Abstract
Isolated forest patches of Nothofagus antarctica (ñire) are frequent in the Patagonian forest-steppe ecotone. These remnants, also called relicts (R), are separate from the continuous forests (C). Over the past century, these ecotonal forests have been impacted by anthropogenic activities, including fires, logging, and cattle ranching. In order to identify in N. antarctica ring-width records the variations in tree growth associated with documented changes in land use, five sites were selected in Santa Cruz, Argentina. In each site, increment cores from R and C were collected. We developed individual chronologies, and the relationships between regional climate variations and N. antarctica growth were established for each forest type and site. The similarities/differences between site-paired chronologies (R-C) were estimated by calculating moving correlation coefficients lagged by 1 year. N. antarctica regional growth was directly related to precipitation during the current growing season (November–December; r = 0.34, n = 62, p < 0.01), and inversely related to temperature (December–March; r = −0.58, n = 62, p < 0.001). Since the middle of the twentieth century, a progressive decrease has been recorded in regional radial growth, consistent with an increase in summer temperature and a decrease in spring precipitation. In the context of this regional response of N. antarctica to climate, differences in growth patterns between R and C were associated with past changes in land use. Overall, the largest differences between R and C chronologies were concurrent with the settlement of cattle ranches and the associated use of forests. Conversely, similarities between R and C records increased after the establishment of protected areas and during the implementation of similar management practices in both forest types. Our research provides the first dendrochronological records from Nothofagus antarctica for the Argentinean Patagonia and represents one of the first efforts to identify in tree-rings past changes in livestock practices in southern South America.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Forest-steppe ecotone
- Relict and continuous forest
- Dendrochronology
- Climate-tree growth relationship
- Historical and cultural use of the land
- Dendroclimatology
1 Introduction
The genus Nothofagus is widely distributed in the continental and insular sectors bordering the South Pacific Ocean, particularly in the mid-latitudes of Australasia and South America (Veblen et al. 1996). Nothofagus antarctica (G. Forster) Oerster (ñire or ñirre) and Nothofagus pumilio (Poepp. and Endl.) Krasser (lenga) have the most extensive geographic distributions of the genus in South America, covering 2200 km along the Andes from 35° 36’ S in Central Chile to 55° S in Tierra del Fuego (Donoso 1993; Lara et al. 2001). The altitudinal range of these species extends from sea level to the upper tree line (Veblen et al. 1996; Roig 1999).
It is known that the Andes induce a marked precipitation gradient across Patagonia (Paruelo et al. 1998). The Nothofagus forests are located in the western, most humid areas of this gradient (Moore 1983; Donoso 1993). Along the west-east precipitation gradient in southern Patagonia, N. antarctica forests develop within a precipitation range that varies between 3000 mm to less than 800 mm over a west-to-east distance of only 50 km (Veblen et al. 1996; Paruelo et al. 1998). In response to its high phenotypic plasticity, N. antarctica presents a large ecological amplitude (Ramírez et al. 1985) growing from wet meadows (mallines) to dry areas in the forest-steppe ecotone. Furthermore, N. antarctica grows at flat-low valley sites as well as on steep slopes with strong and persistent winds (Veblen et al. 1996; Donoso et al. 2006; Kreps et al. 2012). In addition, N. antarctica tolerates winter frosts up to −22 °C (Alberdi 1995). In consequence, N. antarctica shows a great adaptive environmental capacity and multiple phenotypic responses. Under suitable site conditions (moderate temperature, adequate humidity, well-drained and fertile soils), N. antarctica is an erect tree of up to 20 m in height (Marazana et al. 1984; Ramírez et al. 1985; Veblen et al. 1996). By contrast, under extreme soil moisture conditions or at the upper treeline, it becomes a small tree or shrub of approximately 2–3 m height. Shrubby individuals (krumholz) are common in upper tree lines due to abundant snow deposition in winter (Pérez Moreau 1959; Veblen et al. 1996; Donoso et al. 2006). In the forest-steppe ecotone, it is common to observe trees up to 10 m coexisting with individuals less than 3 m in height (Marazana et al. 1984; Ramírez et al. 1985).
In the forest-steppe ecotone, N. antarctica usually forms the eastern boundary of the arboreal vegetation. In this ecotone, trees group into a mosaic of small monospecific patches of forests that alternate with the steppe in elevated areas and with meadows or mallines in depressions (Roig 1999; Peri and Ormaechea 2013). These remaining patches, called relicts (R), are marginal forest communities spatially disconnected with the continuous forests (C) located towards the west over the Andes mountain range (Fig. 15.1).
Photograph showing sectors of the relict (black arrow) and continuous (grey arrow) forests of N. antarctica in Estancia Cancha Carrera (site 3). The relict forests are represented by isolated patches bordered by steppe vegetation, while the most developed continuous forest extends all along the foothills of the Andes
The ecotones are considered very dynamic habitats where the initial impacts of environmental changes become more evident (di Castri et al. 1988; Neilson 1991). In this sense, several authors (Iverson et al. 2004; Hampe and Petit 2005) claim that the populations of tree species located on their distribution margins are very sensitive to environmental variations. Due to the high perimeter/surface ratio, trees at the ecotonal patches are exposed to more severe stress and higher rates of change than trees in continuous forests (Murcia 1995).
Ecotones have often been heavily impacted by human activities, particularly at the low elevation forest-grassland transitions. Changes in cultural land use have greatly influenced their dynamics (Kitzberger 2012). In southern Patagonia, the N. antarctica relicts have been subjected to different cultural uses, and only 14% of these are included in protected areas. The European immigration in the late-nineteenth and early-twentieth centuries caused the demise of the native Aonikenk hunter-gatherer population and the rapid establishment of livestock farming (Bandieri 2005). The new socio-economic system gradually modified the forests in the region (Barbería 1994, 1995; Schweitzer 2011). The N. antarctica forests were intensely used during the European colonization inducing a substantial reduction in the total area of the ecotonal forests. Among the various anthropogenic causes related to the decrease in N. antarctica forest extension were the opening of new roads, unplanned settlements, intensive grazing, forest fires, and logging (Montaña 1982; Manacorda and Bonvissuto 2001). Livestock causes the browsing of arboreal species and limits the natural regeneration of the forest. Livestock also facilitates the introduction of exotic plants that compete with native species (Christensen and Emborg 1996; SAyDS 2005). There have also been registered increments of certain understory taxa resistant to browsing, such as the Berberis sp. (Rajchenberg et al. 2014). As a consequence, the overconsumption and unsustainable use of natural resources led to the accelerated transformation of the environment (Schweitzer 2011). Concurrent with these vegetation changes, a general warming trend at the extreme south of South America (45–55° S) has become evident during the last century (Rosenblüth et al. 1997; Villalba et al. 2003; SAyDS 2005) concurrent with a decrease in precipitation (Aravena and Luckman 2009). These climatic changes, in combination with other local anthropic factors, may have also contribute to the fragmentation of native forests along the region.
