Abstract
The selective use of magnetic resonance imaging (MRI) has provided high resolution images of soft tissue for the management of cancer and complicated fistula-in-ano. It also has provided a better understanding of normal anorectal anatomy and the functional interaction of the pelvic floor and perineal structures during provocative maneuvers such as defecation, squeezing, and straining. Its use in rectal cancer has better defined the role of neoadjuvant therapies during follow-up. In complex cryptogenic fistula and perianal Crohn’s disease, MRI has assisted in the use of new reoperative techniques such as the LIFT procedure (ligation of intersphincteric fistula tract), fistula plugs, and fibrin glues, as well as in identifying patients best suited to advancement anoplasty and bowel diversion. This chapter outlines the normal (magnetic resonance) anatomy of the anorectum and the role of MRI in staging rectal cancer and anorectal fistulas, the interpretation of which has proven essential in the reconstructive/reoperative case.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Magnetic resonance imaging (MRI) is important as a guide for the treatment of anorectal diseases, of which the two most important are rectal cancer and perianal fistulas. In recent years, it has become apparent that MRI of the anorectum is of value for the optimal treatment of rectal cancer. With the use of MRI for local staging, the treatment of rectal cancer can be tailored according to risk of recurrence. In this way, treatment benefits can be weighed against treatment-related complications in the search for the best possible treatment for every individual patient. With regard to anorectal fistulas, we now know that the optimal treatment is preceded by MRI in some complicated and recurrent cases. MRI is able to identify complex tracts of fistulas and possible related abscesses and can guide surgical exploration, in this way reducing the risk for incomplete resection of the fistula and related abscesses and thus recurrent disease. The purpose of this chapter is to describe the (magnetic resonance) anatomy of the anorectum, the role of MRI in staging rectal cancer and anorectal fistulas, and the imaging-guided treatment implications that can influence the complicated primary and reconstructive/reoperative case.
Anatomy
The Anorectum in Relation to Other Organs in the Pelvis
The rectum extends from the anorectal junction to the rectosigmoid junction and is 12–15 cm long, depending on whether the starting point of measurement is the anal verge, dentate line, or the anorectal junction. The rectosigmoid junction is located at the level of the third sacral vertebra. The anal canal is located caudal to the anorectal junction, where it bends sharply posteriorly (Fig. 4.1). The anus and rectum are located in the posterior part of the pelvis, just anterior to the sacral and coccygeal vertebrae and posterior to the vagina and uterus in women and the prostate and seminal vesicles in men (Fig. 4.2).
Axial T2-weighted, fast spin echo (TR/TE 342/150 ms) magnetic resonance image of a male pelvis. This image shows the close relation of the organs in the pelvis. On the posterior side, the sacral bone is visible (S); on both lateral sides, the head of the femur (H) is visible. The lumen of the rectum (L) is hypointense. The rectum itself contains an isointense circular rectal tumor (black arrowhead). On the anterior side of the rectum with its surrounding fat, the seminal vesicles (VS), prostate (P) and bladder (B) are located. The mesorectum of this patient with rectal cancer also contains a lymph node (white arrowhead)
Mesorectal Compartment
The mesorectal fascia (Fig. 4.3) envelops the mesorectal compartment containing the rectum and the mesorectal fat comprising blood vessels lymph nodes and lymphatic vessels. This region is referred to as the mesorectum. The normal rectal wall is depicted as two layers on T2-weighted magnetic resonance images. The hypointense layer corresponds to the muscularis propria recti and the inner isointense layer represents the mucosa. When the rectal wall is inflamed (i.e., in inflammatory bowel disease or as a reaction to radiotherapy), it becomes edematous, which is depicted by a third layer with high signal intensity between the inner and outer layer (Fig. 4.4).
Sagittal T2-weighted, fast spin echo (TR/TE 3427/150 ms) magnetic resonance image of a male patient with rectal cancer shows a high signal-intensity layer (white arrowheads), indicating submucosal edema. This layer is situated between the inner mucosal layer (black arrowheads) and the muscularis propria recti (black arrows) (Reprinted with permission from Lahaye et al. [1])
On T2-weighted magnetic resonance images, the mesorectal fat is seen as a high signal intensity structure. The mesorectal fat surrounds the rectal wall but does not have the same thickness throughout its length and circumferential location. On the anterior side, the layer of mesorectal fat is thinner; therefore, the anterior rectal wall is in close proximity to the genital organs, which consist of the prostate and seminal vesicles in men and the vagina and cervix in women. In rectal cancer, this close relationship as well as the distal tapering of the mesorectal fat makes invasion of the anterior organs as well as the pelvic floor more likely (Fig. 4.5).
