Abstract
Nonalcoholic Fatty liver Disease (NAFLD) is characterized by increased fat storage in the hepatocytes without a history of excessive alcohol consumption. NAFLD is a global health problem, rising in parallel to the increase of the prevalence of obesity and metabolic syndrome. Some patients with NAFLD may be clinically stabile, while others may progress to Non-Alcoholic Steatohepatitis (NASH), fibrosis and even cirrhosis of the liver. However, most people with NAFLD do not develop severe liver disease but have an increased risk of cardiovascular mortality and morbidity. Numerous pharmacological agents have been investigated so far, for the treatment of NAFLD. However, other than weight loss, healthy eating and increased physical activity, there is no effective and approved pharmacological approach for the treatment of NAFLD. Nutraceuticals have anti-inflammatory, anti-oxidant and insulin sensitizing effects, some of which are in used for centuries. Apart from their effects on cardiovascular health, they are under investigation for the management of NAFLD as well. There is an abundance of clinical data on the effects of nutraceuticals as potential therapeutic agents for NAFLD. However, most of these data are derived from small trials with considerable heterogeneity in inclusion criteria, sample size, and study duration. There is need for further randomized controlled trials, to widely support the use of nutraceuticals in NAFLD treatment. This chapter aims to review the current evidence about the effect of nutraceuticals on the treatment of NAFLD.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction and Definition
Nonalcoholic Fatty liver Disease (NAFLD) is a clinical spectrum of liver disorders characterized by increased fat storage in the hepatocytes without a history of excessive alcohol consumption. The disorders in the spectrum of NAFLD range from Nonalcoholic Fatty Liver (NAFL), namely steatosis without significant inflammation or fibrosis; to nonalcoholic steatohepatitis (NASH) with varying degrees of inflammation, injury or fibrosis; and finally, to Cirrhosis [1,2,3].
Epidemiology
Different reports from different geographical regions give the NAFLD incidence about 10–50 per 1000 person-years and a global prevalence around 25% [4,5,6,7,8,9]. The population data for NASH is quite few, as the gold standard diagnostic method, liver biopsy, cannot be performed for epidemiological purposes. However, it can be estimated that 7–30% of NAFLD patients who undergo liver biopsies have NASH. This indicates that the overall prevalence of NASH is likely to be between 1.5% and 6.45% [4]. The prevalence of NAFLD become highest in the fourth to fifth decades [10, 11]. There are conflicting reports about the gender distribution of NAFLD. Some reports suggest that NAFLD is more prevalent in men [12, 13]. While others mention it is more common in women [14, 15]. Ethnicity may be a predictor for NAFLD [16,17,18]. The prevalence appears to be higher in Hispanics than the Caucasian and the African American population [17]. The high prevalence of obesity in the Hispanic population may be a reason for the ethnic variations in the NAFLD frequency. Some genetic modifiers of disease severity may also have role in the ethnic variations of NAFLD frequency [19].
The risk of NAFLD is significantly increased in patients with Metabolic Syndrome mainly in patients with obesity [20, 21]. The whole spectrum of visceral adiposity, from overweight to severe obesity, is associated with the increased risk of NAFLD. More than 95% of subjects who undergo bariatric surgery are reported to have NAFLD [22]. The prevalence is also high in subjects with type 2 diabetes. About one to two third of patients with type 2 diabetes are estimated to have NAFLD [23]. Finally, more than half of patients with dyslipidemia have NAFLD. Patients with high TG and low HDL Cholesterol levels are the ones with the highest frequency of NAFLD [24, 25]. The lipoprotein phenotypes are more atherogenic in patients with NASH than those with NAFL [26].
Diagnosis
NAFLD can only be diagnosed after ruling out several etiological factors such as chronic alcohol intake (>20 gr/day for woman and > 30 gr/day for man), long term use of steatogenic drugs and hereditary disorders [1]. Liver enzymes are often abnormal in NAFLD. However, liver enzyme elevation by itself is not sufficient for the diagnosis of NAFLD. There are many cases with normal liver enzyme levels and there are many different causes of liver enzyme elevation other than the NAFLD. Ultrasound imaging is the most common diagnostic test, which detects NAFLD when the 10% of the hepatocytes have fat deposits [27]. Proton magnetic resonance spectroscopy (HMRS) and quantitative fat/water selective magnetic resonance imaging (MRI) are among the other diagnostic tests [28] which are less frequently used. Vibration controlled transient elastography is a common method to grade fibrosis based on liver stiffness and is also developed to the severity of hepatic steatosis [29]. Liver histology is the gold standard test which can establish NAFLD when 5% of hepatocytes have fat depots. However, it is not necessary to perform liver biopsy for every case with an ultrasound finding consistent with NAFLD. When other etiologies of liver disease cannot be not ruled out, or when the patient is at high risk of cirrhosis, liver biopsy would be necessary for the definite diagnosis.
Pathogenesis
The pathogenesis of NAFLD involves both genetic and environmental factors which in combination cause the metabolic alterations in the hepatocyte. The so called ‘Multiple Hit’ theory puts forward the role of multiple etiopathological factors on a genetic background [30]. The genetic and epigenetic factors not only determine the transition period from NAFL to NASH but also cause inflammation and fibrosis from the very beginning of the process [30]. The main pathology is the TG deposition in the hepatocytes, which occurs due to the higher fatty acid intake than the consumption. The 75% of the total TG depot is exogenous, while only 25% is produced in liver by the de novo lipogenesis [31, 32]. The amount of lipogenesis is significantly increased, and fatty acid beta oxidation rate is impaired in patients with NAFLD. The TG transformation to the very low density lipoproteins (VLDL) is also decreased in patients with NAFLD [32]. It is estimated that one third of patients with NAFL progress to NASH. This progression occurs due to the maladaptation of liver to the increased fatty acid input and involves the sequences of lipotoxicity, inflammation, increased hepatocellular dysfunction and fibrosis [32, 33].
Clinical Picture
The clinical and pathological features of NAFLD are diverse. The spectrum starts from noninflammatory deposition of fatty acids and may progress to fibrosis and hepatocellular carcinoma (HCC) [34, 35]. NASH is the second leading cause of liver disease among adults awaiting liver transplantation [36]. Inflammation and fibrosis are the most important factors playing role in the liver related mortality and morbidity [37,38,39]. Genetic factors play significant role in the severity of inflammation [40]. The mechanism of the progression of NAFL to NASH is not clear. Prospective studies show that progression to NASH is observed in about 40% of cases with NAFLD, while progression to fibrosis without inflammation is reported in the 40% [41]. Mortality is significantly higher in patients with NAFLD which includes mostly the cardiovascular deaths [1, 42]. HCC may occur in every patient with cirrhosis. HCC patients with a background of NAFLD have shorter surveys when compared to HCC patients without NAFLD. They are older and they generally have cardiovascular comorbidities [43]. Interestingly, about half of these HCC patients do not have a significant clinical picture of cirrhosis [44]. Especially patients with metabolic syndrome may develop HCC without fibrosis [45].
Treatment Strategies in Patients with NAFLD
There are two major aims of managing patients with NAFLD. The first one is reducing the cardiovascular risk and the other is preventing the progression of liver disease. Therefore, a wise approach should always involve lifestyle modification while taking into account the comorbidities and the stage of the liver disease [46].
Lifestyle Modification
Living a healthy life is the first and foremost strategy to prevent NAFLD. Weight reduction significantly improves liver histology, liver enzymes, serum insulin levels and quality of life in patients with NAFLD [47,48,49]. Hepatic steatosis and NAFLD activity score (NAS) significantly improve in patients after a modest (>5%) weight loss [47, 50]. There is robust data showing that moderate to severe alcohol intake increase disease progression [51]. Although there is no evidence about the effect of light alcohol intake in NAFLD, it would be a prudent approach to recommend people to refrain from alcohol intake. Cardiorespiratory fitness has a strong relation with the risk of fatty liver, especially in the population with obesity [52]. Exercise increases fatty acid oxidation, reduce fatty acid synthesis, and prevents mitochondrial and hepatocellular damage [53]. Exercise programs of 2–3 sessions a week of 30–60 min over a period of 6–12 weeks are recommended for patients with NAFLD [1].
Treatment of the Components of Metabolic Syndrome
Metabolic syndrome grows on top of insulin resistance and obesity. Therefore, treatment regimens establishing insulin sensitivity and weight lose have the potential to improve metabolic syndrome and NAFLD [54,55,56]. Metformin is the most common insulin sensitizer. However, it’s not effective in NAFLD treatment [57]. There is more robust data for Pioglitazone. Pioglitazone treatment significantly improves ballooning degeneration, lobular inflammation, steatosis and combined necroinflammation in patients with NASH [58].
If patients cannot lose enough weight with diet and exercise, medical treatment of obesity and bariatric surgery are considered as further strategies. Medical agents for weight loss may improve liver enzymes and liver histology. Orlistate improves biochemical indicators of liver damage, while it is far from being a first-choice drug for the management of NAFLD or NASH [59]. On the other hand, Liraglutide treatment may be a promising agent to prevent fibrosis progression and resolve NASH [60]. Patients can be referred for bariatric surgery, if they do not meet weight loss goals after 6 months of lifestyle interventions and medical treatment. Especially for patients with NASH or advanced fibrosis bariatric surgery is a promising treatment strategy. Bariatric surgery decreases steatosis and inflammation and improves fibrosis score [61].
Liver Specific Treatments in NAFLD
Numerous liver specific pharmacological agents with anti-inflammatory, antioxidant, antifibrotic or insulin sensitizing properties have been investigated for NAFLD treatment. However, there is so far no single pharmacological agent which is highly effective and safe. Therefore, pharmacotherapy should be reserved for patients with NASH and with significant fibrosis (stage ≥2) [28]. Current evidence shows that nondiabetic patients with biopsy proven NASH and fibrosis can be treated with vitamin E (800 IU/day) replacement [46, 62]. As there are reports about the increased all-cause mortality in patients taking high dosage vitamin E [63, 64], the risk and benefits of treatment should be discussed with the patient before the treatment. Pioglitazone or Liraglutide can be preferred on top of metformin treatment in patients with diabetes mellitus and biopsy proven NASH [58, 60].
