X, Wang J, Yue T, Li Z (2017a) Surface molecularly imprinted polymer capped Mn-doped ZnS quantum dots as a phosphorescent nanosensor for detecting patulin in apple juice. Food Chem 232:145–154. https://doi.org/10.1016/j.foodchem.2017.03.156
Zhang X et al (2017b) Multiplex lateral flow immunoassays based on amorphous carbon nanoparticles for detecting three fusarium mycotoxins in maize. J Agric Food Chem 65:8063–8071
Zhang L, Dou X-W, Zhang C, Logrieco AF, Yang M-H (2018a) A review of current methods for analysis of mycotoxins in herbal medicines. Toxins 10:65. https://doi.org/10.3390/toxins10020065
Zhang X, Yu X, Wang J, Wang Q, Meng H, Wang Z (2018b) One-step core/multishell quantum dots-based fluoroimmunoassay for screening of deoxynivalenol in maize. Food Anal Methods 11:2569–2578. https://doi.org/10.1007/s12161-018-1198-x
Zhang Z, Zeng C, Peng B (2019) Adsorption properties of magnetic carbon nanotubes for patulin removal from aqueous solution systems. Food Control 102:1–10
Zhao Y, Luo Y, Li T, Song Q (2014) Au NPs driven electrochemiluminescence aptasensors for sensitive detection of fumonisin B1. RSC Adv 4:57709–57714
Zhao Y, Yang Y, Luo Y, Yang X, Li M, Song Q (2015) Double detection of mycotoxins based on SERS labels embedded Ag@ Au core–shell nanoparticles. ACS Appl Mater Interfaces 7:21780–21786
Zhao Y, Liu R, Sun W, Lv L, Guo Z (2018) Ochratoxin A detection platform based on signal amplification by Exonuclease III and fluorescence quenching by gold nanoparticles. Sensors Actuators B Chem 255:1640–1645. https://doi.org/10.1016/j.snb.2017.08.176
Zhong L, Carere J, Lu Z, Lu F, Zhou T (2018) Patulin in apples and apple-based food products: the burdens and the mitigation strategies. Toxins 10:475. https://doi.org/10.3390/toxins10110475
Acknowledgments
The author (K.P.) is thankful to the National Science Foundation (NSF/CREST HRD-1547754) for financial support.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Editor(s) (if applicable) and The Author(s), under exclusive licence to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Manubolu, M., Goodla, L., Ludsin, S.A., Jayakumar, T., Fraker, M., Pathakoti, K. (2021). Nanotechnology-Based Detection and Remediation of Mycotoxins for Food and Agriculture Applications. In: Kumar, V., Guleria, P., Ranjan, S., Dasgupta, N., Lichtfouse, E. (eds) Nanosensors for Environment, Food and Agriculture Vol. 1. Environmental Chemistry for a Sustainable World, vol 60. Springer, Cham. https://doi.org/10.1007/978-3-030-63245-8_9
Download citation
DOI: https://doi.org/10.1007/978-3-030-63245-8_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-63244-1
Online ISBN: 978-3-030-63245-8
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)