Coronal T2-weighted, fast spin echo (TR/TE 3427/150 ms) magnetic resonance image of a patient with rectal cancer showing the distal tapering of the mesorectum, which entails a close relationship of the distal rectal wall and the muscles of the pelvic floor (white dotted line) (Reprinted with permission from Lahaye et al. [1])
The mesorectal fascia itself is depicted on T2-weighted magnetic resonance images as an hypointense fine line surrounding the mesorectal fat (see Fig. 4.3) [2]. Anteriorly, the fascia becomes thicker at the point where it separates the fat from the seminal vesicles in men and the vagina in women. At this point, the fascia is referred to as Denonvilliers’ fascia. Posteriorly, the presacral fascia of Waldeyer, visible as a hypointense signal, is located between the mesorectal fascia and the sacrum. During surgical resection of the rectum, the plane between these two fasciae is the plane of resection. The upper two thirds of the (meso)rectum are enveloped by the peritoneum on the anterior and lateral sides. Anteriorly, the peritoneum is reflected at the height of the seminal vesicles in men and the cervix/posterior vaginal wall in women to form the rectovesical or rectouterine pouch (of Douglas).
Anal Sphincter
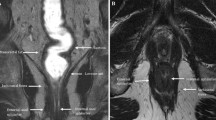
The muscular part of the anal canal is formed by the internal and external anal sphincters, with a longitudinal muscle layer lying in between. More proximally, this sphincter complex is surrounded by the levator ani complex, which forms an important part of the pelvic floor (Fig. 4.6). The main innervation of the levator ani complex and the external anal sphincter is the pudendal nerve. The internal anal sphincter is innervated by the parasympathetic nerve fibers arising from S2 to S4.
Coronal gadolinium contrast–enhanced, T1-weighted, fast spin echo magnetic resonance image. The muscles of the anal canal are indicated: (a) the internal sphincter, (b) the longitudinal muscle layer, (c) the external sphincter, (d) puborectalis muscle, (e) m. submucosae ani, (f) levator ani muscle, (g) intersphincteric space, (h) ischioanal (or ischiorectal) space, (i) supralevator space, and (j) thin muscle layer located within the submucosa
Vascular Supply
The main blood supply of the rectum arrives via the superior rectal artery, arising from the inferior mesenteric artery. The superior rectal artery divides into a right and left branch, feeding both sides of the rectum. The superior rectal vein runs dorsally parallel to the artery. The superior rectal artery is depicted as a hypointense structure on T2-weighted MRI (Fig. 4.7). The distal part of the rectum also is supplied by the middle rectal artery, arising from the internal iliac artery. The internal iliac artery and vein are situated on the lateral pelvic sidewall. The inferior rectal artery arises from the pudendal artery, whereas the latter is a branch of the internal iliac artery. For surgeons, the presence of the presacral venous plexus, situated just behind Waldeyers’ fascia, is of importance because accidental injury can lead to profuse bleeding with great difficulty in hemostasis.
Lymphatic Drainage
The main lymphatic drainage of the rectum follows the superior rectal vein and the inferior mesenteric vein to the para-aortic nodes. Mesorectal lymph nodes are mainly situated in the lateral and posterior part of the mesorectum [3]. Further lymphatic drainage of the rectum, especially the low rectum, is through the drainage system along the middle rectal artery and vein, the so-called lateral pelvic nodes. Nodal metastases do occur in these nodes, usually in low rectal tumors, and most often are associated with involved mesorectal nodes (Fig. 4.8) [4, 5].
Axial T2-weighted, fast spin echo (TR/TE 3427/150 ms) magnetic resonance image. There is a large lymph node suspected to be malignant outside the mesorectum (white arrow) in the region of the middle rectal artery. There are also nodes located within the mesorectum (black arrows). The left node is not large and has a sharp border and an homogeneous signal, so it could be a benign node; however, the right mesorectal node is large, has an irregular border and a heterogeneous signal, and is therefore a malignant node. R rectal tumor, U uterus
Nerves
Innervation of anorectal, urinary, and sexual function is located in the pelvis. Sympathetic as well as parasympathetic nerves course along the walls of the pelvis in close proximity to the mesorectum, making invasion by tumor in the proximity of the nerves and injury during surgery more likely [6, 7]. The superior hypogastric plexus is located at the level of the sacral promontory, dorsal to the rectum. It divides into left and right hypogastric nerves, which run around the rectum along the pelvic side wall to the bladder and the genital organs. At the level of S2–S5, these nerve fibers are accompanied by parasympathetic fibers, the nervi erigenti, which follow the same route to the bladder and the genital viscera. The sympathetic and parasympathetic nerve fibers together form the right and left inferior hypogastric plexus. This inferior hypogastric plexus cannot be depicted on MRI.