Nutraceuticals for NAFLD Treatment
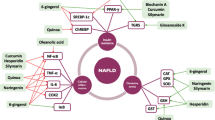
As there is no highly effective pharmacological treatment strategy for NAFLD, the effects of nutraceuticals on liver functions and histology have received much attention [65]. Numerous nutraceutical agents have been investigated for the treatment of NAFLD so far [66,67,68]. Phytochemicals, mono or Polyunsaturated fatty acids, antioxidant vitamins or minerals are the common bioactive molecules of nutraceuticals [69]. These active organic compounds are responsible for the protective effects of nutraceuticals by their positive effects on inflammation, oxidant stress and insulin resistance or by regulating the imbalance of the gut microbiota [67, 68]. Their effects on liver functions and liver histology have been observed in several clinical studies. Some nutraceuticals seem to have positive effects on liver enzymes and lower liver fat content [66]. However, despite the abundance of promising nutraceutical agents, the number of long-term, prospective randomized controlled clinical trials are not enough. The current evidence on the effects of nutraceuticals in NAFLD/NASH treatment is discussed below.
Silymarin
Silymarin, an ancient medicinal plant, is a flavonoid polyphenol, which is produced from the extracts of milk thistle or St. Mary’s thistle (Silybum marianum). Silymarin and its active ingredient silybin have antioxidant, anti-inflammatory and anti-fibrotic effects [70, 71]. Silymarin reduces fibrogenesis, stimulates liver regeneration and inhibits hepatic stellate cell activation [71]. Because of its significant biological effects, silymarin is one of the most common nutraceuticals for liver disorders [72]. The oral bioavailability of silymarin is not high because of the poor intestinal absorption and fast first pass metabolism in liver. Several studies have been performed with silymarin with different follow up periods, different dosages and combinations. An observational study has shown that, 6 months treatment with a complex of silybin- vitamin E and phospholipids decreases liver enzymes, HOMA-IR levels and improves the ultrasound grade of NAFLD [73]. Silymarin and Vitamin E combination, implemented for 6 months, decreases transaminases, without significant improvements in the noninvasive markers, such as fatty liver index, lipid accumulation product and NAFLD-fibrosis Score [74]. Another report of 3 months long treatment with silymarin shows significant improvement in insulin resistance and the indirect markers of hepatosteatosis such as hepatic steatosis index and lipid accumulation product [75]. Combination of silymarin, phosphatidylcholine and vitamin E improved liver enzymes and ultrasonographical scoring of NAFLD in a 12 months long multicenter randomized double blind study in patients with histological diagnosis of NAFLD/NASH. Liver biopsies, performed in a subgroup both before and after the study period, showed significant improvements in the severity of steatosis, lobular inflammation, ballooning and fibrosis [76]. Another clinical study, twice daily intake of a food supplement containing vitamin E, L-glutation, L-systein, L-methionin and silybum marianum significantly improved liver enzyme levels and the ultrasonographical severity of NAFLD in 3 months [77]. Another placebo-controlled study of silymarin showed no improvement in liver enzymes but a significant improvement in biometric parameters such as waist circumference, body mass index and the sonographically measured size of right hepatic lobe [78]. A metanalysis which involves eight randomized controlled studies showed that silymarin has positive efficacy to reduce transaminases in patients with NAFLD [79].
In conclusion, according to the current data, silymarin supplementation appears to have beneficial effects on the NAFLD treatment. The high safety profile of silymarin may be an advantage in the long-term use of this product. Silymarin is mentioned as a potentially useful treatment for NASH in AsiaPacific guidelines [80]. However, in order to obtain a global approval, larger scientific evidence is warranted to find out optimal dose and duration and to implement silymarin in children and adult population with NAFLD.
Vitamin E
Vitamin E is a potent antioxidant fat soluble vitamin, abundantly found in sunflower oil, palm oil, rice bran oil, olive oil, nuts and grains [81]. Because oxidative stress has a significant role in the pathogenesis of NAFLD, vitamin E is largely investigated in NAFLD treatment. In comparison to the healthy population, Vitamin E levels are reported to be significantly lower in patients with NAFLD [82]. No difference is established between patients with simple steatosis or NASH [82]. Vitamin E replacement in patients with NAFLD show significant improvements in liver enzymes [83, 84].
There is also data about the effects of vitamin E replacement on liver histology. Significant improvement in fibrosis score without any change in inflammation is reported in a randomized, double blind, placebo-controlled study of adult patients with NASH [85]. PIVENS (Pioglitazone versus Vitamin E versus Placebo for the Treatment of Nondiabetic Patients with Nonalcoholic Steatohepatitis) was a multicenter, randomized, placebo controlled, double blind, phase 3 study. Vitamin E 800 IU daily was given to 84 nondiabetic patients with NASH for 96 weeks. The results of the PIVENS study show significant decreases in ALT levels and improvements in histopathological findings such as steatosis, lobular inflammation and ballooning degeneration in the Vitamin E arm when compared to placebo or pioglitazone arms [86]. TONIC (Effect of vitamin E or metformin for treatment of nonalcoholic fatty liver disease in children and adolescents) was a double-blind, double-dummy, placebo-controlled clinical trial performed in 173 children or adolescents with biopsy-confirmed NAFLD. Sustained reduction in ALT level were not established but significant histological improvements in hepatocellular ballooning scores and NAFLD activity scores were obtained after 96 weeks of treatment of Vitamin E (800 IU/day) when compared to metformin (1000 mg/day) or placebo [87]. A metanalysis of five randomized controlled studies has shown that vitamin E replacement significantly improves liver functions and liver histology in children and adult patients with NAFLD/NASH [88].
In conclusion, there is limited data on the effects of Vitamin E replacement in patients with NAFLD. In accordance to the results of PIVENS study, current guidelines recommend vitamin E (800 IU/day) replacement in nondiabetic patients with biopsy proven NASH [28, 89]. However, due to the unexpected data on increased all-cause mortality [63, 64] or prostate cancer risk [90], vitamin E replacement should not be unanimously recommended to all patients with NAFLD or NASH.
Vitamin D
Vitamin D is a steroid hormone playing role in bone mineral metabolism, immune response regulation, cell differentiation and inflammation. Only 10% of circulating vitamin D is derived from the food intake while the rest is de-novo synthesized by the epidermal effect of ultraviolet (UV) B radiation. The UV B effect converts 7-dehydrocholesterol to cholecalciferol which is sequentially hydroxylated in liver and kidney to its final active form 1,25-hydroxy-cholecalciferol or Calcitriol [91,92,93]. Vitamin D deficiency may have role in insulin resistance and increase the risk of Metabolic Syndrome and Type 2 diabetes. Several studies show increased prevalence of vitamin D deficiency in patients with metabolic syndrome. Also, people with biopsy diagnosed NASH have lower vitamin D levels with a negative correlation to the severity of liver fibrosis [94, 95]. Similar findings were also reported in patients with NAFLD and normal liver enzymes [96]. Vitamin receptors are expressed in hepatocytes and vitamin receptor interaction may have a role in the progression of NASH [97].
Despite the evidence about the role of vitamin D deficiency in NAFLD, there are controversial reports about the beneficial effects of vitamin D replacement in the improvement of fatty liver. In a double-blind randomized placebo controlled clinical study, vitamin D replacement (50,000 IU/weekly for 4 months to 53 patients) did not cause significant improvements in liver enzymes, insulin sensitivity or the severity of hepatosteatosis [98]. Another clinical study of vitamin D replacement (2000 IU daily) also did not show significant improvements on metabolic parameters and the severity of steatosis in 42 patients with NAFLD [99]. Likewise, vitamin D replacement (25,000 IU/weekly) for 24 weeks did not improve liver enzymes, insulin resistance and histological findings in biopsy proven patients with NASH [100]. Another randomized double-blind controlled study reported that there may be significant improvements in liver enzymes, lipid parameters and insulin sensitivity in patients with NAFLD when calcium is added to vitamin D replacement [101]. A metanalysis of 974 patients with NAFLD has shown that, the vitamin D levels were not different when patients with high and low NAS and patients with high and low fibrosis scores compared [102]. Therefore, although vitamin D deficiency appears to have role in the pathogenesis of NAFLD, it is not clear that vitamin D has a histopathological effect on NAFLD [102].
In summary, the data about the relationship between vitamin D deficiency and the pathogenesis of NAFLD is inconclusive. The inconsistencies between different reports may be related to the methodological differences between these studies. Therefore, larger and longer-term prospective studies are warranted to better understand the direct effects of vitamin D replacement on disease progression.
Carnitine
L-carnitine, is an endogenous precursor of the carnitine-palmitoyltransferase-1. L-carnitine plays critical roles in the transport of fatty acids into mitochondria, cell membrane stabilization, and lowering of serum lipid levels. It regulates the energy imbalance in cells where much of the energy is produced from the fatty acid oxidation. Also, L-carnitine regulates immune response. L-carnitine replacement is expected to have beneficial effects in several metabolic disorders such as obesity, type 2 diabetes and liver cirrhosis [103,104,105].
In a placebo controlled clinical study, oral L- carnitine replacement (2 g/daily for 24 weeks) significantly reduced liver enzymes, insulin resistance and inflammatory parameters in 24 weeks in patients with NASH. The control biopsy has shown that NASH activity index, defined by steatosis, parenchymal inflammation and hepatocellular injury and fibrosis score significantly improved [106]. However, another randomized controlled study reported that 52 weeks long L-carnitine replacement (500 mg twice daily) did not show significant alterations in liver enzymes and liver ultrasound findings about steatosis [107].
So far, the data about the effects of L-carnitine replacement on NAFLD pathogenesis is controversial. More data is warranted to better understand the effect of long-term L-carnitine replacement on liver pathology.
Omega-3 Fatty Acids
Adipose tissue has the ability to effectively deposit free fatty acids (FFA). Obesity is a major cause of lipotoxicity of FFA in liver and pancreas. The exposure to FFA activates inflammatory pathways, impairs cellular communication and results in cellular dysfunction. Metabolic disorders such as insulin resistance and type 2 diabetes ensue as a result of chronic lipotoxicity [108]. Omega-3 fatty acids are essential polyunsaturated fatty acids (PUFA), which are not produced in vivo or which are not derived from alfa-linolenic acid. The most common omega-3 FFAs are Docosahexaenoic acid (DHA) and Eicosapentaenoic acid (EPA). Fish oil is a suitable source for omega-3 fatty acids. The most important effect of omega-3 FFAs is the cellular regulation of the metabolism from lipogenesis and triacylglycerol deposition to the fatty acid oxidation. PUFAs regulate lipid metabolism by reducing lipogenesis via their effects on Peroxisome proliferator-activated receptor alfa (PPAR- α) and the transcription of sterol regulatory element binding protein 1c (SREMP-1). This mechanism facilitates FFA oxidation and reduces hepatosteatosis. Insulin resistance can be improved by the anti-inflammatory effects of PUFAs [109].
There are several studies about the role of PUFA replacement on the improvement of NAFLD. A clinical study about 12 months of PUFA replacement reports significant improvements in TG and ALT levels and the ultrasound grade of steatosis [110]. Similar findings were also reported in several randomized controlled studies of PUFA (2 g/daily) replacement [111, 112]. PUFA appears to lower serum TG levels without lowering liver TG content [113].