It is well known that tree rings can record information about changes in the environmental factors affecting tree growth. However, few studies have been conducted in South America to determine variations in tree rings associated with past changes in land use related to livestock (but see Morales et al. 2005; Morales and Villalba 2006; Mundo et al. 2017). The objectives of this study were to use ring-width chronologies from N. antarctica to identify the species response to climatic variations and determine the influence of different land use practices between relict and continuous forests in Santa Cruz, Argentina. To reach these goals, we first established the relationships between N. antarctica tree growth and the regional climate. Based on these relationships, we postulated that historical changes in stocking density and harvesting intensity have led to important differences in the radial growth of N. antarctica at isolated and continuous forests in the eastern limit of its natural distribution. Changes in cultural land use may provide clues to understand the differences observed in tree growth between nearby stands. Although more than 300 tree-ring chronologies have been developed from different species in Argentina (Villalba et al. 1997; Villalba et al. 2003; Roig and Villalba 2008; Boninsegna et al. 2009), to the best of our knowledge, there are no dendrochronological records from N. antarctica trees. In consequence, the present study provides the first chronologies for N. antarctica in the Argentinean Patagonia.
2 Methods
2.1 Study Area
Located in southern South America, Patagonia presents a cold climate. In Santa Cruz, there are two main geo-climatic units: the Andes dominated by ice and forests with relatively abundant rainfalls to the west, and the Patagonian steppe dominated by grasslands with dry climate to the east (Díaz 2005). The difficulty of analyzing climatic variability in the region lies in the lack of weather stations, or the short extension of available records (Paruelo et al. 1998; Díaz 2005; Garreaud et al. 2013; Soto-Rogel and Aravena 2017). In the study area, the mean annual temperature is 6 °C, with a mean maximum of 17 °C in January and a mean minimum of −5 °C in July (Kreps et al. 2012). The reported absolute minimum temperature for the region is −20 °C (Paruelo et al. 1998). Total annual rainfalls at the study sites is estimated around 400 mm ± 150 mm. In this region, strong westerly winds prevail throughout the year but intensify during the spring and summer (Coronato and Del Valle 1993; González and Vera 2010).
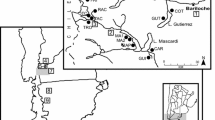
We selected pairs of relict (R) and continuous forests (C) of N. antarctica in five sites in southwestern Santa Cruz province, Argentina. These forests are distributed in a latitudinal range of 467 km, between 47° 04´ W and 51° 05´ S in southern Patagonia (Fig. 15.2). Table 15.1 summarizes the most relevant climate data and the location of each study site.
Geographical distribution of the study sites are indicated by white dots: (1) Perito Moreno National Park, (2) Río Guanaco – Los Glaciares National Park, (3) Estancia Cancha Carrera, (4) Punta Gruesa Natural Reserve and (5) Estancia Rincón de los Morros. Protected areas (shown in shaded blue). Satellite images (framed in white) for the three study sites in which good-quality chronologies were developed are also included. Relict and continuous forests in these three images are indicated by black and grey dots, respectively
2.2 Tree-Ring Data and Chronology Development
Dendrochronological samplings were conducted during the interval 2012–2016 in relict (R) and continuous (C) forests at five study areas (Fig. 15.1). In each site, 20 dominant individuals were selected, and 2 cores were obtained by tree using increment borers. Even though individuals with the best health status were chosen, in many cases only one sample could be recovered per tree. N. antarctica commonly presents tortuous stems of poor sanitary status with rotten centers. For the most part, samples were taken close to the root collar of the trees. However, when sampling at the collar was impeded by logistical problems, cores were extracted at the lowest possible height on the stem.
The samples were processed according to conventional techniques used in dendrochronology (Stokes and Smiley 1968). Tree rings were dated under a binocular magnifier, and the ring widths were measured with an accuracy of 0.001 mm using a Velmex machine (Fig. 15.3). After that, the quality of the measurements was examined using the COFECHA software (Holmes 1983). Finally, the ARSTAN program (Cook 1985) was used to develop raw, standard and residual chronologies from each pair of N. antarctica stands (R and C) at the five study sites. In addition, all series were combined to develop three additional chronologies: two independent chronologies for relict (R) and continuous (C) forests, and a regional chronology including all series from the five study areas. This set of chronologies was developed to validate different hypotheses. Thus, to identify differences in tree growth between relicts and continuous forests due to changes in land use, we developed chronologies in both forest types at each study area. Regional scale chronologies were elaborated to determine the existence of a common growth signal among all sampled trees and to establish differences between relicts and continuous forests .
An incremental core of N. antarctica showing the annual bands (white arrows), clearly discernible in the wood of this species. Although this is a diffuse porous species, a common feature of most Nothofagus species, the vessels at the beginning of the growing season are larger and their cell walls thinner than those formed later in the growing season. Fibers are abundant at the end of the growing season and show thick cell walls
2.3 Climate-Tree Growth Relationship
To determine the climate influences on the N. antarctica radial growth, the residual ring-width chronologies from the three well-replicated sites (sites 2, 3 and 4; Fig. 15.2) were compared with the instrumental temperature and precipitation records available for the region. The correlation function is a simple method for comparing the interannual variations in tree-ring chronologies with monthly interannual fluctuations in climate (Blasing et al. 1984). The statistical relationship between ring-width and climate variable was examined over the common 1952–2014 period (62 years) between the chronologies and the instrumental data. In addition, since the growth in a given year may be influenced by the climatic conditions during the previous years (Fritts 1976), the comparison period analyzed comprised 20 months, starting in September of the previous year until April of the year in which the rings were formed. Based on the results of the correlation function comparing variations between the regional chronology and the regional climate, we proceeded to group the monthly temperature and precipitation records into seasonal means including the months most strongly related to growth. To carry out these calculations, the Microsoft Excel spreadsheet (2013) was used.
