Abstract
Cardiac electrophysiology frequently employs fluoroscopy in both diagnostic and therapeutic procedures. Electrophysiologists need to be aware of the different degrees of exposure associated with each procedure and utilise a personal dosimeter to monitor exposure. In general, diagnostic procedures have lower radiation exposure compared to ablative procedures. Atrial fibrillation ablation has the highest radiation exposure of all electrophysiologic procedures. Radiation exposure has been shown to pose a cumulative risk of cancer and a threshold-based risk for tissue and organ injury. Radiation safety protocols must be utilised to minimise exposure to radiation during the procedure. Measures to reduce radiation exposure include equipment customisation, workflow adaptations, protective gear and effective use of non-radiation techniques. Pregnancy presents a unique challenge and monitoring is essential to prevent harmful consequences to both the mother and the foetus.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Ever since the first ablation procedure was performed by Dr. Scheinman in 1981 the field of interventional electrophysiology has made significant progress [1]. Electrophysiology (EP) studies and radiofrequency catheter ablation (RFCA) procedures are widely performed worldwide. Radiofrequency ablation has been established as a first-line treatment for supraventricular arrhythmias and one of the accepted treatment modalities for more complex atrial and ventricular arrhythmias [2,3,4]. Traditionally, EP procedures are performed in cardiac catheter laboratories and fluoroscopy is used as the key imaging modality to guide positioning cardiac catheters. As a consequence, electrophysiologists are exposed to considerable ionising radiation as scatter emitted by their patients (Fig. 2.1) [5]. In fact electrophysiologists seem to have even higher amount of radiation exposure compared to other medical personnel including interventional radiologists [5,6,7]. Several reports about possible radiation-related serious complications amongst cardiologists who were previously exposed to ionising radiation have heightened the awareness about the need for better understanding and optimal utilisation of available technologies in cardiac catheter labs [8,9,10]. Females comprise 11% of electrophysiology trainees as well as a significant proportion of electrophysiology lab personnel [11]. Misconceptions regarding the risk of occupational radiation exposure on pregnancy may lead to changes in family and career planning. Proper monitoring is essential to prevent harm to both the foetus and the mother.
Quantification of Ionising Radiation
Radiation exposure can be quantified using different measures. Commonly used terminology includes absorbed dose, cumulative air kerma, dose area product (DAP) also referred to as kerma area product (KAP) , equivalent dose, personal dose equivalent and effective dose. These terms are defined by the International Commission on Radiological Protection (ICRP) [12].
Absorbed dose is the energy absorbed per unit mass of the absorbing tissue. It is expressed in grey units. One grey (Gy) equates to one joule of energy per kilogram of absorbing tissue.
Cumulative air kerma is a quantitative measure of X-ray energy delivered to air at the interventional reference point (15 cm from the isocentre in the direction of the focal spot).
DAP is a product of the radiation dose to air and cross-sectional X-ray beam area at any given distance from the X-ray source. It is independent of the distance from the X-ray source. DAP is expressed as Gy cm2.
Equivalent dose is a measure of biological effect of ionising radiation. To allow comparisons of the risk of harm due to different ionising radiations, each type of radiation is assigned a radiation weighting factor (W R) as a measure of its biological effect. Equivalent dose is the product of absorbed dose and radiation weighting factor. Equivalent dose is measured in sieverts (Sv) and denotes potency of radiation (for example one grey of gamma rays is equivalent to 1 Sv whilst one grey of alpha rays is equivalent to 20 Sv).
Effective dose (ED) takes into consideration the potency as well as the probability of a tissue being affected per unit dose of radiation (e.g. the same amount of equivalent dose would equate to 20 times more equivalent dose to ovaries compared to skin). Effective dose is a tissue-weighted sum of all equivalent dose in different tissues of human body. Radio sensitivity in human tissues is highest in highly mitotic or undifferentiated tissues such as bone marrow, lens, basal epidermis and gonads. It reduces with age and as a consequence young children exposed to radiation are much more likely to be affected than the older ones [13, 14]. Effective doses are related to the whole-body risk associated with any given deposition of ionising radiation in an individual.
Personal dose equivalent is measured by the personal occupational dosimeters and is expressed in millisieverts. It gives an estimate of the organ doses.
Hazards from Exposure to Ionising Radiation
The harmful effects of ionising radiation are well known and can be classified as stochastic and deterministic effects.
Stochastic effects are not related to a particular threshold level of radiation exposure; however the risk of their occurrence increases with a lifetime cumulative increase in radiation exposure and severity independent of the dose. Typical example of a stochastic effect of ionising radiation is the risk of developing malignancy and genetic effect being the other. Stochastic risk from exposure to ionising radiation can be expressed using the concept of “effective dose”, a term first used by Wolfgang Jacobi in 1975 [15].
The excessive relative risk of cancer mortality according to International Commission of Radiological Protection (ICRP) is 5% per Sv and 0.2% per Sv for heritable effects.