Another clinical study showed that DHA (250 mg/day and 500 mg/day) significantly lowered hepatosteatosis and serum triglycerides and improved insulin sensitivity in 60 children with NAFLD [114]. A metanalysis of 9 clinical studies showed that PUFA can significantly but modestly improve liver enzymes and hepatic fat content [115].
In conclusion there are well designed but heterogeneous clinical studies about the role of PUFAs in the pathogenesis of NAFLD. Also, the number of studies involving histopathological assessments are limited. Finally, the optimal dosage, treatment period and long-term safety data are not established.
Vitamin C
Vitamin C is a water-soluble antioxidant and a free radical scavenger which plays role in infection control and cellular development. Vitamin C is not synthesized in the body and taken mainly form the fruit and vegetables. Cross-sectional population studies have shown that about 10–20% of the western population may have Vitamin C deficiency [116, 117]. Low vitamin C levels are not only related to NAFLD but also the risk of hypertension, cancer and atherosclerotic cardiovascular diseases [118, 119].
Animal studies report that vitamin C deficiency results in elevation of plasma and liver lipids, and increased oxidative stress, inflammation and fibrosis in liver, and vitamin C replacement reduces hepatic oxidative stress [120]. There are controversial reports from the epidemiological studies about the relation between vitamin C intake and NAFLD. According to some reports people with NAFLD have low vitamin C intake [121, 122] while others do not report any relationship at all [122,123,125].
Direct examination of the effect of Vitamin C replacement in comparison to placebo has not been performed in patients with NAFLD so far. However, studies which combine Vitamin C in combination to other vitamins have been conducted. A 12 month, double-blinded, randomized controlled trial examined the differences between combined treatment with Vitamin C (500 mg/day) and vitamin E (600 IU/day) versus placebo in children [126]. The change in liver enzymes, lipid levels or the liver brightness in the ultrasound were not significantly different in the treatment groups [126].
Another study in patients with NASH with 6 months of treatment with vitamin C (1000 IU daily) and vitamin E (1 gr daily) caused significant histological improvements without any alteration in liver enzymes [85]. Another study, with 4 years treatment of combined vitamin C (1 gr/day) and vitamin E (1000 IU) showed improvements in hepatosteatosis [127]. However, it is not easy to discern whether these beneficial effects are due to Vitamin C itself or Vitamin E or the combination. In conclusion, the present data is not enough to recommend vitamin C replacement to patients with NAFLD.
Coenzyme Q10
Coenzyme Q10 is an antioxidant and anti-inflammatory molecule and a promising agent in NAFLD treatment. Coenzyme Q10 is abundant in striated muscle, heart and liver, but present in all cells of the organism [128]. Main limitation of Coenzyme Q10 treatment is its low bioavailability. Therefore, high dosages are necessary for the establishment of therapeutic effects. On the other side Coenzyme Q10 has high safety profile and low risk of drug interaction [129]. A 3 weeks long, randomized, double blind, placebo-controlled study of 100 mg Coenzyme Q10 has shown significant improvements in liver enzymes, and hsCRP levels in patients with NAFLD [130]. Also, Coenzyme Q10 replacement significantly improved lipid abnormalities in patients with NAFLD [131].
Berberine
Berberine , also called berberine hydrochloride, is an alkaloid extract of Berberis vulgaris, traditional Medicine from China and India. Studies have shown that berberine can improve blood glucose and lipid levels and regulate immune responses [132, 133]. Animal studies have shown that berberine can decrease insulin resistance, lower hepatic and adipose tissue inflammation [134, 135]. Clinical studies with short term (2–4 months) berberine (500 mg/day) replacement has shown improvements in the indirect markers of hepatosteatosis such as Hepatic Steatosis Index and Lipid Accumulation Product [136]. A metanalysis of 6 randomized controlled studies has shown improvements in lipid parameters, insulin resistance and the grade of hepatic steatosis. However, these studies were performed with high dosages of berberine (1000–1500 mg/day) which caused significant intestinal side effects [137].
Curcumin
Curcumin is an extract of Curcuma longa. It improves insulin sensitivity, reduces oxidative stress and inflammation [137,138,139,141]. Curcumine has low bioavailability. Therefore, in most of the clinical studies of curcumin, piperine like molecules were added to improve its bioavailability. In patients with metabolic syndrome, curcumin reduced the serum levels of proinflammatory cytokines (TNF-α, IL-6, IL-1β, and MCP-1), increase anti-inflammatory adiponectine levels [139, 140] and improve Blood lipids [141]. Curcumin ingestion (1000 mg/day divided into two doses) for 8 weeks decrease, liver fat content, reduce liver enzyme levels, and increase hepatic vein flow with a reduction in portal vein diameter and liver volume [142].
Resveratrol
The natural nonflavonoid polyphenol compound resveratrol is an antioxidant, vasoprotective and insulin sensitizing agent abundant in nuts, berries and grape peel [143, 144]. Conflicting reports are present about the effects of resveratrol in patients with NAFLD. A placebo controlled double blind study of resveratrol replacement (500 mg/day for 12 months) did not result in any significant alteration in anthropometric measures, insulin sensitivity and lipid parameters, but reduced serum ALT levels and improved hepatic steatosis [145]. Another study of resveratrol replacement (600 mg/day for 3 months) resulted in decreased insulin resistance, blood glucose and lipids [146]. Furthermore, resveratrol improved the balance between important pro-inflammatory and anti-inflammatory cytokines [146]. On the contrary, another study of resveratrol replacement with much higher dosages (3000 mg/day for 8 weeks) did not reduce insulin resistance, steatosis, or abdominal fat distribution, but increased liver enzymes [147]. The discrepancies with the results of the clinical studies are probably related to differences between the resveratrol dosage and the study periods. As the previous reports are not consistent, long-term clinical studies will be informative about the safety and efficacy of resveratrol in the treatment of NAFLD.
Salvia Miltiorrhiza
Salvia Miltiorrhiza is a traditional Asian herbal medication, which is used in China in the treatment of cardiovascular diseases for more than 2000 years. A metanalysis of 8 controlled studies has shown that, Salvia miltiorrhiza dry extract supplementation significantly improves plasma transaminases and radiological severity of hepatosteatosis [148].
Probiotics
The dysbiosis of gut microbiota can take part in the pathogenesis of liver diseases. Probiotics, Prebiotics and symbiotics modulate gut microbiota and they are potential therapeutic agents for NAFLD [149].
Several studies performed with probiotics or symbiotics have shown significant improvements in insulin resistance, liver enzymes and the severity of steatosis in patients with NAFLD. But different probiotics were used in different amounts with different periods in these studies. Therefore, it is not easy to discern a specific probiotic type, dosage and treatment period for [150].
Randomized controlled studies with L. Acidophilus, B. Lactis, L. Bulgaris, S. Thermophilus or L. rhamnosus showed significant improvements in liver enzymes in children and adults [150,151,153]. Another study in children has shown that 4 months long replacement of bifidobacteria, lactobacilli and S. thermophilus improves the severity of steatosis in liver [154].
In conclusion, probiotics can improve gut microbiota and liver pathology. However, there is problem in the standardization of results from different probiotics. The evidence is promising about the effect of agents targeting gut microbiota in NAFLD treatment.
Anthocyanins
Anthocyanins , are water soluble bioactive flavonoid compounds. Nutrients rich in Anthocyanins improve oxidative stress, dyslipidemia and hepatic steatosis in experimental NASH models. Replacement of purified anthocyanin (320 mg/day) significantly improved insulin resistance and liver enzymes in patients with NAFLD [155]. However, we need more data, especially on histological improvements, in order to get benefit of Anthocyanins on NAFLD [156].
Betaine
Betaine is an important methyl donor. It is either synthesized de novo or taken by the diet. Animal studies has shown that betaine replacement can prevent liver fat deposition [157, 158]. The effects of Betaine replacement were also investigated in a small number of clinical studies.
There appears to be a negative correlation between plasma betaine concentrations and the severity of NAFLD [159]. Eight weeks of Betaine replacement reduced 25% of liver steatosis and improved liver enzymes of patients with NASH [160]. However, another randomized controlled study of 12 months long oral betaine replacement did not show any significant effect on liver enzymes and liver histology [161].
Camellia Sinensis
Camellia Sinensis (Green Tea) is the leaf extract of the green tea plant Camellia Sinensis. Green Tea has been under scrutiny especially for its effects on cardiovascular diseases [162]. Green tea ingestion improves exercise performance, increase fat oxidation, and prevent obesity in mice [163, 164]. In recent years Green Tea is also investigated for the treatment of NAFLD. In two randomized controlled studies, Green Tea extract (500 mg/day for 12 weeks) significantly improved liver enzymes, insulin resistance and the ultrasound findings of liver steatosis when compared to the placebo arm [165, 166]. Another randomized controlled study with 12 weeks of green tea (700 ml/day) containing high density catechins improved liver fat content and inflammation by reducing oxidative stress in patients with NAFLD [167].
Coffee
Coffee consumption decreases insulin resistance and oxidative stress in hepatocytes and have anti-inflammatory and antifibrotic effects in animal models [168, 169]. The role of coffee consumption on NAFLD has been investigated in several clinical studies. A significant negative correlation between the amount of coffee consumption and fibrosis scores were established in patients with biopsy proven NAFLD [170]. A 7 years follow up study has shown that coffee consumption prevents the progression of liver fibrosis [171]. The real-life data also show that healthy patients have higher coffee consumption rates when compared to patients with NAFLD [172]. A metanalysis has shown that the risks of NAFLD and NASH are significantly lower in patients with regular daily coffee consumption [173].
Spirulina
Spirulina (Arthrospira platensis), is a cyanobacterium which has long been used as a food supplement. FDA approved Spirulina as a food supplement as it has a high safety profile and a rich source of proteins, vitamins, minerals, carotenoids, and phytocyanins. Spirulina has hypolipidemic, hypoglycemic, anti-viral, hepatoprotective, anti-inflammatory, anti-oxidant, anti-neoplastic, and immunomodulatory effects [174]. Spirulina replacement decreases liver enzymes and improves fibrosis in the animal models of NAFLD [175]. In a clinical study, 6 months of treatment with spirulina (6 gr/day) significantly improved lipid parameters, insulin resistance and liver enzymes, without any alteration in the severity of hepatosteatosis [176]. There is very limited data about the effect of spirulina in NAFLD treatment. Further studies with spirulina, especially on its effects on liver histology, are warranted.