Rectal Cancer
Colorectal cancer was diagnosed in almost 300,000 patients in Europe in 2006. It is the third most common cancer in men and women taken together and the second most common cause of cancer-related death [8, 9]. About 30 % of all colorectal cancers are located in the rectum. Apart from cancer-related death, rectal cancer is especially notorious for local recurrence, which is accompanied by severe morbidity. The treatment of rectal cancer, therefore, focuses on optimal local and distant control. In past decades, this treatment has undergone major developments both in surgery and neoadjuvant therapy. With regard to surgery, the introduction of total mesorectal excision (TME) has significantly reduced the local recurrence rate [10]. In TME, the rectum is removed along with its surrounding fat, including lymph nodes and blood vessels, through which tumor cells can spread, using a sharp surgical technique along the outside of the mesorectal fascia, creating a mesorectal package. This improvement in surgical technique was driven by the notion that the distance of the tumor to the circumferential resection margin (CRM), as judged by the pathologist, was an important factor in determining local recurrence rates [11]. At the same time, Scandinavian trials have shown that preoperative radiotherapy in addition to conventional surgery is more effective than postoperative radiotherapy for reduction of local recurrence. The Dutch TME trial [12] as well as the MRC CR07 trial [13] further showed that, even with good TME surgery, neoadjuvant 5 × 5 Gy short-course radiation therapy has a favorable effect on local recurrence rates for the whole group; hence, in the Netherlands and Scandinavia, 5 × 5 Gy before TME has become the standard treatment for all patients with rectal cancer. A subgroup analysis of the Dutch TME trial also showed that a short course of preoperative radiation (5 × 5 Gy) does not improve outcome in stage I tumors (T1-2N0) because these tumors already have a favorable prognosis without radiotherapy [12]. Because it was a subgroup analysis and because of the difficulties with nodal staging, clinicians were reluctant to withhold radiotherapy from these patients. On the other hand, for more advanced tumors – those with an involved CRM – it has been shown that the recurrence rates were too high, even after a preoperative short course of radiation [14]. To avoid local recurrences in this group, more aggressive neoadjuvant treatment has been advocated. In 1991, Krook et al. [15] showed the advantage of the addition of chemotherapy to adjuvant radiotherapy for reduction of local recurrence and the improvement of overall survival of patients with more advanced tumors. Two other studies proposed chemotherapy plus radiation therapy in a neoadjuvant setting because it not only improved the local control, [16, 17] but also showed a better adherence to treatment when compared with postoperative chemotherapy [16]. In 2004, Sauer et al. [18] confirmed that this combined chemoradiation is more effective in terms of local control and toxicity when given preoperatively compared with postoperatively. Locally advanced rectal cancer is, therefore, preferably treated with long-course neoadjuvant radiation, with chemotherapy as a radiosensitizer.
The selection of these “more advanced tumors” with a high risk for circumferential margin involvement has long been made based on clinical examination (“fixed tumors”) and endosonography, when available. Clinical and endosonographic staging is not very accurate for assessing tumors with involved CRMs. In the Dutch TME trial, where the intent was to exclude clinically fixed (“locally advanced”) tumors, there was a rate of involved margins of 16 %, often due to the inclusion of large “advanced tumors” that were understaged. With the introduction of MRI at the beginning of this millennium to the workup of patients with rectal cancer, the mesorectal fascia – the anticipated CRM – could accurately be visualized. Tumors that were growing into or near the mesorectal fascia could be better identified with this approach [2, 19–24]. This has been a significant development because of the importance of the circumferential resection plane, which had been previously established [11].
Apart from the CRM, the presence of nodal disease is an important risk factor for local recurrence. Until recently, imaging modalities lacked sufficient accuracy for nodal prediction because size was the main predictive criterion, and in rectal cancer, lymph node micrometastases have been known to occur in nodes with a maximal diameter smaller than 5 mm [25]. The introduction of lymph node–specific contrast agents for MRI offered the hope of better lymph node prediction [26]. These developments in MRI related to accurate prediction of the CRM as well as promising results with regard to nodal staging could lead to a more differentiated treatment approach in rectal cancer rather than one uniform treatment for all. Of all available treatment options, one that best fits an individual patient can be chosen on the basis of an assessment of the risk for local recurrence. Theoretically, this differentiated treatment leads to optimal local control with minimal treatment-related morbidity for the whole group of rectal cancer patients.
MRI-based, tailored treatment of rectal cancer and its surgical outcomes have been studied in a prospective multicenter study in the Netherlands; the definitive results of the 3-year local recurrence rate will soon be available. In this study, all rectal cancer patients received preoperative MRI, on which the important risk factors for recurrence (i.e., CRM, nodal status, and tumor height) were identified. Based on the MRI findings, stratification was performed as follows: surgery alone for the low-risk cases, 5 × 5 Gy radiotherapy plus surgery for the intermediate risk cases, and a long course of preoperative chemoradiotherapy (CRT) for the high-risk cases. The purpose of this study was to show that differentiated treatment for patients with rectal cancer provides optimal treatment with minimal treatment-related complications. The preliminary results of this study show an actuarial 2-year local recurrence rate of 2.8 %, which proves that this differential treatment is safe [27].
Today, however, other studies are exploring even more directed treatment, exploiting the sometimes phenomenal responses to neoadjuvant CRT. Complete response rates of up to 25 % have been described, and an even larger proportion of patients have considerable downsizing or downstaging [28]. The current trend is toward minimally invasive, organ-sparing surgery in the good responders after CRT and even a wait-and-see policy in the complete responders [29–31]. The accuracy of using MRI for restaging tumors after CRT is of great importance in this scenario.