2.4 Cultural Land Use Changes
Land uses changes in the forest-steppe ecotone in Santa Cruz were compiled from several historical documents, including the Memories from the National Park Administration, scientific publications on regional anthropological and archaeological studies, management plans of protected areas, technical reports and documents from the Santa Cruz Cadastral Department. Bibliography and files deposited at the Provincial Historical Archive in Río Gallegos were also consulted. Open interviews were also conducted among the residents and cattle ranch administrators.
Finally, the standard version of the tree-ring width chronologies from N. antarctica, obtained for R and C forests at each site, were compared by calculating the correlation coefficient in segments of 20 years lagged by 1-year between consecutive segments. To carry out these calculations, the Microsoft Excel spreadsheet (2013) was used. Finally, we analyzed the consistency between historical changes in land use and in growth patterns .
3 Results
3.1 Chronology Development
Eleven chronologies from N. antarctica were developed in Santa Cruz, one from each site and forest type (R2, C2, R3, C3, R4, C4, R5, and C5), with the exception of site 1 (Perito Moreno National Park), where samples were not cross-dated. Two other chronologies were developed using all the cross-dated series of each forest type (relicts and continuous respectively) and, finally, a regional chronology with all cross-dated series in southwestern Santa Cruz (Fig. 15.4).
Composite N. antarctica width chronologies in their standard versions. The left and right axes represent the tree-ring width index (dimensionless) and the number of series per year (replication), respectively; in black is represented the regional relict forest (R); in light grey the regional continuous forest (C) and in blue the regional chronology. Forest type and site codes are listed in Table 15.1
The longest chronology, extended for 206 years, is from the Cancha Carrera relict forest (R3). On the other hand, the shortest chronology is from Rincón de los Morros continuous forest (C5), and it extended for 42 years. The mean length of all chronologies per site was 122 ± 54 years long (Table 15.2).
The composite R chronology was considerably longer than the composite C chronology (206 and 125 years, respectively). The regional chronology began in 1807 with 1 sample, reached 10 samples in 1888, a maximum of 133 samples between 1992 and 1995, and ended with 73 samples in 2014 (Fig. 15.5).
N. antarctica tree-ring width chronologies in their standard version. The left axis represents the tree-ring width index (dimensionless); the right axis represents the number of series per year (replication); in black are represented the relict forests (R), and in light grey are represented the continuous forests (C). Forest type and site codes are listed in Table 15.1
3.2 Climate-Tree Growth Relationship
The patterns of temperature variability are relatively uniform across our study area. Thus, the most extensive local temperature record in Torre Glacier (from 2002 to the present) is near the town of El Chaltén. This is significantly correlated with gridded data from ERA-Interin across most southern Patagonia (Fig. 15.S1). The extensive field of spatial correlation includes the Argentinean weather stations of Esquel and Río Gallegos and Punta Arenas in Chile (Fig. 15.S1). Located to the north, east and south of the study sites, respectively, these weather stations provide the most extensive and homogeneous records in southern Patagonia. Therefore, the mean from the monthly temperature deviations from these three stations were composed in a single regional record, and it was used to compare with tree-ring variations. All temperature series were normalized over the 1952–2014 common period. The use of normalized deviations to develop the regional temperature record implies that each meteorological station has similar contribution in the regional mean temperature.
Unlike the extended spatial correlation pattern of temperature for Torre Glacier, the spatial pattern of precipitation is much more limited. The abundant precipitation on the western and eastern slopes of the Andes sharply contrasts with the reduced rains on the Patagonian plateau. Thus, the local precipitations measured at Los Huemules station, also nearby to El Chaltén town (from 2006 to the present), have a much more spatially reduced representability pattern than temperature (Fig. 15.S2). Because there are no long (>20 years) and homogeneous precipitation records in the forest-steppe ecotone in Santa Cruz province, the monthly precipitation from Punta Arenas were compared with the N. antarctica chronologies here developed. Precipitation variations in Punta Arenas from December to August are significantly related to ERA-Interin precipitation over the study sites (Fig. 15.S3).
The correlation functions between climate and regional tree-ring records were estimated over the 1952–2014 common period (62 years). In all these cases, it is observed that N. antarctica growth responds to both temperature and precipitation levels. The previous growing season temperatures are directly associated to the tree-ring growth, while the current growing season temperatures are inversely related to the tree-ring growth.
Conversely, it is observed that N. antarctica growth is inversely related to previous precipitation levels and directly related to current growing season precipitation levels. There is even a negative trend in N. antarctica growth due to rain during the winter (July). Besides, the R responses, compared to the C responses, are most closely associated to the regional pattern (Fig. 15.6).
Correlation functions between N. antarctica tree growth and regional climate. The red bars represent the relationship with temperature, and the blue bars represent the relationship with precipitation. (a) Relict forests. (b) Continuous forests. (c) Forests in the entire study area (regional). Dotted lines indicate 95% confidence levels
Regional temperatures during the growing season (December to March) are significantly inverse related to tree growth (r = −0.58, n = 62, p < 0.001). Above-average temperatures increase evapotranspiration, reducing the water available for tree growth. In contrast, the accumulated precipitation during November and December was positively correlated with N. antarctica growth (r = 0.34, n = 62, p < 0.01). Thus, abundant rainfalls at the beginning of the growing season favor tree-ring growth. Since the 1950s to the present, a negative trend has been recorded in the N. antarctica radial growth. These pattern is consistent with a regional increase in the mean annual temperature and, consequently, with a regional water deficit (Fig. 15.7).
Comparison between variations in N. antarctica regional tree growth and climate during the 1952–2013 period. (a) Variations in summer temperature (December to March; red line) in southern Patagonia showing the abrupt increase close to 0.5 °C around 1976/77. The temperature means for the periods before and after the 1976/77 climatic jump are indicated. (b) Regional temperature deviations from December to March (red line) and N. antarctica tree-ring width variations (grey line). (c) Punta Arenas accumulated precipitation for November and December (blue line) and N. antarctica tree-ring width variations (grey line). To facilitate visual comparison, the temperature axis has been inverted in (b). The correlation coefficients between climate and tree-ring variations are shown for the 1952–2013 period in (b) and (c), and the periods before and after the 1976/77 temperature jump in (b) and (c), respectively
3.3 Cultural Land Use Changes
The local historical documents for each site showed substantial changes in land use over time (Table 15.3). These land use changes were used to interpret the differences in radial growth variations between the pairs of R and C chronologies at each site (2–5).