Dose area product has been shown to correlate well with the total ionising energy imparted to a patient and total scatter radiation and therefore to the risk of stochastic effects such as malignancy both to patient and medical workers [16,17,18,19,20,21,22].
Deterministic effects only occur once a threshold of exposure has been exceeded. The severity of deterministic effects increases as the dose of exposure increases. Deterministic effects are caused by significant cell damage or death. The physical effects will occur when the cell death burden is large enough to cause obvious functional impairment of a tissue or an organ. Radiological measurement that has been used to estimate deterministic effects is cumulative air kerma (Gy). Some of the recognised deterministic effects of radiation include erythema, necrosis and epilation of skin, cataract, reduced fertility and acute radiation syndrome. The skin and eyes are most vulnerable to X-rays with a suggested threshold radiation dose (for deterministic effects) of 2 Gy and 500 μGy, respectively [23,24,25,26].
Monitoring Radiation Exposure in Electrophysiology Lab
The ICRP recommends the use of two personal dosimeters for staff exposed to radiation: one to be worn on the trunk of the body inside lead apron at about waist level and the other worn outside lead apron at collar or left shoulder. Inside dosimeter estimates radiation to internal organs including reproductive organs. However it may be an overestimate as it does not account for further attenuation of radiation through superficial tissues [11]. The outer dosimeter estimates radiation exposure to thyroid, lens, head and neck.
Female medical personnel working in cardiac labs should be encouraged to declare pregnancy as early as possible as it would help monitor ED to foetus. Additional dosimeter at the level of abdomen could be used to assess foetal exposure [27].
Compliance with dosimeter use is essential and should be made mandatory. Passive dosimeters are usually read monthly. Records of exposure are kept for as long as required by national legislation/guidelines, and may depend on the type of radiation worker. Please note that it is not generally possible to establish whether an individual’s cancer has been caused by radiation exposure. The causal relationship between radiation exposure and cancer incidence is only detectable through statistical analysis of large population samples. In some very rare circumstances it may be possible to conclude (on balance of probabilities) that an individual’s cancer is linked to their occupational exposure. This might be possible when the cancer is rare in the general population but observed in disproportionate numbers in an occupational group having very specific exposures to radiation, and the individual in question has had the specific exposure.
Historical Data About Radiation Exposure in Electrophysiology Procedures
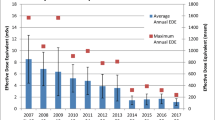
Most of the studies looking at radiation exposure in electrophysiology labs are patient centric with main focus on the radiation exposure to the patient [28,29,30]. Very few studies were aimed at physician radiation exposure [7, 31, 32]. Higher radiation exposure both to the patient and physician was noted with complex ablation procedures [33, 34]. It was also observed that there was operator variability; however average operator dose was quantitatively related to average patient dose. It was also noted that there was reduction in radiation exposure over time with development of newer non-fluoroscopic three-dimensional navigation and electroanatomical mapping systems [35].
Electrophysiologists have been shown to have high radiation exposure with a median of 4.3 mSv per year (3.5–6.1) whilst interventional cardiologists averaged 3.3 mSv (2.0–19.6 mSv) and average background radiation received by a European resident is around 2.1 mSv per year [7, 36]. Casella and colleagues examined the trend in radiation exposure amongst the various electrophysiological procedures in a retrospective study of 8150 patients over a 7-year period. There was a significant decrease in radiation exposure in all procedures except cardiac resynchronisation therapy . This demonstrates the remarkable improvement in procedural influences, protective equipment, as well as operator experience and compliance with protection protocols. Radiation exposures of the various electrophysiological procedures in the year 2010 and 2016 are summarised in Table 2.1. The fact that vast improvement was seen in ablation procedures and specially complex ablations, namely atrial fibrillation and ventricular tachycardia ablations, is testament to the increasing use of non-fluoroscopic techniques and their ability to reduce radiation exposure [37].
Measures to Reduce Radiation Exposure in Electrophysiology Laboratory
There are three principles that govern the use of radiation in the electrophysiology lab: responsibility, justification and optimisation . It is the physician’s responsibility to limit the exposure to radiation by utilising the knowledge of procedural, operator and patient influences on radiation exposure. The use and dose of radiation must be justified in view of the patient’s clinical context. The benefits of the procedure and the need for fluoroscopy must be carefully weighed against the risk of exposure to radiation. In addition, all factors must be optimised to ensure the minimum exposure to radiation without compromising the safety and efficacy of the procedure [38].
Various measures to reduce radiation exposure during electrophysiology procedures can be grouped under the following categories which include
-
1.
Awareness and education resulting in a change in culture
-
2.
Use of protective gear in cath labs (including thyroid shields, lead glasses, etc.)
-
3.
Effective use of radiological views
-
4.
Effective use of available fluoroscopic technology (such as collimation and lower pulse rates)
-
5.
Effective adaptation and implementation of local and national policies
-
6.
Effective use of mapping systems and newer technologies in electrophysiology labs
-
7.