Staging of Rectal Cancer with MRI
T Stage
The T stage of rectal cancer is divided into four categories. A T1 tumor is confined to the submucosa; T2 refers to a tumor that invades the muscularis propria, T3 invades the mesorectal fat, and T4 invades surrounding organs. The accuracy of MRI for the prediction of T stage shows a wide variability in different studies, ranging from 67 to 83 % [32]. This wide range is partly explained by the inaccuracy of MRI in reliably distinguishing between T1 and T2 tumors because the submucosal layer is not depicted. For this distinction, endorectal ultrasound is more reliable [32]. The other reason is the difficulty of MRI to distinguishing between T2 and T3 tumors. With an intact hypointense muscular wall, the tumor most often does not invade the mesorectal fat (positive predictive value, 86–91 %) [33]. However, in cases of desmoplastic reaction in borderline T2 or T3 tumors, MRI has difficulty in distinguishing accurately between desmoplastic reaction with (pT3) or without (pT2) viable tumor cells (Fig. 4.9a, b). However, large T3 tumors and T4 tumors can be accurately selected using MRI, with sensitivities of 74 and 82 % and specificities of 76 and 96 %, respectively [34].
(a and b), Axial contrast-enhanced, T1-weighted, turbo spin echo (TR/TE 612/15 ms) magnetic resonance images. Both show a desmoplastic reaction to a rectal tumor (black arrows), although it is impossible to differentiate between desmoplastic reaction with (b) tumor cells (therefore a T3 tumor) and desmoplastic reaction without (a) tumor cells (therefore, a T2 tumor). Note: Both images also show mesorectal lymph nodes. R rectal tumor, b bladder (Reprinted with permission from Beets-Tan et al. [21])
Circumferential Resection Margin
The mesorectal fascia – the surgeon’s plane of resection in rectal cancer surgery – is clearly depicted as a hypointense line on transverse T2-weighed MRI. Therefore, the CRM can be accurately measured and predicted. This has been shown in a meta-analysis of seven single-center studies, with sensitivities of 60–88 % and specificities between 73 and 100 % [1], and has been confirmed by a large, prospective, multicenter study by the Mercury Study Group, showing an overall accuracy of 88 %, suggesting that the prediction of the CRM is reliable in experienced hands (Fig. 4.10) [35].
Nodal Disease
Information about both mesorectal and extramesorectal nodal disease is important for treatment decisions for patients with rectal cancer. Historically, size criteria were used to differentiate between benign and malignant lymph nodes in rectal cancer. This is a rather limited approach because nodal metastases also occur in small nodes (<5 mm), and in rectal cancer these are the majority of metastatic nodes [25]. Additional criteria, such as border and heterogeneity, have been used to improve the prediction of nodal disease. A benign node is more likely to have a sharp border and homogeneous signal intensity, whereas malignant nodes have an irregular border with heterogeneous signal intensity. With these criteria, performance improves, with sensitivity between 36 and 85 % and a specificity of 95–100 % [36, 37]. It is, however, important to realize that these criteria are more difficult to apply in small nodes. The use of different contrast agents as well as diffusion-weighted MRI for the accurate prediction of nodal status are still under investigation. Ultrasmall paramagnetic iron-oxide, a lymph node–specific magnetic resonance contrast agent, has proven reliable, with a high negative predictive value in expert as well as in general hands [38]. Despite these encouraging results, ultrasmall paramagnetic iron-oxide is not available for general clinical use.
Restaging After Neoadjuvant Chemoradiation Therapy
In locally advanced tumors that respond well to neoadjuvant therapy, the surgeon might consider less extensive surgery when the tumor has regressed from an initially anticipated involved mesorectal fascia. Restaging is even more essential when local excision to preserve organs and nerves or even a wait-and-see policy is considered for patients with complete or near complete tumor response.
yT and yN Stage
MRI staging after CRT has only limited accuracy for both T and N stages [39–41]. The main problem for prediction of T stage after CRT is the formation of hypointense scar tissue. It is impossible to differentiate between such scar tissue alone and scar tissue with residual tumor cells. Therefore, a considerable amount of overstaging is inevitable when interpreting images after CRT when the radiologist wishes to be “on the safe side.” However, for local treatment purposes, the main issue is to identify those tumors limited to the bowel wall without positive lymph nodes so that the exact T and N stage is less relevant. A recent study has shown that the prediction of a ypT0–2 tumor (confined to the bowel wall and therefore suitable for local excision) is accurate in expert as well as in general hands when combining morphologic features as well as volumetric analysis of MRI before and after CRT [33]. The positive predictive value has been reported as high as 94 %. Another prospective multicenter study showed that, with the use of dedicated contrast-enhanced MRI, it is possible to select ypT0–2/ypN0 tumors, with a low risk of understaging. This means that safe selection of patients suitable for local excision is possible [42]. The safe prediction of ypN0 status has been confirmed by Lahaye et al. [43].
Tumor Regression from the Mesorectal Fascia
A recent analysis of a multicenter study by the Mercury Study Group showed an overall accuracy of 77 % for prediction of CRM involvement after chemoradiation, with an especially high negative predictive value of 98 % [44]. This high negative predictive value is the multicenter confirmation of the results of two single-center studies published previously [45]. Vliegen et al. focused on the patterns of CRM involvement or regression after chemoradiation and indicated that there are four different patterns: (1) development of a fat pad larger than 2 mm, (2) development of spiculations from the tumor to the mesorectal fascia, (3) development of diffuse hypointense fibrotic tissue, and (4) persistence of diffuse iso- or hyperintense tissue. Patterns 1 and 2 have a high likelihood of tumor regression away from the mesorectal fascia and thus a free resection margin, whereas pattern 3 has a 50 % chance of persistent involvement of the fascia, and pattern 4 predicts definite persistent involvement of the fascia, indicating that a wider excision is inevitably required.