At each sampling site, we registered several periods with opposite trends in radial growth between the R and C chronologies (Fig. 15.8). In general, the largest differences between the growth patterns from R and C occurred in periods concurrent with the settlement of the estancias, i.e. large rural establishments for the extensive breeding of cattle or sheep. This extensive settlement process was associated with the intensification of the use of forests. For example, in Estancia Cancha Carrera (site 3), we observed very low correlation values between R and C chronologies from the first two decades of the twentieth century. These low correlations were concurrent with the settlement of Cancha Carrera affecting the C forests. Around 1945 similar breeding practices were applied in both stands (R and C), and after that period, correlation values increased sharply (Fig. 15.8b). Conversely, records were similar since the establishment of governmental protected areas, or during periods when the intensity of land use was similar in R and C. For example, towards 1980 in Punta Gruesa Natural Reserve (site 4) the anthropic activity decreased. Since then, there has been a drastic increase in the correlation between the R and C chronologies, which continues after the creation of the Punta Gruesa Natural Reserve in 2002 (Fig. 15.8c).
Comparisons between R and C tree-ring width chronologies obtained at each site: (a) Río Guanaco – Los Glaciares National Park, (b) Estancia Cancha Carrera, (c) Punta Gruesa Provincial Natural Reserve and (d) Estancia Rincón de los Morros. Growth patterns (relict forests; black lines, continuous forests; grey lines) and the history of land uses (historical events mentioned in Table 15.3; grey dotted lines on the X-axis). Variations in the correlation coefficient between chronologies evaluated in 20 years lagged between them by 1 year (red lines). Correlation patterns (red arrows). Changes in growth patterns (grey arrows). Dotted red lines indicate 99% confidence levels
4 Discussion
In this study, we developed the first dendrochronological records for N. antarctica in Argentina. The new chronologies contribute to the expansion of the dendrochronological network and provide new knowledge of ecological processes and climatic variability across southern Patagonia (Villalba et al. 2009). The maximum reported age of 259 years for N. antarctica in Tierra del Fuego (Ivancich 2013) is a bit longer than those found in our study for Santa Cruz (206 years). This suggests that the maximum ages reached by N. antarctica in the southern extreme of South America would be around 200–300 years old. Furthermore, the intercorrelation between the series that integrate the regional chronology is similar to those reported for N. pumilio in the same region (Aravena et al. 2002b; Villalba et al. 2003; Suarez 2010).
The collection of samples for providing well-replicated dendrochronological records of N. antarctica is problematic. The difficulties are in part due to the stem architecture, which commonly has a sympodial growth, with trunks branched from the base. Minor fires used for opening the forest canopy and browsing at early stages of growth cause morphological changes in N. antarctica trees, including branching from the base (Veblen and Lorenz 1987; Aravena et al. 2002a). In addition, browsing affects tree-growth rates by reducing the tree photosynthetic biomass (Veblen et al. 1992; Relva and Veblen 1998; Martínez-Pastur et al. 1999; Pulido et al. 2000; Srur et al. 2013). In addition, these forests usually present deficient phytosanitary conditions (Fig. 15.9). N. antarctica trees frequently have hollow trunk centers and abundant rotting wood. There are reports about several species of fungi that cause chestnut and white rot in the xylem of several Nothofagus species (Rajchenberg et al. 2014). One of the most conspicuous fungus genus is Cyttaria, which develops obligatory parasitism with Nothofagus. Cyttaria hyphae alter the morphology of N. antarctica’s vascular system and generate extrusions that totally or partially surround the stem (Ipinza et al. 1989). Another very frequent interaction occurs between Nothofagus trees and Misodendrum species. Although Misodendron presence is not always a direct indication of tree decline (Rodríguez-Catón and Villalba 2018), these hemiparasitic plants may affect the Nothofagus radial-growth rates (Velásquez et al. 2012).
The well-defined tree rings in N. antarctica make this species suitable for developing good chronological records. However, the sampling effort was high at most sites with only 41% of the collected radii cross-dated and used for the chronology development. In addition to the poor health, the endogenous dynamics of the N. antarctica related to site and competition factors rather than climate reduce the interannual common signal in tree growth (Wigley et al. 1984). Some local factors, such as anthropogenic fires, gap canopy dynamics, local defoliation episodes, and differences in the browsing intensity between nearby forest sectors may contribute to the lack of a common signal between trees. In spite of the mentioned limitations, the radial growth of N. antarctica is sensitive to regional climatic variability. This characteristic offers the opportunity to use the N. antarctica ring-width variations to estimate past changes in environmental fluctuations. The N. antarctica radial growth is positively correlated with spring precipitation. Radial growth is favored by abundant precipitations at the beginning of the growing season (November and December). On the other hand, tree-ring widths are narrower when the temperature during the growing season (December–March) is above average (Fig. 15.6). Our results are consistent with those reported by Lara et al. (2005), Suarez (2010) and Jara (2011) for other Nothofagus species in dry Patagonian environments.
The stronger relationship between tree growth and temperature (r = −0.58) in comparison with precipitation (r = 0.34) is related to the lower spatial representativeness of the Punta Arenas precipitation record in relation to the regional temperature pattern (Figs. 15.S1 and 15.S3). It is known that precipitation is more spatially and temporally variable than temperature (Aravena and Luckman 2009). Therefore, we can assume that precipitation records in the proximity of the sampling sites would provide stronger relationships between rainfall and N. antarctica growth.
The regional chronology showed a progressive decrease in the radial growth of N. antarctica from the 1950s to present, which is consistent with the increase in summer temperatures since 1977 and the negative trend in precipitation starting in the 1990 across the region (Fig. 15.7). The increase in water deficit, exacerbated by higher summer temperatures and scarce precipitation, reduces tree growth. Similar negative trends in radial growth have been documented in several studies for other Nothofagus species in the Patagonian Andes (Villalba et al. 2003; Jara 2011; Villalba et al. 2012; Soto-Rogel and Aravena 2017). Interestingly, the relationships between tree growth and temperature were much stronger prior to the documented climatic jump of 1976/77 (Villalba et al. 2003). As a consequence of the shift from the negative to positive phase of the Pacific Decadal Oscillation in 1976/77, an abrupt increase in temperature was recorded all along the coast of South America, including Patagonia (Villalba et al. 2003; Vuille et al. 2015). Based on the regional temperature variations, this 1976/77 jump was associated with an approximate 0.5 °C increase in summer temperatures (Dec-Mar) throughout our study area (Fig. 15.7). Comparatively to the long-term 1952–2013 relationship between tree growth and climate (r = −0.58), it was much stronger during the colder interval from 1952 to 1976 (r = −0.72). In contrast, the relationship between tree growth and precipitation becomes stronger during the more recent, relatively warmer interval starting in 1977 (r = 0.34 and r = 0.45 for the long-term 1952–2013 and 1977–2013 intervals, respectively). These results suggest changes in the intensity of the relationships between climate and N. antarctica tree growth modulated by recent climate changes.