Continued research to facilitate eventual zero-radiation procedures
Awareness and Education Resulting in a Change in Culture
Unlike other professionals such as professional pilots involved with radiation exposure electrophysiologists seem to be less educated about radiation hazards and ways to minimise them [35]. Electrophysiology trainees need to be educated about awareness and need to use radiation as low as reasonably achievable (ALARA principle) [39, 40], a concept that has been proposed by the International Commission for Radiation Protection to guide responsible radiation use.
Education about radiation exposure hazards and ways to reduce it should start very early in electrophysiology training and should be made mandatory. Electrophysiologists should be well equipped to utilise alternative non-fluoroscopic imaging modalities at every possible stage during the procedure. Proper documentation of radiation doses and compliance with dosimeters should be ensured by the local authorities. It has been shown that use of fluoroscopy is not strictly a function of experience and that the awareness of the harm correlated with X-rays and the presence of a background in radiation safety are the cornerstones for reducing radiation exposure in interventional cardiology [41, 42].
Use of Protective Gear in Cath Labs (Fig. 2.2)
Lead aprons (minimum of 0.5 mm lead equivalence in front and 0.25 mm at the rear) have been shown to reduce radiation dose by over 95% for the operator [43, 44]. In addition, wearing a thyroid shield and lead glasses or the use of a radiation protection cabin and avoiding direct hand exposure help reduce radiation doses significantly. Professional cataract is a well-known problem in invasive cardiology workers and is distinguished from naturally occurring cataract forms as it occurs in the posterior pole of the lens [45, 46]. Use of protective eyewear (leaded glasses with side panels) is highly recommended [47, 48]. Under-table lead shielding and over-table glass shielding help reduce scatter radiation to the operator and hence should be used routinely during EP procedures.
Effective Use of Radiological Views
For a given amount of beam energy the resultant image quality is inversely proportional to the amount of tissue and distance it has to traverse. In lateral (or craniocaudal) angulations, X-rays cross more tissues, which increases attenuation and decreases image quality [49]. To compensate, the system increases the beam energy to maintain image quality. As a result there is more radiation to the patient as well as operator. The operator receives most of the radiation as a result of scatter from the patient. The operator is likely to receive higher radiation dose when standing on the same side of the patient as the tube (as a result of scatter from the patient) [50] and therefore when operating from right side of the patient the operator receives more radiation in a left lateral view compared to the right lateral view. Traditionally when ablation is carried out most operators view catheter position in a left anterior oblique view. Care should be taken that lead shied is protecting the operator during these lateral angulated views. Overall one should be mindful of the fact that higher radiation is being used during the use of angulated views [51]. Also operators have to be cautious using angulated views in patients with high body mass index as it would result in much higher doses of radiation [52].
Effective Use of Available Fluoroscopic Technology
Electrophysiologists can reduce radiation to the patient and scatter radiation for themselves in turn by effective use of available fluoroscopic technology. Effective use of various strategies available with modern X-ray units can considerably reduce radiation dose without significant change in fluoroscopy time. Use of lower pulse frequency and shorter pulse durations has been shown to reduce radiation dose [36, 53, 54]. This can be achieved by using lower frame rates such as 3–4 fps. Removal of the secondary radiation grid and programming an ultra-low pulsed fluoroscopy rate during ablation procedures were associated with significant reductions in radiation exposure [36]. Confining the X-ray beam to small area of interest can be achieved by using collimation. Collimation reduces DAP as it reduces cross-sectional area at any given distance from the source. During interventional electrophysiology procedure collimation has been shown to significantly reduce radiation exposure without compromising on the image quality. These benefits have been more marked in simple ablation procedures where smaller areas are of interest during ablation [55]. Asymmetric collimation can achieve another 60–80% reduction compared with normal collimation [56]. Whilst fluoroscopy is used for catheter placement, cine is used to acquire and store images. Cine acquisitions account for majority of radiation exposure despite representing a smaller fraction of X-ray tube operation time [57, 58]. Cine acquisitions can be minimised or even avoided by using the stored fluoroscopy feature (saving the last fluoroscopic loop) [59].
The X-ray tube output is proportional to the distance between the tube and the detector. The detector should therefore be lowered onto the patient throughout the procedure (Fig. 2.3). A 15% reduction in radiation dose was noted with a combination of elevation of table and lowering detector by 10 cm [60]. Frame rate settings are programmable between 25–30 and 6 fps. Still lower frame rates may be achieved by using triggered fluoroscopy. Lower frame rates are linearly related to lower radiation doses.
Diagrammatic representation of possible ways to reduce scatter radiation in an EP lab. Scattered radiation can be reduced by reducing the X-ray field size, the path length of the X-ray beam through the patient (i.e. projection) and the exposure parameters (e.g. pulse rate). The patient-detector distance is minimised and the focus-patient distance is maximised (within the constraints of available focus-to-detector distances) in the first instance to reduce magnification (image quality) and patient dose. The lower patient dose arises from lower exposure parameters, which in turn reduces scatter. In the case of interventional X-ray units with an isocentre, the focus-patient distance is essentially determined by the unit’s geometry; the operator however is able to control the position of the detector
Effective Adaptation and Implementation of Local and National Policies
European Commission has recognised the need to educate and train medical personnel involved in procedures using ionising radiation. The current Medical Exposure Directive (MED) outlines the justification and optimisation of radiological procedures, distribution of responsibilities, training of medical staff, procedural aspects and equipment use. Under the EURATOM Treaty the European Commission is responsible for the protection of patients and other individuals in medical facilities [61]. It does this by consistently updating the Basic Safety Standards Directive which regulates, amongst other items, the safe use of ionising radiation in medical applications.