S-adenosyl-L-methionine
S-adenosylmethionine (SAM) , is an organic molecule unanimously found in every living cell. Current data about the effect of SAM in the pathogenesis of NAFLD are derived from experimental animal models. Chronic liver SAM deficiency may play role in the development of NASH, and SAM replacement may be effective in prevention of NAFLD in experimental animals [177]. There is however, limited clinical evidence about the role of SAM replacement on the pathogenesis of NAFLD [178].
Astaxanthin
Astaxanthin is an antioxidant caroten derivative, which inhibits lipid peroxidation more powerfully than vitamin E. It is regarded as a possible nutraceutical alternative to vitamin E [65]. Astaxanthin more significantly reduce lipogenesis, insulin resistance, liver inflammation and fibrogenesis when compared to vitamin E in the experimental models [179]. With the preliminary data, Astaxanthin appears to be the ideal antioxidant for the prevention of liver injury induced by NAFLD. However, there is insufficient human data about the effect of Astaxanthin in NAFLD [180].
Conclusion
In conclusion, numerous nutraceuticals are currently in use in different therapeutic forms such as food ingredients, dietary supplements, herbal teas or over the counter pills. Many clinical studies have been performed so far, to investigate the effects of nutraceuticals on NAFLD. There are several technical problems in quite a lot of these reports, such as small sample sizes, short study periods, lack of placebo arms or overestimation of the benefits. However, there is also high-quality evidence derived from randomized, placebo controlled prospective studies. According to these data at least some of these nutraceuticals such as Silmarin, Curcumin, Green Tea or Vitamin E have promising metabolic advantages and may improve liver functions or liver histology. However, there is hardly enough data about the optimal dosage, optimal treatment period and long-term safety data for most of these nutraceuticals. Therefore, time is needed to reproduce long term, randomized controlled studies before the widespread use of nutraceuticals for the NAFLD treatment.
References
Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, Rinella M, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67:328–57. https://doi.org/10.1002/hep.29367.
Sheth S, Gordon F, Chopra S. Nonalcoholic steatohepatitis. Ann Intern Med. 1997;126:137. https://doi.org/10.7326/0003-4819-126-2-199701150-00008.
Caldwell SH, Crespo DM. The spectrum expanded: cryptogenic cirrhosis and the natural history of non-alcoholic fatty liver disease. Powell EE, Cooksley WGE, Hanson R, Searle J, Halliday JW, Powell LW. The natural history of nonalcoholic steatohepatitis: a follow-up study of forty-two patients for up to 21 years [Hepatology 1990; 11:74–80]. J Hepatol 2004;40:578–84. https://doi.org/10.1016/j.jhep.2004.02.013.
Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease—meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64:73–84. https://doi.org/10.1002/hep.28431.
Sung K-C, Wild SH, Byrne CD. Development of new fatty liver, or resolution of existing fatty liver, over five years of follow-up, and risk of incident hypertension. J Hepatol. 2014;60:1040–5. https://doi.org/10.1016/j.jhep.2014.01.009.
Tsuneto A, Hida A, Sera N, Imaizumi M, Ichimaru S, Nakashima E, et al. Fatty liver incidence and predictive variables. Hypertens Res. 2010;33:638. https://doi.org/10.1038/hr.2010.45.
Wong V, Wong G, Yeung D, Lau T, Chan C, Chim A, et al. Incidence of non-alcoholic fatty liver disease in Hong Kong: a population study with paired proton-magnetic resonance spectroscopy. J Hepatol. 2015;62:182–9. https://doi.org/10.1016/j.jhep.2014.08.041.
Whalley S, Puvanachandra P, Desai A, Kennedy H. Hepatology outpatient service provision in secondary care: a study of liver disease incidence and resource costs. Clin Med. 2007;7:119–24. https://doi.org/10.7861/clinmedicine.7-2-119.
Zelber-Sagi S, Lotan R, Shlomai A, Webb M, Harrari G, Buch A, et al. Predictors for incidence and remission of NAFLD in the general population during a seven-year prospective follow-up. J Hepatol. 2012;56:1145–51. https://doi.org/10.1016/j.jhep.2011.12.011.
Estes C, Razavi H, Loomba R, Younossi Z, Sanyal AJ. Modeling the epidemic of nonalcoholic fatty liver disease demonstrates an exponential increase in burden of disease. Hepatology. 2018;67:123–33. https://doi.org/10.1002/hep.29466.
Koehler EM, Schouten J, Hansen BE, van Rooij F, Hofman A, Stricker BH, et al. Prevalence and risk factors of non-alcoholic fatty liver disease in the elderly: results from the Rotterdam study. J Hepatol. 2012;57:1305–11. https://doi.org/10.1016/j.jhep.2012.07.028.
Arun J, Clements RH, Lazenby AJ, Leeth RR, Abrams GA. The prevalence of nonalcoholic steatohepatitis is greater in morbidly obese men compared to women. Obes Surg. 2006;16:1351–8. https://doi.org/10.1381/096089206778663715.
Zelber-Sagi S, Nitzan-Kaluski D, Halpern Z, Oren R. Prevalence of primary non-alcoholic fatty liver disease in a population-based study and its association with biochemical and anthropometric measures. Liver Int. 2006;26:856–63. https://doi.org/10.1111/j.1478-3231.2006.01311.x.
Ludwig J, Viggiano T, McGill D, Oh B. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin Proc. 1980;55:434–8.
Angulo P, Keach JC, Batts KP, Lindor KD. Independent predictors of liver fibrosis in patients with nonalcoholic steatohepatitis. Hepatology. 1999;30:1356–62. https://doi.org/10.1002/hep.510300604.
Agbim U, Carr RM, Pickett-Blakely O, Dagogo-Jack S. Ethnic disparities in adiposity: focus on non-alcoholic fatty liver disease, visceral, and generalized obesity. Curr Obes Rep. 2019;8(3):1–12. https://doi.org/10.1007/s13679-019-00349-x.
Browning JD, Szczepaniak LS, Dobbins R, Nuremberg P, Horton JD, Cohen JC, et al. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology. 2004;40:1387–95. https://doi.org/10.1002/hep.20466.
Nazare J-A, Smith JD, Borel A-L, Haffner SM, Balkau B, Ross R, et al. Ethnic influences on the relations between abdominal subcutaneous and visceral adiposity, liver fat, and cardiometabolic risk profile: the International Study of Prediction of Intra-Abdominal Adiposity and Its Relationship With Cardiometabolic Risk/Intra-Abdominal Adiposity. Am J Clin Nutr. 2012;96:714–26. https://doi.org/10.3945/ajcn.112.035758.
Dongiovanni P, Anstee Q, Valenti L. Genetic predisposition in NAFLD and NASH: impact on severity of liver disease and response to treatment. Curr Pharm Design. 2013;19:5219–38. https://doi.org/10.2174/13816128113199990381.
Byrne CD, Targher G. NAFLD: a multisystem disease. J Hepatol. 2015;62:S47–64. https://doi.org/10.1016/j.jhep.2014.12.012.
Yki-Järvinen H. Non-alcoholic fatty liver disease as a cause and a consequence of metabolic syndrome. Lancet Diabetes Endocrinol. 2014;2:901–10. https://doi.org/10.1016/s2213-8587(14)70032-4.
Sasaki A, Nitta H, Otsuka K, Umemura A, Baba S, Obuchi T, et al. Bariatric surgery and non-alcoholic fatty liver disease: current and potential future treatments. Front Endocrinol. 2014;5:164. https://doi.org/10.3389/fendo.2014.00164.
Leite NC, Salles GF, Araujo AL, Villela-Nogueira CA, Cardoso CR. Prevalence and associated factors of non-alcoholic fatty liver disease in patients with type-2 diabetes mellitus. Liver Int. 2009;29:113–9. https://doi.org/10.1111/j.1478-3231.2008.01718.x.
Assy N, Kaita K, Mymin D, Levy C, Rosser B, Minuk G. Fatty infiltration of liver in hyperlipidemic patients. Dig Dis Sci. 2000;45:1929–34. https://doi.org/10.1023/a:1005661516165.
Wu K-T, Kuo P-L, Su S-B, Chen Y-Y, Yeh M-L, Huang C-I, et al. Nonalcoholic fatty liver disease severity is associated with the ratios of total cholesterol and triglycerides to high-density lipoprotein cholesterol. J Clin Lipidol. 2016;10:420–425.e1. https://doi.org/10.1016/j.jacl.2015.12.026.
Sonmez A, Nikolic D, Dogru T, Ercin C, Genc H, Cesur M, et al. Low- and high-density lipoprotein subclasses in subjects with nonalcoholic fatty liver disease. J Clin Lipidol. 2015;9:576–82. https://doi.org/10.1016/j.jacl.2015.03.010.
Ballestri S, Nascimbeni F, Baldelli E, Marrazzo A, Romagnoli D, Targher G, et al. Ultrasonographic fatty liver indicator detects mild steatosis and correlates with metabolic/histological parameters in various liver diseases. Metabolism. 2017;72:57–65. https://doi.org/10.1016/j.metabol.2017.04.003.
European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL–EASD–EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J Hepatol. 2016;64:1388–402. https://doi.org/10.1016/j.jhep.2015.11.004.
Shi K, Tang J, Zhu X, Ying L, Li D, Gao J, et al. Controlled attenuation parameter for the detection of steatosis severity in chronic liver disease: a meta-analysis of diagnostic accuracy. J Gastroenterol Hepatol. 2014;29:1149–58. https://doi.org/10.1111/jgh.12519.
Lonardo A, Nascimbeni F, Maurantonio M, Marrazzo A, Rinaldi L, Adinolfi L. Nonalcoholic fatty liver disease: evolving paradigms. World J Gastroenterol. 2017;23:6571–92. https://doi.org/10.3748/wjg.v23.i36.6571.
Donnelly KL, Smith CI, Schwarzenberg SJ, Jessurun J, Boldt MD, Parks EJ. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J Clin Invest. 2005;115:1343–51. https://doi.org/10.1172/jci23621.
Arab J, Arrese M, Trauner M. Recent insights into the pathogenesis of nonalcoholic fatty liver disease. Annu Rev Pathol Mech Dis. 2018;13:321–50. https://doi.org/10.1146/annurev-pathol-020117-043617.
Trauner M, Arrese M, Wagner M. Fatty liver and lipotoxicity. Biochimica Et Biophysica Acta Bba – Mol Cell Biol Lipids. 2010;1801:299–310. https://doi.org/10.1016/j.bbalip.2009.10.007.
Lonardo A, Nascimbeni F, Targher G, Bernardi M, Bonino F, et al. AISF position paper on nonalcoholic fatty liver disease (NAFLD): updates and future directions. Dig Liver Dis. 2017;49:471–83. https://doi.org/10.1016/j.dld.2017.01.147.
Farrell GC, Larter CZ. Nonalcoholic fatty liver disease: from steatosis to cirrhosis. Hepatology. 2006;43:S99–112. https://doi.org/10.1002/hep.20973.