Magnetic Resonance Protocol for Rectal Cancer Imaging
New-generation body phased-array coils have a high signal-to-noise ratio and provide high spatial and contrast resolution and therefore are the standard for rectal cancer imaging. Bowel or other preparation is not necessary. Gadolinium is not helpful for prediction of T stage and CRM [47]. Endorectal contrast is not preferred because it has been shown to stretch the bowel wall and as a result compress the mesorectal fat, making the prediction of T and N stage and, even more importantly, the CRM, difficult. Two-dimensional T2-weighted fast spin echo is used in three planes. It is performed first in the sagittal plane, and the axial and coronal planes are angled perpendicularly to the tumor, as shown on the sagittal MRI. This is important to avoid a partial volume effect that makes it impossible to reliably predict T stage or to measure the distance from the tumor to the mesorectal fascia. Thin slices (4 mm or less) are used and the field of view needs to cover the promontory to the anal canal in a craniocaudal direction and the pubic bone to the sacral bone in an anteroposterior direction.
Anorectal Fistulas
Anorectal fistulas consist of extra-anatomical tracts between the rectum or anal canal and the perineal skin. These tracts develop due to infection of the anal glands in the subepithelial layer of the anal canal. The spread of the infection, caused by an expanding abscess, can lead to the formation of a fistula. Anal fistulas can consist of a simple or primary track or a complex track that comprises a fistula track with secondary extensions, including abscesses. The latter more often occur in patients with Crohn’s disease and recurrent fistula, a topic covered by Dr. Dana Sands in Chap. 22.
Fistulas are classified into four different subtypes, as proposed by Parks et al. [48] in 1976. The first type is the intersphincteric fistula, which courses between the internal and external anal sphincter down to the perianal skin, in close proximity to the anus. The transsphincteric fistula courses through the internal and external anal sphincters to the ischiorectal space. The suprasphincteric fistula courses in a cranial direction, bending around the puborectal sling and then coursing down through the ischiorectal fat. The extrasphincteric fistula penetrates both the internal and external anal sphincter at a high level (supralevator), then splits in the ischiorectal fat and courses both downward to the perianal skin and upward to the rectum in the ischiorectal fossa.
Anal fistulas have a tendency to recur after surgery, most commonly because of missed extensions and internal openings. It now is recognized that preoperative imaging, in particular MRI, is able to identify fistulas and associated abscesses in complex tracks that would otherwise have been missed by surgeons, most notably in the multiply reoperated case. Preoperative imaging has been shown to influence subsequent surgery and significantly diminish the chance of recurrent disease, and hence overall successful outcome, as a result [49, 50].
Imaging of fistula-in-ano traditionally was performed by fistulography, but this has been shown to be unreliable [51]. The most important drawback of fistulography is that it does not show the relation of the fistula to the anal sphincter complex because the latter is not depicted on the fistulogram and can only be inferred. Furthermore, when fistulas are blocked by debris, they are not depicted on the fistulogram, giving the surgeon an incomplete view of a possible complex fistula.
In 2004, Buchanan et al. [50] published a study comparing endoanal ultrasound (EAUS), digital examination, and MRI for the visualization of fistulas and showed a superior accuracy of classification for MRI (90 %) when compared with EAUS (81 %) and digital examination (61 %), although EAUS is superior for visualization of the internal fistula opening (accuracy of EAUS versus MRI: 97 % versus 91 %). The superior results of MRI over EAUS were confirmed in a systematic review by Sahni et al. [52] in 2008.
There has been some discussion in the literature about the use of endoanal versus external phased-array coils in MRI of fistula-in-ano. Endoanal coils are probably somewhat more accurate in defining the primary fistula track [53], but complex tracks as well as secondary abscesses are often located outside the field of view of the endoanal coil [54, 55]. These are the collections that tend to be overlooked during surgery and for which MRI was proven to have additional benefit.
Magnetic Resonance Protocol for Anorectal Fistula Imaging
Similar to rectal imaging, fistula imaging with an external phased-array coil results in the high signal-to-noise ratio necessary for high-resolution MRI. Two-dimensional T2-weighted fast spin echo is used in the sagittal plane to plan the additional axial and coronal planes of imaging. The anal canal is tilted a little forward compared with the vertical axis of the patient. It is of great importance to angle the axial and coronal planes perpendicular to the anal canal because partly oblique images of the anal canal make identification of anatomical structures and the fistula track difficult. Fat-suppression images (short tau inversion recovery) are used as well as T2-weighted fast spin echo (FSE) for optimal detection of the (bright) fistula tracks. The field of view should extend at least from the presacral space to the perineum. Sometimes fistula tracks extend further upward or downward (even as far as the subcutaneous tissue in one or both legs), making a larger field of view necessary. With fistula tracks and collections that are present in the supralevator space, it is sometimes necessary to create additional axial and coronal images perpendicular to the rectum to better appreciate the relation of the rectum to the internal opening. Examples of a transsphincteric and extrasphincteric fistula as demonstrated with T2-weighted spin-echo MRI are shown in Fig. 4.11a, b.