The comparative analysis between the R and C chronologies reveals periods of poor relationships between the chronologies, consistent with different forest stand histories (Fig. 15.8; Table 15.3). The most remarkable differences in the N. antarctica growth patterns are associated with the local history of cultural land use. For Perito Moreno National Park (site 1), it was not feasible to cross-date the samples. This reflects the impacts of persistent anthropogenic changes since the beginning of the twentieth century when the first cattle ranches were established in the region. At that time, the economy of Estancia El Rincón was based on livestock production and firewood sale. For the intensive extraction of wood from the N. antarctica forest, a network of trails was built in the area. During our surveys, abundant tree stumps were observed in the steppe matrix located between R and C, which evidences the recent anthropic fragmentation of these forests. In 1937, the area was declared a Natural Reserve and in 1945 this area became part of the Perito Moreno National Park. However, it was not until 1975 that the cattle were effectively removed from the area, and in 1982 firewood extraction was finally interrupted.
At the Sectional Río Guanaco in Los Glaciares National Park (site 2; Fig. 15.8a), we observed an abrupt increase in the correlation between the R and C chronologies which started in the 1920s. Around 1926 the main facilities for the cattle ranch La Irene were built, and livestock and forest practices were unified in R and C forests. Although Los Glaciares National Park was created in 1937, cattle were not removed from the Rio Guanaco’s area. In contrast, in the 1960s the owners increased six times the stocking. This reduced N. antarctica growth, affecting R to a greater extent than C forest. In 2010, the cattle were definitively removed, which is consistent with a period of stabilization in tree growth.
In Estancia Cancha Carrera (site 3; Fig. 15.8b), the cattle ranch was established at the end of the nineteenth century. The livestock and forestry activities focused on C, and R remained without human intervention until 1945. Coinciding with this first period, higher growth rates occurred in R than in C. Results showed a significant increase in the correlation after 1945, probably due to the establishment of four paddocks with different use intensities. This subdivision caused an increase in the livestock numbers in C and the first introduction of cattle in R. In 2000, a little ranch was installed in R at the time that the National Route 40 was been paved. All these changes provoked and intensified the use of firewood for heating. The proximity of the forest to the new route facilitates the extraction of firewood from R. These events, coupled with the regional increase in temperature, induced the extremely low growth in R observed during the last decade.
The first records of permanent use in Punta Gruesa Natural Reserve (site 4; Fig. 15.8c) date back to the end of the nineteenth century. At that time, the Sociedad Explotadora de Tierra del Fuego, the most important cattle company in the history of Chilean-Argentinean Patagonia, started working in the area. Historically, C has been more intensively intervened than the R. While R is isolated in a sector without any access trail and with a high slope (>30°), C has been intensely used for the extraction of firewood. Moreover, C was exposed to roads that were previously used to transport livestock to Chile. The relationships between chronologies were poor until the 1940s, when they became more similar to each other. It was precisely in 1940 that the exploitation of the area by Yacimientos Carboníferos Fiscales (National Coal Company) began. As a consequence, logging for trail building started as well as the construction of urban and rural infrastructure. The use of wood for house heating and steam engines also increased. In 1980, a farm breeding livestock was established in the area with a limited number of animals and remains until today. Results showed an increase in the correlation between chronologies since 1945, following the establishment of Yacimientos Carboníferos Fiscales, and the persistent decrease in radial growth, reaching a minimum growth at C in 2003. In 2002, the Punta Gruesa Natural Reserve was created and logging prohibited in the area. In recent years, the growth rate tends to stabilize.
Finally, the Estancia Rincón de Los Morros (site 5; Fig. 15.8d) represents an area with a permanent history of intense anthropogenic uses and changes. Since the arrival of the Europeans, the economy was based on extensive livestock production and the extraction of wood by selective logging. As a result, the trees present in the area are young (<69 years), so that the chronologies obtained were short and strongly correlated. This led to the assumption that the intensive anthropogenic uses in Estancia Rincón de Los Morros have a lasting impact on the N. antarctica longevity and growth pattern.
The presence of livestock in the forest causes persistent browsing, soil erosion in overgrazed sites and damages in the root systems, leading to a decrease in primary forest productivity (Teague and Smit 1992; Quinteros 2018). Browsing also generates changes in the composition, structure and functional diversity of the understory plant community. In particular, it induces the occurrence of a shrub stratum and the increase of browsing resistant invasive species (Relva et al. 2009; Arias-Sepúlveda and Chillo 2017). Other authors have reported dominance of herbs and annual grasses in overgrazed sites (Gowda et al. 2012; Peri et al. 2016). As a consequence of the understory plant community changes, the decomposition rate (Vázquez 2002; Arias-Sepúlveda and Chillo 2017) the nutrient cycling (Relva and Núñez 2014), and the net primary productivity can be altered (Lencinas et al. 2011). In addition, the livestock production includes practices to increase the forage availability by opening the forest canopy with small fires, thinning and dead tree removal (Somlo et al. 1997; Peri et al. 2016; Amoroso et al. 2018). As a result of the increase in livestock density at N. antarctica stands, the intensification of human practices in neighboring forests is inevitable. These induces in turn, important changes in the growth patterns not related to climatic variability.
In conclusion, our work reports on the first tree-ring chronologies from N. antarctica developed for the Argentinean sector in southern Patagonia. Given the common tortuous form of N. antarctica trees in the forest-steppe ecotone and the poor health conditions of these forests, the development of tree-ring chronologies requires a sampling strategy including a large number of trees. Only 41% of the collected samples were cross-dated and included in the chronologies. At the forest-steppe ecotone, the interannual variations in N. antarctica radial growth are particularly sensitive to water availability during the growing season. The N. antarctica growth increased with the increase in the water supply. However, these relationships between climate and tree growth seem to be unstable and vary according to the prevailing climatic conditions (cold versus warm periods). In the recent warmer decades following the 1976/77 climatic shift in the Pacific Decadal Oscillation (PDO) (Mantua and Hare 2002), the radial growth seems to be more closely related to spring rainfall variations. Finally, the interactions between land use changes and climate variability considered in this study provide a valid approach to interpret past variations in N. antarctica tree-ring in the forest-steppe ecotone in the Patagonian Andes foothills.