Directive 2013/59/Euratom—protection against ionising radiation—states that the member states system of radiation protection shall be based on the principles of justification (benefits outweigh risks) and optimisation (exposure to as few, and as low as possible: dose constraints) and
Dose limitations (on the individual’s level) are as follows:
-
For effective dose limit: 20 mSv in any single year
-
In special circumstances: up to 50 mSv in a single year (the average annual dose over any 5 consecutive years must not exceed 20 mSv)
-
Eyes: 20 mSv in a year or 100 mSv in any 5 consecutive years
-
Skin and extremities: 500 mSv per year
The onus of medical staff’s awareness and protection from radiation exposure lies with the employer. Accordingly, the major cardiovascular associations have published detailed recommendations about X-ray use and suggested practical ways to reduce it [62].
Effective Use of Mapping Systems and Newer Technologies in Electrophysiology Labs
Electroanatomic mapping systems (EMS) have been developed for facilitating catheter localisation, three-dimensional cardiac chamber description, obtaining three-dimensional electrical activation sequences and voltage map within a cardiac chamber (Fig. 2.4). The two EMS that are mostly used include the NavX system (St. Jude/Abbott) which uses low-amplitude high-frequency current fields [63] and CARTO (Biosense Webster) which uses magnetic fields and impedance measurements for catheter localisation [64]. These are also called “non-fluoroscopic” mapping systems as they do not utilise fluoroscopy. Whilst EMS greatly facilitates identification of appropriate ablation sites during complex ablation procedures, operators need greater understanding of underlying principles and appropriate experience in their use. Use of EMS has been shown to reduce radiation dose during ablation procedures [53, 65, 66]. EMS can be further enhanced by integration of 3D imaging obtained using cardiac MRI or CT prior to procedure [67].
Remote navigation systems have been developed on the premise that it would provide better precision movement of ablation catheter and improve outcomes by enabling more effective lesions. The only system currently in use allows remote navigation by moving special catheter by a static magnetic field [68] (Niobe system from Stereotaxis). Whilst remote navigation does not reduce the radiation dose for the patient, it can facilitate near-zero radiation for the operator. However, as the image integration of the system allowed for better orientation of the operator, significant reduction of the patient’s radiation exposure has been demonstrated for a number of different arrhythmias [67,68,70].
Continued Innovation to Facilitate Eventual Zero-Radiation Procedures
Whilst near-zero-radiation ablation procedures have been shown to be feasible amongst paediatric population they are still a work in progress amongst adult cohorts. In our experience it has been feasible to perform complex ablation procedures (including transseptal punctures) using no fluoroscopy with the aid of EMS and alternate imaging modalities such as transoesophageal echocardiography. The key step in transseptal punctures is to visualise the transseptal needle, which can be “faked” on the EMS system [71]. By using a radiofrequency needle (NRG, Baylis Medical) with an isolated tip that serves as small electrode, the exact location of the needle tip on the 3D reconstruction of the right atrium can be achieved.
Radiation Effects on Fertility and Pregnancy
Radiation is toxic to tissues including reproductive organs. However, data is unfortunately scarce in humans. The testes are very sensitive to ionising radiation and therefore sperm count data provide a useful source of information on the effect of ionising radiation on the testes. In a study of healthy volunteer prisoners, aged 22–50 years, who were scheduled to undergo vasectomy, radiation exposure as low as 150 mGy resulted in significant suppression in sperm production [72]. In addition, there was transient abolition of sperm production in most men after exposure to a dose of 500 mGy, with higher doses leading to more prolonged suppression of spermatogenesis. Higher doses also lead to histological changes in the testes [72]. With regard to sperm quality, exposure to ionising radiation in male healthcare workers leads to a significant decrease in sperm quality as evidenced by decreased motility, decreased viability and altered morphological features [73]. Based on the available data, Rowley and colleagues adjudged that short-term exposure to ionising radiation of the magnitude of 110–150 mGy results in transient loss of spermatogenesis, whilst exposure to larger doses of 2000 mGy or higher over a prolonged period may lead to male infertility [74].