Satapathy SK, Sanyal AJ. Epidemiology and natural history of nonalcoholic fatty liver disease. Semin Liver Dis. 2015;35:221–35. https://doi.org/10.1055/s-0035-1562943.
Ekstedt M, Hagström H, Nasr P, Fredrikson M, Stål P, Kechagias S, et al. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology. 2015;61:1547–54. https://doi.org/10.1002/hep.27368.
Younossi Z, Henry L. Contribution of alcoholic and nonalcoholic fatty liver disease to the burden of liver-related morbidity and mortality. Gastroenterology. 2016;150:1778–85. https://doi.org/10.1053/j.gastro.2016.03.005.
Angulo P, Kleiner DE, Dam-Larsen S, Adams LA, Bjornsson ES, Charatcharoenwitthaya P, et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology. 2015;149:389–397.e10. https://doi.org/10.1053/j.gastro.2015.04.043.
Pelusi S, Cespiati A, Rametta R, Pennisi G, Mannisto V, Rosso C, et al. Prevalence and risk factors if significant fibrosis in patients with nonalcoholic fatty liver without steatohepatitis. Clin Gastroenterol Hepatol. 2019;17(11):2310–2319.e6. https://doi.org/10.1016/j.cgh.2019.01.027.
McPherson S, Hardy T, Henderson E, Burt AD, Day CP, Anstee QM. Evidence of NAFLD progression from steatosis to fibrosing-steatohepatitis using paired biopsies: implications for prognosis and clinical management. J Hepatol. 2015;62:1148–55. https://doi.org/10.1016/j.jhep.2014.11.034.
Stahl EP, Dhindsa DS, Lee SK, Sandesara PB, Chalasani NP, Sperling LS. Nonalcoholic fatty liver disease and the heart JACC state-of-the-art review. J Am Coll Cardiol. 2019;73:948–63. https://doi.org/10.1016/j.jacc.2018.11.050.
Mohamad B, Shah V, Onyshchenko M, Elshamy M, Aucejo F, Lopez R, et al. Characterization of hepatocellular carcinoma (HCC) in non-alcoholic fatty liver disease (NAFLD) patients without cirrhosis. Hepatol Int. 2016;10:632–9. https://doi.org/10.1007/s12072-015-9679-0.
Piscaglia F, Svegliati-Baroni G, Barchetti A, Pecorelli A, Marinelli S, Tiribelli C, et al. Clinical patterns of hepatocellular carcinoma in nonalcoholic fatty liver disease: a multicenter prospective study. Hepatology. 2016;63:827–38. https://doi.org/10.1002/hep.28368.
Paradis V, Zalinski S, Chelbi E, Guedj N, Degos F, Vilgrain V, et al. Hepatocellular carcinomas in patients with metabolic syndrome often develop without significant liver fibrosis: a pathological analysis. Hepatology. 2009;49:851–9. https://doi.org/10.1002/hep.22734.
Dyson J, Anstee Q, McPherson S. Non-alcoholic fatty liver disease: a practical approach to treatment. Frontline Gastroenterol. 2014;5:277. https://doi.org/10.1136/flgastro-2013-100404.
Romero-Gómez M, Zelber-Sagi S, Trenell M. Treatment of NAFLD with diet, physical activity and exercise. J Hepatol. 2017;67:829–46. https://doi.org/10.1016/j.jhep.2017.05.016.
Petersen K, Dufour S, Befroy D, Lehrke M, Hendler RE, Shulman GI. Reversal of nonalcoholic hepatic steatosis, hepatic insulin resistance, and hyperglycemia by moderate weight reduction in patients with type 2 diabetes. Diabetes. 2005;54:603–8. https://doi.org/10.2337/diabetes.54.3.603.
Promrat K, Kleiner DE, Niemeier HM, Jackvony E, Kearns M, Wands JR, et al. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology. 2010;51:121–9. https://doi.org/10.1002/hep.23276.
Musso G, Cassader M, Rosina F, Gambino R. Impact of current treatments on liver disease, glucose metabolism and cardiovascular risk in non-alcoholic fatty liver disease (NAFLD): a systematic review and meta-analysis of randomised trials. Diabetologia. 2012;55:885–904. https://doi.org/10.1007/s00125-011-2446-4.
Ekstedt M, Franzén LE, Holmqvist M, Bendtsen P, Mathiesen UL, Bodemar G, et al. Alcohol consumption is associated with progression of hepatic fibrosis in non-alcoholic fatty liver disease. Scand J Gastroenterol. 2009;44:366–74. https://doi.org/10.1080/00365520802555991.
Pälve KS, Pahkala K, Suomela E, Aatola H, Hulkkonen J, Juonala M, et al. Cardiorespiratory fitness and risk of fatty liver. Med Sci Sports Exerc. 2017;49:1834–41. https://doi.org/10.1249/mss.0000000000001288.
van der Windt DJ, Sud V, Zhang H, Tsung A, Huang H. The effects of physical exercise on fatty liver disease. Van Der Wind Dirk J. 2017;18:89–101. https://doi.org/10.3727/105221617x15124844266408.
Benedict M, Zhang X. Non-alcoholic fatty liver disease: an expanded review. World J Hepatol. 2017;9:715–32. https://doi.org/10.4254/wjh.v9.i16.715.
Wong V. Obesity, fatty liver and liver cancer. Adv Exp Med Biol. 2018;1061:149–57. https://doi.org/10.1007/978-981-10-8684-7_12.
Hung C, Bodenheimer HC. Current treatment of nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. Clin Liver Dis. 2018;22:175–87. https://doi.org/10.1016/j.cld.2017.08.012.
Said A, Akhter A. Meta-analysis of randomized controlled trials of pharmacologic agents in non-alcoholic steatohepatitis. Ann Hepatol. 2017;16:538–47. https://doi.org/10.5604/01.3001.0010.0284.
Boettcher E, Csako G, Pucino F, Wesley R, Loomba R. Meta-analysis: pioglitazone improves liver histology and fibrosis in patients with non-alcoholic steatohepatitis. Aliment Pharmacol Ther. 2012;35:66–75. https://doi.org/10.1111/j.1365-2036.2011.04912.x.
Wang H, Wang L, Cheng Y, Xia Z, Liao Y, Cao J. Efficacy of orlistat in non-alcoholic fatty liver disease: a systematic review and meta-analysis. Biomed Rep. 2018;9:90–6. https://doi.org/10.3892/br.2018.1100.
Armstrong M, Gaunt P, Aithal GP, Barton D, Hull D, Parker R, et al. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet. 2016;387:679–90. https://doi.org/10.1016/s0140-6736(15)00803-x.
Uehara D, Seki Y, Kakizaki S, Horiguchi N, Tojima H, Yamazaki Y, et al. Long-term results of bariatric surgery for non-alcoholic fatty liver disease/non-alcoholic steatohepatitis treatment in morbidly obese Japanese patients. Obes Surg. 2019;29:1195–201. https://doi.org/10.1007/s11695-018-03641-2.
Perazzo H, Dufour J. The therapeutic landscape of non-alcoholic steatohepatitis. Liver Int. 2017;37:634–47. https://doi.org/10.1111/liv.13270.
Bjelakovic G, Nikolova D, Gluud L, Simonetti RG, Gluud C. Mortality in randomized trials of antioxidant supplements for primary and secondary prevention: systematic review and meta-analysis. JAMA. 2007;297:842–57. https://doi.org/10.1001/jama.297.8.842.
Miller ER, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, Guallar EL. Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. ACC Curr J Rev. 2005;14:17. https://doi.org/10.1016/j.accreview.2005.04.017.
Cicero AF, Colletti A, Bellentani S. Nutraceutical approach to non-alcoholic fatty liver disease (NAFLD): the available clinical evidence. Nutrients. 2018;10:1153. https://doi.org/10.3390/nu10091153.
Liu Z, Xie L, Zhu J, Li GQ, Grant SJ, Liu J. Herbal medicines for fatty liver diseases. Cochrane Database Syst Rev. 2013;8:CD009059. https://doi.org/10.1002/14651858.cd009059.pub2.
Ben M, Polimeni L, Baratta F, Pastori D, Angelico F. The role of nutraceuticals for the treatment of non-alcoholic fatty liver disease. Br J Clin Pharmacol. 2017;83:88–95. https://doi.org/10.1111/bcp.12899.
Figueiredo P, Inada A, Fernandes M, Arakaki D, de Freitas K, de Guimarães R, et al. An overview of novel dietary supplements and food ingredients in patients with metabolic syndrome and non-alcoholic fatty liver disease. Molecules. 2018;23:877. https://doi.org/10.3390/molecules23040877.
Bahadoran Z, Golzarand M, Mirmiran P, Saadati N, Azizi F. The association of dietary phytochemical index and cardiometabolic risk factors in adults: Tehran lipid and glucose study. J Hum Nutr Diet. 2013;26:145–53. https://doi.org/10.1111/jhn.12048.
Loguercio C, Festi D. Silybin and the liver: from basic research to clinical practice. World J Gastroenterol. 2011;17:2288–301. https://doi.org/10.3748/wjg.v17.i18.2288.
Trappoliere M, Caligiuri A, Schmid M, Bertolani C, Failli P, Vizzutti F, et al. Silybin, a component of sylimarin, exerts anti-inflammatory and anti-fibrogenic effects on human hepatic stellate cells. J Hepatol. 2009;50:1102–11. https://doi.org/10.1016/j.jhep.2009.02.023.
Abenavoli L, Capasso R, Milic N, Capasso F. Milk thistle in liver diseases: past, present, future. Phytother Res. 2010;24:1423–32. https://doi.org/10.1002/ptr.3207.
Federico A, Trappoliere M, Tuccillo C, de Sio I, Leva DA, Blanco DC, et al. A new silybin-vitamin E-phospholipid complex improves insulin resistance and liver damage in patients with non-alcoholic fatty liver disease: preliminary observations. Gut. 2006;55:901. https://doi.org/10.1136/gut.2006.091967.
Aller R, Laserna C, Rojo M, Mora N, Sánchez C, Pina M, et al. Role of the PNPLA3 polymorphism rs738409 on silymarin + vitamin E response in subjects with non-alcoholic fatty liver disease. Rev Esp Enferm Dig. 2018;110(10):634–40. https://doi.org/10.17235/reed.2018.5602/2018.
Surai PF. Silymarin as a natural antioxidant: an overview of the current evidence and perspectives. Antioxidants. 2015;4:204–47. https://doi.org/10.3390/antiox4010204.