(a) Sagittal T2-weighted, turbo spin echo (TR/TE 3427/150 ms) magnetic resonance image showing a hyperintense transsphincteric fistula track (white arrowhead) on the posterior side of the anal canal (black arrowhead). The sphincter is indicated by a white arrow. (b) Coronal T2-weighted, turbo spin echo (TR/TE 3427/150 ms) magnetic resonance image showing an hyperintense extrasphincteric fistula track (black arrow). The track ends in an abscess (white arrow) on the left pelvic floor (Reprinted with permission from Beets-Tan et al. [56])
Conclusion and Future Prospects
MRI of anorectal diseases is of invaluable importance for treatment decisions for patients with both rectal cancer and anorectal fistula and for the management of the primary and reoperative case. For rectal cancer, a combination of new neoadjuvant treatment regimens in locally advanced tumors and dedicated (magnetic resonance) imaging to evaluate the extension of disease before and after neoadjuvant treatment has led to an individually based treatment, combining optimal outcome with minimal treatment-related morbidity. Ongoing research of treatment focuses on organ-saving procedures and the ability of MRI to select the well-responding patients who are eligible for these treatment options. With improvement of imaging technology and diagnostic accuracy, it will be safe to further implement treatment strategies involving local excision of small tumors (either primary tumors or tumors downstaged after neoadjuvant CRT) or even to adopt a wait-and-see policy in those with a complete response after CRT.
For anorectal fistula, dedicated imaging is important to guide surgery, with the aim of eradicating complex fistula tracks, consequently reducing the chance for recurrences while preserving function. The demonstration of supralevator disease alters the surgical approach toward primary repair of the internal opening and the sphincter complex, where injudicious drainage through the ischiorectal fossa may result in iatrogenic extra- or suprasphincteric fistulas that are then difficult to manage successfully.
References
Lahaye MJ, Engelen SM, Nelemans PJ, Beets GL, van de Velde CJ, van Engelshoven JM, et al. Imaging for predicting the risk factors – the circumferential resection margin and nodal disease – of local recurrence in rectal cancer: a meta-analysis. Semin Ultrasound CT MR. 2005;26(4):259–68.
Bissett IP, Fernando CC, Hough DM, Cowan BR, Chau KY, Young AA, et al. Identification of the fascia propria by magnetic resonance imaging and its relevance to preoperative assessment of rectal cancer. Dis Colon Rectum. 2001;44:259–65.
Dworak O. Morphology of lymph nodes in the resected rectum of patients with rectal carcinoma. Pathol Res Pract. 1991;187:1020–4.
Moriya Y, Sugihara K, Akasu T, Fujita S. Importance of extended lymphadenectomy with lateral node dissection for advanced lower rectal cancer. World J Surg. 1997;21:728–32.
Engelen SM, Beets-Tan RG, Lahaye MJ, Kessels AG, Beets GL. Location of involved mesorectal and extramesorectal lymph nodes in patients with primary rectal cancer: preoperative assessment with MR imaging. Eur J Surg Oncol. 2008;34:776–81.
Church JM, Raudkivi PJ, Hill GL. The surgical anatomy of the rectum – a review with particular relevance to the hazards of rectal mobilisation. Int J Colorectal Dis. 1987;2:158–66.
Havenga K, DeRuiter MC, Enker WE, Welvaart K. Anatomical basis of autonomic nerve-preserving total mesorectal excision for rectal cancer. Br J Surg. 1996;83:384–8.
Boyle P, Ferlay J. Mortality and survival in breast and colorectal cancer. Nat Clin Pract Oncol. 2005;2:424–5.
Ferlay J, Autier P, Boniol M, Heanue M, Colombet M, Boyle P. Estimates of the cancer incidence and mortality in Europe in 2006. Ann Oncol. 2007;18:581–92.
Heald RJ, Ryall RD. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet. 1986;1(8496):1479–82.
Nagtegaal ID, Quirke P. What is the role for the circumferential margin in the modern treatment of rectal cancer? J Clin Oncol. 2008;26:303–12.
Kapiteijn E, Marijnen CA, Nagtegaal ID, Putter H, Steup WH, Wiggers T, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med. 2001;345:638–46.
Sebag-Montefiore D, Steele RJ, Quirke P, Grieve R, Khanna S, Monson JR. Routine short course pre-op radiotherapy or selective post-op chemoradiotherapy for resectable rectal cancer? Preliminary results of the MRC CR 07 randomized trial. J Clin Oncol. 2006;24(18S):3511.
Marijnen CA, Nagtegaal ID, Kapiteijn E, Kranenbarg EK, Noordijk EM, van Krieken JH, et al. Radiotherapy does not compensate for positive resection margins in rectal cancer patients: report of a multicenter randomized trial. Int J Radiat Oncol Biol Phys. 2003;55:1311–20.
Krook JE, Moertel CG, Gunderson LL, Wieand HS, Collins RT, Beart RW, et al. Effective surgical adjuvant therapy for high-risk rectal carcinoma. N Engl J Med. 1991;324:709–15.
Bosset JF, Collette L, Calais G, Mineur L, Maingon P, Radosevic-Jelic L, et al. Chemotherapy with preoperative radiotherapy in rectal cancer. N Engl J Med. 2006;355:1114–23.