The exposure of a pregnant operator to ionising radiation continues to be a difficult issue to handle. As a conservative measure, the radiation dose recorded on the dosimeter inside the apron, which should be worn on the abdomen, is used to estimate the radiation exposure of the foetus. Whilst the mother’s abdomen will provide some shielding, this should not be taken into account [62]. Low doses of radiation as would be expected from operators who are properly protected and follow all of the abovementioned recommendations to reduce exposure do not pose an increased risk of prenatal complications or malformations beyond the incidence seen in the general population [75]. Therefore, proper and precise monitoring is essential to ensure that such doses are not exceeded. Current guidelines emphasise that the highest risk occurs in the first trimester, which is the time of organogenesis. Malformations are known to occur with exposures of around 100 mGy [75]. Current guidelines state that at the recommended exposure levels, malformations should not occur. Furthermore, current data suggests no alterations in mental development of children who are exposed to radiation at recommended doses in utero [62]. The greatest uncertainty resides in the risk of childhood malignancy in children, who are exposed to radiation at recommended doses in utero. However, based on the currently available data, there appears to be a small increase in risk which is of uncertain significance [75]. Further research in this area would certainly be welcome.
Current guidelines recommend that the dose of radiation after declaration of pregnancy not exceed 1 mSv. However, the authors state that this does not preclude the electrophysiologist from continuing to work in the electrophysiology lab. In order to do so, the guidelines recommend that the following occur: (1) the electrophysiologist should understand the risks of further radiation exposure and willingly elect to continue working in the electrophysiology lab; (2) to monitor the dose to the foetus, an extra dosimeter should be worn at the level of the abdomen monthly; the exposure dose values should be relayed to the electrophysiologist on a regular basis; (3) the electrophysiologist must be aware of all the aforementioned methods to reduce radiation exposure and should attempt to regulate her workload in fluoroscopy-guided procedures; and (4) a dedicated radiation monitoring and protection programme should exist in the hospital, led by a competent expert [27].
The pregnant electrophysiologist should employ constant radiation awareness, appropriate use of monitoring and radioprotection measures to minimise exposure to the lowest possible level during pregnancy [62]. A significantly greater amount of scattered radiation occurs during device implantation compared to ablation procedures. Therefore, great care should be taken to optimise positioning and shielding in order to reduce radiation exposure. A two-piece wrap around lead skirt and vest should be used with a minimum of 0.5 mm lead equivalence in the front [62]. As mentioned, 3D mapping systems, remote navigation systems as well as protection cabins should be utilised when available [76].
Summary and Outlook for the Future
Using all the options of modern fluoroscopy systems and wearing all personal protection gear available, daily work in the EP lab can be performed without significant increase of the professional risk. However, it is in the greatest personal interest of every operator to protect himself or herself as much as possible and apply ALARA principle whenever possible. Using all available 3D imaging information and applying ZERO-radiation techniques if possible is only a matter of dedicated training. Low personal exposure for the operator always equates to low exposure for the patient and vice versa, such that any attempt of radiation reduction will always benefit both parties. Future 3D EMS should aim to eliminate the need to use X-rays completely, which would open the opportunity to perform invasive procedures outside cath labs.
References
Scheinman MM, Morady F, Hess DS, Gonzalez R. Catheter-induced ablation of the atrioventricular junction to control refractory supraventricular arrhythmias. JAMA. 1982;248:851.
Al-Khatib SM, Stevenson WG, Ackerman MJ, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Heart Rhythm. 2018;15:e190–e252 [published online ahead of print 30 Oct 2017]. https://doi.org/10.1016/j.hrthm.2017.10.035.
Calkins H, Hindricks G, Cappato R, et al. 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation: executive summary. Europace. 2018;20(1):157–208.
Page RL, Joglar JA, Caldwell MA, et al. 2015 ACC/AHA/HRS guideline for the management of adult patients with supraventricular tachycardia: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2016;67:e27–e115.
Kim KP, Donald LM, Balter S, Kleinerman RA, Linet MS, Kwon D, et al. Occupational radiation doses to operators performing cardiac catheterization procedures. Health Phys. 2008;94:211–27.
Picano E, Vañó E, Rehani MM, Cuocolo A, Mont L, Bodi V, Bar O, Maccia C, Pierard L, Sicari R, Plein S, Mahrholdt H, Lancellotti P, Knuuti J, Heidbuchel H, Di Mario C, Badano LP. The appropriate and justified use of medical radiation in cardiovascular imaging: a position document of the ESC Associations of Cardiovascular Imaging, Percutaneous Cardiovascular Interventions and Electrophysiology. Eur Heart J. 2014;35:665–72.
Venneri L, Rossi F, Botto N, Andreassi MG, Salcone N, Emad A, Lazzeri M, Gori C, Vano E, Picano E. Cancer risk from professional exposure in staff working in cardiac catheterization laboratory: insights from the national research council’s biological effects of ionizing radiation VII report. Am Heart J. 2009;157:118–24.
Buchanan GL, Chieffo A, Mehilli J, Mikhail GW, Mauri F, Presbitero P, et al. The occupational effects of interventional cardiology: results from the WIN for safety survey. EuroIntervention. 2012;8:658–63.
Marinskis G, Bongiorni MG, Dagres N, Lewalter T, Pison L, Blomstrom-Lundqvist C. Scientific Initiative Committee, European Heart Rhythm Association. X-ray exposure hazards for physicians performing ablation procedures and device implantation: results of the European Heart Rhythm Association survey. Europace. 2013;15:444–6.