Loguercio C, Andreone P, Brisc C, Brisc M, Bugianesi E, Chiaramonte M, et al. Silybin combined with phosphatidylcholine and vitamin E in patients with nonalcoholic fatty liver disease: a randomized controlled trial. Free Radic Biol Med. 2012;52:1658–65. https://doi.org/10.1016/j.freeradbiomed.2012.02.008.
Cacciapuoti F, Scognamiglio A, Palumbo R, Forte R, Cacciapuoti F. Silymarin in non alcoholic fatty liver disease. World J Hepatol. 2013;5:109–13. https://doi.org/10.4254/wjh.v5.i3.109.
Sorrentino G, Crispino P, Coppola D, Stefano G. Efficacy of lifestyle changes in subjects with non-alcoholic liver steatosis and metabolic syndrome may be improved with an antioxidant nutraceutical: a controlled clinical study. Drugs R&D. 2015;15:21–5. https://doi.org/10.1007/s40268-015-0084-x.
Zhong S, Fan Y, Yan Q, Fan X, Wu B, Han Y, et al. The therapeutic effect of silymarin in the treatment of nonalcoholic fatty disease. Medicine. 2017;96:e9061. https://doi.org/10.1097/md.0000000000009061.
Chitturi S, Wong V, Chan W, Wong G, Wong S, Sollano J, et al. The Asia–Pacific working party on non-alcoholic fatty liver disease guidelines 2017—part 2: management and special groups. Eur J Gastroenterol Hepatol. 2018;33:86–98. https://doi.org/10.1111/jgh.13856.
Wong S, Chin K-Y, Suhaimi F, Ahmad F, Ima-Nirwana S. Vitamin E as a potential interventional treatment for metabolic syndrome: evidence from animal and human studies. Front Pharmacol. 2017;8:444. https://doi.org/10.3389/fphar.2017.00444.
Pastori D, Baratta F, Carnevale R, Cangemi R, Ben M, Bucci T, et al. Similar reduction of cholesterol-adjusted vitamin E serum levels in simple steatosis and non-alcoholic steatohepatitis. Clin Transl Gastroenterol. 2015;6:e113. https://doi.org/10.1038/ctg.2015.43.
Kawanaka M, Mahmood S, Niiyama G, Izumi A, Kamei A, Ikeda H, et al. Control of oxidative stress and reduction in biochemical markers by vitamin E treatment in patients with nonalcoholic steatohepatitis: a pilot study. Hepatol Res. 2004;29:39–41. https://doi.org/10.1016/j.hepres.2004.02.002.
Kim G, Chung J, Lee J, Ok K, Jang E, Kim J, et al. Effect of vitamin E in nonalcoholic fatty liver disease with metabolic syndrome: a propensity score-matched cohort study. Clin Mol Hepatol. 2015;21:379–86. https://doi.org/10.3350/cmh.2015.21.4.379.
Harrison SA, Torgerson S, Hayashi P, Ward J, Schenker S. Vitamin E and vitamin C treatment improves fibrosis in patients with nonalcoholic steatohepatitis. Am J Gastroenterol. 2003;98:ajg2003574. https://doi.org/10.1111/j.1572-0241.2003.08699.x.
Sanyal AJ, Chalasani N, Kowdley KV, McCullough A, Diehl A, Bass NM, et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. New Engl J Med. 2010;362:1675–85. https://doi.org/10.1056/nejmoa0907929.
Lavine JE, Schwimmer JB, Natta ML, Molleston JP, Murray KF, Rosenthal P, et al. Effect of vitamin E or metformin for treatment of nonalcoholic fatty liver disease in children and adolescents: the TONIC randomized controlled trial. JAMA. 2011;305:1659–68. https://doi.org/10.1001/jama.2011.520.
Sato K, Gosho M, Yamamoto T, Kobayashi Y, Ishii N, Ohashi T, et al. Vitamin E has a beneficial effect on nonalcoholic fatty liver disease: a meta-analysis of randomized controlled trials. Nutrition. 2015;31:923–30. https://doi.org/10.1016/j.nut.2014.11.018.
Blond E, Disse E, Cuerq C, Drai J, Valette P-J, Laville M, et al. EASL–EASD–EASO clinical practice guidelines for the management of non-alcoholic fatty liver disease in severely obese people: do they lead to over-referral? Diabetologia. 2017;60:1218–22. https://doi.org/10.1007/s00125-017-4264-9.
Klein EA, Thompson IM, Tangen CM, Crowley JJ, Lucia SM, Goodman PJ, et al. Vitamin E and the risk of prostate cancer: the selenium and vitamin E cancer prevention trial (SELECT). JAMA. 2011;306:1549–56. https://doi.org/10.1001/jama.2011.1437.
Pinelli NR, Jaber LA, Brown MB, Herman WH. Serum 25-Hydroxy vitamin D and insulin resistance, metabolic syndrome, and glucose intolerance among Arab Americans. Diabetes Care. 2010;33:1373–5. https://doi.org/10.2337/dc09-2199.
Eliades M, Spyrou E. Vitamin D: a new player in non-alcoholic fatty liver disease? World J Gastroenterol. 2015;21:1718–27. https://doi.org/10.3748/wjg.v21.i6.1718.
Targher G, Bertolini L, Scala L, Cigolini M, Zenari L, Falezza G, et al. Associations between serum 25-hydroxyvitamin D3 concentrations and liver histology in patients with non-alcoholic fatty liver disease. Nutr Metab Cardiovasc Dis. 2007;17:517–24. https://doi.org/10.1016/j.numecd.2006.04.002.
Keane JT, Elangovan H, Stokes RA, Gunton JE. Vitamin D and the liver—correlation or cause? Nutrients. 2018;10:496. https://doi.org/10.3390/nu10040496.
Salehpour A, Hosseinpanah F, Shidfar F, Vafa M, Razaghi M, Dehghani S, et al. A 12-week double-blind randomized clinical trial of vitamin D3supplementation on body fat mass in healthy overweight and obese women. Nutr J. 2012;11:78. https://doi.org/10.1186/1475-2891-11-78.
Barchetta I, Angelico F, Ben M, Baroni M, Pozzilli P, Morini S, et al. Strong association between non alcoholic fatty liver disease (NAFLD) and low 25(OH) vitamin D levels in an adult population with normal serum liver enzymes. BMC Med. 2011;9:85. https://doi.org/10.1186/1741-7015-9-85.
Barchetta I, Carotti S, Labbadia G, Gentilucci U, Muda A, Angelico F, et al. Liver vitamin D receptor, CYP2R1, and CYP27A1 expression: relationship with liver histology and vitamin D3 levels in patients with nonalcoholic steatohepatitis or hepatitis C virus. Hepatology. 2012;56:2180–7. https://doi.org/10.1002/hep.25930.
Sharifi N, Amani R, Hajiani E, Cheraghian B. Does vitamin D improve liver enzymes, oxidative stress, and inflammatory biomarkers in adults with non-alcoholic fatty liver disease? A randomized clinical trial. Endocrine. 2014;47:70–80. https://doi.org/10.1007/s12020-014-0336-5.
Barchetta I, Ben M, Angelico F, Martino M, Fraioli A, Torre G, et al. No effects of oral vitamin D supplementation on non-alcoholic fatty liver disease in patients with type 2 diabetes: a randomized, double-blind, placebo-controlled trial. BMC Med. 2016;14:92. https://doi.org/10.1186/s12916-016-0638-y.
Kitson MT, Pham A, Gordon A, Kemp W, Roberts SK. High-dose vitamin D supplementation and liver histology in NASH. Gut. 2016;65:717. https://doi.org/10.1136/gutjnl-2015-310417.
Amiri H, Agah S, Azar J, Hosseini S, Shidfar F, Mousavi S. Effect of daily calcitriol supplementation with and without calcium on disease regression in non-alcoholic fatty liver patients following an energy-restricted diet: randomized, controlled, double-blind trial. Clin Nutr. 2017;36:1490–7. https://doi.org/10.1016/j.clnu.2016.09.020.
Jaruvongvanich V, Ahuja W, Sanguankeo A, Wijarnpreecha K, Upala S. Vitamin D and histologic severity of nonalcoholic fatty liver disease: a systematic review and meta-analysis. Dig Liver Dis. 2017;49:618–22. https://doi.org/10.1016/j.dld.2017.02.003.
Gülçin İ. Antioxidant and antiradical activities of l-carnitine. Life Sci. 2006;78:803–11. https://doi.org/10.1016/j.lfs.2005.05.103.
Cuturic M, Abramson RK, Moran RR, Hardin JW, Frank EM, Sellers AA. Serum carnitine levels and levocarnitine supplementation in institutionalized Huntington’s disease patients. Neurol Sci. 2013;34:93–8. https://doi.org/10.1007/s10072-012-0952-x.
Flanagan JL, Simmons PA, Vehige J, Willcox MD, Garrett Q. Role of carnitine in disease. Nutr Metab. 2010;7:30. https://doi.org/10.1186/1743-7075-7-30.
Malaguarnera M, Gargante M, Russo C, Antic T, Vacante M, Malaguarnera M, et al. L-carnitine supplementation to diet: a new tool in treatment of nonalcoholic Steatohepatitis—a randomized and controlled clinical trial. Am J Gastroenterol. 2010;105:1338. https://doi.org/10.1038/ajg.2009.719.
Somi MH, Fatahi E, Panahi J, Havasian MR, Judaki A. Data from a randomized and controlled trial of LCarnitine prescription for the treatment for non- alcoholic fatty liver disease. Bioinformation. 2014;10:575–9. https://doi.org/10.6026/97320630010575.
van Herpen NA, Schrauwen-Hinderling VB. Lipid accumulation in non-adipose tissue and lipotoxicity. Physiol Behav. 2008;94:231–41. https://doi.org/10.1016/j.physbeh.2007.11.049.
Shahidi F, Ambigaipalan P. Omega-3 Polyunsaturated Fatty Acids and Their Health Benefits. Annu Rev Food Sci Technol. 2018;9:345–381.
Capanni M, Calella F, Ini M, Genise S, Raimondi L, Bedogni G, et al. Prolonged n-3 polyunsaturated fatty acid supplementation ameliorates hepatic steatosis in patients with non-alcoholic fatty liver disease: a pilot study. Aliment Pharmacol Ther. 2006;23:1143–51. https://doi.org/10.1111/j.1365-2036.2006.02885.x.
Spadaro L, Magliocco O, Spampinato D, Piro S, Oliveri C, Alagona C, et al. Effects of n-3 polyunsaturated fatty acids in subjects with nonalcoholic fatty liver disease. Dig Liver Dis. 2008;40:194–9. https://doi.org/10.1016/j.dld.2007.10.003.