Gerard JP, Conroy T, Bonnetain F, Bouche O, Chapet O, Closon-Dejardin MT, et al. Preoperative radiotherapy with or without concurrent fluorouracil and leucovorin in T3-4 rectal cancers: results of FFCD 9203. J Clin Oncol. 2006;24:4620–5.
Sauer R, Becker H, Hohenberger W, Rodel C, Wittekind C, Fietkau R, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med. 2004;351:1731–40.
Brown G, Richards CJ, Newcombe RG, Dallimore NS, Radcliffe AG, Carey DP, et al. Rectal carcinoma: thin-section MR imaging for staging in 28 patients. Radiology. 1999;211:215–22.
Blomqvist L, Machado M, Rubio C, Gabrielsson N, Granqvist S, Goldman S, et al. Rectal tumour staging: MR imaging using pelvic phased-array and endorectal coils vs endoscopic ultrasonography. Eur Radiol. 2000;10:653–60.
Beets-Tan RG, Beets GL, Vliegen RF, Kessels AG, Van Boven H, De Bruine A, et al. Accuracy of magnetic resonance imaging in prediction of tumour-free resection margin in rectal cancer surgery. Lancet. 2001;357(9255):497–504.
Beets-Tan RG, Beets GL. Rectal cancer: how accurate can imaging predict the T stage and the circumferential resection margin? Int J Colorectal Dis. 2003;18:385–91.
Peschaud F, Cuenod CA, Benoist S, Julie C, Beauchet A, Siauve N, et al. Accuracy of magnetic resonance imaging in rectal cancer depends on location of the tumor. Dis Colon Rectum. 2005;48:1603–9.
Salerno G, Daniels IR, Moran BJ, Wotherspoon A, Brown G. Clarifying margins in the multidisciplinary management of rectal cancer: the MERCURY experience. Clin Radiol. 2006;61:916–23.
Wang C, Zhou Z, Wang Z, Zheng Y, Zhao G, Yu Y, et al. Patterns of neoplastic foci and lymph node micrometastasis within the mesorectum. Langenbecks Arch Chir. 2005;39:312–8.
Will O, Purkayastha S, Chan C, Athanasiou T, Darzi AW, Gedroyc W, et al. Diagnostic precision of nanoparticle-enhanced MRI for lymph-node metastases: a meta-analysis. Lancet Oncol. 2006;7:52–60.
Engelen SM, Lahaye MJ, Beets-Tan RG, Jansen RL, Lammering G, De Bruine AP, et al. Tailored treatment of primary rectal cancer based on MRI: does it reduce the number of incomplete resections? Ann Oncol. 2008;19 Suppl 1:i17–8.
O’Neill BD, Brown G, Heald RJ, Cunningham D, Tait DM. Non-operative treatment after neoadjuvant chemoradiotherapy for rectal cancer. Lancet Oncol. 2007;8:625–33.
Habr-Gama A, Perez RO, Proscurshim I, Campos FG, Nadalin W, Kiss D, et al. Patterns of failure and survival for nonoperative treatment of stage c0 distal rectal cancer following neoadjuvant chemoradiation therapy. J Gastrointest Surg. 2006;10:1319–29.
Lezoche G, Baldarelli M, Guerrieri M, Paganini AM, De Sanctis A, Bartolacci S, et al. A prospective randomized study with a 5-year minimum follow-up evaluation of transanal endoscopic microsurgery versus laparoscopic total mesorectal excision after neoadjuvant therapy. Surg Endosc. 2008;22:352–8.
Maas M, Nelemans PJ, Valentini V, Das P, Rodel C, Kuo LJ, et al. Long-term outcome in patients with a pathological complete response after chemoradiation for rectal cancer: a pooled analysis of individual patient data. Lancet Oncol. 2010;11:835–44.
Beets-Tan RG, Beets GL. Rectal cancer: review with emphasis on MR imaging. Radiology. 2004;232:335–46.
Dresen RC, Beets GL, Rutten HJ, Engelen SM, Lahaye MJ, Vliegen RF. Locally advanced rectal cancer: MR imaging for restaging after neoadjuvant radiation therapy with concomitant chemotherapy. Part I. Are we able to predict tumor confined to the rectal wall? Radiology. 2009;252:71–80.
Bipat S, Glas AS, Slors FJ, Zwinderman AH, Bossuyt PM, Stoker J. Rectal cancer: local staging and assessment of lymph node involvement with endoluminal US, CT, and MR imaging – a meta-analysis. Radiology. 2004;232:773–83.
MERCURY Study Group. Extramural depth of tumor invasion at thin-section MR in patients with rectal cancer: results of the MERCURY study. Radiology. 2007;243:132–9.
Brown G, Richards CJ, Bourne MW, Newcombe RG, Radcliffe AG, Dallimore NS, et al. Morphologic predictors of lymph node status in rectal cancer with use of high-spatial-resolution MR imaging with histopathologic comparison. Radiology. 2003;227:371–7.
Kim JH, Beets GL, Kim MJ, Kessels AG, Beets-Tan RG. High-resolution MR imaging for nodal staging in rectal cancer: are there any criteria in addition to the size? Eur J Radiol. 2004;52:78–83.