References
Alberdi M (1995) Ecofisiología de especies leñosas de los bosques hidrófilos templados de chile: resistencia a la sequía y bajas temperaturas. En: Armesto J, Villagrán C, Arroyo M (eds) Ecología de los bosques nativos de Chile. Universidad de Chile, Santiago, p 420
Amoroso M, Chillo V, Alcala V et al (2018) Efecto del manejo silvopastoril sobre la estructura y dinámica poblacional de bosques mixtos de ciprés de la cordillera (Austrocedrus chilensis) y coihue (Nothofagus dombeyi). Revista Ecosistemas 27(3):33–40. https://doi.org/10.7818/ECOS.1502
Aravena JC, Luckman BH (2009) Spatio-temporal rainfall patterns in southern South America. Int J Climato 129(14):2106–2120. https://doi.org/10.1002/joc.1761
Aravena JC, Carmona M, Pérez C et al (2002a) Cambios en la riqueza de especies arbóreas, estructura de rodales y propiedades del suelo en una cronosecuencia sucesional en el norte de la Isla de Chiloé, Chile. Revista Chilena de Historia Natural 75(2):339–360. https://doi.org/10.4067/s0716-078x2002000200007
Aravena JC, Lara A, Wolodarsky-Franke A et al (2002b) Tree-ring growth patterns and temperature reconstruction from Nothofagus pumilio (Fagaceae) forests at the upper tree line of southern Chilean Patagonia. Rev Chil Hist Nat 75(2):361–376. https://doi.org/10.4067/S0716-078X2002000200008
Arias-Sepúlveda JEA, Chillo V (2017) Cambios en la diversidad funcional del sotobosque y la tasa de descomposición frente a diferentes intensidades de uso silvopastoril en el noroeste de la Patagonia, Argentina. Ecol Austral 27(1):029–038
Bandieri S (2005) Asuntos de familia. La construcción del poder en la Patagonia: el caso de Neuquén. Boletín del Instituto de Historia Argentina y Americana Dr. Emilio Ravignani 28:65–94
Barbería EM (1994) El extremo austral sudamericano.: Ocupación y relaciones de los territorios argentinos y chilenos, 1880–1920. Estud Front 33:185–212
Barbería EM (1995) Los dueños de la tierra en la Patagonia Austral: 1880–1920. Universidad federal de la Patagonia Austral, Santa Cruz
Blasing TJ, Solomon AM, Duvick DN (1984) Response functions revisited. Tree-Ring Bull 44:1–15
Boninsegna JA, Argollo J, Aravena JC et al (2009) Dendroclimatological reconstructions in South America: a review. Palaeogr Palaeocl 281(3–4):210–228. https://doi.org/10.1016/j.palaeo.2009.07.020
Christensen M, Emborg J (1996) Biodiversity in natural versus managed forest in Denmark. Forest Ecol Manag 85(1–3):47–51. https://doi.org/10.1016/s0378-1127(96)03749-8
Cook ER (1985) A time series analysis approach to tree-ring standardization. Ph. D. dissertation, University of Arizona, Arizona, p 183
Coronato FR, Del Valle HF (1993) Methodological comparison in the estimate of fluvial erosion in an arid closed basin of northeastern Patagonia. J Arid Environ 24(3):231–239. https://doi.org/10.1006/jare.1993.1021
Di Castri F, Hansen A, Holland M (1988). A new look at ecotones: emerging international projects on landscape boundaries. Biology Internat. Special issue 17, Internet. Union of Biol Sci, pp 1–163
Díaz BG (2005) Uso múltiple de recursos naturales renovables en la Cuenca Río de las Vueltas, provincia de Santa Cruz. (Tesis de Mg. Sci. Manejo de Cuencas Hidrográficas). Universidad Nacional de La Plata. Argentina, p 195
Donoso C (1993) Bosques templados de Chile y Argentina. Variación, estructura y dinámica. Ecología Forestal. Editorial Universitaria, Santiago
Donoso C, Steinke L, Premoli A (2006) Nothofagus antarctica (G. Forster) Oerst. ñirre, ñire, ñiré, anís (Tierra Del Fuego) ñirre: de ngërü (Mapudungun): zorro. En: Donoso C (Ed.), Las especies arbóreas de los bosques templados de Chile y Argentina. Autoecología. Ed. Cuneo, M. Santiago, Chile
Fritts HC (1976) Tree rings and climate. Academic Press, London
Garreaud R, Lopez P, Minvielle M et al (2013) Large-scale control on the Patagonian climate. J Clim 26(1):215–230. https://doi.org/10.1175/JCLI-D-12-00001.1
González MH, Vera CS (2010) On the interannual wintertime rainfall variability in the southern Andes. Int J Climatol 30(5):643–657. https://doi.org/10.1002/joc.1910
Gowda JH, Kitzberger T, Premoli AC (2012) Landscape responses to a century of land use along the northern Patagonian forest-steppe transition. Plant Ecol 213(2):259–272. https://doi.org/10.1007/s11258-011-9972-5
Hampe A, Petit RJ (2005) Conserving biodiversity under climate change: the rear edge matters. Ecol Lett 8(5):461–467. https://doi.org/10.1111/j.1461-0248.2005.00739.x
Hijmans RJ, Cameron SE, Parra JL et al (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25:1965–1978. https://doi.org/10.1002/joc.1276
Holmes RL (1983) Computer-assisted quality control. Tree-Ring Bull 43:69–78
Ipinza R, Pérez F, Kappes A (1989) Cyttaria espinosae Lloyd., un hongo de interés en fitopatología, alimentación y evolución de los bosques de Nothofagus en Chile. Bol Soc Micol Madrid 13:31–48
Ivancich HS (2013) Relaciones entre la estructura forestal y el crecimiento del bosque de Nothofagus antarctica en gradientes de edad y calidad de sitio. Tesis doctoral, Facultad de Ciencias Agrarias y Forestales, Universidad Nacional de La Plata, Argentina
Iverson LR, Schwartz MW, Prasad AM (2004) How fast and far might tree species migrate in the eastern United States due to climate change? Glob Ecol Biogeogr 13(3):209–219. https://doi.org/10.1111/j.1466-822X.2004.00093.x
Jara JCL (2011) Relaciones entre el crecimiento de Nothofagus betuloides y el clima local y de gran escala en bosques subantárticos de la Isla Navarino. (Tesis de grado). Universidad Austral de Chile, Valdivia
Kitzberger T (2012) Ecotones as complex arenas of disturbance, climate, and human impacts: the trans-Andean forest-steppe ecotone of northern Patagonia. In: Ecotones between forest and grassland. Springer, New York, pp 59–88