Roguin A, Goldstein J, Bar O. Brain tumours among interventional cardiologists: a cause for alarm? Report of four new cases from two cities and a review of the literature. EuroIntervention. 2012;7(9):1081–6.
Sarkozy A, De Potter T, Heidbuchel H, Ernst S, Kosiuk J, et al. Occupational radiation exposure in the electrophysiology laboratory with a focus on personnel with reproductive potential and during pregnancy: a European Heart Rhythm Association (EHRA) consensus document endorsed by the Heart Rhythm Society (HRS). Europace. 2017;19(12):1909–22.
ICRP. Compendium of dose coefficients based on ICRP publication 60. ICRP publication 119. Ann ICRP. 2012;41(Suppl).
Modan B, Keinan L, Blumstein T, et al. Cancer following cardiac catheterization in childhood. Int J Epidemiol. 2000;29:424–8.
Pearce MS, Salotti JA, Little MP, et al. Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: a retrospective cohort study. Lancet. 2012;380:499–505.
Jacobi W. The concept of effective dose—a proposal for the combination of organ doses. Radiat Environ Biophys. 1975;12:101–9.
Shrimpton PC, Wall BF, Jones DG, Fisher ES. The measurement of energy imparted to patients during diagnostic X-ray examinations using the Diamentor exposure-area product meter. Phys Med Biol. 1984;29:1199–208.
Heron J. Estimation of effective dose to the patient during medical X-ray examinations from measurements of the dose-area product. Phys Med Biol. 1992;37:2117–26.
Theocharopoulos N, Perisinakis K, Damilakis J, Varveris H, Gourtsoyiannis N. Comparison of four methods for assessing patient effective dose from radiological examinations. Med Phys. 2002;29:2070–9.
Jaco JW, Miller DL. Measuring and monitoring radiation dose during fluoroscopically guided procedures. Tech Vasc Interv Radiol. 2010;13:188–93.
Schueler BA, Vrieze TJ, Bjarnason H, Stanson AW. An investigation of operator exposure in interventional radiology. Radiographics. 2006;26:1533–41.
Servomaa A, Karppinen J. The dose-area product and assessment of the occupational dose in interventional radiology. Radiat Prot Dosim. 2001;96:235–6.
Williams JR. Scatter dose estimation based on dose-area product and the specification of radiation barriers. Br J Radiol. 1996;69:1032–7.
Koenig TR, Mettler FA, Wagner LK. Skin injuries from fluoroscopically guided procedures: part 2, review of 73 cases and recommendations for minimizing dose delivered to patient. AJR Am J Roentgenol. 2001;177:13–20.
Wagner LK, McNeese MD, Marx MV, et al. Severe skin reactions from interventional fluoroscopy: case report and review of the literature. Radiology. 1999;213:773–6.
Koenig TR, Wolff D, Mettler FA, et al. Skin injuries from fluoroscopically guided procedures: part 1, characteristics of radiation injury. AJR Am J Roentgenol. 2001;177:3–11.
Ciraj-Bjelac O, Rehani MM, Sim KH, et al. Risk for radiation induced cataract for staff in interventional cardiology: is there reason for concern? Catheter Cardiovasc Interv. 2010;76:826–34.
Rehani MM, Ciraj-Bjelac O, Vano E, Miller DL, Walsh S, Giordano BD, et al. International Commission on Radiological Protection. ICRP publication 117. Radiological protection in fluoroscopically guided procedures performed outside the imaging department. Ann ICRP. 2010;40:1–102.
Kovoor P, Ricciardello M, Collins L, Uther JB, Ross DL. Risk to patients from radiation associated with radiofrequency ablation for supraventricular tachycardia. Circulation. 1998;98:1534–40.
Lickfett L, Mahesh M, Vasamreddy C, et al. Radiation exposure during catheter ablation of atrial fibrillation. Circulation. 2004;110:3003–10.
Rosenthal LS, Mahesh M, Beck TJ, et al. Predictors of fluoroscopy time and estimated radiation exposure during radiofrequency catheter ablation procedures. Am J Cardiol. 1998;82:451–8.
Balter S. Stray radiation in the cardiac catheterisation laboratory. Radiat Prot Dosim. 2001;94:183.
Vano E. Radiation exposure to cardiologists: how it could be reduced. Heart. 2003;89:1123.
Macle L, Weerasooriya R, Jais P, et al. Radiation exposure during radiofrequency catheter ablation for atrial fibrillation. Pacing Clin Electrophysiol. 2003;26:288–91.
Voskoboinik A, Kalman ES, Savicky Y, Sparks PB, Morton JB, Lee G, Kistler PM, Kalman JM. Reduction in radiation dose for atrial fibrillation ablation over time: a 12-year single-center experience of 2344 patients. Heart Rhythm. 2017;14:810–6.
Sporton SC, Earley MJ, Nathan AW, Schilling RJ. Electroanatomic versus fluoroscopic mapping for catheter ablation procedures: a prospective randomized study. J Cardiovasc Electrophysiol. 2004;15:310–5.