Lahaye MJ, Engelen SM, Kessels AG, de Bruine AP, von Meyenfeldt MF, van Engelshoven JM, et al. USPIO-enhanced MR imaging for nodal staging in patients with primary rectal cancer: predictive criteria. Radiology. 2008;246:804–11.
Chen CC, Lee RC, Lin JK, Wang LW, Yang SH. How accurate is magnetic resonance imaging in restaging rectal cancer in patients receiving preoperative combined chemoradiotherapy? Dis Colon Rectum. 2005;48:722–8.
Kuo LJ, Chern MC, Tsou MH, Liu MC, Jian JJ, Chen CM, et al. Interpretation of magnetic resonance imaging for locally advanced rectal carcinoma after preoperative chemoradiation therapy. Dis Colon Rectum. 2005;48:23–8.
Allen SD, Padhani AR, Dzik-Jurasz AS, Glynne-Jones R. Rectal carcinoma: MRI with histologic correlation before and after chemoradiation therapy. AJR Am J Roentgenol. 2007;188:442–51.
Engelen SM, Beets-Tan RG, Lahaye MJ, Lammering G, Jansen RL, van Dam RM, et al. MRI after chemoradiotherapy of rectal cancer: a useful tool to select patients for local excision. Dis Colon Rectum. 2010;53:979–86.
Lahaye MJ, Beets GL, Engelen SM, Kessels AG, de Bruine AP, Kwee HW. Locally advanced rectal cancer: MR imaging for restaging after neoadjuvant radiation therapy with concomitant chemotherapy. Part II. What are the criteria to predict involved lymph nodes? Radiology. 2009;252:81–91.
Mercury Study Group. Diagnostic accuracy of preoperative magnetic resonance imaging in predicting curative resection of rectal cancer: prospective observational study. BMJ. 2006;333(7572):779.
Vliegen RF, Beets GL, Lammering G, Dresen RC, Rutten HJ, Kessels AG, et al. Mesorectal fascia invasion after neoadjuvant chemotherapy and radiation therapy for locally advanced rectal cancer: accuracy of MR imaging for prediction. Radiology. 2008;246:454–62.
Kulkarni T, Gollins S, Maw A, Hobson P, Byrne R, Widdowson D. Magnetic resonance imaging in rectal cancer downstaged using neoadjuvant chemoradiation: accuracy of prediction of tumour stage and circumferential resection margin status. Colorectal Dis. 2008;10:479–89.
Vliegen RF, Beets GL, von Meyenfeldt MF, Kessels AG, Lemaire EE, van Engelshoven JM, et al. Rectal cancer: MR imaging in local staging – is gadolinium-based contrast material helpful? Radiology. 2005;234:179–88.
Parks AG, Gordon PH, Hardcastle JD. A classification of fistula-in-ano. Br J Surg. 1976;63:1–12.
Beets-Tan RG, Beets GL, van der Hoop AG, Kessels AG, Vliegen RF, Baeten CG, et al. Preoperative MR imaging of anal fistulas: does it really help the surgeon? Radiology. 2001;218:75–84.
Buchanan GN, Halligan S, Bartram CI, Williams AB, Tarroni D, Cohen CR. Clinical examination, endosonography, and MR imaging in preoperative assessment of fistula in ano: comparison with outcome-based reference standard. Radiology. 2004;233:674–81.
Kuijpers HC, Schulpen T. Fistulography for fistula-in-ano. Is it useful? Dis Colon Rectum. 1985;28:103–4.
Sahni VA, Ahmad R, Burling D. Which method is best for imaging of perianal fistula? Abdom Imaging. 2008;33:26–30.
de Souza NM, Gilderdale DJ, Coutts GA, Puni R, Steiner RE. MRI of fistula-in-ano: a comparison of endoanal coil with external phased array coil techniques. J Comput Assist Tomogr. 1998;22:357–63.
Stoker J, Hussain SM, van Kempen D, Elevelt AJ, Lameris JS. Endoanal coil in MR imaging of anal fistulas. AJR Am J Roentgenol. 1996;166:360–2.
Halligan S, Bartram CI. MR imaging of fistula in ano: are endoanal coils the gold standard? AJR Am J Roentgenol. 1998;171:407–12.
Beets-Tan RG, Beets GL, van der Hoop AG, Kessels AG, Vliegen RF, Baeten CG, van Engelshoven JM. Preoperative magnetic resonance imaging of anal fistulas: does it really help the surgeon? Radiology. 2001;218(1):75–84.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer-Verlag London
About this chapter
Cite this chapter
Engelen, S.M.E., Beets, G.L., Beets-Tan, R.G.H. (2013). Magnetic Resonance Imaging of Anorectal Diseases. In: Zbar, A., Madoff, R., Wexner, S. (eds) Reconstructive Surgery of the Rectum, Anus and Perineum. Springer, London. https://doi.org/10.1007/978-1-84882-413-3_4
Download citation
DOI: https://doi.org/10.1007/978-1-84882-413-3_4
Published:
Publisher Name: Springer, London
Print ISBN: 978-1-84882-412-6
Online ISBN: 978-1-84882-413-3
eBook Packages: MedicineMedicine (R0)