Kreps G, Martínez PGM, Peri PL (2012) Cambio climático en Patagonia sur. Escenarios futuros en el manejo de los recursos naturales. INTA (ed), Santa Cruz, Argentina
Lara A, Aravena JC, Villalba R et al (2001) Dendroclimatology of high-elevation Nothofagus pumilio forests at their northern distribution limit in the Central Andes of Chile. Can J For Res 31(6):925–936. https://doi.org/10.1139/x00-208
Lara A, Villalba R, Wolodarsky-Franke A et al (2005) Spatial and temporal variation in Nothofagus pumilio growth at tree line along its latitudinal range (35 40′-55 S) in the Chilean Andes. J Biogeogr 32(5):879–893. https://doi.org/10.1111/j.1365-2699.2005.01191.x
Lencinas MV, Martínez PGM, Gallo E et al (2011) Alternative silvicultural practices with variable retention to improve understory plant diversity conservation in southern Patagonian forests. For Ecol Manag 262(7):1236–1250. https://doi.org/10.1016/j.foreco.2011.06.021
Manacorda M, Bonvissuto G (2001) Uso silvopastoril de los bosques de ñire (Nothofagus antarctica) en Río Negro, Patagonia Argentina. Revista Forestal Centroamericana 35:41–44
Mantua NJ, Hare SR (2002) The Pacific decadal oscillation. J Oceanogr 58(1):35–44. https://doi.org/10.1023/A:1015820616384
Marazana O, Verzino G, Ocampo JB et al (1984) Situación actual de los nativos de la Provincia de Santa Cruz: descripción manejo y estado sanitario. Informe Técnico Consejo Agrario Provincial, Córdoba p 33
Martínez-Pastur GM, Peri PL, Fernández C et al (1999) Desarrollo de la regeneración a lo largo del ciclo del manejo forestal de un bosque de Nothofagus pumilio: incidencia del ramoneo de Lama guanicoe. Bosque 20(2):47–53
Montaña C (1982) Las comunidades de ñire (Nothofagus antarctica (Forst.) Oerst.) de la cuenca del río Manso Superior (Río Negro, Argentina). Tesis Doctoral, Universidad Nacional de Córdoba, Córdoba, Argentina
Moore DM (1983) The flora of the Fuego-Patagonian cordilleras: its origins and affinities. Rev Chil Hist Nat 56(123–136):123–136
Morales M, Villalba R (2006) Climatic and anthropogenic influences on the dynamics of Prosopis ferox forests in the Quebrada de Humahuaca, Jujuy, Argentina. In: Land use change and mountain biodiversity. CRC Press, Boca Raton, pp 275–282
Morales MS, Villalba R, Boninsegna JA (2005) Climate, land-use and Prosopis ferox recruitment in the Quebrada de Humahuaca, Jujuy, Argentina. Dendrochronologia 22(3):169–174. https://doi.org/10.1016/j.dendro.2005.05.004
Mundo IA, Villalba R, Veblen TT et al (2017) Fire history in southern Patagonia: human and climate influences on fire activity in Nothofagus pumilio forests. Ecosphere 8(9). https://doi.org/10.1002/ecs2.1932
Murcia C (1995) Edge effects in fragmented forests: implications for conservation. Trends Ecol Evol 10(2):58–62. https://doi.org/10.1016/S0169-5347(00)88977-6
Neilson RP (1991) Climatic constraints and issues of scale controlling regional biomes. In: Ecotones. Springer, Boston, pp 31–51
Paruelo MJ, Beltrán A, Jobbágy E et al (1998) The climate of Patagonia: general patterns and controls on biotic processes. Ecol Austral 8:85–101
Pérez Moreau R (1959) Reseña Botánica sobre el Lago Argentino. Instituto Nacional del Hielo Continental Patagónico, Buenos Aires
Peri PL, Ormaechea S (2013) Relevamiento de los bosques nativos de ñire (Nothofagus antarctica) en Santa Cruz: base para su conservación y manejo. Ediciones INTA, Buenos Aires
Peri PL, Hansen NE, Bahamonde HA et al (2016) Silvopastoral systems under native forest in Patagonia Argentina. In: Silvopastoral systems in southern South America. Springer, pp 117–168
Pulido FJ, Díaz B, Martínez-Pastur GM (2000) Incidencia del ramoneo del guanaco (Lama guanicoe Müller) sobre la regeneración temprana en bosques de lenga [Nothofagus pumilio (Poepp et Endl) Krasser] de Tierra del Fuego, Argentina. Forest Syst 9(2):381–394
Quinteros CP (2018) Propiedades ecosistémicas de bosques de Nothofagus pumilio afectadas por diferente intensidad de uso ganadero en Chubut, Patagonia Argentina. Revista Ecosistemas 27(3):24–32. https://doi.org/10.7818/ECOS.1483
Rajchenberg M, Barroetaveña C, Orellana I (2014) Relevamiento de hongos con fines de conocimiento ambiental, turístico y eventualmente productivo. Centro De Investigación Y Extensión Forestal Andino Patagónico (CIEFAP), Esquel, p 96
Ramírez C, Correa M, Figueroa H et al (1985) Variación del hábito y hábitat de Nothofagus antarctica en el centro de Chile. Bosque 6(2):55–73
Relva MA, Núñez MA (2014) Factores que facilitan y retrasan la invasión de coníferas exóticas en la Isla Victoria. Ecol Austral 24(2):145–153
Relva MA, Veblen TT (1998) Impacts of introduced large herbivores on Austrocedrus chilensis forests in northern Patagonia, Argentina. For Ecol Manag 108(1–2):27–40. https://doi.org/10.1016/s0378-1127(97)00313-7
Relva MA, Westerholm CL, Kitzberger T (2009) Effects of introduced ungulates on forest understory communities in northern Patagonia are modified by timing and severity of stand mortality. Plant Ecol 201(1):11–22. https://doi.org/10.1007/s11258-008-9528-5
Rodríguez-Catón M, Villalba R (2018) Indicadores del decaimiento en bosques de Nothofagus pumilio en el norte de la Patagonia, Argentina. Madera Bosques 24(2):e2421588. https://doi.org/10.21829/myb.2018.2421588
Roig FA (1999) La vegetación de la Patagonia. En: Correa M (Dir.) Flora patagónica. Colección Científica del INTA 8(1):48–166
Roig FA, Villalba R (2008) Understanding climate from Patagonian tree rings. Dev Quat Sci 11:411–435. https://doi.org/10.1016/S1571-0866(07)10021-X