Rogers DP, England F, Lozhkin K, Lowe MD, Lambiase PD, Chow AW. Improving safety in the electrophysiology laboratory using a simple radiation dose reduction strategy: a study of 1007 radiofrequency ablation procedures. Heart. 2011;97(5):366–70.
Casella M, Dello Russo A, Russo E, Catto V, Pizzamiglio F, Zucchetti M, et al. X-ray exposure in cardiac electrophysiology: a retrospective analysis in 8150 patients over 7 years of activity in a modern, large-volume laboratory. J Am Heart Assoc. 2018;7(11):e008233.
Vom J, Williams I. Justification of radiographic examinations: what are the key issues? J Med Radiat Sci. 2017;64(3):212–9.
Chambers CE, Fetterly KA, Holzer R, Lin PJ, Blankenship JC, Balter S, et al. Radiation safety program for the cardiac catheterization laboratory. Catheter Cardiovasc Interv. 2011;77(4):546–56.
Ernst S, Castellano I. Radiation exposure and safety for the electrophysiologist. Curr Cardiol Rep. 2013;15(10):402.
Fetterly KA, Mathew V, Lennon R, Bell MR, Holmes DR Jr, Rihal CS. Radiation dose reduction in the invasive cardiovascular laboratory: implementing a culture and philosophy of radiation safety. JACC Cardiovasc Interv. 2012;5:866–73.
Estner HL, Grazia Bongiorni M, Chen J, Dagres N, Hernandez-Madrid A, Blomstrom-Lundqvist C. Use of fluoroscopy in clinical electrophysiology in Europe: results of the European Heart Rhythm Association Survey. Europace. 2015;17:1149–52.
Miller DL, Vano E, Bartal G, Balter S, Dixon R, Padovani R, Schueler B, Cardella JF, de Baere T. Occupational radiation protection in interventional radiology: a joint guideline of the Cardiovascular and Interventional Radiology Society of Europe and the Society of Interventional Radiology. Cardiovasc Intervent Radiol. 2010;33:230–9.
de Souza E, de Macedo Soares JP. Occupational and technical correlations of interventional radiology. J Vasc Bras. 2008;7:341–50.
Vano E, Kleiman NJ, Duran A, Rehani MM, Echeverri D, Cabrera M. Radiation cataract risk in interventional cardiology personnel. Radiat Res. 2010;174:490–5.
Ciraj-Bjelac O, Rehani M, Minamoto A, Sim KH, Liew HB, Vano E. Radiation-induced eye lens changes and risk for cataract in interventional cardiology. Cardiology. 2012;123:168–71.
Jacob S, Donadille L, Maccia C, Bar O, Boveda S, Laurier D, Bernier MO. Eye lens radiation exposure to interventional cardiologists: a retrospective assessment of cumulative doses. Radiat Prot Dosim. 2013;153:282–93.
Principi S, Delgado Soler C, Ginjaume M, Beltran Vilagrasa M, Rovira Escutia JJ, Duch MA. Eye lens dose in interventional cardiology. Radiat Prot Dosim. 2015;165:289–93.
Hirshfeld JW Jr, Balter S, Brinker JA, et al. ACCF/AHA/HRS/SCAI clinical competence statement on physician knowledge to optimize patient safety and image quality in fluoroscopically guided invasive cardiovascular procedures. J Am Coll Cardiol. 2004;44:2259–82.
Theocharopoulos N, Damilakis J, Perisinakis K, Manios E, Vardas P, Gourtsoyiannis N. Occupational exposure in the electrophysiology laboratory: quantifying and minimizing radiation burden. Br J Radiol. 2006;79:644–51.
Beston S, Efstathopolous EP, Katritsis D, Faulkner K, Panayiotakis G. Patient radiation doses during cardiac catheterization procedures. Br J Radiol. 1998;71(846):634–9.
Agarwal S, Parashar A, Bajaj NS, et al. Relationship of beam angulation and radiation exposure in the cardiac catheterization laboratory. JACC Cardiovasc Interv. 2014;7:558–66.
Wittkampf FH, Wever EF, Vos K, Geleijns J, Schalij MJ, van der Tol J, et al. Reduction of radiation exposure in the cardiac electrophysiology laboratory. Pacing Clin Electrophysiol. 2000;23:1638–44.
Davies AG, Cowen AR, Kengyelics SM, Moore J, Pepper C, Cowan C, et al. X-ray dose reduction in fluoroscopically guided electrophysiology procedures. Pacing Clin Electrophysiol. 2006;29:262–71.
Walters TE, Kistler PM, Morton JB, Sparks PB, Halloran K, Kalman JM. Impact of collimation on radiation exposure during interventional electrophysiology. Europace. 2012;14(11):1670.
De Buck S, La Gerche A, Ector J, et al. Asymmetric collimation can significantly reduce patient radiation dose during pulmonary vein isolation. Europace. 2012;14:437–44.