Sofi F, Giangrandi I, Cesari F, Corsani I, Abbate R, Gensini G, et al. Effects of a 1-year dietary intervention with n-3 polyunsaturated fatty acid-enriched olive oil on non-alcoholic fatty liver disease patients: a preliminary study. Int J Food Sci Nutr. 2010;61:792–802. https://doi.org/10.3109/09637486.2010.487480.
Vega G, Chandalia M, Szczepaniak LS, Grundy SM. Effects of N-3 fatty acids on hepatic triglyceride content in humans. J Investig Med. 2008;56:780. https://doi.org/10.2310/jim.0b013e318177024d.
Nobili V, Bedogni G, Alisi A, Pietrobattista A, Risé P, Galli C, et al. Docosahexaenoic acid supplementation decreases liver fat content in children with non-alcoholic fatty liver disease: double-blind randomised controlled clinical trial. Arch Dis Child. 2011;96:350. https://doi.org/10.1136/adc.2010.192401.
Parker HM, Johnson NA, Burdon CA, Cohn JS, O’Connor HT, George J. Omega-3 supplementation and non-alcoholic fatty liver disease: a systematic review and meta-analysis. J Hepatol. 2012;56:944–51. https://doi.org/10.1016/j.jhep.2011.08.018.
Wrieden WL, Hannah MK, Bolton-Smith C, Tavendale R, Morrison C, Tunstall-Pedoe H. Plasma vitamin C and food choice in the third Glasgow MONICA population survey. J Epidemiol Commun H. 2000;54:355. https://doi.org/10.1136/jech.54.5.355.
Hampl JS, Taylor CA, Johnston CS. Vitamin C deficiency and depletion in the United States: the third national health and nutrition examination survey, 1988 to 1994. Am J Public Health. 2004;94:870–5. https://doi.org/10.2105/ajph.94.5.870.
Chen G, Lu D, Pang Z, Liu Q. Vitamin C intake, circulating vitamin C and risk of stroke: a meta-analysis of prospective studies. J Am Heart Assoc. 2013;2:e000329. https://doi.org/10.1161/jaha.113.000329.
Moser MA, Chun OK. Vitamin C and heart health: a review based on findings from epidemiologic studies. Int J Mol Sci. 2016;17:1328. https://doi.org/10.3390/ijms17081328.
Ipsen D, Tveden-Nyborg P, Lykkesfeldt J. Does vitamin C deficiency promote fatty liver disease development? Nutrients. 2014;6:5473–99. https://doi.org/10.3390/nu6125473.
Ferolla S, Ferrari T, Lima M, Reis T, Tavares W Jr, Couto O, et al. Dietary patterns in Brazilian patients with non-alcoholic fatty liver disease: a cross-sectional study. Clinics. 2013;68:11–7. https://doi.org/10.6061/clinics/2013(01)oa03.
Musso G, Gambino R, Michieli F, Cassader M, Rizzetto M, Durazzo M, et al. Dietary habits and their relations to insulin resistance and postprandial lipemia in nonalcoholic steatohepatitis. Hepatology. 2003;37:909–16. https://doi.org/10.1053/jhep.2003.50132.
Mager D, Patterson C, So S, Rogenstein C, Wykes L, Roberts E. Dietary and physical activity patterns in children with fatty liver. Eur J Clin Nutr. 2010;64:628. https://doi.org/10.1038/ejcn.2010.35.
Silva HE, Arendt BM, Noureldin SA, Therapondos G, Guindi M, Allard JP. A cross-sectional study assessing dietary intake and physical activity in Canadian patients with nonalcoholic fatty liver disease vs healthy controls. J Acad Nutr Diet. 2014;114:1181–94. https://doi.org/10.1016/j.jand.2014.01.009.
Madan K, Bhardwaj P, Thareja S, Gupta SD, Saraya A. Oxidant stress and antioxidant status among patients with nonalcoholic fatty liver disease (NAFLD). J Clin Gastroenterol. 2006;40:930–5. https://doi.org/10.1097/01.mcg.0000212608.59090.08.
Nobili V, Manco M, Devito R, Ciampalini P, Piemonte F, Marcellini M. Effect of vitamin E on aminotransferase levels and insulin resistance in children with non-alcoholic fatty liver disease. Aliment Pharmacol Ther. 2006;24:1553–61. https://doi.org/10.1111/j.1365-2036.2006.03161.x.
Arad Y, Spadaro LA, Roth M, Newstein D, Guerci AD. Treatment of asymptomatic adults with elevated coronary calcium scores with atorvastatin, vitamin C, and vitamin E: the St. Francis heart study randomized clinical trial. ACC Curr J Rev. 2005;14:10. https://doi.org/10.1016/j.accreview.2005.09.028.
Ayers J, Cook J, Koenig RA, Sisson EM, Dixon DL. Recent developments in the role of coenzyme Q10 for coronary heart disease: a systematic review. Curr Atheroscler Rep. 2018;20:29. https://doi.org/10.1007/s11883-018-0730-1.
Gutierrez-Mariscal FM, Yubero-Serrano EM, Villalba JM, Lopez-Miranda J. Coenzyme Q10: from bench to clinic in aging diseases, a translational review. Crit Rev Food Sci. 2018;2018:1–63. https://doi.org/10.1080/10408398.2018.1442316.
Farsi F, Mohammadshahi M, Alavinejad P, Rezazadeh A, Zarei M, Engali K. Functions of coenzyme Q10 supplementation on liver enzymes, markers of systemic inflammation, and adipokines in patients affected by nonalcoholic fatty liver disease: a double-blind, placebo-controlled, randomized clinical trial. J Am Coll Nutr. 2015;35:346–53. https://doi.org/10.1080/07315724.2015.1021057.
Sharifi N, Tabrizi R, Moosazadeh M, Mirhosseini N, Lankarani KB, Akbari M, et al. The effects of coenzyme Q10 supplementation on lipid profiles among patients with metabolic diseases: a systematic review and meta-analysis of randomized controlled trials. Curr Pharm Design. 2018;17(1):123. https://doi.org/10.2174/1381612824666180406104516.
Wei S, Zhang M, Yu Y, Lan X, Yao F, Yan X, et al. Berberine attenuates development of the hepatic gluconeogenesis and lipid metabolism disorder in type 2 diabetic mice and in palmitate-incubated HepG2 cells through suppression of the HNF-4α miR122 pathway. PLoS One. 2016;11:e0152097. https://doi.org/10.1371/journal.pone.0152097.
Vuddanda P, Chakraborty S, Singh S. Berberine: a potential phytochemical with multispectrum therapeutic activities. Expert Opin Investig Drugs. 2010;19:1297–307. https://doi.org/10.1517/13543784.2010.517745.
Guo T, Woo S-L, Guo X, Li H, Zheng J, Botchlett R, et al. Berberine ameliorates hepatic steatosis and suppresses liver and adipose tissue inflammation in mice with diet-induced obesity. Sci Rep-Uk. 2016;6:22612. https://doi.org/10.1038/srep22612.
Cao Y, Pan Q, Cai W, Shen F, Chen G-Y, Xu L-M, et al. Modulation of gut microbiota by berberine improves steatohepatitis in high-fat diet-fed BALB/C mice. Arch Iran Med. 2016;19:197–203.
Cicero AF, Baggioni A. Anti-inflammatory nutraceuticals and chronic diseases. Adv Exp Med Biol. 2016;928:27–45. https://doi.org/10.1007/978-3-319-41334-1_2.
Wei X, Wang C, Hao S, Song H, Yang L. The therapeutic effect of Berberine in the treatment of nonalcoholic fatty liver disease: a meta-analysis. Evid Based Complement Alternat Med. 2016;2016:3593951. https://doi.org/10.1155/2016/3593951.
Panahi Y, Hosseini M, Khalili N, Naimi E, Majeed M, Sahebkar A. Antioxidant and anti-inflammatory effects of curcuminoid-piperine combination in subjects with metabolic syndrome: a randomized controlled trial and an updated meta-analysis. Clin Nutr. 2015;34:1101–8. https://doi.org/10.1016/j.clnu.2014.12.019.
Panahi Y, Hosseini M, Khalili N, Naimi E, Simental-Mendía LE, Majeed M, et al. Effects of curcumin on serum cytokine concentrations in subjects with metabolic syndrome: a post-hoc analysis of a randomized controlled trial. Biomed Pharmacother. 2016;82:578–82. https://doi.org/10.1016/j.biopha.2016.05.037.
Panahi Y, Hosseini M, Khalili N, Naimi E, Soflaei S, Majeed M, et al. Effects of supplementation with curcumin on serum adipokine concentrations: a randomized controlled trial. Nutrition. 2016;32:1116–22. https://doi.org/10.1016/j.nut.2016.03.018.
Panahi Y, Khalili N, Hosseini M, Abbasinazari M, Sahebkar A. Lipid-modifying effects of adjunctive therapy with curcuminoids–piperine combination in patients with metabolic syndrome: results of a randomized controlled trial. Complement Ther Med. 2014;22:851–7. https://doi.org/10.1016/j.ctim.2014.07.006.
Panahi Y, Kianpour P, Mohtashami R, Jafari R, Simental-Mendía LE, Sahebkar A. Efficacy and safety of phytosomal curcumin in non-alcoholic fatty liver disease: a randomized controlled trial. Drug Res. 2017;67:244–51. https://doi.org/10.1055/s-0043-100019.
Sivaprakasapillai B, Edirisinghe I, Randolph J, Steinberg F, Kappagoda T. Effect of grape seed extract on blood pressure in subjects with the metabolic syndrome. Metabolism. 2009;58:1743–6. https://doi.org/10.1016/j.metabol.2009.05.030.
Zhao L, Guo X, Wang O, Zhang H, Wang Y, Zhou F, et al. Fructose and glucose combined with free fatty acids induce metabolic disorders in HepG2 cell: a new model to study the impacts of high-fructose/sucrose and high-fat diets in vitro. Mol Nutr Food Res. 2016;60:909–21. https://doi.org/10.1002/mnfr.201500635.
Faghihzadeh F, Adibi P, Hekmatdoost A. The effects of resveratrol supplementation on cardiovascular risk factors in patients with non-alcoholic fatty liver disease: a randomised, double-blind, placebo-controlled study. Br J Nutr. 2015;114:796–803. https://doi.org/10.1017/s0007114515002433.
Chen S, Zhao X, Ran L, Wan J, Wang X, Qin Y, et al. Resveratrol improves insulin resistance, glucose and lipid metabolism in patients with non-alcoholic fatty liver disease: a randomized controlled trial. Dig Liver Dis. 2015;47:226–32. https://doi.org/10.1016/j.dld.2014.11.015.
Chachay VS, Macdonald GA, Martin JH, Whitehead JP, O’Moore–Sullivan TM, Lee P, et al. Resveratrol does not benefit patients with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol. 2014;12:2092–2103.e6. https://doi.org/10.1016/j.cgh.2014.02.024.