Rosenblüth B, Fuenzalida HA, Aceituno P (1997) Recent temperature variations in southern South America. Int J Climatol 17(1):67–85. https://doi.org/10.1002/(SICI)1097-0088(199701)17:1<67:AID-JOC120>3.0.CO;2-G
SAyDS (2005) Primer Inventario Nacional de Bosques Nativos. Ministerio de Salud y Ambiente de la Nación – Secretaría de Ambiente y Desarrollo Sustentable, Buenos Aires
Schweitzer A (2011) Fronteras, recursos naturales y crisis en la Patagonia sur argentina. In: Integración geoestratégica, seguridad, fronteras y migración en América Latina, editado por Sandoval, J., Álvarez, R. Saavedra, L. Editorial INREDH, Ecuador, pp 33–69
Somlo R, Bonvissuto G, Schlichter T et al (1997) Silvopastoral use of Argentine Patagonian forests. In: Temperate agroforestry systems. CAB International, Wallingford; New York, pp 237–250
Soto-Rogel P, Aravena JC (2017) Potencial dendroclimático de Nothofagus betuloides en la Cordillera de Darwin, Tierra del Fuego, Chile. Bosque 38(1):155–168. https://doi.org/10.4067/S0717-92002017000100016
Srur AM, Golluscio RA, Villalba R et al (2013) Grazing-induced morphological and growth rate changes in Anarthrophyllum rigidum, a Patagonian leguminous shrub. Dendrochronologia 31(3):223–227. https://doi.org/10.1016/j.dendro.2013.02.002
Stokes MA, Smiley TL (1968) An introduction to tree-ring dating. University of Chicago Press, Chicago
Suarez ML (2010) Tree-ring records from Nothofagus dombeyi: a preliminary chronology network in Northern Patagonia, Argentina. Dendrochronologia 28(2):65–72. https://doi.org/10.1016/j.dendro.2009.11.001
Teague WR, Smit GN (1992) Relations between woody and herbaceous components and the effects of bush-clearing in southern African savannas. J Grassl Soc South Afr 9(2):60–71. https://doi.org/10.1080/02566702.1992.9648301
Vázquez DP (2002) Multiple effects of introduced mammalian herbivores in a temperate forest. Biol Invasions 4(1–2):175–191. https://doi.org/10.1023/A:1020522923905
Veblen TT, Lorenz DC (1987) Post-fire stand development of Austrocedrus-Nothofagus forests in northern Patagonia. Vegetatio 71(2):113–126. https://doi.org/10.1007/BF00044825
Veblen TT, Kitzberger T, Lara A (1992) Disturbance and forest dynamics along a transect from Andean rain forest to Patagonian shrubland. J Veg Sci 3(4):507–520. https://doi.org/10.2307/3235807
Veblen TT, Donoso C, Kitzberger T (1996) Ecology of southern Chilean and Argentinean Nothofagus forests. In: Veblen TT, Hill R, Read J (eds) The ecology and biogeography of Nothofagus forests. Yale University Press, Connecticut, pp 293–353
Velásquez C, Henríquez JM, Aravena JC (2012) Damage caused by mistletoe Misodendrum Punctulatum Banks Ex Dc. on architecture and radial growth of Nothofagus pumilio (Poepp. et Endl.) Krasser forests of southern Chile. Austral Ecol 37(7):816–824. https://doi.org/10.1111/j.1442-9993.2011.02342.x
Villalba R, Boninsegna JA, Veblen TT et al (1997) Recent trends in tree-ring records from high elevation sites in the Andes of northern Patagonia. In: Climatic change at high elevation sites. Springer, Dordrecht, pp 193–222
Villalba R, Lara A, Boninsegna JA et al (2003) Large-scale temperature changes across the southern Andes: 20th century variations in the context of the past 400 years. Clim Chang 59:177–232. https://doi.org/10.1007/978-94-015-1252-7_10
Villalba R, Grosjean M, Kiefer T (2009) Long-term multi-proxy climate reconstructions and dynamics in South America (LOTRED-SA): state of the art and perspectives. Palaeogeogr Palaeoclimatol Palaeoecol 281(3–4):175–179. https://doi.org/10.1016/j.palaeo.2009.08.007
Villalba R, Lara A, Masiokas MH et al (2012) Unusual southern hemisphere tree growth patterns induced by changes in the southern annular mode. Nat Geosci 5:793–798. https://doi.org/10.1038/ngeo1613
Vuille M, Franquist E, Garreaud R et al (2015) Impact of the global warming hiatus on Andean temperature. J Geophys Res Atmos 120(9):3745–3757. https://doi.org/10.1002/2015JD023126
Wigley TM, Briffa KR, Jones PD (1984) On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J Clim Appl Meteorol 23(2):201–213. https://doi.org/10.1175/1520-0450(1984)023<0201:OTAVOC>2.0.CO;2
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
1 Electronic Supplementary Material

Fig. 15.S1
Spatial correlation pattern between monthly surface temperature in Torre Glacier (IANIGLA-CONICET) (black dot) and gridded data from the ERA Interim re-analysis during the 2002–2016 period. The location of the meteorological stations from Esquel, Río Gallegos and Punta Arenas are indicated by grey dots. Study sites are numbered within white dots (PNG 2186 KB)

Fig. 15.S2
Spatial correlation pattern between monthly precipitation from Los Huemules station (black dot) and gridded data from the ERA Interim re-analysis during the 2006–2016 period. The location of the meteorological station from Punta Arenas is indicate by a grey dot. Study sites are numbered within white dots (PNG 2037 KB)

Fig. 15.S3
Spatial correlation pattern between seasonal (Dec-Aug) precipitation from Punta Arenas station (black dot) and gridded data from the ERA Interim re-analysis during the 1979–2017 period. The location of the study sites are numbered within white dots (PNG 651 kb)
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Vettese, E.S., Villalba, R., Ibáñez, I.A.O., Peri, P.L. (2020). Tree-Growth Variations of Nothofagus antarctica Related to Climate and Land Use Changes in Southern Patagonia, Argentina. In: Pompa-García, M., Camarero, J. (eds) Latin American Dendroecology. Springer, Cham. https://doi.org/10.1007/978-3-030-36930-9_15
Download citation
DOI: https://doi.org/10.1007/978-3-030-36930-9_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-36929-3
Online ISBN: 978-3-030-36930-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)