Betsou S, Efstathopoulos EP, Katritsis D, Faulkner K, Panayiotakis G. Patient radiation doses during cardiac catheterization procedures. Br J Radiol. 1998;71:634–9.
Efstathopoulos E, Karvouni E, Kottou S, et al. Patient dosimetry during coronary interventions: a comprehensive analysis. Am Heart J. 2004;147:468–75.
Olcay A, Guler E, Karaca IO, Omaygenc MO, Kizilirmak F, Olgun E, Yenipinar E, Cakmak HA, Duman D. Comparison of fluoro and cine coronary angiography: balancing acceptable outcomes with a reduction in radiation dose. J Invasive Cardiol. 2015;27:199–202.
JCS Joint Working Group. Guidelines for radiation safety in interventional cardiology (JCS 2006). Digest version. Circ J. 2010;74:2760–85.
Smith PH. EC directive: 97/43/Euratom. Br J Radiol. 1998;71:108.
Heidbuchel H, Wittkampf FH, Vano E, Ernst S, Schilling R, Picano E, Mont L, Jais P, de Bono J, Piorkowski C, Saad E, Femenia F. Practical ways to reduce radiation dose for patients and staff during device implantations and electrophysiological procedures. Europace. 2014;16:946.
Wittkampf F, Wever E, Derksen R, Wilde A, Ramanna H, Hauer R, et al. LocaLisa: new technique for real-time 3-dimensional localization of regular intracardiac electrodes. Circulation. 1999;99:1312–7.
Gepstein L, Hayam G, Ben-Haim SA. A novel method for nonfluoroscopic catheter-based electroanatomical mapping of the heart. In vitro and in vivo accuracy results. Circulation. 1997;95:1611–22.
Kottkamp H, Hugl B, Krauss B, Wetzel U, Fleck A, Schuler G, et al. Electromagnetic versus fluoroscopic mapping of the inferior isthmus for ablation of typical atrial flutter: a prospective randomized study. Circulation. 2000;102:2082–6.
Earley MJ, Showkathali R, Alzetani M, Kistler PM, Gupta D, Abrams DJ, et al. Radiofrequency ablation of arrhythmias guided by non-fluoroscopic catheter location: a prospective randomized trial. Eur Heart J. 2006;27:1223–9.
Reddy VY, Malchano ZJ, Neuzil P. Early clinical experience with CARTO-merge for integration of 3D-CT imaging with real-time mapping to guide catheter ablation of atrial fibrillation. Heart Rhythm. 2005;2:S160.
Ernst S, Ouyang F, Linder C, et al. Initial experience with remote catheter ablation using a novel magnetic navigation system: magnetic remote catheter ablation. Circulation. 2004;109(12):1472–5.
Ueda A, Suman-Horduna I, Mantziari L, Gujic M, Marchese P, Ho SY, Babu-Narayan SV, Ernst S. Contemporary outcomes of supraventricular tachycardia ablation in congenital heart disease: a single-center experience in 116 patients. Circ Arrhythm Electrophysiol. 2013;6:606–13.
Schwagten BK, Szili-Torok T, Rivero-Ayerza M. Usefulness of remote magnetic navigation for ablation of ventricular arrhythmias originating from outflow regions. Neth Heart J. 2009;17:245–9.
Guarguali S, Cazzoli I, Kempny A, Gatzoulis M, Ernst S. A new technique for zero fluoroscopy atrial fibrillation ablation without the use of intracardiac echocardiography. J Am Coll Cardiol Clin Electrophysiol. 2018;4:1647–8.
Clifton DK, Bremner WJ. The effect of testicular x-irradiation on spermatogenesis in man. A comparison with the mouse. J Androl. 1983;4(6):387–92.
Kumar D, Salian SR, Kalthur G, Uppangala S, Kumari S, Challapalli S, et al. Semen abnormalities, sperm DNA damage and global hypermethylation in health workers occupationally exposed to ionizing radiation. PLoS One. 2013;8(7):e69927.
Rowley MJ, Leach DR, Warner GA, Heller CG. Effect of graded doses of ionizing radiation on the human testis. Radiat Res. 1974;59(3):665–78.
The 2007 Recommendations of the International Commission on Radiological Protection. ICRP publication 103. Ann ICRP. 2007;37(2–4):1–332.
AlTurki A, Proietti R. Remote magnetic navigation versus contact force technology: the two faces of the ablation lesion. Pacing Clin Electrophysiol. 2018;41(5):447–9.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Nagarajan, D.V., AlTurki, A., Ernst, S. (2019). Radiation Exposure and Safety for the Electrophysiologist. In: Proietti, R., Wang, Y., Yao, Y., Zhong, G., Lin Wu, S., Ayala-Paredes, F. (eds) Cardiac Electrophysiology Without Fluoroscopy. Springer, Cham. https://doi.org/10.1007/978-3-030-16992-3_2
Download citation
DOI: https://doi.org/10.1007/978-3-030-16992-3_2
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-16991-6
Online ISBN: 978-3-030-16992-3
eBook Packages: MedicineMedicine (R0)