Peng H, He Y, Zheng G, Zhang W, Yao Z, Xie W. Meta-analysis of traditional herbal medicine in the treatment of nonalcoholic fatty liver disease. Cell Mol Biol (Noisy-le-Grand). 2016;62:88–95.
Ma J, Zhou Q, Li H. Gut microbiota and nonalcoholic fatty liver disease: insights on mechanisms and therapy. Nutrients. 2017;9:1124. https://doi.org/10.3390/nu9101124.
Lavekar AS, Raje DV, Manohar T, Lavekar AA. Role of probiotics in the treatment of nonalcoholic fatty liver disease: a meta-analysis. Euroasian J Hepato-Gastroenterol. 2017;7:130–7. https://doi.org/10.5005/jp-journals-10018-1233.
Aller R, Luis DD, Izaola O, Conde R, Sagrado GM, Primo D, et al. Effect of a probiotic on liver aminotransferases in nonalcoholic fatty liver disease patients: a double blind randomized clinical trial. Eur Rev Med Pharmacol Sci. 2011;15:1090–5.
Vajro P, Mandato C, Licenziati M, Franzese A, Vitale D, Lenta S, et al. Effects of Lactobacillus rhamnosus strain GG in pediatric obesity-related liver disease. J Pediatr Gastroenterol Nutr. 2011;52:740–3. https://doi.org/10.1097/mpg.0b013e31821f9b85.
Nabavi S, Rafraf M, Somi MH, Homayouni-Rad A, Asghari-Jafarabadi M. Effects of probiotic yogurt consumption on metabolic factors in individuals with nonalcoholic fatty liver disease. J Dairy Sci. 2014;97:7386–93. https://doi.org/10.3168/jds.2014-8500.
Alisi A, Bedogni G, Baviera G, Giorgio V, Porro E, Paris C, et al. Randomised clinical trial: the beneficial effects of VSL#3 in obese children with non-alcoholic steatohepatitis. Aliment Pharmacol Ther. 2014;39:1276–85. https://doi.org/10.1111/apt.12758.
Suda I, Ishikawa F, Hatakeyama M, Miyawaki M, Kudo T, Hirano K, et al. Intake of purple sweet potato beverage affects on serum hepatic biomarker levels of healthy adult men with borderline hepatitis. Eur J Clin Nutr. 2007;62:1602674. https://doi.org/10.1038/sj.ejcn.1602674.
Zhang P-W, Chen F-X, Li D, Ling W-H, Guo H-H. A CONSORT-compliant, randomized, double-blind, placebo-controlled pilot trial of purified anthocyanin in patients with nonalcoholic fatty liver disease. Medicine. 2015;94:e758. https://doi.org/10.1097/md.0000000000000758.
Deminice R, da Silva RP, Lamarre SG, Kelly KB, Jacobs RL, Brosnan ME, et al. Betaine supplementation prevents fatty liver induced by a high-fat diet: effects on one-carbon metabolism. Amino Acids. 2015;47:839–46. https://doi.org/10.1007/s00726-014-1913-x.
Kawakami S, Han K-H, Nakamura Y, Shi K, Kitano T, Aritsuka T, et al. Effects of dietary supplementation with betaine on a nonalcoholic steatohepatitis (NASH) mouse model. J Nutr Sci Vitaminol. 2012;58:371–5. https://doi.org/10.3177/jnsv.58.371.
Chen Y, Liu Y, Zhou R, Chen X, Wang C, Tan X, et al. Associations of gut-flora-dependent metabolite trimethylamine-N-oxide, betaine and choline with non-alcoholic fatty liver disease in adults. Sci Rep-Uk. 2016;6:19076. https://doi.org/10.1038/srep19076.
Miglio F, Rovati L, Santoro A, Setnikar I. Efficacy and safety of oral betaine glucuronate in non-alcoholic steatohepatitis. Arzneimittelforschung. 2011;50:722–7. https://doi.org/10.1055/s-0031-1300279.
Abdelmalek MF, Sanderson SO, Angulo P, Soldevila-Pico C, Liu C, Peter J, et al. Betaine for nonalcoholic fatty liver disease: results of a randomized placebo-controlled trial. Hepatology. 2009;50:1818–26. https://doi.org/10.1002/hep.23239.
Venables MC, Hulston CJ, Cox HR, Jeukendrup AE. Green tea extract ingestion, fat oxidation, and glucose tolerance in healthy humans. Am J Clin Nutr. 2008;87:778–84. https://doi.org/10.1093/ajcn/87.3.778.
Shimotoyodome A, Haramizu S, Inaba K, Murase T, Tokimitsu I. Exercise and green tea extract stimulate fat oxidation and prevent obesity in mice. Med Sci Sports Exerc. 2005;37:1884–92. https://doi.org/10.1249/01.mss.0000178062.66981.a8.
Murase T, Haramizu S, Shimotoyodome A, Nagasawa A, Tokimitsu I. Green tea extract improves endurance capacity and increases muscle lipid oxidation in mice. Am J Phys Regul Integr Comp Phys. 2005;288:R708–15. https://doi.org/10.1152/ajpregu.00693.2004.
Pezeshki A, Safi S, Feizi A, Askari G, Karami F. The effect of green tea extract supplementation on liver enzymes in patients with nonalcoholic fatty liver disease. Int J Prev Med. 2015;6:131. https://doi.org/10.4103/2008-7802.173051.
Hussain M, Habib-Ur-Rehman AL. Therapeutic benefits of green tea extract on various parameters in non-alcoholic fatty liver disease patients. Pak J Med Sci. 2017;33:931–6. https://doi.org/10.12669/pjms.334.12571.
Sakata R, Nakamura T, Torimura T, Ueno T, Sata M. Green tea with high-density catechins improves liver function and fat infiltration in non-alcoholic fatty liver disease (NAFLD) patients: a double-blind placebo-controlled study. Int J Mol Med. 2013;32:989–94. https://doi.org/10.3892/ijmm.2013.1503.
Salomone F, Volti G, Vitaglione P, Morisco F, Fogliano V, Zappalà A, et al. Coffee enhances the expression of chaperones and antioxidant proteins in rats with nonalcoholic fatty liver disease. Transl Res. 2014;163:593–602. https://doi.org/10.1016/j.trsl.2013.12.001.
Watanabe S, Takahashi T, Ogawa H, Uehara H, Tsunematsu T, Baba H, et al. Daily coffee intake inhibits pancreatic beta cell damage and nonalcoholic steatohepatitis in a mouse model of spontaneous metabolic syndrome, Tsumura-Suzuki obese diabetic mice. Metab Syndr Relat Disord. 2017;15:170–7. https://doi.org/10.1089/met.2016.0114.
Bambha K, Wilson LA, Unalp A, Loomba R, Neuschwander-Tetri BA, Brunt EM, et al. Coffee consumption in NAFLD patients with lower insulin resistance is associated with lower risk of severe fibrosis. Liver Int. 2014;34:1250–8. https://doi.org/10.1111/liv.12379.
Zelber-Sagi S, Salomone F, Webb M, Lotan R, Yeshua H, Halpern Z, et al. Coffee consumption and nonalcoholic fatty liver onset: a prospective study in the general population. Transl Res. 2015;165:428–36. https://doi.org/10.1016/j.trsl.2014.10.008.
Gutiérrez-Grobe Y, Chávez-Tapia N, Sánchez-Valle V, Gavilanes-Espinar J, Ponciano-Rodríguez G, Uribe M, et al. High coffee intake is associated with lower grade nonalcoholic fatty liver disease: the role of peripheral antioxidant activity. Ann Hepatol. 2012;11:350–5. https://doi.org/10.1016/s1665-2681(19)30931-7.
Wijarnpreecha K, Thongprayoon C, Ungprasert P. Coffee consumption and risk of nonalcoholic fatty liver disease. Eur J Gastroenterol Hepatol. 2017;29:e8–12. https://doi.org/10.1097/meg.0000000000000776.
Karkos P, Leong S, Karkos C, Sivaji N, Assimakopoulos D. Spirulina in clinical practice: evidence-based human applications. Evid Based Complement Alternat Med. 2011;2011:531053. https://doi.org/10.1093/ecam/nen058.
Coué M, Tesse A, Falewée J, Aguesse A, Croyal M, Fizanne L, et al. Spirulina liquid extract protects against fibrosis related to non-alcoholic steatohepatitis and increases ursodeoxycholic acid. Nutrients. 2019;11:194. https://doi.org/10.3390/nu11010194.
Mazokopakis EE, Papadomanolaki MG, Fousteris AA, Kotsiris DA, Lampadakis IM, Ganotakis ES. The hepatoprotective and hypolipidemic effects of Spirulina (Arthrospira platensis) supplementation in a Cretan population with non-alcoholic fatty liver disease: a prospective pilot study. Ann Gastroenterol. 2014;27:387–94.
Mora SI, García-Román J, Gómez-Ñañez I, García-Román R. Chronic liver diseases and the potential use of S-adenosyl-L-methionine as a hepatoprotector. Eur J Gastroenterol Hepatol. 2018;30:893–900. https://doi.org/10.1097/meg.0000000000001141.
Zubiete-Franco I, García-Rodríguez J, Martínez-Uña M, Martínez-Lopez N, Woodhoo A, Juan V, et al. Methionine and S-adenosylmethionine levels are critical regulators of PP2A activity modulating lipophagy during steatosis. J Hepatol. 2016;64:409–18. https://doi.org/10.1016/j.jhep.2015.08.037.
Ni Y, Nagashimada M, Zhuge F, Zhan L, Nagata N, Tsutsui A, et al. Astaxanthin prevents and reverses diet-induced insulin resistance and steatohepatitis in mice: a comparison with vitamin E. Sci Rep-Uk. 2015;5:srep17192. https://doi.org/10.1038/srep17192.
Chen G, Ni Y, Nagata N, Xu L, Ota T. Micronutrient antioxidants and nonalcoholic fatty liver disease. Int J Mol Sci. 2016;17:1379. https://doi.org/10.3390/ijms17091379.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Sonmez, A., Ercin, C.N., Cesur, M., Dogru, T. (2021). Nutraceuticals for Non-alcoholic Fatty Liver Disease. In: Cicero, A.F., Rizzo, M. (eds) Nutraceuticals and Cardiovascular Disease. Contemporary Cardiology. Humana, Cham. https://doi.org/10.1007/978-3-030-62632-7_9
Download citation
DOI: https://doi.org/10.1007/978-3-030-62632-7_9
Published:
Publisher Name: Humana, Cham
Print ISBN: 978-3-030-62631-0
Online ISBN: 978-3-030-62632-7
eBook Packages: MedicineMedicine (R0)